User login
PCOS ups risk of heart complications during delivery period
Pregnant women with polycystic ovary syndrome (PCOS) appear to be at significantly increased risk of experiencing cardiac complications while hospitalized during and after delivery.
An estimated 5 million women of childbearing age in the United States have PCOS, a hormone disorder linked to infertility. PCOS is also known to contribute to the development of cardiometabolic abnormalities like high cholesterol and high blood pressure, which are associated with acute cardiovascular complications during delivery.
But a study, published online in the Journal of the American Heart Association, found that even after accounting for pre-eclampsia, age, comorbidities, and race, PCOS was linked to a 76% increased risk for heart failure, a 79% higher risk of a weakened heart, and an 82% increased risk of having blood clots in the hours and days around giving birth in hospital settings, compared with women without PCOS.
“Perhaps women need a closer follow-up during their pregnancy,” said Erin Michos, MD, MHS, associate director of preventive cardiology at Johns Hopkins Medicine, Baltimore, and a co-author of the study. “They’re counseled about the difficulties of getting pregnant, but what about when they get pregnant?”
Hospitalizations of women with PCOS were also associated with longer stays (3 vs. 2 days) and higher costs ($4,901 vs. $3616; P < .01), compared with women without PCOS.
Over the 17-year analysis period, the number of women with PCOS rose from 569 per 100,000 deliveries to 15,349 per 100,000 deliveries. The researchers attributed the increase in part to greater awareness and diagnosis of the disorder. Dr. Michos and her colleagues used the National Inpatient Sample, managed by the Agency for Healthcare Research and Quality, to pull claims data for women who gave birth in hospitals between 2002 and 2019.
Solutions?
Dr. Michos said there may be more prevention work from og.gyns. to both educate patients about their heart risks during the delivery process and also to refer them to relevant cardiac specialists.
“These women may seek out a gynecologist because of the symptoms, perhaps irregular menses, but along with that should come counseling of the long-term cardiovascular complication,” Dr. Michos said. “And after a pregnancy there should be a good handoff to a primary care provider, so they get a cardiovascular assessment.”
Lifestyle management before, during, and after pregnancy can help prevent the onset of the long-term consequences of cardiac complications during delivery, according to Valerie Baker, MD, director of the division of reproductive endocrinology and infertility at Hopkins Medicine, and her colleagues in a viewpoint published in the journal Fertility and Sterility.
“Once women with PCOS are identified by screening to be at higher risk for [cardiovascular disease], the foundational approach should be lifestyle management followed by statin therapy,” Dr. Baker’s group wrote. “These interventions should include dietary management and physical activity, especially for those who are prediabetic.”
The current study came on the heels of a June 14 meta-analysis by Dr. Michos’ group that found that women with PCOS may be twice as likely as those without PCOS to have coronary artery calcification, a precursor to atherosclerosis and a sign of the early onset of cardiovascular disease.
“We shouldn’t assume that all women of reproductive age are low risk,” Dr. Michos said. “This is the window of time that we can reshape the trajectory early in life.”
The study was supported by the Amato Fund for Women’s Cardiovascular Health research at Johns Hopkins University and through grant support from the American Heart Association (940166). Dr. Michos reported advisory board participation for AstraZeneca, Amarin, Novartis, Novo Nordisk, Bayer, Boehringer Ingelheim, Esperion, and Pfizer. Study coauthor Michael Honigberg, MD, reported consulting fees from CRISPR Therapeutics, unrelated to the present work. The remaining authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pregnant women with polycystic ovary syndrome (PCOS) appear to be at significantly increased risk of experiencing cardiac complications while hospitalized during and after delivery.
An estimated 5 million women of childbearing age in the United States have PCOS, a hormone disorder linked to infertility. PCOS is also known to contribute to the development of cardiometabolic abnormalities like high cholesterol and high blood pressure, which are associated with acute cardiovascular complications during delivery.
But a study, published online in the Journal of the American Heart Association, found that even after accounting for pre-eclampsia, age, comorbidities, and race, PCOS was linked to a 76% increased risk for heart failure, a 79% higher risk of a weakened heart, and an 82% increased risk of having blood clots in the hours and days around giving birth in hospital settings, compared with women without PCOS.
“Perhaps women need a closer follow-up during their pregnancy,” said Erin Michos, MD, MHS, associate director of preventive cardiology at Johns Hopkins Medicine, Baltimore, and a co-author of the study. “They’re counseled about the difficulties of getting pregnant, but what about when they get pregnant?”
Hospitalizations of women with PCOS were also associated with longer stays (3 vs. 2 days) and higher costs ($4,901 vs. $3616; P < .01), compared with women without PCOS.
Over the 17-year analysis period, the number of women with PCOS rose from 569 per 100,000 deliveries to 15,349 per 100,000 deliveries. The researchers attributed the increase in part to greater awareness and diagnosis of the disorder. Dr. Michos and her colleagues used the National Inpatient Sample, managed by the Agency for Healthcare Research and Quality, to pull claims data for women who gave birth in hospitals between 2002 and 2019.
Solutions?
Dr. Michos said there may be more prevention work from og.gyns. to both educate patients about their heart risks during the delivery process and also to refer them to relevant cardiac specialists.
“These women may seek out a gynecologist because of the symptoms, perhaps irregular menses, but along with that should come counseling of the long-term cardiovascular complication,” Dr. Michos said. “And after a pregnancy there should be a good handoff to a primary care provider, so they get a cardiovascular assessment.”
Lifestyle management before, during, and after pregnancy can help prevent the onset of the long-term consequences of cardiac complications during delivery, according to Valerie Baker, MD, director of the division of reproductive endocrinology and infertility at Hopkins Medicine, and her colleagues in a viewpoint published in the journal Fertility and Sterility.
“Once women with PCOS are identified by screening to be at higher risk for [cardiovascular disease], the foundational approach should be lifestyle management followed by statin therapy,” Dr. Baker’s group wrote. “These interventions should include dietary management and physical activity, especially for those who are prediabetic.”
The current study came on the heels of a June 14 meta-analysis by Dr. Michos’ group that found that women with PCOS may be twice as likely as those without PCOS to have coronary artery calcification, a precursor to atherosclerosis and a sign of the early onset of cardiovascular disease.
“We shouldn’t assume that all women of reproductive age are low risk,” Dr. Michos said. “This is the window of time that we can reshape the trajectory early in life.”
The study was supported by the Amato Fund for Women’s Cardiovascular Health research at Johns Hopkins University and through grant support from the American Heart Association (940166). Dr. Michos reported advisory board participation for AstraZeneca, Amarin, Novartis, Novo Nordisk, Bayer, Boehringer Ingelheim, Esperion, and Pfizer. Study coauthor Michael Honigberg, MD, reported consulting fees from CRISPR Therapeutics, unrelated to the present work. The remaining authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pregnant women with polycystic ovary syndrome (PCOS) appear to be at significantly increased risk of experiencing cardiac complications while hospitalized during and after delivery.
An estimated 5 million women of childbearing age in the United States have PCOS, a hormone disorder linked to infertility. PCOS is also known to contribute to the development of cardiometabolic abnormalities like high cholesterol and high blood pressure, which are associated with acute cardiovascular complications during delivery.
But a study, published online in the Journal of the American Heart Association, found that even after accounting for pre-eclampsia, age, comorbidities, and race, PCOS was linked to a 76% increased risk for heart failure, a 79% higher risk of a weakened heart, and an 82% increased risk of having blood clots in the hours and days around giving birth in hospital settings, compared with women without PCOS.
“Perhaps women need a closer follow-up during their pregnancy,” said Erin Michos, MD, MHS, associate director of preventive cardiology at Johns Hopkins Medicine, Baltimore, and a co-author of the study. “They’re counseled about the difficulties of getting pregnant, but what about when they get pregnant?”
Hospitalizations of women with PCOS were also associated with longer stays (3 vs. 2 days) and higher costs ($4,901 vs. $3616; P < .01), compared with women without PCOS.
Over the 17-year analysis period, the number of women with PCOS rose from 569 per 100,000 deliveries to 15,349 per 100,000 deliveries. The researchers attributed the increase in part to greater awareness and diagnosis of the disorder. Dr. Michos and her colleagues used the National Inpatient Sample, managed by the Agency for Healthcare Research and Quality, to pull claims data for women who gave birth in hospitals between 2002 and 2019.
Solutions?
Dr. Michos said there may be more prevention work from og.gyns. to both educate patients about their heart risks during the delivery process and also to refer them to relevant cardiac specialists.
“These women may seek out a gynecologist because of the symptoms, perhaps irregular menses, but along with that should come counseling of the long-term cardiovascular complication,” Dr. Michos said. “And after a pregnancy there should be a good handoff to a primary care provider, so they get a cardiovascular assessment.”
Lifestyle management before, during, and after pregnancy can help prevent the onset of the long-term consequences of cardiac complications during delivery, according to Valerie Baker, MD, director of the division of reproductive endocrinology and infertility at Hopkins Medicine, and her colleagues in a viewpoint published in the journal Fertility and Sterility.
“Once women with PCOS are identified by screening to be at higher risk for [cardiovascular disease], the foundational approach should be lifestyle management followed by statin therapy,” Dr. Baker’s group wrote. “These interventions should include dietary management and physical activity, especially for those who are prediabetic.”
The current study came on the heels of a June 14 meta-analysis by Dr. Michos’ group that found that women with PCOS may be twice as likely as those without PCOS to have coronary artery calcification, a precursor to atherosclerosis and a sign of the early onset of cardiovascular disease.
“We shouldn’t assume that all women of reproductive age are low risk,” Dr. Michos said. “This is the window of time that we can reshape the trajectory early in life.”
The study was supported by the Amato Fund for Women’s Cardiovascular Health research at Johns Hopkins University and through grant support from the American Heart Association (940166). Dr. Michos reported advisory board participation for AstraZeneca, Amarin, Novartis, Novo Nordisk, Bayer, Boehringer Ingelheim, Esperion, and Pfizer. Study coauthor Michael Honigberg, MD, reported consulting fees from CRISPR Therapeutics, unrelated to the present work. The remaining authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New European guidelines ‘drastically’ reduce statin eligibility
New risk thresholds used to guide statin therapy for primary prevention of atherosclerotic cardiovascular disease in the latest European guidelines dramatically reduce eligibility for statin use in low-risk countries, a new study has found.
The authors reported that and essentially eliminate a statin indication in women.
“We have guidelines in place to try to prevent cardiovascular disease but the risk threshold in this new guideline means that almost nobody qualifies for treatment in many countries, which will lead to almost no prevention of future cardiovascular disease in those countries,” lead author Martin Bødtker Mortensen, MD, PhD, Aarhus (Denmark) University Hospital, commented in an interview.
“We argue that the risk thresholds need to be lowered to get the statin eligibility in European countries to be in line with thresholds in the U.K. and U.S., which are based on randomized, controlled trials,” he added.
The study was published online in JAMA Cardiology.
An accompanying editorial describes the results of the study as “alarming,” and, if confirmed, said the guidelines should be revisited to “prevent a step backwards in the use of statins in primary prevention.”
For the study, Dr. Mortensen and colleagues set out to compare the clinical performance of the new European prevention guidelines with American College of Cardiology/American Heart Association, United Kingdom–National Institute for Health and Care Excellence, and the 2019 European guidelines in a contemporary European cohort of 66,909 apparently healthy individuals from the Copenhagen General Population Study.
During the 9-year follow-up, a range of 2,962-4,277 nonfatal and fatal cardiovascular events was observed, as defined by the models in the various guidelines.
Results showed that although the new 2021 European guidelines introduced a new and improved risk model, known as SCORE2, the updated age-specific recommendations dramatically reduced eligibility for a class I recommendation for statin therapy to only 4% of individuals, aged 40-69 years, and less than 1% of women.
This is in sharp contrast to the previous 2019 European guidelines as well as current UK-NICE and US-ACC/AHA guidelines that provide class I/strong recommendations to 20%, 26%, and 34% of individuals, respectively, with better clinical performance in both men and women, the authors report.
The researchers also reported other analyses in which the sensitivity of the new European guidelines was improved considerably by lowering the treatment thresholds.
Dr. Mortensen explained to this news organization that the original SCORE risk model used in ESC guidelines was problematic as it only predicts the 10-year risk of fatal atherosclerotic cardiovascular events, whereas those from the United States and United Kingdom used both fatal and nonfatal cardiovascular events.
“Now the ESC has updated its model and the new model is much better in that it predicts both fatal and nonfatal events, and the predicted risk correlates well with the actual risk. So that’s a big step forward. However, the new thresholds for statin treatment are far too high for low-risk European countries because very few individuals will now qualify for statin therapy,” he said.
“The problem is that, if we use these guidelines, the vast majority of those individuals who will develop cardiovascular disease within 10 years will not be assigned statin therapy that can reduce this risk. There will be lots of individuals who are at high risk of cardiovascular disease, but these guidelines will not identify them as needing to take a statin,” Dr. Mortensen commented.
“If we use the U.K. or U.S. guidelines, far more people in these low-risk European countries would be eligible for statin therapy and we would prevent far more events than if we use the new ESC guidelines,” he added.
Dr. Mortensen explained that the problem arises from having four different risk score models in Europe for areas at different risk, but they all use the same risk thresholds for statin treatment.
“In general, Eastern European countries have higher risk than Western European countries, so these guidelines may work quite well in Eastern European countries but in low-risk Western European countries, where the low-risk score model is used, very few people will qualify for statin therapy,” he said.
While Dr. Mortensen is not against the idea of different risk models in areas that have different risks, he says this needs to be accompanied by different risk thresholds in the different risk areas.
Asked whether there is an argument that most individuals in low-risk countries may not need to take a statin, Dr. Mortensen countered: “One of the reasons the risk is low in many of these European countries is the high use of preventative medication. So, if a threshold that is too high is used most people will not take a statin anymore and the risk in these countries will increase again.”
Authors of the accompanying editorial, Ann Marie Navar, MD, PhD, University of Texas Southwestern Medical Center, Dallas; Gregg C. Fonarow, MD, University of California, Los Angeles; and Michael J. Pencina, PhD, Duke University Medical Center, Durham, N.C., agreed with Dr. Mortensen that the problems appear to arise from use of a risk score that is highly influenced by regional cardiovascular burden.
They point out that under the current guidelines, a 55-year-old woman (smoker; systolic blood pressure, 130 mm Hg; non–HDL cholesterol, 4.0 mmol/L) would have a 10-year predicted risk of having a cardiovascular event of 5% in Denmark but a predicted risk of 18% in Romania.
“While there may be regional differences in environmental risk factors, location alone should not cause a fourfold difference in an individual’s predicted cardiovascular risk,” they wrote.
