User login
Focus on antivirals, vaccines as monkeypox continues
Since the first case of monkeypox on May 6, reports of outbreaks have come from multiple countries, with the United Kingdom, Spain, and Portugal in the lead, followed by Canada, Israel, and Australia, among others. The United States has reported cases in Boston and New York, and presumed cases have occurred in Utah and Florida. As of May 25, close to 350 cases, either suspected (83) or confirmed (265), have been reported globally.
Monkeypox outbreaks have previously been confined to Central and West Africa, except for an impressively large outbreak in the United States in 2003, during which 47 people were infected across six states. The epidemic was traced to a Gambian rat, rope squirrels, and dormice that had been imported from Ghana as pets and that had infected prairie dogs at a large wholesale pet store.
“It’s amazing how many of these viruses – COVID, now monkeypox and others – [exist]. They’re out there in the wild in the animal reservoir,” said Dennis Hruby, PhD, executive VP/chief scientific officer and scientific founder of SIGA Technologies.
“When it comes to the human population, they sometimes behave in ways we’re not expecting. That and a few mutations change those strains and pathogenicity and can be pandemic,” he told this news organization.
Now that the virus is pandemic, there is an urgent interest in medicines and vaccines that might halt its spread.
Smallpox drug tecovirimat
SIGA’s drug is tecovirimat, initially known as ST-246 and now branded as TPOXX. The U.S. Food and Drug Administration approved an oral formulation to treat smallpox in 2018. While smallpox was eradicated by 1980, there have been ongoing concerns about its potential use in a bioterrorism attack.
Tecovirimat is also approved for smallpox in Canada. In Europe, the approval includes treatment of monkeypox, cowpox, and complications from immunization with vaccinia. On May 19, the FDA approved an IV formulation of tecovirimat for those unable to tolerate oral medications.
In a press release, SIGA notes that tecovirimat was “developed through funding and collaboration with the Biomedical Advanced Research and Development Authority (BARDA) at the U.S. Department of Health & Human Services, as well as early-stage development supported by the National Institutes of Health, US Centers for Disease Control and Prevention, and the Department of Defense. Tecovirimat is stockpiled by the U.S. Government to mitigate the impact of a potential outbreak or bioterror attack.”
SIGA adds that, under Project Bioshield, “the United States maintains a stockpile of 1.7 million courses in the Strategic National Stockpile.” The drug is only available through the government’s stockpile.
Tecovirimat works by preventing the viruses from reproducing by interfering with a protein, VP37. The virus cannot escape the cell and so cannot infect other cells, Dr. Hruby explained.
Tecovirimat was developed under the FDA’s so-called Animal Rule, which allows approval on the basis of animal studies when human efficacy studies are unethical or impractical.
In a placebo-controlled human pharmacokinetic and safety study, only 2% of the 359 who received TPOXX had to have treatment stopped because of adverse reactions, a rate similar to placebo. The most common reactions (≥2%) were headache, nausea, and abdominal pain. Significant drug interactions were found with the coadministration of repaglinide and midazolam.
Of note is that tecovirimat’s efficacy may be reduced in immunocompromised patients. The smallpox vaccine is contraindicated for those who are immunocompromised. Those people should be offered vaccinia immune globulin.
With monkeypox, “the earlier the disease is recognized and you start treating, [the] more effective,” said Dr. Hruby. “In a monkey model which, much like humans, if we treat early on as the first lesions emerged or even several days after the lesions emerged, we see close to 100% protection.”
The other alternative drug for smallpox and (likely) monkeypox is Chimerix’s brincidofovir (BCV, Tembexa), a lipid conjugate of cidofovir, a drug for cytomegalovirus. Brincidofovir has a better safety profile than cidofovir and was also approved under the Animal Rule.
UpToDate suggests that tecovirimat is the drug of choice for monkeypox. They note that for severely infected patients, it can be combined with brincidofovir after consultation with the CDC or state health department officials.
Two vaccines available
Two vaccines are currently available. The oldest is ACAM2000, a replication-competent vaccine that replaced Dryvax, whose use was stopped in 1977, the last year in which naturally occurring cases of smallpox occurred. ACAM2000 is used to immunize military recruits. It was produced by Sanofi and is now produced by Emergent Biosolutions. Being a live vaccinia vaccine, it is contraindicated for people who are immunocompromised or pregnant, as well as for children and those with eczema, because serious and occasionally fatal reactions have occurred. Because of unexpected cardiac complications in first responders who received Dryvax, having a history of cardiac disease or significant risk factors is considered a contraindication to replication-competent (live) vaccination except in the setting of a bioterrorism event.
ACAM2000 is not FDA approved for monkeypox, but it is readily available. The United States stockpile has more than 100 million doses, according to the CDC.
“ACAM is not very different from Dryvax in terms of safety profile,” Melvin Sanicas, MD, a vaccinologist and health educator, told this news organization.
The newest option is a replication-deficient modified vaccinia Ankara vaccine called Jynneos in the United States (Imvanex in Europe; Imvamune in Canada). The vaccine is made by Denmark-based Bavarian Nordic. The FDA approved Jynneos in 2019. It, too, is available through BARDA’s stockpiles; 1,000 doses are available now and more are on order.
In the current monkeypox outbreak, Jynneos has been offered to higher-risk contacts in the United Kingdom. The CDC is planning to provide it to high-risk contacts of infected persons in the United States. This strategy is called “ring vaccination,” through which only close contacts are immunized initially. The rings are then enlarged to include more people as needed. Ring vaccination works well for easily identified diseases such as monkeypox and in situations in which there are few cases. It has been used very effectively for smallpox and Ebola.
Jynneos is not associated with the same risks as the live vaccine. In solicited reactions, injection-site reactions were common. Other reported systemic symptoms were muscle pain (42.8%), headache (34.8%), fatigue (30.4%), nausea (17.3%), and chills (10.4%).
Other vaccines are expected to be developed. Moderna has just thrown its hat into the ring, announcing it is beginning preclinical trials for monkeypox.
Prolonged close contact
Monkeypox is spread by large droplets or contact with infected lesions or body fluids. It’s thought to require prolonged close contact. In an email interview, Dr. Sanicas told this news organization that the “contact can be with (1) skin lesions of an infected person, (2) respiratory droplets in prolonged face-to-face contact, (3) fomites. The cases in the United Kingdom are in men having sex with men, but it does not mean the disease is now sexually transmitted. People do not need to have sex to be infected, but of course, sexual contact means there is prolonged contact.” The household transmission rate is less than 10%.
Dr. Sanicas confirmed that, as with smallpox, monkeypox could be transmitted by contact with clothing or bedding that has been contaminated through contact with the infected lesions, as smallpox was transmitted to Native Americans by colonizers. Airborne transmission is a theoretical possibility but is not considered likely. Being a DNA virus, monkeypox is less likely to mutate than COVID. “If it were as infectious as flu or coronavirus, there would be more infections and outbreaks in countries where MPX [monkeypox] is endemic in Western Africa or Congo Basin,” said Dr. Sanicas.
Fortunately, this clade of monkeypox, which appears to have originated in West Africa, is estimated to have a mortality rate of about 1%. In contrast, the Congo Basin clade has a death rate of up to 10%.
Dr. Sanicas concluded, “Be cautious, but there’s no need for further fear and panic on top of what we have for COVID-19. Monkeypox is not COVID and will not cause the same devastation/death/lockdowns as COVID-19.”
Dr. Hruby is an employee and stockholder of SIGA. Dr. Sanicas reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Since the first case of monkeypox on May 6, reports of outbreaks have come from multiple countries, with the United Kingdom, Spain, and Portugal in the lead, followed by Canada, Israel, and Australia, among others. The United States has reported cases in Boston and New York, and presumed cases have occurred in Utah and Florida. As of May 25, close to 350 cases, either suspected (83) or confirmed (265), have been reported globally.
Monkeypox outbreaks have previously been confined to Central and West Africa, except for an impressively large outbreak in the United States in 2003, during which 47 people were infected across six states. The epidemic was traced to a Gambian rat, rope squirrels, and dormice that had been imported from Ghana as pets and that had infected prairie dogs at a large wholesale pet store.
“It’s amazing how many of these viruses – COVID, now monkeypox and others – [exist]. They’re out there in the wild in the animal reservoir,” said Dennis Hruby, PhD, executive VP/chief scientific officer and scientific founder of SIGA Technologies.
“When it comes to the human population, they sometimes behave in ways we’re not expecting. That and a few mutations change those strains and pathogenicity and can be pandemic,” he told this news organization.
Now that the virus is pandemic, there is an urgent interest in medicines and vaccines that might halt its spread.
Smallpox drug tecovirimat
SIGA’s drug is tecovirimat, initially known as ST-246 and now branded as TPOXX. The U.S. Food and Drug Administration approved an oral formulation to treat smallpox in 2018. While smallpox was eradicated by 1980, there have been ongoing concerns about its potential use in a bioterrorism attack.
Tecovirimat is also approved for smallpox in Canada. In Europe, the approval includes treatment of monkeypox, cowpox, and complications from immunization with vaccinia. On May 19, the FDA approved an IV formulation of tecovirimat for those unable to tolerate oral medications.
In a press release, SIGA notes that tecovirimat was “developed through funding and collaboration with the Biomedical Advanced Research and Development Authority (BARDA) at the U.S. Department of Health & Human Services, as well as early-stage development supported by the National Institutes of Health, US Centers for Disease Control and Prevention, and the Department of Defense. Tecovirimat is stockpiled by the U.S. Government to mitigate the impact of a potential outbreak or bioterror attack.”
SIGA adds that, under Project Bioshield, “the United States maintains a stockpile of 1.7 million courses in the Strategic National Stockpile.” The drug is only available through the government’s stockpile.
Tecovirimat works by preventing the viruses from reproducing by interfering with a protein, VP37. The virus cannot escape the cell and so cannot infect other cells, Dr. Hruby explained.
Tecovirimat was developed under the FDA’s so-called Animal Rule, which allows approval on the basis of animal studies when human efficacy studies are unethical or impractical.
In a placebo-controlled human pharmacokinetic and safety study, only 2% of the 359 who received TPOXX had to have treatment stopped because of adverse reactions, a rate similar to placebo. The most common reactions (≥2%) were headache, nausea, and abdominal pain. Significant drug interactions were found with the coadministration of repaglinide and midazolam.
Of note is that tecovirimat’s efficacy may be reduced in immunocompromised patients. The smallpox vaccine is contraindicated for those who are immunocompromised. Those people should be offered vaccinia immune globulin.
With monkeypox, “the earlier the disease is recognized and you start treating, [the] more effective,” said Dr. Hruby. “In a monkey model which, much like humans, if we treat early on as the first lesions emerged or even several days after the lesions emerged, we see close to 100% protection.”
The other alternative drug for smallpox and (likely) monkeypox is Chimerix’s brincidofovir (BCV, Tembexa), a lipid conjugate of cidofovir, a drug for cytomegalovirus. Brincidofovir has a better safety profile than cidofovir and was also approved under the Animal Rule.
UpToDate suggests that tecovirimat is the drug of choice for monkeypox. They note that for severely infected patients, it can be combined with brincidofovir after consultation with the CDC or state health department officials.
Two vaccines available
Two vaccines are currently available. The oldest is ACAM2000, a replication-competent vaccine that replaced Dryvax, whose use was stopped in 1977, the last year in which naturally occurring cases of smallpox occurred. ACAM2000 is used to immunize military recruits. It was produced by Sanofi and is now produced by Emergent Biosolutions. Being a live vaccinia vaccine, it is contraindicated for people who are immunocompromised or pregnant, as well as for children and those with eczema, because serious and occasionally fatal reactions have occurred. Because of unexpected cardiac complications in first responders who received Dryvax, having a history of cardiac disease or significant risk factors is considered a contraindication to replication-competent (live) vaccination except in the setting of a bioterrorism event.
ACAM2000 is not FDA approved for monkeypox, but it is readily available. The United States stockpile has more than 100 million doses, according to the CDC.
“ACAM is not very different from Dryvax in terms of safety profile,” Melvin Sanicas, MD, a vaccinologist and health educator, told this news organization.
The newest option is a replication-deficient modified vaccinia Ankara vaccine called Jynneos in the United States (Imvanex in Europe; Imvamune in Canada). The vaccine is made by Denmark-based Bavarian Nordic. The FDA approved Jynneos in 2019. It, too, is available through BARDA’s stockpiles; 1,000 doses are available now and more are on order.
In the current monkeypox outbreak, Jynneos has been offered to higher-risk contacts in the United Kingdom. The CDC is planning to provide it to high-risk contacts of infected persons in the United States. This strategy is called “ring vaccination,” through which only close contacts are immunized initially. The rings are then enlarged to include more people as needed. Ring vaccination works well for easily identified diseases such as monkeypox and in situations in which there are few cases. It has been used very effectively for smallpox and Ebola.
Jynneos is not associated with the same risks as the live vaccine. In solicited reactions, injection-site reactions were common. Other reported systemic symptoms were muscle pain (42.8%), headache (34.8%), fatigue (30.4%), nausea (17.3%), and chills (10.4%).
Other vaccines are expected to be developed. Moderna has just thrown its hat into the ring, announcing it is beginning preclinical trials for monkeypox.
Prolonged close contact
Monkeypox is spread by large droplets or contact with infected lesions or body fluids. It’s thought to require prolonged close contact. In an email interview, Dr. Sanicas told this news organization that the “contact can be with (1) skin lesions of an infected person, (2) respiratory droplets in prolonged face-to-face contact, (3) fomites. The cases in the United Kingdom are in men having sex with men, but it does not mean the disease is now sexually transmitted. People do not need to have sex to be infected, but of course, sexual contact means there is prolonged contact.” The household transmission rate is less than 10%.
Dr. Sanicas confirmed that, as with smallpox, monkeypox could be transmitted by contact with clothing or bedding that has been contaminated through contact with the infected lesions, as smallpox was transmitted to Native Americans by colonizers. Airborne transmission is a theoretical possibility but is not considered likely. Being a DNA virus, monkeypox is less likely to mutate than COVID. “If it were as infectious as flu or coronavirus, there would be more infections and outbreaks in countries where MPX [monkeypox] is endemic in Western Africa or Congo Basin,” said Dr. Sanicas.
Fortunately, this clade of monkeypox, which appears to have originated in West Africa, is estimated to have a mortality rate of about 1%. In contrast, the Congo Basin clade has a death rate of up to 10%.
Dr. Sanicas concluded, “Be cautious, but there’s no need for further fear and panic on top of what we have for COVID-19. Monkeypox is not COVID and will not cause the same devastation/death/lockdowns as COVID-19.”
Dr. Hruby is an employee and stockholder of SIGA. Dr. Sanicas reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Since the first case of monkeypox on May 6, reports of outbreaks have come from multiple countries, with the United Kingdom, Spain, and Portugal in the lead, followed by Canada, Israel, and Australia, among others. The United States has reported cases in Boston and New York, and presumed cases have occurred in Utah and Florida. As of May 25, close to 350 cases, either suspected (83) or confirmed (265), have been reported globally.
Monkeypox outbreaks have previously been confined to Central and West Africa, except for an impressively large outbreak in the United States in 2003, during which 47 people were infected across six states. The epidemic was traced to a Gambian rat, rope squirrels, and dormice that had been imported from Ghana as pets and that had infected prairie dogs at a large wholesale pet store.
“It’s amazing how many of these viruses – COVID, now monkeypox and others – [exist]. They’re out there in the wild in the animal reservoir,” said Dennis Hruby, PhD, executive VP/chief scientific officer and scientific founder of SIGA Technologies.
“When it comes to the human population, they sometimes behave in ways we’re not expecting. That and a few mutations change those strains and pathogenicity and can be pandemic,” he told this news organization.
Now that the virus is pandemic, there is an urgent interest in medicines and vaccines that might halt its spread.
Smallpox drug tecovirimat
SIGA’s drug is tecovirimat, initially known as ST-246 and now branded as TPOXX. The U.S. Food and Drug Administration approved an oral formulation to treat smallpox in 2018. While smallpox was eradicated by 1980, there have been ongoing concerns about its potential use in a bioterrorism attack.
Tecovirimat is also approved for smallpox in Canada. In Europe, the approval includes treatment of monkeypox, cowpox, and complications from immunization with vaccinia. On May 19, the FDA approved an IV formulation of tecovirimat for those unable to tolerate oral medications.
In a press release, SIGA notes that tecovirimat was “developed through funding and collaboration with the Biomedical Advanced Research and Development Authority (BARDA) at the U.S. Department of Health & Human Services, as well as early-stage development supported by the National Institutes of Health, US Centers for Disease Control and Prevention, and the Department of Defense. Tecovirimat is stockpiled by the U.S. Government to mitigate the impact of a potential outbreak or bioterror attack.”
SIGA adds that, under Project Bioshield, “the United States maintains a stockpile of 1.7 million courses in the Strategic National Stockpile.” The drug is only available through the government’s stockpile.
Tecovirimat works by preventing the viruses from reproducing by interfering with a protein, VP37. The virus cannot escape the cell and so cannot infect other cells, Dr. Hruby explained.
Tecovirimat was developed under the FDA’s so-called Animal Rule, which allows approval on the basis of animal studies when human efficacy studies are unethical or impractical.
In a placebo-controlled human pharmacokinetic and safety study, only 2% of the 359 who received TPOXX had to have treatment stopped because of adverse reactions, a rate similar to placebo. The most common reactions (≥2%) were headache, nausea, and abdominal pain. Significant drug interactions were found with the coadministration of repaglinide and midazolam.
Of note is that tecovirimat’s efficacy may be reduced in immunocompromised patients. The smallpox vaccine is contraindicated for those who are immunocompromised. Those people should be offered vaccinia immune globulin.
With monkeypox, “the earlier the disease is recognized and you start treating, [the] more effective,” said Dr. Hruby. “In a monkey model which, much like humans, if we treat early on as the first lesions emerged or even several days after the lesions emerged, we see close to 100% protection.”
The other alternative drug for smallpox and (likely) monkeypox is Chimerix’s brincidofovir (BCV, Tembexa), a lipid conjugate of cidofovir, a drug for cytomegalovirus. Brincidofovir has a better safety profile than cidofovir and was also approved under the Animal Rule.
UpToDate suggests that tecovirimat is the drug of choice for monkeypox. They note that for severely infected patients, it can be combined with brincidofovir after consultation with the CDC or state health department officials.
Two vaccines available
Two vaccines are currently available. The oldest is ACAM2000, a replication-competent vaccine that replaced Dryvax, whose use was stopped in 1977, the last year in which naturally occurring cases of smallpox occurred. ACAM2000 is used to immunize military recruits. It was produced by Sanofi and is now produced by Emergent Biosolutions. Being a live vaccinia vaccine, it is contraindicated for people who are immunocompromised or pregnant, as well as for children and those with eczema, because serious and occasionally fatal reactions have occurred. Because of unexpected cardiac complications in first responders who received Dryvax, having a history of cardiac disease or significant risk factors is considered a contraindication to replication-competent (live) vaccination except in the setting of a bioterrorism event.
ACAM2000 is not FDA approved for monkeypox, but it is readily available. The United States stockpile has more than 100 million doses, according to the CDC.
“ACAM is not very different from Dryvax in terms of safety profile,” Melvin Sanicas, MD, a vaccinologist and health educator, told this news organization.
The newest option is a replication-deficient modified vaccinia Ankara vaccine called Jynneos in the United States (Imvanex in Europe; Imvamune in Canada). The vaccine is made by Denmark-based Bavarian Nordic. The FDA approved Jynneos in 2019. It, too, is available through BARDA’s stockpiles; 1,000 doses are available now and more are on order.
In the current monkeypox outbreak, Jynneos has been offered to higher-risk contacts in the United Kingdom. The CDC is planning to provide it to high-risk contacts of infected persons in the United States. This strategy is called “ring vaccination,” through which only close contacts are immunized initially. The rings are then enlarged to include more people as needed. Ring vaccination works well for easily identified diseases such as monkeypox and in situations in which there are few cases. It has been used very effectively for smallpox and Ebola.
Jynneos is not associated with the same risks as the live vaccine. In solicited reactions, injection-site reactions were common. Other reported systemic symptoms were muscle pain (42.8%), headache (34.8%), fatigue (30.4%), nausea (17.3%), and chills (10.4%).
