User login
Cardiology News is an independent news source that provides cardiologists with timely and relevant news and commentary about clinical developments and the impact of health care policy on cardiology and the cardiologist's practice. Cardiology News Digital Network is the online destination and multimedia properties of Cardiology News, the independent news publication for cardiologists. Cardiology news is the leading source of news and commentary about clinical developments in cardiology as well as health care policy and regulations that affect the cardiologist's practice. Cardiology News Digital Network is owned by Frontline Medical Communications.
Systemic Bias in AI Models May Undermine Diagnostic Accuracy
Systematically biased artificial intelligence (AI) models did not improve clinicians’ accuracy in diagnosing hospitalized patients, based on data from more than 450 clinicians.
“Artificial Intelligence (AI) could support clinicians in their diagnostic decisions of hospitalized patients but could also be biased and cause potential harm,” said Sarah Jabbour, MSE, a PhD candidate in computer science and engineering at the University of Michigan, Ann Arbor, in an interview.
“Regulatory guidance has suggested that the use of AI explanations could mitigate these harms, but the effectiveness of using AI explanations has not been established,” she said.
To examine whether AI explanations can be effective in mitigating the potential harms of systemic bias in AI models, Ms. Jabbour and colleagues conducted a randomized clinical vignette survey study. The survey was administered between April 2022 and January 2023 across 13 states, and the study population included hospitalist physicians, nurse practitioners, and physician assistants. The results were published in JAMA.
Participants were randomized to AI predictions with AI explanations (226 clinicians) or without AI explanations (231 clinicians).
The primary outcome was diagnostic accuracy for pneumonia, heart failure, and chronic obstructive pulmonary disease, defined as the number of correct diagnoses over the total number of assessments, the researchers wrote.
The clinicians viewed nine clinical vignettes of patients hospitalized with acute respiratory failure, including their presenting symptoms, physical examination, laboratory results, and chest radiographs. Clinicians viewed two vignettes with no AI model input to establish baseline diagnostic accuracy. They made three assessments in each vignette, one for each diagnosis. The order of the vignettes was two without AI predictions (to establish baseline diagnostic accuracy), six with AI predictions, and one with a clinical consultation by a hypothetical colleague. The vignettes included standard and systematically biased AI models.
The baseline diagnostic accuracy was 73% for the diagnoses of pneumonia, heart failure, and chronic obstructive pulmonary disease. Clinicians’ accuracy increased by 2.9% when they viewed a standard diagnostic AI model without explanations and by 4.4% when they viewed models with AI explanations.
However, clinicians’ accuracy decreased by 11.3% after viewing systematically biased AI model predictions without explanations compared with baseline, and biased AI model predictions with explanations decreased accuracy by 9.1%.
The decrease in accuracy with systematically biased AI predictions without explanations was mainly attributable to a decrease in the participants’ diagnostic specificity, the researchers noted, but the addition of explanations did little to improve it, the researchers said.
Potentially Useful but Still Imperfect
The findings were limited by several factors including the use of a web-based survey, which differs from surveys in a clinical setting, the researchers wrote. Other limitations included the younger than average study population, and the focus on the clinicians making treatment decisions, vs other clinicians who might have a better understanding of the AI explanations.
“In our study, explanations were presented in a way that were considered to be obvious, where the AI model was completely focused on areas of the chest X-rays unrelated to the clinical condition,” Ms. Jabbour told this news organization. “We hypothesized that if presented with such explanations, the participants in our study would notice that the model was behaving incorrectly and not rely on its predictions. This was surprisingly not the case, and the explanations when presented alongside biased AI predictions had seemingly no effect in mitigating clinicians’ overreliance on biased AI,” she said.
“AI is being developed at an extraordinary rate, and our study shows that it has the potential to improve clinical decision-making. At the same time, it could harm clinical decision-making when biased,” Ms. Jabbour said. “We must be thoughtful about how to carefully integrate AI into clinical workflows, with the goal of improving clinical care while not introducing systematic errors or harming patients,” she added.
Looking ahead, “There are several potential research areas that could be explored,” said Ms. Jabbour. “Researchers should focus on careful validation of AI models to identify biased model behavior prior to deployment. AI researchers should also continue including and communicating with clinicians during the development of AI tools to better understand clinicians’ needs and how they interact with AI,” she said. “This is not an exhaustive list of research directions, and it will take much discussion between experts across disciplines such as AI, human computer interaction, and medicine to ultimately deploy AI safely into clinical care.”
Don’t Overestimate AI
“With the increasing use of artificial intelligence and machine learning in other spheres, there has been an increase in interest in exploring how they can be utilized to improve clinical outcomes,” said Suman Pal, MD, assistant professor in the division of hospital medicine at the University of New Mexico, Albuquerque, in an interview. “However, concerns remain regarding the possible harms and ways to mitigate them,” said Dr. Pal, who was not involved in the current study.
In the current study, “It was interesting to note that explanations did not significantly mitigate the decrease in clinician accuracy from systematically biased AI model predictions,” Dr. Pal said.
“For the clinician, the findings of this study caution against overreliance on AI in clinical decision-making, especially because of the risk of exacerbating existing health disparities due to systemic inequities in existing literature,” Dr. Pal told this news organization.
“Additional research is needed to explore how clinicians can be better trained in identifying both the utility and the limitations of AI and into methods of validation and continuous quality checks with integration of AI into clinical workflows,” he noted.
The study was funded by the National Heart, Lung, and Blood Institute. The researchers had no financial conflicts to disclose. Dr. Pal had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Systematically biased artificial intelligence (AI) models did not improve clinicians’ accuracy in diagnosing hospitalized patients, based on data from more than 450 clinicians.
“Artificial Intelligence (AI) could support clinicians in their diagnostic decisions of hospitalized patients but could also be biased and cause potential harm,” said Sarah Jabbour, MSE, a PhD candidate in computer science and engineering at the University of Michigan, Ann Arbor, in an interview.
“Regulatory guidance has suggested that the use of AI explanations could mitigate these harms, but the effectiveness of using AI explanations has not been established,” she said.
To examine whether AI explanations can be effective in mitigating the potential harms of systemic bias in AI models, Ms. Jabbour and colleagues conducted a randomized clinical vignette survey study. The survey was administered between April 2022 and January 2023 across 13 states, and the study population included hospitalist physicians, nurse practitioners, and physician assistants. The results were published in JAMA.
Participants were randomized to AI predictions with AI explanations (226 clinicians) or without AI explanations (231 clinicians).
The primary outcome was diagnostic accuracy for pneumonia, heart failure, and chronic obstructive pulmonary disease, defined as the number of correct diagnoses over the total number of assessments, the researchers wrote.
The clinicians viewed nine clinical vignettes of patients hospitalized with acute respiratory failure, including their presenting symptoms, physical examination, laboratory results, and chest radiographs. Clinicians viewed two vignettes with no AI model input to establish baseline diagnostic accuracy. They made three assessments in each vignette, one for each diagnosis. The order of the vignettes was two without AI predictions (to establish baseline diagnostic accuracy), six with AI predictions, and one with a clinical consultation by a hypothetical colleague. The vignettes included standard and systematically biased AI models.
The baseline diagnostic accuracy was 73% for the diagnoses of pneumonia, heart failure, and chronic obstructive pulmonary disease. Clinicians’ accuracy increased by 2.9% when they viewed a standard diagnostic AI model without explanations and by 4.4% when they viewed models with AI explanations.
However, clinicians’ accuracy decreased by 11.3% after viewing systematically biased AI model predictions without explanations compared with baseline, and biased AI model predictions with explanations decreased accuracy by 9.1%.
The decrease in accuracy with systematically biased AI predictions without explanations was mainly attributable to a decrease in the participants’ diagnostic specificity, the researchers noted, but the addition of explanations did little to improve it, the researchers said.
Potentially Useful but Still Imperfect
The findings were limited by several factors including the use of a web-based survey, which differs from surveys in a clinical setting, the researchers wrote. Other limitations included the younger than average study population, and the focus on the clinicians making treatment decisions, vs other clinicians who might have a better understanding of the AI explanations.
“In our study, explanations were presented in a way that were considered to be obvious, where the AI model was completely focused on areas of the chest X-rays unrelated to the clinical condition,” Ms. Jabbour told this news organization. “We hypothesized that if presented with such explanations, the participants in our study would notice that the model was behaving incorrectly and not rely on its predictions. This was surprisingly not the case, and the explanations when presented alongside biased AI predictions had seemingly no effect in mitigating clinicians’ overreliance on biased AI,” she said.
“AI is being developed at an extraordinary rate, and our study shows that it has the potential to improve clinical decision-making. At the same time, it could harm clinical decision-making when biased,” Ms. Jabbour said. “We must be thoughtful about how to carefully integrate AI into clinical workflows, with the goal of improving clinical care while not introducing systematic errors or harming patients,” she added.
Looking ahead, “There are several potential research areas that could be explored,” said Ms. Jabbour. “Researchers should focus on careful validation of AI models to identify biased model behavior prior to deployment. AI researchers should also continue including and communicating with clinicians during the development of AI tools to better understand clinicians’ needs and how they interact with AI,” she said. “This is not an exhaustive list of research directions, and it will take much discussion between experts across disciplines such as AI, human computer interaction, and medicine to ultimately deploy AI safely into clinical care.”
Don’t Overestimate AI
“With the increasing use of artificial intelligence and machine learning in other spheres, there has been an increase in interest in exploring how they can be utilized to improve clinical outcomes,” said Suman Pal, MD, assistant professor in the division of hospital medicine at the University of New Mexico, Albuquerque, in an interview. “However, concerns remain regarding the possible harms and ways to mitigate them,” said Dr. Pal, who was not involved in the current study.
In the current study, “It was interesting to note that explanations did not significantly mitigate the decrease in clinician accuracy from systematically biased AI model predictions,” Dr. Pal said.
“For the clinician, the findings of this study caution against overreliance on AI in clinical decision-making, especially because of the risk of exacerbating existing health disparities due to systemic inequities in existing literature,” Dr. Pal told this news organization.
“Additional research is needed to explore how clinicians can be better trained in identifying both the utility and the limitations of AI and into methods of validation and continuous quality checks with integration of AI into clinical workflows,” he noted.
The study was funded by the National Heart, Lung, and Blood Institute. The researchers had no financial conflicts to disclose. Dr. Pal had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Systematically biased artificial intelligence (AI) models did not improve clinicians’ accuracy in diagnosing hospitalized patients, based on data from more than 450 clinicians.
“Artificial Intelligence (AI) could support clinicians in their diagnostic decisions of hospitalized patients but could also be biased and cause potential harm,” said Sarah Jabbour, MSE, a PhD candidate in computer science and engineering at the University of Michigan, Ann Arbor, in an interview.
“Regulatory guidance has suggested that the use of AI explanations could mitigate these harms, but the effectiveness of using AI explanations has not been established,” she said.
To examine whether AI explanations can be effective in mitigating the potential harms of systemic bias in AI models, Ms. Jabbour and colleagues conducted a randomized clinical vignette survey study. The survey was administered between April 2022 and January 2023 across 13 states, and the study population included hospitalist physicians, nurse practitioners, and physician assistants. The results were published in JAMA.
Participants were randomized to AI predictions with AI explanations (226 clinicians) or without AI explanations (231 clinicians).
The primary outcome was diagnostic accuracy for pneumonia, heart failure, and chronic obstructive pulmonary disease, defined as the number of correct diagnoses over the total number of assessments, the researchers wrote.
The clinicians viewed nine clinical vignettes of patients hospitalized with acute respiratory failure, including their presenting symptoms, physical examination, laboratory results, and chest radiographs. Clinicians viewed two vignettes with no AI model input to establish baseline diagnostic accuracy. They made three assessments in each vignette, one for each diagnosis. The order of the vignettes was two without AI predictions (to establish baseline diagnostic accuracy), six with AI predictions, and one with a clinical consultation by a hypothetical colleague. The vignettes included standard and systematically biased AI models.
The baseline diagnostic accuracy was 73% for the diagnoses of pneumonia, heart failure, and chronic obstructive pulmonary disease. Clinicians’ accuracy increased by 2.9% when they viewed a standard diagnostic AI model without explanations and by 4.4% when they viewed models with AI explanations.
However, clinicians’ accuracy decreased by 11.3% after viewing systematically biased AI model predictions without explanations compared with baseline, and biased AI model predictions with explanations decreased accuracy by 9.1%.
The decrease in accuracy with systematically biased AI predictions without explanations was mainly attributable to a decrease in the participants’ diagnostic specificity, the researchers noted, but the addition of explanations did little to improve it, the researchers said.
Potentially Useful but Still Imperfect
The findings were limited by several factors including the use of a web-based survey, which differs from surveys in a clinical setting, the researchers wrote. Other limitations included the younger than average study population, and the focus on the clinicians making treatment decisions, vs other clinicians who might have a better understanding of the AI explanations.
“In our study, explanations were presented in a way that were considered to be obvious, where the AI model was completely focused on areas of the chest X-rays unrelated to the clinical condition,” Ms. Jabbour told this news organization. “We hypothesized that if presented with such explanations, the participants in our study would notice that the model was behaving incorrectly and not rely on its predictions. This was surprisingly not the case, and the explanations when presented alongside biased AI predictions had seemingly no effect in mitigating clinicians’ overreliance on biased AI,” she said.
“AI is being developed at an extraordinary rate, and our study shows that it has the potential to improve clinical decision-making. At the same time, it could harm clinical decision-making when biased,” Ms. Jabbour said. “We must be thoughtful about how to carefully integrate AI into clinical workflows, with the goal of improving clinical care while not introducing systematic errors or harming patients,” she added.
Looking ahead, “There are several potential research areas that could be explored,” said Ms. Jabbour. “Researchers should focus on careful validation of AI models to identify biased model behavior prior to deployment. AI researchers should also continue including and communicating with clinicians during the development of AI tools to better understand clinicians’ needs and how they interact with AI,” she said. “This is not an exhaustive list of research directions, and it will take much discussion between experts across disciplines such as AI, human computer interaction, and medicine to ultimately deploy AI safely into clinical care.”
Don’t Overestimate AI
“With the increasing use of artificial intelligence and machine learning in other spheres, there has been an increase in interest in exploring how they can be utilized to improve clinical outcomes,” said Suman Pal, MD, assistant professor in the division of hospital medicine at the University of New Mexico, Albuquerque, in an interview. “However, concerns remain regarding the possible harms and ways to mitigate them,” said Dr. Pal, who was not involved in the current study.
In the current study, “It was interesting to note that explanations did not significantly mitigate the decrease in clinician accuracy from systematically biased AI model predictions,” Dr. Pal said.
“For the clinician, the findings of this study caution against overreliance on AI in clinical decision-making, especially because of the risk of exacerbating existing health disparities due to systemic inequities in existing literature,” Dr. Pal told this news organization.
“Additional research is needed to explore how clinicians can be better trained in identifying both the utility and the limitations of AI and into methods of validation and continuous quality checks with integration of AI into clinical workflows,” he noted.
The study was funded by the National Heart, Lung, and Blood Institute. The researchers had no financial conflicts to disclose. Dr. Pal had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
FROM JAMA
Should BP Guidelines Be Sex-Specific?
This transcript has been edited for clarity.
This is Dr. JoAnn Manson, professor of medicine at Harvard Medical School and Brigham and Women’s Hospital. Current BP guidelines are sex-agnostic.
This study was done in the large-scale nationally representative NHANES cohort. It included more than 53,000 US men and women. The average age was about 45 years, with an average duration of follow-up of 9.5 years. During that time, about 2400 cardiovascular (CVD) deaths were documented at baseline. The BP was measured three times, and the results were averaged. About 20% of the cohort were taking antihypertensive medications, and 80% were not.
Sex differences were observed in the association between BP and CVD mortality. The systolic BP associated with the lowest risk for CVD death was 110-119 mm Hg in men and 100-109 mm Hg in women. In men, however, compared with a reference category of systolic BP of 100-109 mm Hg, the risk for CVD death began to increase significantly at a systolic BP ≥ 160 mm Hg, at which point, the hazard ratio was 1.76, or 76% higher risk.
In women, the risk for CVD death began to increase significantly at a lower threshold. Compared with a reference category of systolic BP of 100-109 mm Hg, women whose systolic BP was 130-139 mm Hg had a significant 61% increase in CVD death, and among those with a systolic BP of 140-159 mm Hg, the risk was increased by 75%. With a systolic BP ≥ 160 mm Hg, CVD deaths among women were more than doubled, with a hazard ratio of 2.13.
Overall, these findings suggest sex differences, with women having an increased risk for CVD death beginning at a lower elevation of their systolic BP. For diastolic BP, both men and women showed the typical U-shaped curve and the diastolic BP associated with the lowest risk for CVD death was 70-80 mm Hg.
If these findings can be replicated with additional research and other large-scale cohort studies, and randomized trials show differences in lowering BP, then sex-specific BP guidelines could have advantages and should be seriously considered. Furthermore, some of the CVD risk scores and risk modeling should perhaps use sex-specific blood pressure thresholds.Dr. Manson received study pill donation and infrastructure support from Mars Symbioscience (for the COSMOS trial).
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
This is Dr. JoAnn Manson, professor of medicine at Harvard Medical School and Brigham and Women’s Hospital. Current BP guidelines are sex-agnostic.
This study was done in the large-scale nationally representative NHANES cohort. It included more than 53,000 US men and women. The average age was about 45 years, with an average duration of follow-up of 9.5 years. During that time, about 2400 cardiovascular (CVD) deaths were documented at baseline. The BP was measured three times, and the results were averaged. About 20% of the cohort were taking antihypertensive medications, and 80% were not.
Sex differences were observed in the association between BP and CVD mortality. The systolic BP associated with the lowest risk for CVD death was 110-119 mm Hg in men and 100-109 mm Hg in women. In men, however, compared with a reference category of systolic BP of 100-109 mm Hg, the risk for CVD death began to increase significantly at a systolic BP ≥ 160 mm Hg, at which point, the hazard ratio was 1.76, or 76% higher risk.
In women, the risk for CVD death began to increase significantly at a lower threshold. Compared with a reference category of systolic BP of 100-109 mm Hg, women whose systolic BP was 130-139 mm Hg had a significant 61% increase in CVD death, and among those with a systolic BP of 140-159 mm Hg, the risk was increased by 75%. With a systolic BP ≥ 160 mm Hg, CVD deaths among women were more than doubled, with a hazard ratio of 2.13.
Overall, these findings suggest sex differences, with women having an increased risk for CVD death beginning at a lower elevation of their systolic BP. For diastolic BP, both men and women showed the typical U-shaped curve and the diastolic BP associated with the lowest risk for CVD death was 70-80 mm Hg.
If these findings can be replicated with additional research and other large-scale cohort studies, and randomized trials show differences in lowering BP, then sex-specific BP guidelines could have advantages and should be seriously considered. Furthermore, some of the CVD risk scores and risk modeling should perhaps use sex-specific blood pressure thresholds.Dr. Manson received study pill donation and infrastructure support from Mars Symbioscience (for the COSMOS trial).
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
This is Dr. JoAnn Manson, professor of medicine at Harvard Medical School and Brigham and Women’s Hospital. Current BP guidelines are sex-agnostic.
This study was done in the large-scale nationally representative NHANES cohort. It included more than 53,000 US men and women. The average age was about 45 years, with an average duration of follow-up of 9.5 years. During that time, about 2400 cardiovascular (CVD) deaths were documented at baseline. The BP was measured three times, and the results were averaged. About 20% of the cohort were taking antihypertensive medications, and 80% were not.
Sex differences were observed in the association between BP and CVD mortality. The systolic BP associated with the lowest risk for CVD death was 110-119 mm Hg in men and 100-109 mm Hg in women. In men, however, compared with a reference category of systolic BP of 100-109 mm Hg, the risk for CVD death began to increase significantly at a systolic BP ≥ 160 mm Hg, at which point, the hazard ratio was 1.76, or 76% higher risk.
In women, the risk for CVD death began to increase significantly at a lower threshold. Compared with a reference category of systolic BP of 100-109 mm Hg, women whose systolic BP was 130-139 mm Hg had a significant 61% increase in CVD death, and among those with a systolic BP of 140-159 mm Hg, the risk was increased by 75%. With a systolic BP ≥ 160 mm Hg, CVD deaths among women were more than doubled, with a hazard ratio of 2.13.
Overall, these findings suggest sex differences, with women having an increased risk for CVD death beginning at a lower elevation of their systolic BP. For diastolic BP, both men and women showed the typical U-shaped curve and the diastolic BP associated with the lowest risk for CVD death was 70-80 mm Hg.
If these findings can be replicated with additional research and other large-scale cohort studies, and randomized trials show differences in lowering BP, then sex-specific BP guidelines could have advantages and should be seriously considered. Furthermore, some of the CVD risk scores and risk modeling should perhaps use sex-specific blood pressure thresholds.Dr. Manson received study pill donation and infrastructure support from Mars Symbioscience (for the COSMOS trial).
A version of this article appeared on Medscape.com.