The editorialists also elaborated on Dr. Mortensen’s point that the new guideline creates a system that eventually becomes a victim of its own success.
“As countries are successful in implementing statin therapy to lower CVD, CVD rates drop, and progressively fewer individuals are then eligible for the very therapy that contributed to the decline in CVD in the first place,” they noted.
The editorialists called for the analysis to be replicated in other low-risk countries and extended to higher-risk regions, with a focus on potential overtreatment of men and older adults.
“If confirmed, the present findings should be a catalyst for the ESC to revisit or augment their current guidelines to prevent a step backward in the use of statins in primary prevention,” they concluded.
This news organization asked the ESC for a response to the findings, but did not comment by press time.
This work was supported by the Lundbeck Foundation, Herlev and Gentofte Hospital, Copenhagen University Hospital, the Copenhagen County Foundation, and Aarhus University, Denmark. Dr. Mortensen reported no disclosures.
A version of this article first appeared on Medscape.com.
New risk thresholds used to guide statin therapy for primary prevention of atherosclerotic cardiovascular disease in the latest European guidelines dramatically reduce eligibility for statin use in low-risk countries, a new study has found.
The authors reported that and essentially eliminate a statin indication in women.
“We have guidelines in place to try to prevent cardiovascular disease but the risk threshold in this new guideline means that almost nobody qualifies for treatment in many countries, which will lead to almost no prevention of future cardiovascular disease in those countries,” lead author Martin Bødtker Mortensen, MD, PhD, Aarhus (Denmark) University Hospital, commented in an interview.
“We argue that the risk thresholds need to be lowered to get the statin eligibility in European countries to be in line with thresholds in the U.K. and U.S., which are based on randomized, controlled trials,” he added.
The study was published online in JAMA Cardiology.
An accompanying editorial describes the results of the study as “alarming,” and, if confirmed, said the guidelines should be revisited to “prevent a step backwards in the use of statins in primary prevention.”
For the study, Dr. Mortensen and colleagues set out to compare the clinical performance of the new European prevention guidelines with American College of Cardiology/American Heart Association, United Kingdom–National Institute for Health and Care Excellence, and the 2019 European guidelines in a contemporary European cohort of 66,909 apparently healthy individuals from the Copenhagen General Population Study.
During the 9-year follow-up, a range of 2,962-4,277 nonfatal and fatal cardiovascular events was observed, as defined by the models in the various guidelines.
Results showed that although the new 2021 European guidelines introduced a new and improved risk model, known as SCORE2, the updated age-specific recommendations dramatically reduced eligibility for a class I recommendation for statin therapy to only 4% of individuals, aged 40-69 years, and less than 1% of women.
This is in sharp contrast to the previous 2019 European guidelines as well as current UK-NICE and US-ACC/AHA guidelines that provide class I/strong recommendations to 20%, 26%, and 34% of individuals, respectively, with better clinical performance in both men and women, the authors report.
The researchers also reported other analyses in which the sensitivity of the new European guidelines was improved considerably by lowering the treatment thresholds.
Dr. Mortensen explained to this news organization that the original SCORE risk model used in ESC guidelines was problematic as it only predicts the 10-year risk of fatal atherosclerotic cardiovascular events, whereas those from the United States and United Kingdom used both fatal and nonfatal cardiovascular events.
“Now the ESC has updated its model and the new model is much better in that it predicts both fatal and nonfatal events, and the predicted risk correlates well with the actual risk. So that’s a big step forward. However, the new thresholds for statin treatment are far too high for low-risk European countries because very few individuals will now qualify for statin therapy,” he said.
“The problem is that, if we use these guidelines, the vast majority of those individuals who will develop cardiovascular disease within 10 years will not be assigned statin therapy that can reduce this risk. There will be lots of individuals who are at high risk of cardiovascular disease, but these guidelines will not identify them as needing to take a statin,” Dr. Mortensen commented.
“If we use the U.K. or U.S. guidelines, far more people in these low-risk European countries would be eligible for statin therapy and we would prevent far more events than if we use the new ESC guidelines,” he added.
Dr. Mortensen explained that the problem arises from having four different risk score models in Europe for areas at different risk, but they all use the same risk thresholds for statin treatment.
“In general, Eastern European countries have higher risk than Western European countries, so these guidelines may work quite well in Eastern European countries but in low-risk Western European countries, where the low-risk score model is used, very few people will qualify for statin therapy,” he said.
While Dr. Mortensen is not against the idea of different risk models in areas that have different risks, he says this needs to be accompanied by different risk thresholds in the different risk areas.
Asked whether there is an argument that most individuals in low-risk countries may not need to take a statin, Dr. Mortensen countered: “One of the reasons the risk is low in many of these European countries is the high use of preventative medication. So, if a threshold that is too high is used most people will not take a statin anymore and the risk in these countries will increase again.”
Authors of the accompanying editorial, Ann Marie Navar, MD, PhD, University of Texas Southwestern Medical Center, Dallas; Gregg C. Fonarow, MD, University of California, Los Angeles; and Michael J. Pencina, PhD, Duke University Medical Center, Durham, N.C., agreed with Dr. Mortensen that the problems appear to arise from use of a risk score that is highly influenced by regional cardiovascular burden.
They point out that under the current guidelines, a 55-year-old woman (smoker; systolic blood pressure, 130 mm Hg; non–HDL cholesterol, 4.0 mmol/L) would have a 10-year predicted risk of having a cardiovascular event of 5% in Denmark but a predicted risk of 18% in Romania.
“While there may be regional differences in environmental risk factors, location alone should not cause a fourfold difference in an individual’s predicted cardiovascular risk,” they wrote.
The editorialists also elaborated on Dr. Mortensen’s point that the new guideline creates a system that eventually becomes a victim of its own success.
“As countries are successful in implementing statin therapy to lower CVD, CVD rates drop, and progressively fewer individuals are then eligible for the very therapy that contributed to the decline in CVD in the first place,” they noted.
The editorialists called for the analysis to be replicated in other low-risk countries and extended to higher-risk regions, with a focus on potential overtreatment of men and older adults.
“If confirmed, the present findings should be a catalyst for the ESC to revisit or augment their current guidelines to prevent a step backward in the use of statins in primary prevention,” they concluded.
This news organization asked the ESC for a response to the findings, but did not comment by press time.
This work was supported by the Lundbeck Foundation, Herlev and Gentofte Hospital, Copenhagen University Hospital, the Copenhagen County Foundation, and Aarhus University, Denmark. Dr. Mortensen reported no disclosures.
A version of this article first appeared on Medscape.com.
New risk thresholds used to guide statin therapy for primary prevention of atherosclerotic cardiovascular disease in the latest European guidelines dramatically reduce eligibility for statin use in low-risk countries, a new study has found.
The authors reported that and essentially eliminate a statin indication in women.
“We have guidelines in place to try to prevent cardiovascular disease but the risk threshold in this new guideline means that almost nobody qualifies for treatment in many countries, which will lead to almost no prevention of future cardiovascular disease in those countries,” lead author Martin Bødtker Mortensen, MD, PhD, Aarhus (Denmark) University Hospital, commented in an interview.
“We argue that the risk thresholds need to be lowered to get the statin eligibility in European countries to be in line with thresholds in the U.K. and U.S., which are based on randomized, controlled trials,” he added.
The study was published online in JAMA Cardiology.
An accompanying editorial describes the results of the study as “alarming,” and, if confirmed, said the guidelines should be revisited to “prevent a step backwards in the use of statins in primary prevention.”
For the study, Dr. Mortensen and colleagues set out to compare the clinical performance of the new European prevention guidelines with American College of Cardiology/American Heart Association, United Kingdom–National Institute for Health and Care Excellence, and the 2019 European guidelines in a contemporary European cohort of 66,909 apparently healthy individuals from the Copenhagen General Population Study.
During the 9-year follow-up, a range of 2,962-4,277 nonfatal and fatal cardiovascular events was observed, as defined by the models in the various guidelines.
Results showed that although the new 2021 European guidelines introduced a new and improved risk model, known as SCORE2, the updated age-specific recommendations dramatically reduced eligibility for a class I recommendation for statin therapy to only 4% of individuals, aged 40-69 years, and less than 1% of women.
This is in sharp contrast to the previous 2019 European guidelines as well as current UK-NICE and US-ACC/AHA guidelines that provide class I/strong recommendations to 20%, 26%, and 34% of individuals, respectively, with better clinical performance in both men and women, the authors report.
The researchers also reported other analyses in which the sensitivity of the new European guidelines was improved considerably by lowering the treatment thresholds.
Dr. Mortensen explained to this news organization that the original SCORE risk model used in ESC guidelines was problematic as it only predicts the 10-year risk of fatal atherosclerotic cardiovascular events, whereas those from the United States and United Kingdom used both fatal and nonfatal cardiovascular events.
“Now the ESC has updated its model and the new model is much better in that it predicts both fatal and nonfatal events, and the predicted risk correlates well with the actual risk. So that’s a big step forward. However, the new thresholds for statin treatment are far too high for low-risk European countries because very few individuals will now qualify for statin therapy,” he said.
“The problem is that, if we use these guidelines, the vast majority of those individuals who will develop cardiovascular disease within 10 years will not be assigned statin therapy that can reduce this risk. There will be lots of individuals who are at high risk of cardiovascular disease, but these guidelines will not identify them as needing to take a statin,” Dr. Mortensen commented.
“If we use the U.K. or U.S. guidelines, far more people in these low-risk European countries would be eligible for statin therapy and we would prevent far more events than if we use the new ESC guidelines,” he added.
Dr. Mortensen explained that the problem arises from having four different risk score models in Europe for areas at different risk, but they all use the same risk thresholds for statin treatment.
“In general, Eastern European countries have higher risk than Western European countries, so these guidelines may work quite well in Eastern European countries but in low-risk Western European countries, where the low-risk score model is used, very few people will qualify for statin therapy,” he said.
While Dr. Mortensen is not against the idea of different risk models in areas that have different risks, he says this needs to be accompanied by different risk thresholds in the different risk areas.
Asked whether there is an argument that most individuals in low-risk countries may not need to take a statin, Dr. Mortensen countered: “One of the reasons the risk is low in many of these European countries is the high use of preventative medication. So, if a threshold that is too high is used most people will not take a statin anymore and the risk in these countries will increase again.”
Authors of the accompanying editorial, Ann Marie Navar, MD, PhD, University of Texas Southwestern Medical Center, Dallas; Gregg C. Fonarow, MD, University of California, Los Angeles; and Michael J. Pencina, PhD, Duke University Medical Center, Durham, N.C., agreed with Dr. Mortensen that the problems appear to arise from use of a risk score that is highly influenced by regional cardiovascular burden.
They point out that under the current guidelines, a 55-year-old woman (smoker; systolic blood pressure, 130 mm Hg; non–HDL cholesterol, 4.0 mmol/L) would have a 10-year predicted risk of having a cardiovascular event of 5% in Denmark but a predicted risk of 18% in Romania.
“While there may be regional differences in environmental risk factors, location alone should not cause a fourfold difference in an individual’s predicted cardiovascular risk,” they wrote.
The editorialists also elaborated on Dr. Mortensen’s point that the new guideline creates a system that eventually becomes a victim of its own success.
“As countries are successful in implementing statin therapy to lower CVD, CVD rates drop, and progressively fewer individuals are then eligible for the very therapy that contributed to the decline in CVD in the first place,” they noted.
The editorialists called for the analysis to be replicated in other low-risk countries and extended to higher-risk regions, with a focus on potential overtreatment of men and older adults.
“If confirmed, the present findings should be a catalyst for the ESC to revisit or augment their current guidelines to prevent a step backward in the use of statins in primary prevention,” they concluded.
This news organization asked the ESC for a response to the findings, but did not comment by press time.
This work was supported by the Lundbeck Foundation, Herlev and Gentofte Hospital, Copenhagen University Hospital, the Copenhagen County Foundation, and Aarhus University, Denmark. Dr. Mortensen reported no disclosures.
A version of this article first appeared on Medscape.com.
FROM JAMA CARDIOLOGY
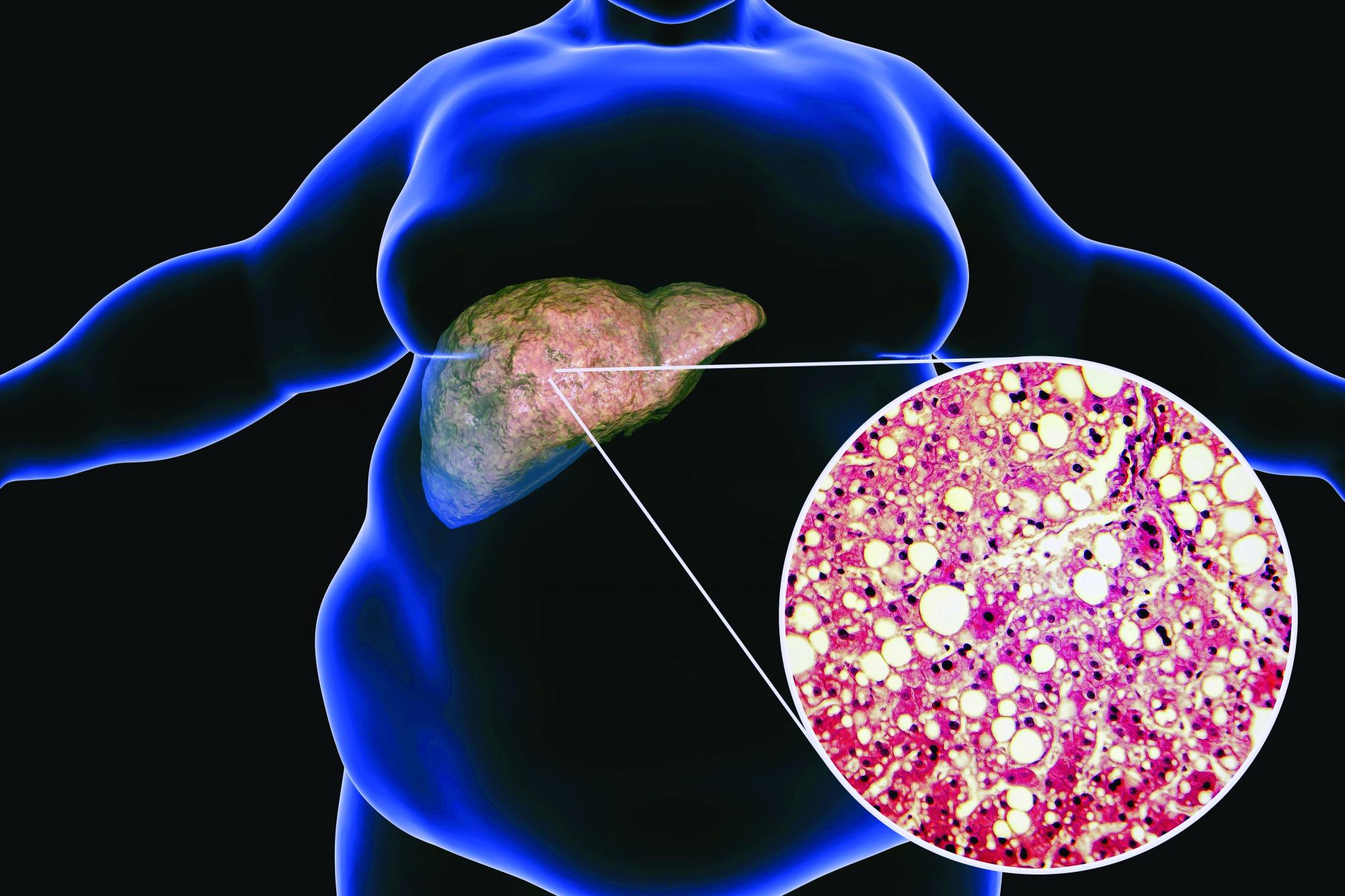
‘Not their fault:’ Obesity warrants long-term management
This transcript has been edited for clarity.