Other vaccines are expected to be developed. Moderna has just thrown its hat into the ring, announcing it is beginning preclinical trials for monkeypox.
Prolonged close contact
Monkeypox is spread by large droplets or contact with infected lesions or body fluids. It’s thought to require prolonged close contact. In an email interview, Dr. Sanicas told this news organization that the “contact can be with (1) skin lesions of an infected person, (2) respiratory droplets in prolonged face-to-face contact, (3) fomites. The cases in the United Kingdom are in men having sex with men, but it does not mean the disease is now sexually transmitted. People do not need to have sex to be infected, but of course, sexual contact means there is prolonged contact.” The household transmission rate is less than 10%.
Dr. Sanicas confirmed that, as with smallpox, monkeypox could be transmitted by contact with clothing or bedding that has been contaminated through contact with the infected lesions, as smallpox was transmitted to Native Americans by colonizers. Airborne transmission is a theoretical possibility but is not considered likely. Being a DNA virus, monkeypox is less likely to mutate than COVID. “If it were as infectious as flu or coronavirus, there would be more infections and outbreaks in countries where MPX [monkeypox] is endemic in Western Africa or Congo Basin,” said Dr. Sanicas.
Fortunately, this clade of monkeypox, which appears to have originated in West Africa, is estimated to have a mortality rate of about 1%. In contrast, the Congo Basin clade has a death rate of up to 10%.
Dr. Sanicas concluded, “Be cautious, but there’s no need for further fear and panic on top of what we have for COVID-19. Monkeypox is not COVID and will not cause the same devastation/death/lockdowns as COVID-19.”
Dr. Hruby is an employee and stockholder of SIGA. Dr. Sanicas reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA, AMA prepare for potential COVID-19 shots for children younger than 6
Regulators and the nation’s largest physician organization took separate steps in recent days to prepare for expected authorization of use of COVID-19 vaccines in children younger than age 6.
The Food and Drug Administration on May 23 announced its Vaccines and Related Biological Products Advisory Committee will meet June 15 to discuss expanding the use of COVID vaccines from Pfizer and Moderna.
The panel will examine a request from Pfizer and its partner BioNTech for an emergency use authorization (EUA) of its vaccine to cover children ages 6 months through 4 years. The EUA expansion for the Moderna shot would cover children ages 6 months through 5 years, the FDA said.
Many parents and physicians have been urging regulators to clear COVID shots for young children, among whom rates of infection are high.
The American Medical Association in February announced an update of its Current Procedural Terminology (CPT) to prepare for an eventual FDA clearance of the Pfizer-BioNTech shot for children aged 6 months to younger than 5 years. On May 19, the association announced a new CPT update to prepare for FDA clearance for use of the Moderna COVID-19 vaccine for children 6 months through 5 years.
“Extending COVID-19 vaccination protection to approximately 18 million young children will significantly reduce their risk of COVID-19 infection, hospitalization, and death, and give their parents incredible peace of mind,” Gerald Harmon, MD, AMA’s president, said in a statement. “We strongly urge all parents to get their infants and toddlers vaccinated as soon as they are eligible for a COVID-19 vaccine.”
Both the Moderna and the Pfizer-BioNTech COVID vaccines would be given to these young children in low doses.
On May 23, Pfizer announced results from a phase 2/3 trial evaluating a series of three shots of its vaccine in children ages 6 months to younger than 5 years.
Vaccine efficacy, which was a secondary endpoint in this study, was 80.3% in this age group, Pfizer said. The analysis was based on 10 symptomatic cases of COVID-19. The trial’s protocol specifies a formal analysis will be performed when at least 21 cases have accrued from 7 days after the third dose. The company said it would share final data on the effectiveness of the vaccine once the results are available.
Moderna on April 28 issued a statement with details about testing of its vaccine in young children. Vaccine efficacy was estimated at about 51% for children aged 6 months to younger than 2 years and 37% for the children aged 2 years to younger than 6. Paul Burton, MD, Moderna’s chief medical officer, spoke about this rate during a May 1 appearance on CBS’ Face the Nation.
“What it means for parents, for caregivers, is that if they give the Moderna vaccine to these little kids, they would basically cut in half the risk of that child getting symptomatic COVID,” Dr. Burton said in the interview. “Now, the number, 50%, I know is often lower than we are used to seeing with our vaccine, but it’s because this study was conducted during a time of Omicron.”
The FDA’s vaccine advisory committee also will meet on June 14 discuss potential use under an EUA of Moderna’s COVID vaccine for children and teenagers aged 6-17 years. The Pfizer-BioNTech vaccine already is authorized under an EUA for people aged 5 years and older.
The FDA has to date granted both conditional clearances, or EUAs, and regular approvals for COVID vaccines.
EUAs are meant to be temporary, allowing for rapid introduction of medicines in response to public health crises such as the pandemic. The FDA also uses EUAs to provide initial clearances of additional indications for products, as would be the case with the authorizations Moderna and Pfizer-BioNTech are seeking for their COVID vaccines.
Companies that want to continue to sell EUA-cleared products or promote EUA-cleared indications beyond the time of the public health crisis must seek regular approvals.
The FDA cleared the Pfizer-BioNTech and Moderna COVID vaccines under EUAs in December 2020. The agency then granted a regular approval for the Pfizer-BioNTech vaccine for people ages 16 and older in August 2021 based on more robust data. Regular approval for the Moderna vaccine for people ages 18 and older followed in January 2022.
Varied reactions among parents
Attitudes in the United States about pediatric COVID vaccines are far from uniform.
The initial uptake has disappointed physicians and researchers, who have been urging wider use of the COVID vaccination among children and teens for whom the FDA already has granted a clearance. Many parents are hesitating to bring their children for the COVID vaccines, according to the Centers for Disease Control and Prevention. Only 35.4% of children ages 5-11 had received at least one dose of a COVID vaccine, CDC staff said during a meeting.
Yet many other parents are demanding this medicine for their young children, urging the FDA to move quickly to clear COVID shots.
A private Facebook group called “Protect Their Future: A Call to Action for COVID Vaccines in Kids <5” boasts about 6,200 members. Many parents and physicians have used Twitter in recent months to press for a speedy review of COVID vaccines for the youngest children, often using the hashtag #immunizeunder5s. A group called Protect Their Future, which uses @ImmunizeUnder5s as its Twitter handle, had 5,288 followers as of the afternoon of May 23.
A special panel of the House of Representatives, the Select Subcommittee on the Coronavirus Crisis, on May 23 joined those tweeting about the need to soon authorize COVID vaccines for very young children.
“Parents have been waiting many months for vaccines for their young children,” the subcommittee tweeted. “They deserve to hear from @US_FDA why this lengthy process has been in children’s best interests.”
A version of this article first appeared on Medscape.com.
Regulators and the nation’s largest physician organization took separate steps in recent days to prepare for expected authorization of use of COVID-19 vaccines in children younger than age 6.
The Food and Drug Administration on May 23 announced its Vaccines and Related Biological Products Advisory Committee will meet June 15 to discuss expanding the use of COVID vaccines from Pfizer and Moderna.
The panel will examine a request from Pfizer and its partner BioNTech for an emergency use authorization (EUA) of its vaccine to cover children ages 6 months through 4 years. The EUA expansion for the Moderna shot would cover children ages 6 months through 5 years, the FDA said.
Many parents and physicians have been urging regulators to clear COVID shots for young children, among whom rates of infection are high.
The American Medical Association in February announced an update of its Current Procedural Terminology (CPT) to prepare for an eventual FDA clearance of the Pfizer-BioNTech shot for children aged 6 months to younger than 5 years. On May 19, the association announced a new CPT update to prepare for FDA clearance for use of the Moderna COVID-19 vaccine for children 6 months through 5 years.
“Extending COVID-19 vaccination protection to approximately 18 million young children will significantly reduce their risk of COVID-19 infection, hospitalization, and death, and give their parents incredible peace of mind,” Gerald Harmon, MD, AMA’s president, said in a statement. “We strongly urge all parents to get their infants and toddlers vaccinated as soon as they are eligible for a COVID-19 vaccine.”
Both the Moderna and the Pfizer-BioNTech COVID vaccines would be given to these young children in low doses.
On May 23, Pfizer announced results from a phase 2/3 trial evaluating a series of three shots of its vaccine in children ages 6 months to younger than 5 years.
Vaccine efficacy, which was a secondary endpoint in this study, was 80.3% in this age group, Pfizer said. The analysis was based on 10 symptomatic cases of COVID-19. The trial’s protocol specifies a formal analysis will be performed when at least 21 cases have accrued from 7 days after the third dose. The company said it would share final data on the effectiveness of the vaccine once the results are available.
Moderna on April 28 issued a statement with details about testing of its vaccine in young children. Vaccine efficacy was estimated at about 51% for children aged 6 months to younger than 2 years and 37% for the children aged 2 years to younger than 6. Paul Burton, MD, Moderna’s chief medical officer, spoke about this rate during a May 1 appearance on CBS’ Face the Nation.
“What it means for parents, for caregivers, is that if they give the Moderna vaccine to these little kids, they would basically cut in half the risk of that child getting symptomatic COVID,” Dr. Burton said in the interview. “Now, the number, 50%, I know is often lower than we are used to seeing with our vaccine, but it’s because this study was conducted during a time of Omicron.”
The FDA’s vaccine advisory committee also will meet on June 14 discuss potential use under an EUA of Moderna’s COVID vaccine for children and teenagers aged 6-17 years. The Pfizer-BioNTech vaccine already is authorized under an EUA for people aged 5 years and older.
The FDA has to date granted both conditional clearances, or EUAs, and regular approvals for COVID vaccines.
EUAs are meant to be temporary, allowing for rapid introduction of medicines in response to public health crises such as the pandemic. The FDA also uses EUAs to provide initial clearances of additional indications for products, as would be the case with the authorizations Moderna and Pfizer-BioNTech are seeking for their COVID vaccines.
Companies that want to continue to sell EUA-cleared products or promote EUA-cleared indications beyond the time of the public health crisis must seek regular approvals.
The FDA cleared the Pfizer-BioNTech and Moderna COVID vaccines under EUAs in December 2020. The agency then granted a regular approval for the Pfizer-BioNTech vaccine for people ages 16 and older in August 2021 based on more robust data. Regular approval for the Moderna vaccine for people ages 18 and older followed in January 2022.
Varied reactions among parents
Attitudes in the United States about pediatric COVID vaccines are far from uniform.
The initial uptake has disappointed physicians and researchers, who have been urging wider use of the COVID vaccination among children and teens for whom the FDA already has granted a clearance. Many parents are hesitating to bring their children for the COVID vaccines, according to the Centers for Disease Control and Prevention. Only 35.4% of children ages 5-11 had received at least one dose of a COVID vaccine, CDC staff said during a meeting.
Yet many other parents are demanding this medicine for their young children, urging the FDA to move quickly to clear COVID shots.
A private Facebook group called “Protect Their Future: A Call to Action for COVID Vaccines in Kids <5” boasts about 6,200 members. Many parents and physicians have used Twitter in recent months to press for a speedy review of COVID vaccines for the youngest children, often using the hashtag #immunizeunder5s. A group called Protect Their Future, which uses @ImmunizeUnder5s as its Twitter handle, had 5,288 followers as of the afternoon of May 23.
A special panel of the House of Representatives, the Select Subcommittee on the Coronavirus Crisis, on May 23 joined those tweeting about the need to soon authorize COVID vaccines for very young children.
“Parents have been waiting many months for vaccines for their young children,” the subcommittee tweeted. “They deserve to hear from @US_FDA why this lengthy process has been in children’s best interests.”
A version of this article first appeared on Medscape.com.
Regulators and the nation’s largest physician organization took separate steps in recent days to prepare for expected authorization of use of COVID-19 vaccines in children younger than age 6.
The Food and Drug Administration on May 23 announced its Vaccines and Related Biological Products Advisory Committee will meet June 15 to discuss expanding the use of COVID vaccines from Pfizer and Moderna.
The panel will examine a request from Pfizer and its partner BioNTech for an emergency use authorization (EUA) of its vaccine to cover children ages 6 months through 4 years. The EUA expansion for the Moderna shot would cover children ages 6 months through 5 years, the FDA said.
Many parents and physicians have been urging regulators to clear COVID shots for young children, among whom rates of infection are high.
The American Medical Association in February announced an update of its Current Procedural Terminology (CPT) to prepare for an eventual FDA clearance of the Pfizer-BioNTech shot for children aged 6 months to younger than 5 years. On May 19, the association announced a new CPT update to prepare for FDA clearance for use of the Moderna COVID-19 vaccine for children 6 months through 5 years.
“Extending COVID-19 vaccination protection to approximately 18 million young children will significantly reduce their risk of COVID-19 infection, hospitalization, and death, and give their parents incredible peace of mind,” Gerald Harmon, MD, AMA’s president, said in a statement. “We strongly urge all parents to get their infants and toddlers vaccinated as soon as they are eligible for a COVID-19 vaccine.”
Both the Moderna and the Pfizer-BioNTech COVID vaccines would be given to these young children in low doses.
On May 23, Pfizer announced results from a phase 2/3 trial evaluating a series of three shots of its vaccine in children ages 6 months to younger than 5 years.
Vaccine efficacy, which was a secondary endpoint in this study, was 80.3% in this age group, Pfizer said. The analysis was based on 10 symptomatic cases of COVID-19. The trial’s protocol specifies a formal analysis will be performed when at least 21 cases have accrued from 7 days after the third dose. The company said it would share final data on the effectiveness of the vaccine once the results are available.
Moderna on April 28 issued a statement with details about testing of its vaccine in young children. Vaccine efficacy was estimated at about 51% for children aged 6 months to younger than 2 years and 37% for the children aged 2 years to younger than 6. Paul Burton, MD, Moderna’s chief medical officer, spoke about this rate during a May 1 appearance on CBS’ Face the Nation.
“What it means for parents, for caregivers, is that if they give the Moderna vaccine to these little kids, they would basically cut in half the risk of that child getting symptomatic COVID,” Dr. Burton said in the interview. “Now, the number, 50%, I know is often lower than we are used to seeing with our vaccine, but it’s because this study was conducted during a time of Omicron.”
The FDA’s vaccine advisory committee also will meet on June 14 discuss potential use under an EUA of Moderna’s COVID vaccine for children and teenagers aged 6-17 years. The Pfizer-BioNTech vaccine already is authorized under an EUA for people aged 5 years and older.
The FDA has to date granted both conditional clearances, or EUAs, and regular approvals for COVID vaccines.
EUAs are meant to be temporary, allowing for rapid introduction of medicines in response to public health crises such as the pandemic. The FDA also uses EUAs to provide initial clearances of additional indications for products, as would be the case with the authorizations Moderna and Pfizer-BioNTech are seeking for their COVID vaccines.
Companies that want to continue to sell EUA-cleared products or promote EUA-cleared indications beyond the time of the public health crisis must seek regular approvals.
The FDA cleared the Pfizer-BioNTech and Moderna COVID vaccines under EUAs in December 2020. The agency then granted a regular approval for the Pfizer-BioNTech vaccine for people ages 16 and older in August 2021 based on more robust data. Regular approval for the Moderna vaccine for people ages 18 and older followed in January 2022.
Varied reactions among parents
Attitudes in the United States about pediatric COVID vaccines are far from uniform.
The initial uptake has disappointed physicians and researchers, who have been urging wider use of the COVID vaccination among children and teens for whom the FDA already has granted a clearance. Many parents are hesitating to bring their children for the COVID vaccines, according to the Centers for Disease Control and Prevention. Only 35.4% of children ages 5-11 had received at least one dose of a COVID vaccine, CDC staff said during a meeting.
Yet many other parents are demanding this medicine for their young children, urging the FDA to move quickly to clear COVID shots.
A private Facebook group called “Protect Their Future: A Call to Action for COVID Vaccines in Kids <5” boasts about 6,200 members. Many parents and physicians have used Twitter in recent months to press for a speedy review of COVID vaccines for the youngest children, often using the hashtag #immunizeunder5s. A group called Protect Their Future, which uses @ImmunizeUnder5s as its Twitter handle, had 5,288 followers as of the afternoon of May 23.
A special panel of the House of Representatives, the Select Subcommittee on the Coronavirus Crisis, on May 23 joined those tweeting about the need to soon authorize COVID vaccines for very young children.
“Parents have been waiting many months for vaccines for their young children,” the subcommittee tweeted. “They deserve to hear from @US_FDA why this lengthy process has been in children’s best interests.”
A version of this article first appeared on Medscape.com.
Rabies: CDC updates and simplifies preexposure prophylaxis vaccination recommendations
Each year, there are about 59,000 deaths from rabies globally. Most of these occur outside the United States and are the result of dog bites. Since infection with rabies is almost always fatal, there has been considerable attention given to vaccinating people at high risk before likely exposure and responding immediately to those bitten by a rabid animal.
The Centers for Disease Control and Prevention recently revised its preexposure prophylaxis (PrEP) recommendations for rabies. Under the previous 2008 guidelines, PrEP injections were given on days 0, 7, and 21 and cost more than $1,100.
The first two groups are those with very high risk of occupational exposures – either working with rabies virus in the laboratory or working with or having contact with bats or performing animal necropsies. They are now advised to get two doses of rabies vaccine on days 0 and 7. The lab workers should have titers checked every 6 months to ensure that they remain adequately protected. And a booster should be given if the titer drops to < 0.5 IU/mL. The second group, with bat exposures, should have titers checked every 2 years.
Risk category 3 is those with long-term (> 3 years) exposure to mammals other than bats that might be rabid. This group would include veterinarians, wildlife biologists, animal control officers, and spelunkers (cavers). Category 3 also includes travelers who may encounter rabid dogs, which is not a risk in the United States. They would get the same initial two doses. The new recommendations for a third dose are based either on a titer drawn 1-3 years later being < 0.5 IU/mL or choosing to give a booster between 3 weeks and 3 years after the second dose.
The same groups are covered in risk group 4, but these are expected to have less than 3 years of potential exposure after PrEP. They would receive two doses on days 0 and 7.
Finally, group 5, at the lowest risk, includes most of the U.S. population. They do not require any PrEP.
Agam Rao, MD, CAPT, U.S. Public Health Service, CDC, told this news organization that the CDC’s Advisory Committee on Immunization Practices (ACIP) has been working on updating the 2008 rabies PrEP recommendations for several years. The committee wanted the new guideline to be “as easily followable as possible but also based on the evidence itself.”
There were two significant problems the committee tried to address. “One was that travelers who book their travel on kind of short notice don’t have enough time to get that third dose, which at the earliest can be given on day 21,” Dr. Rao said.
The second problem is that “a three-dose series [is] just really expensive. And what we found from data that had been published since the last ACIP recommendations is that fewer people than we recommend get vaccinated were getting vaccinated. So hopefully, the two-dose series helps with that.”
The ACIP used an adapted Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach to determine the certainty of the evidence for immunogenicity. The ACIP also used an evidence to recommendations (EtR) framework. “This incorporates a lot of other factors like the acceptability, usability, equity, all of these other variables that are important to the evidence being translated into recommendations,” Dr. Rao said. A table details their analysis.
Rabies expert Thiravat Hemachudha, MD, professor of neurology at WHO Collaborating Centre for Research and Training on Viral Zoonoses, Chulalongkorn University Hospital, Bangkok, told this news organization via email that “the ACIP relies mostly on serology, whereas the rest of the world cannot afford the test or testing may not be available.”
He added: “The issue of ‘long-term immunogenicity’ after receiving [PrEP is] an anamnestic response. All standard tissue culture rabies vaccines with appropriate dosage and route of delivery, either IM or ID, are considered safe and effective. There are many studies in Asian countries confirming that with only one primary series of PrEP, ID or IM with reduced doses, can produce immunity for as long as 20 years. Therefore, serology check is not necessary in general populations in rabies endemic countries where most of the rabies deaths occur. Investigation of all death cases was performed in Thailand and did not reveal any failure. Cases with PrEP in the past who died did not receive a booster after exposure.”
