User login
FDA pulls U.S. authorization for Eli Lilly’s COVID drug bebtelovimab
the Food and Drug Administration said, citing it is not expected to neutralize the dominant BQ.1 and BQ.1.1 subvariants of Omicron.
The announcement on Nov. 30 takes away authorization from the last COVID-19 monoclonal antibody treatment, leaving Pfizer’s antiviral drug Paxlovid, Merck’s Lagevrio, and Gilead Sciences’ Veklury as treatments for the disease, besides convalescent plasma for some patients.
AstraZeneca’s monoclonal antibody Evusheld is also authorized for protection against COVID-19 infection in some people.
Eli Lilly and its authorized distributors have paused commercial distribution of the monoclonal antibody until further notice from the agency, while the U.S. government has also paused fulfillment of any pending requests under its scheme to help uninsured and underinsured Americans access the drug.
The drug, which was discovered by Abcellera and commercialized by Eli Lilly, received an authorization from the FDA in February.
BQ.1 and BQ.1.1 have become the dominant strains in the United States after a steady increase in prevalence over the last 2 months, surpassing Omicron’s BA.5 subvariant, which had driven cases earlier in the year.
The subvariants accounted for around 57% of the cases nationally, as per government data last week.
Reuters Health Information © 2022
the Food and Drug Administration said, citing it is not expected to neutralize the dominant BQ.1 and BQ.1.1 subvariants of Omicron.
The announcement on Nov. 30 takes away authorization from the last COVID-19 monoclonal antibody treatment, leaving Pfizer’s antiviral drug Paxlovid, Merck’s Lagevrio, and Gilead Sciences’ Veklury as treatments for the disease, besides convalescent plasma for some patients.
AstraZeneca’s monoclonal antibody Evusheld is also authorized for protection against COVID-19 infection in some people.
Eli Lilly and its authorized distributors have paused commercial distribution of the monoclonal antibody until further notice from the agency, while the U.S. government has also paused fulfillment of any pending requests under its scheme to help uninsured and underinsured Americans access the drug.
The drug, which was discovered by Abcellera and commercialized by Eli Lilly, received an authorization from the FDA in February.
BQ.1 and BQ.1.1 have become the dominant strains in the United States after a steady increase in prevalence over the last 2 months, surpassing Omicron’s BA.5 subvariant, which had driven cases earlier in the year.
The subvariants accounted for around 57% of the cases nationally, as per government data last week.
Reuters Health Information © 2022
the Food and Drug Administration said, citing it is not expected to neutralize the dominant BQ.1 and BQ.1.1 subvariants of Omicron.
The announcement on Nov. 30 takes away authorization from the last COVID-19 monoclonal antibody treatment, leaving Pfizer’s antiviral drug Paxlovid, Merck’s Lagevrio, and Gilead Sciences’ Veklury as treatments for the disease, besides convalescent plasma for some patients.
AstraZeneca’s monoclonal antibody Evusheld is also authorized for protection against COVID-19 infection in some people.
Eli Lilly and its authorized distributors have paused commercial distribution of the monoclonal antibody until further notice from the agency, while the U.S. government has also paused fulfillment of any pending requests under its scheme to help uninsured and underinsured Americans access the drug.
The drug, which was discovered by Abcellera and commercialized by Eli Lilly, received an authorization from the FDA in February.
BQ.1 and BQ.1.1 have become the dominant strains in the United States after a steady increase in prevalence over the last 2 months, surpassing Omicron’s BA.5 subvariant, which had driven cases earlier in the year.
The subvariants accounted for around 57% of the cases nationally, as per government data last week.
Reuters Health Information © 2022
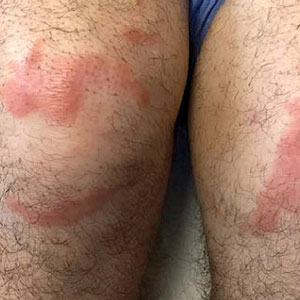
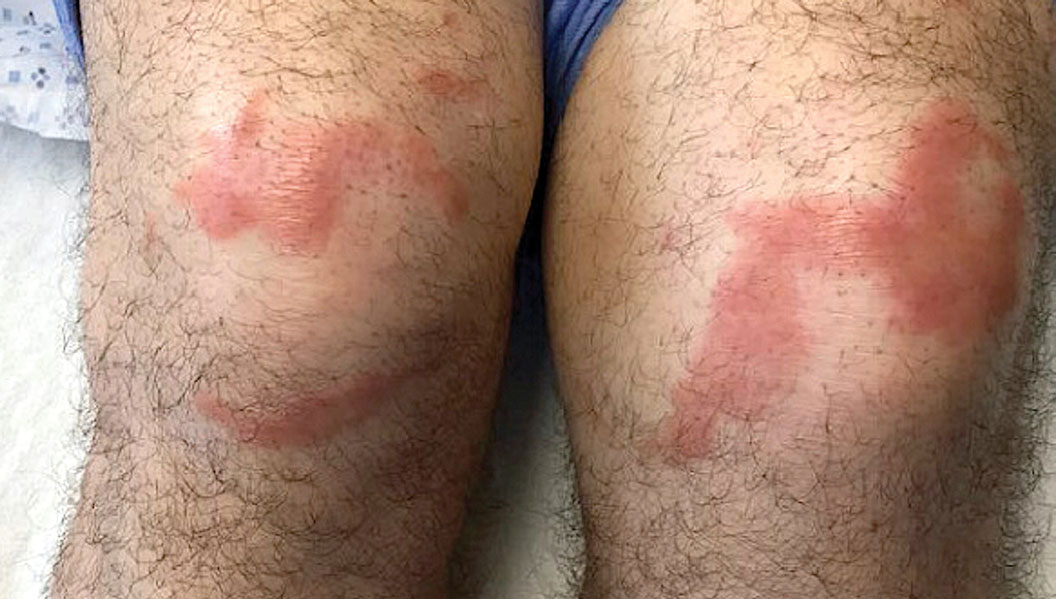
Multiple Annular Erythematous Plaques
The Diagnosis: Mid-Borderline Multibacillary Leprosy
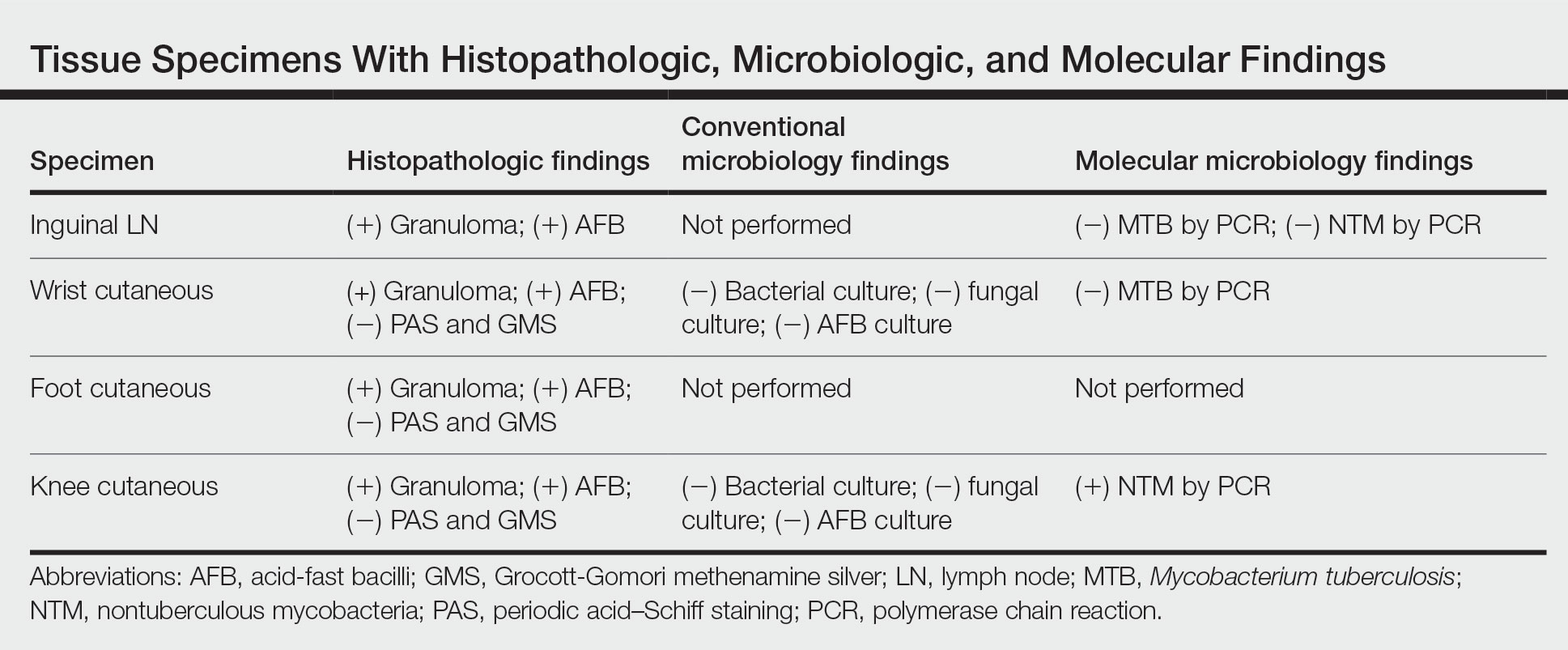
The biopsies showed a granulomatous dermatitis involving the dermis and subcutaneous adipose tissue (Figure, A). Fite staining also revealed numerous acid-fast bacilli (AFB) throughout the dermis (Figure, B); however, polymerase chain reaction (PCR) for Mycobacterium tuberculosis was negative, and concomitant AFB tissue culture showed no growth after 8 weeks of incubation from the left wrist biopsy (Table). Interestingly, a left inguinal lymph node biopsy performed 6 months prior to presentation that helped to establish the diagnosis of follicular lymphoma also revealed nonnecrotizing granulomas and the presence of rare AFB; this formalin-fixed specimen subsequently tested negative for M tuberculosis and nontuberculous mycobacteria (NTM) by broad-range PCR. Due to a high index of suspicion, another unpreserved skin biopsy of the right knee was sent for NTM testing with PCR. Primers to 16S ribosomal RNA and the beta subunit of RNA polymerase, rpoB, gene detected Mycobacterium leprae DNA, leading to the diagnosis of mid-borderline (or borderline-borderline) multibacillary leprosy. Our patient subsequently reported subtle hypoesthesia of the plaques on the knees. He recalled eating undercooked armadillo meat in the southern United States more than 30 years prior to admission. In addition, he had a history of being incarcerated in the northeastern United States. This case was reported to the National Hansen’s Disease Program, and our patient was started on a 2-year course of daily clarithromycin, daily minocycline, and once-monthly moxifloxacin. His family also was evaluated and did not have any skin lesions concerning for leprosy.
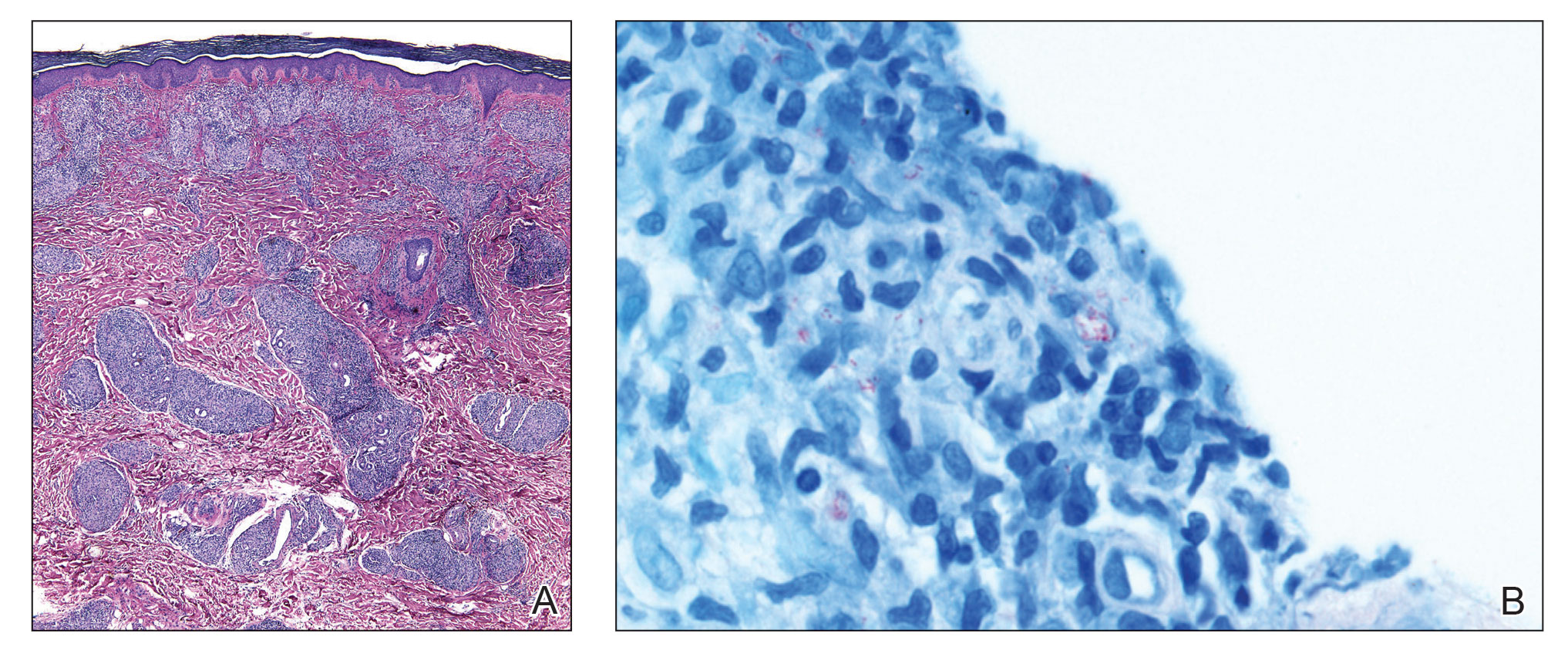
Leprosy is a major global health concern, transmitted via breaks in the skin, respiratory secretions, and contact with armadillos. It continues to be endemic in India, Brazil, and Indonesia.1 In the United States where leprosy is nonendemic, 159 new cases were detected in 2020; the most notable risk factors in the United States are armadillo exposure and travel history.2,3Mycobacterium leprae are intracellular bacilli that preferentially infect macrophages and Schwann cells, resulting in erythematous or hypopigmented skin lesions that often are anesthetic. Mycobacterium leprae has the longest doubling time of all bacteria with unknown in vitro growth requirements and a typical in vivo incubation period of 2 to 10 years.4 Therefore, in vitro cultures will yield no growth, as seen in our case. In our patient, Fite stain showed acid-fast organisms in multiple tissue specimens, but AFB cultures demonstrated no growth after 8 weeks of incubation. Although clinicopathologic correlation is most important, PCR analysis can help to assist in the diagnosis of leprosy. Unpreserved tissue should be used when possible, as the fixation process may adversely affect the analytic sensitivity of subsequent PCR-based assays.5 In our case, NTM were not detected by PCR in the inguinal lymph node specimen despite demonstrating rare AFB staining. This result likely was multifactorial, including the effect of formalin fixation and paraffin embedding as well as concomitant low biomass.
Leprosy is known as a great imitator, and clinical manifestations (both neurologic and cutaneous) depend on host immune response to the mycobacteria. Although tuberculoid leprosy (associated with T helper type 1 immune response) is distinguished by few asymmetric, well-demarcated, and often hypopigmented plaques, lepromatous leprosy (associated with T helper type 2 response) is characterized by numerous symmetric and poorly defined lesions. Borderline leprosy, as seen in our patient, is the most common type of leprosy and shows features of both tuberculoid and lepromatous leprosy.4 It also may be particularly difficult to diagnose.6,7 Borderline-borderline leprosy involves lesions that mostly are of the lepromatous type and symmetric but also may include raised plaques, as in tuberculoid leprosy.4 Plaques in an annular configuration with central clearing, as seen in our patient, are considered suggestive.8 Histopathology of borderline-borderline leprosy lesions shows subepidermal clear zones, and granulomas are more diffuse than in tuberculoid leprosy.4
Given the noncaseating granulomatous dermatitis seen on histopathology and the relatively higher incidence of sarcoidosis in our region of practice, our initial differential included sarcoidosis and other granulomatous disorders such as granuloma annulare. Interestingly, sarcoidosis has been misdiagnosed as leprosy on multiple occasions in countries where leprosy is endemic.9,10 Localized cutaneous leishmaniasis typically presents with infiltrated plaques and nodules that may ulcerate; diffuse and disseminated as well as mucocutaneous presentations may occur depending on the species and severity of infection. Parasitized macrophages containing amastigotes may be seen in the dermis highlighted by CD1a immunostaining. Mycosis fungoides presents as papulosquamous patches or plaques, often favoring sunprotected sites; the hypopigmented variant may mimic the central clearing seen in leprosy.
The diagnosis of leprosy can be challenging due to varying clinical presentation; indolent growth of the causative organism; and indeterminate nature of stains, including the Fite stain. Although leprosy is an uncommon diagnosis, this case underscores the need to keep it in the differential of granulomatous dermatoses in the appropriate clinical setting, particularly in patients with risk factors for exposure.8
- Blok DJ, De Vlas SJ, Richardus JH. Global elimination of leprosy by 2020: are we on track? Parasit Vectors. 2015;8:548. doi:10.1186/s13071-015-1143-4
- National Hansen’s disease (leprosy) program caring and curing since 1894. Health Resources and Services Administration website. Published April 13, 2017. Accessed November 17, 2022. https://www.hrsa.gov/hansens-disease/index.html
- Aslam S, Peraza J, Mekaiel A, et al. Major risk factors for leprosy in a non-endemic area of the United States: a case series. IDCases. 2019;17:E00557. doi:10.1016/j.idcr.2019.e00557
- Kundakci N, Erdem C. Leprosy: a great imitator. Clin Dermatol. 2019;37:200-212. doi:10.1016/j.clindermatol.2019.01.002
- Marchetti G, Gori A, Catozzi L, et al. Evaluation of PCR in detection of Mycobacterium tuberculosis from formalin-fixed, paraffin-embedded tissues: comparison of four amplification assays. J Clin Microbiol. 1998;36:1512-1517.
