User login
Biologics, Women, and Pregnancy: What’s Known?
As the and the child’s development.
“I get asked a lot about fertility,” Vivian Shi, MD, associate professor of dermatology at the University of Arkansas, Little Rock, said at MedscapeLive’s Women’s and Pediatric Dermatology Seminar. Patients want to know, she said, if they go on a specific drug, whether it will affect their chances of conceiving and what else they need to know about safety.
She told the audience what she tells her patients: The answers are not complete but are evolving at a steady pace.
“Putting this talk together was kind of like a scavenger hunt,” said Dr. Shi, who gathered data from pregnancy exposure registries, published research, the Food and Drug Administration, and other sources on biologics. As more studies emerge each year, she said, recommendations will become stronger for considering treatment by certain biological drugs, taking into account effects on fertility, pregnancy, lactation, and the infant.
Among the biologics commonly used in dermatology are:
- Tumor necrosis factor (TNF) inhibitors (etanercept, adalimumab, infliximab, certolizumab).
- Interleukin (IL)–12 and -23 antagonist (ustekinumab).
- IL-17 antagonists (ixekizumab, secukinumab, brodalumab).
- IL-23 antagonists (risankizumab, tildrakizumab, guselkumab).
- IL-4, -13 antagonist (dupilumab) and IL-13 antagonist (tralokinumab).
- CD20-directed cytolytic antibody (rituximab).
To help with decision-making, Dr. Shi discussed the relatively new FDA labeling regulations as well as pregnancy exposure registries, research studies, and recommendations.
FDA pregnancy risk summaries
Under the previous system of classification of drugs in pregnancy, the FDA rated drugs as A, B, C, D, X. These categories ranged from showing no risks to the fetus to clear risk, but were oversimplistic and confusing, Dr. Shi said. Category C was especially confusing, as a drug with no animal or human data was put in the same category as a drug with adverse fetal effects on animals, she noted.
However, effective June 30, 2015, the FDA replaced pregnancy categories with risk summaries by medication. As of June, 2020, all prescription drugs were to remove pregnancy letter labeling. The risk summaries note human data when they are available and also note when no data are available. This information, Dr. Shi said, originates from many sources, including studies published in the medical literature, postmarketing studies conducted by companies, and pregnancy exposure registries, conducted by some companies and others. The FDA does not endorse any specific registries, but does post a list of such registries. Another helpful resource, she said, is Mother to Baby, a service of the nonprofit Organization of Teratology Information Specialists (OTIS).
Known, not known
Citing published literature, Dr. Shi said that TNF inhibitors have the most robust safety data from preconception to after birth. Less is known, she said, about the reproductive safety effects of other biologics used for dermatologic conditions, as they are newer than the anti-TNF medicines.
She reviewed a variety of research studies evaluating the safety of biologics during pregnancy and beyond. Highlights include results from a large registry, the Psoriasis Longitudinal Assessment and Registry (PSOLAR), of 298 pregnancies in about 220 women from 2007 to 2019, looking at 13 different biologics. The overall and live-birth outcomes in the women on biologics for psoriasis were similar to those for the general population and the rate of congenital anomalies was 0.8%, researchers reported in 2021, lower than the generally cited annual figure of U.S. births.
Studies evaluating biologics for nondermatologic conditions suggest safety. A prospective cohort study of women who took adalimumab in pregnancy (for rheumatoid arthritis or Crohn’s disease) found no increased risk for birth defects. In another study looking at women who were breastfeeding, researchers found no increased risk of infections or delay in developmental milestones in the children of women taking biologics for inflammatory bowel disease, compared with those not on the medications.
A report using data from the World Health Organization concludes that dupilumab appears to be safe during pregnancy, based on an evaluation of 36 pregnancy-related reports among more than 37,000 unique adverse event reports related to dupilumab in a global database.
Recommendations about biologic use from different organizations don’t always mesh, Dr. Shi said, noting that European guidelines tend to be stricter, as some reviews show.
If a mother is exposed to any biologic therapy other than certolizumab during the third trimester, after 27 weeks, Dr. Shi said, “you want to consider avoiding a live vaccine for the first 6 months of the baby’s life.” It turns out, she said, the only recommended live vaccine during that period is the rotavirus vaccine, and she suggests doctors recommend postponing that one until the babies are older if women have been on biologics other than certolizumab.
Her other take-home messages: TNF inhibitors have the most robust safety data from before conception through lactation. Under current guidelines, certolizumab is viewed as the safest to use throughout pregnancy. Dr. Shi’s message to her colleagues fielding the same questions she gets from patients: “There is more data coming out every year. Ultimately, we will have better information to inform our patients.”
At the conference, Lawrence F. Eichenfield, MD, a course director and professor of dermatology and pediatrics at the University of California, San Diego and Rady Children’s Hospital San Diego, encouraged Dr. Shi to write up her presentation as a resource for other dermatologists – which she said is in progress.
Medscape Live and this news organization are owned by the same parent company. Dr. Shi disclosed consulting and investigative and research funding from several pharmaceutical firms, but not directly related to the content of her presentation.
As the and the child’s development.
“I get asked a lot about fertility,” Vivian Shi, MD, associate professor of dermatology at the University of Arkansas, Little Rock, said at MedscapeLive’s Women’s and Pediatric Dermatology Seminar. Patients want to know, she said, if they go on a specific drug, whether it will affect their chances of conceiving and what else they need to know about safety.
She told the audience what she tells her patients: The answers are not complete but are evolving at a steady pace.
“Putting this talk together was kind of like a scavenger hunt,” said Dr. Shi, who gathered data from pregnancy exposure registries, published research, the Food and Drug Administration, and other sources on biologics. As more studies emerge each year, she said, recommendations will become stronger for considering treatment by certain biological drugs, taking into account effects on fertility, pregnancy, lactation, and the infant.
Among the biologics commonly used in dermatology are:
- Tumor necrosis factor (TNF) inhibitors (etanercept, adalimumab, infliximab, certolizumab).
- Interleukin (IL)–12 and -23 antagonist (ustekinumab).
- IL-17 antagonists (ixekizumab, secukinumab, brodalumab).
- IL-23 antagonists (risankizumab, tildrakizumab, guselkumab).
- IL-4, -13 antagonist (dupilumab) and IL-13 antagonist (tralokinumab).
- CD20-directed cytolytic antibody (rituximab).
To help with decision-making, Dr. Shi discussed the relatively new FDA labeling regulations as well as pregnancy exposure registries, research studies, and recommendations.
FDA pregnancy risk summaries
Under the previous system of classification of drugs in pregnancy, the FDA rated drugs as A, B, C, D, X. These categories ranged from showing no risks to the fetus to clear risk, but were oversimplistic and confusing, Dr. Shi said. Category C was especially confusing, as a drug with no animal or human data was put in the same category as a drug with adverse fetal effects on animals, she noted.
However, effective June 30, 2015, the FDA replaced pregnancy categories with risk summaries by medication. As of June, 2020, all prescription drugs were to remove pregnancy letter labeling. The risk summaries note human data when they are available and also note when no data are available. This information, Dr. Shi said, originates from many sources, including studies published in the medical literature, postmarketing studies conducted by companies, and pregnancy exposure registries, conducted by some companies and others. The FDA does not endorse any specific registries, but does post a list of such registries. Another helpful resource, she said, is Mother to Baby, a service of the nonprofit Organization of Teratology Information Specialists (OTIS).
Known, not known
Citing published literature, Dr. Shi said that TNF inhibitors have the most robust safety data from preconception to after birth. Less is known, she said, about the reproductive safety effects of other biologics used for dermatologic conditions, as they are newer than the anti-TNF medicines.
She reviewed a variety of research studies evaluating the safety of biologics during pregnancy and beyond. Highlights include results from a large registry, the Psoriasis Longitudinal Assessment and Registry (PSOLAR), of 298 pregnancies in about 220 women from 2007 to 2019, looking at 13 different biologics. The overall and live-birth outcomes in the women on biologics for psoriasis were similar to those for the general population and the rate of congenital anomalies was 0.8%, researchers reported in 2021, lower than the generally cited annual figure of U.S. births.
Studies evaluating biologics for nondermatologic conditions suggest safety. A prospective cohort study of women who took adalimumab in pregnancy (for rheumatoid arthritis or Crohn’s disease) found no increased risk for birth defects. In another study looking at women who were breastfeeding, researchers found no increased risk of infections or delay in developmental milestones in the children of women taking biologics for inflammatory bowel disease, compared with those not on the medications.
A report using data from the World Health Organization concludes that dupilumab appears to be safe during pregnancy, based on an evaluation of 36 pregnancy-related reports among more than 37,000 unique adverse event reports related to dupilumab in a global database.
Recommendations about biologic use from different organizations don’t always mesh, Dr. Shi said, noting that European guidelines tend to be stricter, as some reviews show.
If a mother is exposed to any biologic therapy other than certolizumab during the third trimester, after 27 weeks, Dr. Shi said, “you want to consider avoiding a live vaccine for the first 6 months of the baby’s life.” It turns out, she said, the only recommended live vaccine during that period is the rotavirus vaccine, and she suggests doctors recommend postponing that one until the babies are older if women have been on biologics other than certolizumab.
Her other take-home messages: TNF inhibitors have the most robust safety data from before conception through lactation. Under current guidelines, certolizumab is viewed as the safest to use throughout pregnancy. Dr. Shi’s message to her colleagues fielding the same questions she gets from patients: “There is more data coming out every year. Ultimately, we will have better information to inform our patients.”
At the conference, Lawrence F. Eichenfield, MD, a course director and professor of dermatology and pediatrics at the University of California, San Diego and Rady Children’s Hospital San Diego, encouraged Dr. Shi to write up her presentation as a resource for other dermatologists – which she said is in progress.
Medscape Live and this news organization are owned by the same parent company. Dr. Shi disclosed consulting and investigative and research funding from several pharmaceutical firms, but not directly related to the content of her presentation.
As the and the child’s development.
“I get asked a lot about fertility,” Vivian Shi, MD, associate professor of dermatology at the University of Arkansas, Little Rock, said at MedscapeLive’s Women’s and Pediatric Dermatology Seminar. Patients want to know, she said, if they go on a specific drug, whether it will affect their chances of conceiving and what else they need to know about safety.
She told the audience what she tells her patients: The answers are not complete but are evolving at a steady pace.
“Putting this talk together was kind of like a scavenger hunt,” said Dr. Shi, who gathered data from pregnancy exposure registries, published research, the Food and Drug Administration, and other sources on biologics. As more studies emerge each year, she said, recommendations will become stronger for considering treatment by certain biological drugs, taking into account effects on fertility, pregnancy, lactation, and the infant.
Among the biologics commonly used in dermatology are:
- Tumor necrosis factor (TNF) inhibitors (etanercept, adalimumab, infliximab, certolizumab).
- Interleukin (IL)–12 and -23 antagonist (ustekinumab).
- IL-17 antagonists (ixekizumab, secukinumab, brodalumab).
- IL-23 antagonists (risankizumab, tildrakizumab, guselkumab).
- IL-4, -13 antagonist (dupilumab) and IL-13 antagonist (tralokinumab).
- CD20-directed cytolytic antibody (rituximab).
To help with decision-making, Dr. Shi discussed the relatively new FDA labeling regulations as well as pregnancy exposure registries, research studies, and recommendations.
FDA pregnancy risk summaries
Under the previous system of classification of drugs in pregnancy, the FDA rated drugs as A, B, C, D, X. These categories ranged from showing no risks to the fetus to clear risk, but were oversimplistic and confusing, Dr. Shi said. Category C was especially confusing, as a drug with no animal or human data was put in the same category as a drug with adverse fetal effects on animals, she noted.
However, effective June 30, 2015, the FDA replaced pregnancy categories with risk summaries by medication. As of June, 2020, all prescription drugs were to remove pregnancy letter labeling. The risk summaries note human data when they are available and also note when no data are available. This information, Dr. Shi said, originates from many sources, including studies published in the medical literature, postmarketing studies conducted by companies, and pregnancy exposure registries, conducted by some companies and others. The FDA does not endorse any specific registries, but does post a list of such registries. Another helpful resource, she said, is Mother to Baby, a service of the nonprofit Organization of Teratology Information Specialists (OTIS).
Known, not known
Citing published literature, Dr. Shi said that TNF inhibitors have the most robust safety data from preconception to after birth. Less is known, she said, about the reproductive safety effects of other biologics used for dermatologic conditions, as they are newer than the anti-TNF medicines.
She reviewed a variety of research studies evaluating the safety of biologics during pregnancy and beyond. Highlights include results from a large registry, the Psoriasis Longitudinal Assessment and Registry (PSOLAR), of 298 pregnancies in about 220 women from 2007 to 2019, looking at 13 different biologics. The overall and live-birth outcomes in the women on biologics for psoriasis were similar to those for the general population and the rate of congenital anomalies was 0.8%, researchers reported in 2021, lower than the generally cited annual figure of U.S. births.
Studies evaluating biologics for nondermatologic conditions suggest safety. A prospective cohort study of women who took adalimumab in pregnancy (for rheumatoid arthritis or Crohn’s disease) found no increased risk for birth defects. In another study looking at women who were breastfeeding, researchers found no increased risk of infections or delay in developmental milestones in the children of women taking biologics for inflammatory bowel disease, compared with those not on the medications.
A report using data from the World Health Organization concludes that dupilumab appears to be safe during pregnancy, based on an evaluation of 36 pregnancy-related reports among more than 37,000 unique adverse event reports related to dupilumab in a global database.
Recommendations about biologic use from different organizations don’t always mesh, Dr. Shi said, noting that European guidelines tend to be stricter, as some reviews show.
If a mother is exposed to any biologic therapy other than certolizumab during the third trimester, after 27 weeks, Dr. Shi said, “you want to consider avoiding a live vaccine for the first 6 months of the baby’s life.” It turns out, she said, the only recommended live vaccine during that period is the rotavirus vaccine, and she suggests doctors recommend postponing that one until the babies are older if women have been on biologics other than certolizumab.
Her other take-home messages: TNF inhibitors have the most robust safety data from before conception through lactation. Under current guidelines, certolizumab is viewed as the safest to use throughout pregnancy. Dr. Shi’s message to her colleagues fielding the same questions she gets from patients: “There is more data coming out every year. Ultimately, we will have better information to inform our patients.”
At the conference, Lawrence F. Eichenfield, MD, a course director and professor of dermatology and pediatrics at the University of California, San Diego and Rady Children’s Hospital San Diego, encouraged Dr. Shi to write up her presentation as a resource for other dermatologists – which she said is in progress.
Medscape Live and this news organization are owned by the same parent company. Dr. Shi disclosed consulting and investigative and research funding from several pharmaceutical firms, but not directly related to the content of her presentation.
FROM MEDSCAPELIVE WOMEN’S & PEDIATRIC DERMATOLOGY SEMINAR
‘Medical maximizers’ dole out unneeded antibiotics for ASB
So why did you get that prescription?
The Infectious Diseases Society of America recommends against antibiotics in this scenario, with exceptions for patients who are pregnant or undergoing certain urologic procedures.
Antibiotics for asymptomatic bacteriuria (ASB) generally do not help; are costly; and can cause side effects, Clostridioides difficile infection, and antibiotic resistance.
