User login
C. difficile linked to surgery risk in pediatric Crohn’s
In pediatric Crohn’s disease, a Clostridioides difficile infection detected within the first year after diagnosis is associated with a shorter time to first bowel resection surgery, according to a study that included both a retrospective and prospective analysis. The researchers also found evidence that changes in methionine biosynthesis and depletion of beneficial bacteria may contribute to risk of surgery.
C. difficile infection (CDI) disproportionately affects individuals with inflammatory bowel disease (IBD). Pediatric IBD patients have a 34% risk of recurrent CDI infection, compared with 7.5% in the general population. Previous research found that adults with ulcerative colitis and CDI are at more risk of colectomy, but the finding has not been replicated in children.
In a study published in Inflammatory Bowel Diseases, researchers led by Jennifer Hellmann and Lee Denson of the University of Cincinnati conducted a single-center retrospective analysis of 75 pediatric Crohn’s disease patients. They also conducted a prospective study of 70 pediatric Crohn’s disease patients, using shotgun metagenome sequencing to examine the relationship between microbiota composition and C. difficile carriage or surgery history.
Nineteen percent of patients tested positive for C. difficile. Use of antibiotics was associated with C. difficile (odds ratio, 7.9; P = .02). Of patients who underwent C. difficile testing in the first year, 23 went on to have surgery: 21% who were C. difficile negative required surgery, compared with 67% of those who were positive (hazard ratio, 4.4; P = .0003). The mean time to surgery was 527 days for C. difficile–positive patients and 1,268 days for those who were negative.
A multivariate regression analysis on 54 patients with complete data sets showed that the presence of C. difficile was associated with increased risk of surgery (OR, 16.2; P = .0006). When the analysis was run on all 73 patients, using null value for missing data, the results were similar (OR, 9.17; P = .008).
Shotgun sequencing found that 47 of 114 bacterial species that were associated with the presence of C. difficile were also associated with prior surgery for Crohn’s disease. Species included some that may play a role in mucosal homeostasis, such as Bifidobacterium breve and several Alistipes and Ruminococcus species. That suggests that a reduction in the numbers of these taxa may be associated with C. difficile presence and surgical risk.
The researchers also found that methionine synthesis pathways were depressed in C. difficile–positive and surgery patients. Methionine may bolster antioxidant capacity and improve villus morphology. IBD patients with dysbiosis and those experiencing Crohn’s disease exacerbations have been shown to have decreased methionine pathway activity, suggesting methionine biosynthesis changes have clinical relevance.
The study was funded by the National Institutes of Health.
SOURCE: Hellmann J et al. Inflamm Bowel Dis. 2020. doi: 10.1093/ibd/izz263.
In pediatric Crohn’s disease, a Clostridioides difficile infection detected within the first year after diagnosis is associated with a shorter time to first bowel resection surgery, according to a study that included both a retrospective and prospective analysis. The researchers also found evidence that changes in methionine biosynthesis and depletion of beneficial bacteria may contribute to risk of surgery.
C. difficile infection (CDI) disproportionately affects individuals with inflammatory bowel disease (IBD). Pediatric IBD patients have a 34% risk of recurrent CDI infection, compared with 7.5% in the general population. Previous research found that adults with ulcerative colitis and CDI are at more risk of colectomy, but the finding has not been replicated in children.
In a study published in Inflammatory Bowel Diseases, researchers led by Jennifer Hellmann and Lee Denson of the University of Cincinnati conducted a single-center retrospective analysis of 75 pediatric Crohn’s disease patients. They also conducted a prospective study of 70 pediatric Crohn’s disease patients, using shotgun metagenome sequencing to examine the relationship between microbiota composition and C. difficile carriage or surgery history.
Nineteen percent of patients tested positive for C. difficile. Use of antibiotics was associated with C. difficile (odds ratio, 7.9; P = .02). Of patients who underwent C. difficile testing in the first year, 23 went on to have surgery: 21% who were C. difficile negative required surgery, compared with 67% of those who were positive (hazard ratio, 4.4; P = .0003). The mean time to surgery was 527 days for C. difficile–positive patients and 1,268 days for those who were negative.
A multivariate regression analysis on 54 patients with complete data sets showed that the presence of C. difficile was associated with increased risk of surgery (OR, 16.2; P = .0006). When the analysis was run on all 73 patients, using null value for missing data, the results were similar (OR, 9.17; P = .008).
Shotgun sequencing found that 47 of 114 bacterial species that were associated with the presence of C. difficile were also associated with prior surgery for Crohn’s disease. Species included some that may play a role in mucosal homeostasis, such as Bifidobacterium breve and several Alistipes and Ruminococcus species. That suggests that a reduction in the numbers of these taxa may be associated with C. difficile presence and surgical risk.
The researchers also found that methionine synthesis pathways were depressed in C. difficile–positive and surgery patients. Methionine may bolster antioxidant capacity and improve villus morphology. IBD patients with dysbiosis and those experiencing Crohn’s disease exacerbations have been shown to have decreased methionine pathway activity, suggesting methionine biosynthesis changes have clinical relevance.
The study was funded by the National Institutes of Health.
SOURCE: Hellmann J et al. Inflamm Bowel Dis. 2020. doi: 10.1093/ibd/izz263.
In pediatric Crohn’s disease, a Clostridioides difficile infection detected within the first year after diagnosis is associated with a shorter time to first bowel resection surgery, according to a study that included both a retrospective and prospective analysis. The researchers also found evidence that changes in methionine biosynthesis and depletion of beneficial bacteria may contribute to risk of surgery.
C. difficile infection (CDI) disproportionately affects individuals with inflammatory bowel disease (IBD). Pediatric IBD patients have a 34% risk of recurrent CDI infection, compared with 7.5% in the general population. Previous research found that adults with ulcerative colitis and CDI are at more risk of colectomy, but the finding has not been replicated in children.
In a study published in Inflammatory Bowel Diseases, researchers led by Jennifer Hellmann and Lee Denson of the University of Cincinnati conducted a single-center retrospective analysis of 75 pediatric Crohn’s disease patients. They also conducted a prospective study of 70 pediatric Crohn’s disease patients, using shotgun metagenome sequencing to examine the relationship between microbiota composition and C. difficile carriage or surgery history.
Nineteen percent of patients tested positive for C. difficile. Use of antibiotics was associated with C. difficile (odds ratio, 7.9; P = .02). Of patients who underwent C. difficile testing in the first year, 23 went on to have surgery: 21% who were C. difficile negative required surgery, compared with 67% of those who were positive (hazard ratio, 4.4; P = .0003). The mean time to surgery was 527 days for C. difficile–positive patients and 1,268 days for those who were negative.
A multivariate regression analysis on 54 patients with complete data sets showed that the presence of C. difficile was associated with increased risk of surgery (OR, 16.2; P = .0006). When the analysis was run on all 73 patients, using null value for missing data, the results were similar (OR, 9.17; P = .008).
Shotgun sequencing found that 47 of 114 bacterial species that were associated with the presence of C. difficile were also associated with prior surgery for Crohn’s disease. Species included some that may play a role in mucosal homeostasis, such as Bifidobacterium breve and several Alistipes and Ruminococcus species. That suggests that a reduction in the numbers of these taxa may be associated with C. difficile presence and surgical risk.
The researchers also found that methionine synthesis pathways were depressed in C. difficile–positive and surgery patients. Methionine may bolster antioxidant capacity and improve villus morphology. IBD patients with dysbiosis and those experiencing Crohn’s disease exacerbations have been shown to have decreased methionine pathway activity, suggesting methionine biosynthesis changes have clinical relevance.
The study was funded by the National Institutes of Health.
SOURCE: Hellmann J et al. Inflamm Bowel Dis. 2020. doi: 10.1093/ibd/izz263.
Fecal transplant linked to reduced C. difficile mortality
Vancomycin followed by fecal microbiota transplant (FMT) was associated with reduced Clostridioides difficile (C. diff)-related mortality in patients hospitalized with refractory severe or fulminant C. diff infection (CDI) at a single center. The improvements came after Indiana University implemented an FMT option in 2013.
About 8% of C. diff patients develop severe or fulminant CDI (SFCDI), which can lead to toxic colon and multiorgan failure. Surgery is the current recommended treatment for these patients if they are refractory to vancomycin, but 30-day mortality is above 40%. FMT is recommended for recurrent CDI, and it achieves cure rates greater than 80%, along with fewer relapses compared with anti-CDI antibiotic therapy.
FMT has been shown to be effective for SFCDI, with a 91% cure rate for serious CDI and 66% for fulminant CDI.
In the study published in the September issue of Clinical Gastroenterology and Hepatology, researchers led by Yao-Wen Cheng, MD, and Monika Fischer, MD, of Indiana University, assessed the effect of FMT on SFCDI after their institution adopted it as a treatment protocol for SFCDI. Patients could receive FMT if there was evidence that their SFCDI was refractory, or if they had two or more CDI recurrences. The treatment includes oral vancomycin and pseudomembrane-driven sequential FMT.
Two hundred five patients were admitted before FMT implementation, 225 after. Fifty patients received FMT because of refractory SFCDI. A median of two FMTs was conducted per patient. 21 other patients received FMT for nonrefractory SFCDI or other conditions, including 18 patients with multiple recurrent CDI.
Thirty-day CDI-related mortality dropped after FMT implementation (4.4% versus 10.2%; P =.02). This was true in both the fulminant subset (9.1% versus 21.3%; P =.015) and the refractory group (12.1% versus 43.2%; P < .001).
The researchers used segmented logistic regression to determine if the improved outcomes could be due to nontreatment factors that varied over time, and found that the difference in CDI-related mortality was eliminated except for refractory SFCDI patients (odds of mortality after FMT implementation, 0.09; P =.023). There was no significant difference between those receiving non-CDI antibiotics (4.8%) and those who did not (6.9%; P =.75).
FMT was associated with lower frequency of CDI-related colectomy overall (2.7% versus 6.8%; P =.041), as well as in the fulminant (5.5% versus 15.7%; P =.017) and refractory subgroups (7.6% versus 31.8%; P =.001).
The findings follow another study that showed improved 3-month mortality for FMT among patients hospitalized with severe CDI (12.1% versus 42.2%; P < .003).
The results underscore the utility of FMT for SFCDI, and suggest it might have the most benefit in refractory SFCDI. The authors believe that FMT should be an alternative to colectomy when first-line anti-CDI antibiotics are partially or completely ineffective. In the absence of FMT, patients who go on to fail vancomycin or fidaxomicin will likely continue to be managed medically, with up to 80% mortality, or through salvage colectomy, with postsurgical morality rates of 30-40%.
Although a randomized trial could answer the question of FMT efficacy more definitively, it is unlikely to be conducted for ethical reasons.
“Further investigation is required to clearly define FMT’s role and timing in the clinical course of severe and fulminant CDI. However, our study suggests that FMT should be offered to patients with severe and fulminant CDI who do not respond to a 5-day course of anti-CDI antibiotics and may be considered in lieu of or before colectomy,” the researchers wrote.
No source of funding was disclosed.
SOURCE: Cheng YW et al. Clin Gastroenterol Hepatol. 2020;18:2234-43. doi: 10.1016/j.cgh.2019.12.029.
Vancomycin followed by fecal microbiota transplant (FMT) was associated with reduced Clostridioides difficile (C. diff)-related mortality in patients hospitalized with refractory severe or fulminant C. diff infection (CDI) at a single center. The improvements came after Indiana University implemented an FMT option in 2013.
About 8% of C. diff patients develop severe or fulminant CDI (SFCDI), which can lead to toxic colon and multiorgan failure. Surgery is the current recommended treatment for these patients if they are refractory to vancomycin, but 30-day mortality is above 40%. FMT is recommended for recurrent CDI, and it achieves cure rates greater than 80%, along with fewer relapses compared with anti-CDI antibiotic therapy.
FMT has been shown to be effective for SFCDI, with a 91% cure rate for serious CDI and 66% for fulminant CDI.
In the study published in the September issue of Clinical Gastroenterology and Hepatology, researchers led by Yao-Wen Cheng, MD, and Monika Fischer, MD, of Indiana University, assessed the effect of FMT on SFCDI after their institution adopted it as a treatment protocol for SFCDI. Patients could receive FMT if there was evidence that their SFCDI was refractory, or if they had two or more CDI recurrences. The treatment includes oral vancomycin and pseudomembrane-driven sequential FMT.
Two hundred five patients were admitted before FMT implementation, 225 after. Fifty patients received FMT because of refractory SFCDI. A median of two FMTs was conducted per patient. 21 other patients received FMT for nonrefractory SFCDI or other conditions, including 18 patients with multiple recurrent CDI.
Thirty-day CDI-related mortality dropped after FMT implementation (4.4% versus 10.2%; P =.02). This was true in both the fulminant subset (9.1% versus 21.3%; P =.015) and the refractory group (12.1% versus 43.2%; P < .001).
The researchers used segmented logistic regression to determine if the improved outcomes could be due to nontreatment factors that varied over time, and found that the difference in CDI-related mortality was eliminated except for refractory SFCDI patients (odds of mortality after FMT implementation, 0.09; P =.023). There was no significant difference between those receiving non-CDI antibiotics (4.8%) and those who did not (6.9%; P =.75).
FMT was associated with lower frequency of CDI-related colectomy overall (2.7% versus 6.8%; P =.041), as well as in the fulminant (5.5% versus 15.7%; P =.017) and refractory subgroups (7.6% versus 31.8%; P =.001).
The findings follow another study that showed improved 3-month mortality for FMT among patients hospitalized with severe CDI (12.1% versus 42.2%; P < .003).
The results underscore the utility of FMT for SFCDI, and suggest it might have the most benefit in refractory SFCDI. The authors believe that FMT should be an alternative to colectomy when first-line anti-CDI antibiotics are partially or completely ineffective. In the absence of FMT, patients who go on to fail vancomycin or fidaxomicin will likely continue to be managed medically, with up to 80% mortality, or through salvage colectomy, with postsurgical morality rates of 30-40%.
Although a randomized trial could answer the question of FMT efficacy more definitively, it is unlikely to be conducted for ethical reasons.
“Further investigation is required to clearly define FMT’s role and timing in the clinical course of severe and fulminant CDI. However, our study suggests that FMT should be offered to patients with severe and fulminant CDI who do not respond to a 5-day course of anti-CDI antibiotics and may be considered in lieu of or before colectomy,” the researchers wrote.
No source of funding was disclosed.
SOURCE: Cheng YW et al. Clin Gastroenterol Hepatol. 2020;18:2234-43. doi: 10.1016/j.cgh.2019.12.029.
Vancomycin followed by fecal microbiota transplant (FMT) was associated with reduced Clostridioides difficile (C. diff)-related mortality in patients hospitalized with refractory severe or fulminant C. diff infection (CDI) at a single center. The improvements came after Indiana University implemented an FMT option in 2013.