Is It Time to Air Grievances?
‘Twas the night before Festivus and all through the house, everyone was griping.
In case you’ve only been watching Friends reruns lately, Festivus is a holiday that originated 25 years ago in the last season of Seinfeld. George’s father created it as an alternative to Christmas hype. In addition to an aluminum pole, the holiday features the annual airing of grievances, when one is encouraged to voice complaints. Aluminum poles haven’t replaced Christmas trees, but the spirit of Festivus is still with us in the widespread airing of grievances in 2023.
Complaining isn’t just a post-pandemic problem. Hector spends quite a bit of time complaining about Paris in the Iliad. That was a few pandemics ago. And repining is ubiquitous in literature — as human as walking on two limbs it seems. Ostensibly, we complain to effect change: Something is wrong and we expect it to be different. But that’s not the whole story. No one believes the weather will improve or the Patriots will play better because we complain about them. So why do we bother?
Even if nothing changes on the outside, it does seem to alter our internal state, serving a healthy psychological function. Putting to words what is aggravating can have the same benefit of deep breathing. We describe it as “getting something off our chest” because that’s what it feels like. We feel unburdened just by saying it out loud. Think about the last time you complained: Cranky staff, prior auths, Medicare, disrespectful patients, many of your colleagues will nod in agreement, validating your feelings and making you feel less isolated.
There are also maladaptive reasons for whining. It’s obviously an elementary way to get attention or to remove responsibility. It can also be a political weapon (office politics included). It’s such a potent way to connect that it’s used to build alliances and clout. “Washington is doing a great job,” said no candidate ever. No, if you want to get people on your side, find something irritating and complain to everyone how annoying it is. This solidifies “us” versus “them,” which can harm organizations and families alike.
Yet, eliminating all complaints is neither feasible, nor probably advisable. You could try to make your office a complaint-free zone, but the likely result would be to push any griping to the remote corners where you can no longer hear them. These criticisms might have uncovered missed opportunities, identify problems, and even improve cohesion if done in a safe and transparent setting. If they are left unaddressed or if the underlying culture isn’t sound, then they can propagate and lead to factions that harm productivity.
Griping is as much part of the holiday season as jingle bells and jelly donuts. I don’t believe complaining is up now because people were grumpier in 2023. Rather I think people just craved connection more than ever. So join in: Traffic after the time change, Tesla service, (super) late patients, prior auths, perioral dermatitis, post-COVID telogen effluvium.
I feel better.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on X (formerly Twitter). Write to him at dermnews@mdedge.com.
‘Twas the night before Festivus and all through the house, everyone was griping.
In case you’ve only been watching Friends reruns lately, Festivus is a holiday that originated 25 years ago in the last season of Seinfeld. George’s father created it as an alternative to Christmas hype. In addition to an aluminum pole, the holiday features the annual airing of grievances, when one is encouraged to voice complaints. Aluminum poles haven’t replaced Christmas trees, but the spirit of Festivus is still with us in the widespread airing of grievances in 2023.
Complaining isn’t just a post-pandemic problem. Hector spends quite a bit of time complaining about Paris in the Iliad. That was a few pandemics ago. And repining is ubiquitous in literature — as human as walking on two limbs it seems. Ostensibly, we complain to effect change: Something is wrong and we expect it to be different. But that’s not the whole story. No one believes the weather will improve or the Patriots will play better because we complain about them. So why do we bother?
Even if nothing changes on the outside, it does seem to alter our internal state, serving a healthy psychological function. Putting to words what is aggravating can have the same benefit of deep breathing. We describe it as “getting something off our chest” because that’s what it feels like. We feel unburdened just by saying it out loud. Think about the last time you complained: Cranky staff, prior auths, Medicare, disrespectful patients, many of your colleagues will nod in agreement, validating your feelings and making you feel less isolated.
There are also maladaptive reasons for whining. It’s obviously an elementary way to get attention or to remove responsibility. It can also be a political weapon (office politics included). It’s such a potent way to connect that it’s used to build alliances and clout. “Washington is doing a great job,” said no candidate ever. No, if you want to get people on your side, find something irritating and complain to everyone how annoying it is. This solidifies “us” versus “them,” which can harm organizations and families alike.
Yet, eliminating all complaints is neither feasible, nor probably advisable. You could try to make your office a complaint-free zone, but the likely result would be to push any griping to the remote corners where you can no longer hear them. These criticisms might have uncovered missed opportunities, identify problems, and even improve cohesion if done in a safe and transparent setting. If they are left unaddressed or if the underlying culture isn’t sound, then they can propagate and lead to factions that harm productivity.
Griping is as much part of the holiday season as jingle bells and jelly donuts. I don’t believe complaining is up now because people were grumpier in 2023. Rather I think people just craved connection more than ever. So join in: Traffic after the time change, Tesla service, (super) late patients, prior auths, perioral dermatitis, post-COVID telogen effluvium.
I feel better.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on X (formerly Twitter). Write to him at dermnews@mdedge.com.
‘Twas the night before Festivus and all through the house, everyone was griping.
In case you’ve only been watching Friends reruns lately, Festivus is a holiday that originated 25 years ago in the last season of Seinfeld. George’s father created it as an alternative to Christmas hype. In addition to an aluminum pole, the holiday features the annual airing of grievances, when one is encouraged to voice complaints. Aluminum poles haven’t replaced Christmas trees, but the spirit of Festivus is still with us in the widespread airing of grievances in 2023.
Complaining isn’t just a post-pandemic problem. Hector spends quite a bit of time complaining about Paris in the Iliad. That was a few pandemics ago. And repining is ubiquitous in literature — as human as walking on two limbs it seems. Ostensibly, we complain to effect change: Something is wrong and we expect it to be different. But that’s not the whole story. No one believes the weather will improve or the Patriots will play better because we complain about them. So why do we bother?
Even if nothing changes on the outside, it does seem to alter our internal state, serving a healthy psychological function. Putting to words what is aggravating can have the same benefit of deep breathing. We describe it as “getting something off our chest” because that’s what it feels like. We feel unburdened just by saying it out loud. Think about the last time you complained: Cranky staff, prior auths, Medicare, disrespectful patients, many of your colleagues will nod in agreement, validating your feelings and making you feel less isolated.
There are also maladaptive reasons for whining. It’s obviously an elementary way to get attention or to remove responsibility. It can also be a political weapon (office politics included). It’s such a potent way to connect that it’s used to build alliances and clout. “Washington is doing a great job,” said no candidate ever. No, if you want to get people on your side, find something irritating and complain to everyone how annoying it is. This solidifies “us” versus “them,” which can harm organizations and families alike.
Yet, eliminating all complaints is neither feasible, nor probably advisable. You could try to make your office a complaint-free zone, but the likely result would be to push any griping to the remote corners where you can no longer hear them. These criticisms might have uncovered missed opportunities, identify problems, and even improve cohesion if done in a safe and transparent setting. If they are left unaddressed or if the underlying culture isn’t sound, then they can propagate and lead to factions that harm productivity.
Griping is as much part of the holiday season as jingle bells and jelly donuts. I don’t believe complaining is up now because people were grumpier in 2023. Rather I think people just craved connection more than ever. So join in: Traffic after the time change, Tesla service, (super) late patients, prior auths, perioral dermatitis, post-COVID telogen effluvium.
I feel better.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on X (formerly Twitter). Write to him at dermnews@mdedge.com.
Where Is the ‘Microbiome Revolution’ Headed Next?
Human microbiome research has progressed in leaps and bounds over the past decades, from pivotal studies begun in the 1970s to the launch of the Human Microbiome Project in 2007. Breakthroughs have laid the groundwork for more recent clinical applications, such as fecal microbiota transplantation (FMT), and advanced techniques to explore new therapeutic pathways. Yet the “microbiome revolution” is just getting started, according to professor Martin J. Blaser, MD, one of the field’s pioneers.
, says Dr. Blaser, who holds the Henry Rutgers Chair of the Human Microbiome and is director of the Center for Advanced Biotechnology and Medicine at Rutgers University in New Brunswick, New Jersey.
Dr. Blaser is the author of Missing Microbes: How the Overuse of Antibiotics Is Fueling Our Modern Plagues, serves as chair of the Presidential Advisory Council on Combating Antibiotic-Resistant Bacteria and is a member of the scientific advisory board of the biotech startup Micronoma.
In this interview, which has been condensed and edited for clarity, Dr. Blaser discusses where we’re at now and where he sees the microbiome field evolving in the coming years.
Highlighting the Most Promising Applications
Which recent studies on the link between the human microbiome and disease have you found particularly promising?
There have been a number of studies, including our own, focusing on the gut-kidney axis. The gut microbiome produces, or detoxifies, metabolites that are toxic to the kidney: for example, those involved in the formation of kidney stones and in the worsening of uremia.
Altering the microbiome to reduce the uremic toxins and the nidus for stone formation is a very promising field of research.
What other disease states may be amenable to microbiome-based interventions?
There are diseases that are caused by known genetic mutations. Yet, for nearly all of them, there is great variation in clinical outcomes, which might be classed as genes multiplied by environment interactions.
It seems likely to me that microbiome variation could account for some proportion of those differences for some genetic diseases.
It’s now well established that altering the microbiome with FMT is a successful intervention for recurrent Clostridioides difficile infections. What do you see as the next disease states where FMT could prove successful?
If you go to ClinicalTrials.gov, you will find that that there are 471 trials registered using FMT. This is across a broad range of illnesses, including metabolic, immunological, autoimmune, inflammatory, degenerative, and neoplastic diseases.
Which will be the next condition showing marked efficacy is anyone’s guess. That is why we must do clinical trials to assess what works and what does not, regardless of specific illness.
The donor’s microbiome appears to be vital to engraftment success, with “superdonors” even being identified. What factors do you think primarily influence microbiome engraftment?
There is an emerging science about this question, driven in part by classical ecological theory.
Right now, we are using FMT as if one size fits all. But this probably would not provide optimal treatment for all. Just as we type blood donors and recipients before the blood transfusion, one could easily imagine a parallel kind of procedure.
Are there any diseases where it’s just too far-fetched to think altering the microbiome could make a difference?
The link between the microbiome and human health is so pervasive that there are few conditions that are out of the realm of possibility. It really is a frontier.
Not that the microbiome causes everything, but by understanding and manipulating the microbiome, we could at least palliate, or slow down, particular pathologic processes.
For all the major causes of death in the United States — cardiovascular disease, cancer, dementia and neurogenerative diseases, diabetes, and lung, liver, and kidney diseases — there is ongoing investigation of the microbiome. A greater promise would be to prevent or cure these illnesses.
Predicting the Next Stages of the ‘Microbiome Revolution’
Do you believe we are at a turning point with the microbiome in terms of being able to manipulate or engineer it?
The microbiome is a scientific frontier that has an impact across the biosphere. It is a broad frontier involving human and veterinary medicine, agriculture, and the environment. Knowledge is increasing incrementally, as expected.
Are we at the point yet where doctors should be incorporating microbiome-related lifestyle changes for people with or at risk for cancer, heart disease, Alzheimer’s disease, or other chronic conditions?
Although we are still in the early stages of the “microbiome revolution,” which I first wrote about in EMBO Reports in 2006 and then again in the Journal of Clinical Investigation in 2014, I think important advances for all of these conditions are coming our way in the next 5-10 years.
How are prebiotics, probiotics, and postbiotics being used to shape the microbiome?
This is a very important and active area in clinical investigation, which needs to be ramped up.
Tens of millions of people are using probiotics and prebiotics every day for vague indications, and which have only infrequently been tested in robust clinical trials. So, there is a disconnect between what’s being claimed with the bulk of the probiotics at present and what we’ll actually know in the future.
How do you think the microbiome will stack up to other factors influencing health, such as genetics, exercise, and nutrition?
All are important, but unlike genetics, the microbiome is tractable, like diet and exercise.
It is essentially impossible to change one’s genome, but that might become more likely before too long. However, we can easily change someone’s microbiome through dietary means, for example. Once we know the ground rules, there will be many options. Right now, it is mostly one-offs, but as the scientific basis broadens, much more will be possible.
In the future, do you think we’ll be able to look at a person’s microbiome and tell what his or her risk of developing disease is, similar to the way we use gene panels now?
Yes, but we will need scientific advances to teach us what are the important biomarkers in general and in particular people. This will be one area of precision medicine.
Lessons From Decades at the Forefront
You’ve been involved in this research for over 30 years, and the majority has focused on the human microbiome and its role in disease. When did it become apparent to you that this research had unique therapeutic promise?
From the very start, there was always the potential to harness the microbiome to improve human health. In fact, I wrote a perspective in PNAS on that theme in 2010.
The key is to understand the biology of the microbiome, and from the scientific study comes new preventives and new treatments. Right now, there are many “probiotic” products on the market. Probiotics have a great future, but most of what is out there has not been rigorously tested for effectiveness.
Was there a particular series of studies that occurred before the launch of the Human Microbiome Project and brought us to the current era?
The studies in the 1970s-1980s by Carl Woese using 16S rRNA genes to understand phylogeny and evolution opened up the field of DNA sequencing to consider bacterial evolution and issues of ancestry.
A key subject of your research and the focus of your book is antibiotic-resistant bacteria. What did this work teach you about describing the science of antibiotic resistance to the general public?
People don’t care very much about antibiotic resistance. They think that affects other people, mostly. In contrast, they care about their own health and their children’s health.
The more that the data show that using antibiotics can be harmful to health in some circumstances, the more that use will diminish. We need more transparency about benefits and costs.
Are there any common misconceptions about the microbiome that you hear from the general public, or even clinicians, that you would like to see greater efforts to dispel?
The public and the medical profession are in love with probiotics, buying them by the tens of millions. But as stated before, they are very diverse and mostly untested for efficacy.
The next step is to test specific formulations to see which ones work, and for whom, and which ones don’t. That would be a big advance.
A version of this article appeared on Medscape.com.
Human microbiome research has progressed in leaps and bounds over the past decades, from pivotal studies begun in the 1970s to the launch of the Human Microbiome Project in 2007. Breakthroughs have laid the groundwork for more recent clinical applications, such as fecal microbiota transplantation (FMT), and advanced techniques to explore new therapeutic pathways. Yet the “microbiome revolution” is just getting started, according to professor Martin J. Blaser, MD, one of the field’s pioneers.
, says Dr. Blaser, who holds the Henry Rutgers Chair of the Human Microbiome and is director of the Center for Advanced Biotechnology and Medicine at Rutgers University in New Brunswick, New Jersey.
Dr. Blaser is the author of Missing Microbes: How the Overuse of Antibiotics Is Fueling Our Modern Plagues, serves as chair of the Presidential Advisory Council on Combating Antibiotic-Resistant Bacteria and is a member of the scientific advisory board of the biotech startup Micronoma.
In this interview, which has been condensed and edited for clarity, Dr. Blaser discusses where we’re at now and where he sees the microbiome field evolving in the coming years.
Highlighting the Most Promising Applications
Which recent studies on the link between the human microbiome and disease have you found particularly promising?
There have been a number of studies, including our own, focusing on the gut-kidney axis. The gut microbiome produces, or detoxifies, metabolites that are toxic to the kidney: for example, those involved in the formation of kidney stones and in the worsening of uremia.
Altering the microbiome to reduce the uremic toxins and the nidus for stone formation is a very promising field of research.
What other disease states may be amenable to microbiome-based interventions?
There are diseases that are caused by known genetic mutations. Yet, for nearly all of them, there is great variation in clinical outcomes, which might be classed as genes multiplied by environment interactions.
It seems likely to me that microbiome variation could account for some proportion of those differences for some genetic diseases.
It’s now well established that altering the microbiome with FMT is a successful intervention for recurrent Clostridioides difficile infections. What do you see as the next disease states where FMT could prove successful?
If you go to ClinicalTrials.gov, you will find that that there are 471 trials registered using FMT. This is across a broad range of illnesses, including metabolic, immunological, autoimmune, inflammatory, degenerative, and neoplastic diseases.
Which will be the next condition showing marked efficacy is anyone’s guess. That is why we must do clinical trials to assess what works and what does not, regardless of specific illness.
The donor’s microbiome appears to be vital to engraftment success, with “superdonors” even being identified. What factors do you think primarily influence microbiome engraftment?
There is an emerging science about this question, driven in part by classical ecological theory.
Right now, we are using FMT as if one size fits all. But this probably would not provide optimal treatment for all. Just as we type blood donors and recipients before the blood transfusion, one could easily imagine a parallel kind of procedure.
Are there any diseases where it’s just too far-fetched to think altering the microbiome could make a difference?
The link between the microbiome and human health is so pervasive that there are few conditions that are out of the realm of possibility. It really is a frontier.
Not that the microbiome causes everything, but by understanding and manipulating the microbiome, we could at least palliate, or slow down, particular pathologic processes.
For all the major causes of death in the United States — cardiovascular disease, cancer, dementia and neurogenerative diseases, diabetes, and lung, liver, and kidney diseases — there is ongoing investigation of the microbiome. A greater promise would be to prevent or cure these illnesses.
Predicting the Next Stages of the ‘Microbiome Revolution’
Do you believe we are at a turning point with the microbiome in terms of being able to manipulate or engineer it?
The microbiome is a scientific frontier that has an impact across the biosphere. It is a broad frontier involving human and veterinary medicine, agriculture, and the environment. Knowledge is increasing incrementally, as expected.
Are we at the point yet where doctors should be incorporating microbiome-related lifestyle changes for people with or at risk for cancer, heart disease, Alzheimer’s disease, or other chronic conditions?
Although we are still in the early stages of the “microbiome revolution,” which I first wrote about in EMBO Reports in 2006 and then again in the Journal of Clinical Investigation in 2014, I think important advances for all of these conditions are coming our way in the next 5-10 years.
How are prebiotics, probiotics, and postbiotics being used to shape the microbiome?
This is a very important and active area in clinical investigation, which needs to be ramped up.
Tens of millions of people are using probiotics and prebiotics every day for vague indications, and which have only infrequently been tested in robust clinical trials. So, there is a disconnect between what’s being claimed with the bulk of the probiotics at present and what we’ll actually know in the future.
How do you think the microbiome will stack up to other factors influencing health, such as genetics, exercise, and nutrition?
All are important, but unlike genetics, the microbiome is tractable, like diet and exercise.
It is essentially impossible to change one’s genome, but that might become more likely before too long. However, we can easily change someone’s microbiome through dietary means, for example. Once we know the ground rules, there will be many options. Right now, it is mostly one-offs, but as the scientific basis broadens, much more will be possible.
In the future, do you think we’ll be able to look at a person’s microbiome and tell what his or her risk of developing disease is, similar to the way we use gene panels now?
Yes, but we will need scientific advances to teach us what are the important biomarkers in general and in particular people. This will be one area of precision medicine.
Lessons From Decades at the Forefront
You’ve been involved in this research for over 30 years, and the majority has focused on the human microbiome and its role in disease. When did it become apparent to you that this research had unique therapeutic promise?
From the very start, there was always the potential to harness the microbiome to improve human health. In fact, I wrote a perspective in PNAS on that theme in 2010.
The key is to understand the biology of the microbiome, and from the scientific study comes new preventives and new treatments. Right now, there are many “probiotic” products on the market. Probiotics have a great future, but most of what is out there has not been rigorously tested for effectiveness.
Was there a particular series of studies that occurred before the launch of the Human Microbiome Project and brought us to the current era?
The studies in the 1970s-1980s by Carl Woese using 16S rRNA genes to understand phylogeny and evolution opened up the field of DNA sequencing to consider bacterial evolution and issues of ancestry.
A key subject of your research and the focus of your book is antibiotic-resistant bacteria. What did this work teach you about describing the science of antibiotic resistance to the general public?
People don’t care very much about antibiotic resistance. They think that affects other people, mostly. In contrast, they care about their own health and their children’s health.
The more that the data show that using antibiotics can be harmful to health in some circumstances, the more that use will diminish. We need more transparency about benefits and costs.
Are there any common misconceptions about the microbiome that you hear from the general public, or even clinicians, that you would like to see greater efforts to dispel?
The public and the medical profession are in love with probiotics, buying them by the tens of millions. But as stated before, they are very diverse and mostly untested for efficacy.
The next step is to test specific formulations to see which ones work, and for whom, and which ones don’t. That would be a big advance.
A version of this article appeared on Medscape.com.
Human microbiome research has progressed in leaps and bounds over the past decades, from pivotal studies begun in the 1970s to the launch of the Human Microbiome Project in 2007. Breakthroughs have laid the groundwork for more recent clinical applications, such as fecal microbiota transplantation (FMT), and advanced techniques to explore new therapeutic pathways. Yet the “microbiome revolution” is just getting started, according to professor Martin J. Blaser, MD, one of the field’s pioneers.
, says Dr. Blaser, who holds the Henry Rutgers Chair of the Human Microbiome and is director of the Center for Advanced Biotechnology and Medicine at Rutgers University in New Brunswick, New Jersey.