Dr. Rao offered one additional suggestion to clinicians faced with an urgent need to get a rabies titer: “They really should reach out to the lab (with all the information) before they send the specimen for the titer check ... so that the testing can be facilitated. All of these laboratories have the capacity to do stat and ASAP testing ... Clinicians do not know that they can call laboratories directly and expedite this sort of testing.”
Dr. Rao emphasized that PrEP does not eliminate the need for postexposure prophylaxis (PEP). Still, it eliminates the need for rabies immunoglobulin and decreases the number of vaccine doses required for PEP. “I hope more people will take advantage of the titer checks and potentially save the patient some money,” she concluded.
Dr. Rao and Dr. Hemachudha have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Each year, there are about 59,000 deaths from rabies globally. Most of these occur outside the United States and are the result of dog bites. Since infection with rabies is almost always fatal, there has been considerable attention given to vaccinating people at high risk before likely exposure and responding immediately to those bitten by a rabid animal.
The Centers for Disease Control and Prevention recently revised its preexposure prophylaxis (PrEP) recommendations for rabies. Under the previous 2008 guidelines, PrEP injections were given on days 0, 7, and 21 and cost more than $1,100.
The first two groups are those with very high risk of occupational exposures – either working with rabies virus in the laboratory or working with or having contact with bats or performing animal necropsies. They are now advised to get two doses of rabies vaccine on days 0 and 7. The lab workers should have titers checked every 6 months to ensure that they remain adequately protected. And a booster should be given if the titer drops to < 0.5 IU/mL. The second group, with bat exposures, should have titers checked every 2 years.
Risk category 3 is those with long-term (> 3 years) exposure to mammals other than bats that might be rabid. This group would include veterinarians, wildlife biologists, animal control officers, and spelunkers (cavers). Category 3 also includes travelers who may encounter rabid dogs, which is not a risk in the United States. They would get the same initial two doses. The new recommendations for a third dose are based either on a titer drawn 1-3 years later being < 0.5 IU/mL or choosing to give a booster between 3 weeks and 3 years after the second dose.
The same groups are covered in risk group 4, but these are expected to have less than 3 years of potential exposure after PrEP. They would receive two doses on days 0 and 7.
Finally, group 5, at the lowest risk, includes most of the U.S. population. They do not require any PrEP.
Agam Rao, MD, CAPT, U.S. Public Health Service, CDC, told this news organization that the CDC’s Advisory Committee on Immunization Practices (ACIP) has been working on updating the 2008 rabies PrEP recommendations for several years. The committee wanted the new guideline to be “as easily followable as possible but also based on the evidence itself.”
There were two significant problems the committee tried to address. “One was that travelers who book their travel on kind of short notice don’t have enough time to get that third dose, which at the earliest can be given on day 21,” Dr. Rao said.
The second problem is that “a three-dose series [is] just really expensive. And what we found from data that had been published since the last ACIP recommendations is that fewer people than we recommend get vaccinated were getting vaccinated. So hopefully, the two-dose series helps with that.”
The ACIP used an adapted Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach to determine the certainty of the evidence for immunogenicity. The ACIP also used an evidence to recommendations (EtR) framework. “This incorporates a lot of other factors like the acceptability, usability, equity, all of these other variables that are important to the evidence being translated into recommendations,” Dr. Rao said. A table details their analysis.
Rabies expert Thiravat Hemachudha, MD, professor of neurology at WHO Collaborating Centre for Research and Training on Viral Zoonoses, Chulalongkorn University Hospital, Bangkok, told this news organization via email that “the ACIP relies mostly on serology, whereas the rest of the world cannot afford the test or testing may not be available.”
He added: “The issue of ‘long-term immunogenicity’ after receiving [PrEP is] an anamnestic response. All standard tissue culture rabies vaccines with appropriate dosage and route of delivery, either IM or ID, are considered safe and effective. There are many studies in Asian countries confirming that with only one primary series of PrEP, ID or IM with reduced doses, can produce immunity for as long as 20 years. Therefore, serology check is not necessary in general populations in rabies endemic countries where most of the rabies deaths occur. Investigation of all death cases was performed in Thailand and did not reveal any failure. Cases with PrEP in the past who died did not receive a booster after exposure.”
Dr. Rao offered one additional suggestion to clinicians faced with an urgent need to get a rabies titer: “They really should reach out to the lab (with all the information) before they send the specimen for the titer check ... so that the testing can be facilitated. All of these laboratories have the capacity to do stat and ASAP testing ... Clinicians do not know that they can call laboratories directly and expedite this sort of testing.”
Dr. Rao emphasized that PrEP does not eliminate the need for postexposure prophylaxis (PEP). Still, it eliminates the need for rabies immunoglobulin and decreases the number of vaccine doses required for PEP. “I hope more people will take advantage of the titer checks and potentially save the patient some money,” she concluded.
Dr. Rao and Dr. Hemachudha have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Each year, there are about 59,000 deaths from rabies globally. Most of these occur outside the United States and are the result of dog bites. Since infection with rabies is almost always fatal, there has been considerable attention given to vaccinating people at high risk before likely exposure and responding immediately to those bitten by a rabid animal.
The Centers for Disease Control and Prevention recently revised its preexposure prophylaxis (PrEP) recommendations for rabies. Under the previous 2008 guidelines, PrEP injections were given on days 0, 7, and 21 and cost more than $1,100.
The first two groups are those with very high risk of occupational exposures – either working with rabies virus in the laboratory or working with or having contact with bats or performing animal necropsies. They are now advised to get two doses of rabies vaccine on days 0 and 7. The lab workers should have titers checked every 6 months to ensure that they remain adequately protected. And a booster should be given if the titer drops to < 0.5 IU/mL. The second group, with bat exposures, should have titers checked every 2 years.
Risk category 3 is those with long-term (> 3 years) exposure to mammals other than bats that might be rabid. This group would include veterinarians, wildlife biologists, animal control officers, and spelunkers (cavers). Category 3 also includes travelers who may encounter rabid dogs, which is not a risk in the United States. They would get the same initial two doses. The new recommendations for a third dose are based either on a titer drawn 1-3 years later being < 0.5 IU/mL or choosing to give a booster between 3 weeks and 3 years after the second dose.
The same groups are covered in risk group 4, but these are expected to have less than 3 years of potential exposure after PrEP. They would receive two doses on days 0 and 7.
Finally, group 5, at the lowest risk, includes most of the U.S. population. They do not require any PrEP.
Agam Rao, MD, CAPT, U.S. Public Health Service, CDC, told this news organization that the CDC’s Advisory Committee on Immunization Practices (ACIP) has been working on updating the 2008 rabies PrEP recommendations for several years. The committee wanted the new guideline to be “as easily followable as possible but also based on the evidence itself.”
There were two significant problems the committee tried to address. “One was that travelers who book their travel on kind of short notice don’t have enough time to get that third dose, which at the earliest can be given on day 21,” Dr. Rao said.
The second problem is that “a three-dose series [is] just really expensive. And what we found from data that had been published since the last ACIP recommendations is that fewer people than we recommend get vaccinated were getting vaccinated. So hopefully, the two-dose series helps with that.”
The ACIP used an adapted Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach to determine the certainty of the evidence for immunogenicity. The ACIP also used an evidence to recommendations (EtR) framework. “This incorporates a lot of other factors like the acceptability, usability, equity, all of these other variables that are important to the evidence being translated into recommendations,” Dr. Rao said. A table details their analysis.
Rabies expert Thiravat Hemachudha, MD, professor of neurology at WHO Collaborating Centre for Research and Training on Viral Zoonoses, Chulalongkorn University Hospital, Bangkok, told this news organization via email that “the ACIP relies mostly on serology, whereas the rest of the world cannot afford the test or testing may not be available.”
He added: “The issue of ‘long-term immunogenicity’ after receiving [PrEP is] an anamnestic response. All standard tissue culture rabies vaccines with appropriate dosage and route of delivery, either IM or ID, are considered safe and effective. There are many studies in Asian countries confirming that with only one primary series of PrEP, ID or IM with reduced doses, can produce immunity for as long as 20 years. Therefore, serology check is not necessary in general populations in rabies endemic countries where most of the rabies deaths occur. Investigation of all death cases was performed in Thailand and did not reveal any failure. Cases with PrEP in the past who died did not receive a booster after exposure.”
Dr. Rao offered one additional suggestion to clinicians faced with an urgent need to get a rabies titer: “They really should reach out to the lab (with all the information) before they send the specimen for the titer check ... so that the testing can be facilitated. All of these laboratories have the capacity to do stat and ASAP testing ... Clinicians do not know that they can call laboratories directly and expedite this sort of testing.”
Dr. Rao emphasized that PrEP does not eliminate the need for postexposure prophylaxis (PEP). Still, it eliminates the need for rabies immunoglobulin and decreases the number of vaccine doses required for PEP. “I hope more people will take advantage of the titer checks and potentially save the patient some money,” she concluded.
Dr. Rao and Dr. Hemachudha have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA authorizes Pfizer’s COVID booster for kids ages 5 to 11
emergency use authorization (EUA), allowing the Pfizer-BioNTech COVID-19 booster shot for children ages 5 to 11 who are at least 5 months out from their first vaccine series.
According to the most recent data from the Centers for Disease Control and Prevention, 28.6% of children in this age group have received both initial doses of Pfizer’s COVID-19 vaccine, and 35.3% have received their first dose.
Pfizer’s vaccine trial involving 4,500 children showed few side effects among children younger than 12 who received a booster, or third dose, according to a company statement.
Pfizer asked the FDA for an amended authorization in April, after submitting data showing that a third dose in children between 5 and 11 raised antibodies targeting the Omicron variant by 36 times.
“While it has largely been the case that COVID-19 tends to be less severe in children than adults, the omicron wave has seen more kids getting sick with the disease and being hospitalized, and children may also experience longer-term effects, even following initially mild disease,” FDA Commissioner Robert M. Califf, MD, said in a news release.
A study done by the New York State Department of Health showed the effectiveness of Pfizer’s two-dose vaccine series fell from 68% to 12% 4-5 months after the second dose was given to children 5 to 11 during the Omicron surge. A CDC study published in March also showed that the Pfizer shot reduced the risk of Omicron by 31% in children 5 to 11, a significantly lower rate than for kids 12 to 15, who had a 59% risk reduction after receiving two doses.
To some experts, this data suggest an even greater need for children under 12 to be eligible for a third dose.
“Since authorizing the vaccine for children down to 5 years of age in October 2021, emerging data suggest that vaccine effectiveness against COVID-19 wanes after the second dose of the vaccine in all authorized populations,” says Peter Marks, MD, PhD, the director of the FDA’s Center for Biologics Evaluation and Research.
The CDC still needs to sign off on the shots before they can be allowed. The agency’s Advisory Committee on Immunization Practices is set to meet on May 19 to discuss boosters in this age group.
FDA advisory panels plan to meet next month to discuss allowing Pfizer’s and Moderna’s COVID-19 vaccines for children under 6 years old.
A version of this article first appeared on WebMD.com.
emergency use authorization (EUA), allowing the Pfizer-BioNTech COVID-19 booster shot for children ages 5 to 11 who are at least 5 months out from their first vaccine series.
According to the most recent data from the Centers for Disease Control and Prevention, 28.6% of children in this age group have received both initial doses of Pfizer’s COVID-19 vaccine, and 35.3% have received their first dose.
Pfizer’s vaccine trial involving 4,500 children showed few side effects among children younger than 12 who received a booster, or third dose, according to a company statement.
Pfizer asked the FDA for an amended authorization in April, after submitting data showing that a third dose in children between 5 and 11 raised antibodies targeting the Omicron variant by 36 times.
“While it has largely been the case that COVID-19 tends to be less severe in children than adults, the omicron wave has seen more kids getting sick with the disease and being hospitalized, and children may also experience longer-term effects, even following initially mild disease,” FDA Commissioner Robert M. Califf, MD, said in a news release.
A study done by the New York State Department of Health showed the effectiveness of Pfizer’s two-dose vaccine series fell from 68% to 12% 4-5 months after the second dose was given to children 5 to 11 during the Omicron surge. A CDC study published in March also showed that the Pfizer shot reduced the risk of Omicron by 31% in children 5 to 11, a significantly lower rate than for kids 12 to 15, who had a 59% risk reduction after receiving two doses.
To some experts, this data suggest an even greater need for children under 12 to be eligible for a third dose.
“Since authorizing the vaccine for children down to 5 years of age in October 2021, emerging data suggest that vaccine effectiveness against COVID-19 wanes after the second dose of the vaccine in all authorized populations,” says Peter Marks, MD, PhD, the director of the FDA’s Center for Biologics Evaluation and Research.
The CDC still needs to sign off on the shots before they can be allowed. The agency’s Advisory Committee on Immunization Practices is set to meet on May 19 to discuss boosters in this age group.
FDA advisory panels plan to meet next month to discuss allowing Pfizer’s and Moderna’s COVID-19 vaccines for children under 6 years old.
A version of this article first appeared on WebMD.com.
emergency use authorization (EUA), allowing the Pfizer-BioNTech COVID-19 booster shot for children ages 5 to 11 who are at least 5 months out from their first vaccine series.
According to the most recent data from the Centers for Disease Control and Prevention, 28.6% of children in this age group have received both initial doses of Pfizer’s COVID-19 vaccine, and 35.3% have received their first dose.
Pfizer’s vaccine trial involving 4,500 children showed few side effects among children younger than 12 who received a booster, or third dose, according to a company statement.
Pfizer asked the FDA for an amended authorization in April, after submitting data showing that a third dose in children between 5 and 11 raised antibodies targeting the Omicron variant by 36 times.
“While it has largely been the case that COVID-19 tends to be less severe in children than adults, the omicron wave has seen more kids getting sick with the disease and being hospitalized, and children may also experience longer-term effects, even following initially mild disease,” FDA Commissioner Robert M. Califf, MD, said in a news release.
A study done by the New York State Department of Health showed the effectiveness of Pfizer’s two-dose vaccine series fell from 68% to 12% 4-5 months after the second dose was given to children 5 to 11 during the Omicron surge. A CDC study published in March also showed that the Pfizer shot reduced the risk of Omicron by 31% in children 5 to 11, a significantly lower rate than for kids 12 to 15, who had a 59% risk reduction after receiving two doses.
To some experts, this data suggest an even greater need for children under 12 to be eligible for a third dose.
“Since authorizing the vaccine for children down to 5 years of age in October 2021, emerging data suggest that vaccine effectiveness against COVID-19 wanes after the second dose of the vaccine in all authorized populations,” says Peter Marks, MD, PhD, the director of the FDA’s Center for Biologics Evaluation and Research.
The CDC still needs to sign off on the shots before they can be allowed. The agency’s Advisory Committee on Immunization Practices is set to meet on May 19 to discuss boosters in this age group.
FDA advisory panels plan to meet next month to discuss allowing Pfizer’s and Moderna’s COVID-19 vaccines for children under 6 years old.
A version of this article first appeared on WebMD.com.
HPV strains covered by the vaccine have declined greatly in the U.S.
Twelve years after the human papillomavirus (HPV) vaccination program was introduced in the United States, the overall prevalence of cancer-causing HPV strains covered by the vaccine dropped by 85% among females – 90% among vaccinated females and 74% among unvaccinated females – a strong sign of herd immunity, a new analysis of a nationally representative database is showing.
“HPV vaccination is working well,” Hannah Rosenblum, MD, Centers for Disease Control and Prevention, Atlanta, told this news organization in an email.
“Twelve years after introduction of HPV vaccination in the United States, national data demonstrate increasing impact among females and strong herd effects among unvaccinated females,” she added. “[Although] vaccination coverage and completion of the recommended dose in the United States is lower than coverage with other adolescent vaccinations, HPV vaccination is the best way to prevent HPV infections that can lead to several cancers in both females and males.”
The study was published online in Annals of Internal Medicine.
NHANES survey
The authors used data from the National Health and Nutrition Examination Survey (NHANES) to examine the four HPV types in the quadrivalent vaccine before 2003 and 2006 (the pre-vaccine era) and then again between 2007-2010, 2011-2014, and 2015-2018 (the vaccine era). For females, they analyzed demographic and HPV prevalence data across each 4-year era.
“Analyses were limited to sexually experienced participants, to ensure that all those included had an opportunity for HPV exposure, and to participants aged 14-24 years with adequate self-collected cervicovaginal specimens,” the authors explain.
This resulted in a sample size of 3,197 females. Demographic and HPV prevalence data were also collected from males but only during the 2013-2016 era, because those are the only years for which male HPV typing data are available in NHANES. Again, analyses were limited to sexually experienced males aged 14-24 years with adequate self-collected penile specimens, which resulted in a sample size of 661 males.
Over the 12 years of follow-up for females, there was a steady increase in females reporting having received at least one dose of the HPV vaccine – from slightly over 25% during 2007-12 to 59% during 2015-2018. The percentage of males who reported having at least one HPV dose also increased, from 29.5% in 2016 to 34.5% in 2018.
During the earliest vaccine era (2007-2010), the prevalence of the four HPV strains covered by the vaccine was 7.3% among vaccinated females, compared with 20.4% among unvaccinated females. “By 2015 to 2018, the prevalence was 2.8% (prevalence ratio, 0.16; 95% confidence interval, 0.07-0.39). The prevalence of the four vaccine-covered types was only 1.9% in vaccinated females, compared with 4.8% in unvaccinated females (PR, 0.40; 95% CI, 0.11-1.41).
In contrast, the prevalence of HPV types that were not covered by the vaccine showed little change – from 51.1% in the pre-vaccine era to 47.6% during 2015-2018 (PR, 0.93; 95% CI, 0.80-1.08). The authors considered this a good sign because it indicates that vaccine-type HPV infections are not being replaced with other oncogenic HPV infections. Between 2013 and 2016, the difference in the prevalence of the four HPV vaccine types was smaller at 1.8% among vaccinated males and 3.5% among unvaccinated males (PR, 0.49; 95% CI, 0.11-2.20).
Again, the prevalence of non-HPV vaccine types was not significantly different between vaccinated and unvaccinated males: 30.7% versus 34.3%.
During the vaccine era, effectiveness for females ranged from 60% to 84%. For males, vaccine effectiveness could only be evaluated for the single 4-year period from 2013 to 2016, and it was estimated at 51%. Dr. Rosenblum noted that vaccine efficacy estimates were lower on this national survey than the almost 100% efficacy rates observed in clinical trials in both males and females.
“This might be due in part to many participants receiving the vaccine at an older age than is recommended when they could have been infected [with HPV] at the time of vaccination,” Dr. Rosenblum said. She also noted that because males were incorporated into the HPV vaccination program years after females, they likely also experienced strong herd effects from the vaccine, making it challenging to estimate vaccine effectiveness.
Dr. Rosenblum also noted that there have already been documented declines in cervical precancers and high-grade vulvar and vaginal precancers, as well as genital warts and juvenile-onset recurrent respiratory papillomatosis. At the same time, the incidence of cervical precancers has recently declined among U.S. females in their late teens and early 20s – “likely reflecting the impact of vaccination,” she said.
“This study is good news for the United States HPV vaccination program, and all efforts are needed to ensure that children and adolescents receive routinely recommended vaccinations [including vaccination against HPV],” Dr. Rosenblum added.
Editorial comment
Commenting on the findings, Rebecca Perkins, MD, Boston University School of Medicine, and colleagues point out that the COVID-19 pandemic has led to disruptions in HPV vaccination programs and has reversed much of the progress made in recent years. “During the pandemic, providers and health systems have deprioritized adolescent vaccination and particularly HPV vaccination, which in turn has led to more severe drops for HPV vaccination than for other adolescent vaccinations, and for adolescent vaccination, compared with early childhood and adult vaccinations,” Dr. Perkins and colleagues write in an accompanying editorial.
Thus, the need to compensate for the cumulative deficit of missed vaccinations over the past 2 years has created a “serious and urgent threat” to cancer prevention efforts – “a shortfall from which it may take a decade to recover,” the editorialists predict. To try and reverse this trend, several practices have been shown to improve HPV vaccination rates.
The first is a strong provider recommendation such as, “Your child is due for an HPV vaccine today.” The second is to give standing orders to allow nurses and medical assistants to administer vaccinations without requiring intervention by a physician. Lastly, programs to remind patients when vaccines are due and to recall them for appointments also work well.