It’s important to remember and to think about the first time when patients with obesity come to see us: What have they faced? What have been their struggles? What shame and blame and bias have they faced?
One of the first things that I do when a patient comes to see me is invite them to share their weight journey with me. I ask them to tell me about their struggles, about what’s worked and what hasn’t worked, what they would like, and what their health goals are.
As they share their stories, I look for the opportunity to share with them that obesity is not their fault, but that it’s biology driving their body to carry extra weight and their body is super smart. Neither their body nor their brain want them to starve.
Our bodies evolved during a time where there was food scarcity and the potential of famine. We have a complex system that was designed to make sure that we always held on to extra weight, specifically extra fat, because that’s how we store energy. In the current obesogenic environment, what happens is our bodies carry extra weight, or specifically, extra fat.
Again, I say to them, this is biology. Your body’s doing exactly what it was designed to do. Your body’s very smart, but now we have to figure out how to help your body want to carry less fat because it is impacting your health. This is not your fault. Having obesity is not your fault any more than having diabetes or hypertension is anyone’s fault. Now it’s time for all of us to use highly effective tools that target the pathophysiology of obesity.
When a patient comes to me for weight management or to help them treat their obesity, I listen to them, and I look for clues as to what might help that specific patient. Every patient deserves to have individualized treatment. One medicine may be right for one person, another medicine may be right for another, and surgery may be right for another patient. I really try to listen and hear what that patient is telling me.
What we as providers really need is tools – different options – to be able to provide for our patients and basically present them with different options, and then guide them toward the best therapy for them. Whether it’s semaglutide or tirzepatide potentially in the future, these types of medications are excellent options for our patients. They’re highly effective tools with safe profiles.
A question that I often get from providers or patients is, “Well, Doctor, I’ve lost the weight now. How long should I take this medicine? Can I stop it now?”
Then, we have a conversation, and we actually usually have this conversation even before we start the medicine. Basically, we talk about the fact that obesity is a chronic disease. There’s no cure for obesity. Because it’s a chronic disease, we need to treat it like we would treat any other chronic disease.
The example that I often use is, if you have a patient who has hypertension and you start them on an antihypertensive medication, what happens? Their blood pressure goes down. It improves. Now, if their blood pressure is improved with a specific antihypertensive, would you stop that medicine? What would happen if you stopped that antihypertensive? Well, their blood pressure would go up, and we wouldn’t be surprised.
In the same way, if you have a patient who has obesity and you start that patient on an antiobesity medication, and their weight decreases, and their body fat mass at that point decreases, what would happen if you stop that medicine? They lost the weight, but you stop the medicine. Well, their weight gain comes back. They regain the weight.
We should not be surprised that weight gain occurs when we stop the treatment. That really underscores the fact that treatment needs to be continued. If a patient is started on an antiobesity medication and they lose weight, that medication needs to be continued to maintain that weight loss.
Basically, we eat food and our body responds by releasing these hormones. The hormones are made in our gut and in our pancreas and these hormones inform our brain. Are we hungry? Are we full? Where are we with our homeostatic set point of fat mass? Based on that, our brain is like the sensor or the thermostat.
Obesity is a chronic, treatable disease. We should treat obesity as we treat any other chronic disease, with effective and safe approaches that target underlying disease mechanisms. These results in the SURMOUNT-1 trial underscore that tirzepatide may be doing just that. Remarkably, 9 in 10 individuals with obesity lost weight while taking tirzepatide. These results are impressive. They’re an important step forward in potentially expanding effective therapeutic options for people with obesity.
Dr. Jastreboff is an associate professor of medicine and pediatrics at Yale University, New Haven, Conn., and director of weight management and obesity prevention at Yale Stress Center. She reported conducting trials with Eli Lilly, Novo Nordisk, and Rhythm Pharmaceuticals; serving on scientific advisory boards for Ely Lilly, Intellihealth, Novo Nordisk, Pfizer, Rhythm Pharmaceuticals, and WW; and consulting for Boehringer Ingelheim and Scholar Rock.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
It’s important to remember and to think about the first time when patients with obesity come to see us: What have they faced? What have been their struggles? What shame and blame and bias have they faced?
One of the first things that I do when a patient comes to see me is invite them to share their weight journey with me. I ask them to tell me about their struggles, about what’s worked and what hasn’t worked, what they would like, and what their health goals are.
As they share their stories, I look for the opportunity to share with them that obesity is not their fault, but that it’s biology driving their body to carry extra weight and their body is super smart. Neither their body nor their brain want them to starve.
Our bodies evolved during a time where there was food scarcity and the potential of famine. We have a complex system that was designed to make sure that we always held on to extra weight, specifically extra fat, because that’s how we store energy. In the current obesogenic environment, what happens is our bodies carry extra weight, or specifically, extra fat.
Again, I say to them, this is biology. Your body’s doing exactly what it was designed to do. Your body’s very smart, but now we have to figure out how to help your body want to carry less fat because it is impacting your health. This is not your fault. Having obesity is not your fault any more than having diabetes or hypertension is anyone’s fault. Now it’s time for all of us to use highly effective tools that target the pathophysiology of obesity.
When a patient comes to me for weight management or to help them treat their obesity, I listen to them, and I look for clues as to what might help that specific patient. Every patient deserves to have individualized treatment. One medicine may be right for one person, another medicine may be right for another, and surgery may be right for another patient. I really try to listen and hear what that patient is telling me.
What we as providers really need is tools – different options – to be able to provide for our patients and basically present them with different options, and then guide them toward the best therapy for them. Whether it’s semaglutide or tirzepatide potentially in the future, these types of medications are excellent options for our patients. They’re highly effective tools with safe profiles.
A question that I often get from providers or patients is, “Well, Doctor, I’ve lost the weight now. How long should I take this medicine? Can I stop it now?”
Then, we have a conversation, and we actually usually have this conversation even before we start the medicine. Basically, we talk about the fact that obesity is a chronic disease. There’s no cure for obesity. Because it’s a chronic disease, we need to treat it like we would treat any other chronic disease.
The example that I often use is, if you have a patient who has hypertension and you start them on an antihypertensive medication, what happens? Their blood pressure goes down. It improves. Now, if their blood pressure is improved with a specific antihypertensive, would you stop that medicine? What would happen if you stopped that antihypertensive? Well, their blood pressure would go up, and we wouldn’t be surprised.
In the same way, if you have a patient who has obesity and you start that patient on an antiobesity medication, and their weight decreases, and their body fat mass at that point decreases, what would happen if you stop that medicine? They lost the weight, but you stop the medicine. Well, their weight gain comes back. They regain the weight.
We should not be surprised that weight gain occurs when we stop the treatment. That really underscores the fact that treatment needs to be continued. If a patient is started on an antiobesity medication and they lose weight, that medication needs to be continued to maintain that weight loss.
Basically, we eat food and our body responds by releasing these hormones. The hormones are made in our gut and in our pancreas and these hormones inform our brain. Are we hungry? Are we full? Where are we with our homeostatic set point of fat mass? Based on that, our brain is like the sensor or the thermostat.
Obesity is a chronic, treatable disease. We should treat obesity as we treat any other chronic disease, with effective and safe approaches that target underlying disease mechanisms. These results in the SURMOUNT-1 trial underscore that tirzepatide may be doing just that. Remarkably, 9 in 10 individuals with obesity lost weight while taking tirzepatide. These results are impressive. They’re an important step forward in potentially expanding effective therapeutic options for people with obesity.
Dr. Jastreboff is an associate professor of medicine and pediatrics at Yale University, New Haven, Conn., and director of weight management and obesity prevention at Yale Stress Center. She reported conducting trials with Eli Lilly, Novo Nordisk, and Rhythm Pharmaceuticals; serving on scientific advisory boards for Ely Lilly, Intellihealth, Novo Nordisk, Pfizer, Rhythm Pharmaceuticals, and WW; and consulting for Boehringer Ingelheim and Scholar Rock.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
It’s important to remember and to think about the first time when patients with obesity come to see us: What have they faced? What have been their struggles? What shame and blame and bias have they faced?
One of the first things that I do when a patient comes to see me is invite them to share their weight journey with me. I ask them to tell me about their struggles, about what’s worked and what hasn’t worked, what they would like, and what their health goals are.
As they share their stories, I look for the opportunity to share with them that obesity is not their fault, but that it’s biology driving their body to carry extra weight and their body is super smart. Neither their body nor their brain want them to starve.
Our bodies evolved during a time where there was food scarcity and the potential of famine. We have a complex system that was designed to make sure that we always held on to extra weight, specifically extra fat, because that’s how we store energy. In the current obesogenic environment, what happens is our bodies carry extra weight, or specifically, extra fat.
Again, I say to them, this is biology. Your body’s doing exactly what it was designed to do. Your body’s very smart, but now we have to figure out how to help your body want to carry less fat because it is impacting your health. This is not your fault. Having obesity is not your fault any more than having diabetes or hypertension is anyone’s fault. Now it’s time for all of us to use highly effective tools that target the pathophysiology of obesity.
When a patient comes to me for weight management or to help them treat their obesity, I listen to them, and I look for clues as to what might help that specific patient. Every patient deserves to have individualized treatment. One medicine may be right for one person, another medicine may be right for another, and surgery may be right for another patient. I really try to listen and hear what that patient is telling me.
What we as providers really need is tools – different options – to be able to provide for our patients and basically present them with different options, and then guide them toward the best therapy for them. Whether it’s semaglutide or tirzepatide potentially in the future, these types of medications are excellent options for our patients. They’re highly effective tools with safe profiles.
A question that I often get from providers or patients is, “Well, Doctor, I’ve lost the weight now. How long should I take this medicine? Can I stop it now?”
Then, we have a conversation, and we actually usually have this conversation even before we start the medicine. Basically, we talk about the fact that obesity is a chronic disease. There’s no cure for obesity. Because it’s a chronic disease, we need to treat it like we would treat any other chronic disease.
The example that I often use is, if you have a patient who has hypertension and you start them on an antihypertensive medication, what happens? Their blood pressure goes down. It improves. Now, if their blood pressure is improved with a specific antihypertensive, would you stop that medicine? What would happen if you stopped that antihypertensive? Well, their blood pressure would go up, and we wouldn’t be surprised.
In the same way, if you have a patient who has obesity and you start that patient on an antiobesity medication, and their weight decreases, and their body fat mass at that point decreases, what would happen if you stop that medicine? They lost the weight, but you stop the medicine. Well, their weight gain comes back. They regain the weight.
We should not be surprised that weight gain occurs when we stop the treatment. That really underscores the fact that treatment needs to be continued. If a patient is started on an antiobesity medication and they lose weight, that medication needs to be continued to maintain that weight loss.
Basically, we eat food and our body responds by releasing these hormones. The hormones are made in our gut and in our pancreas and these hormones inform our brain. Are we hungry? Are we full? Where are we with our homeostatic set point of fat mass? Based on that, our brain is like the sensor or the thermostat.
Obesity is a chronic, treatable disease. We should treat obesity as we treat any other chronic disease, with effective and safe approaches that target underlying disease mechanisms. These results in the SURMOUNT-1 trial underscore that tirzepatide may be doing just that. Remarkably, 9 in 10 individuals with obesity lost weight while taking tirzepatide. These results are impressive. They’re an important step forward in potentially expanding effective therapeutic options for people with obesity.
Dr. Jastreboff is an associate professor of medicine and pediatrics at Yale University, New Haven, Conn., and director of weight management and obesity prevention at Yale Stress Center. She reported conducting trials with Eli Lilly, Novo Nordisk, and Rhythm Pharmaceuticals; serving on scientific advisory boards for Ely Lilly, Intellihealth, Novo Nordisk, Pfizer, Rhythm Pharmaceuticals, and WW; and consulting for Boehringer Ingelheim and Scholar Rock.
A version of this article first appeared on Medscape.com.
New AHA checklist: Only one in five adults has optimal heart health
About 80% of American adults have low to moderate cardiovascular (CV) health based on the American Heart Association checklist for optimal heart health, which now includes healthy sleep as an essential component for heart health.
With the addition of sleep, “Life’s Essential 8” replaces the AHA’s “Life’s Simple 7” checklist.
“The new metric of sleep duration reflects the latest research findings: Sleep impacts overall health, and people who have healthier sleep patterns manage health factors such as weight, blood pressure, or risk for type 2 diabetes more effectively,” AHA President Donald M. Lloyd-Jones, MD, said in a news release.
“In addition, advances in ways to measure sleep, such as with wearable devices, now offer people the ability to reliably and routinely monitor their sleep habits at home,” said Dr. Lloyd-Jones, chair of the department of preventive medicine at Northwestern University in Chicago.
The AHA Presidential Advisory – Life’s Essential 8: Updating and Enhancing the American Heart Association’s Construct on Cardiovascular Health – was published online in the journal Circulation.
A companion paper published simultaneously in Circulation reports the first study using Life’s Essential 8.
Overall, the results show that CV health of the U.S. population is “suboptimal, and we see important differences across age and sociodemographic groups,” Dr. Lloyd-Jones said.
Refining Life’s Simple 7
The AHA first defined the seven metrics for optimal CV health in 2010. After 12 years and more than 2,400 scientific papers on the topic, new discoveries in CV health and ways to measure it provided an opportunity to revisit each health component in more detail and provide updates as needed, the AHA explains.