Dr. Blaser is the author of Missing Microbes: How the Overuse of Antibiotics Is Fueling Our Modern Plagues, serves as chair of the Presidential Advisory Council on Combating Antibiotic-Resistant Bacteria and is a member of the scientific advisory board of the biotech startup Micronoma.
In this interview, which has been condensed and edited for clarity, Dr. Blaser discusses where we’re at now and where he sees the microbiome field evolving in the coming years.
Highlighting the Most Promising Applications
Which recent studies on the link between the human microbiome and disease have you found particularly promising?
There have been a number of studies, including our own, focusing on the gut-kidney axis. The gut microbiome produces, or detoxifies, metabolites that are toxic to the kidney: for example, those involved in the formation of kidney stones and in the worsening of uremia.
Altering the microbiome to reduce the uremic toxins and the nidus for stone formation is a very promising field of research.
What other disease states may be amenable to microbiome-based interventions?
There are diseases that are caused by known genetic mutations. Yet, for nearly all of them, there is great variation in clinical outcomes, which might be classed as genes multiplied by environment interactions.
It seems likely to me that microbiome variation could account for some proportion of those differences for some genetic diseases.
It’s now well established that altering the microbiome with FMT is a successful intervention for recurrent Clostridioides difficile infections. What do you see as the next disease states where FMT could prove successful?
If you go to ClinicalTrials.gov, you will find that that there are 471 trials registered using FMT. This is across a broad range of illnesses, including metabolic, immunological, autoimmune, inflammatory, degenerative, and neoplastic diseases.
Which will be the next condition showing marked efficacy is anyone’s guess. That is why we must do clinical trials to assess what works and what does not, regardless of specific illness.
The donor’s microbiome appears to be vital to engraftment success, with “superdonors” even being identified. What factors do you think primarily influence microbiome engraftment?
There is an emerging science about this question, driven in part by classical ecological theory.
Right now, we are using FMT as if one size fits all. But this probably would not provide optimal treatment for all. Just as we type blood donors and recipients before the blood transfusion, one could easily imagine a parallel kind of procedure.
Are there any diseases where it’s just too far-fetched to think altering the microbiome could make a difference?
The link between the microbiome and human health is so pervasive that there are few conditions that are out of the realm of possibility. It really is a frontier.
Not that the microbiome causes everything, but by understanding and manipulating the microbiome, we could at least palliate, or slow down, particular pathologic processes.
For all the major causes of death in the United States — cardiovascular disease, cancer, dementia and neurogenerative diseases, diabetes, and lung, liver, and kidney diseases — there is ongoing investigation of the microbiome. A greater promise would be to prevent or cure these illnesses.
Predicting the Next Stages of the ‘Microbiome Revolution’
Do you believe we are at a turning point with the microbiome in terms of being able to manipulate or engineer it?
The microbiome is a scientific frontier that has an impact across the biosphere. It is a broad frontier involving human and veterinary medicine, agriculture, and the environment. Knowledge is increasing incrementally, as expected.
Are we at the point yet where doctors should be incorporating microbiome-related lifestyle changes for people with or at risk for cancer, heart disease, Alzheimer’s disease, or other chronic conditions?
Although we are still in the early stages of the “microbiome revolution,” which I first wrote about in EMBO Reports in 2006 and then again in the Journal of Clinical Investigation in 2014, I think important advances for all of these conditions are coming our way in the next 5-10 years.
How are prebiotics, probiotics, and postbiotics being used to shape the microbiome?
This is a very important and active area in clinical investigation, which needs to be ramped up.
Tens of millions of people are using probiotics and prebiotics every day for vague indications, and which have only infrequently been tested in robust clinical trials. So, there is a disconnect between what’s being claimed with the bulk of the probiotics at present and what we’ll actually know in the future.
How do you think the microbiome will stack up to other factors influencing health, such as genetics, exercise, and nutrition?
All are important, but unlike genetics, the microbiome is tractable, like diet and exercise.
It is essentially impossible to change one’s genome, but that might become more likely before too long. However, we can easily change someone’s microbiome through dietary means, for example. Once we know the ground rules, there will be many options. Right now, it is mostly one-offs, but as the scientific basis broadens, much more will be possible.
In the future, do you think we’ll be able to look at a person’s microbiome and tell what his or her risk of developing disease is, similar to the way we use gene panels now?
Yes, but we will need scientific advances to teach us what are the important biomarkers in general and in particular people. This will be one area of precision medicine.
Lessons From Decades at the Forefront
You’ve been involved in this research for over 30 years, and the majority has focused on the human microbiome and its role in disease. When did it become apparent to you that this research had unique therapeutic promise?
From the very start, there was always the potential to harness the microbiome to improve human health. In fact, I wrote a perspective in PNAS on that theme in 2010.
The key is to understand the biology of the microbiome, and from the scientific study comes new preventives and new treatments. Right now, there are many “probiotic” products on the market. Probiotics have a great future, but most of what is out there has not been rigorously tested for effectiveness.
Was there a particular series of studies that occurred before the launch of the Human Microbiome Project and brought us to the current era?
The studies in the 1970s-1980s by Carl Woese using 16S rRNA genes to understand phylogeny and evolution opened up the field of DNA sequencing to consider bacterial evolution and issues of ancestry.
A key subject of your research and the focus of your book is antibiotic-resistant bacteria. What did this work teach you about describing the science of antibiotic resistance to the general public?
People don’t care very much about antibiotic resistance. They think that affects other people, mostly. In contrast, they care about their own health and their children’s health.
The more that the data show that using antibiotics can be harmful to health in some circumstances, the more that use will diminish. We need more transparency about benefits and costs.
Are there any common misconceptions about the microbiome that you hear from the general public, or even clinicians, that you would like to see greater efforts to dispel?
The public and the medical profession are in love with probiotics, buying them by the tens of millions. But as stated before, they are very diverse and mostly untested for efficacy.
The next step is to test specific formulations to see which ones work, and for whom, and which ones don’t. That would be a big advance.
A version of this article appeared on Medscape.com.
Thiazide Diuretics May Promote Hyponatremia
Adults who used thiazide diuretics for hypertension were more likely than were those who used nonthiazide agents to develop hyponatremia within 2 years of starting treatment, a new study of more than 180,000 people has found.
Although thiazide diuretics generally are well-tolerated in the routine treatment of uncomplicated hypertension, severe adverse effects are possible, and their frequency has not been examined, according to Niklas Worm Andersson, MD, of Statens Serum Institut, in Copenhagen, Denmark, and his colleagues.
“Thiazide diuretics are commonly used drugs for the treatment of uncomplicated hypertension, and hyponatremia is a known potential side effect to thiazide treatment, but the frequency of this adverse event is inconsistently reported across drug labels,” Dr. Andersson told this news organization.
Product labels for thiazide diuretics list hyponatremia as a potential adverse event that can occur rarely (defined as a range from less than 1 in 10,000 to less than 1 in 100 individuals), but the extent of the burden is unclear given the wide range of symptoms of the condition, the researchers write.
In a study published in Annals of Internal Medicine, Dr. Andersson and his colleagues reviewed data from population-based registries in Denmark of adults aged 40 years or older with uncomplicated hypertension, no recent prescriptions for antihypertensives, and no previous history of hyponatremia. They emulated two target trials. One trial compared the incidence of hyponatremia in new users of bendroflumethiazide (BFZ) vs a calcium-channel blocker (CCB). The other emulation compared the incidence of hyponatremia in new users of hydrochlorothiazide (HCTZ) plus a renin-angiotensin system (RAS) inhibitor vs a RAS inhibitor without HCTZ.
The primary outcome was hyponatremia, defined as blood sodium < 130 mmol/L, within 2 years of starting treatment.
The 2-year incidence of hyponatremia for the two thiazide diuretics was 3.83% for BFZ and 3.51% for HCTZ-RAS inhibitor. The risk difference in the incidence of hyponatremia was 1.35% for BFZ vs CCB and 1.38% for HCTZ-RAS inhibitor vs RAS inhibitor, the researchers reported.
The study population included 37,786 new users of BFZ who were compared with 44,963 new users of CCBs as well as 11,943 new users of HCTZ-RAS inhibitors who were compared with 85,784 new users of RAS inhibitors only.
Overall, older age and a greater number of comorbidities increased the cumulative hyponatremia in new users of thiazide-based hypertensives. The risk differences among individuals aged 80 years or older were 4.80% in the BFZ vs CCB study and 5.52% in the HCTZ-RAS inhibitor vs RAS inhibitor study. Among participants with three or more comorbidities, the risk differences in the two studies were 5.24% and 2.91%, respectively, Dr. Andersson’s group found.
The findings were limited by several factors, mainly the potential for confounding on the basis of the assumption that filled prescriptions equaled drug use, the researchers noted. Other limitations included the focus on new users and a Danish population only, which might limit generalizability, and a lack of data on blood pressure measures.
However, the results suggest a greater risk for hyponatremia with thiazide diuretics than what the drug labels indicate, especially early in treatment, the researchers concluded.
Data Reinforce Need for Vigilance in the Clinic
“Our findings highlight the continued need for clinical awareness and monitoring of this adverse drug reaction; particularly during the first months of treatment, in persons who are older or who have comorbidities,” Dr. Andersson told this news organization. “Further mapping of potential subpopulations at risk in terms of specific comorbidities is important to improve the prevention of this adverse event.”
“The thiazide diuretics have been recommended as first-line therapy for hypertension, and it was important to evaluate the potential development of hyponatremia, especially in the older patients given the potentially serious health effects caused by hyponatremia,” said Noel Deep, MD, a general internist in private practice in Antigo, Wisconsin. Dr. Deep, who was not involved in the study, also serves as chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo.
The current study findings were not surprising, Dr. Deep added. “I have seen this occur in my patients, especially in the older female patients,” he said. “The results reinforce my practice of monitoring the electrolytes and renal function in 1-2 weeks after starting a thiazide diuretic, and then at regular intervals.”
In practice, clinicians should be aware of the potential development of hyponatremia and monitor and address the electrolyte abnormalities, Dr. Deep said. “While thiazide and thiazide-like diuretics are an important component of our treatment options for patients with hypertension and other conditions, we should also ensure that we are cognizant of and address the potential side effects or electrolyte imbalances caused by the medications.”
The study was funded by the Independent Research Fund Denmark, Helsefonden, Dagmar Marshalls Fond, Gangstedfonden, A.P. Møller and Chastine Mc-Kinney Møller Foundation, Brødrene Hartmanns Fond, and Snedkermester Sophus Jacobsen og hustru Astrid Jacobsens Fond.
The researchers had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Adults who used thiazide diuretics for hypertension were more likely than were those who used nonthiazide agents to develop hyponatremia within 2 years of starting treatment, a new study of more than 180,000 people has found.
Although thiazide diuretics generally are well-tolerated in the routine treatment of uncomplicated hypertension, severe adverse effects are possible, and their frequency has not been examined, according to Niklas Worm Andersson, MD, of Statens Serum Institut, in Copenhagen, Denmark, and his colleagues.
“Thiazide diuretics are commonly used drugs for the treatment of uncomplicated hypertension, and hyponatremia is a known potential side effect to thiazide treatment, but the frequency of this adverse event is inconsistently reported across drug labels,” Dr. Andersson told this news organization.
Product labels for thiazide diuretics list hyponatremia as a potential adverse event that can occur rarely (defined as a range from less than 1 in 10,000 to less than 1 in 100 individuals), but the extent of the burden is unclear given the wide range of symptoms of the condition, the researchers write.
In a study published in Annals of Internal Medicine, Dr. Andersson and his colleagues reviewed data from population-based registries in Denmark of adults aged 40 years or older with uncomplicated hypertension, no recent prescriptions for antihypertensives, and no previous history of hyponatremia. They emulated two target trials. One trial compared the incidence of hyponatremia in new users of bendroflumethiazide (BFZ) vs a calcium-channel blocker (CCB). The other emulation compared the incidence of hyponatremia in new users of hydrochlorothiazide (HCTZ) plus a renin-angiotensin system (RAS) inhibitor vs a RAS inhibitor without HCTZ.
The primary outcome was hyponatremia, defined as blood sodium < 130 mmol/L, within 2 years of starting treatment.
The 2-year incidence of hyponatremia for the two thiazide diuretics was 3.83% for BFZ and 3.51% for HCTZ-RAS inhibitor. The risk difference in the incidence of hyponatremia was 1.35% for BFZ vs CCB and 1.38% for HCTZ-RAS inhibitor vs RAS inhibitor, the researchers reported.
The study population included 37,786 new users of BFZ who were compared with 44,963 new users of CCBs as well as 11,943 new users of HCTZ-RAS inhibitors who were compared with 85,784 new users of RAS inhibitors only.
Overall, older age and a greater number of comorbidities increased the cumulative hyponatremia in new users of thiazide-based hypertensives. The risk differences among individuals aged 80 years or older were 4.80% in the BFZ vs CCB study and 5.52% in the HCTZ-RAS inhibitor vs RAS inhibitor study. Among participants with three or more comorbidities, the risk differences in the two studies were 5.24% and 2.91%, respectively, Dr. Andersson’s group found.
The findings were limited by several factors, mainly the potential for confounding on the basis of the assumption that filled prescriptions equaled drug use, the researchers noted. Other limitations included the focus on new users and a Danish population only, which might limit generalizability, and a lack of data on blood pressure measures.
However, the results suggest a greater risk for hyponatremia with thiazide diuretics than what the drug labels indicate, especially early in treatment, the researchers concluded.
Data Reinforce Need for Vigilance in the Clinic
“Our findings highlight the continued need for clinical awareness and monitoring of this adverse drug reaction; particularly during the first months of treatment, in persons who are older or who have comorbidities,” Dr. Andersson told this news organization. “Further mapping of potential subpopulations at risk in terms of specific comorbidities is important to improve the prevention of this adverse event.”
“The thiazide diuretics have been recommended as first-line therapy for hypertension, and it was important to evaluate the potential development of hyponatremia, especially in the older patients given the potentially serious health effects caused by hyponatremia,” said Noel Deep, MD, a general internist in private practice in Antigo, Wisconsin. Dr. Deep, who was not involved in the study, also serves as chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo.
The current study findings were not surprising, Dr. Deep added. “I have seen this occur in my patients, especially in the older female patients,” he said. “The results reinforce my practice of monitoring the electrolytes and renal function in 1-2 weeks after starting a thiazide diuretic, and then at regular intervals.”
In practice, clinicians should be aware of the potential development of hyponatremia and monitor and address the electrolyte abnormalities, Dr. Deep said. “While thiazide and thiazide-like diuretics are an important component of our treatment options for patients with hypertension and other conditions, we should also ensure that we are cognizant of and address the potential side effects or electrolyte imbalances caused by the medications.”
The study was funded by the Independent Research Fund Denmark, Helsefonden, Dagmar Marshalls Fond, Gangstedfonden, A.P. Møller and Chastine Mc-Kinney Møller Foundation, Brødrene Hartmanns Fond, and Snedkermester Sophus Jacobsen og hustru Astrid Jacobsens Fond.
The researchers had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Adults who used thiazide diuretics for hypertension were more likely than were those who used nonthiazide agents to develop hyponatremia within 2 years of starting treatment, a new study of more than 180,000 people has found.
Although thiazide diuretics generally are well-tolerated in the routine treatment of uncomplicated hypertension, severe adverse effects are possible, and their frequency has not been examined, according to Niklas Worm Andersson, MD, of Statens Serum Institut, in Copenhagen, Denmark, and his colleagues.
“Thiazide diuretics are commonly used drugs for the treatment of uncomplicated hypertension, and hyponatremia is a known potential side effect to thiazide treatment, but the frequency of this adverse event is inconsistently reported across drug labels,” Dr. Andersson told this news organization.
Product labels for thiazide diuretics list hyponatremia as a potential adverse event that can occur rarely (defined as a range from less than 1 in 10,000 to less than 1 in 100 individuals), but the extent of the burden is unclear given the wide range of symptoms of the condition, the researchers write.
In a study published in Annals of Internal Medicine, Dr. Andersson and his colleagues reviewed data from population-based registries in Denmark of adults aged 40 years or older with uncomplicated hypertension, no recent prescriptions for antihypertensives, and no previous history of hyponatremia. They emulated two target trials. One trial compared the incidence of hyponatremia in new users of bendroflumethiazide (BFZ) vs a calcium-channel blocker (CCB). The other emulation compared the incidence of hyponatremia in new users of hydrochlorothiazide (HCTZ) plus a renin-angiotensin system (RAS) inhibitor vs a RAS inhibitor without HCTZ.
The primary outcome was hyponatremia, defined as blood sodium < 130 mmol/L, within 2 years of starting treatment.
The 2-year incidence of hyponatremia for the two thiazide diuretics was 3.83% for BFZ and 3.51% for HCTZ-RAS inhibitor. The risk difference in the incidence of hyponatremia was 1.35% for BFZ vs CCB and 1.38% for HCTZ-RAS inhibitor vs RAS inhibitor, the researchers reported.
The study population included 37,786 new users of BFZ who were compared with 44,963 new users of CCBs as well as 11,943 new users of HCTZ-RAS inhibitors who were compared with 85,784 new users of RAS inhibitors only.
Overall, older age and a greater number of comorbidities increased the cumulative hyponatremia in new users of thiazide-based hypertensives. The risk differences among individuals aged 80 years or older were 4.80% in the BFZ vs CCB study and 5.52% in the HCTZ-RAS inhibitor vs RAS inhibitor study. Among participants with three or more comorbidities, the risk differences in the two studies were 5.24% and 2.91%, respectively, Dr. Andersson’s group found.
The findings were limited by several factors, mainly the potential for confounding on the basis of the assumption that filled prescriptions equaled drug use, the researchers noted. Other limitations included the focus on new users and a Danish population only, which might limit generalizability, and a lack of data on blood pressure measures.
However, the results suggest a greater risk for hyponatremia with thiazide diuretics than what the drug labels indicate, especially early in treatment, the researchers concluded.
Data Reinforce Need for Vigilance in the Clinic
“Our findings highlight the continued need for clinical awareness and monitoring of this adverse drug reaction; particularly during the first months of treatment, in persons who are older or who have comorbidities,” Dr. Andersson told this news organization. “Further mapping of potential subpopulations at risk in terms of specific comorbidities is important to improve the prevention of this adverse event.”
“The thiazide diuretics have been recommended as first-line therapy for hypertension, and it was important to evaluate the potential development of hyponatremia, especially in the older patients given the potentially serious health effects caused by hyponatremia,” said Noel Deep, MD, a general internist in private practice in Antigo, Wisconsin. Dr. Deep, who was not involved in the study, also serves as chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo.
The current study findings were not surprising, Dr. Deep added. “I have seen this occur in my patients, especially in the older female patients,” he said. “The results reinforce my practice of monitoring the electrolytes and renal function in 1-2 weeks after starting a thiazide diuretic, and then at regular intervals.”
In practice, clinicians should be aware of the potential development of hyponatremia and monitor and address the electrolyte abnormalities, Dr. Deep said. “While thiazide and thiazide-like diuretics are an important component of our treatment options for patients with hypertension and other conditions, we should also ensure that we are cognizant of and address the potential side effects or electrolyte imbalances caused by the medications.”
The study was funded by the Independent Research Fund Denmark, Helsefonden, Dagmar Marshalls Fond, Gangstedfonden, A.P. Møller and Chastine Mc-Kinney Møller Foundation, Brødrene Hartmanns Fond, and Snedkermester Sophus Jacobsen og hustru Astrid Jacobsens Fond.
The researchers had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE
ED Visits for Diabetes on the Rise in the US
Emergency department (ED) visits by adults with diabetes increased by more than 25% since 2012, with the highest rates among Blacks and those aged over 65 years, a new data brief from the Centers for Disease Control and Prevention’s National Center for Health Statistics shows.
In 2021, diabetes was the eighth leading cause of death in the United States, according to the brief, published online on December 19, 2023. Its frequency is increasing in young people, and increasing age is a risk factor for hospitalization.
The latest data show that in 2020-2021, the overall annual ED visit rate was 72.2 visits per 1000 adults with diabetes, with no significant difference in terms of sex (75.1 visits per 1000 women vs 69.1 visits per 1000 men). By race/ethnicity, Blacks had the highest rates, at 135.5 visits per 1000 adults, followed by Whites (69.9) and Hispanics (52.3). The rates increased with age for both women and men, and among the three race/ethnic groups.
Comorbidities Count
The most ED visits were made by patients with diabetes and two to four other chronic conditions (541.4 visits per 1000 visits). Rates for patients without other chronic conditions were the lowest (90.2).
Among individuals with diabetes aged 18-44 years, ED visit rates were the highest for those with two to four other chronic conditions (402.0) and lowest among those with five or more other conditions (93.8).
Among patients aged 45-64 years, ED visit rates were the highest for those with two to four other chronic conditions (526.4) and lowest for those without other conditions (87.7). In the 65 years and older group, rates were the highest for individuals with two to four other chronic conditions (605.2), followed by five or more conditions (217.7), one other condition (140.6), and no other conditions (36.5).
