User login
Predicting RSV’s Role in the Upcoming Winter Respiratory Season
For children younger than 5 years old, RSV is the main drive — approximately 2,000,000 outpatient/ED visits and about 75,000 hospitalizations annually. RSV disease ranges from upper respiratory tract infections, eg, in older children and healthy adults, to more severe lower tract disease in young children and the elderly. Premature infants and high-risk groups are particularly prone to severe disease.1 Up to 300 pediatric RSV deaths occur yearly. “Normal” RSV seasons start in mid-November, peak in late December-January, and end after April. Note: More drawn out seasons occur in southern latitudes, eg Texas or Florida. But lately RSV seasons have been anything but normal.
2015-2016 to 2022-2023
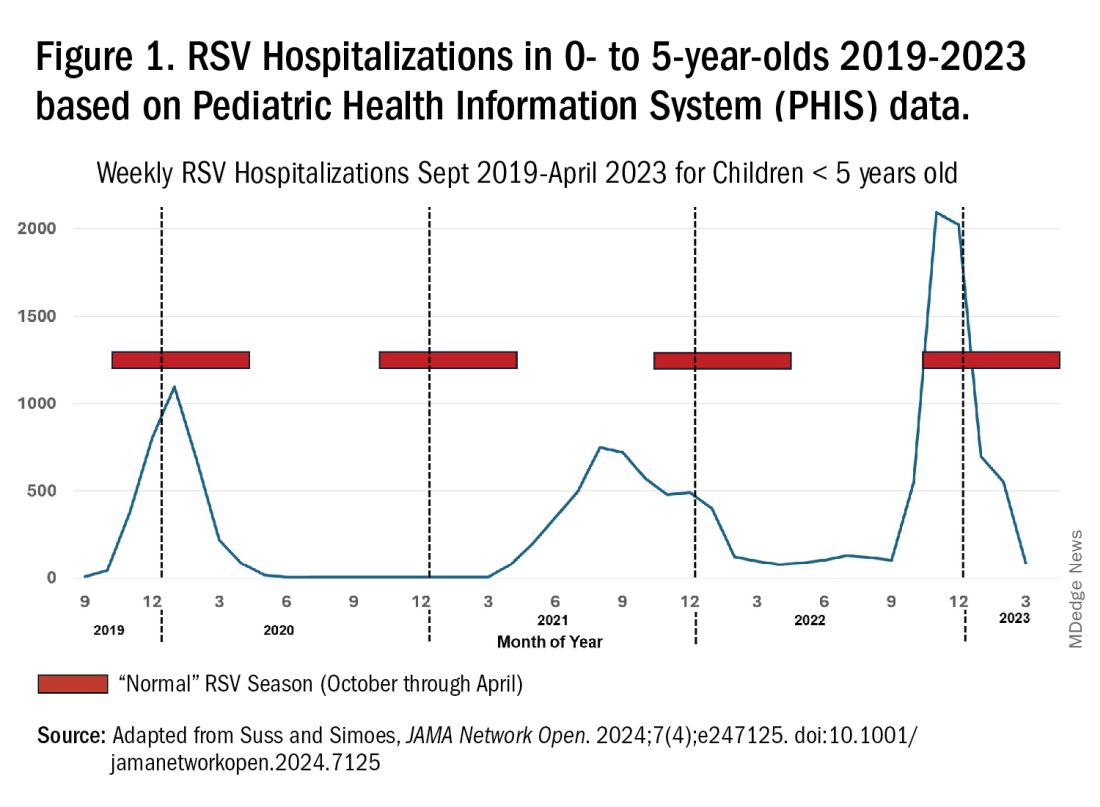
RSV data from the Pediatric Health Information System (PHIS), collected at over 49 US children’s hospitals during 2015 to early 2023, show how crazy RSV seasons have been lately.2 The involved months, intensity, and duration of four prepandemic seasons were pretty “normal” (Figure 1). The 2019-2020 season started normally, peaked in January 2020, and was slowing as expected by February. But when SARS-Cov-2 restrictions kicked in during mid-March, RSV detections tanked to almost nothing (ditto other respiratory viruses). A near 14-month RSV hiatus meant that the 2020-2021 RSV season never materialized. However, RSV was not done with us in 2021. It rebounded in May with weekly hospitalizations peaking in late July; this “rebound season” lasted 9 months, not dropping to baseline until February 2022 (Figure 1).
I guess we should have expected a post-pandemic “disturbance in the Force,” as Yoda once said; but I sure didn’t see a prolonged summer/fall/early winter RSV season coming. It was like two “normal” seasons mashed up into one late-but-long season. Not to be outdone, the 2022-2023 RSV season started early (September) and hospitalizations skyrocketed to peak in November at over twice the peak number from any year since 2015, overloading hospitals (influenza and SARS-Cov-2 seasons were co-circulating). The season terminated early though (March 2023).
Okay, so RSV seasonality/intensity were weird post pandemic, but was anything else different? Some 2021-2023 data suggest more RSV disease in older children, rather than the usual younger than 18 month-olds going through their first winter.3 More medically attended RSV in older ages (2-4 years of life) may have been due to the pandemic year without RSV circulation distorting herd immunity, ie older children remained RSV naive. Other data suggest the apparent increase was really just more frequent multiplex viral testing in older children triggered by SARS-CoV-2 co-circulation.4 More data are needed to decide.
CDC 2023-2024 RESP-NET data
The 2023-2024 winter surge (Figure 2), as measured by RESP-NET’s cumulative RSV, influenza and SARS-CoV-2 hospitalization rates for 0- to 5-year-olds,5 shows that all three viruses’ seasonal months were normal-ish: late October 2023 start, late December-early January peak, and mid-May 2024 return to baseline. RSV season was approximately 22% less severe by area-under-the-curve calculations compared with 2022-2023, but still worse than prepandemic years.6 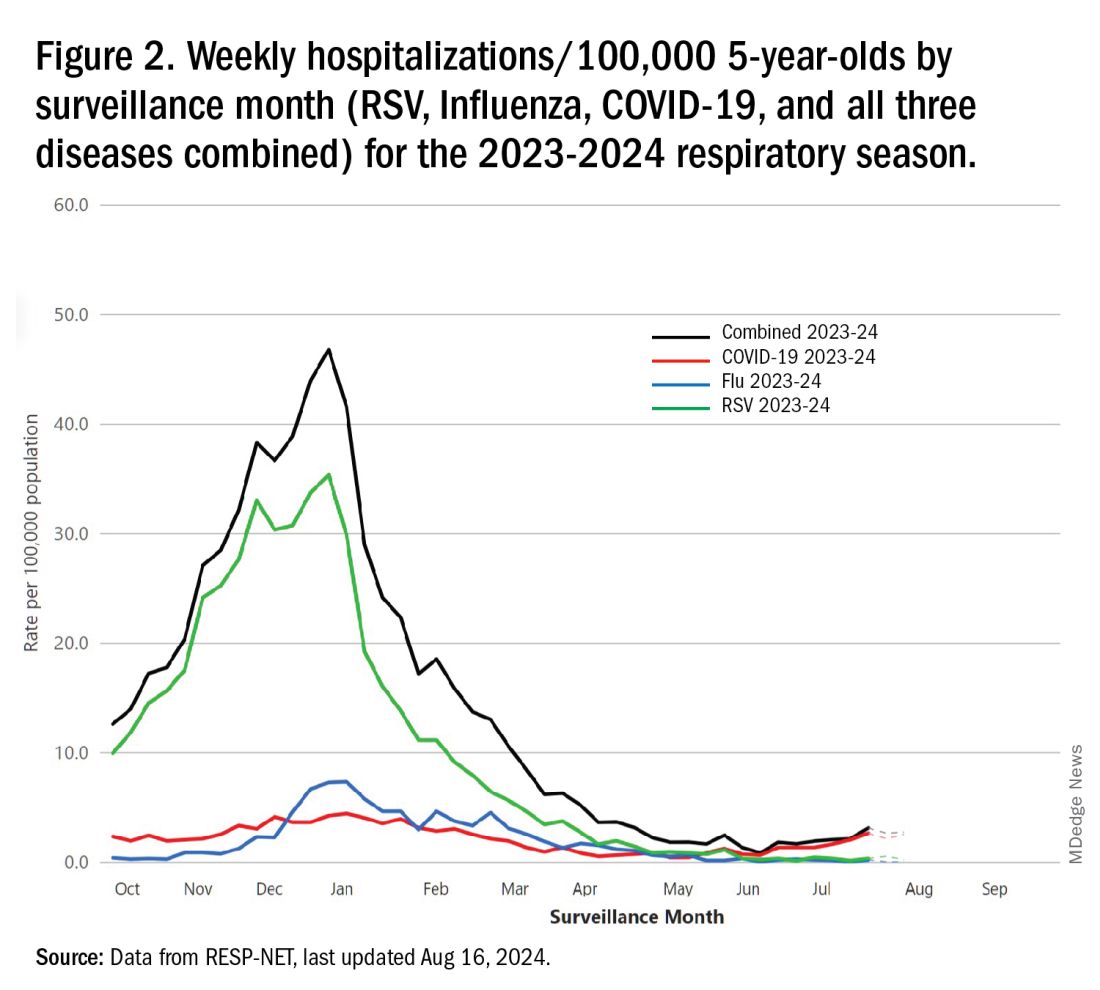
One wonders if the 2022-2023 RSV season might have been worse but for use of the limited supply of nirsevimab.7
Viral Parade
Now we ready ourselves for the 2024-2025 respiratory surge, wondering what nature has in store for us. Will the usual “respiratory virus parade” occur? Will rhinovirus and parainfluenza prevalence bump after a few weeks of schools being in session, adding to the now-usual summer/fall SARS-CoV-2 surge? Note: Twenty-seven states as of Aug. 16 had high SARS-CoV-2 detection in wastewater. Will RSV and influenza start sometime in October/November, peak in January (along with rising SARS-CoV2 activity), followed by a second parainfluenza bump as SARS-CoV-2, influenza, and RSV drop off in April/May? Further, will RSV and influenza seasons be more or less severe than the last 2 years?
Prediction
The overall 2024-2025 respiratory season will be less severe than the past 2 years and hopefully than recent prepandemic years. What is the blueprint for a milder season? First, herd immunity to non-RSV and non-influenza viruses (parainfluenza, rhinovirus, metapneumovirus, adenovirus) in older children should be normalized after 2 years back to usual social activity. So, I expect no mega-seasons from them. The emerging SARS-CoV-2 virus (LB.1) is immunologically close to its recent still-circulating ancestors (KP.2, KP.2.3, KP.3 and KP.3.1.1), so existing SARS-CoV2 herd immunity along with recommended booster vaccine uptake should keep the lid on SARS-CoV2.
Influenza Could Be the Bad News
Which type will dominate? Will a drift/shift occur or vaccine-mismatch reduce vaccine effectiveness? Can we get at least half the population influenza vaccinated, given the vaccine fatigue permeating the US population? The influenza season now underway in the Southern Hemisphere usually helps us predict our season. The Australian May-August 2024 experience (still on an upward trajectory for severity in mid-August) saw no drift/shift or vaccine mismatch. However, this 2024 season has been as severe as 2022 (their worst in a decade). That said, more than 95% has been type A (mostly H1N1 but H3N2 increased in July). So, if our overall 2024-2025 respiratory season is not milder, influenza is the most likely culprit. To reduce chances of influenza being the fly-in-the-ointment, we need to be particularly proactive with seasonal influenza vaccine which is back to the traditional trivalent formulation (one H1N1, one H3N2, and one B type).8 All of this could go out the window if avian influenza becomes more transmissible, but that seems unlikely at present.
Mild RSV Season?
RSV season should be blunted because of the increased use of both the remarkably effective CDC-recommended maternal RSV vaccine9 (one dose during pregnancy weeks 32 through 36, administered September through January) and of nirsevimab (up to 90% reduction in hospitalizations and ED visits).10 (See Figure 3.) 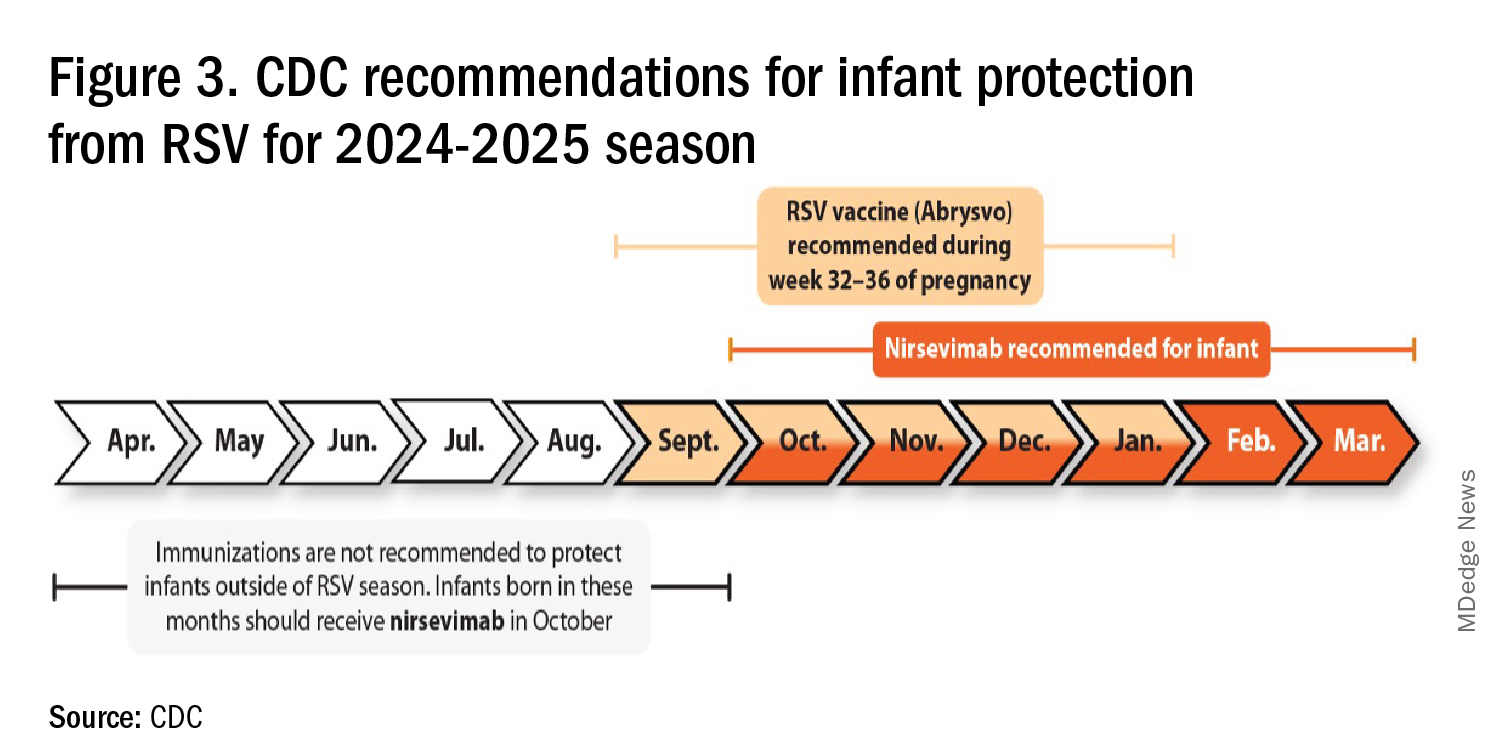
I also expect residual disease to occur mostly in younger than 18 month-olds (the “normal” aged population experiencing their first winter), who received no passive immunity (mother RSV unvaccinated and child did not receive nirsevimab). Some disease will still occur in high-risk infants/children. However, unlike active vaccination strategies, a competent immune system is not required to benefit from passive antibody, whether transplacental or directly administered.
Deep Thought
What if the traditional RSV seasonal hospitalization surge fails to materialize this season? It could happen. If we could get high acceptance/uptake of maternal vaccine and infant nirsevimab, RSV season could resemble the dramatic drop in rotavirus disease the second year after rotavirus vaccine introduction. We could be asking ourselves — “What happened to RSV?”
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Missouri. He said he had no relevant financial disclosures. Email him at pdnews@mdedge.com.
References
1. CDC. RSV in Infants and Young Children. Respiratory Syncytial Virus Infection (RSV). June 18, 2024. https://www.cdc.gov/rsv/infants-young-children/index.html.
2. Suss RJ and Simões EAF. Respiratory Syncytial Virus Hospital-Based Burden of Disease in Children Younger Than 5 Respiratory Syncytial Virus Hospital-Based Burden of Disease in Children Younger Than 5 Years, 2015-2022. JAMA Netw Open. 2024;7(4):e247125. doi:10.1001/jamanetworkopen.2024.7125.
3. Winthrop ZA et al. Pediatric Respiratory Syncytial Virus Hospitalizations and Respiratory Support After the COVID-19 Pandemic. JAMA Netw Open. 2024;7(6):e2416852. doi:10.1001/jamanetworkopen.2024.16852.
4. Petros BA et al. Increased Pediatric RSV Case Counts Following the Emergence of SARS-CoV-2 Are Attributable to Increased Testing. medRxiv [Preprint]. 2024 Feb 12:2024.02.06.24302387. doi: 10.1101/2024.02.06.24302387.
5. Rates of Laboratory-Confirmed RSV, COVID-19, and Flu Hospitalizations from the RESP-NET Surveillance Systems. Centers for Disease Control and Prevention. https://data.cdc.gov/Public-Health-Surveillance/Rates-of-Laboratory-Confirmed-RSV-COVID-19-and-Flu/kvib-3txy/about_data.
6. CDC. Evaluating the 2023-2024 Respiratory Disease Season Outlook. CFA: Qualitative Assessments. August 14, 2024. https://www.cdc.gov/cfa-qualitative-assessments/php/data-research/2023-2024-season-outlook-retro.html.
7. Health Alert Network (HAN). Limited Availability of Nirsevimab in the United States—Interim CDC Recommendations to Protect Infants from Respiratory Syncytial Virus (RSV) during the 2023–2024 Respiratory Virus Season. October 23, 2023. https://emergency.cdc.gov/han/2023/han00499.asp.
8. CDC. Information for the 2024-2025 Flu Season. Centers for Disease Control and Prevention. March 14, 2024. https://www.cdc.gov/flu/season/faq-flu-season-2024-2025.htm.
9. Kampmann B et al. Bivalent Prefusion F Vaccine in Pregnancy to Prevent RSV Illness in Infants. N Engl J Med. 2023 Apr 20;388(16):1451-1464. doi: 10.1056/NEJMoa2216480.
10. Moline HL. Early Estimate of Nirsevimab Effectiveness for Prevention of Respiratory Syncytial Virus–Associated Hospitalization Among Infants Entering Their First Respiratory Syncytial Virus Season — New Vaccine Surveillance Network, October 2023–February 2024. MMWR Morb Mortal Wkly Rep. 2024;73. doi: 10.15585/mmwr.mm7309a4.
For children younger than 5 years old, RSV is the main drive — approximately 2,000,000 outpatient/ED visits and about 75,000 hospitalizations annually. RSV disease ranges from upper respiratory tract infections, eg, in older children and healthy adults, to more severe lower tract disease in young children and the elderly. Premature infants and high-risk groups are particularly prone to severe disease.1 Up to 300 pediatric RSV deaths occur yearly. “Normal” RSV seasons start in mid-November, peak in late December-January, and end after April. Note: More drawn out seasons occur in southern latitudes, eg Texas or Florida. But lately RSV seasons have been anything but normal.
2015-2016 to 2022-2023
RSV data from the Pediatric Health Information System (PHIS), collected at over 49 US children’s hospitals during 2015 to early 2023, show how crazy RSV seasons have been lately.2 The involved months, intensity, and duration of four prepandemic seasons were pretty “normal” (Figure 1). The 2019-2020 season started normally, peaked in January 2020, and was slowing as expected by February. But when SARS-Cov-2 restrictions kicked in during mid-March, RSV detections tanked to almost nothing (ditto other respiratory viruses). A near 14-month RSV hiatus meant that the 2020-2021 RSV season never materialized. However, RSV was not done with us in 2021. It rebounded in May with weekly hospitalizations peaking in late July; this “rebound season” lasted 9 months, not dropping to baseline until February 2022 (Figure 1).
