User login
Children and COVID: Decline of summer surge continues
The continuing decline in COVID-19 incidence suggests the latest surge has peaked as new cases in children dropped for the 4th consecutive week, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
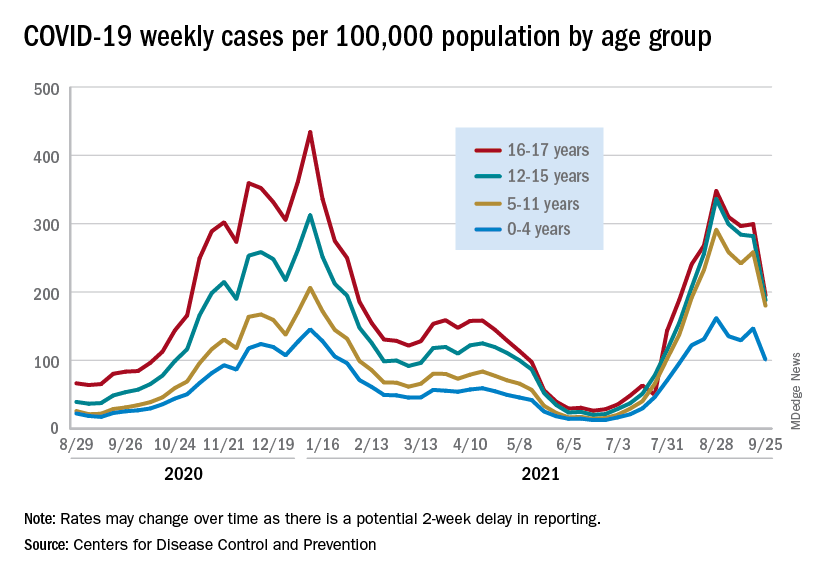
Preliminary data from the Centers for Disease Control and Prevention, however, show an uptick in new cases in late September, largely among younger children, that may indicate otherwise. Those data have a potential 2-week reporting delay, the CDC said on its COVID Data Tracker, so the most recent points on the graph (see above) could still go up.
. Those new cases made up almost 27% of all cases for the week, and the nearly 5.9 million child cases that have been reported since the start of the pandemic represent 16.2% of cases among Americans of all ages, the two groups said in their weekly COVID-19 report.
The CDC data on new cases by age group suggest that younger children have borne a heavier burden in the summer surge of COVID than they did last winter. The rate of new cases was not as high for 16- and 17-year-olds in the summer, but the other age groups all reached higher peaks than in the winter, including the 12- to 15-year-olds, who have been getting vaccinated since May, according to the COVID Data Tracker.
With vaccination approval getting closer for children under age 12 years, initiation in those already eligible continues to slide. Those aged 12-15 made up just 6.9% of new vaccinations during the 2 weeks from Sept. 21 to Oct. 4, and that figure has been dropping since July 13-26, when it was 14.1%. Vaccine initiation among 16- and 17-year-olds over that time has dropped by almost half, from 5.4% to 2.9%, the CDC data show.
All the vaccinations so far add up to this: Almost 55% of those aged 12-15 have gotten at least one dose of COVID vaccine, as have over 62% of those aged 16-17, and 52% of the older group is fully vaccinated, as is 44% of the younger group. Altogether, 10.8 million children were fully vaccinated as of Oct. 4, including those under 12 who may be participating in clinical trials or had a birth date entered incorrectly, the CDC said.
The continuing decline in COVID-19 incidence suggests the latest surge has peaked as new cases in children dropped for the 4th consecutive week, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.

Preliminary data from the Centers for Disease Control and Prevention, however, show an uptick in new cases in late September, largely among younger children, that may indicate otherwise. Those data have a potential 2-week reporting delay, the CDC said on its COVID Data Tracker, so the most recent points on the graph (see above) could still go up.
. Those new cases made up almost 27% of all cases for the week, and the nearly 5.9 million child cases that have been reported since the start of the pandemic represent 16.2% of cases among Americans of all ages, the two groups said in their weekly COVID-19 report.
The CDC data on new cases by age group suggest that younger children have borne a heavier burden in the summer surge of COVID than they did last winter. The rate of new cases was not as high for 16- and 17-year-olds in the summer, but the other age groups all reached higher peaks than in the winter, including the 12- to 15-year-olds, who have been getting vaccinated since May, according to the COVID Data Tracker.
With vaccination approval getting closer for children under age 12 years, initiation in those already eligible continues to slide. Those aged 12-15 made up just 6.9% of new vaccinations during the 2 weeks from Sept. 21 to Oct. 4, and that figure has been dropping since July 13-26, when it was 14.1%. Vaccine initiation among 16- and 17-year-olds over that time has dropped by almost half, from 5.4% to 2.9%, the CDC data show.
All the vaccinations so far add up to this: Almost 55% of those aged 12-15 have gotten at least one dose of COVID vaccine, as have over 62% of those aged 16-17, and 52% of the older group is fully vaccinated, as is 44% of the younger group. Altogether, 10.8 million children were fully vaccinated as of Oct. 4, including those under 12 who may be participating in clinical trials or had a birth date entered incorrectly, the CDC said.
The continuing decline in COVID-19 incidence suggests the latest surge has peaked as new cases in children dropped for the 4th consecutive week, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.

Preliminary data from the Centers for Disease Control and Prevention, however, show an uptick in new cases in late September, largely among younger children, that may indicate otherwise. Those data have a potential 2-week reporting delay, the CDC said on its COVID Data Tracker, so the most recent points on the graph (see above) could still go up.
. Those new cases made up almost 27% of all cases for the week, and the nearly 5.9 million child cases that have been reported since the start of the pandemic represent 16.2% of cases among Americans of all ages, the two groups said in their weekly COVID-19 report.
The CDC data on new cases by age group suggest that younger children have borne a heavier burden in the summer surge of COVID than they did last winter. The rate of new cases was not as high for 16- and 17-year-olds in the summer, but the other age groups all reached higher peaks than in the winter, including the 12- to 15-year-olds, who have been getting vaccinated since May, according to the COVID Data Tracker.
With vaccination approval getting closer for children under age 12 years, initiation in those already eligible continues to slide. Those aged 12-15 made up just 6.9% of new vaccinations during the 2 weeks from Sept. 21 to Oct. 4, and that figure has been dropping since July 13-26, when it was 14.1%. Vaccine initiation among 16- and 17-year-olds over that time has dropped by almost half, from 5.4% to 2.9%, the CDC data show.
All the vaccinations so far add up to this: Almost 55% of those aged 12-15 have gotten at least one dose of COVID vaccine, as have over 62% of those aged 16-17, and 52% of the older group is fully vaccinated, as is 44% of the younger group. Altogether, 10.8 million children were fully vaccinated as of Oct. 4, including those under 12 who may be participating in clinical trials or had a birth date entered incorrectly, the CDC said.
COVID-19: Two more cases of mucosal skin ulcers reported in male teens
Irish A similar case in an adolescent, also with ulcers affecting the mouth and penis, was reported earlier in 2021 in the United States.
“Our cases show that a swab for COVID-19 can be added to the list of investigations for mucosal and cutaneous rashes in children and probably adults,” said dermatologist Stephanie Bowe, MD, of South Infirmary-Victoria University Hospital in Cork, Ireland, in an interview. “Our patients seemed to improve with IV steroids, but there is not enough data to recommend them to all patients or for use in the different cutaneous presentations associated with COVID-19.”
The new case reports were presented at the 2021 meeting of the World Congress of Pediatric Dermatology and published in Pediatric Dermatology.
Researchers have noted that skin disorders linked to COVID-19 infection are different than those in adults. In children, the conditions include morbilliform rash, pernio-like acral lesions, urticaria, macular erythema, vesicular eruption, papulosquamous eruption, and retiform purpura. “The pathogenesis of each is not fully understood but likely related to the inflammatory response to COVID-19 and the various pathways within the body, which become activated,” Dr. Bowe said.
The first patient, a 17-year-old boy, presented at clinic 6 days after he’d been confirmed to be infected with COVID-19 and 8 days after developing fever and cough. “He had a 2-day history of conjunctivitis and ulceration of his oral mucosa, erythematous circumferential erosions of the glans penis with no other cutaneous findings,” the authors write in the report.
The boy “was distressed and embarrassed about his genital ulceration and also found eating very painful due to his oral ulceration,” Dr. Bowe said.
The second patient, a 14-year-old boy, was hospitalized 7 days after a positive COVID-19 test and 9 days after developing cough and fever. “He had a 5-day history of ulceration of the oral mucosa with mild conjunctivitis,” the authors wrote. “Ulceration of the glans penis developed on day 2 of admission.”
The 14-year-old was sicker than the 17-year-old boy, Dr. Bowe said. “He was unable to tolerate an oral diet for several days and had exquisite pain and vomiting with his coughing fits.”
This patient had a history of recurrent herpes labialis, but it’s unclear whether herpes simplex virus (HSV) played a role in the COVID-19–related case. “There is a possibility that the patient was more susceptible to viral cutaneous reactions during COVID-19 infection, but we didn’t have any definite history of HSV infection at the time of mucositis,” Dr. Bowe said. “We also didn’t have any swabs positive for HSV even though several were done at the time.”
Both patients received IV steroids – hydrocortisone at 100 mg 3 times daily for 3 days. This treatment was used “because of deterioration in symptoms and COVID-19 infection,” Dr. Bowe said. “IV steroids were used for respiratory symptoms of COVID-19, so we felt these cutaneous symptoms may have also been caused by an inflammatory response and might benefit from steroids. There was very little literature about this specific situation, though.”
She added that intravenous steroids wouldn’t be appropriate for most pediatric patients, and noted that “their use is controversial in the literature for erythema multiforme and RIME.”
In addition, the patients received betamethasone valerate 0.1% ointment once daily, hydrocortisone 2.5 mg buccal tablets 4 times daily, analgesia with acetaminophen and ibuprofen, and intravenous hydration. The first patient also received prednisolone 1% eye drops, while the second patient was given lidocaine hydrochloride mouthwash and total parenteral nutrition for 5 days.
The patients were discharged after 4 and 14 days, respectively.
Dermatologists in Massachusetts reported a similar case earlier in 2021 in a 17-year-old boy who was positive for COVID-19 and presented with “shallow erosions of the vermilion lips and hard palate, circumferential erythematous erosions of the periurethral glans penis, and five small vesicles on the trunk and upper extremities.”
The patient received betamethasone valerate 0.1% ointment for the lips and penis, intraoral dexamethasone solution, viscous lidocaine, acetaminophen, and ibuprofen. He also received oral prednisone at approximately 1 mg/kg daily for 4 consecutive days after worsening oral pain. A recurrence of oral pain 3 months later was resolved with a higher and longer treatment with oral prednisone.
Dermatologists have also reported cases of erythema multiforme lesions of the mucosa in adults with COVID-19. One case was reported in Iran, and the other in France.
The authors report no study funding and disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Irish A similar case in an adolescent, also with ulcers affecting the mouth and penis, was reported earlier in 2021 in the United States.
“Our cases show that a swab for COVID-19 can be added to the list of investigations for mucosal and cutaneous rashes in children and probably adults,” said dermatologist Stephanie Bowe, MD, of South Infirmary-Victoria University Hospital in Cork, Ireland, in an interview. “Our patients seemed to improve with IV steroids, but there is not enough data to recommend them to all patients or for use in the different cutaneous presentations associated with COVID-19.”
The new case reports were presented at the 2021 meeting of the World Congress of Pediatric Dermatology and published in Pediatric Dermatology.
Researchers have noted that skin disorders linked to COVID-19 infection are different than those in adults. In children, the conditions include morbilliform rash, pernio-like acral lesions, urticaria, macular erythema, vesicular eruption, papulosquamous eruption, and retiform purpura. “The pathogenesis of each is not fully understood but likely related to the inflammatory response to COVID-19 and the various pathways within the body, which become activated,” Dr. Bowe said.
The first patient, a 17-year-old boy, presented at clinic 6 days after he’d been confirmed to be infected with COVID-19 and 8 days after developing fever and cough. “He had a 2-day history of conjunctivitis and ulceration of his oral mucosa, erythematous circumferential erosions of the glans penis with no other cutaneous findings,” the authors write in the report.
The boy “was distressed and embarrassed about his genital ulceration and also found eating very painful due to his oral ulceration,” Dr. Bowe said.
The second patient, a 14-year-old boy, was hospitalized 7 days after a positive COVID-19 test and 9 days after developing cough and fever. “He had a 5-day history of ulceration of the oral mucosa with mild conjunctivitis,” the authors wrote. “Ulceration of the glans penis developed on day 2 of admission.”
The 14-year-old was sicker than the 17-year-old boy, Dr. Bowe said. “He was unable to tolerate an oral diet for several days and had exquisite pain and vomiting with his coughing fits.”
This patient had a history of recurrent herpes labialis, but it’s unclear whether herpes simplex virus (HSV) played a role in the COVID-19–related case. “There is a possibility that the patient was more susceptible to viral cutaneous reactions during COVID-19 infection, but we didn’t have any definite history of HSV infection at the time of mucositis,” Dr. Bowe said. “We also didn’t have any swabs positive for HSV even though several were done at the time.”
Both patients received IV steroids – hydrocortisone at 100 mg 3 times daily for 3 days. This treatment was used “because of deterioration in symptoms and COVID-19 infection,” Dr. Bowe said. “IV steroids were used for respiratory symptoms of COVID-19, so we felt these cutaneous symptoms may have also been caused by an inflammatory response and might benefit from steroids. There was very little literature about this specific situation, though.”
She added that intravenous steroids wouldn’t be appropriate for most pediatric patients, and noted that “their use is controversial in the literature for erythema multiforme and RIME.”
In addition, the patients received betamethasone valerate 0.1% ointment once daily, hydrocortisone 2.5 mg buccal tablets 4 times daily, analgesia with acetaminophen and ibuprofen, and intravenous hydration. The first patient also received prednisolone 1% eye drops, while the second patient was given lidocaine hydrochloride mouthwash and total parenteral nutrition for 5 days.
The patients were discharged after 4 and 14 days, respectively.
Dermatologists in Massachusetts reported a similar case earlier in 2021 in a 17-year-old boy who was positive for COVID-19 and presented with “shallow erosions of the vermilion lips and hard palate, circumferential erythematous erosions of the periurethral glans penis, and five small vesicles on the trunk and upper extremities.”
The patient received betamethasone valerate 0.1% ointment for the lips and penis, intraoral dexamethasone solution, viscous lidocaine, acetaminophen, and ibuprofen. He also received oral prednisone at approximately 1 mg/kg daily for 4 consecutive days after worsening oral pain. A recurrence of oral pain 3 months later was resolved with a higher and longer treatment with oral prednisone.
Dermatologists have also reported cases of erythema multiforme lesions of the mucosa in adults with COVID-19. One case was reported in Iran, and the other in France.
The authors report no study funding and disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Irish A similar case in an adolescent, also with ulcers affecting the mouth and penis, was reported earlier in 2021 in the United States.
“Our cases show that a swab for COVID-19 can be added to the list of investigations for mucosal and cutaneous rashes in children and probably adults,” said dermatologist Stephanie Bowe, MD, of South Infirmary-Victoria University Hospital in Cork, Ireland, in an interview. “Our patients seemed to improve with IV steroids, but there is not enough data to recommend them to all patients or for use in the different cutaneous presentations associated with COVID-19.”
The new case reports were presented at the 2021 meeting of the World Congress of Pediatric Dermatology and published in Pediatric Dermatology.
Researchers have noted that skin disorders linked to COVID-19 infection are different than those in adults. In children, the conditions include morbilliform rash, pernio-like acral lesions, urticaria, macular erythema, vesicular eruption, papulosquamous eruption, and retiform purpura. “The pathogenesis of each is not fully understood but likely related to the inflammatory response to COVID-19 and the various pathways within the body, which become activated,” Dr. Bowe said.
The first patient, a 17-year-old boy, presented at clinic 6 days after he’d been confirmed to be infected with COVID-19 and 8 days after developing fever and cough. “He had a 2-day history of conjunctivitis and ulceration of his oral mucosa, erythematous circumferential erosions of the glans penis with no other cutaneous findings,” the authors write in the report.
The boy “was distressed and embarrassed about his genital ulceration and also found eating very painful due to his oral ulceration,” Dr. Bowe said.
The second patient, a 14-year-old boy, was hospitalized 7 days after a positive COVID-19 test and 9 days after developing cough and fever. “He had a 5-day history of ulceration of the oral mucosa with mild conjunctivitis,” the authors wrote. “Ulceration of the glans penis developed on day 2 of admission.”
The 14-year-old was sicker than the 17-year-old boy, Dr. Bowe said. “He was unable to tolerate an oral diet for several days and had exquisite pain and vomiting with his coughing fits.”
This patient had a history of recurrent herpes labialis, but it’s unclear whether herpes simplex virus (HSV) played a role in the COVID-19–related case. “There is a possibility that the patient was more susceptible to viral cutaneous reactions during COVID-19 infection, but we didn’t have any definite history of HSV infection at the time of mucositis,” Dr. Bowe said. “We also didn’t have any swabs positive for HSV even though several were done at the time.”
Both patients received IV steroids – hydrocortisone at 100 mg 3 times daily for 3 days. This treatment was used “because of deterioration in symptoms and COVID-19 infection,” Dr. Bowe said. “IV steroids were used for respiratory symptoms of COVID-19, so we felt these cutaneous symptoms may have also been caused by an inflammatory response and might benefit from steroids. There was very little literature about this specific situation, though.”
She added that intravenous steroids wouldn’t be appropriate for most pediatric patients, and noted that “their use is controversial in the literature for erythema multiforme and RIME.”
In addition, the patients received betamethasone valerate 0.1% ointment once daily, hydrocortisone 2.5 mg buccal tablets 4 times daily, analgesia with acetaminophen and ibuprofen, and intravenous hydration. The first patient also received prednisolone 1% eye drops, while the second patient was given lidocaine hydrochloride mouthwash and total parenteral nutrition for 5 days.
The patients were discharged after 4 and 14 days, respectively.
Dermatologists in Massachusetts reported a similar case earlier in 2021 in a 17-year-old boy who was positive for COVID-19 and presented with “shallow erosions of the vermilion lips and hard palate, circumferential erythematous erosions of the periurethral glans penis, and five small vesicles on the trunk and upper extremities.”