About 8% of C. diff patients develop severe or fulminant CDI (SFCDI), which can lead to toxic colon and multiorgan failure. Surgery is the current recommended treatment for these patients if they are refractory to vancomycin, but 30-day mortality is above 40%. FMT is recommended for recurrent CDI, and it achieves cure rates greater than 80%, along with fewer relapses compared with anti-CDI antibiotic therapy.
FMT has been shown to be effective for SFCDI, with a 91% cure rate for serious CDI and 66% for fulminant CDI.
In the study published in the September issue of Clinical Gastroenterology and Hepatology, researchers led by Yao-Wen Cheng, MD, and Monika Fischer, MD, of Indiana University, assessed the effect of FMT on SFCDI after their institution adopted it as a treatment protocol for SFCDI. Patients could receive FMT if there was evidence that their SFCDI was refractory, or if they had two or more CDI recurrences. The treatment includes oral vancomycin and pseudomembrane-driven sequential FMT.
Two hundred five patients were admitted before FMT implementation, 225 after. Fifty patients received FMT because of refractory SFCDI. A median of two FMTs was conducted per patient. 21 other patients received FMT for nonrefractory SFCDI or other conditions, including 18 patients with multiple recurrent CDI.
Thirty-day CDI-related mortality dropped after FMT implementation (4.4% versus 10.2%; P =.02). This was true in both the fulminant subset (9.1% versus 21.3%; P =.015) and the refractory group (12.1% versus 43.2%; P < .001).
The researchers used segmented logistic regression to determine if the improved outcomes could be due to nontreatment factors that varied over time, and found that the difference in CDI-related mortality was eliminated except for refractory SFCDI patients (odds of mortality after FMT implementation, 0.09; P =.023). There was no significant difference between those receiving non-CDI antibiotics (4.8%) and those who did not (6.9%; P =.75).
FMT was associated with lower frequency of CDI-related colectomy overall (2.7% versus 6.8%; P =.041), as well as in the fulminant (5.5% versus 15.7%; P =.017) and refractory subgroups (7.6% versus 31.8%; P =.001).
The findings follow another study that showed improved 3-month mortality for FMT among patients hospitalized with severe CDI (12.1% versus 42.2%; P < .003).
The results underscore the utility of FMT for SFCDI, and suggest it might have the most benefit in refractory SFCDI. The authors believe that FMT should be an alternative to colectomy when first-line anti-CDI antibiotics are partially or completely ineffective. In the absence of FMT, patients who go on to fail vancomycin or fidaxomicin will likely continue to be managed medically, with up to 80% mortality, or through salvage colectomy, with postsurgical morality rates of 30-40%.
Although a randomized trial could answer the question of FMT efficacy more definitively, it is unlikely to be conducted for ethical reasons.
“Further investigation is required to clearly define FMT’s role and timing in the clinical course of severe and fulminant CDI. However, our study suggests that FMT should be offered to patients with severe and fulminant CDI who do not respond to a 5-day course of anti-CDI antibiotics and may be considered in lieu of or before colectomy,” the researchers wrote.
No source of funding was disclosed.
SOURCE: Cheng YW et al. Clin Gastroenterol Hepatol. 2020;18:2234-43. doi: 10.1016/j.cgh.2019.12.029.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
What’s in a number? 697,633 children with COVID-19
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
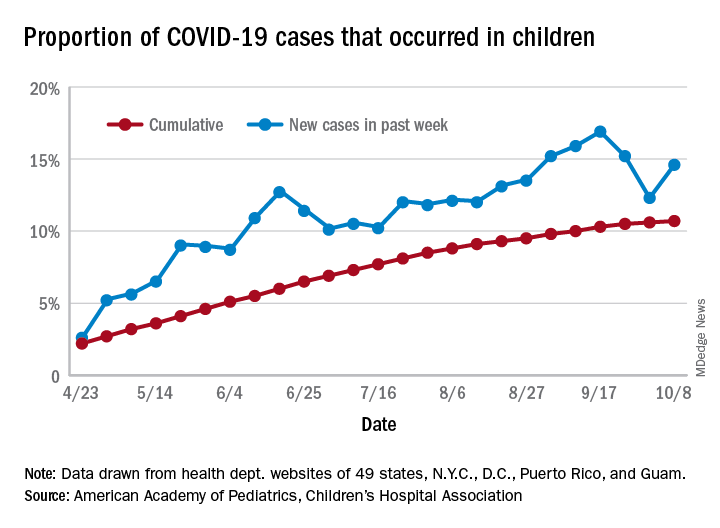
For the week, 14.6% of all COVID-19 cases reported in the United States occurred in children, after 2 consecutive weeks of declines that saw the proportion drop from 16.9% to 12.3%. The cumulative rate of child cases for the entire pandemic is 10.7%, with total child cases in the United States now up to 697,633 and cases among all ages at just over 6.5 million, the AAP and the CHA said Oct. 12 in their weekly COVID-19 report.
Nationally, there were 927 cases reported per 100,000 children as of Oct. 8, with rates at the state level varying from 176 per 100,000 in Vermont to 2,221 per 100,000 in North Dakota. Two other states were over 2,000 cases per 100,000 children: Tennessee (2,155) and South Carolina (2,116), based on data from the health departments of 49 states (New York does not report age distribution), as well as the District of Columbia, New York City, Puerto Rico, and Guam.
Severe illness continues to be rare in children, and national (25 states and New York City) hospitalization rates dropped in the last week. The proportion of hospitalizations occurring in children slipped from a pandemic high of 1.8% the previous week to 1.7% during the week of Oct. 8, and the rate of hospitalizations for children with COVID-19 was down to 1.4% from 1.6% the week before and 1.9% on Sept. 3, the AAP and the CHA said.
Mortality data from 42 states and New York City also show a decline. For the third consecutive week, children represented just 0.06% of all COVID-19 deaths in the United States, down from a high of 0.07% on Sept. 17. Only 0.02% of all cases in children have resulted in death, and that figure has been dropping since early June, when it reached 0.06%, according to the AAP/CHA report. As of Oct. 8, there have been 115 total deaths reported in children.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

For the week, 14.6% of all COVID-19 cases reported in the United States occurred in children, after 2 consecutive weeks of declines that saw the proportion drop from 16.9% to 12.3%. The cumulative rate of child cases for the entire pandemic is 10.7%, with total child cases in the United States now up to 697,633 and cases among all ages at just over 6.5 million, the AAP and the CHA said Oct. 12 in their weekly COVID-19 report.
Nationally, there were 927 cases reported per 100,000 children as of Oct. 8, with rates at the state level varying from 176 per 100,000 in Vermont to 2,221 per 100,000 in North Dakota. Two other states were over 2,000 cases per 100,000 children: Tennessee (2,155) and South Carolina (2,116), based on data from the health departments of 49 states (New York does not report age distribution), as well as the District of Columbia, New York City, Puerto Rico, and Guam.
Severe illness continues to be rare in children, and national (25 states and New York City) hospitalization rates dropped in the last week. The proportion of hospitalizations occurring in children slipped from a pandemic high of 1.8% the previous week to 1.7% during the week of Oct. 8, and the rate of hospitalizations for children with COVID-19 was down to 1.4% from 1.6% the week before and 1.9% on Sept. 3, the AAP and the CHA said.
Mortality data from 42 states and New York City also show a decline. For the third consecutive week, children represented just 0.06% of all COVID-19 deaths in the United States, down from a high of 0.07% on Sept. 17. Only 0.02% of all cases in children have resulted in death, and that figure has been dropping since early June, when it reached 0.06%, according to the AAP/CHA report. As of Oct. 8, there have been 115 total deaths reported in children.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

For the week, 14.6% of all COVID-19 cases reported in the United States occurred in children, after 2 consecutive weeks of declines that saw the proportion drop from 16.9% to 12.3%. The cumulative rate of child cases for the entire pandemic is 10.7%, with total child cases in the United States now up to 697,633 and cases among all ages at just over 6.5 million, the AAP and the CHA said Oct. 12 in their weekly COVID-19 report.
Nationally, there were 927 cases reported per 100,000 children as of Oct. 8, with rates at the state level varying from 176 per 100,000 in Vermont to 2,221 per 100,000 in North Dakota. Two other states were over 2,000 cases per 100,000 children: Tennessee (2,155) and South Carolina (2,116), based on data from the health departments of 49 states (New York does not report age distribution), as well as the District of Columbia, New York City, Puerto Rico, and Guam.
Severe illness continues to be rare in children, and national (25 states and New York City) hospitalization rates dropped in the last week. The proportion of hospitalizations occurring in children slipped from a pandemic high of 1.8% the previous week to 1.7% during the week of Oct. 8, and the rate of hospitalizations for children with COVID-19 was down to 1.4% from 1.6% the week before and 1.9% on Sept. 3, the AAP and the CHA said.
Mortality data from 42 states and New York City also show a decline. For the third consecutive week, children represented just 0.06% of all COVID-19 deaths in the United States, down from a high of 0.07% on Sept. 17. Only 0.02% of all cases in children have resulted in death, and that figure has been dropping since early June, when it reached 0.06%, according to the AAP/CHA report. As of Oct. 8, there have been 115 total deaths reported in children.
Flu vaccine significantly cuts pediatric hospitalizations
Unlike previous studies focused on vaccine effectiveness (VE) in ambulatory care office visits, Angela P. Campbell, MD, MPH, and associates have uncovered evidence of the overall benefit influenza vaccines play in reducing hospitalizations and emergency department visits in pediatric influenza patients.
“Our data provide important VE estimates against severe influenza in children,” the researchers noted in Pediatrics, adding that the findings “provide important evidence supporting the annual recommendation that all children 6 months and older should receive influenza vaccination.”
Dr. Campbell and colleagues collected ongoing surveillance data from the New Vaccine Surveillance Network (NVSN), which is a network of pediatric hospitals across seven cities, including Kansas City, Mo.; Rochester, N.Y.; Cincinnati; Pittsburgh; Nashville, Tenn.; Houston; and Seattle. The influenza season encompassed the period Nov. 7, 2018 to June 21, 2019.
A total of 2,748 hospitalized children and 2,676 children who had completed ED visits that did not lead to hospitalization were included. Once those under 6 months were excluded, 1,792 hospitalized children were included in the VE analysis; of these, 226 (13%) tested positive for influenza infection, including 211 (93%) with influenza A viruses and 15 (7%) with influenza B viruses. Fully 1,611 of the patients (90%), had verified vaccine status, while 181 (10%) had solely parental reported vaccine status. The researchers reported 88 (5%) of the patients received mechanical ventilation and 7 (<1%) died.
Most noteworthy, They further estimated a significant reduction in hospitalizations linked to A(H3N2) and A(H1N1)pdm09 viruses, even in the presence of circulating A(H3N2) viruses that differed from the A(H3N2) vaccine component.
Studies from other countries during the same time period showed that while “significant protection against influenza-associated ambulatory care visits and hospitalizations among children infected with A(H1N1)pdm09 viruses” was observed, the same could not be said for protection against A(H3N2) viruses, which varied among pediatric outpatients in the United States (24%), in England (17% outpatient; 31% inpatient), Europe (46%), and Canada (48%). They explained that such variation in vaccine protection is multifactorial, and includes virus-, host-, and environment-related factors. They also noted that regional variations in circulating viruses, host factors including age, imprinting, and previous vaccination could explain the study’s finding of vaccine protection against both A(H1N1)pdm09 and A(H3N2) viruses.
When comparing VE estimates between ED visits and hospitalizations, the researchers observed one significant difference, that “hospitalized children likely represent more medically complex patients, with 58% having underlying medical conditions and 38% reporting at lease one hospitalization in the past year, compared with 28% and 14% respectively, among ED participants.”
Strengths of the study included the prospective multisite enrollment that provided data across diverse locations and representation from pediatric hospitalizations and ED care, which were not previously strongly represented in the literature. The single-season study with small sample size was considered a limitation, as was the inability to evaluate full and partial vaccine status. Vaccine data also were limited for many of the ED patients observed.
Dr. Campbell and colleagues did caution that while they consider their test-negative design optimal for evaluating both hospitalized and ED patients, they feel their results should not be “interpreted as VE against influenza-associated ambulatory care visits or infections that are not medically attended.”
In a separate interview, Michael E. Pichichero, MD, director of the Rochester General Hospital Research Institute and a clinical professor of pediatrics at the University of Rochester (N.Y.), observed: “There are really no surprises here. A well done contemporary study confirms again the benefits of annual influenza vaccinations for children. Viral coinfections involving SARS-CoV-2 and influenza have been reported from Australia to cause heightened illnesses. That observation provides further impetus for parents to have their children receive influenza vaccinations.”
The researchers cited multiple sources of financial support for their ongoing work, including Sanofi, Quidel, Moderna, Karius, GlaxoSmithKline, Merck, AstraZeneca, and Pfizer. Funding for this study was supported by the Centers for Disease Control and Prevention. Dr. Pichichero said he had no relevant financial disclosures.
SOURCE: Campbell AP et al. Pediatrics. 2020. doi: 10.1542/peds.2020-1368.
Unlike previous studies focused on vaccine effectiveness (VE) in ambulatory care office visits, Angela P. Campbell, MD, MPH, and associates have uncovered evidence of the overall benefit influenza vaccines play in reducing hospitalizations and emergency department visits in pediatric influenza patients.
“Our data provide important VE estimates against severe influenza in children,” the researchers noted in Pediatrics, adding that the findings “provide important evidence supporting the annual recommendation that all children 6 months and older should receive influenza vaccination.”
Dr. Campbell and colleagues collected ongoing surveillance data from the New Vaccine Surveillance Network (NVSN), which is a network of pediatric hospitals across seven cities, including Kansas City, Mo.; Rochester, N.Y.; Cincinnati; Pittsburgh; Nashville, Tenn.; Houston; and Seattle. The influenza season encompassed the period Nov. 7, 2018 to June 21, 2019.
A total of 2,748 hospitalized children and 2,676 children who had completed ED visits that did not lead to hospitalization were included. Once those under 6 months were excluded, 1,792 hospitalized children were included in the VE analysis; of these, 226 (13%) tested positive for influenza infection, including 211 (93%) with influenza A viruses and 15 (7%) with influenza B viruses. Fully 1,611 of the patients (90%), had verified vaccine status, while 181 (10%) had solely parental reported vaccine status. The researchers reported 88 (5%) of the patients received mechanical ventilation and 7 (<1%) died.
Most noteworthy, They further estimated a significant reduction in hospitalizations linked to A(H3N2) and A(H1N1)pdm09 viruses, even in the presence of circulating A(H3N2) viruses that differed from the A(H3N2) vaccine component.