I guess we should have expected a post-pandemic “disturbance in the Force,” as Yoda once said; but I sure didn’t see a prolonged summer/fall/early winter RSV season coming. It was like two “normal” seasons mashed up into one late-but-long season. Not to be outdone, the 2022-2023 RSV season started early (September) and hospitalizations skyrocketed to peak in November at over twice the peak number from any year since 2015, overloading hospitals (influenza and SARS-Cov-2 seasons were co-circulating). The season terminated early though (March 2023).
Okay, so RSV seasonality/intensity were weird post pandemic, but was anything else different? Some 2021-2023 data suggest more RSV disease in older children, rather than the usual younger than 18 month-olds going through their first winter.3 More medically attended RSV in older ages (2-4 years of life) may have been due to the pandemic year without RSV circulation distorting herd immunity, ie older children remained RSV naive. Other data suggest the apparent increase was really just more frequent multiplex viral testing in older children triggered by SARS-CoV-2 co-circulation.4 More data are needed to decide.
CDC 2023-2024 RESP-NET data
The 2023-2024 winter surge (Figure 2), as measured by RESP-NET’s cumulative RSV, influenza and SARS-CoV-2 hospitalization rates for 0- to 5-year-olds,5 shows that all three viruses’ seasonal months were normal-ish: late October 2023 start, late December-early January peak, and mid-May 2024 return to baseline. RSV season was approximately 22% less severe by area-under-the-curve calculations compared with 2022-2023, but still worse than prepandemic years.6 
One wonders if the 2022-2023 RSV season might have been worse but for use of the limited supply of nirsevimab.7
Viral Parade
Now we ready ourselves for the 2024-2025 respiratory surge, wondering what nature has in store for us. Will the usual “respiratory virus parade” occur? Will rhinovirus and parainfluenza prevalence bump after a few weeks of schools being in session, adding to the now-usual summer/fall SARS-CoV-2 surge? Note: Twenty-seven states as of Aug. 16 had high SARS-CoV-2 detection in wastewater. Will RSV and influenza start sometime in October/November, peak in January (along with rising SARS-CoV2 activity), followed by a second parainfluenza bump as SARS-CoV-2, influenza, and RSV drop off in April/May? Further, will RSV and influenza seasons be more or less severe than the last 2 years?
Prediction
The overall 2024-2025 respiratory season will be less severe than the past 2 years and hopefully than recent prepandemic years. What is the blueprint for a milder season? First, herd immunity to non-RSV and non-influenza viruses (parainfluenza, rhinovirus, metapneumovirus, adenovirus) in older children should be normalized after 2 years back to usual social activity. So, I expect no mega-seasons from them. The emerging SARS-CoV-2 virus (LB.1) is immunologically close to its recent still-circulating ancestors (KP.2, KP.2.3, KP.3 and KP.3.1.1), so existing SARS-CoV2 herd immunity along with recommended booster vaccine uptake should keep the lid on SARS-CoV2.
Influenza Could Be the Bad News
Which type will dominate? Will a drift/shift occur or vaccine-mismatch reduce vaccine effectiveness? Can we get at least half the population influenza vaccinated, given the vaccine fatigue permeating the US population? The influenza season now underway in the Southern Hemisphere usually helps us predict our season. The Australian May-August 2024 experience (still on an upward trajectory for severity in mid-August) saw no drift/shift or vaccine mismatch. However, this 2024 season has been as severe as 2022 (their worst in a decade). That said, more than 95% has been type A (mostly H1N1 but H3N2 increased in July). So, if our overall 2024-2025 respiratory season is not milder, influenza is the most likely culprit. To reduce chances of influenza being the fly-in-the-ointment, we need to be particularly proactive with seasonal influenza vaccine which is back to the traditional trivalent formulation (one H1N1, one H3N2, and one B type).8 All of this could go out the window if avian influenza becomes more transmissible, but that seems unlikely at present.
Mild RSV Season?
RSV season should be blunted because of the increased use of both the remarkably effective CDC-recommended maternal RSV vaccine9 (one dose during pregnancy weeks 32 through 36, administered September through January) and of nirsevimab (up to 90% reduction in hospitalizations and ED visits).10 (See Figure 3.) 
I also expect residual disease to occur mostly in younger than 18 month-olds (the “normal” aged population experiencing their first winter), who received no passive immunity (mother RSV unvaccinated and child did not receive nirsevimab). Some disease will still occur in high-risk infants/children. However, unlike active vaccination strategies, a competent immune system is not required to benefit from passive antibody, whether transplacental or directly administered.
Deep Thought
What if the traditional RSV seasonal hospitalization surge fails to materialize this season? It could happen. If we could get high acceptance/uptake of maternal vaccine and infant nirsevimab, RSV season could resemble the dramatic drop in rotavirus disease the second year after rotavirus vaccine introduction. We could be asking ourselves — “What happened to RSV?”
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Missouri. He said he had no relevant financial disclosures. Email him at pdnews@mdedge.com.
References
1. CDC. RSV in Infants and Young Children. Respiratory Syncytial Virus Infection (RSV). June 18, 2024. https://www.cdc.gov/rsv/infants-young-children/index.html.
2. Suss RJ and Simões EAF. Respiratory Syncytial Virus Hospital-Based Burden of Disease in Children Younger Than 5 Respiratory Syncytial Virus Hospital-Based Burden of Disease in Children Younger Than 5 Years, 2015-2022. JAMA Netw Open. 2024;7(4):e247125. doi:10.1001/jamanetworkopen.2024.7125.
3. Winthrop ZA et al. Pediatric Respiratory Syncytial Virus Hospitalizations and Respiratory Support After the COVID-19 Pandemic. JAMA Netw Open. 2024;7(6):e2416852. doi:10.1001/jamanetworkopen.2024.16852.
4. Petros BA et al. Increased Pediatric RSV Case Counts Following the Emergence of SARS-CoV-2 Are Attributable to Increased Testing. medRxiv [Preprint]. 2024 Feb 12:2024.02.06.24302387. doi: 10.1101/2024.02.06.24302387.
5. Rates of Laboratory-Confirmed RSV, COVID-19, and Flu Hospitalizations from the RESP-NET Surveillance Systems. Centers for Disease Control and Prevention. https://data.cdc.gov/Public-Health-Surveillance/Rates-of-Laboratory-Confirmed-RSV-COVID-19-and-Flu/kvib-3txy/about_data.
6. CDC. Evaluating the 2023-2024 Respiratory Disease Season Outlook. CFA: Qualitative Assessments. August 14, 2024. https://www.cdc.gov/cfa-qualitative-assessments/php/data-research/2023-2024-season-outlook-retro.html.
7. Health Alert Network (HAN). Limited Availability of Nirsevimab in the United States—Interim CDC Recommendations to Protect Infants from Respiratory Syncytial Virus (RSV) during the 2023–2024 Respiratory Virus Season. October 23, 2023. https://emergency.cdc.gov/han/2023/han00499.asp.
8. CDC. Information for the 2024-2025 Flu Season. Centers for Disease Control and Prevention. March 14, 2024. https://www.cdc.gov/flu/season/faq-flu-season-2024-2025.htm.
9. Kampmann B et al. Bivalent Prefusion F Vaccine in Pregnancy to Prevent RSV Illness in Infants. N Engl J Med. 2023 Apr 20;388(16):1451-1464. doi: 10.1056/NEJMoa2216480.
10. Moline HL. Early Estimate of Nirsevimab Effectiveness for Prevention of Respiratory Syncytial Virus–Associated Hospitalization Among Infants Entering Their First Respiratory Syncytial Virus Season — New Vaccine Surveillance Network, October 2023–February 2024. MMWR Morb Mortal Wkly Rep. 2024;73. doi: 10.15585/mmwr.mm7309a4.
For children younger than 5 years old, RSV is the main drive — approximately 2,000,000 outpatient/ED visits and about 75,000 hospitalizations annually. RSV disease ranges from upper respiratory tract infections, eg, in older children and healthy adults, to more severe lower tract disease in young children and the elderly. Premature infants and high-risk groups are particularly prone to severe disease.1 Up to 300 pediatric RSV deaths occur yearly. “Normal” RSV seasons start in mid-November, peak in late December-January, and end after April. Note: More drawn out seasons occur in southern latitudes, eg Texas or Florida. But lately RSV seasons have been anything but normal.
2015-2016 to 2022-2023
RSV data from the Pediatric Health Information System (PHIS), collected at over 49 US children’s hospitals during 2015 to early 2023, show how crazy RSV seasons have been lately.2 The involved months, intensity, and duration of four prepandemic seasons were pretty “normal” (Figure 1). The 2019-2020 season started normally, peaked in January 2020, and was slowing as expected by February. But when SARS-Cov-2 restrictions kicked in during mid-March, RSV detections tanked to almost nothing (ditto other respiratory viruses). A near 14-month RSV hiatus meant that the 2020-2021 RSV season never materialized. However, RSV was not done with us in 2021. It rebounded in May with weekly hospitalizations peaking in late July; this “rebound season” lasted 9 months, not dropping to baseline until February 2022 (Figure 1).
I guess we should have expected a post-pandemic “disturbance in the Force,” as Yoda once said; but I sure didn’t see a prolonged summer/fall/early winter RSV season coming. It was like two “normal” seasons mashed up into one late-but-long season. Not to be outdone, the 2022-2023 RSV season started early (September) and hospitalizations skyrocketed to peak in November at over twice the peak number from any year since 2015, overloading hospitals (influenza and SARS-Cov-2 seasons were co-circulating). The season terminated early though (March 2023).
Okay, so RSV seasonality/intensity were weird post pandemic, but was anything else different? Some 2021-2023 data suggest more RSV disease in older children, rather than the usual younger than 18 month-olds going through their first winter.3 More medically attended RSV in older ages (2-4 years of life) may have been due to the pandemic year without RSV circulation distorting herd immunity, ie older children remained RSV naive. Other data suggest the apparent increase was really just more frequent multiplex viral testing in older children triggered by SARS-CoV-2 co-circulation.4 More data are needed to decide.
CDC 2023-2024 RESP-NET data
The 2023-2024 winter surge (Figure 2), as measured by RESP-NET’s cumulative RSV, influenza and SARS-CoV-2 hospitalization rates for 0- to 5-year-olds,5 shows that all three viruses’ seasonal months were normal-ish: late October 2023 start, late December-early January peak, and mid-May 2024 return to baseline. RSV season was approximately 22% less severe by area-under-the-curve calculations compared with 2022-2023, but still worse than prepandemic years.6 
One wonders if the 2022-2023 RSV season might have been worse but for use of the limited supply of nirsevimab.7
Viral Parade
Now we ready ourselves for the 2024-2025 respiratory surge, wondering what nature has in store for us. Will the usual “respiratory virus parade” occur? Will rhinovirus and parainfluenza prevalence bump after a few weeks of schools being in session, adding to the now-usual summer/fall SARS-CoV-2 surge? Note: Twenty-seven states as of Aug. 16 had high SARS-CoV-2 detection in wastewater. Will RSV and influenza start sometime in October/November, peak in January (along with rising SARS-CoV2 activity), followed by a second parainfluenza bump as SARS-CoV-2, influenza, and RSV drop off in April/May? Further, will RSV and influenza seasons be more or less severe than the last 2 years?
Prediction
The overall 2024-2025 respiratory season will be less severe than the past 2 years and hopefully than recent prepandemic years. What is the blueprint for a milder season? First, herd immunity to non-RSV and non-influenza viruses (parainfluenza, rhinovirus, metapneumovirus, adenovirus) in older children should be normalized after 2 years back to usual social activity. So, I expect no mega-seasons from them. The emerging SARS-CoV-2 virus (LB.1) is immunologically close to its recent still-circulating ancestors (KP.2, KP.2.3, KP.3 and KP.3.1.1), so existing SARS-CoV2 herd immunity along with recommended booster vaccine uptake should keep the lid on SARS-CoV2.
Influenza Could Be the Bad News
Which type will dominate? Will a drift/shift occur or vaccine-mismatch reduce vaccine effectiveness? Can we get at least half the population influenza vaccinated, given the vaccine fatigue permeating the US population? The influenza season now underway in the Southern Hemisphere usually helps us predict our season. The Australian May-August 2024 experience (still on an upward trajectory for severity in mid-August) saw no drift/shift or vaccine mismatch. However, this 2024 season has been as severe as 2022 (their worst in a decade). That said, more than 95% has been type A (mostly H1N1 but H3N2 increased in July). So, if our overall 2024-2025 respiratory season is not milder, influenza is the most likely culprit. To reduce chances of influenza being the fly-in-the-ointment, we need to be particularly proactive with seasonal influenza vaccine which is back to the traditional trivalent formulation (one H1N1, one H3N2, and one B type).8 All of this could go out the window if avian influenza becomes more transmissible, but that seems unlikely at present.
Mild RSV Season?
RSV season should be blunted because of the increased use of both the remarkably effective CDC-recommended maternal RSV vaccine9 (one dose during pregnancy weeks 32 through 36, administered September through January) and of nirsevimab (up to 90% reduction in hospitalizations and ED visits).10 (See Figure 3.) 
I also expect residual disease to occur mostly in younger than 18 month-olds (the “normal” aged population experiencing their first winter), who received no passive immunity (mother RSV unvaccinated and child did not receive nirsevimab). Some disease will still occur in high-risk infants/children. However, unlike active vaccination strategies, a competent immune system is not required to benefit from passive antibody, whether transplacental or directly administered.
Deep Thought
What if the traditional RSV seasonal hospitalization surge fails to materialize this season? It could happen. If we could get high acceptance/uptake of maternal vaccine and infant nirsevimab, RSV season could resemble the dramatic drop in rotavirus disease the second year after rotavirus vaccine introduction. We could be asking ourselves — “What happened to RSV?”
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Missouri. He said he had no relevant financial disclosures. Email him at pdnews@mdedge.com.
References
1. CDC. RSV in Infants and Young Children. Respiratory Syncytial Virus Infection (RSV). June 18, 2024. https://www.cdc.gov/rsv/infants-young-children/index.html.
2. Suss RJ and Simões EAF. Respiratory Syncytial Virus Hospital-Based Burden of Disease in Children Younger Than 5 Respiratory Syncytial Virus Hospital-Based Burden of Disease in Children Younger Than 5 Years, 2015-2022. JAMA Netw Open. 2024;7(4):e247125. doi:10.1001/jamanetworkopen.2024.7125.
3. Winthrop ZA et al. Pediatric Respiratory Syncytial Virus Hospitalizations and Respiratory Support After the COVID-19 Pandemic. JAMA Netw Open. 2024;7(6):e2416852. doi:10.1001/jamanetworkopen.2024.16852.
4. Petros BA et al. Increased Pediatric RSV Case Counts Following the Emergence of SARS-CoV-2 Are Attributable to Increased Testing. medRxiv [Preprint]. 2024 Feb 12:2024.02.06.24302387. doi: 10.1101/2024.02.06.24302387.
5. Rates of Laboratory-Confirmed RSV, COVID-19, and Flu Hospitalizations from the RESP-NET Surveillance Systems. Centers for Disease Control and Prevention. https://data.cdc.gov/Public-Health-Surveillance/Rates-of-Laboratory-Confirmed-RSV-COVID-19-and-Flu/kvib-3txy/about_data.
6. CDC. Evaluating the 2023-2024 Respiratory Disease Season Outlook. CFA: Qualitative Assessments. August 14, 2024. https://www.cdc.gov/cfa-qualitative-assessments/php/data-research/2023-2024-season-outlook-retro.html.
7. Health Alert Network (HAN). Limited Availability of Nirsevimab in the United States—Interim CDC Recommendations to Protect Infants from Respiratory Syncytial Virus (RSV) during the 2023–2024 Respiratory Virus Season. October 23, 2023. https://emergency.cdc.gov/han/2023/han00499.asp.
8. CDC. Information for the 2024-2025 Flu Season. Centers for Disease Control and Prevention. March 14, 2024. https://www.cdc.gov/flu/season/faq-flu-season-2024-2025.htm.
9. Kampmann B et al. Bivalent Prefusion F Vaccine in Pregnancy to Prevent RSV Illness in Infants. N Engl J Med. 2023 Apr 20;388(16):1451-1464. doi: 10.1056/NEJMoa2216480.
10. Moline HL. Early Estimate of Nirsevimab Effectiveness for Prevention of Respiratory Syncytial Virus–Associated Hospitalization Among Infants Entering Their First Respiratory Syncytial Virus Season — New Vaccine Surveillance Network, October 2023–February 2024. MMWR Morb Mortal Wkly Rep. 2024;73. doi: 10.15585/mmwr.mm7309a4.
Whooping Cough Likely on Pace for a 5-Year High
Like many diseases, whooping cough reached record low levels during the early days of the COVID pandemic.
More than 10,000 cases of whooping cough have been reported in the United States so far this year, and weekly reports say cases have more than tripled 2023 levels as of June, according to the Centers for Disease Control and Prevention (CDC). In 2023, there were 2815 cases reported during the entire year.
“The number of reported cases this year is close to what was seen at the same time in 2019, prior to the pandemic,” the CDC reported. There were 18,617 cases of whooping cough in 2019.
There were 259 cases reported nationwide for the week ending Aug. 3, with nearly half occurring in the mid-Atlantic region. Public health officials believe the resurgence of whooping cough is likely due to declining vaccination rates, mainly due to the missed vaccines during the height of the COVID pandemic. The diphtheria, tetanus, and pertussis vaccines (DTaP) have been given together since the 1940s, typically during infancy and again during early childhood. In 1941, there were more than 220,000 cases of whooping cough.
Whooping cough is caused by the bacteria Bordetella pertussis. The bacteria attach to tiny, hair-like extensions in the upper respiratory system called cilia, and toxins released by them damage the cilia and cause airways to swell. Early symptoms are similar to the common cold, but the condition eventually leads to coughing fits and a high-pitched “whoop” sound made when inhaling after a fit subsides. Coughing fits can be so severe that people can fracture a rib.
Vaccinated people may get a less severe illness, compared to unvaccinated people, the CDC says. Babies and children are particularly at risk for severe and even potentially deadly complications. About one in three babies under age 1 who get whooping cough will need to be hospitalized, and among those hospitalized babies, 1 in 100 die from complications.
A version of this article appeared on WebMD.com.
Like many diseases, whooping cough reached record low levels during the early days of the COVID pandemic.
More than 10,000 cases of whooping cough have been reported in the United States so far this year, and weekly reports say cases have more than tripled 2023 levels as of June, according to the Centers for Disease Control and Prevention (CDC). In 2023, there were 2815 cases reported during the entire year.
“The number of reported cases this year is close to what was seen at the same time in 2019, prior to the pandemic,” the CDC reported. There were 18,617 cases of whooping cough in 2019.
There were 259 cases reported nationwide for the week ending Aug. 3, with nearly half occurring in the mid-Atlantic region. Public health officials believe the resurgence of whooping cough is likely due to declining vaccination rates, mainly due to the missed vaccines during the height of the COVID pandemic. The diphtheria, tetanus, and pertussis vaccines (DTaP) have been given together since the 1940s, typically during infancy and again during early childhood. In 1941, there were more than 220,000 cases of whooping cough.
Whooping cough is caused by the bacteria Bordetella pertussis. The bacteria attach to tiny, hair-like extensions in the upper respiratory system called cilia, and toxins released by them damage the cilia and cause airways to swell. Early symptoms are similar to the common cold, but the condition eventually leads to coughing fits and a high-pitched “whoop” sound made when inhaling after a fit subsides. Coughing fits can be so severe that people can fracture a rib.
Vaccinated people may get a less severe illness, compared to unvaccinated people, the CDC says. Babies and children are particularly at risk for severe and even potentially deadly complications. About one in three babies under age 1 who get whooping cough will need to be hospitalized, and among those hospitalized babies, 1 in 100 die from complications.
A version of this article appeared on WebMD.com.
Like many diseases, whooping cough reached record low levels during the early days of the COVID pandemic.
More than 10,000 cases of whooping cough have been reported in the United States so far this year, and weekly reports say cases have more than tripled 2023 levels as of June, according to the Centers for Disease Control and Prevention (CDC). In 2023, there were 2815 cases reported during the entire year.
“The number of reported cases this year is close to what was seen at the same time in 2019, prior to the pandemic,” the CDC reported. There were 18,617 cases of whooping cough in 2019.
