User login
Aquagenic Wrinkling Among Skin-Related Signs of Cystic Fibrosis
TOPLINE:
METHODOLOGY:
- Patients with CF, caused by a mutation in the CF Transmembrane Conductance Regulator (CFTR) gene, can develop diverse dermatologic manifestations.
- Researchers reviewed the literature and provided their own clinical experience regarding dermatologic manifestations of CF.
- They also reviewed the cutaneous side effects of CFTR modulators and antibiotics used to treat CF.
TAKEAWAY:
- Aquagenic wrinkling of the palm is common in individuals with CF, affecting up to 80% of patients (and 25% of CF gene carriers), and can be an early manifestation of CF. Treatments include topical medications (such as aluminum chloride, corticosteroids, and salicylic acid), botulinum toxin injections, and recently, CFTR-modulating treatments.
- CF nutrient deficiency dermatitis, often in a diaper distribution, usually appears in infancy and, before newborn screening was available, was sometimes the first sign of CF in some cases. It usually resolves with an adequate diet, pancreatic enzymes, and/or nutritional supplements. Zinc and essential fatty acid deficiencies can lead to acrodermatitis enteropathica–like symptoms and psoriasiform rashes, respectively.
- CF is also associated with vascular disorders, including cutaneous and, rarely, systemic vasculitis. Treatment includes topical and oral steroids and immune-modulating therapies.
- CFTR modulators, now the most common and highly effective treatment for CF, are associated with several skin reactions, which can be managed with treatments that include topical steroids and oral antihistamines. Frequent antibiotic treatment can also trigger skin reactions.
IN PRACTICE:
“Recognition and familiarity with dermatologic clinical manifestations of CF are important for multidisciplinary care” for patients with CF, the authors wrote, adding that “dermatology providers may play a significant role in the diagnosis and management of CF cutaneous comorbidities.”
SOURCE:
Aaron D. Smith, BS, from the University of Virginia (UVA) School of Medicine, Charlottesville, and coauthors were from the departments of dermatology and pulmonology/critical care medicine at UVA. The study was published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The authors did not make a comment about the limitations of their review.
DISCLOSURES:
No funding was received for the review. The authors had no disclosures.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Patients with CF, caused by a mutation in the CF Transmembrane Conductance Regulator (CFTR) gene, can develop diverse dermatologic manifestations.
- Researchers reviewed the literature and provided their own clinical experience regarding dermatologic manifestations of CF.
- They also reviewed the cutaneous side effects of CFTR modulators and antibiotics used to treat CF.
TAKEAWAY:
- Aquagenic wrinkling of the palm is common in individuals with CF, affecting up to 80% of patients (and 25% of CF gene carriers), and can be an early manifestation of CF. Treatments include topical medications (such as aluminum chloride, corticosteroids, and salicylic acid), botulinum toxin injections, and recently, CFTR-modulating treatments.
- CF nutrient deficiency dermatitis, often in a diaper distribution, usually appears in infancy and, before newborn screening was available, was sometimes the first sign of CF in some cases. It usually resolves with an adequate diet, pancreatic enzymes, and/or nutritional supplements. Zinc and essential fatty acid deficiencies can lead to acrodermatitis enteropathica–like symptoms and psoriasiform rashes, respectively.
- CF is also associated with vascular disorders, including cutaneous and, rarely, systemic vasculitis. Treatment includes topical and oral steroids and immune-modulating therapies.
- CFTR modulators, now the most common and highly effective treatment for CF, are associated with several skin reactions, which can be managed with treatments that include topical steroids and oral antihistamines. Frequent antibiotic treatment can also trigger skin reactions.
IN PRACTICE:
“Recognition and familiarity with dermatologic clinical manifestations of CF are important for multidisciplinary care” for patients with CF, the authors wrote, adding that “dermatology providers may play a significant role in the diagnosis and management of CF cutaneous comorbidities.”
SOURCE:
Aaron D. Smith, BS, from the University of Virginia (UVA) School of Medicine, Charlottesville, and coauthors were from the departments of dermatology and pulmonology/critical care medicine at UVA. The study was published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The authors did not make a comment about the limitations of their review.
DISCLOSURES:
No funding was received for the review. The authors had no disclosures.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Patients with CF, caused by a mutation in the CF Transmembrane Conductance Regulator (CFTR) gene, can develop diverse dermatologic manifestations.
- Researchers reviewed the literature and provided their own clinical experience regarding dermatologic manifestations of CF.
- They also reviewed the cutaneous side effects of CFTR modulators and antibiotics used to treat CF.
TAKEAWAY:
- Aquagenic wrinkling of the palm is common in individuals with CF, affecting up to 80% of patients (and 25% of CF gene carriers), and can be an early manifestation of CF. Treatments include topical medications (such as aluminum chloride, corticosteroids, and salicylic acid), botulinum toxin injections, and recently, CFTR-modulating treatments.
- CF nutrient deficiency dermatitis, often in a diaper distribution, usually appears in infancy and, before newborn screening was available, was sometimes the first sign of CF in some cases. It usually resolves with an adequate diet, pancreatic enzymes, and/or nutritional supplements. Zinc and essential fatty acid deficiencies can lead to acrodermatitis enteropathica–like symptoms and psoriasiform rashes, respectively.
- CF is also associated with vascular disorders, including cutaneous and, rarely, systemic vasculitis. Treatment includes topical and oral steroids and immune-modulating therapies.
- CFTR modulators, now the most common and highly effective treatment for CF, are associated with several skin reactions, which can be managed with treatments that include topical steroids and oral antihistamines. Frequent antibiotic treatment can also trigger skin reactions.
IN PRACTICE:
“Recognition and familiarity with dermatologic clinical manifestations of CF are important for multidisciplinary care” for patients with CF, the authors wrote, adding that “dermatology providers may play a significant role in the diagnosis and management of CF cutaneous comorbidities.”
SOURCE:
Aaron D. Smith, BS, from the University of Virginia (UVA) School of Medicine, Charlottesville, and coauthors were from the departments of dermatology and pulmonology/critical care medicine at UVA. The study was published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The authors did not make a comment about the limitations of their review.
DISCLOSURES:
No funding was received for the review. The authors had no disclosures.
A version of this article first appeared on Medscape.com.
Survey Spotlights Identification of Dermatologic Adverse Events From Cancer Therapies
“New cancer therapies have brought a diversity of treatment-related dermatologic adverse events (dAEs) beyond those experienced with conventional chemotherapy, which has demanded an evolving assessment of toxicities,” researchers led by Nicole R. LeBoeuf, MD, MPH, of the Department of Dermatology at Brigham and Women’s Hospital and the Center for Cutaneous Oncology at the Dana-Farber Brigham Cancer Center, Boston, wrote in a poster presented at the American Academy of Dermatology annual meeting.
The authors noted that “Version 5.0 of the Common Terminology Criteria for Adverse Events (CTCAE v5.0)” serves as the current, broadly accepted criteria for classification and grading during routine medical care and clinical trials. But despite extensive utilization of CTCAE, there is little data regarding its application.”
To evaluate how CTCAE is being used in clinical practice, they sent a four-case survey of dAEs to 81 dermatologists and 182 medical oncologists at six US-based academic institutions. For three of the cases, respondents were asked to classify and grade morbilliform, psoriasiform, and papulopustular rashes based on a review of photographs and text descriptions. For the fourth case, respondents were asked to grade a dAE using only a clinic note text description. The researchers used chi-square tests in R software to compare survey responses.
Compared with medical oncologists, dermatologists were significantly more likely to provide correct responses in characterizing morbilliform and psoriasiform eruptions. “As low as 12%” of medical oncologists were correct, and “as low as 87%” of dermatologists were correct (P < .001). Similarly, dermatologists were significantly more likely to grade the psoriasiform, papulopustular, and written cases correctly compared with medical oncologists (P < .001 for all associations).
“These cases demonstrated poor concordance of classification and grading between specialties and across medical oncology,” the authors concluded in their poster, noting that 87% of medical oncologists were interested in additional educational tools on dAEs. “With correct classification as low as 12%, medical oncologists may have more difficulty delivering appropriate, toxicity-specific therapy and may consider banal eruptions dangerous.”
Poor concordance of grading among the two groups of clinicians “raises the question of whether CTCAE v5.0 is an appropriate determinant for patient continuation on therapy or in trials,” they added. “As anticancer therapy becomes more complex — with new toxicities from novel agents and combinations — we must ensure we have a grading system that is valid across investigators and does not harm patients by instituting unnecessary treatment stops.”
Future studies, they said, “can explore what interventions beyond involvement of dermatologists improve classification and grading in practice.”
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study, noted that with the continued expansion and introduction of new targeted and immunotherapies in the oncology space, “you can be sure we will continue to appreciate the importance and value of the field of supportive oncodermatology, as hair, skin, and nails are almost guaranteed collateral damage in this story.
“Ensuring early identification and consistent grading severity is not only important for the plethora of patients who are currently developing the litany of cutaneous adverse events but to evaluate potential mitigation strategies and even push along countermeasures down the FDA approval pathway,” Dr. Friedman said. In this study, the investigators demonstrated that work “is sorely needed, not just in dermatology but even more so for our colleagues across the aisle. A central tenet of supportive oncodermatology must also be education for all stakeholders, and the good news is our oncology partners will welcome it.”
Dr. LeBoeuf disclosed that she is a consultant to and has received honoraria from Bayer, Seattle Genetics, Sanofi, Silverback, Fortress Biotech, and Synox Therapeutics outside the submitted work. No other authors reported having financial disclosures. Dr. Friedman directs the supportive oncodermatology program at GW that received independent funding from La Roche-Posay.
A version of this article first appeared on Medscape.com.
“New cancer therapies have brought a diversity of treatment-related dermatologic adverse events (dAEs) beyond those experienced with conventional chemotherapy, which has demanded an evolving assessment of toxicities,” researchers led by Nicole R. LeBoeuf, MD, MPH, of the Department of Dermatology at Brigham and Women’s Hospital and the Center for Cutaneous Oncology at the Dana-Farber Brigham Cancer Center, Boston, wrote in a poster presented at the American Academy of Dermatology annual meeting.
The authors noted that “Version 5.0 of the Common Terminology Criteria for Adverse Events (CTCAE v5.0)” serves as the current, broadly accepted criteria for classification and grading during routine medical care and clinical trials. But despite extensive utilization of CTCAE, there is little data regarding its application.”
To evaluate how CTCAE is being used in clinical practice, they sent a four-case survey of dAEs to 81 dermatologists and 182 medical oncologists at six US-based academic institutions. For three of the cases, respondents were asked to classify and grade morbilliform, psoriasiform, and papulopustular rashes based on a review of photographs and text descriptions. For the fourth case, respondents were asked to grade a dAE using only a clinic note text description. The researchers used chi-square tests in R software to compare survey responses.
Compared with medical oncologists, dermatologists were significantly more likely to provide correct responses in characterizing morbilliform and psoriasiform eruptions. “As low as 12%” of medical oncologists were correct, and “as low as 87%” of dermatologists were correct (P < .001). Similarly, dermatologists were significantly more likely to grade the psoriasiform, papulopustular, and written cases correctly compared with medical oncologists (P < .001 for all associations).
“These cases demonstrated poor concordance of classification and grading between specialties and across medical oncology,” the authors concluded in their poster, noting that 87% of medical oncologists were interested in additional educational tools on dAEs. “With correct classification as low as 12%, medical oncologists may have more difficulty delivering appropriate, toxicity-specific therapy and may consider banal eruptions dangerous.”
Poor concordance of grading among the two groups of clinicians “raises the question of whether CTCAE v5.0 is an appropriate determinant for patient continuation on therapy or in trials,” they added. “As anticancer therapy becomes more complex — with new toxicities from novel agents and combinations — we must ensure we have a grading system that is valid across investigators and does not harm patients by instituting unnecessary treatment stops.”
Future studies, they said, “can explore what interventions beyond involvement of dermatologists improve classification and grading in practice.”
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study, noted that with the continued expansion and introduction of new targeted and immunotherapies in the oncology space, “you can be sure we will continue to appreciate the importance and value of the field of supportive oncodermatology, as hair, skin, and nails are almost guaranteed collateral damage in this story.
“Ensuring early identification and consistent grading severity is not only important for the plethora of patients who are currently developing the litany of cutaneous adverse events but to evaluate potential mitigation strategies and even push along countermeasures down the FDA approval pathway,” Dr. Friedman said. In this study, the investigators demonstrated that work “is sorely needed, not just in dermatology but even more so for our colleagues across the aisle. A central tenet of supportive oncodermatology must also be education for all stakeholders, and the good news is our oncology partners will welcome it.”
Dr. LeBoeuf disclosed that she is a consultant to and has received honoraria from Bayer, Seattle Genetics, Sanofi, Silverback, Fortress Biotech, and Synox Therapeutics outside the submitted work. No other authors reported having financial disclosures. Dr. Friedman directs the supportive oncodermatology program at GW that received independent funding from La Roche-Posay.