Studies from other countries during the same time period showed that while “significant protection against influenza-associated ambulatory care visits and hospitalizations among children infected with A(H1N1)pdm09 viruses” was observed, the same could not be said for protection against A(H3N2) viruses, which varied among pediatric outpatients in the United States (24%), in England (17% outpatient; 31% inpatient), Europe (46%), and Canada (48%). They explained that such variation in vaccine protection is multifactorial, and includes virus-, host-, and environment-related factors. They also noted that regional variations in circulating viruses, host factors including age, imprinting, and previous vaccination could explain the study’s finding of vaccine protection against both A(H1N1)pdm09 and A(H3N2) viruses.
When comparing VE estimates between ED visits and hospitalizations, the researchers observed one significant difference, that “hospitalized children likely represent more medically complex patients, with 58% having underlying medical conditions and 38% reporting at lease one hospitalization in the past year, compared with 28% and 14% respectively, among ED participants.”
Strengths of the study included the prospective multisite enrollment that provided data across diverse locations and representation from pediatric hospitalizations and ED care, which were not previously strongly represented in the literature. The single-season study with small sample size was considered a limitation, as was the inability to evaluate full and partial vaccine status. Vaccine data also were limited for many of the ED patients observed.
Dr. Campbell and colleagues did caution that while they consider their test-negative design optimal for evaluating both hospitalized and ED patients, they feel their results should not be “interpreted as VE against influenza-associated ambulatory care visits or infections that are not medically attended.”
In a separate interview, Michael E. Pichichero, MD, director of the Rochester General Hospital Research Institute and a clinical professor of pediatrics at the University of Rochester (N.Y.), observed: “There are really no surprises here. A well done contemporary study confirms again the benefits of annual influenza vaccinations for children. Viral coinfections involving SARS-CoV-2 and influenza have been reported from Australia to cause heightened illnesses. That observation provides further impetus for parents to have their children receive influenza vaccinations.”
The researchers cited multiple sources of financial support for their ongoing work, including Sanofi, Quidel, Moderna, Karius, GlaxoSmithKline, Merck, AstraZeneca, and Pfizer. Funding for this study was supported by the Centers for Disease Control and Prevention. Dr. Pichichero said he had no relevant financial disclosures.
SOURCE: Campbell AP et al. Pediatrics. 2020. doi: 10.1542/peds.2020-1368.
Unlike previous studies focused on vaccine effectiveness (VE) in ambulatory care office visits, Angela P. Campbell, MD, MPH, and associates have uncovered evidence of the overall benefit influenza vaccines play in reducing hospitalizations and emergency department visits in pediatric influenza patients.
“Our data provide important VE estimates against severe influenza in children,” the researchers noted in Pediatrics, adding that the findings “provide important evidence supporting the annual recommendation that all children 6 months and older should receive influenza vaccination.”
Dr. Campbell and colleagues collected ongoing surveillance data from the New Vaccine Surveillance Network (NVSN), which is a network of pediatric hospitals across seven cities, including Kansas City, Mo.; Rochester, N.Y.; Cincinnati; Pittsburgh; Nashville, Tenn.; Houston; and Seattle. The influenza season encompassed the period Nov. 7, 2018 to June 21, 2019.
A total of 2,748 hospitalized children and 2,676 children who had completed ED visits that did not lead to hospitalization were included. Once those under 6 months were excluded, 1,792 hospitalized children were included in the VE analysis; of these, 226 (13%) tested positive for influenza infection, including 211 (93%) with influenza A viruses and 15 (7%) with influenza B viruses. Fully 1,611 of the patients (90%), had verified vaccine status, while 181 (10%) had solely parental reported vaccine status. The researchers reported 88 (5%) of the patients received mechanical ventilation and 7 (<1%) died.
Most noteworthy, They further estimated a significant reduction in hospitalizations linked to A(H3N2) and A(H1N1)pdm09 viruses, even in the presence of circulating A(H3N2) viruses that differed from the A(H3N2) vaccine component.
Studies from other countries during the same time period showed that while “significant protection against influenza-associated ambulatory care visits and hospitalizations among children infected with A(H1N1)pdm09 viruses” was observed, the same could not be said for protection against A(H3N2) viruses, which varied among pediatric outpatients in the United States (24%), in England (17% outpatient; 31% inpatient), Europe (46%), and Canada (48%). They explained that such variation in vaccine protection is multifactorial, and includes virus-, host-, and environment-related factors. They also noted that regional variations in circulating viruses, host factors including age, imprinting, and previous vaccination could explain the study’s finding of vaccine protection against both A(H1N1)pdm09 and A(H3N2) viruses.
When comparing VE estimates between ED visits and hospitalizations, the researchers observed one significant difference, that “hospitalized children likely represent more medically complex patients, with 58% having underlying medical conditions and 38% reporting at lease one hospitalization in the past year, compared with 28% and 14% respectively, among ED participants.”
Strengths of the study included the prospective multisite enrollment that provided data across diverse locations and representation from pediatric hospitalizations and ED care, which were not previously strongly represented in the literature. The single-season study with small sample size was considered a limitation, as was the inability to evaluate full and partial vaccine status. Vaccine data also were limited for many of the ED patients observed.
Dr. Campbell and colleagues did caution that while they consider their test-negative design optimal for evaluating both hospitalized and ED patients, they feel their results should not be “interpreted as VE against influenza-associated ambulatory care visits or infections that are not medically attended.”
In a separate interview, Michael E. Pichichero, MD, director of the Rochester General Hospital Research Institute and a clinical professor of pediatrics at the University of Rochester (N.Y.), observed: “There are really no surprises here. A well done contemporary study confirms again the benefits of annual influenza vaccinations for children. Viral coinfections involving SARS-CoV-2 and influenza have been reported from Australia to cause heightened illnesses. That observation provides further impetus for parents to have their children receive influenza vaccinations.”
The researchers cited multiple sources of financial support for their ongoing work, including Sanofi, Quidel, Moderna, Karius, GlaxoSmithKline, Merck, AstraZeneca, and Pfizer. Funding for this study was supported by the Centers for Disease Control and Prevention. Dr. Pichichero said he had no relevant financial disclosures.
SOURCE: Campbell AP et al. Pediatrics. 2020. doi: 10.1542/peds.2020-1368.
FROM PEDIATRICS
‘Profound human toll’ in excess deaths from COVID-19 calculated in two studies
However, additional deaths could be indirectly related because people avoided emergency care during the pandemic, new research shows.
Deaths linked to COVID-19 varied by state and phase of the pandemic, as reported in a study from researchers at Virginia Commonwealth University and Yale University that was published online October 12 in JAMA.
Another study published online simultaneously in JAMA took more of an international perspective. Investigators from the University of Pennsylvania and Harvard University found that in America there were more excess deaths and there was higher all-cause mortality during the pandemic than in 18 other countries.
Although the ongoing number of deaths attributable to COVID-19 continues to garner attention, there can be a lag of weeks or months in how long it takes some public health agencies to update their figures.
“For the public at large, the take-home message is twofold: that the number of deaths caused by the pandemic exceeds publicly reported COVID-19 death counts by 20% and that states that reopened or lifted restrictions early suffered a protracted surge in excess deaths that extended into the summer,” lead author of the US-focused study, Steven H. Woolf, MD, MPH, told Medscape Medical News.
The take-away for physicians is in the bigger picture – it is likely that the COVID-19 pandemic is responsible for deaths from other conditions as well. “Surges in COVID-19 were accompanied by an increase in deaths attributed to other causes, such as heart disease and Alzheimer’s disease and dementia,” said Woolf, director emeritus and senior adviser at the Center on Society and Health and professor in the Department of Family Medicine and Population Health at the Virginia Commonwealth University School of Medicine in Richmond, Virginia.
The investigators identified 225,530 excess US deaths in the 5 months from March to July. They report that 67% were directly attributable to COVID-19.
Deaths linked to COVID-19 included those in which the disease was listed as an underlying or contributing cause. US total death rates are “remarkably consistent” year after year, and the investigators calculated a 20% overall jump in mortality.
The study included data from the National Center for Health Statistics and the US Census Bureau for 48 states and the District of Columbia. Connecticut and North Carolina were excluded because of missing data.
Woolf and colleagues also found statistically higher rates of deaths from two other causes, heart disease and Alzheimer’s disease/dementia.
Altered states
New York, New Jersey, Massachusetts, Louisiana, Arizona, Mississippi, Maryland, Delaware, Rhode Island, and Michigan had the highest per capita excess death rates. Three states experienced the shortest epidemics during the study period: New York, New Jersey, and Massachusetts.
Some lessons could be learned by looking at how individual states managed large numbers of people with COVID-19. “Although we suspected that states that reopened early might have put themselves at risk of a pandemic surge, the consistency with which that occurred and the devastating numbers of deaths they suffered was a surprise,” Woolf said.
“The goal of our study is not to look in the rearview mirror and lament what happened months ago but to learn the lesson going forward: Our country will be unable to take control of this pandemic without more robust efforts to control community spread,” Woolf said. “Our study found that states that did this well, such as New York and New Jersey, experienced large surges but bent the curve and were back to baseline in less than 10 weeks.
“If we could do this as a country, countless lives could be saved.”
A global perspective
The United States experienced high mortality linked to COVID-19, as well as high all-cause mortality, compared with 18 other countries, as reported in the study by University of Pennsylvania and Harvard University researchers.
The United States ranked third, with 72 deaths per 100,000 people, among countries with moderate or high mortality. Although perhaps not surprising given the state of SARS-CoV-2 infection across the United States, a question remains as to what extent the relatively high mortality rate is linked to early outbreaks vs “poor long-term response,” the researchers note.
Alyssa Bilinski, MSc, and lead author Ezekiel J. Emanuel, MD, PhD, chair of the Department of Medical Ethics and Health Policy at the University of Pennsylvania Perelman School of Medicine in Philadelphia, calculated the difference in COVID-19 deaths among countries through Sept. 19, 2020. On this date, the United States reported a total 198,589 COVID-19 deaths.
They calculated that, if the US death rates were similar to those in Australia, the United States would have experienced 187,661 fewer COVID-19 deaths. If similar to those of Canada, there would have been 117,622 fewer deaths in the United States.
The US death rate was lower than six other countries with high COVID-19 mortality in the early spring, including Belgium, Spain, and the United Kingdom. However, after May 10, the per capita mortality rate in the United States exceeded the others.
Between May 10 and Sept. 19, the death rate in Italy was 9.1 per 100,000, vs 36.9 per 100,000.
“After the first peak in early spring, US death rates from COVID-19 and from all causes remained higher than even countries with high COVID-19 mortality,” the researchers note. “This may have been a result of several factors, including weak public health infrastructure and a decentralized, inconsistent US response to the pandemic.”
“Mortifying and motivating”
Woolf and colleagues estimate that more than 225,000 excess deaths occurred in recent months; this represents a 20% increase over expected deaths, note Harvey V. Fineberg, MD, PhD, of the Gordon and Betty Moore Foundation, in an accompanying editorial in JAMA.
“Importantly, a condition such as COVID-19 can contribute both directly and indirectly to excess mortality,” he writes.
Although the direct contribution to the mortality rates by those infected is straightforward, “the indirect contribution may relate to circumstances or choices due to the COVID-19 pandemic: for example, a patient who develops symptoms of a stroke is too concerned about COVID-19 to go to the emergency department, and a potentially reversible condition becomes fatal.”
Fineberg notes that “a general indication of the death toll from COVID-19 and the excess deaths related to the pandemic, as presented by Woolf et al, are sufficiently mortifying and motivating.”
“Profound human toll”
“The importance of the estimate by Woolf et al – which suggests that for the entirety of 2020, more than 400,000 excess deaths will occur – cannot be overstated, because it accounts for what could be declines in some causes of death, like motor vehicle crashes, but increases in others, like myocardial infarction,” write Howard Bauchner, MD, editor in chief of JAMA, and Phil B. Fontanarosa, MD, MBA, executive editor of JAMA, in another accompanying editorial.
“These deaths reflect a true measure of the human cost of the Great Pandemic of 2020,” they add.
The study from Emanuel and Bilinski was notable for calculating the excess COVID-19 and all-cause mortality to Sept. 2020, they note. “After the initial peak in early spring, US death rates from COVID-19 and from all causes remained higher than rates in countries with high COVID-19 mortality.”
“Few people will forget the Great Pandemic of 2020, where and how they lived, how it substantially changed their lives, and for many, the profound human toll it has taken,” Bauchner and Fontanarosa write.
The study by Woolf and colleagues was supported by National Center for Advancing Translational Sciences, the National Institute on Aging, and the National Institute of Allergy and Infectious Diseases. The study by Bilinski and Emanuel was partially funded by the Colton Foundation. Woolf, Emanuel, Fineberg, Bauchner, and Fontanarosa have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
However, additional deaths could be indirectly related because people avoided emergency care during the pandemic, new research shows.
Deaths linked to COVID-19 varied by state and phase of the pandemic, as reported in a study from researchers at Virginia Commonwealth University and Yale University that was published online October 12 in JAMA.
Another study published online simultaneously in JAMA took more of an international perspective. Investigators from the University of Pennsylvania and Harvard University found that in America there were more excess deaths and there was higher all-cause mortality during the pandemic than in 18 other countries.
Although the ongoing number of deaths attributable to COVID-19 continues to garner attention, there can be a lag of weeks or months in how long it takes some public health agencies to update their figures.
“For the public at large, the take-home message is twofold: that the number of deaths caused by the pandemic exceeds publicly reported COVID-19 death counts by 20% and that states that reopened or lifted restrictions early suffered a protracted surge in excess deaths that extended into the summer,” lead author of the US-focused study, Steven H. Woolf, MD, MPH, told Medscape Medical News.
The take-away for physicians is in the bigger picture – it is likely that the COVID-19 pandemic is responsible for deaths from other conditions as well. “Surges in COVID-19 were accompanied by an increase in deaths attributed to other causes, such as heart disease and Alzheimer’s disease and dementia,” said Woolf, director emeritus and senior adviser at the Center on Society and Health and professor in the Department of Family Medicine and Population Health at the Virginia Commonwealth University School of Medicine in Richmond, Virginia.
The investigators identified 225,530 excess US deaths in the 5 months from March to July. They report that 67% were directly attributable to COVID-19.
Deaths linked to COVID-19 included those in which the disease was listed as an underlying or contributing cause. US total death rates are “remarkably consistent” year after year, and the investigators calculated a 20% overall jump in mortality.
The study included data from the National Center for Health Statistics and the US Census Bureau for 48 states and the District of Columbia. Connecticut and North Carolina were excluded because of missing data.
Woolf and colleagues also found statistically higher rates of deaths from two other causes, heart disease and Alzheimer’s disease/dementia.