“Using evidence-based methods and redoubling our efforts to prioritize HPV vaccination will be crucial to ensuring that we do not lose a generation to preventable HPV-associated cancer,” write Dr. Perkins and colleagues.
The study authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Twelve years after the human papillomavirus (HPV) vaccination program was introduced in the United States, the overall prevalence of cancer-causing HPV strains covered by the vaccine dropped by 85% among females – 90% among vaccinated females and 74% among unvaccinated females – a strong sign of herd immunity, a new analysis of a nationally representative database is showing.
“HPV vaccination is working well,” Hannah Rosenblum, MD, Centers for Disease Control and Prevention, Atlanta, told this news organization in an email.
“Twelve years after introduction of HPV vaccination in the United States, national data demonstrate increasing impact among females and strong herd effects among unvaccinated females,” she added. “[Although] vaccination coverage and completion of the recommended dose in the United States is lower than coverage with other adolescent vaccinations, HPV vaccination is the best way to prevent HPV infections that can lead to several cancers in both females and males.”
The study was published online in Annals of Internal Medicine.
NHANES survey
The authors used data from the National Health and Nutrition Examination Survey (NHANES) to examine the four HPV types in the quadrivalent vaccine before 2003 and 2006 (the pre-vaccine era) and then again between 2007-2010, 2011-2014, and 2015-2018 (the vaccine era). For females, they analyzed demographic and HPV prevalence data across each 4-year era.
“Analyses were limited to sexually experienced participants, to ensure that all those included had an opportunity for HPV exposure, and to participants aged 14-24 years with adequate self-collected cervicovaginal specimens,” the authors explain.
This resulted in a sample size of 3,197 females. Demographic and HPV prevalence data were also collected from males but only during the 2013-2016 era, because those are the only years for which male HPV typing data are available in NHANES. Again, analyses were limited to sexually experienced males aged 14-24 years with adequate self-collected penile specimens, which resulted in a sample size of 661 males.
Over the 12 years of follow-up for females, there was a steady increase in females reporting having received at least one dose of the HPV vaccine – from slightly over 25% during 2007-12 to 59% during 2015-2018. The percentage of males who reported having at least one HPV dose also increased, from 29.5% in 2016 to 34.5% in 2018.
During the earliest vaccine era (2007-2010), the prevalence of the four HPV strains covered by the vaccine was 7.3% among vaccinated females, compared with 20.4% among unvaccinated females. “By 2015 to 2018, the prevalence was 2.8% (prevalence ratio, 0.16; 95% confidence interval, 0.07-0.39). The prevalence of the four vaccine-covered types was only 1.9% in vaccinated females, compared with 4.8% in unvaccinated females (PR, 0.40; 95% CI, 0.11-1.41).
In contrast, the prevalence of HPV types that were not covered by the vaccine showed little change – from 51.1% in the pre-vaccine era to 47.6% during 2015-2018 (PR, 0.93; 95% CI, 0.80-1.08). The authors considered this a good sign because it indicates that vaccine-type HPV infections are not being replaced with other oncogenic HPV infections. Between 2013 and 2016, the difference in the prevalence of the four HPV vaccine types was smaller at 1.8% among vaccinated males and 3.5% among unvaccinated males (PR, 0.49; 95% CI, 0.11-2.20).
Again, the prevalence of non-HPV vaccine types was not significantly different between vaccinated and unvaccinated males: 30.7% versus 34.3%.
During the vaccine era, effectiveness for females ranged from 60% to 84%. For males, vaccine effectiveness could only be evaluated for the single 4-year period from 2013 to 2016, and it was estimated at 51%. Dr. Rosenblum noted that vaccine efficacy estimates were lower on this national survey than the almost 100% efficacy rates observed in clinical trials in both males and females.
“This might be due in part to many participants receiving the vaccine at an older age than is recommended when they could have been infected [with HPV] at the time of vaccination,” Dr. Rosenblum said. She also noted that because males were incorporated into the HPV vaccination program years after females, they likely also experienced strong herd effects from the vaccine, making it challenging to estimate vaccine effectiveness.
Dr. Rosenblum also noted that there have already been documented declines in cervical precancers and high-grade vulvar and vaginal precancers, as well as genital warts and juvenile-onset recurrent respiratory papillomatosis. At the same time, the incidence of cervical precancers has recently declined among U.S. females in their late teens and early 20s – “likely reflecting the impact of vaccination,” she said.
“This study is good news for the United States HPV vaccination program, and all efforts are needed to ensure that children and adolescents receive routinely recommended vaccinations [including vaccination against HPV],” Dr. Rosenblum added.
Editorial comment
Commenting on the findings, Rebecca Perkins, MD, Boston University School of Medicine, and colleagues point out that the COVID-19 pandemic has led to disruptions in HPV vaccination programs and has reversed much of the progress made in recent years. “During the pandemic, providers and health systems have deprioritized adolescent vaccination and particularly HPV vaccination, which in turn has led to more severe drops for HPV vaccination than for other adolescent vaccinations, and for adolescent vaccination, compared with early childhood and adult vaccinations,” Dr. Perkins and colleagues write in an accompanying editorial.
Thus, the need to compensate for the cumulative deficit of missed vaccinations over the past 2 years has created a “serious and urgent threat” to cancer prevention efforts – “a shortfall from which it may take a decade to recover,” the editorialists predict. To try and reverse this trend, several practices have been shown to improve HPV vaccination rates.
The first is a strong provider recommendation such as, “Your child is due for an HPV vaccine today.” The second is to give standing orders to allow nurses and medical assistants to administer vaccinations without requiring intervention by a physician. Lastly, programs to remind patients when vaccines are due and to recall them for appointments also work well.
“Using evidence-based methods and redoubling our efforts to prioritize HPV vaccination will be crucial to ensuring that we do not lose a generation to preventable HPV-associated cancer,” write Dr. Perkins and colleagues.
The study authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Twelve years after the human papillomavirus (HPV) vaccination program was introduced in the United States, the overall prevalence of cancer-causing HPV strains covered by the vaccine dropped by 85% among females – 90% among vaccinated females and 74% among unvaccinated females – a strong sign of herd immunity, a new analysis of a nationally representative database is showing.
“HPV vaccination is working well,” Hannah Rosenblum, MD, Centers for Disease Control and Prevention, Atlanta, told this news organization in an email.
“Twelve years after introduction of HPV vaccination in the United States, national data demonstrate increasing impact among females and strong herd effects among unvaccinated females,” she added. “[Although] vaccination coverage and completion of the recommended dose in the United States is lower than coverage with other adolescent vaccinations, HPV vaccination is the best way to prevent HPV infections that can lead to several cancers in both females and males.”
The study was published online in Annals of Internal Medicine.
NHANES survey
The authors used data from the National Health and Nutrition Examination Survey (NHANES) to examine the four HPV types in the quadrivalent vaccine before 2003 and 2006 (the pre-vaccine era) and then again between 2007-2010, 2011-2014, and 2015-2018 (the vaccine era). For females, they analyzed demographic and HPV prevalence data across each 4-year era.
“Analyses were limited to sexually experienced participants, to ensure that all those included had an opportunity for HPV exposure, and to participants aged 14-24 years with adequate self-collected cervicovaginal specimens,” the authors explain.
This resulted in a sample size of 3,197 females. Demographic and HPV prevalence data were also collected from males but only during the 2013-2016 era, because those are the only years for which male HPV typing data are available in NHANES. Again, analyses were limited to sexually experienced males aged 14-24 years with adequate self-collected penile specimens, which resulted in a sample size of 661 males.
Over the 12 years of follow-up for females, there was a steady increase in females reporting having received at least one dose of the HPV vaccine – from slightly over 25% during 2007-12 to 59% during 2015-2018. The percentage of males who reported having at least one HPV dose also increased, from 29.5% in 2016 to 34.5% in 2018.
During the earliest vaccine era (2007-2010), the prevalence of the four HPV strains covered by the vaccine was 7.3% among vaccinated females, compared with 20.4% among unvaccinated females. “By 2015 to 2018, the prevalence was 2.8% (prevalence ratio, 0.16; 95% confidence interval, 0.07-0.39). The prevalence of the four vaccine-covered types was only 1.9% in vaccinated females, compared with 4.8% in unvaccinated females (PR, 0.40; 95% CI, 0.11-1.41).
In contrast, the prevalence of HPV types that were not covered by the vaccine showed little change – from 51.1% in the pre-vaccine era to 47.6% during 2015-2018 (PR, 0.93; 95% CI, 0.80-1.08). The authors considered this a good sign because it indicates that vaccine-type HPV infections are not being replaced with other oncogenic HPV infections. Between 2013 and 2016, the difference in the prevalence of the four HPV vaccine types was smaller at 1.8% among vaccinated males and 3.5% among unvaccinated males (PR, 0.49; 95% CI, 0.11-2.20).
Again, the prevalence of non-HPV vaccine types was not significantly different between vaccinated and unvaccinated males: 30.7% versus 34.3%.
During the vaccine era, effectiveness for females ranged from 60% to 84%. For males, vaccine effectiveness could only be evaluated for the single 4-year period from 2013 to 2016, and it was estimated at 51%. Dr. Rosenblum noted that vaccine efficacy estimates were lower on this national survey than the almost 100% efficacy rates observed in clinical trials in both males and females.
“This might be due in part to many participants receiving the vaccine at an older age than is recommended when they could have been infected [with HPV] at the time of vaccination,” Dr. Rosenblum said. She also noted that because males were incorporated into the HPV vaccination program years after females, they likely also experienced strong herd effects from the vaccine, making it challenging to estimate vaccine effectiveness.
Dr. Rosenblum also noted that there have already been documented declines in cervical precancers and high-grade vulvar and vaginal precancers, as well as genital warts and juvenile-onset recurrent respiratory papillomatosis. At the same time, the incidence of cervical precancers has recently declined among U.S. females in their late teens and early 20s – “likely reflecting the impact of vaccination,” she said.
“This study is good news for the United States HPV vaccination program, and all efforts are needed to ensure that children and adolescents receive routinely recommended vaccinations [including vaccination against HPV],” Dr. Rosenblum added.
Editorial comment
Commenting on the findings, Rebecca Perkins, MD, Boston University School of Medicine, and colleagues point out that the COVID-19 pandemic has led to disruptions in HPV vaccination programs and has reversed much of the progress made in recent years. “During the pandemic, providers and health systems have deprioritized adolescent vaccination and particularly HPV vaccination, which in turn has led to more severe drops for HPV vaccination than for other adolescent vaccinations, and for adolescent vaccination, compared with early childhood and adult vaccinations,” Dr. Perkins and colleagues write in an accompanying editorial.
Thus, the need to compensate for the cumulative deficit of missed vaccinations over the past 2 years has created a “serious and urgent threat” to cancer prevention efforts – “a shortfall from which it may take a decade to recover,” the editorialists predict. To try and reverse this trend, several practices have been shown to improve HPV vaccination rates.
The first is a strong provider recommendation such as, “Your child is due for an HPV vaccine today.” The second is to give standing orders to allow nurses and medical assistants to administer vaccinations without requiring intervention by a physician. Lastly, programs to remind patients when vaccines are due and to recall them for appointments also work well.
“Using evidence-based methods and redoubling our efforts to prioritize HPV vaccination will be crucial to ensuring that we do not lose a generation to preventable HPV-associated cancer,” write Dr. Perkins and colleagues.
The study authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE
Pfizer COVID vaccine performs well in youth with rheumatic diseases
The Pfizer-BioNTech mRNA vaccine (Comirnaty) showed a good safety profile with minimal short-term side effects and no negative impact on disease activity in a cohort of adolescents and young adults with rheumatic diseases, according to research presented at the annual scientific meeting of the Childhood Arthritis and Rheumatology Research Alliance, held virtually this year.
Only 3% of patients experience a severe transient adverse event, according to Merav Heshin-Bekenstein, MD, of Dana-Dwek Children’s Hospital at the Tel Aviv Sourasky Medical Center in Israel. The findings were published in Rheumatology.
“We found that the mRNA Pfizer vaccine was immunogenic and induced an adequate humoral immune response in adolescent patients,” Dr. Heshin-Bekenstein told CARRA attendees. “It was definitely comparable to healthy controls and practically all patients were seropositive following the second vaccine, except for one patient with long-standing systemic sclerosis.”
The findings were not necessarily surprising but were encouraging to Melissa S. Oliver, MD, assistant professor of clinical pediatrics in the division of pediatric rheumatology at Indiana University, Indianapolis. Dr. Oliver wasn’t part of the study team.
“We know that the COVID vaccines in healthy adolescents have shown good efficacy with minimal side effects, and it’s good to see that this study showed that in those with rheumatic diseases on immunosuppressive therapy,” Dr. Oliver told this news organization.
Until now, the data on COVID-19 vaccines in teens with rheumatic illnesses has been limited, she said, so “many pediatric rheumatologists only have the data from adult studies to go on or personal experience with their own cohort of patients.”
But the high immunogenicity seen in the study was a pleasant surprise to Beth H. Rutstein, MD, assistant professor of clinical pediatrics in the division of rheumatology at Children’s Hospital of Philadelphia and the Perelman School of Medicine at the University of Pennsylvania.
“I was both surprised and thrilled with Dr. Heshin-Bekenstein’s findings suggesting near-universal seroconversion for patients with rheumatic disease regardless of underlying diagnosis or immunomodulatory therapy regimen, as much of the adult data has suggested a poorer seroconversion rate” and lower antibody titers in adults with similar illnesses, Dr. Rutstein said in an interview.
The study “provides essential reassurance that vaccination against COVID-19 does not increase the risk of disease flare or worsen disease severity scores,” said Dr. Rutstein, who was not associated with the research. “Rather than speaking purely anecdotally with our patients and their families, we can refer to the science – which is always more reassuring for both our patients and ourselves.”
Study included diverse conditions and therapies
Risk factors for poor outcomes with COVID-19 in children include obesity, cardiovascular disease, chronic lung disease, diabetes, and asthma, Dr. Heshin-Bekenstein told CARRA attendees. Multisystem inflammatory syndrome in children (MIS-C) and long COVID are also potential complications of COVID-19 with less understood risk factors.
Although COVID-19 is most often mild in children, certain severe, systemic rheumatic diseases increase hospitalization risk, including systemic lupus erythematosus (SLE) and vasculitis. Evidence has also shown that COVID-19 infection increases the risk of disease flare in teens with juvenile-onset rheumatic diseases, so it’s “crucial to prevent COVID-19 disease in this population,” Dr. Heshin-Bekenstein said.
Her study therefore aimed to assess the safety and immunogenicity of the Pfizer mRNA vaccine for teens with juvenile-onset rheumatic diseases and those taking immunomodulatory medications. The international prospective multicenter study ran from April to November 2021 at three pediatric rheumatology clinics in Israel and one in Slovenia. Endpoints included short-term side effects, vaccination impact on clinical disease activity, immunogenicity at 2-9 weeks after the second dose, and, secondarily, efficacy against COVID-19 infection.
The 91 participants included adolescents aged 12-18 and young adults aged 18-21. Nearly half of the participants (46%) had juvenile idiopathic arthritis (JIA), and 14% had SLE. Other participants’ conditions included systemic vasculitis, idiopathic uveitis, inflammatory bowel disease–related arthritis, systemic or localized scleroderma, juvenile dermatomyositis, or an autoinflammatory disease. Participants’ mean disease duration was 4.8 years.
The researchers compared the patients with a control group of 40 individuals with similar demographics but without rheumatic disease. The researchers used the LIAISON quantitative assay to assess serum IgG antibody levels against the SARS-CoV-2 spike protein in both groups.
Eight in 10 participants with rheumatic disease were taking an immunomodulatory medication, including a conventional synthetic disease-modifying antirheumatic drug (csDMARD) in 40%, a biologic DMARD in 37%, tumor necrosis factor (TNF) inhibitors in 32%, hydroxychloroquine (HCQ) in 19%, glucocorticoids in 14%, and mycophenolate in 11%. A smaller proportion were on other biologics: JAK inhibitors in 6.6%, anti-CD20 drugs in 4.4%, and an IL-6 inhibitor in 1%.
Side effects similar in both groups
None of the side effects reported by participants were statistically different between those with rheumatic disease and the control group. Localized pain was the most common side effect, reported by 73%-79% of participants after each dose. About twice as many participants with rheumatic disease experienced muscle aches and joint pains, compared with the control group, but the differences were not significant. Fever occurred more often in those with rheumatic disease (6%, five cases) than without (3%, one case). One-third of those with rheumatic disease felt tiredness, compared with 20% of the control group.
None of the healthy controls were hospitalized after vaccination, but three rheumatic patients were, including two after the first dose. Both were 17 years old, had systemic vasculitis with granulomatosis with polyangiitis (GPA), and were taking rituximab (Rituxan). One patient experienced acute onset of chronic renal failure, fever, dehydration, and high C-reactive protein within hours of vaccination. The other experienced new onset of pulmonary hemorrhage a week after vaccination.
In addition, a 14-year-old female with lupus, taking only HCQ, went to the emergency department with fever, headache, vomiting, and joint pain 1 day after the second vaccine dose. She had normal inflammatory markers and no change in disease activity score, and she was discharged with low-dose steroids tapered after 2 weeks.
Immune response high in patients with rheumatic disease
Immunogenicity was similar in both groups, with 97% seropositivity in the rheumatic disease group and 100% in the control group. Average IgG titers were 242 in the rheumatic group and 388 in the control group (P < .0001). Seropositivity was 88% in those taking mycophenolate with another drug (100% with mycophenolate monotherapy), 90% with HCQ, 94% with any csDMARDs and another drug (100% with csDMARD monotherapy), and 100% for all other drugs. During 3 months’ follow-up after vaccination, there were no COVID-19 cases among the participants.
Dr. Heshin-Bekenstein noted that their results showed better immunogenicity in teens, compared with adults, for two specific drugs. Seropositivity in teens taking methotrexate (Rheumatrex, Trexall) or rituximab was 100% in this study, compared with 84% in adults taking methotrexate and 39% in adults taking rituximab in a previous study. However, only three patients in this study were taking rituximab, and only seven were taking methotrexate.
The study’s heterogenous population was both a strength and a weakness of the study. “Due to the diversity of rheumatic diseases and medications included in this cohort, it was not possible to draw significant conclusions regarding the impact of the immunomodulatory medications and type of disease” on titers, Dr. Heshin-Bekenstein told attendees.
Still, “I think as pediatric rheumatologists, we can feel reassured in recommending the COVID-19 vaccine to our patients,” Dr. Oliver said. “I will add that every patient is different, and everyone should have a conversation with their physician about receiving the COVID-19 vaccine.” Dr. Oliver said she discusses vaccination, including COVID vaccination, with every patient, and it’s been challenging to address concerns in the midst of so much misinformation circulating about the vaccine.
These findings do raise questions about whether it’s still necessary to hold immunomodulatory medications to get the vaccine,” Dr. Rutstein said.
“Many families are nervous to pause their medications before and after the vaccine as is currently recommended for many therapies by the American College of Rheumatology, and I do share that concern for some of my patients with more clinically unstable disease, so I try to work with each family to decide on best timing and have delayed or deferred the series until some patients are on a steady dose of a new immunomodulatory medication if it has been recently started,” Dr. Rutstein said. “This is one of the reasons why Dr. Heshin-Bekenstein’s study is so important – we may be holding medications that can be safely continued and even further decrease the risk of disease flare.”
None of the physicians have disclosed any relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Pfizer-BioNTech mRNA vaccine (Comirnaty) showed a good safety profile with minimal short-term side effects and no negative impact on disease activity in a cohort of adolescents and young adults with rheumatic diseases, according to research presented at the annual scientific meeting of the Childhood Arthritis and Rheumatology Research Alliance, held virtually this year.
Only 3% of patients experience a severe transient adverse event, according to Merav Heshin-Bekenstein, MD, of Dana-Dwek Children’s Hospital at the Tel Aviv Sourasky Medical Center in Israel. The findings were published in Rheumatology.
“We found that the mRNA Pfizer vaccine was immunogenic and induced an adequate humoral immune response in adolescent patients,” Dr. Heshin-Bekenstein told CARRA attendees. “It was definitely comparable to healthy controls and practically all patients were seropositive following the second vaccine, except for one patient with long-standing systemic sclerosis.”