A version of this article first appeared on Medscape.com.
“New cancer therapies have brought a diversity of treatment-related dermatologic adverse events (dAEs) beyond those experienced with conventional chemotherapy, which has demanded an evolving assessment of toxicities,” researchers led by Nicole R. LeBoeuf, MD, MPH, of the Department of Dermatology at Brigham and Women’s Hospital and the Center for Cutaneous Oncology at the Dana-Farber Brigham Cancer Center, Boston, wrote in a poster presented at the American Academy of Dermatology annual meeting.
The authors noted that “Version 5.0 of the Common Terminology Criteria for Adverse Events (CTCAE v5.0)” serves as the current, broadly accepted criteria for classification and grading during routine medical care and clinical trials. But despite extensive utilization of CTCAE, there is little data regarding its application.”
To evaluate how CTCAE is being used in clinical practice, they sent a four-case survey of dAEs to 81 dermatologists and 182 medical oncologists at six US-based academic institutions. For three of the cases, respondents were asked to classify and grade morbilliform, psoriasiform, and papulopustular rashes based on a review of photographs and text descriptions. For the fourth case, respondents were asked to grade a dAE using only a clinic note text description. The researchers used chi-square tests in R software to compare survey responses.
Compared with medical oncologists, dermatologists were significantly more likely to provide correct responses in characterizing morbilliform and psoriasiform eruptions. “As low as 12%” of medical oncologists were correct, and “as low as 87%” of dermatologists were correct (P < .001). Similarly, dermatologists were significantly more likely to grade the psoriasiform, papulopustular, and written cases correctly compared with medical oncologists (P < .001 for all associations).
“These cases demonstrated poor concordance of classification and grading between specialties and across medical oncology,” the authors concluded in their poster, noting that 87% of medical oncologists were interested in additional educational tools on dAEs. “With correct classification as low as 12%, medical oncologists may have more difficulty delivering appropriate, toxicity-specific therapy and may consider banal eruptions dangerous.”
Poor concordance of grading among the two groups of clinicians “raises the question of whether CTCAE v5.0 is an appropriate determinant for patient continuation on therapy or in trials,” they added. “As anticancer therapy becomes more complex — with new toxicities from novel agents and combinations — we must ensure we have a grading system that is valid across investigators and does not harm patients by instituting unnecessary treatment stops.”
Future studies, they said, “can explore what interventions beyond involvement of dermatologists improve classification and grading in practice.”
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study, noted that with the continued expansion and introduction of new targeted and immunotherapies in the oncology space, “you can be sure we will continue to appreciate the importance and value of the field of supportive oncodermatology, as hair, skin, and nails are almost guaranteed collateral damage in this story.
“Ensuring early identification and consistent grading severity is not only important for the plethora of patients who are currently developing the litany of cutaneous adverse events but to evaluate potential mitigation strategies and even push along countermeasures down the FDA approval pathway,” Dr. Friedman said. In this study, the investigators demonstrated that work “is sorely needed, not just in dermatology but even more so for our colleagues across the aisle. A central tenet of supportive oncodermatology must also be education for all stakeholders, and the good news is our oncology partners will welcome it.”
Dr. LeBoeuf disclosed that she is a consultant to and has received honoraria from Bayer, Seattle Genetics, Sanofi, Silverback, Fortress Biotech, and Synox Therapeutics outside the submitted work. No other authors reported having financial disclosures. Dr. Friedman directs the supportive oncodermatology program at GW that received independent funding from La Roche-Posay.
A version of this article first appeared on Medscape.com.
FROM AAD 2024
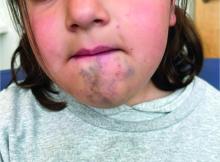
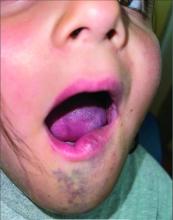
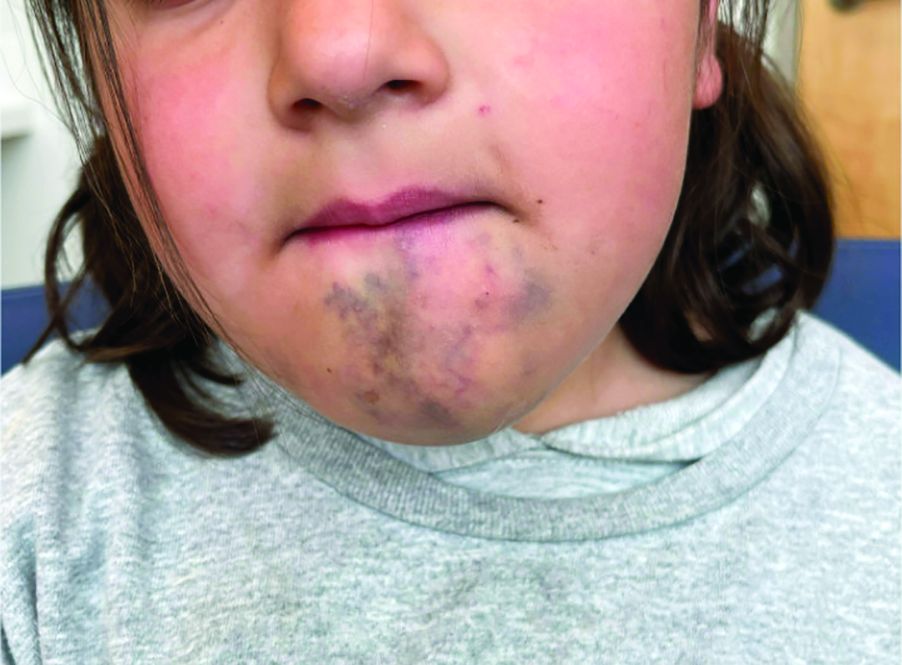
A 6-Year-Old Female Presents With a Bruise-Like Lesion on the Lip, Tongue, and Chin Area Present Since Birth
Diagnosis: Venous Malformation
Although present at birth, they are not always clinically evident early in life. They also tend to grow with the child without spontaneous regression, causing potential cosmetic concerns or complications from impingement on surrounding tissue.
Venous malformations appear with a bluish color appearing beneath the skin and can vary significantly in size and severity. Venous malformations are compressible and characterized by low to stagnant blood flow, which can spontaneously thrombose. Clinically, this may cause pain, swelling, skin changes, tissue and limb overgrowth, or functional impairment depending on location and size.
Venous malformations result from disorganized angiogenesis secondary to sporadic mutations in somatic cells. The most common implicated gene is TEK, a receptor tyrosine kinase. PIK3CA has also been involved. Both genes are involved in the PI3K/AKT/mTOR pathway, which regulates cell growth, proliferation, and angiogenesis. In venous endothelial cells, abnormal angiogenesis and vessel maturation may lead to venous malformation formation. Dysplastic vessels frequently separate from normal veins but may be contiguous with the deep venous system.
Diagnosis involves clinical history and physical examination. Imaging with ultrasound and magnetic resonance imaging (MRI) may be utilized. While ultrasound may be preferred for superficial venous malformations, MRI or MRI with MR angiography (MRA) is the preferred method for venous malformation assessment. Genetic testing may be appropriate for complex malformations, as classification of lesions by underlying mutation may allow targeted therapy.
This patient’s past MRI and MRA findings were consistent with a venous malformation.
Treatment
Venous malformations rarely regress spontaneously. Treatment is required if venous malformations are symptomatic, which may include pain, swelling, deformity, thrombosis, or interference with daily activities of living. Treatment plans require consideration of patient goals of care. The main categories of therapy are embolization/sclerotherapy, surgical resection, and molecular targeted therapy.
Sclerotherapy is a well-tolerated and efficacious first-line therapy. It can be used as either nonsurgical curative therapy or preoperative adjunct therapy to minimize blood loss before surgical resection. While surgical resection may cause scarring, multimodal approaches with sclerotherapy or laser therapy can decrease complications. Molecular therapies aim to reduce vascular proliferation and symptoms. Referral to hematology/oncology for evaluation and consideration of chemotherapeutic agents may be required. Sirolimus has been shown in mice models to inhibit an endothelial cell tyrosine kinase receptor that plays a role in venous malformation growth. Multiple studies have proved its efficacy in managing complicated vascular anomalies, including venous malformations. Alpelisib is an inhibitor of PI3KCA, which is part of the pathway that contributes to venous malformation formation. Dactolisib, a dual inhibitor of the PI3KA and mTOR pathways, and rebastinib, a TEK inhibitor, are being investigated.
Differential Diagnoses
The differential diagnosis includes dermal melanocytosis, nevus of Ota, hemangioma of infancy, and ashy dermatosis. In addition, venous malformations can be part of more complex vascular malformations.
Dermal melanocytosis, also known as Mongolian spots, are blue-gray patches of discoloration on the skin that appear at birth or shortly after. They result from the arrest of dermal melanocytes in the dermis during fetal life and tissue modeling. They are commonly observed in those of Asian or African descent with darker skin types. Most often, they are located in the lumbar or sacral-gluteal region. Unlike venous malformations, they are benign and do not involve vascular abnormalities. They typically fade over time.
Nevus of Ota is a benign congenital condition that presents with blue-gray or brown patches of pigmentation on the skin around the eyes, cheeks, and forehead. They are dermal melanocytes with a speckled instead of uniform appearance. Nevus of Ota primarily affects individuals of Asian descent and typically presents in the trigeminal nerve distribution region. Treatment can be done to minimize deformity, generally with pigmented laser surgery.
Hemangiomas of infancy are common benign tumors of infancy caused by endothelial cell proliferation. They are characterized by rapid growth followed by spontaneous involution within the first year of life and for several years. Hemangiomas can be superficial, deep, or mixed with features of both superficial and deep. Superficial hemangiomas present as raised, lobulated, and bright red while deep hemangiomas present as a bluish-hued nodule, plaque, or tumor. They are diagnosed clinically but skin biopsies and imaging can confirm the suspected diagnosis. While hemangiomas may self-resolve, complicated hemangiomas can be treated with topical timolol, oral propranolol, topical and intralesional corticosteroids, pulsed-dye laser, and surgical resection.
Ashy dermatosis is a term for asymptomatic, gray-blue or ashy patches distributed symmetrically on the trunk, head, neck, and upper extremities. It primarily affects individuals with darker skin types (Fitzpatrick III-V), and is more common in patients with Hispanic, Asian, or African backgrounds. The direct cause of ashy dermatosis is unknown but it is thought to be linked to drug ingestion, genetics, infection, and immune-mediated mechanisms. The general treatment includes topical corticosteroids, clofazimine, topical calcineurin inhibitors, oral dapsone, phototherapy, topical retinoids, or isotretinoin to reduce inflammation and pigmentation.
Danny Lee and Samuel Le serve as research fellows and Jolina Bui as research associate in the Pediatric Dermatology Division of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is Distinguished Professor of Dermatology and Pediatrics and Vice-Chair of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. The authors have no relevant financial disclosures.
Suggested Reading
Agarwal P, Patel BC. Nevus of Ota and Ito. [Updated 2023 Jul 10]. In: StatPearls [Internet]. StatPearls Publishing; 2024.
Dompmartin A et al. The VASCERN-VASCA Working Group Diagnostic and Management Pathways for Venous Malformations. J Vasc Anom (Phila). 2023 Mar 23;4(2):e064.
Dompmartin A et al. Venous malformation: Update on aetiopathogenesis, diagnosis and management. Phlebology. 2010 Oct;25(5):224-235.
Gupta D, Thappa DM. Mongolian spots. Indian J Dermatol Venereol Leprol. 2013 Jul-Aug;79(4):469-478.
Krowchuk DP et al. Clinical Practice Guideline for the Management of Infantile Hemangiomas. Pediatrics. 2019 Jan;143(1):e20183475.
Nguyen K, Khachemoune A. Ashy dermatosis: A review. Dermatol Online J. 2019 May 15;25(5):13030/qt44f462s8.
Patel ND, Chong AT et al. Venous Malformations. Semin Intervent Radiol. 2022 Dec 20;39(5):498-507.
Diagnosis: Venous Malformation
Although present at birth, they are not always clinically evident early in life. They also tend to grow with the child without spontaneous regression, causing potential cosmetic concerns or complications from impingement on surrounding tissue.
Venous malformations appear with a bluish color appearing beneath the skin and can vary significantly in size and severity. Venous malformations are compressible and characterized by low to stagnant blood flow, which can spontaneously thrombose. Clinically, this may cause pain, swelling, skin changes, tissue and limb overgrowth, or functional impairment depending on location and size.
Venous malformations result from disorganized angiogenesis secondary to sporadic mutations in somatic cells. The most common implicated gene is TEK, a receptor tyrosine kinase. PIK3CA has also been involved. Both genes are involved in the PI3K/AKT/mTOR pathway, which regulates cell growth, proliferation, and angiogenesis. In venous endothelial cells, abnormal angiogenesis and vessel maturation may lead to venous malformation formation. Dysplastic vessels frequently separate from normal veins but may be contiguous with the deep venous system.