Notably, the ED visit rates for those with two to four or five or more other chronic conditions increased with age, whereas visits for those with no other chronic conditions or one other condition decreased with age.
Decade-Long Trend
ED visit rates among adults with diabetes increased throughout the past decade, from 48.6 visits per 1000 adults in 2012 to 74.9 per 1000 adults in 2021. Rates for those aged 65 and older were higher than all other age groups, increasing from 113.4 to 156.8. Increases were also seen among those aged 45-64 years (53.1 in 2012 to 89.2 in 2021) and 18-44 (20.9 in 2012 to 26.4 in 2016, then plateauing from 2016-2021).
Data are based on a sample of 4051 ED visits, representing about 18,238,000 average annual visits made by adults with diabetes to nonfederal, general, and short-stay hospitals during 2020-2021.
Taken together, these most recent estimates “show an increasing trend in rates by adults with diabetes in the ED setting,” the authors concluded.
A version of this article appeared on Medscape.com.
Emergency department (ED) visits by adults with diabetes increased by more than 25% since 2012, with the highest rates among Blacks and those aged over 65 years, a new data brief from the Centers for Disease Control and Prevention’s National Center for Health Statistics shows.
In 2021, diabetes was the eighth leading cause of death in the United States, according to the brief, published online on December 19, 2023. Its frequency is increasing in young people, and increasing age is a risk factor for hospitalization.
The latest data show that in 2020-2021, the overall annual ED visit rate was 72.2 visits per 1000 adults with diabetes, with no significant difference in terms of sex (75.1 visits per 1000 women vs 69.1 visits per 1000 men). By race/ethnicity, Blacks had the highest rates, at 135.5 visits per 1000 adults, followed by Whites (69.9) and Hispanics (52.3). The rates increased with age for both women and men, and among the three race/ethnic groups.
Comorbidities Count
The most ED visits were made by patients with diabetes and two to four other chronic conditions (541.4 visits per 1000 visits). Rates for patients without other chronic conditions were the lowest (90.2).
Among individuals with diabetes aged 18-44 years, ED visit rates were the highest for those with two to four other chronic conditions (402.0) and lowest among those with five or more other conditions (93.8).
Among patients aged 45-64 years, ED visit rates were the highest for those with two to four other chronic conditions (526.4) and lowest for those without other conditions (87.7). In the 65 years and older group, rates were the highest for individuals with two to four other chronic conditions (605.2), followed by five or more conditions (217.7), one other condition (140.6), and no other conditions (36.5).
Notably, the ED visit rates for those with two to four or five or more other chronic conditions increased with age, whereas visits for those with no other chronic conditions or one other condition decreased with age.
Decade-Long Trend
ED visit rates among adults with diabetes increased throughout the past decade, from 48.6 visits per 1000 adults in 2012 to 74.9 per 1000 adults in 2021. Rates for those aged 65 and older were higher than all other age groups, increasing from 113.4 to 156.8. Increases were also seen among those aged 45-64 years (53.1 in 2012 to 89.2 in 2021) and 18-44 (20.9 in 2012 to 26.4 in 2016, then plateauing from 2016-2021).
Data are based on a sample of 4051 ED visits, representing about 18,238,000 average annual visits made by adults with diabetes to nonfederal, general, and short-stay hospitals during 2020-2021.
Taken together, these most recent estimates “show an increasing trend in rates by adults with diabetes in the ED setting,” the authors concluded.
A version of this article appeared on Medscape.com.
Emergency department (ED) visits by adults with diabetes increased by more than 25% since 2012, with the highest rates among Blacks and those aged over 65 years, a new data brief from the Centers for Disease Control and Prevention’s National Center for Health Statistics shows.
In 2021, diabetes was the eighth leading cause of death in the United States, according to the brief, published online on December 19, 2023. Its frequency is increasing in young people, and increasing age is a risk factor for hospitalization.
The latest data show that in 2020-2021, the overall annual ED visit rate was 72.2 visits per 1000 adults with diabetes, with no significant difference in terms of sex (75.1 visits per 1000 women vs 69.1 visits per 1000 men). By race/ethnicity, Blacks had the highest rates, at 135.5 visits per 1000 adults, followed by Whites (69.9) and Hispanics (52.3). The rates increased with age for both women and men, and among the three race/ethnic groups.
Comorbidities Count
The most ED visits were made by patients with diabetes and two to four other chronic conditions (541.4 visits per 1000 visits). Rates for patients without other chronic conditions were the lowest (90.2).
Among individuals with diabetes aged 18-44 years, ED visit rates were the highest for those with two to four other chronic conditions (402.0) and lowest among those with five or more other conditions (93.8).
Among patients aged 45-64 years, ED visit rates were the highest for those with two to four other chronic conditions (526.4) and lowest for those without other conditions (87.7). In the 65 years and older group, rates were the highest for individuals with two to four other chronic conditions (605.2), followed by five or more conditions (217.7), one other condition (140.6), and no other conditions (36.5).
Notably, the ED visit rates for those with two to four or five or more other chronic conditions increased with age, whereas visits for those with no other chronic conditions or one other condition decreased with age.
Decade-Long Trend
ED visit rates among adults with diabetes increased throughout the past decade, from 48.6 visits per 1000 adults in 2012 to 74.9 per 1000 adults in 2021. Rates for those aged 65 and older were higher than all other age groups, increasing from 113.4 to 156.8. Increases were also seen among those aged 45-64 years (53.1 in 2012 to 89.2 in 2021) and 18-44 (20.9 in 2012 to 26.4 in 2016, then plateauing from 2016-2021).
Data are based on a sample of 4051 ED visits, representing about 18,238,000 average annual visits made by adults with diabetes to nonfederal, general, and short-stay hospitals during 2020-2021.
Taken together, these most recent estimates “show an increasing trend in rates by adults with diabetes in the ED setting,” the authors concluded.
A version of this article appeared on Medscape.com.
How to Reduce Cardiovascular Morbidity and Mortality in Psoriasis and PsA
Patients with psoriatic disease have significantly higher risks of myocardial infarction, stroke, and cardiovascular mortality than does the general population, yet research consistently paints what dermatologist Joel M. Gelfand, MD, calls an “abysmal” picture: Only a minority of patients with psoriatic disease know about their increased risks, only a minority of dermatologists and rheumatologists screen for cardiovascular risk factors like lipid levels and blood pressure, and only a minority of patients diagnosed with hyperlipidemia are adequately treated with statin therapy.
In the literature and at medical meetings, Dr. Gelfand and others who have studied cardiovascular disease (CVD) comorbidity and physician practices have been urging dermatologists and rheumatologists to play a more consistent and active role in primary cardiovascular prevention for patients with psoriatic disease, who are up to 50% more likely than patients without it to develop CVD and who tend to have atherosclerosis at earlier ages.
According to the 2019 joint American Academy of Dermatology (AAD)–National Psoriasis Foundation (NPF) guidelines for managing psoriasis “with awareness and attention to comorbidities,” this means not only ensuring that all patients with psoriasis receive standard CV risk assessment (screening for hypertension, diabetes, and hyperlipidemia), but also recognizing that patients who are candidates for systemic therapy or phototherapy — or who have psoriasis involving > 10% of body surface area — may benefit from earlier and more frequent screening.
CV risk and premature mortality rises with the severity of skin disease, and patients with psoriatic arthritis (PsA) are believed to have risk levels similar to patients with moderate-severe psoriasis, cardiologist Michael S. Garshick, MD, director of the cardio-rheumatology program at New York University Langone Health, said in an interview.
In a recent survey study of 100 patients seen at NYU Langone Health’s psoriasis specialty clinic, only one-third indicated they had been advised by their physicians to be screened for CV risk factors, and only one-third reported having been told of the connection between psoriasis and CVD risk. Dr. Garshick shared the unpublished findings at the annual research symposium of the NPF in October.
Similarly, data from the National Ambulatory Medical Care Survey shows that just 16% of psoriasis-related visits to dermatology providers from 2007 to 2016 involved screening for CV risk factors. Screening rates were 11% for body mass index, 7.4% for blood pressure, 2.9% for cholesterol, and 1.7% for glucose, Dr. Gelfand and coauthors reported in 2023. .
Such findings are concerning because research shows that fewer than a quarter of patients with psoriasis have a primary care visit within a year of establishing care with their physicians, and that, overall, fewer than half of commercially insured adults under age 65 visit a primary care physician each year, according to John S. Barbieri, MD, of the department of dermatology at Brigham and Women’s Hospital in Boston. He included these findings when reporting in 2022 on a survey study on CVD screening.
In many cases, dermatologists and rheumatologists may be the primary providers for patients with psoriatic disease. So, “the question is, how can the dermatologist or rheumatologist use their interactions as a touchpoint to improve the patient’s well-being?” Dr. Barbieri said in an interview.
For the dermatologist, educating patients about the higher CVD risk fits well into conversations about “how there may be inflammation inside the body as well as in the skin,” he said. “Talk about cardiovascular risk just as you talk about PsA risk.” Both specialists, he added, can incorporate blood pressure readings and look for opportunities to measure lipid levels and hemoglobin A1c (HbA1c). These labs can easily be integrated into a biologic work-up.
“The hard part — and this needs to be individualized — is how do you want to handle [abnormal readings]? Do you want to take on a lot of the ownership and calculate [10-year CVD] risk scores and then counsel patients accordingly?” Dr. Barbieri said. “Or do you want to try to refer, and encourage them to work with their PCP? There a high-touch version and a low-touch version of how you can turn screening into action, into a care plan.”
Beyond traditional risk elevation, the primary care hand-off
Rheumatologists “in general may be more apt to screen for cardiovascular disease” as a result of their internal medicine residency training, and “we’re generally more comfortable prescribing ... if we need to,” said Alexis R. Ogdie, MD, a rheumatologist at the Hospital of the University of Pennsylvania, Philadelphia, and director of the Penn Psoriatic Arthritis Clinic.
Referral to a preventive cardiologist for management of abnormal lab results or ongoing monitoring and prevention is ideal, but when hand-offs to primary care physicians are made — the more common scenario — education is important. “A common problem is that there is underrecognition of the cardiovascular risk being elevated in our patients,” she said, above and beyond risk posed by traditional risk factors such as dyslipidemia, hypertension, metabolic syndrome, and obesity, all of which have been shown to occur more frequently in patients with psoriatic disease than in the general population.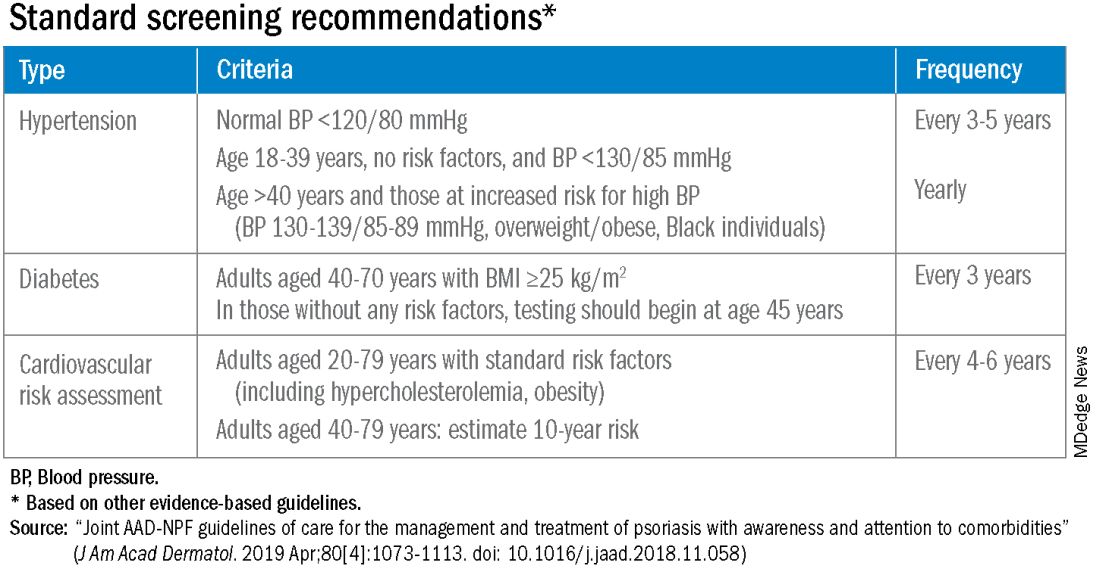
Risk stratification guides CVD prevention in the general population, and “if you use typical scores for cardiovascular risk, they may underestimate risk for our patients with PsA,” said Dr. Ogdie, who has reported on CV risk in patients with PsA. “Relative to what the patient’s perceived risk is, they may be treated similarly (to the general population). But relative to their actual risk, they’re undertreated.”
The 2019 AAD-NPF psoriasis guidelines recommend utilizing a 1.5 multiplication factor in risk score models, such as the American College of Cardiology’s Atherosclerotic Cardiovascular Disease (ASCVD) Risk Estimator, when the patient has a body surface area >10% or is a candidate for systemic therapy or phototherapy.
Similarly, the 2018 American Heart Association (AHA)-ACC Guideline on the Management of Blood Cholesterol defines psoriasis, along with RA, metabolic syndrome, HIV, and other diseases, as a “cardiovascular risk enhancer” that should be factored into assessments of ASCVD risk. (The guideline does not specify a psoriasis severity threshold.)
“It’s the first time the specialty [of cardiology] has said, ‘pay attention to a skin disease,’ ” Dr. Gelfand said at the NPF meeting.
Using the 1.5 multiplication factor, a patient who otherwise would be classified in the AHA/ACC guideline as “borderline risk,” with a 10-year ASCVD risk of 5% to <7.5%, would instead have an “intermediate” 10-year ASCVD risk of ≥7.5% to <20%. Application of the AHA-ACC “risk enhancer” would have a similar effect.
For management, the main impact of psoriasis being considered a risk enhancer is that “it lowers the threshold for treatment with standard cardiovascular prevention medications such as statins.”
In general, “we should be taking a more aggressive approach to the management of traditional cardiovascular risk factors” in patients with psoriatic disease, he said. Instead of telling a patient with mildly elevated blood pressure, ‘I’ll see you in a year or two,’ or a patient entering a prediabetic stage to “watch what you eat, and I’ll see you in a couple of years,” clinicians need to be more vigilant.
“It’s about recognizing that these traditional cardiometabolic risk factors, synergistically with psoriasis, can start enhancing CV risk at an earlier age than we might expect,” said Dr. Garshick, whose 2021 review of CV risk in psoriasis describes how the inflammatory milieu in psoriasis is linked to atherosclerosis development.
Cardiologists are aware of this, but “many primary care physicians are not. It takes time for medical knowledge to diffuse,” Dr. Gelfand said. “Tell the PCP, in notes or in a form letter, that there is a higher risk of CV disease, and reference the AHA/ACC guidelines,” he advised. “You don’t want your patient to go to their doctor and the doctor to [be uninformed].”
‘Patients trust us’
Dr. Gelfand has been at the forefront of research on psoriasis and heart disease. A study he coauthored in 2006, for instance, documented an independent risk of MI, with adjusted relative risks of 1.29 and 3.10 for a 30-year-old patient with mild or severe disease, respectively, and higher risks for a 60-year-old. In 2010, he and coinvestigators found that severe psoriasis was an independent risk factor for CV mortality (HR, 1.57) after adjusting for age, sex, smoking, diabetes, hypertension, and hyperlipidemia.
Today, along with Dr. Barbieri, Dr. Ogdie, and others, he is studying the feasibility and efficacy of a proposed national, “centralized care coordinator” model of care whereby dermatologists and rheumatologists would educate the patient, order lipid and HbA1c measurements as medically appropriate, and then refer patients as needed to a care coordinator. The care coordinator would calculate a 10-year CVD risk score and counsel the patient on possible next steps.
In a pilot study of 85 patients at four sites, 92% of patients followed through on their physician’s recommendations to have labs drawn, and 86% indicated the model was acceptable and feasible. A total of 27% of patients had “newly identified, previously undiagnosed, elevated cardiovascular disease risk,” and exploratory effectiveness results indicated a successful reduction in predicted CVD risk in patients who started statins, Dr. Gelfand reported at the NPF meeting.
With funding from the NPF, a larger, single-arm, pragmatic “CP3” trial (NCT05908240) is enrolling 525 patients with psoriasis at 10-20 academic and nonacademic dermatology sites across the United States to further test the model. The primary endpoint will be the change in LDL cholesterol measured at 6 months among people with a 10-year risk ≥5%. Secondary endpoints will cover improvement in disease severity and quality of life, behavior modification, patient experience, and other issues.
“We have only 10-15 minutes [with patients] ... a care coordinator who is empathetic and understanding and [informed] could make a big difference,” Dr. Gelfand said at the NPF meeting. If findings are positive, the model would be tested in rheumatology sites as well. The hope, he said, is that the NPF would be able to fund an in-house care coordinator(s) for the long-term.
Notably, a patient survey conducted as part of exploratory research leading up to the care coordinator project showed that patients trust their dermatologist or rheumatologist for CVD education and screening. Among 160 patients with psoriasis and 162 patients with PsA, 76% and 90% agreed that “I would like it if my dermatologist/rheumatologist educated me about my risk of heart disease,” and 60% and 75%, respectively, agree that “it would be convenient for me to have my cholesterol checked by my dermatologist/rheumatologist.”
“Patients trust us,” Dr. Gelfand said at the NPF meeting. “And the pilot study shows us that patients are motivated.”
Taking an individualized, holistic, longitudinal approach
“Sometimes you do have to triage bit,” Dr. Gelfand said in an interview. “For a young person with normal body weight who doesn’t smoke and has mild psoriasis, one could just educate and advise that they see their primary care physician” for monitoring.
“But for the same patient who is obese, maybe smokes, and doesn’t have a primary care physician, I’d order labs,” he said. “You don’t want a patient walking out the door with an [undiagnosed] LDL of 160 or hypertension.”
Age is also an important consideration, as excess CVD risk associated with autoimmune diseases like psoriasis rises with age, Dr. Gelfand said during a seminar on psoriasis and PsA held at NYU Langone in December. For a young person, typically, “I need to focus on education and lifestyle … setting them on a healthy lifestyle trajectory,” he said. “Once they get to 40, from 40 to 75 or so, that’s a sweet spot for medical intervention to lower cardiovascular risk.”
Even at older ages, however, lipid management is not the be-all and end-all, he said in the interview. “We have to be holistic.”
One advantage of having highly successful therapies for psoriasis, and to a lesser extent PsA, is the time that becomes available during follow-up visits — once disease is under control — to “focus on other things,” he said. Waiting until disease is under control to discuss diet, exercise, or smoking, for instance, makes sense anyway, he said. “You don’t want to overwhelm patients with too much to do at once.”
Indeed, said dermatologist Robert E. Kalb, MD, of the Buffalo Medical Group in Buffalo, NY, “patients have an open mind [about discussing cardiovascular disease risk], but it is not high on their radar. Most of them just want to get their skin clear.” (Dr. Kalb participated in the care coordinator pilot study, and said in an interview that since its completion, he has been more routinely ordering relevant labs.)
Rheumatologists are less fortunate with highly successful therapies, but “over the continuum of care, we do have time in office visits” to discuss issues like smoking, exercise, and lifestyle, Dr. Ogdie said. “I think of each of those pieces as part of our job.”
In the future, as researchers learn more about the impact of psoriasis and PsA treatments on CVD risk, it may be possible to tailor treatments or to prescribe treatments knowing that the therapies could reduce risk. Observational and epidemiologic data suggest that tumor necrosis factor-alpha inhibitor therapy over 3 years reduces the risk of MI, and that patients whose psoriasis is treated have reduced aortic inflammation, improved myocardial strain, and reduced coronary plaque burden, Dr. Garshick said at the NPF meeting.
“But when we look at the randomized controlled trials, they’re actually inconclusive that targeting inflammation in psoriatic disease reduces surrogates of cardiovascular disease,” he said. Dr. Garshick’s own research focuses on platelet and endothelial biology in psoriasis.
Dr. Barbieri reported he had no relevant disclosures. Dr. Garshick reported consulting fees from Bristol-Myers Squibb, Kiniksa, Horizon Therapeutics, and Agepha. Dr. Ogdie reported financial relationships with AbbVie, Amgen, Bristol-Myers Squibb, Celgene, Eli Lilly, Gilead, GlaxoSmithKline, Janssen, Novartis, Pfizer, Takeda, and UCB. Dr. Gelfand reported serving as a consultant for AbbVie, Artax, Bristol-Myers Squibb, GlaxoSmithKline, and other companies.
Patients with psoriatic disease have significantly higher risks of myocardial infarction, stroke, and cardiovascular mortality than does the general population, yet research consistently paints what dermatologist Joel M. Gelfand, MD, calls an “abysmal” picture: Only a minority of patients with psoriatic disease know about their increased risks, only a minority of dermatologists and rheumatologists screen for cardiovascular risk factors like lipid levels and blood pressure, and only a minority of patients diagnosed with hyperlipidemia are adequately treated with statin therapy.
