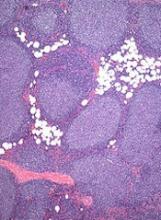
User login
The chief complaint
In medical school, they taught us to learn the patient’s chief complaint.
In dermatology the presenting complaint is on the outside, where the skin is. The chief complaint is often deeper.
Sandra
“How are your parents?”
“Getting older. I’m over their house every day. It’s always something.
“My husband had a stroke this year. Our daughter – she’s a nurse – made him get help. ‘You’re not talking right,’ she said to him. You’re going to the hospital right now.’
“Stan’s at home. He can’t work construction anymore. When I get back from taking care of my parents, I take care of him.”
Sandra’s moles are normal. Who is taking care of her?
Grigoriy
“I’ve had a hard life,” says Grigoriy, apropos of nothing.
“How?”
“My father was important in the Communist party. Stalin purged him in 1938. I was a teenager. “They kept me in a cell of one room for 15 years.”
“Why did they put you in jail?”
“I was my father’s son.”
Phil
Phil is in for his annual. He looks robust, but thinner.
“Sorry I missed last year,” he says. “I was clearing my throat a lot. An ENT doctor found that I had cancer of the vocal cords. I got 39 radiation sessions. They said I would handle them OK, but afterward, I’d feel awful. They were right.
“I lost 20 pounds,” says Phil. “But now I’m getting back to myself.” His smile is broad, but uncertain.
Fred
Fred’s rash is impressive: big, purple blotches all over. Could be a drug eruption, only he takes no drugs.
“It may be viral,” I say.
“Can I visit my Dad in Providence Sunday?” he asks. “It’s Father’s Day.”
“I’m not sure …”
“Dad has cancer of the esophagus. They’re hoping that chemo may buy him a little time.”
I tell Fred to wash carefully. Some things can’t be rescheduled.
Emily
Emily’s Mom has left me a note to read before I see her daughter. It lists Emily’s five psychoactive medications.
Emily is lying on her back and does not sit up. Her gaze is vague and unfocused.
Emily has moderate papular acne on her cheeks. That is her presenting complaint. It is not her chief complaint. As for what her mother goes through, I can barely imagine.
Brenda
Brenda comes for 6-month skin checks. Usually with her husband Glen, but not today.
“Glen’s not so well,” Brenda says. The doctors diagnosed him with MS. They’re vague about how fast it will progress. I guess they don’t know.
“To tell the truth, Glen’s pretty depressed. But he doesn’t want to talk to anyone about it. Do you know a psychiatrist who specializes in MS patients? Glen might take your advice.”
Tom
“It’s been a tough year. Eddie died. You saw him years ago, I think.”
I actually remember Eddie. A troubled kid with terrible acne. He had one visit, never came back.
“I was walking in a mountain field in Cambodia when I got the word,” says Tom. “My ex called me. ‘Tom died,’ she said. ‘Drug overdose. Come home.’
“Every year I walk through Cambodia and Myanmar for a month,” says Tom. “Just to be alone. The people there are nice. They let me be.
“Eddie was a good boy. He hung with the wrong crowd. He made a mistake, and he could never get past it. I think of him every day.”
Frank
Frank doesn’t pick. Frank gouges. He’s been gouging his forearms for years. Intralesional steroids help a little. But he can’t stop.
“I guess it’s stress,” Frank says.
“How about avoiding stress?” I ask, with a smile.
Frank breaks down and weeps.
“I’m sorry,” he says. He gathers himself. “My wife has breast cancer. Mammogram showed a spot 4 years ago. Then it grew. It’s already stage four. Our kids are teenagers.”
Frank breaks down again. He apologizes again. “I’m so sorry for being like this.” Again he weeps, again he apologizes. “I shouldn’t act like this,” he says. “I’m sorry.”
I am sorry, too. Very sorry, indeed. But for patients’ true chief complaints, often incidental to the superficial presenting ones, all I can do is listen. It’s not much, but it’s the best I can do. For many patients, over many years, listening has been the most I’ve had to offer.
Dr. Rockoff practices dermatology in Brookline, Mass., and is a longtime contributor to Dermatology News. He serves on the clinical faculty at Tufts University, Boston, and has taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available at amazon.com and barnesandnoble.com. Write to him at dermnews@mdedge.com.
In medical school, they taught us to learn the patient’s chief complaint.
In dermatology the presenting complaint is on the outside, where the skin is. The chief complaint is often deeper.
Sandra
“How are your parents?”
“Getting older. I’m over their house every day. It’s always something.
“My husband had a stroke this year. Our daughter – she’s a nurse – made him get help. ‘You’re not talking right,’ she said to him. You’re going to the hospital right now.’
“Stan’s at home. He can’t work construction anymore. When I get back from taking care of my parents, I take care of him.”
Sandra’s moles are normal. Who is taking care of her?
Grigoriy
“I’ve had a hard life,” says Grigoriy, apropos of nothing.
“How?”
“My father was important in the Communist party. Stalin purged him in 1938. I was a teenager. “They kept me in a cell of one room for 15 years.”
“Why did they put you in jail?”
“I was my father’s son.”
Phil
Phil is in for his annual. He looks robust, but thinner.
“Sorry I missed last year,” he says. “I was clearing my throat a lot. An ENT doctor found that I had cancer of the vocal cords. I got 39 radiation sessions. They said I would handle them OK, but afterward, I’d feel awful. They were right.
“I lost 20 pounds,” says Phil. “But now I’m getting back to myself.” His smile is broad, but uncertain.
Fred
Fred’s rash is impressive: big, purple blotches all over. Could be a drug eruption, only he takes no drugs.
“It may be viral,” I say.
“Can I visit my Dad in Providence Sunday?” he asks. “It’s Father’s Day.”
“I’m not sure …”
“Dad has cancer of the esophagus. They’re hoping that chemo may buy him a little time.”
I tell Fred to wash carefully. Some things can’t be rescheduled.
Emily
Emily’s Mom has left me a note to read before I see her daughter. It lists Emily’s five psychoactive medications.
Emily is lying on her back and does not sit up. Her gaze is vague and unfocused.
Emily has moderate papular acne on her cheeks. That is her presenting complaint. It is not her chief complaint. As for what her mother goes through, I can barely imagine.
Brenda
Brenda comes for 6-month skin checks. Usually with her husband Glen, but not today.
“Glen’s not so well,” Brenda says. The doctors diagnosed him with MS. They’re vague about how fast it will progress. I guess they don’t know.
“To tell the truth, Glen’s pretty depressed. But he doesn’t want to talk to anyone about it. Do you know a psychiatrist who specializes in MS patients? Glen might take your advice.”
Tom
“It’s been a tough year. Eddie died. You saw him years ago, I think.”
I actually remember Eddie. A troubled kid with terrible acne. He had one visit, never came back.
“I was walking in a mountain field in Cambodia when I got the word,” says Tom. “My ex called me. ‘Tom died,’ she said. ‘Drug overdose. Come home.’
“Every year I walk through Cambodia and Myanmar for a month,” says Tom. “Just to be alone. The people there are nice. They let me be.
“Eddie was a good boy. He hung with the wrong crowd. He made a mistake, and he could never get past it. I think of him every day.”
Frank
Frank doesn’t pick. Frank gouges. He’s been gouging his forearms for years. Intralesional steroids help a little. But he can’t stop.
“I guess it’s stress,” Frank says.
“How about avoiding stress?” I ask, with a smile.
Frank breaks down and weeps.
“I’m sorry,” he says. He gathers himself. “My wife has breast cancer. Mammogram showed a spot 4 years ago. Then it grew. It’s already stage four. Our kids are teenagers.”
Frank breaks down again. He apologizes again. “I’m so sorry for being like this.” Again he weeps, again he apologizes. “I shouldn’t act like this,” he says. “I’m sorry.”
I am sorry, too. Very sorry, indeed. But for patients’ true chief complaints, often incidental to the superficial presenting ones, all I can do is listen. It’s not much, but it’s the best I can do. For many patients, over many years, listening has been the most I’ve had to offer.
Dr. Rockoff practices dermatology in Brookline, Mass., and is a longtime contributor to Dermatology News. He serves on the clinical faculty at Tufts University, Boston, and has taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available at amazon.com and barnesandnoble.com. Write to him at dermnews@mdedge.com.
In medical school, they taught us to learn the patient’s chief complaint.
In dermatology the presenting complaint is on the outside, where the skin is. The chief complaint is often deeper.
Sandra
“How are your parents?”
“Getting older. I’m over their house every day. It’s always something.
“My husband had a stroke this year. Our daughter – she’s a nurse – made him get help. ‘You’re not talking right,’ she said to him. You’re going to the hospital right now.’
“Stan’s at home. He can’t work construction anymore. When I get back from taking care of my parents, I take care of him.”
Sandra’s moles are normal. Who is taking care of her?
Grigoriy
“I’ve had a hard life,” says Grigoriy, apropos of nothing.
“How?”
“My father was important in the Communist party. Stalin purged him in 1938. I was a teenager. “They kept me in a cell of one room for 15 years.”
“Why did they put you in jail?”
“I was my father’s son.”
Phil
Phil is in for his annual. He looks robust, but thinner.
“Sorry I missed last year,” he says. “I was clearing my throat a lot. An ENT doctor found that I had cancer of the vocal cords. I got 39 radiation sessions. They said I would handle them OK, but afterward, I’d feel awful. They were right.
“I lost 20 pounds,” says Phil. “But now I’m getting back to myself.” His smile is broad, but uncertain.
Fred
Fred’s rash is impressive: big, purple blotches all over. Could be a drug eruption, only he takes no drugs.
“It may be viral,” I say.
“Can I visit my Dad in Providence Sunday?” he asks. “It’s Father’s Day.”
“I’m not sure …”
“Dad has cancer of the esophagus. They’re hoping that chemo may buy him a little time.”
I tell Fred to wash carefully. Some things can’t be rescheduled.
Emily
Emily’s Mom has left me a note to read before I see her daughter. It lists Emily’s five psychoactive medications.
Emily is lying on her back and does not sit up. Her gaze is vague and unfocused.
Emily has moderate papular acne on her cheeks. That is her presenting complaint. It is not her chief complaint. As for what her mother goes through, I can barely imagine.
Brenda
Brenda comes for 6-month skin checks. Usually with her husband Glen, but not today.
“Glen’s not so well,” Brenda says. The doctors diagnosed him with MS. They’re vague about how fast it will progress. I guess they don’t know.
“To tell the truth, Glen’s pretty depressed. But he doesn’t want to talk to anyone about it. Do you know a psychiatrist who specializes in MS patients? Glen might take your advice.”
Tom
“It’s been a tough year. Eddie died. You saw him years ago, I think.”
I actually remember Eddie. A troubled kid with terrible acne. He had one visit, never came back.
“I was walking in a mountain field in Cambodia when I got the word,” says Tom. “My ex called me. ‘Tom died,’ she said. ‘Drug overdose. Come home.’
“Every year I walk through Cambodia and Myanmar for a month,” says Tom. “Just to be alone. The people there are nice. They let me be.
“Eddie was a good boy. He hung with the wrong crowd. He made a mistake, and he could never get past it. I think of him every day.”
Frank
Frank doesn’t pick. Frank gouges. He’s been gouging his forearms for years. Intralesional steroids help a little. But he can’t stop.