Diagnosis involves clinical history and physical examination. Imaging with ultrasound and magnetic resonance imaging (MRI) may be utilized. While ultrasound may be preferred for superficial venous malformations, MRI or MRI with MR angiography (MRA) is the preferred method for venous malformation assessment. Genetic testing may be appropriate for complex malformations, as classification of lesions by underlying mutation may allow targeted therapy.
This patient’s past MRI and MRA findings were consistent with a venous malformation.
Treatment
Venous malformations rarely regress spontaneously. Treatment is required if venous malformations are symptomatic, which may include pain, swelling, deformity, thrombosis, or interference with daily activities of living. Treatment plans require consideration of patient goals of care. The main categories of therapy are embolization/sclerotherapy, surgical resection, and molecular targeted therapy.
Sclerotherapy is a well-tolerated and efficacious first-line therapy. It can be used as either nonsurgical curative therapy or preoperative adjunct therapy to minimize blood loss before surgical resection. While surgical resection may cause scarring, multimodal approaches with sclerotherapy or laser therapy can decrease complications. Molecular therapies aim to reduce vascular proliferation and symptoms. Referral to hematology/oncology for evaluation and consideration of chemotherapeutic agents may be required. Sirolimus has been shown in mice models to inhibit an endothelial cell tyrosine kinase receptor that plays a role in venous malformation growth. Multiple studies have proved its efficacy in managing complicated vascular anomalies, including venous malformations. Alpelisib is an inhibitor of PI3KCA, which is part of the pathway that contributes to venous malformation formation. Dactolisib, a dual inhibitor of the PI3KA and mTOR pathways, and rebastinib, a TEK inhibitor, are being investigated.
Differential Diagnoses
The differential diagnosis includes dermal melanocytosis, nevus of Ota, hemangioma of infancy, and ashy dermatosis. In addition, venous malformations can be part of more complex vascular malformations.
Dermal melanocytosis, also known as Mongolian spots, are blue-gray patches of discoloration on the skin that appear at birth or shortly after. They result from the arrest of dermal melanocytes in the dermis during fetal life and tissue modeling. They are commonly observed in those of Asian or African descent with darker skin types. Most often, they are located in the lumbar or sacral-gluteal region. Unlike venous malformations, they are benign and do not involve vascular abnormalities. They typically fade over time.
Nevus of Ota is a benign congenital condition that presents with blue-gray or brown patches of pigmentation on the skin around the eyes, cheeks, and forehead. They are dermal melanocytes with a speckled instead of uniform appearance. Nevus of Ota primarily affects individuals of Asian descent and typically presents in the trigeminal nerve distribution region. Treatment can be done to minimize deformity, generally with pigmented laser surgery.
Hemangiomas of infancy are common benign tumors of infancy caused by endothelial cell proliferation. They are characterized by rapid growth followed by spontaneous involution within the first year of life and for several years. Hemangiomas can be superficial, deep, or mixed with features of both superficial and deep. Superficial hemangiomas present as raised, lobulated, and bright red while deep hemangiomas present as a bluish-hued nodule, plaque, or tumor. They are diagnosed clinically but skin biopsies and imaging can confirm the suspected diagnosis. While hemangiomas may self-resolve, complicated hemangiomas can be treated with topical timolol, oral propranolol, topical and intralesional corticosteroids, pulsed-dye laser, and surgical resection.
Ashy dermatosis is a term for asymptomatic, gray-blue or ashy patches distributed symmetrically on the trunk, head, neck, and upper extremities. It primarily affects individuals with darker skin types (Fitzpatrick III-V), and is more common in patients with Hispanic, Asian, or African backgrounds. The direct cause of ashy dermatosis is unknown but it is thought to be linked to drug ingestion, genetics, infection, and immune-mediated mechanisms. The general treatment includes topical corticosteroids, clofazimine, topical calcineurin inhibitors, oral dapsone, phototherapy, topical retinoids, or isotretinoin to reduce inflammation and pigmentation.
Danny Lee and Samuel Le serve as research fellows and Jolina Bui as research associate in the Pediatric Dermatology Division of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is Distinguished Professor of Dermatology and Pediatrics and Vice-Chair of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. The authors have no relevant financial disclosures.
Suggested Reading
Agarwal P, Patel BC. Nevus of Ota and Ito. [Updated 2023 Jul 10]. In: StatPearls [Internet]. StatPearls Publishing; 2024.
Dompmartin A et al. The VASCERN-VASCA Working Group Diagnostic and Management Pathways for Venous Malformations. J Vasc Anom (Phila). 2023 Mar 23;4(2):e064.
Dompmartin A et al. Venous malformation: Update on aetiopathogenesis, diagnosis and management. Phlebology. 2010 Oct;25(5):224-235.
Gupta D, Thappa DM. Mongolian spots. Indian J Dermatol Venereol Leprol. 2013 Jul-Aug;79(4):469-478.
Krowchuk DP et al. Clinical Practice Guideline for the Management of Infantile Hemangiomas. Pediatrics. 2019 Jan;143(1):e20183475.
Nguyen K, Khachemoune A. Ashy dermatosis: A review. Dermatol Online J. 2019 May 15;25(5):13030/qt44f462s8.
Patel ND, Chong AT et al. Venous Malformations. Semin Intervent Radiol. 2022 Dec 20;39(5):498-507.
Diagnosis: Venous Malformation
Although present at birth, they are not always clinically evident early in life. They also tend to grow with the child without spontaneous regression, causing potential cosmetic concerns or complications from impingement on surrounding tissue.
Venous malformations appear with a bluish color appearing beneath the skin and can vary significantly in size and severity. Venous malformations are compressible and characterized by low to stagnant blood flow, which can spontaneously thrombose. Clinically, this may cause pain, swelling, skin changes, tissue and limb overgrowth, or functional impairment depending on location and size.
Venous malformations result from disorganized angiogenesis secondary to sporadic mutations in somatic cells. The most common implicated gene is TEK, a receptor tyrosine kinase. PIK3CA has also been involved. Both genes are involved in the PI3K/AKT/mTOR pathway, which regulates cell growth, proliferation, and angiogenesis. In venous endothelial cells, abnormal angiogenesis and vessel maturation may lead to venous malformation formation. Dysplastic vessels frequently separate from normal veins but may be contiguous with the deep venous system.
Diagnosis involves clinical history and physical examination. Imaging with ultrasound and magnetic resonance imaging (MRI) may be utilized. While ultrasound may be preferred for superficial venous malformations, MRI or MRI with MR angiography (MRA) is the preferred method for venous malformation assessment. Genetic testing may be appropriate for complex malformations, as classification of lesions by underlying mutation may allow targeted therapy.
This patient’s past MRI and MRA findings were consistent with a venous malformation.
Treatment
Venous malformations rarely regress spontaneously. Treatment is required if venous malformations are symptomatic, which may include pain, swelling, deformity, thrombosis, or interference with daily activities of living. Treatment plans require consideration of patient goals of care. The main categories of therapy are embolization/sclerotherapy, surgical resection, and molecular targeted therapy.
Sclerotherapy is a well-tolerated and efficacious first-line therapy. It can be used as either nonsurgical curative therapy or preoperative adjunct therapy to minimize blood loss before surgical resection. While surgical resection may cause scarring, multimodal approaches with sclerotherapy or laser therapy can decrease complications. Molecular therapies aim to reduce vascular proliferation and symptoms. Referral to hematology/oncology for evaluation and consideration of chemotherapeutic agents may be required. Sirolimus has been shown in mice models to inhibit an endothelial cell tyrosine kinase receptor that plays a role in venous malformation growth. Multiple studies have proved its efficacy in managing complicated vascular anomalies, including venous malformations. Alpelisib is an inhibitor of PI3KCA, which is part of the pathway that contributes to venous malformation formation. Dactolisib, a dual inhibitor of the PI3KA and mTOR pathways, and rebastinib, a TEK inhibitor, are being investigated.
Differential Diagnoses
The differential diagnosis includes dermal melanocytosis, nevus of Ota, hemangioma of infancy, and ashy dermatosis. In addition, venous malformations can be part of more complex vascular malformations.
Dermal melanocytosis, also known as Mongolian spots, are blue-gray patches of discoloration on the skin that appear at birth or shortly after. They result from the arrest of dermal melanocytes in the dermis during fetal life and tissue modeling. They are commonly observed in those of Asian or African descent with darker skin types. Most often, they are located in the lumbar or sacral-gluteal region. Unlike venous malformations, they are benign and do not involve vascular abnormalities. They typically fade over time.
Nevus of Ota is a benign congenital condition that presents with blue-gray or brown patches of pigmentation on the skin around the eyes, cheeks, and forehead. They are dermal melanocytes with a speckled instead of uniform appearance. Nevus of Ota primarily affects individuals of Asian descent and typically presents in the trigeminal nerve distribution region. Treatment can be done to minimize deformity, generally with pigmented laser surgery.
Hemangiomas of infancy are common benign tumors of infancy caused by endothelial cell proliferation. They are characterized by rapid growth followed by spontaneous involution within the first year of life and for several years. Hemangiomas can be superficial, deep, or mixed with features of both superficial and deep. Superficial hemangiomas present as raised, lobulated, and bright red while deep hemangiomas present as a bluish-hued nodule, plaque, or tumor. They are diagnosed clinically but skin biopsies and imaging can confirm the suspected diagnosis. While hemangiomas may self-resolve, complicated hemangiomas can be treated with topical timolol, oral propranolol, topical and intralesional corticosteroids, pulsed-dye laser, and surgical resection.
Ashy dermatosis is a term for asymptomatic, gray-blue or ashy patches distributed symmetrically on the trunk, head, neck, and upper extremities. It primarily affects individuals with darker skin types (Fitzpatrick III-V), and is more common in patients with Hispanic, Asian, or African backgrounds. The direct cause of ashy dermatosis is unknown but it is thought to be linked to drug ingestion, genetics, infection, and immune-mediated mechanisms. The general treatment includes topical corticosteroids, clofazimine, topical calcineurin inhibitors, oral dapsone, phototherapy, topical retinoids, or isotretinoin to reduce inflammation and pigmentation.
Danny Lee and Samuel Le serve as research fellows and Jolina Bui as research associate in the Pediatric Dermatology Division of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is Distinguished Professor of Dermatology and Pediatrics and Vice-Chair of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. The authors have no relevant financial disclosures.
Suggested Reading
Agarwal P, Patel BC. Nevus of Ota and Ito. [Updated 2023 Jul 10]. In: StatPearls [Internet]. StatPearls Publishing; 2024.
Dompmartin A et al. The VASCERN-VASCA Working Group Diagnostic and Management Pathways for Venous Malformations. J Vasc Anom (Phila). 2023 Mar 23;4(2):e064.
Dompmartin A et al. Venous malformation: Update on aetiopathogenesis, diagnosis and management. Phlebology. 2010 Oct;25(5):224-235.
Gupta D, Thappa DM. Mongolian spots. Indian J Dermatol Venereol Leprol. 2013 Jul-Aug;79(4):469-478.
Krowchuk DP et al. Clinical Practice Guideline for the Management of Infantile Hemangiomas. Pediatrics. 2019 Jan;143(1):e20183475.
Nguyen K, Khachemoune A. Ashy dermatosis: A review. Dermatol Online J. 2019 May 15;25(5):13030/qt44f462s8.
Patel ND, Chong AT et al. Venous Malformations. Semin Intervent Radiol. 2022 Dec 20;39(5):498-507.
A 6-year-old girl presents with a bruise-like lesion on the lip, tongue, and chin area present since birth. The family states that her tongue has been increasing in size and is painful. On physical exam, she presents with left lower mucosal lip fullness and an overlying violaceous hue extending into the oral mucosa and onto the left tongue. The left portion of the dorsal tongue displays an increased thickness and bluish discoloration and there is a pink, smooth papule on the left anterolateral tongue.
Study Evaluates CVD, Mortality Risks In Patients With Prurigo Nodularis
TOPLINE:
, particularly among women and White patients.
METHODOLOGY:
- Studies have shown increased risks for cardiovascular diseases in patients with PN, but limited sample sizes have hindered further subgroup analysis. Given PN’s pronounced sex and ethnicity skew, it is important to examine underrepresented groups to accurately assess their cardiovascular risk.
- In this propensity-score matched analysis, researchers identified 64,801 patients (59.44% women) with PN using electronic health reports from the Global Collaborative Network of TriNetX and matched to individuals without PN.
- Researchers calculated risks for 15 cardiovascular endpoints and all-cause mortality within 10 years of diagnosis. Major adverse cardiovascular events (MACE) included acute cerebral and myocardial infarction (MI), heart failure, ventricular arrhythmia, and sudden cardiac death.
TAKEAWAY:
- Patients with PN showed a higher risk for death (hazard ratio [HR], 1.1243) and MACE (HR, 1.117) (P < .0001 for both).