Altered states
New York, New Jersey, Massachusetts, Louisiana, Arizona, Mississippi, Maryland, Delaware, Rhode Island, and Michigan had the highest per capita excess death rates. Three states experienced the shortest epidemics during the study period: New York, New Jersey, and Massachusetts.
Some lessons could be learned by looking at how individual states managed large numbers of people with COVID-19. “Although we suspected that states that reopened early might have put themselves at risk of a pandemic surge, the consistency with which that occurred and the devastating numbers of deaths they suffered was a surprise,” Woolf said.
“The goal of our study is not to look in the rearview mirror and lament what happened months ago but to learn the lesson going forward: Our country will be unable to take control of this pandemic without more robust efforts to control community spread,” Woolf said. “Our study found that states that did this well, such as New York and New Jersey, experienced large surges but bent the curve and were back to baseline in less than 10 weeks.
“If we could do this as a country, countless lives could be saved.”
A global perspective
The United States experienced high mortality linked to COVID-19, as well as high all-cause mortality, compared with 18 other countries, as reported in the study by University of Pennsylvania and Harvard University researchers.
The United States ranked third, with 72 deaths per 100,000 people, among countries with moderate or high mortality. Although perhaps not surprising given the state of SARS-CoV-2 infection across the United States, a question remains as to what extent the relatively high mortality rate is linked to early outbreaks vs “poor long-term response,” the researchers note.
Alyssa Bilinski, MSc, and lead author Ezekiel J. Emanuel, MD, PhD, chair of the Department of Medical Ethics and Health Policy at the University of Pennsylvania Perelman School of Medicine in Philadelphia, calculated the difference in COVID-19 deaths among countries through Sept. 19, 2020. On this date, the United States reported a total 198,589 COVID-19 deaths.
They calculated that, if the US death rates were similar to those in Australia, the United States would have experienced 187,661 fewer COVID-19 deaths. If similar to those of Canada, there would have been 117,622 fewer deaths in the United States.
The US death rate was lower than six other countries with high COVID-19 mortality in the early spring, including Belgium, Spain, and the United Kingdom. However, after May 10, the per capita mortality rate in the United States exceeded the others.
Between May 10 and Sept. 19, the death rate in Italy was 9.1 per 100,000, vs 36.9 per 100,000.
“After the first peak in early spring, US death rates from COVID-19 and from all causes remained higher than even countries with high COVID-19 mortality,” the researchers note. “This may have been a result of several factors, including weak public health infrastructure and a decentralized, inconsistent US response to the pandemic.”
“Mortifying and motivating”
Woolf and colleagues estimate that more than 225,000 excess deaths occurred in recent months; this represents a 20% increase over expected deaths, note Harvey V. Fineberg, MD, PhD, of the Gordon and Betty Moore Foundation, in an accompanying editorial in JAMA.
“Importantly, a condition such as COVID-19 can contribute both directly and indirectly to excess mortality,” he writes.
Although the direct contribution to the mortality rates by those infected is straightforward, “the indirect contribution may relate to circumstances or choices due to the COVID-19 pandemic: for example, a patient who develops symptoms of a stroke is too concerned about COVID-19 to go to the emergency department, and a potentially reversible condition becomes fatal.”
Fineberg notes that “a general indication of the death toll from COVID-19 and the excess deaths related to the pandemic, as presented by Woolf et al, are sufficiently mortifying and motivating.”
“Profound human toll”
“The importance of the estimate by Woolf et al – which suggests that for the entirety of 2020, more than 400,000 excess deaths will occur – cannot be overstated, because it accounts for what could be declines in some causes of death, like motor vehicle crashes, but increases in others, like myocardial infarction,” write Howard Bauchner, MD, editor in chief of JAMA, and Phil B. Fontanarosa, MD, MBA, executive editor of JAMA, in another accompanying editorial.
“These deaths reflect a true measure of the human cost of the Great Pandemic of 2020,” they add.
The study from Emanuel and Bilinski was notable for calculating the excess COVID-19 and all-cause mortality to Sept. 2020, they note. “After the initial peak in early spring, US death rates from COVID-19 and from all causes remained higher than rates in countries with high COVID-19 mortality.”
“Few people will forget the Great Pandemic of 2020, where and how they lived, how it substantially changed their lives, and for many, the profound human toll it has taken,” Bauchner and Fontanarosa write.
The study by Woolf and colleagues was supported by National Center for Advancing Translational Sciences, the National Institute on Aging, and the National Institute of Allergy and Infectious Diseases. The study by Bilinski and Emanuel was partially funded by the Colton Foundation. Woolf, Emanuel, Fineberg, Bauchner, and Fontanarosa have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
However, additional deaths could be indirectly related because people avoided emergency care during the pandemic, new research shows.
Deaths linked to COVID-19 varied by state and phase of the pandemic, as reported in a study from researchers at Virginia Commonwealth University and Yale University that was published online October 12 in JAMA.
Another study published online simultaneously in JAMA took more of an international perspective. Investigators from the University of Pennsylvania and Harvard University found that in America there were more excess deaths and there was higher all-cause mortality during the pandemic than in 18 other countries.
Although the ongoing number of deaths attributable to COVID-19 continues to garner attention, there can be a lag of weeks or months in how long it takes some public health agencies to update their figures.
“For the public at large, the take-home message is twofold: that the number of deaths caused by the pandemic exceeds publicly reported COVID-19 death counts by 20% and that states that reopened or lifted restrictions early suffered a protracted surge in excess deaths that extended into the summer,” lead author of the US-focused study, Steven H. Woolf, MD, MPH, told Medscape Medical News.
The take-away for physicians is in the bigger picture – it is likely that the COVID-19 pandemic is responsible for deaths from other conditions as well. “Surges in COVID-19 were accompanied by an increase in deaths attributed to other causes, such as heart disease and Alzheimer’s disease and dementia,” said Woolf, director emeritus and senior adviser at the Center on Society and Health and professor in the Department of Family Medicine and Population Health at the Virginia Commonwealth University School of Medicine in Richmond, Virginia.
The investigators identified 225,530 excess US deaths in the 5 months from March to July. They report that 67% were directly attributable to COVID-19.
Deaths linked to COVID-19 included those in which the disease was listed as an underlying or contributing cause. US total death rates are “remarkably consistent” year after year, and the investigators calculated a 20% overall jump in mortality.
The study included data from the National Center for Health Statistics and the US Census Bureau for 48 states and the District of Columbia. Connecticut and North Carolina were excluded because of missing data.
Woolf and colleagues also found statistically higher rates of deaths from two other causes, heart disease and Alzheimer’s disease/dementia.
Altered states
New York, New Jersey, Massachusetts, Louisiana, Arizona, Mississippi, Maryland, Delaware, Rhode Island, and Michigan had the highest per capita excess death rates. Three states experienced the shortest epidemics during the study period: New York, New Jersey, and Massachusetts.
Some lessons could be learned by looking at how individual states managed large numbers of people with COVID-19. “Although we suspected that states that reopened early might have put themselves at risk of a pandemic surge, the consistency with which that occurred and the devastating numbers of deaths they suffered was a surprise,” Woolf said.
“The goal of our study is not to look in the rearview mirror and lament what happened months ago but to learn the lesson going forward: Our country will be unable to take control of this pandemic without more robust efforts to control community spread,” Woolf said. “Our study found that states that did this well, such as New York and New Jersey, experienced large surges but bent the curve and were back to baseline in less than 10 weeks.
“If we could do this as a country, countless lives could be saved.”
A global perspective
The United States experienced high mortality linked to COVID-19, as well as high all-cause mortality, compared with 18 other countries, as reported in the study by University of Pennsylvania and Harvard University researchers.
The United States ranked third, with 72 deaths per 100,000 people, among countries with moderate or high mortality. Although perhaps not surprising given the state of SARS-CoV-2 infection across the United States, a question remains as to what extent the relatively high mortality rate is linked to early outbreaks vs “poor long-term response,” the researchers note.
Alyssa Bilinski, MSc, and lead author Ezekiel J. Emanuel, MD, PhD, chair of the Department of Medical Ethics and Health Policy at the University of Pennsylvania Perelman School of Medicine in Philadelphia, calculated the difference in COVID-19 deaths among countries through Sept. 19, 2020. On this date, the United States reported a total 198,589 COVID-19 deaths.
They calculated that, if the US death rates were similar to those in Australia, the United States would have experienced 187,661 fewer COVID-19 deaths. If similar to those of Canada, there would have been 117,622 fewer deaths in the United States.
The US death rate was lower than six other countries with high COVID-19 mortality in the early spring, including Belgium, Spain, and the United Kingdom. However, after May 10, the per capita mortality rate in the United States exceeded the others.
Between May 10 and Sept. 19, the death rate in Italy was 9.1 per 100,000, vs 36.9 per 100,000.
“After the first peak in early spring, US death rates from COVID-19 and from all causes remained higher than even countries with high COVID-19 mortality,” the researchers note. “This may have been a result of several factors, including weak public health infrastructure and a decentralized, inconsistent US response to the pandemic.”
“Mortifying and motivating”
Woolf and colleagues estimate that more than 225,000 excess deaths occurred in recent months; this represents a 20% increase over expected deaths, note Harvey V. Fineberg, MD, PhD, of the Gordon and Betty Moore Foundation, in an accompanying editorial in JAMA.
“Importantly, a condition such as COVID-19 can contribute both directly and indirectly to excess mortality,” he writes.
Although the direct contribution to the mortality rates by those infected is straightforward, “the indirect contribution may relate to circumstances or choices due to the COVID-19 pandemic: for example, a patient who develops symptoms of a stroke is too concerned about COVID-19 to go to the emergency department, and a potentially reversible condition becomes fatal.”
Fineberg notes that “a general indication of the death toll from COVID-19 and the excess deaths related to the pandemic, as presented by Woolf et al, are sufficiently mortifying and motivating.”
“Profound human toll”
“The importance of the estimate by Woolf et al – which suggests that for the entirety of 2020, more than 400,000 excess deaths will occur – cannot be overstated, because it accounts for what could be declines in some causes of death, like motor vehicle crashes, but increases in others, like myocardial infarction,” write Howard Bauchner, MD, editor in chief of JAMA, and Phil B. Fontanarosa, MD, MBA, executive editor of JAMA, in another accompanying editorial.
“These deaths reflect a true measure of the human cost of the Great Pandemic of 2020,” they add.
The study from Emanuel and Bilinski was notable for calculating the excess COVID-19 and all-cause mortality to Sept. 2020, they note. “After the initial peak in early spring, US death rates from COVID-19 and from all causes remained higher than rates in countries with high COVID-19 mortality.”
“Few people will forget the Great Pandemic of 2020, where and how they lived, how it substantially changed their lives, and for many, the profound human toll it has taken,” Bauchner and Fontanarosa write.
The study by Woolf and colleagues was supported by National Center for Advancing Translational Sciences, the National Institute on Aging, and the National Institute of Allergy and Infectious Diseases. The study by Bilinski and Emanuel was partially funded by the Colton Foundation. Woolf, Emanuel, Fineberg, Bauchner, and Fontanarosa have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Fourteen-day sports hiatus recommended for children after COVID-19
Children should not return to sports for 14 days after exposure to COVID-19, and those with moderate symptoms should undergo an electrocardiogram before returning, according to the American Academy of Pediatrics.
said Susannah Briskin, MD, a pediatric sports medicine specialist at Rainbow Babies and Children’s Hospital in Cleveland.
“There has been emerging evidence about cases of myocarditis occurring in athletes, including athletes who are asymptomatic with COVID-19,” she said in an interview.
The update aligns the AAP recommendations with those from the American College of Cardiologists, she added.
Recent imaging studies have turned up signs of myocarditis in athletes recovering from mild or asymptomatic cases of COVID-19 and have prompted calls for clearer guidelines about imaging studies and return to play.
Viral myocarditis poses a risk to athletes because it can lead to potentially fatal arrhythmias, Dr. Briskin said.
Although children benefit from participating in sports, these activities also put them at risk of contracting COVID-19 and spreading it to others, the guidance noted.
To balance the risks and benefits, the academy proposed guidelines that vary depending on the severity of the presentation.
In the first category are patients with a severe presentation (hypotension, arrhythmias, need for intubation or extracorporeal membrane oxygenation support, kidney or cardiac failure) or with multisystem inflammatory syndrome. Clinicians should treat these patients as though they have myocarditis. Patients should be restricted from engaging in sports and other exercise for 3-6 months, the guidance stated.
The primary care physician and “appropriate pediatric medical subspecialist, preferably in consultation with a pediatric cardiologist,” should clear them before they return to activities. In examining patients for return to play, clinicians should focus on cardiac symptoms, including chest pain, shortness of breath, fatigue, palpitations, or syncope, the guidance said.
In another category are patients with cardiac symptoms, those with concerning findings on examination, and those with moderate symptoms of COVID-19, including prolonged fever. These patients should undergo an ECG and possibly be referred to a pediatric cardiologist, the guidelines said. These symptoms must be absent for at least 14 days before these patients can return to sports, and the athletes should obtain clearance from their primary care physicians before they resume.
In a third category are patients who have been infected with SARS-CoV-2 or who have had close contact with someone who was infected but who have not themselves experienced symptoms. These athletes should refrain from sports for at least 14 days, the guidelines said.
Children who don’t fall into any of these categories should not be tested for the virus or antibodies to it before participation in sports, the academy said.
The guidelines don’t vary depending on the sport. But the academy has issued separate guidance for parents and guardians to help them evaluate the risk for COVID-19 transmission by sport.
Athletes participating in “sports that have greater amount of contact time or proximity to people would be at higher risk for contracting COVID-19,” Dr. Briskin said. “But I think that’s all fairly common sense, given the recommendations for non–sport-related activity just in terms of social distancing and masking.”
The new guidance called on sports organizers to minimize contact by, for example, modifying drills and conditioning. It recommended that athletes wear masks except during vigorous exercise or when participating in water sports, as well as in other circumstances in which the mask could become a safety hazard.
They also recommended using handwashing stations or hand sanitizer, avoiding contact with shared surfaces, and avoiding small rooms and areas with poor ventilation.
Dr. Briskin disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Children should not return to sports for 14 days after exposure to COVID-19, and those with moderate symptoms should undergo an electrocardiogram before returning, according to the American Academy of Pediatrics.
said Susannah Briskin, MD, a pediatric sports medicine specialist at Rainbow Babies and Children’s Hospital in Cleveland.
“There has been emerging evidence about cases of myocarditis occurring in athletes, including athletes who are asymptomatic with COVID-19,” she said in an interview.
The update aligns the AAP recommendations with those from the American College of Cardiologists, she added.
Recent imaging studies have turned up signs of myocarditis in athletes recovering from mild or asymptomatic cases of COVID-19 and have prompted calls for clearer guidelines about imaging studies and return to play.