“I guess it’s stress,” Frank says.
“How about avoiding stress?” I ask, with a smile.
Frank breaks down and weeps.
“I’m sorry,” he says. He gathers himself. “My wife has breast cancer. Mammogram showed a spot 4 years ago. Then it grew. It’s already stage four. Our kids are teenagers.”
Frank breaks down again. He apologizes again. “I’m so sorry for being like this.” Again he weeps, again he apologizes. “I shouldn’t act like this,” he says. “I’m sorry.”
I am sorry, too. Very sorry, indeed. But for patients’ true chief complaints, often incidental to the superficial presenting ones, all I can do is listen. It’s not much, but it’s the best I can do. For many patients, over many years, listening has been the most I’ve had to offer.
Dr. Rockoff practices dermatology in Brookline, Mass., and is a longtime contributor to Dermatology News. He serves on the clinical faculty at Tufts University, Boston, and has taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available at amazon.com and barnesandnoble.com. Write to him at dermnews@mdedge.com.
Am I going to die?
Every cancer diagnosis starts out as anything but. That’s what I was thinking when I met Sue Marcus (not her real name) in the emergency department one Friday afternoon.
“You know,” she said in a matter of fact manner. “I think I might have the flu.”
“Yes,” I said. “You might.”
“I was in Vegas a week ago, and the person next to me was sick.”
“It’s definitely possible. That’s one of the things we’ll test for.”
“I mean ... what else can it be?”
I had information Sue didn’t yet. All her blood counts were disturbingly low. On top of that, the lab had picked up several atypical appearing cells. They could be reactive, in the setting of infection, or they could be blasts – a new diagnosis of leukemia.
I paused.
“I’m going to say this now, so you know what we are looking for. There’s a lot more information we still need.”
“But?”
“But, another possibility is that it is cancer.”
“Cancer?”
“Leukemia, maybe.”
I explained the next steps. We would have flow cytometry by that night, but it’s not a perfect test. We were limited, actually, by logistics. It was a Friday night. We would do a bone marrow biopsy on Monday. It would be several days before we’d know with certainty.
That evening, the hematology fellow and I looked at the slide under the microscope. We agreed with the lab.
“That’s a blast, isn’t it?”
“Yes, I think it is.”
But they were not completely classic, and they were sporadic. It wasn’t enough to say for sure. Meanwhile, Sue, understandably, wanted answers.
“What did you see? Is it cancer, or not?”
There was flow cytometry that night, showing an abnormal population of cells. And then, finally, there was a bone marrow biopsy clinching the diagnosis.
We explained what the path ahead looked like: Hospitalization for a month. Chemotherapy, then a repeat bone marrow biopsy to look for response. Then more chemotherapy. Chances of remission. Long-term implications.
As we gathered more information, Sue’s questions evolved.
“Is it curable?”
“Can I go back to work?”
And one day, a week into treatment, she asked, “Am I going to die?”
Over my last 3 years as an internal medicine resident, I’ve been humbled by how often patients turned to me for answers to some of the most difficult questions I could imagine. Sometimes, the questions seemed purely factual, but often, they took a more existential bent: How should I spend my last months? What should I do now? Patients have asked me if they are going to die, along with when, how, and even why.
I’ve wrestled with how to communicate candidly, treading a delicate balance between being as up-front as possible while also recognizing the uncertainty inherent in predicting. I’ve struggled with walking the tightrope of delivering bad news while also emphasizing compassion and support.
I chose hematology and oncology in part because of the gravity of these interactions. I enjoy being a primary doctor for a complex, sick, patient population who are grappling with physically and emotionally challenging illness. I value longitudinal relationships with patients I know well. I found the medicine of hematology and oncology interdisciplinary, the details high stakes, and the big questions always at play. If it was meaningful work I sought, cancer became the ultimate question that mattered.
The changing landscape of cancer care is making these conversations substantially more difficult. A central tenet of medicine is truthfulness: setting the stage for what is going on and what to anticipate. It’s explaining the nuances of the upcoming treatment options, while also addressing what a person’s life may look like down the road. It’s understanding what matters most to a patient, understanding what therapeutic choices we can offer – and then trying to reconcile them, as best as we can.
This has always been hard. But it’s getting harder. The options we have to treat cancer are expanding rapidly as immunotherapy competes with the basics of chemotherapy, radiation, and surgery. We work alongside researchers looking to change the paradigm, collecting information on outcomes and side effects as we go along. We are learning and we are applying what we learn – in real time – on real people willing to try.
How can we speak honestly about a prognosis when our data are limited and our tools are in continuous flux? How can we prepare someone for what lies ahead when we are still trying to grasp what today looks like?
All the while, medical uncertainties are amplified by a complex system with many moving parts. It’s a system in which some patients cannot afford care, in which insurance companies may deny necessary treatment, and where families may come together or fall apart in the face of incredible adversity. There are factors outside the scope of pure medicine that make the path ahead all the hazier and navigating it all the more challenging.
In July, I began my hematology and oncology fellowship. I am caring for patients with a range of cancers, and all of that goes along with the weight of those diagnoses. Learning the most up-to-date management in a constantly evolving landscape will be an ongoing skill. That my patients allow me into their most vulnerable moments – and trust me with them – is a gift.
For patients like Sue, there sometimes remain more questions than answers. She recently underwent her third round of chemotherapy and endured multiple blood clots. With her insurance covering only limited interventions, she is deciding what to focus on and where to receive her care. Her story, like many others, continues to be written.
I am deeply aware of the difficulties that are a part of the world of cancer – and the heartbreak. How can we hold up scans triumphantly showing no recurrence in some patients while others suffer one failed treatment after another? When should we push for more therapy and when should we shift our efforts toward comfort? What should we prioritize – medically and personally – if time is limited?
These questions are hard, but I cannot think of any that are more meaningful.
This is a column about patients and uncertainty as I pursue my hematology and oncology fellowship and grapple with these questions. I look forward to sharing them with you each month.
Dr. Yurkiewicz is a fellow in hematology and oncology at Stanford (Calif.) University. Follow her on Twitter @ilanayurkiewicz.
Every cancer diagnosis starts out as anything but. That’s what I was thinking when I met Sue Marcus (not her real name) in the emergency department one Friday afternoon.
“You know,” she said in a matter of fact manner. “I think I might have the flu.”
“Yes,” I said. “You might.”
“I was in Vegas a week ago, and the person next to me was sick.”
“It’s definitely possible. That’s one of the things we’ll test for.”
“I mean ... what else can it be?”
I had information Sue didn’t yet. All her blood counts were disturbingly low. On top of that, the lab had picked up several atypical appearing cells. They could be reactive, in the setting of infection, or they could be blasts – a new diagnosis of leukemia.
I paused.
“I’m going to say this now, so you know what we are looking for. There’s a lot more information we still need.”
“But?”
“But, another possibility is that it is cancer.”
“Cancer?”
“Leukemia, maybe.”
I explained the next steps. We would have flow cytometry by that night, but it’s not a perfect test. We were limited, actually, by logistics. It was a Friday night. We would do a bone marrow biopsy on Monday. It would be several days before we’d know with certainty.
That evening, the hematology fellow and I looked at the slide under the microscope. We agreed with the lab.
“That’s a blast, isn’t it?”
“Yes, I think it is.”
But they were not completely classic, and they were sporadic. It wasn’t enough to say for sure. Meanwhile, Sue, understandably, wanted answers.
“What did you see? Is it cancer, or not?”
There was flow cytometry that night, showing an abnormal population of cells. And then, finally, there was a bone marrow biopsy clinching the diagnosis.
We explained what the path ahead looked like: Hospitalization for a month. Chemotherapy, then a repeat bone marrow biopsy to look for response. Then more chemotherapy. Chances of remission. Long-term implications.
As we gathered more information, Sue’s questions evolved.
“Is it curable?”
“Can I go back to work?”
And one day, a week into treatment, she asked, “Am I going to die?”
Over my last 3 years as an internal medicine resident, I’ve been humbled by how often patients turned to me for answers to some of the most difficult questions I could imagine. Sometimes, the questions seemed purely factual, but often, they took a more existential bent: How should I spend my last months? What should I do now? Patients have asked me if they are going to die, along with when, how, and even why.
I’ve wrestled with how to communicate candidly, treading a delicate balance between being as up-front as possible while also recognizing the uncertainty inherent in predicting. I’ve struggled with walking the tightrope of delivering bad news while also emphasizing compassion and support.
I chose hematology and oncology in part because of the gravity of these interactions. I enjoy being a primary doctor for a complex, sick, patient population who are grappling with physically and emotionally challenging illness. I value longitudinal relationships with patients I know well. I found the medicine of hematology and oncology interdisciplinary, the details high stakes, and the big questions always at play. If it was meaningful work I sought, cancer became the ultimate question that mattered.
The changing landscape of cancer care is making these conversations substantially more difficult. A central tenet of medicine is truthfulness: setting the stage for what is going on and what to anticipate. It’s explaining the nuances of the upcoming treatment options, while also addressing what a person’s life may look like down the road. It’s understanding what matters most to a patient, understanding what therapeutic choices we can offer – and then trying to reconcile them, as best as we can.
This has always been hard. But it’s getting harder. The options we have to treat cancer are expanding rapidly as immunotherapy competes with the basics of chemotherapy, radiation, and surgery. We work alongside researchers looking to change the paradigm, collecting information on outcomes and side effects as we go along. We are learning and we are applying what we learn – in real time – on real people willing to try.
How can we speak honestly about a prognosis when our data are limited and our tools are in continuous flux? How can we prepare someone for what lies ahead when we are still trying to grasp what today looks like?
All the while, medical uncertainties are amplified by a complex system with many moving parts. It’s a system in which some patients cannot afford care, in which insurance companies may deny necessary treatment, and where families may come together or fall apart in the face of incredible adversity. There are factors outside the scope of pure medicine that make the path ahead all the hazier and navigating it all the more challenging.
In July, I began my hematology and oncology fellowship. I am caring for patients with a range of cancers, and all of that goes along with the weight of those diagnoses. Learning the most up-to-date management in a constantly evolving landscape will be an ongoing skill. That my patients allow me into their most vulnerable moments – and trust me with them – is a gift.
For patients like Sue, there sometimes remain more questions than answers. She recently underwent her third round of chemotherapy and endured multiple blood clots. With her insurance covering only limited interventions, she is deciding what to focus on and where to receive her care. Her story, like many others, continues to be written.
I am deeply aware of the difficulties that are a part of the world of cancer – and the heartbreak. How can we hold up scans triumphantly showing no recurrence in some patients while others suffer one failed treatment after another? When should we push for more therapy and when should we shift our efforts toward comfort? What should we prioritize – medically and personally – if time is limited?
These questions are hard, but I cannot think of any that are more meaningful.
This is a column about patients and uncertainty as I pursue my hematology and oncology fellowship and grapple with these questions. I look forward to sharing them with you each month.
Dr. Yurkiewicz is a fellow in hematology and oncology at Stanford (Calif.) University. Follow her on Twitter @ilanayurkiewicz.
Every cancer diagnosis starts out as anything but. That’s what I was thinking when I met Sue Marcus (not her real name) in the emergency department one Friday afternoon.
“You know,” she said in a matter of fact manner. “I think I might have the flu.”
“Yes,” I said. “You might.”