- PN was also associated with a higher risk for heart failure (HR, 1.062), thrombotic venous disease (HR, 1.26), angina pectoris (HR, 1.096), and peripheral arterial diseases (HR, 1.082) (P < .0001 for all) and for acute MI (HR, 1.11; P = .0015) and valve disorders (HR, 1.08; P = .0018).
- White patients with PN had a significantly increased risk for MACE, death, heart failure, cardiac arrest, vascular diseases, and acute MI, but this was not observed in people of color.
- Women exhibited a higher risk for MACE, heart failure, peripheral artery disease, acute MI, conduction disease, and valve disorders, while men did not have an increased risk for major or acute cardiovascular events. Both men and women had a higher risk for death, chronic ischemic heart disease, and venous disease.
IN PRACTICE:
“Although no novel PN-specific treatment rationale can be derived from the presented data, the potential risk of subsequent cardiovascular disease should be considered in the care of patients with PN, which includes screening and optimal management of other additional cardiovascular risk factors,” the authors wrote.
LIMITATIONS:
Retrospective observational design introduced inherent biases. Misdiagnosis or false coding in electronic health records could affect the data accuracy and ethnicity-specific analyses.
SOURCE:
This work, led by Henning Olbrich, from the Department of Dermatology, University of Lübeck, Germany, was published online in eBioMedicine.
DISCLOSURES:
The study was supported by the University of Lübeck, the Deutsche Forschungsgemeinschaft, and the State of Schleswig-Holstein. One author declared financial ties outside this work, and one author is an employee of TriNetX.
A version of this article appeared on Medscape.com.
TOPLINE:
, particularly among women and White patients.
METHODOLOGY:
- Studies have shown increased risks for cardiovascular diseases in patients with PN, but limited sample sizes have hindered further subgroup analysis. Given PN’s pronounced sex and ethnicity skew, it is important to examine underrepresented groups to accurately assess their cardiovascular risk.
- In this propensity-score matched analysis, researchers identified 64,801 patients (59.44% women) with PN using electronic health reports from the Global Collaborative Network of TriNetX and matched to individuals without PN.
- Researchers calculated risks for 15 cardiovascular endpoints and all-cause mortality within 10 years of diagnosis. Major adverse cardiovascular events (MACE) included acute cerebral and myocardial infarction (MI), heart failure, ventricular arrhythmia, and sudden cardiac death.
TAKEAWAY:
- Patients with PN showed a higher risk for death (hazard ratio [HR], 1.1243) and MACE (HR, 1.117) (P < .0001 for both).
- PN was also associated with a higher risk for heart failure (HR, 1.062), thrombotic venous disease (HR, 1.26), angina pectoris (HR, 1.096), and peripheral arterial diseases (HR, 1.082) (P < .0001 for all) and for acute MI (HR, 1.11; P = .0015) and valve disorders (HR, 1.08; P = .0018).
- White patients with PN had a significantly increased risk for MACE, death, heart failure, cardiac arrest, vascular diseases, and acute MI, but this was not observed in people of color.
- Women exhibited a higher risk for MACE, heart failure, peripheral artery disease, acute MI, conduction disease, and valve disorders, while men did not have an increased risk for major or acute cardiovascular events. Both men and women had a higher risk for death, chronic ischemic heart disease, and venous disease.
IN PRACTICE:
“Although no novel PN-specific treatment rationale can be derived from the presented data, the potential risk of subsequent cardiovascular disease should be considered in the care of patients with PN, which includes screening and optimal management of other additional cardiovascular risk factors,” the authors wrote.
LIMITATIONS:
Retrospective observational design introduced inherent biases. Misdiagnosis or false coding in electronic health records could affect the data accuracy and ethnicity-specific analyses.
SOURCE:
This work, led by Henning Olbrich, from the Department of Dermatology, University of Lübeck, Germany, was published online in eBioMedicine.
DISCLOSURES:
The study was supported by the University of Lübeck, the Deutsche Forschungsgemeinschaft, and the State of Schleswig-Holstein. One author declared financial ties outside this work, and one author is an employee of TriNetX.
A version of this article appeared on Medscape.com.
TOPLINE:
, particularly among women and White patients.
METHODOLOGY:
- Studies have shown increased risks for cardiovascular diseases in patients with PN, but limited sample sizes have hindered further subgroup analysis. Given PN’s pronounced sex and ethnicity skew, it is important to examine underrepresented groups to accurately assess their cardiovascular risk.
- In this propensity-score matched analysis, researchers identified 64,801 patients (59.44% women) with PN using electronic health reports from the Global Collaborative Network of TriNetX and matched to individuals without PN.
- Researchers calculated risks for 15 cardiovascular endpoints and all-cause mortality within 10 years of diagnosis. Major adverse cardiovascular events (MACE) included acute cerebral and myocardial infarction (MI), heart failure, ventricular arrhythmia, and sudden cardiac death.
TAKEAWAY:
- Patients with PN showed a higher risk for death (hazard ratio [HR], 1.1243) and MACE (HR, 1.117) (P < .0001 for both).
- PN was also associated with a higher risk for heart failure (HR, 1.062), thrombotic venous disease (HR, 1.26), angina pectoris (HR, 1.096), and peripheral arterial diseases (HR, 1.082) (P < .0001 for all) and for acute MI (HR, 1.11; P = .0015) and valve disorders (HR, 1.08; P = .0018).
- White patients with PN had a significantly increased risk for MACE, death, heart failure, cardiac arrest, vascular diseases, and acute MI, but this was not observed in people of color.
- Women exhibited a higher risk for MACE, heart failure, peripheral artery disease, acute MI, conduction disease, and valve disorders, while men did not have an increased risk for major or acute cardiovascular events. Both men and women had a higher risk for death, chronic ischemic heart disease, and venous disease.
IN PRACTICE:
“Although no novel PN-specific treatment rationale can be derived from the presented data, the potential risk of subsequent cardiovascular disease should be considered in the care of patients with PN, which includes screening and optimal management of other additional cardiovascular risk factors,” the authors wrote.
LIMITATIONS:
Retrospective observational design introduced inherent biases. Misdiagnosis or false coding in electronic health records could affect the data accuracy and ethnicity-specific analyses.
SOURCE:
This work, led by Henning Olbrich, from the Department of Dermatology, University of Lübeck, Germany, was published online in eBioMedicine.
DISCLOSURES:
The study was supported by the University of Lübeck, the Deutsche Forschungsgemeinschaft, and the State of Schleswig-Holstein. One author declared financial ties outside this work, and one author is an employee of TriNetX.
A version of this article appeared on Medscape.com.
Consider a Four-Step Approach to Shared Decision-Making in Pediatric Dermatology
SAN DIEGO — In the clinical experience of Kelly M. Cordoro, MD, .
“SDM is a cornerstone of person-centered care,” Dr. Cordoro, professor of dermatology and pediatrics at the University of California, San Francisco, said at the Society for Pediatric Dermatology meeting, held in advance of the annual meeting of the American Academy of Dermatology. “We do it all the time. It can be patient-led, clinician-led, or a patient/family dyad approach. If we do it well, it can improve outcomes. Patients report more satisfying interactions with their care team. It brings adolescent patients especially a sense of independence and they adapt faster to their illness.”
Conditions such as acne, psoriasis, and atopic dermatitis have multiple treatment options, often without a single best choice. The ideal treatment depends on disease characteristics (extent, sites affected, symptoms, and natural history), the patient (age, comorbidities, overall disease burden), therapies (safety, efficacy, duration, and adverse events), and preferences (logistics, time, shots vs. pills, etc.). “These factors vary between patients and within the same patient over time, and at each step along the course of the condition, SDM approaches are relevant,” she said.
AHRQ’s Five-Step Approach
The Agency for Healthcare Research and Quality developed a five-step approach to SDM known as SHARE: Seek your patient’s participation; Help your patient explore and compare treatment options; Assess your patient’s values and preferences; Reach a decision with your patient, and Evaluate your patient’s decision. “We do this all the time in practice with adult patients, but may not label it as SDM,” said Dr. Cordoro, chief and fellowship director of pediatric dermatology at UCSF.
“Where it gets a little murkier is in pediatric decision-making, which is a complex type of surrogate decision-making.” In this situation the patient — a minor — does not have full autonomy. The challenge for caregivers is that giving or withholding permission for interventions is a difficult role. “Their job is to protect the patient’s well-being while empowering them toward independence,” she said. “It can be hard for caregivers to understand complex information.” The challenge for clinicians, she continued, is to know when to invite SDM. This requires relational and sharp communication skills. “We must consider our patient’s/family’s health literacy and be sure the information we share is understood,” she said. “What are the social and structural determinants of health that are going to influence decision-making? You want to move into a relationship like this with cultural humility so you can understand what their preferences are and how they’re seeing the problem. Because there’s no universal agreement on the age at which minors should be deemed decision-making competent in health care, the approach is nuanced and depends on each individual patient and family.”
Dr. Cordoro proposed the following four-step approach to SDM to use in pediatric dermatology:
Step 1: Share relevant information about the condition and treatment options in a clear and understandable manner. The average US resident is at the seventh-to eighth-grade level, “so we have to avoid medical jargon and use plain language,” Dr. Cordoro said. Then, use the teach-back approach to assess their understanding. “Ask, ‘What is your understanding of the most important points that we talked about?’ Or, ‘Please share with me what you heard so I’m sure we all understand the plan.’ Using these techniques will reduce the barriers to care such as health literacy.”
Step 2: Solicit and understand patient/patient family perspectives, preferences and priorities. The goal here is to uncover their beliefs, concerns, and assumptions that may influence their decisions. “Be mindful of power asymmetry,” she noted. “Many families still believe the doctor is the boss and they are there to be told what to do. Be clear that the patient has a say. Talk directly to the patient about their interests if developmentally appropriate.”
Step 3: Invite patients/family into a shared decision-making conversation. Consider statements like, “There are many reasonable options here. Let’s work together to come up with the decision that’s right for you.” Or, “Let’s start by exploring your specific goals and concerns. As you think about the options I just talked to you about, what’s important to you?” Or, “Do you want to think about this decision with anyone else?”
Step 4: Check back in frequently. Pause between significant points and check in. “See how they’re doing during the conversation,” she said. “At future appointments, remember to solicit their input on additional decisions.”
In Dr. Cordoro’s opinion, one potential pitfall of SDM is an over-reliance on patient decision aids. “Very few are available in dermatology,” she said. “Some are relevant but none specifically to pediatric dermatology. They are often complex and require a high reading comprehension level. This disadvantages patients and families with low health literacy. Keep it clear and simple. Your patients will appreciate it.”
Dr. Cordoro reported having no relevant disclosures.
SAN DIEGO — In the clinical experience of Kelly M. Cordoro, MD, .
“SDM is a cornerstone of person-centered care,” Dr. Cordoro, professor of dermatology and pediatrics at the University of California, San Francisco, said at the Society for Pediatric Dermatology meeting, held in advance of the annual meeting of the American Academy of Dermatology. “We do it all the time. It can be patient-led, clinician-led, or a patient/family dyad approach. If we do it well, it can improve outcomes. Patients report more satisfying interactions with their care team. It brings adolescent patients especially a sense of independence and they adapt faster to their illness.”
Conditions such as acne, psoriasis, and atopic dermatitis have multiple treatment options, often without a single best choice. The ideal treatment depends on disease characteristics (extent, sites affected, symptoms, and natural history), the patient (age, comorbidities, overall disease burden), therapies (safety, efficacy, duration, and adverse events), and preferences (logistics, time, shots vs. pills, etc.). “These factors vary between patients and within the same patient over time, and at each step along the course of the condition, SDM approaches are relevant,” she said.
AHRQ’s Five-Step Approach
The Agency for Healthcare Research and Quality developed a five-step approach to SDM known as SHARE: Seek your patient’s participation; Help your patient explore and compare treatment options; Assess your patient’s values and preferences; Reach a decision with your patient, and Evaluate your patient’s decision. “We do this all the time in practice with adult patients, but may not label it as SDM,” said Dr. Cordoro, chief and fellowship director of pediatric dermatology at UCSF.
“Where it gets a little murkier is in pediatric decision-making, which is a complex type of surrogate decision-making.” In this situation the patient — a minor — does not have full autonomy. The challenge for caregivers is that giving or withholding permission for interventions is a difficult role. “Their job is to protect the patient’s well-being while empowering them toward independence,” she said. “It can be hard for caregivers to understand complex information.” The challenge for clinicians, she continued, is to know when to invite SDM. This requires relational and sharp communication skills. “We must consider our patient’s/family’s health literacy and be sure the information we share is understood,” she said. “What are the social and structural determinants of health that are going to influence decision-making? You want to move into a relationship like this with cultural humility so you can understand what their preferences are and how they’re seeing the problem. Because there’s no universal agreement on the age at which minors should be deemed decision-making competent in health care, the approach is nuanced and depends on each individual patient and family.”