The patient received betamethasone valerate 0.1% ointment for the lips and penis, intraoral dexamethasone solution, viscous lidocaine, acetaminophen, and ibuprofen. He also received oral prednisone at approximately 1 mg/kg daily for 4 consecutive days after worsening oral pain. A recurrence of oral pain 3 months later was resolved with a higher and longer treatment with oral prednisone.
Dermatologists have also reported cases of erythema multiforme lesions of the mucosa in adults with COVID-19. One case was reported in Iran, and the other in France.
The authors report no study funding and disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Extension study finds dupilumab effective for up to 1 year in teens with AD
in a phase 3, open-label extension trial, researchers reported.
At 1 year, 86% of 50 remaining patients with weights under 60 kg (132 lb) had achieved 75% improvement on the Eczema Area and Severity Index (EASI-75, and 77% of 51 remaining patients with weights over 60 kg reached that level of clearance. Only 5 (1.7%) of 294 patients had serious treatment-emergent adverse events (TEAEs).
The findings back up a perception that patients can stay on dupilumab for some time instead of having to switch from one biologic to another after a few years, study coauthor Eric Simpson, MD, professor of dermatology, Oregon Health & Science University, Portland, said in an interview. He added that the drug’s long-term safety profile is “very reassuring.”
The industry-funded findings of the study were released in a poster at the 2021 meeting of the World Congress of Pediatric Dermatology.
The FDA approved dupilumab (Dupixent), an interleukin-4 receptor alpha antagonist, for treating AD in adults in 2017; it is now approved for treating patients ages 6 years and older with moderate to severe atopic dermatitis whose disease is not adequately controlled with topicals.
The new study tracked patients who received at least 300 mg dupilumab subcutaneously every 4 weeks. The dose could be increased if needed to improve clinical response to once every 2 weeks (200 mg if baseline weight was <60 kg; 300 mg if ≥60 kg).
At 52 weeks, 37% of 52 patients with weights under 60 kg reached an Investigator Global Assessment (IGA) of 0/1, a level that had been fairly steady since week 16 (n = 146). Among 51 heavier patients, 49% reached an IGA of 0/1 at 52 weeks; this percentage grew steadily since baseline.
The mean percentage change in EASI was –87% in the lower-weight group (n = 50) at 52 weeks and –80.1% in the larger-weight group (n = 51). The majority of the reduction in EASI occurred in the first 4 weeks of treatment.
At 52 weeks, the mean Children’s Dermatology Life Quality Index level, which judges the effect of AD on life, was judged as “small” (low) in 71 patients. At baseline, the mean level among 189 patients was “moderate.” The levels dipped below “moderate” at week 4 and never rose above “small” after that.
“Treatment-emergent adverse events reported in ≥5% of patients were nasopharyngitis (21.1%), AD (19.4%), upper respiratory tract infection (12.4%), headache (9.4%), and oropharyngeal pain (5.7%),” the investigators wrote in the poster. They add that 6.7% of patients experienced injection-site reactions, and 8.7% of patients experienced treatment-emergent “narrow conjunctivitis,” which includes conjunctivitis, allergic conjunctivitis, bacterial conjunctivitis, viral conjunctivitis, and atopic keratoconjunctivitis.
Dr. Simpson noted that cases of conjunctivitis fell over time. It’s not clear why this adverse effect appears, he said.
He said that the findings reflect his own experience in clinic. Many of his adolescent patients took part in early dupilumab trials, he said, and dozens have been taking the drug for more than 5 years. “They just seem to get better and better,” he said.
University of Minnesota, Minneapolis, dermatologist Sheilagh Maguiness, MD, who wasn’t involved with the study, said in an interview that dupilumab remains “the safest, most effective and evidence-based therapy we had for children with moderate to severe atopic dermatitis.”
The new study’s findings are “very reassuring,” she said, and similar to those in a 2021 report that tracked long-term use of the drug in children aged 6-11.
Like Dr. Simpson, Dr. Maguiness said many pediatric patients at her clinic have stayed on the drug for more than 5 years. They still have “sustained improvement in skin disease and in their quality of life as well”
There are, however, still questions about dupilumab treatment. “For children who have responded well, when could we consider dose reduction or discontinuation? I have done this successfully just a handful of times, but I would love to see data about what percentage of pediatric patients experience rebound disease after coming off the drug and after what duration of treatment,” she said. “Another mystery that will be very interesting to unravel is the question as to whether or not early treatment with dupilumab may attenuate other atopic diseases.”
Dr. Maguiness added that “another issue specific to pediatric use of dupilumab is the recommendation surrounding vaccinations. This is an issue that should be studied in terms of antibody response and safety surrounding vaccinations, particularly as we are eagerly awaiting a pediatric FDA approval for the COVID-19 vaccine in children.”
She also urged colleagues to push back against insurers who resist paying for dupilumab. “Whether prescribing this medication on or off label, insurance companies are often requiring patients to try and fail other traditional immunosuppressive medications such as methotrexate, cyclosporine, or to pursue phototherapy,” she said. “Oftentimes, these are not practical or even safe options for children for a multitude of reasons. Don’t be shy about advocating for your patients by second- or even third-level appeals to try and gain approval for children who are in need of treatment.”
The study was funded by Sanofi Genzyme and Regeneron Pharmaceuticals. The study authors reported various disclosures. Dr. Simpson reported investigator and consultant fee relationships from various pharmaceutical companies. Dr. Maguiness was an investigator for one of the initial pediatric dupilumab trials.
A version of this article first appeared on Medscape.com.
in a phase 3, open-label extension trial, researchers reported.
At 1 year, 86% of 50 remaining patients with weights under 60 kg (132 lb) had achieved 75% improvement on the Eczema Area and Severity Index (EASI-75, and 77% of 51 remaining patients with weights over 60 kg reached that level of clearance. Only 5 (1.7%) of 294 patients had serious treatment-emergent adverse events (TEAEs).
The findings back up a perception that patients can stay on dupilumab for some time instead of having to switch from one biologic to another after a few years, study coauthor Eric Simpson, MD, professor of dermatology, Oregon Health & Science University, Portland, said in an interview. He added that the drug’s long-term safety profile is “very reassuring.”
The industry-funded findings of the study were released in a poster at the 2021 meeting of the World Congress of Pediatric Dermatology.
The FDA approved dupilumab (Dupixent), an interleukin-4 receptor alpha antagonist, for treating AD in adults in 2017; it is now approved for treating patients ages 6 years and older with moderate to severe atopic dermatitis whose disease is not adequately controlled with topicals.
The new study tracked patients who received at least 300 mg dupilumab subcutaneously every 4 weeks. The dose could be increased if needed to improve clinical response to once every 2 weeks (200 mg if baseline weight was <60 kg; 300 mg if ≥60 kg).
At 52 weeks, 37% of 52 patients with weights under 60 kg reached an Investigator Global Assessment (IGA) of 0/1, a level that had been fairly steady since week 16 (n = 146). Among 51 heavier patients, 49% reached an IGA of 0/1 at 52 weeks; this percentage grew steadily since baseline.
The mean percentage change in EASI was –87% in the lower-weight group (n = 50) at 52 weeks and –80.1% in the larger-weight group (n = 51). The majority of the reduction in EASI occurred in the first 4 weeks of treatment.
At 52 weeks, the mean Children’s Dermatology Life Quality Index level, which judges the effect of AD on life, was judged as “small” (low) in 71 patients. At baseline, the mean level among 189 patients was “moderate.” The levels dipped below “moderate” at week 4 and never rose above “small” after that.
“Treatment-emergent adverse events reported in ≥5% of patients were nasopharyngitis (21.1%), AD (19.4%), upper respiratory tract infection (12.4%), headache (9.4%), and oropharyngeal pain (5.7%),” the investigators wrote in the poster. They add that 6.7% of patients experienced injection-site reactions, and 8.7% of patients experienced treatment-emergent “narrow conjunctivitis,” which includes conjunctivitis, allergic conjunctivitis, bacterial conjunctivitis, viral conjunctivitis, and atopic keratoconjunctivitis.
Dr. Simpson noted that cases of conjunctivitis fell over time. It’s not clear why this adverse effect appears, he said.
He said that the findings reflect his own experience in clinic. Many of his adolescent patients took part in early dupilumab trials, he said, and dozens have been taking the drug for more than 5 years. “They just seem to get better and better,” he said.
University of Minnesota, Minneapolis, dermatologist Sheilagh Maguiness, MD, who wasn’t involved with the study, said in an interview that dupilumab remains “the safest, most effective and evidence-based therapy we had for children with moderate to severe atopic dermatitis.”
The new study’s findings are “very reassuring,” she said, and similar to those in a 2021 report that tracked long-term use of the drug in children aged 6-11.
Like Dr. Simpson, Dr. Maguiness said many pediatric patients at her clinic have stayed on the drug for more than 5 years. They still have “sustained improvement in skin disease and in their quality of life as well”
There are, however, still questions about dupilumab treatment. “For children who have responded well, when could we consider dose reduction or discontinuation? I have done this successfully just a handful of times, but I would love to see data about what percentage of pediatric patients experience rebound disease after coming off the drug and after what duration of treatment,” she said. “Another mystery that will be very interesting to unravel is the question as to whether or not early treatment with dupilumab may attenuate other atopic diseases.”
Dr. Maguiness added that “another issue specific to pediatric use of dupilumab is the recommendation surrounding vaccinations. This is an issue that should be studied in terms of antibody response and safety surrounding vaccinations, particularly as we are eagerly awaiting a pediatric FDA approval for the COVID-19 vaccine in children.”
She also urged colleagues to push back against insurers who resist paying for dupilumab. “Whether prescribing this medication on or off label, insurance companies are often requiring patients to try and fail other traditional immunosuppressive medications such as methotrexate, cyclosporine, or to pursue phototherapy,” she said. “Oftentimes, these are not practical or even safe options for children for a multitude of reasons. Don’t be shy about advocating for your patients by second- or even third-level appeals to try and gain approval for children who are in need of treatment.”
The study was funded by Sanofi Genzyme and Regeneron Pharmaceuticals. The study authors reported various disclosures. Dr. Simpson reported investigator and consultant fee relationships from various pharmaceutical companies. Dr. Maguiness was an investigator for one of the initial pediatric dupilumab trials.
A version of this article first appeared on Medscape.com.
in a phase 3, open-label extension trial, researchers reported.
At 1 year, 86% of 50 remaining patients with weights under 60 kg (132 lb) had achieved 75% improvement on the Eczema Area and Severity Index (EASI-75, and 77% of 51 remaining patients with weights over 60 kg reached that level of clearance. Only 5 (1.7%) of 294 patients had serious treatment-emergent adverse events (TEAEs).
The findings back up a perception that patients can stay on dupilumab for some time instead of having to switch from one biologic to another after a few years, study coauthor Eric Simpson, MD, professor of dermatology, Oregon Health & Science University, Portland, said in an interview. He added that the drug’s long-term safety profile is “very reassuring.”
The industry-funded findings of the study were released in a poster at the 2021 meeting of the World Congress of Pediatric Dermatology.
The FDA approved dupilumab (Dupixent), an interleukin-4 receptor alpha antagonist, for treating AD in adults in 2017; it is now approved for treating patients ages 6 years and older with moderate to severe atopic dermatitis whose disease is not adequately controlled with topicals.
The new study tracked patients who received at least 300 mg dupilumab subcutaneously every 4 weeks. The dose could be increased if needed to improve clinical response to once every 2 weeks (200 mg if baseline weight was <60 kg; 300 mg if ≥60 kg).
At 52 weeks, 37% of 52 patients with weights under 60 kg reached an Investigator Global Assessment (IGA) of 0/1, a level that had been fairly steady since week 16 (n = 146). Among 51 heavier patients, 49% reached an IGA of 0/1 at 52 weeks; this percentage grew steadily since baseline.
The mean percentage change in EASI was –87% in the lower-weight group (n = 50) at 52 weeks and –80.1% in the larger-weight group (n = 51). The majority of the reduction in EASI occurred in the first 4 weeks of treatment.
At 52 weeks, the mean Children’s Dermatology Life Quality Index level, which judges the effect of AD on life, was judged as “small” (low) in 71 patients. At baseline, the mean level among 189 patients was “moderate.” The levels dipped below “moderate” at week 4 and never rose above “small” after that.
“Treatment-emergent adverse events reported in ≥5% of patients were nasopharyngitis (21.1%), AD (19.4%), upper respiratory tract infection (12.4%), headache (9.4%), and oropharyngeal pain (5.7%),” the investigators wrote in the poster. They add that 6.7% of patients experienced injection-site reactions, and 8.7% of patients experienced treatment-emergent “narrow conjunctivitis,” which includes conjunctivitis, allergic conjunctivitis, bacterial conjunctivitis, viral conjunctivitis, and atopic keratoconjunctivitis.
Dr. Simpson noted that cases of conjunctivitis fell over time. It’s not clear why this adverse effect appears, he said.
He said that the findings reflect his own experience in clinic. Many of his adolescent patients took part in early dupilumab trials, he said, and dozens have been taking the drug for more than 5 years. “They just seem to get better and better,” he said.
University of Minnesota, Minneapolis, dermatologist Sheilagh Maguiness, MD, who wasn’t involved with the study, said in an interview that dupilumab remains “the safest, most effective and evidence-based therapy we had for children with moderate to severe atopic dermatitis.”
The new study’s findings are “very reassuring,” she said, and similar to those in a 2021 report that tracked long-term use of the drug in children aged 6-11.
Like Dr. Simpson, Dr. Maguiness said many pediatric patients at her clinic have stayed on the drug for more than 5 years. They still have “sustained improvement in skin disease and in their quality of life as well”
There are, however, still questions about dupilumab treatment. “For children who have responded well, when could we consider dose reduction or discontinuation? I have done this successfully just a handful of times, but I would love to see data about what percentage of pediatric patients experience rebound disease after coming off the drug and after what duration of treatment,” she said. “Another mystery that will be very interesting to unravel is the question as to whether or not early treatment with dupilumab may attenuate other atopic diseases.”
Dr. Maguiness added that “another issue specific to pediatric use of dupilumab is the recommendation surrounding vaccinations. This is an issue that should be studied in terms of antibody response and safety surrounding vaccinations, particularly as we are eagerly awaiting a pediatric FDA approval for the COVID-19 vaccine in children.”
She also urged colleagues to push back against insurers who resist paying for dupilumab. “Whether prescribing this medication on or off label, insurance companies are often requiring patients to try and fail other traditional immunosuppressive medications such as methotrexate, cyclosporine, or to pursue phototherapy,” she said. “Oftentimes, these are not practical or even safe options for children for a multitude of reasons. Don’t be shy about advocating for your patients by second- or even third-level appeals to try and gain approval for children who are in need of treatment.”
The study was funded by Sanofi Genzyme and Regeneron Pharmaceuticals. The study authors reported various disclosures. Dr. Simpson reported investigator and consultant fee relationships from various pharmaceutical companies. Dr. Maguiness was an investigator for one of the initial pediatric dupilumab trials.
A version of this article first appeared on Medscape.com.
More than half of U.S. children under 6 years show detectable blood lead levels
Lead poisoning remains a significant threat to the health of young children in the United States, based on data from blood tests of more than 1 million children.
Any level of lead is potentially harmful, although blood lead levels have decreased over the past several decades in part because of the elimination of lead from many consumer products, as well as from gas, paint, and plumbing fixtures, wrote Marissa Hauptman, MD, of Boston Children’s Hospital and colleagues.
However, “numerous environmental sources of legacy lead still exist,” and children living in poverty and in older housing in particular remain at increased risk for lead exposure, they noted.
In a study published in JAMA Pediatrics, the researchers analyzed deidentified results from blood lead tests performed at a single clinical laboratory for 1,141,441 children younger than 6 years between Oct. 1, 2018, and Feb. 29, 2020. The mean age of the children was 2.3 years; approximately half were boys.
Overall, 50.5% of the children tested (576,092 children) had detectable blood lead levels (BLLs), defined as 1.0 mcg/dL or higher, and 1.9% (21,172 children) had elevated BLLs, defined as 5.0 mcg/dL or higher.
In multivariate analysis, both detectable BLLs and elevated BLLs were significantly more common among children with public insurance (adjusted odds ratios, 2.01 and 1.08, respectively).
Children in the highest vs. lowest quintile of pre-1950s housing had significantly greater odds of both detectable and elevated BLLs (aOR, 1.65 and aOR, 3.06); those in the highest vs. lowest quintiles of poverty showed similarly increased risk of detectable and elevated BLLs (aOR, 1.89 and aOR, 1.99, respectively; P < .001 for all).
When the data were broken out by ZIP code, children in predominantly Black non-Hispanic and non-Latino neighborhoods were more likely than those living in other ZIP codes to have detectable BLLs (aOR, 1.13), but less likely to have elevated BLLs (aOR, 0.83). States with the highest overall proportions of children with detectable BLLs were Nebraska (83%), Missouri (82%), and Michigan (78%).
The study findings were limited by several factors, especially the potential for selection bias because of the use of a single reference laboratory (Quest Diagnostics), that does not perform all lead testing in the United States, the researchers noted. Other limitations included variability in testing at the state level, and the use of ZIP code–level data to estimate race, ethnicity, housing, and poverty, they said.
However, the results suggest that lead exposure remains a problem in young children, with significant disparities at the individual and community level, and national efforts must focus on further reductions of lead exposure in areas of highest risk, they concluded.
Step up lead elimination efforts
“The removal of lead from gasoline and new paint produced a precipitous decrease in blood lead levels from a population mean of 17 mcg/dL (all ages) in 1976 to 4 mcg/dL in the early 1990s to less than 2 mcg/dL today,” wrote Philip J. Landrigan, MD, of Boston College and David Bellinger, PhD, of Harvard University, Boston, in an accompanying editorial. However, “The findings from this study underscore the urgent need to eliminate all sources of lead exposure from U.S. children’s environments,” and highlight the persistent disparities in children’s lead exposure, they said.
The authors emphasized the need to remove existing lead paint from U.S. homes, as not only the paint itself, but the dust that enters the environment as the pain wears over time, continue to account for most detectable and elevated BLLs in children. A comprehensive lead paint removal effort would be an investment that would protect children now and would protect future generations, they emphasized. They proposed “creating a lead paint removal workforce through federally supported partnerships between city governments and major unions,” that would not only protect children from disease and disability, but could potentially provide jobs and vocational programs that would have a significant impact on communities.