“I was in Vegas a week ago, and the person next to me was sick.”
“It’s definitely possible. That’s one of the things we’ll test for.”
“I mean ... what else can it be?”
I had information Sue didn’t yet. All her blood counts were disturbingly low. On top of that, the lab had picked up several atypical appearing cells. They could be reactive, in the setting of infection, or they could be blasts – a new diagnosis of leukemia.
I paused.
“I’m going to say this now, so you know what we are looking for. There’s a lot more information we still need.”
“But?”
“But, another possibility is that it is cancer.”
“Cancer?”
“Leukemia, maybe.”
I explained the next steps. We would have flow cytometry by that night, but it’s not a perfect test. We were limited, actually, by logistics. It was a Friday night. We would do a bone marrow biopsy on Monday. It would be several days before we’d know with certainty.
That evening, the hematology fellow and I looked at the slide under the microscope. We agreed with the lab.
“That’s a blast, isn’t it?”
“Yes, I think it is.”
But they were not completely classic, and they were sporadic. It wasn’t enough to say for sure. Meanwhile, Sue, understandably, wanted answers.
“What did you see? Is it cancer, or not?”
There was flow cytometry that night, showing an abnormal population of cells. And then, finally, there was a bone marrow biopsy clinching the diagnosis.
We explained what the path ahead looked like: Hospitalization for a month. Chemotherapy, then a repeat bone marrow biopsy to look for response. Then more chemotherapy. Chances of remission. Long-term implications.
As we gathered more information, Sue’s questions evolved.
“Is it curable?”
“Can I go back to work?”
And one day, a week into treatment, she asked, “Am I going to die?”
Over my last 3 years as an internal medicine resident, I’ve been humbled by how often patients turned to me for answers to some of the most difficult questions I could imagine. Sometimes, the questions seemed purely factual, but often, they took a more existential bent: How should I spend my last months? What should I do now? Patients have asked me if they are going to die, along with when, how, and even why.
I’ve wrestled with how to communicate candidly, treading a delicate balance between being as up-front as possible while also recognizing the uncertainty inherent in predicting. I’ve struggled with walking the tightrope of delivering bad news while also emphasizing compassion and support.
I chose hematology and oncology in part because of the gravity of these interactions. I enjoy being a primary doctor for a complex, sick, patient population who are grappling with physically and emotionally challenging illness. I value longitudinal relationships with patients I know well. I found the medicine of hematology and oncology interdisciplinary, the details high stakes, and the big questions always at play. If it was meaningful work I sought, cancer became the ultimate question that mattered.
The changing landscape of cancer care is making these conversations substantially more difficult. A central tenet of medicine is truthfulness: setting the stage for what is going on and what to anticipate. It’s explaining the nuances of the upcoming treatment options, while also addressing what a person’s life may look like down the road. It’s understanding what matters most to a patient, understanding what therapeutic choices we can offer – and then trying to reconcile them, as best as we can.
This has always been hard. But it’s getting harder. The options we have to treat cancer are expanding rapidly as immunotherapy competes with the basics of chemotherapy, radiation, and surgery. We work alongside researchers looking to change the paradigm, collecting information on outcomes and side effects as we go along. We are learning and we are applying what we learn – in real time – on real people willing to try.
How can we speak honestly about a prognosis when our data are limited and our tools are in continuous flux? How can we prepare someone for what lies ahead when we are still trying to grasp what today looks like?
All the while, medical uncertainties are amplified by a complex system with many moving parts. It’s a system in which some patients cannot afford care, in which insurance companies may deny necessary treatment, and where families may come together or fall apart in the face of incredible adversity. There are factors outside the scope of pure medicine that make the path ahead all the hazier and navigating it all the more challenging.
In July, I began my hematology and oncology fellowship. I am caring for patients with a range of cancers, and all of that goes along with the weight of those diagnoses. Learning the most up-to-date management in a constantly evolving landscape will be an ongoing skill. That my patients allow me into their most vulnerable moments – and trust me with them – is a gift.
For patients like Sue, there sometimes remain more questions than answers. She recently underwent her third round of chemotherapy and endured multiple blood clots. With her insurance covering only limited interventions, she is deciding what to focus on and where to receive her care. Her story, like many others, continues to be written.
I am deeply aware of the difficulties that are a part of the world of cancer – and the heartbreak. How can we hold up scans triumphantly showing no recurrence in some patients while others suffer one failed treatment after another? When should we push for more therapy and when should we shift our efforts toward comfort? What should we prioritize – medically and personally – if time is limited?
These questions are hard, but I cannot think of any that are more meaningful.
This is a column about patients and uncertainty as I pursue my hematology and oncology fellowship and grapple with these questions. I look forward to sharing them with you each month.
Dr. Yurkiewicz is a fellow in hematology and oncology at Stanford (Calif.) University. Follow her on Twitter @ilanayurkiewicz.
Advances in Hematology and Oncology (August 2018)
Click here to access August 2018 Advances In Hematology and Oncology Digital Edition.
Table of Contents
- VHA Practice Guideline Recommendations for Diffuse Gliomas
- Positivity Rates in Head and Neck Cancer in the VA
- Prevalence of Cancer in Thyroid Nodules in a Veteran Population
- Fatal Drug-Resistant Invasive Infection in a 56-Year-Old Immunosuppressed Man
- Immune Checkpoint Inhibitors for Urothelial Cancer: An Update on New Therapies
- Research News: RCC. Adrenal Tumors, Myeloma Therapy, Cancer Clusters, Hodgkin Lymphoma
- Coordination of Prostate Cance Care Between Primary Care and Oncology
- Open Clinical Trials for Patients With Lung Cancers

Click here to access August 2018 Advances In Hematology and Oncology Digital Edition.
Table of Contents
- VHA Practice Guideline Recommendations for Diffuse Gliomas
- Positivity Rates in Head and Neck Cancer in the VA
- Prevalence of Cancer in Thyroid Nodules in a Veteran Population
- Fatal Drug-Resistant Invasive Infection in a 56-Year-Old Immunosuppressed Man
- Immune Checkpoint Inhibitors for Urothelial Cancer: An Update on New Therapies
- Research News: RCC. Adrenal Tumors, Myeloma Therapy, Cancer Clusters, Hodgkin Lymphoma
- Coordination of Prostate Cance Care Between Primary Care and Oncology
- Open Clinical Trials for Patients With Lung Cancers

Click here to access August 2018 Advances In Hematology and Oncology Digital Edition.
Table of Contents
- VHA Practice Guideline Recommendations for Diffuse Gliomas
- Positivity Rates in Head and Neck Cancer in the VA
- Prevalence of Cancer in Thyroid Nodules in a Veteran Population
- Fatal Drug-Resistant Invasive Infection in a 56-Year-Old Immunosuppressed Man
- Immune Checkpoint Inhibitors for Urothelial Cancer: An Update on New Therapies
- Research News: RCC. Adrenal Tumors, Myeloma Therapy, Cancer Clusters, Hodgkin Lymphoma
- Coordination of Prostate Cance Care Between Primary Care and Oncology
- Open Clinical Trials for Patients With Lung Cancers

Breast Implant Rupture After Radiation
The rupture rate for breast implants is about 10% at 10 years after insertion. That means women aged ≥ 70 years have a greater risk of rupture. For women who had breast augmentation or reconstruction before the advent of fifth-generation implants, there are no specific recommendations regarding follow-up and very little guidance in the literature about management for those who have had implants after radiation, say clinicians from Mayo Clinic.
They report on a 74-year-old patient who was treated for breast cancer in 1987 and 1988. She underwent lumpectomy, adjuvant unilateral radiation, a right simple mastectomy, left modified radical mastectomy, and implant-based reconstruction. Nearly 30 years later, she felt an asymmetry in 1 breast. Magnetic resonance imaging and ultrasound revealed that both implants had ruptured.
It is well known, the clinicians say, that complications of postmastectomy radiotherapy include capsular contracture, infection, and loss of prosthesis in implant-based reconstruction. Studies have shown that fibrosis, a hallmark of chronic radiation therapy, can show up even several years after radiotherapy—underscoring the importance of long-term follow-up for these patients. Moreover, the fact that the consequences of silicone on irradiated mastectomy flaps is unknown posed a further challenge.
While the cause of their patient’s implant rupture is unknown, the clinicians say it is “very likely” that delayed-onset fibrosis and capsular contracture secondary to radiation played a role. Such complications, though rare, should be kept in mind, the clinicians advise, when evaluating patients who had radiation and implants.
Source:
Molinar VE, Sabbagh MD, Manrique OJ. BMJ Case Rep. 2018; pii: bcr-2018-224578.
doi: 10.1136/bcr-2018-224578.
The rupture rate for breast implants is about 10% at 10 years after insertion. That means women aged ≥ 70 years have a greater risk of rupture. For women who had breast augmentation or reconstruction before the advent of fifth-generation implants, there are no specific recommendations regarding follow-up and very little guidance in the literature about management for those who have had implants after radiation, say clinicians from Mayo Clinic.
They report on a 74-year-old patient who was treated for breast cancer in 1987 and 1988. She underwent lumpectomy, adjuvant unilateral radiation, a right simple mastectomy, left modified radical mastectomy, and implant-based reconstruction. Nearly 30 years later, she felt an asymmetry in 1 breast. Magnetic resonance imaging and ultrasound revealed that both implants had ruptured.
It is well known, the clinicians say, that complications of postmastectomy radiotherapy include capsular contracture, infection, and loss of prosthesis in implant-based reconstruction. Studies have shown that fibrosis, a hallmark of chronic radiation therapy, can show up even several years after radiotherapy—underscoring the importance of long-term follow-up for these patients. Moreover, the fact that the consequences of silicone on irradiated mastectomy flaps is unknown posed a further challenge.
While the cause of their patient’s implant rupture is unknown, the clinicians say it is “very likely” that delayed-onset fibrosis and capsular contracture secondary to radiation played a role. Such complications, though rare, should be kept in mind, the clinicians advise, when evaluating patients who had radiation and implants.
Source:
Molinar VE, Sabbagh MD, Manrique OJ. BMJ Case Rep. 2018; pii: bcr-2018-224578.
doi: 10.1136/bcr-2018-224578.
The rupture rate for breast implants is about 10% at 10 years after insertion. That means women aged ≥ 70 years have a greater risk of rupture. For women who had breast augmentation or reconstruction before the advent of fifth-generation implants, there are no specific recommendations regarding follow-up and very little guidance in the literature about management for those who have had implants after radiation, say clinicians from Mayo Clinic.
They report on a 74-year-old patient who was treated for breast cancer in 1987 and 1988. She underwent lumpectomy, adjuvant unilateral radiation, a right simple mastectomy, left modified radical mastectomy, and implant-based reconstruction. Nearly 30 years later, she felt an asymmetry in 1 breast. Magnetic resonance imaging and ultrasound revealed that both implants had ruptured.
It is well known, the clinicians say, that complications of postmastectomy radiotherapy include capsular contracture, infection, and loss of prosthesis in implant-based reconstruction. Studies have shown that fibrosis, a hallmark of chronic radiation therapy, can show up even several years after radiotherapy—underscoring the importance of long-term follow-up for these patients. Moreover, the fact that the consequences of silicone on irradiated mastectomy flaps is unknown posed a further challenge.
