User login
Consider home subcutaneous immune globulin for refractory dermatomyositis
Home-based subcutaneous immune globulin therapy is a promising alternative to intravenous immune globulin therapy for patients with refractory dermatomyositis or polymyositis, Anna Postolova, MD, MPH, declared at the 2021 Rheumatology Winter Clinical Symposium.
“This is really exciting. I think in the years to come we may see a change to having our patients be able to do immune globulin therapy at home,” said Dr. Postolova, a rheumatologist and allergist/immunologist at Stanford (Calif.) Health Care.
“The technology is there. I think our patients might feel more comfortable getting immune globulin at home,” she said. “I would love to switch more patients from IVIg to SCIg [subcutaneous immune globulin] in my practice.”
A few caveats: SCIg remains off label for treatment of dermatomyositis (DM) or polymyositis (PM). Its approved indication is as replacement therapy in patients with primary or secondary immunodeficiency diseases. IVIg is approved for this indication, but is also approved for DM/PM refractory to high-dose corticosteroids and immunosuppressants. Yet SCIg is clearly effective for these autoimmune inflammatory diseases, albeit to date the supporting evidence comes chiefly from observational studies and anecdotal experience.
“I don’t know if insurers will cover it, but they should because it’s obviously a lot cheaper to do it at home,” she noted.
SCIg advantages
SCIg offers compelling advantages over IVIg in addition to its substantially lower cost. These include far fewer systemic side effects, shorter infusion time, greater bioavailability, and better quality of life. Patients self-administer SCIg at home, avoiding the inconvenience of IVIg therapy, which entails travel time for once-monthly hospitalization or long hours spent in an infusion center, she explained.
French investigators recently documented a previously unappreciated further advantage of home-based SCIg. They convened a focus group of patients with DM or PM experienced with both IVIg and home SCIg and determined that participants uniformly preferred home SCIg. The patients cited a new and welcome feeling of autonomy and control.
“All patients with experience of IVIg and SCIg expressed a clear preference for SCIg, which was described to be easy, less disruptive for daily life, well tolerated, and less time-consuming. Preference was mainly related to a restoration of autonomy. Home-based self-administration reinforced the feeling of independence,” according to the investigators.
Available products
Six preparations of SCIg are commercially available. Most are in 10% concentration, as are all IVIg products. However, a 20% formulation of SCIg known as Hizentra allows for a smaller infusion volume and quicker completion of a treatment session. And one SCIg product, HyQvia, uses recombinant human hyaluronidase-facilitated 10% immune globulin, allowing home infusion of large volumes of sustained-release immune globulin on a once-monthly basis.
The relatively recent introduction of home SCIg for treatment of autoimmune inflammatory diseases, including DM, PM, and chronic inflammatory demyelinating polyneuropathy, has been pioneered mainly by European investigators. The treatment is often given by programmable mechanical pump once weekly. Italian investigators have reported efficacy in DM using 0.2 g/kg per week, which is about half the monthly total dose of IVIg employed. The infusion rate is 10-40 mL/hour, with a volume of around 35 mL per injection site.
Alternatively, SCIg can be delivered by rapid push infusions of smaller volumes with a syringe two or three times per week; that’s the regimen that was used at 2 g/kg over the course of a month by patients in the French focus group study, who didn’t mind the more frequent dosing.
“As they have had severe long-lasting symptoms, SCIg was perceived as a curative rather than a preventive therapy,” according to the French investigators.
More than 40% of patients experience adverse reactions to IVIg. These often involve headaches, nausea, back or abdominal pain, arthralgias, and/or difficulty breathing. Thromboembolic events and acute renal failure occur occasionally. For this reason, many physicians give a prophylactic dose of corticosteroids an hour before a patient’s first dose of IVIg. These systemic side effects are so rare with SCIg that Dr. Postolova has never pretreated with steroids, even though the main reason she resorts to the home therapy is a patient’s track record of poor tolerance of IVIg. The lower abdomen and thigh are the most commonly used subcutaneous infusion sites. Mild local infusion site reactions are fairly common.
Formulating IVIg and SCIg is a complex process that entails plasma procurement and pooling, fractionation, and purification. It takes 10,000-60,000 plasma donations to make one lot of IVIg. Donations are accepted only from repeated donors. Samples are held for 6 months and tested for infectious agents. However, efforts are underway to develop bioengineered recombinant immune globulin products that don’t require donated plasma. These products are being designed to capture and enhance the most important mechanisms of benefit of plasma-derived immunoglobulins using Fc fragments that target key receptors, rather than relying on full-length immune globulin. The goal is enhanced efficacy at much lower doses than with IVIg or SCIg.
Dr. Postolova reported having no financial conflicts regarding her presentation.
Home-based subcutaneous immune globulin therapy is a promising alternative to intravenous immune globulin therapy for patients with refractory dermatomyositis or polymyositis, Anna Postolova, MD, MPH, declared at the 2021 Rheumatology Winter Clinical Symposium.
“This is really exciting. I think in the years to come we may see a change to having our patients be able to do immune globulin therapy at home,” said Dr. Postolova, a rheumatologist and allergist/immunologist at Stanford (Calif.) Health Care.
“The technology is there. I think our patients might feel more comfortable getting immune globulin at home,” she said. “I would love to switch more patients from IVIg to SCIg [subcutaneous immune globulin] in my practice.”
A few caveats: SCIg remains off label for treatment of dermatomyositis (DM) or polymyositis (PM). Its approved indication is as replacement therapy in patients with primary or secondary immunodeficiency diseases. IVIg is approved for this indication, but is also approved for DM/PM refractory to high-dose corticosteroids and immunosuppressants. Yet SCIg is clearly effective for these autoimmune inflammatory diseases, albeit to date the supporting evidence comes chiefly from observational studies and anecdotal experience.
“I don’t know if insurers will cover it, but they should because it’s obviously a lot cheaper to do it at home,” she noted.
SCIg advantages
SCIg offers compelling advantages over IVIg in addition to its substantially lower cost. These include far fewer systemic side effects, shorter infusion time, greater bioavailability, and better quality of life. Patients self-administer SCIg at home, avoiding the inconvenience of IVIg therapy, which entails travel time for once-monthly hospitalization or long hours spent in an infusion center, she explained.
French investigators recently documented a previously unappreciated further advantage of home-based SCIg. They convened a focus group of patients with DM or PM experienced with both IVIg and home SCIg and determined that participants uniformly preferred home SCIg. The patients cited a new and welcome feeling of autonomy and control.
“All patients with experience of IVIg and SCIg expressed a clear preference for SCIg, which was described to be easy, less disruptive for daily life, well tolerated, and less time-consuming. Preference was mainly related to a restoration of autonomy. Home-based self-administration reinforced the feeling of independence,” according to the investigators.
Available products
Six preparations of SCIg are commercially available. Most are in 10% concentration, as are all IVIg products. However, a 20% formulation of SCIg known as Hizentra allows for a smaller infusion volume and quicker completion of a treatment session. And one SCIg product, HyQvia, uses recombinant human hyaluronidase-facilitated 10% immune globulin, allowing home infusion of large volumes of sustained-release immune globulin on a once-monthly basis.
The relatively recent introduction of home SCIg for treatment of autoimmune inflammatory diseases, including DM, PM, and chronic inflammatory demyelinating polyneuropathy, has been pioneered mainly by European investigators. The treatment is often given by programmable mechanical pump once weekly. Italian investigators have reported efficacy in DM using 0.2 g/kg per week, which is about half the monthly total dose of IVIg employed. The infusion rate is 10-40 mL/hour, with a volume of around 35 mL per injection site.
Alternatively, SCIg can be delivered by rapid push infusions of smaller volumes with a syringe two or three times per week; that’s the regimen that was used at 2 g/kg over the course of a month by patients in the French focus group study, who didn’t mind the more frequent dosing.
“As they have had severe long-lasting symptoms, SCIg was perceived as a curative rather than a preventive therapy,” according to the French investigators.
More than 40% of patients experience adverse reactions to IVIg. These often involve headaches, nausea, back or abdominal pain, arthralgias, and/or difficulty breathing. Thromboembolic events and acute renal failure occur occasionally. For this reason, many physicians give a prophylactic dose of corticosteroids an hour before a patient’s first dose of IVIg. These systemic side effects are so rare with SCIg that Dr. Postolova has never pretreated with steroids, even though the main reason she resorts to the home therapy is a patient’s track record of poor tolerance of IVIg. The lower abdomen and thigh are the most commonly used subcutaneous infusion sites. Mild local infusion site reactions are fairly common.
Formulating IVIg and SCIg is a complex process that entails plasma procurement and pooling, fractionation, and purification. It takes 10,000-60,000 plasma donations to make one lot of IVIg. Donations are accepted only from repeated donors. Samples are held for 6 months and tested for infectious agents. However, efforts are underway to develop bioengineered recombinant immune globulin products that don’t require donated plasma. These products are being designed to capture and enhance the most important mechanisms of benefit of plasma-derived immunoglobulins using Fc fragments that target key receptors, rather than relying on full-length immune globulin. The goal is enhanced efficacy at much lower doses than with IVIg or SCIg.
Dr. Postolova reported having no financial conflicts regarding her presentation.
Home-based subcutaneous immune globulin therapy is a promising alternative to intravenous immune globulin therapy for patients with refractory dermatomyositis or polymyositis, Anna Postolova, MD, MPH, declared at the 2021 Rheumatology Winter Clinical Symposium.
“This is really exciting. I think in the years to come we may see a change to having our patients be able to do immune globulin therapy at home,” said Dr. Postolova, a rheumatologist and allergist/immunologist at Stanford (Calif.) Health Care.
“The technology is there. I think our patients might feel more comfortable getting immune globulin at home,” she said. “I would love to switch more patients from IVIg to SCIg [subcutaneous immune globulin] in my practice.”
A few caveats: SCIg remains off label for treatment of dermatomyositis (DM) or polymyositis (PM). Its approved indication is as replacement therapy in patients with primary or secondary immunodeficiency diseases. IVIg is approved for this indication, but is also approved for DM/PM refractory to high-dose corticosteroids and immunosuppressants. Yet SCIg is clearly effective for these autoimmune inflammatory diseases, albeit to date the supporting evidence comes chiefly from observational studies and anecdotal experience.
“I don’t know if insurers will cover it, but they should because it’s obviously a lot cheaper to do it at home,” she noted.
SCIg advantages
SCIg offers compelling advantages over IVIg in addition to its substantially lower cost. These include far fewer systemic side effects, shorter infusion time, greater bioavailability, and better quality of life. Patients self-administer SCIg at home, avoiding the inconvenience of IVIg therapy, which entails travel time for once-monthly hospitalization or long hours spent in an infusion center, she explained.
French investigators recently documented a previously unappreciated further advantage of home-based SCIg. They convened a focus group of patients with DM or PM experienced with both IVIg and home SCIg and determined that participants uniformly preferred home SCIg. The patients cited a new and welcome feeling of autonomy and control.
“All patients with experience of IVIg and SCIg expressed a clear preference for SCIg, which was described to be easy, less disruptive for daily life, well tolerated, and less time-consuming. Preference was mainly related to a restoration of autonomy. Home-based self-administration reinforced the feeling of independence,” according to the investigators.
Available products
Six preparations of SCIg are commercially available. Most are in 10% concentration, as are all IVIg products. However, a 20% formulation of SCIg known as Hizentra allows for a smaller infusion volume and quicker completion of a treatment session. And one SCIg product, HyQvia, uses recombinant human hyaluronidase-facilitated 10% immune globulin, allowing home infusion of large volumes of sustained-release immune globulin on a once-monthly basis.
The relatively recent introduction of home SCIg for treatment of autoimmune inflammatory diseases, including DM, PM, and chronic inflammatory demyelinating polyneuropathy, has been pioneered mainly by European investigators. The treatment is often given by programmable mechanical pump once weekly. Italian investigators have reported efficacy in DM using 0.2 g/kg per week, which is about half the monthly total dose of IVIg employed. The infusion rate is 10-40 mL/hour, with a volume of around 35 mL per injection site.
Alternatively, SCIg can be delivered by rapid push infusions of smaller volumes with a syringe two or three times per week; that’s the regimen that was used at 2 g/kg over the course of a month by patients in the French focus group study, who didn’t mind the more frequent dosing.
“As they have had severe long-lasting symptoms, SCIg was perceived as a curative rather than a preventive therapy,” according to the French investigators.
More than 40% of patients experience adverse reactions to IVIg. These often involve headaches, nausea, back or abdominal pain, arthralgias, and/or difficulty breathing. Thromboembolic events and acute renal failure occur occasionally. For this reason, many physicians give a prophylactic dose of corticosteroids an hour before a patient’s first dose of IVIg. These systemic side effects are so rare with SCIg that Dr. Postolova has never pretreated with steroids, even though the main reason she resorts to the home therapy is a patient’s track record of poor tolerance of IVIg. The lower abdomen and thigh are the most commonly used subcutaneous infusion sites. Mild local infusion site reactions are fairly common.
Formulating IVIg and SCIg is a complex process that entails plasma procurement and pooling, fractionation, and purification. It takes 10,000-60,000 plasma donations to make one lot of IVIg. Donations are accepted only from repeated donors. Samples are held for 6 months and tested for infectious agents. However, efforts are underway to develop bioengineered recombinant immune globulin products that don’t require donated plasma. These products are being designed to capture and enhance the most important mechanisms of benefit of plasma-derived immunoglobulins using Fc fragments that target key receptors, rather than relying on full-length immune globulin. The goal is enhanced efficacy at much lower doses than with IVIg or SCIg.
Dr. Postolova reported having no financial conflicts regarding her presentation.
FROM RWCS 2021
Checkpoint inhibitors’ ‘big picture’ safety shown with preexisting autoimmune diseases
Patients with advanced melanoma and preexisting autoimmune diseases (AIDs) who were treated with immune checkpoint inhibitors (ICIs) responded well and did not suffer more grade 3 or higher immune-related adverse events than patients without an AID, a new study finds, although some concerns were raised regarding patients with inflammatory bowel disease (IBD).
“To our knowledge, this is the first study to bridge this knowledge gap by presenting ‘real-world’ data on the safety and efficacy of ICI on a national scale,” wrote Monique K. van der Kooij, MD, of Leiden (the Netherlands) University Medical Center and coauthors. The study was published online in Annals of Internal Medicine.
To investigate ICI use and response among this specific subset of melanoma patients, the researchers launched a nationwide cohort study set in the Netherlands. Data were gathered via the Dutch Melanoma Treatment Registry (DMTR), in which 4,367 patients with advanced melanoma were enrolled between July 2013 and July 2018.
Within that cohort, 415 (9.5%) had preexisting AIDs. Nearly 55% had rheumatologic AIDs (n = 227) – which included RA, systemic lupus erythematosus, scleroderma, sarcoidosis, and vasculitis – with the next most frequent being endocrine AID (n = 143) and IBD (n = 55). Patients with AID were older than patients without (67 vs. 63 years) and were more likely to be female (53% vs. 41%).
The ICIs used in the study included anti-CTLA4 (ipilimumab), anti–programmed death 1 (PD-1) (nivolumab or pembrolizumab), or a combination of nivolumab and ipilimumab. Of the patients with AID, 55% (n = 228) were treated with ICI, compared with 58% of patients without AID. A total of 87 AID patients were treated with anti-CTLA4, 187 received anti-PD-1, and 34 received the combination. The combination was not readily available in the Netherlands until 2017, the authors stated, acknowledging that it may be wise to revisit its effects in the coming years.
Incidence of immune-related adverse events
The incidence of immune-related adverse events (irAEs) grade 3 and above for patients with and without AID who were given anti-CTLA4 was 30%. The incidence rate of irAEs was also similar for patients with (17%; 95% confidence interval, 12%-23%) and without (13%; 95% CI, 12%-15%) AID on anti-PD-1. Patients with AIDs who took anti-PD-1 therapy discontinued it more often because of toxicity than did the patients without AIDs.
The combination group had irAE incidence rates of 44% (95% CI, 27%-62%) for patients with AID, compared with 48% (95% CI, 43%-53%) for patients without AIDs. Overall, no patients with AIDs on ICIs died of toxicity, compared with three deaths among patients without AID on anti-CTLA4, five deaths among patients on anti-PD-1, and one patient on the combination.
Patients with IBD had a notably higher risk of anti-PD-1–induced colitis (19%; 95% CI, 7%-37%), compared with patients with other AIDs (3%; 95% CI, 0%-6%) and patients without AIDs (2%; 95% CI, 2%-3%). IBD patients were also more likely than all other groups on ICIs to stop treatment because of toxicity, leading the researchers to note that “close monitoring in patients with IBD is advised.”
Overall survival after diagnosis was similar in patients with AIDs (median, 13 months; 95% CI, 10-16 months) and without (median, 14 months; 95% CI, 13-15 months), as was the objective response rate to anti-CTLA4 treatment (10% vs. 16%), anti-PD-1 treatment (40% vs. 44%), and combination therapy (39% vs. 43%).
Study largely bypasses the effects of checkpoint inhibitors on RA patients
“For detail, you can’t look to this study,” Anne R. Bass, MD, of the division of rheumatology at the Hospital for Special Surgery in New York, said in an interview. “But for a big-picture look at ‘how safe are checkpoint inhibitors,’ I think it’s an important one.”
Dr. Bass noted that the investigators lumped certain elements together and bypassed others, including their focus on grade 3 or higher adverse events. That was a decision the authors themselves recognized as a potential limitation of their research.
“Understandably, they were worried about life-threatening adverse events, and that’s fine,” she said. But for patients with arthritis who flare, their events are usually grade 2 or even grade 1 and therefore not captured or analyzed in the study. “This does not really address the risk of flare in an RA patient.”
She also questioned their grouping of AIDs, with a bevy of rheumatic diseases categorized as one cluster and the “other” group being particularly broad in its inclusion of “all AIDs not listed” – though only eight patients were placed into that group.
That said, the researchers relied on an oncology database, not one aimed at AID or adverse events. “The numbers are so much bigger than any other study in this area that’s been done,” she said. “It’s both a strength and a weakness of this kind of database.”
Indeed, the authors considered their use of nationwide, population-based data from the DMTR a benefit, calling it “a strength of our approach.”
The DMTR was funded by a grant from the Netherlands Organization for Health Research and Development and sponsored by Bristol-Myers Squibb, Novartis, Roche Nederland, Merck Sharp & Dohme, and Pierre Fabre via the Dutch Institute for Clinical Auditing.
Patients with advanced melanoma and preexisting autoimmune diseases (AIDs) who were treated with immune checkpoint inhibitors (ICIs) responded well and did not suffer more grade 3 or higher immune-related adverse events than patients without an AID, a new study finds, although some concerns were raised regarding patients with inflammatory bowel disease (IBD).
“To our knowledge, this is the first study to bridge this knowledge gap by presenting ‘real-world’ data on the safety and efficacy of ICI on a national scale,” wrote Monique K. van der Kooij, MD, of Leiden (the Netherlands) University Medical Center and coauthors. The study was published online in Annals of Internal Medicine.
To investigate ICI use and response among this specific subset of melanoma patients, the researchers launched a nationwide cohort study set in the Netherlands. Data were gathered via the Dutch Melanoma Treatment Registry (DMTR), in which 4,367 patients with advanced melanoma were enrolled between July 2013 and July 2018.
Within that cohort, 415 (9.5%) had preexisting AIDs. Nearly 55% had rheumatologic AIDs (n = 227) – which included RA, systemic lupus erythematosus, scleroderma, sarcoidosis, and vasculitis – with the next most frequent being endocrine AID (n = 143) and IBD (n = 55). Patients with AID were older than patients without (67 vs. 63 years) and were more likely to be female (53% vs. 41%).