“We felt it was the right time to conduct a comprehensive review of the latest research to refine the existing metrics and consider any new metrics that add value to assessing cardiovascular health for all people,” Dr. Lloyd-Jones said.
Four of the original metrics have been redefined for consistency with newer clinical guidelines or compatibility with new measurement tools, and the scoring system can now also be applied to anyone ages 2 and older. Here is a snapshot of Life’s Essential 8 metrics, including updates.
1. Diet (updated)
The tool includes a new guide to assess diet quality for adults and children at the individual and population level. At the population level, dietary assessment is based on daily intake of elements in the Dietary Approaches to Stop Hypertension (DASH) eating pattern. For individuals, the Mediterranean Eating Pattern for Americans (MEPA) is used to assess and monitor cardiovascular health.
2. Physical activity (no changes)
Physical activity continues to be measured by the total number of minutes of moderate or vigorous physical activity per week, as defined by the U.S. Physical Activity Guidelines for Americans (2nd edition). The optimal level is 150 minutes (2.5 hours) of moderate physical activity or more per week or 75 minutes per week of vigorous-intensity physical activity for adults; 420 minutes (7 hours) or more per week for children ages 6 and older; and age-specific modifications for younger children.
3. Nicotine exposure (updated)
Use of inhaled nicotine-delivery systems, which includes e-cigarettes or vaping devices, has been added since the previous metric monitored only traditional, combustible cigarettes. This reflects use by adults and youth and their implications on long-term health. Second-hand smoke exposure for children and adults has also been added.
4. Sleep duration (new)
Sleep duration is associated with CV health. Measured by average hours of sleep per night, the ideal level is 7-9 hours daily for adults. Ideal daily sleep ranges for children are 10-16 hours per 24 hours for ages 5 and younger; 9-12 hours for ages 6-12 years; and 8-10 hours for ages 13-18 years.
5. Body mass index (no changes)
The AHA acknowledges that body mass index (BMI) is an imperfect metric. Yet, because it’s easily calculated and widely available, BMI continues as a “reasonable” gauge to assess weight categories that may lead to health problems. BMI of 18.5-24.9 is associated with the highest levels of CV health. The AHA notes that BMI ranges and the subsequent health risks associated with them may differ among people from diverse racial or ethnic backgrounds or ancestry. This aligns with the World Health Organization recommendations to adjust BMI ranges for people of Asian or Pacific Islander ancestry because recent evidence indicates their risk of conditions such as CVD or type 2 diabetes is higher at a lower BMI.
6. Blood lipids (updated)
The metric for blood lipids (cholesterol and triglycerides) is updated to use non-HDL cholesterol as the preferred number to monitor, rather than total cholesterol. This shift is made because non-HDL cholesterol can be measured without fasting beforehand (thereby increasing its availability at any time of day and implementation at more appointments) and reliably calculated among all people.
7. Blood glucose (updated)
This metric is expanded to include the option of hemoglobin A1c readings or blood glucose levels for people with or without type 1 or 2 diabetes or prediabetes.
8. Blood pressure (no changes)
Blood pressure criteria remain unchanged from 2017 guidance that established levels less than 120/80 mm Hg as optimal, and defined hypertension as 130-139 mm Hg systolic pressure or 80-89 mm Hg diastolic pressure.
‘Concerning’ new data
Results of the first study using Life’s Essential 8 show that the overall CV health of the U.S. population is “well below ideal,” with 80% of adults scoring at a low or moderate level, the researchers report.
Data for the analysis came from 2013-2018 U.S. National Health and Nutrition Examination surveys (NHANES) of more than 13,500 adults aged 20-79 years and nearly 9,900 children aged 2-19 years. Among the key findings:
- The average CV health score based on Life’s Essential 8 was 64.7 for adults and 65.5 for children – in the moderate range on the 0-100 scale.
- Only 0.45% of adults had a perfect score of 100; 20% had high CV health (score of 80 or higher), 63% moderate (score of 50-79), and 18% had low CV health (score of less than 50).
- Adult women had higher average CV health scores (67) compared with men (62.5).
- In general, adults scored lowest in the areas of diet, physical activity, and BMI.
- CV health scores were generally lower at older ages.
- Non-Hispanic Asian Americans had a higher average CV health score than other racial/ethnic groups. Non-Hispanic Whites had the second highest average CV health score, followed, in order, by Hispanic (other than Mexican), Mexican, and non-Hispanic Blacks.
- Children’s diet scores were low, at an average of 40.6.
- Adult sociodemographic groups varied notably in CV health scores for diet, nicotine exposure, blood glucose, and blood pressure.
“These data represent the first look at the cardiovascular health of the U.S. population using the AHA’s new Life’s Essential 8 scoring algorithm,” Dr. Lloyd-Jones said.
“Life’s Essential 8 is a major step forward in our ability to identify when cardiovascular health can be preserved and when it is suboptimal. It should energize efforts to improve cardiovascular health for all people and at every life stage,” Dr. Lloyd-Jones added.
“Analyses like this can help policymakers, communities, clinicians, and the public to understand the opportunities to intervene to improve and maintain optimal cardiovascular health across the life course,” he said.
This research had no commercial funding. The authors have no reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
About 80% of American adults have low to moderate cardiovascular (CV) health based on the American Heart Association checklist for optimal heart health, which now includes healthy sleep as an essential component for heart health.
With the addition of sleep, “Life’s Essential 8” replaces the AHA’s “Life’s Simple 7” checklist.
“The new metric of sleep duration reflects the latest research findings: Sleep impacts overall health, and people who have healthier sleep patterns manage health factors such as weight, blood pressure, or risk for type 2 diabetes more effectively,” AHA President Donald M. Lloyd-Jones, MD, said in a news release.
“In addition, advances in ways to measure sleep, such as with wearable devices, now offer people the ability to reliably and routinely monitor their sleep habits at home,” said Dr. Lloyd-Jones, chair of the department of preventive medicine at Northwestern University in Chicago.
The AHA Presidential Advisory – Life’s Essential 8: Updating and Enhancing the American Heart Association’s Construct on Cardiovascular Health – was published online in the journal Circulation.
A companion paper published simultaneously in Circulation reports the first study using Life’s Essential 8.
Overall, the results show that CV health of the U.S. population is “suboptimal, and we see important differences across age and sociodemographic groups,” Dr. Lloyd-Jones said.
Refining Life’s Simple 7
The AHA first defined the seven metrics for optimal CV health in 2010. After 12 years and more than 2,400 scientific papers on the topic, new discoveries in CV health and ways to measure it provided an opportunity to revisit each health component in more detail and provide updates as needed, the AHA explains.
“We felt it was the right time to conduct a comprehensive review of the latest research to refine the existing metrics and consider any new metrics that add value to assessing cardiovascular health for all people,” Dr. Lloyd-Jones said.
Four of the original metrics have been redefined for consistency with newer clinical guidelines or compatibility with new measurement tools, and the scoring system can now also be applied to anyone ages 2 and older. Here is a snapshot of Life’s Essential 8 metrics, including updates.
1. Diet (updated)
The tool includes a new guide to assess diet quality for adults and children at the individual and population level. At the population level, dietary assessment is based on daily intake of elements in the Dietary Approaches to Stop Hypertension (DASH) eating pattern. For individuals, the Mediterranean Eating Pattern for Americans (MEPA) is used to assess and monitor cardiovascular health.
2. Physical activity (no changes)
Physical activity continues to be measured by the total number of minutes of moderate or vigorous physical activity per week, as defined by the U.S. Physical Activity Guidelines for Americans (2nd edition). The optimal level is 150 minutes (2.5 hours) of moderate physical activity or more per week or 75 minutes per week of vigorous-intensity physical activity for adults; 420 minutes (7 hours) or more per week for children ages 6 and older; and age-specific modifications for younger children.
3. Nicotine exposure (updated)
Use of inhaled nicotine-delivery systems, which includes e-cigarettes or vaping devices, has been added since the previous metric monitored only traditional, combustible cigarettes. This reflects use by adults and youth and their implications on long-term health. Second-hand smoke exposure for children and adults has also been added.
4. Sleep duration (new)
Sleep duration is associated with CV health. Measured by average hours of sleep per night, the ideal level is 7-9 hours daily for adults. Ideal daily sleep ranges for children are 10-16 hours per 24 hours for ages 5 and younger; 9-12 hours for ages 6-12 years; and 8-10 hours for ages 13-18 years.
5. Body mass index (no changes)
The AHA acknowledges that body mass index (BMI) is an imperfect metric. Yet, because it’s easily calculated and widely available, BMI continues as a “reasonable” gauge to assess weight categories that may lead to health problems. BMI of 18.5-24.9 is associated with the highest levels of CV health. The AHA notes that BMI ranges and the subsequent health risks associated with them may differ among people from diverse racial or ethnic backgrounds or ancestry. This aligns with the World Health Organization recommendations to adjust BMI ranges for people of Asian or Pacific Islander ancestry because recent evidence indicates their risk of conditions such as CVD or type 2 diabetes is higher at a lower BMI.
6. Blood lipids (updated)
The metric for blood lipids (cholesterol and triglycerides) is updated to use non-HDL cholesterol as the preferred number to monitor, rather than total cholesterol. This shift is made because non-HDL cholesterol can be measured without fasting beforehand (thereby increasing its availability at any time of day and implementation at more appointments) and reliably calculated among all people.
7. Blood glucose (updated)
This metric is expanded to include the option of hemoglobin A1c readings or blood glucose levels for people with or without type 1 or 2 diabetes or prediabetes.
8. Blood pressure (no changes)
Blood pressure criteria remain unchanged from 2017 guidance that established levels less than 120/80 mm Hg as optimal, and defined hypertension as 130-139 mm Hg systolic pressure or 80-89 mm Hg diastolic pressure.
‘Concerning’ new data
Results of the first study using Life’s Essential 8 show that the overall CV health of the U.S. population is “well below ideal,” with 80% of adults scoring at a low or moderate level, the researchers report.
Data for the analysis came from 2013-2018 U.S. National Health and Nutrition Examination surveys (NHANES) of more than 13,500 adults aged 20-79 years and nearly 9,900 children aged 2-19 years. Among the key findings:
- The average CV health score based on Life’s Essential 8 was 64.7 for adults and 65.5 for children – in the moderate range on the 0-100 scale.
- Only 0.45% of adults had a perfect score of 100; 20% had high CV health (score of 80 or higher), 63% moderate (score of 50-79), and 18% had low CV health (score of less than 50).
- Adult women had higher average CV health scores (67) compared with men (62.5).
- In general, adults scored lowest in the areas of diet, physical activity, and BMI.
- CV health scores were generally lower at older ages.
- Non-Hispanic Asian Americans had a higher average CV health score than other racial/ethnic groups. Non-Hispanic Whites had the second highest average CV health score, followed, in order, by Hispanic (other than Mexican), Mexican, and non-Hispanic Blacks.
- Children’s diet scores were low, at an average of 40.6.
- Adult sociodemographic groups varied notably in CV health scores for diet, nicotine exposure, blood glucose, and blood pressure.
“These data represent the first look at the cardiovascular health of the U.S. population using the AHA’s new Life’s Essential 8 scoring algorithm,” Dr. Lloyd-Jones said.
“Life’s Essential 8 is a major step forward in our ability to identify when cardiovascular health can be preserved and when it is suboptimal. It should energize efforts to improve cardiovascular health for all people and at every life stage,” Dr. Lloyd-Jones added.
“Analyses like this can help policymakers, communities, clinicians, and the public to understand the opportunities to intervene to improve and maintain optimal cardiovascular health across the life course,” he said.
This research had no commercial funding. The authors have no reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
About 80% of American adults have low to moderate cardiovascular (CV) health based on the American Heart Association checklist for optimal heart health, which now includes healthy sleep as an essential component for heart health.
With the addition of sleep, “Life’s Essential 8” replaces the AHA’s “Life’s Simple 7” checklist.
“The new metric of sleep duration reflects the latest research findings: Sleep impacts overall health, and people who have healthier sleep patterns manage health factors such as weight, blood pressure, or risk for type 2 diabetes more effectively,” AHA President Donald M. Lloyd-Jones, MD, said in a news release.
“In addition, advances in ways to measure sleep, such as with wearable devices, now offer people the ability to reliably and routinely monitor their sleep habits at home,” said Dr. Lloyd-Jones, chair of the department of preventive medicine at Northwestern University in Chicago.
The AHA Presidential Advisory – Life’s Essential 8: Updating and Enhancing the American Heart Association’s Construct on Cardiovascular Health – was published online in the journal Circulation.
A companion paper published simultaneously in Circulation reports the first study using Life’s Essential 8.
Overall, the results show that CV health of the U.S. population is “suboptimal, and we see important differences across age and sociodemographic groups,” Dr. Lloyd-Jones said.
Refining Life’s Simple 7
The AHA first defined the seven metrics for optimal CV health in 2010. After 12 years and more than 2,400 scientific papers on the topic, new discoveries in CV health and ways to measure it provided an opportunity to revisit each health component in more detail and provide updates as needed, the AHA explains.
“We felt it was the right time to conduct a comprehensive review of the latest research to refine the existing metrics and consider any new metrics that add value to assessing cardiovascular health for all people,” Dr. Lloyd-Jones said.
Four of the original metrics have been redefined for consistency with newer clinical guidelines or compatibility with new measurement tools, and the scoring system can now also be applied to anyone ages 2 and older. Here is a snapshot of Life’s Essential 8 metrics, including updates.
1. Diet (updated)
The tool includes a new guide to assess diet quality for adults and children at the individual and population level. At the population level, dietary assessment is based on daily intake of elements in the Dietary Approaches to Stop Hypertension (DASH) eating pattern. For individuals, the Mediterranean Eating Pattern for Americans (MEPA) is used to assess and monitor cardiovascular health.
2. Physical activity (no changes)
Physical activity continues to be measured by the total number of minutes of moderate or vigorous physical activity per week, as defined by the U.S. Physical Activity Guidelines for Americans (2nd edition). The optimal level is 150 minutes (2.5 hours) of moderate physical activity or more per week or 75 minutes per week of vigorous-intensity physical activity for adults; 420 minutes (7 hours) or more per week for children ages 6 and older; and age-specific modifications for younger children.
3. Nicotine exposure (updated)
Use of inhaled nicotine-delivery systems, which includes e-cigarettes or vaping devices, has been added since the previous metric monitored only traditional, combustible cigarettes. This reflects use by adults and youth and their implications on long-term health. Second-hand smoke exposure for children and adults has also been added.
4. Sleep duration (new)
Sleep duration is associated with CV health. Measured by average hours of sleep per night, the ideal level is 7-9 hours daily for adults. Ideal daily sleep ranges for children are 10-16 hours per 24 hours for ages 5 and younger; 9-12 hours for ages 6-12 years; and 8-10 hours for ages 13-18 years.