While the cause of their patient’s implant rupture is unknown, the clinicians say it is “very likely” that delayed-onset fibrosis and capsular contracture secondary to radiation played a role. Such complications, though rare, should be kept in mind, the clinicians advise, when evaluating patients who had radiation and implants.
Source:
Molinar VE, Sabbagh MD, Manrique OJ. BMJ Case Rep. 2018; pii: bcr-2018-224578.
doi: 10.1136/bcr-2018-224578.
Multifaceted pharmacist intervention may reduce postdischarge ED visits and readmissions
Clinical question: Can a multifaceted intervention by a clinical pharmacist reduce the rate of ED visits and readmission over the subsequent 180 days?
Background: The period following an inpatient admission contains many potential risks for patients, among them the risk for adverse drug events. Approximately 45% of readmissions from adverse drug reactions are thought to be avoidable.
Study design: Multicentered, single-blinded, randomized, control trial, from September 2013 to April 2015.
Setting: Four acute inpatient hospitals in Denmark.
Synopsis: 1,467 adult patients being admitted for an acute hospitalization on a minimum of five medications were randomized to receive usual care, a basic intervention (medication review by a clinical pharmacist), or an extended intervention (medication review, three motivational interviews, and follow-up with the primary care physician, pharmacy and, if appropriate, nursing home by a clinical pharmacist). The primary endpoints were readmission within 30 days or 180 days, ED visits within 180 days, and a composite endpoint of readmission or ED visit within 180 days post discharge. For these endpoints, the basic intervention group had no statistically significant difference from the usual-care group. The extended intervention group had significantly lower rates of readmission within 30 days and 180 days, as well as the primary composite endpoint compared to the usual-care group (P less than .05 for all comparisons). For the extended intervention, the number needed to treat for the main composite endpoint was 12.
Bottom line: For patients admitted to the hospital, an extended intervention by a clinical pharmacist resulted in a significant reduction in readmissions.
Citation: Ravn-Nielsen LV et al. Effect of an in-hospital multifaceted clinical pharmacist intervention on the risk of readmission. JAMA Intern Med. 2018;178(3):375-82.
Dr. Biddick is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, Boston.
Clinical question: Can a multifaceted intervention by a clinical pharmacist reduce the rate of ED visits and readmission over the subsequent 180 days?
Background: The period following an inpatient admission contains many potential risks for patients, among them the risk for adverse drug events. Approximately 45% of readmissions from adverse drug reactions are thought to be avoidable.
Study design: Multicentered, single-blinded, randomized, control trial, from September 2013 to April 2015.
Setting: Four acute inpatient hospitals in Denmark.
Synopsis: 1,467 adult patients being admitted for an acute hospitalization on a minimum of five medications were randomized to receive usual care, a basic intervention (medication review by a clinical pharmacist), or an extended intervention (medication review, three motivational interviews, and follow-up with the primary care physician, pharmacy and, if appropriate, nursing home by a clinical pharmacist). The primary endpoints were readmission within 30 days or 180 days, ED visits within 180 days, and a composite endpoint of readmission or ED visit within 180 days post discharge. For these endpoints, the basic intervention group had no statistically significant difference from the usual-care group. The extended intervention group had significantly lower rates of readmission within 30 days and 180 days, as well as the primary composite endpoint compared to the usual-care group (P less than .05 for all comparisons). For the extended intervention, the number needed to treat for the main composite endpoint was 12.
Bottom line: For patients admitted to the hospital, an extended intervention by a clinical pharmacist resulted in a significant reduction in readmissions.
Citation: Ravn-Nielsen LV et al. Effect of an in-hospital multifaceted clinical pharmacist intervention on the risk of readmission. JAMA Intern Med. 2018;178(3):375-82.
Dr. Biddick is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, Boston.
Clinical question: Can a multifaceted intervention by a clinical pharmacist reduce the rate of ED visits and readmission over the subsequent 180 days?
Background: The period following an inpatient admission contains many potential risks for patients, among them the risk for adverse drug events. Approximately 45% of readmissions from adverse drug reactions are thought to be avoidable.
Study design: Multicentered, single-blinded, randomized, control trial, from September 2013 to April 2015.
Setting: Four acute inpatient hospitals in Denmark.
Synopsis: 1,467 adult patients being admitted for an acute hospitalization on a minimum of five medications were randomized to receive usual care, a basic intervention (medication review by a clinical pharmacist), or an extended intervention (medication review, three motivational interviews, and follow-up with the primary care physician, pharmacy and, if appropriate, nursing home by a clinical pharmacist). The primary endpoints were readmission within 30 days or 180 days, ED visits within 180 days, and a composite endpoint of readmission or ED visit within 180 days post discharge. For these endpoints, the basic intervention group had no statistically significant difference from the usual-care group. The extended intervention group had significantly lower rates of readmission within 30 days and 180 days, as well as the primary composite endpoint compared to the usual-care group (P less than .05 for all comparisons). For the extended intervention, the number needed to treat for the main composite endpoint was 12.
Bottom line: For patients admitted to the hospital, an extended intervention by a clinical pharmacist resulted in a significant reduction in readmissions.
Citation: Ravn-Nielsen LV et al. Effect of an in-hospital multifaceted clinical pharmacist intervention on the risk of readmission. JAMA Intern Med. 2018;178(3):375-82.
Dr. Biddick is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, Boston.
Treatment improves PFS in early stage FL
A multidrug regimen can improve upon involved-field radiotherapy (IFRT) in patients with early stage follicular lymphoma (FL), according to research published in the Journal of Clinical Oncology.
FL patients who received IFRT plus cyclophosphamide, vincristine, and prednisolone (CVP)—with or without rituximab—had a significant improvement in progression-free survival (PFS) compared to patients who received standard treatment with IFRT alone.
However, there was no significant difference in overall survival (OS) between the treatment arms.
“This is the first successful randomized study ever to be conducted in early stage follicular lymphoma comparing standard therapy to standard therapy plus effective chemotherapy or immunochemotherapy,” said Michael MacManus, MBBCh, of Peter MacCallum Cancer Centre in Melbourne, Victoria, Australia.
“It shows that the initial treatment received by patients can significantly affect their long-term chance of staying free from disease. Moving forward, we are interested in determining whether there is a benefit in overall long-term survival for patients treated with the combination with further follow-up, and if there is any way to predict if a person will benefit from combined treatment based on analyses of blood or biopsy specimens.”
Dr MacManus and his colleagues studied 150 patients with stage I to II, low-grade FL who were enrolled in this trial between 2000 and 2012.
At randomization, the patients’ median age was 57, 52% were male, 75% had stage I disease, and 48% had PET staging.
Half of patients (n=75) were randomized to receive IFRT (30-36 Gy) alone, and half were randomized to IFRT (30-36 Gy) plus 6 cycles of CVP. From 2006 on, patients in the CVP arm received rituximab (R) as well (n=31).
Baseline characteristics were well-balanced between the treatment arms.
Efficacy
The median follow-up was 9.6 years (range, 3.1 to 15.8 years).
PFS was significantly better among patients randomized to receive CVP±R (hazard ratio [HR]=0.57; P=0.033). The estimated 10-year PFS rate was 41% in the IFRT arm and 59% in the CVP±R arm.
Patients randomized to receive CVP plus R (n=31) had significantly better PFS than patients randomized to receive IFRT alone (n=31) over the same time period (HR=0.26; P=0.045).
There were 10 deaths in the IRFT arm and 5 in the CVP±R arm, but there was no significant difference in OS between the arms (HR=0.62; P=0.40). The 10-year OS rate was 86% in the IFRT arm and 95% in the CVP±R arm.
There was no significant between-arm difference in transformation to aggressive lymphoma (P=0.1). Transformation occurred in 10 patients in the IFRT arm and 4 in the CVP±R arm.
Safety
There were 148 patients from both arms who ultimately received IFRT, and 69 patients who received CVP±R.
Grade 2 toxicities occurring in more than 10% of IFRT recipients included upper gastrointestinal (n=27; 18%), skin (n=21; 14%), and mucous membrane (n=19; 12%) toxicity. One IFRT recipient had grade 3 mucositis, and 1 had grade 4 esophageal/pharyngeal mucosal toxicity.
Grade 3 toxicities occurring in at least 2 patients in the CVP±R arm included neutropenia (n=10; 14%), infection (n=8; 12%), diarrhea (n=3; 4%), elevated gamma-glutamyl transferase (n=3; 4%), fatigue (n=3; 4%), and febrile neutropenia (n=3; 4%).
Three patients (4%) in the CVP±R arm had acute grade 3 neuropathy related to vincristine. Ten patients (14%) had grade 4 neutropenia.
The most common late toxicities for the entire patient cohort were salivary gland (n=8; 5%) and skin (n=4; 3%) toxicities.
Grade 3 lung and menopausal toxicities occurred in 1 patient each. Two patients had late grade 3 vincristine neuropathy. One patient who had grade 3 neuropathy during chemotherapy progressed to grade 4.
A multidrug regimen can improve upon involved-field radiotherapy (IFRT) in patients with early stage follicular lymphoma (FL), according to research published in the Journal of Clinical Oncology.
FL patients who received IFRT plus cyclophosphamide, vincristine, and prednisolone (CVP)—with or without rituximab—had a significant improvement in progression-free survival (PFS) compared to patients who received standard treatment with IFRT alone.
However, there was no significant difference in overall survival (OS) between the treatment arms.
“This is the first successful randomized study ever to be conducted in early stage follicular lymphoma comparing standard therapy to standard therapy plus effective chemotherapy or immunochemotherapy,” said Michael MacManus, MBBCh, of Peter MacCallum Cancer Centre in Melbourne, Victoria, Australia.
“It shows that the initial treatment received by patients can significantly affect their long-term chance of staying free from disease. Moving forward, we are interested in determining whether there is a benefit in overall long-term survival for patients treated with the combination with further follow-up, and if there is any way to predict if a person will benefit from combined treatment based on analyses of blood or biopsy specimens.”
Dr MacManus and his colleagues studied 150 patients with stage I to II, low-grade FL who were enrolled in this trial between 2000 and 2012.
At randomization, the patients’ median age was 57, 52% were male, 75% had stage I disease, and 48% had PET staging.
Half of patients (n=75) were randomized to receive IFRT (30-36 Gy) alone, and half were randomized to IFRT (30-36 Gy) plus 6 cycles of CVP. From 2006 on, patients in the CVP arm received rituximab (R) as well (n=31).
Baseline characteristics were well-balanced between the treatment arms.
Efficacy
The median follow-up was 9.6 years (range, 3.1 to 15.8 years).
PFS was significantly better among patients randomized to receive CVP±R (hazard ratio [HR]=0.57; P=0.033). The estimated 10-year PFS rate was 41% in the IFRT arm and 59% in the CVP±R arm.
Patients randomized to receive CVP plus R (n=31) had significantly better PFS than patients randomized to receive IFRT alone (n=31) over the same time period (HR=0.26; P=0.045).
There were 10 deaths in the IRFT arm and 5 in the CVP±R arm, but there was no significant difference in OS between the arms (HR=0.62; P=0.40). The 10-year OS rate was 86% in the IFRT arm and 95% in the CVP±R arm.
There was no significant between-arm difference in transformation to aggressive lymphoma (P=0.1). Transformation occurred in 10 patients in the IFRT arm and 4 in the CVP±R arm.