The ICIs used in the study included anti-CTLA4 (ipilimumab), anti–programmed death 1 (PD-1) (nivolumab or pembrolizumab), or a combination of nivolumab and ipilimumab. Of the patients with AID, 55% (n = 228) were treated with ICI, compared with 58% of patients without AID. A total of 87 AID patients were treated with anti-CTLA4, 187 received anti-PD-1, and 34 received the combination. The combination was not readily available in the Netherlands until 2017, the authors stated, acknowledging that it may be wise to revisit its effects in the coming years.
Incidence of immune-related adverse events
The incidence of immune-related adverse events (irAEs) grade 3 and above for patients with and without AID who were given anti-CTLA4 was 30%. The incidence rate of irAEs was also similar for patients with (17%; 95% confidence interval, 12%-23%) and without (13%; 95% CI, 12%-15%) AID on anti-PD-1. Patients with AIDs who took anti-PD-1 therapy discontinued it more often because of toxicity than did the patients without AIDs.
The combination group had irAE incidence rates of 44% (95% CI, 27%-62%) for patients with AID, compared with 48% (95% CI, 43%-53%) for patients without AIDs. Overall, no patients with AIDs on ICIs died of toxicity, compared with three deaths among patients without AID on anti-CTLA4, five deaths among patients on anti-PD-1, and one patient on the combination.
Patients with IBD had a notably higher risk of anti-PD-1–induced colitis (19%; 95% CI, 7%-37%), compared with patients with other AIDs (3%; 95% CI, 0%-6%) and patients without AIDs (2%; 95% CI, 2%-3%). IBD patients were also more likely than all other groups on ICIs to stop treatment because of toxicity, leading the researchers to note that “close monitoring in patients with IBD is advised.”
Overall survival after diagnosis was similar in patients with AIDs (median, 13 months; 95% CI, 10-16 months) and without (median, 14 months; 95% CI, 13-15 months), as was the objective response rate to anti-CTLA4 treatment (10% vs. 16%), anti-PD-1 treatment (40% vs. 44%), and combination therapy (39% vs. 43%).
Study largely bypasses the effects of checkpoint inhibitors on RA patients
“For detail, you can’t look to this study,” Anne R. Bass, MD, of the division of rheumatology at the Hospital for Special Surgery in New York, said in an interview. “But for a big-picture look at ‘how safe are checkpoint inhibitors,’ I think it’s an important one.”
Dr. Bass noted that the investigators lumped certain elements together and bypassed others, including their focus on grade 3 or higher adverse events. That was a decision the authors themselves recognized as a potential limitation of their research.
“Understandably, they were worried about life-threatening adverse events, and that’s fine,” she said. But for patients with arthritis who flare, their events are usually grade 2 or even grade 1 and therefore not captured or analyzed in the study. “This does not really address the risk of flare in an RA patient.”
She also questioned their grouping of AIDs, with a bevy of rheumatic diseases categorized as one cluster and the “other” group being particularly broad in its inclusion of “all AIDs not listed” – though only eight patients were placed into that group.
That said, the researchers relied on an oncology database, not one aimed at AID or adverse events. “The numbers are so much bigger than any other study in this area that’s been done,” she said. “It’s both a strength and a weakness of this kind of database.”
Indeed, the authors considered their use of nationwide, population-based data from the DMTR a benefit, calling it “a strength of our approach.”
The DMTR was funded by a grant from the Netherlands Organization for Health Research and Development and sponsored by Bristol-Myers Squibb, Novartis, Roche Nederland, Merck Sharp & Dohme, and Pierre Fabre via the Dutch Institute for Clinical Auditing.
Patients with advanced melanoma and preexisting autoimmune diseases (AIDs) who were treated with immune checkpoint inhibitors (ICIs) responded well and did not suffer more grade 3 or higher immune-related adverse events than patients without an AID, a new study finds, although some concerns were raised regarding patients with inflammatory bowel disease (IBD).
“To our knowledge, this is the first study to bridge this knowledge gap by presenting ‘real-world’ data on the safety and efficacy of ICI on a national scale,” wrote Monique K. van der Kooij, MD, of Leiden (the Netherlands) University Medical Center and coauthors. The study was published online in Annals of Internal Medicine.
To investigate ICI use and response among this specific subset of melanoma patients, the researchers launched a nationwide cohort study set in the Netherlands. Data were gathered via the Dutch Melanoma Treatment Registry (DMTR), in which 4,367 patients with advanced melanoma were enrolled between July 2013 and July 2018.
Within that cohort, 415 (9.5%) had preexisting AIDs. Nearly 55% had rheumatologic AIDs (n = 227) – which included RA, systemic lupus erythematosus, scleroderma, sarcoidosis, and vasculitis – with the next most frequent being endocrine AID (n = 143) and IBD (n = 55). Patients with AID were older than patients without (67 vs. 63 years) and were more likely to be female (53% vs. 41%).
The ICIs used in the study included anti-CTLA4 (ipilimumab), anti–programmed death 1 (PD-1) (nivolumab or pembrolizumab), or a combination of nivolumab and ipilimumab. Of the patients with AID, 55% (n = 228) were treated with ICI, compared with 58% of patients without AID. A total of 87 AID patients were treated with anti-CTLA4, 187 received anti-PD-1, and 34 received the combination. The combination was not readily available in the Netherlands until 2017, the authors stated, acknowledging that it may be wise to revisit its effects in the coming years.
Incidence of immune-related adverse events
The incidence of immune-related adverse events (irAEs) grade 3 and above for patients with and without AID who were given anti-CTLA4 was 30%. The incidence rate of irAEs was also similar for patients with (17%; 95% confidence interval, 12%-23%) and without (13%; 95% CI, 12%-15%) AID on anti-PD-1. Patients with AIDs who took anti-PD-1 therapy discontinued it more often because of toxicity than did the patients without AIDs.
The combination group had irAE incidence rates of 44% (95% CI, 27%-62%) for patients with AID, compared with 48% (95% CI, 43%-53%) for patients without AIDs. Overall, no patients with AIDs on ICIs died of toxicity, compared with three deaths among patients without AID on anti-CTLA4, five deaths among patients on anti-PD-1, and one patient on the combination.
Patients with IBD had a notably higher risk of anti-PD-1–induced colitis (19%; 95% CI, 7%-37%), compared with patients with other AIDs (3%; 95% CI, 0%-6%) and patients without AIDs (2%; 95% CI, 2%-3%). IBD patients were also more likely than all other groups on ICIs to stop treatment because of toxicity, leading the researchers to note that “close monitoring in patients with IBD is advised.”
Overall survival after diagnosis was similar in patients with AIDs (median, 13 months; 95% CI, 10-16 months) and without (median, 14 months; 95% CI, 13-15 months), as was the objective response rate to anti-CTLA4 treatment (10% vs. 16%), anti-PD-1 treatment (40% vs. 44%), and combination therapy (39% vs. 43%).
Study largely bypasses the effects of checkpoint inhibitors on RA patients
“For detail, you can’t look to this study,” Anne R. Bass, MD, of the division of rheumatology at the Hospital for Special Surgery in New York, said in an interview. “But for a big-picture look at ‘how safe are checkpoint inhibitors,’ I think it’s an important one.”
Dr. Bass noted that the investigators lumped certain elements together and bypassed others, including their focus on grade 3 or higher adverse events. That was a decision the authors themselves recognized as a potential limitation of their research.
“Understandably, they were worried about life-threatening adverse events, and that’s fine,” she said. But for patients with arthritis who flare, their events are usually grade 2 or even grade 1 and therefore not captured or analyzed in the study. “This does not really address the risk of flare in an RA patient.”
She also questioned their grouping of AIDs, with a bevy of rheumatic diseases categorized as one cluster and the “other” group being particularly broad in its inclusion of “all AIDs not listed” – though only eight patients were placed into that group.
That said, the researchers relied on an oncology database, not one aimed at AID or adverse events. “The numbers are so much bigger than any other study in this area that’s been done,” she said. “It’s both a strength and a weakness of this kind of database.”
Indeed, the authors considered their use of nationwide, population-based data from the DMTR a benefit, calling it “a strength of our approach.”
The DMTR was funded by a grant from the Netherlands Organization for Health Research and Development and sponsored by Bristol-Myers Squibb, Novartis, Roche Nederland, Merck Sharp & Dohme, and Pierre Fabre via the Dutch Institute for Clinical Auditing.
FROM ANNALS OF INTERNAL MEDICINE
Cumulative exposure to high-potency topical steroid doses drives osteoporosis fractures
In support of previously published case reports, in a dose-response relationship.
In a stepwise manner, the hazard ratios for major osteoporotic fracture (MOF) were found to start climbing incrementally for those with a cumulative topical steroid dose equivalent of more than 500 g of mometasone furoate when compared with exposure of 200-499 g, according to the team of investigators from the University of Copenhagen.
“Use of these drugs is very common, and we found an estimated population-attributable risk of as much as 4.3%,” the investigators reported in the study, published in JAMA Dermatology.
The retrospective cohort study drew data from the Danish National Patient Registry, which covers 99% of the country’s population. It was linked to the Danish National Prescription Registry, which captures data on pharmacy-dispensed medications. Data collected from the beginning of 2003 to the end of 2017 were evaluated.
Exposures to potent or very potent topical corticosteroids were converted into a single standard with potency equivalent to 1 mg/g of mometasone furoate. Four strata of exposure were compared to a reference exposure of 200-499 g. These were 500-999 g, 1,000-1,999 g, 2,000-9,999 g, and 10,000 g or greater.
For the first strata, the small increased risk for MOF did not reach significance (HR, 1.01; 95% confidence interval, 0.99-1.03), but each of the others did. These climbed from a 5% greater risk (HR 1.05 95% CI 1.02-1.08) for a cumulative exposure of 1,000 to 1,999 g, to a 10% greater risk (HR, 1.10; 95% CI, 1.07-1.13) for a cumulative exposure of 2,000-9,999 g, and finally to a 27% greater risk (HR, 1.27; 95% CI, 1.19-1.35) for a cumulative exposure of 10,000 g or higher.
The study included more than 700,000 individuals exposed to topical mometasone at a potency equivalent of 200 g or more over the study period. The reference group (200-499 g) was the largest (317,907 individuals). The first strata (500-999 g) included 186,359 patients; the second (1,000-1,999 g), 111,203 patients; the third (2,000-9,999 g), 94,334 patients; and the fifth (10,000 g or more), 13,448 patients.
“A 3% increase in the relative risk of osteoporosis and MOF was observed per doubling of the TCS dose,” according to the investigators.
Patients exposed to doses of high-potency topical steroids that put them at risk of MOF is limited but substantial, according to the senior author, Alexander Egeberg, MD, PhD, of the department of dermatology and allergy at Herlev and Gentofte Hospital, Copenhagen.
“It is true that the risk is modest for the average user of topical steroids,” Dr. Egeberg said in an interview. However, despite the fact that topical steroids are intended for short-term use, “2% of all our users had been exposed to the equivalent of 10,000 g of mometasone, which mean 100 tubes of 100 g.”
If the other two strata at significantly increased risk of MOF (greater than 1,000 g) are included, an additional 28% of all users are facing the potential for clinically significant osteoporosis, according to the Danish data.
The adverse effect of steroids on bone metabolism has been established previously, and several studies have linked systemic corticosteroid exposure, including inhaled corticosteroids, with increased risk of osteoporotic fracture. For example, one study showed that patients with chronic obstructive pulmonary disease on daily inhaled doses of the equivalent of fluticasone at or above 1,000 mcg for more than 4 years had about a 10% increased risk of MOF relative to those not exposed.
The data associate topical steroids with increased risk of osteoporotic fracture, but Dr. Egeberg said osteoporosis is not the only reason to use topical steroids prudently.
“It is important to keep in mind that osteoporosis and fractures are at the extreme end of the side-effect profile and that other side effects, such as striae formation, skin thinning, and dysregulated diabetes, can occur with much lower quantities of topical steroids,” Dr. Egeberg said
For avoiding this risk, “there are no specific cutoffs” recommended for topical steroids in current guidelines, but dermatologists should be aware that many of the indications for topical steroids, such as psoriasis and atopic dermatitis, involve skin with an impaired barrier function, exposing patients to an increased likelihood of absorption, according to Dr. Egeberg.
“A general rule of thumb that we use is that, if a patient with persistent disease activity requires a new prescription of the equivalent of 100 g mometasone every 1-2 months, it might be worth considering if there is a suitable alternative,” Dr. Egeberg said.
In an accompanying editorial, Rebecca D. Jackson, MD, of the division of endocrinology, diabetes, and metabolism in the department of internal medicine at Ohio State University, Columbus, agreed that no guidelines specific to avoiding the risks of topical corticosteroids are currently available, but she advised clinicians to be considering these risks nonetheless. In general, she suggested that topical steroids, like oral steroids, should be used at “the lowest dose for the shortest duration necessary to manage the underlying medical condition.”
The correlation between topical corticosteroids and increased risk of osteoporotic fracture, although not established previously in a large study, is not surprising, according to Victoria Werth, MD, chief of dermatology at the Philadelphia Veterans Affairs Hospital and professor of dermatology at the University of Pennsylvania, also in Philadelphia.
“Systemic absorption of potent topical steroids has previously been demonstrated with a rapid decrease in serum cortisol levels,” Dr. Werth said in an interview. She indicated that concern about the risk of osteoporosis imposed by use of potent steroids over large body surface areas is appropriate.
To minimize this risk, “it is reasonable to use the lowest dose of steroid possible and to try to substitute other medications when possible,” she said.
Dr. Egeberg reported financial relationships with Abbvie, Almirall, Bristol-Myers Squibb, Dermavant Sciences, Galderma, Janssen Pharmaceuticals, Eli Lilly, Novartis, Pfizer, Samsung, Bioepis, and UCB. Five authors had disclosures related to some of those pharmaceutical companies and/or others. Dr. Jackson had no disclosures.
In support of previously published case reports, in a dose-response relationship.
In a stepwise manner, the hazard ratios for major osteoporotic fracture (MOF) were found to start climbing incrementally for those with a cumulative topical steroid dose equivalent of more than 500 g of mometasone furoate when compared with exposure of 200-499 g, according to the team of investigators from the University of Copenhagen.
“Use of these drugs is very common, and we found an estimated population-attributable risk of as much as 4.3%,” the investigators reported in the study, published in JAMA Dermatology.
The retrospective cohort study drew data from the Danish National Patient Registry, which covers 99% of the country’s population. It was linked to the Danish National Prescription Registry, which captures data on pharmacy-dispensed medications. Data collected from the beginning of 2003 to the end of 2017 were evaluated.
Exposures to potent or very potent topical corticosteroids were converted into a single standard with potency equivalent to 1 mg/g of mometasone furoate. Four strata of exposure were compared to a reference exposure of 200-499 g. These were 500-999 g, 1,000-1,999 g, 2,000-9,999 g, and 10,000 g or greater.
For the first strata, the small increased risk for MOF did not reach significance (HR, 1.01; 95% confidence interval, 0.99-1.03), but each of the others did. These climbed from a 5% greater risk (HR 1.05 95% CI 1.02-1.08) for a cumulative exposure of 1,000 to 1,999 g, to a 10% greater risk (HR, 1.10; 95% CI, 1.07-1.13) for a cumulative exposure of 2,000-9,999 g, and finally to a 27% greater risk (HR, 1.27; 95% CI, 1.19-1.35) for a cumulative exposure of 10,000 g or higher.
The study included more than 700,000 individuals exposed to topical mometasone at a potency equivalent of 200 g or more over the study period. The reference group (200-499 g) was the largest (317,907 individuals). The first strata (500-999 g) included 186,359 patients; the second (1,000-1,999 g), 111,203 patients; the third (2,000-9,999 g), 94,334 patients; and the fifth (10,000 g or more), 13,448 patients.
“A 3% increase in the relative risk of osteoporosis and MOF was observed per doubling of the TCS dose,” according to the investigators.
Patients exposed to doses of high-potency topical steroids that put them at risk of MOF is limited but substantial, according to the senior author, Alexander Egeberg, MD, PhD, of the department of dermatology and allergy at Herlev and Gentofte Hospital, Copenhagen.
“It is true that the risk is modest for the average user of topical steroids,” Dr. Egeberg said in an interview. However, despite the fact that topical steroids are intended for short-term use, “2% of all our users had been exposed to the equivalent of 10,000 g of mometasone, which mean 100 tubes of 100 g.”
If the other two strata at significantly increased risk of MOF (greater than 1,000 g) are included, an additional 28% of all users are facing the potential for clinically significant osteoporosis, according to the Danish data.
The adverse effect of steroids on bone metabolism has been established previously, and several studies have linked systemic corticosteroid exposure, including inhaled corticosteroids, with increased risk of osteoporotic fracture. For example, one study showed that patients with chronic obstructive pulmonary disease on daily inhaled doses of the equivalent of fluticasone at or above 1,000 mcg for more than 4 years had about a 10% increased risk of MOF relative to those not exposed.
The data associate topical steroids with increased risk of osteoporotic fracture, but Dr. Egeberg said osteoporosis is not the only reason to use topical steroids prudently.
“It is important to keep in mind that osteoporosis and fractures are at the extreme end of the side-effect profile and that other side effects, such as striae formation, skin thinning, and dysregulated diabetes, can occur with much lower quantities of topical steroids,” Dr. Egeberg said
For avoiding this risk, “there are no specific cutoffs” recommended for topical steroids in current guidelines, but dermatologists should be aware that many of the indications for topical steroids, such as psoriasis and atopic dermatitis, involve skin with an impaired barrier function, exposing patients to an increased likelihood of absorption, according to Dr. Egeberg.
“A general rule of thumb that we use is that, if a patient with persistent disease activity requires a new prescription of the equivalent of 100 g mometasone every 1-2 months, it might be worth considering if there is a suitable alternative,” Dr. Egeberg said.
In an accompanying editorial, Rebecca D. Jackson, MD, of the division of endocrinology, diabetes, and metabolism in the department of internal medicine at Ohio State University, Columbus, agreed that no guidelines specific to avoiding the risks of topical corticosteroids are currently available, but she advised clinicians to be considering these risks nonetheless. In general, she suggested that topical steroids, like oral steroids, should be used at “the lowest dose for the shortest duration necessary to manage the underlying medical condition.”
The correlation between topical corticosteroids and increased risk of osteoporotic fracture, although not established previously in a large study, is not surprising, according to Victoria Werth, MD, chief of dermatology at the Philadelphia Veterans Affairs Hospital and professor of dermatology at the University of Pennsylvania, also in Philadelphia.
“Systemic absorption of potent topical steroids has previously been demonstrated with a rapid decrease in serum cortisol levels,” Dr. Werth said in an interview. She indicated that concern about the risk of osteoporosis imposed by use of potent steroids over large body surface areas is appropriate.
To minimize this risk, “it is reasonable to use the lowest dose of steroid possible and to try to substitute other medications when possible,” she said.
Dr. Egeberg reported financial relationships with Abbvie, Almirall, Bristol-Myers Squibb, Dermavant Sciences, Galderma, Janssen Pharmaceuticals, Eli Lilly, Novartis, Pfizer, Samsung, Bioepis, and UCB. Five authors had disclosures related to some of those pharmaceutical companies and/or others. Dr. Jackson had no disclosures.
In support of previously published case reports, in a dose-response relationship.
In a stepwise manner, the hazard ratios for major osteoporotic fracture (MOF) were found to start climbing incrementally for those with a cumulative topical steroid dose equivalent of more than 500 g of mometasone furoate when compared with exposure of 200-499 g, according to the team of investigators from the University of Copenhagen.
“Use of these drugs is very common, and we found an estimated population-attributable risk of as much as 4.3%,” the investigators reported in the study, published in JAMA Dermatology.
The retrospective cohort study drew data from the Danish National Patient Registry, which covers 99% of the country’s population. It was linked to the Danish National Prescription Registry, which captures data on pharmacy-dispensed medications. Data collected from the beginning of 2003 to the end of 2017 were evaluated.