The findings were not necessarily surprising but were encouraging to Melissa S. Oliver, MD, assistant professor of clinical pediatrics in the division of pediatric rheumatology at Indiana University, Indianapolis. Dr. Oliver wasn’t part of the study team.
“We know that the COVID vaccines in healthy adolescents have shown good efficacy with minimal side effects, and it’s good to see that this study showed that in those with rheumatic diseases on immunosuppressive therapy,” Dr. Oliver told this news organization.
Until now, the data on COVID-19 vaccines in teens with rheumatic illnesses has been limited, she said, so “many pediatric rheumatologists only have the data from adult studies to go on or personal experience with their own cohort of patients.”
But the high immunogenicity seen in the study was a pleasant surprise to Beth H. Rutstein, MD, assistant professor of clinical pediatrics in the division of rheumatology at Children’s Hospital of Philadelphia and the Perelman School of Medicine at the University of Pennsylvania.
“I was both surprised and thrilled with Dr. Heshin-Bekenstein’s findings suggesting near-universal seroconversion for patients with rheumatic disease regardless of underlying diagnosis or immunomodulatory therapy regimen, as much of the adult data has suggested a poorer seroconversion rate” and lower antibody titers in adults with similar illnesses, Dr. Rutstein said in an interview.
The study “provides essential reassurance that vaccination against COVID-19 does not increase the risk of disease flare or worsen disease severity scores,” said Dr. Rutstein, who was not associated with the research. “Rather than speaking purely anecdotally with our patients and their families, we can refer to the science – which is always more reassuring for both our patients and ourselves.”
Study included diverse conditions and therapies
Risk factors for poor outcomes with COVID-19 in children include obesity, cardiovascular disease, chronic lung disease, diabetes, and asthma, Dr. Heshin-Bekenstein told CARRA attendees. Multisystem inflammatory syndrome in children (MIS-C) and long COVID are also potential complications of COVID-19 with less understood risk factors.
Although COVID-19 is most often mild in children, certain severe, systemic rheumatic diseases increase hospitalization risk, including systemic lupus erythematosus (SLE) and vasculitis. Evidence has also shown that COVID-19 infection increases the risk of disease flare in teens with juvenile-onset rheumatic diseases, so it’s “crucial to prevent COVID-19 disease in this population,” Dr. Heshin-Bekenstein said.
Her study therefore aimed to assess the safety and immunogenicity of the Pfizer mRNA vaccine for teens with juvenile-onset rheumatic diseases and those taking immunomodulatory medications. The international prospective multicenter study ran from April to November 2021 at three pediatric rheumatology clinics in Israel and one in Slovenia. Endpoints included short-term side effects, vaccination impact on clinical disease activity, immunogenicity at 2-9 weeks after the second dose, and, secondarily, efficacy against COVID-19 infection.
The 91 participants included adolescents aged 12-18 and young adults aged 18-21. Nearly half of the participants (46%) had juvenile idiopathic arthritis (JIA), and 14% had SLE. Other participants’ conditions included systemic vasculitis, idiopathic uveitis, inflammatory bowel disease–related arthritis, systemic or localized scleroderma, juvenile dermatomyositis, or an autoinflammatory disease. Participants’ mean disease duration was 4.8 years.
The researchers compared the patients with a control group of 40 individuals with similar demographics but without rheumatic disease. The researchers used the LIAISON quantitative assay to assess serum IgG antibody levels against the SARS-CoV-2 spike protein in both groups.
Eight in 10 participants with rheumatic disease were taking an immunomodulatory medication, including a conventional synthetic disease-modifying antirheumatic drug (csDMARD) in 40%, a biologic DMARD in 37%, tumor necrosis factor (TNF) inhibitors in 32%, hydroxychloroquine (HCQ) in 19%, glucocorticoids in 14%, and mycophenolate in 11%. A smaller proportion were on other biologics: JAK inhibitors in 6.6%, anti-CD20 drugs in 4.4%, and an IL-6 inhibitor in 1%.
Side effects similar in both groups
None of the side effects reported by participants were statistically different between those with rheumatic disease and the control group. Localized pain was the most common side effect, reported by 73%-79% of participants after each dose. About twice as many participants with rheumatic disease experienced muscle aches and joint pains, compared with the control group, but the differences were not significant. Fever occurred more often in those with rheumatic disease (6%, five cases) than without (3%, one case). One-third of those with rheumatic disease felt tiredness, compared with 20% of the control group.
None of the healthy controls were hospitalized after vaccination, but three rheumatic patients were, including two after the first dose. Both were 17 years old, had systemic vasculitis with granulomatosis with polyangiitis (GPA), and were taking rituximab (Rituxan). One patient experienced acute onset of chronic renal failure, fever, dehydration, and high C-reactive protein within hours of vaccination. The other experienced new onset of pulmonary hemorrhage a week after vaccination.
In addition, a 14-year-old female with lupus, taking only HCQ, went to the emergency department with fever, headache, vomiting, and joint pain 1 day after the second vaccine dose. She had normal inflammatory markers and no change in disease activity score, and she was discharged with low-dose steroids tapered after 2 weeks.
Immune response high in patients with rheumatic disease
Immunogenicity was similar in both groups, with 97% seropositivity in the rheumatic disease group and 100% in the control group. Average IgG titers were 242 in the rheumatic group and 388 in the control group (P < .0001). Seropositivity was 88% in those taking mycophenolate with another drug (100% with mycophenolate monotherapy), 90% with HCQ, 94% with any csDMARDs and another drug (100% with csDMARD monotherapy), and 100% for all other drugs. During 3 months’ follow-up after vaccination, there were no COVID-19 cases among the participants.
Dr. Heshin-Bekenstein noted that their results showed better immunogenicity in teens, compared with adults, for two specific drugs. Seropositivity in teens taking methotrexate (Rheumatrex, Trexall) or rituximab was 100% in this study, compared with 84% in adults taking methotrexate and 39% in adults taking rituximab in a previous study. However, only three patients in this study were taking rituximab, and only seven were taking methotrexate.
The study’s heterogenous population was both a strength and a weakness of the study. “Due to the diversity of rheumatic diseases and medications included in this cohort, it was not possible to draw significant conclusions regarding the impact of the immunomodulatory medications and type of disease” on titers, Dr. Heshin-Bekenstein told attendees.
Still, “I think as pediatric rheumatologists, we can feel reassured in recommending the COVID-19 vaccine to our patients,” Dr. Oliver said. “I will add that every patient is different, and everyone should have a conversation with their physician about receiving the COVID-19 vaccine.” Dr. Oliver said she discusses vaccination, including COVID vaccination, with every patient, and it’s been challenging to address concerns in the midst of so much misinformation circulating about the vaccine.
These findings do raise questions about whether it’s still necessary to hold immunomodulatory medications to get the vaccine,” Dr. Rutstein said.
“Many families are nervous to pause their medications before and after the vaccine as is currently recommended for many therapies by the American College of Rheumatology, and I do share that concern for some of my patients with more clinically unstable disease, so I try to work with each family to decide on best timing and have delayed or deferred the series until some patients are on a steady dose of a new immunomodulatory medication if it has been recently started,” Dr. Rutstein said. “This is one of the reasons why Dr. Heshin-Bekenstein’s study is so important – we may be holding medications that can be safely continued and even further decrease the risk of disease flare.”
None of the physicians have disclosed any relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Pfizer-BioNTech mRNA vaccine (Comirnaty) showed a good safety profile with minimal short-term side effects and no negative impact on disease activity in a cohort of adolescents and young adults with rheumatic diseases, according to research presented at the annual scientific meeting of the Childhood Arthritis and Rheumatology Research Alliance, held virtually this year.
Only 3% of patients experience a severe transient adverse event, according to Merav Heshin-Bekenstein, MD, of Dana-Dwek Children’s Hospital at the Tel Aviv Sourasky Medical Center in Israel. The findings were published in Rheumatology.
“We found that the mRNA Pfizer vaccine was immunogenic and induced an adequate humoral immune response in adolescent patients,” Dr. Heshin-Bekenstein told CARRA attendees. “It was definitely comparable to healthy controls and practically all patients were seropositive following the second vaccine, except for one patient with long-standing systemic sclerosis.”
The findings were not necessarily surprising but were encouraging to Melissa S. Oliver, MD, assistant professor of clinical pediatrics in the division of pediatric rheumatology at Indiana University, Indianapolis. Dr. Oliver wasn’t part of the study team.
“We know that the COVID vaccines in healthy adolescents have shown good efficacy with minimal side effects, and it’s good to see that this study showed that in those with rheumatic diseases on immunosuppressive therapy,” Dr. Oliver told this news organization.
Until now, the data on COVID-19 vaccines in teens with rheumatic illnesses has been limited, she said, so “many pediatric rheumatologists only have the data from adult studies to go on or personal experience with their own cohort of patients.”
But the high immunogenicity seen in the study was a pleasant surprise to Beth H. Rutstein, MD, assistant professor of clinical pediatrics in the division of rheumatology at Children’s Hospital of Philadelphia and the Perelman School of Medicine at the University of Pennsylvania.
“I was both surprised and thrilled with Dr. Heshin-Bekenstein’s findings suggesting near-universal seroconversion for patients with rheumatic disease regardless of underlying diagnosis or immunomodulatory therapy regimen, as much of the adult data has suggested a poorer seroconversion rate” and lower antibody titers in adults with similar illnesses, Dr. Rutstein said in an interview.
The study “provides essential reassurance that vaccination against COVID-19 does not increase the risk of disease flare or worsen disease severity scores,” said Dr. Rutstein, who was not associated with the research. “Rather than speaking purely anecdotally with our patients and their families, we can refer to the science – which is always more reassuring for both our patients and ourselves.”
Study included diverse conditions and therapies
Risk factors for poor outcomes with COVID-19 in children include obesity, cardiovascular disease, chronic lung disease, diabetes, and asthma, Dr. Heshin-Bekenstein told CARRA attendees. Multisystem inflammatory syndrome in children (MIS-C) and long COVID are also potential complications of COVID-19 with less understood risk factors.
Although COVID-19 is most often mild in children, certain severe, systemic rheumatic diseases increase hospitalization risk, including systemic lupus erythematosus (SLE) and vasculitis. Evidence has also shown that COVID-19 infection increases the risk of disease flare in teens with juvenile-onset rheumatic diseases, so it’s “crucial to prevent COVID-19 disease in this population,” Dr. Heshin-Bekenstein said.
Her study therefore aimed to assess the safety and immunogenicity of the Pfizer mRNA vaccine for teens with juvenile-onset rheumatic diseases and those taking immunomodulatory medications. The international prospective multicenter study ran from April to November 2021 at three pediatric rheumatology clinics in Israel and one in Slovenia. Endpoints included short-term side effects, vaccination impact on clinical disease activity, immunogenicity at 2-9 weeks after the second dose, and, secondarily, efficacy against COVID-19 infection.
The 91 participants included adolescents aged 12-18 and young adults aged 18-21. Nearly half of the participants (46%) had juvenile idiopathic arthritis (JIA), and 14% had SLE. Other participants’ conditions included systemic vasculitis, idiopathic uveitis, inflammatory bowel disease–related arthritis, systemic or localized scleroderma, juvenile dermatomyositis, or an autoinflammatory disease. Participants’ mean disease duration was 4.8 years.
The researchers compared the patients with a control group of 40 individuals with similar demographics but without rheumatic disease. The researchers used the LIAISON quantitative assay to assess serum IgG antibody levels against the SARS-CoV-2 spike protein in both groups.
Eight in 10 participants with rheumatic disease were taking an immunomodulatory medication, including a conventional synthetic disease-modifying antirheumatic drug (csDMARD) in 40%, a biologic DMARD in 37%, tumor necrosis factor (TNF) inhibitors in 32%, hydroxychloroquine (HCQ) in 19%, glucocorticoids in 14%, and mycophenolate in 11%. A smaller proportion were on other biologics: JAK inhibitors in 6.6%, anti-CD20 drugs in 4.4%, and an IL-6 inhibitor in 1%.
Side effects similar in both groups
None of the side effects reported by participants were statistically different between those with rheumatic disease and the control group. Localized pain was the most common side effect, reported by 73%-79% of participants after each dose. About twice as many participants with rheumatic disease experienced muscle aches and joint pains, compared with the control group, but the differences were not significant. Fever occurred more often in those with rheumatic disease (6%, five cases) than without (3%, one case). One-third of those with rheumatic disease felt tiredness, compared with 20% of the control group.
None of the healthy controls were hospitalized after vaccination, but three rheumatic patients were, including two after the first dose. Both were 17 years old, had systemic vasculitis with granulomatosis with polyangiitis (GPA), and were taking rituximab (Rituxan). One patient experienced acute onset of chronic renal failure, fever, dehydration, and high C-reactive protein within hours of vaccination. The other experienced new onset of pulmonary hemorrhage a week after vaccination.
In addition, a 14-year-old female with lupus, taking only HCQ, went to the emergency department with fever, headache, vomiting, and joint pain 1 day after the second vaccine dose. She had normal inflammatory markers and no change in disease activity score, and she was discharged with low-dose steroids tapered after 2 weeks.
Immune response high in patients with rheumatic disease
Immunogenicity was similar in both groups, with 97% seropositivity in the rheumatic disease group and 100% in the control group. Average IgG titers were 242 in the rheumatic group and 388 in the control group (P < .0001). Seropositivity was 88% in those taking mycophenolate with another drug (100% with mycophenolate monotherapy), 90% with HCQ, 94% with any csDMARDs and another drug (100% with csDMARD monotherapy), and 100% for all other drugs. During 3 months’ follow-up after vaccination, there were no COVID-19 cases among the participants.
Dr. Heshin-Bekenstein noted that their results showed better immunogenicity in teens, compared with adults, for two specific drugs. Seropositivity in teens taking methotrexate (Rheumatrex, Trexall) or rituximab was 100% in this study, compared with 84% in adults taking methotrexate and 39% in adults taking rituximab in a previous study. However, only three patients in this study were taking rituximab, and only seven were taking methotrexate.
The study’s heterogenous population was both a strength and a weakness of the study. “Due to the diversity of rheumatic diseases and medications included in this cohort, it was not possible to draw significant conclusions regarding the impact of the immunomodulatory medications and type of disease” on titers, Dr. Heshin-Bekenstein told attendees.
Still, “I think as pediatric rheumatologists, we can feel reassured in recommending the COVID-19 vaccine to our patients,” Dr. Oliver said. “I will add that every patient is different, and everyone should have a conversation with their physician about receiving the COVID-19 vaccine.” Dr. Oliver said she discusses vaccination, including COVID vaccination, with every patient, and it’s been challenging to address concerns in the midst of so much misinformation circulating about the vaccine.
These findings do raise questions about whether it’s still necessary to hold immunomodulatory medications to get the vaccine,” Dr. Rutstein said.
“Many families are nervous to pause their medications before and after the vaccine as is currently recommended for many therapies by the American College of Rheumatology, and I do share that concern for some of my patients with more clinically unstable disease, so I try to work with each family to decide on best timing and have delayed or deferred the series until some patients are on a steady dose of a new immunomodulatory medication if it has been recently started,” Dr. Rutstein said. “This is one of the reasons why Dr. Heshin-Bekenstein’s study is so important – we may be holding medications that can be safely continued and even further decrease the risk of disease flare.”
None of the physicians have disclosed any relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CARRA 2022
Measles outbreaks: Protecting your patients during international travel
The U.S. immunization program is one of the best public health success stories. Physicians who provide care for children are familiar with the routine childhood immunization schedule and administer a measles-containing vaccine at age-appropriate times. Thanks to its rigorous implementation and acceptance, endemic measles (absence of continuous virus transmission for > 1 year) was eliminated in the U.S. in 2000. Loss of this status was in jeopardy in 2019 when 22 measles outbreaks occurred in 17 states (7 were multistate outbreaks). That year, 1,163 cases were reported.1 Most cases occurred in unvaccinated persons (89%) and 81 cases were imported of which 54 were in U.S. citizens returning from international travel. All outbreaks were linked to travel. Fortunately, the outbreaks were controlled prior to the elimination deadline, or the United States would have lost its measles elimination status. Restrictions on travel because of COVID-19 have relaxed significantly since the introduction of COVID-19 vaccines, resulting in increased regional and international travel. Multiple countries, including the United States noted a decline in routine immunizations rates during the last 2 years. Recent U.S. data for the 2020-2021 school year indicates that MMR immunizations rates (two doses) for kindergarteners declined to 93.9% (range 78.9% to > 98.9%), while the overall percentage of those students with an exemption remained low at 2.2%. Vaccine coverage greater than 95% was reported in only 16 states. Coverage of less than 90% was reported in seven states and the District of Columbia (Georgia, Idaho, Kentucky, Maryland, Minnesota, Ohio, and Wisconsin).2 Vaccine coverage should be 95% or higher to maintain herd immunity and control outbreaks.
Why is measles prevention so important? Many physicians practicing in the United States today have never seen a case or know its potential complications. I saw my first case as a resident in an immigrant child. It took our training director to point out the subtle signs and symptoms. It was the first time I saw Kolpik spots. Measles is transmitted person to person via large respiratory droplets and less often by airborne spread. It is highly contagious for susceptible individuals with an attack rate of 90%. In this case, a medical student on the team developed symptoms about 10 days later. Six years would pass before I diagnosed my next case of measles. An HIV patient acquired it after close contact with someone who was in the prodromal stage. He presented with the 3 C’s: Cough, coryza, and conjunctivitis, in addition to fever and an erythematous rash. He did not recover from complications of the disease.
Prior to the routine administration of a measles vaccine, 3-4 million cases with almost 500 deaths occurred annually in the United States. Worldwide, 35 million cases and more than 6 million deaths occurred each year. Here, most patients recover completely; however, complications including otitis media, pneumonia, croup, and encephalitis can develop. Complications commonly occur in immunocompromised individuals and young children. Groups with the highest fatality rates include children aged less than 5 years, immunocompromised persons, and pregnant women. Worldwide, fatality rates are dependent on the patients underlying nutritional and health status in addition to the quality of health care available.3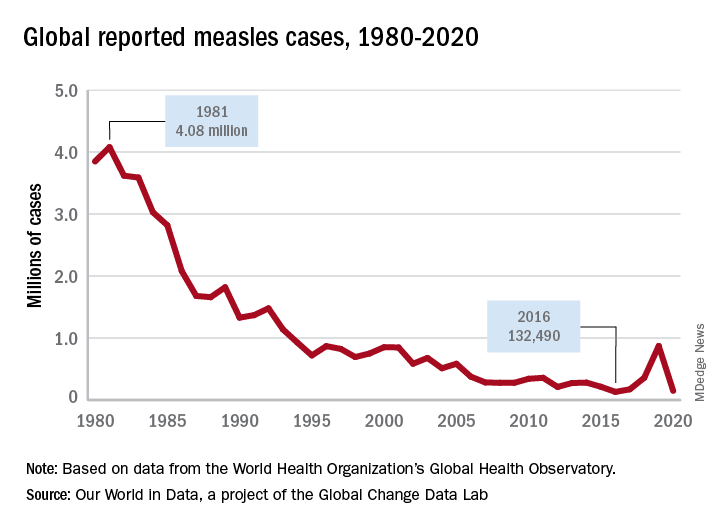
Measles vaccine was licensed in 1963 and cases began to decline (Figure 1). There was a resurgence in 1989 but it was not limited to the United States. The cause of the U.S. resurgence was multifactorial: Widespread viral transmission among unvaccinated preschool-age children residing in inner cities, outbreaks in vaccinated school-age children, outbreaks in students and personnel on college campuses, and primary vaccine failure (2%-5% of recipients failed to have an adequate response). In 1989, to help prevent future outbreaks, the United States recommended a two-dose schedule for measles and in 1993, the Vaccines for Children Program, a federally funded program, was established to improve access to vaccines for all children.
What is going on internationally?
Figure 2 lists the top 10 countries with current measles outbreaks.