Safety
There were 148 patients from both arms who ultimately received IFRT, and 69 patients who received CVP±R.
Grade 2 toxicities occurring in more than 10% of IFRT recipients included upper gastrointestinal (n=27; 18%), skin (n=21; 14%), and mucous membrane (n=19; 12%) toxicity. One IFRT recipient had grade 3 mucositis, and 1 had grade 4 esophageal/pharyngeal mucosal toxicity.
Grade 3 toxicities occurring in at least 2 patients in the CVP±R arm included neutropenia (n=10; 14%), infection (n=8; 12%), diarrhea (n=3; 4%), elevated gamma-glutamyl transferase (n=3; 4%), fatigue (n=3; 4%), and febrile neutropenia (n=3; 4%).
Three patients (4%) in the CVP±R arm had acute grade 3 neuropathy related to vincristine. Ten patients (14%) had grade 4 neutropenia.
The most common late toxicities for the entire patient cohort were salivary gland (n=8; 5%) and skin (n=4; 3%) toxicities.
Grade 3 lung and menopausal toxicities occurred in 1 patient each. Two patients had late grade 3 vincristine neuropathy. One patient who had grade 3 neuropathy during chemotherapy progressed to grade 4.
A multidrug regimen can improve upon involved-field radiotherapy (IFRT) in patients with early stage follicular lymphoma (FL), according to research published in the Journal of Clinical Oncology.
FL patients who received IFRT plus cyclophosphamide, vincristine, and prednisolone (CVP)—with or without rituximab—had a significant improvement in progression-free survival (PFS) compared to patients who received standard treatment with IFRT alone.
However, there was no significant difference in overall survival (OS) between the treatment arms.
“This is the first successful randomized study ever to be conducted in early stage follicular lymphoma comparing standard therapy to standard therapy plus effective chemotherapy or immunochemotherapy,” said Michael MacManus, MBBCh, of Peter MacCallum Cancer Centre in Melbourne, Victoria, Australia.
“It shows that the initial treatment received by patients can significantly affect their long-term chance of staying free from disease. Moving forward, we are interested in determining whether there is a benefit in overall long-term survival for patients treated with the combination with further follow-up, and if there is any way to predict if a person will benefit from combined treatment based on analyses of blood or biopsy specimens.”
Dr MacManus and his colleagues studied 150 patients with stage I to II, low-grade FL who were enrolled in this trial between 2000 and 2012.
At randomization, the patients’ median age was 57, 52% were male, 75% had stage I disease, and 48% had PET staging.
Half of patients (n=75) were randomized to receive IFRT (30-36 Gy) alone, and half were randomized to IFRT (30-36 Gy) plus 6 cycles of CVP. From 2006 on, patients in the CVP arm received rituximab (R) as well (n=31).
Baseline characteristics were well-balanced between the treatment arms.
Efficacy
The median follow-up was 9.6 years (range, 3.1 to 15.8 years).
PFS was significantly better among patients randomized to receive CVP±R (hazard ratio [HR]=0.57; P=0.033). The estimated 10-year PFS rate was 41% in the IFRT arm and 59% in the CVP±R arm.
Patients randomized to receive CVP plus R (n=31) had significantly better PFS than patients randomized to receive IFRT alone (n=31) over the same time period (HR=0.26; P=0.045).
There were 10 deaths in the IRFT arm and 5 in the CVP±R arm, but there was no significant difference in OS between the arms (HR=0.62; P=0.40). The 10-year OS rate was 86% in the IFRT arm and 95% in the CVP±R arm.
There was no significant between-arm difference in transformation to aggressive lymphoma (P=0.1). Transformation occurred in 10 patients in the IFRT arm and 4 in the CVP±R arm.
Safety
There were 148 patients from both arms who ultimately received IFRT, and 69 patients who received CVP±R.
Grade 2 toxicities occurring in more than 10% of IFRT recipients included upper gastrointestinal (n=27; 18%), skin (n=21; 14%), and mucous membrane (n=19; 12%) toxicity. One IFRT recipient had grade 3 mucositis, and 1 had grade 4 esophageal/pharyngeal mucosal toxicity.
Grade 3 toxicities occurring in at least 2 patients in the CVP±R arm included neutropenia (n=10; 14%), infection (n=8; 12%), diarrhea (n=3; 4%), elevated gamma-glutamyl transferase (n=3; 4%), fatigue (n=3; 4%), and febrile neutropenia (n=3; 4%).
Three patients (4%) in the CVP±R arm had acute grade 3 neuropathy related to vincristine. Ten patients (14%) had grade 4 neutropenia.
The most common late toxicities for the entire patient cohort were salivary gland (n=8; 5%) and skin (n=4; 3%) toxicities.
Grade 3 lung and menopausal toxicities occurred in 1 patient each. Two patients had late grade 3 vincristine neuropathy. One patient who had grade 3 neuropathy during chemotherapy progressed to grade 4.
Adult CCSs report financial hardships
Health-related financial hardship is common among adult survivors of childhood cancer, according to a study published in the Journal of the National Cancer Institute.
Researchers analyzed more than 2800 long-term childhood cancer survivors (CCSs) and found that 65% had financial challenges related to their cancer diagnosis.
“These findings suggest primary care doctors and oncologists should routinely screen childhood cancer survivors for possible financial hardship,” said I-Chan Huang, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
Specifically, Dr Huang recommends that healthcare providers routinely ask CCSs if they are unable to purchase medications, ever skip appointments for economic reasons, or worry about how to pay their medical bills.
For this study, Dr Huang and his colleagues analyzed data from 2811 CCSs. The subjects had a mean age of 31.8 (range, 18 to 65) and were a mean of 23.6 years from cancer diagnosis. Most (57.8%) had been diagnosed with hematologic malignancies, 32.0% with solid tumors, and 10.1% with central nervous system malignancies.
All subjects had been treated at St. Jude and enrolled in the St. Jude LIFE study. Participants return to St. Jude periodically for several days of clinical and functional assessments. Data for this study were collected during the CCSs’ first St. Jude LIFE evaluations.
Assessing hardship
The researchers measured 3 types of financial hardship—material, psychological, and coping/behavioral.
About 1 in 5 CCSs (22.4%) reported material financial hardship. In other words, their cancer had an impact on their financial situation.
More than half of CCSs (51.1%) reported psychological hardship—concern about their ability to pay for medical expenses.
And 33% of CCSs reported coping/behavioral hardship—an inability to see a doctor or go to the hospital due to finances.
Roughly 65% of CCSs reported at least 1 type of financial hardship.
All 3 types of hardship were significantly associated with somatization (all P<0.001), anxiety (all P<0.001), depression (all P<0.001), suicidal thoughts (all P<0.05), and difficulty in retirement planning (all P<0.001).
Furthermore, CCSs who reported financial hardship had significantly lower health-related quality of life (P<0.001 for all 3 domains), sensation abnormality (all P<0.001), pulmonary symptoms (all P<0.05), and cardiac symptoms (all P<0.05).
Predicting hardship
Intensive cancer treatment, chronic health conditions, second cancers, age at the time of study evaluation, education level, and annual household income were all significantly associated with a greater risk of financial hardship.
CCSs age 40 and older had an increased risk of psychological and coping/behavioral hardship (P<0.001 for both domains).
CCSs with an annual household income of less than $40,000 had an increased risk of material, psychological, and coping/behavioral hardship, compared to CCSs with an income of $80,000 or more (P<0.001 for all domains).
CCSs who did not obtain a high school diploma had an increased risk of material (P<0.001), psychological (P<0.01), and coping/behavioral hardship (P<0.001) compared to college graduates.
CCSs who received cancer treatments associated with a high-risk disease burden (vs low-risk) had an increased risk of material (P=0.01) and psychological (P=0.004) hardship.
Health conditions associated with material financial hardship included grade 2-4 myocardial infarction (P<0.001), peripheral neuropathy (P<0.001), subsequent neoplasm (P<0.001), seizure (P=0.007), reproductive disorders (P=0.01), stroke (P=0.02), amputation (P=0.02), upper gastrointestinal disease (P=0.04), and hearing loss (P=0.05).
Grade 2-4 myocardial infarction and reproductive disorders were significantly associated with psychological financial hardship (P=0.02 for both).
“Severe late effects that emerge early in life and disrupt education and training opportunities are a double hit for survivors,” Dr Huang said. “These health problems decrease the survivors’ earning mobility and financial security later in life. The phenomenon leaves them at risk for poor health and psychological outcomes compared to healthier survivors.”
Health-related financial hardship is common among adult survivors of childhood cancer, according to a study published in the Journal of the National Cancer Institute.
Researchers analyzed more than 2800 long-term childhood cancer survivors (CCSs) and found that 65% had financial challenges related to their cancer diagnosis.
“These findings suggest primary care doctors and oncologists should routinely screen childhood cancer survivors for possible financial hardship,” said I-Chan Huang, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
Specifically, Dr Huang recommends that healthcare providers routinely ask CCSs if they are unable to purchase medications, ever skip appointments for economic reasons, or worry about how to pay their medical bills.
For this study, Dr Huang and his colleagues analyzed data from 2811 CCSs. The subjects had a mean age of 31.8 (range, 18 to 65) and were a mean of 23.6 years from cancer diagnosis. Most (57.8%) had been diagnosed with hematologic malignancies, 32.0% with solid tumors, and 10.1% with central nervous system malignancies.
All subjects had been treated at St. Jude and enrolled in the St. Jude LIFE study. Participants return to St. Jude periodically for several days of clinical and functional assessments. Data for this study were collected during the CCSs’ first St. Jude LIFE evaluations.
Assessing hardship
The researchers measured 3 types of financial hardship—material, psychological, and coping/behavioral.
About 1 in 5 CCSs (22.4%) reported material financial hardship. In other words, their cancer had an impact on their financial situation.
More than half of CCSs (51.1%) reported psychological hardship—concern about their ability to pay for medical expenses.
And 33% of CCSs reported coping/behavioral hardship—an inability to see a doctor or go to the hospital due to finances.
Roughly 65% of CCSs reported at least 1 type of financial hardship.
All 3 types of hardship were significantly associated with somatization (all P<0.001), anxiety (all P<0.001), depression (all P<0.001), suicidal thoughts (all P<0.05), and difficulty in retirement planning (all P<0.001).
Furthermore, CCSs who reported financial hardship had significantly lower health-related quality of life (P<0.001 for all 3 domains), sensation abnormality (all P<0.001), pulmonary symptoms (all P<0.05), and cardiac symptoms (all P<0.05).
Predicting hardship
Intensive cancer treatment, chronic health conditions, second cancers, age at the time of study evaluation, education level, and annual household income were all significantly associated with a greater risk of financial hardship.
CCSs age 40 and older had an increased risk of psychological and coping/behavioral hardship (P<0.001 for both domains).
CCSs with an annual household income of less than $40,000 had an increased risk of material, psychological, and coping/behavioral hardship, compared to CCSs with an income of $80,000 or more (P<0.001 for all domains).
CCSs who did not obtain a high school diploma had an increased risk of material (P<0.001), psychological (P<0.01), and coping/behavioral hardship (P<0.001) compared to college graduates.
CCSs who received cancer treatments associated with a high-risk disease burden (vs low-risk) had an increased risk of material (P=0.01) and psychological (P=0.004) hardship.