5. Body mass index (no changes)
The AHA acknowledges that body mass index (BMI) is an imperfect metric. Yet, because it’s easily calculated and widely available, BMI continues as a “reasonable” gauge to assess weight categories that may lead to health problems. BMI of 18.5-24.9 is associated with the highest levels of CV health. The AHA notes that BMI ranges and the subsequent health risks associated with them may differ among people from diverse racial or ethnic backgrounds or ancestry. This aligns with the World Health Organization recommendations to adjust BMI ranges for people of Asian or Pacific Islander ancestry because recent evidence indicates their risk of conditions such as CVD or type 2 diabetes is higher at a lower BMI.
6. Blood lipids (updated)
The metric for blood lipids (cholesterol and triglycerides) is updated to use non-HDL cholesterol as the preferred number to monitor, rather than total cholesterol. This shift is made because non-HDL cholesterol can be measured without fasting beforehand (thereby increasing its availability at any time of day and implementation at more appointments) and reliably calculated among all people.
7. Blood glucose (updated)
This metric is expanded to include the option of hemoglobin A1c readings or blood glucose levels for people with or without type 1 or 2 diabetes or prediabetes.
8. Blood pressure (no changes)
Blood pressure criteria remain unchanged from 2017 guidance that established levels less than 120/80 mm Hg as optimal, and defined hypertension as 130-139 mm Hg systolic pressure or 80-89 mm Hg diastolic pressure.
‘Concerning’ new data
Results of the first study using Life’s Essential 8 show that the overall CV health of the U.S. population is “well below ideal,” with 80% of adults scoring at a low or moderate level, the researchers report.
Data for the analysis came from 2013-2018 U.S. National Health and Nutrition Examination surveys (NHANES) of more than 13,500 adults aged 20-79 years and nearly 9,900 children aged 2-19 years. Among the key findings:
- The average CV health score based on Life’s Essential 8 was 64.7 for adults and 65.5 for children – in the moderate range on the 0-100 scale.
- Only 0.45% of adults had a perfect score of 100; 20% had high CV health (score of 80 or higher), 63% moderate (score of 50-79), and 18% had low CV health (score of less than 50).
- Adult women had higher average CV health scores (67) compared with men (62.5).
- In general, adults scored lowest in the areas of diet, physical activity, and BMI.
- CV health scores were generally lower at older ages.
- Non-Hispanic Asian Americans had a higher average CV health score than other racial/ethnic groups. Non-Hispanic Whites had the second highest average CV health score, followed, in order, by Hispanic (other than Mexican), Mexican, and non-Hispanic Blacks.
- Children’s diet scores were low, at an average of 40.6.
- Adult sociodemographic groups varied notably in CV health scores for diet, nicotine exposure, blood glucose, and blood pressure.
“These data represent the first look at the cardiovascular health of the U.S. population using the AHA’s new Life’s Essential 8 scoring algorithm,” Dr. Lloyd-Jones said.
“Life’s Essential 8 is a major step forward in our ability to identify when cardiovascular health can be preserved and when it is suboptimal. It should energize efforts to improve cardiovascular health for all people and at every life stage,” Dr. Lloyd-Jones added.
“Analyses like this can help policymakers, communities, clinicians, and the public to understand the opportunities to intervene to improve and maintain optimal cardiovascular health across the life course,” he said.
This research had no commercial funding. The authors have no reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CIRCULATION
Heart attack care not equal for women and people of color
Radiating chest pain, shortness of breath, nausea, lightheadedness. Everyone knows the telltale signs of a myocardial infarction. Yet a new study shows that despite this widespread recognition, heart attacks aren’t attended to quickly across the board. Historically, the study says, women and people of color wait longer to access emergency care for a heart attack.
Researchers from the University of California, San Francisco published these findings in the Annals of Emergency Medicine. The study used the Office of Statewide Health Planning and Development dataset to gather information on 453,136 cases of heart attack in California between 2005 and 2015. They found that over time, differences in timely treatment between the demographics narrowed, but the gap still existed.
The study defined timely treatment as receiving care for a heart attack within 3 days of admission to a hospital. Women and people of color were found to wait 3 days or more to receive care than their White male counterparts. A disparity of this sort can cause ripples of health effects across society, ripples that doctors should be aware of, says lead author Juan Carlos Montoy, MD. Dr. Montoy was “sadly surprised by our findings that disparities for women and for Black patients only decreased slightly or not at all over time.”
In the study, the team separated the dataset between the two primary types of heart attack: ST-segment elevation myocardial infarction (STEMI), caused by blood vessel blockage, and non–ST-segment elevation myocardial infarction (NSTEMI), caused by a narrowing or temporary blockage of the artery.
Regardless of the type of heart attack, the standard first step in treatment is a coronary angiogram. After finding out where blood flow is disrupted using the angiogram, a physician can proceed with treatment.
But when looking back, the team found that it took a while for many patients to receive this first step in treatment. In 2005, 50% of men and 35.7% of women with STEMI and 45% of men and 33.1% of women with NSTEMI had a timely angiography. In the same year, 46% of White patients and 31.2% of Black patients with STEMI underwent timely angiography.
By 2015, timely treatment increased across the board, but there were still discrepancies, with 76.7% of men and 66.8% of women with STEMI undergoing timely angiography and 56.3% of men and 45.9% of women with NSTEMI undergoing timely angiography. Also in 2015, 75.2% of White patients and 69.2% of Black patients underwent timely angiography for STEMI.
Although differences in care decreased between the demographics, the gap still exists. Whereas this dataset only extends to 2015, this trend may still persist today, says Robert Glatter, MD, an emergency medicine physician at Lenox Hill Hospital, New York, who was not involved in the study. Therefore, physicians need to consider this bias when treating patients. “The bottom line is that we continue to have much work to do to achieve equality in managing not only medical conditions but treating people who have them equally,” Dr. Glatter said.
“Raising awareness of ongoing inequality in care related to gender and ethnic disparities is critical to drive change in our institutions,” he emphasized. “We simply cannot accept the status quo.”
The study was funded by the National Heart, Lung, and Blood Institute of the National Institutes of Health. Dr. Glatter and the authors declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Radiating chest pain, shortness of breath, nausea, lightheadedness. Everyone knows the telltale signs of a myocardial infarction. Yet a new study shows that despite this widespread recognition, heart attacks aren’t attended to quickly across the board. Historically, the study says, women and people of color wait longer to access emergency care for a heart attack.
Researchers from the University of California, San Francisco published these findings in the Annals of Emergency Medicine. The study used the Office of Statewide Health Planning and Development dataset to gather information on 453,136 cases of heart attack in California between 2005 and 2015. They found that over time, differences in timely treatment between the demographics narrowed, but the gap still existed.
The study defined timely treatment as receiving care for a heart attack within 3 days of admission to a hospital. Women and people of color were found to wait 3 days or more to receive care than their White male counterparts. A disparity of this sort can cause ripples of health effects across society, ripples that doctors should be aware of, says lead author Juan Carlos Montoy, MD. Dr. Montoy was “sadly surprised by our findings that disparities for women and for Black patients only decreased slightly or not at all over time.”
In the study, the team separated the dataset between the two primary types of heart attack: ST-segment elevation myocardial infarction (STEMI), caused by blood vessel blockage, and non–ST-segment elevation myocardial infarction (NSTEMI), caused by a narrowing or temporary blockage of the artery.
Regardless of the type of heart attack, the standard first step in treatment is a coronary angiogram. After finding out where blood flow is disrupted using the angiogram, a physician can proceed with treatment.
But when looking back, the team found that it took a while for many patients to receive this first step in treatment. In 2005, 50% of men and 35.7% of women with STEMI and 45% of men and 33.1% of women with NSTEMI had a timely angiography. In the same year, 46% of White patients and 31.2% of Black patients with STEMI underwent timely angiography.
By 2015, timely treatment increased across the board, but there were still discrepancies, with 76.7% of men and 66.8% of women with STEMI undergoing timely angiography and 56.3% of men and 45.9% of women with NSTEMI undergoing timely angiography. Also in 2015, 75.2% of White patients and 69.2% of Black patients underwent timely angiography for STEMI.
Although differences in care decreased between the demographics, the gap still exists. Whereas this dataset only extends to 2015, this trend may still persist today, says Robert Glatter, MD, an emergency medicine physician at Lenox Hill Hospital, New York, who was not involved in the study. Therefore, physicians need to consider this bias when treating patients. “The bottom line is that we continue to have much work to do to achieve equality in managing not only medical conditions but treating people who have them equally,” Dr. Glatter said.
“Raising awareness of ongoing inequality in care related to gender and ethnic disparities is critical to drive change in our institutions,” he emphasized. “We simply cannot accept the status quo.”
The study was funded by the National Heart, Lung, and Blood Institute of the National Institutes of Health. Dr. Glatter and the authors declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Radiating chest pain, shortness of breath, nausea, lightheadedness. Everyone knows the telltale signs of a myocardial infarction. Yet a new study shows that despite this widespread recognition, heart attacks aren’t attended to quickly across the board. Historically, the study says, women and people of color wait longer to access emergency care for a heart attack.
Researchers from the University of California, San Francisco published these findings in the Annals of Emergency Medicine. The study used the Office of Statewide Health Planning and Development dataset to gather information on 453,136 cases of heart attack in California between 2005 and 2015. They found that over time, differences in timely treatment between the demographics narrowed, but the gap still existed.
The study defined timely treatment as receiving care for a heart attack within 3 days of admission to a hospital. Women and people of color were found to wait 3 days or more to receive care than their White male counterparts. A disparity of this sort can cause ripples of health effects across society, ripples that doctors should be aware of, says lead author Juan Carlos Montoy, MD. Dr. Montoy was “sadly surprised by our findings that disparities for women and for Black patients only decreased slightly or not at all over time.”
In the study, the team separated the dataset between the two primary types of heart attack: ST-segment elevation myocardial infarction (STEMI), caused by blood vessel blockage, and non–ST-segment elevation myocardial infarction (NSTEMI), caused by a narrowing or temporary blockage of the artery.
Regardless of the type of heart attack, the standard first step in treatment is a coronary angiogram. After finding out where blood flow is disrupted using the angiogram, a physician can proceed with treatment.
But when looking back, the team found that it took a while for many patients to receive this first step in treatment. In 2005, 50% of men and 35.7% of women with STEMI and 45% of men and 33.1% of women with NSTEMI had a timely angiography. In the same year, 46% of White patients and 31.2% of Black patients with STEMI underwent timely angiography.
By 2015, timely treatment increased across the board, but there were still discrepancies, with 76.7% of men and 66.8% of women with STEMI undergoing timely angiography and 56.3% of men and 45.9% of women with NSTEMI undergoing timely angiography. Also in 2015, 75.2% of White patients and 69.2% of Black patients underwent timely angiography for STEMI.
Although differences in care decreased between the demographics, the gap still exists. Whereas this dataset only extends to 2015, this trend may still persist today, says Robert Glatter, MD, an emergency medicine physician at Lenox Hill Hospital, New York, who was not involved in the study. Therefore, physicians need to consider this bias when treating patients. “The bottom line is that we continue to have much work to do to achieve equality in managing not only medical conditions but treating people who have them equally,” Dr. Glatter said.
“Raising awareness of ongoing inequality in care related to gender and ethnic disparities is critical to drive change in our institutions,” he emphasized. “We simply cannot accept the status quo.”
The study was funded by the National Heart, Lung, and Blood Institute of the National Institutes of Health. Dr. Glatter and the authors declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF EMERGENCY MEDICINE
New biomarker data add to concerns over REDUCE-IT trial
A new analysis of the REDUCE-IT study has reignited concerns that the benefit shown by the high-dose fish oil product in the study, icosapent ethyl (Vascepa, Amarin), may have been related to harms caused by the placebo mineral oil.
Results show that allocation to icosapent ethyl had minimal effects on a series of biomarkers associated with atherosclerotic disease, whereas levels of these biomarkers increased among those allocated to mineral oil.
At 12 months, the median percent increases from baseline in the mineral oil group were 1.5% for homocysteine, 2.2% for lipoprotein(a), 10.9% for oxidized low-density-lipoprotein (LDL) cholesterol, 16.2% for interleukin (IL)-6, 18.5% for lipoprotein-associated phospholipase A2, 21.9% for high-sensitivity C-reactive protein (hsCRP), and 28.9% for IL-1β. The changes were similar at 24 months. However, in the icosapent ethyl group, there were minimal changes in these biomarkers at 12 and 24 months.
The study was published online in Circulation.
The authors, led by Paul Ridker, MD, Brigham & Women’s Hospital, Boston, do not voice much opinion on what the results mean, concluding that “the effect of these findings on the interpretation of the REDUCE-IT trial results remains unclear and will require further investigation.”
They also say that a second icosapent ethyl trial using a nonmineral oil comparator “would help resolve ongoing controversy.”
However, the authors are a mixed group; Dr. Ridker and some of his coauthors were not part of the original REDUCE-IT trial, whereas other coauthors were members of the REDUCE-IT steering committee, and one was an employee of Amarin.
Lead investigator of the REDUCE-IT trial, Deepak Bhatt, MD, also from Brigham & Women’s Hospital, who is the senior author of the current study, played down the new findings, saying they did not offer much new incremental information on mechanistic insight.
“These are small degrees of biomarker increases on an absolute level we are seeing with the placebo that would not be expected to produce harm,” Dr. Bhatt told this news organization. He also said the Circulation peer-review process had removed some of the discussion, which could lead to some “misinterpretation” of the authors’ views.
Dr. Ridker was unavailable for further comment.
The publication of this study has inevitably poured fuel on the fire regarding the controversy that has long dogged the REDUCE-IT trial, with questions about the large reduction in event rates seen with icosapent ethyl without an obvious mechanistic explanation.
‘Smoking gun’
One of the loudest critics of the study, Steve Nissen, MD, Cleveland Clinic, described the new findings as “the closest thing I’ve seen to a smoking gun in medicine for a long, long time.”
“The result of this new analysis shows that mineral oil increases virtually every inflammatory and lipid marker that they measured,” he commented.
“There are a lot of theories, but the bottom line is that something really bad happened in the mineral-oil group, which makes icosapent ethyl look efficacious. In my view, this needs to be reviewed by the FDA for consideration of removing the label claim for cardiovascular benefit.”
Other experts in the field not directly involved in the study voiced concern about these new findings, adding to calls for another trial.
In a Twitter thread on the issue, Harlan Krumholz, MD, describes the Circulation publication as “an exceptionally important article,” adding that it is “time to rethink this drug.”