Exposures to potent or very potent topical corticosteroids were converted into a single standard with potency equivalent to 1 mg/g of mometasone furoate. Four strata of exposure were compared to a reference exposure of 200-499 g. These were 500-999 g, 1,000-1,999 g, 2,000-9,999 g, and 10,000 g or greater.
For the first strata, the small increased risk for MOF did not reach significance (HR, 1.01; 95% confidence interval, 0.99-1.03), but each of the others did. These climbed from a 5% greater risk (HR 1.05 95% CI 1.02-1.08) for a cumulative exposure of 1,000 to 1,999 g, to a 10% greater risk (HR, 1.10; 95% CI, 1.07-1.13) for a cumulative exposure of 2,000-9,999 g, and finally to a 27% greater risk (HR, 1.27; 95% CI, 1.19-1.35) for a cumulative exposure of 10,000 g or higher.
The study included more than 700,000 individuals exposed to topical mometasone at a potency equivalent of 200 g or more over the study period. The reference group (200-499 g) was the largest (317,907 individuals). The first strata (500-999 g) included 186,359 patients; the second (1,000-1,999 g), 111,203 patients; the third (2,000-9,999 g), 94,334 patients; and the fifth (10,000 g or more), 13,448 patients.
“A 3% increase in the relative risk of osteoporosis and MOF was observed per doubling of the TCS dose,” according to the investigators.
Patients exposed to doses of high-potency topical steroids that put them at risk of MOF is limited but substantial, according to the senior author, Alexander Egeberg, MD, PhD, of the department of dermatology and allergy at Herlev and Gentofte Hospital, Copenhagen.
“It is true that the risk is modest for the average user of topical steroids,” Dr. Egeberg said in an interview. However, despite the fact that topical steroids are intended for short-term use, “2% of all our users had been exposed to the equivalent of 10,000 g of mometasone, which mean 100 tubes of 100 g.”
If the other two strata at significantly increased risk of MOF (greater than 1,000 g) are included, an additional 28% of all users are facing the potential for clinically significant osteoporosis, according to the Danish data.
The adverse effect of steroids on bone metabolism has been established previously, and several studies have linked systemic corticosteroid exposure, including inhaled corticosteroids, with increased risk of osteoporotic fracture. For example, one study showed that patients with chronic obstructive pulmonary disease on daily inhaled doses of the equivalent of fluticasone at or above 1,000 mcg for more than 4 years had about a 10% increased risk of MOF relative to those not exposed.
The data associate topical steroids with increased risk of osteoporotic fracture, but Dr. Egeberg said osteoporosis is not the only reason to use topical steroids prudently.
“It is important to keep in mind that osteoporosis and fractures are at the extreme end of the side-effect profile and that other side effects, such as striae formation, skin thinning, and dysregulated diabetes, can occur with much lower quantities of topical steroids,” Dr. Egeberg said
For avoiding this risk, “there are no specific cutoffs” recommended for topical steroids in current guidelines, but dermatologists should be aware that many of the indications for topical steroids, such as psoriasis and atopic dermatitis, involve skin with an impaired barrier function, exposing patients to an increased likelihood of absorption, according to Dr. Egeberg.
“A general rule of thumb that we use is that, if a patient with persistent disease activity requires a new prescription of the equivalent of 100 g mometasone every 1-2 months, it might be worth considering if there is a suitable alternative,” Dr. Egeberg said.
In an accompanying editorial, Rebecca D. Jackson, MD, of the division of endocrinology, diabetes, and metabolism in the department of internal medicine at Ohio State University, Columbus, agreed that no guidelines specific to avoiding the risks of topical corticosteroids are currently available, but she advised clinicians to be considering these risks nonetheless. In general, she suggested that topical steroids, like oral steroids, should be used at “the lowest dose for the shortest duration necessary to manage the underlying medical condition.”
The correlation between topical corticosteroids and increased risk of osteoporotic fracture, although not established previously in a large study, is not surprising, according to Victoria Werth, MD, chief of dermatology at the Philadelphia Veterans Affairs Hospital and professor of dermatology at the University of Pennsylvania, also in Philadelphia.
“Systemic absorption of potent topical steroids has previously been demonstrated with a rapid decrease in serum cortisol levels,” Dr. Werth said in an interview. She indicated that concern about the risk of osteoporosis imposed by use of potent steroids over large body surface areas is appropriate.
To minimize this risk, “it is reasonable to use the lowest dose of steroid possible and to try to substitute other medications when possible,” she said.
Dr. Egeberg reported financial relationships with Abbvie, Almirall, Bristol-Myers Squibb, Dermavant Sciences, Galderma, Janssen Pharmaceuticals, Eli Lilly, Novartis, Pfizer, Samsung, Bioepis, and UCB. Five authors had disclosures related to some of those pharmaceutical companies and/or others. Dr. Jackson had no disclosures.
FROM JAMA DERMATOLOGY
FDA approves first drug that protects against chemo-induced myelosuppression
A novel drug that offers multilineage protection from chemotherapy-induced myelosuppression has been approved by the Food and Drug Administration.
The drug, trilaciclib (Cosela, G1 Therapeutics) is administered intravenously as a 30-minute infusion within 4 hours prior to the start of chemotherapy. It is indicated specifically for use in adults with extensive-stage small-cell lung cancer (ES-SCLC) who are receiving chemotherapy.
Trilaciclib is a CDK4/6 inhibitor, and this action appears to protect normal bone marrow cells from the harmful effects of chemotherapy.
“For patients with extensive-stage small-cell lung cancer, protecting bone marrow function may help make their chemotherapy safer and allow them to complete their course of treatment on time and according to plan,” Albert Deisseroth, MD, PhD, of the FDA’s Center for Drug Evaluation and Research, said in an FDA press release.
First drug of its type
Trilaciclib “is the first and only therapy designed to help protect bone marrow (myeloprotection) when administered prior to treatment with chemotherapy,” according to the drug’s manufacturer.
Myelosuppression is one of the most severe adverse effects of chemotherapy, and it can be life-threatening. It can increase the risk of infection and lead to severe anemia and/or bleeding.
“These complications impact patients’ quality of life and may also result in chemotherapy dose reductions and delays,” Jeffrey Crawford, MD, of Duke Cancer Institute, Durham, N.C., said in a company press release.
“To date, approaches have included the use of growth factor agents to accelerate blood cell recovery after the bone marrow injury has occurred, along with antibiotics and transfusions as needed. By contrast, trilaciclib provides the first proactive approach to myelosuppression through a unique mechanism of action that helps protect the bone marrow from damage by chemotherapy.”
Approval based on randomized, placebo-controlled trials
The approval of trilaciclib is based on data from three randomized, double-blind, placebo-controlled studies, involving a total of 245 patients with ES-SCLC.
These patients were being treated with chemotherapy regimens that were based on the combination of carboplatin and etoposide (with or without the immunotherapy atezolizumab) or regimens that were based on topotecan.
Before receiving the chemotherapy, patients were randomly assigned to receive trilaciclib or placebo.
Results showed that patients who had received an infusion of trilaciclib before receiving chemotherapy had a lower chance of developing severe neutropenia compared with patients who received a placebo, the FDA noted. In addition, among the patients who did develop severe neutropenia, this had a shorter duration among patients who received trilaciclib than among those who received placebo.
The most common side effects of trilaciclib were fatigue; low levels of calcium, potassium, and phosphate in the blood; increased levels of aspartate aminotransferase; headache; and pneumonia.
The FDA noted that patients should also be advised about injection site reactions, acute drug hypersensitivity, interstitial lung disease/pneumonitis, and embryo-fetal toxicity.
The approval received a priority review, based on the drug’s breakthrough therapy designation. As is common for such products, the company plans postmarketing activities that will assess the effects of trilaciclib on disease progression or survival with at least a 2-year follow up. This clinical trial is scheduled to start in 2022.
A version of this article first appeared on Medscape.com.
A novel drug that offers multilineage protection from chemotherapy-induced myelosuppression has been approved by the Food and Drug Administration.
The drug, trilaciclib (Cosela, G1 Therapeutics) is administered intravenously as a 30-minute infusion within 4 hours prior to the start of chemotherapy. It is indicated specifically for use in adults with extensive-stage small-cell lung cancer (ES-SCLC) who are receiving chemotherapy.
Trilaciclib is a CDK4/6 inhibitor, and this action appears to protect normal bone marrow cells from the harmful effects of chemotherapy.
“For patients with extensive-stage small-cell lung cancer, protecting bone marrow function may help make their chemotherapy safer and allow them to complete their course of treatment on time and according to plan,” Albert Deisseroth, MD, PhD, of the FDA’s Center for Drug Evaluation and Research, said in an FDA press release.
First drug of its type
Trilaciclib “is the first and only therapy designed to help protect bone marrow (myeloprotection) when administered prior to treatment with chemotherapy,” according to the drug’s manufacturer.
Myelosuppression is one of the most severe adverse effects of chemotherapy, and it can be life-threatening. It can increase the risk of infection and lead to severe anemia and/or bleeding.
“These complications impact patients’ quality of life and may also result in chemotherapy dose reductions and delays,” Jeffrey Crawford, MD, of Duke Cancer Institute, Durham, N.C., said in a company press release.
“To date, approaches have included the use of growth factor agents to accelerate blood cell recovery after the bone marrow injury has occurred, along with antibiotics and transfusions as needed. By contrast, trilaciclib provides the first proactive approach to myelosuppression through a unique mechanism of action that helps protect the bone marrow from damage by chemotherapy.”
Approval based on randomized, placebo-controlled trials
The approval of trilaciclib is based on data from three randomized, double-blind, placebo-controlled studies, involving a total of 245 patients with ES-SCLC.
These patients were being treated with chemotherapy regimens that were based on the combination of carboplatin and etoposide (with or without the immunotherapy atezolizumab) or regimens that were based on topotecan.
Before receiving the chemotherapy, patients were randomly assigned to receive trilaciclib or placebo.
Results showed that patients who had received an infusion of trilaciclib before receiving chemotherapy had a lower chance of developing severe neutropenia compared with patients who received a placebo, the FDA noted. In addition, among the patients who did develop severe neutropenia, this had a shorter duration among patients who received trilaciclib than among those who received placebo.
The most common side effects of trilaciclib were fatigue; low levels of calcium, potassium, and phosphate in the blood; increased levels of aspartate aminotransferase; headache; and pneumonia.
The FDA noted that patients should also be advised about injection site reactions, acute drug hypersensitivity, interstitial lung disease/pneumonitis, and embryo-fetal toxicity.
The approval received a priority review, based on the drug’s breakthrough therapy designation. As is common for such products, the company plans postmarketing activities that will assess the effects of trilaciclib on disease progression or survival with at least a 2-year follow up. This clinical trial is scheduled to start in 2022.
A version of this article first appeared on Medscape.com.
A novel drug that offers multilineage protection from chemotherapy-induced myelosuppression has been approved by the Food and Drug Administration.
The drug, trilaciclib (Cosela, G1 Therapeutics) is administered intravenously as a 30-minute infusion within 4 hours prior to the start of chemotherapy. It is indicated specifically for use in adults with extensive-stage small-cell lung cancer (ES-SCLC) who are receiving chemotherapy.
Trilaciclib is a CDK4/6 inhibitor, and this action appears to protect normal bone marrow cells from the harmful effects of chemotherapy.
“For patients with extensive-stage small-cell lung cancer, protecting bone marrow function may help make their chemotherapy safer and allow them to complete their course of treatment on time and according to plan,” Albert Deisseroth, MD, PhD, of the FDA’s Center for Drug Evaluation and Research, said in an FDA press release.
First drug of its type
Trilaciclib “is the first and only therapy designed to help protect bone marrow (myeloprotection) when administered prior to treatment with chemotherapy,” according to the drug’s manufacturer.
Myelosuppression is one of the most severe adverse effects of chemotherapy, and it can be life-threatening. It can increase the risk of infection and lead to severe anemia and/or bleeding.
“These complications impact patients’ quality of life and may also result in chemotherapy dose reductions and delays,” Jeffrey Crawford, MD, of Duke Cancer Institute, Durham, N.C., said in a company press release.
“To date, approaches have included the use of growth factor agents to accelerate blood cell recovery after the bone marrow injury has occurred, along with antibiotics and transfusions as needed. By contrast, trilaciclib provides the first proactive approach to myelosuppression through a unique mechanism of action that helps protect the bone marrow from damage by chemotherapy.”
Approval based on randomized, placebo-controlled trials
The approval of trilaciclib is based on data from three randomized, double-blind, placebo-controlled studies, involving a total of 245 patients with ES-SCLC.
These patients were being treated with chemotherapy regimens that were based on the combination of carboplatin and etoposide (with or without the immunotherapy atezolizumab) or regimens that were based on topotecan.
Before receiving the chemotherapy, patients were randomly assigned to receive trilaciclib or placebo.
Results showed that patients who had received an infusion of trilaciclib before receiving chemotherapy had a lower chance of developing severe neutropenia compared with patients who received a placebo, the FDA noted. In addition, among the patients who did develop severe neutropenia, this had a shorter duration among patients who received trilaciclib than among those who received placebo.
The most common side effects of trilaciclib were fatigue; low levels of calcium, potassium, and phosphate in the blood; increased levels of aspartate aminotransferase; headache; and pneumonia.
The FDA noted that patients should also be advised about injection site reactions, acute drug hypersensitivity, interstitial lung disease/pneumonitis, and embryo-fetal toxicity.
The approval received a priority review, based on the drug’s breakthrough therapy designation. As is common for such products, the company plans postmarketing activities that will assess the effects of trilaciclib on disease progression or survival with at least a 2-year follow up. This clinical trial is scheduled to start in 2022.
A version of this article first appeared on Medscape.com.
Semaglutide for weight loss? A good first STEP, with caveats
The phase 3a STEP 1 trial that investigated the use of semaglutide (Novo Nordisk), a glucagonlike peptide–1 (GLP-1) agonist, for weight loss is aptly named, some say.
“In sum, we have a long way to go to control the obesity epidemic ... but on the face of it, the STEP 1 trial (like its name) is a good beginning,” wrote coeditorialists Julie R. Ingelfinger, MD, from Harvard Medical School, Boston, and a deputy editor of the New England Journal of Medicine, and Clifford J. Rosen, MD, from Tufts University School of Medicine, also in Boston.
The trial findings by John P.H. Wilding, DM, University of Liverpool (England), and colleagues and an accompanying editorial were published online Feb. 10, 2021, in the New England Journal of Medicine.
“The results are encouraging, with significantly more patients in the semaglutide group having clinically important weight loss,” Dr. Ingelfinger and Dr. Rosen stressed.
However, they also cautioned that “despite the positive results of this trial, the present study has some important limitations” and “there are concerns, including adverse events (mostly gastrointestinal – nausea, sometimes vomiting, and diarrhea) related primarily to the class of the agent.”
Two U.K. experts drew similar takeaways, speaking to the U.K. Science Media Centre.
“This was a well-designed study with unequivocal findings,” which showed that semaglutide “is indeed likely to be a game-changer in the fight against obesity,” according to Baptiste Leurent, PhD, London School of Hygiene and Tropical Medicine.
However, if the drug is approved at this dose for this use, patients would need close monitoring for gastrointestinal disorders, and “we also need to better understand what is happening once the treatment is stopped, and whether it could be taken for a shorter period of time.”
Sir Stephen O’Rahilly, MD, MRC Metabolic Diseases Unit, University of Cambridge (England), pointed out that “GLP-1 is made by cells in the intestine and levels increase in the blood after a meal, providing some of the signal to the brain that tells us we are ‘full,’ ” so GLP-1 agonists have been studied as appetite suppressants, in addition to their approved use to treat type 2 diabetes.
Only about 4.5% of participants in STEP 1 stopped taking semaglutide because of gastrointestinal issues, he noted, although more participants in that group reported problems with gallstones, which can follow rapid weight loss.
And “unlike some previous appetite suppressant drugs which caused significant psychological and psychiatric side effects, there is no evidence that semaglutide has any adverse effects of that nature,” Dr. O’Rahilly noted.
In sum, he said, “this is the start of a new era for obesity drug development with the future direction being to achieve levels of weight loss comparable to semaglutide, while having fewer side effects.”
‘Pressing need’ to address obesity; semaglutide filed for obesity
There is a “pressing need” to address the worldwide increase in obesity and weight-related coexisting conditions, Dr. Ingelfinger and Dr. Rosen noted.
Sustained long-term weight loss with diet and exercise is challenging; behavioral weight-loss strategies “fail more often than not,” bariatric surgery is invasive and often followed by eventual weight regain, they wrote.
In addition, said Dr. Wilding and colleagues, the “use of available [weight-loss] medications remains limited by modest efficacy, safety concerns, and cost.”
Subcutaneous semaglutide, approved for treating type 2 diabetes (as Ozempic) in adults at doses of up to 1 mg/week, induced weight loss at higher doses. The current study is part of the global Semaglutide Treatment Effect in People With Obesity program of four trials (STEP 1, 2, 3, and 4) that aimed to test the safety and efficacy of subcutaneous semaglutide 2.4 mg/week for weight loss.
Topline results from STEP 1 were presented June 4, 2020.
And as reported earlier, results from STEP 3 – a 68-week trial of semaglutide versus placebo in 611 participants who all received very intensive diet and exercise counseling – were presented at the virtual ObesityWeek 2020 meeting.
The four trials of semaglutide for weight loss have been completed and the data were submitted to the Food and Drug Administration on Dec. 4, 2020 (with a decision expected within 6 months) and to the European Medicines Agency on Dec. 18, 2020.
Most patients had 5% weight loss with semaglutide
The STEP 1 trial enrolled 1,961 adults with a body mass index (BMI) of at least 30 kg/m2 or at least 27 with at least one weight-related coexisting condition, but without type 2 diabetes, at 129 sites in 16 countries in Asia, Europe, North America, and South America.
Participants were a mean age of 47 and three-quarters were women. Most participants were White (76%), followed by Asian (13%), Black or African American (6%), or other (5%).
On average, they had a BMI of 38 and weighed 105 kg. Three-quarters had one or more coexisting conditions.
Participants were randomized to receive semaglutide (1,306 patients) or placebo (655 patients), added to lifestyle intervention.
Everyone received 17 monthly individual counseling sessions during which they learned about adhering to a diet with a 500-calorie/day deficit, were encouraged to build up to walking 150 minutes each week, and recorded their daily diet and exercise (in a diary or using an app).
Semaglutide was administered with a prefilled pen injector at a dose of 0.25 mg/week for the first 4 weeks, escalated to 2.4 mg/week by week 16 (or lower if the patient had unacceptable side effects).
At 68 weeks, participants in the semaglutide versus placebo group had greater mean weight loss (14.9% vs. 2.4%, or 15.3 kg vs. 2.6 kg).
Participants in the semaglutide versus placebo group were much more likely to have lost at least 5% of their initial weight (86% vs. 31.5%) or at least 10% of their initial weight (69.1% vs. 12.0%), or at least 15% of their initial weight (50.5% vs. 4.9%; P < .001 for all three comparisons).
About 80% of participants adhered to the study treatment. A third of participants in the semaglutide group who completed the study lost at least 20% of their initial weight, which approaches the 20%-30% reported weight loss 1-3 years after sleeve gastrectomy, the researchers noted.
Participants in the semaglutide group also had greater improvements in waist circumference and levels of hemoglobin A1c, C-reactive protein (a marker of inflammation), and fasting lipids, as well as in physical function scores on SF-36 and IWQOL-Lite-CT questionnaires.
In their editorial, Dr. Ingelfinger and Dr. Rosen noted that “daily oral semaglutide [already approved in 7-mg and 14-mg doses for the treatment of type 2 diabetes as Rybelsus] might be more appealing to many people,” as a weight-loss medication than a once-weekly subcutaneous dose. Semaglutide is the first GLP-1 agonist available as an oral agent.
The ongoing Semaglutide Effects on Heart Disease and Stroke in Patients With Overweight or Obesity (SELECT) trial (with expected completion in 2023) will shed light on cardiovascular outcomes after 2.5-5 years.