Health conditions associated with material financial hardship included grade 2-4 myocardial infarction (P<0.001), peripheral neuropathy (P<0.001), subsequent neoplasm (P<0.001), seizure (P=0.007), reproductive disorders (P=0.01), stroke (P=0.02), amputation (P=0.02), upper gastrointestinal disease (P=0.04), and hearing loss (P=0.05).
Grade 2-4 myocardial infarction and reproductive disorders were significantly associated with psychological financial hardship (P=0.02 for both).
“Severe late effects that emerge early in life and disrupt education and training opportunities are a double hit for survivors,” Dr Huang said. “These health problems decrease the survivors’ earning mobility and financial security later in life. The phenomenon leaves them at risk for poor health and psychological outcomes compared to healthier survivors.”
Health-related financial hardship is common among adult survivors of childhood cancer, according to a study published in the Journal of the National Cancer Institute.
Researchers analyzed more than 2800 long-term childhood cancer survivors (CCSs) and found that 65% had financial challenges related to their cancer diagnosis.
“These findings suggest primary care doctors and oncologists should routinely screen childhood cancer survivors for possible financial hardship,” said I-Chan Huang, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
Specifically, Dr Huang recommends that healthcare providers routinely ask CCSs if they are unable to purchase medications, ever skip appointments for economic reasons, or worry about how to pay their medical bills.
For this study, Dr Huang and his colleagues analyzed data from 2811 CCSs. The subjects had a mean age of 31.8 (range, 18 to 65) and were a mean of 23.6 years from cancer diagnosis. Most (57.8%) had been diagnosed with hematologic malignancies, 32.0% with solid tumors, and 10.1% with central nervous system malignancies.
All subjects had been treated at St. Jude and enrolled in the St. Jude LIFE study. Participants return to St. Jude periodically for several days of clinical and functional assessments. Data for this study were collected during the CCSs’ first St. Jude LIFE evaluations.
Assessing hardship
The researchers measured 3 types of financial hardship—material, psychological, and coping/behavioral.
About 1 in 5 CCSs (22.4%) reported material financial hardship. In other words, their cancer had an impact on their financial situation.
More than half of CCSs (51.1%) reported psychological hardship—concern about their ability to pay for medical expenses.
And 33% of CCSs reported coping/behavioral hardship—an inability to see a doctor or go to the hospital due to finances.
Roughly 65% of CCSs reported at least 1 type of financial hardship.
All 3 types of hardship were significantly associated with somatization (all P<0.001), anxiety (all P<0.001), depression (all P<0.001), suicidal thoughts (all P<0.05), and difficulty in retirement planning (all P<0.001).
Furthermore, CCSs who reported financial hardship had significantly lower health-related quality of life (P<0.001 for all 3 domains), sensation abnormality (all P<0.001), pulmonary symptoms (all P<0.05), and cardiac symptoms (all P<0.05).
Predicting hardship
Intensive cancer treatment, chronic health conditions, second cancers, age at the time of study evaluation, education level, and annual household income were all significantly associated with a greater risk of financial hardship.
CCSs age 40 and older had an increased risk of psychological and coping/behavioral hardship (P<0.001 for both domains).
CCSs with an annual household income of less than $40,000 had an increased risk of material, psychological, and coping/behavioral hardship, compared to CCSs with an income of $80,000 or more (P<0.001 for all domains).
CCSs who did not obtain a high school diploma had an increased risk of material (P<0.001), psychological (P<0.01), and coping/behavioral hardship (P<0.001) compared to college graduates.
CCSs who received cancer treatments associated with a high-risk disease burden (vs low-risk) had an increased risk of material (P=0.01) and psychological (P=0.004) hardship.
Health conditions associated with material financial hardship included grade 2-4 myocardial infarction (P<0.001), peripheral neuropathy (P<0.001), subsequent neoplasm (P<0.001), seizure (P=0.007), reproductive disorders (P=0.01), stroke (P=0.02), amputation (P=0.02), upper gastrointestinal disease (P=0.04), and hearing loss (P=0.05).
Grade 2-4 myocardial infarction and reproductive disorders were significantly associated with psychological financial hardship (P=0.02 for both).
“Severe late effects that emerge early in life and disrupt education and training opportunities are a double hit for survivors,” Dr Huang said. “These health problems decrease the survivors’ earning mobility and financial security later in life. The phenomenon leaves them at risk for poor health and psychological outcomes compared to healthier survivors.”
Inhibitor receives breakthrough designation for AML
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to quizartinib, an investigational FLT3 inhibitor, for the treatment of adults with relapsed/refractory FLT3-ITD acute myeloid leukemia (AML).
The FDA granted quizartinib breakthrough designation based on results from the phase 3 QuANTUM-R study, which were presented at the 23rd Congress of the European Hematology Association in June.
QuANTUM-R enrolled adults with FLT3-ITD AML (at least 3% FLT3-ITD allelic ratio) who had refractory disease or had relapsed within 6 months of their first complete remission.
Patients were randomized to receive once-daily treatment with quizartinib (n=245) or a salvage chemotherapy regimen (n=122)—low-dose cytarabine (LoDAC, n=29); combination mitoxantrone, etoposide, and cytarabine (MEC, n=40); or combination fludarabine, cytarabine, and idarubicin (FLAG-IDA, n=53).
Responders could proceed to hematopoietic stem cell transplant (HSCT), and those in the quizartinib arm could resume quizartinib after HSCT. Thirty-two percent of quizartinib-treated patients and 12% of the chemotherapy group went on to HSCT.
The median follow-up was 23.5 months. The efficacy results include all randomized patients.
The overall response rate was 69% in the quizartinib arm and 30% in the chemotherapy arm. The complete response (CR) rate was 4% and 1%, respectively; the rate of CR with incomplete platelet recovery was 4% and 0%, respectively; and the rate of CR with incomplete hematologic recovery was 40% and 26%, respectively.
The median overall survival was 6.2 months in the quizartinib arm and 4.7 months in the chemotherapy arm (hazard ratio=0.76, P=0.0177). The 1-year overall survival rate was 27% and 20%, respectively.
The median event-free survival was 6.0 weeks in the quizartinib arm and 3.7 weeks in the chemotherapy arm (hazard ratio=0.90, P=0.1071).
The safety results include only patients who received their assigned treatment—241 patients who received quizartinib and 94 who received salvage chemotherapy (22 on LoDAC, 25 on MEC, and 47 on FLAG-IDA).
Grade 3 or higher hematologic treatment-emergent adverse events occurring in at least 5% of patients (in the quizartinib and chemotherapy groups, respectively) included thrombocytopenia (35% and 34%), anemia (30% and 29%), neutropenia (32% and 25%), febrile neutropenia (31% and 21%), and leukopenia (17% and 16%).
Grade 3 or higher nonhematologic treatment-emergent adverse events occurring in at least 5% of patients (in the quizartinib and chemotherapy groups, respectively) included fatigue (8% and 1%), hypokalemia (12% and 9%), sepsis/septic shock (16% and 18%), dyspnea (5% for both), hypophosphatemia (5% for both), and pneumonia (12% and 9%).
Three percent of patients in the quizartinib arm had grade 3 QTcF prolongation, and 2 patients discontinued quizartinib due to QTcF prolongation.
About breakthrough designation
Breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
The designation entitles the company developing a therapy to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need.
Other designations for quizartinib
In addition to breakthrough therapy designation, quizartinib has fast track and orphan drug designations from the FDA.
The FDA’s fast track development program is designed to expedite clinical development and submission of applications for products with the potential to treat serious or life-threatening conditions and address unmet medical needs.
Fast track designation facilitates frequent interactions with the FDA review team, including meetings to discuss the product’s development plan and written communications about issues such as trial design and use of biomarkers.
Products that receive fast track designation may be eligible for accelerated approval and priority review if relevant criteria are met. Such products may also be eligible for rolling review, which allows a developer to submit individual sections of a product’s application for review as they are ready, rather than waiting until all sections are complete.
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to quizartinib, an investigational FLT3 inhibitor, for the treatment of adults with relapsed/refractory FLT3-ITD acute myeloid leukemia (AML).
The FDA granted quizartinib breakthrough designation based on results from the phase 3 QuANTUM-R study, which were presented at the 23rd Congress of the European Hematology Association in June.
QuANTUM-R enrolled adults with FLT3-ITD AML (at least 3% FLT3-ITD allelic ratio) who had refractory disease or had relapsed within 6 months of their first complete remission.
Patients were randomized to receive once-daily treatment with quizartinib (n=245) or a salvage chemotherapy regimen (n=122)—low-dose cytarabine (LoDAC, n=29); combination mitoxantrone, etoposide, and cytarabine (MEC, n=40); or combination fludarabine, cytarabine, and idarubicin (FLAG-IDA, n=53).
Responders could proceed to hematopoietic stem cell transplant (HSCT), and those in the quizartinib arm could resume quizartinib after HSCT. Thirty-two percent of quizartinib-treated patients and 12% of the chemotherapy group went on to HSCT.
The median follow-up was 23.5 months. The efficacy results include all randomized patients.
The overall response rate was 69% in the quizartinib arm and 30% in the chemotherapy arm. The complete response (CR) rate was 4% and 1%, respectively; the rate of CR with incomplete platelet recovery was 4% and 0%, respectively; and the rate of CR with incomplete hematologic recovery was 40% and 26%, respectively.
The median overall survival was 6.2 months in the quizartinib arm and 4.7 months in the chemotherapy arm (hazard ratio=0.76, P=0.0177). The 1-year overall survival rate was 27% and 20%, respectively.
The median event-free survival was 6.0 weeks in the quizartinib arm and 3.7 weeks in the chemotherapy arm (hazard ratio=0.90, P=0.1071).
The safety results include only patients who received their assigned treatment—241 patients who received quizartinib and 94 who received salvage chemotherapy (22 on LoDAC, 25 on MEC, and 47 on FLAG-IDA).
Grade 3 or higher hematologic treatment-emergent adverse events occurring in at least 5% of patients (in the quizartinib and chemotherapy groups, respectively) included thrombocytopenia (35% and 34%), anemia (30% and 29%), neutropenia (32% and 25%), febrile neutropenia (31% and 21%), and leukopenia (17% and 16%).
Grade 3 or higher nonhematologic treatment-emergent adverse events occurring in at least 5% of patients (in the quizartinib and chemotherapy groups, respectively) included fatigue (8% and 1%), hypokalemia (12% and 9%), sepsis/septic shock (16% and 18%), dyspnea (5% for both), hypophosphatemia (5% for both), and pneumonia (12% and 9%).
Three percent of patients in the quizartinib arm had grade 3 QTcF prolongation, and 2 patients discontinued quizartinib due to QTcF prolongation.
About breakthrough designation
Breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
The designation entitles the company developing a therapy to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need.
Other designations for quizartinib
In addition to breakthrough therapy designation, quizartinib has fast track and orphan drug designations from the FDA.
The FDA’s fast track development program is designed to expedite clinical development and submission of applications for products with the potential to treat serious or life-threatening conditions and address unmet medical needs.
Fast track designation facilitates frequent interactions with the FDA review team, including meetings to discuss the product’s development plan and written communications about issues such as trial design and use of biomarkers.
Products that receive fast track designation may be eligible for accelerated approval and priority review if relevant criteria are met. Such products may also be eligible for rolling review, which allows a developer to submit individual sections of a product’s application for review as they are ready, rather than waiting until all sections are complete.