Dr. Cordoro proposed the following four-step approach to SDM to use in pediatric dermatology:
Step 1: Share relevant information about the condition and treatment options in a clear and understandable manner. The average US resident is at the seventh-to eighth-grade level, “so we have to avoid medical jargon and use plain language,” Dr. Cordoro said. Then, use the teach-back approach to assess their understanding. “Ask, ‘What is your understanding of the most important points that we talked about?’ Or, ‘Please share with me what you heard so I’m sure we all understand the plan.’ Using these techniques will reduce the barriers to care such as health literacy.”
Step 2: Solicit and understand patient/patient family perspectives, preferences and priorities. The goal here is to uncover their beliefs, concerns, and assumptions that may influence their decisions. “Be mindful of power asymmetry,” she noted. “Many families still believe the doctor is the boss and they are there to be told what to do. Be clear that the patient has a say. Talk directly to the patient about their interests if developmentally appropriate.”
Step 3: Invite patients/family into a shared decision-making conversation. Consider statements like, “There are many reasonable options here. Let’s work together to come up with the decision that’s right for you.” Or, “Let’s start by exploring your specific goals and concerns. As you think about the options I just talked to you about, what’s important to you?” Or, “Do you want to think about this decision with anyone else?”
Step 4: Check back in frequently. Pause between significant points and check in. “See how they’re doing during the conversation,” she said. “At future appointments, remember to solicit their input on additional decisions.”
In Dr. Cordoro’s opinion, one potential pitfall of SDM is an over-reliance on patient decision aids. “Very few are available in dermatology,” she said. “Some are relevant but none specifically to pediatric dermatology. They are often complex and require a high reading comprehension level. This disadvantages patients and families with low health literacy. Keep it clear and simple. Your patients will appreciate it.”
Dr. Cordoro reported having no relevant disclosures.
SAN DIEGO — In the clinical experience of Kelly M. Cordoro, MD, .
“SDM is a cornerstone of person-centered care,” Dr. Cordoro, professor of dermatology and pediatrics at the University of California, San Francisco, said at the Society for Pediatric Dermatology meeting, held in advance of the annual meeting of the American Academy of Dermatology. “We do it all the time. It can be patient-led, clinician-led, or a patient/family dyad approach. If we do it well, it can improve outcomes. Patients report more satisfying interactions with their care team. It brings adolescent patients especially a sense of independence and they adapt faster to their illness.”
Conditions such as acne, psoriasis, and atopic dermatitis have multiple treatment options, often without a single best choice. The ideal treatment depends on disease characteristics (extent, sites affected, symptoms, and natural history), the patient (age, comorbidities, overall disease burden), therapies (safety, efficacy, duration, and adverse events), and preferences (logistics, time, shots vs. pills, etc.). “These factors vary between patients and within the same patient over time, and at each step along the course of the condition, SDM approaches are relevant,” she said.
AHRQ’s Five-Step Approach
The Agency for Healthcare Research and Quality developed a five-step approach to SDM known as SHARE: Seek your patient’s participation; Help your patient explore and compare treatment options; Assess your patient’s values and preferences; Reach a decision with your patient, and Evaluate your patient’s decision. “We do this all the time in practice with adult patients, but may not label it as SDM,” said Dr. Cordoro, chief and fellowship director of pediatric dermatology at UCSF.
“Where it gets a little murkier is in pediatric decision-making, which is a complex type of surrogate decision-making.” In this situation the patient — a minor — does not have full autonomy. The challenge for caregivers is that giving or withholding permission for interventions is a difficult role. “Their job is to protect the patient’s well-being while empowering them toward independence,” she said. “It can be hard for caregivers to understand complex information.” The challenge for clinicians, she continued, is to know when to invite SDM. This requires relational and sharp communication skills. “We must consider our patient’s/family’s health literacy and be sure the information we share is understood,” she said. “What are the social and structural determinants of health that are going to influence decision-making? You want to move into a relationship like this with cultural humility so you can understand what their preferences are and how they’re seeing the problem. Because there’s no universal agreement on the age at which minors should be deemed decision-making competent in health care, the approach is nuanced and depends on each individual patient and family.”
Dr. Cordoro proposed the following four-step approach to SDM to use in pediatric dermatology:
Step 1: Share relevant information about the condition and treatment options in a clear and understandable manner. The average US resident is at the seventh-to eighth-grade level, “so we have to avoid medical jargon and use plain language,” Dr. Cordoro said. Then, use the teach-back approach to assess their understanding. “Ask, ‘What is your understanding of the most important points that we talked about?’ Or, ‘Please share with me what you heard so I’m sure we all understand the plan.’ Using these techniques will reduce the barriers to care such as health literacy.”
Step 2: Solicit and understand patient/patient family perspectives, preferences and priorities. The goal here is to uncover their beliefs, concerns, and assumptions that may influence their decisions. “Be mindful of power asymmetry,” she noted. “Many families still believe the doctor is the boss and they are there to be told what to do. Be clear that the patient has a say. Talk directly to the patient about their interests if developmentally appropriate.”
Step 3: Invite patients/family into a shared decision-making conversation. Consider statements like, “There are many reasonable options here. Let’s work together to come up with the decision that’s right for you.” Or, “Let’s start by exploring your specific goals and concerns. As you think about the options I just talked to you about, what’s important to you?” Or, “Do you want to think about this decision with anyone else?”
Step 4: Check back in frequently. Pause between significant points and check in. “See how they’re doing during the conversation,” she said. “At future appointments, remember to solicit their input on additional decisions.”
In Dr. Cordoro’s opinion, one potential pitfall of SDM is an over-reliance on patient decision aids. “Very few are available in dermatology,” she said. “Some are relevant but none specifically to pediatric dermatology. They are often complex and require a high reading comprehension level. This disadvantages patients and families with low health literacy. Keep it clear and simple. Your patients will appreciate it.”
Dr. Cordoro reported having no relevant disclosures.
FROM AAD 2024
New HIV Infections After Vampire Facials at Unlicensed Spa
At least three clients of an unlicensed spa in New Mexico contracted HIV after receiving platelet-rich plasma (PRP) microneedling facials, according to an investigation by the US Centers for Disease Control and Prevention (CDC).
The investigation, spanning 5 years with parts of it still ongoing, has resulted in the closure of the spa and is raising questions about public safety in cosmetic clinics.
Though transmission of HIV by unsterile injection practices is a known risk, this is the first time it has been linked to cosmetic injection services, said Anna Stadelman-Behar, PhD, MPH, of the CDC’s Epidemic Intelligence Service.
Sometimes called a vampire facial, the PRP treatment involves taking a patient’s own blood and separating it in a centrifuge. The portion containing a high concentration of platelets is then reinjected with a syringe or microneedling device.
“The idea is that when you inject this concentrated amount of platelets, the growth factors that the platelets release help to stimulate the regenerative nature of that area,” said Anthony Rossi, MD, professor of dermatology at Weill Cornell Medical College in New York, and attending dermatologist at Memorial Sloan Kettering Cancer Center.
The infections under investigation first came to light when a woman was diagnosed with HIV with no known risk factors for the disease other than exposure to microneedling facials at a cosmetic spa.
The New Mexico Department of Health and the CDC launched an investigation of the spa and discovered a litany of “gross violations of infection control practices,” said Dr. Stadelman-Behar.
Infection-Control Violations
At the spa in New Mexico, investigators found:
- On a kitchen counter, a centrifuge, a heating dry bath, and a rack of unlabeled tubes containing blood
- In a refrigerator, unlabeled tubes of blood and medical injectables including botox and lidocaine stored along with food
- Unwrapped syringes in drawers, on counters, and discarded in regular trash cans
- No autoclave for steam sterilization on the premises
- Only surface cleaning for procedure equipment with ammonium chloride disinfecting spray and benzalkonium chloride disinfecting wipes after each client visit
- Disposable electric desiccator tips cleaned only by alcohol immersion to be reused
The spa’s owner operated without appropriate licenses at multiple locations and did not have an appointment scheduling system that stored client contact information.
Investigators contacted as many people as they could find and launched a large-scale community outreach effort to find more.
In total, four clients and one intimate partner of a client were diagnosed with HIV during the investigation, but one client and her partner were determined to likely have been infected before the spa visit.
It is not clear whether the infections were due to unlabeled contaminated blood products being given to the wrong client or contamination on shared needles. Investigators did not have the authority to collect specimens during their site visit that would have allowed them to study that.
“We can’t definitively say what the route of contamination was,” noted Dr. Stadelman-Behar.
Anne Chapas, MD, a board-certified dermatologist, and instructor at Mount Sinai Hospital in New York, added that just because a procedure is cosmetic, that doesn’t mean it is not medical. “Personally, I feel it should only be done by medical practitioners who understand the risks.”
A Medical Procedure
PRP microneedling has been used extensively in orthopedic surgery to promote joint regeneration. For the past 10 years, it has also been used in dermatology to treat hair loss from alopecia, to augment wound healing, and cosmetically to reduce facial wrinkles.
It is generally done in a doctor’s office or medical spa, and the procedure takes about half an hour.
Dr. Stadelman-Behar said that this ongoing investigation highlights the importance of front-line healthcare workers using their clinical expertise to help identify potential new routes of transmission for infections. “It was provider-led intuition that sparked this investigation, so it’s important to let the department of health know if there is something amiss with any of the exposures that the patient might have had,” she said.
A version of this article appeared on Medscape.com.
At least three clients of an unlicensed spa in New Mexico contracted HIV after receiving platelet-rich plasma (PRP) microneedling facials, according to an investigation by the US Centers for Disease Control and Prevention (CDC).
The investigation, spanning 5 years with parts of it still ongoing, has resulted in the closure of the spa and is raising questions about public safety in cosmetic clinics.
Though transmission of HIV by unsterile injection practices is a known risk, this is the first time it has been linked to cosmetic injection services, said Anna Stadelman-Behar, PhD, MPH, of the CDC’s Epidemic Intelligence Service.
Sometimes called a vampire facial, the PRP treatment involves taking a patient’s own blood and separating it in a centrifuge. The portion containing a high concentration of platelets is then reinjected with a syringe or microneedling device.
“The idea is that when you inject this concentrated amount of platelets, the growth factors that the platelets release help to stimulate the regenerative nature of that area,” said Anthony Rossi, MD, professor of dermatology at Weill Cornell Medical College in New York, and attending dermatologist at Memorial Sloan Kettering Cancer Center.
The infections under investigation first came to light when a woman was diagnosed with HIV with no known risk factors for the disease other than exposure to microneedling facials at a cosmetic spa.
The New Mexico Department of Health and the CDC launched an investigation of the spa and discovered a litany of “gross violations of infection control practices,” said Dr. Stadelman-Behar.
Infection-Control Violations
At the spa in New Mexico, investigators found:
- On a kitchen counter, a centrifuge, a heating dry bath, and a rack of unlabeled tubes containing blood
- In a refrigerator, unlabeled tubes of blood and medical injectables including botox and lidocaine stored along with food
- Unwrapped syringes in drawers, on counters, and discarded in regular trash cans
- No autoclave for steam sterilization on the premises
- Only surface cleaning for procedure equipment with ammonium chloride disinfecting spray and benzalkonium chloride disinfecting wipes after each client visit
- Disposable electric desiccator tips cleaned only by alcohol immersion to be reused
The spa’s owner operated without appropriate licenses at multiple locations and did not have an appointment scheduling system that stored client contact information.
Investigators contacted as many people as they could find and launched a large-scale community outreach effort to find more.
In total, four clients and one intimate partner of a client were diagnosed with HIV during the investigation, but one client and her partner were determined to likely have been infected before the spa visit.
It is not clear whether the infections were due to unlabeled contaminated blood products being given to the wrong client or contamination on shared needles. Investigators did not have the authority to collect specimens during their site visit that would have allowed them to study that.
“We can’t definitively say what the route of contamination was,” noted Dr. Stadelman-Behar.
Anne Chapas, MD, a board-certified dermatologist, and instructor at Mount Sinai Hospital in New York, added that just because a procedure is cosmetic, that doesn’t mean it is not medical. “Personally, I feel it should only be done by medical practitioners who understand the risks.”
A Medical Procedure
PRP microneedling has been used extensively in orthopedic surgery to promote joint regeneration. For the past 10 years, it has also been used in dermatology to treat hair loss from alopecia, to augment wound healing, and cosmetically to reduce facial wrinkles.
It is generally done in a doctor’s office or medical spa, and the procedure takes about half an hour.
Dr. Stadelman-Behar said that this ongoing investigation highlights the importance of front-line healthcare workers using their clinical expertise to help identify potential new routes of transmission for infections. “It was provider-led intuition that sparked this investigation, so it’s important to let the department of health know if there is something amiss with any of the exposures that the patient might have had,” she said.
A version of this article appeared on Medscape.com.
At least three clients of an unlicensed spa in New Mexico contracted HIV after receiving platelet-rich plasma (PRP) microneedling facials, according to an investigation by the US Centers for Disease Control and Prevention (CDC).