GI disorders and ‘important limitations’
More participants in the semaglutide than the placebo group reported gastrointestinal disorders (typically nausea, diarrhea, vomiting, and constipation; 74.2% vs. 47.9%), which were mostly transient and mild to moderate in severity, but also led to more treatment discontinuation (7.0% vs. 3.1%).
More patients in the semaglutide versus placebo group had a gall bladder–related disorder (2.6% vs. 1.2%, mostly cholelithiasis) and mild acute pancreatitis (3 vs. 0 participants), but there were no between-group differences in neoplasms.
Dr. Wilding and colleagues acknowledge the limitations of the study, including the fact that it enrolled mainly women, mainly non-White participants, was relatively short, and excluded patients with type 2 diabetes.
Mean placebo-corrected weight loss with 2.4 mg/weekly subcutaneous semaglutide was greater than with 3.0 mg once-daily subcutaneous liraglutide (Saxenda, Novo Nordisk) – the only GLP-1 agonist approved for weight management – in the 56-week SCALE trial (12.4% vs. 4.5%); however, the two studies had different populations.
The study was supported by Novo Nordisk. Dr. Ingelfinger is a deputy editor and Dr. Rosen is an associate editor of the New England Journal of Medicine. Dr. Ingelfinger, Dr. Rosen, and Dr. Leurent have reported no relevant financial relationships. Dr. O’Rahilly has a current research collaboration with Novo Nordisk scientists in an unrelated area and has been a consultant for the company.
A version of this article first appeared on Medscape.com.
The phase 3a STEP 1 trial that investigated the use of semaglutide (Novo Nordisk), a glucagonlike peptide–1 (GLP-1) agonist, for weight loss is aptly named, some say.
“In sum, we have a long way to go to control the obesity epidemic ... but on the face of it, the STEP 1 trial (like its name) is a good beginning,” wrote coeditorialists Julie R. Ingelfinger, MD, from Harvard Medical School, Boston, and a deputy editor of the New England Journal of Medicine, and Clifford J. Rosen, MD, from Tufts University School of Medicine, also in Boston.
The trial findings by John P.H. Wilding, DM, University of Liverpool (England), and colleagues and an accompanying editorial were published online Feb. 10, 2021, in the New England Journal of Medicine.
“The results are encouraging, with significantly more patients in the semaglutide group having clinically important weight loss,” Dr. Ingelfinger and Dr. Rosen stressed.
However, they also cautioned that “despite the positive results of this trial, the present study has some important limitations” and “there are concerns, including adverse events (mostly gastrointestinal – nausea, sometimes vomiting, and diarrhea) related primarily to the class of the agent.”
Two U.K. experts drew similar takeaways, speaking to the U.K. Science Media Centre.
“This was a well-designed study with unequivocal findings,” which showed that semaglutide “is indeed likely to be a game-changer in the fight against obesity,” according to Baptiste Leurent, PhD, London School of Hygiene and Tropical Medicine.
However, if the drug is approved at this dose for this use, patients would need close monitoring for gastrointestinal disorders, and “we also need to better understand what is happening once the treatment is stopped, and whether it could be taken for a shorter period of time.”
Sir Stephen O’Rahilly, MD, MRC Metabolic Diseases Unit, University of Cambridge (England), pointed out that “GLP-1 is made by cells in the intestine and levels increase in the blood after a meal, providing some of the signal to the brain that tells us we are ‘full,’ ” so GLP-1 agonists have been studied as appetite suppressants, in addition to their approved use to treat type 2 diabetes.
Only about 4.5% of participants in STEP 1 stopped taking semaglutide because of gastrointestinal issues, he noted, although more participants in that group reported problems with gallstones, which can follow rapid weight loss.
And “unlike some previous appetite suppressant drugs which caused significant psychological and psychiatric side effects, there is no evidence that semaglutide has any adverse effects of that nature,” Dr. O’Rahilly noted.
In sum, he said, “this is the start of a new era for obesity drug development with the future direction being to achieve levels of weight loss comparable to semaglutide, while having fewer side effects.”
‘Pressing need’ to address obesity; semaglutide filed for obesity
There is a “pressing need” to address the worldwide increase in obesity and weight-related coexisting conditions, Dr. Ingelfinger and Dr. Rosen noted.
Sustained long-term weight loss with diet and exercise is challenging; behavioral weight-loss strategies “fail more often than not,” bariatric surgery is invasive and often followed by eventual weight regain, they wrote.
In addition, said Dr. Wilding and colleagues, the “use of available [weight-loss] medications remains limited by modest efficacy, safety concerns, and cost.”
Subcutaneous semaglutide, approved for treating type 2 diabetes (as Ozempic) in adults at doses of up to 1 mg/week, induced weight loss at higher doses. The current study is part of the global Semaglutide Treatment Effect in People With Obesity program of four trials (STEP 1, 2, 3, and 4) that aimed to test the safety and efficacy of subcutaneous semaglutide 2.4 mg/week for weight loss.
Topline results from STEP 1 were presented June 4, 2020.
And as reported earlier, results from STEP 3 – a 68-week trial of semaglutide versus placebo in 611 participants who all received very intensive diet and exercise counseling – were presented at the virtual ObesityWeek 2020 meeting.
The four trials of semaglutide for weight loss have been completed and the data were submitted to the Food and Drug Administration on Dec. 4, 2020 (with a decision expected within 6 months) and to the European Medicines Agency on Dec. 18, 2020.
Most patients had 5% weight loss with semaglutide
The STEP 1 trial enrolled 1,961 adults with a body mass index (BMI) of at least 30 kg/m2 or at least 27 with at least one weight-related coexisting condition, but without type 2 diabetes, at 129 sites in 16 countries in Asia, Europe, North America, and South America.
Participants were a mean age of 47 and three-quarters were women. Most participants were White (76%), followed by Asian (13%), Black or African American (6%), or other (5%).
On average, they had a BMI of 38 and weighed 105 kg. Three-quarters had one or more coexisting conditions.
Participants were randomized to receive semaglutide (1,306 patients) or placebo (655 patients), added to lifestyle intervention.
Everyone received 17 monthly individual counseling sessions during which they learned about adhering to a diet with a 500-calorie/day deficit, were encouraged to build up to walking 150 minutes each week, and recorded their daily diet and exercise (in a diary or using an app).
Semaglutide was administered with a prefilled pen injector at a dose of 0.25 mg/week for the first 4 weeks, escalated to 2.4 mg/week by week 16 (or lower if the patient had unacceptable side effects).
At 68 weeks, participants in the semaglutide versus placebo group had greater mean weight loss (14.9% vs. 2.4%, or 15.3 kg vs. 2.6 kg).
Participants in the semaglutide versus placebo group were much more likely to have lost at least 5% of their initial weight (86% vs. 31.5%) or at least 10% of their initial weight (69.1% vs. 12.0%), or at least 15% of their initial weight (50.5% vs. 4.9%; P < .001 for all three comparisons).
About 80% of participants adhered to the study treatment. A third of participants in the semaglutide group who completed the study lost at least 20% of their initial weight, which approaches the 20%-30% reported weight loss 1-3 years after sleeve gastrectomy, the researchers noted.
Participants in the semaglutide group also had greater improvements in waist circumference and levels of hemoglobin A1c, C-reactive protein (a marker of inflammation), and fasting lipids, as well as in physical function scores on SF-36 and IWQOL-Lite-CT questionnaires.
In their editorial, Dr. Ingelfinger and Dr. Rosen noted that “daily oral semaglutide [already approved in 7-mg and 14-mg doses for the treatment of type 2 diabetes as Rybelsus] might be more appealing to many people,” as a weight-loss medication than a once-weekly subcutaneous dose. Semaglutide is the first GLP-1 agonist available as an oral agent.
The ongoing Semaglutide Effects on Heart Disease and Stroke in Patients With Overweight or Obesity (SELECT) trial (with expected completion in 2023) will shed light on cardiovascular outcomes after 2.5-5 years.
GI disorders and ‘important limitations’
More participants in the semaglutide than the placebo group reported gastrointestinal disorders (typically nausea, diarrhea, vomiting, and constipation; 74.2% vs. 47.9%), which were mostly transient and mild to moderate in severity, but also led to more treatment discontinuation (7.0% vs. 3.1%).
More patients in the semaglutide versus placebo group had a gall bladder–related disorder (2.6% vs. 1.2%, mostly cholelithiasis) and mild acute pancreatitis (3 vs. 0 participants), but there were no between-group differences in neoplasms.
Dr. Wilding and colleagues acknowledge the limitations of the study, including the fact that it enrolled mainly women, mainly non-White participants, was relatively short, and excluded patients with type 2 diabetes.
Mean placebo-corrected weight loss with 2.4 mg/weekly subcutaneous semaglutide was greater than with 3.0 mg once-daily subcutaneous liraglutide (Saxenda, Novo Nordisk) – the only GLP-1 agonist approved for weight management – in the 56-week SCALE trial (12.4% vs. 4.5%); however, the two studies had different populations.
The study was supported by Novo Nordisk. Dr. Ingelfinger is a deputy editor and Dr. Rosen is an associate editor of the New England Journal of Medicine. Dr. Ingelfinger, Dr. Rosen, and Dr. Leurent have reported no relevant financial relationships. Dr. O’Rahilly has a current research collaboration with Novo Nordisk scientists in an unrelated area and has been a consultant for the company.
A version of this article first appeared on Medscape.com.
The phase 3a STEP 1 trial that investigated the use of semaglutide (Novo Nordisk), a glucagonlike peptide–1 (GLP-1) agonist, for weight loss is aptly named, some say.
“In sum, we have a long way to go to control the obesity epidemic ... but on the face of it, the STEP 1 trial (like its name) is a good beginning,” wrote coeditorialists Julie R. Ingelfinger, MD, from Harvard Medical School, Boston, and a deputy editor of the New England Journal of Medicine, and Clifford J. Rosen, MD, from Tufts University School of Medicine, also in Boston.
The trial findings by John P.H. Wilding, DM, University of Liverpool (England), and colleagues and an accompanying editorial were published online Feb. 10, 2021, in the New England Journal of Medicine.
“The results are encouraging, with significantly more patients in the semaglutide group having clinically important weight loss,” Dr. Ingelfinger and Dr. Rosen stressed.
However, they also cautioned that “despite the positive results of this trial, the present study has some important limitations” and “there are concerns, including adverse events (mostly gastrointestinal – nausea, sometimes vomiting, and diarrhea) related primarily to the class of the agent.”
Two U.K. experts drew similar takeaways, speaking to the U.K. Science Media Centre.
“This was a well-designed study with unequivocal findings,” which showed that semaglutide “is indeed likely to be a game-changer in the fight against obesity,” according to Baptiste Leurent, PhD, London School of Hygiene and Tropical Medicine.
However, if the drug is approved at this dose for this use, patients would need close monitoring for gastrointestinal disorders, and “we also need to better understand what is happening once the treatment is stopped, and whether it could be taken for a shorter period of time.”
Sir Stephen O’Rahilly, MD, MRC Metabolic Diseases Unit, University of Cambridge (England), pointed out that “GLP-1 is made by cells in the intestine and levels increase in the blood after a meal, providing some of the signal to the brain that tells us we are ‘full,’ ” so GLP-1 agonists have been studied as appetite suppressants, in addition to their approved use to treat type 2 diabetes.
Only about 4.5% of participants in STEP 1 stopped taking semaglutide because of gastrointestinal issues, he noted, although more participants in that group reported problems with gallstones, which can follow rapid weight loss.
And “unlike some previous appetite suppressant drugs which caused significant psychological and psychiatric side effects, there is no evidence that semaglutide has any adverse effects of that nature,” Dr. O’Rahilly noted.
In sum, he said, “this is the start of a new era for obesity drug development with the future direction being to achieve levels of weight loss comparable to semaglutide, while having fewer side effects.”
‘Pressing need’ to address obesity; semaglutide filed for obesity
There is a “pressing need” to address the worldwide increase in obesity and weight-related coexisting conditions, Dr. Ingelfinger and Dr. Rosen noted.
Sustained long-term weight loss with diet and exercise is challenging; behavioral weight-loss strategies “fail more often than not,” bariatric surgery is invasive and often followed by eventual weight regain, they wrote.
In addition, said Dr. Wilding and colleagues, the “use of available [weight-loss] medications remains limited by modest efficacy, safety concerns, and cost.”
Subcutaneous semaglutide, approved for treating type 2 diabetes (as Ozempic) in adults at doses of up to 1 mg/week, induced weight loss at higher doses. The current study is part of the global Semaglutide Treatment Effect in People With Obesity program of four trials (STEP 1, 2, 3, and 4) that aimed to test the safety and efficacy of subcutaneous semaglutide 2.4 mg/week for weight loss.
Topline results from STEP 1 were presented June 4, 2020.
And as reported earlier, results from STEP 3 – a 68-week trial of semaglutide versus placebo in 611 participants who all received very intensive diet and exercise counseling – were presented at the virtual ObesityWeek 2020 meeting.
The four trials of semaglutide for weight loss have been completed and the data were submitted to the Food and Drug Administration on Dec. 4, 2020 (with a decision expected within 6 months) and to the European Medicines Agency on Dec. 18, 2020.
Most patients had 5% weight loss with semaglutide
The STEP 1 trial enrolled 1,961 adults with a body mass index (BMI) of at least 30 kg/m2 or at least 27 with at least one weight-related coexisting condition, but without type 2 diabetes, at 129 sites in 16 countries in Asia, Europe, North America, and South America.
Participants were a mean age of 47 and three-quarters were women. Most participants were White (76%), followed by Asian (13%), Black or African American (6%), or other (5%).
On average, they had a BMI of 38 and weighed 105 kg. Three-quarters had one or more coexisting conditions.
Participants were randomized to receive semaglutide (1,306 patients) or placebo (655 patients), added to lifestyle intervention.
Everyone received 17 monthly individual counseling sessions during which they learned about adhering to a diet with a 500-calorie/day deficit, were encouraged to build up to walking 150 minutes each week, and recorded their daily diet and exercise (in a diary or using an app).
Semaglutide was administered with a prefilled pen injector at a dose of 0.25 mg/week for the first 4 weeks, escalated to 2.4 mg/week by week 16 (or lower if the patient had unacceptable side effects).
At 68 weeks, participants in the semaglutide versus placebo group had greater mean weight loss (14.9% vs. 2.4%, or 15.3 kg vs. 2.6 kg).
Participants in the semaglutide versus placebo group were much more likely to have lost at least 5% of their initial weight (86% vs. 31.5%) or at least 10% of their initial weight (69.1% vs. 12.0%), or at least 15% of their initial weight (50.5% vs. 4.9%; P < .001 for all three comparisons).
About 80% of participants adhered to the study treatment. A third of participants in the semaglutide group who completed the study lost at least 20% of their initial weight, which approaches the 20%-30% reported weight loss 1-3 years after sleeve gastrectomy, the researchers noted.
Participants in the semaglutide group also had greater improvements in waist circumference and levels of hemoglobin A1c, C-reactive protein (a marker of inflammation), and fasting lipids, as well as in physical function scores on SF-36 and IWQOL-Lite-CT questionnaires.
In their editorial, Dr. Ingelfinger and Dr. Rosen noted that “daily oral semaglutide [already approved in 7-mg and 14-mg doses for the treatment of type 2 diabetes as Rybelsus] might be more appealing to many people,” as a weight-loss medication than a once-weekly subcutaneous dose. Semaglutide is the first GLP-1 agonist available as an oral agent.
The ongoing Semaglutide Effects on Heart Disease and Stroke in Patients With Overweight or Obesity (SELECT) trial (with expected completion in 2023) will shed light on cardiovascular outcomes after 2.5-5 years.
GI disorders and ‘important limitations’
More participants in the semaglutide than the placebo group reported gastrointestinal disorders (typically nausea, diarrhea, vomiting, and constipation; 74.2% vs. 47.9%), which were mostly transient and mild to moderate in severity, but also led to more treatment discontinuation (7.0% vs. 3.1%).
More patients in the semaglutide versus placebo group had a gall bladder–related disorder (2.6% vs. 1.2%, mostly cholelithiasis) and mild acute pancreatitis (3 vs. 0 participants), but there were no between-group differences in neoplasms.
Dr. Wilding and colleagues acknowledge the limitations of the study, including the fact that it enrolled mainly women, mainly non-White participants, was relatively short, and excluded patients with type 2 diabetes.
Mean placebo-corrected weight loss with 2.4 mg/weekly subcutaneous semaglutide was greater than with 3.0 mg once-daily subcutaneous liraglutide (Saxenda, Novo Nordisk) – the only GLP-1 agonist approved for weight management – in the 56-week SCALE trial (12.4% vs. 4.5%); however, the two studies had different populations.
The study was supported by Novo Nordisk. Dr. Ingelfinger is a deputy editor and Dr. Rosen is an associate editor of the New England Journal of Medicine. Dr. Ingelfinger, Dr. Rosen, and Dr. Leurent have reported no relevant financial relationships. Dr. O’Rahilly has a current research collaboration with Novo Nordisk scientists in an unrelated area and has been a consultant for the company.
A version of this article first appeared on Medscape.com.
Antidepressants may scupper efficacy of MDMA for PTSD
Pooled data from four phase 2 trials reveal that patients with recent SSRI exposure were significantly more likely to continue to meet PTSD diagnostic criteria after methylenedioxymethamphetamine (MDMA)-assisted psychotherapy than their peers who had not recently taken SSRIs.
Although preliminary, the findings have implications for clinical practice if MDMA-assisted psychotherapy is approved by the Food and Drug Administration, Allison Feduccia, PhD, study coauthor and founder of the education platform Psychedelic.Support, said in an interview.
“As psychedelic medicines become available, it’s going to be important that we try to understand what factors impact the response rate and if there are ways that we can improve the treatment outcomes. Allowing for a longer period for tapering completely off SSRIs before initiating MDMA sessions might increase the effectiveness of MDMA,” Dr. Feduccia said.
The study was published online Nov. 20, 2020, in Psychopharmacology (doi: 10.1007/s00213-020-05710-w).
Reduced response
The primary mechanism of action of MDMA involves the same reuptake transporters that are targeted by antidepressant medications commonly prescribed for PTSD. These medications include SSRIs, serotonin-norepinephrine reuptake inhibitors (SNRIs), NRIs, and norepinephrine-dopamine reuptake inhibitors (NDRIs).
Prior research shows that, when MDMA is coadministered with a reuptake inhibitor, subjective and psychological effects of the therapy are attenuated.
The researchers sought to determine whether or not recent tapering off of an antidepressant that targets the same primary binding sites as MDMA would affect treatment response. They analyzed data on 50 adults who underwent two sessions of MDMA-assisted psychotherapy in phase 2 clinical trials.
For 16 of these patients, SSRI therapy was tapered off prior to the MDMA sessions. For 34 patients, SSRI therapy was not tapered off, because the patients had not been taking the medication at the time of initial study screening (nontaper group).
The taper protocols specified that medications be tapered gradually over a period of weeks to minimize withdrawal symptoms and for them to be discontinued at least five half-lives of each drug prior to MDMA administration.
Demographics, baseline PTSD, and depression severity were similar between the taper and the nontaper groups. Participants in the studies had chronic PTSD (symptoms lasting >6 months). Severity scores on the Clinician-Administered PTSD Scale for DSM IV (CAPS-IV) were at least 50.
After MDMA-assisted psychotherapy, the nontaper group had significantly lower (better) CAPS-IV total scores, compared with the taper group (mean, 45.7 vs. 70.3; P = .009).
About two-thirds (63.6%) of the nontaper group no longer met PTSD criteria after MDMA-assisted therapy, compared with only 25% of those in the taper group.
The nontaper group also had lower depression symptom severity scores on the Beck Depression Inventory–II, compared with the taper group (mean, 12.7 vs. 22.6; P = .010).