Still, antibiotic treatment for asymptomatic bacteriuria remains common, despite guidelines.
And when researchers recently surveyed 551 primary care clinicians to see which ones would inappropriately prescribe antibiotics for a positive urine culture, the answer was most of them: 71%.
“Regardless of years in practice, training background, or professional degree, most clinicians indicated that they would prescribe antibiotics for asymptomatic bacteriuria,” the researchers reported in JAMA Network Open.
Some groups of clinicians seemed especially likely to prescribe antibiotics unnecessarily.
“Medical maximizers” – clinicians who prefer treatment even when its value is ambiguous – and family medicine clinicians were more likely to prescribe antibiotics in response to a hypothetical case.
On the other hand, resident physicians and clinicians in the U.S. Pacific Northwest were less likely to provide antibiotics inappropriately, the researchers found.
Study author Jonathan D. Baghdadi, MD, PhD, with the department of epidemiology and public health at the University of Maryland and the Veterans Affairs Maryland Healthcare System in Baltimore, summed up the findings on Twitter: “ ... who prescribes antibiotics for asymptomatic bacteriuria? The answer is most primary care clinicians in every category, but it’s more common among clinicians who want to ‘do everything.’ ”
Dr. Baghdadi said the gaps reflect problems with the medical system rather than individual clinicians.
“I don’t believe that individual clinicians knowingly choose to prescribe inappropriate antibiotics in defiance of guidelines,” Dr. Baghdadi told this news organization. “Clinical decision-making is complicated, and the decision to prescribe inappropriate antibiotics depends on patient expectations, clinician perception of patient expectations, time pressure in the clinic, regional variation in medical practice, the culture of antibiotic use, and likely in some cases the perception that doing more is better.”
In addition, researchers have used various definitions of ASB over time and in different contexts, he said.
What to do for Mr. Williams?
To examine clinician attitudes and characteristics associated with prescribing antibiotics for asymptomatic bacteriuria, Dr. Baghdadi and his colleagues analyzed survey responses from 490 physicians and 61 advanced practice clinicians.
Study participants completed tests that measure numeracy, risk-taking preferences, burnout, and tendency to maximize care. They were presented with four hypothetical clinical scenarios, including a case of asymptomatic bacteriuria: “Mr. Williams, a 65-year-old man, comes to the office for follow-up of his osteoarthritis. He has noted foul-smelling urine and no pain or difficulty with urination. A urine dipstick shows trace blood. He has no particular preference for testing and wants your advice.”
Clinicians who had been in practice for at least 10 years were more likely to prescribe antibiotics (82%) to “Mr. Williams” than were those with 3-9 years in practice (73%) or less than 3 years in practice (64%).
Of 120 clinicians with a background in family medicine, 85% said they would have prescribed antibiotics, versus 62% of 207 clinicians with a background in internal medicine.
Nurse practitioners and physician assistants were more likely to prescribe antibiotics (90%) than were attending (78%) and resident physicians (63%).
In one analysis, a background in family medicine was associated with nearly three times higher odds of prescribing antibiotics. And a high “medical maximizer” score was associated with about twice the odds of prescribing the medications.
Meanwhile, resident physicians and clinicians in the Pacific Northwest had a lower likelihood of prescribing antibiotics, with odds ratios of 0.57 and 0.49, respectively.
The respondents who prescribed antibiotics estimated a 90% probability of UTI, whereas those who did not prescribe antibiotics estimated a 15% probability of the condition.
Breaking a habit
Some prescribers may know not to treat asymptomatic bacteriuria but mistakenly consider certain findings to be symptoms of UTI.
Bradley Langford, PharmD, an antimicrobial stewardship expert with Public Health Ontario, said in his experience, most clinicians who say they know not to treat ASB incorrectly believe that cloudy urine, altered cognition, and other nonspecific symptoms indicate a UTI.
“The fact that most clinicians would treat ASB suggests that there is still a lot of work to do to improve antimicrobial stewardship, particularly outside of the hospital setting,” Dr. Langford told this news organization.
Avoiding unnecessary antibiotics is important not just because of the lack of benefit, but also because of the potential harms, said Dr. Langford. He has created a list of rebuttals for commonly given reasons for testing and treating asymptomatic bacteriuria.
“Using antibiotics for ASB can counterintuitively increase the risk for symptomatic UTI due to the disruption of protective local microflora, allowing for the growth of more pathogenic/resistant organisms,” he said.
One approach to addressing the problem: Don’t test urine in the first place if patients are asymptomatic. Virtual learning sessions have been shown to reduce urine culturing and urinary antibiotic prescribing in long-term care homes, Dr. Langford noted.
Updated training for health care professionals from the outset may also be key, and the lower rate of prescribing intent among resident physicians is reassuring, he said.
A role for patients
Patients could also help decrease the inappropriate use of antibiotics.
“Be clear with your doctor about your expectations for the health care interaction, including whether you are expecting to receive antibiotics,” Dr. Baghdadi said. “Your doctor may assume you contacted them because you wanted a prescription. If you are not expecting antibiotics, you should feel free to say so. And if you are asymptomatic, you may not need antibiotics, even if the urine culture is positive.”
The study was funded by a grant from the National Institutes of Health, and Dr. Baghdadi received grant support from the University of Maryland, Baltimore Institute for Clinical and Translational Research. Coauthors disclosed government grants and ties to Memorial Sloan Kettering Cancer Center, Vedanta Biosciences, Opentrons, and Fimbrion. Dr. Langford reported no relevant financial conflicts of interest.
A version of this article first appeared on Medscape.com.
So why did you get that prescription?
The Infectious Diseases Society of America recommends against antibiotics in this scenario, with exceptions for patients who are pregnant or undergoing certain urologic procedures.
Antibiotics for asymptomatic bacteriuria (ASB) generally do not help; are costly; and can cause side effects, Clostridioides difficile infection, and antibiotic resistance.
Still, antibiotic treatment for asymptomatic bacteriuria remains common, despite guidelines.
And when researchers recently surveyed 551 primary care clinicians to see which ones would inappropriately prescribe antibiotics for a positive urine culture, the answer was most of them: 71%.
“Regardless of years in practice, training background, or professional degree, most clinicians indicated that they would prescribe antibiotics for asymptomatic bacteriuria,” the researchers reported in JAMA Network Open.
Some groups of clinicians seemed especially likely to prescribe antibiotics unnecessarily.
“Medical maximizers” – clinicians who prefer treatment even when its value is ambiguous – and family medicine clinicians were more likely to prescribe antibiotics in response to a hypothetical case.
On the other hand, resident physicians and clinicians in the U.S. Pacific Northwest were less likely to provide antibiotics inappropriately, the researchers found.
Study author Jonathan D. Baghdadi, MD, PhD, with the department of epidemiology and public health at the University of Maryland and the Veterans Affairs Maryland Healthcare System in Baltimore, summed up the findings on Twitter: “ ... who prescribes antibiotics for asymptomatic bacteriuria? The answer is most primary care clinicians in every category, but it’s more common among clinicians who want to ‘do everything.’ ”
Dr. Baghdadi said the gaps reflect problems with the medical system rather than individual clinicians.
“I don’t believe that individual clinicians knowingly choose to prescribe inappropriate antibiotics in defiance of guidelines,” Dr. Baghdadi told this news organization. “Clinical decision-making is complicated, and the decision to prescribe inappropriate antibiotics depends on patient expectations, clinician perception of patient expectations, time pressure in the clinic, regional variation in medical practice, the culture of antibiotic use, and likely in some cases the perception that doing more is better.”
In addition, researchers have used various definitions of ASB over time and in different contexts, he said.
What to do for Mr. Williams?
To examine clinician attitudes and characteristics associated with prescribing antibiotics for asymptomatic bacteriuria, Dr. Baghdadi and his colleagues analyzed survey responses from 490 physicians and 61 advanced practice clinicians.
Study participants completed tests that measure numeracy, risk-taking preferences, burnout, and tendency to maximize care. They were presented with four hypothetical clinical scenarios, including a case of asymptomatic bacteriuria: “Mr. Williams, a 65-year-old man, comes to the office for follow-up of his osteoarthritis. He has noted foul-smelling urine and no pain or difficulty with urination. A urine dipstick shows trace blood. He has no particular preference for testing and wants your advice.”
Clinicians who had been in practice for at least 10 years were more likely to prescribe antibiotics (82%) to “Mr. Williams” than were those with 3-9 years in practice (73%) or less than 3 years in practice (64%).
Of 120 clinicians with a background in family medicine, 85% said they would have prescribed antibiotics, versus 62% of 207 clinicians with a background in internal medicine.
Nurse practitioners and physician assistants were more likely to prescribe antibiotics (90%) than were attending (78%) and resident physicians (63%).
In one analysis, a background in family medicine was associated with nearly three times higher odds of prescribing antibiotics. And a high “medical maximizer” score was associated with about twice the odds of prescribing the medications.
Meanwhile, resident physicians and clinicians in the Pacific Northwest had a lower likelihood of prescribing antibiotics, with odds ratios of 0.57 and 0.49, respectively.
The respondents who prescribed antibiotics estimated a 90% probability of UTI, whereas those who did not prescribe antibiotics estimated a 15% probability of the condition.
Breaking a habit
Some prescribers may know not to treat asymptomatic bacteriuria but mistakenly consider certain findings to be symptoms of UTI.
Bradley Langford, PharmD, an antimicrobial stewardship expert with Public Health Ontario, said in his experience, most clinicians who say they know not to treat ASB incorrectly believe that cloudy urine, altered cognition, and other nonspecific symptoms indicate a UTI.
“The fact that most clinicians would treat ASB suggests that there is still a lot of work to do to improve antimicrobial stewardship, particularly outside of the hospital setting,” Dr. Langford told this news organization.
Avoiding unnecessary antibiotics is important not just because of the lack of benefit, but also because of the potential harms, said Dr. Langford. He has created a list of rebuttals for commonly given reasons for testing and treating asymptomatic bacteriuria.
“Using antibiotics for ASB can counterintuitively increase the risk for symptomatic UTI due to the disruption of protective local microflora, allowing for the growth of more pathogenic/resistant organisms,” he said.
One approach to addressing the problem: Don’t test urine in the first place if patients are asymptomatic. Virtual learning sessions have been shown to reduce urine culturing and urinary antibiotic prescribing in long-term care homes, Dr. Langford noted.
Updated training for health care professionals from the outset may also be key, and the lower rate of prescribing intent among resident physicians is reassuring, he said.
A role for patients
Patients could also help decrease the inappropriate use of antibiotics.
“Be clear with your doctor about your expectations for the health care interaction, including whether you are expecting to receive antibiotics,” Dr. Baghdadi said. “Your doctor may assume you contacted them because you wanted a prescription. If you are not expecting antibiotics, you should feel free to say so. And if you are asymptomatic, you may not need antibiotics, even if the urine culture is positive.”
The study was funded by a grant from the National Institutes of Health, and Dr. Baghdadi received grant support from the University of Maryland, Baltimore Institute for Clinical and Translational Research. Coauthors disclosed government grants and ties to Memorial Sloan Kettering Cancer Center, Vedanta Biosciences, Opentrons, and Fimbrion. Dr. Langford reported no relevant financial conflicts of interest.
A version of this article first appeared on Medscape.com.
So why did you get that prescription?
The Infectious Diseases Society of America recommends against antibiotics in this scenario, with exceptions for patients who are pregnant or undergoing certain urologic procedures.
Antibiotics for asymptomatic bacteriuria (ASB) generally do not help; are costly; and can cause side effects, Clostridioides difficile infection, and antibiotic resistance.
Still, antibiotic treatment for asymptomatic bacteriuria remains common, despite guidelines.
And when researchers recently surveyed 551 primary care clinicians to see which ones would inappropriately prescribe antibiotics for a positive urine culture, the answer was most of them: 71%.
“Regardless of years in practice, training background, or professional degree, most clinicians indicated that they would prescribe antibiotics for asymptomatic bacteriuria,” the researchers reported in JAMA Network Open.
Some groups of clinicians seemed especially likely to prescribe antibiotics unnecessarily.
“Medical maximizers” – clinicians who prefer treatment even when its value is ambiguous – and family medicine clinicians were more likely to prescribe antibiotics in response to a hypothetical case.
On the other hand, resident physicians and clinicians in the U.S. Pacific Northwest were less likely to provide antibiotics inappropriately, the researchers found.
Study author Jonathan D. Baghdadi, MD, PhD, with the department of epidemiology and public health at the University of Maryland and the Veterans Affairs Maryland Healthcare System in Baltimore, summed up the findings on Twitter: “ ... who prescribes antibiotics for asymptomatic bacteriuria? The answer is most primary care clinicians in every category, but it’s more common among clinicians who want to ‘do everything.’ ”
Dr. Baghdadi said the gaps reflect problems with the medical system rather than individual clinicians.
“I don’t believe that individual clinicians knowingly choose to prescribe inappropriate antibiotics in defiance of guidelines,” Dr. Baghdadi told this news organization. “Clinical decision-making is complicated, and the decision to prescribe inappropriate antibiotics depends on patient expectations, clinician perception of patient expectations, time pressure in the clinic, regional variation in medical practice, the culture of antibiotic use, and likely in some cases the perception that doing more is better.”
In addition, researchers have used various definitions of ASB over time and in different contexts, he said.
What to do for Mr. Williams?
To examine clinician attitudes and characteristics associated with prescribing antibiotics for asymptomatic bacteriuria, Dr. Baghdadi and his colleagues analyzed survey responses from 490 physicians and 61 advanced practice clinicians.
Study participants completed tests that measure numeracy, risk-taking preferences, burnout, and tendency to maximize care. They were presented with four hypothetical clinical scenarios, including a case of asymptomatic bacteriuria: “Mr. Williams, a 65-year-old man, comes to the office for follow-up of his osteoarthritis. He has noted foul-smelling urine and no pain or difficulty with urination. A urine dipstick shows trace blood. He has no particular preference for testing and wants your advice.”
Clinicians who had been in practice for at least 10 years were more likely to prescribe antibiotics (82%) to “Mr. Williams” than were those with 3-9 years in practice (73%) or less than 3 years in practice (64%).
Of 120 clinicians with a background in family medicine, 85% said they would have prescribed antibiotics, versus 62% of 207 clinicians with a background in internal medicine.
Nurse practitioners and physician assistants were more likely to prescribe antibiotics (90%) than were attending (78%) and resident physicians (63%).
In one analysis, a background in family medicine was associated with nearly three times higher odds of prescribing antibiotics. And a high “medical maximizer” score was associated with about twice the odds of prescribing the medications.
Meanwhile, resident physicians and clinicians in the Pacific Northwest had a lower likelihood of prescribing antibiotics, with odds ratios of 0.57 and 0.49, respectively.
The respondents who prescribed antibiotics estimated a 90% probability of UTI, whereas those who did not prescribe antibiotics estimated a 15% probability of the condition.
Breaking a habit
Some prescribers may know not to treat asymptomatic bacteriuria but mistakenly consider certain findings to be symptoms of UTI.
Bradley Langford, PharmD, an antimicrobial stewardship expert with Public Health Ontario, said in his experience, most clinicians who say they know not to treat ASB incorrectly believe that cloudy urine, altered cognition, and other nonspecific symptoms indicate a UTI.