In the literature and at medical meetings, Dr. Gelfand and others who have studied cardiovascular disease (CVD) comorbidity and physician practices have been urging dermatologists and rheumatologists to play a more consistent and active role in primary cardiovascular prevention for patients with psoriatic disease, who are up to 50% more likely than patients without it to develop CVD and who tend to have atherosclerosis at earlier ages.
According to the 2019 joint American Academy of Dermatology (AAD)–National Psoriasis Foundation (NPF) guidelines for managing psoriasis “with awareness and attention to comorbidities,” this means not only ensuring that all patients with psoriasis receive standard CV risk assessment (screening for hypertension, diabetes, and hyperlipidemia), but also recognizing that patients who are candidates for systemic therapy or phototherapy — or who have psoriasis involving > 10% of body surface area — may benefit from earlier and more frequent screening.
CV risk and premature mortality rises with the severity of skin disease, and patients with psoriatic arthritis (PsA) are believed to have risk levels similar to patients with moderate-severe psoriasis, cardiologist Michael S. Garshick, MD, director of the cardio-rheumatology program at New York University Langone Health, said in an interview.
In a recent survey study of 100 patients seen at NYU Langone Health’s psoriasis specialty clinic, only one-third indicated they had been advised by their physicians to be screened for CV risk factors, and only one-third reported having been told of the connection between psoriasis and CVD risk. Dr. Garshick shared the unpublished findings at the annual research symposium of the NPF in October.
Similarly, data from the National Ambulatory Medical Care Survey shows that just 16% of psoriasis-related visits to dermatology providers from 2007 to 2016 involved screening for CV risk factors. Screening rates were 11% for body mass index, 7.4% for blood pressure, 2.9% for cholesterol, and 1.7% for glucose, Dr. Gelfand and coauthors reported in 2023. .
Such findings are concerning because research shows that fewer than a quarter of patients with psoriasis have a primary care visit within a year of establishing care with their physicians, and that, overall, fewer than half of commercially insured adults under age 65 visit a primary care physician each year, according to John S. Barbieri, MD, of the department of dermatology at Brigham and Women’s Hospital in Boston. He included these findings when reporting in 2022 on a survey study on CVD screening.
In many cases, dermatologists and rheumatologists may be the primary providers for patients with psoriatic disease. So, “the question is, how can the dermatologist or rheumatologist use their interactions as a touchpoint to improve the patient’s well-being?” Dr. Barbieri said in an interview.
For the dermatologist, educating patients about the higher CVD risk fits well into conversations about “how there may be inflammation inside the body as well as in the skin,” he said. “Talk about cardiovascular risk just as you talk about PsA risk.” Both specialists, he added, can incorporate blood pressure readings and look for opportunities to measure lipid levels and hemoglobin A1c (HbA1c). These labs can easily be integrated into a biologic work-up.
“The hard part — and this needs to be individualized — is how do you want to handle [abnormal readings]? Do you want to take on a lot of the ownership and calculate [10-year CVD] risk scores and then counsel patients accordingly?” Dr. Barbieri said. “Or do you want to try to refer, and encourage them to work with their PCP? There a high-touch version and a low-touch version of how you can turn screening into action, into a care plan.”
Beyond traditional risk elevation, the primary care hand-off
Rheumatologists “in general may be more apt to screen for cardiovascular disease” as a result of their internal medicine residency training, and “we’re generally more comfortable prescribing ... if we need to,” said Alexis R. Ogdie, MD, a rheumatologist at the Hospital of the University of Pennsylvania, Philadelphia, and director of the Penn Psoriatic Arthritis Clinic.
Referral to a preventive cardiologist for management of abnormal lab results or ongoing monitoring and prevention is ideal, but when hand-offs to primary care physicians are made — the more common scenario — education is important. “A common problem is that there is underrecognition of the cardiovascular risk being elevated in our patients,” she said, above and beyond risk posed by traditional risk factors such as dyslipidemia, hypertension, metabolic syndrome, and obesity, all of which have been shown to occur more frequently in patients with psoriatic disease than in the general population.
Risk stratification guides CVD prevention in the general population, and “if you use typical scores for cardiovascular risk, they may underestimate risk for our patients with PsA,” said Dr. Ogdie, who has reported on CV risk in patients with PsA. “Relative to what the patient’s perceived risk is, they may be treated similarly (to the general population). But relative to their actual risk, they’re undertreated.”
The 2019 AAD-NPF psoriasis guidelines recommend utilizing a 1.5 multiplication factor in risk score models, such as the American College of Cardiology’s Atherosclerotic Cardiovascular Disease (ASCVD) Risk Estimator, when the patient has a body surface area >10% or is a candidate for systemic therapy or phototherapy.
Similarly, the 2018 American Heart Association (AHA)-ACC Guideline on the Management of Blood Cholesterol defines psoriasis, along with RA, metabolic syndrome, HIV, and other diseases, as a “cardiovascular risk enhancer” that should be factored into assessments of ASCVD risk. (The guideline does not specify a psoriasis severity threshold.)
“It’s the first time the specialty [of cardiology] has said, ‘pay attention to a skin disease,’ ” Dr. Gelfand said at the NPF meeting.
Using the 1.5 multiplication factor, a patient who otherwise would be classified in the AHA/ACC guideline as “borderline risk,” with a 10-year ASCVD risk of 5% to <7.5%, would instead have an “intermediate” 10-year ASCVD risk of ≥7.5% to <20%. Application of the AHA-ACC “risk enhancer” would have a similar effect.
For management, the main impact of psoriasis being considered a risk enhancer is that “it lowers the threshold for treatment with standard cardiovascular prevention medications such as statins.”
In general, “we should be taking a more aggressive approach to the management of traditional cardiovascular risk factors” in patients with psoriatic disease, he said. Instead of telling a patient with mildly elevated blood pressure, ‘I’ll see you in a year or two,’ or a patient entering a prediabetic stage to “watch what you eat, and I’ll see you in a couple of years,” clinicians need to be more vigilant.
“It’s about recognizing that these traditional cardiometabolic risk factors, synergistically with psoriasis, can start enhancing CV risk at an earlier age than we might expect,” said Dr. Garshick, whose 2021 review of CV risk in psoriasis describes how the inflammatory milieu in psoriasis is linked to atherosclerosis development.
Cardiologists are aware of this, but “many primary care physicians are not. It takes time for medical knowledge to diffuse,” Dr. Gelfand said. “Tell the PCP, in notes or in a form letter, that there is a higher risk of CV disease, and reference the AHA/ACC guidelines,” he advised. “You don’t want your patient to go to their doctor and the doctor to [be uninformed].”
‘Patients trust us’
Dr. Gelfand has been at the forefront of research on psoriasis and heart disease. A study he coauthored in 2006, for instance, documented an independent risk of MI, with adjusted relative risks of 1.29 and 3.10 for a 30-year-old patient with mild or severe disease, respectively, and higher risks for a 60-year-old. In 2010, he and coinvestigators found that severe psoriasis was an independent risk factor for CV mortality (HR, 1.57) after adjusting for age, sex, smoking, diabetes, hypertension, and hyperlipidemia.
Today, along with Dr. Barbieri, Dr. Ogdie, and others, he is studying the feasibility and efficacy of a proposed national, “centralized care coordinator” model of care whereby dermatologists and rheumatologists would educate the patient, order lipid and HbA1c measurements as medically appropriate, and then refer patients as needed to a care coordinator. The care coordinator would calculate a 10-year CVD risk score and counsel the patient on possible next steps.
In a pilot study of 85 patients at four sites, 92% of patients followed through on their physician’s recommendations to have labs drawn, and 86% indicated the model was acceptable and feasible. A total of 27% of patients had “newly identified, previously undiagnosed, elevated cardiovascular disease risk,” and exploratory effectiveness results indicated a successful reduction in predicted CVD risk in patients who started statins, Dr. Gelfand reported at the NPF meeting.
With funding from the NPF, a larger, single-arm, pragmatic “CP3” trial (NCT05908240) is enrolling 525 patients with psoriasis at 10-20 academic and nonacademic dermatology sites across the United States to further test the model. The primary endpoint will be the change in LDL cholesterol measured at 6 months among people with a 10-year risk ≥5%. Secondary endpoints will cover improvement in disease severity and quality of life, behavior modification, patient experience, and other issues.
“We have only 10-15 minutes [with patients] ... a care coordinator who is empathetic and understanding and [informed] could make a big difference,” Dr. Gelfand said at the NPF meeting. If findings are positive, the model would be tested in rheumatology sites as well. The hope, he said, is that the NPF would be able to fund an in-house care coordinator(s) for the long-term.
Notably, a patient survey conducted as part of exploratory research leading up to the care coordinator project showed that patients trust their dermatologist or rheumatologist for CVD education and screening. Among 160 patients with psoriasis and 162 patients with PsA, 76% and 90% agreed that “I would like it if my dermatologist/rheumatologist educated me about my risk of heart disease,” and 60% and 75%, respectively, agree that “it would be convenient for me to have my cholesterol checked by my dermatologist/rheumatologist.”
“Patients trust us,” Dr. Gelfand said at the NPF meeting. “And the pilot study shows us that patients are motivated.”
Taking an individualized, holistic, longitudinal approach
“Sometimes you do have to triage bit,” Dr. Gelfand said in an interview. “For a young person with normal body weight who doesn’t smoke and has mild psoriasis, one could just educate and advise that they see their primary care physician” for monitoring.
“But for the same patient who is obese, maybe smokes, and doesn’t have a primary care physician, I’d order labs,” he said. “You don’t want a patient walking out the door with an [undiagnosed] LDL of 160 or hypertension.”
Age is also an important consideration, as excess CVD risk associated with autoimmune diseases like psoriasis rises with age, Dr. Gelfand said during a seminar on psoriasis and PsA held at NYU Langone in December. For a young person, typically, “I need to focus on education and lifestyle … setting them on a healthy lifestyle trajectory,” he said. “Once they get to 40, from 40 to 75 or so, that’s a sweet spot for medical intervention to lower cardiovascular risk.”
Even at older ages, however, lipid management is not the be-all and end-all, he said in the interview. “We have to be holistic.”
One advantage of having highly successful therapies for psoriasis, and to a lesser extent PsA, is the time that becomes available during follow-up visits — once disease is under control — to “focus on other things,” he said. Waiting until disease is under control to discuss diet, exercise, or smoking, for instance, makes sense anyway, he said. “You don’t want to overwhelm patients with too much to do at once.”
Indeed, said dermatologist Robert E. Kalb, MD, of the Buffalo Medical Group in Buffalo, NY, “patients have an open mind [about discussing cardiovascular disease risk], but it is not high on their radar. Most of them just want to get their skin clear.” (Dr. Kalb participated in the care coordinator pilot study, and said in an interview that since its completion, he has been more routinely ordering relevant labs.)
Rheumatologists are less fortunate with highly successful therapies, but “over the continuum of care, we do have time in office visits” to discuss issues like smoking, exercise, and lifestyle, Dr. Ogdie said. “I think of each of those pieces as part of our job.”
In the future, as researchers learn more about the impact of psoriasis and PsA treatments on CVD risk, it may be possible to tailor treatments or to prescribe treatments knowing that the therapies could reduce risk. Observational and epidemiologic data suggest that tumor necrosis factor-alpha inhibitor therapy over 3 years reduces the risk of MI, and that patients whose psoriasis is treated have reduced aortic inflammation, improved myocardial strain, and reduced coronary plaque burden, Dr. Garshick said at the NPF meeting.
“But when we look at the randomized controlled trials, they’re actually inconclusive that targeting inflammation in psoriatic disease reduces surrogates of cardiovascular disease,” he said. Dr. Garshick’s own research focuses on platelet and endothelial biology in psoriasis.
Dr. Barbieri reported he had no relevant disclosures. Dr. Garshick reported consulting fees from Bristol-Myers Squibb, Kiniksa, Horizon Therapeutics, and Agepha. Dr. Ogdie reported financial relationships with AbbVie, Amgen, Bristol-Myers Squibb, Celgene, Eli Lilly, Gilead, GlaxoSmithKline, Janssen, Novartis, Pfizer, Takeda, and UCB. Dr. Gelfand reported serving as a consultant for AbbVie, Artax, Bristol-Myers Squibb, GlaxoSmithKline, and other companies.
Patients with psoriatic disease have significantly higher risks of myocardial infarction, stroke, and cardiovascular mortality than does the general population, yet research consistently paints what dermatologist Joel M. Gelfand, MD, calls an “abysmal” picture: Only a minority of patients with psoriatic disease know about their increased risks, only a minority of dermatologists and rheumatologists screen for cardiovascular risk factors like lipid levels and blood pressure, and only a minority of patients diagnosed with hyperlipidemia are adequately treated with statin therapy.
In the literature and at medical meetings, Dr. Gelfand and others who have studied cardiovascular disease (CVD) comorbidity and physician practices have been urging dermatologists and rheumatologists to play a more consistent and active role in primary cardiovascular prevention for patients with psoriatic disease, who are up to 50% more likely than patients without it to develop CVD and who tend to have atherosclerosis at earlier ages.
According to the 2019 joint American Academy of Dermatology (AAD)–National Psoriasis Foundation (NPF) guidelines for managing psoriasis “with awareness and attention to comorbidities,” this means not only ensuring that all patients with psoriasis receive standard CV risk assessment (screening for hypertension, diabetes, and hyperlipidemia), but also recognizing that patients who are candidates for systemic therapy or phototherapy — or who have psoriasis involving > 10% of body surface area — may benefit from earlier and more frequent screening.
CV risk and premature mortality rises with the severity of skin disease, and patients with psoriatic arthritis (PsA) are believed to have risk levels similar to patients with moderate-severe psoriasis, cardiologist Michael S. Garshick, MD, director of the cardio-rheumatology program at New York University Langone Health, said in an interview.
In a recent survey study of 100 patients seen at NYU Langone Health’s psoriasis specialty clinic, only one-third indicated they had been advised by their physicians to be screened for CV risk factors, and only one-third reported having been told of the connection between psoriasis and CVD risk. Dr. Garshick shared the unpublished findings at the annual research symposium of the NPF in October.
Similarly, data from the National Ambulatory Medical Care Survey shows that just 16% of psoriasis-related visits to dermatology providers from 2007 to 2016 involved screening for CV risk factors. Screening rates were 11% for body mass index, 7.4% for blood pressure, 2.9% for cholesterol, and 1.7% for glucose, Dr. Gelfand and coauthors reported in 2023. .
Such findings are concerning because research shows that fewer than a quarter of patients with psoriasis have a primary care visit within a year of establishing care with their physicians, and that, overall, fewer than half of commercially insured adults under age 65 visit a primary care physician each year, according to John S. Barbieri, MD, of the department of dermatology at Brigham and Women’s Hospital in Boston. He included these findings when reporting in 2022 on a survey study on CVD screening.
In many cases, dermatologists and rheumatologists may be the primary providers for patients with psoriatic disease. So, “the question is, how can the dermatologist or rheumatologist use their interactions as a touchpoint to improve the patient’s well-being?” Dr. Barbieri said in an interview.
For the dermatologist, educating patients about the higher CVD risk fits well into conversations about “how there may be inflammation inside the body as well as in the skin,” he said. “Talk about cardiovascular risk just as you talk about PsA risk.” Both specialists, he added, can incorporate blood pressure readings and look for opportunities to measure lipid levels and hemoglobin A1c (HbA1c). These labs can easily be integrated into a biologic work-up.
“The hard part — and this needs to be individualized — is how do you want to handle [abnormal readings]? Do you want to take on a lot of the ownership and calculate [10-year CVD] risk scores and then counsel patients accordingly?” Dr. Barbieri said. “Or do you want to try to refer, and encourage them to work with their PCP? There a high-touch version and a low-touch version of how you can turn screening into action, into a care plan.”
Beyond traditional risk elevation, the primary care hand-off
Rheumatologists “in general may be more apt to screen for cardiovascular disease” as a result of their internal medicine residency training, and “we’re generally more comfortable prescribing ... if we need to,” said Alexis R. Ogdie, MD, a rheumatologist at the Hospital of the University of Pennsylvania, Philadelphia, and director of the Penn Psoriatic Arthritis Clinic.
Referral to a preventive cardiologist for management of abnormal lab results or ongoing monitoring and prevention is ideal, but when hand-offs to primary care physicians are made — the more common scenario — education is important. “A common problem is that there is underrecognition of the cardiovascular risk being elevated in our patients,” she said, above and beyond risk posed by traditional risk factors such as dyslipidemia, hypertension, metabolic syndrome, and obesity, all of which have been shown to occur more frequently in patients with psoriatic disease than in the general population.
Risk stratification guides CVD prevention in the general population, and “if you use typical scores for cardiovascular risk, they may underestimate risk for our patients with PsA,” said Dr. Ogdie, who has reported on CV risk in patients with PsA. “Relative to what the patient’s perceived risk is, they may be treated similarly (to the general population). But relative to their actual risk, they’re undertreated.”
The 2019 AAD-NPF psoriasis guidelines recommend utilizing a 1.5 multiplication factor in risk score models, such as the American College of Cardiology’s Atherosclerotic Cardiovascular Disease (ASCVD) Risk Estimator, when the patient has a body surface area >10% or is a candidate for systemic therapy or phototherapy.
Similarly, the 2018 American Heart Association (AHA)-ACC Guideline on the Management of Blood Cholesterol defines psoriasis, along with RA, metabolic syndrome, HIV, and other diseases, as a “cardiovascular risk enhancer” that should be factored into assessments of ASCVD risk. (The guideline does not specify a psoriasis severity threshold.)
“It’s the first time the specialty [of cardiology] has said, ‘pay attention to a skin disease,’ ” Dr. Gelfand said at the NPF meeting.
Using the 1.5 multiplication factor, a patient who otherwise would be classified in the AHA/ACC guideline as “borderline risk,” with a 10-year ASCVD risk of 5% to <7.5%, would instead have an “intermediate” 10-year ASCVD risk of ≥7.5% to <20%. Application of the AHA-ACC “risk enhancer” would have a similar effect.
For management, the main impact of psoriasis being considered a risk enhancer is that “it lowers the threshold for treatment with standard cardiovascular prevention medications such as statins.”
In general, “we should be taking a more aggressive approach to the management of traditional cardiovascular risk factors” in patients with psoriatic disease, he said. Instead of telling a patient with mildly elevated blood pressure, ‘I’ll see you in a year or two,’ or a patient entering a prediabetic stage to “watch what you eat, and I’ll see you in a couple of years,” clinicians need to be more vigilant.
“It’s about recognizing that these traditional cardiometabolic risk factors, synergistically with psoriasis, can start enhancing CV risk at an earlier age than we might expect,” said Dr. Garshick, whose 2021 review of CV risk in psoriasis describes how the inflammatory milieu in psoriasis is linked to atherosclerosis development.
Cardiologists are aware of this, but “many primary care physicians are not. It takes time for medical knowledge to diffuse,” Dr. Gelfand said. “Tell the PCP, in notes or in a form letter, that there is a higher risk of CV disease, and reference the AHA/ACC guidelines,” he advised. “You don’t want your patient to go to their doctor and the doctor to [be uninformed].”
‘Patients trust us’
Dr. Gelfand has been at the forefront of research on psoriasis and heart disease. A study he coauthored in 2006, for instance, documented an independent risk of MI, with adjusted relative risks of 1.29 and 3.10 for a 30-year-old patient with mild or severe disease, respectively, and higher risks for a 60-year-old. In 2010, he and coinvestigators found that severe psoriasis was an independent risk factor for CV mortality (HR, 1.57) after adjusting for age, sex, smoking, diabetes, hypertension, and hyperlipidemia.
Today, along with Dr. Barbieri, Dr. Ogdie, and others, he is studying the feasibility and efficacy of a proposed national, “centralized care coordinator” model of care whereby dermatologists and rheumatologists would educate the patient, order lipid and HbA1c measurements as medically appropriate, and then refer patients as needed to a care coordinator. The care coordinator would calculate a 10-year CVD risk score and counsel the patient on possible next steps.
In a pilot study of 85 patients at four sites, 92% of patients followed through on their physician’s recommendations to have labs drawn, and 86% indicated the model was acceptable and feasible. A total of 27% of patients had “newly identified, previously undiagnosed, elevated cardiovascular disease risk,” and exploratory effectiveness results indicated a successful reduction in predicted CVD risk in patients who started statins, Dr. Gelfand reported at the NPF meeting.
With funding from the NPF, a larger, single-arm, pragmatic “CP3” trial (NCT05908240) is enrolling 525 patients with psoriasis at 10-20 academic and nonacademic dermatology sites across the United States to further test the model. The primary endpoint will be the change in LDL cholesterol measured at 6 months among people with a 10-year risk ≥5%. Secondary endpoints will cover improvement in disease severity and quality of life, behavior modification, patient experience, and other issues.
“We have only 10-15 minutes [with patients] ... a care coordinator who is empathetic and understanding and [informed] could make a big difference,” Dr. Gelfand said at the NPF meeting. If findings are positive, the model would be tested in rheumatology sites as well. The hope, he said, is that the NPF would be able to fund an in-house care coordinator(s) for the long-term.
