User login
Higher stroke rates seen among patients with COVID-19 compared with influenza
, according to a retrospective cohort study conducted at New York–Presbyterian Hospital and Weill Cornell Medicine, New York. “These findings suggest that clinicians should be vigilant for symptoms and signs of acute ischemic stroke in patients with COVID-19 so that time-sensitive interventions, such as thrombolysis and thrombectomy, can be instituted if possible to reduce the burden of long-term disability,” wrote Alexander E. Merkler and colleagues. Their report is in JAMA Neurology.
While several recent publications have “raised the possibility” of this link, none have had an appropriate control group, noted Dr. Merkler of the department of neurology, Weill Cornell Medicine. “Further elucidation of thrombotic mechanisms in patients with COVID-19 may yield better strategies to prevent disabling thrombotic complications like ischemic stroke,” he added.
An increased risk of stroke
The study included 1,916 adults with confirmed COVID-19 (median age 64 years) who were either hospitalized or visited an emergency department between March 4 and May 2, 2020. These cases were compared with a historical cohort of 1,486 patients (median age 62 years) who were hospitalized with laboratory-confirmed influenza A or B between January 1, 2016, and May 31, 2018.
Among the patients with COVID-19, a diagnosis of cerebrovascular disease during hospitalization, a brain computed tomography (CT), or brain magnetic resonance imaging (MRI) was an indication of possible ischemic stroke. These records were then independently reviewed by two board-certified attending neurologists (with a third resolving any disagreement) to adjudicate a final stroke diagnosis. In the influenza cohort, the Cornell Acute Stroke Academic Registry (CAESAR) was used to ascertain ischemic strokes.
The study identified 31 patients with stroke among the COVID-19 cohort (1.6%; 95% confidence interval, 1.1%-2.3%) and 3 in the influenza cohort (0.2%; 95% CI, 0.0%-0.6%). After adjustment for age, sex, and race, stroke risk was almost 8 times higher in the COVID-19 cohort (OR, 7.6; 95% CI, 2.3-25.2).
This association “persisted across multiple sensitivity analyses, with the magnitude of relative associations ranging from 4.0 to 9,” wrote the authors. “This included a sensitivity analysis that adjusted for the number of vascular risk factors and ICU admissions (OR, 4.6; 95% CI, 1.4-15.7).”
The median age of patients with COVID-19 and stroke was 69 years, and the median duration of COVID-19 symptom onset to stroke diagnosis was 16 days. Stroke symptoms were the presenting complaint in only 26% of the patients, while the remainder developing stroke while hospitalized, and more than a third (35%) of all strokes occurred in patients who were mechanically ventilated with severe COVID-19. Inpatient mortality was considerably higher among patients with COVID-19 with stroke versus without (32% vs. 14%; P = .003).
In patients with COVID-19 “most ischemic strokes occurred in older age groups, those with traditional stroke risk factors, and people of color,” wrote the authors. “We also noted that initial plasma D-dimer levels were nearly 3-fold higher in those who received a diagnosis of ischemic stroke than in those who did not” (1.930 mcg/mL vs. 0.682 mcg/mL).
The authors suggested several possible explanations for the elevated risk of stroke in COVID-19. Acute viral illnesses are known to trigger inflammation, and COVID-19 in particular is associated with “a vigorous inflammatory response accompanied by coagulopathy, with elevated D-dimer levels and the frequent presence of antiphospholipid antibodies,” they wrote. The infection is also associated with more severe respiratory syndrome compared with influenza, as well as a heightened risk for complications such as atrial arrhythmias, myocardial infarction, heart failure, myocarditis, and venous thromboses, all of which likely contribute to the risk of ischemic stroke.”
COVID or conventional risk factors?
Asked to comment on the study, Benedict Michael, MBChB (Hons), MRCP (Neurol), PhD, from the United Kingdom’s Coronerve Studies Group, a collaborative initiative to study the neurological features of COVID-19, said in an interview that “this study suggests many cases of stroke are occurring in older patients with multiple existing conventional and well recognized risks for stroke, and may simply represent decompensation during sepsis.”
Dr. Michael, a senior clinician scientist fellow at the University of Liverpool and an honorary consultant neurologist at the Walton Centre, was the senior author on a recently published UK-wide surveillance study on the neurological and neuropsychiatric complications of COVID-19 (Lancet Psychiatry. 2020 Jun 25. doi: 10.1016/S2215-0366[20]30287-X).
He said among patients in the New York study, “those with COVID and a stroke appeared to have many conventional risk factors for stroke (and often at higher percentages than COVID patients without a stroke), e.g. hypertension, overweight, diabetes, hyperlipidemia, existing vascular disease affecting the coronary arteries and atrial fibrillation. To establish evidence-based treatment pathways, clearly further studies are needed to determine the biological mechanisms underlying the seemingly higher rate of stroke with COVID-19 than influenza; but this must especially focus on those younger patients without conventional risk factors for stroke (which are largely not included in this study).”
SOURCE: Merkler AE et al. JAMA Neurol. doi: 10.1001/jamaneurol.2020.2730.
, according to a retrospective cohort study conducted at New York–Presbyterian Hospital and Weill Cornell Medicine, New York. “These findings suggest that clinicians should be vigilant for symptoms and signs of acute ischemic stroke in patients with COVID-19 so that time-sensitive interventions, such as thrombolysis and thrombectomy, can be instituted if possible to reduce the burden of long-term disability,” wrote Alexander E. Merkler and colleagues. Their report is in JAMA Neurology.
While several recent publications have “raised the possibility” of this link, none have had an appropriate control group, noted Dr. Merkler of the department of neurology, Weill Cornell Medicine. “Further elucidation of thrombotic mechanisms in patients with COVID-19 may yield better strategies to prevent disabling thrombotic complications like ischemic stroke,” he added.
An increased risk of stroke
The study included 1,916 adults with confirmed COVID-19 (median age 64 years) who were either hospitalized or visited an emergency department between March 4 and May 2, 2020. These cases were compared with a historical cohort of 1,486 patients (median age 62 years) who were hospitalized with laboratory-confirmed influenza A or B between January 1, 2016, and May 31, 2018.
Among the patients with COVID-19, a diagnosis of cerebrovascular disease during hospitalization, a brain computed tomography (CT), or brain magnetic resonance imaging (MRI) was an indication of possible ischemic stroke. These records were then independently reviewed by two board-certified attending neurologists (with a third resolving any disagreement) to adjudicate a final stroke diagnosis. In the influenza cohort, the Cornell Acute Stroke Academic Registry (CAESAR) was used to ascertain ischemic strokes.
The study identified 31 patients with stroke among the COVID-19 cohort (1.6%; 95% confidence interval, 1.1%-2.3%) and 3 in the influenza cohort (0.2%; 95% CI, 0.0%-0.6%). After adjustment for age, sex, and race, stroke risk was almost 8 times higher in the COVID-19 cohort (OR, 7.6; 95% CI, 2.3-25.2).
This association “persisted across multiple sensitivity analyses, with the magnitude of relative associations ranging from 4.0 to 9,” wrote the authors. “This included a sensitivity analysis that adjusted for the number of vascular risk factors and ICU admissions (OR, 4.6; 95% CI, 1.4-15.7).”
The median age of patients with COVID-19 and stroke was 69 years, and the median duration of COVID-19 symptom onset to stroke diagnosis was 16 days. Stroke symptoms were the presenting complaint in only 26% of the patients, while the remainder developing stroke while hospitalized, and more than a third (35%) of all strokes occurred in patients who were mechanically ventilated with severe COVID-19. Inpatient mortality was considerably higher among patients with COVID-19 with stroke versus without (32% vs. 14%; P = .003).
In patients with COVID-19 “most ischemic strokes occurred in older age groups, those with traditional stroke risk factors, and people of color,” wrote the authors. “We also noted that initial plasma D-dimer levels were nearly 3-fold higher in those who received a diagnosis of ischemic stroke than in those who did not” (1.930 mcg/mL vs. 0.682 mcg/mL).
The authors suggested several possible explanations for the elevated risk of stroke in COVID-19. Acute viral illnesses are known to trigger inflammation, and COVID-19 in particular is associated with “a vigorous inflammatory response accompanied by coagulopathy, with elevated D-dimer levels and the frequent presence of antiphospholipid antibodies,” they wrote. The infection is also associated with more severe respiratory syndrome compared with influenza, as well as a heightened risk for complications such as atrial arrhythmias, myocardial infarction, heart failure, myocarditis, and venous thromboses, all of which likely contribute to the risk of ischemic stroke.”
COVID or conventional risk factors?
Asked to comment on the study, Benedict Michael, MBChB (Hons), MRCP (Neurol), PhD, from the United Kingdom’s Coronerve Studies Group, a collaborative initiative to study the neurological features of COVID-19, said in an interview that “this study suggests many cases of stroke are occurring in older patients with multiple existing conventional and well recognized risks for stroke, and may simply represent decompensation during sepsis.”
Dr. Michael, a senior clinician scientist fellow at the University of Liverpool and an honorary consultant neurologist at the Walton Centre, was the senior author on a recently published UK-wide surveillance study on the neurological and neuropsychiatric complications of COVID-19 (Lancet Psychiatry. 2020 Jun 25. doi: 10.1016/S2215-0366[20]30287-X).
He said among patients in the New York study, “those with COVID and a stroke appeared to have many conventional risk factors for stroke (and often at higher percentages than COVID patients without a stroke), e.g. hypertension, overweight, diabetes, hyperlipidemia, existing vascular disease affecting the coronary arteries and atrial fibrillation. To establish evidence-based treatment pathways, clearly further studies are needed to determine the biological mechanisms underlying the seemingly higher rate of stroke with COVID-19 than influenza; but this must especially focus on those younger patients without conventional risk factors for stroke (which are largely not included in this study).”
SOURCE: Merkler AE et al. JAMA Neurol. doi: 10.1001/jamaneurol.2020.2730.
, according to a retrospective cohort study conducted at New York–Presbyterian Hospital and Weill Cornell Medicine, New York. “These findings suggest that clinicians should be vigilant for symptoms and signs of acute ischemic stroke in patients with COVID-19 so that time-sensitive interventions, such as thrombolysis and thrombectomy, can be instituted if possible to reduce the burden of long-term disability,” wrote Alexander E. Merkler and colleagues. Their report is in JAMA Neurology.
While several recent publications have “raised the possibility” of this link, none have had an appropriate control group, noted Dr. Merkler of the department of neurology, Weill Cornell Medicine. “Further elucidation of thrombotic mechanisms in patients with COVID-19 may yield better strategies to prevent disabling thrombotic complications like ischemic stroke,” he added.
An increased risk of stroke
The study included 1,916 adults with confirmed COVID-19 (median age 64 years) who were either hospitalized or visited an emergency department between March 4 and May 2, 2020. These cases were compared with a historical cohort of 1,486 patients (median age 62 years) who were hospitalized with laboratory-confirmed influenza A or B between January 1, 2016, and May 31, 2018.
Among the patients with COVID-19, a diagnosis of cerebrovascular disease during hospitalization, a brain computed tomography (CT), or brain magnetic resonance imaging (MRI) was an indication of possible ischemic stroke. These records were then independently reviewed by two board-certified attending neurologists (with a third resolving any disagreement) to adjudicate a final stroke diagnosis. In the influenza cohort, the Cornell Acute Stroke Academic Registry (CAESAR) was used to ascertain ischemic strokes.
The study identified 31 patients with stroke among the COVID-19 cohort (1.6%; 95% confidence interval, 1.1%-2.3%) and 3 in the influenza cohort (0.2%; 95% CI, 0.0%-0.6%). After adjustment for age, sex, and race, stroke risk was almost 8 times higher in the COVID-19 cohort (OR, 7.6; 95% CI, 2.3-25.2).
This association “persisted across multiple sensitivity analyses, with the magnitude of relative associations ranging from 4.0 to 9,” wrote the authors. “This included a sensitivity analysis that adjusted for the number of vascular risk factors and ICU admissions (OR, 4.6; 95% CI, 1.4-15.7).”
The median age of patients with COVID-19 and stroke was 69 years, and the median duration of COVID-19 symptom onset to stroke diagnosis was 16 days. Stroke symptoms were the presenting complaint in only 26% of the patients, while the remainder developing stroke while hospitalized, and more than a third (35%) of all strokes occurred in patients who were mechanically ventilated with severe COVID-19. Inpatient mortality was considerably higher among patients with COVID-19 with stroke versus without (32% vs. 14%; P = .003).
In patients with COVID-19 “most ischemic strokes occurred in older age groups, those with traditional stroke risk factors, and people of color,” wrote the authors. “We also noted that initial plasma D-dimer levels were nearly 3-fold higher in those who received a diagnosis of ischemic stroke than in those who did not” (1.930 mcg/mL vs. 0.682 mcg/mL).
The authors suggested several possible explanations for the elevated risk of stroke in COVID-19. Acute viral illnesses are known to trigger inflammation, and COVID-19 in particular is associated with “a vigorous inflammatory response accompanied by coagulopathy, with elevated D-dimer levels and the frequent presence of antiphospholipid antibodies,” they wrote. The infection is also associated with more severe respiratory syndrome compared with influenza, as well as a heightened risk for complications such as atrial arrhythmias, myocardial infarction, heart failure, myocarditis, and venous thromboses, all of which likely contribute to the risk of ischemic stroke.”
COVID or conventional risk factors?
Asked to comment on the study, Benedict Michael, MBChB (Hons), MRCP (Neurol), PhD, from the United Kingdom’s Coronerve Studies Group, a collaborative initiative to study the neurological features of COVID-19, said in an interview that “this study suggests many cases of stroke are occurring in older patients with multiple existing conventional and well recognized risks for stroke, and may simply represent decompensation during sepsis.”
Dr. Michael, a senior clinician scientist fellow at the University of Liverpool and an honorary consultant neurologist at the Walton Centre, was the senior author on a recently published UK-wide surveillance study on the neurological and neuropsychiatric complications of COVID-19 (Lancet Psychiatry. 2020 Jun 25. doi: 10.1016/S2215-0366[20]30287-X).
He said among patients in the New York study, “those with COVID and a stroke appeared to have many conventional risk factors for stroke (and often at higher percentages than COVID patients without a stroke), e.g. hypertension, overweight, diabetes, hyperlipidemia, existing vascular disease affecting the coronary arteries and atrial fibrillation. To establish evidence-based treatment pathways, clearly further studies are needed to determine the biological mechanisms underlying the seemingly higher rate of stroke with COVID-19 than influenza; but this must especially focus on those younger patients without conventional risk factors for stroke (which are largely not included in this study).”
SOURCE: Merkler AE et al. JAMA Neurol. doi: 10.1001/jamaneurol.2020.2730.
FROM JAMA NEUROLOGY
Anticoagulation in cirrhosis: Best practices
Background: Alterations to the coagulation cascade put cirrhotic patients at higher risk for bleeding and thrombotic complications.
Study design: Expert review.
Setting: Literature review.
Synopsis: The authors provide 12 best practice recommendations, including use blood products sparingly in the absence of active bleeding out of concern for raising portal pressures; low-risk paracentesis, thoracentesis, and upper endoscopy do not require routine correction of thrombocytopenia or coagulopathy; for active bleeding or high-risk procedures, correct hematocrit to above 25%, platelets to more than 50,000, and fibrinogen to above 120 mg/dL; the risk of thrombosis, including venous thromboembolism and portal vein thrombosis, is high in these patients despite elevated INR values.
As such, pharmacologic VTE prophylaxis is often underutilized in patients admitted with cirrhosis; for patients requiring therapeutic anticoagulation, direct oral anticoagulants are safe in stable patients with mild cirrhosis, but should be avoided in Child-Pugh B and C patients.
Bottom line: Cirrhotic patients do not require routine correction of coagulopathy prior to low-risk procedures.
Citation: O’Leary JG et al. AGA Clinical Practice Update: Coagulation in cirrhosis. Gastroenterology. 2019. doi: 10.1053/j.gastro.2019.03.070.
Dr. Lublin is a hospitalist at the University of Colorado at Denver, Aurora.
Background: Alterations to the coagulation cascade put cirrhotic patients at higher risk for bleeding and thrombotic complications.
Study design: Expert review.
Setting: Literature review.
Synopsis: The authors provide 12 best practice recommendations, including use blood products sparingly in the absence of active bleeding out of concern for raising portal pressures; low-risk paracentesis, thoracentesis, and upper endoscopy do not require routine correction of thrombocytopenia or coagulopathy; for active bleeding or high-risk procedures, correct hematocrit to above 25%, platelets to more than 50,000, and fibrinogen to above 120 mg/dL; the risk of thrombosis, including venous thromboembolism and portal vein thrombosis, is high in these patients despite elevated INR values.
As such, pharmacologic VTE prophylaxis is often underutilized in patients admitted with cirrhosis; for patients requiring therapeutic anticoagulation, direct oral anticoagulants are safe in stable patients with mild cirrhosis, but should be avoided in Child-Pugh B and C patients.
Bottom line: Cirrhotic patients do not require routine correction of coagulopathy prior to low-risk procedures.
Citation: O’Leary JG et al. AGA Clinical Practice Update: Coagulation in cirrhosis. Gastroenterology. 2019. doi: 10.1053/j.gastro.2019.03.070.
Dr. Lublin is a hospitalist at the University of Colorado at Denver, Aurora.
Background: Alterations to the coagulation cascade put cirrhotic patients at higher risk for bleeding and thrombotic complications.
Study design: Expert review.
Setting: Literature review.
Synopsis: The authors provide 12 best practice recommendations, including use blood products sparingly in the absence of active bleeding out of concern for raising portal pressures; low-risk paracentesis, thoracentesis, and upper endoscopy do not require routine correction of thrombocytopenia or coagulopathy; for active bleeding or high-risk procedures, correct hematocrit to above 25%, platelets to more than 50,000, and fibrinogen to above 120 mg/dL; the risk of thrombosis, including venous thromboembolism and portal vein thrombosis, is high in these patients despite elevated INR values.
As such, pharmacologic VTE prophylaxis is often underutilized in patients admitted with cirrhosis; for patients requiring therapeutic anticoagulation, direct oral anticoagulants are safe in stable patients with mild cirrhosis, but should be avoided in Child-Pugh B and C patients.
Bottom line: Cirrhotic patients do not require routine correction of coagulopathy prior to low-risk procedures.
Citation: O’Leary JG et al. AGA Clinical Practice Update: Coagulation in cirrhosis. Gastroenterology. 2019. doi: 10.1053/j.gastro.2019.03.070.
Dr. Lublin is a hospitalist at the University of Colorado at Denver, Aurora.
Inhaled treprostinil improves walk distance in patients with ILD-associated pulmonary hypertension
over 16 weeks, compared with patients who used a placebo inhaler, results of a phase 3 trial showed.
Among 326 patients with pulmonary hypertension (PH) associated with interstitial lung disease (ILD), those who were randomly assigned to treatment with treprostinil had a placebo-corrected median difference from baseline in 6-minute walk distance of 21 m (P = .004), reported Steven D. Nathan, MD, from Inova Fairfax Hospital in Falls Church, Va., on behalf of coinvestigators in the INCREASE study (NCT02630316).
“These results support an additional treatment avenue, and might herald a shift in the clinical management of patients with interstitial lung disease,” he said in the American Thoracic Society’s virtual clinical trial session.
“This was an outstanding presentation and outstanding results. I personally am very excited, because this is a field where I work,” commented Martin Kolb, MD, PhD, from McMaster University, Hamilton, Ont., the facilitator for the online presentation.
The INCREASE trial compared inhaled treprostinil dose four times daily with placebo in patients with a CT scan–confirmed diagnosis of World Health Organization group 3 PH within 6 months before randomization who had evidence of diffuse parenchymal lung disease. Eligible patients could have any form of ILD or combined pulmonary fibrosis and emphysema.
Key inclusion criteria included right-heart catheterization within the previous year with documented pulmonary vascular resistance greater than 3 Wood units, pulmonary capillary wedge pressure 15 mm Hg or less, and mean pulmonary arterial pressure 25 mm Hg or higher.
Patients also had to have a 6-minute walk distance of at least 100 m and have stable disease while on an optimized dose of medications for underlying lung disease. Patients with group 3 connective tissue disease had to have baseline forced vital capacity of less than 70%.
The final study cohorts included patients with idiopathic interstitial pneumonias, chronic hypersensitivity pneumonitis, connective tissue disease, combined pulmonary fibrosis and emphysema, and occupational lung disease.
The patients were randomized to receive either inhaled treprostinil at a starting dose of 6 mcg/breath four times daily or to placebo (163 patients in each arm). All patients started the study drug at a dose of three breaths four times daily during waking hours. Dose escalations – adding 1 additional breath four times daily – were allowed every 3 days, up to a target dose of 9 breaths (54 mcg) four times daily, and a maximum of 12 breaths (72 mcg) four times daily as clinically tolerated.
A total of 130 patients assigned to treprostinil and 128 assigned to placebo completed 16 weeks of therapy and assessment.
As noted before, patients assigned to treprostinil had a placebo-corrected median difference from baseline in peak 6-minute walk distance, as measured by Hodges-Lehmann estimation, of 21 m (P = .004). An analysis of the same parameter using mixed model repeated measurement showed a placebo-corrected difference from baseline in peak 6-minute walk distance of 31.12 m (P < .001).
Secondary endpoints that were significantly better with treprostinil, compared with placebo, included improvements in N-terminal of the prohormone brain natriuretic peptide, a longer time to clinical worsening, and improvements in peak 6-minute walk distance week 12, and trough 6-minute walk distance at week 15.
Treprostinil was associated with a 39% reduction in risk of clinical worsening (P = .04). In all, 37 patients on treprostinil (22.7%) and 54 on placebo (33.1%) experienced clinical worsening.
For the exploratory endpoints of change in patient reported quality of life as measured by the St. George’s Respiratory Questionnaire, or in peak distance saturation product, however, there were no significant differences between the groups.