There were 259 cases reported nationwide for the week ending Aug. 3, with nearly half occurring in the mid-Atlantic region. Public health officials believe the resurgence of whooping cough is likely due to declining vaccination rates, mainly due to the missed vaccines during the height of the COVID pandemic. The diphtheria, tetanus, and pertussis vaccines (DTaP) have been given together since the 1940s, typically during infancy and again during early childhood. In 1941, there were more than 220,000 cases of whooping cough.
Whooping cough is caused by the bacteria Bordetella pertussis. The bacteria attach to tiny, hair-like extensions in the upper respiratory system called cilia, and toxins released by them damage the cilia and cause airways to swell. Early symptoms are similar to the common cold, but the condition eventually leads to coughing fits and a high-pitched “whoop” sound made when inhaling after a fit subsides. Coughing fits can be so severe that people can fracture a rib.
Vaccinated people may get a less severe illness, compared to unvaccinated people, the CDC says. Babies and children are particularly at risk for severe and even potentially deadly complications. About one in three babies under age 1 who get whooping cough will need to be hospitalized, and among those hospitalized babies, 1 in 100 die from complications.
A version of this article appeared on WebMD.com.
Viral Season 2024-2025: Try for An Ounce of Prevention
We are quickly approaching the typical cold and flu season. But can we call anything typical since 2020? Since 2020, there have been different recommendations for prevention, testing, return to work, and treatment since our world was rocked by the pandemic. Now that we are in the “post-pandemic” era, family physicians and other primary care professionals are the front line for discussions on prevention, evaluation, and treatment of the typical upper-respiratory infections, influenza, and COVID-19.
Let’s start with prevention. We have all heard the old adage, an ounce of prevention is worth a pound of cure. In primary care, we need to focus on prevention. Vaccination is often one of our best tools against the myriad of infections we are hoping to help patients prevent during cold and flu season. Most recently, we have fall vaccinations aimed to prevent COVID-19, influenza, and respiratory syncytial virus (RSV).
The number and timing of each of these vaccinations has different recommendations based on a variety of factors including age, pregnancy status, and whether or not the patient is immunocompromised. For the 2024-2025 season, the Centers for Disease Control and Prevention has recommended updated vaccines for both influenza and COVID-19.1
They have also updated the RSV vaccine recommendations to “People 75 or older, or between 60-74 with certain chronic health conditions or living in a nursing home should get one dose of the RSV vaccine to provide an extra layer of protection.”2
In addition to vaccines as prevention, there is also hygiene, staying home when sick and away from others who are sick, following guidelines for where and when to wear a face mask, and the general tools of eating well, and getting sufficient sleep and exercise to help maintain the healthiest immune system.
Despite the best of intentions, there will still be many who experience viral infections in this upcoming season. The CDC is currently recommending persons to stay away from others for at least 24 hours after their symptoms improve and they are fever-free without antipyretics. In addition to isolation while sick, general symptom management is something that we can recommend for all of these illnesses.
There is more to consider, though, as our patients face these illnesses. The first question is how to determine the diagnosis — and if that diagnosis is even necessary. Unfortunately, many of these viral illnesses can look the same. They can all cause fevers, chills, and other upper respiratory symptoms. They are all fairly contagious. All of these viruses can cause serious illness associated with additional complications. It is not truly possible to determine which virus someone has by symptoms alone, our patients can have multiple viruses at the same time and diagnosis of one does not preclude having another.3
Instead, we truly do need a test for diagnosis. In-office testing is available for RSV, influenza, and COVID-19. Additionally, despite not being as freely available as they were during the pandemic, patients are able to do home COVID tests and then call in with their results. At the time of writing this, at-home rapid influenza tests have also been approved by the FDA but are not yet readily available to the public. These tests are important for determining if the patient is eligible for treatment. Both influenza and COVID-19 have antiviral treatments available to help decrease the severity of the illness and potentially the length of illness and time contagious. According to the CDC, both treatments are underutilized.
This could be because of a lack of testing and diagnosis. It may also be because of a lack of familiarity with the available treatments.4,5
Influenza treatment is recommended as soon as possible for those with suspected or confirmed diagnosis, immediately for anyone hospitalized, anyone with severe, complicated, or progressing illness, and for those at high risk of severe illness including but not limited to those under 2 years old, those over 65, those who are pregnant, and those with many chronic conditions.
Treatment can also be used for those who are not high risk when diagnosed within 48 hours. In the United States, four antivirals are recommended to treat influenza: oseltamivir phosphate, zanamivir, peramivir, and baloxavir marboxil. For COVID-19, treatments are also available for mild or moderate disease in those at risk for severe disease. Both remdesivir and nimatrelvir with ritonavir are treatment options that can be used for COVID-19 infection. Unfortunately, no specific antiviral is available for the other viral illnesses we see often during this season.
In primary care, we have some important roles to play. We need to continue to discuss all methods of prevention. Not only do vaccine recommendations change at least annually, our patients’ situations change and we have to reassess them. Additionally, people often need to hear things more than once before committing — so it never hurts to continue having those conversations. Combining the conversation about vaccines with other prevention measures is also important so that it does not seem like we are only recommending one thing. We should also start talking about treatment options before our patients are sick. We can communicate what is available as long as they let us know they are sick early. We can also be there to help our patients determine when they are at risk for severe illness and when they should consider a higher level of care.
The availability of home testing gives us the opportunity to provide these treatments via telehealth and even potentially in times when these illnesses are everywhere — with standing orders with our clinical teams. Although it is a busy time for us in the clinic, “cold and flu” season is definitely one of those times when our primary care relationship can truly help our patients.
References
1. CDC Recommends Updated 2024-2025 COVID-19 and Flu Vaccines for Fall/Winter Virus Season. https://www.cdc.gov/media/releases/2024/s-t0627-vaccine-recommendations.html. Accessed August 8, 2024. Source: Centers for Disease Control and Prevention.
2. CDC Updates RSV Vaccination Recommendation for Adults. https://www.cdc.gov/media/releases/2024/s-0626-vaccination-adults.html. Accessed August 8, 2024. Source: Centers for Disease Control and Prevention.
3. Similarities and Differences between Flu and COVID-19. https://www.cdc.gov/flu/symptoms/flu-vs-covid19.htm. Accessed August 8, 2024. Source: Centers for Disease Control and Prevention, National Center for Immunization and Respiratory Diseases.
4. Respiratory Virus Guidance. https://www.cdc.gov/respiratory-viruses/guidance/index.html. Accessed August 9, 2024. Source: National Center for Immunization and Respiratory Diseases.
5. Provider Toolkit: Preparing Patients for the Fall and Winter Virus Season. https://www.cdc.gov/respiratory-viruses/hcp/tools-resources/index.html. Accessed August 9, 2024. Source: Centers for Disease Control and Prevention.
We are quickly approaching the typical cold and flu season. But can we call anything typical since 2020? Since 2020, there have been different recommendations for prevention, testing, return to work, and treatment since our world was rocked by the pandemic. Now that we are in the “post-pandemic” era, family physicians and other primary care professionals are the front line for discussions on prevention, evaluation, and treatment of the typical upper-respiratory infections, influenza, and COVID-19.
Let’s start with prevention. We have all heard the old adage, an ounce of prevention is worth a pound of cure. In primary care, we need to focus on prevention. Vaccination is often one of our best tools against the myriad of infections we are hoping to help patients prevent during cold and flu season. Most recently, we have fall vaccinations aimed to prevent COVID-19, influenza, and respiratory syncytial virus (RSV).
The number and timing of each of these vaccinations has different recommendations based on a variety of factors including age, pregnancy status, and whether or not the patient is immunocompromised. For the 2024-2025 season, the Centers for Disease Control and Prevention has recommended updated vaccines for both influenza and COVID-19.1
They have also updated the RSV vaccine recommendations to “People 75 or older, or between 60-74 with certain chronic health conditions or living in a nursing home should get one dose of the RSV vaccine to provide an extra layer of protection.”2
In addition to vaccines as prevention, there is also hygiene, staying home when sick and away from others who are sick, following guidelines for where and when to wear a face mask, and the general tools of eating well, and getting sufficient sleep and exercise to help maintain the healthiest immune system.
Despite the best of intentions, there will still be many who experience viral infections in this upcoming season. The CDC is currently recommending persons to stay away from others for at least 24 hours after their symptoms improve and they are fever-free without antipyretics. In addition to isolation while sick, general symptom management is something that we can recommend for all of these illnesses.
There is more to consider, though, as our patients face these illnesses. The first question is how to determine the diagnosis — and if that diagnosis is even necessary. Unfortunately, many of these viral illnesses can look the same. They can all cause fevers, chills, and other upper respiratory symptoms. They are all fairly contagious. All of these viruses can cause serious illness associated with additional complications. It is not truly possible to determine which virus someone has by symptoms alone, our patients can have multiple viruses at the same time and diagnosis of one does not preclude having another.3
Instead, we truly do need a test for diagnosis. In-office testing is available for RSV, influenza, and COVID-19. Additionally, despite not being as freely available as they were during the pandemic, patients are able to do home COVID tests and then call in with their results. At the time of writing this, at-home rapid influenza tests have also been approved by the FDA but are not yet readily available to the public. These tests are important for determining if the patient is eligible for treatment. Both influenza and COVID-19 have antiviral treatments available to help decrease the severity of the illness and potentially the length of illness and time contagious. According to the CDC, both treatments are underutilized.
This could be because of a lack of testing and diagnosis. It may also be because of a lack of familiarity with the available treatments.4,5
Influenza treatment is recommended as soon as possible for those with suspected or confirmed diagnosis, immediately for anyone hospitalized, anyone with severe, complicated, or progressing illness, and for those at high risk of severe illness including but not limited to those under 2 years old, those over 65, those who are pregnant, and those with many chronic conditions.
Treatment can also be used for those who are not high risk when diagnosed within 48 hours. In the United States, four antivirals are recommended to treat influenza: oseltamivir phosphate, zanamivir, peramivir, and baloxavir marboxil. For COVID-19, treatments are also available for mild or moderate disease in those at risk for severe disease. Both remdesivir and nimatrelvir with ritonavir are treatment options that can be used for COVID-19 infection. Unfortunately, no specific antiviral is available for the other viral illnesses we see often during this season.
In primary care, we have some important roles to play. We need to continue to discuss all methods of prevention. Not only do vaccine recommendations change at least annually, our patients’ situations change and we have to reassess them. Additionally, people often need to hear things more than once before committing — so it never hurts to continue having those conversations. Combining the conversation about vaccines with other prevention measures is also important so that it does not seem like we are only recommending one thing. We should also start talking about treatment options before our patients are sick. We can communicate what is available as long as they let us know they are sick early. We can also be there to help our patients determine when they are at risk for severe illness and when they should consider a higher level of care.
The availability of home testing gives us the opportunity to provide these treatments via telehealth and even potentially in times when these illnesses are everywhere — with standing orders with our clinical teams. Although it is a busy time for us in the clinic, “cold and flu” season is definitely one of those times when our primary care relationship can truly help our patients.
References
1. CDC Recommends Updated 2024-2025 COVID-19 and Flu Vaccines for Fall/Winter Virus Season. https://www.cdc.gov/media/releases/2024/s-t0627-vaccine-recommendations.html. Accessed August 8, 2024. Source: Centers for Disease Control and Prevention.
2. CDC Updates RSV Vaccination Recommendation for Adults. https://www.cdc.gov/media/releases/2024/s-0626-vaccination-adults.html. Accessed August 8, 2024. Source: Centers for Disease Control and Prevention.
3. Similarities and Differences between Flu and COVID-19. https://www.cdc.gov/flu/symptoms/flu-vs-covid19.htm. Accessed August 8, 2024. Source: Centers for Disease Control and Prevention, National Center for Immunization and Respiratory Diseases.
4. Respiratory Virus Guidance. https://www.cdc.gov/respiratory-viruses/guidance/index.html. Accessed August 9, 2024. Source: National Center for Immunization and Respiratory Diseases.
5. Provider Toolkit: Preparing Patients for the Fall and Winter Virus Season. https://www.cdc.gov/respiratory-viruses/hcp/tools-resources/index.html. Accessed August 9, 2024. Source: Centers for Disease Control and Prevention.
We are quickly approaching the typical cold and flu season. But can we call anything typical since 2020? Since 2020, there have been different recommendations for prevention, testing, return to work, and treatment since our world was rocked by the pandemic. Now that we are in the “post-pandemic” era, family physicians and other primary care professionals are the front line for discussions on prevention, evaluation, and treatment of the typical upper-respiratory infections, influenza, and COVID-19.
Let’s start with prevention. We have all heard the old adage, an ounce of prevention is worth a pound of cure. In primary care, we need to focus on prevention. Vaccination is often one of our best tools against the myriad of infections we are hoping to help patients prevent during cold and flu season. Most recently, we have fall vaccinations aimed to prevent COVID-19, influenza, and respiratory syncytial virus (RSV).
The number and timing of each of these vaccinations has different recommendations based on a variety of factors including age, pregnancy status, and whether or not the patient is immunocompromised. For the 2024-2025 season, the Centers for Disease Control and Prevention has recommended updated vaccines for both influenza and COVID-19.1
They have also updated the RSV vaccine recommendations to “People 75 or older, or between 60-74 with certain chronic health conditions or living in a nursing home should get one dose of the RSV vaccine to provide an extra layer of protection.”2
In addition to vaccines as prevention, there is also hygiene, staying home when sick and away from others who are sick, following guidelines for where and when to wear a face mask, and the general tools of eating well, and getting sufficient sleep and exercise to help maintain the healthiest immune system.
Despite the best of intentions, there will still be many who experience viral infections in this upcoming season. The CDC is currently recommending persons to stay away from others for at least 24 hours after their symptoms improve and they are fever-free without antipyretics. In addition to isolation while sick, general symptom management is something that we can recommend for all of these illnesses.
There is more to consider, though, as our patients face these illnesses. The first question is how to determine the diagnosis — and if that diagnosis is even necessary. Unfortunately, many of these viral illnesses can look the same. They can all cause fevers, chills, and other upper respiratory symptoms. They are all fairly contagious. All of these viruses can cause serious illness associated with additional complications. It is not truly possible to determine which virus someone has by symptoms alone, our patients can have multiple viruses at the same time and diagnosis of one does not preclude having another.3
Instead, we truly do need a test for diagnosis. In-office testing is available for RSV, influenza, and COVID-19. Additionally, despite not being as freely available as they were during the pandemic, patients are able to do home COVID tests and then call in with their results. At the time of writing this, at-home rapid influenza tests have also been approved by the FDA but are not yet readily available to the public. These tests are important for determining if the patient is eligible for treatment. Both influenza and COVID-19 have antiviral treatments available to help decrease the severity of the illness and potentially the length of illness and time contagious. According to the CDC, both treatments are underutilized.
This could be because of a lack of testing and diagnosis. It may also be because of a lack of familiarity with the available treatments.4,5
Influenza treatment is recommended as soon as possible for those with suspected or confirmed diagnosis, immediately for anyone hospitalized, anyone with severe, complicated, or progressing illness, and for those at high risk of severe illness including but not limited to those under 2 years old, those over 65, those who are pregnant, and those with many chronic conditions.
Treatment can also be used for those who are not high risk when diagnosed within 48 hours. In the United States, four antivirals are recommended to treat influenza: oseltamivir phosphate, zanamivir, peramivir, and baloxavir marboxil. For COVID-19, treatments are also available for mild or moderate disease in those at risk for severe disease. Both remdesivir and nimatrelvir with ritonavir are treatment options that can be used for COVID-19 infection. Unfortunately, no specific antiviral is available for the other viral illnesses we see often during this season.
In primary care, we have some important roles to play. We need to continue to discuss all methods of prevention. Not only do vaccine recommendations change at least annually, our patients’ situations change and we have to reassess them. Additionally, people often need to hear things more than once before committing — so it never hurts to continue having those conversations. Combining the conversation about vaccines with other prevention measures is also important so that it does not seem like we are only recommending one thing. We should also start talking about treatment options before our patients are sick. We can communicate what is available as long as they let us know they are sick early. We can also be there to help our patients determine when they are at risk for severe illness and when they should consider a higher level of care.
The availability of home testing gives us the opportunity to provide these treatments via telehealth and even potentially in times when these illnesses are everywhere — with standing orders with our clinical teams. Although it is a busy time for us in the clinic, “cold and flu” season is definitely one of those times when our primary care relationship can truly help our patients.
References
1. CDC Recommends Updated 2024-2025 COVID-19 and Flu Vaccines for Fall/Winter Virus Season. https://www.cdc.gov/media/releases/2024/s-t0627-vaccine-recommendations.html. Accessed August 8, 2024. Source: Centers for Disease Control and Prevention.
2. CDC Updates RSV Vaccination Recommendation for Adults. https://www.cdc.gov/media/releases/2024/s-0626-vaccination-adults.html. Accessed August 8, 2024. Source: Centers for Disease Control and Prevention.
3. Similarities and Differences between Flu and COVID-19. https://www.cdc.gov/flu/symptoms/flu-vs-covid19.htm. Accessed August 8, 2024. Source: Centers for Disease Control and Prevention, National Center for Immunization and Respiratory Diseases.
4. Respiratory Virus Guidance. https://www.cdc.gov/respiratory-viruses/guidance/index.html. Accessed August 9, 2024. Source: National Center for Immunization and Respiratory Diseases.
5. Provider Toolkit: Preparing Patients for the Fall and Winter Virus Season. https://www.cdc.gov/respiratory-viruses/hcp/tools-resources/index.html. Accessed August 9, 2024. Source: Centers for Disease Control and Prevention.
Study Links Newer Shingles Vaccine to Delayed Dementia Diagnosis
The study builds on previous observations of a reduction in dementia risk with the older live shingles vaccine and reports a delay in dementia diagnosis of 164 days with the newer recombinant version, compared with the live vaccine.
“Given the prevalence of dementia, a delay of 164 days in diagnosis would not be a trivial effect at the public health level. It’s a big enough effect that if there is a causality it feels meaningful,” said senior author Paul Harrison, DM, FRCPsych, professor of psychiatry at the University of Oxford, Oxford, England.
But Dr. Harrison stressed that the study had not proven that the shingles vaccine reduced dementia risk.
“The design of the study allows us to do away with many of the confounding effects we usually see in observational studies, but this is still an observational study, and as such it cannot prove a definite causal effect,” he said.
The study was published online on July 25 in Nature Medicine.
‘Natural Experiment’
Given the risk for deleterious consequences of shingles, vaccination is now recommended for older adults in many countries. The previously used live shingles vaccine (Zostavax) is being replaced in most countries with the new recombinant shingles vaccine (Shingrix), which is more effective at preventing shingles infection.
The current study made use of a “natural experiment” in the United States, which switched over from use of the live vaccine to the recombinant vaccine in October 2017.
Researchers used electronic heath records to compare the incidence of a dementia diagnosis in individuals who received the live shingles vaccine prior to October 2017 with those who received the recombinant version after the United States made the switch.
They also used propensity score matching to further control for confounding factors, comparing 103,837 individuals who received a first dose of the live shingles vaccine between October 2014 and September 2017 with the same number of matched people who received the recombinant vaccine between November 2017 and October 2020.
Results showed that within the 6 years after vaccination, the recombinant vaccine was associated with a delay in the diagnosis of dementia, compared with the live vaccine. Specifically, receiving the recombinant vaccine was associated with a 17% increase in diagnosis-free time, translating to 164 additional days lived without a diagnosis of dementia in those subsequently affected.
As an additional control, the researchers also found significantly lower risks for dementia in individuals receiving the new recombinant shingles vaccine vs two other vaccines commonly used in older people: influenza and tetanus/diphtheria/pertussis vaccines, with increases in diagnosis-free time of 14%-27%.
Reduced Risk or Delayed Diagnosis?
Speaking at a Science Media Centre press conference on the study, lead author Maxime Taquet, PhD, FRCPsych, clinical lecturer in psychiatry at the University of Oxford, noted that the total number of dementia cases were similar in the two shingles vaccine groups by the end of the 6-year follow-up period but there was a difference in the time at which they received a diagnosis of dementia.
“The study suggests that rather than actually reducing dementia risk, the recombinant vaccine delays the onset of dementia compared to the live vaccine in patients who go on to develop the condition,” he explained.
But when comparing the recombinant vaccine with the influenza and tetanus/diphtheria/pertussis vaccines there was a clear reduction in dementia risk itself, Dr. Taquet reported.