- Pawar M, Zawar V. Mid-borderline leprosy masquerading as an overlap syndrome. Rheumatology (Oxford). 2018;57:1686-1688. doi:10.1093 /rheumatology/key125
- Day W, Prodanovic E. Borderline lepromatous leprosy masking as tinea versicolor. Int J Dermatol. 2019;58:E125-E126. doi:10.1111/ijd.14439
- Lastória JC, de Abreu MAMM. Leprosy: review of the epidemiological, clinical, and etiopathogenic aspects: part 1. An Bras Dermatol. 2014;89:205-218. doi:10.1590/abd1806-4841.20142450
- Kaushik A, Vinay K, Narang T, et al. Ichthyosiform sarcoidosis: a mimic of leprosy? Clin Exp Dermatol. 2019;44:677-680. doi:10.1111/ced.13863
- Chowdhary KN, Rao R, Priya P, et al. Cutaneous sarcoidosis misdiagnosed as leprosy. report of two cases and review of literature. Indian J Lepr. 2016;88:177-183.
The Diagnosis: Mid-Borderline Multibacillary Leprosy
The biopsies showed a granulomatous dermatitis involving the dermis and subcutaneous adipose tissue (Figure, A). Fite staining also revealed numerous acid-fast bacilli (AFB) throughout the dermis (Figure, B); however, polymerase chain reaction (PCR) for Mycobacterium tuberculosis was negative, and concomitant AFB tissue culture showed no growth after 8 weeks of incubation from the left wrist biopsy (Table). Interestingly, a left inguinal lymph node biopsy performed 6 months prior to presentation that helped to establish the diagnosis of follicular lymphoma also revealed nonnecrotizing granulomas and the presence of rare AFB; this formalin-fixed specimen subsequently tested negative for M tuberculosis and nontuberculous mycobacteria (NTM) by broad-range PCR. Due to a high index of suspicion, another unpreserved skin biopsy of the right knee was sent for NTM testing with PCR. Primers to 16S ribosomal RNA and the beta subunit of RNA polymerase, rpoB, gene detected Mycobacterium leprae DNA, leading to the diagnosis of mid-borderline (or borderline-borderline) multibacillary leprosy. Our patient subsequently reported subtle hypoesthesia of the plaques on the knees. He recalled eating undercooked armadillo meat in the southern United States more than 30 years prior to admission. In addition, he had a history of being incarcerated in the northeastern United States. This case was reported to the National Hansen’s Disease Program, and our patient was started on a 2-year course of daily clarithromycin, daily minocycline, and once-monthly moxifloxacin. His family also was evaluated and did not have any skin lesions concerning for leprosy.

Leprosy is a major global health concern, transmitted via breaks in the skin, respiratory secretions, and contact with armadillos. It continues to be endemic in India, Brazil, and Indonesia.1 In the United States where leprosy is nonendemic, 159 new cases were detected in 2020; the most notable risk factors in the United States are armadillo exposure and travel history.2,3Mycobacterium leprae are intracellular bacilli that preferentially infect macrophages and Schwann cells, resulting in erythematous or hypopigmented skin lesions that often are anesthetic. Mycobacterium leprae has the longest doubling time of all bacteria with unknown in vitro growth requirements and a typical in vivo incubation period of 2 to 10 years.4 Therefore, in vitro cultures will yield no growth, as seen in our case. In our patient, Fite stain showed acid-fast organisms in multiple tissue specimens, but AFB cultures demonstrated no growth after 8 weeks of incubation. Although clinicopathologic correlation is most important, PCR analysis can help to assist in the diagnosis of leprosy. Unpreserved tissue should be used when possible, as the fixation process may adversely affect the analytic sensitivity of subsequent PCR-based assays.5 In our case, NTM were not detected by PCR in the inguinal lymph node specimen despite demonstrating rare AFB staining. This result likely was multifactorial, including the effect of formalin fixation and paraffin embedding as well as concomitant low biomass.

Leprosy is known as a great imitator, and clinical manifestations (both neurologic and cutaneous) depend on host immune response to the mycobacteria. Although tuberculoid leprosy (associated with T helper type 1 immune response) is distinguished by few asymmetric, well-demarcated, and often hypopigmented plaques, lepromatous leprosy (associated with T helper type 2 response) is characterized by numerous symmetric and poorly defined lesions. Borderline leprosy, as seen in our patient, is the most common type of leprosy and shows features of both tuberculoid and lepromatous leprosy.4 It also may be particularly difficult to diagnose.6,7 Borderline-borderline leprosy involves lesions that mostly are of the lepromatous type and symmetric but also may include raised plaques, as in tuberculoid leprosy.4 Plaques in an annular configuration with central clearing, as seen in our patient, are considered suggestive.8 Histopathology of borderline-borderline leprosy lesions shows subepidermal clear zones, and granulomas are more diffuse than in tuberculoid leprosy.4
Given the noncaseating granulomatous dermatitis seen on histopathology and the relatively higher incidence of sarcoidosis in our region of practice, our initial differential included sarcoidosis and other granulomatous disorders such as granuloma annulare. Interestingly, sarcoidosis has been misdiagnosed as leprosy on multiple occasions in countries where leprosy is endemic.9,10 Localized cutaneous leishmaniasis typically presents with infiltrated plaques and nodules that may ulcerate; diffuse and disseminated as well as mucocutaneous presentations may occur depending on the species and severity of infection. Parasitized macrophages containing amastigotes may be seen in the dermis highlighted by CD1a immunostaining. Mycosis fungoides presents as papulosquamous patches or plaques, often favoring sunprotected sites; the hypopigmented variant may mimic the central clearing seen in leprosy.
The diagnosis of leprosy can be challenging due to varying clinical presentation; indolent growth of the causative organism; and indeterminate nature of stains, including the Fite stain. Although leprosy is an uncommon diagnosis, this case underscores the need to keep it in the differential of granulomatous dermatoses in the appropriate clinical setting, particularly in patients with risk factors for exposure.8
The Diagnosis: Mid-Borderline Multibacillary Leprosy
The biopsies showed a granulomatous dermatitis involving the dermis and subcutaneous adipose tissue (Figure, A). Fite staining also revealed numerous acid-fast bacilli (AFB) throughout the dermis (Figure, B); however, polymerase chain reaction (PCR) for Mycobacterium tuberculosis was negative, and concomitant AFB tissue culture showed no growth after 8 weeks of incubation from the left wrist biopsy (Table). Interestingly, a left inguinal lymph node biopsy performed 6 months prior to presentation that helped to establish the diagnosis of follicular lymphoma also revealed nonnecrotizing granulomas and the presence of rare AFB; this formalin-fixed specimen subsequently tested negative for M tuberculosis and nontuberculous mycobacteria (NTM) by broad-range PCR. Due to a high index of suspicion, another unpreserved skin biopsy of the right knee was sent for NTM testing with PCR. Primers to 16S ribosomal RNA and the beta subunit of RNA polymerase, rpoB, gene detected Mycobacterium leprae DNA, leading to the diagnosis of mid-borderline (or borderline-borderline) multibacillary leprosy. Our patient subsequently reported subtle hypoesthesia of the plaques on the knees. He recalled eating undercooked armadillo meat in the southern United States more than 30 years prior to admission. In addition, he had a history of being incarcerated in the northeastern United States. This case was reported to the National Hansen’s Disease Program, and our patient was started on a 2-year course of daily clarithromycin, daily minocycline, and once-monthly moxifloxacin. His family also was evaluated and did not have any skin lesions concerning for leprosy.

Leprosy is a major global health concern, transmitted via breaks in the skin, respiratory secretions, and contact with armadillos. It continues to be endemic in India, Brazil, and Indonesia.1 In the United States where leprosy is nonendemic, 159 new cases were detected in 2020; the most notable risk factors in the United States are armadillo exposure and travel history.2,3Mycobacterium leprae are intracellular bacilli that preferentially infect macrophages and Schwann cells, resulting in erythematous or hypopigmented skin lesions that often are anesthetic. Mycobacterium leprae has the longest doubling time of all bacteria with unknown in vitro growth requirements and a typical in vivo incubation period of 2 to 10 years.4 Therefore, in vitro cultures will yield no growth, as seen in our case. In our patient, Fite stain showed acid-fast organisms in multiple tissue specimens, but AFB cultures demonstrated no growth after 8 weeks of incubation. Although clinicopathologic correlation is most important, PCR analysis can help to assist in the diagnosis of leprosy. Unpreserved tissue should be used when possible, as the fixation process may adversely affect the analytic sensitivity of subsequent PCR-based assays.5 In our case, NTM were not detected by PCR in the inguinal lymph node specimen despite demonstrating rare AFB staining. This result likely was multifactorial, including the effect of formalin fixation and paraffin embedding as well as concomitant low biomass.

Leprosy is known as a great imitator, and clinical manifestations (both neurologic and cutaneous) depend on host immune response to the mycobacteria. Although tuberculoid leprosy (associated with T helper type 1 immune response) is distinguished by few asymmetric, well-demarcated, and often hypopigmented plaques, lepromatous leprosy (associated with T helper type 2 response) is characterized by numerous symmetric and poorly defined lesions. Borderline leprosy, as seen in our patient, is the most common type of leprosy and shows features of both tuberculoid and lepromatous leprosy.4 It also may be particularly difficult to diagnose.6,7 Borderline-borderline leprosy involves lesions that mostly are of the lepromatous type and symmetric but also may include raised plaques, as in tuberculoid leprosy.4 Plaques in an annular configuration with central clearing, as seen in our patient, are considered suggestive.8 Histopathology of borderline-borderline leprosy lesions shows subepidermal clear zones, and granulomas are more diffuse than in tuberculoid leprosy.4
Given the noncaseating granulomatous dermatitis seen on histopathology and the relatively higher incidence of sarcoidosis in our region of practice, our initial differential included sarcoidosis and other granulomatous disorders such as granuloma annulare. Interestingly, sarcoidosis has been misdiagnosed as leprosy on multiple occasions in countries where leprosy is endemic.9,10 Localized cutaneous leishmaniasis typically presents with infiltrated plaques and nodules that may ulcerate; diffuse and disseminated as well as mucocutaneous presentations may occur depending on the species and severity of infection. Parasitized macrophages containing amastigotes may be seen in the dermis highlighted by CD1a immunostaining. Mycosis fungoides presents as papulosquamous patches or plaques, often favoring sunprotected sites; the hypopigmented variant may mimic the central clearing seen in leprosy.
The diagnosis of leprosy can be challenging due to varying clinical presentation; indolent growth of the causative organism; and indeterminate nature of stains, including the Fite stain. Although leprosy is an uncommon diagnosis, this case underscores the need to keep it in the differential of granulomatous dermatoses in the appropriate clinical setting, particularly in patients with risk factors for exposure.8
- Blok DJ, De Vlas SJ, Richardus JH. Global elimination of leprosy by 2020: are we on track? Parasit Vectors. 2015;8:548. doi:10.1186/s13071-015-1143-4
- National Hansen’s disease (leprosy) program caring and curing since 1894. Health Resources and Services Administration website. Published April 13, 2017. Accessed November 17, 2022. https://www.hrsa.gov/hansens-disease/index.html
- Aslam S, Peraza J, Mekaiel A, et al. Major risk factors for leprosy in a non-endemic area of the United States: a case series. IDCases. 2019;17:E00557. doi:10.1016/j.idcr.2019.e00557
- Kundakci N, Erdem C. Leprosy: a great imitator. Clin Dermatol. 2019;37:200-212. doi:10.1016/j.clindermatol.2019.01.002
- Marchetti G, Gori A, Catozzi L, et al. Evaluation of PCR in detection of Mycobacterium tuberculosis from formalin-fixed, paraffin-embedded tissues: comparison of four amplification assays. J Clin Microbiol. 1998;36:1512-1517.
- Pawar M, Zawar V. Mid-borderline leprosy masquerading as an overlap syndrome. Rheumatology (Oxford). 2018;57:1686-1688. doi:10.1093 /rheumatology/key125
- Day W, Prodanovic E. Borderline lepromatous leprosy masking as tinea versicolor. Int J Dermatol. 2019;58:E125-E126. doi:10.1111/ijd.14439
- Lastória JC, de Abreu MAMM. Leprosy: review of the epidemiological, clinical, and etiopathogenic aspects: part 1. An Bras Dermatol. 2014;89:205-218. doi:10.1590/abd1806-4841.20142450
- Kaushik A, Vinay K, Narang T, et al. Ichthyosiform sarcoidosis: a mimic of leprosy? Clin Exp Dermatol. 2019;44:677-680. doi:10.1111/ced.13863
- Chowdhary KN, Rao R, Priya P, et al. Cutaneous sarcoidosis misdiagnosed as leprosy. report of two cases and review of literature. Indian J Lepr. 2016;88:177-183.
- Blok DJ, De Vlas SJ, Richardus JH. Global elimination of leprosy by 2020: are we on track? Parasit Vectors. 2015;8:548. doi:10.1186/s13071-015-1143-4
- National Hansen’s disease (leprosy) program caring and curing since 1894. Health Resources and Services Administration website. Published April 13, 2017. Accessed November 17, 2022. https://www.hrsa.gov/hansens-disease/index.html
- Aslam S, Peraza J, Mekaiel A, et al. Major risk factors for leprosy in a non-endemic area of the United States: a case series. IDCases. 2019;17:E00557. doi:10.1016/j.idcr.2019.e00557
- Kundakci N, Erdem C. Leprosy: a great imitator. Clin Dermatol. 2019;37:200-212. doi:10.1016/j.clindermatol.2019.01.002
- Marchetti G, Gori A, Catozzi L, et al. Evaluation of PCR in detection of Mycobacterium tuberculosis from formalin-fixed, paraffin-embedded tissues: comparison of four amplification assays. J Clin Microbiol. 1998;36:1512-1517.
- Pawar M, Zawar V. Mid-borderline leprosy masquerading as an overlap syndrome. Rheumatology (Oxford). 2018;57:1686-1688. doi:10.1093 /rheumatology/key125
- Day W, Prodanovic E. Borderline lepromatous leprosy masking as tinea versicolor. Int J Dermatol. 2019;58:E125-E126. doi:10.1111/ijd.14439
- Lastória JC, de Abreu MAMM. Leprosy: review of the epidemiological, clinical, and etiopathogenic aspects: part 1. An Bras Dermatol. 2014;89:205-218. doi:10.1590/abd1806-4841.20142450
- Kaushik A, Vinay K, Narang T, et al. Ichthyosiform sarcoidosis: a mimic of leprosy? Clin Exp Dermatol. 2019;44:677-680. doi:10.1111/ced.13863
- Chowdhary KN, Rao R, Priya P, et al. Cutaneous sarcoidosis misdiagnosed as leprosy. report of two cases and review of literature. Indian J Lepr. 2016;88:177-183.
A 59-year-old man was admitted to the medical ward with multiple annular erythematous plaques and polyarthralgia of several months’ duration. His medical history included low-grade stage IIA follicular lymphoma diagnosed 6 months prior to presentation, substance abuse with opiates and cocaine, coronary artery disease, ascending aortic aneurysm, and chronic lower back pain. Physical examination revealed multiple red to red-brown papules and plaques, some in an annular configuration, that were distributed on the cheeks, left wrist, knees, dorsal feet, and soles. Bilateral inguinal lymphadenopathy also was noted. Serological testing for HIV, hepatitis B and C viruses, Treponema pallidum, Borrelia burgdorferi, and tuberculosis assay were negative. Arthrocentesis of the left wrist 1 week prior to admission noted 5333 nucleated cells/μL (reference range, <3000 cells/μL) and no crystals; culture of the fluid was sterile. Skin biopsies of plaques on the left wrist, left dorsal foot, and right knee were obtained for histopathologic analysis.
RSV surge stuns parents and strains providers, but doctors offer help
RSV cases peaked in mid-November, according to the latest Centers for Disease Control and Prevention data, with RSV-associated hospitalizations in the United States among patients 0-4 years having maxed out five times higher than they were at the same time in 2021. These surges strained providers and left parents scrambling for care. Fortunately, pediatric hospitalizations appear to be subsiding.
In interviews, the parents of the child who had a severe case of RSV reflected on their son’s bout with the illness, and doctors described challenges to dealing with the surge in RSV cases this season. The physicians also offered advice on how recognize and respond to future cases of the virus.
Sebastian Witt’s story
“I didn’t even know what RSV was,” said Malte Witt, whose son, Sebastian, 2, was recently hospitalized for RSV in Denver.
Mr. Witt and his wife, Emily Witt, both 32, thought they were dealing with a typical cold until Sebastian’s condition dramatically deteriorated about 36 hours after symptom onset.
“He basically just slumped over and collapsed, coughing uncontrollably,” Mr. Witt said in an interview. “He couldn’t catch his breath.”
The Witts rushed Sebastian to the ED at Children’s Hospital Colorado, expecting to see a doctor immediately. Instead, they spent the night in an overcrowded waiting room alongside many other families in the same situation.
“There was no room for anyone to sit anywhere,” Mr. Witt said. “There were people sitting on the floor. I counted maybe six children hooked up to oxygen when we walked in.”
After waiting approximately 45 minutes, a nurse checked Sebastian’s oxygen saturation. The readings were 79%-83%. This range is significantly below thresholds for supplemental oxygen described by most pediatric guidelines, which range from 90 to 94%.
The nurse connected Sebastian to bottled oxygen in the waiting room, and a recheck 4 hours later showed that his oxygen saturation had improved.
But the improvement didn’t last.
“At roughly hour 10 in the waiting room – it was 4 in the morning – you could tell that Seb was exhausted, really not acting like himself,” Mr. Witt said. “We thought maybe it’s just late at night, he hasn’t really slept. But then Emily noticed that his oxygen tank had run out.”
Mr. Witt told a nurse, and after another check revealed low oxygen saturation, Sebastian was finally admitted.
Early RSV surge strains pediatric providers
With RSV-associated hospitalizations peaking at 48 per 100,000 children, Colorado has been among the states hardest hit by the virus. New Mexico – where hospitalizations peaked at 56.4 per 100,000 children – comes in second. Even in states like California, where hospitalization rates have been almost 10-fold lower than New Mexico, pediatric providers have been stretched to their limits.