“Another really interesting” observation, said Dr. Feduccia, is that the expected increases in systolic and diastolic blood pressure following MDMA administration were reduced in the taper group, compared with the nontaper group.
“This suggests that MDMA didn’t have the same physiological response in individuals who tapered SSRIs. This should be followed up,” she said.
The investigators offerred several potential mechanisms for the negative effect of recent SSRI use on MDMA-assisted psychotherapy for PTSD.
These include the down-regulation of binding sites (serotonin, dopamine, and/or norepinephrine) related to SSRI use, reduced MDMA treatment-relevant increases in blood pressure in patients with recent SSRI use, and the possibility that withdrawal symptoms from SSRIs may reduce the effectiveness of MDMA psychotherapy.
Important clinical implications
In a comment, Steven R. Thorp, PhD, professor at Alliant International University, San Diego, said the findings are “very interesting” and likely “not well known.”
“There has been great interest in MDMA-assisted psychotherapy in recent years, and if this finding is replicated, it will have important implications for that research,” Dr. Thorp said.
“Although psychotherapy is often preferred by clients with PTSD, compared to medications, and typically shows efficacy that is as strong or stronger (and longer lasting) than medications, many individuals with PTSD are provided with medication only,” Dr. Thorp noted.
“This study suggests that, in addition to the other potential disadvantages of medications (e.g., cost, side effects, potential for addiction), those who take SSRIs, SNRIs, NRIs, and NDRIs for PTSD may also benefit less from MDMA-assisted psychotherapy,” Dr. Thorp added.
The four phase 2 studies used in the analysis were sponsored by the Multidisciplinary Association for Psychedelic Studies, a nonprofit organization. Dr. Feduccia received salary support for full-time employment with MAPS Public Benefit Corporation. Dr. Thorp disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pooled data from four phase 2 trials reveal that patients with recent SSRI exposure were significantly more likely to continue to meet PTSD diagnostic criteria after methylenedioxymethamphetamine (MDMA)-assisted psychotherapy than their peers who had not recently taken SSRIs.
Although preliminary, the findings have implications for clinical practice if MDMA-assisted psychotherapy is approved by the Food and Drug Administration, Allison Feduccia, PhD, study coauthor and founder of the education platform Psychedelic.Support, said in an interview.
“As psychedelic medicines become available, it’s going to be important that we try to understand what factors impact the response rate and if there are ways that we can improve the treatment outcomes. Allowing for a longer period for tapering completely off SSRIs before initiating MDMA sessions might increase the effectiveness of MDMA,” Dr. Feduccia said.
The study was published online Nov. 20, 2020, in Psychopharmacology (doi: 10.1007/s00213-020-05710-w).
Reduced response
The primary mechanism of action of MDMA involves the same reuptake transporters that are targeted by antidepressant medications commonly prescribed for PTSD. These medications include SSRIs, serotonin-norepinephrine reuptake inhibitors (SNRIs), NRIs, and norepinephrine-dopamine reuptake inhibitors (NDRIs).
Prior research shows that, when MDMA is coadministered with a reuptake inhibitor, subjective and psychological effects of the therapy are attenuated.
The researchers sought to determine whether or not recent tapering off of an antidepressant that targets the same primary binding sites as MDMA would affect treatment response. They analyzed data on 50 adults who underwent two sessions of MDMA-assisted psychotherapy in phase 2 clinical trials.
For 16 of these patients, SSRI therapy was tapered off prior to the MDMA sessions. For 34 patients, SSRI therapy was not tapered off, because the patients had not been taking the medication at the time of initial study screening (nontaper group).
The taper protocols specified that medications be tapered gradually over a period of weeks to minimize withdrawal symptoms and for them to be discontinued at least five half-lives of each drug prior to MDMA administration.
Demographics, baseline PTSD, and depression severity were similar between the taper and the nontaper groups. Participants in the studies had chronic PTSD (symptoms lasting >6 months). Severity scores on the Clinician-Administered PTSD Scale for DSM IV (CAPS-IV) were at least 50.
After MDMA-assisted psychotherapy, the nontaper group had significantly lower (better) CAPS-IV total scores, compared with the taper group (mean, 45.7 vs. 70.3; P = .009).
About two-thirds (63.6%) of the nontaper group no longer met PTSD criteria after MDMA-assisted therapy, compared with only 25% of those in the taper group.
The nontaper group also had lower depression symptom severity scores on the Beck Depression Inventory–II, compared with the taper group (mean, 12.7 vs. 22.6; P = .010).
“Another really interesting” observation, said Dr. Feduccia, is that the expected increases in systolic and diastolic blood pressure following MDMA administration were reduced in the taper group, compared with the nontaper group.
“This suggests that MDMA didn’t have the same physiological response in individuals who tapered SSRIs. This should be followed up,” she said.
The investigators offerred several potential mechanisms for the negative effect of recent SSRI use on MDMA-assisted psychotherapy for PTSD.
These include the down-regulation of binding sites (serotonin, dopamine, and/or norepinephrine) related to SSRI use, reduced MDMA treatment-relevant increases in blood pressure in patients with recent SSRI use, and the possibility that withdrawal symptoms from SSRIs may reduce the effectiveness of MDMA psychotherapy.
Important clinical implications
In a comment, Steven R. Thorp, PhD, professor at Alliant International University, San Diego, said the findings are “very interesting” and likely “not well known.”
“There has been great interest in MDMA-assisted psychotherapy in recent years, and if this finding is replicated, it will have important implications for that research,” Dr. Thorp said.
“Although psychotherapy is often preferred by clients with PTSD, compared to medications, and typically shows efficacy that is as strong or stronger (and longer lasting) than medications, many individuals with PTSD are provided with medication only,” Dr. Thorp noted.
“This study suggests that, in addition to the other potential disadvantages of medications (e.g., cost, side effects, potential for addiction), those who take SSRIs, SNRIs, NRIs, and NDRIs for PTSD may also benefit less from MDMA-assisted psychotherapy,” Dr. Thorp added.
The four phase 2 studies used in the analysis were sponsored by the Multidisciplinary Association for Psychedelic Studies, a nonprofit organization. Dr. Feduccia received salary support for full-time employment with MAPS Public Benefit Corporation. Dr. Thorp disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pooled data from four phase 2 trials reveal that patients with recent SSRI exposure were significantly more likely to continue to meet PTSD diagnostic criteria after methylenedioxymethamphetamine (MDMA)-assisted psychotherapy than their peers who had not recently taken SSRIs.
Although preliminary, the findings have implications for clinical practice if MDMA-assisted psychotherapy is approved by the Food and Drug Administration, Allison Feduccia, PhD, study coauthor and founder of the education platform Psychedelic.Support, said in an interview.
“As psychedelic medicines become available, it’s going to be important that we try to understand what factors impact the response rate and if there are ways that we can improve the treatment outcomes. Allowing for a longer period for tapering completely off SSRIs before initiating MDMA sessions might increase the effectiveness of MDMA,” Dr. Feduccia said.
The study was published online Nov. 20, 2020, in Psychopharmacology (doi: 10.1007/s00213-020-05710-w).
Reduced response
The primary mechanism of action of MDMA involves the same reuptake transporters that are targeted by antidepressant medications commonly prescribed for PTSD. These medications include SSRIs, serotonin-norepinephrine reuptake inhibitors (SNRIs), NRIs, and norepinephrine-dopamine reuptake inhibitors (NDRIs).
Prior research shows that, when MDMA is coadministered with a reuptake inhibitor, subjective and psychological effects of the therapy are attenuated.
The researchers sought to determine whether or not recent tapering off of an antidepressant that targets the same primary binding sites as MDMA would affect treatment response. They analyzed data on 50 adults who underwent two sessions of MDMA-assisted psychotherapy in phase 2 clinical trials.
For 16 of these patients, SSRI therapy was tapered off prior to the MDMA sessions. For 34 patients, SSRI therapy was not tapered off, because the patients had not been taking the medication at the time of initial study screening (nontaper group).
The taper protocols specified that medications be tapered gradually over a period of weeks to minimize withdrawal symptoms and for them to be discontinued at least five half-lives of each drug prior to MDMA administration.
Demographics, baseline PTSD, and depression severity were similar between the taper and the nontaper groups. Participants in the studies had chronic PTSD (symptoms lasting >6 months). Severity scores on the Clinician-Administered PTSD Scale for DSM IV (CAPS-IV) were at least 50.
After MDMA-assisted psychotherapy, the nontaper group had significantly lower (better) CAPS-IV total scores, compared with the taper group (mean, 45.7 vs. 70.3; P = .009).
About two-thirds (63.6%) of the nontaper group no longer met PTSD criteria after MDMA-assisted therapy, compared with only 25% of those in the taper group.
The nontaper group also had lower depression symptom severity scores on the Beck Depression Inventory–II, compared with the taper group (mean, 12.7 vs. 22.6; P = .010).
“Another really interesting” observation, said Dr. Feduccia, is that the expected increases in systolic and diastolic blood pressure following MDMA administration were reduced in the taper group, compared with the nontaper group.
“This suggests that MDMA didn’t have the same physiological response in individuals who tapered SSRIs. This should be followed up,” she said.
The investigators offerred several potential mechanisms for the negative effect of recent SSRI use on MDMA-assisted psychotherapy for PTSD.
These include the down-regulation of binding sites (serotonin, dopamine, and/or norepinephrine) related to SSRI use, reduced MDMA treatment-relevant increases in blood pressure in patients with recent SSRI use, and the possibility that withdrawal symptoms from SSRIs may reduce the effectiveness of MDMA psychotherapy.
Important clinical implications
In a comment, Steven R. Thorp, PhD, professor at Alliant International University, San Diego, said the findings are “very interesting” and likely “not well known.”
“There has been great interest in MDMA-assisted psychotherapy in recent years, and if this finding is replicated, it will have important implications for that research,” Dr. Thorp said.
“Although psychotherapy is often preferred by clients with PTSD, compared to medications, and typically shows efficacy that is as strong or stronger (and longer lasting) than medications, many individuals with PTSD are provided with medication only,” Dr. Thorp noted.
“This study suggests that, in addition to the other potential disadvantages of medications (e.g., cost, side effects, potential for addiction), those who take SSRIs, SNRIs, NRIs, and NDRIs for PTSD may also benefit less from MDMA-assisted psychotherapy,” Dr. Thorp added.
The four phase 2 studies used in the analysis were sponsored by the Multidisciplinary Association for Psychedelic Studies, a nonprofit organization. Dr. Feduccia received salary support for full-time employment with MAPS Public Benefit Corporation. Dr. Thorp disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
‘Unprecedented’ long-term survival after immunotherapy in pretreated NSCLC
Longer-term survival with immunotherapy for patients with non–small cell lung cancer (NSCLC) is once again being applauded by experts in the field.
This time, the data come from trials that tested immunotherapy in the second-line setting for patients who had experienced disease progression with platinum-based chemotherapy. The latest 5-year follow-up from two landmark trials, one with pembrolizumab, the other with nivolumab, show that the survival benefit can persist for years after treatment is stopped.
“These are unprecedented data,” Fred R. Hirsch, MD, PhD, executive director of the Center for Thoracic Oncology at the Tisch Cancer Institute, New York, said in an interview. He was not involved in either trial and was approached for comment.
Pembrolizumab survival data
The new longer-term data on pembrolizumab come from the KEYNOTE-010 trial, which included more than 1,000 patients with advanced NSCLC who had previously undergone treatment with platinum-based chemotherapy. The patients were randomly assigned to receive either pembrolizumab or docetaxel for 2 years.
This is the latest update on data from this trial, which has been described as “really extraordinary.”
The 5-year overall survival rates were more than doubled in the pembrolizumab groups, compared with the docetaxel group, reported Roy Herbst, MD, PhD, department of medical oncology, Yale Comprehensive Cancer Center, New Haven, Conn.. He was presenting the new data at the recent World Conference on Lung Cancer 2020.
Overall results for patients with programmed death-ligand 1 (PD-L1) Tumor Proportion Score (TPS) expression greater than 1% show that 15.6% of the pembrolizumab group were still alive at 5 years versus 6.5% of the docetaxel group.
The results were even better among patients who had high PD-L1 TPS expression (>50%): in this subgroup, 25% of the patients who received pembrolizumab were still alive versus 8.2% of those who received docetaxel.
In addition, at 5 years, 9.4% of patients who received pembrolizumab were disease free versus 0.7% of the patients who received docetaxel, Dr. Herbst reported.
Dr. Hirsch commented that the 5-year survival rate of 25% among patients with high PD-L1 expression who underwent treatment with pembrolizumab is “great progress in lung cancer treatment, there is no doubt about it.”
He noted that the results also show that “numerically,” it matters whether patients have low PD-L1 expression. “We know from first-line studies that pembrolizumab monotherapy is effective in high PD-L1–expressing tumors, so these data fit very well,” he said.
At the meeting, Dr. Herbst summarized his presentation on pembrolizumab for patients with NSCLC who had previously undergone treatment, saying that, “with 5 years of follow-up, we continue to see a clinically meaningful improvement in overall survival and PFS [progression-free survival].
“Pembrolizumab monotherapy is a standard of care in patients with immunotherapy-naive or previously treated PD-L1–positive advanced non–small cell lung cancer,” Herbst stated.
Dr. Hirsch was largely in agreement. He believes that, for patients with a PD-L1 TPS of at least 50%, the standard of care “is practically pembrolizumab monotherapy, unless there are certain circumstances where you would add chemotherapy,” such as for patients with a high tumor volume, “where you want to see a very quick response.”
Dr. Hirsch pointed out, however, that currently most patients with high PD-L1–expressing tumors are given pembrolizumab in the first line, which begs the question as to what to give those who experience disease progression after immunotherapy.
“That is an open space,” he said. “There is a lot of studies going on in what we call the immunotherapy-refractory patients.
“We don’t have clear guidance for clinical practice yet,” he commented. He noted that there are several options: “Do you continue with chemotherapy? Do you continue with chemotherapy plus another immunotherapy? Do you switch to another immunotherapy?”
Commenting on Twitter, Stephen V. Liu, MD, director of thoracic oncology at Georgetown University, Washington, said the results were “very exciting.”
However, he wondered whether the results suggest that patients with high PD-L1 expression “may be able to stop” receiving pembrolizumab, whereas those with disease of lower expression “may need longer therapy.”
H. Jack West, MD, medical director of the thoracic oncology program, Swedish Cancer Institute, Seattle, said on Twitter that, to him, the “most impressive” aspect was the “new insight about patients stopping pembro after 2 years but still having two-thirds with sustained response.”
He added that he would “love to learn which patients can stop therapy and when, or whether we can do infrequent maintenance IO [immunotherapy].”
Nivolumab survival data
The data on nivolumab come from a pooled analysis of 5-year data on 854 patients from CheckMate 057 and CheckMate 017. The analysis was published in the Journal of Clinical Oncology on Jan. 15, 2021.
Both of these trials compared nivolumab with docetaxel for patients with NSCLC who had experienced disease progression with platinum-based chemotherapy.
The pooled analysis showed that the 5-year overall survival rate was more than fivefold greater with nivolumab than with docetaxel, at 13.4% versus 2.6%.
Moreover, more than 80% of patients who had not experienced progression with the immunotherapy at 2 years were still alive at 5 years. The percentage rose to more than 90% among those who had not experienced progression at 3 years.
Lead author Julie R. Brahmer, MD, Sidney Kimmel Comprehensive Cancer Center, Johns Hopkins University, Baltimore, and colleagues said the results “demonstrate that nivolumab can provide long-term survival benefit with durable responses and a tolerable safety profile in patients with previously treated, advanced NSCLC.
“Furthermore, some patients appear to maintain prolonged disease control even after stopping systemic therapy,” they noted.
Dr. Hirsch commented that, although the survival rates with nivolumab were slightly lower than reported with pembrolizumab in KEYNOTE-010, they could still be “within the range.” He added that “I wouldn’t conclude that pembrolizumab is better than nivolumab.”
Many factors may account for these differences, he suggested, including differences in the patient populations or simply differences in the numbers of patients included.
For him, the “main point” of the new data from both trials is that immunotherapy has shown “tremendous progress, compared to chemotherapy.”
KEYNOTE-010 was sponsored by Merck Sharp & Dohme. CheckMate 017 and CheckMate057 were sponsored by Bristol-Myers Squibb. Dr. Herbst has relationships with Jun Shi Pharmaceuticals, AstraZeneca, Genentech, Merck, Pfizer, AbbVie, Biodesix, Bristol-Myers Squibb, Eli Lilly, EMD Serono, Heat Biologics, Loxo, Nektar, NextCure, Novartis, Sanofi, Seattle Genetics, Shire, Spectrum Pharmaceuticals, Symphogen, Tesaro, Neon Therapeutics, Infinity Pharmaceuticals, Armo Biosciences, Genmab, Halozyme, and Tocagen. Dr. Brahmer has relationships with Roche/Genentech, Bristol-Myers Squibb, Lilly, Celgene, Syndax, Janssen Oncology, Merck, Amgen, Genentech, AstraZeneca, Incyte, Spectrum Pharmaceuticals, Revolution, and Roche/Genentech.
A version of this article first appeared on Medscape.com.
Longer-term survival with immunotherapy for patients with non–small cell lung cancer (NSCLC) is once again being applauded by experts in the field.
This time, the data come from trials that tested immunotherapy in the second-line setting for patients who had experienced disease progression with platinum-based chemotherapy. The latest 5-year follow-up from two landmark trials, one with pembrolizumab, the other with nivolumab, show that the survival benefit can persist for years after treatment is stopped.
“These are unprecedented data,” Fred R. Hirsch, MD, PhD, executive director of the Center for Thoracic Oncology at the Tisch Cancer Institute, New York, said in an interview. He was not involved in either trial and was approached for comment.
Pembrolizumab survival data
The new longer-term data on pembrolizumab come from the KEYNOTE-010 trial, which included more than 1,000 patients with advanced NSCLC who had previously undergone treatment with platinum-based chemotherapy. The patients were randomly assigned to receive either pembrolizumab or docetaxel for 2 years.
This is the latest update on data from this trial, which has been described as “really extraordinary.”
The 5-year overall survival rates were more than doubled in the pembrolizumab groups, compared with the docetaxel group, reported Roy Herbst, MD, PhD, department of medical oncology, Yale Comprehensive Cancer Center, New Haven, Conn.. He was presenting the new data at the recent World Conference on Lung Cancer 2020.
Overall results for patients with programmed death-ligand 1 (PD-L1) Tumor Proportion Score (TPS) expression greater than 1% show that 15.6% of the pembrolizumab group were still alive at 5 years versus 6.5% of the docetaxel group.
The results were even better among patients who had high PD-L1 TPS expression (>50%): in this subgroup, 25% of the patients who received pembrolizumab were still alive versus 8.2% of those who received docetaxel.
In addition, at 5 years, 9.4% of patients who received pembrolizumab were disease free versus 0.7% of the patients who received docetaxel, Dr. Herbst reported.
Dr. Hirsch commented that the 5-year survival rate of 25% among patients with high PD-L1 expression who underwent treatment with pembrolizumab is “great progress in lung cancer treatment, there is no doubt about it.”
He noted that the results also show that “numerically,” it matters whether patients have low PD-L1 expression. “We know from first-line studies that pembrolizumab monotherapy is effective in high PD-L1–expressing tumors, so these data fit very well,” he said.
At the meeting, Dr. Herbst summarized his presentation on pembrolizumab for patients with NSCLC who had previously undergone treatment, saying that, “with 5 years of follow-up, we continue to see a clinically meaningful improvement in overall survival and PFS [progression-free survival].
“Pembrolizumab monotherapy is a standard of care in patients with immunotherapy-naive or previously treated PD-L1–positive advanced non–small cell lung cancer,” Herbst stated.
Dr. Hirsch was largely in agreement. He believes that, for patients with a PD-L1 TPS of at least 50%, the standard of care “is practically pembrolizumab monotherapy, unless there are certain circumstances where you would add chemotherapy,” such as for patients with a high tumor volume, “where you want to see a very quick response.”