“It might well be that the live vaccine has a potential effect on the risk of dementia itself and therefore the recombinant vaccine only shows a delay in dementia compared to the live vaccine, but both of them might decrease the overall risk of dementia,” he suggested.
But the researchers cautioned that this study could not prove causality.
“While the two groups were very carefully matched in terms of factors that might influence the development of dementia, we still have to be cautious before assuming that the vaccine is indeed causally reducing the risk of onset of dementia,” Dr. Harrison warned.
The researchers say the results would need to be confirmed in a randomized trial, which may have to be conducted in a slightly younger age group, as currently shingles vaccine is recommended for all older individuals in the United Kingdom.
Vaccine recommendations vary from country to country, Dr. Harrison added. In the United States, the Centers for Disease Control and Prevention recommends the recombinant shingles vaccine for all adults aged 50 years or older.
In the meantime, it would be interesting to see whether further observational studies in other countries find similar results as this US study, Dr. Harrison said.
Mechanism Uncertain
Speculating on a possible mechanism behind the findings, Dr. Harrison suggested two plausible explanations.
“First, it is thought that the herpes virus could be one of many factors that could promote dementia, so a vaccine that stops reactivation of this virus might therefore be delaying that process,” he noted.
The other possibility is that adjuvants included in the recombinant vaccine to stimulate the immune system might have played a role.
“We don’t have any data on the mechanism, and thus study did not address that, so further studies are needed to look into this,” Dr. Harrison said.
Stronger Effect in Women
Another intriguing finding is that the association with the recombinant vaccine and delayed dementia diagnosis seemed to be stronger in women vs men.
In the original study of the live shingles vaccine, a protective effect against dementia was shown only in women.
In the current study, the delay in dementia diagnosis was seen in both sexes but was stronger in women, showing a 22% increased time without dementia in women versus a 13% increased time in men with the recombinant versus the live vaccine.
As expected, the recombinant vaccine was associated with a lower risk for shingles disease vs the live vaccine (2.5% versus 3.5%), but women did not have a better response than men did in this respect.
“The better protection against shingles with the recombinant vaccine was similar in men and women, an observation that might be one reason to question the possible mechanism behind the dementia effect being better suppression of the herpes zoster virus by the recombinant vaccine,” Dr. Harrison commented.
Though these findings are not likely to lead to any immediate changes in policy regarding the shingles vaccine, Dr. Harrison said it would be interesting to see whether uptake of the vaccine increased after this study.
He estimated that, currently in the United Kingdom, about 60% of older adults choose to have the shingles vaccine. A 2020 study in the United States found that only about one-third of US adults over 60 had received the vaccine.
“It will be interesting to see if that figure increases after these data are publicized, but I am not recommending that people have the vaccine specifically to lower their risk of dementia because of the caveats about the study that we have discussed,” he commented.
Outside Experts Positive
Outside experts, providing comment to the Science Media Centre, welcomed the new research.
“ The study is very well-conducted and adds to previous data indicating that vaccination against shingles is associated with lower dementia risk. More research is needed in future to determine why this vaccine is associated with lower dementia risk,” said Tara Spires-Jones, FMedSci, president of the British Neuroscience Association.
The high number of patients in the study and the adjustments for potential confounders are also strong points, noted Andrew Doig, PhD, professor of biochemistry, University of Manchester, Manchester, England.
“This is a significant result, comparable in effectiveness to the recent antibody drugs for Alzheimer’s disease,” Dr. Doig said. “Administering the recombinant shingles vaccine could well be a simple and cheap way to lower the risk of Alzheimer’s disease.”
Dr. Doig noted that a link between herpes zoster infection and the onset of dementia has been suspected for some time, and a trial of the antiviral drug valacyclovir against Alzheimer’s disease is currently underway.
In regard to the shingles vaccine, he said a placebo-controlled trial would be needed to prove causality.
“We also need to see how many years the effect might last and whether we should vaccinate people at a younger age. We know that the path to Alzheimer’s can start decades before any symptoms are apparent, so the vaccine might be even more effective if given to people in their 40s or 50s,” he said.
Dr. Harrison and Dr. Taquet reported no disclosures. Dr. Doig is a founder, director, and consultant for PharmaKure, which works on Alzheimer’s drugs and diagnostics. Other commentators declared no disclosures.
A version of this article first appeared on Medscape.com.
The study builds on previous observations of a reduction in dementia risk with the older live shingles vaccine and reports a delay in dementia diagnosis of 164 days with the newer recombinant version, compared with the live vaccine.
“Given the prevalence of dementia, a delay of 164 days in diagnosis would not be a trivial effect at the public health level. It’s a big enough effect that if there is a causality it feels meaningful,” said senior author Paul Harrison, DM, FRCPsych, professor of psychiatry at the University of Oxford, Oxford, England.
But Dr. Harrison stressed that the study had not proven that the shingles vaccine reduced dementia risk.
“The design of the study allows us to do away with many of the confounding effects we usually see in observational studies, but this is still an observational study, and as such it cannot prove a definite causal effect,” he said.
The study was published online on July 25 in Nature Medicine.
‘Natural Experiment’
Given the risk for deleterious consequences of shingles, vaccination is now recommended for older adults in many countries. The previously used live shingles vaccine (Zostavax) is being replaced in most countries with the new recombinant shingles vaccine (Shingrix), which is more effective at preventing shingles infection.
The current study made use of a “natural experiment” in the United States, which switched over from use of the live vaccine to the recombinant vaccine in October 2017.
Researchers used electronic heath records to compare the incidence of a dementia diagnosis in individuals who received the live shingles vaccine prior to October 2017 with those who received the recombinant version after the United States made the switch.
They also used propensity score matching to further control for confounding factors, comparing 103,837 individuals who received a first dose of the live shingles vaccine between October 2014 and September 2017 with the same number of matched people who received the recombinant vaccine between November 2017 and October 2020.
Results showed that within the 6 years after vaccination, the recombinant vaccine was associated with a delay in the diagnosis of dementia, compared with the live vaccine. Specifically, receiving the recombinant vaccine was associated with a 17% increase in diagnosis-free time, translating to 164 additional days lived without a diagnosis of dementia in those subsequently affected.
As an additional control, the researchers also found significantly lower risks for dementia in individuals receiving the new recombinant shingles vaccine vs two other vaccines commonly used in older people: influenza and tetanus/diphtheria/pertussis vaccines, with increases in diagnosis-free time of 14%-27%.
Reduced Risk or Delayed Diagnosis?
Speaking at a Science Media Centre press conference on the study, lead author Maxime Taquet, PhD, FRCPsych, clinical lecturer in psychiatry at the University of Oxford, noted that the total number of dementia cases were similar in the two shingles vaccine groups by the end of the 6-year follow-up period but there was a difference in the time at which they received a diagnosis of dementia.
“The study suggests that rather than actually reducing dementia risk, the recombinant vaccine delays the onset of dementia compared to the live vaccine in patients who go on to develop the condition,” he explained.
But when comparing the recombinant vaccine with the influenza and tetanus/diphtheria/pertussis vaccines there was a clear reduction in dementia risk itself, Dr. Taquet reported.
“It might well be that the live vaccine has a potential effect on the risk of dementia itself and therefore the recombinant vaccine only shows a delay in dementia compared to the live vaccine, but both of them might decrease the overall risk of dementia,” he suggested.
But the researchers cautioned that this study could not prove causality.
“While the two groups were very carefully matched in terms of factors that might influence the development of dementia, we still have to be cautious before assuming that the vaccine is indeed causally reducing the risk of onset of dementia,” Dr. Harrison warned.
The researchers say the results would need to be confirmed in a randomized trial, which may have to be conducted in a slightly younger age group, as currently shingles vaccine is recommended for all older individuals in the United Kingdom.
Vaccine recommendations vary from country to country, Dr. Harrison added. In the United States, the Centers for Disease Control and Prevention recommends the recombinant shingles vaccine for all adults aged 50 years or older.
In the meantime, it would be interesting to see whether further observational studies in other countries find similar results as this US study, Dr. Harrison said.
Mechanism Uncertain
Speculating on a possible mechanism behind the findings, Dr. Harrison suggested two plausible explanations.
“First, it is thought that the herpes virus could be one of many factors that could promote dementia, so a vaccine that stops reactivation of this virus might therefore be delaying that process,” he noted.
The other possibility is that adjuvants included in the recombinant vaccine to stimulate the immune system might have played a role.
“We don’t have any data on the mechanism, and thus study did not address that, so further studies are needed to look into this,” Dr. Harrison said.
Stronger Effect in Women
Another intriguing finding is that the association with the recombinant vaccine and delayed dementia diagnosis seemed to be stronger in women vs men.
In the original study of the live shingles vaccine, a protective effect against dementia was shown only in women.
In the current study, the delay in dementia diagnosis was seen in both sexes but was stronger in women, showing a 22% increased time without dementia in women versus a 13% increased time in men with the recombinant versus the live vaccine.
As expected, the recombinant vaccine was associated with a lower risk for shingles disease vs the live vaccine (2.5% versus 3.5%), but women did not have a better response than men did in this respect.
“The better protection against shingles with the recombinant vaccine was similar in men and women, an observation that might be one reason to question the possible mechanism behind the dementia effect being better suppression of the herpes zoster virus by the recombinant vaccine,” Dr. Harrison commented.
Though these findings are not likely to lead to any immediate changes in policy regarding the shingles vaccine, Dr. Harrison said it would be interesting to see whether uptake of the vaccine increased after this study.
He estimated that, currently in the United Kingdom, about 60% of older adults choose to have the shingles vaccine. A 2020 study in the United States found that only about one-third of US adults over 60 had received the vaccine.
“It will be interesting to see if that figure increases after these data are publicized, but I am not recommending that people have the vaccine specifically to lower their risk of dementia because of the caveats about the study that we have discussed,” he commented.
Outside Experts Positive
Outside experts, providing comment to the Science Media Centre, welcomed the new research.
“ The study is very well-conducted and adds to previous data indicating that vaccination against shingles is associated with lower dementia risk. More research is needed in future to determine why this vaccine is associated with lower dementia risk,” said Tara Spires-Jones, FMedSci, president of the British Neuroscience Association.
The high number of patients in the study and the adjustments for potential confounders are also strong points, noted Andrew Doig, PhD, professor of biochemistry, University of Manchester, Manchester, England.
“This is a significant result, comparable in effectiveness to the recent antibody drugs for Alzheimer’s disease,” Dr. Doig said. “Administering the recombinant shingles vaccine could well be a simple and cheap way to lower the risk of Alzheimer’s disease.”
Dr. Doig noted that a link between herpes zoster infection and the onset of dementia has been suspected for some time, and a trial of the antiviral drug valacyclovir against Alzheimer’s disease is currently underway.
In regard to the shingles vaccine, he said a placebo-controlled trial would be needed to prove causality.
“We also need to see how many years the effect might last and whether we should vaccinate people at a younger age. We know that the path to Alzheimer’s can start decades before any symptoms are apparent, so the vaccine might be even more effective if given to people in their 40s or 50s,” he said.
Dr. Harrison and Dr. Taquet reported no disclosures. Dr. Doig is a founder, director, and consultant for PharmaKure, which works on Alzheimer’s drugs and diagnostics. Other commentators declared no disclosures.
A version of this article first appeared on Medscape.com.
The study builds on previous observations of a reduction in dementia risk with the older live shingles vaccine and reports a delay in dementia diagnosis of 164 days with the newer recombinant version, compared with the live vaccine.
“Given the prevalence of dementia, a delay of 164 days in diagnosis would not be a trivial effect at the public health level. It’s a big enough effect that if there is a causality it feels meaningful,” said senior author Paul Harrison, DM, FRCPsych, professor of psychiatry at the University of Oxford, Oxford, England.
But Dr. Harrison stressed that the study had not proven that the shingles vaccine reduced dementia risk.
“The design of the study allows us to do away with many of the confounding effects we usually see in observational studies, but this is still an observational study, and as such it cannot prove a definite causal effect,” he said.
The study was published online on July 25 in Nature Medicine.
‘Natural Experiment’
Given the risk for deleterious consequences of shingles, vaccination is now recommended for older adults in many countries. The previously used live shingles vaccine (Zostavax) is being replaced in most countries with the new recombinant shingles vaccine (Shingrix), which is more effective at preventing shingles infection.
The current study made use of a “natural experiment” in the United States, which switched over from use of the live vaccine to the recombinant vaccine in October 2017.
Researchers used electronic heath records to compare the incidence of a dementia diagnosis in individuals who received the live shingles vaccine prior to October 2017 with those who received the recombinant version after the United States made the switch.
They also used propensity score matching to further control for confounding factors, comparing 103,837 individuals who received a first dose of the live shingles vaccine between October 2014 and September 2017 with the same number of matched people who received the recombinant vaccine between November 2017 and October 2020.
Results showed that within the 6 years after vaccination, the recombinant vaccine was associated with a delay in the diagnosis of dementia, compared with the live vaccine. Specifically, receiving the recombinant vaccine was associated with a 17% increase in diagnosis-free time, translating to 164 additional days lived without a diagnosis of dementia in those subsequently affected.
As an additional control, the researchers also found significantly lower risks for dementia in individuals receiving the new recombinant shingles vaccine vs two other vaccines commonly used in older people: influenza and tetanus/diphtheria/pertussis vaccines, with increases in diagnosis-free time of 14%-27%.
Reduced Risk or Delayed Diagnosis?
Speaking at a Science Media Centre press conference on the study, lead author Maxime Taquet, PhD, FRCPsych, clinical lecturer in psychiatry at the University of Oxford, noted that the total number of dementia cases were similar in the two shingles vaccine groups by the end of the 6-year follow-up period but there was a difference in the time at which they received a diagnosis of dementia.
“The study suggests that rather than actually reducing dementia risk, the recombinant vaccine delays the onset of dementia compared to the live vaccine in patients who go on to develop the condition,” he explained.
But when comparing the recombinant vaccine with the influenza and tetanus/diphtheria/pertussis vaccines there was a clear reduction in dementia risk itself, Dr. Taquet reported.
“It might well be that the live vaccine has a potential effect on the risk of dementia itself and therefore the recombinant vaccine only shows a delay in dementia compared to the live vaccine, but both of them might decrease the overall risk of dementia,” he suggested.
But the researchers cautioned that this study could not prove causality.
“While the two groups were very carefully matched in terms of factors that might influence the development of dementia, we still have to be cautious before assuming that the vaccine is indeed causally reducing the risk of onset of dementia,” Dr. Harrison warned.
The researchers say the results would need to be confirmed in a randomized trial, which may have to be conducted in a slightly younger age group, as currently shingles vaccine is recommended for all older individuals in the United Kingdom.
Vaccine recommendations vary from country to country, Dr. Harrison added. In the United States, the Centers for Disease Control and Prevention recommends the recombinant shingles vaccine for all adults aged 50 years or older.
In the meantime, it would be interesting to see whether further observational studies in other countries find similar results as this US study, Dr. Harrison said.
Mechanism Uncertain
Speculating on a possible mechanism behind the findings, Dr. Harrison suggested two plausible explanations.
“First, it is thought that the herpes virus could be one of many factors that could promote dementia, so a vaccine that stops reactivation of this virus might therefore be delaying that process,” he noted.
The other possibility is that adjuvants included in the recombinant vaccine to stimulate the immune system might have played a role.
“We don’t have any data on the mechanism, and thus study did not address that, so further studies are needed to look into this,” Dr. Harrison said.
Stronger Effect in Women
Another intriguing finding is that the association with the recombinant vaccine and delayed dementia diagnosis seemed to be stronger in women vs men.
In the original study of the live shingles vaccine, a protective effect against dementia was shown only in women.
In the current study, the delay in dementia diagnosis was seen in both sexes but was stronger in women, showing a 22% increased time without dementia in women versus a 13% increased time in men with the recombinant versus the live vaccine.
As expected, the recombinant vaccine was associated with a lower risk for shingles disease vs the live vaccine (2.5% versus 3.5%), but women did not have a better response than men did in this respect.
“The better protection against shingles with the recombinant vaccine was similar in men and women, an observation that might be one reason to question the possible mechanism behind the dementia effect being better suppression of the herpes zoster virus by the recombinant vaccine,” Dr. Harrison commented.
Though these findings are not likely to lead to any immediate changes in policy regarding the shingles vaccine, Dr. Harrison said it would be interesting to see whether uptake of the vaccine increased after this study.
He estimated that, currently in the United Kingdom, about 60% of older adults choose to have the shingles vaccine. A 2020 study in the United States found that only about one-third of US adults over 60 had received the vaccine.
“It will be interesting to see if that figure increases after these data are publicized, but I am not recommending that people have the vaccine specifically to lower their risk of dementia because of the caveats about the study that we have discussed,” he commented.
Outside Experts Positive
Outside experts, providing comment to the Science Media Centre, welcomed the new research.
“ The study is very well-conducted and adds to previous data indicating that vaccination against shingles is associated with lower dementia risk. More research is needed in future to determine why this vaccine is associated with lower dementia risk,” said Tara Spires-Jones, FMedSci, president of the British Neuroscience Association.
The high number of patients in the study and the adjustments for potential confounders are also strong points, noted Andrew Doig, PhD, professor of biochemistry, University of Manchester, Manchester, England.
“This is a significant result, comparable in effectiveness to the recent antibody drugs for Alzheimer’s disease,” Dr. Doig said. “Administering the recombinant shingles vaccine could well be a simple and cheap way to lower the risk of Alzheimer’s disease.”
Dr. Doig noted that a link between herpes zoster infection and the onset of dementia has been suspected for some time, and a trial of the antiviral drug valacyclovir against Alzheimer’s disease is currently underway.
In regard to the shingles vaccine, he said a placebo-controlled trial would be needed to prove causality.
“We also need to see how many years the effect might last and whether we should vaccinate people at a younger age. We know that the path to Alzheimer’s can start decades before any symptoms are apparent, so the vaccine might be even more effective if given to people in their 40s or 50s,” he said.
Dr. Harrison and Dr. Taquet reported no disclosures. Dr. Doig is a founder, director, and consultant for PharmaKure, which works on Alzheimer’s drugs and diagnostics. Other commentators declared no disclosures.
A version of this article first appeared on Medscape.com.
FROM NATURE MEDICINE
New Mid-Year Vaccine Recommendations From ACIP
This transcript has been edited for clarity.
ACIP, the CDC’s Advisory Committee on Immunization Practices, met for 3 days in June. New vaccines and new recommendations for respiratory syncytial virus (RSV), flu, COVID, and a new pneumococcal vaccine were revealed.
RSV Protection
We’ll begin with RSV vaccines for adults aged 60 or older. For this group, shared clinical decision-making is out; it no longer applies. New, more specific recommendations from ACIP for RSV vaccines are both age based and risk based. The age-based recommendation applies to those aged 75 or older, who should receive a single RSV vaccine dose. If they have already received a dose under the old recommendation, they don’t need another one, at least for now.
The risk-based recommendation applies to adults from age 60 up to 75, but only for those with risk factors for severe RSV. These risk factors include lung disease, heart disease, immunocompromise, diabetes, obesity with a BMI of 40 or more, neurologic conditions, neuromuscular conditions, chronic kidney disease, liver disorders, hematologic disorders, frailty, and living in a nursing home or other long-term care facility. Those aged 60-75 with these risk factors should receive the RSV vaccine, and those without them should not receive it. The best time to get the RSV vaccine is late summer, but early fall administration with other adult vaccines is allowed and is acceptable.
Vaccine safety concerns were top of mind as ACIP members began their deliberations. Possible safety concerns for RSV vaccines have been detected for Guillain-Barré syndrome, atrial fibrillation, and idiopathic thrombocytopenic purpura. Safety surveillance updates are still interim and inconclusive. These signals still need further study and clarification.
Two RSV vaccines have been on the market: one by Pfizer, called Abrysvo, which does not contain an adjuvant; and another one by GSK, called Arexvy, which does contain an adjuvant. With the recent FDA approval of Moderna’s new mRNA RSV vaccine, mRESVIA, there are now three RSV vaccines licensed for those 60 or older. Arexvy is now FDA approved for adults in their 50s. That just happened in early June, but ACIP doesn’t currently recommend it for this fifty-something age group, even for those at high risk for severe RSV disease. This may change with greater clarification of potential vaccine safety concerns.