Notably, a patient survey conducted as part of exploratory research leading up to the care coordinator project showed that patients trust their dermatologist or rheumatologist for CVD education and screening. Among 160 patients with psoriasis and 162 patients with PsA, 76% and 90% agreed that “I would like it if my dermatologist/rheumatologist educated me about my risk of heart disease,” and 60% and 75%, respectively, agree that “it would be convenient for me to have my cholesterol checked by my dermatologist/rheumatologist.”
“Patients trust us,” Dr. Gelfand said at the NPF meeting. “And the pilot study shows us that patients are motivated.”
Taking an individualized, holistic, longitudinal approach
“Sometimes you do have to triage bit,” Dr. Gelfand said in an interview. “For a young person with normal body weight who doesn’t smoke and has mild psoriasis, one could just educate and advise that they see their primary care physician” for monitoring.
“But for the same patient who is obese, maybe smokes, and doesn’t have a primary care physician, I’d order labs,” he said. “You don’t want a patient walking out the door with an [undiagnosed] LDL of 160 or hypertension.”
Age is also an important consideration, as excess CVD risk associated with autoimmune diseases like psoriasis rises with age, Dr. Gelfand said during a seminar on psoriasis and PsA held at NYU Langone in December. For a young person, typically, “I need to focus on education and lifestyle … setting them on a healthy lifestyle trajectory,” he said. “Once they get to 40, from 40 to 75 or so, that’s a sweet spot for medical intervention to lower cardiovascular risk.”
Even at older ages, however, lipid management is not the be-all and end-all, he said in the interview. “We have to be holistic.”
One advantage of having highly successful therapies for psoriasis, and to a lesser extent PsA, is the time that becomes available during follow-up visits — once disease is under control — to “focus on other things,” he said. Waiting until disease is under control to discuss diet, exercise, or smoking, for instance, makes sense anyway, he said. “You don’t want to overwhelm patients with too much to do at once.”
Indeed, said dermatologist Robert E. Kalb, MD, of the Buffalo Medical Group in Buffalo, NY, “patients have an open mind [about discussing cardiovascular disease risk], but it is not high on their radar. Most of them just want to get their skin clear.” (Dr. Kalb participated in the care coordinator pilot study, and said in an interview that since its completion, he has been more routinely ordering relevant labs.)
Rheumatologists are less fortunate with highly successful therapies, but “over the continuum of care, we do have time in office visits” to discuss issues like smoking, exercise, and lifestyle, Dr. Ogdie said. “I think of each of those pieces as part of our job.”
In the future, as researchers learn more about the impact of psoriasis and PsA treatments on CVD risk, it may be possible to tailor treatments or to prescribe treatments knowing that the therapies could reduce risk. Observational and epidemiologic data suggest that tumor necrosis factor-alpha inhibitor therapy over 3 years reduces the risk of MI, and that patients whose psoriasis is treated have reduced aortic inflammation, improved myocardial strain, and reduced coronary plaque burden, Dr. Garshick said at the NPF meeting.
“But when we look at the randomized controlled trials, they’re actually inconclusive that targeting inflammation in psoriatic disease reduces surrogates of cardiovascular disease,” he said. Dr. Garshick’s own research focuses on platelet and endothelial biology in psoriasis.
Dr. Barbieri reported he had no relevant disclosures. Dr. Garshick reported consulting fees from Bristol-Myers Squibb, Kiniksa, Horizon Therapeutics, and Agepha. Dr. Ogdie reported financial relationships with AbbVie, Amgen, Bristol-Myers Squibb, Celgene, Eli Lilly, Gilead, GlaxoSmithKline, Janssen, Novartis, Pfizer, Takeda, and UCB. Dr. Gelfand reported serving as a consultant for AbbVie, Artax, Bristol-Myers Squibb, GlaxoSmithKline, and other companies.
Alcohol and CV Risk: Both Beneficial and Harmful Effects?
with evidence emerging that alcohol use may both increase and decrease the risk for CVD.
The answer may depend on the presence of circulating metabolites of alcohol, some of which may be beneficial while others may be harmful, new research suggests.
“We adopted an association analysis, looking at 60 metabolites produced during or after alcohol has been metabolized, to see whether those metabolites can link alcohol consumption with CVD,” senior author Jiantao Ma, PhD, MBBS, assistant professor, Division of Nutrition Epidemiology and Data Science, Friedman School, Tufts University, Boston, Massachusetts, said in an interview.
“We found that the relationship is quite complex, with some metabolites showing protective effects against CVD and others showing harmful effects,” said Dr. Ma. “This opens the door for future research because we think that these molecules can help [us] understand the mechanism of the relationship between alcohol and CVD.”
The study was published online in BMC Medicine.
J-Shaped Relationship?
Previous research has painted a confusing picture of the relationship between alcohol consumption and CVD. For example, some studies have suggested that moderate levels of drinking may be hazardous to cardiac health, while others have pointed to potential cardioprotective effects.
Nevertheless, “according to the latest ACC/AHA guidelines regarding alcohol consumption and its relationship to CVD, there is no level of alcohol use that is deemed safe and considered acceptable,” Saurabh Sharma, MD, program director, Internal Medicine Residency Program, and clinical assistant professor of cardiology, Geisinger Commonwealth School of Medicine, Scranton, Pennsylvania, said in an interview.
Older observational data suggested a “J-shaped” relationship between alcohol consumption and cardiovascular risk, such that a low to moderate amount might reduce risk, while higher amounts increase it, said Dr. Sharma, a member of the American College of Cardiology (ACC) Prevention of Cardiovascular Diseases Council.
“But it’s essential to note that these findings were based on observational studies. No randomized controlled trials have provided conclusive evidence supporting the idea that moderate alcohol consumption actively reduces cardiovascular risk,” he said.
The current study is also observational, but it shines a somewhat different spotlight on the subject by examining alcohol consumption–related metabolites, said Dr. Ma — that is, small molecules that are the intermediates or end-products of metabolism in many cellular processes.
Some recent research “shows that alcohol may be harmful or at least has no beneficial effect in CVD prevention,” he said. “Our motivation was to analyze the association using metabolites, genetics, and epigenetics, because we think that these molecules may help us understand some of the mechanisms that underlie the relationship between alcohol consumption and CVD, and partially answer the question of whether alcohol may be harmful or helpful.”
Caution Warranted
Although some previous studies have looked at metabolites, most analyzed alcohol consumption measured at a single time point, “which may not represent habitual or long-term alcohol consumption,” the researchers note.
The team used data derived from 2458 Framingham Heart Study Offspring participants (mean age, 56 ± 9.3 years at the fifth examination; 52% female), calculating the cumulative average alcohol consumption from total intake of beer, wine, and liquor over an average 20-year period. Most participants were overweight, close to one fifth were current smokers, and 636 developed CVD over the study period.
Participants were assessed every 4-8 years, with metabolites measured during the fifth examination.
Linear models were used to investigate the association of alcohol consumption with 211 plasma metabolites, adjusting for a variety of potential confounders, including age, sex, batch, smoking, diet, physical activity, body mass index, and familial relationship.
Sixty metabolites associated with cumulative average alcohol consumption were identified (P < .00024), after adjustment for confounders. Of these, 40 displayed positive associations with the cumulative average alcohol consumption, with the most significant metabolite being cholesteryl palmitoleate (CE16:1), a plasma cholesteryl ester involved with cholesterol metabolism.
One gram per day of higher alcohol consumption was associated with a higher-level CE16:1 in the blood (b = .023). Several other phosphatidylcholine metabolites were also positively associated with alcohol consumption.
On the other hand, 20 metabolites were negatively associated with alcohol consumption, with triacylglycerol 54:4 (TAG 54:4) displaying the most significant association (b = –.017).
The alcoholic beverages were not equal when it came to association with metabolites: 19 metabolites were significantly associated with the cumulative average consumption of beer, 30 with wine, and 32 with liquor. Seven were significantly associated with the cumulative consumption of all three types of drinks.
The researchers conducted survival analysis that identified 10 alcohol-associated metabolites associated with differential CVD risks, after adjusting for confounders. They also built two alcohol consumption–weighted metabolite scores using these 10 metabolites. After adjustment for confounders including CVD risk factors, the two scores had “comparable but opposite” associations with incident CVD, HR 1.11 (95% CI, 1.02-1.21) vs 0.88 (0.78-0.98; both P values = .02).
“We found that seven metabolites were harmful, while three were beneficial, “ Dr. Ma reported.
Dr. Ma cautioned that association “doesn’t represent causation.” On the basis of the findings, however, “we can hypothesize that if you drink a moderate amount of alcohol, you can either increase or decrease your risk of CVD.”
For people with cardiac conditions, “it would be [wise to be] cautious in recommending alcohol consumption,” he said. “For people without cardiac conditions, I would follow the recommendations of the AHA. If people don’t already drink alcohol, we don’t recommend that you start drinking it; and if you already drink, we’d recommend keeping it minimal.”
He cautioned that this is “only one study and we need more research if we are to generate a clearer message to the patient.” At present, perhaps the best message to patients is “to be cautious and warn them that there are potentially harmful effects,” he said.
Mendelian Randomization?
Dr. Sharma, who was not involved in the study, emphasized that it’s “crucial” to recognize that the study “does not alter the established understanding that any level of alcohol consumption poses harm to the heart,” and that “any amount of alcohol consumption has the potential to elevate triglyceride levels, thereby contributing to the increased risk of cardiovascular complications.”
Previously reported cardioprotective benefits “are likely influenced by confounding factors, such as lifestyle and sociodemographic elements,” he speculated.
He noted that observational studies “encounter challenges in disentangling the influence of factors like obesity, lack of exercise, and tobacco use” as well as reverse causality.
“To overcome these limitations, Mendelian randomization emerges as a robust method,” he suggested. “This approach utilizes measured genetic variations with known functions to investigate the causal effect of a modifiable exposure on disease within the framework of observational studies.”
Notably, certain studies using this approach, including one by Larsson and colleagues, and another by Biddinger and associates, “have provided valuable insights by establishing a clear and causal relationship between alcohol consumption and CVD,” he said.
The study was funded by the National Institute of Health’s National Institute on Alcohol Abuse and Alcoholism. Data collection in the Framingham Heart Study was supported by the National Heart, Lung, and Blood Institute. Dr. Ma and coauthors and Dr. Sharma disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
with evidence emerging that alcohol use may both increase and decrease the risk for CVD.
The answer may depend on the presence of circulating metabolites of alcohol, some of which may be beneficial while others may be harmful, new research suggests.
“We adopted an association analysis, looking at 60 metabolites produced during or after alcohol has been metabolized, to see whether those metabolites can link alcohol consumption with CVD,” senior author Jiantao Ma, PhD, MBBS, assistant professor, Division of Nutrition Epidemiology and Data Science, Friedman School, Tufts University, Boston, Massachusetts, said in an interview.
“We found that the relationship is quite complex, with some metabolites showing protective effects against CVD and others showing harmful effects,” said Dr. Ma. “This opens the door for future research because we think that these molecules can help [us] understand the mechanism of the relationship between alcohol and CVD.”
The study was published online in BMC Medicine.
J-Shaped Relationship?
Previous research has painted a confusing picture of the relationship between alcohol consumption and CVD. For example, some studies have suggested that moderate levels of drinking may be hazardous to cardiac health, while others have pointed to potential cardioprotective effects.
Nevertheless, “according to the latest ACC/AHA guidelines regarding alcohol consumption and its relationship to CVD, there is no level of alcohol use that is deemed safe and considered acceptable,” Saurabh Sharma, MD, program director, Internal Medicine Residency Program, and clinical assistant professor of cardiology, Geisinger Commonwealth School of Medicine, Scranton, Pennsylvania, said in an interview.
Older observational data suggested a “J-shaped” relationship between alcohol consumption and cardiovascular risk, such that a low to moderate amount might reduce risk, while higher amounts increase it, said Dr. Sharma, a member of the American College of Cardiology (ACC) Prevention of Cardiovascular Diseases Council.
“But it’s essential to note that these findings were based on observational studies. No randomized controlled trials have provided conclusive evidence supporting the idea that moderate alcohol consumption actively reduces cardiovascular risk,” he said.
The current study is also observational, but it shines a somewhat different spotlight on the subject by examining alcohol consumption–related metabolites, said Dr. Ma — that is, small molecules that are the intermediates or end-products of metabolism in many cellular processes.
Some recent research “shows that alcohol may be harmful or at least has no beneficial effect in CVD prevention,” he said. “Our motivation was to analyze the association using metabolites, genetics, and epigenetics, because we think that these molecules may help us understand some of the mechanisms that underlie the relationship between alcohol consumption and CVD, and partially answer the question of whether alcohol may be harmful or helpful.”
Caution Warranted
Although some previous studies have looked at metabolites, most analyzed alcohol consumption measured at a single time point, “which may not represent habitual or long-term alcohol consumption,” the researchers note.
The team used data derived from 2458 Framingham Heart Study Offspring participants (mean age, 56 ± 9.3 years at the fifth examination; 52% female), calculating the cumulative average alcohol consumption from total intake of beer, wine, and liquor over an average 20-year period. Most participants were overweight, close to one fifth were current smokers, and 636 developed CVD over the study period.
Participants were assessed every 4-8 years, with metabolites measured during the fifth examination.
Linear models were used to investigate the association of alcohol consumption with 211 plasma metabolites, adjusting for a variety of potential confounders, including age, sex, batch, smoking, diet, physical activity, body mass index, and familial relationship.
Sixty metabolites associated with cumulative average alcohol consumption were identified (P < .00024), after adjustment for confounders. Of these, 40 displayed positive associations with the cumulative average alcohol consumption, with the most significant metabolite being cholesteryl palmitoleate (CE16:1), a plasma cholesteryl ester involved with cholesterol metabolism.
One gram per day of higher alcohol consumption was associated with a higher-level CE16:1 in the blood (b = .023). Several other phosphatidylcholine metabolites were also positively associated with alcohol consumption.
On the other hand, 20 metabolites were negatively associated with alcohol consumption, with triacylglycerol 54:4 (TAG 54:4) displaying the most significant association (b = –.017).
The alcoholic beverages were not equal when it came to association with metabolites: 19 metabolites were significantly associated with the cumulative average consumption of beer, 30 with wine, and 32 with liquor. Seven were significantly associated with the cumulative consumption of all three types of drinks.
The researchers conducted survival analysis that identified 10 alcohol-associated metabolites associated with differential CVD risks, after adjusting for confounders. They also built two alcohol consumption–weighted metabolite scores using these 10 metabolites. After adjustment for confounders including CVD risk factors, the two scores had “comparable but opposite” associations with incident CVD, HR 1.11 (95% CI, 1.02-1.21) vs 0.88 (0.78-0.98; both P values = .02).
“We found that seven metabolites were harmful, while three were beneficial, “ Dr. Ma reported.
Dr. Ma cautioned that association “doesn’t represent causation.” On the basis of the findings, however, “we can hypothesize that if you drink a moderate amount of alcohol, you can either increase or decrease your risk of CVD.”
For people with cardiac conditions, “it would be [wise to be] cautious in recommending alcohol consumption,” he said. “For people without cardiac conditions, I would follow the recommendations of the AHA. If people don’t already drink alcohol, we don’t recommend that you start drinking it; and if you already drink, we’d recommend keeping it minimal.”
He cautioned that this is “only one study and we need more research if we are to generate a clearer message to the patient.” At present, perhaps the best message to patients is “to be cautious and warn them that there are potentially harmful effects,” he said.
Mendelian Randomization?
Dr. Sharma, who was not involved in the study, emphasized that it’s “crucial” to recognize that the study “does not alter the established understanding that any level of alcohol consumption poses harm to the heart,” and that “any amount of alcohol consumption has the potential to elevate triglyceride levels, thereby contributing to the increased risk of cardiovascular complications.”
Previously reported cardioprotective benefits “are likely influenced by confounding factors, such as lifestyle and sociodemographic elements,” he speculated.
He noted that observational studies “encounter challenges in disentangling the influence of factors like obesity, lack of exercise, and tobacco use” as well as reverse causality.
“To overcome these limitations, Mendelian randomization emerges as a robust method,” he suggested. “This approach utilizes measured genetic variations with known functions to investigate the causal effect of a modifiable exposure on disease within the framework of observational studies.”
Notably, certain studies using this approach, including one by Larsson and colleagues, and another by Biddinger and associates, “have provided valuable insights by establishing a clear and causal relationship between alcohol consumption and CVD,” he said.
The study was funded by the National Institute of Health’s National Institute on Alcohol Abuse and Alcoholism. Data collection in the Framingham Heart Study was supported by the National Heart, Lung, and Blood Institute. Dr. Ma and coauthors and Dr. Sharma disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
with evidence emerging that alcohol use may both increase and decrease the risk for CVD.
The answer may depend on the presence of circulating metabolites of alcohol, some of which may be beneficial while others may be harmful, new research suggests.
“We adopted an association analysis, looking at 60 metabolites produced during or after alcohol has been metabolized, to see whether those metabolites can link alcohol consumption with CVD,” senior author Jiantao Ma, PhD, MBBS, assistant professor, Division of Nutrition Epidemiology and Data Science, Friedman School, Tufts University, Boston, Massachusetts, said in an interview.
“We found that the relationship is quite complex, with some metabolites showing protective effects against CVD and others showing harmful effects,” said Dr. Ma. “This opens the door for future research because we think that these molecules can help [us] understand the mechanism of the relationship between alcohol and CVD.”
The study was published online in BMC Medicine.
J-Shaped Relationship?
Previous research has painted a confusing picture of the relationship between alcohol consumption and CVD. For example, some studies have suggested that moderate levels of drinking may be hazardous to cardiac health, while others have pointed to potential cardioprotective effects.
Nevertheless, “according to the latest ACC/AHA guidelines regarding alcohol consumption and its relationship to CVD, there is no level of alcohol use that is deemed safe and considered acceptable,” Saurabh Sharma, MD, program director, Internal Medicine Residency Program, and clinical assistant professor of cardiology, Geisinger Commonwealth School of Medicine, Scranton, Pennsylvania, said in an interview.
Older observational data suggested a “J-shaped” relationship between alcohol consumption and cardiovascular risk, such that a low to moderate amount might reduce risk, while higher amounts increase it, said Dr. Sharma, a member of the American College of Cardiology (ACC) Prevention of Cardiovascular Diseases Council.
“But it’s essential to note that these findings were based on observational studies. No randomized controlled trials have provided conclusive evidence supporting the idea that moderate alcohol consumption actively reduces cardiovascular risk,” he said.
The current study is also observational, but it shines a somewhat different spotlight on the subject by examining alcohol consumption–related metabolites, said Dr. Ma — that is, small molecules that are the intermediates or end-products of metabolism in many cellular processes.
Some recent research “shows that alcohol may be harmful or at least has no beneficial effect in CVD prevention,” he said. “Our motivation was to analyze the association using metabolites, genetics, and epigenetics, because we think that these molecules may help us understand some of the mechanisms that underlie the relationship between alcohol consumption and CVD, and partially answer the question of whether alcohol may be harmful or helpful.”
Caution Warranted
Although some previous studies have looked at metabolites, most analyzed alcohol consumption measured at a single time point, “which may not represent habitual or long-term alcohol consumption,” the researchers note.
The team used data derived from 2458 Framingham Heart Study Offspring participants (mean age, 56 ± 9.3 years at the fifth examination; 52% female), calculating the cumulative average alcohol consumption from total intake of beer, wine, and liquor over an average 20-year period. Most participants were overweight, close to one fifth were current smokers, and 636 developed CVD over the study period.
Participants were assessed every 4-8 years, with metabolites measured during the fifth examination.
Linear models were used to investigate the association of alcohol consumption with 211 plasma metabolites, adjusting for a variety of potential confounders, including age, sex, batch, smoking, diet, physical activity, body mass index, and familial relationship.
Sixty metabolites associated with cumulative average alcohol consumption were identified (P < .00024), after adjustment for confounders. Of these, 40 displayed positive associations with the cumulative average alcohol consumption, with the most significant metabolite being cholesteryl palmitoleate (CE16:1), a plasma cholesteryl ester involved with cholesterol metabolism.
One gram per day of higher alcohol consumption was associated with a higher-level CE16:1 in the blood (b = .023). Several other phosphatidylcholine metabolites were also positively associated with alcohol consumption.
On the other hand, 20 metabolites were negatively associated with alcohol consumption, with triacylglycerol 54:4 (TAG 54:4) displaying the most significant association (b = –.017).
The alcoholic beverages were not equal when it came to association with metabolites: 19 metabolites were significantly associated with the cumulative average consumption of beer, 30 with wine, and 32 with liquor. Seven were significantly associated with the cumulative consumption of all three types of drinks.