“The fact that most clinicians would treat ASB suggests that there is still a lot of work to do to improve antimicrobial stewardship, particularly outside of the hospital setting,” Dr. Langford told this news organization.
Avoiding unnecessary antibiotics is important not just because of the lack of benefit, but also because of the potential harms, said Dr. Langford. He has created a list of rebuttals for commonly given reasons for testing and treating asymptomatic bacteriuria.
“Using antibiotics for ASB can counterintuitively increase the risk for symptomatic UTI due to the disruption of protective local microflora, allowing for the growth of more pathogenic/resistant organisms,” he said.
One approach to addressing the problem: Don’t test urine in the first place if patients are asymptomatic. Virtual learning sessions have been shown to reduce urine culturing and urinary antibiotic prescribing in long-term care homes, Dr. Langford noted.
Updated training for health care professionals from the outset may also be key, and the lower rate of prescribing intent among resident physicians is reassuring, he said.
A role for patients
Patients could also help decrease the inappropriate use of antibiotics.
“Be clear with your doctor about your expectations for the health care interaction, including whether you are expecting to receive antibiotics,” Dr. Baghdadi said. “Your doctor may assume you contacted them because you wanted a prescription. If you are not expecting antibiotics, you should feel free to say so. And if you are asymptomatic, you may not need antibiotics, even if the urine culture is positive.”
The study was funded by a grant from the National Institutes of Health, and Dr. Baghdadi received grant support from the University of Maryland, Baltimore Institute for Clinical and Translational Research. Coauthors disclosed government grants and ties to Memorial Sloan Kettering Cancer Center, Vedanta Biosciences, Opentrons, and Fimbrion. Dr. Langford reported no relevant financial conflicts of interest.
A version of this article first appeared on Medscape.com.
Hope for quicker and more accurate endometriosis diagnosis
A new imaging study hopes to make diagnosing endometriosis quicker, more accurate and reduce the need for invasive surgery.
In October 2020 the All Party Parliamentary Group on Endometriosis published a report that included within its recommendations “a commitment to drive down diagnosis times” for women with the condition. On average, it takes around 8 years for a woman to get a diagnosis of endometriosis, a figure, said the authors of the report, that had “not improved in the last decade.”
Indeed, in its report the APPG said that it was seeking a commitment from Governments in all four nations to reduce average diagnosis times with “targets of 4 years or less by 2025, and a year or less by 2030.”
Surgery often needed for endometriosis diagnosis
Endometriosis affects 1 in 10 women between puberty and menopause – 1.5 million in the United Kingdom – often results in multiple general practitioner and accident and emergency department visits, multiple scans, and often laparoscopic surgery to confirm the diagnosis, as there is currently no simple diagnostic test for the condition. One of the main reasons for the delay in diagnosis is the lack of noninvasive tests capable of detecting all endometriosis subtypes – ovarian, superficial, and deep disease.
Now, experts at the Endometriosis CaRe Centre and Nuffield Department of Women’s and Reproductive Health, University of Oxford (England), in collaboration with British life sciences company Serac Healthcare, hope to establish a faster process for diagnosing endometriosis.
Christian Becker, codirector of the Endometriosis CaRe Centre in Oxford, and a study lead, said: “There is an urgent unmet clinical need for a noninvasive marker to identify or rule out endometriosis as it is such a very common disease affecting more than 190 million women worldwide.”
In the study, researchers will investigate whether a 20-minute imaging scan can detect the most common types of endometriosis, which currently require surgery to diagnose. In turn, they hope that earlier diagnosis of the condition will allow women to seek appropriate treatment sooner. They will use an experimental imaging marker – 99mTc-maraciclatide – that binds to areas of inflammation and that can be used in endometriosis to visualize the disease on a scan. The imaging marker has already been used for detecting inflammation in conditions such as rheumatoid arthritis.
Between 2 and 7 days before planned surgery for suspected endometriosis, participants will be invited for an imaging scan, and the team will compare the suspected locations of disease detected on the scan with those seen during surgery to confirm whether this imaging test could be an effective noninvasive method of detecting all endometriosis subtypes.
Doctor visits and repeated investigations reduced
The researchers commented that the potential strengths of the scan lie in the way the imaging marker binds to areas of inflammation, which may allow doctors to distinguish between new and old lesions and detect endometriosis in areas not easily seen during surgery, such as the lung.
They added that the development of a 20-minute imaging test would reduce the need for repeated visits to doctors, for repeated investigations, and for invasive surgery to obtain a diagnosis. This would ultimately “reduce the time taken to confirm or exclude endometriosis,” they pointed out.
Following the publication of the APPG report in October 2020 the group’s then chair, the late Sir David Amess, said: “Without investment in research, a reduction in diagnosis time, and appropriate NHS pathways, those with endometriosis will continue to face huge barriers in accessing the appropriate support at the right time.”
Krina Zondervan, head of department at the Nuffield Department of Women’s and Reproductive Health, University of Oxford, and a study lead, said: “This study highlights that close collaborations between academics, clinicians and industry are important to combine and accelerate discovery and innovation in addressing high-priority areas in women’s health such as endometriosis.”
David Hail, CEO of Serac Healthcare, said: “We are excited about the potential of 99mTc-maraciclatide to diagnose endometriosis noninvasively and delighted to be working with the internationally renowned team at Oxford on this important first study.”
A version of this article first appeared on Medscape UK.
A new imaging study hopes to make diagnosing endometriosis quicker, more accurate and reduce the need for invasive surgery.
In October 2020 the All Party Parliamentary Group on Endometriosis published a report that included within its recommendations “a commitment to drive down diagnosis times” for women with the condition. On average, it takes around 8 years for a woman to get a diagnosis of endometriosis, a figure, said the authors of the report, that had “not improved in the last decade.”
Indeed, in its report the APPG said that it was seeking a commitment from Governments in all four nations to reduce average diagnosis times with “targets of 4 years or less by 2025, and a year or less by 2030.”
Surgery often needed for endometriosis diagnosis
Endometriosis affects 1 in 10 women between puberty and menopause – 1.5 million in the United Kingdom – often results in multiple general practitioner and accident and emergency department visits, multiple scans, and often laparoscopic surgery to confirm the diagnosis, as there is currently no simple diagnostic test for the condition. One of the main reasons for the delay in diagnosis is the lack of noninvasive tests capable of detecting all endometriosis subtypes – ovarian, superficial, and deep disease.
Now, experts at the Endometriosis CaRe Centre and Nuffield Department of Women’s and Reproductive Health, University of Oxford (England), in collaboration with British life sciences company Serac Healthcare, hope to establish a faster process for diagnosing endometriosis.
Christian Becker, codirector of the Endometriosis CaRe Centre in Oxford, and a study lead, said: “There is an urgent unmet clinical need for a noninvasive marker to identify or rule out endometriosis as it is such a very common disease affecting more than 190 million women worldwide.”
In the study, researchers will investigate whether a 20-minute imaging scan can detect the most common types of endometriosis, which currently require surgery to diagnose. In turn, they hope that earlier diagnosis of the condition will allow women to seek appropriate treatment sooner. They will use an experimental imaging marker – 99mTc-maraciclatide – that binds to areas of inflammation and that can be used in endometriosis to visualize the disease on a scan. The imaging marker has already been used for detecting inflammation in conditions such as rheumatoid arthritis.
Between 2 and 7 days before planned surgery for suspected endometriosis, participants will be invited for an imaging scan, and the team will compare the suspected locations of disease detected on the scan with those seen during surgery to confirm whether this imaging test could be an effective noninvasive method of detecting all endometriosis subtypes.
Doctor visits and repeated investigations reduced
The researchers commented that the potential strengths of the scan lie in the way the imaging marker binds to areas of inflammation, which may allow doctors to distinguish between new and old lesions and detect endometriosis in areas not easily seen during surgery, such as the lung.
They added that the development of a 20-minute imaging test would reduce the need for repeated visits to doctors, for repeated investigations, and for invasive surgery to obtain a diagnosis. This would ultimately “reduce the time taken to confirm or exclude endometriosis,” they pointed out.
Following the publication of the APPG report in October 2020 the group’s then chair, the late Sir David Amess, said: “Without investment in research, a reduction in diagnosis time, and appropriate NHS pathways, those with endometriosis will continue to face huge barriers in accessing the appropriate support at the right time.”
Krina Zondervan, head of department at the Nuffield Department of Women’s and Reproductive Health, University of Oxford, and a study lead, said: “This study highlights that close collaborations between academics, clinicians and industry are important to combine and accelerate discovery and innovation in addressing high-priority areas in women’s health such as endometriosis.”
David Hail, CEO of Serac Healthcare, said: “We are excited about the potential of 99mTc-maraciclatide to diagnose endometriosis noninvasively and delighted to be working with the internationally renowned team at Oxford on this important first study.”
A version of this article first appeared on Medscape UK.
A new imaging study hopes to make diagnosing endometriosis quicker, more accurate and reduce the need for invasive surgery.
In October 2020 the All Party Parliamentary Group on Endometriosis published a report that included within its recommendations “a commitment to drive down diagnosis times” for women with the condition. On average, it takes around 8 years for a woman to get a diagnosis of endometriosis, a figure, said the authors of the report, that had “not improved in the last decade.”
Indeed, in its report the APPG said that it was seeking a commitment from Governments in all four nations to reduce average diagnosis times with “targets of 4 years or less by 2025, and a year or less by 2030.”
Surgery often needed for endometriosis diagnosis
Endometriosis affects 1 in 10 women between puberty and menopause – 1.5 million in the United Kingdom – often results in multiple general practitioner and accident and emergency department visits, multiple scans, and often laparoscopic surgery to confirm the diagnosis, as there is currently no simple diagnostic test for the condition. One of the main reasons for the delay in diagnosis is the lack of noninvasive tests capable of detecting all endometriosis subtypes – ovarian, superficial, and deep disease.
Now, experts at the Endometriosis CaRe Centre and Nuffield Department of Women’s and Reproductive Health, University of Oxford (England), in collaboration with British life sciences company Serac Healthcare, hope to establish a faster process for diagnosing endometriosis.
Christian Becker, codirector of the Endometriosis CaRe Centre in Oxford, and a study lead, said: “There is an urgent unmet clinical need for a noninvasive marker to identify or rule out endometriosis as it is such a very common disease affecting more than 190 million women worldwide.”
In the study, researchers will investigate whether a 20-minute imaging scan can detect the most common types of endometriosis, which currently require surgery to diagnose. In turn, they hope that earlier diagnosis of the condition will allow women to seek appropriate treatment sooner. They will use an experimental imaging marker – 99mTc-maraciclatide – that binds to areas of inflammation and that can be used in endometriosis to visualize the disease on a scan. The imaging marker has already been used for detecting inflammation in conditions such as rheumatoid arthritis.
Between 2 and 7 days before planned surgery for suspected endometriosis, participants will be invited for an imaging scan, and the team will compare the suspected locations of disease detected on the scan with those seen during surgery to confirm whether this imaging test could be an effective noninvasive method of detecting all endometriosis subtypes.
Doctor visits and repeated investigations reduced
The researchers commented that the potential strengths of the scan lie in the way the imaging marker binds to areas of inflammation, which may allow doctors to distinguish between new and old lesions and detect endometriosis in areas not easily seen during surgery, such as the lung.
They added that the development of a 20-minute imaging test would reduce the need for repeated visits to doctors, for repeated investigations, and for invasive surgery to obtain a diagnosis. This would ultimately “reduce the time taken to confirm or exclude endometriosis,” they pointed out.
Following the publication of the APPG report in October 2020 the group’s then chair, the late Sir David Amess, said: “Without investment in research, a reduction in diagnosis time, and appropriate NHS pathways, those with endometriosis will continue to face huge barriers in accessing the appropriate support at the right time.”
Krina Zondervan, head of department at the Nuffield Department of Women’s and Reproductive Health, University of Oxford, and a study lead, said: “This study highlights that close collaborations between academics, clinicians and industry are important to combine and accelerate discovery and innovation in addressing high-priority areas in women’s health such as endometriosis.”
David Hail, CEO of Serac Healthcare, said: “We are excited about the potential of 99mTc-maraciclatide to diagnose endometriosis noninvasively and delighted to be working with the internationally renowned team at Oxford on this important first study.”
A version of this article first appeared on Medscape UK.
Women are not being warned that anesthetic may reduce birth pill efficacy
The effectiveness of hormonal contraceptives, including the pill and mini-pill, may be compromised by sugammadex, a drug widely used in anesthesia for reversing neuromuscular blockade induced by rocuronium or vecuronium.
Yet women are not routinely informed that the drug may make their contraception less effective, delegates at Euroanaesthesia, the annual meeting of the European Society of Anaesthesiology and Intensive Care in Milan were told.
New research presented at the meeting supports the authors’ experience that “robust methods for identifying at-risk patients and informing them of the associated risk of contraceptive failures is not common practice across anesthetic departments within the United Kingdom, and likely further afield.”
This is according to a survey of almost 150 anesthetic professionals, including consultants, junior doctors, and physician assistants, working at University College London Hospitals NHS Foundation Trust.
Dr. Neha Passi, Dr. Matt Oliver, and colleagues at the trust’s department of anesthesiology sent out a seven-question survey to their 150 colleagues and received 82 responses, 94% of which claimed awareness of the risk of contraceptive failure with sugammadex. However, 70% of the respondents admitted that they do not routinely discuss this with patients who have received the drug.
Risk with all forms of hormonal contraceptive
Yet current guidance is to inform women of child-bearing age that they have received the drug and, because of increased risk of contraceptive failure, advise those taking oral hormonal contraceptives to follow the missed pill advice in the leaflet that comes with their contraceptives. It also counsels that clinicians should advise women using other types of hormonal contraceptive to use an additional nonhormonal means of contraception for 7 days.
The study authors also carried out a retrospective audit of sugammadex use in the trust and reported that during the 6 weeks covered by the audit, 234 patients were administered sugammadex of whom 65 (28%) were women of childbearing age. Of these, 17 had a medical history that meant they weren’t at risk of pregnancy, but the other 48 should have received advice on the risks of contraceptive failure – however there was no record in the medical notes of such advice having been given for any of the at-risk 48 women.
While sugammadex is the only anesthetic drug known to have this effect, it is recognized to interact with progesterone and so may reduce the effectiveness of hormonal contraceptives, including the progesterone-only pill, combined pill, vaginal rings, implants, and intrauterine devices.
Dr. Passi said: “It is concerning that we are so seldom informing patients of the risk of contraceptive failure following sugammadex use.
“Use of sugammadex is expected to rise as it becomes cheaper in the future, and ensuring that women receiving this medicine are aware it may increase their risk of unwanted pregnancy must be a priority.”
She added: “It is important to note, however, that most patients receiving an anesthetic do not need a muscle relaxant and that sugammadex is one of several drugs available to reverse muscle relaxation.”
Dr. Oliver said: “We only studied one hospital trust but we expect the results to be similar in elsewhere in the U.K.”
In response to their findings, the study’s authors have created patient information leaflets and letters and programmed the trust’s electronic patient record system to identify “at-risk” patients and deliver electronic prompts to the anesthetists caring for them in the perioperative period.
A version of this article first appeared on Medscape UK.