There is also news about protecting babies from RSV. RSV is the most common cause of hospitalization for infants in the United States, and most hospitalizations for RSV are in healthy, full-term infants. We now have two ways to protect babies: a dose of RSV vaccine given to mom, or a dose of the long-acting monoclonal antibody nirsevimab given to the baby. ACIP clarified that those who received a dose of maternal RSV vaccine during a previous pregnancy are not recommended to receive additional doses during future pregnancies, but infants born to those who were vaccinated for RSV during a prior pregnancy can receive nirsevimab, which is recommended for infants up to 8 months of age during their first RSV season, and for high-risk infants and toddlers aged 8-19 months during their second RSV season.
Last RSV season, supplies of nirsevimab were limited and doses had to be prioritized. No supply problems are anticipated for the upcoming season. A study published in March showed that nirsevimab was 90% effective at preventing RSV-associated hospitalization for infants in their first RSV season.
COVID
Here’s what’s new for COVID vaccines. A new-formula COVID vaccine will be ready for fall. ACIP voted unanimously to recommend a dose of the updated 2024-2025 COVID vaccine for everyone aged 6 months or older. This is a universal recommendation, just like the one we have for flu. But understand that even though COVID has waned, it’s still more deadly than flu. Most Americans now have some immunity against COVID, but this immunity wanes with time, and it also wanes as the virus keeps changing. These updated vaccines provide an incremental boost to our immunity for the new formula for fall. FDA has directed manufacturers to use a monovalent JN.1 lineage formula, with a preference for the KP.2 strain.
Older adults (aged 75 or older) and children under 6 months old are hit hardest by COVID. The littlest ones are too young to be vaccinated, but they can get protection from maternal vaccination. The uptake for last year’s COVID vaccine has been disappointing. Only 22.5% of adults and 14% of children received a dose of the updated shot. Focus-group discussions highlight the importance of a physician recommendation. Adults and children who receive a healthcare provider’s recommendation to get the COVID vaccine are more likely to get vaccinated.
Pneumococcal Vaccines
On June 17, 2024, a new pneumococcal vaccine, PCV21, was FDA approved for those aged 18 or older under an accelerated-approval pathway. ACIP voted to keep it simple and recommends PCV21 as an option for adults aged 19 or older who currently have an indication to receive a dose of PCV. This new PCV21 vaccine is indicated for prevention of both invasive pneumococcal disease (IPD) and pneumococcal pneumonia. Its brand name is Capvaxive and it’s made by Merck. IPD includes bacteremia, pneumonia, pneumococcal bacteremia, and meningitis.
There are two basic types of pneumococcal vaccines: polysaccharide vaccines (PPSV), which do not produce memory B cells; and PCV conjugate vaccines, which do trigger memory B-cell production and therefore induce greater long-term immunity. PCV21 covers 11 unique serotypes not in PCV20. This is important because many cases of adult disease are caused by subtypes not covered by other FDA-approved pneumococcal vaccines. PCV21 has greater coverage of the serotypes that cause invasive disease in adults as compared with PCV20. PCV20 covers up to 58% of those strains, while PCV21 covers up to 84% of strains responsible for invasive disease in adults. But there’s one serotype missing in PCV21, which may limit the groups who receive it. PCV21 does not cover serotype 4, a major cause of IPD in certain populations. Adults experiencing homelessness are 100-300 times more likely to develop IPD due to serotype 4. So are adults in Alaska, especially Alaska Natives. They have an 88-fold increase in serotype 4 invasive disease. Serotype 4 is covered by other pneumococcal vaccines, so for these patients, PCV20 is likely a better high-valent conjugate vaccine option than PCV21.
Flu Vaccines
What’s new for flu? Everyone aged 6 months or older needs a seasonal flu vaccination every year. That’s not new, but there are two new things coming this fall: (1) The seasonal flu vaccine is going trivalent. FDA has removed the Yamagata flu B strain because it no longer appears to be circulating. (2) ACIP made a special off-label recommendation to boost flu protection for solid organ transplant recipients ages 18-64 who are on immunosuppressive medications. These high-risk patients now have the off-label option of receiving one of the higher-dose flu vaccines, including high-dose and adjuvanted flu vaccines, which are FDA approved only for those 65 or older.
Sandra Adamson Fryhofer, Adjunct Clinical Associate Professor of Medicine, Emory University School of Medicine, Atlanta, Georgia, has disclosed the following relevant financial relationships: Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for American Medical Association; Medical Association of Atlanta; ACIP liaison. Received income in an amount equal to or greater than $250 from American College of Physicians; Medscape; American Medical Association.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
ACIP, the CDC’s Advisory Committee on Immunization Practices, met for 3 days in June. New vaccines and new recommendations for respiratory syncytial virus (RSV), flu, COVID, and a new pneumococcal vaccine were revealed.
RSV Protection
We’ll begin with RSV vaccines for adults aged 60 or older. For this group, shared clinical decision-making is out; it no longer applies. New, more specific recommendations from ACIP for RSV vaccines are both age based and risk based. The age-based recommendation applies to those aged 75 or older, who should receive a single RSV vaccine dose. If they have already received a dose under the old recommendation, they don’t need another one, at least for now.
The risk-based recommendation applies to adults from age 60 up to 75, but only for those with risk factors for severe RSV. These risk factors include lung disease, heart disease, immunocompromise, diabetes, obesity with a BMI of 40 or more, neurologic conditions, neuromuscular conditions, chronic kidney disease, liver disorders, hematologic disorders, frailty, and living in a nursing home or other long-term care facility. Those aged 60-75 with these risk factors should receive the RSV vaccine, and those without them should not receive it. The best time to get the RSV vaccine is late summer, but early fall administration with other adult vaccines is allowed and is acceptable.
Vaccine safety concerns were top of mind as ACIP members began their deliberations. Possible safety concerns for RSV vaccines have been detected for Guillain-Barré syndrome, atrial fibrillation, and idiopathic thrombocytopenic purpura. Safety surveillance updates are still interim and inconclusive. These signals still need further study and clarification.
Two RSV vaccines have been on the market: one by Pfizer, called Abrysvo, which does not contain an adjuvant; and another one by GSK, called Arexvy, which does contain an adjuvant. With the recent FDA approval of Moderna’s new mRNA RSV vaccine, mRESVIA, there are now three RSV vaccines licensed for those 60 or older. Arexvy is now FDA approved for adults in their 50s. That just happened in early June, but ACIP doesn’t currently recommend it for this fifty-something age group, even for those at high risk for severe RSV disease. This may change with greater clarification of potential vaccine safety concerns.
There is also news about protecting babies from RSV. RSV is the most common cause of hospitalization for infants in the United States, and most hospitalizations for RSV are in healthy, full-term infants. We now have two ways to protect babies: a dose of RSV vaccine given to mom, or a dose of the long-acting monoclonal antibody nirsevimab given to the baby. ACIP clarified that those who received a dose of maternal RSV vaccine during a previous pregnancy are not recommended to receive additional doses during future pregnancies, but infants born to those who were vaccinated for RSV during a prior pregnancy can receive nirsevimab, which is recommended for infants up to 8 months of age during their first RSV season, and for high-risk infants and toddlers aged 8-19 months during their second RSV season.
Last RSV season, supplies of nirsevimab were limited and doses had to be prioritized. No supply problems are anticipated for the upcoming season. A study published in March showed that nirsevimab was 90% effective at preventing RSV-associated hospitalization for infants in their first RSV season.
COVID
Here’s what’s new for COVID vaccines. A new-formula COVID vaccine will be ready for fall. ACIP voted unanimously to recommend a dose of the updated 2024-2025 COVID vaccine for everyone aged 6 months or older. This is a universal recommendation, just like the one we have for flu. But understand that even though COVID has waned, it’s still more deadly than flu. Most Americans now have some immunity against COVID, but this immunity wanes with time, and it also wanes as the virus keeps changing. These updated vaccines provide an incremental boost to our immunity for the new formula for fall. FDA has directed manufacturers to use a monovalent JN.1 lineage formula, with a preference for the KP.2 strain.
Older adults (aged 75 or older) and children under 6 months old are hit hardest by COVID. The littlest ones are too young to be vaccinated, but they can get protection from maternal vaccination. The uptake for last year’s COVID vaccine has been disappointing. Only 22.5% of adults and 14% of children received a dose of the updated shot. Focus-group discussions highlight the importance of a physician recommendation. Adults and children who receive a healthcare provider’s recommendation to get the COVID vaccine are more likely to get vaccinated.
Pneumococcal Vaccines
On June 17, 2024, a new pneumococcal vaccine, PCV21, was FDA approved for those aged 18 or older under an accelerated-approval pathway. ACIP voted to keep it simple and recommends PCV21 as an option for adults aged 19 or older who currently have an indication to receive a dose of PCV. This new PCV21 vaccine is indicated for prevention of both invasive pneumococcal disease (IPD) and pneumococcal pneumonia. Its brand name is Capvaxive and it’s made by Merck. IPD includes bacteremia, pneumonia, pneumococcal bacteremia, and meningitis.
There are two basic types of pneumococcal vaccines: polysaccharide vaccines (PPSV), which do not produce memory B cells; and PCV conjugate vaccines, which do trigger memory B-cell production and therefore induce greater long-term immunity. PCV21 covers 11 unique serotypes not in PCV20. This is important because many cases of adult disease are caused by subtypes not covered by other FDA-approved pneumococcal vaccines. PCV21 has greater coverage of the serotypes that cause invasive disease in adults as compared with PCV20. PCV20 covers up to 58% of those strains, while PCV21 covers up to 84% of strains responsible for invasive disease in adults. But there’s one serotype missing in PCV21, which may limit the groups who receive it. PCV21 does not cover serotype 4, a major cause of IPD in certain populations. Adults experiencing homelessness are 100-300 times more likely to develop IPD due to serotype 4. So are adults in Alaska, especially Alaska Natives. They have an 88-fold increase in serotype 4 invasive disease. Serotype 4 is covered by other pneumococcal vaccines, so for these patients, PCV20 is likely a better high-valent conjugate vaccine option than PCV21.
Flu Vaccines
What’s new for flu? Everyone aged 6 months or older needs a seasonal flu vaccination every year. That’s not new, but there are two new things coming this fall: (1) The seasonal flu vaccine is going trivalent. FDA has removed the Yamagata flu B strain because it no longer appears to be circulating. (2) ACIP made a special off-label recommendation to boost flu protection for solid organ transplant recipients ages 18-64 who are on immunosuppressive medications. These high-risk patients now have the off-label option of receiving one of the higher-dose flu vaccines, including high-dose and adjuvanted flu vaccines, which are FDA approved only for those 65 or older.
Sandra Adamson Fryhofer, Adjunct Clinical Associate Professor of Medicine, Emory University School of Medicine, Atlanta, Georgia, has disclosed the following relevant financial relationships: Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for American Medical Association; Medical Association of Atlanta; ACIP liaison. Received income in an amount equal to or greater than $250 from American College of Physicians; Medscape; American Medical Association.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
ACIP, the CDC’s Advisory Committee on Immunization Practices, met for 3 days in June. New vaccines and new recommendations for respiratory syncytial virus (RSV), flu, COVID, and a new pneumococcal vaccine were revealed.
RSV Protection
We’ll begin with RSV vaccines for adults aged 60 or older. For this group, shared clinical decision-making is out; it no longer applies. New, more specific recommendations from ACIP for RSV vaccines are both age based and risk based. The age-based recommendation applies to those aged 75 or older, who should receive a single RSV vaccine dose. If they have already received a dose under the old recommendation, they don’t need another one, at least for now.
The risk-based recommendation applies to adults from age 60 up to 75, but only for those with risk factors for severe RSV. These risk factors include lung disease, heart disease, immunocompromise, diabetes, obesity with a BMI of 40 or more, neurologic conditions, neuromuscular conditions, chronic kidney disease, liver disorders, hematologic disorders, frailty, and living in a nursing home or other long-term care facility. Those aged 60-75 with these risk factors should receive the RSV vaccine, and those without them should not receive it. The best time to get the RSV vaccine is late summer, but early fall administration with other adult vaccines is allowed and is acceptable.
Vaccine safety concerns were top of mind as ACIP members began their deliberations. Possible safety concerns for RSV vaccines have been detected for Guillain-Barré syndrome, atrial fibrillation, and idiopathic thrombocytopenic purpura. Safety surveillance updates are still interim and inconclusive. These signals still need further study and clarification.
Two RSV vaccines have been on the market: one by Pfizer, called Abrysvo, which does not contain an adjuvant; and another one by GSK, called Arexvy, which does contain an adjuvant. With the recent FDA approval of Moderna’s new mRNA RSV vaccine, mRESVIA, there are now three RSV vaccines licensed for those 60 or older. Arexvy is now FDA approved for adults in their 50s. That just happened in early June, but ACIP doesn’t currently recommend it for this fifty-something age group, even for those at high risk for severe RSV disease. This may change with greater clarification of potential vaccine safety concerns.
There is also news about protecting babies from RSV. RSV is the most common cause of hospitalization for infants in the United States, and most hospitalizations for RSV are in healthy, full-term infants. We now have two ways to protect babies: a dose of RSV vaccine given to mom, or a dose of the long-acting monoclonal antibody nirsevimab given to the baby. ACIP clarified that those who received a dose of maternal RSV vaccine during a previous pregnancy are not recommended to receive additional doses during future pregnancies, but infants born to those who were vaccinated for RSV during a prior pregnancy can receive nirsevimab, which is recommended for infants up to 8 months of age during their first RSV season, and for high-risk infants and toddlers aged 8-19 months during their second RSV season.
Last RSV season, supplies of nirsevimab were limited and doses had to be prioritized. No supply problems are anticipated for the upcoming season. A study published in March showed that nirsevimab was 90% effective at preventing RSV-associated hospitalization for infants in their first RSV season.
COVID
Here’s what’s new for COVID vaccines. A new-formula COVID vaccine will be ready for fall. ACIP voted unanimously to recommend a dose of the updated 2024-2025 COVID vaccine for everyone aged 6 months or older. This is a universal recommendation, just like the one we have for flu. But understand that even though COVID has waned, it’s still more deadly than flu. Most Americans now have some immunity against COVID, but this immunity wanes with time, and it also wanes as the virus keeps changing. These updated vaccines provide an incremental boost to our immunity for the new formula for fall. FDA has directed manufacturers to use a monovalent JN.1 lineage formula, with a preference for the KP.2 strain.
Older adults (aged 75 or older) and children under 6 months old are hit hardest by COVID. The littlest ones are too young to be vaccinated, but they can get protection from maternal vaccination. The uptake for last year’s COVID vaccine has been disappointing. Only 22.5% of adults and 14% of children received a dose of the updated shot. Focus-group discussions highlight the importance of a physician recommendation. Adults and children who receive a healthcare provider’s recommendation to get the COVID vaccine are more likely to get vaccinated.
Pneumococcal Vaccines
On June 17, 2024, a new pneumococcal vaccine, PCV21, was FDA approved for those aged 18 or older under an accelerated-approval pathway. ACIP voted to keep it simple and recommends PCV21 as an option for adults aged 19 or older who currently have an indication to receive a dose of PCV. This new PCV21 vaccine is indicated for prevention of both invasive pneumococcal disease (IPD) and pneumococcal pneumonia. Its brand name is Capvaxive and it’s made by Merck. IPD includes bacteremia, pneumonia, pneumococcal bacteremia, and meningitis.
There are two basic types of pneumococcal vaccines: polysaccharide vaccines (PPSV), which do not produce memory B cells; and PCV conjugate vaccines, which do trigger memory B-cell production and therefore induce greater long-term immunity. PCV21 covers 11 unique serotypes not in PCV20. This is important because many cases of adult disease are caused by subtypes not covered by other FDA-approved pneumococcal vaccines. PCV21 has greater coverage of the serotypes that cause invasive disease in adults as compared with PCV20. PCV20 covers up to 58% of those strains, while PCV21 covers up to 84% of strains responsible for invasive disease in adults. But there’s one serotype missing in PCV21, which may limit the groups who receive it. PCV21 does not cover serotype 4, a major cause of IPD in certain populations. Adults experiencing homelessness are 100-300 times more likely to develop IPD due to serotype 4. So are adults in Alaska, especially Alaska Natives. They have an 88-fold increase in serotype 4 invasive disease. Serotype 4 is covered by other pneumococcal vaccines, so for these patients, PCV20 is likely a better high-valent conjugate vaccine option than PCV21.
Flu Vaccines
What’s new for flu? Everyone aged 6 months or older needs a seasonal flu vaccination every year. That’s not new, but there are two new things coming this fall: (1) The seasonal flu vaccine is going trivalent. FDA has removed the Yamagata flu B strain because it no longer appears to be circulating. (2) ACIP made a special off-label recommendation to boost flu protection for solid organ transplant recipients ages 18-64 who are on immunosuppressive medications. These high-risk patients now have the off-label option of receiving one of the higher-dose flu vaccines, including high-dose and adjuvanted flu vaccines, which are FDA approved only for those 65 or older.
Sandra Adamson Fryhofer, Adjunct Clinical Associate Professor of Medicine, Emory University School of Medicine, Atlanta, Georgia, has disclosed the following relevant financial relationships: Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for American Medical Association; Medical Association of Atlanta; ACIP liaison. Received income in an amount equal to or greater than $250 from American College of Physicians; Medscape; American Medical Association.
A version of this article first appeared on Medscape.com.
Don’t Forget Adult Hepatitis Vaccinations
Hepatitis B (Hep B) is a liver infection against which vaccination was previously recommended for certain eligible risk groups. However, in 2022, the Centers for Disease Control and Prevention (CDC) switched from recommending vaccination for at-risk persons to recommending universal vaccination for children and adults.
That includes infants, children 18 years of age and younger, adults aged 19-59, and people aged 60 and up with risk factors for this viral infection. Even those aged 60 and older without known risk factors for hepatitis B may receive Hep B vaccines.
Risk factors under prior recommendations included potential criminal or stigmatizing behaviors such as injection-drug use, incarceration, or multiple sex partners, which limited risk assessment by providers. The CDC points out that universal adult Hep B vaccination through age 59 obviates the need for the previous approach of risk-factor screening and sensitive disclosures to determine eligibility and could increase vaccination coverage and reduce cases.
“A universal recommendation for Hep B vaccination could increase the number of persons who receive vaccination before the onset of chronic liver disease and other comorbidities (e.g., obesity or diabetes) that might make vaccination less effective,” the CDC stated, noting that patients with chronic liver disease have a reduced immune response to Hep B vaccination. Hep B vaccination also protects against hepatitis D.
“Most people born in the US are vaccinated during infancy, beginning on the first day of life,” Lauren D. Block, MD, an internist at Northwell Health and an assistant professor in the Institute of Health System Science at the Feinstein Institutes for Medical Research at sites in metropolitan New York City, said in an interview.
Typically, Dr. Block added, at-risk persons will have titers drawn to make sure they are immune to Hep B. “Titers can wane over the decades in healthy people, in which case a booster shot may be needed, or a restart of the three-part vaccination series if a person is not sure if they were vaccinated previously.” Those at greater risk include people with weakened immune systems, people with diabetes or on immunosuppressives, healthcare workers, travelers to higher-risk countries, people with multiple sexual partners, and IV drug users.
Although Hep B vaccines have demonstrated safety, immunogenicity, and efficacy during the past four decades, coverage among US adults has been suboptimal, limiting further reduction in infections, the CDC noted.
Hepatitis A
Though not widely endemic to North America, Hep A can be acquired during travel abroad, particularly to developing countries, or through exposure to unsanitary conditions and contaminated food or water.
The CDC recommends routine Hep A vaccination for all children aged 12-23 months, all unvaccinated children and adolescents 2-18 years, and all persons, including those who are pregnant, with increased risk factors for this orally and fecally transmitted infection or at risk for severe disease from it. At-risk groups include international travelers to affected regions, men who have sex with men, incarcerated individuals or group-home residents, injection and non-injection drug users, and homeless persons.
Hepatitis C and E
There is no vaccine for Hep C, and no FDA-approved vaccine in the United States for hepatitis E, although a vaccine for the latter was approved in China in 2012.
As with Hep A, observing strict water, food, and sanitation standards is essential for preventing infection with hepatitis E.
Dr. Block disclosed no competing interests relevant to her comments.
Hepatitis B (Hep B) is a liver infection against which vaccination was previously recommended for certain eligible risk groups. However, in 2022, the Centers for Disease Control and Prevention (CDC) switched from recommending vaccination for at-risk persons to recommending universal vaccination for children and adults.