Elevated lead levels may be underreported
In fact, the situation of children’s lead exposure in the United States may be more severe than indicated by the study findings, given the variation in testing at the state and local levels, said Karalyn Kinsella, MD, a pediatrician in private practice in Cheshire, Conn.
“There are no available lead test kits in our offices, so I do worry that many elevated lead levels will be missed,” she said.
“The recent case of elevated lead levels in drinking water in Flint, Michigan, was largely detected through pediatric clinic screening and showed that elevated lead levels may remain a major issue in some communities,” said Tim Joos, MD, a clinician in combined internal medicine/pediatrics in Seattle, Wash., in an interview.
“It is important to highlight to what extent baseline and point-source lead contamination still exists, monitor progress towards lowering levels, and identify communities at high risk,” Dr. Joos emphasized. “The exact prevalence of elevated lead levels among the general pediatric populations is hard to estimate from this study because of the methodology, which looked at demographic characteristics of the subset of the pediatric population that had venous samples sent to Quest Lab,” he noted.
“As the authors pointed out, it is hard to know what biases went into deciding whether to screen or not, and whether these were confirmatory tests for elevated point of care testing done earlier in the clinic,” said Dr. Joos. “Nonetheless, it does point to the role of poverty and pre-1950s housing in elevated blood lead levels,” he added. “The study also highlights that, as the CDC considers lowering the level for what is considered an ‘elevated blood lead level’ from 5.0 to perhaps 3.5 mcg/dL, we still have a lot more work to do,” he said.
The study was funded by Quest Diagnostics and the company provided salaries to several coauthors during the study. Dr. Hauptmann disclosed support from the National Institutes of Health/National Institute of Environmental Health Sciences during the current study and support from the Agency for Toxic Substances and Disease Registry and the U.S. Environmental Protection Agency unrelated to the current study. Dr. Landrigan had no financial conflicts to disclose. Dr. Bellinger disclosed fees from attorneys for testimony in cases unrelated to the editorial. Dr. Kinsella had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Pediatric News. Dr. Joos had no financial conflicts to disclose, but serves on the Pediatric News Editorial Advisory Board.
Lead poisoning remains a significant threat to the health of young children in the United States, based on data from blood tests of more than 1 million children.
Any level of lead is potentially harmful, although blood lead levels have decreased over the past several decades in part because of the elimination of lead from many consumer products, as well as from gas, paint, and plumbing fixtures, wrote Marissa Hauptman, MD, of Boston Children’s Hospital and colleagues.
However, “numerous environmental sources of legacy lead still exist,” and children living in poverty and in older housing in particular remain at increased risk for lead exposure, they noted.
In a study published in JAMA Pediatrics, the researchers analyzed deidentified results from blood lead tests performed at a single clinical laboratory for 1,141,441 children younger than 6 years between Oct. 1, 2018, and Feb. 29, 2020. The mean age of the children was 2.3 years; approximately half were boys.
Overall, 50.5% of the children tested (576,092 children) had detectable blood lead levels (BLLs), defined as 1.0 mcg/dL or higher, and 1.9% (21,172 children) had elevated BLLs, defined as 5.0 mcg/dL or higher.
In multivariate analysis, both detectable BLLs and elevated BLLs were significantly more common among children with public insurance (adjusted odds ratios, 2.01 and 1.08, respectively).
Children in the highest vs. lowest quintile of pre-1950s housing had significantly greater odds of both detectable and elevated BLLs (aOR, 1.65 and aOR, 3.06); those in the highest vs. lowest quintiles of poverty showed similarly increased risk of detectable and elevated BLLs (aOR, 1.89 and aOR, 1.99, respectively; P < .001 for all).
When the data were broken out by ZIP code, children in predominantly Black non-Hispanic and non-Latino neighborhoods were more likely than those living in other ZIP codes to have detectable BLLs (aOR, 1.13), but less likely to have elevated BLLs (aOR, 0.83). States with the highest overall proportions of children with detectable BLLs were Nebraska (83%), Missouri (82%), and Michigan (78%).
The study findings were limited by several factors, especially the potential for selection bias because of the use of a single reference laboratory (Quest Diagnostics), that does not perform all lead testing in the United States, the researchers noted. Other limitations included variability in testing at the state level, and the use of ZIP code–level data to estimate race, ethnicity, housing, and poverty, they said.
However, the results suggest that lead exposure remains a problem in young children, with significant disparities at the individual and community level, and national efforts must focus on further reductions of lead exposure in areas of highest risk, they concluded.
Step up lead elimination efforts
“The removal of lead from gasoline and new paint produced a precipitous decrease in blood lead levels from a population mean of 17 mcg/dL (all ages) in 1976 to 4 mcg/dL in the early 1990s to less than 2 mcg/dL today,” wrote Philip J. Landrigan, MD, of Boston College and David Bellinger, PhD, of Harvard University, Boston, in an accompanying editorial. However, “The findings from this study underscore the urgent need to eliminate all sources of lead exposure from U.S. children’s environments,” and highlight the persistent disparities in children’s lead exposure, they said.
The authors emphasized the need to remove existing lead paint from U.S. homes, as not only the paint itself, but the dust that enters the environment as the pain wears over time, continue to account for most detectable and elevated BLLs in children. A comprehensive lead paint removal effort would be an investment that would protect children now and would protect future generations, they emphasized. They proposed “creating a lead paint removal workforce through federally supported partnerships between city governments and major unions,” that would not only protect children from disease and disability, but could potentially provide jobs and vocational programs that would have a significant impact on communities.
Elevated lead levels may be underreported
In fact, the situation of children’s lead exposure in the United States may be more severe than indicated by the study findings, given the variation in testing at the state and local levels, said Karalyn Kinsella, MD, a pediatrician in private practice in Cheshire, Conn.
“There are no available lead test kits in our offices, so I do worry that many elevated lead levels will be missed,” she said.
“The recent case of elevated lead levels in drinking water in Flint, Michigan, was largely detected through pediatric clinic screening and showed that elevated lead levels may remain a major issue in some communities,” said Tim Joos, MD, a clinician in combined internal medicine/pediatrics in Seattle, Wash., in an interview.
“It is important to highlight to what extent baseline and point-source lead contamination still exists, monitor progress towards lowering levels, and identify communities at high risk,” Dr. Joos emphasized. “The exact prevalence of elevated lead levels among the general pediatric populations is hard to estimate from this study because of the methodology, which looked at demographic characteristics of the subset of the pediatric population that had venous samples sent to Quest Lab,” he noted.
“As the authors pointed out, it is hard to know what biases went into deciding whether to screen or not, and whether these were confirmatory tests for elevated point of care testing done earlier in the clinic,” said Dr. Joos. “Nonetheless, it does point to the role of poverty and pre-1950s housing in elevated blood lead levels,” he added. “The study also highlights that, as the CDC considers lowering the level for what is considered an ‘elevated blood lead level’ from 5.0 to perhaps 3.5 mcg/dL, we still have a lot more work to do,” he said.
The study was funded by Quest Diagnostics and the company provided salaries to several coauthors during the study. Dr. Hauptmann disclosed support from the National Institutes of Health/National Institute of Environmental Health Sciences during the current study and support from the Agency for Toxic Substances and Disease Registry and the U.S. Environmental Protection Agency unrelated to the current study. Dr. Landrigan had no financial conflicts to disclose. Dr. Bellinger disclosed fees from attorneys for testimony in cases unrelated to the editorial. Dr. Kinsella had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Pediatric News. Dr. Joos had no financial conflicts to disclose, but serves on the Pediatric News Editorial Advisory Board.
Lead poisoning remains a significant threat to the health of young children in the United States, based on data from blood tests of more than 1 million children.
Any level of lead is potentially harmful, although blood lead levels have decreased over the past several decades in part because of the elimination of lead from many consumer products, as well as from gas, paint, and plumbing fixtures, wrote Marissa Hauptman, MD, of Boston Children’s Hospital and colleagues.
However, “numerous environmental sources of legacy lead still exist,” and children living in poverty and in older housing in particular remain at increased risk for lead exposure, they noted.
In a study published in JAMA Pediatrics, the researchers analyzed deidentified results from blood lead tests performed at a single clinical laboratory for 1,141,441 children younger than 6 years between Oct. 1, 2018, and Feb. 29, 2020. The mean age of the children was 2.3 years; approximately half were boys.
Overall, 50.5% of the children tested (576,092 children) had detectable blood lead levels (BLLs), defined as 1.0 mcg/dL or higher, and 1.9% (21,172 children) had elevated BLLs, defined as 5.0 mcg/dL or higher.
In multivariate analysis, both detectable BLLs and elevated BLLs were significantly more common among children with public insurance (adjusted odds ratios, 2.01 and 1.08, respectively).
Children in the highest vs. lowest quintile of pre-1950s housing had significantly greater odds of both detectable and elevated BLLs (aOR, 1.65 and aOR, 3.06); those in the highest vs. lowest quintiles of poverty showed similarly increased risk of detectable and elevated BLLs (aOR, 1.89 and aOR, 1.99, respectively; P < .001 for all).
When the data were broken out by ZIP code, children in predominantly Black non-Hispanic and non-Latino neighborhoods were more likely than those living in other ZIP codes to have detectable BLLs (aOR, 1.13), but less likely to have elevated BLLs (aOR, 0.83). States with the highest overall proportions of children with detectable BLLs were Nebraska (83%), Missouri (82%), and Michigan (78%).
The study findings were limited by several factors, especially the potential for selection bias because of the use of a single reference laboratory (Quest Diagnostics), that does not perform all lead testing in the United States, the researchers noted. Other limitations included variability in testing at the state level, and the use of ZIP code–level data to estimate race, ethnicity, housing, and poverty, they said.
However, the results suggest that lead exposure remains a problem in young children, with significant disparities at the individual and community level, and national efforts must focus on further reductions of lead exposure in areas of highest risk, they concluded.
Step up lead elimination efforts
“The removal of lead from gasoline and new paint produced a precipitous decrease in blood lead levels from a population mean of 17 mcg/dL (all ages) in 1976 to 4 mcg/dL in the early 1990s to less than 2 mcg/dL today,” wrote Philip J. Landrigan, MD, of Boston College and David Bellinger, PhD, of Harvard University, Boston, in an accompanying editorial. However, “The findings from this study underscore the urgent need to eliminate all sources of lead exposure from U.S. children’s environments,” and highlight the persistent disparities in children’s lead exposure, they said.
The authors emphasized the need to remove existing lead paint from U.S. homes, as not only the paint itself, but the dust that enters the environment as the pain wears over time, continue to account for most detectable and elevated BLLs in children. A comprehensive lead paint removal effort would be an investment that would protect children now and would protect future generations, they emphasized. They proposed “creating a lead paint removal workforce through federally supported partnerships between city governments and major unions,” that would not only protect children from disease and disability, but could potentially provide jobs and vocational programs that would have a significant impact on communities.
Elevated lead levels may be underreported
In fact, the situation of children’s lead exposure in the United States may be more severe than indicated by the study findings, given the variation in testing at the state and local levels, said Karalyn Kinsella, MD, a pediatrician in private practice in Cheshire, Conn.
“There are no available lead test kits in our offices, so I do worry that many elevated lead levels will be missed,” she said.
“The recent case of elevated lead levels in drinking water in Flint, Michigan, was largely detected through pediatric clinic screening and showed that elevated lead levels may remain a major issue in some communities,” said Tim Joos, MD, a clinician in combined internal medicine/pediatrics in Seattle, Wash., in an interview.
“It is important to highlight to what extent baseline and point-source lead contamination still exists, monitor progress towards lowering levels, and identify communities at high risk,” Dr. Joos emphasized. “The exact prevalence of elevated lead levels among the general pediatric populations is hard to estimate from this study because of the methodology, which looked at demographic characteristics of the subset of the pediatric population that had venous samples sent to Quest Lab,” he noted.
“As the authors pointed out, it is hard to know what biases went into deciding whether to screen or not, and whether these were confirmatory tests for elevated point of care testing done earlier in the clinic,” said Dr. Joos. “Nonetheless, it does point to the role of poverty and pre-1950s housing in elevated blood lead levels,” he added. “The study also highlights that, as the CDC considers lowering the level for what is considered an ‘elevated blood lead level’ from 5.0 to perhaps 3.5 mcg/dL, we still have a lot more work to do,” he said.
The study was funded by Quest Diagnostics and the company provided salaries to several coauthors during the study. Dr. Hauptmann disclosed support from the National Institutes of Health/National Institute of Environmental Health Sciences during the current study and support from the Agency for Toxic Substances and Disease Registry and the U.S. Environmental Protection Agency unrelated to the current study. Dr. Landrigan had no financial conflicts to disclose. Dr. Bellinger disclosed fees from attorneys for testimony in cases unrelated to the editorial. Dr. Kinsella had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Pediatric News. Dr. Joos had no financial conflicts to disclose, but serves on the Pediatric News Editorial Advisory Board.
FROM JAMA PEDIATRICS
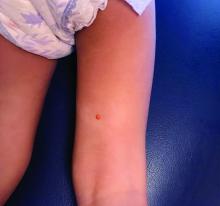
A female toddler presents with an itchy yellow nodule
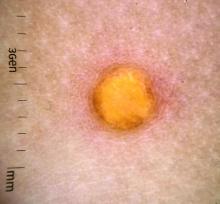
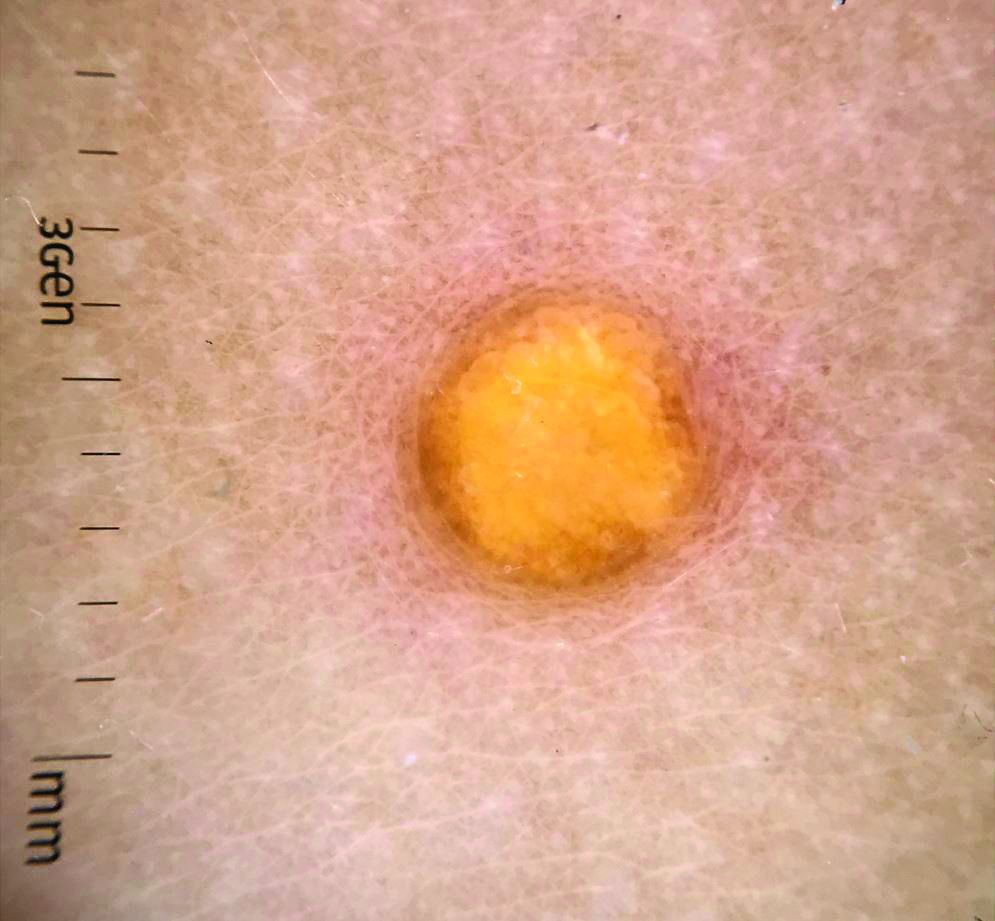
Juvenile xanthogranuloma (JXG) is a benign disorder presenting as firm, yellow-red skin papules or nodules, usually in infancy or early childhood. It derives its name based on its yellowish color and the histologic finding of lipid-filled histiocytes. In fact, it is a form of non-Langerhans’ cell histiocytosis. It most commonly presents on the head, neck, and trunk, but can arise anywhere on the body as demonstrated by this case. While often pink to reddish early on, the characteristic yellow or orange, brown appearance over time is common, occasionally with overlying telangiectasia, and ranging in size from 1 mm to 2 cm. While typically asymptomatic, it is possible for lesions to itch. JXG is usually self-limiting, and spontaneously resolves over several years. On dermoscopy (with polarized light), it has a characteristic “setting sun” appearance because of its central yellow area surrounded by a reddish periphery.
JXGs have been associated with neurofibromatosis-1 and a “triple association” of NF-1, JXG, and juvenile myelomonocytic leukemia (JMML) has been debated. Many cases are diagnosed on clinical grounds without histologic confirmation, so while the absolute incidence is unknown, they are not uncommon.
What is on the differential?
Spitz nevus is a melanocytic lesion which typically presents as a sharply circumscribed, dome-shaped, pink-red or brown papule or nodule, and is composed of large epithelioid and/or spindled cells. These nevi can present with a spectrum of morphology and biologic activity; commonly with benign melanocytic proliferations and a symmetric appearance or, rarely, with atypical tumors or lesions, characterized as Spitzoid melanomas. The yellowish color of JXG is distinct from the appearance of Spitz tumors.
Molluscum contagiosum is a common pox viral infection seen in children that presents with round, flat-topped firm papules on the skin and distinctive whitish centers with or without umbilication. Like JXG, molluscum contagiosum papules may grow over time and cause pruritus. However, this diagnosis is less likely given the absence of other lesions on the skin, lack of known contacts with similar lesions, and yellowish color without a more typical appearance of molluscum.
Dermatofibromas occur in people of all ages, although more commonly between the ages of 20 and 40 and in those with a history of trauma at the lesion. Like JXGs, dermatofibromas tend to be firm, solitary papules or nodules. They usually are hyperpigmented, and classically “dimple when pinched” as they are fixed to the subcutaneous tissue. However, this patient’s age, lack of trauma, and the lesion morphology are not consistent with dermatofibromas.