“Many hospitals are really being overwhelmed with admissions for RSV, both routine RSV – relatively mild hospitalizations with bronchiolitis – as well as kids in the ICU with more severe cases,” said Dean Blumberg, MD, chief of the division of pediatric infectious diseases at UC Davis Health, Sacramento, said in an interview.
Dr. Blumberg believes the severity of the 2022-2023 RSV season is likely COVID related.
“All community-associated respiratory viral infections are out of whack because of the pandemic, and all the masking and social distancing that was occurring,” he said.
This may also explain why older kids are coming down with more severe cases of RSV.
“Some children are getting RSV for the first time as older children,” Dr. Blumberg said, noting that, historically, most children were infected in the first 2 years of life. “There are reports of children 3 or 4 years of age being admitted with their first episode of RSV because of the [COVID] pandemic.”
This year’s RSV season is also notable for arriving early, potentially catching the community off guard, according to Jennifer D. Kusma, MD, a primary care pediatrician at Ann & Robert H. Lurie Children’s Hospital of Chicago.
“People who should have been protected often weren’t protected yet,” Dr. Kusma said in an interview.
Treatments new, old, and unproven
On Nov. 17, in the midst of the RSV surge, the American Academy of Pediatrics issued updated guidance for palivizumab, an RSV-targeting monoclonal antibody labeled for children at risk of severe RSV, including those with pre-existing lung or heart conditions, and infants with a history of premature birth (less than or equal to 35 weeks’ gestational age).
“If RSV disease activity persists at high levels in a given region through the fall and winter, the AAP supports providing more than five consecutive doses of palivizumab to eligible children,” the update stated.
Insurance companies appear to be responding in kind, covering additional doses for children in need.
“[Payers] have agreed that, if [palivizumab] needs to be given for an additional month or 2 or 3, then they’re making a commitment that they’ll reimburse hospitals for providing that,” Dr. Blumberg said.
For ineligible patients, such as Sebastian, who was born prematurely at 36 weeks – 1 week shy of the label requirement – treatment relies upon supportive care with oxygen and IV fluids.
At home, parents are left with simpler options.
Dr. Blumberg and Dr. Kusma recommended keeping children hydrated, maintaining humidified air, and using saline nose drops with bulb suction to clear mucus.
In the Witts’ experience, that last step may be easier said than done.
“Every time a nurse would walk into the room, Sebastian would yell: ‘Go away, doctor! I don’t want snot sucker!’” Mr. Witt said.
“If you over snot-suck, that’s really uncomfortable for the kid, and really hard for you,” Ms. Witt said. “And it doesn’t make much of a difference. It’s just very hard to find a middle ground, where you’re helping and keeping them comfortable.”
Some parents are turning to novel strategies, such as nebulized hypertonic saline, currently marketed on Amazon for children with RSV.
Although the AAP offers a weak recommendation for nebulized hypertonic saline in children hospitalized more than 72 hours, they advise against it in the emergency setting, citing inconsistent findings in clinical trials.
To any parents tempted by thousands of positive Amazon reviews, Dr. Blumberg said, “I wouldn’t waste my money on that.”
Dr. Kusma agreed.
“[Nebulized hypertonic saline] can be irritating,” she said. “It’s saltwater, essentially. If a parent is in the position where they’re worried about their child’s breathing to the point that they think they need to use it, I would err on the side of calling your pediatrician and being seen.”
Going in, coming home
Dr. Kusma said parents should seek medical attention if a child is breathing faster and working harder to get air. Increased work of breathing is characterized by pulling of the skin at the notch where the throat meets the chest bone (tracheal tugging), and flattening of the belly that makes the ribcage more prominent.
Mr. Witt saw these signs in Sebastian. He knew they were significant, because a friend who is a nurse had previously shown him some examples of children who exhibited these symptoms online.
“That’s how I knew that things were actually really dangerous,” Mr. Witt said. “Had she not shown me those videos a month and a half before this happened, I don’t know that we would have hit the alarm bell as quickly as we did.”
After spending their second night and the following day in a cramped preoperative room converted to manage overflow from the emergency department, Sebastian’s condition improved, and he was discharged. The Witts are relieved to be home, but frustrations from their ordeal remain, especially considering the estimated $5,000 in out-of-pocket costs they expect to pay.
“How is this our health care system?” Ms. Witt asked. “This is unbelievable.”
An optimistic outlook
RSV seasons typically demonstrate a clear peak, followed by a decline through the rest of the season, suggesting better times lie ahead; however, this season has been anything but typical.
“I’m hopeful that it will just go away and stay away,” Dr. Kusma said, citing this trend. “But I can’t know for sure.”
To anxious parents, Dr. Blumberg offered an optimistic view of RSV seasons to come.
“There’s hope,” he said. “There are vaccines that are being developed that are very close to FDA approval. So, it’s possible that this time next year, we might have widespread RSV vaccination available for children so that we don’t have to go through this nightmare again.”
Dr. Blumberg and Dr. Kusma disclosed no relevant conflicts of interest.
RSV cases peaked in mid-November, according to the latest Centers for Disease Control and Prevention data, with RSV-associated hospitalizations in the United States among patients 0-4 years having maxed out five times higher than they were at the same time in 2021. These surges strained providers and left parents scrambling for care. Fortunately, pediatric hospitalizations appear to be subsiding.
In interviews, the parents of the child who had a severe case of RSV reflected on their son’s bout with the illness, and doctors described challenges to dealing with the surge in RSV cases this season. The physicians also offered advice on how recognize and respond to future cases of the virus.
Sebastian Witt’s story
“I didn’t even know what RSV was,” said Malte Witt, whose son, Sebastian, 2, was recently hospitalized for RSV in Denver.
Mr. Witt and his wife, Emily Witt, both 32, thought they were dealing with a typical cold until Sebastian’s condition dramatically deteriorated about 36 hours after symptom onset.
“He basically just slumped over and collapsed, coughing uncontrollably,” Mr. Witt said in an interview. “He couldn’t catch his breath.”
The Witts rushed Sebastian to the ED at Children’s Hospital Colorado, expecting to see a doctor immediately. Instead, they spent the night in an overcrowded waiting room alongside many other families in the same situation.
“There was no room for anyone to sit anywhere,” Mr. Witt said. “There were people sitting on the floor. I counted maybe six children hooked up to oxygen when we walked in.”
After waiting approximately 45 minutes, a nurse checked Sebastian’s oxygen saturation. The readings were 79%-83%. This range is significantly below thresholds for supplemental oxygen described by most pediatric guidelines, which range from 90 to 94%.
The nurse connected Sebastian to bottled oxygen in the waiting room, and a recheck 4 hours later showed that his oxygen saturation had improved.
But the improvement didn’t last.
“At roughly hour 10 in the waiting room – it was 4 in the morning – you could tell that Seb was exhausted, really not acting like himself,” Mr. Witt said. “We thought maybe it’s just late at night, he hasn’t really slept. But then Emily noticed that his oxygen tank had run out.”
Mr. Witt told a nurse, and after another check revealed low oxygen saturation, Sebastian was finally admitted.
Early RSV surge strains pediatric providers
With RSV-associated hospitalizations peaking at 48 per 100,000 children, Colorado has been among the states hardest hit by the virus. New Mexico – where hospitalizations peaked at 56.4 per 100,000 children – comes in second. Even in states like California, where hospitalization rates have been almost 10-fold lower than New Mexico, pediatric providers have been stretched to their limits.
“Many hospitals are really being overwhelmed with admissions for RSV, both routine RSV – relatively mild hospitalizations with bronchiolitis – as well as kids in the ICU with more severe cases,” said Dean Blumberg, MD, chief of the division of pediatric infectious diseases at UC Davis Health, Sacramento, said in an interview.
Dr. Blumberg believes the severity of the 2022-2023 RSV season is likely COVID related.
“All community-associated respiratory viral infections are out of whack because of the pandemic, and all the masking and social distancing that was occurring,” he said.
This may also explain why older kids are coming down with more severe cases of RSV.
“Some children are getting RSV for the first time as older children,” Dr. Blumberg said, noting that, historically, most children were infected in the first 2 years of life. “There are reports of children 3 or 4 years of age being admitted with their first episode of RSV because of the [COVID] pandemic.”
This year’s RSV season is also notable for arriving early, potentially catching the community off guard, according to Jennifer D. Kusma, MD, a primary care pediatrician at Ann & Robert H. Lurie Children’s Hospital of Chicago.
“People who should have been protected often weren’t protected yet,” Dr. Kusma said in an interview.
Treatments new, old, and unproven
On Nov. 17, in the midst of the RSV surge, the American Academy of Pediatrics issued updated guidance for palivizumab, an RSV-targeting monoclonal antibody labeled for children at risk of severe RSV, including those with pre-existing lung or heart conditions, and infants with a history of premature birth (less than or equal to 35 weeks’ gestational age).
“If RSV disease activity persists at high levels in a given region through the fall and winter, the AAP supports providing more than five consecutive doses of palivizumab to eligible children,” the update stated.
Insurance companies appear to be responding in kind, covering additional doses for children in need.
“[Payers] have agreed that, if [palivizumab] needs to be given for an additional month or 2 or 3, then they’re making a commitment that they’ll reimburse hospitals for providing that,” Dr. Blumberg said.
For ineligible patients, such as Sebastian, who was born prematurely at 36 weeks – 1 week shy of the label requirement – treatment relies upon supportive care with oxygen and IV fluids.
At home, parents are left with simpler options.
Dr. Blumberg and Dr. Kusma recommended keeping children hydrated, maintaining humidified air, and using saline nose drops with bulb suction to clear mucus.
In the Witts’ experience, that last step may be easier said than done.
“Every time a nurse would walk into the room, Sebastian would yell: ‘Go away, doctor! I don’t want snot sucker!’” Mr. Witt said.
“If you over snot-suck, that’s really uncomfortable for the kid, and really hard for you,” Ms. Witt said. “And it doesn’t make much of a difference. It’s just very hard to find a middle ground, where you’re helping and keeping them comfortable.”
Some parents are turning to novel strategies, such as nebulized hypertonic saline, currently marketed on Amazon for children with RSV.
Although the AAP offers a weak recommendation for nebulized hypertonic saline in children hospitalized more than 72 hours, they advise against it in the emergency setting, citing inconsistent findings in clinical trials.
To any parents tempted by thousands of positive Amazon reviews, Dr. Blumberg said, “I wouldn’t waste my money on that.”
Dr. Kusma agreed.
“[Nebulized hypertonic saline] can be irritating,” she said. “It’s saltwater, essentially. If a parent is in the position where they’re worried about their child’s breathing to the point that they think they need to use it, I would err on the side of calling your pediatrician and being seen.”
Going in, coming home
Dr. Kusma said parents should seek medical attention if a child is breathing faster and working harder to get air. Increased work of breathing is characterized by pulling of the skin at the notch where the throat meets the chest bone (tracheal tugging), and flattening of the belly that makes the ribcage more prominent.
Mr. Witt saw these signs in Sebastian. He knew they were significant, because a friend who is a nurse had previously shown him some examples of children who exhibited these symptoms online.
“That’s how I knew that things were actually really dangerous,” Mr. Witt said. “Had she not shown me those videos a month and a half before this happened, I don’t know that we would have hit the alarm bell as quickly as we did.”
After spending their second night and the following day in a cramped preoperative room converted to manage overflow from the emergency department, Sebastian’s condition improved, and he was discharged. The Witts are relieved to be home, but frustrations from their ordeal remain, especially considering the estimated $5,000 in out-of-pocket costs they expect to pay.
“How is this our health care system?” Ms. Witt asked. “This is unbelievable.”
An optimistic outlook
RSV seasons typically demonstrate a clear peak, followed by a decline through the rest of the season, suggesting better times lie ahead; however, this season has been anything but typical.
“I’m hopeful that it will just go away and stay away,” Dr. Kusma said, citing this trend. “But I can’t know for sure.”
To anxious parents, Dr. Blumberg offered an optimistic view of RSV seasons to come.
“There’s hope,” he said. “There are vaccines that are being developed that are very close to FDA approval. So, it’s possible that this time next year, we might have widespread RSV vaccination available for children so that we don’t have to go through this nightmare again.”
Dr. Blumberg and Dr. Kusma disclosed no relevant conflicts of interest.
RSV cases peaked in mid-November, according to the latest Centers for Disease Control and Prevention data, with RSV-associated hospitalizations in the United States among patients 0-4 years having maxed out five times higher than they were at the same time in 2021. These surges strained providers and left parents scrambling for care. Fortunately, pediatric hospitalizations appear to be subsiding.
In interviews, the parents of the child who had a severe case of RSV reflected on their son’s bout with the illness, and doctors described challenges to dealing with the surge in RSV cases this season. The physicians also offered advice on how recognize and respond to future cases of the virus.
Sebastian Witt’s story
“I didn’t even know what RSV was,” said Malte Witt, whose son, Sebastian, 2, was recently hospitalized for RSV in Denver.
Mr. Witt and his wife, Emily Witt, both 32, thought they were dealing with a typical cold until Sebastian’s condition dramatically deteriorated about 36 hours after symptom onset.
“He basically just slumped over and collapsed, coughing uncontrollably,” Mr. Witt said in an interview. “He couldn’t catch his breath.”
The Witts rushed Sebastian to the ED at Children’s Hospital Colorado, expecting to see a doctor immediately. Instead, they spent the night in an overcrowded waiting room alongside many other families in the same situation.
“There was no room for anyone to sit anywhere,” Mr. Witt said. “There were people sitting on the floor. I counted maybe six children hooked up to oxygen when we walked in.”
After waiting approximately 45 minutes, a nurse checked Sebastian’s oxygen saturation. The readings were 79%-83%. This range is significantly below thresholds for supplemental oxygen described by most pediatric guidelines, which range from 90 to 94%.
The nurse connected Sebastian to bottled oxygen in the waiting room, and a recheck 4 hours later showed that his oxygen saturation had improved.
But the improvement didn’t last.
“At roughly hour 10 in the waiting room – it was 4 in the morning – you could tell that Seb was exhausted, really not acting like himself,” Mr. Witt said. “We thought maybe it’s just late at night, he hasn’t really slept. But then Emily noticed that his oxygen tank had run out.”
Mr. Witt told a nurse, and after another check revealed low oxygen saturation, Sebastian was finally admitted.
Early RSV surge strains pediatric providers
With RSV-associated hospitalizations peaking at 48 per 100,000 children, Colorado has been among the states hardest hit by the virus. New Mexico – where hospitalizations peaked at 56.4 per 100,000 children – comes in second. Even in states like California, where hospitalization rates have been almost 10-fold lower than New Mexico, pediatric providers have been stretched to their limits.
“Many hospitals are really being overwhelmed with admissions for RSV, both routine RSV – relatively mild hospitalizations with bronchiolitis – as well as kids in the ICU with more severe cases,” said Dean Blumberg, MD, chief of the division of pediatric infectious diseases at UC Davis Health, Sacramento, said in an interview.
Dr. Blumberg believes the severity of the 2022-2023 RSV season is likely COVID related.
“All community-associated respiratory viral infections are out of whack because of the pandemic, and all the masking and social distancing that was occurring,” he said.
This may also explain why older kids are coming down with more severe cases of RSV.
“Some children are getting RSV for the first time as older children,” Dr. Blumberg said, noting that, historically, most children were infected in the first 2 years of life. “There are reports of children 3 or 4 years of age being admitted with their first episode of RSV because of the [COVID] pandemic.”
This year’s RSV season is also notable for arriving early, potentially catching the community off guard, according to Jennifer D. Kusma, MD, a primary care pediatrician at Ann & Robert H. Lurie Children’s Hospital of Chicago.
“People who should have been protected often weren’t protected yet,” Dr. Kusma said in an interview.
Treatments new, old, and unproven
On Nov. 17, in the midst of the RSV surge, the American Academy of Pediatrics issued updated guidance for palivizumab, an RSV-targeting monoclonal antibody labeled for children at risk of severe RSV, including those with pre-existing lung or heart conditions, and infants with a history of premature birth (less than or equal to 35 weeks’ gestational age).
“If RSV disease activity persists at high levels in a given region through the fall and winter, the AAP supports providing more than five consecutive doses of palivizumab to eligible children,” the update stated.
Insurance companies appear to be responding in kind, covering additional doses for children in need.
“[Payers] have agreed that, if [palivizumab] needs to be given for an additional month or 2 or 3, then they’re making a commitment that they’ll reimburse hospitals for providing that,” Dr. Blumberg said.
For ineligible patients, such as Sebastian, who was born prematurely at 36 weeks – 1 week shy of the label requirement – treatment relies upon supportive care with oxygen and IV fluids.
At home, parents are left with simpler options.
Dr. Blumberg and Dr. Kusma recommended keeping children hydrated, maintaining humidified air, and using saline nose drops with bulb suction to clear mucus.
In the Witts’ experience, that last step may be easier said than done.
“Every time a nurse would walk into the room, Sebastian would yell: ‘Go away, doctor! I don’t want snot sucker!’” Mr. Witt said.
“If you over snot-suck, that’s really uncomfortable for the kid, and really hard for you,” Ms. Witt said. “And it doesn’t make much of a difference. It’s just very hard to find a middle ground, where you’re helping and keeping them comfortable.”
Some parents are turning to novel strategies, such as nebulized hypertonic saline, currently marketed on Amazon for children with RSV.
Although the AAP offers a weak recommendation for nebulized hypertonic saline in children hospitalized more than 72 hours, they advise against it in the emergency setting, citing inconsistent findings in clinical trials.
To any parents tempted by thousands of positive Amazon reviews, Dr. Blumberg said, “I wouldn’t waste my money on that.”
Dr. Kusma agreed.
“[Nebulized hypertonic saline] can be irritating,” she said. “It’s saltwater, essentially. If a parent is in the position where they’re worried about their child’s breathing to the point that they think they need to use it, I would err on the side of calling your pediatrician and being seen.”
Going in, coming home
Dr. Kusma said parents should seek medical attention if a child is breathing faster and working harder to get air. Increased work of breathing is characterized by pulling of the skin at the notch where the throat meets the chest bone (tracheal tugging), and flattening of the belly that makes the ribcage more prominent.