In addition, treprostinil was associated with a 34% reduction the risk of exacerbation of underlying lung disease, compared with placebo (P = .03).
The safety profile of treprostinil was similar to that seen in other studies of the drug, and most treatment-related adverse events were mild or moderate in severity. Adverse events led to discontinuation in 10% of patients on treprostinil and 8% on placebo.
Serious adverse events were seen in 23.3% and 25.8%, respectively. The most frequently occurring adverse events of any grade included cough, headache, dyspnea, dizziness, nausea, fatigue, diarrhea, throat irritation, and oropharyngeal pain.
There was no evidence of worsened oxygenation or lung function “allaying V/Q mismatch concerns,” Dr. Nathan said, and there was evidence for an improvement in forced vital capacity with treprostinil.
In the question-and-answer portion of the presentation, Dr. Kolb commented that many clinicians, particularly those who treated patients with ILD, question whether a 21-m difference in walk distance makes much of a difference in patient lives. He relayed a question from a viewer asking how Dr. Nathan and associates reconciled their primary endpoint with the finding that there was no difference in patient-reported quality of life.
“I think that the difference in the 6-minute walk test was both statistically significant and clinically meaningful,” Dr. Nathan replied.
He noted that the primary endpoint used a stringent measure, and that less conservative methods of analysis showed a larger difference in benefit favoring treprostinil. He also pointed out that the original study of inhaled treprostinil added to oral therapy for pulmonary arterial hypertension showed a 20-m improvement in walk distance, and that these results were sufficient to get the inhaled formulation approved in the United States (J Am Coll Cardiol. 2010 May. doi: 10.1016/j.jacc.2010.01.027).
Regarding the failure to detect a difference in quality of life, he said that the study was only 16 weeks in length, and that the St. George’s Respiratory Questionnaire was developed for evaluation of patients with chronic obstructive pulmonary disease, “perhaps not the best instrument to use in an ILD PH study.”
The study was funded by United Therapeutics. Dr. Nathan disclosed advisory committee activity/consulting, research support, and speaker fees from the company. Dr. Kolb has previously disclosed financial relationships with various companies, not including United Therapeutics.
over 16 weeks, compared with patients who used a placebo inhaler, results of a phase 3 trial showed.
Among 326 patients with pulmonary hypertension (PH) associated with interstitial lung disease (ILD), those who were randomly assigned to treatment with treprostinil had a placebo-corrected median difference from baseline in 6-minute walk distance of 21 m (P = .004), reported Steven D. Nathan, MD, from Inova Fairfax Hospital in Falls Church, Va., on behalf of coinvestigators in the INCREASE study (NCT02630316).
“These results support an additional treatment avenue, and might herald a shift in the clinical management of patients with interstitial lung disease,” he said in the American Thoracic Society’s virtual clinical trial session.
“This was an outstanding presentation and outstanding results. I personally am very excited, because this is a field where I work,” commented Martin Kolb, MD, PhD, from McMaster University, Hamilton, Ont., the facilitator for the online presentation.
The INCREASE trial compared inhaled treprostinil dose four times daily with placebo in patients with a CT scan–confirmed diagnosis of World Health Organization group 3 PH within 6 months before randomization who had evidence of diffuse parenchymal lung disease. Eligible patients could have any form of ILD or combined pulmonary fibrosis and emphysema.
Key inclusion criteria included right-heart catheterization within the previous year with documented pulmonary vascular resistance greater than 3 Wood units, pulmonary capillary wedge pressure 15 mm Hg or less, and mean pulmonary arterial pressure 25 mm Hg or higher.
Patients also had to have a 6-minute walk distance of at least 100 m and have stable disease while on an optimized dose of medications for underlying lung disease. Patients with group 3 connective tissue disease had to have baseline forced vital capacity of less than 70%.
The final study cohorts included patients with idiopathic interstitial pneumonias, chronic hypersensitivity pneumonitis, connective tissue disease, combined pulmonary fibrosis and emphysema, and occupational lung disease.
The patients were randomized to receive either inhaled treprostinil at a starting dose of 6 mcg/breath four times daily or to placebo (163 patients in each arm). All patients started the study drug at a dose of three breaths four times daily during waking hours. Dose escalations – adding 1 additional breath four times daily – were allowed every 3 days, up to a target dose of 9 breaths (54 mcg) four times daily, and a maximum of 12 breaths (72 mcg) four times daily as clinically tolerated.
A total of 130 patients assigned to treprostinil and 128 assigned to placebo completed 16 weeks of therapy and assessment.
As noted before, patients assigned to treprostinil had a placebo-corrected median difference from baseline in peak 6-minute walk distance, as measured by Hodges-Lehmann estimation, of 21 m (P = .004). An analysis of the same parameter using mixed model repeated measurement showed a placebo-corrected difference from baseline in peak 6-minute walk distance of 31.12 m (P < .001).
Secondary endpoints that were significantly better with treprostinil, compared with placebo, included improvements in N-terminal of the prohormone brain natriuretic peptide, a longer time to clinical worsening, and improvements in peak 6-minute walk distance week 12, and trough 6-minute walk distance at week 15.
Treprostinil was associated with a 39% reduction in risk of clinical worsening (P = .04). In all, 37 patients on treprostinil (22.7%) and 54 on placebo (33.1%) experienced clinical worsening.
For the exploratory endpoints of change in patient reported quality of life as measured by the St. George’s Respiratory Questionnaire, or in peak distance saturation product, however, there were no significant differences between the groups.
In addition, treprostinil was associated with a 34% reduction the risk of exacerbation of underlying lung disease, compared with placebo (P = .03).
The safety profile of treprostinil was similar to that seen in other studies of the drug, and most treatment-related adverse events were mild or moderate in severity. Adverse events led to discontinuation in 10% of patients on treprostinil and 8% on placebo.
Serious adverse events were seen in 23.3% and 25.8%, respectively. The most frequently occurring adverse events of any grade included cough, headache, dyspnea, dizziness, nausea, fatigue, diarrhea, throat irritation, and oropharyngeal pain.
There was no evidence of worsened oxygenation or lung function “allaying V/Q mismatch concerns,” Dr. Nathan said, and there was evidence for an improvement in forced vital capacity with treprostinil.
In the question-and-answer portion of the presentation, Dr. Kolb commented that many clinicians, particularly those who treated patients with ILD, question whether a 21-m difference in walk distance makes much of a difference in patient lives. He relayed a question from a viewer asking how Dr. Nathan and associates reconciled their primary endpoint with the finding that there was no difference in patient-reported quality of life.
“I think that the difference in the 6-minute walk test was both statistically significant and clinically meaningful,” Dr. Nathan replied.
He noted that the primary endpoint used a stringent measure, and that less conservative methods of analysis showed a larger difference in benefit favoring treprostinil. He also pointed out that the original study of inhaled treprostinil added to oral therapy for pulmonary arterial hypertension showed a 20-m improvement in walk distance, and that these results were sufficient to get the inhaled formulation approved in the United States (J Am Coll Cardiol. 2010 May. doi: 10.1016/j.jacc.2010.01.027).
Regarding the failure to detect a difference in quality of life, he said that the study was only 16 weeks in length, and that the St. George’s Respiratory Questionnaire was developed for evaluation of patients with chronic obstructive pulmonary disease, “perhaps not the best instrument to use in an ILD PH study.”
The study was funded by United Therapeutics. Dr. Nathan disclosed advisory committee activity/consulting, research support, and speaker fees from the company. Dr. Kolb has previously disclosed financial relationships with various companies, not including United Therapeutics.
over 16 weeks, compared with patients who used a placebo inhaler, results of a phase 3 trial showed.
Among 326 patients with pulmonary hypertension (PH) associated with interstitial lung disease (ILD), those who were randomly assigned to treatment with treprostinil had a placebo-corrected median difference from baseline in 6-minute walk distance of 21 m (P = .004), reported Steven D. Nathan, MD, from Inova Fairfax Hospital in Falls Church, Va., on behalf of coinvestigators in the INCREASE study (NCT02630316).
“These results support an additional treatment avenue, and might herald a shift in the clinical management of patients with interstitial lung disease,” he said in the American Thoracic Society’s virtual clinical trial session.
“This was an outstanding presentation and outstanding results. I personally am very excited, because this is a field where I work,” commented Martin Kolb, MD, PhD, from McMaster University, Hamilton, Ont., the facilitator for the online presentation.
The INCREASE trial compared inhaled treprostinil dose four times daily with placebo in patients with a CT scan–confirmed diagnosis of World Health Organization group 3 PH within 6 months before randomization who had evidence of diffuse parenchymal lung disease. Eligible patients could have any form of ILD or combined pulmonary fibrosis and emphysema.
Key inclusion criteria included right-heart catheterization within the previous year with documented pulmonary vascular resistance greater than 3 Wood units, pulmonary capillary wedge pressure 15 mm Hg or less, and mean pulmonary arterial pressure 25 mm Hg or higher.
Patients also had to have a 6-minute walk distance of at least 100 m and have stable disease while on an optimized dose of medications for underlying lung disease. Patients with group 3 connective tissue disease had to have baseline forced vital capacity of less than 70%.
The final study cohorts included patients with idiopathic interstitial pneumonias, chronic hypersensitivity pneumonitis, connective tissue disease, combined pulmonary fibrosis and emphysema, and occupational lung disease.
The patients were randomized to receive either inhaled treprostinil at a starting dose of 6 mcg/breath four times daily or to placebo (163 patients in each arm). All patients started the study drug at a dose of three breaths four times daily during waking hours. Dose escalations – adding 1 additional breath four times daily – were allowed every 3 days, up to a target dose of 9 breaths (54 mcg) four times daily, and a maximum of 12 breaths (72 mcg) four times daily as clinically tolerated.
A total of 130 patients assigned to treprostinil and 128 assigned to placebo completed 16 weeks of therapy and assessment.
As noted before, patients assigned to treprostinil had a placebo-corrected median difference from baseline in peak 6-minute walk distance, as measured by Hodges-Lehmann estimation, of 21 m (P = .004). An analysis of the same parameter using mixed model repeated measurement showed a placebo-corrected difference from baseline in peak 6-minute walk distance of 31.12 m (P < .001).
Secondary endpoints that were significantly better with treprostinil, compared with placebo, included improvements in N-terminal of the prohormone brain natriuretic peptide, a longer time to clinical worsening, and improvements in peak 6-minute walk distance week 12, and trough 6-minute walk distance at week 15.
Treprostinil was associated with a 39% reduction in risk of clinical worsening (P = .04). In all, 37 patients on treprostinil (22.7%) and 54 on placebo (33.1%) experienced clinical worsening.
For the exploratory endpoints of change in patient reported quality of life as measured by the St. George’s Respiratory Questionnaire, or in peak distance saturation product, however, there were no significant differences between the groups.
In addition, treprostinil was associated with a 34% reduction the risk of exacerbation of underlying lung disease, compared with placebo (P = .03).
The safety profile of treprostinil was similar to that seen in other studies of the drug, and most treatment-related adverse events were mild or moderate in severity. Adverse events led to discontinuation in 10% of patients on treprostinil and 8% on placebo.
Serious adverse events were seen in 23.3% and 25.8%, respectively. The most frequently occurring adverse events of any grade included cough, headache, dyspnea, dizziness, nausea, fatigue, diarrhea, throat irritation, and oropharyngeal pain.
There was no evidence of worsened oxygenation or lung function “allaying V/Q mismatch concerns,” Dr. Nathan said, and there was evidence for an improvement in forced vital capacity with treprostinil.
In the question-and-answer portion of the presentation, Dr. Kolb commented that many clinicians, particularly those who treated patients with ILD, question whether a 21-m difference in walk distance makes much of a difference in patient lives. He relayed a question from a viewer asking how Dr. Nathan and associates reconciled their primary endpoint with the finding that there was no difference in patient-reported quality of life.
“I think that the difference in the 6-minute walk test was both statistically significant and clinically meaningful,” Dr. Nathan replied.
He noted that the primary endpoint used a stringent measure, and that less conservative methods of analysis showed a larger difference in benefit favoring treprostinil. He also pointed out that the original study of inhaled treprostinil added to oral therapy for pulmonary arterial hypertension showed a 20-m improvement in walk distance, and that these results were sufficient to get the inhaled formulation approved in the United States (J Am Coll Cardiol. 2010 May. doi: 10.1016/j.jacc.2010.01.027).
Regarding the failure to detect a difference in quality of life, he said that the study was only 16 weeks in length, and that the St. George’s Respiratory Questionnaire was developed for evaluation of patients with chronic obstructive pulmonary disease, “perhaps not the best instrument to use in an ILD PH study.”
The study was funded by United Therapeutics. Dr. Nathan disclosed advisory committee activity/consulting, research support, and speaker fees from the company. Dr. Kolb has previously disclosed financial relationships with various companies, not including United Therapeutics.
FROM ATS 2020
Dr. Eric E. Howell assumes new role as CEO of SHM
The Society of Hospital Medicine officially welcomed Eric E. Howell, MD, MHM, as chief executive officer on July 1, 2020. Dr. Howell reports to the Society of Hospital Medicine board of directors and is tasked with ensuring that SHM continues to serve the evolving needs and interests of its members while overseeing the organization’s strategic direction.
“The SHM board of directors is excited to work with Dr. Howell to navigate the future of SHM and of the hospital medicine specialty,” said Danielle Scheurer, MD, MSCR, SFHM, SHM president and chair of the CEO Search Committee. “With his extensive knowledge of the health care landscape and of SHM, Dr. Howell embodies the society’s dedication to empowering hospitalists to be positive change agents in their institutions and in the health care system as a whole.”
Prior to his current role, Dr. Howell served as chief operating officer of SHM for 2 years; in that role, he led senior management’s planning and defined organizational goals to drive growth. As the senior physician adviser to SHM’s Center for Quality Improvement for 5 years, he consulted for the society’s arm that conducts quality improvement programs for hospitalist teams. In addition to being a past president of SHM’s board of directors, he is the course director for the SHM Leadership Academies.
“Now more than ever, SHM has an opportunity to superserve hospitalists and the patients they serve, and I couldn’t be more excited to lead the society into its next chapter,” Dr. Howell said. “Supported by a dedicated member base and innovative staff, I am confident that SHM will continue on its successful path forward and will provide its members with the products, services, and tools that hospitalists need to improve patient care and adapt to the constantly evolving environment.”
In addition to serving in various capacities at SHM, Dr. Howell has served as a professor of medicine in the department of medicine at Johns Hopkins University, Baltimore. He has held multiple titles within the Johns Hopkins medical institutions, including chief of the division of hospital medicine at Johns Hopkins Bayview, section chief of hospital medicine for Johns Hopkins Community Physicians, deputy director of hospital operations for the department of medicine at Johns Hopkins Bayview Medical Center, and chief medical officer of operations at Johns Hopkins Bayview. Dr. Howell joined the Johns Hopkins Bayview hospitalist program in 2000, began the Howard County (Md.) General Hospital hospitalist program in 2010, and oversaw nearly 200 physicians and clinical staff providing patient care in three hospitals. Along with his role as SHM CEO, he will remain a member of the adjunct faculty at Johns Hopkins University.
More recently, Dr. Howell served as chief medical officer for the Baltimore Convention Center Field Hospital, a fully functional, 250-bed hospital created to care for patients in the Baltimore metropolitan area who were suffering from complications from COVID-19.
Dr. Howell received his electrical engineering degree from the University of Maryland, College Park, which has proven instrumental in his mastery of managing and implementing change in the hospital. His research has focused on the relationship between the emergency department and medicine floors, improving communication, throughput and patient outcomes.
The nationwide search process that led to Dr. Howell’s appointment was led by a CEO Search Committee, which included members of the SHM board of directors and was assisted by the executive search firm Spencer Stuart.
Dr. Howell succeeds Laurence Wellikson, MD, MHM, who helped in founding the Society of Hospital Medicine, its first and only CEO since 2000 prior to Dr. Howell’s appointment.
The Society of Hospital Medicine officially welcomed Eric E. Howell, MD, MHM, as chief executive officer on July 1, 2020. Dr. Howell reports to the Society of Hospital Medicine board of directors and is tasked with ensuring that SHM continues to serve the evolving needs and interests of its members while overseeing the organization’s strategic direction.
“The SHM board of directors is excited to work with Dr. Howell to navigate the future of SHM and of the hospital medicine specialty,” said Danielle Scheurer, MD, MSCR, SFHM, SHM president and chair of the CEO Search Committee. “With his extensive knowledge of the health care landscape and of SHM, Dr. Howell embodies the society’s dedication to empowering hospitalists to be positive change agents in their institutions and in the health care system as a whole.”
Prior to his current role, Dr. Howell served as chief operating officer of SHM for 2 years; in that role, he led senior management’s planning and defined organizational goals to drive growth. As the senior physician adviser to SHM’s Center for Quality Improvement for 5 years, he consulted for the society’s arm that conducts quality improvement programs for hospitalist teams. In addition to being a past president of SHM’s board of directors, he is the course director for the SHM Leadership Academies.
“Now more than ever, SHM has an opportunity to superserve hospitalists and the patients they serve, and I couldn’t be more excited to lead the society into its next chapter,” Dr. Howell said. “Supported by a dedicated member base and innovative staff, I am confident that SHM will continue on its successful path forward and will provide its members with the products, services, and tools that hospitalists need to improve patient care and adapt to the constantly evolving environment.”
In addition to serving in various capacities at SHM, Dr. Howell has served as a professor of medicine in the department of medicine at Johns Hopkins University, Baltimore. He has held multiple titles within the Johns Hopkins medical institutions, including chief of the division of hospital medicine at Johns Hopkins Bayview, section chief of hospital medicine for Johns Hopkins Community Physicians, deputy director of hospital operations for the department of medicine at Johns Hopkins Bayview Medical Center, and chief medical officer of operations at Johns Hopkins Bayview. Dr. Howell joined the Johns Hopkins Bayview hospitalist program in 2000, began the Howard County (Md.) General Hospital hospitalist program in 2010, and oversaw nearly 200 physicians and clinical staff providing patient care in three hospitals. Along with his role as SHM CEO, he will remain a member of the adjunct faculty at Johns Hopkins University.
More recently, Dr. Howell served as chief medical officer for the Baltimore Convention Center Field Hospital, a fully functional, 250-bed hospital created to care for patients in the Baltimore metropolitan area who were suffering from complications from COVID-19.
Dr. Howell received his electrical engineering degree from the University of Maryland, College Park, which has proven instrumental in his mastery of managing and implementing change in the hospital. His research has focused on the relationship between the emergency department and medicine floors, improving communication, throughput and patient outcomes.
The nationwide search process that led to Dr. Howell’s appointment was led by a CEO Search Committee, which included members of the SHM board of directors and was assisted by the executive search firm Spencer Stuart.
Dr. Howell succeeds Laurence Wellikson, MD, MHM, who helped in founding the Society of Hospital Medicine, its first and only CEO since 2000 prior to Dr. Howell’s appointment.
The Society of Hospital Medicine officially welcomed Eric E. Howell, MD, MHM, as chief executive officer on July 1, 2020. Dr. Howell reports to the Society of Hospital Medicine board of directors and is tasked with ensuring that SHM continues to serve the evolving needs and interests of its members while overseeing the organization’s strategic direction.
“The SHM board of directors is excited to work with Dr. Howell to navigate the future of SHM and of the hospital medicine specialty,” said Danielle Scheurer, MD, MSCR, SFHM, SHM president and chair of the CEO Search Committee. “With his extensive knowledge of the health care landscape and of SHM, Dr. Howell embodies the society’s dedication to empowering hospitalists to be positive change agents in their institutions and in the health care system as a whole.”
Prior to his current role, Dr. Howell served as chief operating officer of SHM for 2 years; in that role, he led senior management’s planning and defined organizational goals to drive growth. As the senior physician adviser to SHM’s Center for Quality Improvement for 5 years, he consulted for the society’s arm that conducts quality improvement programs for hospitalist teams. In addition to being a past president of SHM’s board of directors, he is the course director for the SHM Leadership Academies.
“Now more than ever, SHM has an opportunity to superserve hospitalists and the patients they serve, and I couldn’t be more excited to lead the society into its next chapter,” Dr. Howell said. “Supported by a dedicated member base and innovative staff, I am confident that SHM will continue on its successful path forward and will provide its members with the products, services, and tools that hospitalists need to improve patient care and adapt to the constantly evolving environment.”
In addition to serving in various capacities at SHM, Dr. Howell has served as a professor of medicine in the department of medicine at Johns Hopkins University, Baltimore. He has held multiple titles within the Johns Hopkins medical institutions, including chief of the division of hospital medicine at Johns Hopkins Bayview, section chief of hospital medicine for Johns Hopkins Community Physicians, deputy director of hospital operations for the department of medicine at Johns Hopkins Bayview Medical Center, and chief medical officer of operations at Johns Hopkins Bayview. Dr. Howell joined the Johns Hopkins Bayview hospitalist program in 2000, began the Howard County (Md.) General Hospital hospitalist program in 2010, and oversaw nearly 200 physicians and clinical staff providing patient care in three hospitals. Along with his role as SHM CEO, he will remain a member of the adjunct faculty at Johns Hopkins University.
More recently, Dr. Howell served as chief medical officer for the Baltimore Convention Center Field Hospital, a fully functional, 250-bed hospital created to care for patients in the Baltimore metropolitan area who were suffering from complications from COVID-19.
Dr. Howell received his electrical engineering degree from the University of Maryland, College Park, which has proven instrumental in his mastery of managing and implementing change in the hospital. His research has focused on the relationship between the emergency department and medicine floors, improving communication, throughput and patient outcomes.
The nationwide search process that led to Dr. Howell’s appointment was led by a CEO Search Committee, which included members of the SHM board of directors and was assisted by the executive search firm Spencer Stuart.
Dr. Howell succeeds Laurence Wellikson, MD, MHM, who helped in founding the Society of Hospital Medicine, its first and only CEO since 2000 prior to Dr. Howell’s appointment.