Viral myocarditis poses a risk to athletes because it can lead to potentially fatal arrhythmias, Dr. Briskin said.
Although children benefit from participating in sports, these activities also put them at risk of contracting COVID-19 and spreading it to others, the guidance noted.
To balance the risks and benefits, the academy proposed guidelines that vary depending on the severity of the presentation.
In the first category are patients with a severe presentation (hypotension, arrhythmias, need for intubation or extracorporeal membrane oxygenation support, kidney or cardiac failure) or with multisystem inflammatory syndrome. Clinicians should treat these patients as though they have myocarditis. Patients should be restricted from engaging in sports and other exercise for 3-6 months, the guidance stated.
The primary care physician and “appropriate pediatric medical subspecialist, preferably in consultation with a pediatric cardiologist,” should clear them before they return to activities. In examining patients for return to play, clinicians should focus on cardiac symptoms, including chest pain, shortness of breath, fatigue, palpitations, or syncope, the guidance said.
In another category are patients with cardiac symptoms, those with concerning findings on examination, and those with moderate symptoms of COVID-19, including prolonged fever. These patients should undergo an ECG and possibly be referred to a pediatric cardiologist, the guidelines said. These symptoms must be absent for at least 14 days before these patients can return to sports, and the athletes should obtain clearance from their primary care physicians before they resume.
In a third category are patients who have been infected with SARS-CoV-2 or who have had close contact with someone who was infected but who have not themselves experienced symptoms. These athletes should refrain from sports for at least 14 days, the guidelines said.
Children who don’t fall into any of these categories should not be tested for the virus or antibodies to it before participation in sports, the academy said.
The guidelines don’t vary depending on the sport. But the academy has issued separate guidance for parents and guardians to help them evaluate the risk for COVID-19 transmission by sport.
Athletes participating in “sports that have greater amount of contact time or proximity to people would be at higher risk for contracting COVID-19,” Dr. Briskin said. “But I think that’s all fairly common sense, given the recommendations for non–sport-related activity just in terms of social distancing and masking.”
The new guidance called on sports organizers to minimize contact by, for example, modifying drills and conditioning. It recommended that athletes wear masks except during vigorous exercise or when participating in water sports, as well as in other circumstances in which the mask could become a safety hazard.
They also recommended using handwashing stations or hand sanitizer, avoiding contact with shared surfaces, and avoiding small rooms and areas with poor ventilation.
Dr. Briskin disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Children should not return to sports for 14 days after exposure to COVID-19, and those with moderate symptoms should undergo an electrocardiogram before returning, according to the American Academy of Pediatrics.
said Susannah Briskin, MD, a pediatric sports medicine specialist at Rainbow Babies and Children’s Hospital in Cleveland.
“There has been emerging evidence about cases of myocarditis occurring in athletes, including athletes who are asymptomatic with COVID-19,” she said in an interview.
The update aligns the AAP recommendations with those from the American College of Cardiologists, she added.
Recent imaging studies have turned up signs of myocarditis in athletes recovering from mild or asymptomatic cases of COVID-19 and have prompted calls for clearer guidelines about imaging studies and return to play.
Viral myocarditis poses a risk to athletes because it can lead to potentially fatal arrhythmias, Dr. Briskin said.
Although children benefit from participating in sports, these activities also put them at risk of contracting COVID-19 and spreading it to others, the guidance noted.
To balance the risks and benefits, the academy proposed guidelines that vary depending on the severity of the presentation.
In the first category are patients with a severe presentation (hypotension, arrhythmias, need for intubation or extracorporeal membrane oxygenation support, kidney or cardiac failure) or with multisystem inflammatory syndrome. Clinicians should treat these patients as though they have myocarditis. Patients should be restricted from engaging in sports and other exercise for 3-6 months, the guidance stated.
The primary care physician and “appropriate pediatric medical subspecialist, preferably in consultation with a pediatric cardiologist,” should clear them before they return to activities. In examining patients for return to play, clinicians should focus on cardiac symptoms, including chest pain, shortness of breath, fatigue, palpitations, or syncope, the guidance said.
In another category are patients with cardiac symptoms, those with concerning findings on examination, and those with moderate symptoms of COVID-19, including prolonged fever. These patients should undergo an ECG and possibly be referred to a pediatric cardiologist, the guidelines said. These symptoms must be absent for at least 14 days before these patients can return to sports, and the athletes should obtain clearance from their primary care physicians before they resume.
In a third category are patients who have been infected with SARS-CoV-2 or who have had close contact with someone who was infected but who have not themselves experienced symptoms. These athletes should refrain from sports for at least 14 days, the guidelines said.
Children who don’t fall into any of these categories should not be tested for the virus or antibodies to it before participation in sports, the academy said.
The guidelines don’t vary depending on the sport. But the academy has issued separate guidance for parents and guardians to help them evaluate the risk for COVID-19 transmission by sport.
Athletes participating in “sports that have greater amount of contact time or proximity to people would be at higher risk for contracting COVID-19,” Dr. Briskin said. “But I think that’s all fairly common sense, given the recommendations for non–sport-related activity just in terms of social distancing and masking.”
The new guidance called on sports organizers to minimize contact by, for example, modifying drills and conditioning. It recommended that athletes wear masks except during vigorous exercise or when participating in water sports, as well as in other circumstances in which the mask could become a safety hazard.
They also recommended using handwashing stations or hand sanitizer, avoiding contact with shared surfaces, and avoiding small rooms and areas with poor ventilation.
Dr. Briskin disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Empagliflozin cut PA pressures in heart failure patients
Elevated pulmonary artery diastolic pressure is “perhaps the best predictor of bad outcomes in patients with heart failure, including hospitalization and death,” and new evidence clearly showed that the sodium-glucose cotransporter 2 (SGLT2) inhibitor empagliflozin cuts this metric in patients by a clinically significant amount, Mikhail Kosiborod, MD, said at the virtual annual scientific meeting of the Heart Failure Society of America.
The evidence he collected from a total of 65 heart failure patients with either reduced or preserved ejection fraction is the first documentation from a randomized, controlled study to show a direct effect by a SGLT2 inhibitor on pulmonary artery (PA) pressures.
Other key findings were that the drop in PA diastolic pressure with empagliflozin treatment compared with placebo became discernible early (within the first 4 weeks on treatment), that the pressure-lowering effect steadily grew over time, and that it showed no link to the intensity of loop diuretic treatment, which held steady during 12 weeks on treatment and 13 weeks of overall monitoring.
The study’s primary endpoint was the change from baseline in PA diastolic pressure after 12 weeks on treatment. The 31 patients who completed the full 12-week course had an average drop in their PA diastolic pressure of about 1.5 mm Hg, compared with 28 patients who completed 12 weeks on placebo. Average PA diastolic pressure at baseline was about 21 mm Hg in both treatment arms, and on treatment this fell by more than 0.5 mm Hg among those who received empagliflozin and rose by close to 1 mm Hg among control patients.
“There appears to be a direct effect of empagliflozin on pulmonary artery pressure that’s not been previously demonstrated” by an SGLT2 inhibitor, Dr. Kosiborod said. “I think this is one mechanism of action” for this drug class. “If you control pulmonary artery filling pressures you can prevent hospitalizations and deaths.”
Small reductions matter
“Small pressure differences are particularly important for pulmonary hypertension,” commented Lynne W. Stevenson, MD, professor of medicine at Vanderbilt University Medical Center in Nashville, Tenn., and the report’s designated discussant.
“In the Vanderbilt heart failure database, patients with a pulmonary artery mean pressure of 20-24 mm Hg had 30% higher mortality than patients with lower pressures,” Dr. Stevenson noted. “This has led to a new definition of pulmonary hypertension, a mean pulmonary artery pressure above at or above 20 mm Hg.”
In Dr. Kosiborod’s study, patients began with an average PA mean pressure of about 30 mm Hg, and empagliflozin treatment led to a reduction in this metric with about the same magnitude as its effect on PA diastolic pressure. Empagliflozin also produced a similar reduction in average PA systolic pressure.
A study built on ambulatory PA monitoring
The results “also provide more proof for the concept of ambulatory hemodynamic monitoring” in patients with heart failure to monitor their status, she added. The study enrolled only patients who had already received a CardioMEMS implant as part of their routine care. This device allows for frequent, noninvasive monitoring of PA pressures. Researchers collected PA pressure data from patients twice daily for the entire 13-week study.
The EMBRACE HF (Empagliflozin Impact on Hemodynamics in Patients With Heart Failure) study enrolled patients with established heart failure, a CardioMEMS implant, and New York Heart Association class II-IV symptoms at any of eight U.S. centers. Patients averaged about 65 years old, and slightly more than half had class III disease, which denotes marked limitation of physical activity.
Despite the brief treatment period, patients who received empagliflozin showed other evidence of benefit including a trend toward improved quality of life scores, reduced levels of two different forms of brain natriuretic peptide, and significant weight loss, compared with controls, that averaged 2.4 kg.
The mechanism by which empagliflozin and other drugs in its class might lower PA filling pressures is unclear, but Dr. Kosiborod stressed that the consistent level of loop diuretic use during the study seems to rule out a diuretic effect from the SGLT2 inhibitor as having a role. A pulmonary vasculature effect is “much more likely,” perhaps mediated through modified endothelial function and vasodilation, he suggested.
EMBRACE HF was funded by Boehringer Ingelheim, the company that markets empagliflozin (Jardiance) along with Eli Lilly. Dr. Kosiborod has received research support and honoraria from Boehringer Ingelheim, and he has received honoraria from several other companies. Dr. Stevenson had no disclosures.
Elevated pulmonary artery diastolic pressure is “perhaps the best predictor of bad outcomes in patients with heart failure, including hospitalization and death,” and new evidence clearly showed that the sodium-glucose cotransporter 2 (SGLT2) inhibitor empagliflozin cuts this metric in patients by a clinically significant amount, Mikhail Kosiborod, MD, said at the virtual annual scientific meeting of the Heart Failure Society of America.
The evidence he collected from a total of 65 heart failure patients with either reduced or preserved ejection fraction is the first documentation from a randomized, controlled study to show a direct effect by a SGLT2 inhibitor on pulmonary artery (PA) pressures.
Other key findings were that the drop in PA diastolic pressure with empagliflozin treatment compared with placebo became discernible early (within the first 4 weeks on treatment), that the pressure-lowering effect steadily grew over time, and that it showed no link to the intensity of loop diuretic treatment, which held steady during 12 weeks on treatment and 13 weeks of overall monitoring.
The study’s primary endpoint was the change from baseline in PA diastolic pressure after 12 weeks on treatment. The 31 patients who completed the full 12-week course had an average drop in their PA diastolic pressure of about 1.5 mm Hg, compared with 28 patients who completed 12 weeks on placebo. Average PA diastolic pressure at baseline was about 21 mm Hg in both treatment arms, and on treatment this fell by more than 0.5 mm Hg among those who received empagliflozin and rose by close to 1 mm Hg among control patients.
“There appears to be a direct effect of empagliflozin on pulmonary artery pressure that’s not been previously demonstrated” by an SGLT2 inhibitor, Dr. Kosiborod said. “I think this is one mechanism of action” for this drug class. “If you control pulmonary artery filling pressures you can prevent hospitalizations and deaths.”
Small reductions matter
“Small pressure differences are particularly important for pulmonary hypertension,” commented Lynne W. Stevenson, MD, professor of medicine at Vanderbilt University Medical Center in Nashville, Tenn., and the report’s designated discussant.
“In the Vanderbilt heart failure database, patients with a pulmonary artery mean pressure of 20-24 mm Hg had 30% higher mortality than patients with lower pressures,” Dr. Stevenson noted. “This has led to a new definition of pulmonary hypertension, a mean pulmonary artery pressure above at or above 20 mm Hg.”
In Dr. Kosiborod’s study, patients began with an average PA mean pressure of about 30 mm Hg, and empagliflozin treatment led to a reduction in this metric with about the same magnitude as its effect on PA diastolic pressure. Empagliflozin also produced a similar reduction in average PA systolic pressure.
A study built on ambulatory PA monitoring
The results “also provide more proof for the concept of ambulatory hemodynamic monitoring” in patients with heart failure to monitor their status, she added. The study enrolled only patients who had already received a CardioMEMS implant as part of their routine care. This device allows for frequent, noninvasive monitoring of PA pressures. Researchers collected PA pressure data from patients twice daily for the entire 13-week study.
The EMBRACE HF (Empagliflozin Impact on Hemodynamics in Patients With Heart Failure) study enrolled patients with established heart failure, a CardioMEMS implant, and New York Heart Association class II-IV symptoms at any of eight U.S. centers. Patients averaged about 65 years old, and slightly more than half had class III disease, which denotes marked limitation of physical activity.
Despite the brief treatment period, patients who received empagliflozin showed other evidence of benefit including a trend toward improved quality of life scores, reduced levels of two different forms of brain natriuretic peptide, and significant weight loss, compared with controls, that averaged 2.4 kg.
The mechanism by which empagliflozin and other drugs in its class might lower PA filling pressures is unclear, but Dr. Kosiborod stressed that the consistent level of loop diuretic use during the study seems to rule out a diuretic effect from the SGLT2 inhibitor as having a role. A pulmonary vasculature effect is “much more likely,” perhaps mediated through modified endothelial function and vasodilation, he suggested.
EMBRACE HF was funded by Boehringer Ingelheim, the company that markets empagliflozin (Jardiance) along with Eli Lilly. Dr. Kosiborod has received research support and honoraria from Boehringer Ingelheim, and he has received honoraria from several other companies. Dr. Stevenson had no disclosures.
Elevated pulmonary artery diastolic pressure is “perhaps the best predictor of bad outcomes in patients with heart failure, including hospitalization and death,” and new evidence clearly showed that the sodium-glucose cotransporter 2 (SGLT2) inhibitor empagliflozin cuts this metric in patients by a clinically significant amount, Mikhail Kosiborod, MD, said at the virtual annual scientific meeting of the Heart Failure Society of America.
The evidence he collected from a total of 65 heart failure patients with either reduced or preserved ejection fraction is the first documentation from a randomized, controlled study to show a direct effect by a SGLT2 inhibitor on pulmonary artery (PA) pressures.
Other key findings were that the drop in PA diastolic pressure with empagliflozin treatment compared with placebo became discernible early (within the first 4 weeks on treatment), that the pressure-lowering effect steadily grew over time, and that it showed no link to the intensity of loop diuretic treatment, which held steady during 12 weeks on treatment and 13 weeks of overall monitoring.