Mr. Witt saw these signs in Sebastian. He knew they were significant, because a friend who is a nurse had previously shown him some examples of children who exhibited these symptoms online.
“That’s how I knew that things were actually really dangerous,” Mr. Witt said. “Had she not shown me those videos a month and a half before this happened, I don’t know that we would have hit the alarm bell as quickly as we did.”
After spending their second night and the following day in a cramped preoperative room converted to manage overflow from the emergency department, Sebastian’s condition improved, and he was discharged. The Witts are relieved to be home, but frustrations from their ordeal remain, especially considering the estimated $5,000 in out-of-pocket costs they expect to pay.
“How is this our health care system?” Ms. Witt asked. “This is unbelievable.”
An optimistic outlook
RSV seasons typically demonstrate a clear peak, followed by a decline through the rest of the season, suggesting better times lie ahead; however, this season has been anything but typical.
“I’m hopeful that it will just go away and stay away,” Dr. Kusma said, citing this trend. “But I can’t know for sure.”
To anxious parents, Dr. Blumberg offered an optimistic view of RSV seasons to come.
“There’s hope,” he said. “There are vaccines that are being developed that are very close to FDA approval. So, it’s possible that this time next year, we might have widespread RSV vaccination available for children so that we don’t have to go through this nightmare again.”
Dr. Blumberg and Dr. Kusma disclosed no relevant conflicts of interest.
Flu, RSV infecting children at staggering rates
There has been no ebb in the flurry of respiratory illnesses infecting America’s youngest children. More pediatric wards across the country are announcing crises as beds reach capacity, the pediatric death toll jumped significantly in the past week, and sometimes children are being infected with more than one virus at a time.
In Oregon, for example, the governor announced an official state of emergency to help hospitals deal with the surge of respiratory viruses. Doernbecher Children’s Hospital in Portland, which houses half of all pediatric ICU beds statewide, moved to “crisis mode” and said every pediatric ICU bed was full, Oregon Public Broadcasting reported.
Last month, pediatricians nationwide called for a similar emergency declaration from the federal government to help them respond to the wave of illnesses, which include influenza and respiratory syncytial virus (RSV).
“What’s concerning to us are not only the number of infections, but the severity of these infections, leading to a high number of emergency room visits and hospitalizations,” pediatric infectious disease expert Oscar G. Gómez-Duarte, MD, PhD, said in a news release.
Five more pediatric deaths due to the flu were logged by the CDC in the past week, bringing the total for pediatric flu deaths this season to 12. Nearly 21 per 100,000 children ages 4 and under are being hospitalized for the flu, which is double the rate for children ages 5-17. Last year at this time, fewer than 1 child under age 4 per 100,000 were being hospitalized for the flu.
RSV rates are also stunningly high.
“If we look at CDC data, the RSV hospitalization rate is 10 times higher than usual for this point in the season,” American Medical Association Vice President Andrea Garcia, JD, said in this week’s AMA podcast. “And 171 out of every 100,000 infants younger than 6 months were hospitalized with RSV for the week ending Nov. 12. That is more than double the RSV hospitalization rate for newborns last year and seven times the rate in 2018, which is the last complete season we saw before the pandemic.”
Dr. Gómez-Duarte said hospitals are admitting children with respiratory illnesses who had otherwise been healthy, and sometimes they are even seeing patients with more than one illness.
“Yes, some children are getting what we call coinfections, where they become infected with more than one virus at a time. In some instances, a child becomes initially infected with flu, begins to recover, and subsequently comes down with rhinovirus (a common cold virus), RSV, or any other respiratory virus,” he said. “These coinfections tend to be more severe than when the child just has one infection.”
A version of this article first appeared on Medscape.com.
There has been no ebb in the flurry of respiratory illnesses infecting America’s youngest children. More pediatric wards across the country are announcing crises as beds reach capacity, the pediatric death toll jumped significantly in the past week, and sometimes children are being infected with more than one virus at a time.
In Oregon, for example, the governor announced an official state of emergency to help hospitals deal with the surge of respiratory viruses. Doernbecher Children’s Hospital in Portland, which houses half of all pediatric ICU beds statewide, moved to “crisis mode” and said every pediatric ICU bed was full, Oregon Public Broadcasting reported.
Last month, pediatricians nationwide called for a similar emergency declaration from the federal government to help them respond to the wave of illnesses, which include influenza and respiratory syncytial virus (RSV).
“What’s concerning to us are not only the number of infections, but the severity of these infections, leading to a high number of emergency room visits and hospitalizations,” pediatric infectious disease expert Oscar G. Gómez-Duarte, MD, PhD, said in a news release.
Five more pediatric deaths due to the flu were logged by the CDC in the past week, bringing the total for pediatric flu deaths this season to 12. Nearly 21 per 100,000 children ages 4 and under are being hospitalized for the flu, which is double the rate for children ages 5-17. Last year at this time, fewer than 1 child under age 4 per 100,000 were being hospitalized for the flu.
RSV rates are also stunningly high.
“If we look at CDC data, the RSV hospitalization rate is 10 times higher than usual for this point in the season,” American Medical Association Vice President Andrea Garcia, JD, said in this week’s AMA podcast. “And 171 out of every 100,000 infants younger than 6 months were hospitalized with RSV for the week ending Nov. 12. That is more than double the RSV hospitalization rate for newborns last year and seven times the rate in 2018, which is the last complete season we saw before the pandemic.”
Dr. Gómez-Duarte said hospitals are admitting children with respiratory illnesses who had otherwise been healthy, and sometimes they are even seeing patients with more than one illness.
“Yes, some children are getting what we call coinfections, where they become infected with more than one virus at a time. In some instances, a child becomes initially infected with flu, begins to recover, and subsequently comes down with rhinovirus (a common cold virus), RSV, or any other respiratory virus,” he said. “These coinfections tend to be more severe than when the child just has one infection.”
A version of this article first appeared on Medscape.com.
There has been no ebb in the flurry of respiratory illnesses infecting America’s youngest children. More pediatric wards across the country are announcing crises as beds reach capacity, the pediatric death toll jumped significantly in the past week, and sometimes children are being infected with more than one virus at a time.
In Oregon, for example, the governor announced an official state of emergency to help hospitals deal with the surge of respiratory viruses. Doernbecher Children’s Hospital in Portland, which houses half of all pediatric ICU beds statewide, moved to “crisis mode” and said every pediatric ICU bed was full, Oregon Public Broadcasting reported.
Last month, pediatricians nationwide called for a similar emergency declaration from the federal government to help them respond to the wave of illnesses, which include influenza and respiratory syncytial virus (RSV).
“What’s concerning to us are not only the number of infections, but the severity of these infections, leading to a high number of emergency room visits and hospitalizations,” pediatric infectious disease expert Oscar G. Gómez-Duarte, MD, PhD, said in a news release.
Five more pediatric deaths due to the flu were logged by the CDC in the past week, bringing the total for pediatric flu deaths this season to 12. Nearly 21 per 100,000 children ages 4 and under are being hospitalized for the flu, which is double the rate for children ages 5-17. Last year at this time, fewer than 1 child under age 4 per 100,000 were being hospitalized for the flu.
RSV rates are also stunningly high.
“If we look at CDC data, the RSV hospitalization rate is 10 times higher than usual for this point in the season,” American Medical Association Vice President Andrea Garcia, JD, said in this week’s AMA podcast. “And 171 out of every 100,000 infants younger than 6 months were hospitalized with RSV for the week ending Nov. 12. That is more than double the RSV hospitalization rate for newborns last year and seven times the rate in 2018, which is the last complete season we saw before the pandemic.”
Dr. Gómez-Duarte said hospitals are admitting children with respiratory illnesses who had otherwise been healthy, and sometimes they are even seeing patients with more than one illness.
“Yes, some children are getting what we call coinfections, where they become infected with more than one virus at a time. In some instances, a child becomes initially infected with flu, begins to recover, and subsequently comes down with rhinovirus (a common cold virus), RSV, or any other respiratory virus,” he said. “These coinfections tend to be more severe than when the child just has one infection.”
A version of this article first appeared on Medscape.com.
Buzzy Lancet long COVID paper under investigation for ‘data errors’
An editorial that accompanied the paper when it was published in January of last year described it as “the first large cohort study with 6-months’ follow-up” of people hospitalized with COVID-19. The article has received plenty of attention since then.
Titled “6-month consequences of COVID-19 in patients discharged from hospital: a cohort study,” the paper has been cited nearly 1,600 times, according to Clarivate’s Web of Science. Altmetric finds references to it in multiple documents from the World Health Organization.
According to the expression of concern, dated November 24, a reader found inconsistencies between the data in the article and a later paper describing the same cohort of patients after a year of follow-up. That discovery sparked an investigation that is still ongoing:
- On Jan 8, 2021, The Lancet published an Article, 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study, by Chaolin Huang and colleagues. 1 On Aug 28, 2021, The Lancet published an Article, 1-year outcomes in hospital survivors with COVID-19: a longitudinal cohort study, by Lixue Huang and colleagues. 2 We received an inquiry from a researcher on data inconsistencies between these two Articles, and we sought an explanation from the corresponding author of the two papers. On Nov 7, 2022, Lancet editors were informed that inconsistencies between the 6-month and the 1-year data were due to “some variables in the dataset used for the 6-month paper were mistakenly disrupted in order”. In view of the extent of these data errors, we now issue an Expression of Concern about the 6-month paper 1 while we investigate further, including further statistical and clinical review of the corrected data. We will update this notice as soon as we have further information.
The corresponding author of both papers, Bin Cao of China’s National Center for Respiratory Medicine and the China-Japan Friendship Hospital in Beijing, has not responded to our request for comment.
A profile of Cao published in Lancet Infectious Diseases last March described him as “a leading researcher in pneumonia and influenza” who “has been instrumental in increasing knowledge about COVID-19.” In addition to the follow-up study of hospitalized COVID patients:
- Cao’s seminal papers during the COVID-19 pandemic include the first report of the clinical characteristics of COVID-19 patients in Wuhan, the description of the risk factors for mortality for adult inpatients, and the results of trials testing the use of antiviral drugs, including lopinavir-ritonavir, to treat COVID-19 in China.
We reached out to The Lancet’s press office and Richard Horton, the journal’s editor-in-chief, and received this statement:
- The Lancet Group treats all communications between editors and authors or readers as confidential. Investigations are continuing, and the Expression of Concern will be updated as soon as we have further information to share. More information about our policies is available here:
This year, The Lancet overtook the New England Journal of Medicine as the medical journal with the highest impact factor, in large part due to the papers it published about COVID-19.
We’ve counted retractions for three of those papers, most notably a paper about the use of the drug hydroxychloroquine that claimed to use medical data from a company called Surgisphere. As Retraction Watch readers may remember, the article was retracted after sleuths questioned if the data were real, and the company would not produce it for review.
This article first appeared on Retraction Watch.
An editorial that accompanied the paper when it was published in January of last year described it as “the first large cohort study with 6-months’ follow-up” of people hospitalized with COVID-19. The article has received plenty of attention since then.
Titled “6-month consequences of COVID-19 in patients discharged from hospital: a cohort study,” the paper has been cited nearly 1,600 times, according to Clarivate’s Web of Science. Altmetric finds references to it in multiple documents from the World Health Organization.
According to the expression of concern, dated November 24, a reader found inconsistencies between the data in the article and a later paper describing the same cohort of patients after a year of follow-up. That discovery sparked an investigation that is still ongoing:
- On Jan 8, 2021, The Lancet published an Article, 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study, by Chaolin Huang and colleagues. 1 On Aug 28, 2021, The Lancet published an Article, 1-year outcomes in hospital survivors with COVID-19: a longitudinal cohort study, by Lixue Huang and colleagues. 2 We received an inquiry from a researcher on data inconsistencies between these two Articles, and we sought an explanation from the corresponding author of the two papers. On Nov 7, 2022, Lancet editors were informed that inconsistencies between the 6-month and the 1-year data were due to “some variables in the dataset used for the 6-month paper were mistakenly disrupted in order”. In view of the extent of these data errors, we now issue an Expression of Concern about the 6-month paper 1 while we investigate further, including further statistical and clinical review of the corrected data. We will update this notice as soon as we have further information.
The corresponding author of both papers, Bin Cao of China’s National Center for Respiratory Medicine and the China-Japan Friendship Hospital in Beijing, has not responded to our request for comment.
A profile of Cao published in Lancet Infectious Diseases last March described him as “a leading researcher in pneumonia and influenza” who “has been instrumental in increasing knowledge about COVID-19.” In addition to the follow-up study of hospitalized COVID patients:
- Cao’s seminal papers during the COVID-19 pandemic include the first report of the clinical characteristics of COVID-19 patients in Wuhan, the description of the risk factors for mortality for adult inpatients, and the results of trials testing the use of antiviral drugs, including lopinavir-ritonavir, to treat COVID-19 in China.
We reached out to The Lancet’s press office and Richard Horton, the journal’s editor-in-chief, and received this statement:
- The Lancet Group treats all communications between editors and authors or readers as confidential. Investigations are continuing, and the Expression of Concern will be updated as soon as we have further information to share. More information about our policies is available here:
This year, The Lancet overtook the New England Journal of Medicine as the medical journal with the highest impact factor, in large part due to the papers it published about COVID-19.
We’ve counted retractions for three of those papers, most notably a paper about the use of the drug hydroxychloroquine that claimed to use medical data from a company called Surgisphere. As Retraction Watch readers may remember, the article was retracted after sleuths questioned if the data were real, and the company would not produce it for review.
This article first appeared on Retraction Watch.
An editorial that accompanied the paper when it was published in January of last year described it as “the first large cohort study with 6-months’ follow-up” of people hospitalized with COVID-19. The article has received plenty of attention since then.
Titled “6-month consequences of COVID-19 in patients discharged from hospital: a cohort study,” the paper has been cited nearly 1,600 times, according to Clarivate’s Web of Science. Altmetric finds references to it in multiple documents from the World Health Organization.
According to the expression of concern, dated November 24, a reader found inconsistencies between the data in the article and a later paper describing the same cohort of patients after a year of follow-up. That discovery sparked an investigation that is still ongoing:
- On Jan 8, 2021, The Lancet published an Article, 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study, by Chaolin Huang and colleagues. 1 On Aug 28, 2021, The Lancet published an Article, 1-year outcomes in hospital survivors with COVID-19: a longitudinal cohort study, by Lixue Huang and colleagues. 2 We received an inquiry from a researcher on data inconsistencies between these two Articles, and we sought an explanation from the corresponding author of the two papers. On Nov 7, 2022, Lancet editors were informed that inconsistencies between the 6-month and the 1-year data were due to “some variables in the dataset used for the 6-month paper were mistakenly disrupted in order”. In view of the extent of these data errors, we now issue an Expression of Concern about the 6-month paper 1 while we investigate further, including further statistical and clinical review of the corrected data. We will update this notice as soon as we have further information.
The corresponding author of both papers, Bin Cao of China’s National Center for Respiratory Medicine and the China-Japan Friendship Hospital in Beijing, has not responded to our request for comment.
A profile of Cao published in Lancet Infectious Diseases last March described him as “a leading researcher in pneumonia and influenza” who “has been instrumental in increasing knowledge about COVID-19.” In addition to the follow-up study of hospitalized COVID patients:
- Cao’s seminal papers during the COVID-19 pandemic include the first report of the clinical characteristics of COVID-19 patients in Wuhan, the description of the risk factors for mortality for adult inpatients, and the results of trials testing the use of antiviral drugs, including lopinavir-ritonavir, to treat COVID-19 in China.
We reached out to The Lancet’s press office and Richard Horton, the journal’s editor-in-chief, and received this statement:
- The Lancet Group treats all communications between editors and authors or readers as confidential. Investigations are continuing, and the Expression of Concern will be updated as soon as we have further information to share. More information about our policies is available here:
This year, The Lancet overtook the New England Journal of Medicine as the medical journal with the highest impact factor, in large part due to the papers it published about COVID-19.
We’ve counted retractions for three of those papers, most notably a paper about the use of the drug hydroxychloroquine that claimed to use medical data from a company called Surgisphere. As Retraction Watch readers may remember, the article was retracted after sleuths questioned if the data were real, and the company would not produce it for review.
This article first appeared on Retraction Watch.
Covid vax prevents death in children regardless of variant
The vaccine’s effectiveness against infection in the short term has been established, as has the waning effectiveness of the vaccine over time, wrote Juan Manuel Castelli, MD, of the Ministry of Health of Argentina, Buenos Aires, and colleagues, in the British Medical Journal.
However, data on the impact of vaccine effectiveness on mortality in children and adolescents are limited, especially during periods of omicron variant dominance, the researchers said.
In their new study, the researchers reviewed data from 844,460 children and adolescents aged 3-17 years from the National Surveillance System and the Nominalized Federal Vaccination Registry of Argentina, during a time that included a period of omicron dominance.
Argentina began vaccinating adolescents aged 12-17 years against COVID-19 in August 2021 and added children aged 3-11 years in October 2021. Those aged 12-17 years who were considered fully vaccinated received two doses of either Pfizer-BioNTech and/or Moderna vaccines, and fully-vaccinated 3- to 11-year-olds received two doses of Sinopharm vaccine.
The average time from the second vaccine dose to a COVID-19 test was 66 days for those aged 12-17 years and 54 days for 3- to 11-year-olds. The researchers matched COVID-19 cases with uninfected controls, and a total of 139,321 cases were included in the analysis.
Overall, the estimated vaccine effectiveness against COVID-19 was 64.2% during a period of delta dominance (61.2% in children aged 3-11 years and 66.8% in adolescents aged 12-17 years).
During a period of omicron dominance, estimated vaccine effectiveness was 19.9% across all ages (15.9% and 26.0% for younger and older age groups, respectively).
Effectiveness of the vaccine decreased over time, regardless of the dominant variant, but the decline was greater during the omicron dominant period, the researchers noted. During the omicron period, effectiveness in children aged 3-11 years decreased from 37.6% at 15-30 days postvaccination to 2.0% at 60 days or longer after vaccination. In adolescents aged 12-17 years, vaccine effectiveness during the omicron period decreased from 55.8% at 15-30 days postvaccination to 12.4% at 60 days or longer after vaccination.