The in-person postpartum blood pressure check: For whose benefit?
CASE Patient questions need for postpartum BP check
Ms. P presents at 28 weeks’ gestation with superimposed preeclampsia. She receives antenatal corticosteroids and titration of her nifedipine, but she is delivered at 29 weeks because of worsening fetal status. Her physician recommends a blood pressure (BP) visit in the office at 7 days postpartum.
She asks, “But can’t I just call you with the BP reading? And what do I do in the meantime?”
Hypertensive disorders of pregnancy and chronic hypertension remain among the leading causes of maternal morbidity and mortality in the United States and worldwide.1 The postpartum period remains a particularly high-risk time since up to 40% of maternal mortality can occur after delivery. To that end, the 2013 American College of Obstetricians and Gynecologists Hypertension in Pregnancy Task Force recommends postpartum follow-up 7 to 10 days after delivery in women with a hypertensive disorder of pregnancy.2
Why we need to find an alternative approach
Unfortunately, these guidelines are both cumbersome and insufficient. Up to one-third of patients do not attend their postpartum visit, particularly those who are young, uninsured, and nonwhite, a list uncomfortably similar to that for women most at risk for adverse outcomes after a high-risk pregnancy. In addition, the 7- to 10-day visit still represents only a single snapshot of the patient’s BP values rather than an ongoing assessment of symptoms or BP elevation over time. Moreover, studies also have shown that BP in both normotensive and hypertensive women often rises by the fifth day postpartum, suggesting that leaving this large window of time without surveillance may miss an opportunity to detect elevated BP in a more timely manner.3
It is time to break the habit of the in-office postpartum BP check and to evaluate the patient where she is and when she needs it. Research in the last 2 years shows that there are several solutions to our case patient’s question.

Solution 1: The provider-driven system
“Of course. Text us your numbers, and you will hear from the doctor if you need to do anything differently.”
One method that addresses both the communication and safety issues inherent in the 7- to 10-day routine in-office BP check is to have the patient send in her BP measurements for direct clinician review.
Researchers at the University of Pennsylvania developed a robust program using their Way to Health platform.4 Participating patients text their BP values twice daily, and they receive automated feedback for all values, with additional human feedback in real time from a clinician for severe-range values (>160 mm Hg systolic or >110 mm Hg diastolic). As an added safety measure, a physician reviews all inputted BPs daily and assesses the need for antihypertensive medication for BPs in the high mild range. Using this protocol, the researchers achieved a significant increase in adherence with the recommendation for reporting a BP value in the first 10 days after discharge (from 44% to 92%) as well as having fewer readmissions in the text-messaging arm (4% vs 0%).
Perhaps most impressive, though, is that the technology use eliminated pre-existing racial disparities in adherence. Black participants were as likely as nonblack participants to report a postpartum BP in the text-messaging system (93% vs 91%) despite being less than half as likely to keep a BP check visit (33% vs 70%).5
A similar solution is in place at the University of Pittsburgh, where a text message system on the Vivify platform is used to deliver patient BP measurements to a centralized monitoring team.6 This program is unique in that, rather than relying on a single physician, it is run through a nurse “call center” that allowed them to expand to 3 hospitals with the use of a single centralized monitoring team. To date, the program has enrolled more than 2,000 patients and achieved patient satisfaction rates greater than 94%.
A final program to consider was developed and piloted at the University of Wisconsin with an added technological advance: the use of a Bluetooth-enabled BP cuff that permits values to be automatically transmitted to a tablet that then uploads the information to a centralized database.7 This database was in turn monitored by trained nurses for safety and initiation or titration of antihypertensive medication as needed. Similar to the experience at the University of Pennsylvania, the researchers found improved adherence with monitoring and a notable reduction in readmissions (3.7% in controls vs 0.5% in the intervention arm). Of note, among those who did receive the ongoing monitoring, severe hypertension occurred in 56 (26.2%) of those patients and did so a mean of 6 days after discharge (that is, prior to when they typically would have seen a provider.)
The promise of such provider-driven systems is that they represent a true chronicle of a patient’s ongoing clinical course rather than a single snapshot of her BP in an artificial environment (and often after the highest risk time period!). In addition, direct monitoring by clinicians ensures an optimal safety profile.
Such systems, however, are also extremely resource intense in terms of both upfront information technology investment and ongoing provider surveillance. The systems above also relied on giving the patients a BP cuff, so it is unclear whether it was the technology support or this simple intervention that yielded the benefits. Nonetheless, the benefits were undeniable, and the financial costs saved by reducing even 1 hospital admission as well as the costs of outpatient surveillance may in the end justify these upfront expenditures.
Continue to: Solution 2: The algorithm-driven system...
Solution 2: The algorithm-driven system
“Sure. Plug your numbers into our system, and you’ll receive an automated response as to what to do next.”
One way to alleviate both the financial and opportunity cost of constant clinician surveillance would be to offload some tasks to algorithmic support. This approach—home BP monitoring accompanied by self-titration of antihypertensive medication—has been validated in outpatient primary care hypertension management in nonpregnant adults and more recently for postpartum patients as well.
In the SNAP-HT trial, investigators randomly assigned women to either usual care or algorithm-driven outpatient BP management.8 While both groups had serial visits (for safety monitoring), those in the experimental arm were advised only by the algorithm for any ongoing titration of medication. At 6 weeks, the investigators found that BPs were lower in the intervention group, and diastolic BPs remained lower at 6 months.
This methodology emphasizes the potential utility of true self-management of hypertension in the postpartum period. It relies, however, on having a highly developed system in place that can receive the data, respond with recommendations, and safely monitor for any aberrations in the feed. Still, this hybrid method may represent the sweet spot: a combination that ensures adequate surveillance while not overburdening the clinician with the simpler, initial steps in postpartum antihypertensive management.
Solution 3: The DIY system
“That’s a good point. I want to hear about your blood pressure readings in the meantime. Here’s what we can do.”
What about the 99% of practicing ObGyns who do not have an entire connected system for remote hypertension monitoring? A number of options can be put in place today with little cost and even less tech know-how (see “Do-it-yourself options for remote blood pressure monitoring,” below). Note that since many of these options would not be monitored in “real time” like the connected systems discussed above, the patient should be given strict parameters for contacting her clinician directly. These do-it-yourself, or DIY, methods are instead best for the purpose of chronic monitoring and medication titration but are still an improvement in communication over the single-serve BP check.
The bottom line
Pregnant women represent one of the most connected, Internet-savvy demographic groups of any patient population: More than three-quarters of pregnant women turn to the Internet for advice during their pregnancy.9,10 In addition, unlike most social determinants of health, such as housing, food access, and health care coverage, access to connected electronic devices differs little across racial lines, suggesting the potential for targeting health care inequities by implementing more—not less—technology into prenatal and postpartum care.
For this generation of new mothers, the in-office postpartum BP check is insufficient, artificial, and simply a waste of everyone’s time. While there is no one-size-fits-all approach, there are many options, and it is up to us as health care providers to facilitate the right care, in the right place, at the right time for our patients. ●
Acknowledgements: The authors would like to thank Haritha Pavuluri, Margaret Oliver, and Samantha Boniface for their assistance in the preparation of this manuscript.
Electronic health record (EHR) messaging
Most EHR systems have some form of patient messaging built in. Consider asking your patient to:
- message her blood pressure measurements every 1 to 2 days
- send a photo of handwritten blood pressure measurements
Vendor text messaging platforms
The year 2020 has seen the entire telehealth space grow tremendously, and platforms such as Doxy.me (https://doxy.me) and Updox (https://www.updox.com) allow secure text messaging with patients.
All-in-one connected vendor solutions
Third-party solutions are available that give the patient a connected blood pressure cuff, scale, and personalized app. For the clinician, these data then can be accessed either independently through a portal or can be integrated into the EHR. Examples of 2 companies include:
- Babyscripts (https://www.getbabyscripts.com)
- Wildflower Health (https://www.wildflowerhealth.com)
Telehealth visits
Scheduling weekly telephone or video visits (while not near the frequency of the above) would still yield greater engagement, and many payors currently reimburse for these visits at rates on par with in-person visits.
- American College of Obstetricians and Gynecologists. ACOG practice bulletin summary, No. 222. Gestational hypertension and preeclampsia. Obstet Gynecol. 2020;135:1492-1495.
- American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Hypertension in pregnancy. Obstet Gynecol. 2013;122:1122-1131.
- Walters BN, Thompson ME, Lee A, et al. Blood pressure in the puerperium. Clin Sci. 1986;71:589-594.
- Hirshberg A, Downes K, Srinivas S. Comparing standard office-based follow-up with text-based remote monitoring in the management of postpartum hypertension: a randomised clinical trial. BMJ Qual Saf. 2018;27:871-877.
- Hirshberg A, Sammel MD, Srinivas SK. Text message remote monitoring reduced racial disparities in postpartum blood pressure ascertainment. Am J Obstet Gynecol. 2019;221:283-285.
- Hauspurg A, Lemon LS, Quinn BA, et al. A postpartum remote hypertension monitoring protocol implemented at the hospital level. Obstet Gynecol. 2019;134:685-691.
- Hoppe KK, Thomas N, Zernick M, et al. Telehealth with remote blood pressure monitoring compared to standard care for postpartum hypertension. Am J Obstet Gynecol. 2020;S0002-9378(20)30554-doi:10.1016/j.ajog.2020.05.027.
- Cairns AE, Tucker KL, Leeson P, et al. Self-management of postnatal hypertension. Hypertension. 2018;72:425-432.
- Pew Research Center. Mobile fact sheet, 2019. https://www.pewresearch.org/internet/fact-sheet/mobile/. Accessed June 16, 2020.
- Sayakhot P, Carolan-Olah M. Internet use by pregnant women seeking pregnancy-related information: a systematic review. BMC Pregnancy Childbirth. 2016;16:65
CASE Patient questions need for postpartum BP check
Ms. P presents at 28 weeks’ gestation with superimposed preeclampsia. She receives antenatal corticosteroids and titration of her nifedipine, but she is delivered at 29 weeks because of worsening fetal status. Her physician recommends a blood pressure (BP) visit in the office at 7 days postpartum.
She asks, “But can’t I just call you with the BP reading? And what do I do in the meantime?”
Hypertensive disorders of pregnancy and chronic hypertension remain among the leading causes of maternal morbidity and mortality in the United States and worldwide.1 The postpartum period remains a particularly high-risk time since up to 40% of maternal mortality can occur after delivery. To that end, the 2013 American College of Obstetricians and Gynecologists Hypertension in Pregnancy Task Force recommends postpartum follow-up 7 to 10 days after delivery in women with a hypertensive disorder of pregnancy.2
Why we need to find an alternative approach
Unfortunately, these guidelines are both cumbersome and insufficient. Up to one-third of patients do not attend their postpartum visit, particularly those who are young, uninsured, and nonwhite, a list uncomfortably similar to that for women most at risk for adverse outcomes after a high-risk pregnancy. In addition, the 7- to 10-day visit still represents only a single snapshot of the patient’s BP values rather than an ongoing assessment of symptoms or BP elevation over time. Moreover, studies also have shown that BP in both normotensive and hypertensive women often rises by the fifth day postpartum, suggesting that leaving this large window of time without surveillance may miss an opportunity to detect elevated BP in a more timely manner.3
It is time to break the habit of the in-office postpartum BP check and to evaluate the patient where she is and when she needs it. Research in the last 2 years shows that there are several solutions to our case patient’s question.

Solution 1: The provider-driven system
“Of course. Text us your numbers, and you will hear from the doctor if you need to do anything differently.”
One method that addresses both the communication and safety issues inherent in the 7- to 10-day routine in-office BP check is to have the patient send in her BP measurements for direct clinician review.
Researchers at the University of Pennsylvania developed a robust program using their Way to Health platform.4 Participating patients text their BP values twice daily, and they receive automated feedback for all values, with additional human feedback in real time from a clinician for severe-range values (>160 mm Hg systolic or >110 mm Hg diastolic). As an added safety measure, a physician reviews all inputted BPs daily and assesses the need for antihypertensive medication for BPs in the high mild range. Using this protocol, the researchers achieved a significant increase in adherence with the recommendation for reporting a BP value in the first 10 days after discharge (from 44% to 92%) as well as having fewer readmissions in the text-messaging arm (4% vs 0%).
Perhaps most impressive, though, is that the technology use eliminated pre-existing racial disparities in adherence. Black participants were as likely as nonblack participants to report a postpartum BP in the text-messaging system (93% vs 91%) despite being less than half as likely to keep a BP check visit (33% vs 70%).5
A similar solution is in place at the University of Pittsburgh, where a text message system on the Vivify platform is used to deliver patient BP measurements to a centralized monitoring team.6 This program is unique in that, rather than relying on a single physician, it is run through a nurse “call center” that allowed them to expand to 3 hospitals with the use of a single centralized monitoring team. To date, the program has enrolled more than 2,000 patients and achieved patient satisfaction rates greater than 94%.
A final program to consider was developed and piloted at the University of Wisconsin with an added technological advance: the use of a Bluetooth-enabled BP cuff that permits values to be automatically transmitted to a tablet that then uploads the information to a centralized database.7 This database was in turn monitored by trained nurses for safety and initiation or titration of antihypertensive medication as needed. Similar to the experience at the University of Pennsylvania, the researchers found improved adherence with monitoring and a notable reduction in readmissions (3.7% in controls vs 0.5% in the intervention arm). Of note, among those who did receive the ongoing monitoring, severe hypertension occurred in 56 (26.2%) of those patients and did so a mean of 6 days after discharge (that is, prior to when they typically would have seen a provider.)
The promise of such provider-driven systems is that they represent a true chronicle of a patient’s ongoing clinical course rather than a single snapshot of her BP in an artificial environment (and often after the highest risk time period!). In addition, direct monitoring by clinicians ensures an optimal safety profile.
Such systems, however, are also extremely resource intense in terms of both upfront information technology investment and ongoing provider surveillance. The systems above also relied on giving the patients a BP cuff, so it is unclear whether it was the technology support or this simple intervention that yielded the benefits. Nonetheless, the benefits were undeniable, and the financial costs saved by reducing even 1 hospital admission as well as the costs of outpatient surveillance may in the end justify these upfront expenditures.
Continue to: Solution 2: The algorithm-driven system...
Solution 2: The algorithm-driven system
“Sure. Plug your numbers into our system, and you’ll receive an automated response as to what to do next.”
One way to alleviate both the financial and opportunity cost of constant clinician surveillance would be to offload some tasks to algorithmic support. This approach—home BP monitoring accompanied by self-titration of antihypertensive medication—has been validated in outpatient primary care hypertension management in nonpregnant adults and more recently for postpartum patients as well.
In the SNAP-HT trial, investigators randomly assigned women to either usual care or algorithm-driven outpatient BP management.8 While both groups had serial visits (for safety monitoring), those in the experimental arm were advised only by the algorithm for any ongoing titration of medication. At 6 weeks, the investigators found that BPs were lower in the intervention group, and diastolic BPs remained lower at 6 months.
This methodology emphasizes the potential utility of true self-management of hypertension in the postpartum period. It relies, however, on having a highly developed system in place that can receive the data, respond with recommendations, and safely monitor for any aberrations in the feed. Still, this hybrid method may represent the sweet spot: a combination that ensures adequate surveillance while not overburdening the clinician with the simpler, initial steps in postpartum antihypertensive management.
Solution 3: The DIY system
“That’s a good point. I want to hear about your blood pressure readings in the meantime. Here’s what we can do.”
What about the 99% of practicing ObGyns who do not have an entire connected system for remote hypertension monitoring? A number of options can be put in place today with little cost and even less tech know-how (see “Do-it-yourself options for remote blood pressure monitoring,” below). Note that since many of these options would not be monitored in “real time” like the connected systems discussed above, the patient should be given strict parameters for contacting her clinician directly. These do-it-yourself, or DIY, methods are instead best for the purpose of chronic monitoring and medication titration but are still an improvement in communication over the single-serve BP check.
The bottom line
Pregnant women represent one of the most connected, Internet-savvy demographic groups of any patient population: More than three-quarters of pregnant women turn to the Internet for advice during their pregnancy.9,10 In addition, unlike most social determinants of health, such as housing, food access, and health care coverage, access to connected electronic devices differs little across racial lines, suggesting the potential for targeting health care inequities by implementing more—not less—technology into prenatal and postpartum care.
For this generation of new mothers, the in-office postpartum BP check is insufficient, artificial, and simply a waste of everyone’s time. While there is no one-size-fits-all approach, there are many options, and it is up to us as health care providers to facilitate the right care, in the right place, at the right time for our patients. ●
Acknowledgements: The authors would like to thank Haritha Pavuluri, Margaret Oliver, and Samantha Boniface for their assistance in the preparation of this manuscript.
Electronic health record (EHR) messaging
Most EHR systems have some form of patient messaging built in. Consider asking your patient to:
- message her blood pressure measurements every 1 to 2 days
- send a photo of handwritten blood pressure measurements
Vendor text messaging platforms
The year 2020 has seen the entire telehealth space grow tremendously, and platforms such as Doxy.me (https://doxy.me) and Updox (https://www.updox.com) allow secure text messaging with patients.
All-in-one connected vendor solutions
Third-party solutions are available that give the patient a connected blood pressure cuff, scale, and personalized app. For the clinician, these data then can be accessed either independently through a portal or can be integrated into the EHR. Examples of 2 companies include:
- Babyscripts (https://www.getbabyscripts.com)
- Wildflower Health (https://www.wildflowerhealth.com)
Telehealth visits
Scheduling weekly telephone or video visits (while not near the frequency of the above) would still yield greater engagement, and many payors currently reimburse for these visits at rates on par with in-person visits.
CASE Patient questions need for postpartum BP check
Ms. P presents at 28 weeks’ gestation with superimposed preeclampsia. She receives antenatal corticosteroids and titration of her nifedipine, but she is delivered at 29 weeks because of worsening fetal status. Her physician recommends a blood pressure (BP) visit in the office at 7 days postpartum.
She asks, “But can’t I just call you with the BP reading? And what do I do in the meantime?”
Hypertensive disorders of pregnancy and chronic hypertension remain among the leading causes of maternal morbidity and mortality in the United States and worldwide.1 The postpartum period remains a particularly high-risk time since up to 40% of maternal mortality can occur after delivery. To that end, the 2013 American College of Obstetricians and Gynecologists Hypertension in Pregnancy Task Force recommends postpartum follow-up 7 to 10 days after delivery in women with a hypertensive disorder of pregnancy.2
Why we need to find an alternative approach
Unfortunately, these guidelines are both cumbersome and insufficient. Up to one-third of patients do not attend their postpartum visit, particularly those who are young, uninsured, and nonwhite, a list uncomfortably similar to that for women most at risk for adverse outcomes after a high-risk pregnancy. In addition, the 7- to 10-day visit still represents only a single snapshot of the patient’s BP values rather than an ongoing assessment of symptoms or BP elevation over time. Moreover, studies also have shown that BP in both normotensive and hypertensive women often rises by the fifth day postpartum, suggesting that leaving this large window of time without surveillance may miss an opportunity to detect elevated BP in a more timely manner.3
It is time to break the habit of the in-office postpartum BP check and to evaluate the patient where she is and when she needs it. Research in the last 2 years shows that there are several solutions to our case patient’s question.

Solution 1: The provider-driven system
“Of course. Text us your numbers, and you will hear from the doctor if you need to do anything differently.”
One method that addresses both the communication and safety issues inherent in the 7- to 10-day routine in-office BP check is to have the patient send in her BP measurements for direct clinician review.
Researchers at the University of Pennsylvania developed a robust program using their Way to Health platform.4 Participating patients text their BP values twice daily, and they receive automated feedback for all values, with additional human feedback in real time from a clinician for severe-range values (>160 mm Hg systolic or >110 mm Hg diastolic). As an added safety measure, a physician reviews all inputted BPs daily and assesses the need for antihypertensive medication for BPs in the high mild range. Using this protocol, the researchers achieved a significant increase in adherence with the recommendation for reporting a BP value in the first 10 days after discharge (from 44% to 92%) as well as having fewer readmissions in the text-messaging arm (4% vs 0%).
Perhaps most impressive, though, is that the technology use eliminated pre-existing racial disparities in adherence. Black participants were as likely as nonblack participants to report a postpartum BP in the text-messaging system (93% vs 91%) despite being less than half as likely to keep a BP check visit (33% vs 70%).5
A similar solution is in place at the University of Pittsburgh, where a text message system on the Vivify platform is used to deliver patient BP measurements to a centralized monitoring team.6 This program is unique in that, rather than relying on a single physician, it is run through a nurse “call center” that allowed them to expand to 3 hospitals with the use of a single centralized monitoring team. To date, the program has enrolled more than 2,000 patients and achieved patient satisfaction rates greater than 94%.
A final program to consider was developed and piloted at the University of Wisconsin with an added technological advance: the use of a Bluetooth-enabled BP cuff that permits values to be automatically transmitted to a tablet that then uploads the information to a centralized database.7 This database was in turn monitored by trained nurses for safety and initiation or titration of antihypertensive medication as needed. Similar to the experience at the University of Pennsylvania, the researchers found improved adherence with monitoring and a notable reduction in readmissions (3.7% in controls vs 0.5% in the intervention arm). Of note, among those who did receive the ongoing monitoring, severe hypertension occurred in 56 (26.2%) of those patients and did so a mean of 6 days after discharge (that is, prior to when they typically would have seen a provider.)
The promise of such provider-driven systems is that they represent a true chronicle of a patient’s ongoing clinical course rather than a single snapshot of her BP in an artificial environment (and often after the highest risk time period!). In addition, direct monitoring by clinicians ensures an optimal safety profile.
Such systems, however, are also extremely resource intense in terms of both upfront information technology investment and ongoing provider surveillance. The systems above also relied on giving the patients a BP cuff, so it is unclear whether it was the technology support or this simple intervention that yielded the benefits. Nonetheless, the benefits were undeniable, and the financial costs saved by reducing even 1 hospital admission as well as the costs of outpatient surveillance may in the end justify these upfront expenditures.