The study’s primary endpoint was the change from baseline in PA diastolic pressure after 12 weeks on treatment. The 31 patients who completed the full 12-week course had an average drop in their PA diastolic pressure of about 1.5 mm Hg, compared with 28 patients who completed 12 weeks on placebo. Average PA diastolic pressure at baseline was about 21 mm Hg in both treatment arms, and on treatment this fell by more than 0.5 mm Hg among those who received empagliflozin and rose by close to 1 mm Hg among control patients.
“There appears to be a direct effect of empagliflozin on pulmonary artery pressure that’s not been previously demonstrated” by an SGLT2 inhibitor, Dr. Kosiborod said. “I think this is one mechanism of action” for this drug class. “If you control pulmonary artery filling pressures you can prevent hospitalizations and deaths.”
Small reductions matter
“Small pressure differences are particularly important for pulmonary hypertension,” commented Lynne W. Stevenson, MD, professor of medicine at Vanderbilt University Medical Center in Nashville, Tenn., and the report’s designated discussant.
“In the Vanderbilt heart failure database, patients with a pulmonary artery mean pressure of 20-24 mm Hg had 30% higher mortality than patients with lower pressures,” Dr. Stevenson noted. “This has led to a new definition of pulmonary hypertension, a mean pulmonary artery pressure above at or above 20 mm Hg.”
In Dr. Kosiborod’s study, patients began with an average PA mean pressure of about 30 mm Hg, and empagliflozin treatment led to a reduction in this metric with about the same magnitude as its effect on PA diastolic pressure. Empagliflozin also produced a similar reduction in average PA systolic pressure.
A study built on ambulatory PA monitoring
The results “also provide more proof for the concept of ambulatory hemodynamic monitoring” in patients with heart failure to monitor their status, she added. The study enrolled only patients who had already received a CardioMEMS implant as part of their routine care. This device allows for frequent, noninvasive monitoring of PA pressures. Researchers collected PA pressure data from patients twice daily for the entire 13-week study.
The EMBRACE HF (Empagliflozin Impact on Hemodynamics in Patients With Heart Failure) study enrolled patients with established heart failure, a CardioMEMS implant, and New York Heart Association class II-IV symptoms at any of eight U.S. centers. Patients averaged about 65 years old, and slightly more than half had class III disease, which denotes marked limitation of physical activity.
Despite the brief treatment period, patients who received empagliflozin showed other evidence of benefit including a trend toward improved quality of life scores, reduced levels of two different forms of brain natriuretic peptide, and significant weight loss, compared with controls, that averaged 2.4 kg.
The mechanism by which empagliflozin and other drugs in its class might lower PA filling pressures is unclear, but Dr. Kosiborod stressed that the consistent level of loop diuretic use during the study seems to rule out a diuretic effect from the SGLT2 inhibitor as having a role. A pulmonary vasculature effect is “much more likely,” perhaps mediated through modified endothelial function and vasodilation, he suggested.
EMBRACE HF was funded by Boehringer Ingelheim, the company that markets empagliflozin (Jardiance) along with Eli Lilly. Dr. Kosiborod has received research support and honoraria from Boehringer Ingelheim, and he has received honoraria from several other companies. Dr. Stevenson had no disclosures.
FROM HFSA 2020
Remdesivir effective, well-tolerated in final trial report
Drug beats placebo across multiple endpoints in COVID-19 patients
In May 2020, remdesivir received Food and Drug Administration approval for emergency treatment of severe COVID-19 on the basis of a preliminary report on this trial. In August 2020, the FDA expanded the indication to include all hospitalized adult and pediatric patients with suspected or laboratory-confirmed COVID-19 infection irrespective of severity.
“Our findings were consistent with the findings of the preliminary report: a 10-day course of remdesivir was superior to placebo in the treatment of hospitalized patients with COVID-19,” reported a team of investigators led by John H. Beigel, MD, of the Division of Microbiology and Infectious Diseases at the National Institute of Allergy and Infectious Diseases, in the New England Journal of Medicine.
The drug’s broadened indication was not based on the ACTT-1 trial, according to Dr. Beigel. “Other data have demonstrated that remdesivir shortens recovery in patients with lower acuity. In our study, evidence of pneumonia was an enrollment requirement,” he explained in an interview.
In the newly published final ACTT-1 data, the median time to recovery was 10 days for those on active therapy versus 15 days for those randomized to placebo. With a rate ratio of 1.29 (P less than .001), this translated to a recovery that was about one third faster.
In this final report, remdesivir’s significant advantage over placebo regarding the trial’s primary endpoint was reinforced by efficacy on multiple secondary endpoints.
This benefits on multiple secondary endpoints included a 50% greater odds ratio (OR, 1.5; 95% CI, 1.2-1.9) of significant clinical improvement by day 15 after adjustment for baseline severity, a shorter initial length of hospital stay (12 vs. 17 days) and fewer days on oxygen supplementation (13 vs. 21 days) for the subgroup of patients on oxygen at enrollment.
Although the numerically lower mortality in the remdesivir arm (6.75 vs. 11.9%) did not reach statistical significance, Dr. Beigel said, “mortality was moving in the same direction as the other key endpoints.”
According to the study investigators, the types of rates of adverse events on remdesivir, which inhibits viral replication, “were generally similar in the remdesivir and placebo groups.”
In ACTT-1, 1,062 patients were randomized to remdesivir (200 mg loading dose followed by 100 mg daily for up to 9 days) or placebo. Patients were enrolled at study sites in North America, Europe, and Asia.
The data of ACTT-1 confirm a benefit from remdesivir in hospitalized COVID-19 patients with severe disease, but Dr. Beigel said he agrees with the current FDA indication that supports treatment in any hospitalized COVID-19 patient.
“We saw bigger benefits in patients with more severe infections. The benefits are not as large in patients with mild disease, but I think remdesivir should be considered in any hospitalized patient,” Dr. Beigel said.
This point of view is shared.
“I would give this drug to anyone in the hospital infected with COVID-19 assuming there was an ample supply and no need for rationing,” said Donna E. Sweet, MD, professor of internal medicine, University of Kansas, Wichita. She noted that this study has implications for hospital and hospital staff, as well as for patients.
“This type of reduction in recovery time means a reduction in potential exposures to hospital staff, a reduced need for PPE [personal protective equipment], and it will free up beds in the ICU [intensive care unit],” said Dr. Sweet, who also serves as an editorial advisory board member for Internal Medicine News.
An infectious disease specialist at the University of Minnesota also considers remdesivir to have an important role for conserving resources that deserves emphasis.
The reduction in time to recovery “is of benefit to the health system by maintaining hospital bed capacity,” said David R. Boulware, MD, professor of medicine at the University of Minnesota, Minneapolis.
According to his reading of the available data, including those from ACTT-1, the benefit appears to be greatest in those with a moderate degree of illness, which he defined as “sick enough to be hospitalized and require oxygen, yet not severely sick [and] requiring a ventilator or [extracorporeal membrane oxygenation].”
This does not preclude a benefit in those with more severe or milder disease, but patients with mild disease “are likely to recover regardless – or despite – whatever therapy they receive,” he said.
Dr. Beigel, the principal investigator of this trial, reports no potential conflicts of interest.
SOURCE: Beigel JH et al. N Engl J Med. 2020 Oct 8. doi: 10.1056/NEJMoa2007764.
Drug beats placebo across multiple endpoints in COVID-19 patients
Drug beats placebo across multiple endpoints in COVID-19 patients
In May 2020, remdesivir received Food and Drug Administration approval for emergency treatment of severe COVID-19 on the basis of a preliminary report on this trial. In August 2020, the FDA expanded the indication to include all hospitalized adult and pediatric patients with suspected or laboratory-confirmed COVID-19 infection irrespective of severity.
“Our findings were consistent with the findings of the preliminary report: a 10-day course of remdesivir was superior to placebo in the treatment of hospitalized patients with COVID-19,” reported a team of investigators led by John H. Beigel, MD, of the Division of Microbiology and Infectious Diseases at the National Institute of Allergy and Infectious Diseases, in the New England Journal of Medicine.
The drug’s broadened indication was not based on the ACTT-1 trial, according to Dr. Beigel. “Other data have demonstrated that remdesivir shortens recovery in patients with lower acuity. In our study, evidence of pneumonia was an enrollment requirement,” he explained in an interview.
In the newly published final ACTT-1 data, the median time to recovery was 10 days for those on active therapy versus 15 days for those randomized to placebo. With a rate ratio of 1.29 (P less than .001), this translated to a recovery that was about one third faster.
In this final report, remdesivir’s significant advantage over placebo regarding the trial’s primary endpoint was reinforced by efficacy on multiple secondary endpoints.
This benefits on multiple secondary endpoints included a 50% greater odds ratio (OR, 1.5; 95% CI, 1.2-1.9) of significant clinical improvement by day 15 after adjustment for baseline severity, a shorter initial length of hospital stay (12 vs. 17 days) and fewer days on oxygen supplementation (13 vs. 21 days) for the subgroup of patients on oxygen at enrollment.
Although the numerically lower mortality in the remdesivir arm (6.75 vs. 11.9%) did not reach statistical significance, Dr. Beigel said, “mortality was moving in the same direction as the other key endpoints.”
According to the study investigators, the types of rates of adverse events on remdesivir, which inhibits viral replication, “were generally similar in the remdesivir and placebo groups.”
In ACTT-1, 1,062 patients were randomized to remdesivir (200 mg loading dose followed by 100 mg daily for up to 9 days) or placebo. Patients were enrolled at study sites in North America, Europe, and Asia.
The data of ACTT-1 confirm a benefit from remdesivir in hospitalized COVID-19 patients with severe disease, but Dr. Beigel said he agrees with the current FDA indication that supports treatment in any hospitalized COVID-19 patient.
“We saw bigger benefits in patients with more severe infections. The benefits are not as large in patients with mild disease, but I think remdesivir should be considered in any hospitalized patient,” Dr. Beigel said.
This point of view is shared.
“I would give this drug to anyone in the hospital infected with COVID-19 assuming there was an ample supply and no need for rationing,” said Donna E. Sweet, MD, professor of internal medicine, University of Kansas, Wichita. She noted that this study has implications for hospital and hospital staff, as well as for patients.
“This type of reduction in recovery time means a reduction in potential exposures to hospital staff, a reduced need for PPE [personal protective equipment], and it will free up beds in the ICU [intensive care unit],” said Dr. Sweet, who also serves as an editorial advisory board member for Internal Medicine News.
An infectious disease specialist at the University of Minnesota also considers remdesivir to have an important role for conserving resources that deserves emphasis.
The reduction in time to recovery “is of benefit to the health system by maintaining hospital bed capacity,” said David R. Boulware, MD, professor of medicine at the University of Minnesota, Minneapolis.
According to his reading of the available data, including those from ACTT-1, the benefit appears to be greatest in those with a moderate degree of illness, which he defined as “sick enough to be hospitalized and require oxygen, yet not severely sick [and] requiring a ventilator or [extracorporeal membrane oxygenation].”
This does not preclude a benefit in those with more severe or milder disease, but patients with mild disease “are likely to recover regardless – or despite – whatever therapy they receive,” he said.
Dr. Beigel, the principal investigator of this trial, reports no potential conflicts of interest.
SOURCE: Beigel JH et al. N Engl J Med. 2020 Oct 8. doi: 10.1056/NEJMoa2007764.
In May 2020, remdesivir received Food and Drug Administration approval for emergency treatment of severe COVID-19 on the basis of a preliminary report on this trial. In August 2020, the FDA expanded the indication to include all hospitalized adult and pediatric patients with suspected or laboratory-confirmed COVID-19 infection irrespective of severity.
“Our findings were consistent with the findings of the preliminary report: a 10-day course of remdesivir was superior to placebo in the treatment of hospitalized patients with COVID-19,” reported a team of investigators led by John H. Beigel, MD, of the Division of Microbiology and Infectious Diseases at the National Institute of Allergy and Infectious Diseases, in the New England Journal of Medicine.
The drug’s broadened indication was not based on the ACTT-1 trial, according to Dr. Beigel. “Other data have demonstrated that remdesivir shortens recovery in patients with lower acuity. In our study, evidence of pneumonia was an enrollment requirement,” he explained in an interview.
In the newly published final ACTT-1 data, the median time to recovery was 10 days for those on active therapy versus 15 days for those randomized to placebo. With a rate ratio of 1.29 (P less than .001), this translated to a recovery that was about one third faster.
In this final report, remdesivir’s significant advantage over placebo regarding the trial’s primary endpoint was reinforced by efficacy on multiple secondary endpoints.
This benefits on multiple secondary endpoints included a 50% greater odds ratio (OR, 1.5; 95% CI, 1.2-1.9) of significant clinical improvement by day 15 after adjustment for baseline severity, a shorter initial length of hospital stay (12 vs. 17 days) and fewer days on oxygen supplementation (13 vs. 21 days) for the subgroup of patients on oxygen at enrollment.
Although the numerically lower mortality in the remdesivir arm (6.75 vs. 11.9%) did not reach statistical significance, Dr. Beigel said, “mortality was moving in the same direction as the other key endpoints.”
According to the study investigators, the types of rates of adverse events on remdesivir, which inhibits viral replication, “were generally similar in the remdesivir and placebo groups.”
In ACTT-1, 1,062 patients were randomized to remdesivir (200 mg loading dose followed by 100 mg daily for up to 9 days) or placebo. Patients were enrolled at study sites in North America, Europe, and Asia.
The data of ACTT-1 confirm a benefit from remdesivir in hospitalized COVID-19 patients with severe disease, but Dr. Beigel said he agrees with the current FDA indication that supports treatment in any hospitalized COVID-19 patient.
“We saw bigger benefits in patients with more severe infections. The benefits are not as large in patients with mild disease, but I think remdesivir should be considered in any hospitalized patient,” Dr. Beigel said.
This point of view is shared.
“I would give this drug to anyone in the hospital infected with COVID-19 assuming there was an ample supply and no need for rationing,” said Donna E. Sweet, MD, professor of internal medicine, University of Kansas, Wichita. She noted that this study has implications for hospital and hospital staff, as well as for patients.
“This type of reduction in recovery time means a reduction in potential exposures to hospital staff, a reduced need for PPE [personal protective equipment], and it will free up beds in the ICU [intensive care unit],” said Dr. Sweet, who also serves as an editorial advisory board member for Internal Medicine News.
An infectious disease specialist at the University of Minnesota also considers remdesivir to have an important role for conserving resources that deserves emphasis.
The reduction in time to recovery “is of benefit to the health system by maintaining hospital bed capacity,” said David R. Boulware, MD, professor of medicine at the University of Minnesota, Minneapolis.
According to his reading of the available data, including those from ACTT-1, the benefit appears to be greatest in those with a moderate degree of illness, which he defined as “sick enough to be hospitalized and require oxygen, yet not severely sick [and] requiring a ventilator or [extracorporeal membrane oxygenation].”