Despite the waning protection against infection, the vaccine’s effectiveness against death from COVID-19 was 66.9% in children aged 3-11 years and 97.6% in adolescents aged 12-17 during the period of omicron dominance, the researchers noted.
The results are consistent with similar studies showing a decreased vaccine effectiveness against infection but a persistent effectiveness against deaths over time, the researchers wrote in the discussion section of their paper.
“Our results suggest that the primary vaccination schedule is effective in preventing mortality in children and adolescents with COVID-19 regardless of the circulating SARS-CoV-2 variant,” the researchers said.
Study limitations and strengths
The study was limited by several factors including the incomplete data on symptoms and hospital admissions, the possible impact of unmeasured confounding variables, and the observational design that prevents conclusions of causality, the researchers noted. However, the results were strengthened by the large sample size and access to detailed vaccination records, they said.
Both heterologous and homologous mRNA vaccine schedules showed similar effectiveness in preventing short-term infection and mortality from COVID-19 during periods of differing dominant variants, they noted.
The study findings support the vaccination of children against COVID-19 as an important public health measure to prevent mortality in children and adolescents, they concluded.
Data support value of vaccination, outside experts say
“COVID vaccines may not be as effective over time as the gene variants in the SARS-CoV-2 virus change,” Adrienne G. Randolph, MD, a pediatrician at Harvard Medical School and Boston Children’s Hospital, said in an interview. “Therefore, it is essential to assess vaccine effectiveness over time to look at effectiveness against variants and duration of effectiveness.” Dr. Randolph, who was not involved in the study, said she was not surprised by the findings, which she described as consistent with data from the United States. “COVID vaccines are very effective against preventing life-threatening disease, but the effectiveness against less severe illness for COVID vaccines is not as effective against Omicron,” she noted.
The take-home message for clinicians is that it’s important to get children vaccinated against COVID to prevent severe and life-threatening illness, said Dr. Randolph. “Although these cases are uncommon in children, it is not possible to predict which children will be the most severely affected by COVID,” she emphasized.
However, “we need more data on the new COVID booster vaccines in children that are designed to be more effective against Omicron’s newer variants,” Dr. Randolph said in an interview. “We also need more data on COVID vaccine effectiveness in the youngest children, under 5 years of age, and data on vaccinating mothers to prevent COVID in infants,” she said.
Tim Joos, MD, a Seattle-based clinician who practices a combination of internal medicine and pediatrics, agreed that future research should continue to assess how the new COVID boosters are faring against new variants, noting that the current study did not include data from children who received the new bivalent vaccine.
“The methodology of this study uses a test negative case control design which is common for estimating vaccine effectiveness post-release of a vaccine, but is subject to biases,” Dr. Joos explained. “These are not the clean effectiveness numbers of the prospective randomized control trials that we are used to hearing about when a vaccine is first being approved.”
“Nevertheless, the study reinforces the initial manufacturers’ studies that the vaccines are effective at preventing infection in the pediatric population,” Dr. Joos said in an interview. The current study also reinforces the effectiveness of vaccines in preventing “the rare but devastating mortality from COVID-19 in the pediatric population.”
Commenting on other research showing an increasing ratio of COVID deaths among vaccinated individuals compared to total COVID deaths, he noted that this finding is “likely reflecting a denominator effect of rapidly declining COVID deaths overall,” partly from the vaccines and partly from immunity after previous natural infection.
The study received no outside funding. The researchers, Dr. Randolph, and Dr. Joos had no financial conflicts to disclose. Dr. Joos serves on the Editorial Advisory Board of Pediatric News.
The vaccine’s effectiveness against infection in the short term has been established, as has the waning effectiveness of the vaccine over time, wrote Juan Manuel Castelli, MD, of the Ministry of Health of Argentina, Buenos Aires, and colleagues, in the British Medical Journal.
However, data on the impact of vaccine effectiveness on mortality in children and adolescents are limited, especially during periods of omicron variant dominance, the researchers said.
In their new study, the researchers reviewed data from 844,460 children and adolescents aged 3-17 years from the National Surveillance System and the Nominalized Federal Vaccination Registry of Argentina, during a time that included a period of omicron dominance.
Argentina began vaccinating adolescents aged 12-17 years against COVID-19 in August 2021 and added children aged 3-11 years in October 2021. Those aged 12-17 years who were considered fully vaccinated received two doses of either Pfizer-BioNTech and/or Moderna vaccines, and fully-vaccinated 3- to 11-year-olds received two doses of Sinopharm vaccine.
The average time from the second vaccine dose to a COVID-19 test was 66 days for those aged 12-17 years and 54 days for 3- to 11-year-olds. The researchers matched COVID-19 cases with uninfected controls, and a total of 139,321 cases were included in the analysis.
Overall, the estimated vaccine effectiveness against COVID-19 was 64.2% during a period of delta dominance (61.2% in children aged 3-11 years and 66.8% in adolescents aged 12-17 years).
During a period of omicron dominance, estimated vaccine effectiveness was 19.9% across all ages (15.9% and 26.0% for younger and older age groups, respectively).
Effectiveness of the vaccine decreased over time, regardless of the dominant variant, but the decline was greater during the omicron dominant period, the researchers noted. During the omicron period, effectiveness in children aged 3-11 years decreased from 37.6% at 15-30 days postvaccination to 2.0% at 60 days or longer after vaccination. In adolescents aged 12-17 years, vaccine effectiveness during the omicron period decreased from 55.8% at 15-30 days postvaccination to 12.4% at 60 days or longer after vaccination.
Despite the waning protection against infection, the vaccine’s effectiveness against death from COVID-19 was 66.9% in children aged 3-11 years and 97.6% in adolescents aged 12-17 during the period of omicron dominance, the researchers noted.
The results are consistent with similar studies showing a decreased vaccine effectiveness against infection but a persistent effectiveness against deaths over time, the researchers wrote in the discussion section of their paper.
“Our results suggest that the primary vaccination schedule is effective in preventing mortality in children and adolescents with COVID-19 regardless of the circulating SARS-CoV-2 variant,” the researchers said.
Study limitations and strengths
The study was limited by several factors including the incomplete data on symptoms and hospital admissions, the possible impact of unmeasured confounding variables, and the observational design that prevents conclusions of causality, the researchers noted. However, the results were strengthened by the large sample size and access to detailed vaccination records, they said.
Both heterologous and homologous mRNA vaccine schedules showed similar effectiveness in preventing short-term infection and mortality from COVID-19 during periods of differing dominant variants, they noted.
The study findings support the vaccination of children against COVID-19 as an important public health measure to prevent mortality in children and adolescents, they concluded.
Data support value of vaccination, outside experts say
“COVID vaccines may not be as effective over time as the gene variants in the SARS-CoV-2 virus change,” Adrienne G. Randolph, MD, a pediatrician at Harvard Medical School and Boston Children’s Hospital, said in an interview. “Therefore, it is essential to assess vaccine effectiveness over time to look at effectiveness against variants and duration of effectiveness.” Dr. Randolph, who was not involved in the study, said she was not surprised by the findings, which she described as consistent with data from the United States. “COVID vaccines are very effective against preventing life-threatening disease, but the effectiveness against less severe illness for COVID vaccines is not as effective against Omicron,” she noted.
The take-home message for clinicians is that it’s important to get children vaccinated against COVID to prevent severe and life-threatening illness, said Dr. Randolph. “Although these cases are uncommon in children, it is not possible to predict which children will be the most severely affected by COVID,” she emphasized.
However, “we need more data on the new COVID booster vaccines in children that are designed to be more effective against Omicron’s newer variants,” Dr. Randolph said in an interview. “We also need more data on COVID vaccine effectiveness in the youngest children, under 5 years of age, and data on vaccinating mothers to prevent COVID in infants,” she said.
Tim Joos, MD, a Seattle-based clinician who practices a combination of internal medicine and pediatrics, agreed that future research should continue to assess how the new COVID boosters are faring against new variants, noting that the current study did not include data from children who received the new bivalent vaccine.
“The methodology of this study uses a test negative case control design which is common for estimating vaccine effectiveness post-release of a vaccine, but is subject to biases,” Dr. Joos explained. “These are not the clean effectiveness numbers of the prospective randomized control trials that we are used to hearing about when a vaccine is first being approved.”
“Nevertheless, the study reinforces the initial manufacturers’ studies that the vaccines are effective at preventing infection in the pediatric population,” Dr. Joos said in an interview. The current study also reinforces the effectiveness of vaccines in preventing “the rare but devastating mortality from COVID-19 in the pediatric population.”
Commenting on other research showing an increasing ratio of COVID deaths among vaccinated individuals compared to total COVID deaths, he noted that this finding is “likely reflecting a denominator effect of rapidly declining COVID deaths overall,” partly from the vaccines and partly from immunity after previous natural infection.
The study received no outside funding. The researchers, Dr. Randolph, and Dr. Joos had no financial conflicts to disclose. Dr. Joos serves on the Editorial Advisory Board of Pediatric News.
The vaccine’s effectiveness against infection in the short term has been established, as has the waning effectiveness of the vaccine over time, wrote Juan Manuel Castelli, MD, of the Ministry of Health of Argentina, Buenos Aires, and colleagues, in the British Medical Journal.
However, data on the impact of vaccine effectiveness on mortality in children and adolescents are limited, especially during periods of omicron variant dominance, the researchers said.
In their new study, the researchers reviewed data from 844,460 children and adolescents aged 3-17 years from the National Surveillance System and the Nominalized Federal Vaccination Registry of Argentina, during a time that included a period of omicron dominance.
Argentina began vaccinating adolescents aged 12-17 years against COVID-19 in August 2021 and added children aged 3-11 years in October 2021. Those aged 12-17 years who were considered fully vaccinated received two doses of either Pfizer-BioNTech and/or Moderna vaccines, and fully-vaccinated 3- to 11-year-olds received two doses of Sinopharm vaccine.
The average time from the second vaccine dose to a COVID-19 test was 66 days for those aged 12-17 years and 54 days for 3- to 11-year-olds. The researchers matched COVID-19 cases with uninfected controls, and a total of 139,321 cases were included in the analysis.
Overall, the estimated vaccine effectiveness against COVID-19 was 64.2% during a period of delta dominance (61.2% in children aged 3-11 years and 66.8% in adolescents aged 12-17 years).
During a period of omicron dominance, estimated vaccine effectiveness was 19.9% across all ages (15.9% and 26.0% for younger and older age groups, respectively).
Effectiveness of the vaccine decreased over time, regardless of the dominant variant, but the decline was greater during the omicron dominant period, the researchers noted. During the omicron period, effectiveness in children aged 3-11 years decreased from 37.6% at 15-30 days postvaccination to 2.0% at 60 days or longer after vaccination. In adolescents aged 12-17 years, vaccine effectiveness during the omicron period decreased from 55.8% at 15-30 days postvaccination to 12.4% at 60 days or longer after vaccination.
Despite the waning protection against infection, the vaccine’s effectiveness against death from COVID-19 was 66.9% in children aged 3-11 years and 97.6% in adolescents aged 12-17 during the period of omicron dominance, the researchers noted.
The results are consistent with similar studies showing a decreased vaccine effectiveness against infection but a persistent effectiveness against deaths over time, the researchers wrote in the discussion section of their paper.
“Our results suggest that the primary vaccination schedule is effective in preventing mortality in children and adolescents with COVID-19 regardless of the circulating SARS-CoV-2 variant,” the researchers said.
Study limitations and strengths
The study was limited by several factors including the incomplete data on symptoms and hospital admissions, the possible impact of unmeasured confounding variables, and the observational design that prevents conclusions of causality, the researchers noted. However, the results were strengthened by the large sample size and access to detailed vaccination records, they said.
Both heterologous and homologous mRNA vaccine schedules showed similar effectiveness in preventing short-term infection and mortality from COVID-19 during periods of differing dominant variants, they noted.
The study findings support the vaccination of children against COVID-19 as an important public health measure to prevent mortality in children and adolescents, they concluded.
Data support value of vaccination, outside experts say
“COVID vaccines may not be as effective over time as the gene variants in the SARS-CoV-2 virus change,” Adrienne G. Randolph, MD, a pediatrician at Harvard Medical School and Boston Children’s Hospital, said in an interview. “Therefore, it is essential to assess vaccine effectiveness over time to look at effectiveness against variants and duration of effectiveness.” Dr. Randolph, who was not involved in the study, said she was not surprised by the findings, which she described as consistent with data from the United States. “COVID vaccines are very effective against preventing life-threatening disease, but the effectiveness against less severe illness for COVID vaccines is not as effective against Omicron,” she noted.
The take-home message for clinicians is that it’s important to get children vaccinated against COVID to prevent severe and life-threatening illness, said Dr. Randolph. “Although these cases are uncommon in children, it is not possible to predict which children will be the most severely affected by COVID,” she emphasized.
However, “we need more data on the new COVID booster vaccines in children that are designed to be more effective against Omicron’s newer variants,” Dr. Randolph said in an interview. “We also need more data on COVID vaccine effectiveness in the youngest children, under 5 years of age, and data on vaccinating mothers to prevent COVID in infants,” she said.
Tim Joos, MD, a Seattle-based clinician who practices a combination of internal medicine and pediatrics, agreed that future research should continue to assess how the new COVID boosters are faring against new variants, noting that the current study did not include data from children who received the new bivalent vaccine.
“The methodology of this study uses a test negative case control design which is common for estimating vaccine effectiveness post-release of a vaccine, but is subject to biases,” Dr. Joos explained. “These are not the clean effectiveness numbers of the prospective randomized control trials that we are used to hearing about when a vaccine is first being approved.”
“Nevertheless, the study reinforces the initial manufacturers’ studies that the vaccines are effective at preventing infection in the pediatric population,” Dr. Joos said in an interview. The current study also reinforces the effectiveness of vaccines in preventing “the rare but devastating mortality from COVID-19 in the pediatric population.”
Commenting on other research showing an increasing ratio of COVID deaths among vaccinated individuals compared to total COVID deaths, he noted that this finding is “likely reflecting a denominator effect of rapidly declining COVID deaths overall,” partly from the vaccines and partly from immunity after previous natural infection.
The study received no outside funding. The researchers, Dr. Randolph, and Dr. Joos had no financial conflicts to disclose. Dr. Joos serves on the Editorial Advisory Board of Pediatric News.
FROM THE BMJ
A new use for dating apps: Chasing STDs
Heather Meador and Anna Herber-Downey use dating apps on the job – and their boss knows it.
Both are public health nurses employed by Linn County Public Health in eastern Iowa. They’ve learned that dating apps are the most efficient way to inform users that people they previously met on the sites may have exposed them to sexually transmitted infections.
A nationwide surge in STIs, also known as STDs – with reported cases of gonorrhea and syphilis increasing 10% and 7%, respectively, from 2019 to 2020, according to the Centers for Disease Control and Prevention – isn’t sparing Iowa. The duo has found that the telephone call, a traditional method of contact tracing, no longer works well.
“When I started 12 years ago, we called everyone,” said Ms. Meador, the county health department’s clinical branch supervisor. “It’s getting harder and harder to just call someone on the phone.”
Even texting is ineffective, they said. And people aren’t necessarily answering messages on Facebook. The dating apps are where they’re at.
Because many people are meeting sex partners online – via sites like Grindr or Snapchat, which are headquartered in West Hollywood and Santa Monica, Calif., respectively – contact tracers often don’t have much information to go on, just a screen name or a picture.
So, about a year ago, Ms. Meador and her colleagues got approval from their bosses at the local level to build profiles on the app, through which they can contact the sex partners of infected people.
Traditionally, contact tracers interview people infected with an STI about their recent encounters and then reach out to those partners to tell them about the potential exposure.
Linn County contact tracers use the apps throughout their workday. Grindr, in particular, relies on geolocation, showing users matches who are close by. So the tracers use the apps when they’re out and about, hoping to wander into the same neighborhoods as the person diagnosed with an STI. Sometimes users “tap” the contract tracers to see whether they’re interested – in dating, that is.
When the public health officials spot someone they’re looking for, they send a message asking for a call. It’s a successful method: Ms. Herber-Downey estimated they make an initial contact 75% of the time.
Linn County’s decision to move online comes as STI rates rise nationally, funding to fight them falls, and people adopt new technologies to meet people and seek fun. “STIs are increasing way faster than the funding we have,” said Leo Parker, director of prevention programs for the National Coalition of STD Directors, all while public health departments – many underfunded – are grappling with new behaviors.
“Social media companies have billions; we have tens of thousands,” said Jeffrey Klausner, MD, MPH, a University of Southern California, Los Angeles, public health professor, who previously served as San Francisco’s director of STD prevention and control services. That funding disparity means few public health departments have staff members who can go online. “It’s only really in major cities that they have anyone who’s tasked for that,” Dr. Klausner said.
Even when departments have enough employees to take on the challenge, institutional support can be lacking. Some public health officials question employees who log into the apps. Dr. Klausner once testified on behalf of a Ventura County, Calif., contact tracer who was fired for using sex sites for work.
But with people migrating online to meet partners, following them there makes sense. “We’re now in a digital age,” Mr. Parker said. Individuals might not be out, or might be questioning their identity, making online venues comfortable, anonymous spaces for romance – which, in turn, means people are harder to reach face-to-face, at least at first.
What’s more, online spaces like Grindr are effective public health tools beyond contact tracing. They can be useful ways to get the word out about public health concerns.
Mr. Parker and the Linn County officials said public service announcements on dating apps – advocating for condom use or sharing the business hours for sexual health clinics – do seem to lead people to services. “We do have individuals coming in, saying, ‘I saw you had free testing. I saw it on Grindr,’ ” Mr. Parker said.
Grindr, which touts itself as the biggest dating app focused on LGBTQ+ people, pushes out messages and information to its members, said Jack Harrison-Quintana, director of Grindr for Equality. That engagement intensified during a 2015 meningitis outbreak among LGBTQ+ communities in Chicago, for example.
During that outbreak, the app sent citywide messages about vaccination. Then Mr. Harrison-Quintana took advantage of the service’s design: Using the site’s geolocating capabilities, Grindr workers targeted messages to specific neighborhoods. “We could go in and really go block to block and say, ‘Is this where the cases are showing up?’ ” he said. If so, they sent more messages to that area.