Continue to: Solution 2: The algorithm-driven system...
Solution 2: The algorithm-driven system
“Sure. Plug your numbers into our system, and you’ll receive an automated response as to what to do next.”
One way to alleviate both the financial and opportunity cost of constant clinician surveillance would be to offload some tasks to algorithmic support. This approach—home BP monitoring accompanied by self-titration of antihypertensive medication—has been validated in outpatient primary care hypertension management in nonpregnant adults and more recently for postpartum patients as well.
In the SNAP-HT trial, investigators randomly assigned women to either usual care or algorithm-driven outpatient BP management.8 While both groups had serial visits (for safety monitoring), those in the experimental arm were advised only by the algorithm for any ongoing titration of medication. At 6 weeks, the investigators found that BPs were lower in the intervention group, and diastolic BPs remained lower at 6 months.
This methodology emphasizes the potential utility of true self-management of hypertension in the postpartum period. It relies, however, on having a highly developed system in place that can receive the data, respond with recommendations, and safely monitor for any aberrations in the feed. Still, this hybrid method may represent the sweet spot: a combination that ensures adequate surveillance while not overburdening the clinician with the simpler, initial steps in postpartum antihypertensive management.
Solution 3: The DIY system
“That’s a good point. I want to hear about your blood pressure readings in the meantime. Here’s what we can do.”
What about the 99% of practicing ObGyns who do not have an entire connected system for remote hypertension monitoring? A number of options can be put in place today with little cost and even less tech know-how (see “Do-it-yourself options for remote blood pressure monitoring,” below). Note that since many of these options would not be monitored in “real time” like the connected systems discussed above, the patient should be given strict parameters for contacting her clinician directly. These do-it-yourself, or DIY, methods are instead best for the purpose of chronic monitoring and medication titration but are still an improvement in communication over the single-serve BP check.
The bottom line
Pregnant women represent one of the most connected, Internet-savvy demographic groups of any patient population: More than three-quarters of pregnant women turn to the Internet for advice during their pregnancy.9,10 In addition, unlike most social determinants of health, such as housing, food access, and health care coverage, access to connected electronic devices differs little across racial lines, suggesting the potential for targeting health care inequities by implementing more—not less—technology into prenatal and postpartum care.
For this generation of new mothers, the in-office postpartum BP check is insufficient, artificial, and simply a waste of everyone’s time. While there is no one-size-fits-all approach, there are many options, and it is up to us as health care providers to facilitate the right care, in the right place, at the right time for our patients. ●
Acknowledgements: The authors would like to thank Haritha Pavuluri, Margaret Oliver, and Samantha Boniface for their assistance in the preparation of this manuscript.
Electronic health record (EHR) messaging
Most EHR systems have some form of patient messaging built in. Consider asking your patient to:
- message her blood pressure measurements every 1 to 2 days
- send a photo of handwritten blood pressure measurements
Vendor text messaging platforms
The year 2020 has seen the entire telehealth space grow tremendously, and platforms such as Doxy.me (https://doxy.me) and Updox (https://www.updox.com) allow secure text messaging with patients.
All-in-one connected vendor solutions
Third-party solutions are available that give the patient a connected blood pressure cuff, scale, and personalized app. For the clinician, these data then can be accessed either independently through a portal or can be integrated into the EHR. Examples of 2 companies include:
- Babyscripts (https://www.getbabyscripts.com)
- Wildflower Health (https://www.wildflowerhealth.com)
Telehealth visits
Scheduling weekly telephone or video visits (while not near the frequency of the above) would still yield greater engagement, and many payors currently reimburse for these visits at rates on par with in-person visits.
- American College of Obstetricians and Gynecologists. ACOG practice bulletin summary, No. 222. Gestational hypertension and preeclampsia. Obstet Gynecol. 2020;135:1492-1495.
- American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Hypertension in pregnancy. Obstet Gynecol. 2013;122:1122-1131.
- Walters BN, Thompson ME, Lee A, et al. Blood pressure in the puerperium. Clin Sci. 1986;71:589-594.
- Hirshberg A, Downes K, Srinivas S. Comparing standard office-based follow-up with text-based remote monitoring in the management of postpartum hypertension: a randomised clinical trial. BMJ Qual Saf. 2018;27:871-877.
- Hirshberg A, Sammel MD, Srinivas SK. Text message remote monitoring reduced racial disparities in postpartum blood pressure ascertainment. Am J Obstet Gynecol. 2019;221:283-285.
- Hauspurg A, Lemon LS, Quinn BA, et al. A postpartum remote hypertension monitoring protocol implemented at the hospital level. Obstet Gynecol. 2019;134:685-691.
- Hoppe KK, Thomas N, Zernick M, et al. Telehealth with remote blood pressure monitoring compared to standard care for postpartum hypertension. Am J Obstet Gynecol. 2020;S0002-9378(20)30554-doi:10.1016/j.ajog.2020.05.027.
- Cairns AE, Tucker KL, Leeson P, et al. Self-management of postnatal hypertension. Hypertension. 2018;72:425-432.
- Pew Research Center. Mobile fact sheet, 2019. https://www.pewresearch.org/internet/fact-sheet/mobile/. Accessed June 16, 2020.
- Sayakhot P, Carolan-Olah M. Internet use by pregnant women seeking pregnancy-related information: a systematic review. BMC Pregnancy Childbirth. 2016;16:65
- American College of Obstetricians and Gynecologists. ACOG practice bulletin summary, No. 222. Gestational hypertension and preeclampsia. Obstet Gynecol. 2020;135:1492-1495.
- American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Hypertension in pregnancy. Obstet Gynecol. 2013;122:1122-1131.
- Walters BN, Thompson ME, Lee A, et al. Blood pressure in the puerperium. Clin Sci. 1986;71:589-594.
- Hirshberg A, Downes K, Srinivas S. Comparing standard office-based follow-up with text-based remote monitoring in the management of postpartum hypertension: a randomised clinical trial. BMJ Qual Saf. 2018;27:871-877.
- Hirshberg A, Sammel MD, Srinivas SK. Text message remote monitoring reduced racial disparities in postpartum blood pressure ascertainment. Am J Obstet Gynecol. 2019;221:283-285.
- Hauspurg A, Lemon LS, Quinn BA, et al. A postpartum remote hypertension monitoring protocol implemented at the hospital level. Obstet Gynecol. 2019;134:685-691.
- Hoppe KK, Thomas N, Zernick M, et al. Telehealth with remote blood pressure monitoring compared to standard care for postpartum hypertension. Am J Obstet Gynecol. 2020;S0002-9378(20)30554-doi:10.1016/j.ajog.2020.05.027.
- Cairns AE, Tucker KL, Leeson P, et al. Self-management of postnatal hypertension. Hypertension. 2018;72:425-432.
- Pew Research Center. Mobile fact sheet, 2019. https://www.pewresearch.org/internet/fact-sheet/mobile/. Accessed June 16, 2020.
- Sayakhot P, Carolan-Olah M. Internet use by pregnant women seeking pregnancy-related information: a systematic review. BMC Pregnancy Childbirth. 2016;16:65
2020 Update on abnormal uterine bleeding
Abnormal uterine bleeding (AUB) continues to be a top reason that women present for gynecologic care. In general, our approach to the management of AUB is to diagnose causes before we prescribe therapy and to offer conservative therapies initially and progress to more invasive measures if indicated.
In this Update, we highlight several new studies that provide evidence for preferential use of certain medical and surgical therapies. In considering conservative therapy for the treatment of AUB, we take a closer look at the efficacy of cyclic progestogens. Another important issue, as more types of endometrial ablation (EA) are being developed and are coming into the market, is the need for additional guidance regarding decisions about EA versus progestin-releasing intrauterine devices (IUDs). Lastly, an unintended consequence of an increased cesarean delivery rate is the development of isthmocele, also known as cesarean scar defect or uterine niche. These defects, which can be bothersome and cause abnormal bleeding, are treated with various techniques. Within the last year, 2 systematic reviews that compare the efficacy of several different approaches and provide guidance have been published.
Is it time to retire cyclic progestogens for the treatment of heavy menstrual bleeding?
Bofill Rodriguez M, Lethaby A, Low C, et al. Cyclical progestogens for heavy menstrual bleeding. Cochrane Database Syst Rev. 2019;(8):CD001016.
In a recent Cochrane Database Systematic Review, Bofill Rodriguez and colleagues looked at the efficacy, safety, and tolerability of oral progestogen therapy for heavy menstrual bleeding.1 They considered progestogen (medroxyprogesterone acetate or norethisterone) in short-cycle use (7 to 10 days in the luteal phase) and long-cycle use (21 days per cycle) in a review of 15 randomized clinical trials (RCTs) that included a total of 1,071 women. As this topic had not been updated in 12 years, this review was essential in demonstrating changes that occurred over the past decade.
The primary outcomes of the analysis were menstrual blood loss and treatment satisfaction. Secondary outcomes included the number of days of bleeding, quality of life, adherence and acceptability of treatment, adverse events, and costs.
Classic progestogens fall short compared with newer approaches
Analysis of the data revealed that short-cycle progestogen was inferior to treatment with tranexamic acid, danazol, and the 65-µg progesterone-releasing IUD (Pg-IUD). Of note, the 65-µg Pg-IUD has been off the market since 2001, and danazol is rarely used in current practice. Furthermore, based on 2 trials, cyclic progestogens demonstrated no clear benefit over nonsteroidal anti-inflammatory drugs. Additionally, long-cycle progestogen therapy was found to be inferior to the 52-mg levonorgestrel-releasing IUD (LNG-IUD), tranexamic acid, and ormeloxifene.
It should be noted that the quality of evidence is still lacking for progestogen therapy, and this study's main limitation is bias, as the women and the researchers were aware of the treatments that were given. This review is helpful, however, for emphasizing the advantage of tranexamic acid and LNG-IUD use in clinical care.
The takeaway. Although it may not necessarily be time to retire the use of cyclic oral progestogens, the 52-mg LNG-IUD or tranexamic acid may be more successful for treating AUB in women who are appropriate candidates.
Cyclic progestogen therapy appears to be less effective for the treatment of AUB when compared with tranexamic acid and the LNG-IUD. It does not appear to be more helpful than nonsteroidal anti-inflammatory drugs. We frequently offer and prescribe tranexamic acid, 1,300 mg 3 times daily, as a medical alternative to hormonal therapy for up to 5 days monthly for women without thromboembolism risk. Lukes and colleagues published an RCT in 2010 that demonstrated a 40% reduction of bleeding in tranexamic acid–treated women compared with an 8.2% reduction in the placebo group.2
Continue to: Endometrial ablation...
Endometrial ablation: New evidence informs when it could (and could not) be the best option
Bergeron C, Laberge PY, Boutin A, et al. Endometrial ablation or resection versus levonorgestrel intra-uterine system for the treatment of women with heavy menstrual bleeding and a normal uterine cavity: a systematic review with meta-analysis. Hum Reprod Update. 2020;26:302-311.
Vitale SG, Ferrero S, Ciebiera M, et al. Hysteroscopic endometrial resection vs hysterectomy for abnormal uterine bleeding: impact on quality of life and sexuality. Evidence from a systematic review of randomized controlled trials. Curr Opin Obstet Gynecol. 2020;32:159-165.
Two systematic reviews evaluated the efficacy of EA in women with abnormal uterine bleeding. One compared EA with the LNG-IUD and reported on safety and efficacy, while the other compared EA with hysterectomy and reported on quality of life.
Bergeron and colleagues reviewed 13 studies that included 884 women to compare the efficacy and safety of EA or resection with the LNG-IUD for the treatment of premenopausal women with AUB.3 They found no significant differences between EA and the LNG-IUD in terms of subsequent hysterectomy (risk ratio [RR] = 1.3; 95% confidence interval [CI], 0.60-2.11). It was not surprising that, when looking at age, EA was associated with a higher risk for hysterectomy in women younger than age 42 (RR = 5.26; 95% CI, 1.21-22.91). Conversely, subsequent hysterectomy was less likely with EA compared to LNG-IUD use in women older than 42 years. However, statistical significance was not reached in the older group (RR = 0.51; 95% CI, 0.21-1.24).
In the systematic review by Vitale and colleagues, 9 studies met inclusion criteria for a comparison of EA and hysterectomy, with the objective of ascertaining improvement in quality of life and several other measures.4
Although there was significant heterogeneity between assessment tools, both treatment groups experienced similar improvements in quality of life during the first year. However, hysterectomy was more advantageous in terms of improving uterine bleeding and satisfaction in the long term when compared with EA.4
As EA is considered, it is important to continue to counsel about the efficacy of the LNG-IUD, as well as its decreased associated morbidity. Additionally, EA is particularly less effective in younger women.
Continue to: Laparoscopy is best approach for isthomocele management, with caveats...
Laparoscopy is best approach for isthomocele management, with caveats
He Y, Zhong J, Zhou W, et al. Four surgical strategies for the treatment of cesarean scar defect: a systematic review and network meta-analysis. J Minim Invasive Gynecol. 2020;27:593-602.
Vitale SG, Ludwin A, Vilos GA, et al. From hysteroscopy to laparoendoscopic surgery: what is the best surgical approach for symptomatic isthmocele? A systematic review and meta-analysis. Arch Gynecol Obstet. 2020;301:33-52.
The isthmocele (cesarean scar defect, uterine niche), a known complication of cesarean delivery, represents a myometrial defect in the anterior uterine wall that often presents as abnormal uterine bleeding. It also can be a site for pregnancy-related complications, such as invasive placentation, placenta previa, and uterine rupture.
Two systematic reviews compared surgical strategies for treating isthmocele, including laparoscopy, hysteroscopy, combined laparoscopy and hysteroscopy, laparotomy, and vaginal repair.
Laparoscopy reduced isthmocele-associated AUB better than other techniques
A review by He and colleagues analyzed data from 10 pertinent studies (4 RCTs and 6 observational studies) that included 858 patients in total.5 Treatments compared were laparoscopy, hysteroscopy, combined laparoscopy with hysteroscopy, and vaginal repair for reduction of AUB and isthmocele and diverticulum depth.
The authors found no difference in intraoperative bleeding between the 4 surgical methods (laparotomy was not included in this review). Hysteroscopic surgery was associated with the shortest operative time, while laparoscopy was the longest surgery. In terms of reducing intermittent abnormal bleeding and scar depth, laparoscopic surgery performed better than the other 3 methods.
Approach considerations in isthmocele repair
Vitale and colleagues conducted a systematic review that included 33 publications (28 focused on a single surgical technique, 5 compared different techniques) to examine the effectiveness and risks of various surgical approaches for isthmocele in women with AUB, infertility, or for prevention of obstetric complications.6
Results of their analysis in general favored a laparoscopic approach for patients who desired future fertility, with an improvement rate of 92.7%. Hysteroscopic correction had an 85% improvement rate, and vaginal correction had an 82.5% improvement rate.
Although there were no high-level data to suggest a threshold for myometrial thickness in recommending a surgical approach, the authors provided a helpful algorithm for choosing a route based on a patient's fertility desires. For the asymptomatic patient, they suggest no treatment. In symptomatic patients, the laparoscopic approach is the gold standard but requires significant laparoscopic surgical skill, and a hysteroscopic approach may be considered as an alternative route if the residual myometrial defect is greater than 2.5 to 3.5 mm. For patients who are not considering future reproduction, hysteroscopy is the gold standard as long as the residual myometrial thickness is greater than 2.5 to 3.5 mm.
The takeaway. Of the several methods used for surgical isthmocele management, the laparoscopic approach reduced intermittent abnormal bleeding and scar depth better than other methods. It also was associated with the longest surgical duration. Hysteroscopic surgery was the quickest procedure to perform and is effective in removing the upper valve to promote the elimination of the hematocele and symptoms of abnormal bleeding; however, it does not change the anatomic aspects of the isthmocele in terms of myometrial thickness. Some authors suggested that deciding on the surgical route should be based on fertility desires and the residual thickness of the myometrium. ●
In terms of isthmocele repair, the laparoscopic approach is preferred in patients who desire fertility, as long as the surgeon possesses the skill set to perform this difficult surgery, and as long as the residual myometrium is thicker than 2.5 to 3.5 mm.
- Bofill Rodriguez M, Lethaby A, Low C, et al. Cyclical progestogens for heavy menstrual bleeding. Cochrane Database Syst Rev. 2019;(8):CD001016.
- Lukes AS, Moore KA, Muse KN, et al. Tranexamic acid treatment for heavy menstrual bleeding: a randomized controlled study. Obstet Gynecol. 2010;116:865-875.
- Bergeron C, Laberge PY, Boutin A, et al. Endometrial ablation or resection versus levonorgestrel intra-uterine system for the treatment of women with heavy menstrual bleeding and a normal uterine cavity: a systematic review with meta-analysis. Hum Reprod Update. 2020;26:302-311.
- Vitale SG, Ferrero S, Ciebiera M, et al. Hysteroscopic endometrial resection vs hysterectomy for abnormal uterine bleeding: impact on quality of life and sexuality. Evidence from a systematic review of randomized controlled trials. Curr Opin Obstet Gynecol. 2020;32:159-165.
- He Y, Zhong J, Zhou W, et al. Four surgical strategies for the treatment of cesarean scar defect: a systematic review and network meta-analysis. J Minim Invasive Gynecol. 2020;27:593-602.
- Vitale SG, Ludwin A, Vilos GA, et al. From hysteroscopy to laparoendoscopic surgery: what is the best surgical approach for symptomatic isthmocele? A systematic review and meta-analysis. Arch Gynecol Obstet. 2020;301:33-52.

Abnormal uterine bleeding (AUB) continues to be a top reason that women present for gynecologic care. In general, our approach to the management of AUB is to diagnose causes before we prescribe therapy and to offer conservative therapies initially and progress to more invasive measures if indicated.
In this Update, we highlight several new studies that provide evidence for preferential use of certain medical and surgical therapies. In considering conservative therapy for the treatment of AUB, we take a closer look at the efficacy of cyclic progestogens. Another important issue, as more types of endometrial ablation (EA) are being developed and are coming into the market, is the need for additional guidance regarding decisions about EA versus progestin-releasing intrauterine devices (IUDs). Lastly, an unintended consequence of an increased cesarean delivery rate is the development of isthmocele, also known as cesarean scar defect or uterine niche. These defects, which can be bothersome and cause abnormal bleeding, are treated with various techniques. Within the last year, 2 systematic reviews that compare the efficacy of several different approaches and provide guidance have been published.
Is it time to retire cyclic progestogens for the treatment of heavy menstrual bleeding?
Bofill Rodriguez M, Lethaby A, Low C, et al. Cyclical progestogens for heavy menstrual bleeding. Cochrane Database Syst Rev. 2019;(8):CD001016.
In a recent Cochrane Database Systematic Review, Bofill Rodriguez and colleagues looked at the efficacy, safety, and tolerability of oral progestogen therapy for heavy menstrual bleeding.1 They considered progestogen (medroxyprogesterone acetate or norethisterone) in short-cycle use (7 to 10 days in the luteal phase) and long-cycle use (21 days per cycle) in a review of 15 randomized clinical trials (RCTs) that included a total of 1,071 women. As this topic had not been updated in 12 years, this review was essential in demonstrating changes that occurred over the past decade.
The primary outcomes of the analysis were menstrual blood loss and treatment satisfaction. Secondary outcomes included the number of days of bleeding, quality of life, adherence and acceptability of treatment, adverse events, and costs.
Classic progestogens fall short compared with newer approaches
Analysis of the data revealed that short-cycle progestogen was inferior to treatment with tranexamic acid, danazol, and the 65-µg progesterone-releasing IUD (Pg-IUD). Of note, the 65-µg Pg-IUD has been off the market since 2001, and danazol is rarely used in current practice. Furthermore, based on 2 trials, cyclic progestogens demonstrated no clear benefit over nonsteroidal anti-inflammatory drugs. Additionally, long-cycle progestogen therapy was found to be inferior to the 52-mg levonorgestrel-releasing IUD (LNG-IUD), tranexamic acid, and ormeloxifene.
It should be noted that the quality of evidence is still lacking for progestogen therapy, and this study's main limitation is bias, as the women and the researchers were aware of the treatments that were given. This review is helpful, however, for emphasizing the advantage of tranexamic acid and LNG-IUD use in clinical care.
The takeaway. Although it may not necessarily be time to retire the use of cyclic oral progestogens, the 52-mg LNG-IUD or tranexamic acid may be more successful for treating AUB in women who are appropriate candidates.
Cyclic progestogen therapy appears to be less effective for the treatment of AUB when compared with tranexamic acid and the LNG-IUD. It does not appear to be more helpful than nonsteroidal anti-inflammatory drugs. We frequently offer and prescribe tranexamic acid, 1,300 mg 3 times daily, as a medical alternative to hormonal therapy for up to 5 days monthly for women without thromboembolism risk. Lukes and colleagues published an RCT in 2010 that demonstrated a 40% reduction of bleeding in tranexamic acid–treated women compared with an 8.2% reduction in the placebo group.2
Continue to: Endometrial ablation...
Endometrial ablation: New evidence informs when it could (and could not) be the best option
Bergeron C, Laberge PY, Boutin A, et al. Endometrial ablation or resection versus levonorgestrel intra-uterine system for the treatment of women with heavy menstrual bleeding and a normal uterine cavity: a systematic review with meta-analysis. Hum Reprod Update. 2020;26:302-311.
Vitale SG, Ferrero S, Ciebiera M, et al. Hysteroscopic endometrial resection vs hysterectomy for abnormal uterine bleeding: impact on quality of life and sexuality. Evidence from a systematic review of randomized controlled trials. Curr Opin Obstet Gynecol. 2020;32:159-165.