This does not preclude a benefit in those with more severe or milder disease, but patients with mild disease “are likely to recover regardless – or despite – whatever therapy they receive,” he said.
Dr. Beigel, the principal investigator of this trial, reports no potential conflicts of interest.
SOURCE: Beigel JH et al. N Engl J Med. 2020 Oct 8. doi: 10.1056/NEJMoa2007764.
More data on impact of corticosteroids on COVID-19 mortality in patients with COPD
, a study of almost 1 million individuals in the United Kingdom has shown.
Patients with chronic obstructive pulmonary disease or asthma who used ICS on a regular basis were more likely to die from COVID-19 than COPD or asthma patients who were prescribed non-ICS therapies, reported co-lead author Anna Schultze, PhD, of London School of Hygiene & Tropical Medicine and colleagues.
Of note, the increased risk of death among ICS users likely stemmed from greater severity of preexisting chronic respiratory conditions, instead of directly from ICS usage, which has little apparent impact on COVID-19 mortality, the investigators wrote in Lancet Respiratory Medicine.
These findings conflict with a hypothesis proposed early in the pandemic: that ICS may protect individuals from SARS-CoV-2 infection and poor outcomes with COVID-19.
According to Megan Conroy, MD, of the department of internal medicine at the Ohio State University Wexner Medical Center, Columbus, this hypothesis was based on some unexpected epidemiological findings.
“In general, we tend to think people with underlying lung disease – like COPD or asthma – to be at higher risk for severe forms of lower respiratory tract infections,” Dr. Conroy said. “Somewhat surprisingly, early data in the pandemic showed patients with COPD and asthma [were] underrepresented [among patients with COVID] when compared to the prevalence of these diseases in the population.”
This raised the possibility of an incidental protective effect from regular ICS therapy, which “had some strong theoretic pathophysiologic basis,” Dr. Conroy said, referring to research that demonstrated ICS-mediated downregulation of SARS-CoV-2 entry receptors ACE2 and TMPRSS2.
Dr. Schultze and colleagues noted that investigators for two ongoing randomized controlled trials (NCT04331054, NCT04330586) are studying ICS as an intervention for COVID-19; but neither trial includes individuals already taking ICS for chronic respiratory disease.
The present observational study therefore aimed to assess mortality risk within this population. Data were drawn from electronic health records and a U.K. national mortality database, with follow-up ranging from March 1 to May 6, 2020. Eligibility required a relevant prescription within 4 months of first follow-up. In the COPD group, patients were prescribed a long-acting beta agonist plus a long-acting muscarinic antagonist (LABA–LAMA), LABA alone, LABA plus ICS, LABA–LAMA plus ICS, or ICS alone (if prescribed LABA within 4 months).
In the asthma group, patients received low/medium-dose ICS, high-dose ICS, or a short-acting beta agonist (SABA) alone. Patients with COPD were at least 35 years of age, while those with asthma were 18 years or older. Hazard ratios were adjusted for a variety of covariates, including respiratory disease–exacerbation history, age, sex, body mass index, hypertension, diabetes, and others.
These eligibility criteria returned 148,557 patients with COPD and 818,490 with asthma.
Patients with COPD who were prescribed ICS plus LABA-LAMA or ICS plus LABA had an increased risk of COVID-19-related death, compared with those who did not receive ICS (adjusted hazard ratio, 1.39; 95% confidence interval, 1.10-1.76). Separate analyses of patients who received a triple combination (LABA–LAMA plus ICS) versus those who took a dual combination (LABA plus ICS) showed that triple-combination therapy was significantly associated with increased COVID-19-related mortality (aHR, 1.43; 95% CI, 1.12-1.83), while dual-combination therapy was less so (aHR, 1.29; 95% CI, 0.96-1.74). Non–COVID-19–related mortality was significantly increased for all COPD patients who were prescribed ICS, with or without adjustment for covariates.
Asthma patients prescribed high-dose ICS instead of SABA alone had a slightly greater risk of COVID-19–related death, based on an adjusted hazard ratio of 1.55 (95% CI, 1.10-2.18). Those with asthma who received low/medium–dose ICS demonstrated a slight trend toward increased mortality risk, but this was not significant (aHR, 1.14; 95% CI, 0.85-1.54). ICS usage in the asthma group was not linked with a significant increase in non–COVID-19–related death.
“In summary, we found no evidence of a beneficial effect of regular ICS use among people with COPD and asthma on COVID-19–related mortality,” the investigators concluded.
In agreement with the investigators, Dr. Conroy said that the increased mortality rate among ICS users should not be misconstrued as a medication-related risk.
“While the study found that those with COPD or asthma taking ICS and high-dose ICS were at an increased risk of death, this could easily be explained by the likelihood that those are the patients who are more likely to have more severe underlying lung disease,” Dr. Conroy said. “While this observational study did attempt to control for exacerbation history, the ability to do so by electronic health records data is certainly imperfect.”
With this in mind, patients with chronic respiratory disease should be encouraged to adhere to their usual treatment regimen, Dr. Conroy added.
“There isn’t evidence to increase or decrease medications just because of the pandemic,” she said. “A patient with asthma or COPD should continue to take the medications that are needed to achieve good control of their lung disease.”
The study was funded by the U.K. Medical Research Council. The investigators reported additional relationships with the Wellcome Trust, the Good Thinking Foundation, the Laura and John Arnold Foundation, and others. Dr. Conroy reported no conflicts of interest.
SOURCE: Schultze A et al. Lancet Respir Med. 2020 Sep 24. doi: 10.1016/ S2213-2600(20)30415-X.
, a study of almost 1 million individuals in the United Kingdom has shown.
Patients with chronic obstructive pulmonary disease or asthma who used ICS on a regular basis were more likely to die from COVID-19 than COPD or asthma patients who were prescribed non-ICS therapies, reported co-lead author Anna Schultze, PhD, of London School of Hygiene & Tropical Medicine and colleagues.
Of note, the increased risk of death among ICS users likely stemmed from greater severity of preexisting chronic respiratory conditions, instead of directly from ICS usage, which has little apparent impact on COVID-19 mortality, the investigators wrote in Lancet Respiratory Medicine.
These findings conflict with a hypothesis proposed early in the pandemic: that ICS may protect individuals from SARS-CoV-2 infection and poor outcomes with COVID-19.
According to Megan Conroy, MD, of the department of internal medicine at the Ohio State University Wexner Medical Center, Columbus, this hypothesis was based on some unexpected epidemiological findings.
“In general, we tend to think people with underlying lung disease – like COPD or asthma – to be at higher risk for severe forms of lower respiratory tract infections,” Dr. Conroy said. “Somewhat surprisingly, early data in the pandemic showed patients with COPD and asthma [were] underrepresented [among patients with COVID] when compared to the prevalence of these diseases in the population.”
This raised the possibility of an incidental protective effect from regular ICS therapy, which “had some strong theoretic pathophysiologic basis,” Dr. Conroy said, referring to research that demonstrated ICS-mediated downregulation of SARS-CoV-2 entry receptors ACE2 and TMPRSS2.
Dr. Schultze and colleagues noted that investigators for two ongoing randomized controlled trials (NCT04331054, NCT04330586) are studying ICS as an intervention for COVID-19; but neither trial includes individuals already taking ICS for chronic respiratory disease.
The present observational study therefore aimed to assess mortality risk within this population. Data were drawn from electronic health records and a U.K. national mortality database, with follow-up ranging from March 1 to May 6, 2020. Eligibility required a relevant prescription within 4 months of first follow-up. In the COPD group, patients were prescribed a long-acting beta agonist plus a long-acting muscarinic antagonist (LABA–LAMA), LABA alone, LABA plus ICS, LABA–LAMA plus ICS, or ICS alone (if prescribed LABA within 4 months).
In the asthma group, patients received low/medium-dose ICS, high-dose ICS, or a short-acting beta agonist (SABA) alone. Patients with COPD were at least 35 years of age, while those with asthma were 18 years or older. Hazard ratios were adjusted for a variety of covariates, including respiratory disease–exacerbation history, age, sex, body mass index, hypertension, diabetes, and others.
These eligibility criteria returned 148,557 patients with COPD and 818,490 with asthma.
Patients with COPD who were prescribed ICS plus LABA-LAMA or ICS plus LABA had an increased risk of COVID-19-related death, compared with those who did not receive ICS (adjusted hazard ratio, 1.39; 95% confidence interval, 1.10-1.76). Separate analyses of patients who received a triple combination (LABA–LAMA plus ICS) versus those who took a dual combination (LABA plus ICS) showed that triple-combination therapy was significantly associated with increased COVID-19-related mortality (aHR, 1.43; 95% CI, 1.12-1.83), while dual-combination therapy was less so (aHR, 1.29; 95% CI, 0.96-1.74). Non–COVID-19–related mortality was significantly increased for all COPD patients who were prescribed ICS, with or without adjustment for covariates.
Asthma patients prescribed high-dose ICS instead of SABA alone had a slightly greater risk of COVID-19–related death, based on an adjusted hazard ratio of 1.55 (95% CI, 1.10-2.18). Those with asthma who received low/medium–dose ICS demonstrated a slight trend toward increased mortality risk, but this was not significant (aHR, 1.14; 95% CI, 0.85-1.54). ICS usage in the asthma group was not linked with a significant increase in non–COVID-19–related death.
“In summary, we found no evidence of a beneficial effect of regular ICS use among people with COPD and asthma on COVID-19–related mortality,” the investigators concluded.
In agreement with the investigators, Dr. Conroy said that the increased mortality rate among ICS users should not be misconstrued as a medication-related risk.
“While the study found that those with COPD or asthma taking ICS and high-dose ICS were at an increased risk of death, this could easily be explained by the likelihood that those are the patients who are more likely to have more severe underlying lung disease,” Dr. Conroy said. “While this observational study did attempt to control for exacerbation history, the ability to do so by electronic health records data is certainly imperfect.”
With this in mind, patients with chronic respiratory disease should be encouraged to adhere to their usual treatment regimen, Dr. Conroy added.
“There isn’t evidence to increase or decrease medications just because of the pandemic,” she said. “A patient with asthma or COPD should continue to take the medications that are needed to achieve good control of their lung disease.”
The study was funded by the U.K. Medical Research Council. The investigators reported additional relationships with the Wellcome Trust, the Good Thinking Foundation, the Laura and John Arnold Foundation, and others. Dr. Conroy reported no conflicts of interest.
SOURCE: Schultze A et al. Lancet Respir Med. 2020 Sep 24. doi: 10.1016/ S2213-2600(20)30415-X.
, a study of almost 1 million individuals in the United Kingdom has shown.
Patients with chronic obstructive pulmonary disease or asthma who used ICS on a regular basis were more likely to die from COVID-19 than COPD or asthma patients who were prescribed non-ICS therapies, reported co-lead author Anna Schultze, PhD, of London School of Hygiene & Tropical Medicine and colleagues.
Of note, the increased risk of death among ICS users likely stemmed from greater severity of preexisting chronic respiratory conditions, instead of directly from ICS usage, which has little apparent impact on COVID-19 mortality, the investigators wrote in Lancet Respiratory Medicine.
These findings conflict with a hypothesis proposed early in the pandemic: that ICS may protect individuals from SARS-CoV-2 infection and poor outcomes with COVID-19.
According to Megan Conroy, MD, of the department of internal medicine at the Ohio State University Wexner Medical Center, Columbus, this hypothesis was based on some unexpected epidemiological findings.
“In general, we tend to think people with underlying lung disease – like COPD or asthma – to be at higher risk for severe forms of lower respiratory tract infections,” Dr. Conroy said. “Somewhat surprisingly, early data in the pandemic showed patients with COPD and asthma [were] underrepresented [among patients with COVID] when compared to the prevalence of these diseases in the population.”
This raised the possibility of an incidental protective effect from regular ICS therapy, which “had some strong theoretic pathophysiologic basis,” Dr. Conroy said, referring to research that demonstrated ICS-mediated downregulation of SARS-CoV-2 entry receptors ACE2 and TMPRSS2.
Dr. Schultze and colleagues noted that investigators for two ongoing randomized controlled trials (NCT04331054, NCT04330586) are studying ICS as an intervention for COVID-19; but neither trial includes individuals already taking ICS for chronic respiratory disease.
The present observational study therefore aimed to assess mortality risk within this population. Data were drawn from electronic health records and a U.K. national mortality database, with follow-up ranging from March 1 to May 6, 2020. Eligibility required a relevant prescription within 4 months of first follow-up. In the COPD group, patients were prescribed a long-acting beta agonist plus a long-acting muscarinic antagonist (LABA–LAMA), LABA alone, LABA plus ICS, LABA–LAMA plus ICS, or ICS alone (if prescribed LABA within 4 months).
In the asthma group, patients received low/medium-dose ICS, high-dose ICS, or a short-acting beta agonist (SABA) alone. Patients with COPD were at least 35 years of age, while those with asthma were 18 years or older. Hazard ratios were adjusted for a variety of covariates, including respiratory disease–exacerbation history, age, sex, body mass index, hypertension, diabetes, and others.
These eligibility criteria returned 148,557 patients with COPD and 818,490 with asthma.
Patients with COPD who were prescribed ICS plus LABA-LAMA or ICS plus LABA had an increased risk of COVID-19-related death, compared with those who did not receive ICS (adjusted hazard ratio, 1.39; 95% confidence interval, 1.10-1.76). Separate analyses of patients who received a triple combination (LABA–LAMA plus ICS) versus those who took a dual combination (LABA plus ICS) showed that triple-combination therapy was significantly associated with increased COVID-19-related mortality (aHR, 1.43; 95% CI, 1.12-1.83), while dual-combination therapy was less so (aHR, 1.29; 95% CI, 0.96-1.74). Non–COVID-19–related mortality was significantly increased for all COPD patients who were prescribed ICS, with or without adjustment for covariates.
Asthma patients prescribed high-dose ICS instead of SABA alone had a slightly greater risk of COVID-19–related death, based on an adjusted hazard ratio of 1.55 (95% CI, 1.10-2.18). Those with asthma who received low/medium–dose ICS demonstrated a slight trend toward increased mortality risk, but this was not significant (aHR, 1.14; 95% CI, 0.85-1.54). ICS usage in the asthma group was not linked with a significant increase in non–COVID-19–related death.
“In summary, we found no evidence of a beneficial effect of regular ICS use among people with COPD and asthma on COVID-19–related mortality,” the investigators concluded.
In agreement with the investigators, Dr. Conroy said that the increased mortality rate among ICS users should not be misconstrued as a medication-related risk.
“While the study found that those with COPD or asthma taking ICS and high-dose ICS were at an increased risk of death, this could easily be explained by the likelihood that those are the patients who are more likely to have more severe underlying lung disease,” Dr. Conroy said. “While this observational study did attempt to control for exacerbation history, the ability to do so by electronic health records data is certainly imperfect.”