“My point is ... once you know you have non-neutral comparator and the effect on risk biomarkers is far from trivial ... then you have introduced substantial uncertainty about the trial result, as conveyed by the authors ... and no one can say what would happen with a neutral comparator,” Dr. Krumholz writes.
In an accompanying editorial in Circulation, Robert Harrington, MD, professor of medicine at Stanford (Calif.) University, concludes that “the hard reality is that we are left with uncertainties and the questions raised by use of the mineral oil as placebo can only be answered by another randomized controlled trial.”
“My main points are that the chosen placebo was not inert (an essential characteristic for an active control–placebo comparison), that the biomarker data show elevations in multiple markers involved with inflammatory pathways associated with atherosclerosis, and that these data create enough uncertainty in the trial interpretation that the best recourse to answer the criticisms is to do another trial with a truly inert placebo,” Dr. Harrington said in an interview.
He added that Dr. Bhatt’s point that the changes in biomarkers are too small to really matter clinically may be right. “But for me, the uncertainties mean that I have less confidence in the true magnitude of the treatment effect than I would have if there were no changes in the inflammatory markers.”
In Circulation, the authors say it is unclear why multiple biomarkers increased over time among REDUCE-IT participants allocated to mineral oil. They note that no substantive changes in these biomarkers were observed in the placebo groups over periods of 3-5 years in other trials, including JUPITER, CIRT, CANTOS, SPIRE, and the STRENGTH trials, which evaluated a different high-dose omega-3 oil product but used corn oil as the placebo.
“The core design of REDUCE-IT does not make it possible to resolve convincingly whether any adverse effects associated with mineral oil use as a comparator may have affected clinical outcomes,” they write.
They point out that regulatory agencies evaluating REDUCE-IT estimated that approximately 3% of the net clinical benefit observed with icosapent ethyl might have been a consequence of adverse biomarker effects on LDL cholesterol and hsCRP attributable to mineral oil. But in the context of an overall 25% relative risk reduction in first events and a 30% reduction in total ischemic events observed, a potential bias of this magnitude, even if doubled in size, would be unlikely to fully attenuate the overall benefit of icosapent ethyl observed.
They add that they are not aware of a method to assess what the potential magnitude might be of a combination of the multiple effects.
New data do not change the debate
“We did a large, well-powered randomized trial, and this paper shouldn’t change anything in how that trial should be interpreted,” Dr. Bhatt said in an interview.
He claims the new biomarkers evaluated in the study are correlated with LDL and CRP, data which have already been reported and analyzed so have limited relevance.
“It’s not really independent biomarker information; this is what we would expect to see when we see small increases in LDL and CRP. So, I don’t think this new information fundamentally changes the debate,” he said.
Dr. Bhatt also pointed out that the study highlights relative increases rather than absolute increases in the biomarkers, making it seem more alarming than is actually the case.
“The paper makes it seem like that there are large increases in these other biomarkers, but the values reported are relative increases and the absolute increases were actually rather small. In many cases, the changes reported are less than the lower limit of quantification of the assay used,” he noted.
He added: “Even if one is unable to get around the placebo issue in the REDUCE-IT trial, there will always be the JELIS trial – a randomized trial with no placebo showing a 19% relative risk reduction. While the biomarker data may be interesting, what really matters in the end is clinical events. And significant reductions in two independent trials should be enough.”
Dr. Bhatt says the REDUCE-IT steering committee does not believe another trial is needed. “Maybe a different population would be good – such as primary prevention, patients without elevated triglycerides – but just repeating REDUCE- IT with a different placebo would be a waste of resources,” he commented.
But Dr. Nissen refuted Dr. Bhatt’s claims.
“These biomarkers are not in the same pathways as LDL and CRP, and these are not small increases. In the CANTOS trial, a monoclonal antibody against interleukin-1β beta showed a significant benefit. The increase in interleukin-1β now reported in REDUCE-IT is exactly the opposite of CANTOS,” he pointed out.
“The FDA did not know about these additional biomarkers when it reviewed the data on LDL and CRP. Now we have new information. It needs to be looked at again,” Dr. Nissen added.
Funding for the study was provided by Amarin Pharma. Dr. Bhatt was the lead investigator of the REDUCE-IT trial. Dr. Nissen was the lead investigator the STRENGTH trial. Further disclosures of the authors can be found in Circulation.
A version of this article first appeared on Medscape.com.
A new analysis of the REDUCE-IT study has reignited concerns that the benefit shown by the high-dose fish oil product in the study, icosapent ethyl (Vascepa, Amarin), may have been related to harms caused by the placebo mineral oil.
Results show that allocation to icosapent ethyl had minimal effects on a series of biomarkers associated with atherosclerotic disease, whereas levels of these biomarkers increased among those allocated to mineral oil.
At 12 months, the median percent increases from baseline in the mineral oil group were 1.5% for homocysteine, 2.2% for lipoprotein(a), 10.9% for oxidized low-density-lipoprotein (LDL) cholesterol, 16.2% for interleukin (IL)-6, 18.5% for lipoprotein-associated phospholipase A2, 21.9% for high-sensitivity C-reactive protein (hsCRP), and 28.9% for IL-1β. The changes were similar at 24 months. However, in the icosapent ethyl group, there were minimal changes in these biomarkers at 12 and 24 months.
The study was published online in Circulation.
The authors, led by Paul Ridker, MD, Brigham & Women’s Hospital, Boston, do not voice much opinion on what the results mean, concluding that “the effect of these findings on the interpretation of the REDUCE-IT trial results remains unclear and will require further investigation.”
They also say that a second icosapent ethyl trial using a nonmineral oil comparator “would help resolve ongoing controversy.”
However, the authors are a mixed group; Dr. Ridker and some of his coauthors were not part of the original REDUCE-IT trial, whereas other coauthors were members of the REDUCE-IT steering committee, and one was an employee of Amarin.
Lead investigator of the REDUCE-IT trial, Deepak Bhatt, MD, also from Brigham & Women’s Hospital, who is the senior author of the current study, played down the new findings, saying they did not offer much new incremental information on mechanistic insight.
“These are small degrees of biomarker increases on an absolute level we are seeing with the placebo that would not be expected to produce harm,” Dr. Bhatt told this news organization. He also said the Circulation peer-review process had removed some of the discussion, which could lead to some “misinterpretation” of the authors’ views.
Dr. Ridker was unavailable for further comment.
The publication of this study has inevitably poured fuel on the fire regarding the controversy that has long dogged the REDUCE-IT trial, with questions about the large reduction in event rates seen with icosapent ethyl without an obvious mechanistic explanation.
‘Smoking gun’
One of the loudest critics of the study, Steve Nissen, MD, Cleveland Clinic, described the new findings as “the closest thing I’ve seen to a smoking gun in medicine for a long, long time.”
“The result of this new analysis shows that mineral oil increases virtually every inflammatory and lipid marker that they measured,” he commented.
“There are a lot of theories, but the bottom line is that something really bad happened in the mineral-oil group, which makes icosapent ethyl look efficacious. In my view, this needs to be reviewed by the FDA for consideration of removing the label claim for cardiovascular benefit.”
Other experts in the field not directly involved in the study voiced concern about these new findings, adding to calls for another trial.
In a Twitter thread on the issue, Harlan Krumholz, MD, describes the Circulation publication as “an exceptionally important article,” adding that it is “time to rethink this drug.”
“My point is ... once you know you have non-neutral comparator and the effect on risk biomarkers is far from trivial ... then you have introduced substantial uncertainty about the trial result, as conveyed by the authors ... and no one can say what would happen with a neutral comparator,” Dr. Krumholz writes.
In an accompanying editorial in Circulation, Robert Harrington, MD, professor of medicine at Stanford (Calif.) University, concludes that “the hard reality is that we are left with uncertainties and the questions raised by use of the mineral oil as placebo can only be answered by another randomized controlled trial.”
“My main points are that the chosen placebo was not inert (an essential characteristic for an active control–placebo comparison), that the biomarker data show elevations in multiple markers involved with inflammatory pathways associated with atherosclerosis, and that these data create enough uncertainty in the trial interpretation that the best recourse to answer the criticisms is to do another trial with a truly inert placebo,” Dr. Harrington said in an interview.
He added that Dr. Bhatt’s point that the changes in biomarkers are too small to really matter clinically may be right. “But for me, the uncertainties mean that I have less confidence in the true magnitude of the treatment effect than I would have if there were no changes in the inflammatory markers.”
In Circulation, the authors say it is unclear why multiple biomarkers increased over time among REDUCE-IT participants allocated to mineral oil. They note that no substantive changes in these biomarkers were observed in the placebo groups over periods of 3-5 years in other trials, including JUPITER, CIRT, CANTOS, SPIRE, and the STRENGTH trials, which evaluated a different high-dose omega-3 oil product but used corn oil as the placebo.
“The core design of REDUCE-IT does not make it possible to resolve convincingly whether any adverse effects associated with mineral oil use as a comparator may have affected clinical outcomes,” they write.
They point out that regulatory agencies evaluating REDUCE-IT estimated that approximately 3% of the net clinical benefit observed with icosapent ethyl might have been a consequence of adverse biomarker effects on LDL cholesterol and hsCRP attributable to mineral oil. But in the context of an overall 25% relative risk reduction in first events and a 30% reduction in total ischemic events observed, a potential bias of this magnitude, even if doubled in size, would be unlikely to fully attenuate the overall benefit of icosapent ethyl observed.
They add that they are not aware of a method to assess what the potential magnitude might be of a combination of the multiple effects.
New data do not change the debate
“We did a large, well-powered randomized trial, and this paper shouldn’t change anything in how that trial should be interpreted,” Dr. Bhatt said in an interview.
He claims the new biomarkers evaluated in the study are correlated with LDL and CRP, data which have already been reported and analyzed so have limited relevance.
“It’s not really independent biomarker information; this is what we would expect to see when we see small increases in LDL and CRP. So, I don’t think this new information fundamentally changes the debate,” he said.
Dr. Bhatt also pointed out that the study highlights relative increases rather than absolute increases in the biomarkers, making it seem more alarming than is actually the case.
“The paper makes it seem like that there are large increases in these other biomarkers, but the values reported are relative increases and the absolute increases were actually rather small. In many cases, the changes reported are less than the lower limit of quantification of the assay used,” he noted.
He added: “Even if one is unable to get around the placebo issue in the REDUCE-IT trial, there will always be the JELIS trial – a randomized trial with no placebo showing a 19% relative risk reduction. While the biomarker data may be interesting, what really matters in the end is clinical events. And significant reductions in two independent trials should be enough.”
Dr. Bhatt says the REDUCE-IT steering committee does not believe another trial is needed. “Maybe a different population would be good – such as primary prevention, patients without elevated triglycerides – but just repeating REDUCE- IT with a different placebo would be a waste of resources,” he commented.
But Dr. Nissen refuted Dr. Bhatt’s claims.
“These biomarkers are not in the same pathways as LDL and CRP, and these are not small increases. In the CANTOS trial, a monoclonal antibody against interleukin-1β beta showed a significant benefit. The increase in interleukin-1β now reported in REDUCE-IT is exactly the opposite of CANTOS,” he pointed out.
“The FDA did not know about these additional biomarkers when it reviewed the data on LDL and CRP. Now we have new information. It needs to be looked at again,” Dr. Nissen added.
Funding for the study was provided by Amarin Pharma. Dr. Bhatt was the lead investigator of the REDUCE-IT trial. Dr. Nissen was the lead investigator the STRENGTH trial. Further disclosures of the authors can be found in Circulation.
A version of this article first appeared on Medscape.com.
A new analysis of the REDUCE-IT study has reignited concerns that the benefit shown by the high-dose fish oil product in the study, icosapent ethyl (Vascepa, Amarin), may have been related to harms caused by the placebo mineral oil.
Results show that allocation to icosapent ethyl had minimal effects on a series of biomarkers associated with atherosclerotic disease, whereas levels of these biomarkers increased among those allocated to mineral oil.
At 12 months, the median percent increases from baseline in the mineral oil group were 1.5% for homocysteine, 2.2% for lipoprotein(a), 10.9% for oxidized low-density-lipoprotein (LDL) cholesterol, 16.2% for interleukin (IL)-6, 18.5% for lipoprotein-associated phospholipase A2, 21.9% for high-sensitivity C-reactive protein (hsCRP), and 28.9% for IL-1β. The changes were similar at 24 months. However, in the icosapent ethyl group, there were minimal changes in these biomarkers at 12 and 24 months.
The study was published online in Circulation.
The authors, led by Paul Ridker, MD, Brigham & Women’s Hospital, Boston, do not voice much opinion on what the results mean, concluding that “the effect of these findings on the interpretation of the REDUCE-IT trial results remains unclear and will require further investigation.”
They also say that a second icosapent ethyl trial using a nonmineral oil comparator “would help resolve ongoing controversy.”
However, the authors are a mixed group; Dr. Ridker and some of his coauthors were not part of the original REDUCE-IT trial, whereas other coauthors were members of the REDUCE-IT steering committee, and one was an employee of Amarin.
Lead investigator of the REDUCE-IT trial, Deepak Bhatt, MD, also from Brigham & Women’s Hospital, who is the senior author of the current study, played down the new findings, saying they did not offer much new incremental information on mechanistic insight.
“These are small degrees of biomarker increases on an absolute level we are seeing with the placebo that would not be expected to produce harm,” Dr. Bhatt told this news organization. He also said the Circulation peer-review process had removed some of the discussion, which could lead to some “misinterpretation” of the authors’ views.
Dr. Ridker was unavailable for further comment.
The publication of this study has inevitably poured fuel on the fire regarding the controversy that has long dogged the REDUCE-IT trial, with questions about the large reduction in event rates seen with icosapent ethyl without an obvious mechanistic explanation.
‘Smoking gun’
One of the loudest critics of the study, Steve Nissen, MD, Cleveland Clinic, described the new findings as “the closest thing I’ve seen to a smoking gun in medicine for a long, long time.”
“The result of this new analysis shows that mineral oil increases virtually every inflammatory and lipid marker that they measured,” he commented.
“There are a lot of theories, but the bottom line is that something really bad happened in the mineral-oil group, which makes icosapent ethyl look efficacious. In my view, this needs to be reviewed by the FDA for consideration of removing the label claim for cardiovascular benefit.”
Other experts in the field not directly involved in the study voiced concern about these new findings, adding to calls for another trial.
In a Twitter thread on the issue, Harlan Krumholz, MD, describes the Circulation publication as “an exceptionally important article,” adding that it is “time to rethink this drug.”
“My point is ... once you know you have non-neutral comparator and the effect on risk biomarkers is far from trivial ... then you have introduced substantial uncertainty about the trial result, as conveyed by the authors ... and no one can say what would happen with a neutral comparator,” Dr. Krumholz writes.