Two systematic reviews evaluated the efficacy of EA in women with abnormal uterine bleeding. One compared EA with the LNG-IUD and reported on safety and efficacy, while the other compared EA with hysterectomy and reported on quality of life.
Bergeron and colleagues reviewed 13 studies that included 884 women to compare the efficacy and safety of EA or resection with the LNG-IUD for the treatment of premenopausal women with AUB.3 They found no significant differences between EA and the LNG-IUD in terms of subsequent hysterectomy (risk ratio [RR] = 1.3; 95% confidence interval [CI], 0.60-2.11). It was not surprising that, when looking at age, EA was associated with a higher risk for hysterectomy in women younger than age 42 (RR = 5.26; 95% CI, 1.21-22.91). Conversely, subsequent hysterectomy was less likely with EA compared to LNG-IUD use in women older than 42 years. However, statistical significance was not reached in the older group (RR = 0.51; 95% CI, 0.21-1.24).
In the systematic review by Vitale and colleagues, 9 studies met inclusion criteria for a comparison of EA and hysterectomy, with the objective of ascertaining improvement in quality of life and several other measures.4
Although there was significant heterogeneity between assessment tools, both treatment groups experienced similar improvements in quality of life during the first year. However, hysterectomy was more advantageous in terms of improving uterine bleeding and satisfaction in the long term when compared with EA.4
As EA is considered, it is important to continue to counsel about the efficacy of the LNG-IUD, as well as its decreased associated morbidity. Additionally, EA is particularly less effective in younger women.
Continue to: Laparoscopy is best approach for isthomocele management, with caveats...
Laparoscopy is best approach for isthomocele management, with caveats
He Y, Zhong J, Zhou W, et al. Four surgical strategies for the treatment of cesarean scar defect: a systematic review and network meta-analysis. J Minim Invasive Gynecol. 2020;27:593-602.
Vitale SG, Ludwin A, Vilos GA, et al. From hysteroscopy to laparoendoscopic surgery: what is the best surgical approach for symptomatic isthmocele? A systematic review and meta-analysis. Arch Gynecol Obstet. 2020;301:33-52.
The isthmocele (cesarean scar defect, uterine niche), a known complication of cesarean delivery, represents a myometrial defect in the anterior uterine wall that often presents as abnormal uterine bleeding. It also can be a site for pregnancy-related complications, such as invasive placentation, placenta previa, and uterine rupture.
Two systematic reviews compared surgical strategies for treating isthmocele, including laparoscopy, hysteroscopy, combined laparoscopy and hysteroscopy, laparotomy, and vaginal repair.
Laparoscopy reduced isthmocele-associated AUB better than other techniques
A review by He and colleagues analyzed data from 10 pertinent studies (4 RCTs and 6 observational studies) that included 858 patients in total.5 Treatments compared were laparoscopy, hysteroscopy, combined laparoscopy with hysteroscopy, and vaginal repair for reduction of AUB and isthmocele and diverticulum depth.
The authors found no difference in intraoperative bleeding between the 4 surgical methods (laparotomy was not included in this review). Hysteroscopic surgery was associated with the shortest operative time, while laparoscopy was the longest surgery. In terms of reducing intermittent abnormal bleeding and scar depth, laparoscopic surgery performed better than the other 3 methods.
Approach considerations in isthmocele repair
Vitale and colleagues conducted a systematic review that included 33 publications (28 focused on a single surgical technique, 5 compared different techniques) to examine the effectiveness and risks of various surgical approaches for isthmocele in women with AUB, infertility, or for prevention of obstetric complications.6
Results of their analysis in general favored a laparoscopic approach for patients who desired future fertility, with an improvement rate of 92.7%. Hysteroscopic correction had an 85% improvement rate, and vaginal correction had an 82.5% improvement rate.
Although there were no high-level data to suggest a threshold for myometrial thickness in recommending a surgical approach, the authors provided a helpful algorithm for choosing a route based on a patient's fertility desires. For the asymptomatic patient, they suggest no treatment. In symptomatic patients, the laparoscopic approach is the gold standard but requires significant laparoscopic surgical skill, and a hysteroscopic approach may be considered as an alternative route if the residual myometrial defect is greater than 2.5 to 3.5 mm. For patients who are not considering future reproduction, hysteroscopy is the gold standard as long as the residual myometrial thickness is greater than 2.5 to 3.5 mm.
The takeaway. Of the several methods used for surgical isthmocele management, the laparoscopic approach reduced intermittent abnormal bleeding and scar depth better than other methods. It also was associated with the longest surgical duration. Hysteroscopic surgery was the quickest procedure to perform and is effective in removing the upper valve to promote the elimination of the hematocele and symptoms of abnormal bleeding; however, it does not change the anatomic aspects of the isthmocele in terms of myometrial thickness. Some authors suggested that deciding on the surgical route should be based on fertility desires and the residual thickness of the myometrium. ●
In terms of isthmocele repair, the laparoscopic approach is preferred in patients who desire fertility, as long as the surgeon possesses the skill set to perform this difficult surgery, and as long as the residual myometrium is thicker than 2.5 to 3.5 mm.

Abnormal uterine bleeding (AUB) continues to be a top reason that women present for gynecologic care. In general, our approach to the management of AUB is to diagnose causes before we prescribe therapy and to offer conservative therapies initially and progress to more invasive measures if indicated.
In this Update, we highlight several new studies that provide evidence for preferential use of certain medical and surgical therapies. In considering conservative therapy for the treatment of AUB, we take a closer look at the efficacy of cyclic progestogens. Another important issue, as more types of endometrial ablation (EA) are being developed and are coming into the market, is the need for additional guidance regarding decisions about EA versus progestin-releasing intrauterine devices (IUDs). Lastly, an unintended consequence of an increased cesarean delivery rate is the development of isthmocele, also known as cesarean scar defect or uterine niche. These defects, which can be bothersome and cause abnormal bleeding, are treated with various techniques. Within the last year, 2 systematic reviews that compare the efficacy of several different approaches and provide guidance have been published.
Is it time to retire cyclic progestogens for the treatment of heavy menstrual bleeding?
Bofill Rodriguez M, Lethaby A, Low C, et al. Cyclical progestogens for heavy menstrual bleeding. Cochrane Database Syst Rev. 2019;(8):CD001016.
In a recent Cochrane Database Systematic Review, Bofill Rodriguez and colleagues looked at the efficacy, safety, and tolerability of oral progestogen therapy for heavy menstrual bleeding.1 They considered progestogen (medroxyprogesterone acetate or norethisterone) in short-cycle use (7 to 10 days in the luteal phase) and long-cycle use (21 days per cycle) in a review of 15 randomized clinical trials (RCTs) that included a total of 1,071 women. As this topic had not been updated in 12 years, this review was essential in demonstrating changes that occurred over the past decade.
The primary outcomes of the analysis were menstrual blood loss and treatment satisfaction. Secondary outcomes included the number of days of bleeding, quality of life, adherence and acceptability of treatment, adverse events, and costs.
Classic progestogens fall short compared with newer approaches
Analysis of the data revealed that short-cycle progestogen was inferior to treatment with tranexamic acid, danazol, and the 65-µg progesterone-releasing IUD (Pg-IUD). Of note, the 65-µg Pg-IUD has been off the market since 2001, and danazol is rarely used in current practice. Furthermore, based on 2 trials, cyclic progestogens demonstrated no clear benefit over nonsteroidal anti-inflammatory drugs. Additionally, long-cycle progestogen therapy was found to be inferior to the 52-mg levonorgestrel-releasing IUD (LNG-IUD), tranexamic acid, and ormeloxifene.
It should be noted that the quality of evidence is still lacking for progestogen therapy, and this study's main limitation is bias, as the women and the researchers were aware of the treatments that were given. This review is helpful, however, for emphasizing the advantage of tranexamic acid and LNG-IUD use in clinical care.
The takeaway. Although it may not necessarily be time to retire the use of cyclic oral progestogens, the 52-mg LNG-IUD or tranexamic acid may be more successful for treating AUB in women who are appropriate candidates.
Cyclic progestogen therapy appears to be less effective for the treatment of AUB when compared with tranexamic acid and the LNG-IUD. It does not appear to be more helpful than nonsteroidal anti-inflammatory drugs. We frequently offer and prescribe tranexamic acid, 1,300 mg 3 times daily, as a medical alternative to hormonal therapy for up to 5 days monthly for women without thromboembolism risk. Lukes and colleagues published an RCT in 2010 that demonstrated a 40% reduction of bleeding in tranexamic acid–treated women compared with an 8.2% reduction in the placebo group.2
Continue to: Endometrial ablation...
Endometrial ablation: New evidence informs when it could (and could not) be the best option
Bergeron C, Laberge PY, Boutin A, et al. Endometrial ablation or resection versus levonorgestrel intra-uterine system for the treatment of women with heavy menstrual bleeding and a normal uterine cavity: a systematic review with meta-analysis. Hum Reprod Update. 2020;26:302-311.
Vitale SG, Ferrero S, Ciebiera M, et al. Hysteroscopic endometrial resection vs hysterectomy for abnormal uterine bleeding: impact on quality of life and sexuality. Evidence from a systematic review of randomized controlled trials. Curr Opin Obstet Gynecol. 2020;32:159-165.
Two systematic reviews evaluated the efficacy of EA in women with abnormal uterine bleeding. One compared EA with the LNG-IUD and reported on safety and efficacy, while the other compared EA with hysterectomy and reported on quality of life.
Bergeron and colleagues reviewed 13 studies that included 884 women to compare the efficacy and safety of EA or resection with the LNG-IUD for the treatment of premenopausal women with AUB.3 They found no significant differences between EA and the LNG-IUD in terms of subsequent hysterectomy (risk ratio [RR] = 1.3; 95% confidence interval [CI], 0.60-2.11). It was not surprising that, when looking at age, EA was associated with a higher risk for hysterectomy in women younger than age 42 (RR = 5.26; 95% CI, 1.21-22.91). Conversely, subsequent hysterectomy was less likely with EA compared to LNG-IUD use in women older than 42 years. However, statistical significance was not reached in the older group (RR = 0.51; 95% CI, 0.21-1.24).
In the systematic review by Vitale and colleagues, 9 studies met inclusion criteria for a comparison of EA and hysterectomy, with the objective of ascertaining improvement in quality of life and several other measures.4
Although there was significant heterogeneity between assessment tools, both treatment groups experienced similar improvements in quality of life during the first year. However, hysterectomy was more advantageous in terms of improving uterine bleeding and satisfaction in the long term when compared with EA.4
As EA is considered, it is important to continue to counsel about the efficacy of the LNG-IUD, as well as its decreased associated morbidity. Additionally, EA is particularly less effective in younger women.
Continue to: Laparoscopy is best approach for isthomocele management, with caveats...
Laparoscopy is best approach for isthomocele management, with caveats
He Y, Zhong J, Zhou W, et al. Four surgical strategies for the treatment of cesarean scar defect: a systematic review and network meta-analysis. J Minim Invasive Gynecol. 2020;27:593-602.
Vitale SG, Ludwin A, Vilos GA, et al. From hysteroscopy to laparoendoscopic surgery: what is the best surgical approach for symptomatic isthmocele? A systematic review and meta-analysis. Arch Gynecol Obstet. 2020;301:33-52.
The isthmocele (cesarean scar defect, uterine niche), a known complication of cesarean delivery, represents a myometrial defect in the anterior uterine wall that often presents as abnormal uterine bleeding. It also can be a site for pregnancy-related complications, such as invasive placentation, placenta previa, and uterine rupture.
Two systematic reviews compared surgical strategies for treating isthmocele, including laparoscopy, hysteroscopy, combined laparoscopy and hysteroscopy, laparotomy, and vaginal repair.
Laparoscopy reduced isthmocele-associated AUB better than other techniques
A review by He and colleagues analyzed data from 10 pertinent studies (4 RCTs and 6 observational studies) that included 858 patients in total.5 Treatments compared were laparoscopy, hysteroscopy, combined laparoscopy with hysteroscopy, and vaginal repair for reduction of AUB and isthmocele and diverticulum depth.
The authors found no difference in intraoperative bleeding between the 4 surgical methods (laparotomy was not included in this review). Hysteroscopic surgery was associated with the shortest operative time, while laparoscopy was the longest surgery. In terms of reducing intermittent abnormal bleeding and scar depth, laparoscopic surgery performed better than the other 3 methods.
Approach considerations in isthmocele repair
Vitale and colleagues conducted a systematic review that included 33 publications (28 focused on a single surgical technique, 5 compared different techniques) to examine the effectiveness and risks of various surgical approaches for isthmocele in women with AUB, infertility, or for prevention of obstetric complications.6
Results of their analysis in general favored a laparoscopic approach for patients who desired future fertility, with an improvement rate of 92.7%. Hysteroscopic correction had an 85% improvement rate, and vaginal correction had an 82.5% improvement rate.
Although there were no high-level data to suggest a threshold for myometrial thickness in recommending a surgical approach, the authors provided a helpful algorithm for choosing a route based on a patient's fertility desires. For the asymptomatic patient, they suggest no treatment. In symptomatic patients, the laparoscopic approach is the gold standard but requires significant laparoscopic surgical skill, and a hysteroscopic approach may be considered as an alternative route if the residual myometrial defect is greater than 2.5 to 3.5 mm. For patients who are not considering future reproduction, hysteroscopy is the gold standard as long as the residual myometrial thickness is greater than 2.5 to 3.5 mm.
The takeaway. Of the several methods used for surgical isthmocele management, the laparoscopic approach reduced intermittent abnormal bleeding and scar depth better than other methods. It also was associated with the longest surgical duration. Hysteroscopic surgery was the quickest procedure to perform and is effective in removing the upper valve to promote the elimination of the hematocele and symptoms of abnormal bleeding; however, it does not change the anatomic aspects of the isthmocele in terms of myometrial thickness. Some authors suggested that deciding on the surgical route should be based on fertility desires and the residual thickness of the myometrium. ●
In terms of isthmocele repair, the laparoscopic approach is preferred in patients who desire fertility, as long as the surgeon possesses the skill set to perform this difficult surgery, and as long as the residual myometrium is thicker than 2.5 to 3.5 mm.
- Bofill Rodriguez M, Lethaby A, Low C, et al. Cyclical progestogens for heavy menstrual bleeding. Cochrane Database Syst Rev. 2019;(8):CD001016.
- Lukes AS, Moore KA, Muse KN, et al. Tranexamic acid treatment for heavy menstrual bleeding: a randomized controlled study. Obstet Gynecol. 2010;116:865-875.
- Bergeron C, Laberge PY, Boutin A, et al. Endometrial ablation or resection versus levonorgestrel intra-uterine system for the treatment of women with heavy menstrual bleeding and a normal uterine cavity: a systematic review with meta-analysis. Hum Reprod Update. 2020;26:302-311.
- Vitale SG, Ferrero S, Ciebiera M, et al. Hysteroscopic endometrial resection vs hysterectomy for abnormal uterine bleeding: impact on quality of life and sexuality. Evidence from a systematic review of randomized controlled trials. Curr Opin Obstet Gynecol. 2020;32:159-165.
- He Y, Zhong J, Zhou W, et al. Four surgical strategies for the treatment of cesarean scar defect: a systematic review and network meta-analysis. J Minim Invasive Gynecol. 2020;27:593-602.
- Vitale SG, Ludwin A, Vilos GA, et al. From hysteroscopy to laparoendoscopic surgery: what is the best surgical approach for symptomatic isthmocele? A systematic review and meta-analysis. Arch Gynecol Obstet. 2020;301:33-52.
- Bofill Rodriguez M, Lethaby A, Low C, et al. Cyclical progestogens for heavy menstrual bleeding. Cochrane Database Syst Rev. 2019;(8):CD001016.
- Lukes AS, Moore KA, Muse KN, et al. Tranexamic acid treatment for heavy menstrual bleeding: a randomized controlled study. Obstet Gynecol. 2010;116:865-875.
- Bergeron C, Laberge PY, Boutin A, et al. Endometrial ablation or resection versus levonorgestrel intra-uterine system for the treatment of women with heavy menstrual bleeding and a normal uterine cavity: a systematic review with meta-analysis. Hum Reprod Update. 2020;26:302-311.
- Vitale SG, Ferrero S, Ciebiera M, et al. Hysteroscopic endometrial resection vs hysterectomy for abnormal uterine bleeding: impact on quality of life and sexuality. Evidence from a systematic review of randomized controlled trials. Curr Opin Obstet Gynecol. 2020;32:159-165.
- He Y, Zhong J, Zhou W, et al. Four surgical strategies for the treatment of cesarean scar defect: a systematic review and network meta-analysis. J Minim Invasive Gynecol. 2020;27:593-602.
- Vitale SG, Ludwin A, Vilos GA, et al. From hysteroscopy to laparoendoscopic surgery: what is the best surgical approach for symptomatic isthmocele? A systematic review and meta-analysis. Arch Gynecol Obstet. 2020;301:33-52.
Should all women with a history of OASI have a mediolateral episiotomy at their subsequent delivery?
Van Bavel J, Ravelli AC, Abu-Hanna A, et al. Risk factors for the recurrence of obstetrical anal sphincter injury and the role of a mediolateral episiotomy: an analysis of a national registry. BJOG. 2020;127:951-956.
EXPERT COMMENTARY
Women with a history of OASI are at increased risk for recurrence in a subsequent delivery. Higher rates of anal and fecal incontinence are reported in women with recurrent OASI (rOASI) compared with women who had an OASI only in their first delivery. Previous studies have reported recurrence rates of 5% to 7%,1 and some suggested that MLE may be protective, but standardized recommendations for mode of delivery and use of MLE currently are not available.
Recently, van Bavel and colleagues sought to determine the rate of rOASI in their population as well as the factors that increase and decrease the risk of this complication.
Details of the study
This cohort study used data from the Dutch Perinatal Registry (Perined) that included 268,607 women who had their first and second deliveries (singleton, term, vertex, < 43 weeks) vaginally in 2000–2009. The study’s primary objective was to determine the rate of rOASI in women who had OASI in their first delivery. The secondary objectives were to identify risk factors for rOASI and to assess the effect of MLE. For the purposes of this study, OASI was defined as subtotal and total rupture of the perineum, or grades 3A-4 as defined by the Royal College of Obstetricians and Gynaecologists.2
Within this cohort, 9,943 women had an OASI in their first delivery (4%), and the rate of rOASI was 5.8% (579 of 9,943). After multivariate analysis, the risk factors for rOASI were birth weight of 4,000 g or greater (odds ratio [OR], 2.1; 95% confidence interval [CI], 1.6–2.6) and duration of the second stage of labor of 30 minutes or longer (OR, 1.8; 95% CI, 1.4–2.3).
The MLE rate was 40.8% (4,054 of 9,943) and was associated with a lower rate of rOASI (OR, 0.3; 95% CI, 0.3–0.4). This association persisted when delivery type was separated into spontaneous and operative vaginal deliveries, with the number of MLEs needed to prevent one rOASI of 22 and 8, respectively. Birth weight of less than 3,000 g also was noted to be protective against rOASI (OR, 0.5; 95% CI, 0.3–0.9).
Based on these findings, as well as comparisons to previous studies, the authors concluded that MLE could be considered for routine use or at least discussed with all women with a prior OASI for prevention of rOASI.
Continue to: Study strengths and limitations...
Study strengths and limitations
A strength of this study was the large number of deliveries and the wide variation of practice included in the registry database, which promotes the generalizability of the results and reduces bias. This also provides an adequate base on which to determine an accurate rate of rOASI in the Dutch population.
One study limitation is that information is not available regarding how the episiotomies were performed (specifically, angle of incision), delivery techniques (“hands on” vs “hands off”), and indication for the episiotomy. Additional limitations suggested are that clinicians who perform an episiotomy may have an inherent bias regarding the protective nature of the procedure and may miss a rOASI due to inadequate examination postprocedure, overestimating its protective effect.
Finally, the relatively high rate of MLE and low rate of cesarean delivery (6.9%) in this study are specific to the Netherlands and do not reflect the obstetric practices used in many other countries. Generalizability of these results in the context of much lower MLE and higher cesarean delivery rates (as in the United States) would therefore be in question.●
Prevention of rOASI is important, as fecal incontinence is debilitating and difficult to treat. While this study provides evidence that MLE may protect against this complication, its results may not be generalizable to all patient or clinician populations. Differences in baseline rate of MLE and cesarean delivery, technique, indication, and comfort with repair—all not evaluated in this study—must be taken into account when counseling OASI patients about their options for delivery and the use of MLE in a subsequent pregnancy.
JAIMEY M. PAULI, MD
- Van Bavel J, Ravelli AC, Abu-Hanna A, et al. Risk factors for the recurrence of obstetrical anal sphincter injury and the role of a mediolateral episiotomy: an analysis of a national registry. BJOG. 2020;127:951-956.
- Royal College of Obstetricians and Gynaecologists. Green-top guideline No. 29: the management of third- and fourth-degree perineal tears. June 2014. https://www.rcog.org.uk/globalassets/documents/guidelines/gtg-29.pdf. Accessed June 12, 2020.
Van Bavel J, Ravelli AC, Abu-Hanna A, et al. Risk factors for the recurrence of obstetrical anal sphincter injury and the role of a mediolateral episiotomy: an analysis of a national registry. BJOG. 2020;127:951-956.
EXPERT COMMENTARY
Women with a history of OASI are at increased risk for recurrence in a subsequent delivery. Higher rates of anal and fecal incontinence are reported in women with recurrent OASI (rOASI) compared with women who had an OASI only in their first delivery. Previous studies have reported recurrence rates of 5% to 7%,1 and some suggested that MLE may be protective, but standardized recommendations for mode of delivery and use of MLE currently are not available.
Recently, van Bavel and colleagues sought to determine the rate of rOASI in their population as well as the factors that increase and decrease the risk of this complication.