Dr. Hirsch pointed out, however, that currently most patients with high PD-L1–expressing tumors are given pembrolizumab in the first line, which begs the question as to what to give those who experience disease progression after immunotherapy.
“That is an open space,” he said. “There is a lot of studies going on in what we call the immunotherapy-refractory patients.
“We don’t have clear guidance for clinical practice yet,” he commented. He noted that there are several options: “Do you continue with chemotherapy? Do you continue with chemotherapy plus another immunotherapy? Do you switch to another immunotherapy?”
Commenting on Twitter, Stephen V. Liu, MD, director of thoracic oncology at Georgetown University, Washington, said the results were “very exciting.”
However, he wondered whether the results suggest that patients with high PD-L1 expression “may be able to stop” receiving pembrolizumab, whereas those with disease of lower expression “may need longer therapy.”
H. Jack West, MD, medical director of the thoracic oncology program, Swedish Cancer Institute, Seattle, said on Twitter that, to him, the “most impressive” aspect was the “new insight about patients stopping pembro after 2 years but still having two-thirds with sustained response.”
He added that he would “love to learn which patients can stop therapy and when, or whether we can do infrequent maintenance IO [immunotherapy].”
Nivolumab survival data
The data on nivolumab come from a pooled analysis of 5-year data on 854 patients from CheckMate 057 and CheckMate 017. The analysis was published in the Journal of Clinical Oncology on Jan. 15, 2021.
Both of these trials compared nivolumab with docetaxel for patients with NSCLC who had experienced disease progression with platinum-based chemotherapy.
The pooled analysis showed that the 5-year overall survival rate was more than fivefold greater with nivolumab than with docetaxel, at 13.4% versus 2.6%.
Moreover, more than 80% of patients who had not experienced progression with the immunotherapy at 2 years were still alive at 5 years. The percentage rose to more than 90% among those who had not experienced progression at 3 years.
Lead author Julie R. Brahmer, MD, Sidney Kimmel Comprehensive Cancer Center, Johns Hopkins University, Baltimore, and colleagues said the results “demonstrate that nivolumab can provide long-term survival benefit with durable responses and a tolerable safety profile in patients with previously treated, advanced NSCLC.
“Furthermore, some patients appear to maintain prolonged disease control even after stopping systemic therapy,” they noted.
Dr. Hirsch commented that, although the survival rates with nivolumab were slightly lower than reported with pembrolizumab in KEYNOTE-010, they could still be “within the range.” He added that “I wouldn’t conclude that pembrolizumab is better than nivolumab.”
Many factors may account for these differences, he suggested, including differences in the patient populations or simply differences in the numbers of patients included.
For him, the “main point” of the new data from both trials is that immunotherapy has shown “tremendous progress, compared to chemotherapy.”
KEYNOTE-010 was sponsored by Merck Sharp & Dohme. CheckMate 017 and CheckMate057 were sponsored by Bristol-Myers Squibb. Dr. Herbst has relationships with Jun Shi Pharmaceuticals, AstraZeneca, Genentech, Merck, Pfizer, AbbVie, Biodesix, Bristol-Myers Squibb, Eli Lilly, EMD Serono, Heat Biologics, Loxo, Nektar, NextCure, Novartis, Sanofi, Seattle Genetics, Shire, Spectrum Pharmaceuticals, Symphogen, Tesaro, Neon Therapeutics, Infinity Pharmaceuticals, Armo Biosciences, Genmab, Halozyme, and Tocagen. Dr. Brahmer has relationships with Roche/Genentech, Bristol-Myers Squibb, Lilly, Celgene, Syndax, Janssen Oncology, Merck, Amgen, Genentech, AstraZeneca, Incyte, Spectrum Pharmaceuticals, Revolution, and Roche/Genentech.
A version of this article first appeared on Medscape.com.
Longer-term survival with immunotherapy for patients with non–small cell lung cancer (NSCLC) is once again being applauded by experts in the field.
This time, the data come from trials that tested immunotherapy in the second-line setting for patients who had experienced disease progression with platinum-based chemotherapy. The latest 5-year follow-up from two landmark trials, one with pembrolizumab, the other with nivolumab, show that the survival benefit can persist for years after treatment is stopped.
“These are unprecedented data,” Fred R. Hirsch, MD, PhD, executive director of the Center for Thoracic Oncology at the Tisch Cancer Institute, New York, said in an interview. He was not involved in either trial and was approached for comment.
Pembrolizumab survival data
The new longer-term data on pembrolizumab come from the KEYNOTE-010 trial, which included more than 1,000 patients with advanced NSCLC who had previously undergone treatment with platinum-based chemotherapy. The patients were randomly assigned to receive either pembrolizumab or docetaxel for 2 years.
This is the latest update on data from this trial, which has been described as “really extraordinary.”
The 5-year overall survival rates were more than doubled in the pembrolizumab groups, compared with the docetaxel group, reported Roy Herbst, MD, PhD, department of medical oncology, Yale Comprehensive Cancer Center, New Haven, Conn.. He was presenting the new data at the recent World Conference on Lung Cancer 2020.
Overall results for patients with programmed death-ligand 1 (PD-L1) Tumor Proportion Score (TPS) expression greater than 1% show that 15.6% of the pembrolizumab group were still alive at 5 years versus 6.5% of the docetaxel group.
The results were even better among patients who had high PD-L1 TPS expression (>50%): in this subgroup, 25% of the patients who received pembrolizumab were still alive versus 8.2% of those who received docetaxel.
In addition, at 5 years, 9.4% of patients who received pembrolizumab were disease free versus 0.7% of the patients who received docetaxel, Dr. Herbst reported.
Dr. Hirsch commented that the 5-year survival rate of 25% among patients with high PD-L1 expression who underwent treatment with pembrolizumab is “great progress in lung cancer treatment, there is no doubt about it.”
He noted that the results also show that “numerically,” it matters whether patients have low PD-L1 expression. “We know from first-line studies that pembrolizumab monotherapy is effective in high PD-L1–expressing tumors, so these data fit very well,” he said.
At the meeting, Dr. Herbst summarized his presentation on pembrolizumab for patients with NSCLC who had previously undergone treatment, saying that, “with 5 years of follow-up, we continue to see a clinically meaningful improvement in overall survival and PFS [progression-free survival].
“Pembrolizumab monotherapy is a standard of care in patients with immunotherapy-naive or previously treated PD-L1–positive advanced non–small cell lung cancer,” Herbst stated.
Dr. Hirsch was largely in agreement. He believes that, for patients with a PD-L1 TPS of at least 50%, the standard of care “is practically pembrolizumab monotherapy, unless there are certain circumstances where you would add chemotherapy,” such as for patients with a high tumor volume, “where you want to see a very quick response.”
Dr. Hirsch pointed out, however, that currently most patients with high PD-L1–expressing tumors are given pembrolizumab in the first line, which begs the question as to what to give those who experience disease progression after immunotherapy.
“That is an open space,” he said. “There is a lot of studies going on in what we call the immunotherapy-refractory patients.
“We don’t have clear guidance for clinical practice yet,” he commented. He noted that there are several options: “Do you continue with chemotherapy? Do you continue with chemotherapy plus another immunotherapy? Do you switch to another immunotherapy?”
Commenting on Twitter, Stephen V. Liu, MD, director of thoracic oncology at Georgetown University, Washington, said the results were “very exciting.”
However, he wondered whether the results suggest that patients with high PD-L1 expression “may be able to stop” receiving pembrolizumab, whereas those with disease of lower expression “may need longer therapy.”
H. Jack West, MD, medical director of the thoracic oncology program, Swedish Cancer Institute, Seattle, said on Twitter that, to him, the “most impressive” aspect was the “new insight about patients stopping pembro after 2 years but still having two-thirds with sustained response.”
He added that he would “love to learn which patients can stop therapy and when, or whether we can do infrequent maintenance IO [immunotherapy].”
Nivolumab survival data
The data on nivolumab come from a pooled analysis of 5-year data on 854 patients from CheckMate 057 and CheckMate 017. The analysis was published in the Journal of Clinical Oncology on Jan. 15, 2021.
Both of these trials compared nivolumab with docetaxel for patients with NSCLC who had experienced disease progression with platinum-based chemotherapy.
The pooled analysis showed that the 5-year overall survival rate was more than fivefold greater with nivolumab than with docetaxel, at 13.4% versus 2.6%.
Moreover, more than 80% of patients who had not experienced progression with the immunotherapy at 2 years were still alive at 5 years. The percentage rose to more than 90% among those who had not experienced progression at 3 years.
Lead author Julie R. Brahmer, MD, Sidney Kimmel Comprehensive Cancer Center, Johns Hopkins University, Baltimore, and colleagues said the results “demonstrate that nivolumab can provide long-term survival benefit with durable responses and a tolerable safety profile in patients with previously treated, advanced NSCLC.
“Furthermore, some patients appear to maintain prolonged disease control even after stopping systemic therapy,” they noted.
Dr. Hirsch commented that, although the survival rates with nivolumab were slightly lower than reported with pembrolizumab in KEYNOTE-010, they could still be “within the range.” He added that “I wouldn’t conclude that pembrolizumab is better than nivolumab.”
Many factors may account for these differences, he suggested, including differences in the patient populations or simply differences in the numbers of patients included.
For him, the “main point” of the new data from both trials is that immunotherapy has shown “tremendous progress, compared to chemotherapy.”
KEYNOTE-010 was sponsored by Merck Sharp & Dohme. CheckMate 017 and CheckMate057 were sponsored by Bristol-Myers Squibb. Dr. Herbst has relationships with Jun Shi Pharmaceuticals, AstraZeneca, Genentech, Merck, Pfizer, AbbVie, Biodesix, Bristol-Myers Squibb, Eli Lilly, EMD Serono, Heat Biologics, Loxo, Nektar, NextCure, Novartis, Sanofi, Seattle Genetics, Shire, Spectrum Pharmaceuticals, Symphogen, Tesaro, Neon Therapeutics, Infinity Pharmaceuticals, Armo Biosciences, Genmab, Halozyme, and Tocagen. Dr. Brahmer has relationships with Roche/Genentech, Bristol-Myers Squibb, Lilly, Celgene, Syndax, Janssen Oncology, Merck, Amgen, Genentech, AstraZeneca, Incyte, Spectrum Pharmaceuticals, Revolution, and Roche/Genentech.
A version of this article first appeared on Medscape.com.
Vedolizumab looks safer than anti-TNF drugs in older adults with IBD
A large analysis of Medicare data from all 50 states suggests that vedolizumab may be just as effective as anti–tumor necrosis factor (anti-TNF) agents in controlling inflammatory bowel disease (IBD) in patients aged over 65 years, with fewer infectious disease hospitalizations.
The study was prompted by the fact that older adults are greatly underrepresented in clinical trials of approved IBD medications. There is a second peak in IBD diagnosis among people in their 50s and 60s, and IBD patients are living longer with more effective medications. So although a significant number of IBD patients are aged 65 years or older, that group encompasses less than 1% of adults in clinical trials, Bharati Kochar, MD, reported at the annual congress of the Crohn’s & Colitis Foundation and the American Gastroenterological Association.
“Therefore, we don’t know how well these medications work and how safe they are specifically in older adults,” said Dr. Kochar, a gastroenterologist at Massachusetts General Hospital and assistant professor of medicine at Harvard Medical School, both in Boston.
The data largely support what had been known mechanistically about vedolizumab. “It suggests that both drugs work well enough to prevent [IBD-related] hospitalizations, but clearly there was a benefit toward the safer medication, Entyvio [vedolizumab], in the infection-related hospitalizations. That’s not the only readout in infections, but it is an important readout because infections that get hospitalized are the ones that predict mortality and disability,” said Matthew Ciorba, MD, who attended the session. Dr. Ciorba is director of the IBD Center at Washington University in St. Louis and was not involved in the study.
“I think this study is reassuring to clinicians. It provides important clinical data that support what we know about the mechanisms of vedolizumab. The safety data we predicted is borne out in this large and well-done study,” said Dr. Ciorba.
The researchers collected a 20% random sample from a 50-state Medicare claims database, including patients who were aged 65 years or older, who had two or more codes for Crohn’s disease or ulcerative colitis, and had 18 months of continuous enrollment. It excluded Medicare Part C patients; those who used ustekinumab, natalizumab, cyclosporine, or tacrolimus during the look back and study period; and those with two or more codes for rheumatoid arthritis, plaque psoriasis, psoriatic arthritis, or ankylosing spondylitis during the study period.
Among those included, 480 patients were on vedolizumab, while 1,152 were on anti-TNF medications. The two groups were broadly similar in their characteristics: Twenty-nine percent of both groups took budesonide, although the anti-TNF group had a higher frequency use of systemic corticosteroids (68% vs. 57%), 5-ASA drugs (62% vs. 42%), and immunomodulators (32% vs. 28%).
There were no significant differences between the two groups with respect to frequency of IBD-related hospitalizations, IBD-related surgery, steroid prescription rate after induction, or all-cause hospitalization. However, infection-related hospitalizations were less frequent in the vedolizumab group (crude incidence, 0.03 vs. 0.05 per person-year; adjusted hazard ratio, 0.47; 95% confidence interval, 0.25-0.86).
“I think it’s important to use your clinical judgment to treat the patient in front of you, and these data should simply help contextualize risk for older IBD patients newly initiating vedolizumab and anti-TNF agents,” said Dr. Kochar. However, recognizing the limitations of any retrospective study based on administrative data, she called for additional research. “There is a vast need for additional large and robust comparative effectiveness and safety studies in older adults of the rapidly proliferating arsenal of IBD medications,” Dr. Kochar concluded.
Dr. Kochar and Dr. Ciorba have no relevant financial disclosures.
A large analysis of Medicare data from all 50 states suggests that vedolizumab may be just as effective as anti–tumor necrosis factor (anti-TNF) agents in controlling inflammatory bowel disease (IBD) in patients aged over 65 years, with fewer infectious disease hospitalizations.
The study was prompted by the fact that older adults are greatly underrepresented in clinical trials of approved IBD medications. There is a second peak in IBD diagnosis among people in their 50s and 60s, and IBD patients are living longer with more effective medications. So although a significant number of IBD patients are aged 65 years or older, that group encompasses less than 1% of adults in clinical trials, Bharati Kochar, MD, reported at the annual congress of the Crohn’s & Colitis Foundation and the American Gastroenterological Association.
“Therefore, we don’t know how well these medications work and how safe they are specifically in older adults,” said Dr. Kochar, a gastroenterologist at Massachusetts General Hospital and assistant professor of medicine at Harvard Medical School, both in Boston.
The data largely support what had been known mechanistically about vedolizumab. “It suggests that both drugs work well enough to prevent [IBD-related] hospitalizations, but clearly there was a benefit toward the safer medication, Entyvio [vedolizumab], in the infection-related hospitalizations. That’s not the only readout in infections, but it is an important readout because infections that get hospitalized are the ones that predict mortality and disability,” said Matthew Ciorba, MD, who attended the session. Dr. Ciorba is director of the IBD Center at Washington University in St. Louis and was not involved in the study.
“I think this study is reassuring to clinicians. It provides important clinical data that support what we know about the mechanisms of vedolizumab. The safety data we predicted is borne out in this large and well-done study,” said Dr. Ciorba.
The researchers collected a 20% random sample from a 50-state Medicare claims database, including patients who were aged 65 years or older, who had two or more codes for Crohn’s disease or ulcerative colitis, and had 18 months of continuous enrollment. It excluded Medicare Part C patients; those who used ustekinumab, natalizumab, cyclosporine, or tacrolimus during the look back and study period; and those with two or more codes for rheumatoid arthritis, plaque psoriasis, psoriatic arthritis, or ankylosing spondylitis during the study period.
Among those included, 480 patients were on vedolizumab, while 1,152 were on anti-TNF medications. The two groups were broadly similar in their characteristics: Twenty-nine percent of both groups took budesonide, although the anti-TNF group had a higher frequency use of systemic corticosteroids (68% vs. 57%), 5-ASA drugs (62% vs. 42%), and immunomodulators (32% vs. 28%).
There were no significant differences between the two groups with respect to frequency of IBD-related hospitalizations, IBD-related surgery, steroid prescription rate after induction, or all-cause hospitalization. However, infection-related hospitalizations were less frequent in the vedolizumab group (crude incidence, 0.03 vs. 0.05 per person-year; adjusted hazard ratio, 0.47; 95% confidence interval, 0.25-0.86).
“I think it’s important to use your clinical judgment to treat the patient in front of you, and these data should simply help contextualize risk for older IBD patients newly initiating vedolizumab and anti-TNF agents,” said Dr. Kochar. However, recognizing the limitations of any retrospective study based on administrative data, she called for additional research. “There is a vast need for additional large and robust comparative effectiveness and safety studies in older adults of the rapidly proliferating arsenal of IBD medications,” Dr. Kochar concluded.
Dr. Kochar and Dr. Ciorba have no relevant financial disclosures.
A large analysis of Medicare data from all 50 states suggests that vedolizumab may be just as effective as anti–tumor necrosis factor (anti-TNF) agents in controlling inflammatory bowel disease (IBD) in patients aged over 65 years, with fewer infectious disease hospitalizations.
The study was prompted by the fact that older adults are greatly underrepresented in clinical trials of approved IBD medications. There is a second peak in IBD diagnosis among people in their 50s and 60s, and IBD patients are living longer with more effective medications. So although a significant number of IBD patients are aged 65 years or older, that group encompasses less than 1% of adults in clinical trials, Bharati Kochar, MD, reported at the annual congress of the Crohn’s & Colitis Foundation and the American Gastroenterological Association.
“Therefore, we don’t know how well these medications work and how safe they are specifically in older adults,” said Dr. Kochar, a gastroenterologist at Massachusetts General Hospital and assistant professor of medicine at Harvard Medical School, both in Boston.
The data largely support what had been known mechanistically about vedolizumab. “It suggests that both drugs work well enough to prevent [IBD-related] hospitalizations, but clearly there was a benefit toward the safer medication, Entyvio [vedolizumab], in the infection-related hospitalizations. That’s not the only readout in infections, but it is an important readout because infections that get hospitalized are the ones that predict mortality and disability,” said Matthew Ciorba, MD, who attended the session. Dr. Ciorba is director of the IBD Center at Washington University in St. Louis and was not involved in the study.
“I think this study is reassuring to clinicians. It provides important clinical data that support what we know about the mechanisms of vedolizumab. The safety data we predicted is borne out in this large and well-done study,” said Dr. Ciorba.
The researchers collected a 20% random sample from a 50-state Medicare claims database, including patients who were aged 65 years or older, who had two or more codes for Crohn’s disease or ulcerative colitis, and had 18 months of continuous enrollment. It excluded Medicare Part C patients; those who used ustekinumab, natalizumab, cyclosporine, or tacrolimus during the look back and study period; and those with two or more codes for rheumatoid arthritis, plaque psoriasis, psoriatic arthritis, or ankylosing spondylitis during the study period.
Among those included, 480 patients were on vedolizumab, while 1,152 were on anti-TNF medications. The two groups were broadly similar in their characteristics: Twenty-nine percent of both groups took budesonide, although the anti-TNF group had a higher frequency use of systemic corticosteroids (68% vs. 57%), 5-ASA drugs (62% vs. 42%), and immunomodulators (32% vs. 28%).
There were no significant differences between the two groups with respect to frequency of IBD-related hospitalizations, IBD-related surgery, steroid prescription rate after induction, or all-cause hospitalization. However, infection-related hospitalizations were less frequent in the vedolizumab group (crude incidence, 0.03 vs. 0.05 per person-year; adjusted hazard ratio, 0.47; 95% confidence interval, 0.25-0.86).