Like XJGs, mastocytomas commonly present in the first 2 years of life with maculopapular or nodular lesions that itch. However, the history of new-onset itch in recent months as the lesion grew larger and the yellow color on dermoscopy are more consistent with JXG.
Eruptive xanthomas typically appear suddenly as multiple erythematous yellow, dome-shaped papules on the extensor surfaces of the extremities, buttocks, and hands. They are usually present with hypertriglyceridemia and are very rare in young children. The presence of a solitary lesion in a 6-month-old patient without a history of lipid abnormalities favors the diagnosis of XJG.
Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Ms. Kleinman is a pediatric dermatology research associate in the division of pediatric and adolescent dermatology, University of California, San Diego, and Rady Children’s Hospital. Dr. Eichenfield and Ms. Kleinman have no relevant financial disclosures.
References
Hernandez-Martin A et al. J Am Acad Dermatol. 1997 Mar;36(3 Pt 1):355-67.
Prendiville J. Lumps, bumps and hamartomas in “Neonatal and Infant Dermatology,” 3rd ed. (Philadelphia: Elsevier, 2015).
Püttgen KB. Juvenile xanthogranuloma. UpToDate, 2021.
Schaffer JV. Am J Clin Dermatol. 2021 Mar;22(2):205-20.
Juvenile xanthogranuloma (JXG) is a benign disorder presenting as firm, yellow-red skin papules or nodules, usually in infancy or early childhood. It derives its name based on its yellowish color and the histologic finding of lipid-filled histiocytes. In fact, it is a form of non-Langerhans’ cell histiocytosis. It most commonly presents on the head, neck, and trunk, but can arise anywhere on the body as demonstrated by this case. While often pink to reddish early on, the characteristic yellow or orange, brown appearance over time is common, occasionally with overlying telangiectasia, and ranging in size from 1 mm to 2 cm. While typically asymptomatic, it is possible for lesions to itch. JXG is usually self-limiting, and spontaneously resolves over several years. On dermoscopy (with polarized light), it has a characteristic “setting sun” appearance because of its central yellow area surrounded by a reddish periphery.
JXGs have been associated with neurofibromatosis-1 and a “triple association” of NF-1, JXG, and juvenile myelomonocytic leukemia (JMML) has been debated. Many cases are diagnosed on clinical grounds without histologic confirmation, so while the absolute incidence is unknown, they are not uncommon.
What is on the differential?
Spitz nevus is a melanocytic lesion which typically presents as a sharply circumscribed, dome-shaped, pink-red or brown papule or nodule, and is composed of large epithelioid and/or spindled cells. These nevi can present with a spectrum of morphology and biologic activity; commonly with benign melanocytic proliferations and a symmetric appearance or, rarely, with atypical tumors or lesions, characterized as Spitzoid melanomas. The yellowish color of JXG is distinct from the appearance of Spitz tumors.
Molluscum contagiosum is a common pox viral infection seen in children that presents with round, flat-topped firm papules on the skin and distinctive whitish centers with or without umbilication. Like JXG, molluscum contagiosum papules may grow over time and cause pruritus. However, this diagnosis is less likely given the absence of other lesions on the skin, lack of known contacts with similar lesions, and yellowish color without a more typical appearance of molluscum.
Dermatofibromas occur in people of all ages, although more commonly between the ages of 20 and 40 and in those with a history of trauma at the lesion. Like JXGs, dermatofibromas tend to be firm, solitary papules or nodules. They usually are hyperpigmented, and classically “dimple when pinched” as they are fixed to the subcutaneous tissue. However, this patient’s age, lack of trauma, and the lesion morphology are not consistent with dermatofibromas.
Like XJGs, mastocytomas commonly present in the first 2 years of life with maculopapular or nodular lesions that itch. However, the history of new-onset itch in recent months as the lesion grew larger and the yellow color on dermoscopy are more consistent with JXG.
Eruptive xanthomas typically appear suddenly as multiple erythematous yellow, dome-shaped papules on the extensor surfaces of the extremities, buttocks, and hands. They are usually present with hypertriglyceridemia and are very rare in young children. The presence of a solitary lesion in a 6-month-old patient without a history of lipid abnormalities favors the diagnosis of XJG.
Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Ms. Kleinman is a pediatric dermatology research associate in the division of pediatric and adolescent dermatology, University of California, San Diego, and Rady Children’s Hospital. Dr. Eichenfield and Ms. Kleinman have no relevant financial disclosures.
References
Hernandez-Martin A et al. J Am Acad Dermatol. 1997 Mar;36(3 Pt 1):355-67.
Prendiville J. Lumps, bumps and hamartomas in “Neonatal and Infant Dermatology,” 3rd ed. (Philadelphia: Elsevier, 2015).
Püttgen KB. Juvenile xanthogranuloma. UpToDate, 2021.
Schaffer JV. Am J Clin Dermatol. 2021 Mar;22(2):205-20.
Juvenile xanthogranuloma (JXG) is a benign disorder presenting as firm, yellow-red skin papules or nodules, usually in infancy or early childhood. It derives its name based on its yellowish color and the histologic finding of lipid-filled histiocytes. In fact, it is a form of non-Langerhans’ cell histiocytosis. It most commonly presents on the head, neck, and trunk, but can arise anywhere on the body as demonstrated by this case. While often pink to reddish early on, the characteristic yellow or orange, brown appearance over time is common, occasionally with overlying telangiectasia, and ranging in size from 1 mm to 2 cm. While typically asymptomatic, it is possible for lesions to itch. JXG is usually self-limiting, and spontaneously resolves over several years. On dermoscopy (with polarized light), it has a characteristic “setting sun” appearance because of its central yellow area surrounded by a reddish periphery.
JXGs have been associated with neurofibromatosis-1 and a “triple association” of NF-1, JXG, and juvenile myelomonocytic leukemia (JMML) has been debated. Many cases are diagnosed on clinical grounds without histologic confirmation, so while the absolute incidence is unknown, they are not uncommon.
What is on the differential?
Spitz nevus is a melanocytic lesion which typically presents as a sharply circumscribed, dome-shaped, pink-red or brown papule or nodule, and is composed of large epithelioid and/or spindled cells. These nevi can present with a spectrum of morphology and biologic activity; commonly with benign melanocytic proliferations and a symmetric appearance or, rarely, with atypical tumors or lesions, characterized as Spitzoid melanomas. The yellowish color of JXG is distinct from the appearance of Spitz tumors.
Molluscum contagiosum is a common pox viral infection seen in children that presents with round, flat-topped firm papules on the skin and distinctive whitish centers with or without umbilication. Like JXG, molluscum contagiosum papules may grow over time and cause pruritus. However, this diagnosis is less likely given the absence of other lesions on the skin, lack of known contacts with similar lesions, and yellowish color without a more typical appearance of molluscum.
Dermatofibromas occur in people of all ages, although more commonly between the ages of 20 and 40 and in those with a history of trauma at the lesion. Like JXGs, dermatofibromas tend to be firm, solitary papules or nodules. They usually are hyperpigmented, and classically “dimple when pinched” as they are fixed to the subcutaneous tissue. However, this patient’s age, lack of trauma, and the lesion morphology are not consistent with dermatofibromas.
Like XJGs, mastocytomas commonly present in the first 2 years of life with maculopapular or nodular lesions that itch. However, the history of new-onset itch in recent months as the lesion grew larger and the yellow color on dermoscopy are more consistent with JXG.
Eruptive xanthomas typically appear suddenly as multiple erythematous yellow, dome-shaped papules on the extensor surfaces of the extremities, buttocks, and hands. They are usually present with hypertriglyceridemia and are very rare in young children. The presence of a solitary lesion in a 6-month-old patient without a history of lipid abnormalities favors the diagnosis of XJG.
Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Ms. Kleinman is a pediatric dermatology research associate in the division of pediatric and adolescent dermatology, University of California, San Diego, and Rady Children’s Hospital. Dr. Eichenfield and Ms. Kleinman have no relevant financial disclosures.
References
Hernandez-Martin A et al. J Am Acad Dermatol. 1997 Mar;36(3 Pt 1):355-67.
Prendiville J. Lumps, bumps and hamartomas in “Neonatal and Infant Dermatology,” 3rd ed. (Philadelphia: Elsevier, 2015).
Püttgen KB. Juvenile xanthogranuloma. UpToDate, 2021.
Schaffer JV. Am J Clin Dermatol. 2021 Mar;22(2):205-20.
Management of pediatric food allergies evolving
The treatment of atopic dermatitis (AD) is undergoing a revolution thanks to biologics. Now, an allergist and a dietitian told pediatric dermatologists that the treatment of a related condition – food allergy – is also undergoing a dramatic transformation as the management approach evolves away from blanket avoidance of allergens.
“Over the past 15 years, we’ve seen a shift from a very passive approach where generally we just advised patients to avoid the things they’re allergic to,” said U.K. pediatric allergist Adam Fox, MBBS, MD, in a presentation at The World Congress of Pediatric Dermatology (WCPD) 2021 Annual Meeting. “Now, we have a much better understanding of how allergy develops and strategies to minimize the risk of allergy happening in the first place,” he said.
According to Carina Venter, PhD, RD, associate professor of pediatrics-allergy/immunology at the University of Colorado, Denver, who also spoke at the conference, an estimated 20% to 30% of patients with AD also have food allergies, and up to 90% of infants with cow’s milk allergy develop skin symptoms.
It may not be necessary for a breastfeeding mother to avoid food allergens if a child is allergic, said Dr. Fox, of Guy’s and St. Thomas’ NHS Foundation Trust, London. “A lot of parents will automatically assume that if their child has an egg or milk allergy, then it’s a good idea to completely eliminate that from their diet if they’re breastfeeding,” but it is “surprisingly uncommon” that this approach makes a difference, he said. “Less goes through the breast milk than people imagine,” he said.
He noted that eliminating foods from the breastfeeding mother’s diet may have negative consequences. “There’s always that risk that if you make life harder for the breastfeeding mom because they’re going to have to avoid all sorts of foods, they’ll be more likely to discontinue breastfeeding. You really need a compelling reason to stop the food.”
As for children themselves, Dr. Fox suggested that there’s often no connection between AD and food allergies. “What will commonly happen when you see and diagnose these kids is that their eczema has been quite significantly undertreated,” he said. “Once you just get them on the right [regimen], they don’t need to be cutting the food out of their diet. It’s just making their life unnecessarily harder.”
Dr. Venter said there may be little choice but to avoid a trigger food if a child develops AD with exposure. However, she noted, it’s important to understand that avoidance of certain foods could make the allergy – and AD – worse. “If you have a child or an adult with atopic dermatitis that’s not controlled by an optimal topical treatment, and you do consider avoidance, we need to be aware that development of more severe IgA-mediated symptoms can happen in a short period of time,” she said.
In a slide that Dr. Venter presented, the dilemma for physicians was expressed this way: “The potential benefit of food avoidance as a management strategy for some patients with AD must now be weighed against the strong evidence that unnecessarily avoiding a food in kids with AD increases the risk of developing anaphylaxis to that food.”
What should pediatric dermatologists do to balance the risks of allergen exposure to the risks that children will develop permanent allergies? Dr. Venter pointed to guidelines about AD that were developed by the U.K.’s National Institute for Health and Care Excellence. She also highlighted the International Milk Allergy in Primary Care recommendations.
She suggested considering creative ways to bypass complete avoidance and boost a child’s tolerance of allergens if possible. “If we’re going to keep a child with eczema on a mold-free diet for a longer period of time, is there perhaps a role for regularly introducing small amounts of yogurt or even small amounts of milk in the child’s diet to at least keep immune tolerance without necessarily aggravating eczema symptoms?”
Dr. Fox has consulted for DBV and Aimmune through his employer, NHS Trust. He serves as president of the British Society for Allergy and Clinical Immunology and as chair of the Allergy UK Health Advisory Board, both of which receive funding from drug companies. Dr. Venter has received support for allergy-related research from the National Peanut Board.
A version of this article first appeared on Medscape.com.
The treatment of atopic dermatitis (AD) is undergoing a revolution thanks to biologics. Now, an allergist and a dietitian told pediatric dermatologists that the treatment of a related condition – food allergy – is also undergoing a dramatic transformation as the management approach evolves away from blanket avoidance of allergens.
“Over the past 15 years, we’ve seen a shift from a very passive approach where generally we just advised patients to avoid the things they’re allergic to,” said U.K. pediatric allergist Adam Fox, MBBS, MD, in a presentation at The World Congress of Pediatric Dermatology (WCPD) 2021 Annual Meeting. “Now, we have a much better understanding of how allergy develops and strategies to minimize the risk of allergy happening in the first place,” he said.
According to Carina Venter, PhD, RD, associate professor of pediatrics-allergy/immunology at the University of Colorado, Denver, who also spoke at the conference, an estimated 20% to 30% of patients with AD also have food allergies, and up to 90% of infants with cow’s milk allergy develop skin symptoms.
It may not be necessary for a breastfeeding mother to avoid food allergens if a child is allergic, said Dr. Fox, of Guy’s and St. Thomas’ NHS Foundation Trust, London. “A lot of parents will automatically assume that if their child has an egg or milk allergy, then it’s a good idea to completely eliminate that from their diet if they’re breastfeeding,” but it is “surprisingly uncommon” that this approach makes a difference, he said. “Less goes through the breast milk than people imagine,” he said.
He noted that eliminating foods from the breastfeeding mother’s diet may have negative consequences. “There’s always that risk that if you make life harder for the breastfeeding mom because they’re going to have to avoid all sorts of foods, they’ll be more likely to discontinue breastfeeding. You really need a compelling reason to stop the food.”
As for children themselves, Dr. Fox suggested that there’s often no connection between AD and food allergies. “What will commonly happen when you see and diagnose these kids is that their eczema has been quite significantly undertreated,” he said. “Once you just get them on the right [regimen], they don’t need to be cutting the food out of their diet. It’s just making their life unnecessarily harder.”
Dr. Venter said there may be little choice but to avoid a trigger food if a child develops AD with exposure. However, she noted, it’s important to understand that avoidance of certain foods could make the allergy – and AD – worse. “If you have a child or an adult with atopic dermatitis that’s not controlled by an optimal topical treatment, and you do consider avoidance, we need to be aware that development of more severe IgA-mediated symptoms can happen in a short period of time,” she said.
In a slide that Dr. Venter presented, the dilemma for physicians was expressed this way: “The potential benefit of food avoidance as a management strategy for some patients with AD must now be weighed against the strong evidence that unnecessarily avoiding a food in kids with AD increases the risk of developing anaphylaxis to that food.”
What should pediatric dermatologists do to balance the risks of allergen exposure to the risks that children will develop permanent allergies? Dr. Venter pointed to guidelines about AD that were developed by the U.K.’s National Institute for Health and Care Excellence. She also highlighted the International Milk Allergy in Primary Care recommendations.
She suggested considering creative ways to bypass complete avoidance and boost a child’s tolerance of allergens if possible. “If we’re going to keep a child with eczema on a mold-free diet for a longer period of time, is there perhaps a role for regularly introducing small amounts of yogurt or even small amounts of milk in the child’s diet to at least keep immune tolerance without necessarily aggravating eczema symptoms?”
Dr. Fox has consulted for DBV and Aimmune through his employer, NHS Trust. He serves as president of the British Society for Allergy and Clinical Immunology and as chair of the Allergy UK Health Advisory Board, both of which receive funding from drug companies. Dr. Venter has received support for allergy-related research from the National Peanut Board.
A version of this article first appeared on Medscape.com.
The treatment of atopic dermatitis (AD) is undergoing a revolution thanks to biologics. Now, an allergist and a dietitian told pediatric dermatologists that the treatment of a related condition – food allergy – is also undergoing a dramatic transformation as the management approach evolves away from blanket avoidance of allergens.
“Over the past 15 years, we’ve seen a shift from a very passive approach where generally we just advised patients to avoid the things they’re allergic to,” said U.K. pediatric allergist Adam Fox, MBBS, MD, in a presentation at The World Congress of Pediatric Dermatology (WCPD) 2021 Annual Meeting. “Now, we have a much better understanding of how allergy develops and strategies to minimize the risk of allergy happening in the first place,” he said.
According to Carina Venter, PhD, RD, associate professor of pediatrics-allergy/immunology at the University of Colorado, Denver, who also spoke at the conference, an estimated 20% to 30% of patients with AD also have food allergies, and up to 90% of infants with cow’s milk allergy develop skin symptoms.
It may not be necessary for a breastfeeding mother to avoid food allergens if a child is allergic, said Dr. Fox, of Guy’s and St. Thomas’ NHS Foundation Trust, London. “A lot of parents will automatically assume that if their child has an egg or milk allergy, then it’s a good idea to completely eliminate that from their diet if they’re breastfeeding,” but it is “surprisingly uncommon” that this approach makes a difference, he said. “Less goes through the breast milk than people imagine,” he said.
He noted that eliminating foods from the breastfeeding mother’s diet may have negative consequences. “There’s always that risk that if you make life harder for the breastfeeding mom because they’re going to have to avoid all sorts of foods, they’ll be more likely to discontinue breastfeeding. You really need a compelling reason to stop the food.”
As for children themselves, Dr. Fox suggested that there’s often no connection between AD and food allergies. “What will commonly happen when you see and diagnose these kids is that their eczema has been quite significantly undertreated,” he said. “Once you just get them on the right [regimen], they don’t need to be cutting the food out of their diet. It’s just making their life unnecessarily harder.”
Dr. Venter said there may be little choice but to avoid a trigger food if a child develops AD with exposure. However, she noted, it’s important to understand that avoidance of certain foods could make the allergy – and AD – worse. “If you have a child or an adult with atopic dermatitis that’s not controlled by an optimal topical treatment, and you do consider avoidance, we need to be aware that development of more severe IgA-mediated symptoms can happen in a short period of time,” she said.
In a slide that Dr. Venter presented, the dilemma for physicians was expressed this way: “The potential benefit of food avoidance as a management strategy for some patients with AD must now be weighed against the strong evidence that unnecessarily avoiding a food in kids with AD increases the risk of developing anaphylaxis to that food.”
What should pediatric dermatologists do to balance the risks of allergen exposure to the risks that children will develop permanent allergies? Dr. Venter pointed to guidelines about AD that were developed by the U.K.’s National Institute for Health and Care Excellence. She also highlighted the International Milk Allergy in Primary Care recommendations.