Details of the study
This cohort study used data from the Dutch Perinatal Registry (Perined) that included 268,607 women who had their first and second deliveries (singleton, term, vertex, < 43 weeks) vaginally in 2000–2009. The study’s primary objective was to determine the rate of rOASI in women who had OASI in their first delivery. The secondary objectives were to identify risk factors for rOASI and to assess the effect of MLE. For the purposes of this study, OASI was defined as subtotal and total rupture of the perineum, or grades 3A-4 as defined by the Royal College of Obstetricians and Gynaecologists.2
Within this cohort, 9,943 women had an OASI in their first delivery (4%), and the rate of rOASI was 5.8% (579 of 9,943). After multivariate analysis, the risk factors for rOASI were birth weight of 4,000 g or greater (odds ratio [OR], 2.1; 95% confidence interval [CI], 1.6–2.6) and duration of the second stage of labor of 30 minutes or longer (OR, 1.8; 95% CI, 1.4–2.3).
The MLE rate was 40.8% (4,054 of 9,943) and was associated with a lower rate of rOASI (OR, 0.3; 95% CI, 0.3–0.4). This association persisted when delivery type was separated into spontaneous and operative vaginal deliveries, with the number of MLEs needed to prevent one rOASI of 22 and 8, respectively. Birth weight of less than 3,000 g also was noted to be protective against rOASI (OR, 0.5; 95% CI, 0.3–0.9).
Based on these findings, as well as comparisons to previous studies, the authors concluded that MLE could be considered for routine use or at least discussed with all women with a prior OASI for prevention of rOASI.
Continue to: Study strengths and limitations...
Study strengths and limitations
A strength of this study was the large number of deliveries and the wide variation of practice included in the registry database, which promotes the generalizability of the results and reduces bias. This also provides an adequate base on which to determine an accurate rate of rOASI in the Dutch population.
One study limitation is that information is not available regarding how the episiotomies were performed (specifically, angle of incision), delivery techniques (“hands on” vs “hands off”), and indication for the episiotomy. Additional limitations suggested are that clinicians who perform an episiotomy may have an inherent bias regarding the protective nature of the procedure and may miss a rOASI due to inadequate examination postprocedure, overestimating its protective effect.
Finally, the relatively high rate of MLE and low rate of cesarean delivery (6.9%) in this study are specific to the Netherlands and do not reflect the obstetric practices used in many other countries. Generalizability of these results in the context of much lower MLE and higher cesarean delivery rates (as in the United States) would therefore be in question.●
Prevention of rOASI is important, as fecal incontinence is debilitating and difficult to treat. While this study provides evidence that MLE may protect against this complication, its results may not be generalizable to all patient or clinician populations. Differences in baseline rate of MLE and cesarean delivery, technique, indication, and comfort with repair—all not evaluated in this study—must be taken into account when counseling OASI patients about their options for delivery and the use of MLE in a subsequent pregnancy.
JAIMEY M. PAULI, MD
Van Bavel J, Ravelli AC, Abu-Hanna A, et al. Risk factors for the recurrence of obstetrical anal sphincter injury and the role of a mediolateral episiotomy: an analysis of a national registry. BJOG. 2020;127:951-956.
EXPERT COMMENTARY
Women with a history of OASI are at increased risk for recurrence in a subsequent delivery. Higher rates of anal and fecal incontinence are reported in women with recurrent OASI (rOASI) compared with women who had an OASI only in their first delivery. Previous studies have reported recurrence rates of 5% to 7%,1 and some suggested that MLE may be protective, but standardized recommendations for mode of delivery and use of MLE currently are not available.
Recently, van Bavel and colleagues sought to determine the rate of rOASI in their population as well as the factors that increase and decrease the risk of this complication.
Details of the study
This cohort study used data from the Dutch Perinatal Registry (Perined) that included 268,607 women who had their first and second deliveries (singleton, term, vertex, < 43 weeks) vaginally in 2000–2009. The study’s primary objective was to determine the rate of rOASI in women who had OASI in their first delivery. The secondary objectives were to identify risk factors for rOASI and to assess the effect of MLE. For the purposes of this study, OASI was defined as subtotal and total rupture of the perineum, or grades 3A-4 as defined by the Royal College of Obstetricians and Gynaecologists.2
Within this cohort, 9,943 women had an OASI in their first delivery (4%), and the rate of rOASI was 5.8% (579 of 9,943). After multivariate analysis, the risk factors for rOASI were birth weight of 4,000 g or greater (odds ratio [OR], 2.1; 95% confidence interval [CI], 1.6–2.6) and duration of the second stage of labor of 30 minutes or longer (OR, 1.8; 95% CI, 1.4–2.3).
The MLE rate was 40.8% (4,054 of 9,943) and was associated with a lower rate of rOASI (OR, 0.3; 95% CI, 0.3–0.4). This association persisted when delivery type was separated into spontaneous and operative vaginal deliveries, with the number of MLEs needed to prevent one rOASI of 22 and 8, respectively. Birth weight of less than 3,000 g also was noted to be protective against rOASI (OR, 0.5; 95% CI, 0.3–0.9).
Based on these findings, as well as comparisons to previous studies, the authors concluded that MLE could be considered for routine use or at least discussed with all women with a prior OASI for prevention of rOASI.
Continue to: Study strengths and limitations...
Study strengths and limitations
A strength of this study was the large number of deliveries and the wide variation of practice included in the registry database, which promotes the generalizability of the results and reduces bias. This also provides an adequate base on which to determine an accurate rate of rOASI in the Dutch population.
One study limitation is that information is not available regarding how the episiotomies were performed (specifically, angle of incision), delivery techniques (“hands on” vs “hands off”), and indication for the episiotomy. Additional limitations suggested are that clinicians who perform an episiotomy may have an inherent bias regarding the protective nature of the procedure and may miss a rOASI due to inadequate examination postprocedure, overestimating its protective effect.
Finally, the relatively high rate of MLE and low rate of cesarean delivery (6.9%) in this study are specific to the Netherlands and do not reflect the obstetric practices used in many other countries. Generalizability of these results in the context of much lower MLE and higher cesarean delivery rates (as in the United States) would therefore be in question.●
Prevention of rOASI is important, as fecal incontinence is debilitating and difficult to treat. While this study provides evidence that MLE may protect against this complication, its results may not be generalizable to all patient or clinician populations. Differences in baseline rate of MLE and cesarean delivery, technique, indication, and comfort with repair—all not evaluated in this study—must be taken into account when counseling OASI patients about their options for delivery and the use of MLE in a subsequent pregnancy.
JAIMEY M. PAULI, MD
- Van Bavel J, Ravelli AC, Abu-Hanna A, et al. Risk factors for the recurrence of obstetrical anal sphincter injury and the role of a mediolateral episiotomy: an analysis of a national registry. BJOG. 2020;127:951-956.
- Royal College of Obstetricians and Gynaecologists. Green-top guideline No. 29: the management of third- and fourth-degree perineal tears. June 2014. https://www.rcog.org.uk/globalassets/documents/guidelines/gtg-29.pdf. Accessed June 12, 2020.
- Van Bavel J, Ravelli AC, Abu-Hanna A, et al. Risk factors for the recurrence of obstetrical anal sphincter injury and the role of a mediolateral episiotomy: an analysis of a national registry. BJOG. 2020;127:951-956.
- Royal College of Obstetricians and Gynaecologists. Green-top guideline No. 29: the management of third- and fourth-degree perineal tears. June 2014. https://www.rcog.org.uk/globalassets/documents/guidelines/gtg-29.pdf. Accessed June 12, 2020.
How effective is elagolix treatment in women with fibroids and HMB?
Simon JA, Al-Hendy A, Archer DF, et al. Elagolix treatment for up to 12 months in women with heavy menstrual bleeding and uterine leiomyomas. Obstet Gynecol. 2020;135:1313-1326.
Expert Commentary
Uterine fibroids are common (occurring in up to 80% of reproductive-age women),1,2 and often associated with heavy menstrual bleeding (HMB). There are surgical and medical options, but typically medical options are used for short periods of time. Elagolix with hormonal add-back therapy was recently approved (May 29, 2020) by the US Food and Drug Administration (FDA) for treatment of HMB in women with uterine fibroids for up to 24 months.
Elagolix is an oral, nonpeptide gonadotropin-releasing hormone antagonist that results in a dose-dependent reduction of gonadotropins and ovarian sex hormones. There are now 2 approved products containing elagolix, with different indications:
- Orilissa. Elagolix was approved in 2018 by the FDA for moderate to severe pain associated with endometriosis. For that indication there are 2 dose options of elagolix (150 mg for up to 2 years and 200 mg for up to 6 months) and there is no hormonal add-back therapy.
- Oriahnn. Elagolix and hormonal add-back therapy was approved in 2020 for HMB associated with uterine fibroids for up to 24 months. The total daily dose of elagolix is 600 mg (elagolix 300 mg in the morning with estradiol 1 mg/norethindrone acetate 0.5 mg and then in the evening elagolix 300 mg and no hormonal add-back).
This new class of drug, GnRH antagonist, is an important one for women’s health, and emerging science will continue to expand its potential uses, such as in reproductive health, as well as long-term efficacy and safety. The difference in daily dose of elagolix for endometriosis (150 mg for 24 months) compared with HMB associated with fibroids (600 mg for 24 months) is why the hormonal add-back therapy is important and allows for protection of bone density.
This is an important manuscript because it highlights a medical option for women with HMB associated with fibroids, which can be used for a long period of time. Further, the improvement in bleeding is both impressive and maintained in the extension study. Approximately 90% of women show improvement in their menstrual bleeding associated with fibroids.
The question of what to do after 24 months of therapy with elagolix and hormonal add-back therapy is an important one, but providers should recognize that the limiting factor with this elagolix and hormonal add-back therapy is bone mineral density (BMD). We will only learn more and more moving forward if this is a clinically meaningful reason for stopping treatment at 24 months. The FDA takes a strict view of safety, and providers must weigh this with the benefit of therapy.
One other highlight between the 2 approved medications is that Orilissa does not have a black box warning, given that there is no hormonal add-back therapy. Oriahnn does have a warning, regarding thromboembolic disorders and vascular events:
- Estrogen and progestin combinations, including Oriahnn, increase the risk of thrombotic or thromboembolic disorders, especially in women at increased risk for these events.
- Oriahnn is contraindicated in women with current or a history of thrombotic or thromboembolic disorders and in women at increased risk for these events, including women over 35 years of age who smoke or women with uncontrolled hypertension.
Continue to: Details about the study...
Details about the study
The study by Simon et al is an extension study (UF-EXTEND), in that women could participate if they had completed 1 of the 2 pivotal studies on elagolix. The pivotal studies (Elaris UF1 and UF2) were both randomized, double-blinded, placebo-controlled studies with up to 6 months of therapy; for UF-EXTEND, however, participants were randomly assigned to either combined elagolix and hormone replacement therapy or elagolix alone for an additional 6 months of therapy. Although it was known that all participants would receive elagolix in UF-EXTEND, those who received hormonal add-back therapy were blinded. All women were then followed up for an additional 12 months after treatment ended.
The efficacy of elagolix was measured by the objective alkaline hematin method for menstrual blood loss with the a priori coprimary endpoints. The elagolix and hormonal add-back therapy group showed objective improvement in menstrual blood loss at 12 months in 87.9% of women in the extension study (89.4% in the elagolix alone group). This compares with 72.2% improvement at 6 months of treatment in the UF1 and UF2 studies for those taking elagolix and hormonal add-back therapy. These findings illustrate maintenance of the efficacy seen within the 6-month pivotal studies using elagolix over an extended amount of time.
The safety of elagolix also was demonstrated in UF-EXTEND. The 3 most common adverse events were similar to those found in Elaris UF1 and UF2 and included hot flushes, headache, and nausea. In the elagolix and hormonal add-back therapy group during the extension study, the percentage with hot flushes was 7%, headache 6%, and nausea 4%. These are small percentages, which is encouraging for providers and women with HMB associated with fibroids.
Effects on bone density
Bone density was evaluated at baseline in the UF1 and UF2 studies, through treatment, and then 12 months after the extended treatment was stopped. The hormonal add-back therapy of estradiol 1 mg/norethindrone acetate 0.5 mg significantly protected bone density. Some women did not have a decrease in bone density, but for those who did the average was less than 5% for the lumbar spine. The lumbar spine is considered the most reactive, so this illustrates the safety that combined therapy offers women with HMB and fibroids.
The lumbar spine is considered the most reactive, so this site is often used as the main focus with BMD studies. As Simon et al show, the lumbar spine mean BMD percent change from baseline for the elagolix with add-back therapy was -1.5% (95% confidence interval [CI], -1.9 to -1.0) in women who received up to 12 months of treatment at month 6 in the extension study. After stopping elagolix with add-back therapy, at 6 months the elagolix with add-back therapy had a Z-score of -0.6% (95% CI, -1.1 to -0.1). This shows a trend toward baseline, or a recovery within a short time from stopping medication.
Continue to: Study strengths and limitations...
Study strengths and limitations
Strengths of this study include its overall design; efficacy endpoints, which were all established a priori; the fact that measurement of menstrual blood loss was done with the objective alkaline hematin method; and the statistical analysis, which is thorough and well presented. This extension study allowed further evaluation of efficacy and safety for elagolix. Although the authors point out that there may be some selection bias in an extension study, the fact that so many women elected to continue into the extended study is a positive reflection of the treatment.
As providers learn of new therapies for management of HMB associated with fibroids, it is important to consider who will benefit the most. In my opinion, any woman with heavy periods associated with fibroids could be a candidate for elagolix with add-back therapy. This treatment is highly effective, well tolerated, and safe. My approach to management includes educating a woman on all potential therapies and this new option of elagolix and add-back therapy is an important one. The decision for an individual woman on how to manage heavy periods associated with fibroids should consider her contraceptive needs, medical issues, and the risk and benefit of individual therapies. ●
Elagolix and hormonal add-back therapy offer a long-term medical option for women with HMB associated with fibroids that is both effective and safe.
ANDREA S. LUKES, MD, MHSc
- Stewart EA, Nicholson WK, Bradley L, et al. The burden of uterine fibroids for African-American women: results of a national survey. J Women’s Health. 2013;22:807-816.
- Baird DD, Dunson DB, Hill MC, et al. High cumulative incidence of uterine leiomyoma in black and white women: ultrasound evidence. Am J Obstet Gynecol. 2003;188:100-107.
Simon JA, Al-Hendy A, Archer DF, et al. Elagolix treatment for up to 12 months in women with heavy menstrual bleeding and uterine leiomyomas. Obstet Gynecol. 2020;135:1313-1326.
Expert Commentary
Uterine fibroids are common (occurring in up to 80% of reproductive-age women),1,2 and often associated with heavy menstrual bleeding (HMB). There are surgical and medical options, but typically medical options are used for short periods of time. Elagolix with hormonal add-back therapy was recently approved (May 29, 2020) by the US Food and Drug Administration (FDA) for treatment of HMB in women with uterine fibroids for up to 24 months.
Elagolix is an oral, nonpeptide gonadotropin-releasing hormone antagonist that results in a dose-dependent reduction of gonadotropins and ovarian sex hormones. There are now 2 approved products containing elagolix, with different indications:
- Orilissa. Elagolix was approved in 2018 by the FDA for moderate to severe pain associated with endometriosis. For that indication there are 2 dose options of elagolix (150 mg for up to 2 years and 200 mg for up to 6 months) and there is no hormonal add-back therapy.
- Oriahnn. Elagolix and hormonal add-back therapy was approved in 2020 for HMB associated with uterine fibroids for up to 24 months. The total daily dose of elagolix is 600 mg (elagolix 300 mg in the morning with estradiol 1 mg/norethindrone acetate 0.5 mg and then in the evening elagolix 300 mg and no hormonal add-back).
This new class of drug, GnRH antagonist, is an important one for women’s health, and emerging science will continue to expand its potential uses, such as in reproductive health, as well as long-term efficacy and safety. The difference in daily dose of elagolix for endometriosis (150 mg for 24 months) compared with HMB associated with fibroids (600 mg for 24 months) is why the hormonal add-back therapy is important and allows for protection of bone density.
This is an important manuscript because it highlights a medical option for women with HMB associated with fibroids, which can be used for a long period of time. Further, the improvement in bleeding is both impressive and maintained in the extension study. Approximately 90% of women show improvement in their menstrual bleeding associated with fibroids.
The question of what to do after 24 months of therapy with elagolix and hormonal add-back therapy is an important one, but providers should recognize that the limiting factor with this elagolix and hormonal add-back therapy is bone mineral density (BMD). We will only learn more and more moving forward if this is a clinically meaningful reason for stopping treatment at 24 months. The FDA takes a strict view of safety, and providers must weigh this with the benefit of therapy.
One other highlight between the 2 approved medications is that Orilissa does not have a black box warning, given that there is no hormonal add-back therapy. Oriahnn does have a warning, regarding thromboembolic disorders and vascular events:
- Estrogen and progestin combinations, including Oriahnn, increase the risk of thrombotic or thromboembolic disorders, especially in women at increased risk for these events.
- Oriahnn is contraindicated in women with current or a history of thrombotic or thromboembolic disorders and in women at increased risk for these events, including women over 35 years of age who smoke or women with uncontrolled hypertension.
Continue to: Details about the study...
Details about the study
The study by Simon et al is an extension study (UF-EXTEND), in that women could participate if they had completed 1 of the 2 pivotal studies on elagolix. The pivotal studies (Elaris UF1 and UF2) were both randomized, double-blinded, placebo-controlled studies with up to 6 months of therapy; for UF-EXTEND, however, participants were randomly assigned to either combined elagolix and hormone replacement therapy or elagolix alone for an additional 6 months of therapy. Although it was known that all participants would receive elagolix in UF-EXTEND, those who received hormonal add-back therapy were blinded. All women were then followed up for an additional 12 months after treatment ended.
The efficacy of elagolix was measured by the objective alkaline hematin method for menstrual blood loss with the a priori coprimary endpoints. The elagolix and hormonal add-back therapy group showed objective improvement in menstrual blood loss at 12 months in 87.9% of women in the extension study (89.4% in the elagolix alone group). This compares with 72.2% improvement at 6 months of treatment in the UF1 and UF2 studies for those taking elagolix and hormonal add-back therapy. These findings illustrate maintenance of the efficacy seen within the 6-month pivotal studies using elagolix over an extended amount of time.
The safety of elagolix also was demonstrated in UF-EXTEND. The 3 most common adverse events were similar to those found in Elaris UF1 and UF2 and included hot flushes, headache, and nausea. In the elagolix and hormonal add-back therapy group during the extension study, the percentage with hot flushes was 7%, headache 6%, and nausea 4%. These are small percentages, which is encouraging for providers and women with HMB associated with fibroids.
Effects on bone density
Bone density was evaluated at baseline in the UF1 and UF2 studies, through treatment, and then 12 months after the extended treatment was stopped. The hormonal add-back therapy of estradiol 1 mg/norethindrone acetate 0.5 mg significantly protected bone density. Some women did not have a decrease in bone density, but for those who did the average was less than 5% for the lumbar spine. The lumbar spine is considered the most reactive, so this illustrates the safety that combined therapy offers women with HMB and fibroids.
The lumbar spine is considered the most reactive, so this site is often used as the main focus with BMD studies. As Simon et al show, the lumbar spine mean BMD percent change from baseline for the elagolix with add-back therapy was -1.5% (95% confidence interval [CI], -1.9 to -1.0) in women who received up to 12 months of treatment at month 6 in the extension study. After stopping elagolix with add-back therapy, at 6 months the elagolix with add-back therapy had a Z-score of -0.6% (95% CI, -1.1 to -0.1). This shows a trend toward baseline, or a recovery within a short time from stopping medication.
Continue to: Study strengths and limitations...
Study strengths and limitations
Strengths of this study include its overall design; efficacy endpoints, which were all established a priori; the fact that measurement of menstrual blood loss was done with the objective alkaline hematin method; and the statistical analysis, which is thorough and well presented. This extension study allowed further evaluation of efficacy and safety for elagolix. Although the authors point out that there may be some selection bias in an extension study, the fact that so many women elected to continue into the extended study is a positive reflection of the treatment.
As providers learn of new therapies for management of HMB associated with fibroids, it is important to consider who will benefit the most. In my opinion, any woman with heavy periods associated with fibroids could be a candidate for elagolix with add-back therapy. This treatment is highly effective, well tolerated, and safe. My approach to management includes educating a woman on all potential therapies and this new option of elagolix and add-back therapy is an important one. The decision for an individual woman on how to manage heavy periods associated with fibroids should consider her contraceptive needs, medical issues, and the risk and benefit of individual therapies. ●
Elagolix and hormonal add-back therapy offer a long-term medical option for women with HMB associated with fibroids that is both effective and safe.
ANDREA S. LUKES, MD, MHSc
Simon JA, Al-Hendy A, Archer DF, et al. Elagolix treatment for up to 12 months in women with heavy menstrual bleeding and uterine leiomyomas. Obstet Gynecol. 2020;135:1313-1326.
Expert Commentary
Uterine fibroids are common (occurring in up to 80% of reproductive-age women),1,2 and often associated with heavy menstrual bleeding (HMB). There are surgical and medical options, but typically medical options are used for short periods of time. Elagolix with hormonal add-back therapy was recently approved (May 29, 2020) by the US Food and Drug Administration (FDA) for treatment of HMB in women with uterine fibroids for up to 24 months.
Elagolix is an oral, nonpeptide gonadotropin-releasing hormone antagonist that results in a dose-dependent reduction of gonadotropins and ovarian sex hormones. There are now 2 approved products containing elagolix, with different indications:
- Orilissa. Elagolix was approved in 2018 by the FDA for moderate to severe pain associated with endometriosis. For that indication there are 2 dose options of elagolix (150 mg for up to 2 years and 200 mg for up to 6 months) and there is no hormonal add-back therapy.
- Oriahnn. Elagolix and hormonal add-back therapy was approved in 2020 for HMB associated with uterine fibroids for up to 24 months. The total daily dose of elagolix is 600 mg (elagolix 300 mg in the morning with estradiol 1 mg/norethindrone acetate 0.5 mg and then in the evening elagolix 300 mg and no hormonal add-back).