That includes infants, children 18 years of age and younger, adults aged 19-59, and people aged 60 and up with risk factors for this viral infection. Even those aged 60 and older without known risk factors for hepatitis B may receive Hep B vaccines.
Risk factors under prior recommendations included potential criminal or stigmatizing behaviors such as injection-drug use, incarceration, or multiple sex partners, which limited risk assessment by providers. The CDC points out that universal adult Hep B vaccination through age 59 obviates the need for the previous approach of risk-factor screening and sensitive disclosures to determine eligibility and could increase vaccination coverage and reduce cases.
“A universal recommendation for Hep B vaccination could increase the number of persons who receive vaccination before the onset of chronic liver disease and other comorbidities (e.g., obesity or diabetes) that might make vaccination less effective,” the CDC stated, noting that patients with chronic liver disease have a reduced immune response to Hep B vaccination. Hep B vaccination also protects against hepatitis D.
“Most people born in the US are vaccinated during infancy, beginning on the first day of life,” Lauren D. Block, MD, an internist at Northwell Health and an assistant professor in the Institute of Health System Science at the Feinstein Institutes for Medical Research at sites in metropolitan New York City, said in an interview.
Typically, Dr. Block added, at-risk persons will have titers drawn to make sure they are immune to Hep B. “Titers can wane over the decades in healthy people, in which case a booster shot may be needed, or a restart of the three-part vaccination series if a person is not sure if they were vaccinated previously.” Those at greater risk include people with weakened immune systems, people with diabetes or on immunosuppressives, healthcare workers, travelers to higher-risk countries, people with multiple sexual partners, and IV drug users.
Although Hep B vaccines have demonstrated safety, immunogenicity, and efficacy during the past four decades, coverage among US adults has been suboptimal, limiting further reduction in infections, the CDC noted.
Hepatitis A
Though not widely endemic to North America, Hep A can be acquired during travel abroad, particularly to developing countries, or through exposure to unsanitary conditions and contaminated food or water.
The CDC recommends routine Hep A vaccination for all children aged 12-23 months, all unvaccinated children and adolescents 2-18 years, and all persons, including those who are pregnant, with increased risk factors for this orally and fecally transmitted infection or at risk for severe disease from it. At-risk groups include international travelers to affected regions, men who have sex with men, incarcerated individuals or group-home residents, injection and non-injection drug users, and homeless persons.
Hepatitis C and E
There is no vaccine for Hep C, and no FDA-approved vaccine in the United States for hepatitis E, although a vaccine for the latter was approved in China in 2012.
As with Hep A, observing strict water, food, and sanitation standards is essential for preventing infection with hepatitis E.
Dr. Block disclosed no competing interests relevant to her comments.
Hepatitis B (Hep B) is a liver infection against which vaccination was previously recommended for certain eligible risk groups. However, in 2022, the Centers for Disease Control and Prevention (CDC) switched from recommending vaccination for at-risk persons to recommending universal vaccination for children and adults.
That includes infants, children 18 years of age and younger, adults aged 19-59, and people aged 60 and up with risk factors for this viral infection. Even those aged 60 and older without known risk factors for hepatitis B may receive Hep B vaccines.
Risk factors under prior recommendations included potential criminal or stigmatizing behaviors such as injection-drug use, incarceration, or multiple sex partners, which limited risk assessment by providers. The CDC points out that universal adult Hep B vaccination through age 59 obviates the need for the previous approach of risk-factor screening and sensitive disclosures to determine eligibility and could increase vaccination coverage and reduce cases.
“A universal recommendation for Hep B vaccination could increase the number of persons who receive vaccination before the onset of chronic liver disease and other comorbidities (e.g., obesity or diabetes) that might make vaccination less effective,” the CDC stated, noting that patients with chronic liver disease have a reduced immune response to Hep B vaccination. Hep B vaccination also protects against hepatitis D.
“Most people born in the US are vaccinated during infancy, beginning on the first day of life,” Lauren D. Block, MD, an internist at Northwell Health and an assistant professor in the Institute of Health System Science at the Feinstein Institutes for Medical Research at sites in metropolitan New York City, said in an interview.
Typically, Dr. Block added, at-risk persons will have titers drawn to make sure they are immune to Hep B. “Titers can wane over the decades in healthy people, in which case a booster shot may be needed, or a restart of the three-part vaccination series if a person is not sure if they were vaccinated previously.” Those at greater risk include people with weakened immune systems, people with diabetes or on immunosuppressives, healthcare workers, travelers to higher-risk countries, people with multiple sexual partners, and IV drug users.
Although Hep B vaccines have demonstrated safety, immunogenicity, and efficacy during the past four decades, coverage among US adults has been suboptimal, limiting further reduction in infections, the CDC noted.
Hepatitis A
Though not widely endemic to North America, Hep A can be acquired during travel abroad, particularly to developing countries, or through exposure to unsanitary conditions and contaminated food or water.
The CDC recommends routine Hep A vaccination for all children aged 12-23 months, all unvaccinated children and adolescents 2-18 years, and all persons, including those who are pregnant, with increased risk factors for this orally and fecally transmitted infection or at risk for severe disease from it. At-risk groups include international travelers to affected regions, men who have sex with men, incarcerated individuals or group-home residents, injection and non-injection drug users, and homeless persons.
Hepatitis C and E
There is no vaccine for Hep C, and no FDA-approved vaccine in the United States for hepatitis E, although a vaccine for the latter was approved in China in 2012.
As with Hep A, observing strict water, food, and sanitation standards is essential for preventing infection with hepatitis E.
Dr. Block disclosed no competing interests relevant to her comments.
Online Diagnosis of Sexually Transmitted Infections? Ethicist Says We Are Nowhere Close
This transcript has been edited for clarity.
There has been a large amount of news lately about dating online and dating apps. Probably the most common way younger people find potential partners is to go online and see who’s there that they might want to meet.
Online dating is also notorious for being full of scammers. There are all kinds of people out there that you have to be careful of, who are trying to rip you off by saying, “Send me money, I’m in trouble,” or “Now that we have a relationship, will you support my particular entrepreneurial idea?” Certainly, dangers are there.
Another danger we don’t talk much about is meeting people who have sexually transmitted diseases. That’s been a problem before websites and before dating apps. I think the opportunity of meeting more people — strangers, people you don’t really know — who may not tell you the truth about their health, and particularly their sexual health, is really out there.
It’s always good medical advice to tell people to practice safe sex, and that often involves a man wearing a condom. It certainly is the case that we want to attend not just to the prevention of unwanted pregnancy but also to the transmission of diseases. I think it’s very important to tell women of reproductive age to get their HPV shot to try to reduce cancers in their reproductive systems, or sometimes in men — anal cancers, or even being a transmitter of disease.
Even then, certainly one wants to recommend that, in an age where some people are going to meet many partners that they don’t know well or don’t have much background with, it’s wise to try to prevent diseases using the vaccines we’ve got, using the contraceptive methods that will prevent disease transmission, and reminding people to ask about sex life.
I did come across a website that just startled me. It’s called HeHealth, and basically it says to men, if you are conscientious about your sex life, take a picture of your penis, send it to us, and we have doctors — I presume they’re US doctors but I don’t know — who will diagnose venereal diseases based on that picture. I presume women could also say, “Before we have sex, or now that we’re approaching that possibility, I want you to send a picture to this company on this website.”
Now, a couple of reminders. I think we all know this, but just because you’re not manifesting symptoms on your reproductive organs doesn’t mean you don’t have a sexual disease. It’s not a reliable measure. Yes, maybe you could have somebody say: “Oh, that looks nasty. I’m not sure you ought to have sex right now, and maybe you should go get some treatment.” This is going to miss many cases and is not a reliable indicator that your partner is safe in terms of not transmitting diseases to you.
It also isn’t clear what they do with these images. Do they keep them? Who can see them? Could they resell them? What sort of privacy protection have you got if you decide to use this?
There’s another issue here, which is, if they misdiagnose someone and you do catch a sexual disease, who’s liable? Can you go after them for using doctors who weren’t competent or transmitting images that weren’t really adequate because you didn’t know how to take that picture properly when you sent that off to them? There are many unknowns.
The bottom line is that we’re in a different world, I think, of romance. We’re in a world where some people are going to meet more partners. Some people are going to meet more strangers. One approach is to have us take pictures of ourselves, send them off to who knows where, and ask for a green light to go ahead and have sexual relations. I don’t think we’re anywhere close to being able to rely on that as a way to avoid the risks of unprotected sexual behavior.
We do know what to do in dealing with patients who are sexually active. First, we have to ask them. Then we’ve got to recommend available vaccinations to prevent the transmission of some cancers, the HPV vaccine. Then they need that reminder about safe sexual practices not only to protect against unwanted pregnancy, but still, in this day and age, to protect against syphilis, which is on the rise, plus HIV, gonorrhea, chlamydia, and other sexually transmissible diseases.
I’m not going to rely on the penis picture to make the world safe for sex. I think we have to still use the old-fashioned techniques of education and prevention to do the best we can.
Dr. Caplan is director of the Division of Medical Ethics at New York University Langone Medical Center, New York City. He reported conflicts of interest with Johnson & Johnson’s Panel for Compassionate Drug Use and Medscape.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
There has been a large amount of news lately about dating online and dating apps. Probably the most common way younger people find potential partners is to go online and see who’s there that they might want to meet.
Online dating is also notorious for being full of scammers. There are all kinds of people out there that you have to be careful of, who are trying to rip you off by saying, “Send me money, I’m in trouble,” or “Now that we have a relationship, will you support my particular entrepreneurial idea?” Certainly, dangers are there.
Another danger we don’t talk much about is meeting people who have sexually transmitted diseases. That’s been a problem before websites and before dating apps. I think the opportunity of meeting more people — strangers, people you don’t really know — who may not tell you the truth about their health, and particularly their sexual health, is really out there.
It’s always good medical advice to tell people to practice safe sex, and that often involves a man wearing a condom. It certainly is the case that we want to attend not just to the prevention of unwanted pregnancy but also to the transmission of diseases. I think it’s very important to tell women of reproductive age to get their HPV shot to try to reduce cancers in their reproductive systems, or sometimes in men — anal cancers, or even being a transmitter of disease.
Even then, certainly one wants to recommend that, in an age where some people are going to meet many partners that they don’t know well or don’t have much background with, it’s wise to try to prevent diseases using the vaccines we’ve got, using the contraceptive methods that will prevent disease transmission, and reminding people to ask about sex life.
I did come across a website that just startled me. It’s called HeHealth, and basically it says to men, if you are conscientious about your sex life, take a picture of your penis, send it to us, and we have doctors — I presume they’re US doctors but I don’t know — who will diagnose venereal diseases based on that picture. I presume women could also say, “Before we have sex, or now that we’re approaching that possibility, I want you to send a picture to this company on this website.”
Now, a couple of reminders. I think we all know this, but just because you’re not manifesting symptoms on your reproductive organs doesn’t mean you don’t have a sexual disease. It’s not a reliable measure. Yes, maybe you could have somebody say: “Oh, that looks nasty. I’m not sure you ought to have sex right now, and maybe you should go get some treatment.” This is going to miss many cases and is not a reliable indicator that your partner is safe in terms of not transmitting diseases to you.
It also isn’t clear what they do with these images. Do they keep them? Who can see them? Could they resell them? What sort of privacy protection have you got if you decide to use this?
There’s another issue here, which is, if they misdiagnose someone and you do catch a sexual disease, who’s liable? Can you go after them for using doctors who weren’t competent or transmitting images that weren’t really adequate because you didn’t know how to take that picture properly when you sent that off to them? There are many unknowns.
The bottom line is that we’re in a different world, I think, of romance. We’re in a world where some people are going to meet more partners. Some people are going to meet more strangers. One approach is to have us take pictures of ourselves, send them off to who knows where, and ask for a green light to go ahead and have sexual relations. I don’t think we’re anywhere close to being able to rely on that as a way to avoid the risks of unprotected sexual behavior.
We do know what to do in dealing with patients who are sexually active. First, we have to ask them. Then we’ve got to recommend available vaccinations to prevent the transmission of some cancers, the HPV vaccine. Then they need that reminder about safe sexual practices not only to protect against unwanted pregnancy, but still, in this day and age, to protect against syphilis, which is on the rise, plus HIV, gonorrhea, chlamydia, and other sexually transmissible diseases.
I’m not going to rely on the penis picture to make the world safe for sex. I think we have to still use the old-fashioned techniques of education and prevention to do the best we can.
Dr. Caplan is director of the Division of Medical Ethics at New York University Langone Medical Center, New York City. He reported conflicts of interest with Johnson & Johnson’s Panel for Compassionate Drug Use and Medscape.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
There has been a large amount of news lately about dating online and dating apps. Probably the most common way younger people find potential partners is to go online and see who’s there that they might want to meet.
Online dating is also notorious for being full of scammers. There are all kinds of people out there that you have to be careful of, who are trying to rip you off by saying, “Send me money, I’m in trouble,” or “Now that we have a relationship, will you support my particular entrepreneurial idea?” Certainly, dangers are there.
Another danger we don’t talk much about is meeting people who have sexually transmitted diseases. That’s been a problem before websites and before dating apps. I think the opportunity of meeting more people — strangers, people you don’t really know — who may not tell you the truth about their health, and particularly their sexual health, is really out there.
It’s always good medical advice to tell people to practice safe sex, and that often involves a man wearing a condom. It certainly is the case that we want to attend not just to the prevention of unwanted pregnancy but also to the transmission of diseases. I think it’s very important to tell women of reproductive age to get their HPV shot to try to reduce cancers in their reproductive systems, or sometimes in men — anal cancers, or even being a transmitter of disease.
Even then, certainly one wants to recommend that, in an age where some people are going to meet many partners that they don’t know well or don’t have much background with, it’s wise to try to prevent diseases using the vaccines we’ve got, using the contraceptive methods that will prevent disease transmission, and reminding people to ask about sex life.
I did come across a website that just startled me. It’s called HeHealth, and basically it says to men, if you are conscientious about your sex life, take a picture of your penis, send it to us, and we have doctors — I presume they’re US doctors but I don’t know — who will diagnose venereal diseases based on that picture. I presume women could also say, “Before we have sex, or now that we’re approaching that possibility, I want you to send a picture to this company on this website.”
Now, a couple of reminders. I think we all know this, but just because you’re not manifesting symptoms on your reproductive organs doesn’t mean you don’t have a sexual disease. It’s not a reliable measure. Yes, maybe you could have somebody say: “Oh, that looks nasty. I’m not sure you ought to have sex right now, and maybe you should go get some treatment.” This is going to miss many cases and is not a reliable indicator that your partner is safe in terms of not transmitting diseases to you.
It also isn’t clear what they do with these images. Do they keep them? Who can see them? Could they resell them? What sort of privacy protection have you got if you decide to use this?
There’s another issue here, which is, if they misdiagnose someone and you do catch a sexual disease, who’s liable? Can you go after them for using doctors who weren’t competent or transmitting images that weren’t really adequate because you didn’t know how to take that picture properly when you sent that off to them? There are many unknowns.
The bottom line is that we’re in a different world, I think, of romance. We’re in a world where some people are going to meet more partners. Some people are going to meet more strangers. One approach is to have us take pictures of ourselves, send them off to who knows where, and ask for a green light to go ahead and have sexual relations. I don’t think we’re anywhere close to being able to rely on that as a way to avoid the risks of unprotected sexual behavior.
We do know what to do in dealing with patients who are sexually active. First, we have to ask them. Then we’ve got to recommend available vaccinations to prevent the transmission of some cancers, the HPV vaccine. Then they need that reminder about safe sexual practices not only to protect against unwanted pregnancy, but still, in this day and age, to protect against syphilis, which is on the rise, plus HIV, gonorrhea, chlamydia, and other sexually transmissible diseases.
I’m not going to rely on the penis picture to make the world safe for sex. I think we have to still use the old-fashioned techniques of education and prevention to do the best we can.
Dr. Caplan is director of the Division of Medical Ethics at New York University Langone Medical Center, New York City. He reported conflicts of interest with Johnson & Johnson’s Panel for Compassionate Drug Use and Medscape.
A version of this article first appeared on Medscape.com.
Timing Pneumococcal Vaccination in Patients with RA Starting Methotrexate: When’s Best?
VIENNA — Pneumococcal vaccination administered 1 month prior to starting methotrexate (MTX) in patients with rheumatoid arthritis (RA) allows a significantly higher immunological response at 1 month and does not affect disease control at 1 year, compared with starting MTX simultaneously with the vaccination, according to data from a randomized trial presented at the annual European Congress of Rheumatology.
“Our patients are more susceptible to infection due to immunosuppressive therapy, and it’s recommended they receive vaccination against pneumococcal infection,” the lead author Jacques Morel, MD, PhD, said in his presentation of results from the VACIMRA study.
Timing the vaccination against pneumococcal disease when initiating MTX in clinical practice has been a point of uncertainty, noted Dr. Morel, a rheumatologist from Centre Hospitalier Universitaire, Montpellier, France.
“How can we deal with this in clinical practice where one recommendation is to vaccine before initiation of methotrexate, but it is also recommended to start methotrexate as soon as the diagnosis of RA is made?” he asked.
Comparing Humoral Response of MTX Started Immediately or 1 Month Post-Vaccination
The prospective, randomized, multicenter trial aimed to compare the rate of humoral immunological response against pneumococcal 13-valent conjugate vaccine (PCV13) in patients with RA who had a Disease Activity Score in 28 joints (DAS28) > 3.2, never taken MTX, and never been vaccinated against pneumococcus. Patients were vaccinated either 1 month before MTX initiation (n = 126) or simultaneously with MTX (n = 123). Oral glucocorticoids were allowed but only at < 10 mg/d. Following PCV13 vaccination, all patients also received the 23-valent pneumococcal polysaccharide vaccine (PPV23) 2 months later.
Concentrations of immunoglobulin (Ig) G antibodies against the 13 serotypes contained within PCV13 were measured using enzyme-linked immunosorbent assay (ELISA) and opsonophagocytic killing assay (OPA) at baseline and during follow-up at 1, 3, 6, and 12 months.
Positive antibody response was defined as a twofold or more increase in the IgG concentration using ELISA. The main outcome was the responder rates at 1 month after PCV13, defined by at least three positive antibody responses out of five of the target PVC13 serotypes (1, 3, 5, 7F, and 19A) using ELISA or OPA. Secondary outcomes included comparisons of the percentage of patients responding to each of the 13 vaccine serotypes at 1 month and after the boost with PPV23 and at 3, 6, and 12 months after vaccination with PCV13. The researchers also measured disease activity, infections, and side effects throughout the study.
Dr. Morel highlighted that all the patients had very early RA of less than 6 months, and that their characteristics at baseline were similar in both groups with 70% women, mean age 55.6 years, RA duration 2 months, 69% positive for anticitrullinated protein antibodies, 21% with erosive disease, and a DAS28 based on C-reactive protein of 4.6.
Response rates in those receiving MTX 1 month after vaccination were significantly higher at 88% with ELISA than those at 75% for immediate vaccination (P < .01) and 96% vs 88% with OPA (P = .02). These responder proportions persisted at the 12-month follow-up measurements, remaining higher in the delayed MTX group for both assays and across the 13 serotypes.
Showing a graph of the antibody responses, Dr. Morel explained that “at 12 months, the curves start to converge, but the difference in antibody titers were still significant for eight of the 13 serotypes.”
Disease Activity Not Adversely Affected by Starting MTX 1 Month Later
Regarding medication doses at 12 months, the cumulative glucocorticoid doses were similar between groups during the follow-up. As expected, the 1-year cumulative dose of MTX was higher in those given the drug immediately after vaccination vs delayed (826 vs 734 mg), but the weekly mean doses of MTX were similar at 3, 6, and 12 months between the two groups, and likewise, the use of targeted disease-modifying antirheumatic drugs (DMARDs) at 1 year was comparable. The cumulative glucocorticoid dose at 12 months was similar at 1716 mg with delayed MTX and 1613 mg with immediate MTX.
Not unexpectedly, at 1 month, DAS28 scores were higher with delayed vs immediate MTX at 3.95 vs 3.38 for DAS28-ESR and 3.54 vs 3.01 for DAS28-CRP (P < .01), but after the first month, DAS28 scores were similar between the two groups.
No significant differences were found between the groups for adverse event rates within 7 days of receiving PCV13, with local and systemic reactions occurring at 60%-61% and 50%-58%, respectively; fever at 0%-4%; and severe infections at 12%.