She suggested considering creative ways to bypass complete avoidance and boost a child’s tolerance of allergens if possible. “If we’re going to keep a child with eczema on a mold-free diet for a longer period of time, is there perhaps a role for regularly introducing small amounts of yogurt or even small amounts of milk in the child’s diet to at least keep immune tolerance without necessarily aggravating eczema symptoms?”
Dr. Fox has consulted for DBV and Aimmune through his employer, NHS Trust. He serves as president of the British Society for Allergy and Clinical Immunology and as chair of the Allergy UK Health Advisory Board, both of which receive funding from drug companies. Dr. Venter has received support for allergy-related research from the National Peanut Board.
A version of this article first appeared on Medscape.com.
Toy soldier syndrome: A consequence of parental cognitive dissonance
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Childhood and adolescence are periods with marked psychobehavioral development of the brain. The sense of self, identity, and role are established. This process is not without risk because brain regions governing reward, impulsivity, and sensation-seeking are relatively more developed and influential than higher-order cognitive regions regulating behavioral inhibition, decision-making, and planning, which continue to mature into one’s early to mid-20s. Consequently, while the developing brain is “under construction” by forging new pathways and taking advantage of its immense neuroplasticity, it is also prone to psychological insults under exposure to stressful events, attitudes, and behaviors, including those that can arise in the family.1
Most people would agree that there is no stronger emotion than parental love. The origins of this powerful biobehavioral bonding with a child have been attributed to maternal release of oxytocin, known colloquially as the “love hormone,” during the birthing process, and to both biological parents experiencing psychosocial attachment with their infant. Therefore, common sense dictates that parents would do anything to protect their offspring, and that no parent would deliberately behave in a manner that harms their child.
Common sense notwithstanding, reports of both child neglect and abuse are common. States have established agencies to protect children from their own parents. The answers to the question “Whose kids are they?” and under what circumstances the state has the authority to warn or reprimand parents, or even temporarily or permanently separate minors from their parents, are complex and vary by state.
In this commentary, we describe harmful actions committed by parents with the intention of protecting the impressionable minds of their children from malevolent forces or intrusive and unhealthy ideas. Second, we examine how to protect a minor from parental actions that are well-meaning but potentially harmful.
Parent-child communication
Delusional family interactions. Originally described in 1877 as “folie à deux,”2 shared madness is an extreme and uncommon parental psychiatric condition harmful to a child’s mental health. It is primarily characterized by parental-initiated delusions shared with the child that are typically persecutory and attributed to danger from vengeful folks or grandiose in nature. The question of whether the “folie” or “madness” is contagious arises due to the propensity of the child to adopt these delusions under an imposed insular or restrictive environment. Separating the child from the environment dominated by the delusional adult usually is sufficient to reverse the symptoms due to reality testing.
Normative familial communication. In contrary to a delusional familial interaction, normative family traditions and values are a unifying psychosocial force and a source of bonding and loyalty from an early age. A ubiquitous example is the support of a local sports team, and the emotional turmoil associated with the team’s wins and losses, accompanied by “hating” a rival. These family rituals are commonly devoid of emotional negative consequences for an impressionable young mind unless the child is exposed to unsportsmanlike emotional, verbal, or aggressive behavior by an adult at home in front of the television or in the stands at a game.
Continue to: Unfortunately...
Unfortunately, the “love-hate” dichotomy rooted in family-generated traditions of loyalty is becoming more evident in today’s turbulent sociopolitical environment. Children and young adolescents are not prepared to cope with the stressful effects of repeated exposure to intense conflictual events at home when parents adopt opposing sociopolitical ideologies. Furthermore, a parent might intentionally expose their child to emotionally conflictual circumstances in the name of a perceived value that might create and exacerbate stress, fear, and self-loathing. Ironically, by doing what a parent believes is right for their child, they might be transforming the child without their consent into a variant of a “toy soldier by proxy.” Such a child is a tool expected to follow the parental pathway and belief system without questioning, or even having the cognitive ability to do so, given their ongoing bio-behavioral and moral developmental phase.3
This new normative exposure to conflictual situations at the will of the parent is not only limited to watching them remotely but also may include participating in what is meant to be a peaceful protest or march. As we all witnessed in 2020, such events can easily deteriorate into unsafe environments rife with lawlessness and uncontrolled violence. This has included clashes between opposing groups who are matched in zeal and conviction, as well as opposition to or endangerment by law enforcement personnel trying to restore order by force. This is not where a responsible parent should take their child. Furthermore, there is the danger of loss of privacy of children exposed by media following their participation in public activity. This may lead to hate mail as that would further confuse and jeopardize a peaceful lifestyle, which is highly desirable for a developing child.
Cognitive dissonance. Have these parents temporarily allowed the limbic system to trump the restraints of the prefrontal cortex, as exhibited by an impulsive and risky behavior driven by poor insight? Have these parents thoughtfully weighed the balance between the merit of a child’s exposure to such conflictual circumstances and the peril of negative emotional consequences? This is illustrated by a mother who has been taking her preadolescent son to demonstrations regularly because “I want him to see how democracy works.”
Might this be a case of cognitive dissonance (CD) that amounts to unwitting mental child abuse if it happens repeatedly? According to the CD theory, there is a tendency to seek consistency between cognitions (eg, beliefs, opinions) and attitudes or behaviors. Inconsistency between these variables is termed “dissonance.”4,5 The importance attached to the dissonant belief affects the severity of the dissonance. The dissonance occurs when a parent must choose between 2 incompatible beliefs or actions. A classic demonstration of CD is when an adult requests that an adolescent follows his instructions (eg, “do not smoke or drink alcohol”), yet the adult does not act accordingly (eg, they smoke or drink). Role modeling demonstrated by such a discrepancy is a cause of confusion in the child. In terms of this article, the CD is between what the parent believes is an important learning experience by exercising the perceived right to pass to the child the parental value system vs compromising the protection of the child by exposing them to the potential negative consequences of a risky situation.
What can parents and therapists do?
Usually, parents mean well. It is important to communicate to parents the importance of refraining from forcing their children to join their battles. Calculating risks based on an intuitive approach is flawed because doing so is based on beliefs and emotions that originated in the limbic system (“I feel that”…) and are neither precise nor accurate.6 Teaching our youth in the school system how to think (eg, the science of logic and history of science) vs what to think (ie, indoctrination) is a key to healthy cognitive development. Furthermore, children need to have the time, space, and opportunities (learning moments) to develop this capacity. It is not until approximately age 16 that abstract thinking capabilities are developed. Cognitive dissonance can be eliminated by reducing the valence of the conflicting beliefs or by removing the conflicting attitude or behavior.
As parents and as mental health professionals, we should carry the necessary burden of responsibility to prevent the risk of “lost childhood” due to parental emotional zeal and righteousness that lead to early exposure to damaging adversity. We cannot afford to turn our children into exploitable tools (ie, toy soldiers) in conflicts they do not fully grasp.
1. Bagot KS, Kaminer Y. Harm reduction for youth in treatment with substance use disorders: one size does not fit all. Curr Addict Rep. 2018;5:379-385.
2. Arnon D, Patel A, Tan GM. The nosological significance of Folie à Deux: a review of the literature. Ann Gen Psychiatry. 2006;5:11.
3. Kohlberg L. The philosophy of moral development: the nature and validity of moral stages. Harper & Row; 1984.
4. Festinger L. A theory of cognitive dissonance. Stanford University Press; 1957.
5. Festinger L. Cognitive dissonance. Sci Am. 1962;207:93-102.
6. Henderson SW, Gerson R, Phillips B. What is “high risk” and what are we actually supposed to do about it? J Am Acad Child Adolesc Psychiatry. 2019;58(6):561-564.
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Childhood and adolescence are periods with marked psychobehavioral development of the brain. The sense of self, identity, and role are established. This process is not without risk because brain regions governing reward, impulsivity, and sensation-seeking are relatively more developed and influential than higher-order cognitive regions regulating behavioral inhibition, decision-making, and planning, which continue to mature into one’s early to mid-20s. Consequently, while the developing brain is “under construction” by forging new pathways and taking advantage of its immense neuroplasticity, it is also prone to psychological insults under exposure to stressful events, attitudes, and behaviors, including those that can arise in the family.1
Most people would agree that there is no stronger emotion than parental love. The origins of this powerful biobehavioral bonding with a child have been attributed to maternal release of oxytocin, known colloquially as the “love hormone,” during the birthing process, and to both biological parents experiencing psychosocial attachment with their infant. Therefore, common sense dictates that parents would do anything to protect their offspring, and that no parent would deliberately behave in a manner that harms their child.
Common sense notwithstanding, reports of both child neglect and abuse are common. States have established agencies to protect children from their own parents. The answers to the question “Whose kids are they?” and under what circumstances the state has the authority to warn or reprimand parents, or even temporarily or permanently separate minors from their parents, are complex and vary by state.
In this commentary, we describe harmful actions committed by parents with the intention of protecting the impressionable minds of their children from malevolent forces or intrusive and unhealthy ideas. Second, we examine how to protect a minor from parental actions that are well-meaning but potentially harmful.
Parent-child communication
Delusional family interactions. Originally described in 1877 as “folie à deux,”2 shared madness is an extreme and uncommon parental psychiatric condition harmful to a child’s mental health. It is primarily characterized by parental-initiated delusions shared with the child that are typically persecutory and attributed to danger from vengeful folks or grandiose in nature. The question of whether the “folie” or “madness” is contagious arises due to the propensity of the child to adopt these delusions under an imposed insular or restrictive environment. Separating the child from the environment dominated by the delusional adult usually is sufficient to reverse the symptoms due to reality testing.
Normative familial communication. In contrary to a delusional familial interaction, normative family traditions and values are a unifying psychosocial force and a source of bonding and loyalty from an early age. A ubiquitous example is the support of a local sports team, and the emotional turmoil associated with the team’s wins and losses, accompanied by “hating” a rival. These family rituals are commonly devoid of emotional negative consequences for an impressionable young mind unless the child is exposed to unsportsmanlike emotional, verbal, or aggressive behavior by an adult at home in front of the television or in the stands at a game.
Continue to: Unfortunately...
Unfortunately, the “love-hate” dichotomy rooted in family-generated traditions of loyalty is becoming more evident in today’s turbulent sociopolitical environment. Children and young adolescents are not prepared to cope with the stressful effects of repeated exposure to intense conflictual events at home when parents adopt opposing sociopolitical ideologies. Furthermore, a parent might intentionally expose their child to emotionally conflictual circumstances in the name of a perceived value that might create and exacerbate stress, fear, and self-loathing. Ironically, by doing what a parent believes is right for their child, they might be transforming the child without their consent into a variant of a “toy soldier by proxy.” Such a child is a tool expected to follow the parental pathway and belief system without questioning, or even having the cognitive ability to do so, given their ongoing bio-behavioral and moral developmental phase.3
This new normative exposure to conflictual situations at the will of the parent is not only limited to watching them remotely but also may include participating in what is meant to be a peaceful protest or march. As we all witnessed in 2020, such events can easily deteriorate into unsafe environments rife with lawlessness and uncontrolled violence. This has included clashes between opposing groups who are matched in zeal and conviction, as well as opposition to or endangerment by law enforcement personnel trying to restore order by force. This is not where a responsible parent should take their child. Furthermore, there is the danger of loss of privacy of children exposed by media following their participation in public activity. This may lead to hate mail as that would further confuse and jeopardize a peaceful lifestyle, which is highly desirable for a developing child.
Cognitive dissonance. Have these parents temporarily allowed the limbic system to trump the restraints of the prefrontal cortex, as exhibited by an impulsive and risky behavior driven by poor insight? Have these parents thoughtfully weighed the balance between the merit of a child’s exposure to such conflictual circumstances and the peril of negative emotional consequences? This is illustrated by a mother who has been taking her preadolescent son to demonstrations regularly because “I want him to see how democracy works.”
Might this be a case of cognitive dissonance (CD) that amounts to unwitting mental child abuse if it happens repeatedly? According to the CD theory, there is a tendency to seek consistency between cognitions (eg, beliefs, opinions) and attitudes or behaviors. Inconsistency between these variables is termed “dissonance.”4,5 The importance attached to the dissonant belief affects the severity of the dissonance. The dissonance occurs when a parent must choose between 2 incompatible beliefs or actions. A classic demonstration of CD is when an adult requests that an adolescent follows his instructions (eg, “do not smoke or drink alcohol”), yet the adult does not act accordingly (eg, they smoke or drink). Role modeling demonstrated by such a discrepancy is a cause of confusion in the child. In terms of this article, the CD is between what the parent believes is an important learning experience by exercising the perceived right to pass to the child the parental value system vs compromising the protection of the child by exposing them to the potential negative consequences of a risky situation.
What can parents and therapists do?
Usually, parents mean well. It is important to communicate to parents the importance of refraining from forcing their children to join their battles. Calculating risks based on an intuitive approach is flawed because doing so is based on beliefs and emotions that originated in the limbic system (“I feel that”…) and are neither precise nor accurate.6 Teaching our youth in the school system how to think (eg, the science of logic and history of science) vs what to think (ie, indoctrination) is a key to healthy cognitive development. Furthermore, children need to have the time, space, and opportunities (learning moments) to develop this capacity. It is not until approximately age 16 that abstract thinking capabilities are developed. Cognitive dissonance can be eliminated by reducing the valence of the conflicting beliefs or by removing the conflicting attitude or behavior.
As parents and as mental health professionals, we should carry the necessary burden of responsibility to prevent the risk of “lost childhood” due to parental emotional zeal and righteousness that lead to early exposure to damaging adversity. We cannot afford to turn our children into exploitable tools (ie, toy soldiers) in conflicts they do not fully grasp.
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Childhood and adolescence are periods with marked psychobehavioral development of the brain. The sense of self, identity, and role are established. This process is not without risk because brain regions governing reward, impulsivity, and sensation-seeking are relatively more developed and influential than higher-order cognitive regions regulating behavioral inhibition, decision-making, and planning, which continue to mature into one’s early to mid-20s. Consequently, while the developing brain is “under construction” by forging new pathways and taking advantage of its immense neuroplasticity, it is also prone to psychological insults under exposure to stressful events, attitudes, and behaviors, including those that can arise in the family.1
Most people would agree that there is no stronger emotion than parental love. The origins of this powerful biobehavioral bonding with a child have been attributed to maternal release of oxytocin, known colloquially as the “love hormone,” during the birthing process, and to both biological parents experiencing psychosocial attachment with their infant. Therefore, common sense dictates that parents would do anything to protect their offspring, and that no parent would deliberately behave in a manner that harms their child.
Common sense notwithstanding, reports of both child neglect and abuse are common. States have established agencies to protect children from their own parents. The answers to the question “Whose kids are they?” and under what circumstances the state has the authority to warn or reprimand parents, or even temporarily or permanently separate minors from their parents, are complex and vary by state.
In this commentary, we describe harmful actions committed by parents with the intention of protecting the impressionable minds of their children from malevolent forces or intrusive and unhealthy ideas. Second, we examine how to protect a minor from parental actions that are well-meaning but potentially harmful.
Parent-child communication
Delusional family interactions. Originally described in 1877 as “folie à deux,”2 shared madness is an extreme and uncommon parental psychiatric condition harmful to a child’s mental health. It is primarily characterized by parental-initiated delusions shared with the child that are typically persecutory and attributed to danger from vengeful folks or grandiose in nature. The question of whether the “folie” or “madness” is contagious arises due to the propensity of the child to adopt these delusions under an imposed insular or restrictive environment. Separating the child from the environment dominated by the delusional adult usually is sufficient to reverse the symptoms due to reality testing.
Normative familial communication. In contrary to a delusional familial interaction, normative family traditions and values are a unifying psychosocial force and a source of bonding and loyalty from an early age. A ubiquitous example is the support of a local sports team, and the emotional turmoil associated with the team’s wins and losses, accompanied by “hating” a rival. These family rituals are commonly devoid of emotional negative consequences for an impressionable young mind unless the child is exposed to unsportsmanlike emotional, verbal, or aggressive behavior by an adult at home in front of the television or in the stands at a game.
Continue to: Unfortunately...
Unfortunately, the “love-hate” dichotomy rooted in family-generated traditions of loyalty is becoming more evident in today’s turbulent sociopolitical environment. Children and young adolescents are not prepared to cope with the stressful effects of repeated exposure to intense conflictual events at home when parents adopt opposing sociopolitical ideologies. Furthermore, a parent might intentionally expose their child to emotionally conflictual circumstances in the name of a perceived value that might create and exacerbate stress, fear, and self-loathing. Ironically, by doing what a parent believes is right for their child, they might be transforming the child without their consent into a variant of a “toy soldier by proxy.” Such a child is a tool expected to follow the parental pathway and belief system without questioning, or even having the cognitive ability to do so, given their ongoing bio-behavioral and moral developmental phase.3
This new normative exposure to conflictual situations at the will of the parent is not only limited to watching them remotely but also may include participating in what is meant to be a peaceful protest or march. As we all witnessed in 2020, such events can easily deteriorate into unsafe environments rife with lawlessness and uncontrolled violence. This has included clashes between opposing groups who are matched in zeal and conviction, as well as opposition to or endangerment by law enforcement personnel trying to restore order by force. This is not where a responsible parent should take their child. Furthermore, there is the danger of loss of privacy of children exposed by media following their participation in public activity. This may lead to hate mail as that would further confuse and jeopardize a peaceful lifestyle, which is highly desirable for a developing child.
Cognitive dissonance. Have these parents temporarily allowed the limbic system to trump the restraints of the prefrontal cortex, as exhibited by an impulsive and risky behavior driven by poor insight? Have these parents thoughtfully weighed the balance between the merit of a child’s exposure to such conflictual circumstances and the peril of negative emotional consequences? This is illustrated by a mother who has been taking her preadolescent son to demonstrations regularly because “I want him to see how democracy works.”
Might this be a case of cognitive dissonance (CD) that amounts to unwitting mental child abuse if it happens repeatedly? According to the CD theory, there is a tendency to seek consistency between cognitions (eg, beliefs, opinions) and attitudes or behaviors. Inconsistency between these variables is termed “dissonance.”4,5 The importance attached to the dissonant belief affects the severity of the dissonance. The dissonance occurs when a parent must choose between 2 incompatible beliefs or actions. A classic demonstration of CD is when an adult requests that an adolescent follows his instructions (eg, “do not smoke or drink alcohol”), yet the adult does not act accordingly (eg, they smoke or drink). Role modeling demonstrated by such a discrepancy is a cause of confusion in the child. In terms of this article, the CD is between what the parent believes is an important learning experience by exercising the perceived right to pass to the child the parental value system vs compromising the protection of the child by exposing them to the potential negative consequences of a risky situation.