Most countries on the list may not be typical travel destinations for tourists; however, they are common destinations for individuals visiting friends and relatives after immigrating to the United States. In contrast to the United States, most countries with limited resources and infrastructure have mass-vaccination campaigns to ensure vaccine administration to large segments of the population. They too have been affected by the COVID-19 pandemic. By report, at least 41 countries delayed implementation of their measles campaign in 2020 and 2021, thus, leading to the potential for even larger outbreaks.4
Progress toward the global elimination of measles is evidenced by the following: All 194 countries now include one dose of measles in their routine schedules; between 2000 and 2019 coverage of one dose of measles increased from 72% to 85% and countries with more than 90% coverage increased from 45% to 63%. Finally, the number of countries offering two doses of measles increased from 50% to 91% and vaccine coverage increased from 18% to 71% over the same time period.3
What can you do for your patients and their parents before they travel abroad?
- Inform all staff that the MMR vaccine can be administered to children as young as 6 months and at times other than those listed on the routine immunization schedule. This will help avoid parents seeking vaccine being denied an appointment.
- Children 6-11 months need 1 dose of MMR. Two additional doses will still need to be administered at the routine time.
- Children 12 months or older need 2 doses of MMR at least 4 weeks apart.
- If yellow fever vaccine is needed, coordinate administration with a travel medicine clinic since both are live vaccines and must be given on the same day.
- Any person born after 1956 should have 2 doses of MMR at least 4 weeks apart if they have no evidence of immunity.
- Encourage parents to always inform you and your staff of any international travel plans.
Moving forward, remember this increased global activity and the presence of inadequately vaccinated individuals/communities keeps the United States at continued risk for measles outbreaks. The source of the next outbreak may only be one plane ride away.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
This article was updated 6/29/22.
References
1. Patel M et al. MMWR. 2019 Oct 11; 68(40):893-6.
2. Seither R et al. MMWR. 2022 Apr 22;71(16):561-8.
3. Gastañaduy PA et al. J Infect Dis. 2021 Sep 30;224(12 Suppl 2):S420-8. doi: 10.1093/infdis/jiaa793.
4. Centers for Disease Control and Prevention. Measles (Rubeola). http://www.CDC.gov/Measles.
The U.S. immunization program is one of the best public health success stories. Physicians who provide care for children are familiar with the routine childhood immunization schedule and administer a measles-containing vaccine at age-appropriate times. Thanks to its rigorous implementation and acceptance, endemic measles (absence of continuous virus transmission for > 1 year) was eliminated in the U.S. in 2000. Loss of this status was in jeopardy in 2019 when 22 measles outbreaks occurred in 17 states (7 were multistate outbreaks). That year, 1,163 cases were reported.1 Most cases occurred in unvaccinated persons (89%) and 81 cases were imported of which 54 were in U.S. citizens returning from international travel. All outbreaks were linked to travel. Fortunately, the outbreaks were controlled prior to the elimination deadline, or the United States would have lost its measles elimination status. Restrictions on travel because of COVID-19 have relaxed significantly since the introduction of COVID-19 vaccines, resulting in increased regional and international travel. Multiple countries, including the United States noted a decline in routine immunizations rates during the last 2 years. Recent U.S. data for the 2020-2021 school year indicates that MMR immunizations rates (two doses) for kindergarteners declined to 93.9% (range 78.9% to > 98.9%), while the overall percentage of those students with an exemption remained low at 2.2%. Vaccine coverage greater than 95% was reported in only 16 states. Coverage of less than 90% was reported in seven states and the District of Columbia (Georgia, Idaho, Kentucky, Maryland, Minnesota, Ohio, and Wisconsin).2 Vaccine coverage should be 95% or higher to maintain herd immunity and control outbreaks.
Why is measles prevention so important? Many physicians practicing in the United States today have never seen a case or know its potential complications. I saw my first case as a resident in an immigrant child. It took our training director to point out the subtle signs and symptoms. It was the first time I saw Kolpik spots. Measles is transmitted person to person via large respiratory droplets and less often by airborne spread. It is highly contagious for susceptible individuals with an attack rate of 90%. In this case, a medical student on the team developed symptoms about 10 days later. Six years would pass before I diagnosed my next case of measles. An HIV patient acquired it after close contact with someone who was in the prodromal stage. He presented with the 3 C’s: Cough, coryza, and conjunctivitis, in addition to fever and an erythematous rash. He did not recover from complications of the disease.
Prior to the routine administration of a measles vaccine, 3-4 million cases with almost 500 deaths occurred annually in the United States. Worldwide, 35 million cases and more than 6 million deaths occurred each year. Here, most patients recover completely; however, complications including otitis media, pneumonia, croup, and encephalitis can develop. Complications commonly occur in immunocompromised individuals and young children. Groups with the highest fatality rates include children aged less than 5 years, immunocompromised persons, and pregnant women. Worldwide, fatality rates are dependent on the patients underlying nutritional and health status in addition to the quality of health care available.3
Measles vaccine was licensed in 1963 and cases began to decline (Figure 1). There was a resurgence in 1989 but it was not limited to the United States. The cause of the U.S. resurgence was multifactorial: Widespread viral transmission among unvaccinated preschool-age children residing in inner cities, outbreaks in vaccinated school-age children, outbreaks in students and personnel on college campuses, and primary vaccine failure (2%-5% of recipients failed to have an adequate response). In 1989, to help prevent future outbreaks, the United States recommended a two-dose schedule for measles and in 1993, the Vaccines for Children Program, a federally funded program, was established to improve access to vaccines for all children.
What is going on internationally?
Figure 2 lists the top 10 countries with current measles outbreaks.

Most countries on the list may not be typical travel destinations for tourists; however, they are common destinations for individuals visiting friends and relatives after immigrating to the United States. In contrast to the United States, most countries with limited resources and infrastructure have mass-vaccination campaigns to ensure vaccine administration to large segments of the population. They too have been affected by the COVID-19 pandemic. By report, at least 41 countries delayed implementation of their measles campaign in 2020 and 2021, thus, leading to the potential for even larger outbreaks.4
Progress toward the global elimination of measles is evidenced by the following: All 194 countries now include one dose of measles in their routine schedules; between 2000 and 2019 coverage of one dose of measles increased from 72% to 85% and countries with more than 90% coverage increased from 45% to 63%. Finally, the number of countries offering two doses of measles increased from 50% to 91% and vaccine coverage increased from 18% to 71% over the same time period.3
What can you do for your patients and their parents before they travel abroad?
- Inform all staff that the MMR vaccine can be administered to children as young as 6 months and at times other than those listed on the routine immunization schedule. This will help avoid parents seeking vaccine being denied an appointment.
- Children 6-11 months need 1 dose of MMR. Two additional doses will still need to be administered at the routine time.
- Children 12 months or older need 2 doses of MMR at least 4 weeks apart.
- If yellow fever vaccine is needed, coordinate administration with a travel medicine clinic since both are live vaccines and must be given on the same day.
- Any person born after 1956 should have 2 doses of MMR at least 4 weeks apart if they have no evidence of immunity.
- Encourage parents to always inform you and your staff of any international travel plans.
Moving forward, remember this increased global activity and the presence of inadequately vaccinated individuals/communities keeps the United States at continued risk for measles outbreaks. The source of the next outbreak may only be one plane ride away.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
This article was updated 6/29/22.
References
1. Patel M et al. MMWR. 2019 Oct 11; 68(40):893-6.
2. Seither R et al. MMWR. 2022 Apr 22;71(16):561-8.
3. Gastañaduy PA et al. J Infect Dis. 2021 Sep 30;224(12 Suppl 2):S420-8. doi: 10.1093/infdis/jiaa793.
4. Centers for Disease Control and Prevention. Measles (Rubeola). http://www.CDC.gov/Measles.
The U.S. immunization program is one of the best public health success stories. Physicians who provide care for children are familiar with the routine childhood immunization schedule and administer a measles-containing vaccine at age-appropriate times. Thanks to its rigorous implementation and acceptance, endemic measles (absence of continuous virus transmission for > 1 year) was eliminated in the U.S. in 2000. Loss of this status was in jeopardy in 2019 when 22 measles outbreaks occurred in 17 states (7 were multistate outbreaks). That year, 1,163 cases were reported.1 Most cases occurred in unvaccinated persons (89%) and 81 cases were imported of which 54 were in U.S. citizens returning from international travel. All outbreaks were linked to travel. Fortunately, the outbreaks were controlled prior to the elimination deadline, or the United States would have lost its measles elimination status. Restrictions on travel because of COVID-19 have relaxed significantly since the introduction of COVID-19 vaccines, resulting in increased regional and international travel. Multiple countries, including the United States noted a decline in routine immunizations rates during the last 2 years. Recent U.S. data for the 2020-2021 school year indicates that MMR immunizations rates (two doses) for kindergarteners declined to 93.9% (range 78.9% to > 98.9%), while the overall percentage of those students with an exemption remained low at 2.2%. Vaccine coverage greater than 95% was reported in only 16 states. Coverage of less than 90% was reported in seven states and the District of Columbia (Georgia, Idaho, Kentucky, Maryland, Minnesota, Ohio, and Wisconsin).2 Vaccine coverage should be 95% or higher to maintain herd immunity and control outbreaks.
Why is measles prevention so important? Many physicians practicing in the United States today have never seen a case or know its potential complications. I saw my first case as a resident in an immigrant child. It took our training director to point out the subtle signs and symptoms. It was the first time I saw Kolpik spots. Measles is transmitted person to person via large respiratory droplets and less often by airborne spread. It is highly contagious for susceptible individuals with an attack rate of 90%. In this case, a medical student on the team developed symptoms about 10 days later. Six years would pass before I diagnosed my next case of measles. An HIV patient acquired it after close contact with someone who was in the prodromal stage. He presented with the 3 C’s: Cough, coryza, and conjunctivitis, in addition to fever and an erythematous rash. He did not recover from complications of the disease.
Prior to the routine administration of a measles vaccine, 3-4 million cases with almost 500 deaths occurred annually in the United States. Worldwide, 35 million cases and more than 6 million deaths occurred each year. Here, most patients recover completely; however, complications including otitis media, pneumonia, croup, and encephalitis can develop. Complications commonly occur in immunocompromised individuals and young children. Groups with the highest fatality rates include children aged less than 5 years, immunocompromised persons, and pregnant women. Worldwide, fatality rates are dependent on the patients underlying nutritional and health status in addition to the quality of health care available.3
Measles vaccine was licensed in 1963 and cases began to decline (Figure 1). There was a resurgence in 1989 but it was not limited to the United States. The cause of the U.S. resurgence was multifactorial: Widespread viral transmission among unvaccinated preschool-age children residing in inner cities, outbreaks in vaccinated school-age children, outbreaks in students and personnel on college campuses, and primary vaccine failure (2%-5% of recipients failed to have an adequate response). In 1989, to help prevent future outbreaks, the United States recommended a two-dose schedule for measles and in 1993, the Vaccines for Children Program, a federally funded program, was established to improve access to vaccines for all children.
What is going on internationally?
Figure 2 lists the top 10 countries with current measles outbreaks.

Most countries on the list may not be typical travel destinations for tourists; however, they are common destinations for individuals visiting friends and relatives after immigrating to the United States. In contrast to the United States, most countries with limited resources and infrastructure have mass-vaccination campaigns to ensure vaccine administration to large segments of the population. They too have been affected by the COVID-19 pandemic. By report, at least 41 countries delayed implementation of their measles campaign in 2020 and 2021, thus, leading to the potential for even larger outbreaks.4
Progress toward the global elimination of measles is evidenced by the following: All 194 countries now include one dose of measles in their routine schedules; between 2000 and 2019 coverage of one dose of measles increased from 72% to 85% and countries with more than 90% coverage increased from 45% to 63%. Finally, the number of countries offering two doses of measles increased from 50% to 91% and vaccine coverage increased from 18% to 71% over the same time period.3
What can you do for your patients and their parents before they travel abroad?
- Inform all staff that the MMR vaccine can be administered to children as young as 6 months and at times other than those listed on the routine immunization schedule. This will help avoid parents seeking vaccine being denied an appointment.
- Children 6-11 months need 1 dose of MMR. Two additional doses will still need to be administered at the routine time.
- Children 12 months or older need 2 doses of MMR at least 4 weeks apart.
- If yellow fever vaccine is needed, coordinate administration with a travel medicine clinic since both are live vaccines and must be given on the same day.
- Any person born after 1956 should have 2 doses of MMR at least 4 weeks apart if they have no evidence of immunity.
- Encourage parents to always inform you and your staff of any international travel plans.
Moving forward, remember this increased global activity and the presence of inadequately vaccinated individuals/communities keeps the United States at continued risk for measles outbreaks. The source of the next outbreak may only be one plane ride away.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
This article was updated 6/29/22.
References
1. Patel M et al. MMWR. 2019 Oct 11; 68(40):893-6.
2. Seither R et al. MMWR. 2022 Apr 22;71(16):561-8.
3. Gastañaduy PA et al. J Infect Dis. 2021 Sep 30;224(12 Suppl 2):S420-8. doi: 10.1093/infdis/jiaa793.
4. Centers for Disease Control and Prevention. Measles (Rubeola). http://www.CDC.gov/Measles.
COVID fallout: ‘Alarming’ dip in routine vax for pregnant women
The percentage of low-income pregnant mothers who received influenza and Tdap vaccinations fell sharply during the COVID-19 pandemic, especially in Black and Hispanic patients, a new study finds.
The percentage of patients who received the influenza vaccines at two Medicaid clinics in Houston dropped from 78% before the pandemic to 61% during it (adjusted odds ratio, 0.38; 95% CI, 0.26-0.53; P < .01), researchers reported at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. The percentage receiving the Tdap vaccine dipped from 85% to 76% (aOR, 0.56; 95% CI, 0.40-0.79; P < .01).
New York–Presbyterian/Weill Cornell Medical Center pediatrician Sallie Permar, MD, PhD, who’s familiar with the study findings, called them “alarming” and said in an interview that they should be “a call to action for providers.”
“Continuing the status quo in our routine preventative health care and clinic operations means that we are losing ground in reduction and elimination of vaccine-preventable diseases,” Dr. Permar said in an interview.
According to corresponding author Bani Ratan, MD, an ob.gyn. with the Baylor College of Medicine, Houston, there’s been little if any previous research into routine, non-COVID vaccination in pregnant women during the pandemic.
For the study, researchers retrospectively analyzed the records of 939 pregnant women who entered prenatal care before 20 weeks (462 from May–November 2019, and 477 from May–November 2020) and delivered at full term.
Among ethnic groups, non-Hispanic Blacks saw the largest decline in influenza vaccines. Among them, the percentage who got them fell from 64% (73/114) to 35% (35/101; aOR, 0.30; 95% CI, 0.17-0.52; P < .01). Only Hispanics had a statistically significant decline in Tdap vaccination (OR, 0.52, 95% CI, 0.34-0.80; P < .01, percentages not provided).
Another study presented at ACOG examined vaccination rates during the pandemic and found that Tdap vaccination rates dipped among pregnant women in a Philadelphia-area health care system.
Possible causes for the decline in routine vaccination include hesitancy linked to the COVID-19 vaccines and fewer office visits because of telemedicine, said Dr. Batan in an interview.
Dr. Permar blamed the role of vaccine misinformation during the pandemic and the mistrust caused by the exclusion of pregnant women from early vaccine trials. She added that “challenges in health care staffing and issues of health care provider burnout that worsened during the pandemic likely contributed to a fraying of the focus on preventive health maintenance simply due to bandwidth of health professionals.”
In a separate study presented at ACOG, researchers at the State University of New York, Syracuse, reported on a survey of 157 pregnant women of whom just 38.2% were vaccinated against COVID-19. Among the unvaccinated, who were more likely to have less education, 66% reported that lack of data about vaccination was their primary concern.
No funding or disclosures are reported by study authors. Dr. Permar reported consulting for Merck, Moderna, GlaxoSmithKline, Pfizer, Dynavax, and Hookipa on cytomegalovirus vaccine programs.
*This story was updated on 5/11/2022.
The percentage of low-income pregnant mothers who received influenza and Tdap vaccinations fell sharply during the COVID-19 pandemic, especially in Black and Hispanic patients, a new study finds.
The percentage of patients who received the influenza vaccines at two Medicaid clinics in Houston dropped from 78% before the pandemic to 61% during it (adjusted odds ratio, 0.38; 95% CI, 0.26-0.53; P < .01), researchers reported at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. The percentage receiving the Tdap vaccine dipped from 85% to 76% (aOR, 0.56; 95% CI, 0.40-0.79; P < .01).
New York–Presbyterian/Weill Cornell Medical Center pediatrician Sallie Permar, MD, PhD, who’s familiar with the study findings, called them “alarming” and said in an interview that they should be “a call to action for providers.”
“Continuing the status quo in our routine preventative health care and clinic operations means that we are losing ground in reduction and elimination of vaccine-preventable diseases,” Dr. Permar said in an interview.
According to corresponding author Bani Ratan, MD, an ob.gyn. with the Baylor College of Medicine, Houston, there’s been little if any previous research into routine, non-COVID vaccination in pregnant women during the pandemic.
For the study, researchers retrospectively analyzed the records of 939 pregnant women who entered prenatal care before 20 weeks (462 from May–November 2019, and 477 from May–November 2020) and delivered at full term.
Among ethnic groups, non-Hispanic Blacks saw the largest decline in influenza vaccines. Among them, the percentage who got them fell from 64% (73/114) to 35% (35/101; aOR, 0.30; 95% CI, 0.17-0.52; P < .01). Only Hispanics had a statistically significant decline in Tdap vaccination (OR, 0.52, 95% CI, 0.34-0.80; P < .01, percentages not provided).
Another study presented at ACOG examined vaccination rates during the pandemic and found that Tdap vaccination rates dipped among pregnant women in a Philadelphia-area health care system.
Possible causes for the decline in routine vaccination include hesitancy linked to the COVID-19 vaccines and fewer office visits because of telemedicine, said Dr. Batan in an interview.
Dr. Permar blamed the role of vaccine misinformation during the pandemic and the mistrust caused by the exclusion of pregnant women from early vaccine trials. She added that “challenges in health care staffing and issues of health care provider burnout that worsened during the pandemic likely contributed to a fraying of the focus on preventive health maintenance simply due to bandwidth of health professionals.”
In a separate study presented at ACOG, researchers at the State University of New York, Syracuse, reported on a survey of 157 pregnant women of whom just 38.2% were vaccinated against COVID-19. Among the unvaccinated, who were more likely to have less education, 66% reported that lack of data about vaccination was their primary concern.
No funding or disclosures are reported by study authors. Dr. Permar reported consulting for Merck, Moderna, GlaxoSmithKline, Pfizer, Dynavax, and Hookipa on cytomegalovirus vaccine programs.
*This story was updated on 5/11/2022.
The percentage of low-income pregnant mothers who received influenza and Tdap vaccinations fell sharply during the COVID-19 pandemic, especially in Black and Hispanic patients, a new study finds.
The percentage of patients who received the influenza vaccines at two Medicaid clinics in Houston dropped from 78% before the pandemic to 61% during it (adjusted odds ratio, 0.38; 95% CI, 0.26-0.53; P < .01), researchers reported at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. The percentage receiving the Tdap vaccine dipped from 85% to 76% (aOR, 0.56; 95% CI, 0.40-0.79; P < .01).
New York–Presbyterian/Weill Cornell Medical Center pediatrician Sallie Permar, MD, PhD, who’s familiar with the study findings, called them “alarming” and said in an interview that they should be “a call to action for providers.”
“Continuing the status quo in our routine preventative health care and clinic operations means that we are losing ground in reduction and elimination of vaccine-preventable diseases,” Dr. Permar said in an interview.
According to corresponding author Bani Ratan, MD, an ob.gyn. with the Baylor College of Medicine, Houston, there’s been little if any previous research into routine, non-COVID vaccination in pregnant women during the pandemic.
For the study, researchers retrospectively analyzed the records of 939 pregnant women who entered prenatal care before 20 weeks (462 from May–November 2019, and 477 from May–November 2020) and delivered at full term.
Among ethnic groups, non-Hispanic Blacks saw the largest decline in influenza vaccines. Among them, the percentage who got them fell from 64% (73/114) to 35% (35/101; aOR, 0.30; 95% CI, 0.17-0.52; P < .01). Only Hispanics had a statistically significant decline in Tdap vaccination (OR, 0.52, 95% CI, 0.34-0.80; P < .01, percentages not provided).