In an accompanying editorial in Circulation, Robert Harrington, MD, professor of medicine at Stanford (Calif.) University, concludes that “the hard reality is that we are left with uncertainties and the questions raised by use of the mineral oil as placebo can only be answered by another randomized controlled trial.”
“My main points are that the chosen placebo was not inert (an essential characteristic for an active control–placebo comparison), that the biomarker data show elevations in multiple markers involved with inflammatory pathways associated with atherosclerosis, and that these data create enough uncertainty in the trial interpretation that the best recourse to answer the criticisms is to do another trial with a truly inert placebo,” Dr. Harrington said in an interview.
He added that Dr. Bhatt’s point that the changes in biomarkers are too small to really matter clinically may be right. “But for me, the uncertainties mean that I have less confidence in the true magnitude of the treatment effect than I would have if there were no changes in the inflammatory markers.”
In Circulation, the authors say it is unclear why multiple biomarkers increased over time among REDUCE-IT participants allocated to mineral oil. They note that no substantive changes in these biomarkers were observed in the placebo groups over periods of 3-5 years in other trials, including JUPITER, CIRT, CANTOS, SPIRE, and the STRENGTH trials, which evaluated a different high-dose omega-3 oil product but used corn oil as the placebo.
“The core design of REDUCE-IT does not make it possible to resolve convincingly whether any adverse effects associated with mineral oil use as a comparator may have affected clinical outcomes,” they write.
They point out that regulatory agencies evaluating REDUCE-IT estimated that approximately 3% of the net clinical benefit observed with icosapent ethyl might have been a consequence of adverse biomarker effects on LDL cholesterol and hsCRP attributable to mineral oil. But in the context of an overall 25% relative risk reduction in first events and a 30% reduction in total ischemic events observed, a potential bias of this magnitude, even if doubled in size, would be unlikely to fully attenuate the overall benefit of icosapent ethyl observed.
They add that they are not aware of a method to assess what the potential magnitude might be of a combination of the multiple effects.
New data do not change the debate
“We did a large, well-powered randomized trial, and this paper shouldn’t change anything in how that trial should be interpreted,” Dr. Bhatt said in an interview.
He claims the new biomarkers evaluated in the study are correlated with LDL and CRP, data which have already been reported and analyzed so have limited relevance.
“It’s not really independent biomarker information; this is what we would expect to see when we see small increases in LDL and CRP. So, I don’t think this new information fundamentally changes the debate,” he said.
Dr. Bhatt also pointed out that the study highlights relative increases rather than absolute increases in the biomarkers, making it seem more alarming than is actually the case.
“The paper makes it seem like that there are large increases in these other biomarkers, but the values reported are relative increases and the absolute increases were actually rather small. In many cases, the changes reported are less than the lower limit of quantification of the assay used,” he noted.
He added: “Even if one is unable to get around the placebo issue in the REDUCE-IT trial, there will always be the JELIS trial – a randomized trial with no placebo showing a 19% relative risk reduction. While the biomarker data may be interesting, what really matters in the end is clinical events. And significant reductions in two independent trials should be enough.”
Dr. Bhatt says the REDUCE-IT steering committee does not believe another trial is needed. “Maybe a different population would be good – such as primary prevention, patients without elevated triglycerides – but just repeating REDUCE- IT with a different placebo would be a waste of resources,” he commented.
But Dr. Nissen refuted Dr. Bhatt’s claims.
“These biomarkers are not in the same pathways as LDL and CRP, and these are not small increases. In the CANTOS trial, a monoclonal antibody against interleukin-1β beta showed a significant benefit. The increase in interleukin-1β now reported in REDUCE-IT is exactly the opposite of CANTOS,” he pointed out.
“The FDA did not know about these additional biomarkers when it reviewed the data on LDL and CRP. Now we have new information. It needs to be looked at again,” Dr. Nissen added.
Funding for the study was provided by Amarin Pharma. Dr. Bhatt was the lead investigator of the REDUCE-IT trial. Dr. Nissen was the lead investigator the STRENGTH trial. Further disclosures of the authors can be found in Circulation.
A version of this article first appeared on Medscape.com.
FROM CIRCULATION
Nordic walking bests other workouts on functional outcome in CVD
Nordic walking was significantly better at improving functional capacity than were moderate- to vigorous-intensity continuous training and high-intensity interval training (HIIT) in a single-center randomized controlled trial.
Participants who did Nordic walking saw better improvements in functional capacity, measured via the 6-minute walk test distances, than did individuals doing either of the other exercise strategies (interaction effect, P = .010).
From baseline to 26 weeks, the average changes in 6-minute walk test distance were 55.6 m and 59.9 m for moderate- to vigorous-intensity continuous training and HIIT, respectively, but 94.2 m in the Nordic walking group, reported Tasuku Terada, PhD, University of Ottawa Heart Institute, Ontario, and colleagues.
Previous research looked at these results at the end of a 12-week supervised exercise intervention and showed that although all three strategies were safe and had positive effects on physical and mental health in these patients, Nordic walking had a better effect in raising the 6-minute walk test scores than did moderate- to vigorous-intensity continuous training and HIIT, the researchers noted.
“This study is a follow-up on the previous study to show that Nordic walking had greater sustained effects even after the observation phase,” from 12 to 26 weeks, Dr. Terada said in an interview.
“Exercise is a medicine to improve the health of patients, but unfortunately, sometimes it is not as often utilized,” Dr. Terada told this news organization.
Giving patients additional exercise modalities is beneficial because not everyone likes HIIT workouts or long continuous walking, Dr. Terada said. “So, if that’s the case, we can recommend Nordic walking as another type of exercise and expect a similar or good impact in functional capacity.”
The results were published online in the Canadian Journal of Cardiology.
“I think it honestly supports the idea that, as many other studies show, physical activity and exercise improve functional capacity no matter how you measure it and have beneficial effects on mental health and quality of life and particularly depression as well,” Carl “Chip” Lavie, MD, University of Queensland, New Orleans, who coauthored an editorial accompanying the publication, said in an interview.
“Clinicians need to get patients to do the type of exercise that they are going to do. A lot of people ask what’s the best exercise, and the best exercise is one that the person is going to do,” Dr. Lavie said.
Nordic walking is an enhanced form of walking that engages the upper and lower body musculatures, noted Dr. Lavie.
“With regard to Nordic walking, I think that now adds an additional option that many people wouldn’t have thought about. For many of the patients that have issues that are musculoskeletal, issues with posture, gait, or balance, using the poles can be a way to allow them to walk much better and increase their speed, and as they do that, they become fitter,” Dr. Lavie continued.
Moreover, these findings support the use of Nordic walking in cardiac rehabilitation programs, the editorialists noted.
Cardiac rehabilitation
The study examined patients with coronary artery disease who underwent cardiac revascularization. They were then referred by their physicians to cardiac rehabilitation.
Participants were randomly assigned to one of the following intervention groups: Nordic walking (n = 30), moderate- to vigorous-intensity continuous training (n = 27), and HIIT (n = 29) for a 12-week period. There was then an additional 14-week observation period after the exercise program. Mean age was 60 years across the intervention groups.
The research team analyzed the extent of participants’ depression with Beck Depression Inventory–II, quality of life with Short Form–36 and HeartQoL, and functional capacity with a 6-minute walk test. They assessed functional capacity, depression, and quality of life at baseline, 12 weeks, and 26 weeks.
Using linear mixed models with extended measures, the study authors evaluated sustained effects, which were between week 12 and week 26, and prolonged effects, which were between baseline and week 26.
From baseline to 26 weeks, participants saw significantly better outcomes in quality of life, depression symptoms, and 6-minute walk test (P < .05).
Physical quality of life and 6-minute walk test distance rose significantly between weeks 12 and 26 (P < .05).
Notably, at week 26, all training groups achieved the minimal clinical threshold difference of 54 m, although participants in the Nordic walking cohort demonstrated significantly greater improvement in outcomes.
Other data indicated the following:
- From baseline to week 12, physical activity levels rose significantly, and this improvement was sustained through the observation period.
- During the observation period, mental component summary significantly declined while physical component summary outcomes improved.
- After completion of cardiac rehabilitation, functional capacity continued to increase significantly.
- Moderate- to vigorous-intensity continuous training, HIIT, and Nordic walking had positive and significant prolonged effects on depression symptoms and general and disease-specific quality of life, with no differences in the extent of improvements between exercise types.
Some limitations of the study include the fact that women comprised a small portion of the study group, which limits the generalizability of these data, the cohort was recruited from a single medical facility, and there was a short follow-up time, the researchers noted.
“Further research is warranted to investigate the efficacy and integration of Nordic walking into home-based exercise after supervised cardiac rehabilitation for maintenance of physical and mental health,” the editorialists concluded.
Dr. Terada, Dr. Lavie, and Dr. Taylor reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Nordic walking was significantly better at improving functional capacity than were moderate- to vigorous-intensity continuous training and high-intensity interval training (HIIT) in a single-center randomized controlled trial.
Participants who did Nordic walking saw better improvements in functional capacity, measured via the 6-minute walk test distances, than did individuals doing either of the other exercise strategies (interaction effect, P = .010).
From baseline to 26 weeks, the average changes in 6-minute walk test distance were 55.6 m and 59.9 m for moderate- to vigorous-intensity continuous training and HIIT, respectively, but 94.2 m in the Nordic walking group, reported Tasuku Terada, PhD, University of Ottawa Heart Institute, Ontario, and colleagues.
Previous research looked at these results at the end of a 12-week supervised exercise intervention and showed that although all three strategies were safe and had positive effects on physical and mental health in these patients, Nordic walking had a better effect in raising the 6-minute walk test scores than did moderate- to vigorous-intensity continuous training and HIIT, the researchers noted.
“This study is a follow-up on the previous study to show that Nordic walking had greater sustained effects even after the observation phase,” from 12 to 26 weeks, Dr. Terada said in an interview.
“Exercise is a medicine to improve the health of patients, but unfortunately, sometimes it is not as often utilized,” Dr. Terada told this news organization.
Giving patients additional exercise modalities is beneficial because not everyone likes HIIT workouts or long continuous walking, Dr. Terada said. “So, if that’s the case, we can recommend Nordic walking as another type of exercise and expect a similar or good impact in functional capacity.”
The results were published online in the Canadian Journal of Cardiology.
“I think it honestly supports the idea that, as many other studies show, physical activity and exercise improve functional capacity no matter how you measure it and have beneficial effects on mental health and quality of life and particularly depression as well,” Carl “Chip” Lavie, MD, University of Queensland, New Orleans, who coauthored an editorial accompanying the publication, said in an interview.
“Clinicians need to get patients to do the type of exercise that they are going to do. A lot of people ask what’s the best exercise, and the best exercise is one that the person is going to do,” Dr. Lavie said.
Nordic walking is an enhanced form of walking that engages the upper and lower body musculatures, noted Dr. Lavie.
“With regard to Nordic walking, I think that now adds an additional option that many people wouldn’t have thought about. For many of the patients that have issues that are musculoskeletal, issues with posture, gait, or balance, using the poles can be a way to allow them to walk much better and increase their speed, and as they do that, they become fitter,” Dr. Lavie continued.
Moreover, these findings support the use of Nordic walking in cardiac rehabilitation programs, the editorialists noted.
Cardiac rehabilitation
The study examined patients with coronary artery disease who underwent cardiac revascularization. They were then referred by their physicians to cardiac rehabilitation.
Participants were randomly assigned to one of the following intervention groups: Nordic walking (n = 30), moderate- to vigorous-intensity continuous training (n = 27), and HIIT (n = 29) for a 12-week period. There was then an additional 14-week observation period after the exercise program. Mean age was 60 years across the intervention groups.
The research team analyzed the extent of participants’ depression with Beck Depression Inventory–II, quality of life with Short Form–36 and HeartQoL, and functional capacity with a 6-minute walk test. They assessed functional capacity, depression, and quality of life at baseline, 12 weeks, and 26 weeks.
Using linear mixed models with extended measures, the study authors evaluated sustained effects, which were between week 12 and week 26, and prolonged effects, which were between baseline and week 26.
From baseline to 26 weeks, participants saw significantly better outcomes in quality of life, depression symptoms, and 6-minute walk test (P < .05).
Physical quality of life and 6-minute walk test distance rose significantly between weeks 12 and 26 (P < .05).
Notably, at week 26, all training groups achieved the minimal clinical threshold difference of 54 m, although participants in the Nordic walking cohort demonstrated significantly greater improvement in outcomes.
Other data indicated the following:
- From baseline to week 12, physical activity levels rose significantly, and this improvement was sustained through the observation period.
- During the observation period, mental component summary significantly declined while physical component summary outcomes improved.
- After completion of cardiac rehabilitation, functional capacity continued to increase significantly.
- Moderate- to vigorous-intensity continuous training, HIIT, and Nordic walking had positive and significant prolonged effects on depression symptoms and general and disease-specific quality of life, with no differences in the extent of improvements between exercise types.
Some limitations of the study include the fact that women comprised a small portion of the study group, which limits the generalizability of these data, the cohort was recruited from a single medical facility, and there was a short follow-up time, the researchers noted.
“Further research is warranted to investigate the efficacy and integration of Nordic walking into home-based exercise after supervised cardiac rehabilitation for maintenance of physical and mental health,” the editorialists concluded.
Dr. Terada, Dr. Lavie, and Dr. Taylor reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Nordic walking was significantly better at improving functional capacity than were moderate- to vigorous-intensity continuous training and high-intensity interval training (HIIT) in a single-center randomized controlled trial.
Participants who did Nordic walking saw better improvements in functional capacity, measured via the 6-minute walk test distances, than did individuals doing either of the other exercise strategies (interaction effect, P = .010).
From baseline to 26 weeks, the average changes in 6-minute walk test distance were 55.6 m and 59.9 m for moderate- to vigorous-intensity continuous training and HIIT, respectively, but 94.2 m in the Nordic walking group, reported Tasuku Terada, PhD, University of Ottawa Heart Institute, Ontario, and colleagues.
Previous research looked at these results at the end of a 12-week supervised exercise intervention and showed that although all three strategies were safe and had positive effects on physical and mental health in these patients, Nordic walking had a better effect in raising the 6-minute walk test scores than did moderate- to vigorous-intensity continuous training and HIIT, the researchers noted.
“This study is a follow-up on the previous study to show that Nordic walking had greater sustained effects even after the observation phase,” from 12 to 26 weeks, Dr. Terada said in an interview.
“Exercise is a medicine to improve the health of patients, but unfortunately, sometimes it is not as often utilized,” Dr. Terada told this news organization.