What can parents and therapists do?
Usually, parents mean well. It is important to communicate to parents the importance of refraining from forcing their children to join their battles. Calculating risks based on an intuitive approach is flawed because doing so is based on beliefs and emotions that originated in the limbic system (“I feel that”…) and are neither precise nor accurate.6 Teaching our youth in the school system how to think (eg, the science of logic and history of science) vs what to think (ie, indoctrination) is a key to healthy cognitive development. Furthermore, children need to have the time, space, and opportunities (learning moments) to develop this capacity. It is not until approximately age 16 that abstract thinking capabilities are developed. Cognitive dissonance can be eliminated by reducing the valence of the conflicting beliefs or by removing the conflicting attitude or behavior.
As parents and as mental health professionals, we should carry the necessary burden of responsibility to prevent the risk of “lost childhood” due to parental emotional zeal and righteousness that lead to early exposure to damaging adversity. We cannot afford to turn our children into exploitable tools (ie, toy soldiers) in conflicts they do not fully grasp.
1. Bagot KS, Kaminer Y. Harm reduction for youth in treatment with substance use disorders: one size does not fit all. Curr Addict Rep. 2018;5:379-385.
2. Arnon D, Patel A, Tan GM. The nosological significance of Folie à Deux: a review of the literature. Ann Gen Psychiatry. 2006;5:11.
3. Kohlberg L. The philosophy of moral development: the nature and validity of moral stages. Harper & Row; 1984.
4. Festinger L. A theory of cognitive dissonance. Stanford University Press; 1957.
5. Festinger L. Cognitive dissonance. Sci Am. 1962;207:93-102.
6. Henderson SW, Gerson R, Phillips B. What is “high risk” and what are we actually supposed to do about it? J Am Acad Child Adolesc Psychiatry. 2019;58(6):561-564.
1. Bagot KS, Kaminer Y. Harm reduction for youth in treatment with substance use disorders: one size does not fit all. Curr Addict Rep. 2018;5:379-385.
2. Arnon D, Patel A, Tan GM. The nosological significance of Folie à Deux: a review of the literature. Ann Gen Psychiatry. 2006;5:11.
3. Kohlberg L. The philosophy of moral development: the nature and validity of moral stages. Harper & Row; 1984.
4. Festinger L. A theory of cognitive dissonance. Stanford University Press; 1957.
5. Festinger L. Cognitive dissonance. Sci Am. 1962;207:93-102.
6. Henderson SW, Gerson R, Phillips B. What is “high risk” and what are we actually supposed to do about it? J Am Acad Child Adolesc Psychiatry. 2019;58(6):561-564.
Worried parents scramble to vaccinate kids despite FDA guidance
One week after reporting promising results from the trial of their COVID-19 vaccine in children ages 5-11, Pfizer and BioNTech announced they’d submitted the data to the Food and Drug Administration. But that hasn’t stopped some parents from discreetly getting their children under age 12 vaccinated.
“The FDA, you never want to get ahead of their judgment,” Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told MSNBC on Sept. 28. “But I would imagine in the next few weeks, they will examine that data and hopefully they’ll give the okay so that we can start vaccinating children, hopefully before the end of October.”
Lying to vaccinate now
More than half of all parents with children under 12 say they plan to get their kids vaccinated, according to a Gallup poll.
And although the FDA and the American Academy of Pediatrics have warned against it, some parents whose children can pass for 12 have lied to get them vaccinated already.
Dawn G. is a mom of two in southwest Missouri, where less than 45% of the population has been fully vaccinated. Her son turns 12 in early October, but in-person school started in mid-August.
“It was scary, thinking of him going to school for even 2 months,” she said. “Some parents thought their kid had a low chance of getting COVID, and their kid died. Nobody expects it to be them.”
In July, she and her husband took their son to a walk-in clinic and lied about his age.
“So many things can happen, from bullying to school shootings, and now this added pandemic risk,” she said. “I’ll do anything I can to protect my child, and a birthdate seems so arbitrary. He’ll be 12 in a matter of weeks. It seems ridiculous that that date would stop me from protecting him.”
In northern California, Carrie S. had a similar thought. When the vaccine was authorized for children ages 12-15 in May, the older of her two children got the shot right away. But her youngest doesn’t turn 12 until November.
“We were tempted to get the younger one vaccinated in May, but it didn’t seem like a rush. We were willing to wait to get the dosage right,” she ssaid. “But as Delta came through, there were no options for online school, the CDC was dropping mask expectations –it seemed like the world was ready to forget the pandemic was happening. It seemed like the least-bad option to get her vaccinated so she could go back to school, and we could find some balance of risk in our lives.”
Adult vs. pediatric doses
For now, experts advise against getting younger children vaccinated, even those who are the size of an adult, because of the way the human immune system develops.
“It’s not really about size,” said Anne Liu, MD, an immunologist and pediatrics professor at Stanford (Calif.) University. “The immune system behaves differently at different ages. Younger kids tend to have a more exuberant innate immune system, which is the part of the immune system that senses danger, even before it has developed a memory response.”
The adult Pfizer-BioNTech vaccine contains 30 mcg of mRNA, while the pediatric dose is just 10 mcg. That smaller dose produces an immune response similar to what’s seen in adults who receive 30 mcg, according to Pfizer.
“We were one of the sites that was involved in the phase 1 trial, a lot of times that’s called a dose-finding trial,” said Michael Smith, MD, a coinvestigator for the COVID vaccine trials done at Duke University. “And basically, if younger kids got a higher dose, they had more of a reaction, so it hurt more. They had fever, they had more redness and swelling at the site of the injection, and they just felt lousy, more than at the lower doses.”
At this point, with Pfizer’s data showing that younger children need a smaller dose, it doesn’t make sense to lie about your child’s age, said Dr. Smith.
“If my two options were having my child get the infection versus getting the vaccine, I’d get the vaccine. But we’re a few weeks away from getting the lower dose approved in kids,” he said. “It’s certainly safer. I don’t expect major, lifelong side effects from the higher dose, but it’s going to hurt, your kid’s going to have a fever, they’re going to feel lousy for a couple days, and they just don’t need that much antigen.”
A version of this article first appeared on WebMD.com.
One week after reporting promising results from the trial of their COVID-19 vaccine in children ages 5-11, Pfizer and BioNTech announced they’d submitted the data to the Food and Drug Administration. But that hasn’t stopped some parents from discreetly getting their children under age 12 vaccinated.
“The FDA, you never want to get ahead of their judgment,” Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told MSNBC on Sept. 28. “But I would imagine in the next few weeks, they will examine that data and hopefully they’ll give the okay so that we can start vaccinating children, hopefully before the end of October.”
Lying to vaccinate now
More than half of all parents with children under 12 say they plan to get their kids vaccinated, according to a Gallup poll.
And although the FDA and the American Academy of Pediatrics have warned against it, some parents whose children can pass for 12 have lied to get them vaccinated already.
Dawn G. is a mom of two in southwest Missouri, where less than 45% of the population has been fully vaccinated. Her son turns 12 in early October, but in-person school started in mid-August.
“It was scary, thinking of him going to school for even 2 months,” she said. “Some parents thought their kid had a low chance of getting COVID, and their kid died. Nobody expects it to be them.”
In July, she and her husband took their son to a walk-in clinic and lied about his age.
“So many things can happen, from bullying to school shootings, and now this added pandemic risk,” she said. “I’ll do anything I can to protect my child, and a birthdate seems so arbitrary. He’ll be 12 in a matter of weeks. It seems ridiculous that that date would stop me from protecting him.”
In northern California, Carrie S. had a similar thought. When the vaccine was authorized for children ages 12-15 in May, the older of her two children got the shot right away. But her youngest doesn’t turn 12 until November.
“We were tempted to get the younger one vaccinated in May, but it didn’t seem like a rush. We were willing to wait to get the dosage right,” she ssaid. “But as Delta came through, there were no options for online school, the CDC was dropping mask expectations –it seemed like the world was ready to forget the pandemic was happening. It seemed like the least-bad option to get her vaccinated so she could go back to school, and we could find some balance of risk in our lives.”
Adult vs. pediatric doses
For now, experts advise against getting younger children vaccinated, even those who are the size of an adult, because of the way the human immune system develops.
“It’s not really about size,” said Anne Liu, MD, an immunologist and pediatrics professor at Stanford (Calif.) University. “The immune system behaves differently at different ages. Younger kids tend to have a more exuberant innate immune system, which is the part of the immune system that senses danger, even before it has developed a memory response.”
The adult Pfizer-BioNTech vaccine contains 30 mcg of mRNA, while the pediatric dose is just 10 mcg. That smaller dose produces an immune response similar to what’s seen in adults who receive 30 mcg, according to Pfizer.
“We were one of the sites that was involved in the phase 1 trial, a lot of times that’s called a dose-finding trial,” said Michael Smith, MD, a coinvestigator for the COVID vaccine trials done at Duke University. “And basically, if younger kids got a higher dose, they had more of a reaction, so it hurt more. They had fever, they had more redness and swelling at the site of the injection, and they just felt lousy, more than at the lower doses.”
At this point, with Pfizer’s data showing that younger children need a smaller dose, it doesn’t make sense to lie about your child’s age, said Dr. Smith.
“If my two options were having my child get the infection versus getting the vaccine, I’d get the vaccine. But we’re a few weeks away from getting the lower dose approved in kids,” he said. “It’s certainly safer. I don’t expect major, lifelong side effects from the higher dose, but it’s going to hurt, your kid’s going to have a fever, they’re going to feel lousy for a couple days, and they just don’t need that much antigen.”
A version of this article first appeared on WebMD.com.
One week after reporting promising results from the trial of their COVID-19 vaccine in children ages 5-11, Pfizer and BioNTech announced they’d submitted the data to the Food and Drug Administration. But that hasn’t stopped some parents from discreetly getting their children under age 12 vaccinated.
“The FDA, you never want to get ahead of their judgment,” Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told MSNBC on Sept. 28. “But I would imagine in the next few weeks, they will examine that data and hopefully they’ll give the okay so that we can start vaccinating children, hopefully before the end of October.”
Lying to vaccinate now
More than half of all parents with children under 12 say they plan to get their kids vaccinated, according to a Gallup poll.
And although the FDA and the American Academy of Pediatrics have warned against it, some parents whose children can pass for 12 have lied to get them vaccinated already.
Dawn G. is a mom of two in southwest Missouri, where less than 45% of the population has been fully vaccinated. Her son turns 12 in early October, but in-person school started in mid-August.
“It was scary, thinking of him going to school for even 2 months,” she said. “Some parents thought their kid had a low chance of getting COVID, and their kid died. Nobody expects it to be them.”
In July, she and her husband took their son to a walk-in clinic and lied about his age.
“So many things can happen, from bullying to school shootings, and now this added pandemic risk,” she said. “I’ll do anything I can to protect my child, and a birthdate seems so arbitrary. He’ll be 12 in a matter of weeks. It seems ridiculous that that date would stop me from protecting him.”
In northern California, Carrie S. had a similar thought. When the vaccine was authorized for children ages 12-15 in May, the older of her two children got the shot right away. But her youngest doesn’t turn 12 until November.
“We were tempted to get the younger one vaccinated in May, but it didn’t seem like a rush. We were willing to wait to get the dosage right,” she ssaid. “But as Delta came through, there were no options for online school, the CDC was dropping mask expectations –it seemed like the world was ready to forget the pandemic was happening. It seemed like the least-bad option to get her vaccinated so she could go back to school, and we could find some balance of risk in our lives.”
Adult vs. pediatric doses
For now, experts advise against getting younger children vaccinated, even those who are the size of an adult, because of the way the human immune system develops.
“It’s not really about size,” said Anne Liu, MD, an immunologist and pediatrics professor at Stanford (Calif.) University. “The immune system behaves differently at different ages. Younger kids tend to have a more exuberant innate immune system, which is the part of the immune system that senses danger, even before it has developed a memory response.”
The adult Pfizer-BioNTech vaccine contains 30 mcg of mRNA, while the pediatric dose is just 10 mcg. That smaller dose produces an immune response similar to what’s seen in adults who receive 30 mcg, according to Pfizer.
“We were one of the sites that was involved in the phase 1 trial, a lot of times that’s called a dose-finding trial,” said Michael Smith, MD, a coinvestigator for the COVID vaccine trials done at Duke University. “And basically, if younger kids got a higher dose, they had more of a reaction, so it hurt more. They had fever, they had more redness and swelling at the site of the injection, and they just felt lousy, more than at the lower doses.”
At this point, with Pfizer’s data showing that younger children need a smaller dose, it doesn’t make sense to lie about your child’s age, said Dr. Smith.
“If my two options were having my child get the infection versus getting the vaccine, I’d get the vaccine. But we’re a few weeks away from getting the lower dose approved in kids,” he said. “It’s certainly safer. I don’t expect major, lifelong side effects from the higher dose, but it’s going to hurt, your kid’s going to have a fever, they’re going to feel lousy for a couple days, and they just don’t need that much antigen.”
A version of this article first appeared on WebMD.com.
Acceptance of biosimilars grows but greater use may hinge on switching, interchangeability studies
It took years for Elle Moxley to get a diagnosis that explained her crippling gastrointestinal pain, digestion problems, fatigue, and hot, red rashes. And after learning in 2016 that she had Crohn’s disease, a chronic inflammation of the digestive tract, she spent more than 4 years trying medications before getting her disease under control with a biologic drug called Remicade.
So Ms. Moxley, 33, was dismayed to receive a notice from her insurer in January that Remicade would no longer be covered as a preferred drug on her plan. Another drug, Inflectra, which the Food and Drug Administration says has no meaningful clinical differences from Remicade, is now preferred. It is a “biosimilar” drug.
“I felt very powerless,” said Ms. Moxley, who recently started a job as a public relations coordinator for Kansas City (Mo.) Public Schools. “I have this decision being made for me and my doctor that’s not in my best interest, and it might knock me out of remission.”
After Ms. Moxley’s first Inflectra infusion in July, she developed a painful rash. It went away after a few days, but she said she continues to feel extremely fatigued and experiences gastrointestinal pain, constipation, diarrhea and nausea.
Many medical professionals look to biosimilar drugs as a way to increase competition and give consumers cheaper options, much as generic drugs do, and they point to the more robust use of these products in Europe to cut costs.
Yet the United States has been slower to adopt biosimilar drugs since the first such medicine was approved in 2015. That’s partly because of concerns raised by patients like Moxley and their doctors, but also because brand-name biologics have kept biosimilars from entering the market. The companies behind the brand-name drugs have used legal actions to extend the life of their patents and incentives that make offering the brand biologic more attractive than offering a biosimilar on a formulary, listing which drugs are covered on an insurance plan.
“It distorts the market and makes it so that patients can’t get access,” said Jinoos Yazdany, MD, MPH, a professor of medicine and chief of the rheumatology division at Zuckerberg San Francisco General Hospital.
The FDA has approved 31 biosimilar medications since 2015, but only about 60% have made it to market, according to an analysis by NORC, a research organization at the University of Chicago.
Remicade’s manufacturer, Johnson & Johnson, and Pfizer, which makes the Remicade biosimilar Inflectra, have been embroiled in a long-running lawsuit over Pfizer’s claims that Johnson & Johnson tried to choke off competition through exclusionary contracts with insurers and other anticompetitive actions. In July, the companies settled the case on undisclosed terms.
In a statement, Pfizer said it would continue to sell Inflectra in the United States but noted ongoing challenges: “Pfizer has begun to see progress in the overall biosimilars marketplace in the U.S. However, changes in policy at a government level and acceptance of biosimilars among key stakeholders are critical to deliver more meaningful uptake so patients and the health care system at large can benefit from the cost savings these medicines may deliver.”
Johnson & Johnson said it is committed to making Remicade available to patients who choose it, which “compels us to compete responsibly on both price and value.”
Biologic medicines, which are generally grown from living organisms such as animal cells or bacteria, are more complex and expensive to manufacture than drugs made from chemicals. In recent years, biologic drugs have become a mainstay of treatment for autoimmune conditions like Crohn’s disease and rheumatoid arthritis, as well as certain cancers and diabetes, among other conditions.
Other drugmakers can’t exactly reproduce these biologic drugs by following chemical recipes as they do for generic versions of conventional drugs.
Instead, biosimilar versions of biologic drugs are generally made from the same types of materials as the original biologics and must be “highly similar” to them to be approved by the FDA. They must have no clinically meaningful differences from the biologic drug, and be just as safe, pure and potent. More than a decade after Congress created an approval pathway for biosimilars, they are widely accepted as safe and effective alternatives to brand biologics.
Medical experts hope that as biosimilars become more widely used they will increasingly provide a brake on drug spending.
From 2015 to 2019, drug spending overall grew 6.1%, while spending on biologics grew more than twice as much – 14.6% – according to a report by IQVIA, a health care analytics company. In 2019, biologics accounted for 43% of drug spending in the United States
Biosimilars provide a roughly 30% discount over brand biologics in the United States but have the potential to reduce spending by more than $100 billion in the next 5 years, the IQVIA analysis found.
In a survey of 602 physicians who prescribe biologic medications, more than three-quarters said they believed biosimilars are just as safe and effective as their biologic counterparts, according to NORC.
But they were less comfortable with switching patients from a brand biologic to a biosimilar. While about half said they were very likely to prescribe a biosimilar to a patient just starting biologic therapy, only 31% said they were very likely to prescribe a biosimilar to a patient already doing well on a brand biologic.
It can be challenging to find a treatment regimen that works for patients with complicated chronic conditions, and physicians and patients often don’t want to rock the boat once that is achieved.
In Ms. Moxley’s case, for example, before her condition stabilized on Remicade, she tried a conventional pill called Lialda, the biologic drug Humira and a lower dose of Remicade.
Some doctors and patients raise concerns that switching between these drugs might cause patients to develop antibodies that cause the drugs to lose effectiveness. They want to see more research about the effects of such switches.