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to quizartinib, an investigational FLT3 inhibitor, for the treatment of adults with relapsed/refractory FLT3-ITD acute myeloid leukemia (AML).
The FDA granted quizartinib breakthrough designation based on results from the phase 3 QuANTUM-R study, which were presented at the 23rd Congress of the European Hematology Association in June.
QuANTUM-R enrolled adults with FLT3-ITD AML (at least 3% FLT3-ITD allelic ratio) who had refractory disease or had relapsed within 6 months of their first complete remission.
Patients were randomized to receive once-daily treatment with quizartinib (n=245) or a salvage chemotherapy regimen (n=122)—low-dose cytarabine (LoDAC, n=29); combination mitoxantrone, etoposide, and cytarabine (MEC, n=40); or combination fludarabine, cytarabine, and idarubicin (FLAG-IDA, n=53).
Responders could proceed to hematopoietic stem cell transplant (HSCT), and those in the quizartinib arm could resume quizartinib after HSCT. Thirty-two percent of quizartinib-treated patients and 12% of the chemotherapy group went on to HSCT.
The median follow-up was 23.5 months. The efficacy results include all randomized patients.
The overall response rate was 69% in the quizartinib arm and 30% in the chemotherapy arm. The complete response (CR) rate was 4% and 1%, respectively; the rate of CR with incomplete platelet recovery was 4% and 0%, respectively; and the rate of CR with incomplete hematologic recovery was 40% and 26%, respectively.
The median overall survival was 6.2 months in the quizartinib arm and 4.7 months in the chemotherapy arm (hazard ratio=0.76, P=0.0177). The 1-year overall survival rate was 27% and 20%, respectively.
The median event-free survival was 6.0 weeks in the quizartinib arm and 3.7 weeks in the chemotherapy arm (hazard ratio=0.90, P=0.1071).
The safety results include only patients who received their assigned treatment—241 patients who received quizartinib and 94 who received salvage chemotherapy (22 on LoDAC, 25 on MEC, and 47 on FLAG-IDA).
Grade 3 or higher hematologic treatment-emergent adverse events occurring in at least 5% of patients (in the quizartinib and chemotherapy groups, respectively) included thrombocytopenia (35% and 34%), anemia (30% and 29%), neutropenia (32% and 25%), febrile neutropenia (31% and 21%), and leukopenia (17% and 16%).
Grade 3 or higher nonhematologic treatment-emergent adverse events occurring in at least 5% of patients (in the quizartinib and chemotherapy groups, respectively) included fatigue (8% and 1%), hypokalemia (12% and 9%), sepsis/septic shock (16% and 18%), dyspnea (5% for both), hypophosphatemia (5% for both), and pneumonia (12% and 9%).
Three percent of patients in the quizartinib arm had grade 3 QTcF prolongation, and 2 patients discontinued quizartinib due to QTcF prolongation.
About breakthrough designation
Breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
The designation entitles the company developing a therapy to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need.
Other designations for quizartinib
In addition to breakthrough therapy designation, quizartinib has fast track and orphan drug designations from the FDA.
The FDA’s fast track development program is designed to expedite clinical development and submission of applications for products with the potential to treat serious or life-threatening conditions and address unmet medical needs.
Fast track designation facilitates frequent interactions with the FDA review team, including meetings to discuss the product’s development plan and written communications about issues such as trial design and use of biomarkers.
Products that receive fast track designation may be eligible for accelerated approval and priority review if relevant criteria are met. Such products may also be eligible for rolling review, which allows a developer to submit individual sections of a product’s application for review as they are ready, rather than waiting until all sections are complete.
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The Mother of All Skin Problems
Each time this 32-year-old woman has a baby—she’s had four to date—she notices that sections of her face darken. Early on, she observed a pattern in which the coming of winter coincided with a lightening of these affected areas—but now the effect lasts year-round, with progressive darkening. She has not tried any products (OTC or prescription) for this problem.
Growing up in the South, the patient and her family spent most summers boating, swimming, and fishing. Her use of sunscreen was sporadic, but she would tan easily regardless.
Her health is good, aside from a 15-year history of smoking.
EXAMINATION
There is excessive hyperpigmentation (brown) on the patient’s face. It follows a mask-like pattern, including her maxilla and the periphery of her face.
Elsewhere, there is abundant evidence of excessive sun exposure, with focal hyperpigmentation and telangiectasias on her arms. She has type IV skin, consistent with her Native American ancestry.
What is the diagnosis?
DISCUSSION
Melasma, also known as chloasma and dubbed the “mask of pregnancy,” is an extremely common problem that results from a combination of naturally dark skin, lots of sun exposure, and increased levels of estrogenic hormones. The latter can result from pregnancy or from oral contraceptive or estrogen replacement therapy use. Another precipitating factor is thyroid disease, certain types of which lead to an increase in melanocytic stimulating hormone.
Melasma, as one might expect, is seen almost exclusively in women, though a rare male is affected. It is especially common among Latina, Native American, and African-American women, whose melanocytes are especially able to produce pigment.
There are several treatments for melasma, none of them perfect, including tretinoin, azelaic acid, chemical peels, dermabrasion, and lasers. The most common treatment is hydroquinone cream, available in the US in both OTC (2%) and prescription (4%) strengths. However, hydroquinone is available OTC in stronger formulations (15% to 20%) in many Central and South American countries; unfortunately, many women who obtain and use these products experience exogenous ochronosis—a worsening or even precipitation of melasma, resulting from excessive production of tyrosinase.
Any treatment must be used in conjunction with rigorous sunscreen application. A full-spectrum product, with titanium dioxide and zinc oxide as the only active ingredients, must be used, because chemical-laden sunscreens don’t do as good a job covering UVA, UVB, and visible light. Convincing women who are unaccustomed to needing sunscreen to use it religiously is part of what makes treating melasma difficult.
The differential for melasma includes postinflammatory hyperpigmentation (eg, following an episode of contact dermatitis) and simple solar lentigines.
This patient was treated with hydroquinone 4% cream bid, plus sunscreen. She was also given information about other treatment options, such as laser and dermabrasion.
TAKE-HOME LEARNING POINTS
- Melasma, also known at chloasma, is quite common, especially among women with darker skin who live in sunny parts of the world.
- It results from a combination of dark skin, an increased level of estrogenic hormones (eg, with pregnancy, birth control pills, or estrogen replacement therapy), and excessive exposure to UV light.
- While hydroquinone cream can be an effective treatment, the maximum strength should be 4%; overuse of stronger concentrations (available in other countries, such as Mexico), can actually cause melasma to worsen.
Each time this 32-year-old woman has a baby—she’s had four to date—she notices that sections of her face darken. Early on, she observed a pattern in which the coming of winter coincided with a lightening of these affected areas—but now the effect lasts year-round, with progressive darkening. She has not tried any products (OTC or prescription) for this problem.
Growing up in the South, the patient and her family spent most summers boating, swimming, and fishing. Her use of sunscreen was sporadic, but she would tan easily regardless.
Her health is good, aside from a 15-year history of smoking.
EXAMINATION
There is excessive hyperpigmentation (brown) on the patient’s face. It follows a mask-like pattern, including her maxilla and the periphery of her face.

Elsewhere, there is abundant evidence of excessive sun exposure, with focal hyperpigmentation and telangiectasias on her arms. She has type IV skin, consistent with her Native American ancestry.
What is the diagnosis?
DISCUSSION
Melasma, also known as chloasma and dubbed the “mask of pregnancy,” is an extremely common problem that results from a combination of naturally dark skin, lots of sun exposure, and increased levels of estrogenic hormones. The latter can result from pregnancy or from oral contraceptive or estrogen replacement therapy use. Another precipitating factor is thyroid disease, certain types of which lead to an increase in melanocytic stimulating hormone.
Melasma, as one might expect, is seen almost exclusively in women, though a rare male is affected. It is especially common among Latina, Native American, and African-American women, whose melanocytes are especially able to produce pigment.
There are several treatments for melasma, none of them perfect, including tretinoin, azelaic acid, chemical peels, dermabrasion, and lasers. The most common treatment is hydroquinone cream, available in the US in both OTC (2%) and prescription (4%) strengths. However, hydroquinone is available OTC in stronger formulations (15% to 20%) in many Central and South American countries; unfortunately, many women who obtain and use these products experience exogenous ochronosis—a worsening or even precipitation of melasma, resulting from excessive production of tyrosinase.
Any treatment must be used in conjunction with rigorous sunscreen application. A full-spectrum product, with titanium dioxide and zinc oxide as the only active ingredients, must be used, because chemical-laden sunscreens don’t do as good a job covering UVA, UVB, and visible light. Convincing women who are unaccustomed to needing sunscreen to use it religiously is part of what makes treating melasma difficult.
The differential for melasma includes postinflammatory hyperpigmentation (eg, following an episode of contact dermatitis) and simple solar lentigines.
This patient was treated with hydroquinone 4% cream bid, plus sunscreen. She was also given information about other treatment options, such as laser and dermabrasion.
TAKE-HOME LEARNING POINTS
- Melasma, also known at chloasma, is quite common, especially among women with darker skin who live in sunny parts of the world.
- It results from a combination of dark skin, an increased level of estrogenic hormones (eg, with pregnancy, birth control pills, or estrogen replacement therapy), and excessive exposure to UV light.
- While hydroquinone cream can be an effective treatment, the maximum strength should be 4%; overuse of stronger concentrations (available in other countries, such as Mexico), can actually cause melasma to worsen.
Each time this 32-year-old woman has a baby—she’s had four to date—she notices that sections of her face darken. Early on, she observed a pattern in which the coming of winter coincided with a lightening of these affected areas—but now the effect lasts year-round, with progressive darkening. She has not tried any products (OTC or prescription) for this problem.
Growing up in the South, the patient and her family spent most summers boating, swimming, and fishing. Her use of sunscreen was sporadic, but she would tan easily regardless.
Her health is good, aside from a 15-year history of smoking.
EXAMINATION
There is excessive hyperpigmentation (brown) on the patient’s face. It follows a mask-like pattern, including her maxilla and the periphery of her face.

Elsewhere, there is abundant evidence of excessive sun exposure, with focal hyperpigmentation and telangiectasias on her arms. She has type IV skin, consistent with her Native American ancestry.
What is the diagnosis?
DISCUSSION
Melasma, also known as chloasma and dubbed the “mask of pregnancy,” is an extremely common problem that results from a combination of naturally dark skin, lots of sun exposure, and increased levels of estrogenic hormones. The latter can result from pregnancy or from oral contraceptive or estrogen replacement therapy use. Another precipitating factor is thyroid disease, certain types of which lead to an increase in melanocytic stimulating hormone.
Melasma, as one might expect, is seen almost exclusively in women, though a rare male is affected. It is especially common among Latina, Native American, and African-American women, whose melanocytes are especially able to produce pigment.
There are several treatments for melasma, none of them perfect, including tretinoin, azelaic acid, chemical peels, dermabrasion, and lasers. The most common treatment is hydroquinone cream, available in the US in both OTC (2%) and prescription (4%) strengths. However, hydroquinone is available OTC in stronger formulations (15% to 20%) in many Central and South American countries; unfortunately, many women who obtain and use these products experience exogenous ochronosis—a worsening or even precipitation of melasma, resulting from excessive production of tyrosinase.