This new class of drug, GnRH antagonist, is an important one for women’s health, and emerging science will continue to expand its potential uses, such as in reproductive health, as well as long-term efficacy and safety. The difference in daily dose of elagolix for endometriosis (150 mg for 24 months) compared with HMB associated with fibroids (600 mg for 24 months) is why the hormonal add-back therapy is important and allows for protection of bone density.
This is an important manuscript because it highlights a medical option for women with HMB associated with fibroids, which can be used for a long period of time. Further, the improvement in bleeding is both impressive and maintained in the extension study. Approximately 90% of women show improvement in their menstrual bleeding associated with fibroids.
The question of what to do after 24 months of therapy with elagolix and hormonal add-back therapy is an important one, but providers should recognize that the limiting factor with this elagolix and hormonal add-back therapy is bone mineral density (BMD). We will only learn more and more moving forward if this is a clinically meaningful reason for stopping treatment at 24 months. The FDA takes a strict view of safety, and providers must weigh this with the benefit of therapy.
One other highlight between the 2 approved medications is that Orilissa does not have a black box warning, given that there is no hormonal add-back therapy. Oriahnn does have a warning, regarding thromboembolic disorders and vascular events:
- Estrogen and progestin combinations, including Oriahnn, increase the risk of thrombotic or thromboembolic disorders, especially in women at increased risk for these events.
- Oriahnn is contraindicated in women with current or a history of thrombotic or thromboembolic disorders and in women at increased risk for these events, including women over 35 years of age who smoke or women with uncontrolled hypertension.
Continue to: Details about the study...
Details about the study
The study by Simon et al is an extension study (UF-EXTEND), in that women could participate if they had completed 1 of the 2 pivotal studies on elagolix. The pivotal studies (Elaris UF1 and UF2) were both randomized, double-blinded, placebo-controlled studies with up to 6 months of therapy; for UF-EXTEND, however, participants were randomly assigned to either combined elagolix and hormone replacement therapy or elagolix alone for an additional 6 months of therapy. Although it was known that all participants would receive elagolix in UF-EXTEND, those who received hormonal add-back therapy were blinded. All women were then followed up for an additional 12 months after treatment ended.
The efficacy of elagolix was measured by the objective alkaline hematin method for menstrual blood loss with the a priori coprimary endpoints. The elagolix and hormonal add-back therapy group showed objective improvement in menstrual blood loss at 12 months in 87.9% of women in the extension study (89.4% in the elagolix alone group). This compares with 72.2% improvement at 6 months of treatment in the UF1 and UF2 studies for those taking elagolix and hormonal add-back therapy. These findings illustrate maintenance of the efficacy seen within the 6-month pivotal studies using elagolix over an extended amount of time.
The safety of elagolix also was demonstrated in UF-EXTEND. The 3 most common adverse events were similar to those found in Elaris UF1 and UF2 and included hot flushes, headache, and nausea. In the elagolix and hormonal add-back therapy group during the extension study, the percentage with hot flushes was 7%, headache 6%, and nausea 4%. These are small percentages, which is encouraging for providers and women with HMB associated with fibroids.
Effects on bone density
Bone density was evaluated at baseline in the UF1 and UF2 studies, through treatment, and then 12 months after the extended treatment was stopped. The hormonal add-back therapy of estradiol 1 mg/norethindrone acetate 0.5 mg significantly protected bone density. Some women did not have a decrease in bone density, but for those who did the average was less than 5% for the lumbar spine. The lumbar spine is considered the most reactive, so this illustrates the safety that combined therapy offers women with HMB and fibroids.
The lumbar spine is considered the most reactive, so this site is often used as the main focus with BMD studies. As Simon et al show, the lumbar spine mean BMD percent change from baseline for the elagolix with add-back therapy was -1.5% (95% confidence interval [CI], -1.9 to -1.0) in women who received up to 12 months of treatment at month 6 in the extension study. After stopping elagolix with add-back therapy, at 6 months the elagolix with add-back therapy had a Z-score of -0.6% (95% CI, -1.1 to -0.1). This shows a trend toward baseline, or a recovery within a short time from stopping medication.
Continue to: Study strengths and limitations...
Study strengths and limitations
Strengths of this study include its overall design; efficacy endpoints, which were all established a priori; the fact that measurement of menstrual blood loss was done with the objective alkaline hematin method; and the statistical analysis, which is thorough and well presented. This extension study allowed further evaluation of efficacy and safety for elagolix. Although the authors point out that there may be some selection bias in an extension study, the fact that so many women elected to continue into the extended study is a positive reflection of the treatment.
As providers learn of new therapies for management of HMB associated with fibroids, it is important to consider who will benefit the most. In my opinion, any woman with heavy periods associated with fibroids could be a candidate for elagolix with add-back therapy. This treatment is highly effective, well tolerated, and safe. My approach to management includes educating a woman on all potential therapies and this new option of elagolix and add-back therapy is an important one. The decision for an individual woman on how to manage heavy periods associated with fibroids should consider her contraceptive needs, medical issues, and the risk and benefit of individual therapies. ●
Elagolix and hormonal add-back therapy offer a long-term medical option for women with HMB associated with fibroids that is both effective and safe.
ANDREA S. LUKES, MD, MHSc
- Stewart EA, Nicholson WK, Bradley L, et al. The burden of uterine fibroids for African-American women: results of a national survey. J Women’s Health. 2013;22:807-816.
- Baird DD, Dunson DB, Hill MC, et al. High cumulative incidence of uterine leiomyoma in black and white women: ultrasound evidence. Am J Obstet Gynecol. 2003;188:100-107.
- Stewart EA, Nicholson WK, Bradley L, et al. The burden of uterine fibroids for African-American women: results of a national survey. J Women’s Health. 2013;22:807-816.
- Baird DD, Dunson DB, Hill MC, et al. High cumulative incidence of uterine leiomyoma in black and white women: ultrasound evidence. Am J Obstet Gynecol. 2003;188:100-107.
Oral danicamtiv improves left atrial contractility in HFrEF
Danicamtiv, a novel oral selective cardiac myosin activator, demonstrated promising beneficial effects on left ventricular systolic function coupled with marked improvements in left atrial volume and function in patients with heart failure with reduced ejection fraction in a phase 2a clinical trial, Adriaan A. Voors, MD, PhD, said at the European Society of Cardiology Heart Failure Discoveries virtual meeting.
Importantly, these improvements weren’t accompanied by any unwelcome significant increase in diastolic stiffness, added Dr. Voors, a cardiologist at the University of Groningen (the Netherlands).
This is a drug whose novel mechanism of action could make it a good fit in combination with existing guideline-recommended therapies known to improve morbidity and mortality in patients with heart failure with reduced ejection fraction (HFrEF), none of which do what danicamtiv does: namely, activates cardiac myosin by enhancing myofibrillar adenosine triphosphatase activity, thereby boosting intrinsic myocardial contractility without any impact upon calcium homeostasis, he explained.
Dr. Voors reported on 40 patients with stable HFrEF and a left ventricular ejection fraction of 35% or less, all on background guideline-directed medical therapy. They were randomized double blind to 7 days of danicamtiv at 50, 75, or 100 mg twice daily, or placebo. A total of 489 ECGs were performed in conjunction with blood draws to measure plasma drug concentrations during the study.
Danicamtiv significantly improved left ventricular stroke volume and global longitudinal and circumferential strain in plasma drug concentration–dependent fashion, while simultaneously decreasing left ventricular end-systolic and end-diastolic diameters. Danicamtiv increased systolic ejection time from 286 milliseconds at baseline by an additional placebo-corrected 15, 36, and 48 milliseconds in patients with low, mid-range, and high drug concentrations.
The cardiac myosin activator’s concentration-dependent salutary effects on left atrial (LA) parameters in this brief study were intriguing, since LA function is often compromised in patients with heart failure and has been shown in prior observational studies to independently predict cardiovascular outcomes, the cardiologist noted. The favorable changes in response to danicamtiv included a reduction in LA minimal volume index and an increase in LA emptying fraction. Also, there were marked improvements in LA function index, by 6.1 and 5.8 points, respectively, in patients with mid- and high drug concentrations, from a baseline of 26 points.
Holter monitoring revealed no increased risk of atrial or ventricular arrhythmias in study participants.
Treatment-emergent adverse events were mild and/or unrelated to treatment and showed no particular pattern. The one serious adverse event in the study was a case of hyperkalemia deemed by investigators to be unrelated to treatment.
Seven of 30 danicamtiv-treated patients developed mild, transient, asymptomatic increases in serum cardiac troponin I and/or high-sensitivity troponin T. Dr. Voors said the significance of this must await further examination in larger clinical trials. A phase 2 clinical trial in patients with HFrEF and paroxysmal or persistent atrial fibrillation is planned in order to learn if chronic therapy with danicamtiv results in sustained LA remodeling and clinical benefits. Another planned phase 2 study will be conducted in patients with selected forms of genetic dilated cardiomyopathy.
Because danicamtiv appears to have no effects on blood pressure, renal function, or electrolytes, Dr. Voors speculated that the drug might prove to be an attractive therapeutic option in patients with advanced refractory heart failure, who often have low blood pressure, poor renal function, and a very low left ventricular ejection fraction.
Discussant Thomas Thum, MD, PhD, commented that danicamtiv has definitely earned an opportunity to show what it can do in larger, long-term clinical trials. He was impressed by the significant increase in systolic ejection time, a good marker for cardiac contractility. But he added that the troponin signal warrants careful scrutiny.
“The slight increase over baseline in the phase 2a study was not correlated with any ECG changes or clinical symptoms. However, whether this is a detrimental biomarker sign of a silent harm to the heart remains to be investigated,” said Dr. Thum, a cardiologist at the Institute of Molecular and Translational Therapeutic Strategies at Hannover (Germany) Medical School.
The phase 2a study finding of a plasma drug concentration–dependent prolongation in isovolumetric relaxation time “warrants some caution in future clinical development in patients with impaired diastolic function,” he added.
Simultaneous with Dr. Voors’ presentation, the study results were published online (Eur J Heart Fail. 2020 Jun 19. doi: 10.1002/ejhf.1933).
The danicamtiv study was sponsored by MyoKardia. Dr. Voors reported receiving research funding from and serving as a consultant to MyoKardia and numerous other companies.
Danicamtiv, a novel oral selective cardiac myosin activator, demonstrated promising beneficial effects on left ventricular systolic function coupled with marked improvements in left atrial volume and function in patients with heart failure with reduced ejection fraction in a phase 2a clinical trial, Adriaan A. Voors, MD, PhD, said at the European Society of Cardiology Heart Failure Discoveries virtual meeting.
Importantly, these improvements weren’t accompanied by any unwelcome significant increase in diastolic stiffness, added Dr. Voors, a cardiologist at the University of Groningen (the Netherlands).
This is a drug whose novel mechanism of action could make it a good fit in combination with existing guideline-recommended therapies known to improve morbidity and mortality in patients with heart failure with reduced ejection fraction (HFrEF), none of which do what danicamtiv does: namely, activates cardiac myosin by enhancing myofibrillar adenosine triphosphatase activity, thereby boosting intrinsic myocardial contractility without any impact upon calcium homeostasis, he explained.
Dr. Voors reported on 40 patients with stable HFrEF and a left ventricular ejection fraction of 35% or less, all on background guideline-directed medical therapy. They were randomized double blind to 7 days of danicamtiv at 50, 75, or 100 mg twice daily, or placebo. A total of 489 ECGs were performed in conjunction with blood draws to measure plasma drug concentrations during the study.
Danicamtiv significantly improved left ventricular stroke volume and global longitudinal and circumferential strain in plasma drug concentration–dependent fashion, while simultaneously decreasing left ventricular end-systolic and end-diastolic diameters. Danicamtiv increased systolic ejection time from 286 milliseconds at baseline by an additional placebo-corrected 15, 36, and 48 milliseconds in patients with low, mid-range, and high drug concentrations.
The cardiac myosin activator’s concentration-dependent salutary effects on left atrial (LA) parameters in this brief study were intriguing, since LA function is often compromised in patients with heart failure and has been shown in prior observational studies to independently predict cardiovascular outcomes, the cardiologist noted. The favorable changes in response to danicamtiv included a reduction in LA minimal volume index and an increase in LA emptying fraction. Also, there were marked improvements in LA function index, by 6.1 and 5.8 points, respectively, in patients with mid- and high drug concentrations, from a baseline of 26 points.
Holter monitoring revealed no increased risk of atrial or ventricular arrhythmias in study participants.
Treatment-emergent adverse events were mild and/or unrelated to treatment and showed no particular pattern. The one serious adverse event in the study was a case of hyperkalemia deemed by investigators to be unrelated to treatment.
Seven of 30 danicamtiv-treated patients developed mild, transient, asymptomatic increases in serum cardiac troponin I and/or high-sensitivity troponin T. Dr. Voors said the significance of this must await further examination in larger clinical trials. A phase 2 clinical trial in patients with HFrEF and paroxysmal or persistent atrial fibrillation is planned in order to learn if chronic therapy with danicamtiv results in sustained LA remodeling and clinical benefits. Another planned phase 2 study will be conducted in patients with selected forms of genetic dilated cardiomyopathy.
Because danicamtiv appears to have no effects on blood pressure, renal function, or electrolytes, Dr. Voors speculated that the drug might prove to be an attractive therapeutic option in patients with advanced refractory heart failure, who often have low blood pressure, poor renal function, and a very low left ventricular ejection fraction.
Discussant Thomas Thum, MD, PhD, commented that danicamtiv has definitely earned an opportunity to show what it can do in larger, long-term clinical trials. He was impressed by the significant increase in systolic ejection time, a good marker for cardiac contractility. But he added that the troponin signal warrants careful scrutiny.
“The slight increase over baseline in the phase 2a study was not correlated with any ECG changes or clinical symptoms. However, whether this is a detrimental biomarker sign of a silent harm to the heart remains to be investigated,” said Dr. Thum, a cardiologist at the Institute of Molecular and Translational Therapeutic Strategies at Hannover (Germany) Medical School.
The phase 2a study finding of a plasma drug concentration–dependent prolongation in isovolumetric relaxation time “warrants some caution in future clinical development in patients with impaired diastolic function,” he added.
Simultaneous with Dr. Voors’ presentation, the study results were published online (Eur J Heart Fail. 2020 Jun 19. doi: 10.1002/ejhf.1933).
The danicamtiv study was sponsored by MyoKardia. Dr. Voors reported receiving research funding from and serving as a consultant to MyoKardia and numerous other companies.
Danicamtiv, a novel oral selective cardiac myosin activator, demonstrated promising beneficial effects on left ventricular systolic function coupled with marked improvements in left atrial volume and function in patients with heart failure with reduced ejection fraction in a phase 2a clinical trial, Adriaan A. Voors, MD, PhD, said at the European Society of Cardiology Heart Failure Discoveries virtual meeting.
Importantly, these improvements weren’t accompanied by any unwelcome significant increase in diastolic stiffness, added Dr. Voors, a cardiologist at the University of Groningen (the Netherlands).
This is a drug whose novel mechanism of action could make it a good fit in combination with existing guideline-recommended therapies known to improve morbidity and mortality in patients with heart failure with reduced ejection fraction (HFrEF), none of which do what danicamtiv does: namely, activates cardiac myosin by enhancing myofibrillar adenosine triphosphatase activity, thereby boosting intrinsic myocardial contractility without any impact upon calcium homeostasis, he explained.
Dr. Voors reported on 40 patients with stable HFrEF and a left ventricular ejection fraction of 35% or less, all on background guideline-directed medical therapy. They were randomized double blind to 7 days of danicamtiv at 50, 75, or 100 mg twice daily, or placebo. A total of 489 ECGs were performed in conjunction with blood draws to measure plasma drug concentrations during the study.
Danicamtiv significantly improved left ventricular stroke volume and global longitudinal and circumferential strain in plasma drug concentration–dependent fashion, while simultaneously decreasing left ventricular end-systolic and end-diastolic diameters. Danicamtiv increased systolic ejection time from 286 milliseconds at baseline by an additional placebo-corrected 15, 36, and 48 milliseconds in patients with low, mid-range, and high drug concentrations.
The cardiac myosin activator’s concentration-dependent salutary effects on left atrial (LA) parameters in this brief study were intriguing, since LA function is often compromised in patients with heart failure and has been shown in prior observational studies to independently predict cardiovascular outcomes, the cardiologist noted. The favorable changes in response to danicamtiv included a reduction in LA minimal volume index and an increase in LA emptying fraction. Also, there were marked improvements in LA function index, by 6.1 and 5.8 points, respectively, in patients with mid- and high drug concentrations, from a baseline of 26 points.
Holter monitoring revealed no increased risk of atrial or ventricular arrhythmias in study participants.
Treatment-emergent adverse events were mild and/or unrelated to treatment and showed no particular pattern. The one serious adverse event in the study was a case of hyperkalemia deemed by investigators to be unrelated to treatment.
Seven of 30 danicamtiv-treated patients developed mild, transient, asymptomatic increases in serum cardiac troponin I and/or high-sensitivity troponin T. Dr. Voors said the significance of this must await further examination in larger clinical trials. A phase 2 clinical trial in patients with HFrEF and paroxysmal or persistent atrial fibrillation is planned in order to learn if chronic therapy with danicamtiv results in sustained LA remodeling and clinical benefits. Another planned phase 2 study will be conducted in patients with selected forms of genetic dilated cardiomyopathy.
Because danicamtiv appears to have no effects on blood pressure, renal function, or electrolytes, Dr. Voors speculated that the drug might prove to be an attractive therapeutic option in patients with advanced refractory heart failure, who often have low blood pressure, poor renal function, and a very low left ventricular ejection fraction.
Discussant Thomas Thum, MD, PhD, commented that danicamtiv has definitely earned an opportunity to show what it can do in larger, long-term clinical trials. He was impressed by the significant increase in systolic ejection time, a good marker for cardiac contractility. But he added that the troponin signal warrants careful scrutiny.
“The slight increase over baseline in the phase 2a study was not correlated with any ECG changes or clinical symptoms. However, whether this is a detrimental biomarker sign of a silent harm to the heart remains to be investigated,” said Dr. Thum, a cardiologist at the Institute of Molecular and Translational Therapeutic Strategies at Hannover (Germany) Medical School.
The phase 2a study finding of a plasma drug concentration–dependent prolongation in isovolumetric relaxation time “warrants some caution in future clinical development in patients with impaired diastolic function,” he added.
Simultaneous with Dr. Voors’ presentation, the study results were published online (Eur J Heart Fail. 2020 Jun 19. doi: 10.1002/ejhf.1933).
The danicamtiv study was sponsored by MyoKardia. Dr. Voors reported receiving research funding from and serving as a consultant to MyoKardia and numerous other companies.
FROM ESC HEART FAILURE 2020
The Fetal Pillow: A new option for delivering the deeply impacted fetal head
Obstetricians know that a cesarean delivery (CD) for a woman with a prolonged second stage and a fetal head deeply impacted in the pelvis is challenging. In this situation, extensions of the uterine incision commonly occur, resulting in prolonged operative time and increased blood loss. Even more harrowing is the inability to deliver the fetal head, necessitating emergency assistance from other clinicians. In this situation, interventions that may be helpful include:
- extend or T the uterine incision
- enlist the aid of a clinician to push up on the fetal head with a vaginal hand (FIGURE 1)
- reverse breech extraction (FIGURE 2), and
- vaginal insertion of a Fetal Pillow prior to starting the delivery.
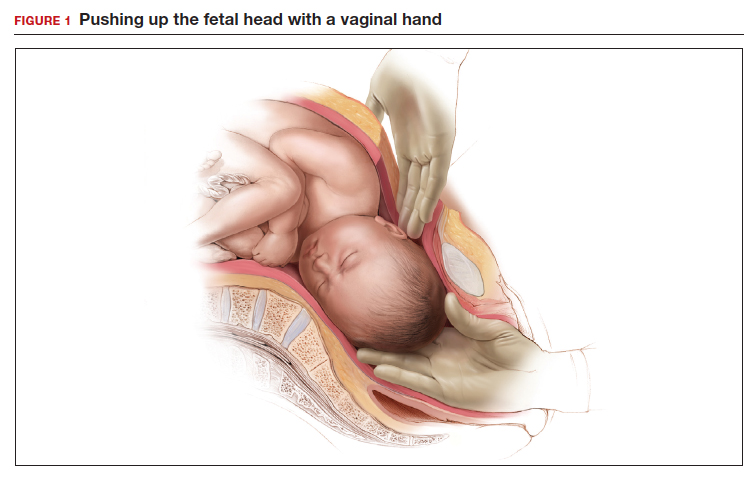
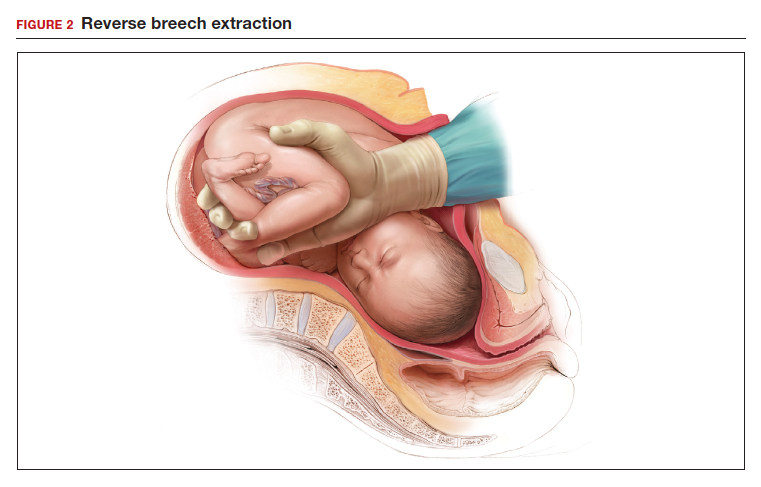
Evidence from clinical trials indicates that reverse breech extraction or insertion of a Fetal Pillow result in the best clinical outcomes.