“I think it’s important to use your clinical judgment to treat the patient in front of you, and these data should simply help contextualize risk for older IBD patients newly initiating vedolizumab and anti-TNF agents,” said Dr. Kochar. However, recognizing the limitations of any retrospective study based on administrative data, she called for additional research. “There is a vast need for additional large and robust comparative effectiveness and safety studies in older adults of the rapidly proliferating arsenal of IBD medications,” Dr. Kochar concluded.
Dr. Kochar and Dr. Ciorba have no relevant financial disclosures.
FROM THE CROHN’S & COLITIS CONGRESS
Prostate drugs tied to lower risk for Parkinson’s disease
new research suggests. Treatment of BPH with terazosin (Hytrin), doxazosin (Cardura), or alfuzosin (Uroxatral), all of which enhance glycolysis, was associated with a lower risk of developing Parkinson’s disease than patients taking a drug used for the same indication, tamsulosin (Flomax), which does not affect glycolysis.
“If giving someone terazosin or similar medications truly reduces their risk of disease, these results could have significant clinical implications for neurologists,” said lead author Jacob E. Simmering, PhD, assistant professor of internal medicine at the University of Iowa, Iowa City.
There are few reliable neuroprotective treatments for Parkinson’s disease, he said. “We can manage some of the symptoms, but we can’t stop it from progressing. If a randomized trial finds the same result, this will provide a new option to slow progression of Parkinson’s disease.”
The pathogenesis of Parkinson’s disease is heterogeneous, however, and not all patients may benefit from glycolysis-enhancing drugs, the investigators noted. Future research will be needed to identify potential candidates for this treatment, and clarify the effects of these drugs, they wrote.
The findings were published online Feb. 1, 2021, in JAMA Neurology.
Time-dependent effects
The major risk factor for Parkinson’s disease is age, which is associated with impaired energy metabolism. Glycolysis is decreased among patients with Parkinson’s disease, yet impaired energy metabolism has not been investigated widely as a pathogenic factor in the disease, the authors wrote.
Studies have indicated that terazosin increases the activity of an enzyme important in glycolysis. Doxazosin and alfuzosin have a similar mechanism of action and enhance energy metabolism. Tamsulosin, a structurally unrelated drug, has the same mechanism of action as the other three drugs, but does not enhance energy metabolism.
In this report, the researchers investigated the hypothesis that patients who received therapy with terazosin, doxazosin, or alfuzosin would have a lower risk of developing Parkinson’s disease than patients receiving tamsulosin. To do that, they used health care utilization data from Denmark and the United States, including the Danish National Prescription Registry, the Danish National Patient Registry, the Danish Civil Registration System, and the Truven Health Analytics MarketScan database.
The investigators searched the records for patients who filled prescriptions for any of the four drugs of interest. They excluded any patients who developed Parkinson’s disease within 1 year of starting medication. Because use of these drugs is rare among women, they included only men in their analysis.
They looked at patient outcomes beginning at 1 year after the initiation of treatment. They also required patients to fill at least two prescriptions before the beginning of follow-up. Patients who switched from tamsulosin to any of the other drugs, or vice versa, were excluded from analysis.
The investigators used propensity-score matching to ensure that patients in the tamsulosin and terazosin/doxazosin/alfuzosin groups were similar in terms of their other potential risk factors. The primary outcome was the development of Parkinson’s disease.
They identified 52,365 propensity score–matched pairs in the Danish registries and 94,883 pairs in the Truven database. The mean age was 67.9 years in the Danish registries and 63.8 years in the Truven database, and follow-up was approximately 5 years and 3 years respectively. Baseline covariates were well balanced between cohorts.
Among Danish patients, those who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease versus those who took tamsulosin (hazard ratio, 0.88). Similarly, patients in the Truven database who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease than those who took tamsulosin (HR, 0.63).
In both cohorts, the risk for Parkinson’s disease among patients receiving terazosin, doxazosin, or alfuzosin, compared with those receiving tamsulosin, decreased with increasing numbers of prescriptions filled. Long-term treatment with any of the three glycolysis-enhancing drugs was associated with greater risk reduction in the Danish (HR, 0.79) and Truven (HR, 0.46) cohorts versus tamsulosin.
Differences in case definitions, which may reflect how Parkinson’s disease was managed, complicate comparisons between the Danish and Truven cohorts, said Dr. Simmering. Another challenge is the source of the data. “The Truven data set was derived from insurance claims from people with private insurance or Medicare supplemental plans,” he said. “This group is quite large but may not be representative of everyone in the United States. We would also only be able to follow people while they were on one insurance plan. If they switched coverage to a company that doesn’t contribute data, we would lose them.”
The Danish database, however, includes all residents of Denmark. Only people who left the country were lost to follow-up.
The results support the hypothesis that increasing energy in cells slows disease progression, Dr. Simmering added. “There are a few conditions, mostly REM sleep disorders, that are associated with future diagnosis of Parkinson’s disease. Right now, we don’t have anything to offer people at elevated risk of Parkinson’s disease that might prevent the disease. If a controlled trial finds that terazosin slows or prevents Parkinson’s disease, we would have something truly protective to offer these patients.”
Biomarker needed
Commenting on the results, Alberto J. Espay, MD, MSc, professor of neurology at the University of Cincinnati Academic Health Center, was cautious. “These findings are of unclear applicability to any particular patient without a biomarker for a deficit of glycolysis that these drugs are presumed to affect,” Dr. Espay said. “Hence, there is no feasible or warranted change in practice as a result of this study.”
Pathogenic mechanisms are heterogeneous among patients with Parkinson’s disease, Dr. Espay added. “We will need to understand who among the large biological universe of Parkinson’s patients may have impaired energy metabolism as a pathogenic mechanism to be selected for a future clinical trial evaluating terazosin, doxazosin, or alfuzosin as a potential disease-modifying intervention.”
Parkinson’s disease is not one disease, but a group of disorders with unique biological abnormalities, said Dr. Espay. “We know so much about ‘Parkinson’s disease’ and next to nothing about the biology of individuals with Parkinson’s disease.”
This situation has enabled the development of symptomatic treatments, such as dopaminergic therapies, but failed to yield disease-modifying treatments, he said.
The University of Iowa contributed funds for this study. Dr. Simmering has received pilot funding from the University of Iowa Institute for Clinical and Translational Science. He had no conflicts of interest to disclose. Dr. Espay disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests. Treatment of BPH with terazosin (Hytrin), doxazosin (Cardura), or alfuzosin (Uroxatral), all of which enhance glycolysis, was associated with a lower risk of developing Parkinson’s disease than patients taking a drug used for the same indication, tamsulosin (Flomax), which does not affect glycolysis.
“If giving someone terazosin or similar medications truly reduces their risk of disease, these results could have significant clinical implications for neurologists,” said lead author Jacob E. Simmering, PhD, assistant professor of internal medicine at the University of Iowa, Iowa City.
There are few reliable neuroprotective treatments for Parkinson’s disease, he said. “We can manage some of the symptoms, but we can’t stop it from progressing. If a randomized trial finds the same result, this will provide a new option to slow progression of Parkinson’s disease.”
The pathogenesis of Parkinson’s disease is heterogeneous, however, and not all patients may benefit from glycolysis-enhancing drugs, the investigators noted. Future research will be needed to identify potential candidates for this treatment, and clarify the effects of these drugs, they wrote.
The findings were published online Feb. 1, 2021, in JAMA Neurology.
Time-dependent effects
The major risk factor for Parkinson’s disease is age, which is associated with impaired energy metabolism. Glycolysis is decreased among patients with Parkinson’s disease, yet impaired energy metabolism has not been investigated widely as a pathogenic factor in the disease, the authors wrote.
Studies have indicated that terazosin increases the activity of an enzyme important in glycolysis. Doxazosin and alfuzosin have a similar mechanism of action and enhance energy metabolism. Tamsulosin, a structurally unrelated drug, has the same mechanism of action as the other three drugs, but does not enhance energy metabolism.
In this report, the researchers investigated the hypothesis that patients who received therapy with terazosin, doxazosin, or alfuzosin would have a lower risk of developing Parkinson’s disease than patients receiving tamsulosin. To do that, they used health care utilization data from Denmark and the United States, including the Danish National Prescription Registry, the Danish National Patient Registry, the Danish Civil Registration System, and the Truven Health Analytics MarketScan database.
The investigators searched the records for patients who filled prescriptions for any of the four drugs of interest. They excluded any patients who developed Parkinson’s disease within 1 year of starting medication. Because use of these drugs is rare among women, they included only men in their analysis.
They looked at patient outcomes beginning at 1 year after the initiation of treatment. They also required patients to fill at least two prescriptions before the beginning of follow-up. Patients who switched from tamsulosin to any of the other drugs, or vice versa, were excluded from analysis.
The investigators used propensity-score matching to ensure that patients in the tamsulosin and terazosin/doxazosin/alfuzosin groups were similar in terms of their other potential risk factors. The primary outcome was the development of Parkinson’s disease.
They identified 52,365 propensity score–matched pairs in the Danish registries and 94,883 pairs in the Truven database. The mean age was 67.9 years in the Danish registries and 63.8 years in the Truven database, and follow-up was approximately 5 years and 3 years respectively. Baseline covariates were well balanced between cohorts.
Among Danish patients, those who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease versus those who took tamsulosin (hazard ratio, 0.88). Similarly, patients in the Truven database who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease than those who took tamsulosin (HR, 0.63).
In both cohorts, the risk for Parkinson’s disease among patients receiving terazosin, doxazosin, or alfuzosin, compared with those receiving tamsulosin, decreased with increasing numbers of prescriptions filled. Long-term treatment with any of the three glycolysis-enhancing drugs was associated with greater risk reduction in the Danish (HR, 0.79) and Truven (HR, 0.46) cohorts versus tamsulosin.
Differences in case definitions, which may reflect how Parkinson’s disease was managed, complicate comparisons between the Danish and Truven cohorts, said Dr. Simmering. Another challenge is the source of the data. “The Truven data set was derived from insurance claims from people with private insurance or Medicare supplemental plans,” he said. “This group is quite large but may not be representative of everyone in the United States. We would also only be able to follow people while they were on one insurance plan. If they switched coverage to a company that doesn’t contribute data, we would lose them.”
The Danish database, however, includes all residents of Denmark. Only people who left the country were lost to follow-up.
The results support the hypothesis that increasing energy in cells slows disease progression, Dr. Simmering added. “There are a few conditions, mostly REM sleep disorders, that are associated with future diagnosis of Parkinson’s disease. Right now, we don’t have anything to offer people at elevated risk of Parkinson’s disease that might prevent the disease. If a controlled trial finds that terazosin slows or prevents Parkinson’s disease, we would have something truly protective to offer these patients.”
Biomarker needed
Commenting on the results, Alberto J. Espay, MD, MSc, professor of neurology at the University of Cincinnati Academic Health Center, was cautious. “These findings are of unclear applicability to any particular patient without a biomarker for a deficit of glycolysis that these drugs are presumed to affect,” Dr. Espay said. “Hence, there is no feasible or warranted change in practice as a result of this study.”
Pathogenic mechanisms are heterogeneous among patients with Parkinson’s disease, Dr. Espay added. “We will need to understand who among the large biological universe of Parkinson’s patients may have impaired energy metabolism as a pathogenic mechanism to be selected for a future clinical trial evaluating terazosin, doxazosin, or alfuzosin as a potential disease-modifying intervention.”
Parkinson’s disease is not one disease, but a group of disorders with unique biological abnormalities, said Dr. Espay. “We know so much about ‘Parkinson’s disease’ and next to nothing about the biology of individuals with Parkinson’s disease.”
This situation has enabled the development of symptomatic treatments, such as dopaminergic therapies, but failed to yield disease-modifying treatments, he said.
The University of Iowa contributed funds for this study. Dr. Simmering has received pilot funding from the University of Iowa Institute for Clinical and Translational Science. He had no conflicts of interest to disclose. Dr. Espay disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests. Treatment of BPH with terazosin (Hytrin), doxazosin (Cardura), or alfuzosin (Uroxatral), all of which enhance glycolysis, was associated with a lower risk of developing Parkinson’s disease than patients taking a drug used for the same indication, tamsulosin (Flomax), which does not affect glycolysis.
“If giving someone terazosin or similar medications truly reduces their risk of disease, these results could have significant clinical implications for neurologists,” said lead author Jacob E. Simmering, PhD, assistant professor of internal medicine at the University of Iowa, Iowa City.
There are few reliable neuroprotective treatments for Parkinson’s disease, he said. “We can manage some of the symptoms, but we can’t stop it from progressing. If a randomized trial finds the same result, this will provide a new option to slow progression of Parkinson’s disease.”
The pathogenesis of Parkinson’s disease is heterogeneous, however, and not all patients may benefit from glycolysis-enhancing drugs, the investigators noted. Future research will be needed to identify potential candidates for this treatment, and clarify the effects of these drugs, they wrote.
The findings were published online Feb. 1, 2021, in JAMA Neurology.
Time-dependent effects
The major risk factor for Parkinson’s disease is age, which is associated with impaired energy metabolism. Glycolysis is decreased among patients with Parkinson’s disease, yet impaired energy metabolism has not been investigated widely as a pathogenic factor in the disease, the authors wrote.
Studies have indicated that terazosin increases the activity of an enzyme important in glycolysis. Doxazosin and alfuzosin have a similar mechanism of action and enhance energy metabolism. Tamsulosin, a structurally unrelated drug, has the same mechanism of action as the other three drugs, but does not enhance energy metabolism.
In this report, the researchers investigated the hypothesis that patients who received therapy with terazosin, doxazosin, or alfuzosin would have a lower risk of developing Parkinson’s disease than patients receiving tamsulosin. To do that, they used health care utilization data from Denmark and the United States, including the Danish National Prescription Registry, the Danish National Patient Registry, the Danish Civil Registration System, and the Truven Health Analytics MarketScan database.
The investigators searched the records for patients who filled prescriptions for any of the four drugs of interest. They excluded any patients who developed Parkinson’s disease within 1 year of starting medication. Because use of these drugs is rare among women, they included only men in their analysis.
They looked at patient outcomes beginning at 1 year after the initiation of treatment. They also required patients to fill at least two prescriptions before the beginning of follow-up. Patients who switched from tamsulosin to any of the other drugs, or vice versa, were excluded from analysis.
The investigators used propensity-score matching to ensure that patients in the tamsulosin and terazosin/doxazosin/alfuzosin groups were similar in terms of their other potential risk factors. The primary outcome was the development of Parkinson’s disease.
They identified 52,365 propensity score–matched pairs in the Danish registries and 94,883 pairs in the Truven database. The mean age was 67.9 years in the Danish registries and 63.8 years in the Truven database, and follow-up was approximately 5 years and 3 years respectively. Baseline covariates were well balanced between cohorts.
Among Danish patients, those who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease versus those who took tamsulosin (hazard ratio, 0.88). Similarly, patients in the Truven database who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease than those who took tamsulosin (HR, 0.63).
In both cohorts, the risk for Parkinson’s disease among patients receiving terazosin, doxazosin, or alfuzosin, compared with those receiving tamsulosin, decreased with increasing numbers of prescriptions filled. Long-term treatment with any of the three glycolysis-enhancing drugs was associated with greater risk reduction in the Danish (HR, 0.79) and Truven (HR, 0.46) cohorts versus tamsulosin.
Differences in case definitions, which may reflect how Parkinson’s disease was managed, complicate comparisons between the Danish and Truven cohorts, said Dr. Simmering. Another challenge is the source of the data. “The Truven data set was derived from insurance claims from people with private insurance or Medicare supplemental plans,” he said. “This group is quite large but may not be representative of everyone in the United States. We would also only be able to follow people while they were on one insurance plan. If they switched coverage to a company that doesn’t contribute data, we would lose them.”
The Danish database, however, includes all residents of Denmark. Only people who left the country were lost to follow-up.
The results support the hypothesis that increasing energy in cells slows disease progression, Dr. Simmering added. “There are a few conditions, mostly REM sleep disorders, that are associated with future diagnosis of Parkinson’s disease. Right now, we don’t have anything to offer people at elevated risk of Parkinson’s disease that might prevent the disease. If a controlled trial finds that terazosin slows or prevents Parkinson’s disease, we would have something truly protective to offer these patients.”
Biomarker needed
Commenting on the results, Alberto J. Espay, MD, MSc, professor of neurology at the University of Cincinnati Academic Health Center, was cautious. “These findings are of unclear applicability to any particular patient without a biomarker for a deficit of glycolysis that these drugs are presumed to affect,” Dr. Espay said. “Hence, there is no feasible or warranted change in practice as a result of this study.”
Pathogenic mechanisms are heterogeneous among patients with Parkinson’s disease, Dr. Espay added. “We will need to understand who among the large biological universe of Parkinson’s patients may have impaired energy metabolism as a pathogenic mechanism to be selected for a future clinical trial evaluating terazosin, doxazosin, or alfuzosin as a potential disease-modifying intervention.”
Parkinson’s disease is not one disease, but a group of disorders with unique biological abnormalities, said Dr. Espay. “We know so much about ‘Parkinson’s disease’ and next to nothing about the biology of individuals with Parkinson’s disease.”
This situation has enabled the development of symptomatic treatments, such as dopaminergic therapies, but failed to yield disease-modifying treatments, he said.
The University of Iowa contributed funds for this study. Dr. Simmering has received pilot funding from the University of Iowa Institute for Clinical and Translational Science. He had no conflicts of interest to disclose. Dr. Espay disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
ColCORONA: More questions than answers for colchicine in COVID-19
Science by press release and preprint has cooled clinician enthusiasm for the use of colchicine in nonhospitalized patients with COVID-19, despite a pressing need for early treatments.
As previously reported by this news organization, a Jan. 22 press release announced that the massive ColCORONA study missed its primary endpoint of hospitalization or death among 4,488 newly diagnosed patients at increased risk for hospitalization.
But it also touted that use of the anti-inflammatory drug significantly reduced the primary endpoint in 4,159 of those patients with polymerase chain reaction–confirmed COVID and led to reductions of 25%, 50%, and 44%, respectively, for hospitalizations, ventilations, and death.
Lead investigator Jean-Claude Tardif, MD, director of the Montreal Heart Institute Research Centre, deemed the findings a “medical breakthrough.”
When the preprint released a few days later, however, newly revealed confidence intervals showed colchicine did not meaningfully reduce the need for mechanical ventilation (odds ratio, 0.50; 95% confidence interval, 0.23-1.07) or death alone (OR, 0.56; 95% CI, 0.19-1.66).
Further, the significant benefit on the primary outcome came at the cost of a fivefold increase in pulmonary embolism (11 vs. 2; P = .01), which was not mentioned in the press release.
“Whether this represents a real phenomenon or simply the play of chance is not known,” Dr. Tardif and colleagues noted later in the preprint.
“I read the preprint on colchicine and I have so many questions,” Aaron E. Glatt, MD, spokesperson for the Infectious Diseases Society of America and chief of infectious diseases, Mount Sinai South Nassau, Hewlett, N.Y., said in an interview. “I’ve been burned too many times with COVID and prefer to see better data.
“People sometimes say if you wait for perfect data, people are going to die,” he said. “Yeah, but we have no idea if people are going to die from getting this drug more than not getting it. That’s what concerns me. How many pulmonary emboli are going to be fatal versus the slight benefit that the study showed?”
The pushback to the non–peer-reviewed data on social media and via emails was so strong that Dr. Tardif posted a nearly 2,000-word letter responding to the many questions at play.
Chief among them was why the trial, originally planned for 6,000 patients, was stopped early by the investigators without consultation with the data safety monitoring board (DSMB).
The explanation in the letter that logistical issues like running the study call center, budget constraints, and a perceived need to quickly communicate the results left some calling foul that the study wasn’t allowed to finish and come to a more definitive conclusion.
“I can be a little bit sympathetic to their cause but at the same time the DSMB should have said no,” said David Boulware, MD, MPH, who led a recent hydroxychloroquine trial in COVID-19. “The problem is we’re sort of left in limbo, where some people kind of believe it and some say it’s not really a thing. So it’s not really moving the needle, as far as guidelines go.”