The investigation, spanning 5 years with parts of it still ongoing, has resulted in the closure of the spa and is raising questions about public safety in cosmetic clinics.
Though transmission of HIV by unsterile injection practices is a known risk, this is the first time it has been linked to cosmetic injection services, said Anna Stadelman-Behar, PhD, MPH, of the CDC’s Epidemic Intelligence Service.
Sometimes called a vampire facial, the PRP treatment involves taking a patient’s own blood and separating it in a centrifuge. The portion containing a high concentration of platelets is then reinjected with a syringe or microneedling device.
“The idea is that when you inject this concentrated amount of platelets, the growth factors that the platelets release help to stimulate the regenerative nature of that area,” said Anthony Rossi, MD, professor of dermatology at Weill Cornell Medical College in New York, and attending dermatologist at Memorial Sloan Kettering Cancer Center.
The infections under investigation first came to light when a woman was diagnosed with HIV with no known risk factors for the disease other than exposure to microneedling facials at a cosmetic spa.
The New Mexico Department of Health and the CDC launched an investigation of the spa and discovered a litany of “gross violations of infection control practices,” said Dr. Stadelman-Behar.
Infection-Control Violations
At the spa in New Mexico, investigators found:
- On a kitchen counter, a centrifuge, a heating dry bath, and a rack of unlabeled tubes containing blood
- In a refrigerator, unlabeled tubes of blood and medical injectables including botox and lidocaine stored along with food
- Unwrapped syringes in drawers, on counters, and discarded in regular trash cans
- No autoclave for steam sterilization on the premises
- Only surface cleaning for procedure equipment with ammonium chloride disinfecting spray and benzalkonium chloride disinfecting wipes after each client visit
- Disposable electric desiccator tips cleaned only by alcohol immersion to be reused
The spa’s owner operated without appropriate licenses at multiple locations and did not have an appointment scheduling system that stored client contact information.
Investigators contacted as many people as they could find and launched a large-scale community outreach effort to find more.
In total, four clients and one intimate partner of a client were diagnosed with HIV during the investigation, but one client and her partner were determined to likely have been infected before the spa visit.
It is not clear whether the infections were due to unlabeled contaminated blood products being given to the wrong client or contamination on shared needles. Investigators did not have the authority to collect specimens during their site visit that would have allowed them to study that.
“We can’t definitively say what the route of contamination was,” noted Dr. Stadelman-Behar.
Anne Chapas, MD, a board-certified dermatologist, and instructor at Mount Sinai Hospital in New York, added that just because a procedure is cosmetic, that doesn’t mean it is not medical. “Personally, I feel it should only be done by medical practitioners who understand the risks.”
A Medical Procedure
PRP microneedling has been used extensively in orthopedic surgery to promote joint regeneration. For the past 10 years, it has also been used in dermatology to treat hair loss from alopecia, to augment wound healing, and cosmetically to reduce facial wrinkles.
It is generally done in a doctor’s office or medical spa, and the procedure takes about half an hour.
Dr. Stadelman-Behar said that this ongoing investigation highlights the importance of front-line healthcare workers using their clinical expertise to help identify potential new routes of transmission for infections. “It was provider-led intuition that sparked this investigation, so it’s important to let the department of health know if there is something amiss with any of the exposures that the patient might have had,” she said.
A version of this article appeared on Medscape.com.
Mpox Presentation Compared in Different Racial, Ethnic Groups
TOPLINE:
.
METHODOLOGY:
- There is limited information on the populations disproportionately affected by the recent global mpox outbreak, particularly in individuals with HIV and racial and ethnic minorities.
- To investigate morphologic and clinical presentations of mpox in diverse populations, researchers conducted a review of the records of 54 individuals (mean age, 42.4 years) diagnosed with mpox at a San Francisco clinic for patients with HIV or at high risk for HIV, between June and October 2022.
- All patients were assigned male at birth, and three identified themselves as transgender women.
- Morphologic descriptions were documented through either photographic evidence or physical examination notes.
TAKEAWAY:
- Pustules or pseudopustules were the most common morphologic finding in 57.1% of the White non-Hispanic patients and 62.5% of the patients of color (P = .72).
- White non-Hispanic patients were more likely to have no prodromal symptoms (50.0% vs 17.5%; P = .02) and were more likely to have genital lesions (78.6% vs 40.0%; P = .01) than patients of color. These differences were significant or nearly significant when White non-Hispanic patients were compared with Hispanic patients but not in other ethnic or racial groups.
- There were no differences in HIV viral loads or CD4 counts between racial and ethnic groups, and no variations in clinical presentations were observed based on CD4 counts.
- Patients with higher HIV viral loads were more likely to have concurrent sexually transmitted infections (57.1% vs 25%; P = .03).
- Symptoms resolved in all patients, regardless of medical intervention, within weeks of initial presentation, and there were no hospitalizations or deaths.
IN PRACTICE:
Considering that HIV viral burden was not significantly different between White non-Hispanic patients and patients of color, the difference in presentation of the prodrome “may indicate disparities in vulnerable populations,” the authors wrote, noting that more research in large groups is needed to confirm their results.
SOURCE:
The study, led by Richard W. Kim, BS, from the University of California San Francisco, was published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
Inclusion of “other” racial category in the records highlighted potential inaccuracies in data representation.
DISCLOSURES:
The study received no external funding. The authors did not declare any competing interests.
A version of this article first appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- There is limited information on the populations disproportionately affected by the recent global mpox outbreak, particularly in individuals with HIV and racial and ethnic minorities.
- To investigate morphologic and clinical presentations of mpox in diverse populations, researchers conducted a review of the records of 54 individuals (mean age, 42.4 years) diagnosed with mpox at a San Francisco clinic for patients with HIV or at high risk for HIV, between June and October 2022.
- All patients were assigned male at birth, and three identified themselves as transgender women.
- Morphologic descriptions were documented through either photographic evidence or physical examination notes.
TAKEAWAY:
- Pustules or pseudopustules were the most common morphologic finding in 57.1% of the White non-Hispanic patients and 62.5% of the patients of color (P = .72).
- White non-Hispanic patients were more likely to have no prodromal symptoms (50.0% vs 17.5%; P = .02) and were more likely to have genital lesions (78.6% vs 40.0%; P = .01) than patients of color. These differences were significant or nearly significant when White non-Hispanic patients were compared with Hispanic patients but not in other ethnic or racial groups.
- There were no differences in HIV viral loads or CD4 counts between racial and ethnic groups, and no variations in clinical presentations were observed based on CD4 counts.
- Patients with higher HIV viral loads were more likely to have concurrent sexually transmitted infections (57.1% vs 25%; P = .03).
- Symptoms resolved in all patients, regardless of medical intervention, within weeks of initial presentation, and there were no hospitalizations or deaths.
IN PRACTICE:
Considering that HIV viral burden was not significantly different between White non-Hispanic patients and patients of color, the difference in presentation of the prodrome “may indicate disparities in vulnerable populations,” the authors wrote, noting that more research in large groups is needed to confirm their results.
SOURCE:
The study, led by Richard W. Kim, BS, from the University of California San Francisco, was published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
Inclusion of “other” racial category in the records highlighted potential inaccuracies in data representation.
DISCLOSURES:
The study received no external funding. The authors did not declare any competing interests.
A version of this article first appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- There is limited information on the populations disproportionately affected by the recent global mpox outbreak, particularly in individuals with HIV and racial and ethnic minorities.
- To investigate morphologic and clinical presentations of mpox in diverse populations, researchers conducted a review of the records of 54 individuals (mean age, 42.4 years) diagnosed with mpox at a San Francisco clinic for patients with HIV or at high risk for HIV, between June and October 2022.
- All patients were assigned male at birth, and three identified themselves as transgender women.
- Morphologic descriptions were documented through either photographic evidence or physical examination notes.
TAKEAWAY:
- Pustules or pseudopustules were the most common morphologic finding in 57.1% of the White non-Hispanic patients and 62.5% of the patients of color (P = .72).
- White non-Hispanic patients were more likely to have no prodromal symptoms (50.0% vs 17.5%; P = .02) and were more likely to have genital lesions (78.6% vs 40.0%; P = .01) than patients of color. These differences were significant or nearly significant when White non-Hispanic patients were compared with Hispanic patients but not in other ethnic or racial groups.
- There were no differences in HIV viral loads or CD4 counts between racial and ethnic groups, and no variations in clinical presentations were observed based on CD4 counts.
- Patients with higher HIV viral loads were more likely to have concurrent sexually transmitted infections (57.1% vs 25%; P = .03).
- Symptoms resolved in all patients, regardless of medical intervention, within weeks of initial presentation, and there were no hospitalizations or deaths.
IN PRACTICE:
Considering that HIV viral burden was not significantly different between White non-Hispanic patients and patients of color, the difference in presentation of the prodrome “may indicate disparities in vulnerable populations,” the authors wrote, noting that more research in large groups is needed to confirm their results.
SOURCE:
The study, led by Richard W. Kim, BS, from the University of California San Francisco, was published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
Inclusion of “other” racial category in the records highlighted potential inaccuracies in data representation.
DISCLOSURES:
The study received no external funding. The authors did not declare any competing interests.
A version of this article first appeared on Medscape.com.
In the Story of the Rubella Virus as a Source of Granulomas, the Plot Is Still Thickening
SAN DIEGO — Approximately 10 years ago in France, high throughput .
Based on accumulating evidence, the Centers for Disease Control and Prevention (CDC) through collaborations with others also recognized this in pediatric patients with inborn errors of immunity, and it is now appropriate for clinicians to consider this etiology when no other infectious agents can be identified, according to Karolyn A. Wanat, MD, professor of dermatology, Medical College of Wisconsin, Milwaukee, who spoke about rubella as a trigger in granulomatous disease at the meeting. “This is a huge evolving area of interest,” said Dr. Wanat, who has been the first author or coauthor on several published papers, including a review article published earlier this year.
In the earliest cases, including those reported in 2014, the cutaneous granulomas presumed to be causally related to vaccine-derived rubella virus were found only in those with a primary immunodeficiency. This is no longer the case. In a collaboration among US clinics, granulomas that had persisted for years in immunocompetent adults were identified, according to Dr. Wanat, the first author of a report on these findings in four adults in 2022. In addition, it now appears that wild-type rubella virus, like vaccine-derived rubella virus, can be the source of the antigenic response that underlies the development of rubella-associated cutaneous granulomas.
The phenotype of these granulomas is comparable to granulomas associated with other infectious agents. On dermatopathology, these commonly feature a robust granulomatous inflammation with multinucleated giant cells and lymphocytic infiltrate. Necrosis and fibrosis are also common.
“These are the types of granulomas that we would be thinking infection. If tissue cultures are negative, we would probably repeat them,” she said, suggesting that suspicion of an infectious etiology would probably remain high even after multiple negative tests.
Of the cases accruing in the United States and elsewhere, most but not all have been linked to inborn errors of immunity. In a 2020 CDC review, the risk of granulomas caused by compromised immunity, such as defects in T cell function, was estimated to be in the range of 0.6% to 2.5%, Dr. Wanat said.
It is now known that primary immunodeficiency is not a prerequisite, but this should not change the perception that the rubella vaccine, which was introduced in 1979, is effective and safe, according to Dr. Wanat. The vaccine is associated with few serious adverse events and is so effective that rubella was eliminated from the United States in 2004 and from the Americas in 2015.
This makes cases of granuloma associated with wild-type rubella virus surprising, but they appear to be exceedingly rare. Whether caused by vaccine exposure or another source, the mechanism of latent development of cutaneous granulomas is consistent with other infectious sources, and is not well understood.
“Rubella is a sneaky virus that can persist in some immunoprivileged sites indefinitely,” Dr. Wanat said. These sites include the eyes, joints, and placenta.
Many initial cases of rubella-associated granulomas occurred on the arms, presumably where the vaccine was administered, despite long intervals between exposure and lesion growth. This interval is often measured in years.
With more cases, it is now understood that involvement of other organs does occur even if the skin is the most common site of antigenic response in patients with immunodeficiency. The liver and lymph nodes represent other tissues that have been affected. Even lesions in the brain have been seen on autopsy.
Based on the benefit-to-risk ratio of a highly effective and successful vaccine, however, the association with a risk of granulomas “should not raise questions” about the value of the vaccine itself, Dr. Wanat noted.
“The proportion of patients who develop these granulomas is very, very low. Yet, the vaccine provides life-long immunity,” she said.
The discovery of granulomas associated with wild type rubella infection was “shocking” based on the supposition that the rubella virus had been eliminated, but this is just one of the unexpected discoveries as the still-evolving science has traced the story of rubella-associated granulomas over the past 10 years.
Cases now include children and adults through advanced ages.
Shedding of the virus and risk of infection to others has been studied but so far, the risk — if it exists — is very low. The evidence includes the many patients who have lived with the granulomas for years, even decades, without any known spread to others.