“We haven’t seen enough studies about patients going from the biologic to the biosimilar and bouncing back and forth,” said Marcus Snow, MD, chair of the American College of Rheumatology’s Committee on Rheumatologic Care. “We don’t want our patients to be guinea pigs.”
Manufacturers of biologic and biosimilar drugs have participated in advertising, exhibit or sponsorship opportunities with the American College of Rheumatology, according to ACR spokesperson Jocelyn Givens.
But studies show a one-time switch from Remicade to a biosimilar like Inflectra does not cause side effects or the development of antibodies, said Ross Maltz, MD, a pediatric gastroenterologist at Nationwide Children’s Hospital in Columbus, Ohio, and former member of the Crohn’s & Colitis Foundation’s National Scientific Advisory Committee. Studies may be conducted by researchers with extensive ties to the industry and funded by drugmakers.
Situations like Ms. Moxley’s are unusual, said Kristine Grow, senior vice president of communications at AHIP, an insurer trade group.
“For patients who have been taking a brand-name biologic for some time, health insurance providers do not typically encourage them to switch to a biosimilar because of a formulary change, and most plans exclude these patients from any changes in cost sharing due to formulary changes,” she said.
Drugmakers can seek approval from the FDA of their biosimilar as interchangeable with a biologic drug, allowing pharmacists, subject to state law, to switch a physician’s prescription from the brand drug, as they often do with generic drugs.
However, the FDA has approved only one biosimilar (Semglee, a form of insulin) as interchangeable with a biologic (Lantus).
Like Ms. Moxley, many other patients using biologics get copay assistance from drug companies, but the money often isn’t enough to cover the full cost. In her old job as a radio reporter, Ms. Moxley said, she hit the $7,000 maximum annual out-of-pocket spending limit for her plan by May.
In her new job, Ms. Moxley has an individual plan with a $4,000 maximum out-of-pocket limit, which she expects to blow past once again within months.
But she received good news recently: Her new plan will cover Remicade.
“I’m still concerned that I will have developed antibodies since my last dose,” she said. “But it feels like a step in the direction of good health again.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
It took years for Elle Moxley to get a diagnosis that explained her crippling gastrointestinal pain, digestion problems, fatigue, and hot, red rashes. And after learning in 2016 that she had Crohn’s disease, a chronic inflammation of the digestive tract, she spent more than 4 years trying medications before getting her disease under control with a biologic drug called Remicade.
So Ms. Moxley, 33, was dismayed to receive a notice from her insurer in January that Remicade would no longer be covered as a preferred drug on her plan. Another drug, Inflectra, which the Food and Drug Administration says has no meaningful clinical differences from Remicade, is now preferred. It is a “biosimilar” drug.
“I felt very powerless,” said Ms. Moxley, who recently started a job as a public relations coordinator for Kansas City (Mo.) Public Schools. “I have this decision being made for me and my doctor that’s not in my best interest, and it might knock me out of remission.”
After Ms. Moxley’s first Inflectra infusion in July, she developed a painful rash. It went away after a few days, but she said she continues to feel extremely fatigued and experiences gastrointestinal pain, constipation, diarrhea and nausea.
Many medical professionals look to biosimilar drugs as a way to increase competition and give consumers cheaper options, much as generic drugs do, and they point to the more robust use of these products in Europe to cut costs.
Yet the United States has been slower to adopt biosimilar drugs since the first such medicine was approved in 2015. That’s partly because of concerns raised by patients like Moxley and their doctors, but also because brand-name biologics have kept biosimilars from entering the market. The companies behind the brand-name drugs have used legal actions to extend the life of their patents and incentives that make offering the brand biologic more attractive than offering a biosimilar on a formulary, listing which drugs are covered on an insurance plan.
“It distorts the market and makes it so that patients can’t get access,” said Jinoos Yazdany, MD, MPH, a professor of medicine and chief of the rheumatology division at Zuckerberg San Francisco General Hospital.
The FDA has approved 31 biosimilar medications since 2015, but only about 60% have made it to market, according to an analysis by NORC, a research organization at the University of Chicago.
Remicade’s manufacturer, Johnson & Johnson, and Pfizer, which makes the Remicade biosimilar Inflectra, have been embroiled in a long-running lawsuit over Pfizer’s claims that Johnson & Johnson tried to choke off competition through exclusionary contracts with insurers and other anticompetitive actions. In July, the companies settled the case on undisclosed terms.
In a statement, Pfizer said it would continue to sell Inflectra in the United States but noted ongoing challenges: “Pfizer has begun to see progress in the overall biosimilars marketplace in the U.S. However, changes in policy at a government level and acceptance of biosimilars among key stakeholders are critical to deliver more meaningful uptake so patients and the health care system at large can benefit from the cost savings these medicines may deliver.”
Johnson & Johnson said it is committed to making Remicade available to patients who choose it, which “compels us to compete responsibly on both price and value.”
Biologic medicines, which are generally grown from living organisms such as animal cells or bacteria, are more complex and expensive to manufacture than drugs made from chemicals. In recent years, biologic drugs have become a mainstay of treatment for autoimmune conditions like Crohn’s disease and rheumatoid arthritis, as well as certain cancers and diabetes, among other conditions.
Other drugmakers can’t exactly reproduce these biologic drugs by following chemical recipes as they do for generic versions of conventional drugs.
Instead, biosimilar versions of biologic drugs are generally made from the same types of materials as the original biologics and must be “highly similar” to them to be approved by the FDA. They must have no clinically meaningful differences from the biologic drug, and be just as safe, pure and potent. More than a decade after Congress created an approval pathway for biosimilars, they are widely accepted as safe and effective alternatives to brand biologics.
Medical experts hope that as biosimilars become more widely used they will increasingly provide a brake on drug spending.
From 2015 to 2019, drug spending overall grew 6.1%, while spending on biologics grew more than twice as much – 14.6% – according to a report by IQVIA, a health care analytics company. In 2019, biologics accounted for 43% of drug spending in the United States
Biosimilars provide a roughly 30% discount over brand biologics in the United States but have the potential to reduce spending by more than $100 billion in the next 5 years, the IQVIA analysis found.
In a survey of 602 physicians who prescribe biologic medications, more than three-quarters said they believed biosimilars are just as safe and effective as their biologic counterparts, according to NORC.
But they were less comfortable with switching patients from a brand biologic to a biosimilar. While about half said they were very likely to prescribe a biosimilar to a patient just starting biologic therapy, only 31% said they were very likely to prescribe a biosimilar to a patient already doing well on a brand biologic.
It can be challenging to find a treatment regimen that works for patients with complicated chronic conditions, and physicians and patients often don’t want to rock the boat once that is achieved.
In Ms. Moxley’s case, for example, before her condition stabilized on Remicade, she tried a conventional pill called Lialda, the biologic drug Humira and a lower dose of Remicade.
Some doctors and patients raise concerns that switching between these drugs might cause patients to develop antibodies that cause the drugs to lose effectiveness. They want to see more research about the effects of such switches.
“We haven’t seen enough studies about patients going from the biologic to the biosimilar and bouncing back and forth,” said Marcus Snow, MD, chair of the American College of Rheumatology’s Committee on Rheumatologic Care. “We don’t want our patients to be guinea pigs.”
Manufacturers of biologic and biosimilar drugs have participated in advertising, exhibit or sponsorship opportunities with the American College of Rheumatology, according to ACR spokesperson Jocelyn Givens.
But studies show a one-time switch from Remicade to a biosimilar like Inflectra does not cause side effects or the development of antibodies, said Ross Maltz, MD, a pediatric gastroenterologist at Nationwide Children’s Hospital in Columbus, Ohio, and former member of the Crohn’s & Colitis Foundation’s National Scientific Advisory Committee. Studies may be conducted by researchers with extensive ties to the industry and funded by drugmakers.
Situations like Ms. Moxley’s are unusual, said Kristine Grow, senior vice president of communications at AHIP, an insurer trade group.
“For patients who have been taking a brand-name biologic for some time, health insurance providers do not typically encourage them to switch to a biosimilar because of a formulary change, and most plans exclude these patients from any changes in cost sharing due to formulary changes,” she said.
Drugmakers can seek approval from the FDA of their biosimilar as interchangeable with a biologic drug, allowing pharmacists, subject to state law, to switch a physician’s prescription from the brand drug, as they often do with generic drugs.
However, the FDA has approved only one biosimilar (Semglee, a form of insulin) as interchangeable with a biologic (Lantus).
Like Ms. Moxley, many other patients using biologics get copay assistance from drug companies, but the money often isn’t enough to cover the full cost. In her old job as a radio reporter, Ms. Moxley said, she hit the $7,000 maximum annual out-of-pocket spending limit for her plan by May.
In her new job, Ms. Moxley has an individual plan with a $4,000 maximum out-of-pocket limit, which she expects to blow past once again within months.
But she received good news recently: Her new plan will cover Remicade.
“I’m still concerned that I will have developed antibodies since my last dose,” she said. “But it feels like a step in the direction of good health again.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
It took years for Elle Moxley to get a diagnosis that explained her crippling gastrointestinal pain, digestion problems, fatigue, and hot, red rashes. And after learning in 2016 that she had Crohn’s disease, a chronic inflammation of the digestive tract, she spent more than 4 years trying medications before getting her disease under control with a biologic drug called Remicade.
So Ms. Moxley, 33, was dismayed to receive a notice from her insurer in January that Remicade would no longer be covered as a preferred drug on her plan. Another drug, Inflectra, which the Food and Drug Administration says has no meaningful clinical differences from Remicade, is now preferred. It is a “biosimilar” drug.
“I felt very powerless,” said Ms. Moxley, who recently started a job as a public relations coordinator for Kansas City (Mo.) Public Schools. “I have this decision being made for me and my doctor that’s not in my best interest, and it might knock me out of remission.”
After Ms. Moxley’s first Inflectra infusion in July, she developed a painful rash. It went away after a few days, but she said she continues to feel extremely fatigued and experiences gastrointestinal pain, constipation, diarrhea and nausea.
Many medical professionals look to biosimilar drugs as a way to increase competition and give consumers cheaper options, much as generic drugs do, and they point to the more robust use of these products in Europe to cut costs.
Yet the United States has been slower to adopt biosimilar drugs since the first such medicine was approved in 2015. That’s partly because of concerns raised by patients like Moxley and their doctors, but also because brand-name biologics have kept biosimilars from entering the market. The companies behind the brand-name drugs have used legal actions to extend the life of their patents and incentives that make offering the brand biologic more attractive than offering a biosimilar on a formulary, listing which drugs are covered on an insurance plan.
“It distorts the market and makes it so that patients can’t get access,” said Jinoos Yazdany, MD, MPH, a professor of medicine and chief of the rheumatology division at Zuckerberg San Francisco General Hospital.
The FDA has approved 31 biosimilar medications since 2015, but only about 60% have made it to market, according to an analysis by NORC, a research organization at the University of Chicago.
Remicade’s manufacturer, Johnson & Johnson, and Pfizer, which makes the Remicade biosimilar Inflectra, have been embroiled in a long-running lawsuit over Pfizer’s claims that Johnson & Johnson tried to choke off competition through exclusionary contracts with insurers and other anticompetitive actions. In July, the companies settled the case on undisclosed terms.
In a statement, Pfizer said it would continue to sell Inflectra in the United States but noted ongoing challenges: “Pfizer has begun to see progress in the overall biosimilars marketplace in the U.S. However, changes in policy at a government level and acceptance of biosimilars among key stakeholders are critical to deliver more meaningful uptake so patients and the health care system at large can benefit from the cost savings these medicines may deliver.”
Johnson & Johnson said it is committed to making Remicade available to patients who choose it, which “compels us to compete responsibly on both price and value.”
Biologic medicines, which are generally grown from living organisms such as animal cells or bacteria, are more complex and expensive to manufacture than drugs made from chemicals. In recent years, biologic drugs have become a mainstay of treatment for autoimmune conditions like Crohn’s disease and rheumatoid arthritis, as well as certain cancers and diabetes, among other conditions.
Other drugmakers can’t exactly reproduce these biologic drugs by following chemical recipes as they do for generic versions of conventional drugs.
Instead, biosimilar versions of biologic drugs are generally made from the same types of materials as the original biologics and must be “highly similar” to them to be approved by the FDA. They must have no clinically meaningful differences from the biologic drug, and be just as safe, pure and potent. More than a decade after Congress created an approval pathway for biosimilars, they are widely accepted as safe and effective alternatives to brand biologics.
Medical experts hope that as biosimilars become more widely used they will increasingly provide a brake on drug spending.
From 2015 to 2019, drug spending overall grew 6.1%, while spending on biologics grew more than twice as much – 14.6% – according to a report by IQVIA, a health care analytics company. In 2019, biologics accounted for 43% of drug spending in the United States
Biosimilars provide a roughly 30% discount over brand biologics in the United States but have the potential to reduce spending by more than $100 billion in the next 5 years, the IQVIA analysis found.
In a survey of 602 physicians who prescribe biologic medications, more than three-quarters said they believed biosimilars are just as safe and effective as their biologic counterparts, according to NORC.
But they were less comfortable with switching patients from a brand biologic to a biosimilar. While about half said they were very likely to prescribe a biosimilar to a patient just starting biologic therapy, only 31% said they were very likely to prescribe a biosimilar to a patient already doing well on a brand biologic.
It can be challenging to find a treatment regimen that works for patients with complicated chronic conditions, and physicians and patients often don’t want to rock the boat once that is achieved.
In Ms. Moxley’s case, for example, before her condition stabilized on Remicade, she tried a conventional pill called Lialda, the biologic drug Humira and a lower dose of Remicade.
Some doctors and patients raise concerns that switching between these drugs might cause patients to develop antibodies that cause the drugs to lose effectiveness. They want to see more research about the effects of such switches.
“We haven’t seen enough studies about patients going from the biologic to the biosimilar and bouncing back and forth,” said Marcus Snow, MD, chair of the American College of Rheumatology’s Committee on Rheumatologic Care. “We don’t want our patients to be guinea pigs.”
Manufacturers of biologic and biosimilar drugs have participated in advertising, exhibit or sponsorship opportunities with the American College of Rheumatology, according to ACR spokesperson Jocelyn Givens.
But studies show a one-time switch from Remicade to a biosimilar like Inflectra does not cause side effects or the development of antibodies, said Ross Maltz, MD, a pediatric gastroenterologist at Nationwide Children’s Hospital in Columbus, Ohio, and former member of the Crohn’s & Colitis Foundation’s National Scientific Advisory Committee. Studies may be conducted by researchers with extensive ties to the industry and funded by drugmakers.
Situations like Ms. Moxley’s are unusual, said Kristine Grow, senior vice president of communications at AHIP, an insurer trade group.
“For patients who have been taking a brand-name biologic for some time, health insurance providers do not typically encourage them to switch to a biosimilar because of a formulary change, and most plans exclude these patients from any changes in cost sharing due to formulary changes,” she said.
Drugmakers can seek approval from the FDA of their biosimilar as interchangeable with a biologic drug, allowing pharmacists, subject to state law, to switch a physician’s prescription from the brand drug, as they often do with generic drugs.
However, the FDA has approved only one biosimilar (Semglee, a form of insulin) as interchangeable with a biologic (Lantus).
Like Ms. Moxley, many other patients using biologics get copay assistance from drug companies, but the money often isn’t enough to cover the full cost. In her old job as a radio reporter, Ms. Moxley said, she hit the $7,000 maximum annual out-of-pocket spending limit for her plan by May.
In her new job, Ms. Moxley has an individual plan with a $4,000 maximum out-of-pocket limit, which she expects to blow past once again within months.
But she received good news recently: Her new plan will cover Remicade.
“I’m still concerned that I will have developed antibodies since my last dose,” she said. “But it feels like a step in the direction of good health again.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Dr. Judy C. Washington shows URM physicians how to lead
For URM physicians, she also imparts a shared experience of being a minority in the field and helps prepare them for the challenges of facing racism or feeling marginalized or not equitably supported in academic life – and for making change.
While family medicine’s demographics have become more diverse over time, and more so than other specialties, they are not yet representative of the U.S. population. Within academia, male physicians who are Black or African American, or Hispanic or Latino, comprised about 4% and 5% of family medicine faculty, respectively, at the end of 2019, according to data from the Association of American Medical Colleges. For women, these numbers were about 9% and 4%, respectively. (Only those with an MD degree exclusively were included in the report.)
“When you have the privilege to serve in leadership, you have the responsibility to reach back and identify and help others who would not otherwise have the opportunity to be recognized,” Dr. Washington said.
Her mentorship work stems in large part from her long-time involvement and leadership roles in the Society of Teachers of Family Medicine (STFM) – roles she considers a pillar of her professional life. She currently serves as president of the STFM Foundation and is associate chief medical officer of the Atlantic Medical Group, a large multisite physician-led organization. She is also coordinator of women’s health for the Overlook Family Medicine Residency Program, which is affiliated with Atlantic Medical Group.
In Dr. Washington’s role as associate chief medical officer of Atlantic Medical Group in Summit, N.J., she focuses on physician engagement, satisfaction, and diversity. She also assists in areas such as population health. For the Overlook Family Medicine Residency Program also in Summit, she precepts residents in the obstetrics clinic and in the family medicine outpatient clinic.
Diana N. Carvajal, MD, MPH, one of Dr. Washington’s mentees, called her an “inspirational leader” for young academic faculty and said she is a familiar speaker at STFM meetings on topics of workforce diversity, equity, and leadership. She is “passionate” about mentorship, Dr. Carvajal said, and has understood “that URMs and women of color were not always getting [the mentorship they need to be successful].”
Guiding future leaders
Ivonne McLean, MD, assistant professor of family and community medicine at Icahn School of Medicine at Mount Sinai, New York, and an attending at a community health center in the Bronx, called Dr. Washington for advice a couple of years ago when she was considering her next career move.
“She took a genuine interest in me. She never said, this is what you should do. But the questions she asked and the examples she gave from her own life were incredibly helpful to me [in deciding to pursue a research fellowship] ... it was a pivotal conversation,” said Dr. McLean, associate director of a reproductive health fellowship and a research fellow in a New York State–funded program.
“From a lived experience angle, she also told me, here are some of the challenges you’ll have as a woman of color, and here are some of the ways you can approach that,” she said.
Dr. Carvajal, also a URM family physician, credits Dr. Washington’s mentorship with the development of a day-long workshop – held before the annual Society of Teachers of Family Medicine (STFM) meeting – on the low and declining rates of Black males in medicine. “We’d planned it as a presentation, and [she heard of it and] helped us expand it,” she said, calling Dr. Washington “warm, welcoming, and encouraging.