Indeed, a Twitter poll by cardiologist James Januzzi Jr., MD, captured the uncertainty, with 28% of respondents saying the trial was “neutral,” 58% saying “maybe but meh,” and 14% saying “colchicine for all.”
Another poll cheekily asked whether ColCORONA was the Gamestop/Reddit equivalent of COVID.
“The press release really didn’t help things because it very much oversold the effect. That, I think, poisoned the well,” said Dr. Boulware, professor of medicine in infectious diseases at the University of Minnesota, Minneapolis.
“The question I’m left with is not whether colchicine works, but who does it work in,” he said. “That’s really the fundamental question because it does seem that there are probably high-risk groups in their trial and others where they benefit, whereas other groups don’t benefit. In the subgroup analysis, there was absolutely no beneficial effect in women.”
According to the authors, the number needed to treat to prevent one death or hospitalization was 71 overall, but 29 for patients with diabetes, 31 for those aged 70 years and older, 53 for patients with respiratory disease, and 25 for those with coronary disease or heart failure.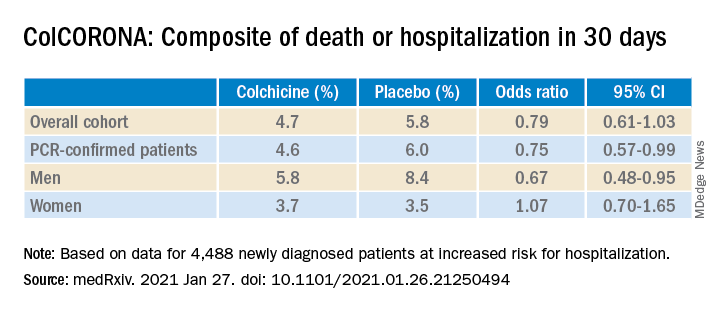
Men are at higher risk overall for poor outcomes. But “the authors didn’t present a multivariable analysis, so it is unclear if another factor, such as a differential prevalence of smoking or cardiovascular risk factors, contributed to the differential benefit,” Rachel Bender Ignacio, MD, MPH, infectious disease specialist, University of Washington, Seattle, said in an interview.
Importantly, in this pragmatic study, duration and severity of symptoms were not reported, observed Dr. Bender Ignacio, who is also a STOP-COVID-2 investigator. “We don’t yet have data as to whether colchicine shortens duration or severity of symptoms or prevents long COVID, so we need more data on that.”
The overall risk for serious adverse events was lower in the colchicine group, but the difference in pulmonary embolism (PE) was striking, she said. This could be caused by a real biologic effect, or it’s possible that persons with shortness of breath and hypoxia, without evident viral pneumonia on chest x-ray after a positive COVID-19 test, were more likely to receive a CT-PE study.
The press release also failed to include information, later noted in the preprint, that the MHI has submitted two patents related to colchicine: “Methods of treating a coronavirus infection using colchicine” and “Early administration of low-dose colchicine after myocardial infarction.”
Reached for clarification, MHI communications adviser Camille Turbide said in an interview that the first patent “simply refers to the novel concept of preventing complications of COVID-19, such as admission to the hospital, with colchicine as tested in the ColCORONA study.”
The second patent, she said, refers to the “novel concept that administering colchicine early after a major adverse cardiovascular event is better than waiting several days,” as supported by the COLCOT study, which Dr. Tardif also led.
The patents are being reviewed by authorities and “Dr. Tardif has waived his rights in these patents and does not stand to benefit financially at all if colchicine becomes used as a treatment for COVID-19,” Ms. Turbide said.
Dr. Tardif did not respond to interview requests for this story. Dr. Glatt said conflicts of interest must be assessed and are “something that is of great concern in any scientific study.”
Cardiologist Steve Nissen, MD, of the Cleveland Clinic said in an interview that, “despite the negative results, the study does suggest that colchicine might have a benefit and should be studied in future trials. These findings are not sufficient evidence to suggest use of the drug in patients infected with COVID-19.”
He noted that adverse effects like diarrhea were expected but that the excess PE was unexpected and needs greater clarification.
“Stopping the trial for administrative reasons is puzzling and undermined the ability of the trial to give a reliable answer,” Dr. Nissen said. “This is a reasonable pilot study that should be viewed as hypothesis generating but inconclusive.”
Several sources said a new trial is unlikely, particularly given the cost and 28 trials already evaluating colchicine. Among these are RECOVERY and COLCOVID, testing whether colchicine can reduce the duration of hospitalization or death in hospitalized patients with COVID-19.
Because there are so many trials ongoing right now, including for antivirals and other immunomodulators, it’s important that, if colchicine comes to routine clinical use, it provides access to treatment for those not able or willing to access clinical trials, rather than impeding clinical trial enrollment, Dr. Bender Ignacio suggested.
“We have already learned the lesson in the pandemic that early adoption of potentially promising therapies can negatively impact our ability to study and develop other promising treatments,” she said.
The trial was coordinated by the Montreal Heart Institute and funded by the government of Quebec; the National Heart, Lung, and Blood Institute of the National Institutes of Health; Montreal philanthropist Sophie Desmarais, and the COVID-19 Therapeutics Accelerator launched by the Bill & Melinda Gates Foundation, Wellcome, and Mastercard. CGI, Dacima, and Pharmascience of Montreal were also collaborators. Dr. Glatt reported no conflicts of interest. Dr. Boulware reported receiving $18 in food and beverages from Gilead Sciences in 2018.
A version of this article first appeared on Medscape.com.
Science by press release and preprint has cooled clinician enthusiasm for the use of colchicine in nonhospitalized patients with COVID-19, despite a pressing need for early treatments.
As previously reported by this news organization, a Jan. 22 press release announced that the massive ColCORONA study missed its primary endpoint of hospitalization or death among 4,488 newly diagnosed patients at increased risk for hospitalization.
But it also touted that use of the anti-inflammatory drug significantly reduced the primary endpoint in 4,159 of those patients with polymerase chain reaction–confirmed COVID and led to reductions of 25%, 50%, and 44%, respectively, for hospitalizations, ventilations, and death.
Lead investigator Jean-Claude Tardif, MD, director of the Montreal Heart Institute Research Centre, deemed the findings a “medical breakthrough.”
When the preprint released a few days later, however, newly revealed confidence intervals showed colchicine did not meaningfully reduce the need for mechanical ventilation (odds ratio, 0.50; 95% confidence interval, 0.23-1.07) or death alone (OR, 0.56; 95% CI, 0.19-1.66).
Further, the significant benefit on the primary outcome came at the cost of a fivefold increase in pulmonary embolism (11 vs. 2; P = .01), which was not mentioned in the press release.
“Whether this represents a real phenomenon or simply the play of chance is not known,” Dr. Tardif and colleagues noted later in the preprint.
“I read the preprint on colchicine and I have so many questions,” Aaron E. Glatt, MD, spokesperson for the Infectious Diseases Society of America and chief of infectious diseases, Mount Sinai South Nassau, Hewlett, N.Y., said in an interview. “I’ve been burned too many times with COVID and prefer to see better data.
“People sometimes say if you wait for perfect data, people are going to die,” he said. “Yeah, but we have no idea if people are going to die from getting this drug more than not getting it. That’s what concerns me. How many pulmonary emboli are going to be fatal versus the slight benefit that the study showed?”
The pushback to the non–peer-reviewed data on social media and via emails was so strong that Dr. Tardif posted a nearly 2,000-word letter responding to the many questions at play.
Chief among them was why the trial, originally planned for 6,000 patients, was stopped early by the investigators without consultation with the data safety monitoring board (DSMB).
The explanation in the letter that logistical issues like running the study call center, budget constraints, and a perceived need to quickly communicate the results left some calling foul that the study wasn’t allowed to finish and come to a more definitive conclusion.
“I can be a little bit sympathetic to their cause but at the same time the DSMB should have said no,” said David Boulware, MD, MPH, who led a recent hydroxychloroquine trial in COVID-19. “The problem is we’re sort of left in limbo, where some people kind of believe it and some say it’s not really a thing. So it’s not really moving the needle, as far as guidelines go.”
Indeed, a Twitter poll by cardiologist James Januzzi Jr., MD, captured the uncertainty, with 28% of respondents saying the trial was “neutral,” 58% saying “maybe but meh,” and 14% saying “colchicine for all.”
Another poll cheekily asked whether ColCORONA was the Gamestop/Reddit equivalent of COVID.
“The press release really didn’t help things because it very much oversold the effect. That, I think, poisoned the well,” said Dr. Boulware, professor of medicine in infectious diseases at the University of Minnesota, Minneapolis.
“The question I’m left with is not whether colchicine works, but who does it work in,” he said. “That’s really the fundamental question because it does seem that there are probably high-risk groups in their trial and others where they benefit, whereas other groups don’t benefit. In the subgroup analysis, there was absolutely no beneficial effect in women.”
According to the authors, the number needed to treat to prevent one death or hospitalization was 71 overall, but 29 for patients with diabetes, 31 for those aged 70 years and older, 53 for patients with respiratory disease, and 25 for those with coronary disease or heart failure.
Men are at higher risk overall for poor outcomes. But “the authors didn’t present a multivariable analysis, so it is unclear if another factor, such as a differential prevalence of smoking or cardiovascular risk factors, contributed to the differential benefit,” Rachel Bender Ignacio, MD, MPH, infectious disease specialist, University of Washington, Seattle, said in an interview.
Importantly, in this pragmatic study, duration and severity of symptoms were not reported, observed Dr. Bender Ignacio, who is also a STOP-COVID-2 investigator. “We don’t yet have data as to whether colchicine shortens duration or severity of symptoms or prevents long COVID, so we need more data on that.”
The overall risk for serious adverse events was lower in the colchicine group, but the difference in pulmonary embolism (PE) was striking, she said. This could be caused by a real biologic effect, or it’s possible that persons with shortness of breath and hypoxia, without evident viral pneumonia on chest x-ray after a positive COVID-19 test, were more likely to receive a CT-PE study.
The press release also failed to include information, later noted in the preprint, that the MHI has submitted two patents related to colchicine: “Methods of treating a coronavirus infection using colchicine” and “Early administration of low-dose colchicine after myocardial infarction.”
Reached for clarification, MHI communications adviser Camille Turbide said in an interview that the first patent “simply refers to the novel concept of preventing complications of COVID-19, such as admission to the hospital, with colchicine as tested in the ColCORONA study.”
The second patent, she said, refers to the “novel concept that administering colchicine early after a major adverse cardiovascular event is better than waiting several days,” as supported by the COLCOT study, which Dr. Tardif also led.
The patents are being reviewed by authorities and “Dr. Tardif has waived his rights in these patents and does not stand to benefit financially at all if colchicine becomes used as a treatment for COVID-19,” Ms. Turbide said.
Dr. Tardif did not respond to interview requests for this story. Dr. Glatt said conflicts of interest must be assessed and are “something that is of great concern in any scientific study.”
Cardiologist Steve Nissen, MD, of the Cleveland Clinic said in an interview that, “despite the negative results, the study does suggest that colchicine might have a benefit and should be studied in future trials. These findings are not sufficient evidence to suggest use of the drug in patients infected with COVID-19.”
He noted that adverse effects like diarrhea were expected but that the excess PE was unexpected and needs greater clarification.
“Stopping the trial for administrative reasons is puzzling and undermined the ability of the trial to give a reliable answer,” Dr. Nissen said. “This is a reasonable pilot study that should be viewed as hypothesis generating but inconclusive.”
Several sources said a new trial is unlikely, particularly given the cost and 28 trials already evaluating colchicine. Among these are RECOVERY and COLCOVID, testing whether colchicine can reduce the duration of hospitalization or death in hospitalized patients with COVID-19.
Because there are so many trials ongoing right now, including for antivirals and other immunomodulators, it’s important that, if colchicine comes to routine clinical use, it provides access to treatment for those not able or willing to access clinical trials, rather than impeding clinical trial enrollment, Dr. Bender Ignacio suggested.
“We have already learned the lesson in the pandemic that early adoption of potentially promising therapies can negatively impact our ability to study and develop other promising treatments,” she said.
The trial was coordinated by the Montreal Heart Institute and funded by the government of Quebec; the National Heart, Lung, and Blood Institute of the National Institutes of Health; Montreal philanthropist Sophie Desmarais, and the COVID-19 Therapeutics Accelerator launched by the Bill & Melinda Gates Foundation, Wellcome, and Mastercard. CGI, Dacima, and Pharmascience of Montreal were also collaborators. Dr. Glatt reported no conflicts of interest. Dr. Boulware reported receiving $18 in food and beverages from Gilead Sciences in 2018.
A version of this article first appeared on Medscape.com.
Science by press release and preprint has cooled clinician enthusiasm for the use of colchicine in nonhospitalized patients with COVID-19, despite a pressing need for early treatments.
As previously reported by this news organization, a Jan. 22 press release announced that the massive ColCORONA study missed its primary endpoint of hospitalization or death among 4,488 newly diagnosed patients at increased risk for hospitalization.
But it also touted that use of the anti-inflammatory drug significantly reduced the primary endpoint in 4,159 of those patients with polymerase chain reaction–confirmed COVID and led to reductions of 25%, 50%, and 44%, respectively, for hospitalizations, ventilations, and death.
Lead investigator Jean-Claude Tardif, MD, director of the Montreal Heart Institute Research Centre, deemed the findings a “medical breakthrough.”
When the preprint released a few days later, however, newly revealed confidence intervals showed colchicine did not meaningfully reduce the need for mechanical ventilation (odds ratio, 0.50; 95% confidence interval, 0.23-1.07) or death alone (OR, 0.56; 95% CI, 0.19-1.66).
Further, the significant benefit on the primary outcome came at the cost of a fivefold increase in pulmonary embolism (11 vs. 2; P = .01), which was not mentioned in the press release.
“Whether this represents a real phenomenon or simply the play of chance is not known,” Dr. Tardif and colleagues noted later in the preprint.
“I read the preprint on colchicine and I have so many questions,” Aaron E. Glatt, MD, spokesperson for the Infectious Diseases Society of America and chief of infectious diseases, Mount Sinai South Nassau, Hewlett, N.Y., said in an interview. “I’ve been burned too many times with COVID and prefer to see better data.
“People sometimes say if you wait for perfect data, people are going to die,” he said. “Yeah, but we have no idea if people are going to die from getting this drug more than not getting it. That’s what concerns me. How many pulmonary emboli are going to be fatal versus the slight benefit that the study showed?”
The pushback to the non–peer-reviewed data on social media and via emails was so strong that Dr. Tardif posted a nearly 2,000-word letter responding to the many questions at play.
Chief among them was why the trial, originally planned for 6,000 patients, was stopped early by the investigators without consultation with the data safety monitoring board (DSMB).
The explanation in the letter that logistical issues like running the study call center, budget constraints, and a perceived need to quickly communicate the results left some calling foul that the study wasn’t allowed to finish and come to a more definitive conclusion.
“I can be a little bit sympathetic to their cause but at the same time the DSMB should have said no,” said David Boulware, MD, MPH, who led a recent hydroxychloroquine trial in COVID-19. “The problem is we’re sort of left in limbo, where some people kind of believe it and some say it’s not really a thing. So it’s not really moving the needle, as far as guidelines go.”
Indeed, a Twitter poll by cardiologist James Januzzi Jr., MD, captured the uncertainty, with 28% of respondents saying the trial was “neutral,” 58% saying “maybe but meh,” and 14% saying “colchicine for all.”
Another poll cheekily asked whether ColCORONA was the Gamestop/Reddit equivalent of COVID.
“The press release really didn’t help things because it very much oversold the effect. That, I think, poisoned the well,” said Dr. Boulware, professor of medicine in infectious diseases at the University of Minnesota, Minneapolis.
“The question I’m left with is not whether colchicine works, but who does it work in,” he said. “That’s really the fundamental question because it does seem that there are probably high-risk groups in their trial and others where they benefit, whereas other groups don’t benefit. In the subgroup analysis, there was absolutely no beneficial effect in women.”
According to the authors, the number needed to treat to prevent one death or hospitalization was 71 overall, but 29 for patients with diabetes, 31 for those aged 70 years and older, 53 for patients with respiratory disease, and 25 for those with coronary disease or heart failure.
Men are at higher risk overall for poor outcomes. But “the authors didn’t present a multivariable analysis, so it is unclear if another factor, such as a differential prevalence of smoking or cardiovascular risk factors, contributed to the differential benefit,” Rachel Bender Ignacio, MD, MPH, infectious disease specialist, University of Washington, Seattle, said in an interview.
Importantly, in this pragmatic study, duration and severity of symptoms were not reported, observed Dr. Bender Ignacio, who is also a STOP-COVID-2 investigator. “We don’t yet have data as to whether colchicine shortens duration or severity of symptoms or prevents long COVID, so we need more data on that.”
The overall risk for serious adverse events was lower in the colchicine group, but the difference in pulmonary embolism (PE) was striking, she said. This could be caused by a real biologic effect, or it’s possible that persons with shortness of breath and hypoxia, without evident viral pneumonia on chest x-ray after a positive COVID-19 test, were more likely to receive a CT-PE study.
The press release also failed to include information, later noted in the preprint, that the MHI has submitted two patents related to colchicine: “Methods of treating a coronavirus infection using colchicine” and “Early administration of low-dose colchicine after myocardial infarction.”
Reached for clarification, MHI communications adviser Camille Turbide said in an interview that the first patent “simply refers to the novel concept of preventing complications of COVID-19, such as admission to the hospital, with colchicine as tested in the ColCORONA study.”
The second patent, she said, refers to the “novel concept that administering colchicine early after a major adverse cardiovascular event is better than waiting several days,” as supported by the COLCOT study, which Dr. Tardif also led.
The patents are being reviewed by authorities and “Dr. Tardif has waived his rights in these patents and does not stand to benefit financially at all if colchicine becomes used as a treatment for COVID-19,” Ms. Turbide said.
Dr. Tardif did not respond to interview requests for this story. Dr. Glatt said conflicts of interest must be assessed and are “something that is of great concern in any scientific study.”
Cardiologist Steve Nissen, MD, of the Cleveland Clinic said in an interview that, “despite the negative results, the study does suggest that colchicine might have a benefit and should be studied in future trials. These findings are not sufficient evidence to suggest use of the drug in patients infected with COVID-19.”
He noted that adverse effects like diarrhea were expected but that the excess PE was unexpected and needs greater clarification.
“Stopping the trial for administrative reasons is puzzling and undermined the ability of the trial to give a reliable answer,” Dr. Nissen said. “This is a reasonable pilot study that should be viewed as hypothesis generating but inconclusive.”
Several sources said a new trial is unlikely, particularly given the cost and 28 trials already evaluating colchicine. Among these are RECOVERY and COLCOVID, testing whether colchicine can reduce the duration of hospitalization or death in hospitalized patients with COVID-19.
Because there are so many trials ongoing right now, including for antivirals and other immunomodulators, it’s important that, if colchicine comes to routine clinical use, it provides access to treatment for those not able or willing to access clinical trials, rather than impeding clinical trial enrollment, Dr. Bender Ignacio suggested.
“We have already learned the lesson in the pandemic that early adoption of potentially promising therapies can negatively impact our ability to study and develop other promising treatments,” she said.
The trial was coordinated by the Montreal Heart Institute and funded by the government of Quebec; the National Heart, Lung, and Blood Institute of the National Institutes of Health; Montreal philanthropist Sophie Desmarais, and the COVID-19 Therapeutics Accelerator launched by the Bill & Melinda Gates Foundation, Wellcome, and Mastercard. CGI, Dacima, and Pharmascience of Montreal were also collaborators. Dr. Glatt reported no conflicts of interest. Dr. Boulware reported receiving $18 in food and beverages from Gilead Sciences in 2018.
A version of this article first appeared on Medscape.com.