The researchers conducted survival analysis that identified 10 alcohol-associated metabolites associated with differential CVD risks, after adjusting for confounders. They also built two alcohol consumption–weighted metabolite scores using these 10 metabolites. After adjustment for confounders including CVD risk factors, the two scores had “comparable but opposite” associations with incident CVD, HR 1.11 (95% CI, 1.02-1.21) vs 0.88 (0.78-0.98; both P values = .02).
“We found that seven metabolites were harmful, while three were beneficial, “ Dr. Ma reported.
Dr. Ma cautioned that association “doesn’t represent causation.” On the basis of the findings, however, “we can hypothesize that if you drink a moderate amount of alcohol, you can either increase or decrease your risk of CVD.”
For people with cardiac conditions, “it would be [wise to be] cautious in recommending alcohol consumption,” he said. “For people without cardiac conditions, I would follow the recommendations of the AHA. If people don’t already drink alcohol, we don’t recommend that you start drinking it; and if you already drink, we’d recommend keeping it minimal.”
He cautioned that this is “only one study and we need more research if we are to generate a clearer message to the patient.” At present, perhaps the best message to patients is “to be cautious and warn them that there are potentially harmful effects,” he said.
Mendelian Randomization?
Dr. Sharma, who was not involved in the study, emphasized that it’s “crucial” to recognize that the study “does not alter the established understanding that any level of alcohol consumption poses harm to the heart,” and that “any amount of alcohol consumption has the potential to elevate triglyceride levels, thereby contributing to the increased risk of cardiovascular complications.”
Previously reported cardioprotective benefits “are likely influenced by confounding factors, such as lifestyle and sociodemographic elements,” he speculated.
He noted that observational studies “encounter challenges in disentangling the influence of factors like obesity, lack of exercise, and tobacco use” as well as reverse causality.
“To overcome these limitations, Mendelian randomization emerges as a robust method,” he suggested. “This approach utilizes measured genetic variations with known functions to investigate the causal effect of a modifiable exposure on disease within the framework of observational studies.”
Notably, certain studies using this approach, including one by Larsson and colleagues, and another by Biddinger and associates, “have provided valuable insights by establishing a clear and causal relationship between alcohol consumption and CVD,” he said.
The study was funded by the National Institute of Health’s National Institute on Alcohol Abuse and Alcoholism. Data collection in the Framingham Heart Study was supported by the National Heart, Lung, and Blood Institute. Dr. Ma and coauthors and Dr. Sharma disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
FROM BMC MEDICINE
Debate grows over facility fees as lawmakers urge greater transparency
Can the US healthcare system learn something about how to operate from car dealerships? Lawrence Kosinski, MD, MBA, a governing board member of American Gastroenterological Association (AGA), believes so.
There’s growing concern in the United States about the lack of clarity surrounding facility fees, which are intended to cover costs of maintaining medical facilities. Dr. Kosinski thinks that Congress should look into the transparency mandate it created for car prices as a model for how to address this.
A 1958 federal law set the stage for the consumer-friendly breakdown of costs and relevant performance data that anyone who has bought a new vehicle in the United States would recognize.
“You look at that and you know exactly what you are paying for,” Dr. Kosinski told this news organization. “In healthcare, we need something like that.”
Novel solutions like Dr. Kosinski’s will be increasingly necessary, as lawmakers on the state and federal level have begun to set their sights on tackling this issue.
The Biden administration in July expressed concern about an increased use of facility fees for healthcare provided at doctors’ offices, saying these additional costs often surprise consumers. House Energy and Commerce Chairwoman Cathy McMorris Rodgers (R-WA) also raised this issue several times this year, including at a May meeting about pending legislation on price transparency for health services, where she mentioned the case of a man who underwent eye surgery in Maine.
“His bill included three separate facility fees totaling $7800 and professional fees totaling $6200,” Ms. Rodgers said. “Why are three facility fees necessary for 1 hour of surgery in one O.R.?”
AGA’s Dr. Kosinski said facility fees cover the additional costs hospitals and clinics face in providing even routine treatments for some patients. For example, colonoscopy for a patient with a body mass index of 50 would pose special challenges for the anesthesiologist.
These factors need to be considered in setting policies on facility fees, he said. But there is no reason hospitals and other sites of medical care can’t make the information about facility fees easy for patients to find and understand, Dr. Kosinski said.
“I’m struggling to see a reason why we can’t be more transparent,” he said.
Big Battles Ahead
There are two connected battles ahead regarding facility fees: Efforts to restrict these additional charges for many medical services and fights over the need for greater transparency in general about health costs.
Senate Health, Education, Labor and Pensions Chairman Bernie Sanders (I-VT) is seeking to broadly restrict facility fees through his pending Primary Care and Health Workforce Act (S. 2840). The measure would block hospitals from charging health plans facility fees for many evaluation, management, and telehealth services.
The American Hospital Association (AHA) opposes it. They argue that the current payment approach rightly accounts for the added costs incurred when hospitals treat patients who are more likely to be ill or have chronic conditions than those seen in independent practices.
AHA said hospitals also need to maintain standby capacity for natural and man-made disasters, public health emergencies, and unexpected traumatic events. In September, AHA launched a television ad campaign to oppose any drive toward site-neutral policies. AHA says reducing the extra payments could cause more hospitals to shut their doors.
But there’s persistent interest in site-neutral payment, the term describing when the same reimbursement is given for care regardless of setting. This would lower pay for hospitals.
Among those pressing for change is an umbrella group of medical organizations known as the Alliance for Site Neutral Payment Reform. Its members include the American Academy of Family Physicians, American Academy of Orthopaedic Surgeons, American College of Physicians, Community Oncology Alliance, and Digestive Health Physicians Association.
And on November 9, Sen. Maggie Hassan (D-NH) argued for eventually including a site-neutral Medicare provision to a major healthcare package that the Senate Finance Committee is putting together.
Sen. Hassan is seeking to end what she called the “the practice of charging patients unfair hospital facility fees for care provided in the off-campus outpatient setting, like at a regular doctor’s office.”
Senate Finance Chairman Ron Wyden (D-OR) and the ranking Republican on the committee, Sen. Mike Crapo (R-ID), told Sen. Hassan they intended to work with her to see if this issue could be addressed in the pending legislative package.
A 2015 budget deal marked the last time Congress took a major step to address the higher cost of services provided in hospital-owned facilities.
Lawmakers then were scrambling to find cuts to offset spending in what became the 2015 Bipartisan Budget Act. This law established site-neutral payments under Medicare for services received at off-campus outpatient departments but exempted hospitals that already ran these kinds of operations or had advanced plans to create them.
Lawmakers are well aware of the potential savings from site-neutral policies and could look in time again to use them as part of a future budget deal.
In fact, in June, Sen. Hassan and Sens. Mike Braun (R-IN) and John Kennedy (R-LA) introduced a bill meant to basically end the exemption given in the 2015 deal to existing hospital outpatient departments, which has allowed higher Medicare payments. In a press release, Braun estimated that their proposed site-neutral change could save taxpayers $40 billion over a decade.
As Debate Continues, States Are Moving Ahead With Changes
Consumer activists have won a few battles this year at the state level about facility fees.
In July, Maine Gov. Janet Mills, a Democrat, signed a law that requires medical organizations to report facility fees to the state, which will share them publicly. Facility fees can pop up after a patient has received an insurance company estimate of the out-of-pocket costs for care.
“Patients receive bills bloated by healthcare providers that overcharge for services and insurance companies that deny claims without explanation,” the Portland Press Herald reported in a 2022 story. “And with little clout to fight back or even negotiate, feeling helpless, they often give up and pay, worn down by a system that is as time-consuming as it is obtuse.”
In May, Colorado enacted a law that will require patient notification about facility fees at many hospitals in the state.
In June, Connecticut expanded its law regarding facility fees and prohibited them for certain routine outpatient healthcare services. A statement from Gov. Ned Lamont’s office said the original intent of these facility fees was to ensure hospitals could maintain the around-the-clock care needed for inpatient and emergency care.
“However, these fees have been increasingly applied to services such as diagnostic testing and other routine services,” the statement said.
But there have been setbacks as well for those seeking to curb facilities.
The Texas Hospital Association (THA) in May said its advocacy defeated a pair of state bills, House bill 1692 and Senate bill 1275, that sought to limit facility fees for outpatient services.
In rallying opposition to these bills, THA said the loss of facility fees would threaten care for patients. Facility fees help cover costs “beyond the doctor’s bill,” such as “lab technicians, interpreters, medical records, security personnel, janitorial staff, and others,” THA said.
More Patients Shopping?
It’s unclear when — or if — Congress and other states will take major steps to reduce additional payments to hospitals for outpatient care.
But the increased use of high deductibles in health plans is driving more consumers to try to understand all of the costs of medical procedures ahead of time and, thus, drawing attention to facility fees, said Charlie Byrge, the chief operating officer of MDsave.
The average annual deductible levels for an individual increased by 3.0% to $2004 from 2020 to 2021 and for a family plan by 3.9% to $3868, according to a federal report. Some people have higher deductibles, exceeding $5000, Mr. Byrge said.
“That’s creating an opportunity for firms that can connect physicians directly with patients who will pay part or all of the costs of a treatment out of pocket,” he told this news organization.
Doctors and hospitals work with MDsave to charge preset prices for certain services, such as colonoscopies and mammograms. Consumers then can shop online to see if they can save. For example, in Nashville, Tennessee, where MDsave is based, the cost of a colonoscopy through MDsave is $2334, about half of the $4714 national average, according to the firm’s website.
This model for pricing routine medical care is akin to those used for other products and services, where companies decide ahead of time what to charge, he said.
“You don’t buy an airline ticket from Southwest or United or Delta and then there’s a bill after the fact because the price of gas went up a little bit on your flight,” Mr. Byrge said.
This will drive more competition among hospitals and clinics, in places where there are several sites of care in a region, Mr. Byrge said. But there are advantages for physicians and hospitals from the MDsave approach, he said.
“They know they’re getting paid upfront. They’re not going through the delays and headaches of the insurance reimbursement process. There are no denials. It’s just an upfront payment, and I think that’s what we’re starting to see the market really moving toward,” he said.
A version of this article appeared on Medscape.com.
Can the US healthcare system learn something about how to operate from car dealerships? Lawrence Kosinski, MD, MBA, a governing board member of American Gastroenterological Association (AGA), believes so.
There’s growing concern in the United States about the lack of clarity surrounding facility fees, which are intended to cover costs of maintaining medical facilities. Dr. Kosinski thinks that Congress should look into the transparency mandate it created for car prices as a model for how to address this.
A 1958 federal law set the stage for the consumer-friendly breakdown of costs and relevant performance data that anyone who has bought a new vehicle in the United States would recognize.
“You look at that and you know exactly what you are paying for,” Dr. Kosinski told this news organization. “In healthcare, we need something like that.”
Novel solutions like Dr. Kosinski’s will be increasingly necessary, as lawmakers on the state and federal level have begun to set their sights on tackling this issue.
The Biden administration in July expressed concern about an increased use of facility fees for healthcare provided at doctors’ offices, saying these additional costs often surprise consumers. House Energy and Commerce Chairwoman Cathy McMorris Rodgers (R-WA) also raised this issue several times this year, including at a May meeting about pending legislation on price transparency for health services, where she mentioned the case of a man who underwent eye surgery in Maine.
“His bill included three separate facility fees totaling $7800 and professional fees totaling $6200,” Ms. Rodgers said. “Why are three facility fees necessary for 1 hour of surgery in one O.R.?”
AGA’s Dr. Kosinski said facility fees cover the additional costs hospitals and clinics face in providing even routine treatments for some patients. For example, colonoscopy for a patient with a body mass index of 50 would pose special challenges for the anesthesiologist.
These factors need to be considered in setting policies on facility fees, he said. But there is no reason hospitals and other sites of medical care can’t make the information about facility fees easy for patients to find and understand, Dr. Kosinski said.
“I’m struggling to see a reason why we can’t be more transparent,” he said.
Big Battles Ahead
There are two connected battles ahead regarding facility fees: Efforts to restrict these additional charges for many medical services and fights over the need for greater transparency in general about health costs.
Senate Health, Education, Labor and Pensions Chairman Bernie Sanders (I-VT) is seeking to broadly restrict facility fees through his pending Primary Care and Health Workforce Act (S. 2840). The measure would block hospitals from charging health plans facility fees for many evaluation, management, and telehealth services.
The American Hospital Association (AHA) opposes it. They argue that the current payment approach rightly accounts for the added costs incurred when hospitals treat patients who are more likely to be ill or have chronic conditions than those seen in independent practices.
AHA said hospitals also need to maintain standby capacity for natural and man-made disasters, public health emergencies, and unexpected traumatic events. In September, AHA launched a television ad campaign to oppose any drive toward site-neutral policies. AHA says reducing the extra payments could cause more hospitals to shut their doors.
But there’s persistent interest in site-neutral payment, the term describing when the same reimbursement is given for care regardless of setting. This would lower pay for hospitals.
Among those pressing for change is an umbrella group of medical organizations known as the Alliance for Site Neutral Payment Reform. Its members include the American Academy of Family Physicians, American Academy of Orthopaedic Surgeons, American College of Physicians, Community Oncology Alliance, and Digestive Health Physicians Association.
And on November 9, Sen. Maggie Hassan (D-NH) argued for eventually including a site-neutral Medicare provision to a major healthcare package that the Senate Finance Committee is putting together.
Sen. Hassan is seeking to end what she called the “the practice of charging patients unfair hospital facility fees for care provided in the off-campus outpatient setting, like at a regular doctor’s office.”
Senate Finance Chairman Ron Wyden (D-OR) and the ranking Republican on the committee, Sen. Mike Crapo (R-ID), told Sen. Hassan they intended to work with her to see if this issue could be addressed in the pending legislative package.
A 2015 budget deal marked the last time Congress took a major step to address the higher cost of services provided in hospital-owned facilities.
Lawmakers then were scrambling to find cuts to offset spending in what became the 2015 Bipartisan Budget Act. This law established site-neutral payments under Medicare for services received at off-campus outpatient departments but exempted hospitals that already ran these kinds of operations or had advanced plans to create them.
Lawmakers are well aware of the potential savings from site-neutral policies and could look in time again to use them as part of a future budget deal.
In fact, in June, Sen. Hassan and Sens. Mike Braun (R-IN) and John Kennedy (R-LA) introduced a bill meant to basically end the exemption given in the 2015 deal to existing hospital outpatient departments, which has allowed higher Medicare payments. In a press release, Braun estimated that their proposed site-neutral change could save taxpayers $40 billion over a decade.
As Debate Continues, States Are Moving Ahead With Changes
Consumer activists have won a few battles this year at the state level about facility fees.
In July, Maine Gov. Janet Mills, a Democrat, signed a law that requires medical organizations to report facility fees to the state, which will share them publicly. Facility fees can pop up after a patient has received an insurance company estimate of the out-of-pocket costs for care.
“Patients receive bills bloated by healthcare providers that overcharge for services and insurance companies that deny claims without explanation,” the Portland Press Herald reported in a 2022 story. “And with little clout to fight back or even negotiate, feeling helpless, they often give up and pay, worn down by a system that is as time-consuming as it is obtuse.”
In May, Colorado enacted a law that will require patient notification about facility fees at many hospitals in the state.
In June, Connecticut expanded its law regarding facility fees and prohibited them for certain routine outpatient healthcare services. A statement from Gov. Ned Lamont’s office said the original intent of these facility fees was to ensure hospitals could maintain the around-the-clock care needed for inpatient and emergency care.
“However, these fees have been increasingly applied to services such as diagnostic testing and other routine services,” the statement said.
But there have been setbacks as well for those seeking to curb facilities.
The Texas Hospital Association (THA) in May said its advocacy defeated a pair of state bills, House bill 1692 and Senate bill 1275, that sought to limit facility fees for outpatient services.
In rallying opposition to these bills, THA said the loss of facility fees would threaten care for patients. Facility fees help cover costs “beyond the doctor’s bill,” such as “lab technicians, interpreters, medical records, security personnel, janitorial staff, and others,” THA said.
More Patients Shopping?
It’s unclear when — or if — Congress and other states will take major steps to reduce additional payments to hospitals for outpatient care.
But the increased use of high deductibles in health plans is driving more consumers to try to understand all of the costs of medical procedures ahead of time and, thus, drawing attention to facility fees, said Charlie Byrge, the chief operating officer of MDsave.
The average annual deductible levels for an individual increased by 3.0% to $2004 from 2020 to 2021 and for a family plan by 3.9% to $3868, according to a federal report. Some people have higher deductibles, exceeding $5000, Mr. Byrge said.
“That’s creating an opportunity for firms that can connect physicians directly with patients who will pay part or all of the costs of a treatment out of pocket,” he told this news organization.
Doctors and hospitals work with MDsave to charge preset prices for certain services, such as colonoscopies and mammograms. Consumers then can shop online to see if they can save. For example, in Nashville, Tennessee, where MDsave is based, the cost of a colonoscopy through MDsave is $2334, about half of the $4714 national average, according to the firm’s website.
This model for pricing routine medical care is akin to those used for other products and services, where companies decide ahead of time what to charge, he said.
“You don’t buy an airline ticket from Southwest or United or Delta and then there’s a bill after the fact because the price of gas went up a little bit on your flight,” Mr. Byrge said.
This will drive more competition among hospitals and clinics, in places where there are several sites of care in a region, Mr. Byrge said. But there are advantages for physicians and hospitals from the MDsave approach, he said.
“They know they’re getting paid upfront. They’re not going through the delays and headaches of the insurance reimbursement process. There are no denials. It’s just an upfront payment, and I think that’s what we’re starting to see the market really moving toward,” he said.
A version of this article appeared on Medscape.com.
Can the US healthcare system learn something about how to operate from car dealerships? Lawrence Kosinski, MD, MBA, a governing board member of American Gastroenterological Association (AGA), believes so.
There’s growing concern in the United States about the lack of clarity surrounding facility fees, which are intended to cover costs of maintaining medical facilities. Dr. Kosinski thinks that Congress should look into the transparency mandate it created for car prices as a model for how to address this.
A 1958 federal law set the stage for the consumer-friendly breakdown of costs and relevant performance data that anyone who has bought a new vehicle in the United States would recognize.
“You look at that and you know exactly what you are paying for,” Dr. Kosinski told this news organization. “In healthcare, we need something like that.”
Novel solutions like Dr. Kosinski’s will be increasingly necessary, as lawmakers on the state and federal level have begun to set their sights on tackling this issue.
The Biden administration in July expressed concern about an increased use of facility fees for healthcare provided at doctors’ offices, saying these additional costs often surprise consumers. House Energy and Commerce Chairwoman Cathy McMorris Rodgers (R-WA) also raised this issue several times this year, including at a May meeting about pending legislation on price transparency for health services, where she mentioned the case of a man who underwent eye surgery in Maine.
“His bill included three separate facility fees totaling $7800 and professional fees totaling $6200,” Ms. Rodgers said. “Why are three facility fees necessary for 1 hour of surgery in one O.R.?”
AGA’s Dr. Kosinski said facility fees cover the additional costs hospitals and clinics face in providing even routine treatments for some patients. For example, colonoscopy for a patient with a body mass index of 50 would pose special challenges for the anesthesiologist.
These factors need to be considered in setting policies on facility fees, he said. But there is no reason hospitals and other sites of medical care can’t make the information about facility fees easy for patients to find and understand, Dr. Kosinski said.
“I’m struggling to see a reason why we can’t be more transparent,” he said.
Big Battles Ahead
There are two connected battles ahead regarding facility fees: Efforts to restrict these additional charges for many medical services and fights over the need for greater transparency in general about health costs.
Senate Health, Education, Labor and Pensions Chairman Bernie Sanders (I-VT) is seeking to broadly restrict facility fees through his pending Primary Care and Health Workforce Act (S. 2840). The measure would block hospitals from charging health plans facility fees for many evaluation, management, and telehealth services.
The American Hospital Association (AHA) opposes it. They argue that the current payment approach rightly accounts for the added costs incurred when hospitals treat patients who are more likely to be ill or have chronic conditions than those seen in independent practices.
AHA said hospitals also need to maintain standby capacity for natural and man-made disasters, public health emergencies, and unexpected traumatic events. In September, AHA launched a television ad campaign to oppose any drive toward site-neutral policies. AHA says reducing the extra payments could cause more hospitals to shut their doors.
But there’s persistent interest in site-neutral payment, the term describing when the same reimbursement is given for care regardless of setting. This would lower pay for hospitals.
Among those pressing for change is an umbrella group of medical organizations known as the Alliance for Site Neutral Payment Reform. Its members include the American Academy of Family Physicians, American Academy of Orthopaedic Surgeons, American College of Physicians, Community Oncology Alliance, and Digestive Health Physicians Association.
And on November 9, Sen. Maggie Hassan (D-NH) argued for eventually including a site-neutral Medicare provision to a major healthcare package that the Senate Finance Committee is putting together.
Sen. Hassan is seeking to end what she called the “the practice of charging patients unfair hospital facility fees for care provided in the off-campus outpatient setting, like at a regular doctor’s office.”