Reverse breech extraction vs the push technique
Although the data are limited, most studies report that compared with pushing up with a vaginal hand (as shown in Figure 1), the reverse breech extraction technique (as shown in Figure 2) is associated with a reduction in extensions of the uterine incision, reduced blood loss, and reduced operative time.1 In a randomized trial, 108 women with obstructed labor undergoing CD in the second stage were randomly assigned to reverse breech extraction or pushing up with a vaginal hand.2 Following the uterine incision, the reverse breech extraction technique is performed by immediately reaching into the upper uterus and grasping the lower portion of the fetal leg and applying gentle traction on the leg until the second leg appeared. The lower legs are then pulled out of the uterus. Standard breech delivery maneuvers are used to deliver the shoulders and head. In the trial, compared with the push technique, reverse breech extraction was associated with fewer extensions of the uterine incision (30% vs 11%; P<.05), less blood loss (899 mL vs 1,257 mL; P<.001), and shorter operative time (56 min vs 89 min, P<.001). Fetal injury was similar with the push and breech extraction techniques (6% and 7%).
In another randomized trial, 192 women undergoing CD for obstructed labor were randomly assigned to reverse breech extraction or pushing the head up with a hand in the vagina.3 Compared with the vaginal push technique, reverse breech extraction was associated with fewer extensions of the uterine incision (19% vs 48%; P = .003), fewer cases of wound infection (2% vs 13%; P = .007), and fewer blood transfusions (2 vs 11; P = .012).
Additional options and adjuvants for facilitating delivery of a fetal head deeply impacted in the pelvis include: using a Coyne spoon, using nitroglycerine or terbutaline to relax the myometrium, breaking the vaginal suction on the fetal head before attempting delivery, keeping the wrist of the delivering hand as straight as possible to reduce uterine incision extensions, and incising the ring (if a Bandl’s ring is detected).
Continue to: The Fetal Pillow...
The Fetal Pillow
The Fetal Pillow (Safe Obstetric Systems, New York, New York) is a single-use fetal cephalic elevation device for managing the deeply impacted fetal head (FIGURE 3). The Fetal Pillow has a firm plastic base upon which is attached a soft silicon balloon. The Fetal Pillow is inserted into the vagina prior to initiating CD and the balloon is filled with 180 mL of saline, causing the fetal head to be pushed to a higher station (FIGURE 4). Use of the Fetal Pillow may be indicated prior to CD in the following situations:
- second stage labor with a deeply impacted head
- second stage labor and failed operative delivery
- occiput posterior position or deep transverse arrest
- absent progress in the first stage between 8 cm and 10 cm with a deeply impacted fetal head or excessive caput of the fetal head.
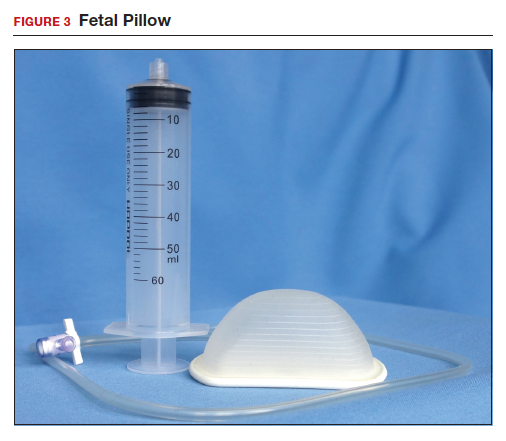
The Fetal Pillow is inserted after completing vaginal preparation for CD and before initiating skin preparation and abdominal draping. The steps for inserting the Fetal Pillow include:
- Use the 60 mL syringe to fully deflate the Fetal Pillow and leave the cock-stop open.
- Fold the Fetal Pillow by squeezing the firm plastic base, and with the patient’s legs in a frog-leg position, place the device in the vagina.
- Allow the firm plastic base to open to a flat position with the base against the posterior vaginal wall and the soft silicon balloon against the fetal head.
- Using pressure on the plastic base, gently push the Fetal Pillow posteriorly toward the sacrum of the mother.
- Use the 60 mL syringe to inflate the balloon with 180 mL of normal saline and close the valve.
- Straighten the patient’s legs and proceed with skin preparation and abdominal draping (FIGURE 4).
When the CD is completed, deflate the balloon by drawing out the saline with the 60 mL syringe and remove the device by hooking a finger around the firm plastic base. The Fetal Pillow is surprisingly easy to use.
Continue to: Effectiveness of the Fetal Pillow...
Effectiveness of the Fetal Pillow
In one randomized trial, 240 women undergoing CD were randomly allocated to a group in which the Fetal Pillow was placed in the vagina and inflated prior to the cesarean and a control group in which the Fetal Pillow was not used.
In another randomized trial, 60 nulliparous women undergoing CD in the second stage of labor had a Fetal Pillow inserted in the vagina and were randomly allocated to inflation of the pillow (Fetal Pillow group) or noninflation of the pillow (control group).5 In this study the mean length of the second stage was 4 hours. Compared with noninflation of the Fetal Pillow, use of the inflated Fetal Pillow was associated with a reduction in grade 3 extension of the uterine incision (extensions into the uterine artery, vagina, or bladder) (0% for inflation vs 13% for noninflation) and fewer difficult plus very difficult deliveries of the fetal head as reported by the surgeon (0% for inflation vs 37% for noninflation). There was no significant difference in blood loss between the two groups (800 mL vs 900 mL). These two randomized studies both reported that the use of the Fetal Pillow was associated with a reduction in grade 3 extensions of the uterine incision and a decrease in the difficulty of delivering the fetal head.
Consider trialing the Fetal Pillow
When a CD is performed after a prolonged second stage of labor, surgical complications are common, including extensions of the uterine incision and difficulty delivering the fetal head. When a grade 3 extension occurs—with tearing of a uterine artery, deep extension into the vagina, or damage to the bladder—the surgical repair can be extraordinarily challenging. Clinical trials report that both reverse breech extraction and the Fetal Pillow can facilitate CD in the setting of a prolonged second stage. For many obstetricians reverse breech extraction is a challenging obstetric maneuver. The insertion and inflation of a Fetal Pillow is a simple procedure. Obstetrician-gynecologists learn by doing. If you have never used the Fetal Pillow, I suggest you consider trialing it in your practice. ●
- Jeve YB, Navti OB, Konje JC. Comparison of techniques used to deliver a deeply impacted fetal head at full dilation: a systematic review and meta-analysis. BJOG. 2016;123:337-345.
- Fasubaa OB, Ezechi OC, Orji EO, et al. Delivery of the impacted head of the fetus at cesarean section after prolonged obstructed labor: a randomised comparative study of two methods. J Obstet Gynaecol. 2002;22:375-378.
- Nooh AM, Abdeldayem HM, Ben-Affan O. Reverse breech extraction versus the standard approach of pushing the impacted fetal head up through the vagina in caesarean section for obstructed labour: a randomised controlled trial. J Obstet Gynaecol. 2017;37:459-463.
- Seal SL, Dey A, Barman SC, et al. Randomized controlled trial of elevation of the fetal head with a fetal pillow during cesarean delivery at full cervical dilatation. Int J Gynaecol Obstet. 2016;133:178-182.
- Lassey SC, Little SE, Saadeh M,et al. Cephalic elevation device for second-stage cesarean delivery: a randomized controlled trial. Obstet Gynecol. 2020;135:879-884.
Obstetricians know that a cesarean delivery (CD) for a woman with a prolonged second stage and a fetal head deeply impacted in the pelvis is challenging. In this situation, extensions of the uterine incision commonly occur, resulting in prolonged operative time and increased blood loss. Even more harrowing is the inability to deliver the fetal head, necessitating emergency assistance from other clinicians. In this situation, interventions that may be helpful include:
- extend or T the uterine incision
- enlist the aid of a clinician to push up on the fetal head with a vaginal hand (FIGURE 1)
- reverse breech extraction (FIGURE 2), and
- vaginal insertion of a Fetal Pillow prior to starting the delivery.


Evidence from clinical trials indicates that reverse breech extraction or insertion of a Fetal Pillow result in the best clinical outcomes.
Reverse breech extraction vs the push technique
Although the data are limited, most studies report that compared with pushing up with a vaginal hand (as shown in Figure 1), the reverse breech extraction technique (as shown in Figure 2) is associated with a reduction in extensions of the uterine incision, reduced blood loss, and reduced operative time.1 In a randomized trial, 108 women with obstructed labor undergoing CD in the second stage were randomly assigned to reverse breech extraction or pushing up with a vaginal hand.2 Following the uterine incision, the reverse breech extraction technique is performed by immediately reaching into the upper uterus and grasping the lower portion of the fetal leg and applying gentle traction on the leg until the second leg appeared. The lower legs are then pulled out of the uterus. Standard breech delivery maneuvers are used to deliver the shoulders and head. In the trial, compared with the push technique, reverse breech extraction was associated with fewer extensions of the uterine incision (30% vs 11%; P<.05), less blood loss (899 mL vs 1,257 mL; P<.001), and shorter operative time (56 min vs 89 min, P<.001). Fetal injury was similar with the push and breech extraction techniques (6% and 7%).
In another randomized trial, 192 women undergoing CD for obstructed labor were randomly assigned to reverse breech extraction or pushing the head up with a hand in the vagina.3 Compared with the vaginal push technique, reverse breech extraction was associated with fewer extensions of the uterine incision (19% vs 48%; P = .003), fewer cases of wound infection (2% vs 13%; P = .007), and fewer blood transfusions (2 vs 11; P = .012).
Additional options and adjuvants for facilitating delivery of a fetal head deeply impacted in the pelvis include: using a Coyne spoon, using nitroglycerine or terbutaline to relax the myometrium, breaking the vaginal suction on the fetal head before attempting delivery, keeping the wrist of the delivering hand as straight as possible to reduce uterine incision extensions, and incising the ring (if a Bandl’s ring is detected).
Continue to: The Fetal Pillow...
The Fetal Pillow
The Fetal Pillow (Safe Obstetric Systems, New York, New York) is a single-use fetal cephalic elevation device for managing the deeply impacted fetal head (FIGURE 3). The Fetal Pillow has a firm plastic base upon which is attached a soft silicon balloon. The Fetal Pillow is inserted into the vagina prior to initiating CD and the balloon is filled with 180 mL of saline, causing the fetal head to be pushed to a higher station (FIGURE 4). Use of the Fetal Pillow may be indicated prior to CD in the following situations:
- second stage labor with a deeply impacted head
- second stage labor and failed operative delivery
- occiput posterior position or deep transverse arrest
- absent progress in the first stage between 8 cm and 10 cm with a deeply impacted fetal head or excessive caput of the fetal head.


The Fetal Pillow is inserted after completing vaginal preparation for CD and before initiating skin preparation and abdominal draping. The steps for inserting the Fetal Pillow include:
- Use the 60 mL syringe to fully deflate the Fetal Pillow and leave the cock-stop open.
- Fold the Fetal Pillow by squeezing the firm plastic base, and with the patient’s legs in a frog-leg position, place the device in the vagina.
- Allow the firm plastic base to open to a flat position with the base against the posterior vaginal wall and the soft silicon balloon against the fetal head.
- Using pressure on the plastic base, gently push the Fetal Pillow posteriorly toward the sacrum of the mother.
- Use the 60 mL syringe to inflate the balloon with 180 mL of normal saline and close the valve.
- Straighten the patient’s legs and proceed with skin preparation and abdominal draping (FIGURE 4).
When the CD is completed, deflate the balloon by drawing out the saline with the 60 mL syringe and remove the device by hooking a finger around the firm plastic base. The Fetal Pillow is surprisingly easy to use.
Continue to: Effectiveness of the Fetal Pillow...
Effectiveness of the Fetal Pillow
In one randomized trial, 240 women undergoing CD were randomly allocated to a group in which the Fetal Pillow was placed in the vagina and inflated prior to the cesarean and a control group in which the Fetal Pillow was not used.
In another randomized trial, 60 nulliparous women undergoing CD in the second stage of labor had a Fetal Pillow inserted in the vagina and were randomly allocated to inflation of the pillow (Fetal Pillow group) or noninflation of the pillow (control group).5 In this study the mean length of the second stage was 4 hours. Compared with noninflation of the Fetal Pillow, use of the inflated Fetal Pillow was associated with a reduction in grade 3 extension of the uterine incision (extensions into the uterine artery, vagina, or bladder) (0% for inflation vs 13% for noninflation) and fewer difficult plus very difficult deliveries of the fetal head as reported by the surgeon (0% for inflation vs 37% for noninflation). There was no significant difference in blood loss between the two groups (800 mL vs 900 mL). These two randomized studies both reported that the use of the Fetal Pillow was associated with a reduction in grade 3 extensions of the uterine incision and a decrease in the difficulty of delivering the fetal head.
Consider trialing the Fetal Pillow
When a CD is performed after a prolonged second stage of labor, surgical complications are common, including extensions of the uterine incision and difficulty delivering the fetal head. When a grade 3 extension occurs—with tearing of a uterine artery, deep extension into the vagina, or damage to the bladder—the surgical repair can be extraordinarily challenging. Clinical trials report that both reverse breech extraction and the Fetal Pillow can facilitate CD in the setting of a prolonged second stage. For many obstetricians reverse breech extraction is a challenging obstetric maneuver. The insertion and inflation of a Fetal Pillow is a simple procedure. Obstetrician-gynecologists learn by doing. If you have never used the Fetal Pillow, I suggest you consider trialing it in your practice. ●
Obstetricians know that a cesarean delivery (CD) for a woman with a prolonged second stage and a fetal head deeply impacted in the pelvis is challenging. In this situation, extensions of the uterine incision commonly occur, resulting in prolonged operative time and increased blood loss. Even more harrowing is the inability to deliver the fetal head, necessitating emergency assistance from other clinicians. In this situation, interventions that may be helpful include:
- extend or T the uterine incision
- enlist the aid of a clinician to push up on the fetal head with a vaginal hand (FIGURE 1)
- reverse breech extraction (FIGURE 2), and
- vaginal insertion of a Fetal Pillow prior to starting the delivery.


Evidence from clinical trials indicates that reverse breech extraction or insertion of a Fetal Pillow result in the best clinical outcomes.
Reverse breech extraction vs the push technique
Although the data are limited, most studies report that compared with pushing up with a vaginal hand (as shown in Figure 1), the reverse breech extraction technique (as shown in Figure 2) is associated with a reduction in extensions of the uterine incision, reduced blood loss, and reduced operative time.1 In a randomized trial, 108 women with obstructed labor undergoing CD in the second stage were randomly assigned to reverse breech extraction or pushing up with a vaginal hand.2 Following the uterine incision, the reverse breech extraction technique is performed by immediately reaching into the upper uterus and grasping the lower portion of the fetal leg and applying gentle traction on the leg until the second leg appeared. The lower legs are then pulled out of the uterus. Standard breech delivery maneuvers are used to deliver the shoulders and head. In the trial, compared with the push technique, reverse breech extraction was associated with fewer extensions of the uterine incision (30% vs 11%; P<.05), less blood loss (899 mL vs 1,257 mL; P<.001), and shorter operative time (56 min vs 89 min, P<.001). Fetal injury was similar with the push and breech extraction techniques (6% and 7%).
In another randomized trial, 192 women undergoing CD for obstructed labor were randomly assigned to reverse breech extraction or pushing the head up with a hand in the vagina.3 Compared with the vaginal push technique, reverse breech extraction was associated with fewer extensions of the uterine incision (19% vs 48%; P = .003), fewer cases of wound infection (2% vs 13%; P = .007), and fewer blood transfusions (2 vs 11; P = .012).
Additional options and adjuvants for facilitating delivery of a fetal head deeply impacted in the pelvis include: using a Coyne spoon, using nitroglycerine or terbutaline to relax the myometrium, breaking the vaginal suction on the fetal head before attempting delivery, keeping the wrist of the delivering hand as straight as possible to reduce uterine incision extensions, and incising the ring (if a Bandl’s ring is detected).
Continue to: The Fetal Pillow...
The Fetal Pillow
The Fetal Pillow (Safe Obstetric Systems, New York, New York) is a single-use fetal cephalic elevation device for managing the deeply impacted fetal head (FIGURE 3). The Fetal Pillow has a firm plastic base upon which is attached a soft silicon balloon. The Fetal Pillow is inserted into the vagina prior to initiating CD and the balloon is filled with 180 mL of saline, causing the fetal head to be pushed to a higher station (FIGURE 4). Use of the Fetal Pillow may be indicated prior to CD in the following situations:
- second stage labor with a deeply impacted head
- second stage labor and failed operative delivery
- occiput posterior position or deep transverse arrest
- absent progress in the first stage between 8 cm and 10 cm with a deeply impacted fetal head or excessive caput of the fetal head.


The Fetal Pillow is inserted after completing vaginal preparation for CD and before initiating skin preparation and abdominal draping. The steps for inserting the Fetal Pillow include:
- Use the 60 mL syringe to fully deflate the Fetal Pillow and leave the cock-stop open.
- Fold the Fetal Pillow by squeezing the firm plastic base, and with the patient’s legs in a frog-leg position, place the device in the vagina.
- Allow the firm plastic base to open to a flat position with the base against the posterior vaginal wall and the soft silicon balloon against the fetal head.
- Using pressure on the plastic base, gently push the Fetal Pillow posteriorly toward the sacrum of the mother.
- Use the 60 mL syringe to inflate the balloon with 180 mL of normal saline and close the valve.
- Straighten the patient’s legs and proceed with skin preparation and abdominal draping (FIGURE 4).
When the CD is completed, deflate the balloon by drawing out the saline with the 60 mL syringe and remove the device by hooking a finger around the firm plastic base. The Fetal Pillow is surprisingly easy to use.
Continue to: Effectiveness of the Fetal Pillow...
Effectiveness of the Fetal Pillow
In one randomized trial, 240 women undergoing CD were randomly allocated to a group in which the Fetal Pillow was placed in the vagina and inflated prior to the cesarean and a control group in which the Fetal Pillow was not used.
In another randomized trial, 60 nulliparous women undergoing CD in the second stage of labor had a Fetal Pillow inserted in the vagina and were randomly allocated to inflation of the pillow (Fetal Pillow group) or noninflation of the pillow (control group).5 In this study the mean length of the second stage was 4 hours. Compared with noninflation of the Fetal Pillow, use of the inflated Fetal Pillow was associated with a reduction in grade 3 extension of the uterine incision (extensions into the uterine artery, vagina, or bladder) (0% for inflation vs 13% for noninflation) and fewer difficult plus very difficult deliveries of the fetal head as reported by the surgeon (0% for inflation vs 37% for noninflation). There was no significant difference in blood loss between the two groups (800 mL vs 900 mL). These two randomized studies both reported that the use of the Fetal Pillow was associated with a reduction in grade 3 extensions of the uterine incision and a decrease in the difficulty of delivering the fetal head.
Consider trialing the Fetal Pillow
When a CD is performed after a prolonged second stage of labor, surgical complications are common, including extensions of the uterine incision and difficulty delivering the fetal head. When a grade 3 extension occurs—with tearing of a uterine artery, deep extension into the vagina, or damage to the bladder—the surgical repair can be extraordinarily challenging. Clinical trials report that both reverse breech extraction and the Fetal Pillow can facilitate CD in the setting of a prolonged second stage. For many obstetricians reverse breech extraction is a challenging obstetric maneuver. The insertion and inflation of a Fetal Pillow is a simple procedure. Obstetrician-gynecologists learn by doing. If you have never used the Fetal Pillow, I suggest you consider trialing it in your practice. ●
- Jeve YB, Navti OB, Konje JC. Comparison of techniques used to deliver a deeply impacted fetal head at full dilation: a systematic review and meta-analysis. BJOG. 2016;123:337-345.
- Fasubaa OB, Ezechi OC, Orji EO, et al. Delivery of the impacted head of the fetus at cesarean section after prolonged obstructed labor: a randomised comparative study of two methods. J Obstet Gynaecol. 2002;22:375-378.
- Nooh AM, Abdeldayem HM, Ben-Affan O. Reverse breech extraction versus the standard approach of pushing the impacted fetal head up through the vagina in caesarean section for obstructed labour: a randomised controlled trial. J Obstet Gynaecol. 2017;37:459-463.
- Seal SL, Dey A, Barman SC, et al. Randomized controlled trial of elevation of the fetal head with a fetal pillow during cesarean delivery at full cervical dilatation. Int J Gynaecol Obstet. 2016;133:178-182.
- Lassey SC, Little SE, Saadeh M,et al. Cephalic elevation device for second-stage cesarean delivery: a randomized controlled trial. Obstet Gynecol. 2020;135:879-884.
- Jeve YB, Navti OB, Konje JC. Comparison of techniques used to deliver a deeply impacted fetal head at full dilation: a systematic review and meta-analysis. BJOG. 2016;123:337-345.
- Fasubaa OB, Ezechi OC, Orji EO, et al. Delivery of the impacted head of the fetus at cesarean section after prolonged obstructed labor: a randomised comparative study of two methods. J Obstet Gynaecol. 2002;22:375-378.
- Nooh AM, Abdeldayem HM, Ben-Affan O. Reverse breech extraction versus the standard approach of pushing the impacted fetal head up through the vagina in caesarean section for obstructed labour: a randomised controlled trial. J Obstet Gynaecol. 2017;37:459-463.
- Seal SL, Dey A, Barman SC, et al. Randomized controlled trial of elevation of the fetal head with a fetal pillow during cesarean delivery at full cervical dilatation. Int J Gynaecol Obstet. 2016;133:178-182.
- Lassey SC, Little SE, Saadeh M,et al. Cephalic elevation device for second-stage cesarean delivery: a randomized controlled trial. Obstet Gynecol. 2020;135:879-884.