The effectiveness of hormonal contraceptives, including the pill and mini-pill, may be compromised by sugammadex, a drug widely used in anesthesia for reversing neuromuscular blockade induced by rocuronium or vecuronium.
Yet women are not routinely informed that the drug may make their contraception less effective, delegates at Euroanaesthesia, the annual meeting of the European Society of Anaesthesiology and Intensive Care in Milan were told.
New research presented at the meeting supports the authors’ experience that “robust methods for identifying at-risk patients and informing them of the associated risk of contraceptive failures is not common practice across anesthetic departments within the United Kingdom, and likely further afield.”
This is according to a survey of almost 150 anesthetic professionals, including consultants, junior doctors, and physician assistants, working at University College London Hospitals NHS Foundation Trust.
Dr. Neha Passi, Dr. Matt Oliver, and colleagues at the trust’s department of anesthesiology sent out a seven-question survey to their 150 colleagues and received 82 responses, 94% of which claimed awareness of the risk of contraceptive failure with sugammadex. However, 70% of the respondents admitted that they do not routinely discuss this with patients who have received the drug.
Risk with all forms of hormonal contraceptive
Yet current guidance is to inform women of child-bearing age that they have received the drug and, because of increased risk of contraceptive failure, advise those taking oral hormonal contraceptives to follow the missed pill advice in the leaflet that comes with their contraceptives. It also counsels that clinicians should advise women using other types of hormonal contraceptive to use an additional nonhormonal means of contraception for 7 days.
The study authors also carried out a retrospective audit of sugammadex use in the trust and reported that during the 6 weeks covered by the audit, 234 patients were administered sugammadex of whom 65 (28%) were women of childbearing age. Of these, 17 had a medical history that meant they weren’t at risk of pregnancy, but the other 48 should have received advice on the risks of contraceptive failure – however there was no record in the medical notes of such advice having been given for any of the at-risk 48 women.
While sugammadex is the only anesthetic drug known to have this effect, it is recognized to interact with progesterone and so may reduce the effectiveness of hormonal contraceptives, including the progesterone-only pill, combined pill, vaginal rings, implants, and intrauterine devices.
Dr. Passi said: “It is concerning that we are so seldom informing patients of the risk of contraceptive failure following sugammadex use.
“Use of sugammadex is expected to rise as it becomes cheaper in the future, and ensuring that women receiving this medicine are aware it may increase their risk of unwanted pregnancy must be a priority.”
She added: “It is important to note, however, that most patients receiving an anesthetic do not need a muscle relaxant and that sugammadex is one of several drugs available to reverse muscle relaxation.”
Dr. Oliver said: “We only studied one hospital trust but we expect the results to be similar in elsewhere in the U.K.”
In response to their findings, the study’s authors have created patient information leaflets and letters and programmed the trust’s electronic patient record system to identify “at-risk” patients and deliver electronic prompts to the anesthetists caring for them in the perioperative period.
A version of this article first appeared on Medscape UK.
The effectiveness of hormonal contraceptives, including the pill and mini-pill, may be compromised by sugammadex, a drug widely used in anesthesia for reversing neuromuscular blockade induced by rocuronium or vecuronium.
Yet women are not routinely informed that the drug may make their contraception less effective, delegates at Euroanaesthesia, the annual meeting of the European Society of Anaesthesiology and Intensive Care in Milan were told.
New research presented at the meeting supports the authors’ experience that “robust methods for identifying at-risk patients and informing them of the associated risk of contraceptive failures is not common practice across anesthetic departments within the United Kingdom, and likely further afield.”
This is according to a survey of almost 150 anesthetic professionals, including consultants, junior doctors, and physician assistants, working at University College London Hospitals NHS Foundation Trust.
Dr. Neha Passi, Dr. Matt Oliver, and colleagues at the trust’s department of anesthesiology sent out a seven-question survey to their 150 colleagues and received 82 responses, 94% of which claimed awareness of the risk of contraceptive failure with sugammadex. However, 70% of the respondents admitted that they do not routinely discuss this with patients who have received the drug.
Risk with all forms of hormonal contraceptive
Yet current guidance is to inform women of child-bearing age that they have received the drug and, because of increased risk of contraceptive failure, advise those taking oral hormonal contraceptives to follow the missed pill advice in the leaflet that comes with their contraceptives. It also counsels that clinicians should advise women using other types of hormonal contraceptive to use an additional nonhormonal means of contraception for 7 days.
The study authors also carried out a retrospective audit of sugammadex use in the trust and reported that during the 6 weeks covered by the audit, 234 patients were administered sugammadex of whom 65 (28%) were women of childbearing age. Of these, 17 had a medical history that meant they weren’t at risk of pregnancy, but the other 48 should have received advice on the risks of contraceptive failure – however there was no record in the medical notes of such advice having been given for any of the at-risk 48 women.
While sugammadex is the only anesthetic drug known to have this effect, it is recognized to interact with progesterone and so may reduce the effectiveness of hormonal contraceptives, including the progesterone-only pill, combined pill, vaginal rings, implants, and intrauterine devices.
Dr. Passi said: “It is concerning that we are so seldom informing patients of the risk of contraceptive failure following sugammadex use.
“Use of sugammadex is expected to rise as it becomes cheaper in the future, and ensuring that women receiving this medicine are aware it may increase their risk of unwanted pregnancy must be a priority.”
She added: “It is important to note, however, that most patients receiving an anesthetic do not need a muscle relaxant and that sugammadex is one of several drugs available to reverse muscle relaxation.”
Dr. Oliver said: “We only studied one hospital trust but we expect the results to be similar in elsewhere in the U.K.”
In response to their findings, the study’s authors have created patient information leaflets and letters and programmed the trust’s electronic patient record system to identify “at-risk” patients and deliver electronic prompts to the anesthetists caring for them in the perioperative period.
A version of this article first appeared on Medscape UK.
FROM EUROANAESTHESIA
Monkeypox largely a mystery for pregnant people
With monkeypox now circulating in the United States, expecting mothers may worry about what might happen if they contract the infection while pregnant.
As of today, 25 cases of monkeypox have been confirmed in the United States since the outbreak began in early May, according to the U.S. Centers for Disease Control and Prevention. Although none of those cases has involved a pregnant person, the World Health Organization says monkeypox can pass from mother to fetus before delivery or to newborns by close contact during and after birth.
The case count could grow as the agency continues to investigate potential infections of the virus. In a conference call Friday, health officials stressed the importance of contact tracing, testing, and vaccine treatment.
As physicians in the United States are scrambling for information on ways to treat patients, a new study, published in Ultrasound in Obstetrics & Gynecology, could help clinicians better care for pregnant people infected with monkeypox. The authors advise consistently monitoring the fetus for infection and conducting regular ultrasounds, among other precautions.
Asma Khalil, MBBCh, MD, a professor of obstetrics and fetal medicine at St. George’s University, London, and lead author of the new study, said the monkeypox outbreak outside Africa caught many clinicians by surprise.
“We quickly realized very few physicians caring for pregnant women knew anything at all about monkeypox and how it affects pregnancy,” Dr. Khalil told this news organization. “Clinicians caring for pregnant women are likely to be faced soon with pregnant women concerned they may have the infection – because they have a rash, for example – or indeed pregnant women who do have the infection.”
According to the CDC, monkeypox can be transmitted through direct contact with the rash, sores, or scabs caused by the virus, as well as contact with clothing, bedding, towels, or other surfaces used by an infected person. Respiratory droplets and oral fluids from a person with monkeypox have also been linked to spread of the virus, as has sexual activity.
Although the condition is rarely fatal, infants and young children are at the greatest risk of developing severe symptoms, health officials said.
The U.S. Food and Drug Administration has approved a monkeypox vaccine, Jynneos (Bavarian Nordic A/S), for general use, but it has not been specifically approved for pregnant people. However, a study of 300 pregnant women who received the vaccine reported no adverse reactions or failed pregnancies linked to the shots.
The new review suggests that women who have a confirmed infection during pregnancy should have a doctor closely monitor the fetus until birth.
If the fetus is over 26 weeks or if the mother is unwell, the fetus should be cared for with heart monitoring, either by a doctor or remotely every 2-3 days. Ultrasounds should be performed regularly to confirm that the fetus is still growing well and that the placenta is functioning properly.
Further into the pregnancy, monitoring should include measurements of the fetus and detailed assessment of the fetal organs and the amniotic fluid. Once the infection is resolved, the risk to the fetus is small, according to Dr. Khalil. However, since data are limited, she recommended an ultrasound scan every 2-4 weeks. At birth, for the protection of the infant and the mother, the baby should be isolated until infection is no longer a risk.
The Royal College of Obstetricians & Gynaecologists is preparing guidance on the management of monkeypox in pregnant people, Dr. Khalil said. The American College of Obstetricians and Gynecologists said it is “relying on the CDC for the time being,” according to a spokesperson for ACOG.
“There is a clear need for further research in this area,” Dr. Khalil said. “The current outbreak is an ideal opportunity to make this happen.”
Dr. Khalil has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
With monkeypox now circulating in the United States, expecting mothers may worry about what might happen if they contract the infection while pregnant.
As of today, 25 cases of monkeypox have been confirmed in the United States since the outbreak began in early May, according to the U.S. Centers for Disease Control and Prevention. Although none of those cases has involved a pregnant person, the World Health Organization says monkeypox can pass from mother to fetus before delivery or to newborns by close contact during and after birth.
The case count could grow as the agency continues to investigate potential infections of the virus. In a conference call Friday, health officials stressed the importance of contact tracing, testing, and vaccine treatment.
As physicians in the United States are scrambling for information on ways to treat patients, a new study, published in Ultrasound in Obstetrics & Gynecology, could help clinicians better care for pregnant people infected with monkeypox. The authors advise consistently monitoring the fetus for infection and conducting regular ultrasounds, among other precautions.
Asma Khalil, MBBCh, MD, a professor of obstetrics and fetal medicine at St. George’s University, London, and lead author of the new study, said the monkeypox outbreak outside Africa caught many clinicians by surprise.
“We quickly realized very few physicians caring for pregnant women knew anything at all about monkeypox and how it affects pregnancy,” Dr. Khalil told this news organization. “Clinicians caring for pregnant women are likely to be faced soon with pregnant women concerned they may have the infection – because they have a rash, for example – or indeed pregnant women who do have the infection.”
According to the CDC, monkeypox can be transmitted through direct contact with the rash, sores, or scabs caused by the virus, as well as contact with clothing, bedding, towels, or other surfaces used by an infected person. Respiratory droplets and oral fluids from a person with monkeypox have also been linked to spread of the virus, as has sexual activity.
Although the condition is rarely fatal, infants and young children are at the greatest risk of developing severe symptoms, health officials said.
The U.S. Food and Drug Administration has approved a monkeypox vaccine, Jynneos (Bavarian Nordic A/S), for general use, but it has not been specifically approved for pregnant people. However, a study of 300 pregnant women who received the vaccine reported no adverse reactions or failed pregnancies linked to the shots.
The new review suggests that women who have a confirmed infection during pregnancy should have a doctor closely monitor the fetus until birth.
If the fetus is over 26 weeks or if the mother is unwell, the fetus should be cared for with heart monitoring, either by a doctor or remotely every 2-3 days. Ultrasounds should be performed regularly to confirm that the fetus is still growing well and that the placenta is functioning properly.
Further into the pregnancy, monitoring should include measurements of the fetus and detailed assessment of the fetal organs and the amniotic fluid. Once the infection is resolved, the risk to the fetus is small, according to Dr. Khalil. However, since data are limited, she recommended an ultrasound scan every 2-4 weeks. At birth, for the protection of the infant and the mother, the baby should be isolated until infection is no longer a risk.
The Royal College of Obstetricians & Gynaecologists is preparing guidance on the management of monkeypox in pregnant people, Dr. Khalil said. The American College of Obstetricians and Gynecologists said it is “relying on the CDC for the time being,” according to a spokesperson for ACOG.
“There is a clear need for further research in this area,” Dr. Khalil said. “The current outbreak is an ideal opportunity to make this happen.”
Dr. Khalil has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
With monkeypox now circulating in the United States, expecting mothers may worry about what might happen if they contract the infection while pregnant.
As of today, 25 cases of monkeypox have been confirmed in the United States since the outbreak began in early May, according to the U.S. Centers for Disease Control and Prevention. Although none of those cases has involved a pregnant person, the World Health Organization says monkeypox can pass from mother to fetus before delivery or to newborns by close contact during and after birth.
The case count could grow as the agency continues to investigate potential infections of the virus. In a conference call Friday, health officials stressed the importance of contact tracing, testing, and vaccine treatment.
As physicians in the United States are scrambling for information on ways to treat patients, a new study, published in Ultrasound in Obstetrics & Gynecology, could help clinicians better care for pregnant people infected with monkeypox. The authors advise consistently monitoring the fetus for infection and conducting regular ultrasounds, among other precautions.
Asma Khalil, MBBCh, MD, a professor of obstetrics and fetal medicine at St. George’s University, London, and lead author of the new study, said the monkeypox outbreak outside Africa caught many clinicians by surprise.
“We quickly realized very few physicians caring for pregnant women knew anything at all about monkeypox and how it affects pregnancy,” Dr. Khalil told this news organization. “Clinicians caring for pregnant women are likely to be faced soon with pregnant women concerned they may have the infection – because they have a rash, for example – or indeed pregnant women who do have the infection.”
According to the CDC, monkeypox can be transmitted through direct contact with the rash, sores, or scabs caused by the virus, as well as contact with clothing, bedding, towels, or other surfaces used by an infected person. Respiratory droplets and oral fluids from a person with monkeypox have also been linked to spread of the virus, as has sexual activity.
Although the condition is rarely fatal, infants and young children are at the greatest risk of developing severe symptoms, health officials said.
The U.S. Food and Drug Administration has approved a monkeypox vaccine, Jynneos (Bavarian Nordic A/S), for general use, but it has not been specifically approved for pregnant people. However, a study of 300 pregnant women who received the vaccine reported no adverse reactions or failed pregnancies linked to the shots.
The new review suggests that women who have a confirmed infection during pregnancy should have a doctor closely monitor the fetus until birth.
If the fetus is over 26 weeks or if the mother is unwell, the fetus should be cared for with heart monitoring, either by a doctor or remotely every 2-3 days. Ultrasounds should be performed regularly to confirm that the fetus is still growing well and that the placenta is functioning properly.
Further into the pregnancy, monitoring should include measurements of the fetus and detailed assessment of the fetal organs and the amniotic fluid. Once the infection is resolved, the risk to the fetus is small, according to Dr. Khalil. However, since data are limited, she recommended an ultrasound scan every 2-4 weeks. At birth, for the protection of the infant and the mother, the baby should be isolated until infection is no longer a risk.
The Royal College of Obstetricians & Gynaecologists is preparing guidance on the management of monkeypox in pregnant people, Dr. Khalil said. The American College of Obstetricians and Gynecologists said it is “relying on the CDC for the time being,” according to a spokesperson for ACOG.
“There is a clear need for further research in this area,” Dr. Khalil said. “The current outbreak is an ideal opportunity to make this happen.”