As for ongoing work in this area, Dr. Wanat said that a histopathological case definition for rubella-associated granulomas is being developed, and she and other investigators are actively seeking new cases to better characterize the disease.
So far, optimal treatment is not well defined. A number of strategies have had limited success or are considered impractical for routine use. One example is a stem cell transplant. In a case Dr. Wanat cited, complete resolution of the skin lesions was achieved with a transplant.
“I am not suggesting that those with localized disease in the skin should undergo a transplant, but it does support the role of the immune system and the potential for a reboot to clear the skin,” she said.
Other therapies associated with benefit in at least some patients include tumor necrosis factor (TNF) inhibitors with dapsone and ribavirin. The risk of adverse events for the latter might again limit its use, Dr. Wanat said.
With awareness, the number of granulomas found to be associated with rubella virus is expected to grow. Dr. Wanat speculated that those areas of the country that not yet have documented a case will do so over time. For idiopathic cases of cutaneous granulomas, rubella should be kept in mind, she said.
Characterizing rubella-associated cutaneous granulomas as “a public health concern,” Dr. Wanat urged clinicians to consider this etiology in lesions that match the phenotype, particularly when other more common infectious agents cannot be identified.
Asked for his perspective, Jeffrey P. North, MD, managing director of the UCSF Dermatopathology, and professor of dermatology and pathology at the University of California, San Francisco, agreed that rubella should be considered as a source of granulomas with a suspected infectious etiology when a pathogen cannot be found.
“It is likely much more common than we know as it has only been recently described and testing for it is limited. I suspected there are a lot of undiagnosed patients suffering from this disease,” Dr. North said in an interview.
“One of the important points for clinicians to consider is that while this has been reported mostly in patients with some form of immunodeficiency, there have also been patients reported to have this condition with no immunodeficiency,” he added. Even though the association between rubella and granulomas was made 10 years ago, awareness is only now spreading, which means the frequency with which rubella leads to granulomas remains uncertain.
“I think we will start to get a better idea of how common this is as more people learn about and testing for it expands,” Dr. North said.
Dr. Wanat reports no potential conflicts of interest. Dr. North reports financial relationships with AdviNow and Kiniksa Pharmaceuticals.
SAN DIEGO — Approximately 10 years ago in France, high throughput .
Based on accumulating evidence, the Centers for Disease Control and Prevention (CDC) through collaborations with others also recognized this in pediatric patients with inborn errors of immunity, and it is now appropriate for clinicians to consider this etiology when no other infectious agents can be identified, according to Karolyn A. Wanat, MD, professor of dermatology, Medical College of Wisconsin, Milwaukee, who spoke about rubella as a trigger in granulomatous disease at the meeting. “This is a huge evolving area of interest,” said Dr. Wanat, who has been the first author or coauthor on several published papers, including a review article published earlier this year.
In the earliest cases, including those reported in 2014, the cutaneous granulomas presumed to be causally related to vaccine-derived rubella virus were found only in those with a primary immunodeficiency. This is no longer the case. In a collaboration among US clinics, granulomas that had persisted for years in immunocompetent adults were identified, according to Dr. Wanat, the first author of a report on these findings in four adults in 2022. In addition, it now appears that wild-type rubella virus, like vaccine-derived rubella virus, can be the source of the antigenic response that underlies the development of rubella-associated cutaneous granulomas.
The phenotype of these granulomas is comparable to granulomas associated with other infectious agents. On dermatopathology, these commonly feature a robust granulomatous inflammation with multinucleated giant cells and lymphocytic infiltrate. Necrosis and fibrosis are also common.
“These are the types of granulomas that we would be thinking infection. If tissue cultures are negative, we would probably repeat them,” she said, suggesting that suspicion of an infectious etiology would probably remain high even after multiple negative tests.
Of the cases accruing in the United States and elsewhere, most but not all have been linked to inborn errors of immunity. In a 2020 CDC review, the risk of granulomas caused by compromised immunity, such as defects in T cell function, was estimated to be in the range of 0.6% to 2.5%, Dr. Wanat said.
It is now known that primary immunodeficiency is not a prerequisite, but this should not change the perception that the rubella vaccine, which was introduced in 1979, is effective and safe, according to Dr. Wanat. The vaccine is associated with few serious adverse events and is so effective that rubella was eliminated from the United States in 2004 and from the Americas in 2015.
This makes cases of granuloma associated with wild-type rubella virus surprising, but they appear to be exceedingly rare. Whether caused by vaccine exposure or another source, the mechanism of latent development of cutaneous granulomas is consistent with other infectious sources, and is not well understood.
“Rubella is a sneaky virus that can persist in some immunoprivileged sites indefinitely,” Dr. Wanat said. These sites include the eyes, joints, and placenta.
Many initial cases of rubella-associated granulomas occurred on the arms, presumably where the vaccine was administered, despite long intervals between exposure and lesion growth. This interval is often measured in years.
With more cases, it is now understood that involvement of other organs does occur even if the skin is the most common site of antigenic response in patients with immunodeficiency. The liver and lymph nodes represent other tissues that have been affected. Even lesions in the brain have been seen on autopsy.
Based on the benefit-to-risk ratio of a highly effective and successful vaccine, however, the association with a risk of granulomas “should not raise questions” about the value of the vaccine itself, Dr. Wanat noted.
“The proportion of patients who develop these granulomas is very, very low. Yet, the vaccine provides life-long immunity,” she said.
The discovery of granulomas associated with wild type rubella infection was “shocking” based on the supposition that the rubella virus had been eliminated, but this is just one of the unexpected discoveries as the still-evolving science has traced the story of rubella-associated granulomas over the past 10 years.
Cases now include children and adults through advanced ages.
Shedding of the virus and risk of infection to others has been studied but so far, the risk — if it exists — is very low. The evidence includes the many patients who have lived with the granulomas for years, even decades, without any known spread to others.
As for ongoing work in this area, Dr. Wanat said that a histopathological case definition for rubella-associated granulomas is being developed, and she and other investigators are actively seeking new cases to better characterize the disease.
So far, optimal treatment is not well defined. A number of strategies have had limited success or are considered impractical for routine use. One example is a stem cell transplant. In a case Dr. Wanat cited, complete resolution of the skin lesions was achieved with a transplant.
“I am not suggesting that those with localized disease in the skin should undergo a transplant, but it does support the role of the immune system and the potential for a reboot to clear the skin,” she said.
Other therapies associated with benefit in at least some patients include tumor necrosis factor (TNF) inhibitors with dapsone and ribavirin. The risk of adverse events for the latter might again limit its use, Dr. Wanat said.
With awareness, the number of granulomas found to be associated with rubella virus is expected to grow. Dr. Wanat speculated that those areas of the country that not yet have documented a case will do so over time. For idiopathic cases of cutaneous granulomas, rubella should be kept in mind, she said.
Characterizing rubella-associated cutaneous granulomas as “a public health concern,” Dr. Wanat urged clinicians to consider this etiology in lesions that match the phenotype, particularly when other more common infectious agents cannot be identified.
Asked for his perspective, Jeffrey P. North, MD, managing director of the UCSF Dermatopathology, and professor of dermatology and pathology at the University of California, San Francisco, agreed that rubella should be considered as a source of granulomas with a suspected infectious etiology when a pathogen cannot be found.
“It is likely much more common than we know as it has only been recently described and testing for it is limited. I suspected there are a lot of undiagnosed patients suffering from this disease,” Dr. North said in an interview.
“One of the important points for clinicians to consider is that while this has been reported mostly in patients with some form of immunodeficiency, there have also been patients reported to have this condition with no immunodeficiency,” he added. Even though the association between rubella and granulomas was made 10 years ago, awareness is only now spreading, which means the frequency with which rubella leads to granulomas remains uncertain.
“I think we will start to get a better idea of how common this is as more people learn about and testing for it expands,” Dr. North said.
Dr. Wanat reports no potential conflicts of interest. Dr. North reports financial relationships with AdviNow and Kiniksa Pharmaceuticals.
SAN DIEGO — Approximately 10 years ago in France, high throughput .
Based on accumulating evidence, the Centers for Disease Control and Prevention (CDC) through collaborations with others also recognized this in pediatric patients with inborn errors of immunity, and it is now appropriate for clinicians to consider this etiology when no other infectious agents can be identified, according to Karolyn A. Wanat, MD, professor of dermatology, Medical College of Wisconsin, Milwaukee, who spoke about rubella as a trigger in granulomatous disease at the meeting. “This is a huge evolving area of interest,” said Dr. Wanat, who has been the first author or coauthor on several published papers, including a review article published earlier this year.
In the earliest cases, including those reported in 2014, the cutaneous granulomas presumed to be causally related to vaccine-derived rubella virus were found only in those with a primary immunodeficiency. This is no longer the case. In a collaboration among US clinics, granulomas that had persisted for years in immunocompetent adults were identified, according to Dr. Wanat, the first author of a report on these findings in four adults in 2022. In addition, it now appears that wild-type rubella virus, like vaccine-derived rubella virus, can be the source of the antigenic response that underlies the development of rubella-associated cutaneous granulomas.
The phenotype of these granulomas is comparable to granulomas associated with other infectious agents. On dermatopathology, these commonly feature a robust granulomatous inflammation with multinucleated giant cells and lymphocytic infiltrate. Necrosis and fibrosis are also common.
“These are the types of granulomas that we would be thinking infection. If tissue cultures are negative, we would probably repeat them,” she said, suggesting that suspicion of an infectious etiology would probably remain high even after multiple negative tests.
Of the cases accruing in the United States and elsewhere, most but not all have been linked to inborn errors of immunity. In a 2020 CDC review, the risk of granulomas caused by compromised immunity, such as defects in T cell function, was estimated to be in the range of 0.6% to 2.5%, Dr. Wanat said.
It is now known that primary immunodeficiency is not a prerequisite, but this should not change the perception that the rubella vaccine, which was introduced in 1979, is effective and safe, according to Dr. Wanat. The vaccine is associated with few serious adverse events and is so effective that rubella was eliminated from the United States in 2004 and from the Americas in 2015.
This makes cases of granuloma associated with wild-type rubella virus surprising, but they appear to be exceedingly rare. Whether caused by vaccine exposure or another source, the mechanism of latent development of cutaneous granulomas is consistent with other infectious sources, and is not well understood.
“Rubella is a sneaky virus that can persist in some immunoprivileged sites indefinitely,” Dr. Wanat said. These sites include the eyes, joints, and placenta.
Many initial cases of rubella-associated granulomas occurred on the arms, presumably where the vaccine was administered, despite long intervals between exposure and lesion growth. This interval is often measured in years.
With more cases, it is now understood that involvement of other organs does occur even if the skin is the most common site of antigenic response in patients with immunodeficiency. The liver and lymph nodes represent other tissues that have been affected. Even lesions in the brain have been seen on autopsy.
Based on the benefit-to-risk ratio of a highly effective and successful vaccine, however, the association with a risk of granulomas “should not raise questions” about the value of the vaccine itself, Dr. Wanat noted.
“The proportion of patients who develop these granulomas is very, very low. Yet, the vaccine provides life-long immunity,” she said.
The discovery of granulomas associated with wild type rubella infection was “shocking” based on the supposition that the rubella virus had been eliminated, but this is just one of the unexpected discoveries as the still-evolving science has traced the story of rubella-associated granulomas over the past 10 years.
Cases now include children and adults through advanced ages.
Shedding of the virus and risk of infection to others has been studied but so far, the risk — if it exists — is very low. The evidence includes the many patients who have lived with the granulomas for years, even decades, without any known spread to others.
As for ongoing work in this area, Dr. Wanat said that a histopathological case definition for rubella-associated granulomas is being developed, and she and other investigators are actively seeking new cases to better characterize the disease.
So far, optimal treatment is not well defined. A number of strategies have had limited success or are considered impractical for routine use. One example is a stem cell transplant. In a case Dr. Wanat cited, complete resolution of the skin lesions was achieved with a transplant.
“I am not suggesting that those with localized disease in the skin should undergo a transplant, but it does support the role of the immune system and the potential for a reboot to clear the skin,” she said.
Other therapies associated with benefit in at least some patients include tumor necrosis factor (TNF) inhibitors with dapsone and ribavirin. The risk of adverse events for the latter might again limit its use, Dr. Wanat said.
With awareness, the number of granulomas found to be associated with rubella virus is expected to grow. Dr. Wanat speculated that those areas of the country that not yet have documented a case will do so over time. For idiopathic cases of cutaneous granulomas, rubella should be kept in mind, she said.
Characterizing rubella-associated cutaneous granulomas as “a public health concern,” Dr. Wanat urged clinicians to consider this etiology in lesions that match the phenotype, particularly when other more common infectious agents cannot be identified.
Asked for his perspective, Jeffrey P. North, MD, managing director of the UCSF Dermatopathology, and professor of dermatology and pathology at the University of California, San Francisco, agreed that rubella should be considered as a source of granulomas with a suspected infectious etiology when a pathogen cannot be found.