Senate Finance Chairman Ron Wyden (D-OR) and the ranking Republican on the committee, Sen. Mike Crapo (R-ID), told Sen. Hassan they intended to work with her to see if this issue could be addressed in the pending legislative package.
A 2015 budget deal marked the last time Congress took a major step to address the higher cost of services provided in hospital-owned facilities.
Lawmakers then were scrambling to find cuts to offset spending in what became the 2015 Bipartisan Budget Act. This law established site-neutral payments under Medicare for services received at off-campus outpatient departments but exempted hospitals that already ran these kinds of operations or had advanced plans to create them.
Lawmakers are well aware of the potential savings from site-neutral policies and could look in time again to use them as part of a future budget deal.
In fact, in June, Sen. Hassan and Sens. Mike Braun (R-IN) and John Kennedy (R-LA) introduced a bill meant to basically end the exemption given in the 2015 deal to existing hospital outpatient departments, which has allowed higher Medicare payments. In a press release, Braun estimated that their proposed site-neutral change could save taxpayers $40 billion over a decade.
As Debate Continues, States Are Moving Ahead With Changes
Consumer activists have won a few battles this year at the state level about facility fees.
In July, Maine Gov. Janet Mills, a Democrat, signed a law that requires medical organizations to report facility fees to the state, which will share them publicly. Facility fees can pop up after a patient has received an insurance company estimate of the out-of-pocket costs for care.
“Patients receive bills bloated by healthcare providers that overcharge for services and insurance companies that deny claims without explanation,” the Portland Press Herald reported in a 2022 story. “And with little clout to fight back or even negotiate, feeling helpless, they often give up and pay, worn down by a system that is as time-consuming as it is obtuse.”
In May, Colorado enacted a law that will require patient notification about facility fees at many hospitals in the state.
In June, Connecticut expanded its law regarding facility fees and prohibited them for certain routine outpatient healthcare services. A statement from Gov. Ned Lamont’s office said the original intent of these facility fees was to ensure hospitals could maintain the around-the-clock care needed for inpatient and emergency care.
“However, these fees have been increasingly applied to services such as diagnostic testing and other routine services,” the statement said.
But there have been setbacks as well for those seeking to curb facilities.
The Texas Hospital Association (THA) in May said its advocacy defeated a pair of state bills, House bill 1692 and Senate bill 1275, that sought to limit facility fees for outpatient services.
In rallying opposition to these bills, THA said the loss of facility fees would threaten care for patients. Facility fees help cover costs “beyond the doctor’s bill,” such as “lab technicians, interpreters, medical records, security personnel, janitorial staff, and others,” THA said.
More Patients Shopping?
It’s unclear when — or if — Congress and other states will take major steps to reduce additional payments to hospitals for outpatient care.
But the increased use of high deductibles in health plans is driving more consumers to try to understand all of the costs of medical procedures ahead of time and, thus, drawing attention to facility fees, said Charlie Byrge, the chief operating officer of MDsave.
The average annual deductible levels for an individual increased by 3.0% to $2004 from 2020 to 2021 and for a family plan by 3.9% to $3868, according to a federal report. Some people have higher deductibles, exceeding $5000, Mr. Byrge said.
“That’s creating an opportunity for firms that can connect physicians directly with patients who will pay part or all of the costs of a treatment out of pocket,” he told this news organization.
Doctors and hospitals work with MDsave to charge preset prices for certain services, such as colonoscopies and mammograms. Consumers then can shop online to see if they can save. For example, in Nashville, Tennessee, where MDsave is based, the cost of a colonoscopy through MDsave is $2334, about half of the $4714 national average, according to the firm’s website.
This model for pricing routine medical care is akin to those used for other products and services, where companies decide ahead of time what to charge, he said.
“You don’t buy an airline ticket from Southwest or United or Delta and then there’s a bill after the fact because the price of gas went up a little bit on your flight,” Mr. Byrge said.
This will drive more competition among hospitals and clinics, in places where there are several sites of care in a region, Mr. Byrge said. But there are advantages for physicians and hospitals from the MDsave approach, he said.
“They know they’re getting paid upfront. They’re not going through the delays and headaches of the insurance reimbursement process. There are no denials. It’s just an upfront payment, and I think that’s what we’re starting to see the market really moving toward,” he said.
A version of this article appeared on Medscape.com.
Catheter-directed strategy improves pulmonary artery occlusion
Use of pharmacomechanical catheter-directory thrombolysis significantly reduced the number of pulmonary artery branches with total or subtotal occlusions in patients with acute pulmonary embolism, based on data from more than 100 individuals.
Reduced distal vascular volume is a significant predictor of 30-day and 90-day mortality in acute pulmonary embolism (PE) patients, and pulmonary obstruction is often the cause, wrote Riyaz Bashir, MD, of Temple University, Philadelphia, Pennsylvania, and colleagues.
, the researchers said.
“The recently published RESCUE (Recombinant tPA by Endovascular Administration for the Treatment of Submassive PE Using CDT for the Reduction of Thrombus Burden) trial showed a 35.9% reduction in PA obstruction using the Refined Modified Miller Index (RMMI), the largest reduction of all published catheter studies with core lab measurement, with similar doses of tissue plasminogen activator (tPA),” the researchers wrote.
The Bashir endovascular catheter was designed to maximize thrombus reduction via a pharmacomechanical infusion. The catheter features an expandable basket of 6 nitinol-reinforced infusion limbs.
“There are three crucial goals that we want to accomplish in patients who have a severe pulmonary embolism,” Dr. Bashir said in an interview. “Those include, in the order of importance, survival, recovery of right ventricular function, and resolution of blocked pulmonary arteries; both segmental and proximal pulmonary arteries,” he said.
Most previous studies have focused on the first two goals, but they still need to evaluate the resolution of PA blockages carefully, said Dr. Bashir. “In our clinical practice, we have seen a large number of patients who develop debilitating shortness of breath from these blockages. We decided to carefully evaluate these blockages before and after pharmacomechanical catheter-directed thrombolysis with the Bashir endovascular catheter using the core lab data from the RESCUE study,” he said.
In the current study published in JACC: Advances), the researchers used baseline and 48-hour posttreatment contrast-enhanced chest computed tomography angiography of adult PE patients with right ventricular dilatation.
The study population included 107 adults with acute intermediate-risk PE who were treated with pharmacomechanical catheter-directory thrombolysis (PM-CDT) at 18 sites in the United States. Of these, 98 had intermediate high-risk PE with elevated troponin and/or brain-type natriuretic peptide (BNP) levels and 102 had bilateral PE.
The primary endpoint was the change in the number of segmental and proximal PA branches with total or subtotal occlusions (defined as > 65%) after 48 hours compared to baseline. Occlusions were assessed using McNemar’s test.
Patients with bilateral PE received two Bashir catheters; those with unilateral PE received one catheter each.
Each patient received a pulse spray of 2 mg of recombinant tPA (r-tPA) into each lung, followed by 5 mg of r-tPA over 5 hours; the total dose was 7 mg of r-tPA for patients with unilateral PEs and 14 mg for those with bilateral PEs, the researchers said. The median times for catheter placement and total procedure were 15 minutes and 54 minutes, respectively.
The number of segmental PA branches with total or subtotal occlusions decreased significantly, from 40.5% at baseline to 11.7% at 48 hours, and proximal PA branch total or subtotal occlusions decreased significantly, from 28.7% at baseline to 11.0% at 48 hours (P < 0.0001 for both).
The magnitude of the reductions in both total and subtotal occlusions of segmental arteries was significantly correlated with the extent of right ventricle recovery (measured by the reduction in right ventricular/left ventricular ratio) with a correlation coefficient of 0.287 (P = .0026); however, this correlation was not observed in the proximal PA arteries (correlation coefficient 0.132, P = .173).
One major bleeding event occurred within 72 hours in a patient who also experienced a device-related left common iliac vein thrombosis while not taking anticoagulation medication, and one death unrelated to PE occurred within 30 days.
“The two findings that surprised me include, first, a more than 70% reduction in total and subtotal occlusions in the segmental arteries with such a low dose of r-tPA and, second, the resolution of the blockages was seen not only in the arteries where the device was placed but also at remote sites away from the location of the catheter,” Dr. Bashir told this news organization.
The findings were limited by several factors including the lack of long-term clinical follow-up outcomes data and lack of comparison groups who underwent other treatments.
However, “This study implies that we now have a safe therapy for these patients that improves survival and right ventricular recovery in addition to dramatically improving blocked pulmonary arteries,” Dr. Bashir said.
As for additional research, “we need all the current and future prospective pulmonary embolism studies to include an assessment of pulmonary artery blockage resolution as an essential endpoint,” he said.
Catheter Expands Treatment Options
The current study, a subgroup analysis of the RESCUE trial, was one of the first to examine the impact of catheter-directed lysis on distal occlusions, study coauthor Parth M. Rali, MD, said in an interview.
To this point, literature has been limited to evaluation for proximal disease, said Dr. Rali, director of thoracic surgery and medicine and part of the Pulmonary Embolism Response Team at Temple University Hospital, Philadelphia.
Dr. Rali said he was encouraged to see confirmation that the BEC catheter, because of its design, works in patients with proximal or distal occlusive disease.
In clinical practice, “the catheter provides an additional option for care in patients with multiple distal occlusive disease when a systemic tissue plasminogen activator (tPA), may put patient at high bleeding risk,” Dr. Rali said.
Looking ahead, a prospective, observational multicenter study would be useful to validate the findings from the post hoc analysis of the current study, he noted.
The study was sponsored by the National Heart, Lung, and Blood Institute, Commonwealth of Pennsylvania, and Thrombolex Inc., a medical device company developing interventional catheter-based therapies for the rapid and effective treatment of acute venous thromboembolic disorders. Dr. Bashir is a cofounder and has an equity interest in Thrombolex Inc. Dr. Rali disclosed serving as a consultant for Thrombolex, Inari Medical, Viz AI, and ThinkSono.
Use of pharmacomechanical catheter-directory thrombolysis significantly reduced the number of pulmonary artery branches with total or subtotal occlusions in patients with acute pulmonary embolism, based on data from more than 100 individuals.
Reduced distal vascular volume is a significant predictor of 30-day and 90-day mortality in acute pulmonary embolism (PE) patients, and pulmonary obstruction is often the cause, wrote Riyaz Bashir, MD, of Temple University, Philadelphia, Pennsylvania, and colleagues.
, the researchers said.
“The recently published RESCUE (Recombinant tPA by Endovascular Administration for the Treatment of Submassive PE Using CDT for the Reduction of Thrombus Burden) trial showed a 35.9% reduction in PA obstruction using the Refined Modified Miller Index (RMMI), the largest reduction of all published catheter studies with core lab measurement, with similar doses of tissue plasminogen activator (tPA),” the researchers wrote.
The Bashir endovascular catheter was designed to maximize thrombus reduction via a pharmacomechanical infusion. The catheter features an expandable basket of 6 nitinol-reinforced infusion limbs.
“There are three crucial goals that we want to accomplish in patients who have a severe pulmonary embolism,” Dr. Bashir said in an interview. “Those include, in the order of importance, survival, recovery of right ventricular function, and resolution of blocked pulmonary arteries; both segmental and proximal pulmonary arteries,” he said.
Most previous studies have focused on the first two goals, but they still need to evaluate the resolution of PA blockages carefully, said Dr. Bashir. “In our clinical practice, we have seen a large number of patients who develop debilitating shortness of breath from these blockages. We decided to carefully evaluate these blockages before and after pharmacomechanical catheter-directed thrombolysis with the Bashir endovascular catheter using the core lab data from the RESCUE study,” he said.
In the current study published in JACC: Advances), the researchers used baseline and 48-hour posttreatment contrast-enhanced chest computed tomography angiography of adult PE patients with right ventricular dilatation.
The study population included 107 adults with acute intermediate-risk PE who were treated with pharmacomechanical catheter-directory thrombolysis (PM-CDT) at 18 sites in the United States. Of these, 98 had intermediate high-risk PE with elevated troponin and/or brain-type natriuretic peptide (BNP) levels and 102 had bilateral PE.
The primary endpoint was the change in the number of segmental and proximal PA branches with total or subtotal occlusions (defined as > 65%) after 48 hours compared to baseline. Occlusions were assessed using McNemar’s test.
Patients with bilateral PE received two Bashir catheters; those with unilateral PE received one catheter each.
Each patient received a pulse spray of 2 mg of recombinant tPA (r-tPA) into each lung, followed by 5 mg of r-tPA over 5 hours; the total dose was 7 mg of r-tPA for patients with unilateral PEs and 14 mg for those with bilateral PEs, the researchers said. The median times for catheter placement and total procedure were 15 minutes and 54 minutes, respectively.
The number of segmental PA branches with total or subtotal occlusions decreased significantly, from 40.5% at baseline to 11.7% at 48 hours, and proximal PA branch total or subtotal occlusions decreased significantly, from 28.7% at baseline to 11.0% at 48 hours (P < 0.0001 for both).
The magnitude of the reductions in both total and subtotal occlusions of segmental arteries was significantly correlated with the extent of right ventricle recovery (measured by the reduction in right ventricular/left ventricular ratio) with a correlation coefficient of 0.287 (P = .0026); however, this correlation was not observed in the proximal PA arteries (correlation coefficient 0.132, P = .173).
One major bleeding event occurred within 72 hours in a patient who also experienced a device-related left common iliac vein thrombosis while not taking anticoagulation medication, and one death unrelated to PE occurred within 30 days.
“The two findings that surprised me include, first, a more than 70% reduction in total and subtotal occlusions in the segmental arteries with such a low dose of r-tPA and, second, the resolution of the blockages was seen not only in the arteries where the device was placed but also at remote sites away from the location of the catheter,” Dr. Bashir told this news organization.
The findings were limited by several factors including the lack of long-term clinical follow-up outcomes data and lack of comparison groups who underwent other treatments.
However, “This study implies that we now have a safe therapy for these patients that improves survival and right ventricular recovery in addition to dramatically improving blocked pulmonary arteries,” Dr. Bashir said.
As for additional research, “we need all the current and future prospective pulmonary embolism studies to include an assessment of pulmonary artery blockage resolution as an essential endpoint,” he said.
Catheter Expands Treatment Options
The current study, a subgroup analysis of the RESCUE trial, was one of the first to examine the impact of catheter-directed lysis on distal occlusions, study coauthor Parth M. Rali, MD, said in an interview.
To this point, literature has been limited to evaluation for proximal disease, said Dr. Rali, director of thoracic surgery and medicine and part of the Pulmonary Embolism Response Team at Temple University Hospital, Philadelphia.
Dr. Rali said he was encouraged to see confirmation that the BEC catheter, because of its design, works in patients with proximal or distal occlusive disease.
In clinical practice, “the catheter provides an additional option for care in patients with multiple distal occlusive disease when a systemic tissue plasminogen activator (tPA), may put patient at high bleeding risk,” Dr. Rali said.
Looking ahead, a prospective, observational multicenter study would be useful to validate the findings from the post hoc analysis of the current study, he noted.
The study was sponsored by the National Heart, Lung, and Blood Institute, Commonwealth of Pennsylvania, and Thrombolex Inc., a medical device company developing interventional catheter-based therapies for the rapid and effective treatment of acute venous thromboembolic disorders. Dr. Bashir is a cofounder and has an equity interest in Thrombolex Inc. Dr. Rali disclosed serving as a consultant for Thrombolex, Inari Medical, Viz AI, and ThinkSono.
Use of pharmacomechanical catheter-directory thrombolysis significantly reduced the number of pulmonary artery branches with total or subtotal occlusions in patients with acute pulmonary embolism, based on data from more than 100 individuals.
Reduced distal vascular volume is a significant predictor of 30-day and 90-day mortality in acute pulmonary embolism (PE) patients, and pulmonary obstruction is often the cause, wrote Riyaz Bashir, MD, of Temple University, Philadelphia, Pennsylvania, and colleagues.
, the researchers said.
“The recently published RESCUE (Recombinant tPA by Endovascular Administration for the Treatment of Submassive PE Using CDT for the Reduction of Thrombus Burden) trial showed a 35.9% reduction in PA obstruction using the Refined Modified Miller Index (RMMI), the largest reduction of all published catheter studies with core lab measurement, with similar doses of tissue plasminogen activator (tPA),” the researchers wrote.
The Bashir endovascular catheter was designed to maximize thrombus reduction via a pharmacomechanical infusion. The catheter features an expandable basket of 6 nitinol-reinforced infusion limbs.
“There are three crucial goals that we want to accomplish in patients who have a severe pulmonary embolism,” Dr. Bashir said in an interview. “Those include, in the order of importance, survival, recovery of right ventricular function, and resolution of blocked pulmonary arteries; both segmental and proximal pulmonary arteries,” he said.
Most previous studies have focused on the first two goals, but they still need to evaluate the resolution of PA blockages carefully, said Dr. Bashir. “In our clinical practice, we have seen a large number of patients who develop debilitating shortness of breath from these blockages. We decided to carefully evaluate these blockages before and after pharmacomechanical catheter-directed thrombolysis with the Bashir endovascular catheter using the core lab data from the RESCUE study,” he said.
In the current study published in JACC: Advances), the researchers used baseline and 48-hour posttreatment contrast-enhanced chest computed tomography angiography of adult PE patients with right ventricular dilatation.
The study population included 107 adults with acute intermediate-risk PE who were treated with pharmacomechanical catheter-directory thrombolysis (PM-CDT) at 18 sites in the United States. Of these, 98 had intermediate high-risk PE with elevated troponin and/or brain-type natriuretic peptide (BNP) levels and 102 had bilateral PE.
The primary endpoint was the change in the number of segmental and proximal PA branches with total or subtotal occlusions (defined as > 65%) after 48 hours compared to baseline. Occlusions were assessed using McNemar’s test.
Patients with bilateral PE received two Bashir catheters; those with unilateral PE received one catheter each.
Each patient received a pulse spray of 2 mg of recombinant tPA (r-tPA) into each lung, followed by 5 mg of r-tPA over 5 hours; the total dose was 7 mg of r-tPA for patients with unilateral PEs and 14 mg for those with bilateral PEs, the researchers said. The median times for catheter placement and total procedure were 15 minutes and 54 minutes, respectively.
The number of segmental PA branches with total or subtotal occlusions decreased significantly, from 40.5% at baseline to 11.7% at 48 hours, and proximal PA branch total or subtotal occlusions decreased significantly, from 28.7% at baseline to 11.0% at 48 hours (P < 0.0001 for both).
The magnitude of the reductions in both total and subtotal occlusions of segmental arteries was significantly correlated with the extent of right ventricle recovery (measured by the reduction in right ventricular/left ventricular ratio) with a correlation coefficient of 0.287 (P = .0026); however, this correlation was not observed in the proximal PA arteries (correlation coefficient 0.132, P = .173).
One major bleeding event occurred within 72 hours in a patient who also experienced a device-related left common iliac vein thrombosis while not taking anticoagulation medication, and one death unrelated to PE occurred within 30 days.
“The two findings that surprised me include, first, a more than 70% reduction in total and subtotal occlusions in the segmental arteries with such a low dose of r-tPA and, second, the resolution of the blockages was seen not only in the arteries where the device was placed but also at remote sites away from the location of the catheter,” Dr. Bashir told this news organization.
The findings were limited by several factors including the lack of long-term clinical follow-up outcomes data and lack of comparison groups who underwent other treatments.
However, “This study implies that we now have a safe therapy for these patients that improves survival and right ventricular recovery in addition to dramatically improving blocked pulmonary arteries,” Dr. Bashir said.
As for additional research, “we need all the current and future prospective pulmonary embolism studies to include an assessment of pulmonary artery blockage resolution as an essential endpoint,” he said.
Catheter Expands Treatment Options
The current study, a subgroup analysis of the RESCUE trial, was one of the first to examine the impact of catheter-directed lysis on distal occlusions, study coauthor Parth M. Rali, MD, said in an interview.
To this point, literature has been limited to evaluation for proximal disease, said Dr. Rali, director of thoracic surgery and medicine and part of the Pulmonary Embolism Response Team at Temple University Hospital, Philadelphia.
Dr. Rali said he was encouraged to see confirmation that the BEC catheter, because of its design, works in patients with proximal or distal occlusive disease.
In clinical practice, “the catheter provides an additional option for care in patients with multiple distal occlusive disease when a systemic tissue plasminogen activator (tPA), may put patient at high bleeding risk,” Dr. Rali said.
Looking ahead, a prospective, observational multicenter study would be useful to validate the findings from the post hoc analysis of the current study, he noted.
The study was sponsored by the National Heart, Lung, and Blood Institute, Commonwealth of Pennsylvania, and Thrombolex Inc., a medical device company developing interventional catheter-based therapies for the rapid and effective treatment of acute venous thromboembolic disorders. Dr. Bashir is a cofounder and has an equity interest in Thrombolex Inc. Dr. Rali disclosed serving as a consultant for Thrombolex, Inari Medical, Viz AI, and ThinkSono.
FROM JACC: ADVANCES