Dr. Khalil has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Women with high-risk pregnancies could die if Roe is overturned
Kendra Joseph of San Antonio, Tex., had given up on the idea of having a second child. At 40 years old, and with a daughter pleading for a sibling, she and her husband were nervous about the risk of trying for another child due to her advanced maternal age. Mrs. Joseph had ended an earlier pregnancy at 15 weeks after finding out her son had Edwards syndrome, a genetic trait that’s fatal in most cases.
Now a new Texas law that bans abortion past 6 weeks would mean that if either she or her baby were at risk of dying, she might still have to carry the baby to term. For Mrs. Joseph, it wasn’t worth the risk at first. Then in February, just as they had decided against another baby, the couple found out they were expecting. She’s thrilled about her pregnancy, but it’s also been a nervewracking few months.
“It’s scary being pregnant anyway,” she says, “but these new restrictions add a layer of stress.”
Twenty-eight states could ban or tightly restrict abortion if the Supreme Court overturns the landmark Roe v. Wade decision. A leaked draft of the court’s opinion has been widely interpreted as signaling that the court will overturn the law. This means that women who are at a higher risk of pregnancy complications or those who have chronic conditions before getting pregnant could be at risk of dying if they can’t get an abortion.
According to the CDC, the maternal mortality rate in the United States in 2020 was 23.8 deaths per 100,000 live births – among the highest in the developed world. The rate is eight times as high as it is in countries like the Netherlands, Norway, and New Zealand.
“Many of the women I take care of have a pregnancy that presents a real and present danger to their health, and this often goes along with the fact that they’re very unlikely to have a healthy baby,” says Chavi Karkowsky, MD, a maternal fetal medicine specialist at Montefiore Medical Center, New York.
Maternal mortality, she says, can be caused by health conditions that some women may not know about before getting pregnant. (For example, finding out she had cervical cancer at a prenatal visit and then having to choose between chemotherapy and her baby.) And there are also life-threatening conditions caused by pregnancy, like preeclampsia, which can cause high blood pressure and kidney damage, as well as gestational diabetes. Research has also shown that the risk of maternal mortality increases with age.
University of Colorado researchers, in a study published in the journal Demography, found that banning abortion nationwide would lead to a 20% increase in maternal death. For Black women, the increase in mortality could be as high as 33%, due to higher rates of poverty and less access to health care, says Amanda Stevenson, PhD, a sociologist at the University of Colorado and one of the study’s authors. Black women in the U.S. are more than three times as likely to die as a result of pregnancy complications due to poor exposure to health care, structural racism, and chronic health conditions, according to the CDC.
If Roe v. Wade is overturned, more women will likely die because remaining pregnant poses a far greater mortality risk for them than the risk associated with an abortion, says Dr. Stevenson.
For women with high-risk pregnancies who need an abortion, traveling out of state puts them at a health risk, says Jamila Perritt, MD, an ob.gyn. in Washington, D.C. and president of Physicians for Reproductive Health. In places where abortion is restricted, it can cause significant delays in accessing medical care. “Abortion is a time-sensitive procedure, and as the pregnancy progresses, it can become increasingly difficult to find a clinic that will provide care,” she says.
She recalls one of her patients who had a heart problem that required a pregnancy to be ended. The patient at first had to travel to find a doctor who could evaluate her unique condition, then go out of state to get an abortion. All the while, the clock was ticking and her health was at risk. In this case, the patient had the money to travel out of state, find child care, and pay for the procedure.
“This was a resourced individual, and while this was difficult for her, it wasn’t impossible,” says Dr. Perritt.
Many of the states with the highest maternal mortality rates, including Louisiana, Texas, Arkansas, Alabama, South Carolina, and Georgia, also plan to strictly limit abortions or ban them completely. Some abortion opponents insist this won’t harm mothers.
“The pro-life movement loves both babies and moms,” says Sarah Zagorski, a spokeswoman for Louisiana Right to Life. “It is a tragedy that Louisiana has high mortality rates among pregnant women. However, legal abortion does not improve these rates.”
But for many women who need an abortion, statewide bans may make it hard to get. This worries Kendra Joseph, who’s now 18 weeks into her pregnancy.
“I try to put the bad things that could happen out of my mind, but it’s really hard when you’re dealing with these totally unnecessary and cruel restrictions. We as women, we’re just losing so much,” she says.
A version of this article first appeared on WebMD.com.
Kendra Joseph of San Antonio, Tex., had given up on the idea of having a second child. At 40 years old, and with a daughter pleading for a sibling, she and her husband were nervous about the risk of trying for another child due to her advanced maternal age. Mrs. Joseph had ended an earlier pregnancy at 15 weeks after finding out her son had Edwards syndrome, a genetic trait that’s fatal in most cases.
Now a new Texas law that bans abortion past 6 weeks would mean that if either she or her baby were at risk of dying, she might still have to carry the baby to term. For Mrs. Joseph, it wasn’t worth the risk at first. Then in February, just as they had decided against another baby, the couple found out they were expecting. She’s thrilled about her pregnancy, but it’s also been a nervewracking few months.
“It’s scary being pregnant anyway,” she says, “but these new restrictions add a layer of stress.”
Twenty-eight states could ban or tightly restrict abortion if the Supreme Court overturns the landmark Roe v. Wade decision. A leaked draft of the court’s opinion has been widely interpreted as signaling that the court will overturn the law. This means that women who are at a higher risk of pregnancy complications or those who have chronic conditions before getting pregnant could be at risk of dying if they can’t get an abortion.
According to the CDC, the maternal mortality rate in the United States in 2020 was 23.8 deaths per 100,000 live births – among the highest in the developed world. The rate is eight times as high as it is in countries like the Netherlands, Norway, and New Zealand.
“Many of the women I take care of have a pregnancy that presents a real and present danger to their health, and this often goes along with the fact that they’re very unlikely to have a healthy baby,” says Chavi Karkowsky, MD, a maternal fetal medicine specialist at Montefiore Medical Center, New York.
Maternal mortality, she says, can be caused by health conditions that some women may not know about before getting pregnant. (For example, finding out she had cervical cancer at a prenatal visit and then having to choose between chemotherapy and her baby.) And there are also life-threatening conditions caused by pregnancy, like preeclampsia, which can cause high blood pressure and kidney damage, as well as gestational diabetes. Research has also shown that the risk of maternal mortality increases with age.
University of Colorado researchers, in a study published in the journal Demography, found that banning abortion nationwide would lead to a 20% increase in maternal death. For Black women, the increase in mortality could be as high as 33%, due to higher rates of poverty and less access to health care, says Amanda Stevenson, PhD, a sociologist at the University of Colorado and one of the study’s authors. Black women in the U.S. are more than three times as likely to die as a result of pregnancy complications due to poor exposure to health care, structural racism, and chronic health conditions, according to the CDC.
If Roe v. Wade is overturned, more women will likely die because remaining pregnant poses a far greater mortality risk for them than the risk associated with an abortion, says Dr. Stevenson.
For women with high-risk pregnancies who need an abortion, traveling out of state puts them at a health risk, says Jamila Perritt, MD, an ob.gyn. in Washington, D.C. and president of Physicians for Reproductive Health. In places where abortion is restricted, it can cause significant delays in accessing medical care. “Abortion is a time-sensitive procedure, and as the pregnancy progresses, it can become increasingly difficult to find a clinic that will provide care,” she says.
She recalls one of her patients who had a heart problem that required a pregnancy to be ended. The patient at first had to travel to find a doctor who could evaluate her unique condition, then go out of state to get an abortion. All the while, the clock was ticking and her health was at risk. In this case, the patient had the money to travel out of state, find child care, and pay for the procedure.
“This was a resourced individual, and while this was difficult for her, it wasn’t impossible,” says Dr. Perritt.
Many of the states with the highest maternal mortality rates, including Louisiana, Texas, Arkansas, Alabama, South Carolina, and Georgia, also plan to strictly limit abortions or ban them completely. Some abortion opponents insist this won’t harm mothers.
“The pro-life movement loves both babies and moms,” says Sarah Zagorski, a spokeswoman for Louisiana Right to Life. “It is a tragedy that Louisiana has high mortality rates among pregnant women. However, legal abortion does not improve these rates.”
But for many women who need an abortion, statewide bans may make it hard to get. This worries Kendra Joseph, who’s now 18 weeks into her pregnancy.
“I try to put the bad things that could happen out of my mind, but it’s really hard when you’re dealing with these totally unnecessary and cruel restrictions. We as women, we’re just losing so much,” she says.
A version of this article first appeared on WebMD.com.
Kendra Joseph of San Antonio, Tex., had given up on the idea of having a second child. At 40 years old, and with a daughter pleading for a sibling, she and her husband were nervous about the risk of trying for another child due to her advanced maternal age. Mrs. Joseph had ended an earlier pregnancy at 15 weeks after finding out her son had Edwards syndrome, a genetic trait that’s fatal in most cases.
Now a new Texas law that bans abortion past 6 weeks would mean that if either she or her baby were at risk of dying, she might still have to carry the baby to term. For Mrs. Joseph, it wasn’t worth the risk at first. Then in February, just as they had decided against another baby, the couple found out they were expecting. She’s thrilled about her pregnancy, but it’s also been a nervewracking few months.
“It’s scary being pregnant anyway,” she says, “but these new restrictions add a layer of stress.”
Twenty-eight states could ban or tightly restrict abortion if the Supreme Court overturns the landmark Roe v. Wade decision. A leaked draft of the court’s opinion has been widely interpreted as signaling that the court will overturn the law. This means that women who are at a higher risk of pregnancy complications or those who have chronic conditions before getting pregnant could be at risk of dying if they can’t get an abortion.
According to the CDC, the maternal mortality rate in the United States in 2020 was 23.8 deaths per 100,000 live births – among the highest in the developed world. The rate is eight times as high as it is in countries like the Netherlands, Norway, and New Zealand.
“Many of the women I take care of have a pregnancy that presents a real and present danger to their health, and this often goes along with the fact that they’re very unlikely to have a healthy baby,” says Chavi Karkowsky, MD, a maternal fetal medicine specialist at Montefiore Medical Center, New York.
Maternal mortality, she says, can be caused by health conditions that some women may not know about before getting pregnant. (For example, finding out she had cervical cancer at a prenatal visit and then having to choose between chemotherapy and her baby.) And there are also life-threatening conditions caused by pregnancy, like preeclampsia, which can cause high blood pressure and kidney damage, as well as gestational diabetes. Research has also shown that the risk of maternal mortality increases with age.
University of Colorado researchers, in a study published in the journal Demography, found that banning abortion nationwide would lead to a 20% increase in maternal death. For Black women, the increase in mortality could be as high as 33%, due to higher rates of poverty and less access to health care, says Amanda Stevenson, PhD, a sociologist at the University of Colorado and one of the study’s authors. Black women in the U.S. are more than three times as likely to die as a result of pregnancy complications due to poor exposure to health care, structural racism, and chronic health conditions, according to the CDC.
If Roe v. Wade is overturned, more women will likely die because remaining pregnant poses a far greater mortality risk for them than the risk associated with an abortion, says Dr. Stevenson.
For women with high-risk pregnancies who need an abortion, traveling out of state puts them at a health risk, says Jamila Perritt, MD, an ob.gyn. in Washington, D.C. and president of Physicians for Reproductive Health. In places where abortion is restricted, it can cause significant delays in accessing medical care. “Abortion is a time-sensitive procedure, and as the pregnancy progresses, it can become increasingly difficult to find a clinic that will provide care,” she says.
She recalls one of her patients who had a heart problem that required a pregnancy to be ended. The patient at first had to travel to find a doctor who could evaluate her unique condition, then go out of state to get an abortion. All the while, the clock was ticking and her health was at risk. In this case, the patient had the money to travel out of state, find child care, and pay for the procedure.
“This was a resourced individual, and while this was difficult for her, it wasn’t impossible,” says Dr. Perritt.
Many of the states with the highest maternal mortality rates, including Louisiana, Texas, Arkansas, Alabama, South Carolina, and Georgia, also plan to strictly limit abortions or ban them completely. Some abortion opponents insist this won’t harm mothers.
“The pro-life movement loves both babies and moms,” says Sarah Zagorski, a spokeswoman for Louisiana Right to Life. “It is a tragedy that Louisiana has high mortality rates among pregnant women. However, legal abortion does not improve these rates.”
But for many women who need an abortion, statewide bans may make it hard to get. This worries Kendra Joseph, who’s now 18 weeks into her pregnancy.
“I try to put the bad things that could happen out of my mind, but it’s really hard when you’re dealing with these totally unnecessary and cruel restrictions. We as women, we’re just losing so much,” she says.
A version of this article first appeared on WebMD.com.
High maternal, fetal morbidity rates in SLE pregnancies
COPENHAGEN – Pregnant women with systemic lupus erythematosus (SLE) are at significantly higher risk of requiring transfusion, developing a cerebrovascular disorder, or developing acute renal failure than pregnant women without SLE, a review of data from an American national sample indicates.
Pregnant women with SLE also have a twofold-higher risk for premature delivery, and a threefold risk of having a fetus with intrauterine growth restriction than their pregnant counterparts without SLE, reported Bella Mehta, MBBS, MS, MD, a rheumatologist at the Hospital for Special Surgery in New York.
“Severe maternal morbidity and fetal morbidity still remain high, but this work can help inform physicians and counsel patients for pregnancy planning and management,” she said at the annual European Congress of Rheumatology.
Although in-hospital maternal and fetal mortality rates for women with SLE have declined over the past 2 decades, the same cannot be said for morbidities, prompting the investigators to conduct a study to determine the proportion of fetal and maternal morbidity in SLE deliveries, compared with non-SLE deliveries over a decade.
Inpatient Sample
Dr. Mehta and colleagues studied retrospective data on 40 million delivery-related admissions from the National Inpatient Sample database. Of these patients, 51,161 had a diagnosis of SLE.
They identified all delivery-related hospital admissions for patients with and without SLE from 2008 through 2017 using diagnostic codes.
The researchers looked at fetal morbidity indicators, including preterm delivery and intrauterine growth restriction, and used the Centers for Disease Control and Prevention standard definition of severe maternal morbidity as “unexpected outcomes of labor and delivery that result in significant short- or long- term consequences to a woman’s health.”
They identified 21 severe maternal morbidity outcomes, including blood transfusion requirements, acute renal failure, eclampsia and disseminated intravascular coagulation, cardiovascular and peripheral vascular disorders, and general medical issues (hysterectomy, shock, sepsis, adult respiratory distress syndrome, severe anesthesia complications, temporary tracheostomy, and ventilation).
Study results
Women with SLE were slightly older at the time of delivery (mean age, 30.05 vs. 29.19 years) and had more comorbidities, according to the Elixhauser Comorbidity Scale, with 97.84% of women in this group having one to four comorbidities, compared with 19.4% of women without SLE.