With this in mind, patients with chronic respiratory disease should be encouraged to adhere to their usual treatment regimen, Dr. Conroy added.
“There isn’t evidence to increase or decrease medications just because of the pandemic,” she said. “A patient with asthma or COPD should continue to take the medications that are needed to achieve good control of their lung disease.”
The study was funded by the U.K. Medical Research Council. The investigators reported additional relationships with the Wellcome Trust, the Good Thinking Foundation, the Laura and John Arnold Foundation, and others. Dr. Conroy reported no conflicts of interest.
SOURCE: Schultze A et al. Lancet Respir Med. 2020 Sep 24. doi: 10.1016/ S2213-2600(20)30415-X.
FROM LANCET RESPIRATORY MEDICINE
COVID-19 vaccine hesitancy ‘somewhat understandable,’ expert says
“I worry that vaccines are going to be sold like magic powder that we sprinkle across the land and make the virus go away,” Paul Offit, MD, said at the virtual American Academy of Pediatrics (AAP) 2020 National Conference. “That’s not true.”
according to Dr. Offit, director of the Vaccine Education Center and an attending physician in the Division of Infectious Diseases at Children’s Hospital of Philadelphia.
“I think we can get a vaccine that’s 75%-80% effective at preventing mild to moderate disease, but that means one of every four people can still get moderate to severe disease,” Dr. Offit continued.
And that’s if there is high uptake of the vaccine, which may not be the case. Recent polls have suggested there is considerable concern about the pending vaccines.
“It’s somewhat understandable,” Dr. Offitt acknowledged, especially given the “frightening” language used to describe vaccine development. Terms such as “warp speed” may suggest that haste might trump safety considerations. Before COVID-19, the fastest vaccine ever developed was for mumps, he said, with the virus isolated in 1963 and a commercial product available in 1967.
Addressing hesitancy in clinics
In a wide-ranging livestream plenary presentation, Dr. Offit, coinventor of a rotavirus vaccine, shed light on SARS-CoV-2 vaccine development and his impressions of vaccine hesitancy among patients and families. He also offered advice for how to reassure those skeptical of the safety and efficacy of any SARS-COV-2 vaccine, given the accelerated development process.
With more than 180 different vaccines in various stages of investigation, Dr. Offit called the effort to develop COVID-19 vaccines “unprecedented.” Part of that is a result of governments relieving pharmaceutical companies of much of the typical financial risk – which often climbs to hundreds of millions of dollars – by underwriting the costs of vaccine development to battle the pandemic-inducing virus, he said.
But this very swiftness is also stoking antivaccine sentiment. Dr. Offit, part of vaccine advisory groups for the National Institutes of Health and U.S. Food and Drug Administration, cited recent research reporting nearly half of American adults definitely or probably would not get a COVID-19 vaccine if it were available today.
“One way you convince skeptics is with data presented in a clear, compassionate, and compelling way,” he said.
“The other group is vaccine cynics, who are basically conspiracy theorists who believe pharmaceutical companies control the world, the government, the medical establishment. I think there’s no talking them down from this.”
Numerous strategies are being used in COVID-19 vaccine development, he noted, including messenger RNA, DNA, viral vectors, purified protein, and whole killed virus. Dr. Offit believes any candidates approved for distribution will likely be in the range of 75% effective at preventing mild to moderate symptoms.
But clinicians should be ready to face immediate questions of safety. “Even if this vaccination is given to 20,000 [trial participants] safely, that’s not 20 million,” Dr. Offit said. “Anyone could reasonably ask questions about if it causes rare, serious side effects.
“The good news is, there are systems in place,” such as adverse event reporting systems, to identify rare events, even those that occur in one in a million vaccine recipients. Reminding patients of that continued surveillance can be reassuring.
Another reassuring point is that COVID-19 vaccine trial participants have included people from many diverse populations, he said. But children, notably absent so far, should be added to trials immediately, Dr. Offit contends.
“This is going to be important when you consider strategies to get children universally back into school,” he said, which is a “critical issue” from both learning and wellness standpoints. “It breaks my heart that we’ve been unable to do this when other countries have.”
Transparency will be paramount
While presenting data transparently to patients is key in helping them accept COVID-19 vaccination, Dr. Offit said, he also believes “telling stories” can be just as effective, if not more so. When the varicella vaccine was approved in 1995, he said, the “uptake the first few years was pretty miserable” until public service messaging emphasized that some children die from chickenpox.
“Fear works,” he said. “You always worry about pushback of something being oversold, but hopefully we’re scared enough about this virus” to convince people that vaccination is wise. “I do think personal stories carry weight on both sides,” Dr. Offit said.
Mark Sawyer, MD, of University of California San Diego School of Medicine and Rady Children’s Hospital in San Diego, California, said Offit’s presentation offered important takeaways for clinicians about how to broach the topic of COVID-19 vaccination with patients and families.
“We need to communicate clearly and transparently to patients about what we do and don’t know” about the vaccines, Dr. Sawyer said in an interview. “We will know if they have common side effects, but we will not know about very rare side effects until we have used the vaccines for a while.
“We will know how well the vaccine works over the short-term, but we won’t know over the long term,” added Dr. Sawyer, a member of the AAP Committee on Infectious Diseases.
“We can reassure the community that SARS-CoV-2 vaccines are being evaluated in trials in the same way and with the same thoroughness as other vaccines have been,” he said. “That should give people confidence that shortcuts are not being taken with regard to safety and effectiveness evaluations.”
Dr. Offit and Dr. Sawyer have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
“I worry that vaccines are going to be sold like magic powder that we sprinkle across the land and make the virus go away,” Paul Offit, MD, said at the virtual American Academy of Pediatrics (AAP) 2020 National Conference. “That’s not true.”
according to Dr. Offit, director of the Vaccine Education Center and an attending physician in the Division of Infectious Diseases at Children’s Hospital of Philadelphia.
“I think we can get a vaccine that’s 75%-80% effective at preventing mild to moderate disease, but that means one of every four people can still get moderate to severe disease,” Dr. Offit continued.
And that’s if there is high uptake of the vaccine, which may not be the case. Recent polls have suggested there is considerable concern about the pending vaccines.
“It’s somewhat understandable,” Dr. Offitt acknowledged, especially given the “frightening” language used to describe vaccine development. Terms such as “warp speed” may suggest that haste might trump safety considerations. Before COVID-19, the fastest vaccine ever developed was for mumps, he said, with the virus isolated in 1963 and a commercial product available in 1967.
Addressing hesitancy in clinics
In a wide-ranging livestream plenary presentation, Dr. Offit, coinventor of a rotavirus vaccine, shed light on SARS-CoV-2 vaccine development and his impressions of vaccine hesitancy among patients and families. He also offered advice for how to reassure those skeptical of the safety and efficacy of any SARS-COV-2 vaccine, given the accelerated development process.
With more than 180 different vaccines in various stages of investigation, Dr. Offit called the effort to develop COVID-19 vaccines “unprecedented.” Part of that is a result of governments relieving pharmaceutical companies of much of the typical financial risk – which often climbs to hundreds of millions of dollars – by underwriting the costs of vaccine development to battle the pandemic-inducing virus, he said.
But this very swiftness is also stoking antivaccine sentiment. Dr. Offit, part of vaccine advisory groups for the National Institutes of Health and U.S. Food and Drug Administration, cited recent research reporting nearly half of American adults definitely or probably would not get a COVID-19 vaccine if it were available today.
“One way you convince skeptics is with data presented in a clear, compassionate, and compelling way,” he said.
“The other group is vaccine cynics, who are basically conspiracy theorists who believe pharmaceutical companies control the world, the government, the medical establishment. I think there’s no talking them down from this.”
Numerous strategies are being used in COVID-19 vaccine development, he noted, including messenger RNA, DNA, viral vectors, purified protein, and whole killed virus. Dr. Offit believes any candidates approved for distribution will likely be in the range of 75% effective at preventing mild to moderate symptoms.
But clinicians should be ready to face immediate questions of safety. “Even if this vaccination is given to 20,000 [trial participants] safely, that’s not 20 million,” Dr. Offit said. “Anyone could reasonably ask questions about if it causes rare, serious side effects.
“The good news is, there are systems in place,” such as adverse event reporting systems, to identify rare events, even those that occur in one in a million vaccine recipients. Reminding patients of that continued surveillance can be reassuring.
Another reassuring point is that COVID-19 vaccine trial participants have included people from many diverse populations, he said. But children, notably absent so far, should be added to trials immediately, Dr. Offit contends.
“This is going to be important when you consider strategies to get children universally back into school,” he said, which is a “critical issue” from both learning and wellness standpoints. “It breaks my heart that we’ve been unable to do this when other countries have.”
Transparency will be paramount
While presenting data transparently to patients is key in helping them accept COVID-19 vaccination, Dr. Offit said, he also believes “telling stories” can be just as effective, if not more so. When the varicella vaccine was approved in 1995, he said, the “uptake the first few years was pretty miserable” until public service messaging emphasized that some children die from chickenpox.
“Fear works,” he said. “You always worry about pushback of something being oversold, but hopefully we’re scared enough about this virus” to convince people that vaccination is wise. “I do think personal stories carry weight on both sides,” Dr. Offit said.
Mark Sawyer, MD, of University of California San Diego School of Medicine and Rady Children’s Hospital in San Diego, California, said Offit’s presentation offered important takeaways for clinicians about how to broach the topic of COVID-19 vaccination with patients and families.
“We need to communicate clearly and transparently to patients about what we do and don’t know” about the vaccines, Dr. Sawyer said in an interview. “We will know if they have common side effects, but we will not know about very rare side effects until we have used the vaccines for a while.
“We will know how well the vaccine works over the short-term, but we won’t know over the long term,” added Dr. Sawyer, a member of the AAP Committee on Infectious Diseases.
“We can reassure the community that SARS-CoV-2 vaccines are being evaluated in trials in the same way and with the same thoroughness as other vaccines have been,” he said. “That should give people confidence that shortcuts are not being taken with regard to safety and effectiveness evaluations.”
Dr. Offit and Dr. Sawyer have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
“I worry that vaccines are going to be sold like magic powder that we sprinkle across the land and make the virus go away,” Paul Offit, MD, said at the virtual American Academy of Pediatrics (AAP) 2020 National Conference. “That’s not true.”
according to Dr. Offit, director of the Vaccine Education Center and an attending physician in the Division of Infectious Diseases at Children’s Hospital of Philadelphia.
“I think we can get a vaccine that’s 75%-80% effective at preventing mild to moderate disease, but that means one of every four people can still get moderate to severe disease,” Dr. Offit continued.
And that’s if there is high uptake of the vaccine, which may not be the case. Recent polls have suggested there is considerable concern about the pending vaccines.
“It’s somewhat understandable,” Dr. Offitt acknowledged, especially given the “frightening” language used to describe vaccine development. Terms such as “warp speed” may suggest that haste might trump safety considerations. Before COVID-19, the fastest vaccine ever developed was for mumps, he said, with the virus isolated in 1963 and a commercial product available in 1967.
Addressing hesitancy in clinics
In a wide-ranging livestream plenary presentation, Dr. Offit, coinventor of a rotavirus vaccine, shed light on SARS-CoV-2 vaccine development and his impressions of vaccine hesitancy among patients and families. He also offered advice for how to reassure those skeptical of the safety and efficacy of any SARS-COV-2 vaccine, given the accelerated development process.
With more than 180 different vaccines in various stages of investigation, Dr. Offit called the effort to develop COVID-19 vaccines “unprecedented.” Part of that is a result of governments relieving pharmaceutical companies of much of the typical financial risk – which often climbs to hundreds of millions of dollars – by underwriting the costs of vaccine development to battle the pandemic-inducing virus, he said.
But this very swiftness is also stoking antivaccine sentiment. Dr. Offit, part of vaccine advisory groups for the National Institutes of Health and U.S. Food and Drug Administration, cited recent research reporting nearly half of American adults definitely or probably would not get a COVID-19 vaccine if it were available today.
“One way you convince skeptics is with data presented in a clear, compassionate, and compelling way,” he said.
“The other group is vaccine cynics, who are basically conspiracy theorists who believe pharmaceutical companies control the world, the government, the medical establishment. I think there’s no talking them down from this.”
Numerous strategies are being used in COVID-19 vaccine development, he noted, including messenger RNA, DNA, viral vectors, purified protein, and whole killed virus. Dr. Offit believes any candidates approved for distribution will likely be in the range of 75% effective at preventing mild to moderate symptoms.
But clinicians should be ready to face immediate questions of safety. “Even if this vaccination is given to 20,000 [trial participants] safely, that’s not 20 million,” Dr. Offit said. “Anyone could reasonably ask questions about if it causes rare, serious side effects.
“The good news is, there are systems in place,” such as adverse event reporting systems, to identify rare events, even those that occur in one in a million vaccine recipients. Reminding patients of that continued surveillance can be reassuring.
Another reassuring point is that COVID-19 vaccine trial participants have included people from many diverse populations, he said. But children, notably absent so far, should be added to trials immediately, Dr. Offit contends.
“This is going to be important when you consider strategies to get children universally back into school,” he said, which is a “critical issue” from both learning and wellness standpoints. “It breaks my heart that we’ve been unable to do this when other countries have.”
Transparency will be paramount
While presenting data transparently to patients is key in helping them accept COVID-19 vaccination, Dr. Offit said, he also believes “telling stories” can be just as effective, if not more so. When the varicella vaccine was approved in 1995, he said, the “uptake the first few years was pretty miserable” until public service messaging emphasized that some children die from chickenpox.
“Fear works,” he said. “You always worry about pushback of something being oversold, but hopefully we’re scared enough about this virus” to convince people that vaccination is wise. “I do think personal stories carry weight on both sides,” Dr. Offit said.
Mark Sawyer, MD, of University of California San Diego School of Medicine and Rady Children’s Hospital in San Diego, California, said Offit’s presentation offered important takeaways for clinicians about how to broach the topic of COVID-19 vaccination with patients and families.
“We need to communicate clearly and transparently to patients about what we do and don’t know” about the vaccines, Dr. Sawyer said in an interview. “We will know if they have common side effects, but we will not know about very rare side effects until we have used the vaccines for a while.
“We will know how well the vaccine works over the short-term, but we won’t know over the long term,” added Dr. Sawyer, a member of the AAP Committee on Infectious Diseases.
“We can reassure the community that SARS-CoV-2 vaccines are being evaluated in trials in the same way and with the same thoroughness as other vaccines have been,” he said. “That should give people confidence that shortcuts are not being taken with regard to safety and effectiveness evaluations.”
Dr. Offit and Dr. Sawyer have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.