That campaign encouraged further efforts from the app, which regularly sends public health messages about everything from COVID-19 to monkeypox to the platform’s base of roughly 11 million monthly users. Grindr also allows users to display their HIV status and indicate whether they’re vaccinated against COVID, monkeypox, and meningitis.
There are a couple of things Grindr won’t do, however. The company won’t allow public health departments to create institutional accounts. And it won’t allow automated notifications about STI exposures to be sent to users.
That’s due to privacy concerns, the company said, despite calls from public health advocates to deploy better messaging features. Grindr believes that a government presence on the app would be too intrusive and that even anonymous notifications would allow users to trace infections back to their source. (When asked about public health officials who join the site on their own, company spokesperson Patrick Lenihan said: “Individuals are free to say something like ‘I’m a public health professional – ask me about my work!’ in their profile and are free to discuss sexual and public health matters however they see fit.”)
Grindr’s position – however disappointing to some in the public health world – reflects a longtime balancing act attempted by the private sector, which aims to square government concerns with users’ privacy interests.
Dr. Klausner pointed to a 1999 syphilis outbreak in San Francisco as one of the first times he saw how those interests could be at odds. The outbreak was traced to an AOL chatroom. Based on his research, Dr. Klausner said it seemed as though people could go online and “get a sex partner faster than you can get a pizza delivered.”
But persuading New York–based Time Warner, eventually AOL’s corporate parent, to cooperate was time-intensive and tricky – gaining entrée into the chatroom required help from the New York attorney general’s office.
The online industry has advanced since then, Dr. Klausner said. He helped one service develop a system to send digital postcards to potentially exposed people. “Congratulations, you got syphilis,” the postcards read. “They were edgy postcards,” he said, although some options were less “snarky.”
Overall, however, the dating app world is still “bifurcated,” he said. For public health efforts, apps that appeal to LGBTQ+ users are generally more helpful than those that predominantly cater to heterosexual clients.
That’s due to the community’s history with sexual health, explained Jen Hecht, a leader of Building Healthy Online Communities, a public health group partnering with dating apps. “Folks in the queer community have – what – 30, 40 years of thinking about HIV?” she said.
Even though STIs affect everyone, “the norm and the expectation is not there” for straight-focused dating apps, she said. Indeed, neither Match Group nor Bumble – the corporations with the biggest apps focused on heterosexual dating, both based in Texas – responded to multiple requests for comment from KHN.
But users, at least so far, seem to appreciate the app-based interventions. Mr. Harrison-Quintana said Grindr has landed on a just-the-facts approach to conveying health information. He has never received any backlash, “which has been very nice.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Heather Meador and Anna Herber-Downey use dating apps on the job – and their boss knows it.
Both are public health nurses employed by Linn County Public Health in eastern Iowa. They’ve learned that dating apps are the most efficient way to inform users that people they previously met on the sites may have exposed them to sexually transmitted infections.
A nationwide surge in STIs, also known as STDs – with reported cases of gonorrhea and syphilis increasing 10% and 7%, respectively, from 2019 to 2020, according to the Centers for Disease Control and Prevention – isn’t sparing Iowa. The duo has found that the telephone call, a traditional method of contact tracing, no longer works well.
“When I started 12 years ago, we called everyone,” said Ms. Meador, the county health department’s clinical branch supervisor. “It’s getting harder and harder to just call someone on the phone.”
Even texting is ineffective, they said. And people aren’t necessarily answering messages on Facebook. The dating apps are where they’re at.
Because many people are meeting sex partners online – via sites like Grindr or Snapchat, which are headquartered in West Hollywood and Santa Monica, Calif., respectively – contact tracers often don’t have much information to go on, just a screen name or a picture.
So, about a year ago, Ms. Meador and her colleagues got approval from their bosses at the local level to build profiles on the app, through which they can contact the sex partners of infected people.
Traditionally, contact tracers interview people infected with an STI about their recent encounters and then reach out to those partners to tell them about the potential exposure.
Linn County contact tracers use the apps throughout their workday. Grindr, in particular, relies on geolocation, showing users matches who are close by. So the tracers use the apps when they’re out and about, hoping to wander into the same neighborhoods as the person diagnosed with an STI. Sometimes users “tap” the contract tracers to see whether they’re interested – in dating, that is.
When the public health officials spot someone they’re looking for, they send a message asking for a call. It’s a successful method: Ms. Herber-Downey estimated they make an initial contact 75% of the time.
Linn County’s decision to move online comes as STI rates rise nationally, funding to fight them falls, and people adopt new technologies to meet people and seek fun. “STIs are increasing way faster than the funding we have,” said Leo Parker, director of prevention programs for the National Coalition of STD Directors, all while public health departments – many underfunded – are grappling with new behaviors.
“Social media companies have billions; we have tens of thousands,” said Jeffrey Klausner, MD, MPH, a University of Southern California, Los Angeles, public health professor, who previously served as San Francisco’s director of STD prevention and control services. That funding disparity means few public health departments have staff members who can go online. “It’s only really in major cities that they have anyone who’s tasked for that,” Dr. Klausner said.
Even when departments have enough employees to take on the challenge, institutional support can be lacking. Some public health officials question employees who log into the apps. Dr. Klausner once testified on behalf of a Ventura County, Calif., contact tracer who was fired for using sex sites for work.
But with people migrating online to meet partners, following them there makes sense. “We’re now in a digital age,” Mr. Parker said. Individuals might not be out, or might be questioning their identity, making online venues comfortable, anonymous spaces for romance – which, in turn, means people are harder to reach face-to-face, at least at first.
What’s more, online spaces like Grindr are effective public health tools beyond contact tracing. They can be useful ways to get the word out about public health concerns.
Mr. Parker and the Linn County officials said public service announcements on dating apps – advocating for condom use or sharing the business hours for sexual health clinics – do seem to lead people to services. “We do have individuals coming in, saying, ‘I saw you had free testing. I saw it on Grindr,’ ” Mr. Parker said.
Grindr, which touts itself as the biggest dating app focused on LGBTQ+ people, pushes out messages and information to its members, said Jack Harrison-Quintana, director of Grindr for Equality. That engagement intensified during a 2015 meningitis outbreak among LGBTQ+ communities in Chicago, for example.
During that outbreak, the app sent citywide messages about vaccination. Then Mr. Harrison-Quintana took advantage of the service’s design: Using the site’s geolocating capabilities, Grindr workers targeted messages to specific neighborhoods. “We could go in and really go block to block and say, ‘Is this where the cases are showing up?’ ” he said. If so, they sent more messages to that area.
That campaign encouraged further efforts from the app, which regularly sends public health messages about everything from COVID-19 to monkeypox to the platform’s base of roughly 11 million monthly users. Grindr also allows users to display their HIV status and indicate whether they’re vaccinated against COVID, monkeypox, and meningitis.
There are a couple of things Grindr won’t do, however. The company won’t allow public health departments to create institutional accounts. And it won’t allow automated notifications about STI exposures to be sent to users.
That’s due to privacy concerns, the company said, despite calls from public health advocates to deploy better messaging features. Grindr believes that a government presence on the app would be too intrusive and that even anonymous notifications would allow users to trace infections back to their source. (When asked about public health officials who join the site on their own, company spokesperson Patrick Lenihan said: “Individuals are free to say something like ‘I’m a public health professional – ask me about my work!’ in their profile and are free to discuss sexual and public health matters however they see fit.”)
Grindr’s position – however disappointing to some in the public health world – reflects a longtime balancing act attempted by the private sector, which aims to square government concerns with users’ privacy interests.
Dr. Klausner pointed to a 1999 syphilis outbreak in San Francisco as one of the first times he saw how those interests could be at odds. The outbreak was traced to an AOL chatroom. Based on his research, Dr. Klausner said it seemed as though people could go online and “get a sex partner faster than you can get a pizza delivered.”
But persuading New York–based Time Warner, eventually AOL’s corporate parent, to cooperate was time-intensive and tricky – gaining entrée into the chatroom required help from the New York attorney general’s office.
The online industry has advanced since then, Dr. Klausner said. He helped one service develop a system to send digital postcards to potentially exposed people. “Congratulations, you got syphilis,” the postcards read. “They were edgy postcards,” he said, although some options were less “snarky.”
Overall, however, the dating app world is still “bifurcated,” he said. For public health efforts, apps that appeal to LGBTQ+ users are generally more helpful than those that predominantly cater to heterosexual clients.
That’s due to the community’s history with sexual health, explained Jen Hecht, a leader of Building Healthy Online Communities, a public health group partnering with dating apps. “Folks in the queer community have – what – 30, 40 years of thinking about HIV?” she said.
Even though STIs affect everyone, “the norm and the expectation is not there” for straight-focused dating apps, she said. Indeed, neither Match Group nor Bumble – the corporations with the biggest apps focused on heterosexual dating, both based in Texas – responded to multiple requests for comment from KHN.
But users, at least so far, seem to appreciate the app-based interventions. Mr. Harrison-Quintana said Grindr has landed on a just-the-facts approach to conveying health information. He has never received any backlash, “which has been very nice.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Heather Meador and Anna Herber-Downey use dating apps on the job – and their boss knows it.
Both are public health nurses employed by Linn County Public Health in eastern Iowa. They’ve learned that dating apps are the most efficient way to inform users that people they previously met on the sites may have exposed them to sexually transmitted infections.
A nationwide surge in STIs, also known as STDs – with reported cases of gonorrhea and syphilis increasing 10% and 7%, respectively, from 2019 to 2020, according to the Centers for Disease Control and Prevention – isn’t sparing Iowa. The duo has found that the telephone call, a traditional method of contact tracing, no longer works well.
“When I started 12 years ago, we called everyone,” said Ms. Meador, the county health department’s clinical branch supervisor. “It’s getting harder and harder to just call someone on the phone.”
Even texting is ineffective, they said. And people aren’t necessarily answering messages on Facebook. The dating apps are where they’re at.
Because many people are meeting sex partners online – via sites like Grindr or Snapchat, which are headquartered in West Hollywood and Santa Monica, Calif., respectively – contact tracers often don’t have much information to go on, just a screen name or a picture.
So, about a year ago, Ms. Meador and her colleagues got approval from their bosses at the local level to build profiles on the app, through which they can contact the sex partners of infected people.
Traditionally, contact tracers interview people infected with an STI about their recent encounters and then reach out to those partners to tell them about the potential exposure.
Linn County contact tracers use the apps throughout their workday. Grindr, in particular, relies on geolocation, showing users matches who are close by. So the tracers use the apps when they’re out and about, hoping to wander into the same neighborhoods as the person diagnosed with an STI. Sometimes users “tap” the contract tracers to see whether they’re interested – in dating, that is.
When the public health officials spot someone they’re looking for, they send a message asking for a call. It’s a successful method: Ms. Herber-Downey estimated they make an initial contact 75% of the time.
Linn County’s decision to move online comes as STI rates rise nationally, funding to fight them falls, and people adopt new technologies to meet people and seek fun. “STIs are increasing way faster than the funding we have,” said Leo Parker, director of prevention programs for the National Coalition of STD Directors, all while public health departments – many underfunded – are grappling with new behaviors.
“Social media companies have billions; we have tens of thousands,” said Jeffrey Klausner, MD, MPH, a University of Southern California, Los Angeles, public health professor, who previously served as San Francisco’s director of STD prevention and control services. That funding disparity means few public health departments have staff members who can go online. “It’s only really in major cities that they have anyone who’s tasked for that,” Dr. Klausner said.
Even when departments have enough employees to take on the challenge, institutional support can be lacking. Some public health officials question employees who log into the apps. Dr. Klausner once testified on behalf of a Ventura County, Calif., contact tracer who was fired for using sex sites for work.
But with people migrating online to meet partners, following them there makes sense. “We’re now in a digital age,” Mr. Parker said. Individuals might not be out, or might be questioning their identity, making online venues comfortable, anonymous spaces for romance – which, in turn, means people are harder to reach face-to-face, at least at first.
What’s more, online spaces like Grindr are effective public health tools beyond contact tracing. They can be useful ways to get the word out about public health concerns.
Mr. Parker and the Linn County officials said public service announcements on dating apps – advocating for condom use or sharing the business hours for sexual health clinics – do seem to lead people to services. “We do have individuals coming in, saying, ‘I saw you had free testing. I saw it on Grindr,’ ” Mr. Parker said.
Grindr, which touts itself as the biggest dating app focused on LGBTQ+ people, pushes out messages and information to its members, said Jack Harrison-Quintana, director of Grindr for Equality. That engagement intensified during a 2015 meningitis outbreak among LGBTQ+ communities in Chicago, for example.
During that outbreak, the app sent citywide messages about vaccination. Then Mr. Harrison-Quintana took advantage of the service’s design: Using the site’s geolocating capabilities, Grindr workers targeted messages to specific neighborhoods. “We could go in and really go block to block and say, ‘Is this where the cases are showing up?’ ” he said. If so, they sent more messages to that area.
That campaign encouraged further efforts from the app, which regularly sends public health messages about everything from COVID-19 to monkeypox to the platform’s base of roughly 11 million monthly users. Grindr also allows users to display their HIV status and indicate whether they’re vaccinated against COVID, monkeypox, and meningitis.
There are a couple of things Grindr won’t do, however. The company won’t allow public health departments to create institutional accounts. And it won’t allow automated notifications about STI exposures to be sent to users.
That’s due to privacy concerns, the company said, despite calls from public health advocates to deploy better messaging features. Grindr believes that a government presence on the app would be too intrusive and that even anonymous notifications would allow users to trace infections back to their source. (When asked about public health officials who join the site on their own, company spokesperson Patrick Lenihan said: “Individuals are free to say something like ‘I’m a public health professional – ask me about my work!’ in their profile and are free to discuss sexual and public health matters however they see fit.”)
Grindr’s position – however disappointing to some in the public health world – reflects a longtime balancing act attempted by the private sector, which aims to square government concerns with users’ privacy interests.
Dr. Klausner pointed to a 1999 syphilis outbreak in San Francisco as one of the first times he saw how those interests could be at odds. The outbreak was traced to an AOL chatroom. Based on his research, Dr. Klausner said it seemed as though people could go online and “get a sex partner faster than you can get a pizza delivered.”
But persuading New York–based Time Warner, eventually AOL’s corporate parent, to cooperate was time-intensive and tricky – gaining entrée into the chatroom required help from the New York attorney general’s office.
The online industry has advanced since then, Dr. Klausner said. He helped one service develop a system to send digital postcards to potentially exposed people. “Congratulations, you got syphilis,” the postcards read. “They were edgy postcards,” he said, although some options were less “snarky.”
Overall, however, the dating app world is still “bifurcated,” he said. For public health efforts, apps that appeal to LGBTQ+ users are generally more helpful than those that predominantly cater to heterosexual clients.
That’s due to the community’s history with sexual health, explained Jen Hecht, a leader of Building Healthy Online Communities, a public health group partnering with dating apps. “Folks in the queer community have – what – 30, 40 years of thinking about HIV?” she said.
Even though STIs affect everyone, “the norm and the expectation is not there” for straight-focused dating apps, she said. Indeed, neither Match Group nor Bumble – the corporations with the biggest apps focused on heterosexual dating, both based in Texas – responded to multiple requests for comment from KHN.
But users, at least so far, seem to appreciate the app-based interventions. Mr. Harrison-Quintana said Grindr has landed on a just-the-facts approach to conveying health information. He has never received any backlash, “which has been very nice.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Newer agents for nosocomial pneumonia: The right drug for the right bug
“The right drug at the right time with the right dose for the right bug for the right duration.” That, said professor Kristina Crothers, MD, is the general guidance for optimizing antibiotic use (while awaiting an infectious disease consult). In her oral presentation at the annual meeting of the American College of Chest Physicians, “Choosing newer antibiotics for nosocomial pneumonia,” Dr. Crothers asked the question: “Beyond the guidelines: When should novel antimicrobials be used?”
Hospital-acquired pneumonia (HAP) and ventilator-associated pneumonia (VAP) are the most common nosocomial infections at 22%, and are the leading cause of death attributable to hospital-acquired infections. They increase mortality by 20%-50%, with an economic burden of about $40,000 per patient. The incidence of multidrug-resistant (MDR) organism infections varies widely by locality, but several factors increase the likelihood: prior broad-spectrum antibiotic exposure within the past 90 days; longer hospitalization; indwelling vascular devices; tracheostomy; and ventilator dependence. The Centers for Disease Control and Prevention lists as “Serious Threat” the HAP/VAP MDR organisms methicillin resistant Staphylococcus aureus (MRSA), Pseudomonas aeruginosa (PSA) with difficult-to-treat-resistance, and beta-lactamase producing Enterobacterales (ESBL). In the category of “Urgent Threat” the CDC lists: carbapenamase-resistant Enterobacterales (CRE) (carbapenamase producing or non–carbapenemase producing), and carbapenem-resistant Acinetobacter (CRAB), according to Dr. Crothers who is at the University of Washington Veterans Affairs Puget Sound Health Care System, Seattle.
Newer antibiotics for HAP/VAP that are still beyond the guidelines include telavancin and tedizolid as gram-positive agents, and as gram-negative ones: ceftazidime-avibactam, ceftolozane-tazobactam, cefiderocol, imipenem-cilastatin-relebactam and meropenem-vaborbactam, she added.
Tedizolid, Dr. Crothers stated, is a novel oxazolidinone, and is an alternative to vancomycin and linezolid for gram-positive HAP/VAP. In the VITAL noninferiority study versus linezolid with 726 patients, it was noninferior to linezolid for 28-day all-cause mortality (28% vs. 26%), but did not achieve noninferiority for investigator-assessed clinical cure (56% vs. 64%).
Televancin, a semisynthetic derivative of vancomycin, in the ATTAIN studies vs. vancomycin had overall similar cure rates. It is FDA-approved for S. aureus HAP/VAP but not other bacterial causes. It should be reserved for those who cannot receive vancomycin or linezolid, with normal renal function, according to Dr. Crothers. Excluded from first-line treatment of gram-positive HAP/VAP are daptomycin, ceftaroline, ceftobiprole, and tigecycline.