Any treatment must be used in conjunction with rigorous sunscreen application. A full-spectrum product, with titanium dioxide and zinc oxide as the only active ingredients, must be used, because chemical-laden sunscreens don’t do as good a job covering UVA, UVB, and visible light. Convincing women who are unaccustomed to needing sunscreen to use it religiously is part of what makes treating melasma difficult.
The differential for melasma includes postinflammatory hyperpigmentation (eg, following an episode of contact dermatitis) and simple solar lentigines.
This patient was treated with hydroquinone 4% cream bid, plus sunscreen. She was also given information about other treatment options, such as laser and dermabrasion.
TAKE-HOME LEARNING POINTS
- Melasma, also known at chloasma, is quite common, especially among women with darker skin who live in sunny parts of the world.
- It results from a combination of dark skin, an increased level of estrogenic hormones (eg, with pregnancy, birth control pills, or estrogen replacement therapy), and excessive exposure to UV light.
- While hydroquinone cream can be an effective treatment, the maximum strength should be 4%; overuse of stronger concentrations (available in other countries, such as Mexico), can actually cause melasma to worsen.
Education: The Mediating Factor in Gun Violence?
I read “Up In Arms About Gun Violence” in the May/June issue of Clinician Reviews with great interest. My response is drawn from research, professional experience, and three particularly formative experiences:
- The birth of a child with learning and behavioral differences requiring an individualized education program and temporary placement in an alternative school.
- Co-leading a parent workshop for my clinical Capstone project.
- A nonclinical position at a public alternative school for children with emotional and behavioral disorders.
In nursing, as in all human services, we strive to be holistic, drawing from science, research, ethics, human development, sociology, anthropology, etc, to function in our roles. Many in nursing have come to value and function well in multidisciplinary teams for the management of individuals with complex needs.
What I have learned in my life—a fact supported by my professional preparation—is that the cycle of poverty, poor health, and quality of life are greatly influenced by education. During my pediatrics rotation and clinical, I learned you must consider family to be the vehicle by which the child may benefit from your clinical services. Therefore, you must reach beyond your setting in an effective and consistent way to engage, connect the home, and then follow up for response. My colleagues are aware of the need to document and the processes currently available for health, social service, and school professionals to report concerns. Too often, obstacles and other influences stand in the way of our ethical duty as professionals; we must find a way to overcome them, both individually and as a group.
We have processes for adolescents aging out of foster care and the developmentally delayed transitioning to sheltered work; can these be expanded?
My experience in a public school district’s alternative school for children in grades 6-12 opened my eyes to the need for the public to understand how these schools function. I likened these schools to the intensive care unit in the acute care setting. Smaller population, high needs, high cost, and dedicated staff but high burnout and turnover. Children in my school came from foster care or struggling homes and often had experienced trauma. Underfunded school districts cut nurses and social workers, cannot attract professional support staff, and choose to distribute money and efforts on the general population in neighborhood schools.
My “professional health care” mindset was embedded in an environment of wounded children, amazing educators, and support staff; powerless to do our best, we held on. But it is not enough to just hold on, sequestering at-risk children out of sight. We can do better.
Continue to: Some ideas to turn this belief into reality
Some ideas to turn this belief into reality:
- Create position statements and recommendations for our licensing and professional certifying bodies to share with their membership.
- Include mandates for communicating those recommendations during certification and relicensing.
- Employ district staff or contracted consulting medical and psychologic evaluators for review of public school students with health and behavioral diagnoses.
- Partner better with schools from the community health, pediatrician, behavioral health, and primary care settings. Kennedy Krieger Institute has partnered with schools in Baltimore—can this be replicated elsewhere?
- Every child enters school with a need for health clearance and a “Guardian Permission Contact Card.” Perhaps we can expand this to develop a process for results of routine assessments to be shared, for the safety and welfare of the child.
- Teach individuals to cope effectively with the anxiety and distress fed by constant exposure to the Internet and social media. This can—and should—be done through multiple venues: religious and social groups and the workplace.
- Inform the public with PowerPoint presentations and seminars, advertising what to look for and where to get help for concerning behaviors; again, this can be done through multiple portals. This, I believe, may also help defuse tensions between parents and schools surrounding children with health and educational differences.
- Have professional health associations deliver a cohesive message to our legislators for gun control, funding for research, and mental and public health.
Dr. Onieal, you are correct in saying that this epidemic of mass shootings is a public health issue. It is a threat as serious as HIV, cancer, lead poisoning, hypertension, and diabetes. We must find a way to stop this epidemic.
Diane Page, RN, MSN, ARNP-C
Sanford, FL
I read “Up In Arms About Gun Violence” in the May/June issue of Clinician Reviews with great interest. My response is drawn from research, professional experience, and three particularly formative experiences:
- The birth of a child with learning and behavioral differences requiring an individualized education program and temporary placement in an alternative school.
- Co-leading a parent workshop for my clinical Capstone project.
- A nonclinical position at a public alternative school for children with emotional and behavioral disorders.
In nursing, as in all human services, we strive to be holistic, drawing from science, research, ethics, human development, sociology, anthropology, etc, to function in our roles. Many in nursing have come to value and function well in multidisciplinary teams for the management of individuals with complex needs.
What I have learned in my life—a fact supported by my professional preparation—is that the cycle of poverty, poor health, and quality of life are greatly influenced by education. During my pediatrics rotation and clinical, I learned you must consider family to be the vehicle by which the child may benefit from your clinical services. Therefore, you must reach beyond your setting in an effective and consistent way to engage, connect the home, and then follow up for response. My colleagues are aware of the need to document and the processes currently available for health, social service, and school professionals to report concerns. Too often, obstacles and other influences stand in the way of our ethical duty as professionals; we must find a way to overcome them, both individually and as a group.
We have processes for adolescents aging out of foster care and the developmentally delayed transitioning to sheltered work; can these be expanded?
My experience in a public school district’s alternative school for children in grades 6-12 opened my eyes to the need for the public to understand how these schools function. I likened these schools to the intensive care unit in the acute care setting. Smaller population, high needs, high cost, and dedicated staff but high burnout and turnover. Children in my school came from foster care or struggling homes and often had experienced trauma. Underfunded school districts cut nurses and social workers, cannot attract professional support staff, and choose to distribute money and efforts on the general population in neighborhood schools.
My “professional health care” mindset was embedded in an environment of wounded children, amazing educators, and support staff; powerless to do our best, we held on. But it is not enough to just hold on, sequestering at-risk children out of sight. We can do better.
Continue to: Some ideas to turn this belief into reality
Some ideas to turn this belief into reality:
- Create position statements and recommendations for our licensing and professional certifying bodies to share with their membership.
- Include mandates for communicating those recommendations during certification and relicensing.
- Employ district staff or contracted consulting medical and psychologic evaluators for review of public school students with health and behavioral diagnoses.
- Partner better with schools from the community health, pediatrician, behavioral health, and primary care settings. Kennedy Krieger Institute has partnered with schools in Baltimore—can this be replicated elsewhere?
- Every child enters school with a need for health clearance and a “Guardian Permission Contact Card.” Perhaps we can expand this to develop a process for results of routine assessments to be shared, for the safety and welfare of the child.
- Teach individuals to cope effectively with the anxiety and distress fed by constant exposure to the Internet and social media. This can—and should—be done through multiple venues: religious and social groups and the workplace.
- Inform the public with PowerPoint presentations and seminars, advertising what to look for and where to get help for concerning behaviors; again, this can be done through multiple portals. This, I believe, may also help defuse tensions between parents and schools surrounding children with health and educational differences.
- Have professional health associations deliver a cohesive message to our legislators for gun control, funding for research, and mental and public health.
Dr. Onieal, you are correct in saying that this epidemic of mass shootings is a public health issue. It is a threat as serious as HIV, cancer, lead poisoning, hypertension, and diabetes. We must find a way to stop this epidemic.
Diane Page, RN, MSN, ARNP-C
Sanford, FL
I read “Up In Arms About Gun Violence” in the May/June issue of Clinician Reviews with great interest. My response is drawn from research, professional experience, and three particularly formative experiences:
- The birth of a child with learning and behavioral differences requiring an individualized education program and temporary placement in an alternative school.
- Co-leading a parent workshop for my clinical Capstone project.
- A nonclinical position at a public alternative school for children with emotional and behavioral disorders.
In nursing, as in all human services, we strive to be holistic, drawing from science, research, ethics, human development, sociology, anthropology, etc, to function in our roles. Many in nursing have come to value and function well in multidisciplinary teams for the management of individuals with complex needs.
What I have learned in my life—a fact supported by my professional preparation—is that the cycle of poverty, poor health, and quality of life are greatly influenced by education. During my pediatrics rotation and clinical, I learned you must consider family to be the vehicle by which the child may benefit from your clinical services. Therefore, you must reach beyond your setting in an effective and consistent way to engage, connect the home, and then follow up for response. My colleagues are aware of the need to document and the processes currently available for health, social service, and school professionals to report concerns. Too often, obstacles and other influences stand in the way of our ethical duty as professionals; we must find a way to overcome them, both individually and as a group.
We have processes for adolescents aging out of foster care and the developmentally delayed transitioning to sheltered work; can these be expanded?
My experience in a public school district’s alternative school for children in grades 6-12 opened my eyes to the need for the public to understand how these schools function. I likened these schools to the intensive care unit in the acute care setting. Smaller population, high needs, high cost, and dedicated staff but high burnout and turnover. Children in my school came from foster care or struggling homes and often had experienced trauma. Underfunded school districts cut nurses and social workers, cannot attract professional support staff, and choose to distribute money and efforts on the general population in neighborhood schools.
My “professional health care” mindset was embedded in an environment of wounded children, amazing educators, and support staff; powerless to do our best, we held on. But it is not enough to just hold on, sequestering at-risk children out of sight. We can do better.
Continue to: Some ideas to turn this belief into reality
Some ideas to turn this belief into reality:
- Create position statements and recommendations for our licensing and professional certifying bodies to share with their membership.
- Include mandates for communicating those recommendations during certification and relicensing.
- Employ district staff or contracted consulting medical and psychologic evaluators for review of public school students with health and behavioral diagnoses.
- Partner better with schools from the community health, pediatrician, behavioral health, and primary care settings. Kennedy Krieger Institute has partnered with schools in Baltimore—can this be replicated elsewhere?
- Every child enters school with a need for health clearance and a “Guardian Permission Contact Card.” Perhaps we can expand this to develop a process for results of routine assessments to be shared, for the safety and welfare of the child.
- Teach individuals to cope effectively with the anxiety and distress fed by constant exposure to the Internet and social media. This can—and should—be done through multiple venues: religious and social groups and the workplace.
- Inform the public with PowerPoint presentations and seminars, advertising what to look for and where to get help for concerning behaviors; again, this can be done through multiple portals. This, I believe, may also help defuse tensions between parents and schools surrounding children with health and educational differences.
- Have professional health associations deliver a cohesive message to our legislators for gun control, funding for research, and mental and public health.
Dr. Onieal, you are correct in saying that this epidemic of mass shootings is a public health issue. It is a threat as serious as HIV, cancer, lead poisoning, hypertension, and diabetes. We must find a way to stop this epidemic.
Diane Page, RN, MSN, ARNP-C
Sanford, FL