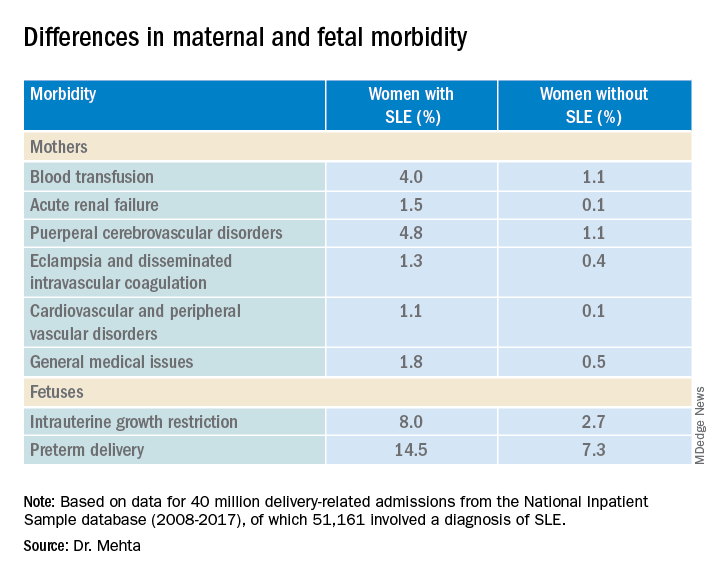
Dr. Mehta acknowledged that the study was limited by the inability to capture outpatient deliveries, although she noted that only about 1.3% of deliveries in the United States occur outside the inpatient setting.
In addition, she noted that the database does not include information on lupus disease activity, Apgar scores, SLE flares, the presence of nephritis, antiphospholipid or anti-Ro/SSA antibodies, or medication use.
A rheumatologist who was not involved in the study said in an interview that the data from this study are in line with those in other recently published studies.
“The problem is that these data were not corrected for further disease activity or drugs,” said Frauke Förger, MD, professor of rheumatology and immunology at the University of Bern (Switzerland), who comoderated the oral abstract session where the data were presented.
She said prospective studies that adjusted for factors such as SLE disease activity and medication use will be required to give clinicians a better understanding of how to manage pregnancies in women with SLE.
The study was supported by an award from Weill Cornell Medicine. Dr. Mehta and Dr. Förger reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
COPENHAGEN – Pregnant women with systemic lupus erythematosus (SLE) are at significantly higher risk of requiring transfusion, developing a cerebrovascular disorder, or developing acute renal failure than pregnant women without SLE, a review of data from an American national sample indicates.
Pregnant women with SLE also have a twofold-higher risk for premature delivery, and a threefold risk of having a fetus with intrauterine growth restriction than their pregnant counterparts without SLE, reported Bella Mehta, MBBS, MS, MD, a rheumatologist at the Hospital for Special Surgery in New York.
“Severe maternal morbidity and fetal morbidity still remain high, but this work can help inform physicians and counsel patients for pregnancy planning and management,” she said at the annual European Congress of Rheumatology.
Although in-hospital maternal and fetal mortality rates for women with SLE have declined over the past 2 decades, the same cannot be said for morbidities, prompting the investigators to conduct a study to determine the proportion of fetal and maternal morbidity in SLE deliveries, compared with non-SLE deliveries over a decade.
Inpatient Sample
Dr. Mehta and colleagues studied retrospective data on 40 million delivery-related admissions from the National Inpatient Sample database. Of these patients, 51,161 had a diagnosis of SLE.
They identified all delivery-related hospital admissions for patients with and without SLE from 2008 through 2017 using diagnostic codes.
The researchers looked at fetal morbidity indicators, including preterm delivery and intrauterine growth restriction, and used the Centers for Disease Control and Prevention standard definition of severe maternal morbidity as “unexpected outcomes of labor and delivery that result in significant short- or long- term consequences to a woman’s health.”
They identified 21 severe maternal morbidity outcomes, including blood transfusion requirements, acute renal failure, eclampsia and disseminated intravascular coagulation, cardiovascular and peripheral vascular disorders, and general medical issues (hysterectomy, shock, sepsis, adult respiratory distress syndrome, severe anesthesia complications, temporary tracheostomy, and ventilation).
Study results
Women with SLE were slightly older at the time of delivery (mean age, 30.05 vs. 29.19 years) and had more comorbidities, according to the Elixhauser Comorbidity Scale, with 97.84% of women in this group having one to four comorbidities, compared with 19.4% of women without SLE.

Dr. Mehta acknowledged that the study was limited by the inability to capture outpatient deliveries, although she noted that only about 1.3% of deliveries in the United States occur outside the inpatient setting.
In addition, she noted that the database does not include information on lupus disease activity, Apgar scores, SLE flares, the presence of nephritis, antiphospholipid or anti-Ro/SSA antibodies, or medication use.
A rheumatologist who was not involved in the study said in an interview that the data from this study are in line with those in other recently published studies.
“The problem is that these data were not corrected for further disease activity or drugs,” said Frauke Förger, MD, professor of rheumatology and immunology at the University of Bern (Switzerland), who comoderated the oral abstract session where the data were presented.
She said prospective studies that adjusted for factors such as SLE disease activity and medication use will be required to give clinicians a better understanding of how to manage pregnancies in women with SLE.
The study was supported by an award from Weill Cornell Medicine. Dr. Mehta and Dr. Förger reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
COPENHAGEN – Pregnant women with systemic lupus erythematosus (SLE) are at significantly higher risk of requiring transfusion, developing a cerebrovascular disorder, or developing acute renal failure than pregnant women without SLE, a review of data from an American national sample indicates.
Pregnant women with SLE also have a twofold-higher risk for premature delivery, and a threefold risk of having a fetus with intrauterine growth restriction than their pregnant counterparts without SLE, reported Bella Mehta, MBBS, MS, MD, a rheumatologist at the Hospital for Special Surgery in New York.
“Severe maternal morbidity and fetal morbidity still remain high, but this work can help inform physicians and counsel patients for pregnancy planning and management,” she said at the annual European Congress of Rheumatology.
Although in-hospital maternal and fetal mortality rates for women with SLE have declined over the past 2 decades, the same cannot be said for morbidities, prompting the investigators to conduct a study to determine the proportion of fetal and maternal morbidity in SLE deliveries, compared with non-SLE deliveries over a decade.
Inpatient Sample
Dr. Mehta and colleagues studied retrospective data on 40 million delivery-related admissions from the National Inpatient Sample database. Of these patients, 51,161 had a diagnosis of SLE.
They identified all delivery-related hospital admissions for patients with and without SLE from 2008 through 2017 using diagnostic codes.
The researchers looked at fetal morbidity indicators, including preterm delivery and intrauterine growth restriction, and used the Centers for Disease Control and Prevention standard definition of severe maternal morbidity as “unexpected outcomes of labor and delivery that result in significant short- or long- term consequences to a woman’s health.”
They identified 21 severe maternal morbidity outcomes, including blood transfusion requirements, acute renal failure, eclampsia and disseminated intravascular coagulation, cardiovascular and peripheral vascular disorders, and general medical issues (hysterectomy, shock, sepsis, adult respiratory distress syndrome, severe anesthesia complications, temporary tracheostomy, and ventilation).
Study results
Women with SLE were slightly older at the time of delivery (mean age, 30.05 vs. 29.19 years) and had more comorbidities, according to the Elixhauser Comorbidity Scale, with 97.84% of women in this group having one to four comorbidities, compared with 19.4% of women without SLE.

Dr. Mehta acknowledged that the study was limited by the inability to capture outpatient deliveries, although she noted that only about 1.3% of deliveries in the United States occur outside the inpatient setting.
In addition, she noted that the database does not include information on lupus disease activity, Apgar scores, SLE flares, the presence of nephritis, antiphospholipid or anti-Ro/SSA antibodies, or medication use.
A rheumatologist who was not involved in the study said in an interview that the data from this study are in line with those in other recently published studies.
“The problem is that these data were not corrected for further disease activity or drugs,” said Frauke Förger, MD, professor of rheumatology and immunology at the University of Bern (Switzerland), who comoderated the oral abstract session where the data were presented.
She said prospective studies that adjusted for factors such as SLE disease activity and medication use will be required to give clinicians a better understanding of how to manage pregnancies in women with SLE.
The study was supported by an award from Weill Cornell Medicine. Dr. Mehta and Dr. Förger reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
AT THE EULAR 2022 CONGRESS
At-home vagus nerve stimulation promising for postpartum depression
At-home, noninvasive auricular vagus nerve stimulation (aVNS) therapy is well-tolerated and associated with a significant reduction in postpartum depressive and anxiety symptoms, new research suggests.
In a small proof-of-concept pilot study of 25 women with postpartum depression receiving 6 weeks of daily aVNS treatment, results showed that 74% achieved response and 61% achieved remission, as shown in reduced scores on the Hamilton Rating Scale for Depression (HAM-D17).
Although invasive electrical stimulation of the vagus nerve was approved by the U.S. Food and Drug Administration for treatment-resistant depression in 2005, it involves risk for implantation, infection, and significant side effects, coinvestigator Kristina M. Deligiannidis, MD, director, Women’s Behavioral Health, Zucker Hillside Hospital, Northwell Health, Glen Oaks, New York, told this news organization.
“This newer approach, transcutaneous auricular VNS, is non-invasive, is well tolerated, and has shown initial efficacy in major depression in men and women,” she said.
The findings were presented at the virtual American Society of Clinical Psychopharmacology (ASCP) Annual Meeting.
Potential alternative to meds
“Given that aVNS is a non-invasive treatment which can be administered at home, we wanted to test if this approach was safe, feasible, and could reduce depressive symptoms in women with postpartum depression, as many of these women have barriers to accessing current treatments,” Dr. Deligiannidis said.
Auricular VNS uses surface skin electrodes to stimulate nerve endings of a branch of the vagus nerve, located on the surface of the outer ear. Those nerve endings travel to the brain where they have been shown to modulate brain communication in areas important for mood and anxiety regulation, she said.
Dr. Deligiannidis noted that evidence-based treatments for postpartum depression include psychotherapies and antidepressants. However, some women have difficulty accessing weekly psychotherapy, and, when antidepressants are indicated, many are reluctant to take them if they are breastfeeding because of concerns about the medications getting into their breast milk, she said.
Although most antidepressants are safe in lactation, many women postpone antidepressant treatment until they have finished breastfeeding, which can postpone their postpartum depression treatment, Dr. Deligiannidis added.
“At home treatments reduce many barriers women have to current treatments, and this intervention [of aVNS] does not impact breastfeeding, as it is not a medication approach,” she said.
The researchers enrolled 25 women (mean age, 33.7 years) diagnosed with postpartum depression. Ten of the women (40%) were on a stable dose of antidepressant medication.
The participants self-administered 6 weeks of open-label aVNS for 15 minutes daily at home. They were then observed without intervention for an additional 2 weeks. The women also completed medical, psychiatric, and safety interviews throughout the study period.
Promising findings
At baseline, the mean HAM-D17 was 18.4 and was similar for those on (17.8) and off (18.9) antidepressants.
By week 6, the mean HAM-D17 total score decreased by 9.7 points overall, compared with baseline score. For participants on antidepressants, the HAM-D17 decreased by 8.7 points; for women off antidepressants, it decreased by 10.3 points.
In addition, 74% of the women achieved a response to the therapy, and 61% achieved remission of their depressive symptoms.
The most common adverse effects were discomfort (n = 5 patients), headache (n = 3), and dizziness (n = 2). All resolved without intervention.
Commenting on the findings, Anita Clayton, MD, professor and chair, department of psychiatry and neurobehavioral sciences, University of Virginia School of Medicine, Charlottesville, said the study was “quite interesting.”
Dr. Clayton, who was not involved with the research, also noted the “pretty high” response and remission rates.
“So, I think this does have promise, and it would be worth doing a study where you look at placebo versus this treatment,” she said.
“Many women are fearful of taking medicines postpartum, even peripartum, unless they have had pre-existing severe depression. This is not a medicine, and it sounds like it could be useful even in people who are pregnant, although it’s harder to do studies in pregnant women,” Dr. Clayton added.
The study was funded by Nesos Corporation. Dr. Deligiannidis received contracted research funds from Nesos Corporation to conduct this study. She also serves as a consultant to Sage Therapeutics, Brii Biosciences, and GH Research. Dr. Clayton reports financial relationships with Dare Bioscience, Janssen, Praxis Precision Medicines, Relmada Therapeutics, Sage Therapeutics, AbbVie, Brii Biosciences, Fabre-Kramer, Field Trip Health, Mind Cure Health, Ovoca Bio, PureTech Health, S1 Biopharma, Takeda/Lundbeck, Vella Bioscience, WCG MedAvante-ProPhase, Ballantine Books/Random House, Changes in Sexual Functioning Questionnaire, Guilford Publications, Euthymics Bioscience, and Mediflix.
A version of this article first appeared on Medscape.com.
At-home, noninvasive auricular vagus nerve stimulation (aVNS) therapy is well-tolerated and associated with a significant reduction in postpartum depressive and anxiety symptoms, new research suggests.
In a small proof-of-concept pilot study of 25 women with postpartum depression receiving 6 weeks of daily aVNS treatment, results showed that 74% achieved response and 61% achieved remission, as shown in reduced scores on the Hamilton Rating Scale for Depression (HAM-D17).
Although invasive electrical stimulation of the vagus nerve was approved by the U.S. Food and Drug Administration for treatment-resistant depression in 2005, it involves risk for implantation, infection, and significant side effects, coinvestigator Kristina M. Deligiannidis, MD, director, Women’s Behavioral Health, Zucker Hillside Hospital, Northwell Health, Glen Oaks, New York, told this news organization.
“This newer approach, transcutaneous auricular VNS, is non-invasive, is well tolerated, and has shown initial efficacy in major depression in men and women,” she said.
The findings were presented at the virtual American Society of Clinical Psychopharmacology (ASCP) Annual Meeting.
Potential alternative to meds
“Given that aVNS is a non-invasive treatment which can be administered at home, we wanted to test if this approach was safe, feasible, and could reduce depressive symptoms in women with postpartum depression, as many of these women have barriers to accessing current treatments,” Dr. Deligiannidis said.
Auricular VNS uses surface skin electrodes to stimulate nerve endings of a branch of the vagus nerve, located on the surface of the outer ear. Those nerve endings travel to the brain where they have been shown to modulate brain communication in areas important for mood and anxiety regulation, she said.
Dr. Deligiannidis noted that evidence-based treatments for postpartum depression include psychotherapies and antidepressants. However, some women have difficulty accessing weekly psychotherapy, and, when antidepressants are indicated, many are reluctant to take them if they are breastfeeding because of concerns about the medications getting into their breast milk, she said.
Although most antidepressants are safe in lactation, many women postpone antidepressant treatment until they have finished breastfeeding, which can postpone their postpartum depression treatment, Dr. Deligiannidis added.
“At home treatments reduce many barriers women have to current treatments, and this intervention [of aVNS] does not impact breastfeeding, as it is not a medication approach,” she said.
The researchers enrolled 25 women (mean age, 33.7 years) diagnosed with postpartum depression. Ten of the women (40%) were on a stable dose of antidepressant medication.
The participants self-administered 6 weeks of open-label aVNS for 15 minutes daily at home. They were then observed without intervention for an additional 2 weeks. The women also completed medical, psychiatric, and safety interviews throughout the study period.
Promising findings
At baseline, the mean HAM-D17 was 18.4 and was similar for those on (17.8) and off (18.9) antidepressants.
By week 6, the mean HAM-D17 total score decreased by 9.7 points overall, compared with baseline score. For participants on antidepressants, the HAM-D17 decreased by 8.7 points; for women off antidepressants, it decreased by 10.3 points.