Another study presented at ACOG examined vaccination rates during the pandemic and found that Tdap vaccination rates dipped among pregnant women in a Philadelphia-area health care system.
Possible causes for the decline in routine vaccination include hesitancy linked to the COVID-19 vaccines and fewer office visits because of telemedicine, said Dr. Batan in an interview.
Dr. Permar blamed the role of vaccine misinformation during the pandemic and the mistrust caused by the exclusion of pregnant women from early vaccine trials. She added that “challenges in health care staffing and issues of health care provider burnout that worsened during the pandemic likely contributed to a fraying of the focus on preventive health maintenance simply due to bandwidth of health professionals.”
In a separate study presented at ACOG, researchers at the State University of New York, Syracuse, reported on a survey of 157 pregnant women of whom just 38.2% were vaccinated against COVID-19. Among the unvaccinated, who were more likely to have less education, 66% reported that lack of data about vaccination was their primary concern.
No funding or disclosures are reported by study authors. Dr. Permar reported consulting for Merck, Moderna, GlaxoSmithKline, Pfizer, Dynavax, and Hookipa on cytomegalovirus vaccine programs.
*This story was updated on 5/11/2022.
FROM ACOG 2022
Flu vaccine linked to lower risk for stroke: INTERSTROKE
in a large new case-control study.
“While influenza vaccination is a cost-effective method to prevent influenza, it is also an effective way to reduce the burden of stroke,” said study author Christopher Schwarzbach, MD, of Ludwigshafen (Germany) Hospital.
“Our results therefore encourage the wider use of influenza vaccination,” he concluded.
Dr. Schwarzbach presented these data from the INTERSTROKE study at the 2022 European Stroke Organisation Conference.
He explained that acute inflammatory disease is thought to increase the risk for cerebrovascular events, and the seasonality of influenza-like illness appears to be associated with the seasonality of cardiovascular and cerebrovascular events. Previous observational studies have also shown a link between influenza vaccination and a reduced risk for stroke.
The current INTERSTROKE study was a large international case-control study conducted between 2007 and 2015 that involved 13,447 cases (patients within 5 days of their first stroke) and a similar number of age- and gender-matched people from 32 countries across the world.
All cases and control subjects were systematically asked whether they had acute febrile illness in the previous 4 weeks and whether they had received an influenza vaccination within the previous year.
Conditional logistical regression was used to quantify the results, with adjustment for 13 different possible confounding factors, including hypertension, activity, smoking, cardiovascular risk factors, and socioeconomic factors.
Results showed that having had an acute febrile illness in the previous 4 weeks was more commonly reported in the patients with an acute ischemic stroke (8.7%) than in control patients (5.6%). After adjustment for confounding factors, this gives an adjusted risk ratio of 1.18, which was of borderline statistical significance (95% confidence limits, 1.01-1.39), Dr. Schwarzbach reported.
The association between recent febrile illness and acute ischemic stroke was stronger when compared with community control subjects (adjusted odds ratio, 2.0), but it was absent when compared with hospital control subjects.
The association was also only apparent in Australia, China, North America, and Western Europe; it was not seen in other parts of the world.
There was no association between acute febrile illness and acute cerebral hemorrhage.
Flu vaccine linked to halving of stroke risk
Having received a flu vaccine in the previous year was strongly associated with a lower risk for any type of stroke (aOR, 0.53), ischemic stroke (aOR, 0.57), and hemorrhagic stroke (aOR, 0.34).
Dr. Schwarzbach noted that these results were also consistent in an extended statistical model that included variables that might reflect a willingness to be vaccinated and when compared with both community and hospital-based control subjects.
The strength of the association between influenza vaccination and reduced risk for stroke was similar when compared with either community or hospital control subjects, and was only moderately stronger during than outside the influenza season.
The association was also seen in all regions of the world apart from Africa and South Asia, Dr. Schwarzbach reported, but he noted that vaccination rates in these two regions were extremely low.
The researchers also found that the magnitude of the associations between flu vaccination and lower risk for stroke were stronger in individuals who had multiple annual vaccinations, with an odds ratio of 0.54 in those who had received a vaccine every year for the previous 5 years, and of 0.79 in those who had received one to four vaccinations in the previous 5 years.
Mechanism: Immune stimulation?
Discussing possible mechanisms behind these results, Dr. Schwarzbach noted that the finding that the association with influenza vaccination and reduced stroke risk was independent of seasonality was surprising. “We had expected the protective effect of vaccination to be bigger during the influenza season, but this wasn’t the case.”
He suggested that one explanation might be the inclusion of regions of the world where this seasonality doesn’t exist.
But he pointed out that the finding of a stronger association between flu vaccination and lower stroke risk in those who had received more vaccinations has given rise to another theory: that it is the stimulation of the immune system rather than the protection of infection against influenza that is the key factor.
In an interview with Dr. Schwarzbach, Guillaume Turk, MD, professor of neurology at GHU Paris, pointed out that causal inferences are always difficult in case-control studies and in clinical epidemiology in general.
“What makes you think that this association between influenza vaccination and decreased risk is causal rather than due to unmeasured confounders? For example, patients who received vaccination may have received more medical attention and may have been more aware of the risk factors for stroke,” he asked.
Dr. Schwarzbach replied: “Yes, this is the issue of healthy user bias, which is always a problem in this type of research and is hard to address.”
“What we tried to do here is to adjust for variables that might influence the willingness of people to get vaccinated,” he added. “These were mainly socioeconomic factors. But, of course, this is something that we can’t rule out.”
Dr. Schwarzbach noted that, for more reliable information on this association, prospective studies are needed.
‘A plausible effect’
Discussing the study after the presentation, William Whiteley, BM, PhD, a clinical epidemiologist at the University of Edinburgh and a consultant neurologist in NHS Lothian, said vaccination was a potentially important way to reduce stroke.
“In this study, there was a plausible effect on reducing stroke incidence from vaccination against influenza, and also a plausible increase in the risk of stroke from having a recent febrile illness, which we have seen in other studies,” he commented.
Dr. Whiteley noted that this observation was particularly relevant now because of the COVID-19 pandemic.
“We’ve all been worried about the risk of heart attack and stroke after COVID, where we’ve seen quite early high risks, and we are also optimistic about the effect of vaccination on reducing those incidences. We’ve seen data from the U.K. that there may be around a 20% reduction in risk of stroke from vaccination. So, it’s all quite plausible, but at the moment it’s all based on observational evidence and we really need some randomized evidence,” he said.
“Vaccination and infections have all sorts of odd confounders,” he added. “People who get vaccines tend to be more healthy than those who don’t get vaccines, so you can start to see quite implausible effects of vaccination on overall mortality, which probably aren’t real, and you probably can’t get rid of that totally with statistical methods.”
Alastair Webb, MD, University of Oxford (England), asked how reliable the current findings were, given that the occurrence of febrile illnesses and receipt of vaccines were all self-reported, and although there was an association for ischemic stroke and febrile illness, this seemed to go in the opposite direction for hemorrhagic stroke. He also noted that the 50% reduction in stroke risk with vaccination in this study seemed “quite a large magnitude of effect.”
Dr. Whiteley replied: “Yes, it is large, but it is promising.” He cited a previous meta-analysis of randomized studies that showed a roughly 25%-35% reduction in vascular events after flu vaccination, but noted that there was a lot of heterogeneity between studies.
“I’m not sure we’re going to see much more randomized evidence, but I think we can probably all agree that having a vaccine against flu or COVID is a good thing for all of us,” Dr. Whiteley concluded.
The INTERSTROKE study was funded by the Canadian Institutes of Health Research, Heart and Stroke Foundation of Canada, Canadian Stroke Network, Health Research Board Ireland, Swedish Research Council, Swedish Heart and Lung Foundation, The Health & Medical Care Committee of the Regional Executive Board, Region Vastra Gotaland (Sweden), AstraZeneca, Boehringer Ingelheim, Pfizer, MSD, Chest, Heart and Stroke Scotland, and The Stroke Association, with support from The UK Stroke Research Network. The authors reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
in a large new case-control study.
“While influenza vaccination is a cost-effective method to prevent influenza, it is also an effective way to reduce the burden of stroke,” said study author Christopher Schwarzbach, MD, of Ludwigshafen (Germany) Hospital.
“Our results therefore encourage the wider use of influenza vaccination,” he concluded.
Dr. Schwarzbach presented these data from the INTERSTROKE study at the 2022 European Stroke Organisation Conference.
He explained that acute inflammatory disease is thought to increase the risk for cerebrovascular events, and the seasonality of influenza-like illness appears to be associated with the seasonality of cardiovascular and cerebrovascular events. Previous observational studies have also shown a link between influenza vaccination and a reduced risk for stroke.
The current INTERSTROKE study was a large international case-control study conducted between 2007 and 2015 that involved 13,447 cases (patients within 5 days of their first stroke) and a similar number of age- and gender-matched people from 32 countries across the world.
All cases and control subjects were systematically asked whether they had acute febrile illness in the previous 4 weeks and whether they had received an influenza vaccination within the previous year.
Conditional logistical regression was used to quantify the results, with adjustment for 13 different possible confounding factors, including hypertension, activity, smoking, cardiovascular risk factors, and socioeconomic factors.
Results showed that having had an acute febrile illness in the previous 4 weeks was more commonly reported in the patients with an acute ischemic stroke (8.7%) than in control patients (5.6%). After adjustment for confounding factors, this gives an adjusted risk ratio of 1.18, which was of borderline statistical significance (95% confidence limits, 1.01-1.39), Dr. Schwarzbach reported.
The association between recent febrile illness and acute ischemic stroke was stronger when compared with community control subjects (adjusted odds ratio, 2.0), but it was absent when compared with hospital control subjects.
The association was also only apparent in Australia, China, North America, and Western Europe; it was not seen in other parts of the world.
There was no association between acute febrile illness and acute cerebral hemorrhage.
Flu vaccine linked to halving of stroke risk
Having received a flu vaccine in the previous year was strongly associated with a lower risk for any type of stroke (aOR, 0.53), ischemic stroke (aOR, 0.57), and hemorrhagic stroke (aOR, 0.34).
Dr. Schwarzbach noted that these results were also consistent in an extended statistical model that included variables that might reflect a willingness to be vaccinated and when compared with both community and hospital-based control subjects.
The strength of the association between influenza vaccination and reduced risk for stroke was similar when compared with either community or hospital control subjects, and was only moderately stronger during than outside the influenza season.
The association was also seen in all regions of the world apart from Africa and South Asia, Dr. Schwarzbach reported, but he noted that vaccination rates in these two regions were extremely low.
The researchers also found that the magnitude of the associations between flu vaccination and lower risk for stroke were stronger in individuals who had multiple annual vaccinations, with an odds ratio of 0.54 in those who had received a vaccine every year for the previous 5 years, and of 0.79 in those who had received one to four vaccinations in the previous 5 years.
Mechanism: Immune stimulation?
Discussing possible mechanisms behind these results, Dr. Schwarzbach noted that the finding that the association with influenza vaccination and reduced stroke risk was independent of seasonality was surprising. “We had expected the protective effect of vaccination to be bigger during the influenza season, but this wasn’t the case.”
He suggested that one explanation might be the inclusion of regions of the world where this seasonality doesn’t exist.
But he pointed out that the finding of a stronger association between flu vaccination and lower stroke risk in those who had received more vaccinations has given rise to another theory: that it is the stimulation of the immune system rather than the protection of infection against influenza that is the key factor.
In an interview with Dr. Schwarzbach, Guillaume Turk, MD, professor of neurology at GHU Paris, pointed out that causal inferences are always difficult in case-control studies and in clinical epidemiology in general.
“What makes you think that this association between influenza vaccination and decreased risk is causal rather than due to unmeasured confounders? For example, patients who received vaccination may have received more medical attention and may have been more aware of the risk factors for stroke,” he asked.
Dr. Schwarzbach replied: “Yes, this is the issue of healthy user bias, which is always a problem in this type of research and is hard to address.”
“What we tried to do here is to adjust for variables that might influence the willingness of people to get vaccinated,” he added. “These were mainly socioeconomic factors. But, of course, this is something that we can’t rule out.”
Dr. Schwarzbach noted that, for more reliable information on this association, prospective studies are needed.
‘A plausible effect’
Discussing the study after the presentation, William Whiteley, BM, PhD, a clinical epidemiologist at the University of Edinburgh and a consultant neurologist in NHS Lothian, said vaccination was a potentially important way to reduce stroke.
“In this study, there was a plausible effect on reducing stroke incidence from vaccination against influenza, and also a plausible increase in the risk of stroke from having a recent febrile illness, which we have seen in other studies,” he commented.
Dr. Whiteley noted that this observation was particularly relevant now because of the COVID-19 pandemic.
“We’ve all been worried about the risk of heart attack and stroke after COVID, where we’ve seen quite early high risks, and we are also optimistic about the effect of vaccination on reducing those incidences. We’ve seen data from the U.K. that there may be around a 20% reduction in risk of stroke from vaccination. So, it’s all quite plausible, but at the moment it’s all based on observational evidence and we really need some randomized evidence,” he said.
“Vaccination and infections have all sorts of odd confounders,” he added. “People who get vaccines tend to be more healthy than those who don’t get vaccines, so you can start to see quite implausible effects of vaccination on overall mortality, which probably aren’t real, and you probably can’t get rid of that totally with statistical methods.”
Alastair Webb, MD, University of Oxford (England), asked how reliable the current findings were, given that the occurrence of febrile illnesses and receipt of vaccines were all self-reported, and although there was an association for ischemic stroke and febrile illness, this seemed to go in the opposite direction for hemorrhagic stroke. He also noted that the 50% reduction in stroke risk with vaccination in this study seemed “quite a large magnitude of effect.”
Dr. Whiteley replied: “Yes, it is large, but it is promising.” He cited a previous meta-analysis of randomized studies that showed a roughly 25%-35% reduction in vascular events after flu vaccination, but noted that there was a lot of heterogeneity between studies.
“I’m not sure we’re going to see much more randomized evidence, but I think we can probably all agree that having a vaccine against flu or COVID is a good thing for all of us,” Dr. Whiteley concluded.
The INTERSTROKE study was funded by the Canadian Institutes of Health Research, Heart and Stroke Foundation of Canada, Canadian Stroke Network, Health Research Board Ireland, Swedish Research Council, Swedish Heart and Lung Foundation, The Health & Medical Care Committee of the Regional Executive Board, Region Vastra Gotaland (Sweden), AstraZeneca, Boehringer Ingelheim, Pfizer, MSD, Chest, Heart and Stroke Scotland, and The Stroke Association, with support from The UK Stroke Research Network. The authors reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
in a large new case-control study.
“While influenza vaccination is a cost-effective method to prevent influenza, it is also an effective way to reduce the burden of stroke,” said study author Christopher Schwarzbach, MD, of Ludwigshafen (Germany) Hospital.
“Our results therefore encourage the wider use of influenza vaccination,” he concluded.
Dr. Schwarzbach presented these data from the INTERSTROKE study at the 2022 European Stroke Organisation Conference.
He explained that acute inflammatory disease is thought to increase the risk for cerebrovascular events, and the seasonality of influenza-like illness appears to be associated with the seasonality of cardiovascular and cerebrovascular events. Previous observational studies have also shown a link between influenza vaccination and a reduced risk for stroke.
The current INTERSTROKE study was a large international case-control study conducted between 2007 and 2015 that involved 13,447 cases (patients within 5 days of their first stroke) and a similar number of age- and gender-matched people from 32 countries across the world.
All cases and control subjects were systematically asked whether they had acute febrile illness in the previous 4 weeks and whether they had received an influenza vaccination within the previous year.
Conditional logistical regression was used to quantify the results, with adjustment for 13 different possible confounding factors, including hypertension, activity, smoking, cardiovascular risk factors, and socioeconomic factors.
Results showed that having had an acute febrile illness in the previous 4 weeks was more commonly reported in the patients with an acute ischemic stroke (8.7%) than in control patients (5.6%). After adjustment for confounding factors, this gives an adjusted risk ratio of 1.18, which was of borderline statistical significance (95% confidence limits, 1.01-1.39), Dr. Schwarzbach reported.
The association between recent febrile illness and acute ischemic stroke was stronger when compared with community control subjects (adjusted odds ratio, 2.0), but it was absent when compared with hospital control subjects.
The association was also only apparent in Australia, China, North America, and Western Europe; it was not seen in other parts of the world.
There was no association between acute febrile illness and acute cerebral hemorrhage.
Flu vaccine linked to halving of stroke risk
Having received a flu vaccine in the previous year was strongly associated with a lower risk for any type of stroke (aOR, 0.53), ischemic stroke (aOR, 0.57), and hemorrhagic stroke (aOR, 0.34).
Dr. Schwarzbach noted that these results were also consistent in an extended statistical model that included variables that might reflect a willingness to be vaccinated and when compared with both community and hospital-based control subjects.
The strength of the association between influenza vaccination and reduced risk for stroke was similar when compared with either community or hospital control subjects, and was only moderately stronger during than outside the influenza season.
The association was also seen in all regions of the world apart from Africa and South Asia, Dr. Schwarzbach reported, but he noted that vaccination rates in these two regions were extremely low.
The researchers also found that the magnitude of the associations between flu vaccination and lower risk for stroke were stronger in individuals who had multiple annual vaccinations, with an odds ratio of 0.54 in those who had received a vaccine every year for the previous 5 years, and of 0.79 in those who had received one to four vaccinations in the previous 5 years.
Mechanism: Immune stimulation?
Discussing possible mechanisms behind these results, Dr. Schwarzbach noted that the finding that the association with influenza vaccination and reduced stroke risk was independent of seasonality was surprising. “We had expected the protective effect of vaccination to be bigger during the influenza season, but this wasn’t the case.”
He suggested that one explanation might be the inclusion of regions of the world where this seasonality doesn’t exist.
But he pointed out that the finding of a stronger association between flu vaccination and lower stroke risk in those who had received more vaccinations has given rise to another theory: that it is the stimulation of the immune system rather than the protection of infection against influenza that is the key factor.
In an interview with Dr. Schwarzbach, Guillaume Turk, MD, professor of neurology at GHU Paris, pointed out that causal inferences are always difficult in case-control studies and in clinical epidemiology in general.
“What makes you think that this association between influenza vaccination and decreased risk is causal rather than due to unmeasured confounders? For example, patients who received vaccination may have received more medical attention and may have been more aware of the risk factors for stroke,” he asked.
Dr. Schwarzbach replied: “Yes, this is the issue of healthy user bias, which is always a problem in this type of research and is hard to address.”
“What we tried to do here is to adjust for variables that might influence the willingness of people to get vaccinated,” he added. “These were mainly socioeconomic factors. But, of course, this is something that we can’t rule out.”
Dr. Schwarzbach noted that, for more reliable information on this association, prospective studies are needed.
‘A plausible effect’
Discussing the study after the presentation, William Whiteley, BM, PhD, a clinical epidemiologist at the University of Edinburgh and a consultant neurologist in NHS Lothian, said vaccination was a potentially important way to reduce stroke.
“In this study, there was a plausible effect on reducing stroke incidence from vaccination against influenza, and also a plausible increase in the risk of stroke from having a recent febrile illness, which we have seen in other studies,” he commented.
Dr. Whiteley noted that this observation was particularly relevant now because of the COVID-19 pandemic.
“We’ve all been worried about the risk of heart attack and stroke after COVID, where we’ve seen quite early high risks, and we are also optimistic about the effect of vaccination on reducing those incidences. We’ve seen data from the U.K. that there may be around a 20% reduction in risk of stroke from vaccination. So, it’s all quite plausible, but at the moment it’s all based on observational evidence and we really need some randomized evidence,” he said.
“Vaccination and infections have all sorts of odd confounders,” he added. “People who get vaccines tend to be more healthy than those who don’t get vaccines, so you can start to see quite implausible effects of vaccination on overall mortality, which probably aren’t real, and you probably can’t get rid of that totally with statistical methods.”