Ceftazidime-avibactam, a third-generation cephalosporin-plus novel beta-lactamase inhibitor has wide activity (Klebsiella pneumoniae, Enterobacter cloacae, Escherichia coli, Serratia marcescens, Proteus mirabilis, PSA and Haemophilus influenzae. It is also active against some extended-spectrum beta-lactamases (ESBLs), ampC beta-lactamases (AmpCs), and K. pneumoniae carbapenemase (KPC)–producing Enterobacterales, but not with metallo-beta-lactamases). Ceftazidime-avibactam is also indicated for HAP/VAP, and has a toxicity profile including nausea, vomiting, and diarrhea.
In the REPROVE trial of ceftazidime-avibactam vs. meropenem for 7-14 days with 527 clinically evaluable patients (37% K. pneumoniae, 30% P. aeruginosa, and 33%-35% VAP), the clinical cure at 21-25 days post randomization was 69% vs. 73%, respectively, with similar adverse events.
Ceftolozane-tazobactam, a novel fifth-generation cephalosporin plus a beta-lactamase inhibitor has activity against PSA including extensively drug-resistant PSA, AmpC, and ESBL-E, but it has limited activity against Acinetobacter and Stenotrophomonas. It is indicated for HAP/VAP, has reduced efficacy with creatine clearance of 50 mL/min or less, increases transaminases and renal impairment, and causes diarrhea. In ASPECT-NP (n = 726) ceftolozane-tazobactam versus meropenem for 8-14 days (HAP/VAP), showed a 28 day-mortality of 24% vs. 25%, respectively, with test of cure at 54% vs. 53% at 7-14 days post therapy. Adverse events were similar between groups.
Imipenem-cilastatin-relebactam, a novel beta-lactamase inhibitor plus carbapenem, is indicated for HAP/VAP and has activity against ESBL, CRE: KPC-producing Enterobacterales, PSA including AmpC. It can cause seizures (requires caution with central nervous system disorders and renal impairment). It increases transaminases, anemia, diarrhea, and reduces potassium and sodium. In RESTORE-IMI 2 (n = 537 with HAP/VAP) it was noninferior for 28-day all-cause mortality vs. piperacillin and tazobactam (16% vs. 21%), with similar adverse events.
Cefiderocol, a siderophore cephalosporin, is indicated for HAP/VAP. It has a wide spectrum of activity: ESBL, CRE, CR PSA, Stenotrophomonas maltophilia, Acinetobacter baumanii, Streptococcus.) It increases transaminases, diarrhea, and atrial fibrillation, and it reduces potassium and magnesium. In APEKS-NP versus linezolid plus cefiderocol or extended meropenem infusion (HAP/VAP n = 292; gram-negative pneumonia = 251; 60% invasive mechanical ventilation) it was noninferior for 14-day all-cause mortality (12.4% vs. 11.6%) with similar adverse events. In CREDIBLE-CR vs. best available therapy for carbapenem-resistant gram-negative infections, clinical cure rates were similar (50% vs. 53% in 59 HAP/VAP patients at 7 days), but with more deaths in the cefiderocol arm. Adverse events were > 90% in both groups and 34% vs. 19% died, mostly with Acinetobacter.
Meropenem-vaborbactam, a novel beta-lactamase inhibitor plus carbapenem, is approved and indicated for HAP/VAP in Europe. It has activity against MDR, Enterobacterales including CRE. Its toxicities include headache, phlebitis/infusion-site reactions and diarrhea. In TANGO-2 versus best available treatment for carbapenem-resistant Enterobacteriaceae (CRE) (n = 77, 47 with confirmed CRE), clinical cure was increased and mortality decreased compared with best available therapy. Treatment- and renal-related adverse events were lower for meropenem-vaborbactam.
In closing, Dr. Crothers cited advice from the paper by Tamma et al. (“Rethinking how Antibiotics are Prescribed” JAMA. 2018) about the need to review findings after therapy has been initiated to confirm the pneumonia diagnosis: Novel agents should be kept in reserve in the absence of MDR risk factors for MRSA and gram-negative bacilli; therapy should be deescalated after 48-72 hours if MDR organisms are not detected; and therapy should be directed to the specific organism detected. Most HAP and VAP in adults can be treated for 7 days, she added.
“Know indications for new therapeutic agents approved for nosocomial pneumonia,” she concluded.
Dr. Crothers reported having no disclosures.
“The right drug at the right time with the right dose for the right bug for the right duration.” That, said professor Kristina Crothers, MD, is the general guidance for optimizing antibiotic use (while awaiting an infectious disease consult). In her oral presentation at the annual meeting of the American College of Chest Physicians, “Choosing newer antibiotics for nosocomial pneumonia,” Dr. Crothers asked the question: “Beyond the guidelines: When should novel antimicrobials be used?”
Hospital-acquired pneumonia (HAP) and ventilator-associated pneumonia (VAP) are the most common nosocomial infections at 22%, and are the leading cause of death attributable to hospital-acquired infections. They increase mortality by 20%-50%, with an economic burden of about $40,000 per patient. The incidence of multidrug-resistant (MDR) organism infections varies widely by locality, but several factors increase the likelihood: prior broad-spectrum antibiotic exposure within the past 90 days; longer hospitalization; indwelling vascular devices; tracheostomy; and ventilator dependence. The Centers for Disease Control and Prevention lists as “Serious Threat” the HAP/VAP MDR organisms methicillin resistant Staphylococcus aureus (MRSA), Pseudomonas aeruginosa (PSA) with difficult-to-treat-resistance, and beta-lactamase producing Enterobacterales (ESBL). In the category of “Urgent Threat” the CDC lists: carbapenamase-resistant Enterobacterales (CRE) (carbapenamase producing or non–carbapenemase producing), and carbapenem-resistant Acinetobacter (CRAB), according to Dr. Crothers who is at the University of Washington Veterans Affairs Puget Sound Health Care System, Seattle.
Newer antibiotics for HAP/VAP that are still beyond the guidelines include telavancin and tedizolid as gram-positive agents, and as gram-negative ones: ceftazidime-avibactam, ceftolozane-tazobactam, cefiderocol, imipenem-cilastatin-relebactam and meropenem-vaborbactam, she added.
Tedizolid, Dr. Crothers stated, is a novel oxazolidinone, and is an alternative to vancomycin and linezolid for gram-positive HAP/VAP. In the VITAL noninferiority study versus linezolid with 726 patients, it was noninferior to linezolid for 28-day all-cause mortality (28% vs. 26%), but did not achieve noninferiority for investigator-assessed clinical cure (56% vs. 64%).
Televancin, a semisynthetic derivative of vancomycin, in the ATTAIN studies vs. vancomycin had overall similar cure rates. It is FDA-approved for S. aureus HAP/VAP but not other bacterial causes. It should be reserved for those who cannot receive vancomycin or linezolid, with normal renal function, according to Dr. Crothers. Excluded from first-line treatment of gram-positive HAP/VAP are daptomycin, ceftaroline, ceftobiprole, and tigecycline.
Ceftazidime-avibactam, a third-generation cephalosporin-plus novel beta-lactamase inhibitor has wide activity (Klebsiella pneumoniae, Enterobacter cloacae, Escherichia coli, Serratia marcescens, Proteus mirabilis, PSA and Haemophilus influenzae. It is also active against some extended-spectrum beta-lactamases (ESBLs), ampC beta-lactamases (AmpCs), and K. pneumoniae carbapenemase (KPC)–producing Enterobacterales, but not with metallo-beta-lactamases). Ceftazidime-avibactam is also indicated for HAP/VAP, and has a toxicity profile including nausea, vomiting, and diarrhea.
In the REPROVE trial of ceftazidime-avibactam vs. meropenem for 7-14 days with 527 clinically evaluable patients (37% K. pneumoniae, 30% P. aeruginosa, and 33%-35% VAP), the clinical cure at 21-25 days post randomization was 69% vs. 73%, respectively, with similar adverse events.
Ceftolozane-tazobactam, a novel fifth-generation cephalosporin plus a beta-lactamase inhibitor has activity against PSA including extensively drug-resistant PSA, AmpC, and ESBL-E, but it has limited activity against Acinetobacter and Stenotrophomonas. It is indicated for HAP/VAP, has reduced efficacy with creatine clearance of 50 mL/min or less, increases transaminases and renal impairment, and causes diarrhea. In ASPECT-NP (n = 726) ceftolozane-tazobactam versus meropenem for 8-14 days (HAP/VAP), showed a 28 day-mortality of 24% vs. 25%, respectively, with test of cure at 54% vs. 53% at 7-14 days post therapy. Adverse events were similar between groups.
Imipenem-cilastatin-relebactam, a novel beta-lactamase inhibitor plus carbapenem, is indicated for HAP/VAP and has activity against ESBL, CRE: KPC-producing Enterobacterales, PSA including AmpC. It can cause seizures (requires caution with central nervous system disorders and renal impairment). It increases transaminases, anemia, diarrhea, and reduces potassium and sodium. In RESTORE-IMI 2 (n = 537 with HAP/VAP) it was noninferior for 28-day all-cause mortality vs. piperacillin and tazobactam (16% vs. 21%), with similar adverse events.
Cefiderocol, a siderophore cephalosporin, is indicated for HAP/VAP. It has a wide spectrum of activity: ESBL, CRE, CR PSA, Stenotrophomonas maltophilia, Acinetobacter baumanii, Streptococcus.) It increases transaminases, diarrhea, and atrial fibrillation, and it reduces potassium and magnesium. In APEKS-NP versus linezolid plus cefiderocol or extended meropenem infusion (HAP/VAP n = 292; gram-negative pneumonia = 251; 60% invasive mechanical ventilation) it was noninferior for 14-day all-cause mortality (12.4% vs. 11.6%) with similar adverse events. In CREDIBLE-CR vs. best available therapy for carbapenem-resistant gram-negative infections, clinical cure rates were similar (50% vs. 53% in 59 HAP/VAP patients at 7 days), but with more deaths in the cefiderocol arm. Adverse events were > 90% in both groups and 34% vs. 19% died, mostly with Acinetobacter.
Meropenem-vaborbactam, a novel beta-lactamase inhibitor plus carbapenem, is approved and indicated for HAP/VAP in Europe. It has activity against MDR, Enterobacterales including CRE. Its toxicities include headache, phlebitis/infusion-site reactions and diarrhea. In TANGO-2 versus best available treatment for carbapenem-resistant Enterobacteriaceae (CRE) (n = 77, 47 with confirmed CRE), clinical cure was increased and mortality decreased compared with best available therapy. Treatment- and renal-related adverse events were lower for meropenem-vaborbactam.
In closing, Dr. Crothers cited advice from the paper by Tamma et al. (“Rethinking how Antibiotics are Prescribed” JAMA. 2018) about the need to review findings after therapy has been initiated to confirm the pneumonia diagnosis: Novel agents should be kept in reserve in the absence of MDR risk factors for MRSA and gram-negative bacilli; therapy should be deescalated after 48-72 hours if MDR organisms are not detected; and therapy should be directed to the specific organism detected. Most HAP and VAP in adults can be treated for 7 days, she added.
“Know indications for new therapeutic agents approved for nosocomial pneumonia,” she concluded.
Dr. Crothers reported having no disclosures.
“The right drug at the right time with the right dose for the right bug for the right duration.” That, said professor Kristina Crothers, MD, is the general guidance for optimizing antibiotic use (while awaiting an infectious disease consult). In her oral presentation at the annual meeting of the American College of Chest Physicians, “Choosing newer antibiotics for nosocomial pneumonia,” Dr. Crothers asked the question: “Beyond the guidelines: When should novel antimicrobials be used?”
Hospital-acquired pneumonia (HAP) and ventilator-associated pneumonia (VAP) are the most common nosocomial infections at 22%, and are the leading cause of death attributable to hospital-acquired infections. They increase mortality by 20%-50%, with an economic burden of about $40,000 per patient. The incidence of multidrug-resistant (MDR) organism infections varies widely by locality, but several factors increase the likelihood: prior broad-spectrum antibiotic exposure within the past 90 days; longer hospitalization; indwelling vascular devices; tracheostomy; and ventilator dependence. The Centers for Disease Control and Prevention lists as “Serious Threat” the HAP/VAP MDR organisms methicillin resistant Staphylococcus aureus (MRSA), Pseudomonas aeruginosa (PSA) with difficult-to-treat-resistance, and beta-lactamase producing Enterobacterales (ESBL). In the category of “Urgent Threat” the CDC lists: carbapenamase-resistant Enterobacterales (CRE) (carbapenamase producing or non–carbapenemase producing), and carbapenem-resistant Acinetobacter (CRAB), according to Dr. Crothers who is at the University of Washington Veterans Affairs Puget Sound Health Care System, Seattle.
Newer antibiotics for HAP/VAP that are still beyond the guidelines include telavancin and tedizolid as gram-positive agents, and as gram-negative ones: ceftazidime-avibactam, ceftolozane-tazobactam, cefiderocol, imipenem-cilastatin-relebactam and meropenem-vaborbactam, she added.
Tedizolid, Dr. Crothers stated, is a novel oxazolidinone, and is an alternative to vancomycin and linezolid for gram-positive HAP/VAP. In the VITAL noninferiority study versus linezolid with 726 patients, it was noninferior to linezolid for 28-day all-cause mortality (28% vs. 26%), but did not achieve noninferiority for investigator-assessed clinical cure (56% vs. 64%).
Televancin, a semisynthetic derivative of vancomycin, in the ATTAIN studies vs. vancomycin had overall similar cure rates. It is FDA-approved for S. aureus HAP/VAP but not other bacterial causes. It should be reserved for those who cannot receive vancomycin or linezolid, with normal renal function, according to Dr. Crothers. Excluded from first-line treatment of gram-positive HAP/VAP are daptomycin, ceftaroline, ceftobiprole, and tigecycline.
Ceftazidime-avibactam, a third-generation cephalosporin-plus novel beta-lactamase inhibitor has wide activity (Klebsiella pneumoniae, Enterobacter cloacae, Escherichia coli, Serratia marcescens, Proteus mirabilis, PSA and Haemophilus influenzae. It is also active against some extended-spectrum beta-lactamases (ESBLs), ampC beta-lactamases (AmpCs), and K. pneumoniae carbapenemase (KPC)–producing Enterobacterales, but not with metallo-beta-lactamases). Ceftazidime-avibactam is also indicated for HAP/VAP, and has a toxicity profile including nausea, vomiting, and diarrhea.
In the REPROVE trial of ceftazidime-avibactam vs. meropenem for 7-14 days with 527 clinically evaluable patients (37% K. pneumoniae, 30% P. aeruginosa, and 33%-35% VAP), the clinical cure at 21-25 days post randomization was 69% vs. 73%, respectively, with similar adverse events.
Ceftolozane-tazobactam, a novel fifth-generation cephalosporin plus a beta-lactamase inhibitor has activity against PSA including extensively drug-resistant PSA, AmpC, and ESBL-E, but it has limited activity against Acinetobacter and Stenotrophomonas. It is indicated for HAP/VAP, has reduced efficacy with creatine clearance of 50 mL/min or less, increases transaminases and renal impairment, and causes diarrhea. In ASPECT-NP (n = 726) ceftolozane-tazobactam versus meropenem for 8-14 days (HAP/VAP), showed a 28 day-mortality of 24% vs. 25%, respectively, with test of cure at 54% vs. 53% at 7-14 days post therapy. Adverse events were similar between groups.
Imipenem-cilastatin-relebactam, a novel beta-lactamase inhibitor plus carbapenem, is indicated for HAP/VAP and has activity against ESBL, CRE: KPC-producing Enterobacterales, PSA including AmpC. It can cause seizures (requires caution with central nervous system disorders and renal impairment). It increases transaminases, anemia, diarrhea, and reduces potassium and sodium. In RESTORE-IMI 2 (n = 537 with HAP/VAP) it was noninferior for 28-day all-cause mortality vs. piperacillin and tazobactam (16% vs. 21%), with similar adverse events.
Cefiderocol, a siderophore cephalosporin, is indicated for HAP/VAP. It has a wide spectrum of activity: ESBL, CRE, CR PSA, Stenotrophomonas maltophilia, Acinetobacter baumanii, Streptococcus.) It increases transaminases, diarrhea, and atrial fibrillation, and it reduces potassium and magnesium. In APEKS-NP versus linezolid plus cefiderocol or extended meropenem infusion (HAP/VAP n = 292; gram-negative pneumonia = 251; 60% invasive mechanical ventilation) it was noninferior for 14-day all-cause mortality (12.4% vs. 11.6%) with similar adverse events. In CREDIBLE-CR vs. best available therapy for carbapenem-resistant gram-negative infections, clinical cure rates were similar (50% vs. 53% in 59 HAP/VAP patients at 7 days), but with more deaths in the cefiderocol arm. Adverse events were > 90% in both groups and 34% vs. 19% died, mostly with Acinetobacter.
Meropenem-vaborbactam, a novel beta-lactamase inhibitor plus carbapenem, is approved and indicated for HAP/VAP in Europe. It has activity against MDR, Enterobacterales including CRE. Its toxicities include headache, phlebitis/infusion-site reactions and diarrhea. In TANGO-2 versus best available treatment for carbapenem-resistant Enterobacteriaceae (CRE) (n = 77, 47 with confirmed CRE), clinical cure was increased and mortality decreased compared with best available therapy. Treatment- and renal-related adverse events were lower for meropenem-vaborbactam.
In closing, Dr. Crothers cited advice from the paper by Tamma et al. (“Rethinking how Antibiotics are Prescribed” JAMA. 2018) about the need to review findings after therapy has been initiated to confirm the pneumonia diagnosis: Novel agents should be kept in reserve in the absence of MDR risk factors for MRSA and gram-negative bacilli; therapy should be deescalated after 48-72 hours if MDR organisms are not detected; and therapy should be directed to the specific organism detected. Most HAP and VAP in adults can be treated for 7 days, she added.
“Know indications for new therapeutic agents approved for nosocomial pneumonia,” she concluded.
Dr. Crothers reported having no disclosures.
FROM CHEST 2022
The surprising failure of vitamin D in deficient kids
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
My basic gripe is that you’ve got all these observational studies linking lower levels of vitamin D to everything from dementia to falls to cancer to COVID infection, and then you do a big randomized trial of supplementation and don’t see an effect.