In addition, 74% of the women achieved a response to the therapy, and 61% achieved remission of their depressive symptoms.
The most common adverse effects were discomfort (n = 5 patients), headache (n = 3), and dizziness (n = 2). All resolved without intervention.
Commenting on the findings, Anita Clayton, MD, professor and chair, department of psychiatry and neurobehavioral sciences, University of Virginia School of Medicine, Charlottesville, said the study was “quite interesting.”
Dr. Clayton, who was not involved with the research, also noted the “pretty high” response and remission rates.
“So, I think this does have promise, and it would be worth doing a study where you look at placebo versus this treatment,” she said.
“Many women are fearful of taking medicines postpartum, even peripartum, unless they have had pre-existing severe depression. This is not a medicine, and it sounds like it could be useful even in people who are pregnant, although it’s harder to do studies in pregnant women,” Dr. Clayton added.
The study was funded by Nesos Corporation. Dr. Deligiannidis received contracted research funds from Nesos Corporation to conduct this study. She also serves as a consultant to Sage Therapeutics, Brii Biosciences, and GH Research. Dr. Clayton reports financial relationships with Dare Bioscience, Janssen, Praxis Precision Medicines, Relmada Therapeutics, Sage Therapeutics, AbbVie, Brii Biosciences, Fabre-Kramer, Field Trip Health, Mind Cure Health, Ovoca Bio, PureTech Health, S1 Biopharma, Takeda/Lundbeck, Vella Bioscience, WCG MedAvante-ProPhase, Ballantine Books/Random House, Changes in Sexual Functioning Questionnaire, Guilford Publications, Euthymics Bioscience, and Mediflix.
A version of this article first appeared on Medscape.com.
At-home, noninvasive auricular vagus nerve stimulation (aVNS) therapy is well-tolerated and associated with a significant reduction in postpartum depressive and anxiety symptoms, new research suggests.
In a small proof-of-concept pilot study of 25 women with postpartum depression receiving 6 weeks of daily aVNS treatment, results showed that 74% achieved response and 61% achieved remission, as shown in reduced scores on the Hamilton Rating Scale for Depression (HAM-D17).
Although invasive electrical stimulation of the vagus nerve was approved by the U.S. Food and Drug Administration for treatment-resistant depression in 2005, it involves risk for implantation, infection, and significant side effects, coinvestigator Kristina M. Deligiannidis, MD, director, Women’s Behavioral Health, Zucker Hillside Hospital, Northwell Health, Glen Oaks, New York, told this news organization.
“This newer approach, transcutaneous auricular VNS, is non-invasive, is well tolerated, and has shown initial efficacy in major depression in men and women,” she said.
The findings were presented at the virtual American Society of Clinical Psychopharmacology (ASCP) Annual Meeting.
Potential alternative to meds
“Given that aVNS is a non-invasive treatment which can be administered at home, we wanted to test if this approach was safe, feasible, and could reduce depressive symptoms in women with postpartum depression, as many of these women have barriers to accessing current treatments,” Dr. Deligiannidis said.
Auricular VNS uses surface skin electrodes to stimulate nerve endings of a branch of the vagus nerve, located on the surface of the outer ear. Those nerve endings travel to the brain where they have been shown to modulate brain communication in areas important for mood and anxiety regulation, she said.
Dr. Deligiannidis noted that evidence-based treatments for postpartum depression include psychotherapies and antidepressants. However, some women have difficulty accessing weekly psychotherapy, and, when antidepressants are indicated, many are reluctant to take them if they are breastfeeding because of concerns about the medications getting into their breast milk, she said.
Although most antidepressants are safe in lactation, many women postpone antidepressant treatment until they have finished breastfeeding, which can postpone their postpartum depression treatment, Dr. Deligiannidis added.
“At home treatments reduce many barriers women have to current treatments, and this intervention [of aVNS] does not impact breastfeeding, as it is not a medication approach,” she said.
The researchers enrolled 25 women (mean age, 33.7 years) diagnosed with postpartum depression. Ten of the women (40%) were on a stable dose of antidepressant medication.
The participants self-administered 6 weeks of open-label aVNS for 15 minutes daily at home. They were then observed without intervention for an additional 2 weeks. The women also completed medical, psychiatric, and safety interviews throughout the study period.
Promising findings
At baseline, the mean HAM-D17 was 18.4 and was similar for those on (17.8) and off (18.9) antidepressants.
By week 6, the mean HAM-D17 total score decreased by 9.7 points overall, compared with baseline score. For participants on antidepressants, the HAM-D17 decreased by 8.7 points; for women off antidepressants, it decreased by 10.3 points.
In addition, 74% of the women achieved a response to the therapy, and 61% achieved remission of their depressive symptoms.
The most common adverse effects were discomfort (n = 5 patients), headache (n = 3), and dizziness (n = 2). All resolved without intervention.
Commenting on the findings, Anita Clayton, MD, professor and chair, department of psychiatry and neurobehavioral sciences, University of Virginia School of Medicine, Charlottesville, said the study was “quite interesting.”
Dr. Clayton, who was not involved with the research, also noted the “pretty high” response and remission rates.
“So, I think this does have promise, and it would be worth doing a study where you look at placebo versus this treatment,” she said.
“Many women are fearful of taking medicines postpartum, even peripartum, unless they have had pre-existing severe depression. This is not a medicine, and it sounds like it could be useful even in people who are pregnant, although it’s harder to do studies in pregnant women,” Dr. Clayton added.
The study was funded by Nesos Corporation. Dr. Deligiannidis received contracted research funds from Nesos Corporation to conduct this study. She also serves as a consultant to Sage Therapeutics, Brii Biosciences, and GH Research. Dr. Clayton reports financial relationships with Dare Bioscience, Janssen, Praxis Precision Medicines, Relmada Therapeutics, Sage Therapeutics, AbbVie, Brii Biosciences, Fabre-Kramer, Field Trip Health, Mind Cure Health, Ovoca Bio, PureTech Health, S1 Biopharma, Takeda/Lundbeck, Vella Bioscience, WCG MedAvante-ProPhase, Ballantine Books/Random House, Changes in Sexual Functioning Questionnaire, Guilford Publications, Euthymics Bioscience, and Mediflix.
A version of this article first appeared on Medscape.com.
Five-year cervical screening interval safe for HPV-negative women
A 5-year cervical screening interval is as safe and effective for women who test negative for human papillomavirus (HPV) as are 3-year intervals, according to a new ‘real life’ study led by King’s College London (KCL) with researchers from the University of Manchester, and the NHS, on behalf of the HPV pilot steering group.
The study, published in The BMJ, used data from the HPV screening pilot to assess rates of detection of high-grade cervical intraepithelial neoplasia (CIN3+) and of cervical cancer following a negative HPV test. It confirmed that 5-yearly screening prevents as many cancers as screening at 3-year intervals, even in women who are not vaccinated against HPV.
Change to primary HPV testing since 2019
Before 2019, the NHS cervical screening program conducted cytology testing first, testing for HPV only if abnormalities were found. In 2019, following reporting of early results of the HPV pilot by the same researchers, the program in England switched to testing for HPV first, on the grounds that since having HPV infection comes before having abnormal cells, HPV testing would detect more women at risk of cervical cancer.
Following the switch to primary HPV testing, the same screening intervals were retained, meaning 3-yearly screening for those aged 24-49 years and testing every 5 years for women aged 50-64 years, or 3 years if they tested positive. However, the National Screening Committee had recommended that invites should be changed from 3 to 5 years for those in the under-50 age group found not to have high-risk HPV at their routine screening test.
For the latest study, funded by Cancer Research UK, the steering group researchers analyzed details for more than 1.3 million women who had attended screening for two rounds of the HPV screening pilot, the first from 2013 to 2016, with a follow-up to the end of 2019. By this time, the data set had doubled in size from the pilot study, and results had been linked with the national cancer registry.
They confirmed that HPV testing was more accurate than a cytology test, irrespective of whether the HPV test assay was DNA- or mRNA-based. With HPV testing, the risk of subsequent cytological changes more than halved overall. Eligible women under 50 who had a negative HPV screen in the first round had a much lower risk of detection of CIN3+ in the second round, with a rate of 1.21 in 1,000, compared with 4.52 in 1,000 after a negative cytology test.
Data support extension of the testing interval
“The study confirms that women in this age group are much less likely to develop clinically relevant cervical lesions and cervical cancer, 3 years after a negative HPV screen, compared with a negative smear test,” the researchers said.
They suggested that most women do not need to be screened as frequently as the current program allows, and that the data support an extension of the screening intervals, regardless of the test assay used, to 5 years after a negative HPV test in women aged 25-49 years, and even longer for women aged 50 years and older.
However, the screening interval for HPV-positive women who have negative HPV tests at early recall should be kept at 3 years, they said.
“These results are very reassuring,” said lead author Matejka Rebolj, PhD, senior epidemiologist at KCL. “They build on previous research that shows that following the introduction of HPV testing for cervical screening, a 5-year interval is at least as safe as the previous 3-year interval. Changing to 5-yearly screening will mean we can prevent just as many cancers as before, while allowing for fewer screens.”
Michelle Mitchell, Cancer Research UK’s chief executive, said: “This large study shows that offering cervical screening using HPV testing effectively prevents cervical cancer, without having to be screened as often. This builds on findings from years of research showing HPV testing is more accurate at predicting who is at risk of developing cervical cancer compared to the previous way of testing. As changes to the screening [programs] are made, they will be monitored to help ensure that cervical screening is as effective as possible for all who take part.”
If HPV is present, testing interval should remain every 3 years
Responding to the study, Theresa Freeman-Wang, MBChB, consultant gynecologist, president of the British Society for Colposcopy and Cervical Pathology, and spokesperson for the Royal College of Obstetricians and Gynaecologists, told this news organization: “England, Scotland, and Wales and many other countries now use HPV primary screening, which is much better at assessing risk than previous methods. HPV testing is more sensitive and accurate, so changes are picked up earlier.
“Studies have confirmed that if someone is HPV negative (i.e., HPV is not present in the screen test), intervals between tests can very safely be increased from 3 to 5 years.
“If HPV is present, then the program will automatically look for any abnormal cells. If there are no abnormalities, the woman will be advised to have a repeat screen test in a year. If the HPV remains present over 3 successive years or if abnormal cells are detected at any stage, she will be referred for a more detailed screening examination called a colposcopy.
“It’s important that with any change like this, there is clear information available to explain what these changes mean.
“We have an effective cervical screening program in the UK that has significantly reduced the number of cases and deaths from this preventable cancer.
“HPV screening every 5 years is safe and to be fully effective it is vital that women take up the invitation for cervical screening when called.”
A version of this article first appeared on Medscape UK.
A 5-year cervical screening interval is as safe and effective for women who test negative for human papillomavirus (HPV) as are 3-year intervals, according to a new ‘real life’ study led by King’s College London (KCL) with researchers from the University of Manchester, and the NHS, on behalf of the HPV pilot steering group.
The study, published in The BMJ, used data from the HPV screening pilot to assess rates of detection of high-grade cervical intraepithelial neoplasia (CIN3+) and of cervical cancer following a negative HPV test. It confirmed that 5-yearly screening prevents as many cancers as screening at 3-year intervals, even in women who are not vaccinated against HPV.
Change to primary HPV testing since 2019
Before 2019, the NHS cervical screening program conducted cytology testing first, testing for HPV only if abnormalities were found. In 2019, following reporting of early results of the HPV pilot by the same researchers, the program in England switched to testing for HPV first, on the grounds that since having HPV infection comes before having abnormal cells, HPV testing would detect more women at risk of cervical cancer.
Following the switch to primary HPV testing, the same screening intervals were retained, meaning 3-yearly screening for those aged 24-49 years and testing every 5 years for women aged 50-64 years, or 3 years if they tested positive. However, the National Screening Committee had recommended that invites should be changed from 3 to 5 years for those in the under-50 age group found not to have high-risk HPV at their routine screening test.
For the latest study, funded by Cancer Research UK, the steering group researchers analyzed details for more than 1.3 million women who had attended screening for two rounds of the HPV screening pilot, the first from 2013 to 2016, with a follow-up to the end of 2019. By this time, the data set had doubled in size from the pilot study, and results had been linked with the national cancer registry.
They confirmed that HPV testing was more accurate than a cytology test, irrespective of whether the HPV test assay was DNA- or mRNA-based. With HPV testing, the risk of subsequent cytological changes more than halved overall. Eligible women under 50 who had a negative HPV screen in the first round had a much lower risk of detection of CIN3+ in the second round, with a rate of 1.21 in 1,000, compared with 4.52 in 1,000 after a negative cytology test.
Data support extension of the testing interval
“The study confirms that women in this age group are much less likely to develop clinically relevant cervical lesions and cervical cancer, 3 years after a negative HPV screen, compared with a negative smear test,” the researchers said.
They suggested that most women do not need to be screened as frequently as the current program allows, and that the data support an extension of the screening intervals, regardless of the test assay used, to 5 years after a negative HPV test in women aged 25-49 years, and even longer for women aged 50 years and older.
However, the screening interval for HPV-positive women who have negative HPV tests at early recall should be kept at 3 years, they said.
“These results are very reassuring,” said lead author Matejka Rebolj, PhD, senior epidemiologist at KCL. “They build on previous research that shows that following the introduction of HPV testing for cervical screening, a 5-year interval is at least as safe as the previous 3-year interval. Changing to 5-yearly screening will mean we can prevent just as many cancers as before, while allowing for fewer screens.”
Michelle Mitchell, Cancer Research UK’s chief executive, said: “This large study shows that offering cervical screening using HPV testing effectively prevents cervical cancer, without having to be screened as often. This builds on findings from years of research showing HPV testing is more accurate at predicting who is at risk of developing cervical cancer compared to the previous way of testing. As changes to the screening [programs] are made, they will be monitored to help ensure that cervical screening is as effective as possible for all who take part.”
If HPV is present, testing interval should remain every 3 years
Responding to the study, Theresa Freeman-Wang, MBChB, consultant gynecologist, president of the British Society for Colposcopy and Cervical Pathology, and spokesperson for the Royal College of Obstetricians and Gynaecologists, told this news organization: “England, Scotland, and Wales and many other countries now use HPV primary screening, which is much better at assessing risk than previous methods. HPV testing is more sensitive and accurate, so changes are picked up earlier.
“Studies have confirmed that if someone is HPV negative (i.e., HPV is not present in the screen test), intervals between tests can very safely be increased from 3 to 5 years.
“If HPV is present, then the program will automatically look for any abnormal cells. If there are no abnormalities, the woman will be advised to have a repeat screen test in a year. If the HPV remains present over 3 successive years or if abnormal cells are detected at any stage, she will be referred for a more detailed screening examination called a colposcopy.
“It’s important that with any change like this, there is clear information available to explain what these changes mean.
“We have an effective cervical screening program in the UK that has significantly reduced the number of cases and deaths from this preventable cancer.
“HPV screening every 5 years is safe and to be fully effective it is vital that women take up the invitation for cervical screening when called.”
A version of this article first appeared on Medscape UK.
A 5-year cervical screening interval is as safe and effective for women who test negative for human papillomavirus (HPV) as are 3-year intervals, according to a new ‘real life’ study led by King’s College London (KCL) with researchers from the University of Manchester, and the NHS, on behalf of the HPV pilot steering group.