Finally, no difference was found in terms of serious adverse events between groups, with one pneumococcal infection with delayed MTX during follow-up, and there were no unexpected side effects observed with the PCV13 and PPV23 vaccinations.
Rheumatologists’ Reactions
Ernest Choy, MD, head of rheumatology and translational research at the Institute of Infection and Immunity, Cardiff University School of Medicine, Cardiff, Wales, asked if any individual showed no humoral response at all rather than a reduced response. “I ask because if there is no humoral response, then they are at very high risk, and there will be clinical relevance to that.”
Dr. Morel replied that “one serotype showed no response, at least according to the assays used, but we don’t know why. We analyzed at the population [level], not at the individual level, so it is difficult to answer the question.”
Another delegate asked what the participants thought about delaying MTX by 1 month. “When we tell the patient we need to vaccinate before we can use methotrexate [because] otherwise, we will reduce the response to the vaccination, then patient accepts it,” said Dr. Morel, adding that, “we allowed a minimal dose of steroids, and we saw from the results that the DAS28 at 1 month had changed.”
Co-moderator Katerina Chatzidionysiou, MD, PhD, a consultant rheumatologist and head of the Clinical Trial Department Rheumatology Unit, Karolinska University Hospital, Stockholm, Sweden, said that “As a physician, I’d feel uncomfortable delaying MTX if they had very active disease even for a short period of time.”
Dr. Morel replied that, “Today, we have so many drugs that can control the disease, for example, the targeted DMARDs. Progression does not show much variation, and we know x-ray progression with today’s drugs is a lot less than previously.”
Dr. Morel reported financial relationships with AbbVie, Amgen, Biogen, Bristol Myers Squibb, Fresenius Kabi, Galapagos, GlaxoSmithKline, Lilly, Medac, Nordic Pharma, Novartis, Pfizer, Sandoz, Sanofi, Boehringer Ingelheim, and Servier. Dr. Choy had no relevant financial relationships of relevance to this study.
A version of this article appeared on Medscape.com.
VIENNA — Pneumococcal vaccination administered 1 month prior to starting methotrexate (MTX) in patients with rheumatoid arthritis (RA) allows a significantly higher immunological response at 1 month and does not affect disease control at 1 year, compared with starting MTX simultaneously with the vaccination, according to data from a randomized trial presented at the annual European Congress of Rheumatology.
“Our patients are more susceptible to infection due to immunosuppressive therapy, and it’s recommended they receive vaccination against pneumococcal infection,” the lead author Jacques Morel, MD, PhD, said in his presentation of results from the VACIMRA study.
Timing the vaccination against pneumococcal disease when initiating MTX in clinical practice has been a point of uncertainty, noted Dr. Morel, a rheumatologist from Centre Hospitalier Universitaire, Montpellier, France.
“How can we deal with this in clinical practice where one recommendation is to vaccine before initiation of methotrexate, but it is also recommended to start methotrexate as soon as the diagnosis of RA is made?” he asked.
Comparing Humoral Response of MTX Started Immediately or 1 Month Post-Vaccination
The prospective, randomized, multicenter trial aimed to compare the rate of humoral immunological response against pneumococcal 13-valent conjugate vaccine (PCV13) in patients with RA who had a Disease Activity Score in 28 joints (DAS28) > 3.2, never taken MTX, and never been vaccinated against pneumococcus. Patients were vaccinated either 1 month before MTX initiation (n = 126) or simultaneously with MTX (n = 123). Oral glucocorticoids were allowed but only at < 10 mg/d. Following PCV13 vaccination, all patients also received the 23-valent pneumococcal polysaccharide vaccine (PPV23) 2 months later.
Concentrations of immunoglobulin (Ig) G antibodies against the 13 serotypes contained within PCV13 were measured using enzyme-linked immunosorbent assay (ELISA) and opsonophagocytic killing assay (OPA) at baseline and during follow-up at 1, 3, 6, and 12 months.
Positive antibody response was defined as a twofold or more increase in the IgG concentration using ELISA. The main outcome was the responder rates at 1 month after PCV13, defined by at least three positive antibody responses out of five of the target PVC13 serotypes (1, 3, 5, 7F, and 19A) using ELISA or OPA. Secondary outcomes included comparisons of the percentage of patients responding to each of the 13 vaccine serotypes at 1 month and after the boost with PPV23 and at 3, 6, and 12 months after vaccination with PCV13. The researchers also measured disease activity, infections, and side effects throughout the study.
Dr. Morel highlighted that all the patients had very early RA of less than 6 months, and that their characteristics at baseline were similar in both groups with 70% women, mean age 55.6 years, RA duration 2 months, 69% positive for anticitrullinated protein antibodies, 21% with erosive disease, and a DAS28 based on C-reactive protein of 4.6.
Response rates in those receiving MTX 1 month after vaccination were significantly higher at 88% with ELISA than those at 75% for immediate vaccination (P < .01) and 96% vs 88% with OPA (P = .02). These responder proportions persisted at the 12-month follow-up measurements, remaining higher in the delayed MTX group for both assays and across the 13 serotypes.
Showing a graph of the antibody responses, Dr. Morel explained that “at 12 months, the curves start to converge, but the difference in antibody titers were still significant for eight of the 13 serotypes.”
Disease Activity Not Adversely Affected by Starting MTX 1 Month Later
Regarding medication doses at 12 months, the cumulative glucocorticoid doses were similar between groups during the follow-up. As expected, the 1-year cumulative dose of MTX was higher in those given the drug immediately after vaccination vs delayed (826 vs 734 mg), but the weekly mean doses of MTX were similar at 3, 6, and 12 months between the two groups, and likewise, the use of targeted disease-modifying antirheumatic drugs (DMARDs) at 1 year was comparable. The cumulative glucocorticoid dose at 12 months was similar at 1716 mg with delayed MTX and 1613 mg with immediate MTX.
Not unexpectedly, at 1 month, DAS28 scores were higher with delayed vs immediate MTX at 3.95 vs 3.38 for DAS28-ESR and 3.54 vs 3.01 for DAS28-CRP (P < .01), but after the first month, DAS28 scores were similar between the two groups.
No significant differences were found between the groups for adverse event rates within 7 days of receiving PCV13, with local and systemic reactions occurring at 60%-61% and 50%-58%, respectively; fever at 0%-4%; and severe infections at 12%.
Finally, no difference was found in terms of serious adverse events between groups, with one pneumococcal infection with delayed MTX during follow-up, and there were no unexpected side effects observed with the PCV13 and PPV23 vaccinations.
Rheumatologists’ Reactions
Ernest Choy, MD, head of rheumatology and translational research at the Institute of Infection and Immunity, Cardiff University School of Medicine, Cardiff, Wales, asked if any individual showed no humoral response at all rather than a reduced response. “I ask because if there is no humoral response, then they are at very high risk, and there will be clinical relevance to that.”
Dr. Morel replied that “one serotype showed no response, at least according to the assays used, but we don’t know why. We analyzed at the population [level], not at the individual level, so it is difficult to answer the question.”
Another delegate asked what the participants thought about delaying MTX by 1 month. “When we tell the patient we need to vaccinate before we can use methotrexate [because] otherwise, we will reduce the response to the vaccination, then patient accepts it,” said Dr. Morel, adding that, “we allowed a minimal dose of steroids, and we saw from the results that the DAS28 at 1 month had changed.”
Co-moderator Katerina Chatzidionysiou, MD, PhD, a consultant rheumatologist and head of the Clinical Trial Department Rheumatology Unit, Karolinska University Hospital, Stockholm, Sweden, said that “As a physician, I’d feel uncomfortable delaying MTX if they had very active disease even for a short period of time.”
Dr. Morel replied that, “Today, we have so many drugs that can control the disease, for example, the targeted DMARDs. Progression does not show much variation, and we know x-ray progression with today’s drugs is a lot less than previously.”
Dr. Morel reported financial relationships with AbbVie, Amgen, Biogen, Bristol Myers Squibb, Fresenius Kabi, Galapagos, GlaxoSmithKline, Lilly, Medac, Nordic Pharma, Novartis, Pfizer, Sandoz, Sanofi, Boehringer Ingelheim, and Servier. Dr. Choy had no relevant financial relationships of relevance to this study.
A version of this article appeared on Medscape.com.
VIENNA — Pneumococcal vaccination administered 1 month prior to starting methotrexate (MTX) in patients with rheumatoid arthritis (RA) allows a significantly higher immunological response at 1 month and does not affect disease control at 1 year, compared with starting MTX simultaneously with the vaccination, according to data from a randomized trial presented at the annual European Congress of Rheumatology.
“Our patients are more susceptible to infection due to immunosuppressive therapy, and it’s recommended they receive vaccination against pneumococcal infection,” the lead author Jacques Morel, MD, PhD, said in his presentation of results from the VACIMRA study.
Timing the vaccination against pneumococcal disease when initiating MTX in clinical practice has been a point of uncertainty, noted Dr. Morel, a rheumatologist from Centre Hospitalier Universitaire, Montpellier, France.
“How can we deal with this in clinical practice where one recommendation is to vaccine before initiation of methotrexate, but it is also recommended to start methotrexate as soon as the diagnosis of RA is made?” he asked.
Comparing Humoral Response of MTX Started Immediately or 1 Month Post-Vaccination
The prospective, randomized, multicenter trial aimed to compare the rate of humoral immunological response against pneumococcal 13-valent conjugate vaccine (PCV13) in patients with RA who had a Disease Activity Score in 28 joints (DAS28) > 3.2, never taken MTX, and never been vaccinated against pneumococcus. Patients were vaccinated either 1 month before MTX initiation (n = 126) or simultaneously with MTX (n = 123). Oral glucocorticoids were allowed but only at < 10 mg/d. Following PCV13 vaccination, all patients also received the 23-valent pneumococcal polysaccharide vaccine (PPV23) 2 months later.
Concentrations of immunoglobulin (Ig) G antibodies against the 13 serotypes contained within PCV13 were measured using enzyme-linked immunosorbent assay (ELISA) and opsonophagocytic killing assay (OPA) at baseline and during follow-up at 1, 3, 6, and 12 months.
Positive antibody response was defined as a twofold or more increase in the IgG concentration using ELISA. The main outcome was the responder rates at 1 month after PCV13, defined by at least three positive antibody responses out of five of the target PVC13 serotypes (1, 3, 5, 7F, and 19A) using ELISA or OPA. Secondary outcomes included comparisons of the percentage of patients responding to each of the 13 vaccine serotypes at 1 month and after the boost with PPV23 and at 3, 6, and 12 months after vaccination with PCV13. The researchers also measured disease activity, infections, and side effects throughout the study.
Dr. Morel highlighted that all the patients had very early RA of less than 6 months, and that their characteristics at baseline were similar in both groups with 70% women, mean age 55.6 years, RA duration 2 months, 69% positive for anticitrullinated protein antibodies, 21% with erosive disease, and a DAS28 based on C-reactive protein of 4.6.
Response rates in those receiving MTX 1 month after vaccination were significantly higher at 88% with ELISA than those at 75% for immediate vaccination (P < .01) and 96% vs 88% with OPA (P = .02). These responder proportions persisted at the 12-month follow-up measurements, remaining higher in the delayed MTX group for both assays and across the 13 serotypes.
Showing a graph of the antibody responses, Dr. Morel explained that “at 12 months, the curves start to converge, but the difference in antibody titers were still significant for eight of the 13 serotypes.”
Disease Activity Not Adversely Affected by Starting MTX 1 Month Later
Regarding medication doses at 12 months, the cumulative glucocorticoid doses were similar between groups during the follow-up. As expected, the 1-year cumulative dose of MTX was higher in those given the drug immediately after vaccination vs delayed (826 vs 734 mg), but the weekly mean doses of MTX were similar at 3, 6, and 12 months between the two groups, and likewise, the use of targeted disease-modifying antirheumatic drugs (DMARDs) at 1 year was comparable. The cumulative glucocorticoid dose at 12 months was similar at 1716 mg with delayed MTX and 1613 mg with immediate MTX.
Not unexpectedly, at 1 month, DAS28 scores were higher with delayed vs immediate MTX at 3.95 vs 3.38 for DAS28-ESR and 3.54 vs 3.01 for DAS28-CRP (P < .01), but after the first month, DAS28 scores were similar between the two groups.
No significant differences were found between the groups for adverse event rates within 7 days of receiving PCV13, with local and systemic reactions occurring at 60%-61% and 50%-58%, respectively; fever at 0%-4%; and severe infections at 12%.
Finally, no difference was found in terms of serious adverse events between groups, with one pneumococcal infection with delayed MTX during follow-up, and there were no unexpected side effects observed with the PCV13 and PPV23 vaccinations.
Rheumatologists’ Reactions
Ernest Choy, MD, head of rheumatology and translational research at the Institute of Infection and Immunity, Cardiff University School of Medicine, Cardiff, Wales, asked if any individual showed no humoral response at all rather than a reduced response. “I ask because if there is no humoral response, then they are at very high risk, and there will be clinical relevance to that.”
Dr. Morel replied that “one serotype showed no response, at least according to the assays used, but we don’t know why. We analyzed at the population [level], not at the individual level, so it is difficult to answer the question.”
Another delegate asked what the participants thought about delaying MTX by 1 month. “When we tell the patient we need to vaccinate before we can use methotrexate [because] otherwise, we will reduce the response to the vaccination, then patient accepts it,” said Dr. Morel, adding that, “we allowed a minimal dose of steroids, and we saw from the results that the DAS28 at 1 month had changed.”
Co-moderator Katerina Chatzidionysiou, MD, PhD, a consultant rheumatologist and head of the Clinical Trial Department Rheumatology Unit, Karolinska University Hospital, Stockholm, Sweden, said that “As a physician, I’d feel uncomfortable delaying MTX if they had very active disease even for a short period of time.”
Dr. Morel replied that, “Today, we have so many drugs that can control the disease, for example, the targeted DMARDs. Progression does not show much variation, and we know x-ray progression with today’s drugs is a lot less than previously.”
Dr. Morel reported financial relationships with AbbVie, Amgen, Biogen, Bristol Myers Squibb, Fresenius Kabi, Galapagos, GlaxoSmithKline, Lilly, Medac, Nordic Pharma, Novartis, Pfizer, Sandoz, Sanofi, Boehringer Ingelheim, and Servier. Dr. Choy had no relevant financial relationships of relevance to this study.
A version of this article appeared on Medscape.com.
FROM EULAR 2024
Predicting and Understanding Vaccine Response Determinants
In this column, I recently discussed the impact of the microbiome on childhood vaccine responses. My group has been expanding our research on the topic of childhood vaccine response and its relationship to infection proneness. Therefore, I want to share new research findings.
Immune responsiveness to vaccines varies among children, leaving some susceptible to infections. We also have evidence that the immune deficiencies that contribute to poor vaccine responsiveness also manifest in children as respiratory infection proneness.
Predicting Vaccine Response in the Neonatal Period
The first 100 days of life is an amazing transition time in early life. During that time, the immune system is highly influenced by environmental factors that generate epigenetic changes affecting vaccine responsiveness. Some publications have used the term “window of opportunity,” because it is thought that interventions to change a negative trajectory to a positive one for vaccine responsiveness have a better potential to be effective. Predicting which children will be poorly responsive to vaccines would be desirable, so those children could be specifically identified for intervention. Doing so in the neonatal age time frame using easy-to-obtain clinical samples would be a bonus.
In our most recent study, we sought to identify cytokine biosignatures in the neonatal period, measured in convenient nasopharyngeal secretions, that predict vaccine responses, measured as antibody levels to various vaccines at 1 year of life. Secondly, we assessed the effect of antibiotic exposures on vaccine responses in the study cohort. Third, we tested for induction of CD4+ T-cell vaccine-specific immune memory at infant age 1 year. Fourth, we studied antigen presenting cells (APCs) at rest and in response to an adjuvant called R848, known to stimulate toll-like receptor (TLR) 7/8 agonist, to assess its effects on the immune cells of low vaccine responder children, compared with other children.1
The study population consisted of 101 infants recruited from two primary care pediatric practices in/near Rochester, New York. Children lived in suburban and rural environments. Enrollment and sampling occurred during 2017-2020. All participants received regularly scheduled childhood vaccinations according to the recommendations by US Centers for Disease Control. Nasopharyngeal swabs were used to collect nasal secretions. Antibody titers against six antigens were measured at approximately 1 year of age from all 72 available blood samples. The protective threshold of the corresponding vaccine antigen divided each vaccine-induced antibody level and the ratio considered a normalized titer. The normalized antibody titers were used to define vaccine responsiveness groups as Low Vaccine Responder (bottom 25th percentile of vaccine responders, n = 18 children), as Normal Vaccine Responder (25-75th percentile of vaccine responders, n = 36 children) and as High Vaccine Responder (top 25th percentile of vaccine responders, n = 18 children).
We found that specific nasal cytokine levels measured at newborn age 1 week old, 2 weeks old, and 3 weeks old were predictive of the vaccine response groupings measured at child age 1 year old, following their primary series of vaccinations. The P values varied between less than .05 to .001.
Five newborns had antibiotic exposure at/near the time of birth; 4 [80%] of the 5 were Low Vaccine Responders vs 1 [2%] of 60 Normal+High Vaccine Responder children, P = .006. Also, the cumulative days of antibiotic exposure up to 1 year was highly associated with low vaccine responders, compared with Normal+High Vaccine Responder children (P = 2 x 10-16).
We found that Low Vaccine Responder infants had reduced vaccine-specific T-helper memory cells producing INFg and IL-2 (Th1 cytokines) and IL-4 (Th2 cytokines), compared with Normal+High Vaccine Responder children. In the absence of sufficient numbers of antigen-specific memory CD4+ T-cells, a child would become unprotected from the target infection that the vaccines were intended to prevent after the antibody levels wane.
We found that Low Vaccine Responder antigen-presenting cells are different from those in normal vaccine responders and they can be distinguished when at rest and when stimulated by a specific adjuvant — R848. Our previous findings suggested that Low Vaccine Responder children have a prolonged neonatal-like immune profile (PNIP).2 Therefore, stimulating the immune system of a Low Vaccine Responder could shift their cellular immune responses to behave like cells of Normal+High Vaccine Responder children.
In summary, we identified cytokine biosignatures measured in nasopharyngeal secretions in the neonatal period that predicted vaccine response groups measured as antibody levels at 1 year of life. We showed that reduced vaccine responsiveness was associated with antibiotic exposure at/near birth and with cumulative exposure during the first year of life. We found that Low Vaccine Responder children at 1 year old have fewer vaccine-specific memory CD4+ Th1 and Th2-cells and that antigen-presenting cells at rest and in response to R848 antigen stimulation differ, compared with Normal+High Vaccine Responder children.
Future work by our group will focus on exploring early-life risk factors that influence differences in vaccine responsiveness and interventions that might shift a child’s responsiveness from low to normal or high.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (New York) General Hospital. He has no conflicts of interest to declare.
References
1. Pichichero ME et al. Variability of Vaccine Responsiveness in Young Children. J Infect Dis. 2023 Nov 22:jiad524. doi: 10.1093/infdis/jiad524.
2. Pichichero ME et al. Functional Immune Cell Differences Associated with Low Vaccine Responses in Infants. J Infect Dis. 2016 Jun 15;213(12):2014-2019. doi: 10.1093/infdis/jiw053.
In this column, I recently discussed the impact of the microbiome on childhood vaccine responses. My group has been expanding our research on the topic of childhood vaccine response and its relationship to infection proneness. Therefore, I want to share new research findings.
Immune responsiveness to vaccines varies among children, leaving some susceptible to infections. We also have evidence that the immune deficiencies that contribute to poor vaccine responsiveness also manifest in children as respiratory infection proneness.
Predicting Vaccine Response in the Neonatal Period
The first 100 days of life is an amazing transition time in early life. During that time, the immune system is highly influenced by environmental factors that generate epigenetic changes affecting vaccine responsiveness. Some publications have used the term “window of opportunity,” because it is thought that interventions to change a negative trajectory to a positive one for vaccine responsiveness have a better potential to be effective. Predicting which children will be poorly responsive to vaccines would be desirable, so those children could be specifically identified for intervention. Doing so in the neonatal age time frame using easy-to-obtain clinical samples would be a bonus.