“That work and collaboration with her and the others she brought [into the process] have resulted in publications and more presentations and strategy building for diversifying the workforce,” said Dr. Carvajal, assistant professor, director of reproductive health education in family medicine, and codirector of the research section, all in the department of family and community medicine at the University of Maryland, Baltimore.
STFM involvement
Dr. Washington, who says that all or almost all of her mentees are now leaders in their academic institutions and communities, has been instrumental in developing STFM’s mentoring programming and in facilitating the organization’s multifaceted URM Initiative.
She has been active in STFM since the start of her academic career, and in 2009, while serving as assistant program director for the residency program in which she’d trained, she joined two other African American women, Monique Y. Davis-Smith, MD, and Joedrecka Brown-Speights, MD, in cochairing the society’s Group on Minority and Multicultural Health.
It was in this space, that Dr. Washington said she “heard people’s stories of being in major academic institutions and not feeling supported, not being given roadmaps to success, not getting assistance with publishing, or just kind of feeling like an outsider ... of not being pulled in.” Hispanic and African American females, in particular, “were feeling marginalized,” she said.
In 2018, having co-led development of the STFM Quality Mentoring Program for URM faculty, Dr. Washington was asked to join the STFM Foundation and subsequently led the STFM Foundation’s fundraising campaign for a new URM Initiative. She exceeded her goal, increasing support for URM participation in meetings and activities, and then participated in an STFM steering committee to create broader and longer-lasting support for URM faculty, community teachers, and medical students and residents going into academic family medicine.
Increasing the percentage of URM family medicine faculty in leadership positions – and raising awareness of structural barriers to achievement – is one of the current pillars of the URM Initiative.
Navigating the ‘minority tax’
As part of her mentoring, Dr. Washington helps URM physicians navigate the minority tax – a term referring to the uncompensated citizenship tasks that are more often assigned to Black and other URM physicians than to White physicians, and that take time away from scholarship, further perpetuating inequities.
“Some of our young faculty members find themselves thrust into being the diversity and inclusion leaders in their institutions at a level at which they feel little power and little buy-in from [leadership],” she noted.
A commentary written by Dr. Washington and several colleagues on the minority tax as it impacts women – and the need to build a “tax shelter” to make academic medicine a more just environment for URM women – was published earlier this year in the Journal of Women’s Health.
She also answers e-mails and fields phone calls from young URM faculty who are mulling career moves and facing other familiar challenges.
Physicians who are URM, and African American physicians in particular, tend to “get pulled into the [often underserved] communities, into the patient care and community service areas,” Dr. Washington explained. “But unless you convert these projects into scholarship and publications, and unless you serve on a national committee outside of your institution, you’re not going to be promoted.”
Dr. Washington helps junior faculty envision themselves 5-plus years down the road, find what she calls scholarly “passion projects,” and prepare themselves for their next steps.
She helps her mentees navigate other parts of the continuum of unconscious bias and racism as well, from microaggressions from colleagues to overt discrimination from patients.
“I spend countless minutes fielding texts and phone calls from those who need support,” she wrote in a blog post. “They are a constant reminder that I must continue to speak up when I get the opportunity to do so.”
A journey through family medicine, and through bias and racism
Dr. Washington’s early days in medicine included graduating from Meharry Medical College in 1983 and the Mountainside Family Practice Residency Program in 1990. Following 6 years of working in a private practice in rural Maryland, she moved to academia, spending 6 years at East Tennessee State University and 4 years at the UMDNJ–New Jersey Medical School in Newark as an assistant professor of family medicine.
As had happened in rural Maryland, bias and racism have too often lurked during her career as a physician.
“I grew up in Alabama so I was pretty much ready to deal with racism in the South,” Dr. Washington said. “What I was not ready for was coming to the Northeast and seeing that you’re marginalized because you’re not invited into the room. Or if you do go into spaces when you’re the only one, you often don’t feel as welcomed as you thought you might be.”
Her ideas and contributions were too often dismissed, she wrote in a 2020 blog entry posted on her LinkedIn page. And during contract negotiations, “I was not aware of all the information that my White colleagues had. They had the advantage of inside information.”
Dr. Washington says that “it took a village” to make her who she is today: teachers in her segregated schools in Alabama, one of her college professors, her best friend in medical school – and STFM, “where the list [of her own mentors] is long.”
For URM physicians, she also imparts a shared experience of being a minority in the field and helps prepare them for the challenges of facing racism or feeling marginalized or not equitably supported in academic life – and for making change.
While family medicine’s demographics have become more diverse over time, and more so than other specialties, they are not yet representative of the U.S. population. Within academia, male physicians who are Black or African American, or Hispanic or Latino, comprised about 4% and 5% of family medicine faculty, respectively, at the end of 2019, according to data from the Association of American Medical Colleges. For women, these numbers were about 9% and 4%, respectively. (Only those with an MD degree exclusively were included in the report.)
“When you have the privilege to serve in leadership, you have the responsibility to reach back and identify and help others who would not otherwise have the opportunity to be recognized,” Dr. Washington said.
Her mentorship work stems in large part from her long-time involvement and leadership roles in the Society of Teachers of Family Medicine (STFM) – roles she considers a pillar of her professional life. She currently serves as president of the STFM Foundation and is associate chief medical officer of the Atlantic Medical Group, a large multisite physician-led organization. She is also coordinator of women’s health for the Overlook Family Medicine Residency Program, which is affiliated with Atlantic Medical Group.
In Dr. Washington’s role as associate chief medical officer of Atlantic Medical Group in Summit, N.J., she focuses on physician engagement, satisfaction, and diversity. She also assists in areas such as population health. For the Overlook Family Medicine Residency Program also in Summit, she precepts residents in the obstetrics clinic and in the family medicine outpatient clinic.
Diana N. Carvajal, MD, MPH, one of Dr. Washington’s mentees, called her an “inspirational leader” for young academic faculty and said she is a familiar speaker at STFM meetings on topics of workforce diversity, equity, and leadership. She is “passionate” about mentorship, Dr. Carvajal said, and has understood “that URMs and women of color were not always getting [the mentorship they need to be successful].”
Guiding future leaders
Ivonne McLean, MD, assistant professor of family and community medicine at Icahn School of Medicine at Mount Sinai, New York, and an attending at a community health center in the Bronx, called Dr. Washington for advice a couple of years ago when she was considering her next career move.
“She took a genuine interest in me. She never said, this is what you should do. But the questions she asked and the examples she gave from her own life were incredibly helpful to me [in deciding to pursue a research fellowship] ... it was a pivotal conversation,” said Dr. McLean, associate director of a reproductive health fellowship and a research fellow in a New York State–funded program.
“From a lived experience angle, she also told me, here are some of the challenges you’ll have as a woman of color, and here are some of the ways you can approach that,” she said.
Dr. Carvajal, also a URM family physician, credits Dr. Washington’s mentorship with the development of a day-long workshop – held before the annual Society of Teachers of Family Medicine (STFM) meeting – on the low and declining rates of Black males in medicine. “We’d planned it as a presentation, and [she heard of it and] helped us expand it,” she said, calling Dr. Washington “warm, welcoming, and encouraging.
“That work and collaboration with her and the others she brought [into the process] have resulted in publications and more presentations and strategy building for diversifying the workforce,” said Dr. Carvajal, assistant professor, director of reproductive health education in family medicine, and codirector of the research section, all in the department of family and community medicine at the University of Maryland, Baltimore.
STFM involvement
Dr. Washington, who says that all or almost all of her mentees are now leaders in their academic institutions and communities, has been instrumental in developing STFM’s mentoring programming and in facilitating the organization’s multifaceted URM Initiative.
She has been active in STFM since the start of her academic career, and in 2009, while serving as assistant program director for the residency program in which she’d trained, she joined two other African American women, Monique Y. Davis-Smith, MD, and Joedrecka Brown-Speights, MD, in cochairing the society’s Group on Minority and Multicultural Health.
It was in this space, that Dr. Washington said she “heard people’s stories of being in major academic institutions and not feeling supported, not being given roadmaps to success, not getting assistance with publishing, or just kind of feeling like an outsider ... of not being pulled in.” Hispanic and African American females, in particular, “were feeling marginalized,” she said.
In 2018, having co-led development of the STFM Quality Mentoring Program for URM faculty, Dr. Washington was asked to join the STFM Foundation and subsequently led the STFM Foundation’s fundraising campaign for a new URM Initiative. She exceeded her goal, increasing support for URM participation in meetings and activities, and then participated in an STFM steering committee to create broader and longer-lasting support for URM faculty, community teachers, and medical students and residents going into academic family medicine.
Increasing the percentage of URM family medicine faculty in leadership positions – and raising awareness of structural barriers to achievement – is one of the current pillars of the URM Initiative.
Navigating the ‘minority tax’
As part of her mentoring, Dr. Washington helps URM physicians navigate the minority tax – a term referring to the uncompensated citizenship tasks that are more often assigned to Black and other URM physicians than to White physicians, and that take time away from scholarship, further perpetuating inequities.
“Some of our young faculty members find themselves thrust into being the diversity and inclusion leaders in their institutions at a level at which they feel little power and little buy-in from [leadership],” she noted.
A commentary written by Dr. Washington and several colleagues on the minority tax as it impacts women – and the need to build a “tax shelter” to make academic medicine a more just environment for URM women – was published earlier this year in the Journal of Women’s Health.
She also answers e-mails and fields phone calls from young URM faculty who are mulling career moves and facing other familiar challenges.
Physicians who are URM, and African American physicians in particular, tend to “get pulled into the [often underserved] communities, into the patient care and community service areas,” Dr. Washington explained. “But unless you convert these projects into scholarship and publications, and unless you serve on a national committee outside of your institution, you’re not going to be promoted.”
Dr. Washington helps junior faculty envision themselves 5-plus years down the road, find what she calls scholarly “passion projects,” and prepare themselves for their next steps.
She helps her mentees navigate other parts of the continuum of unconscious bias and racism as well, from microaggressions from colleagues to overt discrimination from patients.
“I spend countless minutes fielding texts and phone calls from those who need support,” she wrote in a blog post. “They are a constant reminder that I must continue to speak up when I get the opportunity to do so.”
A journey through family medicine, and through bias and racism
Dr. Washington’s early days in medicine included graduating from Meharry Medical College in 1983 and the Mountainside Family Practice Residency Program in 1990. Following 6 years of working in a private practice in rural Maryland, she moved to academia, spending 6 years at East Tennessee State University and 4 years at the UMDNJ–New Jersey Medical School in Newark as an assistant professor of family medicine.
As had happened in rural Maryland, bias and racism have too often lurked during her career as a physician.
“I grew up in Alabama so I was pretty much ready to deal with racism in the South,” Dr. Washington said. “What I was not ready for was coming to the Northeast and seeing that you’re marginalized because you’re not invited into the room. Or if you do go into spaces when you’re the only one, you often don’t feel as welcomed as you thought you might be.”
Her ideas and contributions were too often dismissed, she wrote in a 2020 blog entry posted on her LinkedIn page. And during contract negotiations, “I was not aware of all the information that my White colleagues had. They had the advantage of inside information.”
Dr. Washington says that “it took a village” to make her who she is today: teachers in her segregated schools in Alabama, one of her college professors, her best friend in medical school – and STFM, “where the list [of her own mentors] is long.”
For URM physicians, she also imparts a shared experience of being a minority in the field and helps prepare them for the challenges of facing racism or feeling marginalized or not equitably supported in academic life – and for making change.
While family medicine’s demographics have become more diverse over time, and more so than other specialties, they are not yet representative of the U.S. population. Within academia, male physicians who are Black or African American, or Hispanic or Latino, comprised about 4% and 5% of family medicine faculty, respectively, at the end of 2019, according to data from the Association of American Medical Colleges. For women, these numbers were about 9% and 4%, respectively. (Only those with an MD degree exclusively were included in the report.)
“When you have the privilege to serve in leadership, you have the responsibility to reach back and identify and help others who would not otherwise have the opportunity to be recognized,” Dr. Washington said.
Her mentorship work stems in large part from her long-time involvement and leadership roles in the Society of Teachers of Family Medicine (STFM) – roles she considers a pillar of her professional life. She currently serves as president of the STFM Foundation and is associate chief medical officer of the Atlantic Medical Group, a large multisite physician-led organization. She is also coordinator of women’s health for the Overlook Family Medicine Residency Program, which is affiliated with Atlantic Medical Group.
In Dr. Washington’s role as associate chief medical officer of Atlantic Medical Group in Summit, N.J., she focuses on physician engagement, satisfaction, and diversity. She also assists in areas such as population health. For the Overlook Family Medicine Residency Program also in Summit, she precepts residents in the obstetrics clinic and in the family medicine outpatient clinic.
Diana N. Carvajal, MD, MPH, one of Dr. Washington’s mentees, called her an “inspirational leader” for young academic faculty and said she is a familiar speaker at STFM meetings on topics of workforce diversity, equity, and leadership. She is “passionate” about mentorship, Dr. Carvajal said, and has understood “that URMs and women of color were not always getting [the mentorship they need to be successful].”
Guiding future leaders
Ivonne McLean, MD, assistant professor of family and community medicine at Icahn School of Medicine at Mount Sinai, New York, and an attending at a community health center in the Bronx, called Dr. Washington for advice a couple of years ago when she was considering her next career move.
“She took a genuine interest in me. She never said, this is what you should do. But the questions she asked and the examples she gave from her own life were incredibly helpful to me [in deciding to pursue a research fellowship] ... it was a pivotal conversation,” said Dr. McLean, associate director of a reproductive health fellowship and a research fellow in a New York State–funded program.
“From a lived experience angle, she also told me, here are some of the challenges you’ll have as a woman of color, and here are some of the ways you can approach that,” she said.
Dr. Carvajal, also a URM family physician, credits Dr. Washington’s mentorship with the development of a day-long workshop – held before the annual Society of Teachers of Family Medicine (STFM) meeting – on the low and declining rates of Black males in medicine. “We’d planned it as a presentation, and [she heard of it and] helped us expand it,” she said, calling Dr. Washington “warm, welcoming, and encouraging.
“That work and collaboration with her and the others she brought [into the process] have resulted in publications and more presentations and strategy building for diversifying the workforce,” said Dr. Carvajal, assistant professor, director of reproductive health education in family medicine, and codirector of the research section, all in the department of family and community medicine at the University of Maryland, Baltimore.
STFM involvement
Dr. Washington, who says that all or almost all of her mentees are now leaders in their academic institutions and communities, has been instrumental in developing STFM’s mentoring programming and in facilitating the organization’s multifaceted URM Initiative.
She has been active in STFM since the start of her academic career, and in 2009, while serving as assistant program director for the residency program in which she’d trained, she joined two other African American women, Monique Y. Davis-Smith, MD, and Joedrecka Brown-Speights, MD, in cochairing the society’s Group on Minority and Multicultural Health.
It was in this space, that Dr. Washington said she “heard people’s stories of being in major academic institutions and not feeling supported, not being given roadmaps to success, not getting assistance with publishing, or just kind of feeling like an outsider ... of not being pulled in.” Hispanic and African American females, in particular, “were feeling marginalized,” she said.
In 2018, having co-led development of the STFM Quality Mentoring Program for URM faculty, Dr. Washington was asked to join the STFM Foundation and subsequently led the STFM Foundation’s fundraising campaign for a new URM Initiative. She exceeded her goal, increasing support for URM participation in meetings and activities, and then participated in an STFM steering committee to create broader and longer-lasting support for URM faculty, community teachers, and medical students and residents going into academic family medicine.
Increasing the percentage of URM family medicine faculty in leadership positions – and raising awareness of structural barriers to achievement – is one of the current pillars of the URM Initiative.
Navigating the ‘minority tax’
As part of her mentoring, Dr. Washington helps URM physicians navigate the minority tax – a term referring to the uncompensated citizenship tasks that are more often assigned to Black and other URM physicians than to White physicians, and that take time away from scholarship, further perpetuating inequities.
“Some of our young faculty members find themselves thrust into being the diversity and inclusion leaders in their institutions at a level at which they feel little power and little buy-in from [leadership],” she noted.
A commentary written by Dr. Washington and several colleagues on the minority tax as it impacts women – and the need to build a “tax shelter” to make academic medicine a more just environment for URM women – was published earlier this year in the Journal of Women’s Health.
She also answers e-mails and fields phone calls from young URM faculty who are mulling career moves and facing other familiar challenges.
Physicians who are URM, and African American physicians in particular, tend to “get pulled into the [often underserved] communities, into the patient care and community service areas,” Dr. Washington explained. “But unless you convert these projects into scholarship and publications, and unless you serve on a national committee outside of your institution, you’re not going to be promoted.”
Dr. Washington helps junior faculty envision themselves 5-plus years down the road, find what she calls scholarly “passion projects,” and prepare themselves for their next steps.
She helps her mentees navigate other parts of the continuum of unconscious bias and racism as well, from microaggressions from colleagues to overt discrimination from patients.
“I spend countless minutes fielding texts and phone calls from those who need support,” she wrote in a blog post. “They are a constant reminder that I must continue to speak up when I get the opportunity to do so.”
A journey through family medicine, and through bias and racism
Dr. Washington’s early days in medicine included graduating from Meharry Medical College in 1983 and the Mountainside Family Practice Residency Program in 1990. Following 6 years of working in a private practice in rural Maryland, she moved to academia, spending 6 years at East Tennessee State University and 4 years at the UMDNJ–New Jersey Medical School in Newark as an assistant professor of family medicine.
As had happened in rural Maryland, bias and racism have too often lurked during her career as a physician.
“I grew up in Alabama so I was pretty much ready to deal with racism in the South,” Dr. Washington said. “What I was not ready for was coming to the Northeast and seeing that you’re marginalized because you’re not invited into the room. Or if you do go into spaces when you’re the only one, you often don’t feel as welcomed as you thought you might be.”
Her ideas and contributions were too often dismissed, she wrote in a 2020 blog entry posted on her LinkedIn page. And during contract negotiations, “I was not aware of all the information that my White colleagues had. They had the advantage of inside information.”
Dr. Washington says that “it took a village” to make her who she is today: teachers in her segregated schools in Alabama, one of her college professors, her best friend in medical school – and STFM, “where the list [of her own mentors] is long.”