The study, published in The BMJ, used data from the HPV screening pilot to assess rates of detection of high-grade cervical intraepithelial neoplasia (CIN3+) and of cervical cancer following a negative HPV test. It confirmed that 5-yearly screening prevents as many cancers as screening at 3-year intervals, even in women who are not vaccinated against HPV.
Change to primary HPV testing since 2019
Before 2019, the NHS cervical screening program conducted cytology testing first, testing for HPV only if abnormalities were found. In 2019, following reporting of early results of the HPV pilot by the same researchers, the program in England switched to testing for HPV first, on the grounds that since having HPV infection comes before having abnormal cells, HPV testing would detect more women at risk of cervical cancer.
Following the switch to primary HPV testing, the same screening intervals were retained, meaning 3-yearly screening for those aged 24-49 years and testing every 5 years for women aged 50-64 years, or 3 years if they tested positive. However, the National Screening Committee had recommended that invites should be changed from 3 to 5 years for those in the under-50 age group found not to have high-risk HPV at their routine screening test.
For the latest study, funded by Cancer Research UK, the steering group researchers analyzed details for more than 1.3 million women who had attended screening for two rounds of the HPV screening pilot, the first from 2013 to 2016, with a follow-up to the end of 2019. By this time, the data set had doubled in size from the pilot study, and results had been linked with the national cancer registry.
They confirmed that HPV testing was more accurate than a cytology test, irrespective of whether the HPV test assay was DNA- or mRNA-based. With HPV testing, the risk of subsequent cytological changes more than halved overall. Eligible women under 50 who had a negative HPV screen in the first round had a much lower risk of detection of CIN3+ in the second round, with a rate of 1.21 in 1,000, compared with 4.52 in 1,000 after a negative cytology test.
Data support extension of the testing interval
“The study confirms that women in this age group are much less likely to develop clinically relevant cervical lesions and cervical cancer, 3 years after a negative HPV screen, compared with a negative smear test,” the researchers said.
They suggested that most women do not need to be screened as frequently as the current program allows, and that the data support an extension of the screening intervals, regardless of the test assay used, to 5 years after a negative HPV test in women aged 25-49 years, and even longer for women aged 50 years and older.
However, the screening interval for HPV-positive women who have negative HPV tests at early recall should be kept at 3 years, they said.
“These results are very reassuring,” said lead author Matejka Rebolj, PhD, senior epidemiologist at KCL. “They build on previous research that shows that following the introduction of HPV testing for cervical screening, a 5-year interval is at least as safe as the previous 3-year interval. Changing to 5-yearly screening will mean we can prevent just as many cancers as before, while allowing for fewer screens.”
Michelle Mitchell, Cancer Research UK’s chief executive, said: “This large study shows that offering cervical screening using HPV testing effectively prevents cervical cancer, without having to be screened as often. This builds on findings from years of research showing HPV testing is more accurate at predicting who is at risk of developing cervical cancer compared to the previous way of testing. As changes to the screening [programs] are made, they will be monitored to help ensure that cervical screening is as effective as possible for all who take part.”
If HPV is present, testing interval should remain every 3 years
Responding to the study, Theresa Freeman-Wang, MBChB, consultant gynecologist, president of the British Society for Colposcopy and Cervical Pathology, and spokesperson for the Royal College of Obstetricians and Gynaecologists, told this news organization: “England, Scotland, and Wales and many other countries now use HPV primary screening, which is much better at assessing risk than previous methods. HPV testing is more sensitive and accurate, so changes are picked up earlier.
“Studies have confirmed that if someone is HPV negative (i.e., HPV is not present in the screen test), intervals between tests can very safely be increased from 3 to 5 years.
“If HPV is present, then the program will automatically look for any abnormal cells. If there are no abnormalities, the woman will be advised to have a repeat screen test in a year. If the HPV remains present over 3 successive years or if abnormal cells are detected at any stage, she will be referred for a more detailed screening examination called a colposcopy.
“It’s important that with any change like this, there is clear information available to explain what these changes mean.
“We have an effective cervical screening program in the UK that has significantly reduced the number of cases and deaths from this preventable cancer.
“HPV screening every 5 years is safe and to be fully effective it is vital that women take up the invitation for cervical screening when called.”
A version of this article first appeared on Medscape UK.
FROM THE BMJ
Tin in permanent contraception implants causes toxicity
Essure implants arrived on the market in 2002 as permanent contraception for women older than age 45 years with children. They were recalled in 2017. Presented as an alternative to laparoscopic tubal ligation, this medical device resulted in rare side effects affecting thousands of women, most notably the nervous system, cardiovascular system, endocrine system, and musculoskeletal system.
Implant analysis protocol
“My research focuses on a variety of medical devices, mostly joint replacements, and more specifically, hip replacements. I look at how these materials behave in humans and how the wear debris affects the body,” explained Ana Maria Trunfio-Sfarghiu, bioengineering expert and research associate with the French National Center for Scientific Research at the Lyon National Institute of Applied Sciences’ Contact and Structure Mechanics Laboratory.
“The problems with Essure implants started with a woman who had been using one for about 10 years and was experiencing side effects such as trouble concentrating and focusing, significant vaginal bleeding, extreme tiredness, hair loss, etc. She had the implant removed, and we retrieved it from her gynecologist and analyzed it alongside other implants,” said Ms. Trunfio-Sfarghiu.
“Together with the hospital, we set up an implant analysis protocol. We visited hospital teams to demonstrate how to prepare the biopsies, embedded in paraffin blocks, before sending them to us for analysis. We gave the same specimen preparation instructions for all subjects,” Ms. Trunfio-Sfarghiu explained.
After a year of clinical analysis, the Journal of Trace Elements in Medicine and Biology published an article about 18 cases.
Implant weld corrosion
The Essure implant measures a few centimeters long and resembles a small spring. Once it is released inside the fallopian tube, its goal is to create inflammation and block the tube. It triggers fibrosis, which prevents the sperm from reaching the egg. Premarketing tests had shown that the fibrosis surrounding the implant would keep it from moving. However, the pharmaceutical company hadn’t assessed the mechanical integrity of the spring weld, which was made of silver-tin.
During their analysis in collaboration with the Minapath laboratory, Ms. Trunfio-Sfarghiu’s team found that the weld had corroded and that tin particles had been released into the subjects’ bodies. “The study included about 40 women, and we found tin in all of them,” said Ms. Trunfio-Sfarghiu.
This weld corrosion has several possible consequences. “When the implant degrades, it can travel anywhere in the pelvis, like a needle moving through the body with no apparent destination. The surgeons who operate to remove it describe similar surgeries in military medicine when the patient has been hit by a bullet!”
Organotin toxicity
Although tin is not especially toxic for the body when ingested, it can bind to organic compounds if it passes through to the blood. “When tin binds to a carbon atom, it becomes organotin, a neurotoxin,” said Ms. Trunfio-Sfarghiu.
She said that this organotin can travel to the brain and trigger symptoms like those found in patients with Essure implants. “For the time being, there is insufficient data to assert that we found organotin in all subjects. Another more in-depth study would be needed to assess migration to the brain. For the past 2 years, we have tried to obtain academic funding to continue our research, so far without success. Academic and political authorities seem to be a bit scared of what we’ve found,” said Ms. Trunfio-Sfarghiu.
For her, “it’s how the implant was marketed that is problematic. The implant was designed to create local inflammation, inflammation in itself being difficult to control. Some women need to have their entire uterus and ovaries removed to resolve problems caused by the implant.”
Harm in the United States
Ms. Trunfio-Sfarghiu’s research has helped American victims obtain acknowledgment of their suffering in the United States. “But the harm caused to women by defective implants has yet to be acknowledged in France,” she added.
She explained that Essure was recalled in 2017 because sales were poor, not because it was deemed dangerous. Her conclusion? “No implant that creates inflammation should be authorized, especially if there is a surgical alternative, which there is here: tubal ligation.”
A version of this article appeared on Medscape.com. This article was translated from the Medscape French edition.
Essure implants arrived on the market in 2002 as permanent contraception for women older than age 45 years with children. They were recalled in 2017. Presented as an alternative to laparoscopic tubal ligation, this medical device resulted in rare side effects affecting thousands of women, most notably the nervous system, cardiovascular system, endocrine system, and musculoskeletal system.
Implant analysis protocol
“My research focuses on a variety of medical devices, mostly joint replacements, and more specifically, hip replacements. I look at how these materials behave in humans and how the wear debris affects the body,” explained Ana Maria Trunfio-Sfarghiu, bioengineering expert and research associate with the French National Center for Scientific Research at the Lyon National Institute of Applied Sciences’ Contact and Structure Mechanics Laboratory.
“The problems with Essure implants started with a woman who had been using one for about 10 years and was experiencing side effects such as trouble concentrating and focusing, significant vaginal bleeding, extreme tiredness, hair loss, etc. She had the implant removed, and we retrieved it from her gynecologist and analyzed it alongside other implants,” said Ms. Trunfio-Sfarghiu.
“Together with the hospital, we set up an implant analysis protocol. We visited hospital teams to demonstrate how to prepare the biopsies, embedded in paraffin blocks, before sending them to us for analysis. We gave the same specimen preparation instructions for all subjects,” Ms. Trunfio-Sfarghiu explained.
After a year of clinical analysis, the Journal of Trace Elements in Medicine and Biology published an article about 18 cases.
Implant weld corrosion
The Essure implant measures a few centimeters long and resembles a small spring. Once it is released inside the fallopian tube, its goal is to create inflammation and block the tube. It triggers fibrosis, which prevents the sperm from reaching the egg. Premarketing tests had shown that the fibrosis surrounding the implant would keep it from moving. However, the pharmaceutical company hadn’t assessed the mechanical integrity of the spring weld, which was made of silver-tin.
During their analysis in collaboration with the Minapath laboratory, Ms. Trunfio-Sfarghiu’s team found that the weld had corroded and that tin particles had been released into the subjects’ bodies. “The study included about 40 women, and we found tin in all of them,” said Ms. Trunfio-Sfarghiu.
This weld corrosion has several possible consequences. “When the implant degrades, it can travel anywhere in the pelvis, like a needle moving through the body with no apparent destination. The surgeons who operate to remove it describe similar surgeries in military medicine when the patient has been hit by a bullet!”
Organotin toxicity
Although tin is not especially toxic for the body when ingested, it can bind to organic compounds if it passes through to the blood. “When tin binds to a carbon atom, it becomes organotin, a neurotoxin,” said Ms. Trunfio-Sfarghiu.
She said that this organotin can travel to the brain and trigger symptoms like those found in patients with Essure implants. “For the time being, there is insufficient data to assert that we found organotin in all subjects. Another more in-depth study would be needed to assess migration to the brain. For the past 2 years, we have tried to obtain academic funding to continue our research, so far without success. Academic and political authorities seem to be a bit scared of what we’ve found,” said Ms. Trunfio-Sfarghiu.
For her, “it’s how the implant was marketed that is problematic. The implant was designed to create local inflammation, inflammation in itself being difficult to control. Some women need to have their entire uterus and ovaries removed to resolve problems caused by the implant.”
Harm in the United States
Ms. Trunfio-Sfarghiu’s research has helped American victims obtain acknowledgment of their suffering in the United States. “But the harm caused to women by defective implants has yet to be acknowledged in France,” she added.
She explained that Essure was recalled in 2017 because sales were poor, not because it was deemed dangerous. Her conclusion? “No implant that creates inflammation should be authorized, especially if there is a surgical alternative, which there is here: tubal ligation.”
A version of this article appeared on Medscape.com. This article was translated from the Medscape French edition.
Essure implants arrived on the market in 2002 as permanent contraception for women older than age 45 years with children. They were recalled in 2017. Presented as an alternative to laparoscopic tubal ligation, this medical device resulted in rare side effects affecting thousands of women, most notably the nervous system, cardiovascular system, endocrine system, and musculoskeletal system.
Implant analysis protocol
“My research focuses on a variety of medical devices, mostly joint replacements, and more specifically, hip replacements. I look at how these materials behave in humans and how the wear debris affects the body,” explained Ana Maria Trunfio-Sfarghiu, bioengineering expert and research associate with the French National Center for Scientific Research at the Lyon National Institute of Applied Sciences’ Contact and Structure Mechanics Laboratory.
“The problems with Essure implants started with a woman who had been using one for about 10 years and was experiencing side effects such as trouble concentrating and focusing, significant vaginal bleeding, extreme tiredness, hair loss, etc. She had the implant removed, and we retrieved it from her gynecologist and analyzed it alongside other implants,” said Ms. Trunfio-Sfarghiu.
“Together with the hospital, we set up an implant analysis protocol. We visited hospital teams to demonstrate how to prepare the biopsies, embedded in paraffin blocks, before sending them to us for analysis. We gave the same specimen preparation instructions for all subjects,” Ms. Trunfio-Sfarghiu explained.
After a year of clinical analysis, the Journal of Trace Elements in Medicine and Biology published an article about 18 cases.
Implant weld corrosion
The Essure implant measures a few centimeters long and resembles a small spring. Once it is released inside the fallopian tube, its goal is to create inflammation and block the tube. It triggers fibrosis, which prevents the sperm from reaching the egg. Premarketing tests had shown that the fibrosis surrounding the implant would keep it from moving. However, the pharmaceutical company hadn’t assessed the mechanical integrity of the spring weld, which was made of silver-tin.
During their analysis in collaboration with the Minapath laboratory, Ms. Trunfio-Sfarghiu’s team found that the weld had corroded and that tin particles had been released into the subjects’ bodies. “The study included about 40 women, and we found tin in all of them,” said Ms. Trunfio-Sfarghiu.
This weld corrosion has several possible consequences. “When the implant degrades, it can travel anywhere in the pelvis, like a needle moving through the body with no apparent destination. The surgeons who operate to remove it describe similar surgeries in military medicine when the patient has been hit by a bullet!”
Organotin toxicity
Although tin is not especially toxic for the body when ingested, it can bind to organic compounds if it passes through to the blood. “When tin binds to a carbon atom, it becomes organotin, a neurotoxin,” said Ms. Trunfio-Sfarghiu.
She said that this organotin can travel to the brain and trigger symptoms like those found in patients with Essure implants. “For the time being, there is insufficient data to assert that we found organotin in all subjects. Another more in-depth study would be needed to assess migration to the brain. For the past 2 years, we have tried to obtain academic funding to continue our research, so far without success. Academic and political authorities seem to be a bit scared of what we’ve found,” said Ms. Trunfio-Sfarghiu.
For her, “it’s how the implant was marketed that is problematic. The implant was designed to create local inflammation, inflammation in itself being difficult to control. Some women need to have their entire uterus and ovaries removed to resolve problems caused by the implant.”
Harm in the United States
Ms. Trunfio-Sfarghiu’s research has helped American victims obtain acknowledgment of their suffering in the United States. “But the harm caused to women by defective implants has yet to be acknowledged in France,” she added.
She explained that Essure was recalled in 2017 because sales were poor, not because it was deemed dangerous. Her conclusion? “No implant that creates inflammation should be authorized, especially if there is a surgical alternative, which there is here: tubal ligation.”
A version of this article appeared on Medscape.com. This article was translated from the Medscape French edition.