“It is likely much more common than we know as it has only been recently described and testing for it is limited. I suspected there are a lot of undiagnosed patients suffering from this disease,” Dr. North said in an interview.
“One of the important points for clinicians to consider is that while this has been reported mostly in patients with some form of immunodeficiency, there have also been patients reported to have this condition with no immunodeficiency,” he added. Even though the association between rubella and granulomas was made 10 years ago, awareness is only now spreading, which means the frequency with which rubella leads to granulomas remains uncertain.
“I think we will start to get a better idea of how common this is as more people learn about and testing for it expands,” Dr. North said.
Dr. Wanat reports no potential conflicts of interest. Dr. North reports financial relationships with AdviNow and Kiniksa Pharmaceuticals.
FROM AAD 2024
Gene Therapy for Dystrophic EB: Extension Study Results Reported
and no new safety signals were identified.
The results were presented by Amy S. Paller, MD, during a late-breaking session at the annual meeting of the American Academy of Dermatology.
In May 2023, beremagene geperpavec, marketed as Vyjuvek (formerly known as B-VEC) was approved by the US Food and Drug Administration (FDA) for the treatment of wounds in patients 6 months of age and older with DEB, a rare genetic blistering disorder caused by COL7A1 gene variants. The therapy uses a nonreplicating herpes simplex virus type 1 (HSV-1) vector to deliver the COL7A1 gene directly to skin cells, restoring the COL7 protein fibrils that stabilize skin structure. It is designed to be used repetitively, to heal a single wound, or on more than one wound.
In the pivotal study of patients with DEB, the gene therapy, delivered in a topical gel, was administered once a week for 6 months to one wound and placebo was applied to another wound for each participant. The proportion of wounds treated with beremagene geperpavec that healed was significantly higher than among placebo-treated wounds at 3 and 6 months (68% vs. 23% at 3 months, P = .003) and 65% vs. 26% at 6 months (P = .012), with no serious adverse events related to treatment.
The prospective, open label, uncontrolled extension study included 24 patients from the phase 3 study and 23 treatment-naive patients from five US sites. Their mean age was 16 years (range, 6 months to 46 years).
Of the 47 patients, 29 (62%) were on treatment for more than 1 year (the longest was about 2 years), and the mean duration of treatment was 475 days; 5 patients withdrew from the study for reasons not related to treatment.
Their types of adverse events (AEs) were similar to those seen in the phase 3 study and were consistent with what would be expected in patients with DEB, said Dr. Paller, professor and chair of dermatology, Northwestern University, Chicago. One patient experienced two wound hemorrhages that were possibly related to treatment, but there were no treatment-related AEs, no deaths or treatment discontinuations because of an AE, and no serious AEs thought to be related to treatment.
Wounds that were evaluated in the phase 3 study showed “a high durability of closure with continued treatment,” according to Dr. Paller. There were enough data on 19 of the 24 patients who had been in the phase 3 trial to evaluate wound closure, defined as “complete wound closure based on comparison to the exact wound area selected at baseline” at the beginning of the phase 3 study.
In the extension study, wound closure rates were almost 90% at baseline, 84.2% at 3 months, 61.1% at 6 months, 82.4% at 9 months, and 62.5% at 12 months, which was comparable to the rates observed in the third (86.4%) and sixth (73.7%) months of the phase 3 study, Dr. Paller said.
Patient-reported outcomes indicated that quality of life and satisfaction with treatment were preserved with continued treatment.The extension study was terminated in July 2023, after FDA approval, when patients could be transitioned to the commercially available treatment.Dr. Paller disclosed being an investigator (funds to institution) for multiple pharmaceutical companies, including the manufacturer of beremagene geperpavec, Krystal Biotech, which funded the study.
and no new safety signals were identified.
The results were presented by Amy S. Paller, MD, during a late-breaking session at the annual meeting of the American Academy of Dermatology.
In May 2023, beremagene geperpavec, marketed as Vyjuvek (formerly known as B-VEC) was approved by the US Food and Drug Administration (FDA) for the treatment of wounds in patients 6 months of age and older with DEB, a rare genetic blistering disorder caused by COL7A1 gene variants. The therapy uses a nonreplicating herpes simplex virus type 1 (HSV-1) vector to deliver the COL7A1 gene directly to skin cells, restoring the COL7 protein fibrils that stabilize skin structure. It is designed to be used repetitively, to heal a single wound, or on more than one wound.
In the pivotal study of patients with DEB, the gene therapy, delivered in a topical gel, was administered once a week for 6 months to one wound and placebo was applied to another wound for each participant. The proportion of wounds treated with beremagene geperpavec that healed was significantly higher than among placebo-treated wounds at 3 and 6 months (68% vs. 23% at 3 months, P = .003) and 65% vs. 26% at 6 months (P = .012), with no serious adverse events related to treatment.
The prospective, open label, uncontrolled extension study included 24 patients from the phase 3 study and 23 treatment-naive patients from five US sites. Their mean age was 16 years (range, 6 months to 46 years).
Of the 47 patients, 29 (62%) were on treatment for more than 1 year (the longest was about 2 years), and the mean duration of treatment was 475 days; 5 patients withdrew from the study for reasons not related to treatment.
Their types of adverse events (AEs) were similar to those seen in the phase 3 study and were consistent with what would be expected in patients with DEB, said Dr. Paller, professor and chair of dermatology, Northwestern University, Chicago. One patient experienced two wound hemorrhages that were possibly related to treatment, but there were no treatment-related AEs, no deaths or treatment discontinuations because of an AE, and no serious AEs thought to be related to treatment.
Wounds that were evaluated in the phase 3 study showed “a high durability of closure with continued treatment,” according to Dr. Paller. There were enough data on 19 of the 24 patients who had been in the phase 3 trial to evaluate wound closure, defined as “complete wound closure based on comparison to the exact wound area selected at baseline” at the beginning of the phase 3 study.
In the extension study, wound closure rates were almost 90% at baseline, 84.2% at 3 months, 61.1% at 6 months, 82.4% at 9 months, and 62.5% at 12 months, which was comparable to the rates observed in the third (86.4%) and sixth (73.7%) months of the phase 3 study, Dr. Paller said.
Patient-reported outcomes indicated that quality of life and satisfaction with treatment were preserved with continued treatment.The extension study was terminated in July 2023, after FDA approval, when patients could be transitioned to the commercially available treatment.Dr. Paller disclosed being an investigator (funds to institution) for multiple pharmaceutical companies, including the manufacturer of beremagene geperpavec, Krystal Biotech, which funded the study.
and no new safety signals were identified.
The results were presented by Amy S. Paller, MD, during a late-breaking session at the annual meeting of the American Academy of Dermatology.
In May 2023, beremagene geperpavec, marketed as Vyjuvek (formerly known as B-VEC) was approved by the US Food and Drug Administration (FDA) for the treatment of wounds in patients 6 months of age and older with DEB, a rare genetic blistering disorder caused by COL7A1 gene variants. The therapy uses a nonreplicating herpes simplex virus type 1 (HSV-1) vector to deliver the COL7A1 gene directly to skin cells, restoring the COL7 protein fibrils that stabilize skin structure. It is designed to be used repetitively, to heal a single wound, or on more than one wound.
In the pivotal study of patients with DEB, the gene therapy, delivered in a topical gel, was administered once a week for 6 months to one wound and placebo was applied to another wound for each participant. The proportion of wounds treated with beremagene geperpavec that healed was significantly higher than among placebo-treated wounds at 3 and 6 months (68% vs. 23% at 3 months, P = .003) and 65% vs. 26% at 6 months (P = .012), with no serious adverse events related to treatment.
The prospective, open label, uncontrolled extension study included 24 patients from the phase 3 study and 23 treatment-naive patients from five US sites. Their mean age was 16 years (range, 6 months to 46 years).
Of the 47 patients, 29 (62%) were on treatment for more than 1 year (the longest was about 2 years), and the mean duration of treatment was 475 days; 5 patients withdrew from the study for reasons not related to treatment.
Their types of adverse events (AEs) were similar to those seen in the phase 3 study and were consistent with what would be expected in patients with DEB, said Dr. Paller, professor and chair of dermatology, Northwestern University, Chicago. One patient experienced two wound hemorrhages that were possibly related to treatment, but there were no treatment-related AEs, no deaths or treatment discontinuations because of an AE, and no serious AEs thought to be related to treatment.
Wounds that were evaluated in the phase 3 study showed “a high durability of closure with continued treatment,” according to Dr. Paller. There were enough data on 19 of the 24 patients who had been in the phase 3 trial to evaluate wound closure, defined as “complete wound closure based on comparison to the exact wound area selected at baseline” at the beginning of the phase 3 study.
In the extension study, wound closure rates were almost 90% at baseline, 84.2% at 3 months, 61.1% at 6 months, 82.4% at 9 months, and 62.5% at 12 months, which was comparable to the rates observed in the third (86.4%) and sixth (73.7%) months of the phase 3 study, Dr. Paller said.
Patient-reported outcomes indicated that quality of life and satisfaction with treatment were preserved with continued treatment.The extension study was terminated in July 2023, after FDA approval, when patients could be transitioned to the commercially available treatment.Dr. Paller disclosed being an investigator (funds to institution) for multiple pharmaceutical companies, including the manufacturer of beremagene geperpavec, Krystal Biotech, which funded the study.
FROM AAD 2024
FDA Requests More Information for RDEB Rx Under Review
The Food and Drug Administration (RDEB), requesting more information from the manufacturer.
Pz-cel, which comprises autologous, COL7A1 gene–corrected epidermal sheets, is being evaluated for its ability to enable normal type VII collagen expression in a patient’s skin cells and to facilitate wound healing and pain reduction in wounds in patients with RDEB after a one-time application procedure. The cause of RDEB is a defect in the COL7A1 gene that “results in the inability to produce type VII collagen,” a press release from the manufacturer noted.
On April 22, 2024, the manufacturer Abeona Therapeutics announced that following a meeting with the FDA in March and in a subsequent request for information, the agency requires additional information to satisfy certain Chemistry Manufacturing and Controls requirements before the BLA for pz-cel can be approved. According to a press release from the company, the information pertains to validation requirements for certain manufacturing and release testing methods, including some that were observed during the FDA’s pre-licensing inspection.
The complete response letter did not identify any issues related to the clinical efficacy or safety data in the BLA, and the FDA did not request any new clinical trials or clinical data to support approval, according to the company.
The company anticipates completing the BLA resubmission in the third quarter of 2024. The application is supported by clinical efficacy and safety data from the pivotal phase 3 VIITAL study and a phase 1/2a study in patients with RDEB.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration (RDEB), requesting more information from the manufacturer.
Pz-cel, which comprises autologous, COL7A1 gene–corrected epidermal sheets, is being evaluated for its ability to enable normal type VII collagen expression in a patient’s skin cells and to facilitate wound healing and pain reduction in wounds in patients with RDEB after a one-time application procedure. The cause of RDEB is a defect in the COL7A1 gene that “results in the inability to produce type VII collagen,” a press release from the manufacturer noted.
On April 22, 2024, the manufacturer Abeona Therapeutics announced that following a meeting with the FDA in March and in a subsequent request for information, the agency requires additional information to satisfy certain Chemistry Manufacturing and Controls requirements before the BLA for pz-cel can be approved. According to a press release from the company, the information pertains to validation requirements for certain manufacturing and release testing methods, including some that were observed during the FDA’s pre-licensing inspection.
The complete response letter did not identify any issues related to the clinical efficacy or safety data in the BLA, and the FDA did not request any new clinical trials or clinical data to support approval, according to the company.
The company anticipates completing the BLA resubmission in the third quarter of 2024. The application is supported by clinical efficacy and safety data from the pivotal phase 3 VIITAL study and a phase 1/2a study in patients with RDEB.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration (RDEB), requesting more information from the manufacturer.
Pz-cel, which comprises autologous, COL7A1 gene–corrected epidermal sheets, is being evaluated for its ability to enable normal type VII collagen expression in a patient’s skin cells and to facilitate wound healing and pain reduction in wounds in patients with RDEB after a one-time application procedure. The cause of RDEB is a defect in the COL7A1 gene that “results in the inability to produce type VII collagen,” a press release from the manufacturer noted.
On April 22, 2024, the manufacturer Abeona Therapeutics announced that following a meeting with the FDA in March and in a subsequent request for information, the agency requires additional information to satisfy certain Chemistry Manufacturing and Controls requirements before the BLA for pz-cel can be approved. According to a press release from the company, the information pertains to validation requirements for certain manufacturing and release testing methods, including some that were observed during the FDA’s pre-licensing inspection.
The complete response letter did not identify any issues related to the clinical efficacy or safety data in the BLA, and the FDA did not request any new clinical trials or clinical data to support approval, according to the company.
The company anticipates completing the BLA resubmission in the third quarter of 2024. The application is supported by clinical efficacy and safety data from the pivotal phase 3 VIITAL study and a phase 1/2a study in patients with RDEB.
A version of this article first appeared on Medscape.com.