In our most recent study, we sought to identify cytokine biosignatures in the neonatal period, measured in convenient nasopharyngeal secretions, that predict vaccine responses, measured as antibody levels to various vaccines at 1 year of life. Secondly, we assessed the effect of antibiotic exposures on vaccine responses in the study cohort. Third, we tested for induction of CD4+ T-cell vaccine-specific immune memory at infant age 1 year. Fourth, we studied antigen presenting cells (APCs) at rest and in response to an adjuvant called R848, known to stimulate toll-like receptor (TLR) 7/8 agonist, to assess its effects on the immune cells of low vaccine responder children, compared with other children.1
The study population consisted of 101 infants recruited from two primary care pediatric practices in/near Rochester, New York. Children lived in suburban and rural environments. Enrollment and sampling occurred during 2017-2020. All participants received regularly scheduled childhood vaccinations according to the recommendations by US Centers for Disease Control. Nasopharyngeal swabs were used to collect nasal secretions. Antibody titers against six antigens were measured at approximately 1 year of age from all 72 available blood samples. The protective threshold of the corresponding vaccine antigen divided each vaccine-induced antibody level and the ratio considered a normalized titer. The normalized antibody titers were used to define vaccine responsiveness groups as Low Vaccine Responder (bottom 25th percentile of vaccine responders, n = 18 children), as Normal Vaccine Responder (25-75th percentile of vaccine responders, n = 36 children) and as High Vaccine Responder (top 25th percentile of vaccine responders, n = 18 children).
We found that specific nasal cytokine levels measured at newborn age 1 week old, 2 weeks old, and 3 weeks old were predictive of the vaccine response groupings measured at child age 1 year old, following their primary series of vaccinations. The P values varied between less than .05 to .001.
Five newborns had antibiotic exposure at/near the time of birth; 4 [80%] of the 5 were Low Vaccine Responders vs 1 [2%] of 60 Normal+High Vaccine Responder children, P = .006. Also, the cumulative days of antibiotic exposure up to 1 year was highly associated with low vaccine responders, compared with Normal+High Vaccine Responder children (P = 2 x 10-16).
We found that Low Vaccine Responder infants had reduced vaccine-specific T-helper memory cells producing INFg and IL-2 (Th1 cytokines) and IL-4 (Th2 cytokines), compared with Normal+High Vaccine Responder children. In the absence of sufficient numbers of antigen-specific memory CD4+ T-cells, a child would become unprotected from the target infection that the vaccines were intended to prevent after the antibody levels wane.
We found that Low Vaccine Responder antigen-presenting cells are different from those in normal vaccine responders and they can be distinguished when at rest and when stimulated by a specific adjuvant — R848. Our previous findings suggested that Low Vaccine Responder children have a prolonged neonatal-like immune profile (PNIP).2 Therefore, stimulating the immune system of a Low Vaccine Responder could shift their cellular immune responses to behave like cells of Normal+High Vaccine Responder children.
In summary, we identified cytokine biosignatures measured in nasopharyngeal secretions in the neonatal period that predicted vaccine response groups measured as antibody levels at 1 year of life. We showed that reduced vaccine responsiveness was associated with antibiotic exposure at/near birth and with cumulative exposure during the first year of life. We found that Low Vaccine Responder children at 1 year old have fewer vaccine-specific memory CD4+ Th1 and Th2-cells and that antigen-presenting cells at rest and in response to R848 antigen stimulation differ, compared with Normal+High Vaccine Responder children.
Future work by our group will focus on exploring early-life risk factors that influence differences in vaccine responsiveness and interventions that might shift a child’s responsiveness from low to normal or high.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (New York) General Hospital. He has no conflicts of interest to declare.
References
1. Pichichero ME et al. Variability of Vaccine Responsiveness in Young Children. J Infect Dis. 2023 Nov 22:jiad524. doi: 10.1093/infdis/jiad524.
2. Pichichero ME et al. Functional Immune Cell Differences Associated with Low Vaccine Responses in Infants. J Infect Dis. 2016 Jun 15;213(12):2014-2019. doi: 10.1093/infdis/jiw053.
In this column, I recently discussed the impact of the microbiome on childhood vaccine responses. My group has been expanding our research on the topic of childhood vaccine response and its relationship to infection proneness. Therefore, I want to share new research findings.
Immune responsiveness to vaccines varies among children, leaving some susceptible to infections. We also have evidence that the immune deficiencies that contribute to poor vaccine responsiveness also manifest in children as respiratory infection proneness.
Predicting Vaccine Response in the Neonatal Period
The first 100 days of life is an amazing transition time in early life. During that time, the immune system is highly influenced by environmental factors that generate epigenetic changes affecting vaccine responsiveness. Some publications have used the term “window of opportunity,” because it is thought that interventions to change a negative trajectory to a positive one for vaccine responsiveness have a better potential to be effective. Predicting which children will be poorly responsive to vaccines would be desirable, so those children could be specifically identified for intervention. Doing so in the neonatal age time frame using easy-to-obtain clinical samples would be a bonus.
In our most recent study, we sought to identify cytokine biosignatures in the neonatal period, measured in convenient nasopharyngeal secretions, that predict vaccine responses, measured as antibody levels to various vaccines at 1 year of life. Secondly, we assessed the effect of antibiotic exposures on vaccine responses in the study cohort. Third, we tested for induction of CD4+ T-cell vaccine-specific immune memory at infant age 1 year. Fourth, we studied antigen presenting cells (APCs) at rest and in response to an adjuvant called R848, known to stimulate toll-like receptor (TLR) 7/8 agonist, to assess its effects on the immune cells of low vaccine responder children, compared with other children.1
The study population consisted of 101 infants recruited from two primary care pediatric practices in/near Rochester, New York. Children lived in suburban and rural environments. Enrollment and sampling occurred during 2017-2020. All participants received regularly scheduled childhood vaccinations according to the recommendations by US Centers for Disease Control. Nasopharyngeal swabs were used to collect nasal secretions. Antibody titers against six antigens were measured at approximately 1 year of age from all 72 available blood samples. The protective threshold of the corresponding vaccine antigen divided each vaccine-induced antibody level and the ratio considered a normalized titer. The normalized antibody titers were used to define vaccine responsiveness groups as Low Vaccine Responder (bottom 25th percentile of vaccine responders, n = 18 children), as Normal Vaccine Responder (25-75th percentile of vaccine responders, n = 36 children) and as High Vaccine Responder (top 25th percentile of vaccine responders, n = 18 children).
We found that specific nasal cytokine levels measured at newborn age 1 week old, 2 weeks old, and 3 weeks old were predictive of the vaccine response groupings measured at child age 1 year old, following their primary series of vaccinations. The P values varied between less than .05 to .001.
Five newborns had antibiotic exposure at/near the time of birth; 4 [80%] of the 5 were Low Vaccine Responders vs 1 [2%] of 60 Normal+High Vaccine Responder children, P = .006. Also, the cumulative days of antibiotic exposure up to 1 year was highly associated with low vaccine responders, compared with Normal+High Vaccine Responder children (P = 2 x 10-16).
We found that Low Vaccine Responder infants had reduced vaccine-specific T-helper memory cells producing INFg and IL-2 (Th1 cytokines) and IL-4 (Th2 cytokines), compared with Normal+High Vaccine Responder children. In the absence of sufficient numbers of antigen-specific memory CD4+ T-cells, a child would become unprotected from the target infection that the vaccines were intended to prevent after the antibody levels wane.
We found that Low Vaccine Responder antigen-presenting cells are different from those in normal vaccine responders and they can be distinguished when at rest and when stimulated by a specific adjuvant — R848. Our previous findings suggested that Low Vaccine Responder children have a prolonged neonatal-like immune profile (PNIP).2 Therefore, stimulating the immune system of a Low Vaccine Responder could shift their cellular immune responses to behave like cells of Normal+High Vaccine Responder children.
In summary, we identified cytokine biosignatures measured in nasopharyngeal secretions in the neonatal period that predicted vaccine response groups measured as antibody levels at 1 year of life. We showed that reduced vaccine responsiveness was associated with antibiotic exposure at/near birth and with cumulative exposure during the first year of life. We found that Low Vaccine Responder children at 1 year old have fewer vaccine-specific memory CD4+ Th1 and Th2-cells and that antigen-presenting cells at rest and in response to R848 antigen stimulation differ, compared with Normal+High Vaccine Responder children.
Future work by our group will focus on exploring early-life risk factors that influence differences in vaccine responsiveness and interventions that might shift a child’s responsiveness from low to normal or high.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (New York) General Hospital. He has no conflicts of interest to declare.
References
1. Pichichero ME et al. Variability of Vaccine Responsiveness in Young Children. J Infect Dis. 2023 Nov 22:jiad524. doi: 10.1093/infdis/jiad524.
2. Pichichero ME et al. Functional Immune Cell Differences Associated with Low Vaccine Responses in Infants. J Infect Dis. 2016 Jun 15;213(12):2014-2019. doi: 10.1093/infdis/jiw053.
Could British Columbia Eliminate Cervical Cancer by 2031?
To achieve this goal, the province will also need to reach historically underscreened, equity-seeking populations (ie, Black, indigenous, immigrant, LGBTQ, and disabled patients, and those with sexual trauma) through mailed self-screening HPV tests.
The adoption of both these strategies is essential, according to a modeling study that was published on June 3 in CMAJ, especially because the true impact of HPV vaccination has yet to be fully realized.
“In BC, we have a school-based program to increase vaccine coverage in boys and girls starting in grade 6,” study author Reka Pataky, PhD, a senior research health economist at the Canadian Centre for Applied Research in Cancer Control and BC Cancer in Vancouver, British Columbia, Canada, told this news organization. Dr. Pataky noted that this immunization program was launched in 2008 and that some of the initial cohorts haven›t yet reached the average age of diagnosis, which is between 30 and 59 years.
Three’s a Charm
The investigators undertook a modeling study to determine when and how BC might achieve the elimination of cervical cancer following a transition to HPV-based screening. Elimination was defined as an annual age-standardized incidence rate of < 4.0 per 100,000 women.
Modeling scenarios were developed using the Canadian Partnership Against Cancer’s priority targets, which include increasing HPV vaccination through school-based coverage from 70% to 90%, increasing the probability of ever receiving a screening test from 90% to 95%, increasing the rate of on-time screening from 70% to 90%, and improving follow-up to 95% for colposcopy (currently 88%) and HPV testing (currently 80%). Modeling simulated HPV transmission and the natural history of cervical cancer in the Canadian population and relied upon two reference scenarios: One using BC’s cytology-based screening at the time of analysis, and the other an HPV base-case scenario.
The researchers found that with the status quo (ie, cytology-based screening and no change to vaccination or screening participation rates), BC would not eliminate cervical cancer until 2045. Implementation of HPV-based screening at the current 70% participation rate would achieve elimination in 2034 and prevent 942 cases compared with cytology screening. Increasing the proportion of patients who were ever screened or increasing vaccination coverage would result in cervical cancer elimination by 2033. The time line would be shortened even further (to 2031) through a combination of three strategies (ie, improving recruitment, on-time screening, and follow-up compliance).
Low Incidence, Strained System
The incidence of cervical cancer in Canada is relatively low, accounting for 1.3% of all new female cancers and 1.1% of all female cancer deaths.
“The reason that we have such low rates is because we have organized screening programs,” explained Rachel Kupets, MD, associate professor of gynecologic oncology at the University of Toronto and Sunnybrook Hospital, Toronto. She was not involved in the study.
“We’re starting to see what happens when the system gets strained with lower participation rates. I am starting to see a lot more women with invasive cervical cancer. They’re younger, and their cancers are less curable and less treatable,” she said.
Difficulties with access, interest, and education have contributed to low cervical screening rates among equity-seeking populations, according to Dr. Pataky and Dr. Kupets.
“Self-screening is another tool that can incrementally benefit those folks who wouldn’t otherwise undergo screening or don’t want an invasive test,” said Dr. Kupets. It can also play an increasing role, while current access to primary care services in Canada is at an all-time low. Community outreach through centers, mobile coaches, and nursing stations might help ensure participation by at-risk populations. These measures also could boost follow-up for and education about positive results, said Dr. Kupets.
In a related editorial, Shannon Charlebois, MD, medical editor of CMAJ, and Sarah Kean, MD, assistant professor of gynecologic oncology at the University of Manitoba in Winnipeg, Manitoba, Canada, emphasized the need for mailed HPV self-screening kits to be paid for and integrated into provincial cervical cancer screening programs across Canada to support earlier cervical cancer detection and lower invasive cancer rates.
Dr. Pataky concurred. “There have been discussions about making the big transition from traditional cytology to implementing HPV self-screening,” she said. “We have really effective tools for preventing cervical cancer, and it’s important to not lose sight of that goal.”
The study was funded by the National Institutes of Health. Dr. Pataky and Dr. Kupets reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
To achieve this goal, the province will also need to reach historically underscreened, equity-seeking populations (ie, Black, indigenous, immigrant, LGBTQ, and disabled patients, and those with sexual trauma) through mailed self-screening HPV tests.
The adoption of both these strategies is essential, according to a modeling study that was published on June 3 in CMAJ, especially because the true impact of HPV vaccination has yet to be fully realized.
“In BC, we have a school-based program to increase vaccine coverage in boys and girls starting in grade 6,” study author Reka Pataky, PhD, a senior research health economist at the Canadian Centre for Applied Research in Cancer Control and BC Cancer in Vancouver, British Columbia, Canada, told this news organization. Dr. Pataky noted that this immunization program was launched in 2008 and that some of the initial cohorts haven›t yet reached the average age of diagnosis, which is between 30 and 59 years.
Three’s a Charm
The investigators undertook a modeling study to determine when and how BC might achieve the elimination of cervical cancer following a transition to HPV-based screening. Elimination was defined as an annual age-standardized incidence rate of < 4.0 per 100,000 women.
Modeling scenarios were developed using the Canadian Partnership Against Cancer’s priority targets, which include increasing HPV vaccination through school-based coverage from 70% to 90%, increasing the probability of ever receiving a screening test from 90% to 95%, increasing the rate of on-time screening from 70% to 90%, and improving follow-up to 95% for colposcopy (currently 88%) and HPV testing (currently 80%). Modeling simulated HPV transmission and the natural history of cervical cancer in the Canadian population and relied upon two reference scenarios: One using BC’s cytology-based screening at the time of analysis, and the other an HPV base-case scenario.
The researchers found that with the status quo (ie, cytology-based screening and no change to vaccination or screening participation rates), BC would not eliminate cervical cancer until 2045. Implementation of HPV-based screening at the current 70% participation rate would achieve elimination in 2034 and prevent 942 cases compared with cytology screening. Increasing the proportion of patients who were ever screened or increasing vaccination coverage would result in cervical cancer elimination by 2033. The time line would be shortened even further (to 2031) through a combination of three strategies (ie, improving recruitment, on-time screening, and follow-up compliance).
Low Incidence, Strained System
The incidence of cervical cancer in Canada is relatively low, accounting for 1.3% of all new female cancers and 1.1% of all female cancer deaths.
“The reason that we have such low rates is because we have organized screening programs,” explained Rachel Kupets, MD, associate professor of gynecologic oncology at the University of Toronto and Sunnybrook Hospital, Toronto. She was not involved in the study.
“We’re starting to see what happens when the system gets strained with lower participation rates. I am starting to see a lot more women with invasive cervical cancer. They’re younger, and their cancers are less curable and less treatable,” she said.
Difficulties with access, interest, and education have contributed to low cervical screening rates among equity-seeking populations, according to Dr. Pataky and Dr. Kupets.
“Self-screening is another tool that can incrementally benefit those folks who wouldn’t otherwise undergo screening or don’t want an invasive test,” said Dr. Kupets. It can also play an increasing role, while current access to primary care services in Canada is at an all-time low. Community outreach through centers, mobile coaches, and nursing stations might help ensure participation by at-risk populations. These measures also could boost follow-up for and education about positive results, said Dr. Kupets.
In a related editorial, Shannon Charlebois, MD, medical editor of CMAJ, and Sarah Kean, MD, assistant professor of gynecologic oncology at the University of Manitoba in Winnipeg, Manitoba, Canada, emphasized the need for mailed HPV self-screening kits to be paid for and integrated into provincial cervical cancer screening programs across Canada to support earlier cervical cancer detection and lower invasive cancer rates.
Dr. Pataky concurred. “There have been discussions about making the big transition from traditional cytology to implementing HPV self-screening,” she said. “We have really effective tools for preventing cervical cancer, and it’s important to not lose sight of that goal.”
The study was funded by the National Institutes of Health. Dr. Pataky and Dr. Kupets reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
To achieve this goal, the province will also need to reach historically underscreened, equity-seeking populations (ie, Black, indigenous, immigrant, LGBTQ, and disabled patients, and those with sexual trauma) through mailed self-screening HPV tests.
The adoption of both these strategies is essential, according to a modeling study that was published on June 3 in CMAJ, especially because the true impact of HPV vaccination has yet to be fully realized.
“In BC, we have a school-based program to increase vaccine coverage in boys and girls starting in grade 6,” study author Reka Pataky, PhD, a senior research health economist at the Canadian Centre for Applied Research in Cancer Control and BC Cancer in Vancouver, British Columbia, Canada, told this news organization. Dr. Pataky noted that this immunization program was launched in 2008 and that some of the initial cohorts haven›t yet reached the average age of diagnosis, which is between 30 and 59 years.
Three’s a Charm
The investigators undertook a modeling study to determine when and how BC might achieve the elimination of cervical cancer following a transition to HPV-based screening. Elimination was defined as an annual age-standardized incidence rate of < 4.0 per 100,000 women.
Modeling scenarios were developed using the Canadian Partnership Against Cancer’s priority targets, which include increasing HPV vaccination through school-based coverage from 70% to 90%, increasing the probability of ever receiving a screening test from 90% to 95%, increasing the rate of on-time screening from 70% to 90%, and improving follow-up to 95% for colposcopy (currently 88%) and HPV testing (currently 80%). Modeling simulated HPV transmission and the natural history of cervical cancer in the Canadian population and relied upon two reference scenarios: One using BC’s cytology-based screening at the time of analysis, and the other an HPV base-case scenario.
The researchers found that with the status quo (ie, cytology-based screening and no change to vaccination or screening participation rates), BC would not eliminate cervical cancer until 2045. Implementation of HPV-based screening at the current 70% participation rate would achieve elimination in 2034 and prevent 942 cases compared with cytology screening. Increasing the proportion of patients who were ever screened or increasing vaccination coverage would result in cervical cancer elimination by 2033. The time line would be shortened even further (to 2031) through a combination of three strategies (ie, improving recruitment, on-time screening, and follow-up compliance).
Low Incidence, Strained System
The incidence of cervical cancer in Canada is relatively low, accounting for 1.3% of all new female cancers and 1.1% of all female cancer deaths.
“The reason that we have such low rates is because we have organized screening programs,” explained Rachel Kupets, MD, associate professor of gynecologic oncology at the University of Toronto and Sunnybrook Hospital, Toronto. She was not involved in the study.
“We’re starting to see what happens when the system gets strained with lower participation rates. I am starting to see a lot more women with invasive cervical cancer. They’re younger, and their cancers are less curable and less treatable,” she said.
Difficulties with access, interest, and education have contributed to low cervical screening rates among equity-seeking populations, according to Dr. Pataky and Dr. Kupets.
“Self-screening is another tool that can incrementally benefit those folks who wouldn’t otherwise undergo screening or don’t want an invasive test,” said Dr. Kupets. It can also play an increasing role, while current access to primary care services in Canada is at an all-time low. Community outreach through centers, mobile coaches, and nursing stations might help ensure participation by at-risk populations. These measures also could boost follow-up for and education about positive results, said Dr. Kupets.
In a related editorial, Shannon Charlebois, MD, medical editor of CMAJ, and Sarah Kean, MD, assistant professor of gynecologic oncology at the University of Manitoba in Winnipeg, Manitoba, Canada, emphasized the need for mailed HPV self-screening kits to be paid for and integrated into provincial cervical cancer screening programs across Canada to support earlier cervical cancer detection and lower invasive cancer rates.
Dr. Pataky concurred. “There have been discussions about making the big transition from traditional cytology to implementing HPV self-screening,” she said. “We have really effective tools for preventing cervical cancer, and it’s important to not lose sight of that goal.”
The study was funded by the National Institutes of Health. Dr. Pataky and Dr. Kupets reported no relevant financial relationships.
A version of this article appeared on Medscape.com.