And the explanation is that vitamin D is not necessarily the thing causing these bad outcomes; it’s a bystander – a canary in the coal mine. Your vitamin D level is a marker of your lifestyle; it’s higher in people who eat healthier foods, who exercise, and who spend more time out in the sun.
And yet ... if you were to ask me whether supplementing vitamin D in children with vitamin D deficiency would help them grow better and be healthier, I probably would have been on board for the idea.
And, it looks like, I would have been wrong.
Yes, it’s another negative randomized trial of vitamin D supplementation to add to the seemingly ever-growing body of literature suggesting that your money is better spent on a day at the park rather than buying D3 from your local GNC.
We are talking about this study, appearing in JAMA Pediatrics.
Briefly, 8,851 children from around Ulaanbaatar, Mongolia, were randomized to receive 14,000 international units of vitamin D3 or placebo every week for 3 years.
Before we get into the results of the study, I need to point out that this part of Mongolia has a high rate of vitamin D deficiency. Beyond that, a prior observational study by these authors had shown that lower vitamin D levels were linked to the risk of acquiring latent tuberculosis infection in this area. Other studies have linked vitamin D deficiency with poorer growth metrics in children. Given the global scourge that is TB (around 2 million deaths a year) and childhood malnutrition (around 10% of children around the world), vitamin D supplementation is incredibly attractive as a public health intervention. It is relatively low on side effects and, importantly, it is cheap – and thus scalable.
Back to the study. These kids had pretty poor vitamin D levels at baseline; 95% of them were deficient, based on the accepted standard of levels less than 20 ng/mL. Over 30% were severely deficient, with levels less than 10 ng/mL.
The initial purpose of this study was to see if supplementation would prevent TB, but that analysis, which was published a few months ago, was negative. Vitamin D levels went up dramatically in the intervention group – they were taking their pills – but there was no difference in the rate of latent TB infection, active TB, other respiratory infections, or even serum interferon gamma levels.
Nothing.
But to be fair, the TB seroconversion rate was lower than expected, potentially leading to an underpowered study.
Which brings us to the just-published analysis which moves away from infectious disease to something where vitamin D should have some stronger footing: growth.
Would the kids who were randomized to vitamin D, those same kids who got their vitamin D levels into the normal range over 3 years of supplementation, grow more or grow better than the kids who didn’t?
And, unfortunately, the answer is still no.
At the end of follow-up, height z scores were not different between the groups. BMI z scores were not different between the groups. Pubertal development was not different between the groups. This was true not only overall, but across various subgroups, including analyses of those kids who had vitamin D levels less than 10 ng/mL to start with.
So, what’s going on? There are two very broad possibilities we can endorse. First, there’s the idea that vitamin D supplementation simply doesn’t do much for health. This is supported, now, by a long string of large clinical trials that show no effect across a variety of disease states and predisease states. In other words, the observational data linking low vitamin D to bad outcomes is correlation, not causation.
Or we can take the tack of some vitamin D apologists and decide that this trial just got it wrong. Perhaps the dose wasn’t given correctly, or 3 years isn’t long enough to see a real difference, or the growth metrics were wrong, or vitamin D needs to be given alongside something else to really work and so on. This is fine; no study is perfect and there is always something to criticize, believe me. But we need to be careful not to fall into the baby-and-bathwater fallacy. Just because we think a study could have done something better, or differently, doesn’t mean we can ignore all the results. And as each new randomized trial of vitamin D supplementation comes out, it’s getting harder and harder to believe that these trialists keep getting their methods wrong. Maybe they are just testing something that doesn’t work.
What to do? Well, it should be obvious. If low vitamin D levels are linked to TB rates and poor growth but supplementation doesn’t fix the problem, then we have to fix what is upstream of the problem. We need to boost vitamin D levels not through supplements, but through nutrition, exercise, activity, and getting outside. That’s a randomized trial you can sign me up for any day.
Dr. Wilson is associate professor, department of medicine, Yale University, New Haven, Conn. He reported no relevant conflicts of interest.
A version of this video transcript first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
My basic gripe is that you’ve got all these observational studies linking lower levels of vitamin D to everything from dementia to falls to cancer to COVID infection, and then you do a big randomized trial of supplementation and don’t see an effect.
And the explanation is that vitamin D is not necessarily the thing causing these bad outcomes; it’s a bystander – a canary in the coal mine. Your vitamin D level is a marker of your lifestyle; it’s higher in people who eat healthier foods, who exercise, and who spend more time out in the sun.
And yet ... if you were to ask me whether supplementing vitamin D in children with vitamin D deficiency would help them grow better and be healthier, I probably would have been on board for the idea.
And, it looks like, I would have been wrong.
Yes, it’s another negative randomized trial of vitamin D supplementation to add to the seemingly ever-growing body of literature suggesting that your money is better spent on a day at the park rather than buying D3 from your local GNC.
We are talking about this study, appearing in JAMA Pediatrics.
Briefly, 8,851 children from around Ulaanbaatar, Mongolia, were randomized to receive 14,000 international units of vitamin D3 or placebo every week for 3 years.
Before we get into the results of the study, I need to point out that this part of Mongolia has a high rate of vitamin D deficiency. Beyond that, a prior observational study by these authors had shown that lower vitamin D levels were linked to the risk of acquiring latent tuberculosis infection in this area. Other studies have linked vitamin D deficiency with poorer growth metrics in children. Given the global scourge that is TB (around 2 million deaths a year) and childhood malnutrition (around 10% of children around the world), vitamin D supplementation is incredibly attractive as a public health intervention. It is relatively low on side effects and, importantly, it is cheap – and thus scalable.
Back to the study. These kids had pretty poor vitamin D levels at baseline; 95% of them were deficient, based on the accepted standard of levels less than 20 ng/mL. Over 30% were severely deficient, with levels less than 10 ng/mL.
The initial purpose of this study was to see if supplementation would prevent TB, but that analysis, which was published a few months ago, was negative. Vitamin D levels went up dramatically in the intervention group – they were taking their pills – but there was no difference in the rate of latent TB infection, active TB, other respiratory infections, or even serum interferon gamma levels.
Nothing.
But to be fair, the TB seroconversion rate was lower than expected, potentially leading to an underpowered study.
Which brings us to the just-published analysis which moves away from infectious disease to something where vitamin D should have some stronger footing: growth.
Would the kids who were randomized to vitamin D, those same kids who got their vitamin D levels into the normal range over 3 years of supplementation, grow more or grow better than the kids who didn’t?
And, unfortunately, the answer is still no.
At the end of follow-up, height z scores were not different between the groups. BMI z scores were not different between the groups. Pubertal development was not different between the groups. This was true not only overall, but across various subgroups, including analyses of those kids who had vitamin D levels less than 10 ng/mL to start with.
So, what’s going on? There are two very broad possibilities we can endorse. First, there’s the idea that vitamin D supplementation simply doesn’t do much for health. This is supported, now, by a long string of large clinical trials that show no effect across a variety of disease states and predisease states. In other words, the observational data linking low vitamin D to bad outcomes is correlation, not causation.
Or we can take the tack of some vitamin D apologists and decide that this trial just got it wrong. Perhaps the dose wasn’t given correctly, or 3 years isn’t long enough to see a real difference, or the growth metrics were wrong, or vitamin D needs to be given alongside something else to really work and so on. This is fine; no study is perfect and there is always something to criticize, believe me. But we need to be careful not to fall into the baby-and-bathwater fallacy. Just because we think a study could have done something better, or differently, doesn’t mean we can ignore all the results. And as each new randomized trial of vitamin D supplementation comes out, it’s getting harder and harder to believe that these trialists keep getting their methods wrong. Maybe they are just testing something that doesn’t work.
What to do? Well, it should be obvious. If low vitamin D levels are linked to TB rates and poor growth but supplementation doesn’t fix the problem, then we have to fix what is upstream of the problem. We need to boost vitamin D levels not through supplements, but through nutrition, exercise, activity, and getting outside. That’s a randomized trial you can sign me up for any day.
Dr. Wilson is associate professor, department of medicine, Yale University, New Haven, Conn. He reported no relevant conflicts of interest.
A version of this video transcript first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
My basic gripe is that you’ve got all these observational studies linking lower levels of vitamin D to everything from dementia to falls to cancer to COVID infection, and then you do a big randomized trial of supplementation and don’t see an effect.
And the explanation is that vitamin D is not necessarily the thing causing these bad outcomes; it’s a bystander – a canary in the coal mine. Your vitamin D level is a marker of your lifestyle; it’s higher in people who eat healthier foods, who exercise, and who spend more time out in the sun.
And yet ... if you were to ask me whether supplementing vitamin D in children with vitamin D deficiency would help them grow better and be healthier, I probably would have been on board for the idea.
And, it looks like, I would have been wrong.
Yes, it’s another negative randomized trial of vitamin D supplementation to add to the seemingly ever-growing body of literature suggesting that your money is better spent on a day at the park rather than buying D3 from your local GNC.
We are talking about this study, appearing in JAMA Pediatrics.
Briefly, 8,851 children from around Ulaanbaatar, Mongolia, were randomized to receive 14,000 international units of vitamin D3 or placebo every week for 3 years.
Before we get into the results of the study, I need to point out that this part of Mongolia has a high rate of vitamin D deficiency. Beyond that, a prior observational study by these authors had shown that lower vitamin D levels were linked to the risk of acquiring latent tuberculosis infection in this area. Other studies have linked vitamin D deficiency with poorer growth metrics in children. Given the global scourge that is TB (around 2 million deaths a year) and childhood malnutrition (around 10% of children around the world), vitamin D supplementation is incredibly attractive as a public health intervention. It is relatively low on side effects and, importantly, it is cheap – and thus scalable.
Back to the study. These kids had pretty poor vitamin D levels at baseline; 95% of them were deficient, based on the accepted standard of levels less than 20 ng/mL. Over 30% were severely deficient, with levels less than 10 ng/mL.
The initial purpose of this study was to see if supplementation would prevent TB, but that analysis, which was published a few months ago, was negative. Vitamin D levels went up dramatically in the intervention group – they were taking their pills – but there was no difference in the rate of latent TB infection, active TB, other respiratory infections, or even serum interferon gamma levels.
Nothing.
But to be fair, the TB seroconversion rate was lower than expected, potentially leading to an underpowered study.
Which brings us to the just-published analysis which moves away from infectious disease to something where vitamin D should have some stronger footing: growth.
Would the kids who were randomized to vitamin D, those same kids who got their vitamin D levels into the normal range over 3 years of supplementation, grow more or grow better than the kids who didn’t?
And, unfortunately, the answer is still no.
At the end of follow-up, height z scores were not different between the groups. BMI z scores were not different between the groups. Pubertal development was not different between the groups. This was true not only overall, but across various subgroups, including analyses of those kids who had vitamin D levels less than 10 ng/mL to start with.
So, what’s going on? There are two very broad possibilities we can endorse. First, there’s the idea that vitamin D supplementation simply doesn’t do much for health. This is supported, now, by a long string of large clinical trials that show no effect across a variety of disease states and predisease states. In other words, the observational data linking low vitamin D to bad outcomes is correlation, not causation.
Or we can take the tack of some vitamin D apologists and decide that this trial just got it wrong. Perhaps the dose wasn’t given correctly, or 3 years isn’t long enough to see a real difference, or the growth metrics were wrong, or vitamin D needs to be given alongside something else to really work and so on. This is fine; no study is perfect and there is always something to criticize, believe me. But we need to be careful not to fall into the baby-and-bathwater fallacy. Just because we think a study could have done something better, or differently, doesn’t mean we can ignore all the results. And as each new randomized trial of vitamin D supplementation comes out, it’s getting harder and harder to believe that these trialists keep getting their methods wrong. Maybe they are just testing something that doesn’t work.
What to do? Well, it should be obvious. If low vitamin D levels are linked to TB rates and poor growth but supplementation doesn’t fix the problem, then we have to fix what is upstream of the problem. We need to boost vitamin D levels not through supplements, but through nutrition, exercise, activity, and getting outside. That’s a randomized trial you can sign me up for any day.
Dr. Wilson is associate professor, department of medicine, Yale University, New Haven, Conn. He reported no relevant conflicts of interest.
A version of this video transcript first appeared on Medscape.com.
U.S. flu activity already at mid-season levels
according to the Centers of Disease Control and Prevention.
Nationally, 6% of all outpatient visits were because of flu or flu-like illness for the week of Nov. 13-19, up from 5.8% the previous week, the CDC’s Influenza Division said in its weekly FluView report.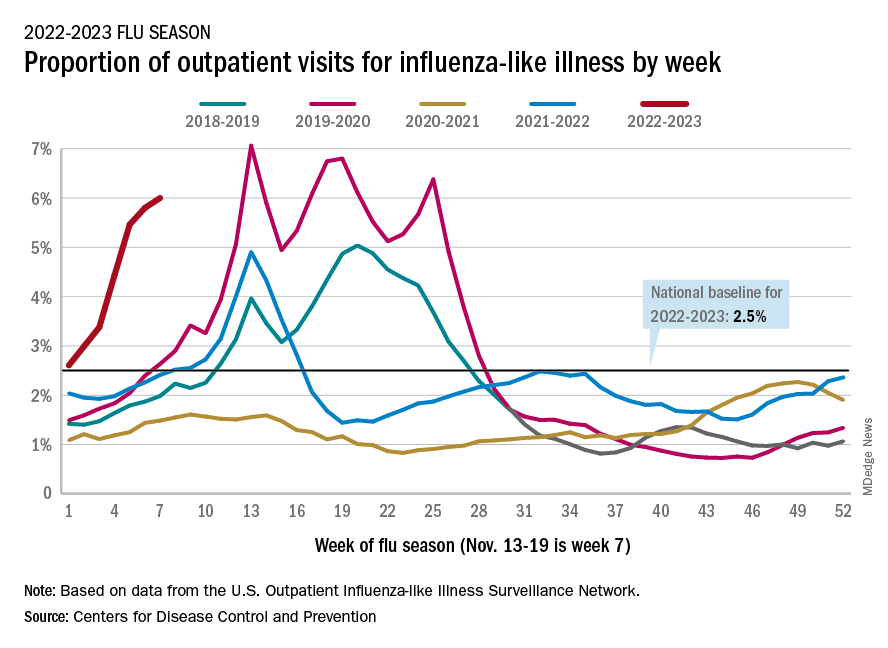
Those figures are the highest recorded in November since 2009, but the peak of the 2009-10 flu season occurred even earlier – the week of Oct. 18-24 – and the rate of flu-like illness had already dropped to just over 4.0% by Nov. 15-21 that year and continued to drop thereafter.
Although COVID-19 and respiratory syncytial virus (RSV) are included in the data from the CDC’s Outpatient Influenza-like Illness Surveillance Network, the agency did note that “seasonal influenza activity is elevated across the country” and estimated that “there have been at least 6.2 million illnesses, 53,000 hospitalizations, and 2,900 deaths from flu” during the 2022-23 season.
Total flu deaths include 11 reported in children as of Nov. 19, and children ages 0-4 had a higher proportion of visits for flu like-illness than other age groups.
The agency also said the cumulative hospitalization rate of 11.3 per 100,000 population “is higher than the rate observed in [the corresponding week of] every previous season since 2010-2011.” Adults 65 years and older have the highest cumulative rate, 25.9 per 100,000, for this year, compared with 20.7 for children 0-4; 11.1 for adults 50-64; 10.3 for children 5-17; and 5.6 for adults 18-49 years old, the CDC said.
A version of this article first appeared on WebMD.com.
according to the Centers of Disease Control and Prevention.
Nationally, 6% of all outpatient visits were because of flu or flu-like illness for the week of Nov. 13-19, up from 5.8% the previous week, the CDC’s Influenza Division said in its weekly FluView report.
Those figures are the highest recorded in November since 2009, but the peak of the 2009-10 flu season occurred even earlier – the week of Oct. 18-24 – and the rate of flu-like illness had already dropped to just over 4.0% by Nov. 15-21 that year and continued to drop thereafter.
Although COVID-19 and respiratory syncytial virus (RSV) are included in the data from the CDC’s Outpatient Influenza-like Illness Surveillance Network, the agency did note that “seasonal influenza activity is elevated across the country” and estimated that “there have been at least 6.2 million illnesses, 53,000 hospitalizations, and 2,900 deaths from flu” during the 2022-23 season.
Total flu deaths include 11 reported in children as of Nov. 19, and children ages 0-4 had a higher proportion of visits for flu like-illness than other age groups.
The agency also said the cumulative hospitalization rate of 11.3 per 100,000 population “is higher than the rate observed in [the corresponding week of] every previous season since 2010-2011.” Adults 65 years and older have the highest cumulative rate, 25.9 per 100,000, for this year, compared with 20.7 for children 0-4; 11.1 for adults 50-64; 10.3 for children 5-17; and 5.6 for adults 18-49 years old, the CDC said.
A version of this article first appeared on WebMD.com.
according to the Centers of Disease Control and Prevention.
Nationally, 6% of all outpatient visits were because of flu or flu-like illness for the week of Nov. 13-19, up from 5.8% the previous week, the CDC’s Influenza Division said in its weekly FluView report.
Those figures are the highest recorded in November since 2009, but the peak of the 2009-10 flu season occurred even earlier – the week of Oct. 18-24 – and the rate of flu-like illness had already dropped to just over 4.0% by Nov. 15-21 that year and continued to drop thereafter.
Although COVID-19 and respiratory syncytial virus (RSV) are included in the data from the CDC’s Outpatient Influenza-like Illness Surveillance Network, the agency did note that “seasonal influenza activity is elevated across the country” and estimated that “there have been at least 6.2 million illnesses, 53,000 hospitalizations, and 2,900 deaths from flu” during the 2022-23 season.
Total flu deaths include 11 reported in children as of Nov. 19, and children ages 0-4 had a higher proportion of visits for flu like-illness than other age groups.
The agency also said the cumulative hospitalization rate of 11.3 per 100,000 population “is higher than the rate observed in [the corresponding week of] every previous season since 2010-2011.” Adults 65 years and older have the highest cumulative rate, 25.9 per 100,000, for this year, compared with 20.7 for children 0-4; 11.1 for adults 50-64; 10.3 for children 5-17; and 5.6 for adults 18-49 years old, the CDC said.
A version of this article first appeared on WebMD.com.