Alastair Webb, MD, University of Oxford (England), asked how reliable the current findings were, given that the occurrence of febrile illnesses and receipt of vaccines were all self-reported, and although there was an association for ischemic stroke and febrile illness, this seemed to go in the opposite direction for hemorrhagic stroke. He also noted that the 50% reduction in stroke risk with vaccination in this study seemed “quite a large magnitude of effect.”
Dr. Whiteley replied: “Yes, it is large, but it is promising.” He cited a previous meta-analysis of randomized studies that showed a roughly 25%-35% reduction in vascular events after flu vaccination, but noted that there was a lot of heterogeneity between studies.
“I’m not sure we’re going to see much more randomized evidence, but I think we can probably all agree that having a vaccine against flu or COVID is a good thing for all of us,” Dr. Whiteley concluded.
The INTERSTROKE study was funded by the Canadian Institutes of Health Research, Heart and Stroke Foundation of Canada, Canadian Stroke Network, Health Research Board Ireland, Swedish Research Council, Swedish Heart and Lung Foundation, The Health & Medical Care Committee of the Regional Executive Board, Region Vastra Gotaland (Sweden), AstraZeneca, Boehringer Ingelheim, Pfizer, MSD, Chest, Heart and Stroke Scotland, and The Stroke Association, with support from The UK Stroke Research Network. The authors reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
Antibiotics use and vaccine antibody levels
In this column I have previously discussed the microbiome and its importance to health, especially as it relates to infections in children. Given the appreciated connection between microbiome and immunity, my group in Rochester, N.Y., recently undertook a study of the effect of antibiotic usage on the immune response to routine early childhood vaccines. In mouse models, it was previously shown that antibiotic exposure induced a reduction in the abundance and diversity of gut microbiota that in turn negatively affected the generation and maintenance of vaccine-induced immunity.1,2 A study from Stanford University was the first experimental human trial of antibiotic effects on vaccine responses. Adult volunteers were given an antibiotic or not before seasonal influenza vaccination and the researchers identified specific bacteria in the gut that were reduced by the antibiotics given. Those normal bacteria in the gut microbiome were shown to provide positive immunity signals to the systemic immune system that potentiated vaccine responses.3
My group conducted the first-ever study in children to explore whether an association existed between antibiotic use and vaccine-induced antibody levels. In the May issue of Pediatrics we report results from 560 children studied.4 From these children, 11,888 serum antibody levels to vaccine antigens were measured. Vaccine-induced antibody levels were determined at various time points after primary vaccination at child age 2, 4, and 6 months and boosters at age 12-18 months for 10 antigens included in four vaccines: DTaP, Hib, IPV, and PCV. The antibody levels to vaccine components were measured to DTaP (diphtheria toxoid, pertussis toxoid, tetanus toxoid, pertactin, and filamentous hemagglutinin), Hib conjugate (polyribosylribitol phosphate), IPV (polio 2), and PCV (serotypes 6B, 14, and 23F). A total of 342 children with 1,678 antibiotic courses prescribed were compared with 218 children with no antibiotic exposures. The predominant antibiotics prescribed were amoxicillin, cefdinir, amoxicillin/clavulanate, and ceftriaxone, since most treatments were for acute otitis media.
Of possible high clinical relevance, we found that from 9 to 24 months of age, children with antibiotic exposure had a higher frequency of vaccine-induced antibody levels below protection compared with children with no antibiotic use, placing them at risk of contracting a vaccine-preventable infection for DTaP antigens DT, TT, and PT and for PCV serotype 14.
For time points where antibody levels were determined within 30 days of completion of a course of antibiotics (recent antibiotic use), individual antibiotics were analyzed for effect on antibody levels below protective levels. Across all vaccine antigens measured, we found that all antibiotics had a negative effect on antibody levels and percentage of children achieving the protective antibody level threshold. Amoxicillin use had a lower association with lower antibody levels than the broader spectrum antibiotics, amoxicillin clavulanate (Augmentin), cefdinir, and ceftriaxone. For children receiving amoxicillin/clavulanate prescriptions, it was possible to compare the effect of shorter versus longer courses and we found that a 5-day course was associated with subprotective antibody levels similar to 10 days of amoxicillin, whereas 10-day amoxicillin/clavulanate was associated with higher frequency of children having subprotective antibody levels (Figure).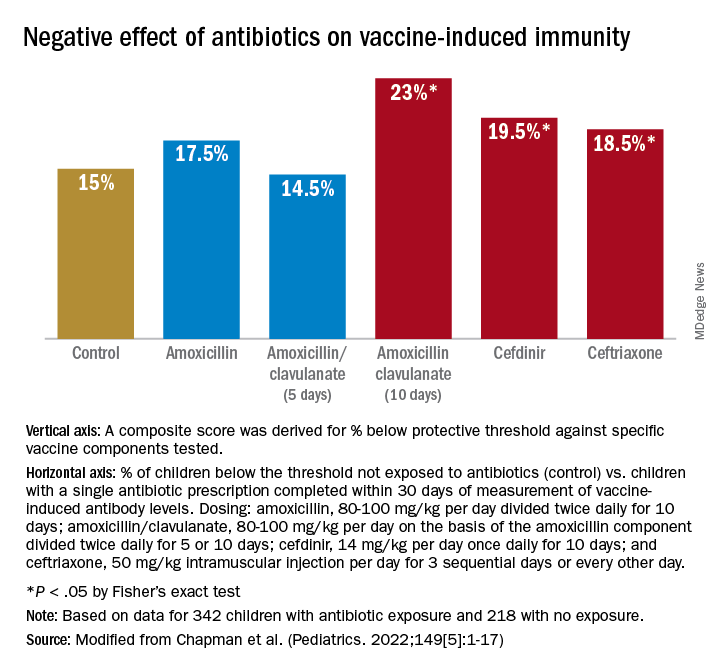
We examined whether accumulation of antibiotic courses in the first year of life had an association with subsequent vaccine-induced antibody levels and found that each antibiotic prescription was associated with a reduction in the median antibody level. For DTaP, each prescription was associated with 5.8% drop in antibody level to the vaccine components. For Hib the drop was 6.8%, IPV was 11.3%, and PCV was 10.4% – all statistically significant. To determine if booster vaccination influenced this association, a second analysis was performed using antibiotic prescriptions up to 15 months of age. We found each antibiotic prescription was associated with a reduction in median vaccine-induced antibody levels for DTaP by 18%, Hib by 21%, IPV by 19%, and PCV by 12% – all statistically significant.
Our study is the first in young children during the early age window where vaccine-induced immunity is established. Antibiotic use was associated with increased frequency of subprotective antibody levels for several vaccines used in children up to 2 years of age. The lower antibody levels could leave children vulnerable to vaccine preventable diseases. Perhaps outbreaks of vaccine-preventable diseases, such as pertussis, may be a consequence of multiple courses of antibiotics suppressing vaccine-induced immunity.
A goal of this study was to explore potential acute and long-term effects of antibiotic exposure on vaccine-induced antibody levels. Accumulated antibiotic courses up to booster immunization was associated with decreased vaccine antibody levels both before and after booster, suggesting that booster immunization was not sufficient to change the negative association with antibiotic exposure. The results were similar for all vaccines tested, suggesting that the specific vaccine formulation was not a factor.
The study has several limitations. The antibiotic prescription data and measurements of vaccine-induced antibody levels were recorded and measured prospectively; however, our analysis was done retrospectively. The group of study children was derived from my private practice in Rochester, N.Y., and may not be broadly representative of all children. The number of vaccine antibody measurements was limited by serum availability at some sampling time points in some children; and sometimes, the serum samples were collected far apart, which weakened our ability to perform longitudinal analyses. We did not collect stool samples from the children so we could not directly study the effect of antibiotic courses on the gut microbiome.
Our study adds new reasons to be cautious about overprescribing antibiotics on an individual child basis because an adverse effect extends to reduction in vaccine responses. This should be explained to parents requesting unnecessary antibiotics for colds and coughs. When antibiotics are necessary, the judicious choice of a narrow-spectrum antibiotic or a shorter duration of a broader spectrum antibiotic may reduce adverse effects on vaccine-induced immunity.
References
1. Valdez Y et al. Influence of the microbiota on vaccine effectiveness. Trends Immunol. 2014;35(11):526-37.
2. Lynn MA et al. Early-life antibiotic-driven dysbiosis leads to dysregulated vaccine immune responses in mice. Cell Host Microbe. 2018;23(5):653-60.e5.
3. Hagan T et al. Antibiotics-driven gut microbiome perturbation alters immunity to vaccines in humans. Cell. 2019;178(6):1313-28.e13.
4. Chapman T et al. Antibiotic use and vaccine antibody levels. Pediatrics. 2022;149(5);1-17. doi: 10.1542/peds.2021-052061.
In this column I have previously discussed the microbiome and its importance to health, especially as it relates to infections in children. Given the appreciated connection between microbiome and immunity, my group in Rochester, N.Y., recently undertook a study of the effect of antibiotic usage on the immune response to routine early childhood vaccines. In mouse models, it was previously shown that antibiotic exposure induced a reduction in the abundance and diversity of gut microbiota that in turn negatively affected the generation and maintenance of vaccine-induced immunity.1,2 A study from Stanford University was the first experimental human trial of antibiotic effects on vaccine responses. Adult volunteers were given an antibiotic or not before seasonal influenza vaccination and the researchers identified specific bacteria in the gut that were reduced by the antibiotics given. Those normal bacteria in the gut microbiome were shown to provide positive immunity signals to the systemic immune system that potentiated vaccine responses.3
My group conducted the first-ever study in children to explore whether an association existed between antibiotic use and vaccine-induced antibody levels. In the May issue of Pediatrics we report results from 560 children studied.4 From these children, 11,888 serum antibody levels to vaccine antigens were measured. Vaccine-induced antibody levels were determined at various time points after primary vaccination at child age 2, 4, and 6 months and boosters at age 12-18 months for 10 antigens included in four vaccines: DTaP, Hib, IPV, and PCV. The antibody levels to vaccine components were measured to DTaP (diphtheria toxoid, pertussis toxoid, tetanus toxoid, pertactin, and filamentous hemagglutinin), Hib conjugate (polyribosylribitol phosphate), IPV (polio 2), and PCV (serotypes 6B, 14, and 23F). A total of 342 children with 1,678 antibiotic courses prescribed were compared with 218 children with no antibiotic exposures. The predominant antibiotics prescribed were amoxicillin, cefdinir, amoxicillin/clavulanate, and ceftriaxone, since most treatments were for acute otitis media.
Of possible high clinical relevance, we found that from 9 to 24 months of age, children with antibiotic exposure had a higher frequency of vaccine-induced antibody levels below protection compared with children with no antibiotic use, placing them at risk of contracting a vaccine-preventable infection for DTaP antigens DT, TT, and PT and for PCV serotype 14.
For time points where antibody levels were determined within 30 days of completion of a course of antibiotics (recent antibiotic use), individual antibiotics were analyzed for effect on antibody levels below protective levels. Across all vaccine antigens measured, we found that all antibiotics had a negative effect on antibody levels and percentage of children achieving the protective antibody level threshold. Amoxicillin use had a lower association with lower antibody levels than the broader spectrum antibiotics, amoxicillin clavulanate (Augmentin), cefdinir, and ceftriaxone. For children receiving amoxicillin/clavulanate prescriptions, it was possible to compare the effect of shorter versus longer courses and we found that a 5-day course was associated with subprotective antibody levels similar to 10 days of amoxicillin, whereas 10-day amoxicillin/clavulanate was associated with higher frequency of children having subprotective antibody levels (Figure).
We examined whether accumulation of antibiotic courses in the first year of life had an association with subsequent vaccine-induced antibody levels and found that each antibiotic prescription was associated with a reduction in the median antibody level. For DTaP, each prescription was associated with 5.8% drop in antibody level to the vaccine components. For Hib the drop was 6.8%, IPV was 11.3%, and PCV was 10.4% – all statistically significant. To determine if booster vaccination influenced this association, a second analysis was performed using antibiotic prescriptions up to 15 months of age. We found each antibiotic prescription was associated with a reduction in median vaccine-induced antibody levels for DTaP by 18%, Hib by 21%, IPV by 19%, and PCV by 12% – all statistically significant.
Our study is the first in young children during the early age window where vaccine-induced immunity is established. Antibiotic use was associated with increased frequency of subprotective antibody levels for several vaccines used in children up to 2 years of age. The lower antibody levels could leave children vulnerable to vaccine preventable diseases. Perhaps outbreaks of vaccine-preventable diseases, such as pertussis, may be a consequence of multiple courses of antibiotics suppressing vaccine-induced immunity.
A goal of this study was to explore potential acute and long-term effects of antibiotic exposure on vaccine-induced antibody levels. Accumulated antibiotic courses up to booster immunization was associated with decreased vaccine antibody levels both before and after booster, suggesting that booster immunization was not sufficient to change the negative association with antibiotic exposure. The results were similar for all vaccines tested, suggesting that the specific vaccine formulation was not a factor.
The study has several limitations. The antibiotic prescription data and measurements of vaccine-induced antibody levels were recorded and measured prospectively; however, our analysis was done retrospectively. The group of study children was derived from my private practice in Rochester, N.Y., and may not be broadly representative of all children. The number of vaccine antibody measurements was limited by serum availability at some sampling time points in some children; and sometimes, the serum samples were collected far apart, which weakened our ability to perform longitudinal analyses. We did not collect stool samples from the children so we could not directly study the effect of antibiotic courses on the gut microbiome.
Our study adds new reasons to be cautious about overprescribing antibiotics on an individual child basis because an adverse effect extends to reduction in vaccine responses. This should be explained to parents requesting unnecessary antibiotics for colds and coughs. When antibiotics are necessary, the judicious choice of a narrow-spectrum antibiotic or a shorter duration of a broader spectrum antibiotic may reduce adverse effects on vaccine-induced immunity.
References
1. Valdez Y et al. Influence of the microbiota on vaccine effectiveness. Trends Immunol. 2014;35(11):526-37.
2. Lynn MA et al. Early-life antibiotic-driven dysbiosis leads to dysregulated vaccine immune responses in mice. Cell Host Microbe. 2018;23(5):653-60.e5.
3. Hagan T et al. Antibiotics-driven gut microbiome perturbation alters immunity to vaccines in humans. Cell. 2019;178(6):1313-28.e13.
4. Chapman T et al. Antibiotic use and vaccine antibody levels. Pediatrics. 2022;149(5);1-17. doi: 10.1542/peds.2021-052061.
In this column I have previously discussed the microbiome and its importance to health, especially as it relates to infections in children. Given the appreciated connection between microbiome and immunity, my group in Rochester, N.Y., recently undertook a study of the effect of antibiotic usage on the immune response to routine early childhood vaccines. In mouse models, it was previously shown that antibiotic exposure induced a reduction in the abundance and diversity of gut microbiota that in turn negatively affected the generation and maintenance of vaccine-induced immunity.1,2 A study from Stanford University was the first experimental human trial of antibiotic effects on vaccine responses. Adult volunteers were given an antibiotic or not before seasonal influenza vaccination and the researchers identified specific bacteria in the gut that were reduced by the antibiotics given. Those normal bacteria in the gut microbiome were shown to provide positive immunity signals to the systemic immune system that potentiated vaccine responses.3
My group conducted the first-ever study in children to explore whether an association existed between antibiotic use and vaccine-induced antibody levels. In the May issue of Pediatrics we report results from 560 children studied.4 From these children, 11,888 serum antibody levels to vaccine antigens were measured. Vaccine-induced antibody levels were determined at various time points after primary vaccination at child age 2, 4, and 6 months and boosters at age 12-18 months for 10 antigens included in four vaccines: DTaP, Hib, IPV, and PCV. The antibody levels to vaccine components were measured to DTaP (diphtheria toxoid, pertussis toxoid, tetanus toxoid, pertactin, and filamentous hemagglutinin), Hib conjugate (polyribosylribitol phosphate), IPV (polio 2), and PCV (serotypes 6B, 14, and 23F). A total of 342 children with 1,678 antibiotic courses prescribed were compared with 218 children with no antibiotic exposures. The predominant antibiotics prescribed were amoxicillin, cefdinir, amoxicillin/clavulanate, and ceftriaxone, since most treatments were for acute otitis media.
Of possible high clinical relevance, we found that from 9 to 24 months of age, children with antibiotic exposure had a higher frequency of vaccine-induced antibody levels below protection compared with children with no antibiotic use, placing them at risk of contracting a vaccine-preventable infection for DTaP antigens DT, TT, and PT and for PCV serotype 14.
For time points where antibody levels were determined within 30 days of completion of a course of antibiotics (recent antibiotic use), individual antibiotics were analyzed for effect on antibody levels below protective levels. Across all vaccine antigens measured, we found that all antibiotics had a negative effect on antibody levels and percentage of children achieving the protective antibody level threshold. Amoxicillin use had a lower association with lower antibody levels than the broader spectrum antibiotics, amoxicillin clavulanate (Augmentin), cefdinir, and ceftriaxone. For children receiving amoxicillin/clavulanate prescriptions, it was possible to compare the effect of shorter versus longer courses and we found that a 5-day course was associated with subprotective antibody levels similar to 10 days of amoxicillin, whereas 10-day amoxicillin/clavulanate was associated with higher frequency of children having subprotective antibody levels (Figure).
We examined whether accumulation of antibiotic courses in the first year of life had an association with subsequent vaccine-induced antibody levels and found that each antibiotic prescription was associated with a reduction in the median antibody level. For DTaP, each prescription was associated with 5.8% drop in antibody level to the vaccine components. For Hib the drop was 6.8%, IPV was 11.3%, and PCV was 10.4% – all statistically significant. To determine if booster vaccination influenced this association, a second analysis was performed using antibiotic prescriptions up to 15 months of age. We found each antibiotic prescription was associated with a reduction in median vaccine-induced antibody levels for DTaP by 18%, Hib by 21%, IPV by 19%, and PCV by 12% – all statistically significant.
Our study is the first in young children during the early age window where vaccine-induced immunity is established. Antibiotic use was associated with increased frequency of subprotective antibody levels for several vaccines used in children up to 2 years of age. The lower antibody levels could leave children vulnerable to vaccine preventable diseases. Perhaps outbreaks of vaccine-preventable diseases, such as pertussis, may be a consequence of multiple courses of antibiotics suppressing vaccine-induced immunity.
A goal of this study was to explore potential acute and long-term effects of antibiotic exposure on vaccine-induced antibody levels. Accumulated antibiotic courses up to booster immunization was associated with decreased vaccine antibody levels both before and after booster, suggesting that booster immunization was not sufficient to change the negative association with antibiotic exposure. The results were similar for all vaccines tested, suggesting that the specific vaccine formulation was not a factor.
The study has several limitations. The antibiotic prescription data and measurements of vaccine-induced antibody levels were recorded and measured prospectively; however, our analysis was done retrospectively. The group of study children was derived from my private practice in Rochester, N.Y., and may not be broadly representative of all children. The number of vaccine antibody measurements was limited by serum availability at some sampling time points in some children; and sometimes, the serum samples were collected far apart, which weakened our ability to perform longitudinal analyses. We did not collect stool samples from the children so we could not directly study the effect of antibiotic courses on the gut microbiome.
Our study adds new reasons to be cautious about overprescribing antibiotics on an individual child basis because an adverse effect extends to reduction in vaccine responses. This should be explained to parents requesting unnecessary antibiotics for colds and coughs. When antibiotics are necessary, the judicious choice of a narrow-spectrum antibiotic or a shorter duration of a broader spectrum antibiotic may reduce adverse effects on vaccine-induced immunity.
References
1. Valdez Y et al. Influence of the microbiota on vaccine effectiveness. Trends Immunol. 2014;35(11):526-37.
2. Lynn MA et al. Early-life antibiotic-driven dysbiosis leads to dysregulated vaccine immune responses in mice. Cell Host Microbe. 2018;23(5):653-60.e5.
3. Hagan T et al. Antibiotics-driven gut microbiome perturbation alters immunity to vaccines in humans. Cell. 2019;178(6):1313-28.e13.
4. Chapman T et al. Antibiotic use and vaccine antibody levels. Pediatrics. 2022;149(5);1-17. doi: 10.1542/peds.2021-052061.