Giving patients additional exercise modalities is beneficial because not everyone likes HIIT workouts or long continuous walking, Dr. Terada said. “So, if that’s the case, we can recommend Nordic walking as another type of exercise and expect a similar or good impact in functional capacity.”
The results were published online in the Canadian Journal of Cardiology.
“I think it honestly supports the idea that, as many other studies show, physical activity and exercise improve functional capacity no matter how you measure it and have beneficial effects on mental health and quality of life and particularly depression as well,” Carl “Chip” Lavie, MD, University of Queensland, New Orleans, who coauthored an editorial accompanying the publication, said in an interview.
“Clinicians need to get patients to do the type of exercise that they are going to do. A lot of people ask what’s the best exercise, and the best exercise is one that the person is going to do,” Dr. Lavie said.
Nordic walking is an enhanced form of walking that engages the upper and lower body musculatures, noted Dr. Lavie.
“With regard to Nordic walking, I think that now adds an additional option that many people wouldn’t have thought about. For many of the patients that have issues that are musculoskeletal, issues with posture, gait, or balance, using the poles can be a way to allow them to walk much better and increase their speed, and as they do that, they become fitter,” Dr. Lavie continued.
Moreover, these findings support the use of Nordic walking in cardiac rehabilitation programs, the editorialists noted.
Cardiac rehabilitation
The study examined patients with coronary artery disease who underwent cardiac revascularization. They were then referred by their physicians to cardiac rehabilitation.
Participants were randomly assigned to one of the following intervention groups: Nordic walking (n = 30), moderate- to vigorous-intensity continuous training (n = 27), and HIIT (n = 29) for a 12-week period. There was then an additional 14-week observation period after the exercise program. Mean age was 60 years across the intervention groups.
The research team analyzed the extent of participants’ depression with Beck Depression Inventory–II, quality of life with Short Form–36 and HeartQoL, and functional capacity with a 6-minute walk test. They assessed functional capacity, depression, and quality of life at baseline, 12 weeks, and 26 weeks.
Using linear mixed models with extended measures, the study authors evaluated sustained effects, which were between week 12 and week 26, and prolonged effects, which were between baseline and week 26.
From baseline to 26 weeks, participants saw significantly better outcomes in quality of life, depression symptoms, and 6-minute walk test (P < .05).
Physical quality of life and 6-minute walk test distance rose significantly between weeks 12 and 26 (P < .05).
Notably, at week 26, all training groups achieved the minimal clinical threshold difference of 54 m, although participants in the Nordic walking cohort demonstrated significantly greater improvement in outcomes.
Other data indicated the following:
- From baseline to week 12, physical activity levels rose significantly, and this improvement was sustained through the observation period.
- During the observation period, mental component summary significantly declined while physical component summary outcomes improved.
- After completion of cardiac rehabilitation, functional capacity continued to increase significantly.
- Moderate- to vigorous-intensity continuous training, HIIT, and Nordic walking had positive and significant prolonged effects on depression symptoms and general and disease-specific quality of life, with no differences in the extent of improvements between exercise types.
Some limitations of the study include the fact that women comprised a small portion of the study group, which limits the generalizability of these data, the cohort was recruited from a single medical facility, and there was a short follow-up time, the researchers noted.
“Further research is warranted to investigate the efficacy and integration of Nordic walking into home-based exercise after supervised cardiac rehabilitation for maintenance of physical and mental health,” the editorialists concluded.
Dr. Terada, Dr. Lavie, and Dr. Taylor reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE CANADIAN JOURNAL OF CARDIOLOGY
Mobile devices ‘addictive by design’: Obesity is one of many health effects
Wireless devices, like smart phones and tablets, appear to induce compulsive or even addictive use in many individuals, leading to adverse health consequences that are likely to be curtailed only through often difficult behavior modification, according to a pediatric endocrinologist’s take on the problem.
While the summary was based in part on the analysis of 234 published papers drawn from the medical literature, the lead author, Nidhi Gupta, MD, said the data reinforce her own clinical experience.
“As a pediatric endocrinologist, the trend in smartphone-associated health disorders, such as obesity, sleep, and behavior issues, worries me,” Dr. Gupta, director of KAP Pediatric Endocrinology, Nashville, Tenn., said at the annual meeting of the Endocrine Society.
Based on her search of the medical literature, the available data raise concern. In one study she cited, for example, each hour per day of screen time was found to translate into a body mass index increase of 0.5 to 0.7 kg/m2 (P < .001).
With this type of progressive rise in BMI comes prediabetes, dyslipidemia, and other metabolic disorders associated with major health risks, including cardiovascular disease. And there are others. Dr. Gupta cited data suggesting screen time before bed disturbs sleep, which has its own set of health risks.
“When I say health, it includes physical health, mental health, and emotional health,” said Dr. Gupta.
In the U.S. and other countries with a growing obesity epidemic, lack of physical activity and unhealthy eating are widely considered the major culprits. Excessive screen time contributes to both.
“When we are engaged with our devices, we are often snacking subconsciously and not very mindful that we are making unhealthy choices,” Dr. Gupta said.
The problem is that there is a vicious circle. Compulsive use of devices follows the same loop as other types of addictive behaviors, according to Dr. Gupta. She traced overuse of wireless devices to the dopaminergic system, which is a powerful neuroendocrine-mediated process of craving, response, and reward.
Like fat, sugar, and salt, which provoke a neuroendocrine reward signal, the chimes and buzzes of a cell phone provide their own cues for reward in the form of a dopamine surge. As a result, these become the “triggers of an irresistible and irrational urge to check our device that makes the dopamine go high in our brain,” Dr. Gupta explained.
Although the vicious cycle can be thwarted by turning off the device, Dr. Gupta characterized this as “impractical” when smartphones are so vital to daily communication. Rather, Dr. Gupta advocated a program of moderation, reserving the phone for useful tasks without succumbing to the siren song of apps that waste time.
The most conspicuous culprit is social media, which Dr. Gupta considers to be among the most Pavlovian triggers of cell phone addiction. However, she acknowledged that participation in social media has its justifications.
“I, myself, use social media for my own branding and marketing,” Dr. Gupta said.
The problem that users have is distinguishing between screen time that does and does not have value, according to Dr. Gupta. She indicated that many of those overusing their smart devices are being driven by the dopaminergic reward system, which is generally divorced from the real goals of life, such as personal satisfaction and activity that is rewarding monetarily or in other ways.
“I am not asking for these devices to be thrown out the window. I am advocating for moderation, balance, and real-life engagement,” Dr. Gupta said at the meeting, held in Atlanta and virtually.
She outlined a long list of practical suggestions, including turning off the alarms, chimes, and messages that engage the user into the vicious dopaminergic-reward system loop. She suggested mindfulness so that the user can distinguish between valuable device use and activity that is simply procrastination.
“The devices are designed to be addictive. They are designed to manipulate our brain,” she said. “Eliminate the reward. Let’s try to make our devices boring, unappealing, or enticing so that they only work as tools.”
The medical literature is filled with data that support the potential harms of excessive screen use, leading many others to make some of the same points. In 2017, Thomas N. Robinson, MD, professor of child health at Stanford (Calif.) University, reviewed data showing an association between screen media exposure and obesity in children and adolescents.
“This is an area crying out for more research,” Dr. Robinson said in an interview. The problem of screen time, sedentary behavior, and weight gain has been an issue since the television was invented, which was the point he made in his 2017 paper, but he agreed that the problem is only getting worse.
“Digital technology has become ubiquitous, touching nearly every aspect of people’s lives,” he said. Yet, as evidence grows that overuse of this technology can be harmful, it is creating a problem without a clear solution.
“There are few data about the efficacy of specific strategies to reduce harmful impacts of digital screen use,” he said.
While some of the solutions that Dr. Gupta described make sense, they are more easily described than executed. The dopaminergic reward system is strong and largely experienced subconsciously. Recruiting patients to recognize that dopaminergic rewards are not rewards in any true sense is already a challenge. Enlisting patients to take the difficult steps to avoid the behavioral cues might be even more difficult.
Dr. Gupta and Dr. Robinson report no potential conflicts of interest.
Wireless devices, like smart phones and tablets, appear to induce compulsive or even addictive use in many individuals, leading to adverse health consequences that are likely to be curtailed only through often difficult behavior modification, according to a pediatric endocrinologist’s take on the problem.
While the summary was based in part on the analysis of 234 published papers drawn from the medical literature, the lead author, Nidhi Gupta, MD, said the data reinforce her own clinical experience.
“As a pediatric endocrinologist, the trend in smartphone-associated health disorders, such as obesity, sleep, and behavior issues, worries me,” Dr. Gupta, director of KAP Pediatric Endocrinology, Nashville, Tenn., said at the annual meeting of the Endocrine Society.
Based on her search of the medical literature, the available data raise concern. In one study she cited, for example, each hour per day of screen time was found to translate into a body mass index increase of 0.5 to 0.7 kg/m2 (P < .001).
With this type of progressive rise in BMI comes prediabetes, dyslipidemia, and other metabolic disorders associated with major health risks, including cardiovascular disease. And there are others. Dr. Gupta cited data suggesting screen time before bed disturbs sleep, which has its own set of health risks.
“When I say health, it includes physical health, mental health, and emotional health,” said Dr. Gupta.
In the U.S. and other countries with a growing obesity epidemic, lack of physical activity and unhealthy eating are widely considered the major culprits. Excessive screen time contributes to both.
“When we are engaged with our devices, we are often snacking subconsciously and not very mindful that we are making unhealthy choices,” Dr. Gupta said.
The problem is that there is a vicious circle. Compulsive use of devices follows the same loop as other types of addictive behaviors, according to Dr. Gupta. She traced overuse of wireless devices to the dopaminergic system, which is a powerful neuroendocrine-mediated process of craving, response, and reward.
Like fat, sugar, and salt, which provoke a neuroendocrine reward signal, the chimes and buzzes of a cell phone provide their own cues for reward in the form of a dopamine surge. As a result, these become the “triggers of an irresistible and irrational urge to check our device that makes the dopamine go high in our brain,” Dr. Gupta explained.
Although the vicious cycle can be thwarted by turning off the device, Dr. Gupta characterized this as “impractical” when smartphones are so vital to daily communication. Rather, Dr. Gupta advocated a program of moderation, reserving the phone for useful tasks without succumbing to the siren song of apps that waste time.
The most conspicuous culprit is social media, which Dr. Gupta considers to be among the most Pavlovian triggers of cell phone addiction. However, she acknowledged that participation in social media has its justifications.
“I, myself, use social media for my own branding and marketing,” Dr. Gupta said.
The problem that users have is distinguishing between screen time that does and does not have value, according to Dr. Gupta. She indicated that many of those overusing their smart devices are being driven by the dopaminergic reward system, which is generally divorced from the real goals of life, such as personal satisfaction and activity that is rewarding monetarily or in other ways.
“I am not asking for these devices to be thrown out the window. I am advocating for moderation, balance, and real-life engagement,” Dr. Gupta said at the meeting, held in Atlanta and virtually.
She outlined a long list of practical suggestions, including turning off the alarms, chimes, and messages that engage the user into the vicious dopaminergic-reward system loop. She suggested mindfulness so that the user can distinguish between valuable device use and activity that is simply procrastination.
“The devices are designed to be addictive. They are designed to manipulate our brain,” she said. “Eliminate the reward. Let’s try to make our devices boring, unappealing, or enticing so that they only work as tools.”
The medical literature is filled with data that support the potential harms of excessive screen use, leading many others to make some of the same points. In 2017, Thomas N. Robinson, MD, professor of child health at Stanford (Calif.) University, reviewed data showing an association between screen media exposure and obesity in children and adolescents.
“This is an area crying out for more research,” Dr. Robinson said in an interview. The problem of screen time, sedentary behavior, and weight gain has been an issue since the television was invented, which was the point he made in his 2017 paper, but he agreed that the problem is only getting worse.
“Digital technology has become ubiquitous, touching nearly every aspect of people’s lives,” he said. Yet, as evidence grows that overuse of this technology can be harmful, it is creating a problem without a clear solution.
“There are few data about the efficacy of specific strategies to reduce harmful impacts of digital screen use,” he said.
While some of the solutions that Dr. Gupta described make sense, they are more easily described than executed. The dopaminergic reward system is strong and largely experienced subconsciously. Recruiting patients to recognize that dopaminergic rewards are not rewards in any true sense is already a challenge. Enlisting patients to take the difficult steps to avoid the behavioral cues might be even more difficult.
Dr. Gupta and Dr. Robinson report no potential conflicts of interest.
Wireless devices, like smart phones and tablets, appear to induce compulsive or even addictive use in many individuals, leading to adverse health consequences that are likely to be curtailed only through often difficult behavior modification, according to a pediatric endocrinologist’s take on the problem.
While the summary was based in part on the analysis of 234 published papers drawn from the medical literature, the lead author, Nidhi Gupta, MD, said the data reinforce her own clinical experience.
“As a pediatric endocrinologist, the trend in smartphone-associated health disorders, such as obesity, sleep, and behavior issues, worries me,” Dr. Gupta, director of KAP Pediatric Endocrinology, Nashville, Tenn., said at the annual meeting of the Endocrine Society.
Based on her search of the medical literature, the available data raise concern. In one study she cited, for example, each hour per day of screen time was found to translate into a body mass index increase of 0.5 to 0.7 kg/m2 (P < .001).
With this type of progressive rise in BMI comes prediabetes, dyslipidemia, and other metabolic disorders associated with major health risks, including cardiovascular disease. And there are others. Dr. Gupta cited data suggesting screen time before bed disturbs sleep, which has its own set of health risks.
“When I say health, it includes physical health, mental health, and emotional health,” said Dr. Gupta.
In the U.S. and other countries with a growing obesity epidemic, lack of physical activity and unhealthy eating are widely considered the major culprits. Excessive screen time contributes to both.
“When we are engaged with our devices, we are often snacking subconsciously and not very mindful that we are making unhealthy choices,” Dr. Gupta said.
The problem is that there is a vicious circle. Compulsive use of devices follows the same loop as other types of addictive behaviors, according to Dr. Gupta. She traced overuse of wireless devices to the dopaminergic system, which is a powerful neuroendocrine-mediated process of craving, response, and reward.
Like fat, sugar, and salt, which provoke a neuroendocrine reward signal, the chimes and buzzes of a cell phone provide their own cues for reward in the form of a dopamine surge. As a result, these become the “triggers of an irresistible and irrational urge to check our device that makes the dopamine go high in our brain,” Dr. Gupta explained.
Although the vicious cycle can be thwarted by turning off the device, Dr. Gupta characterized this as “impractical” when smartphones are so vital to daily communication. Rather, Dr. Gupta advocated a program of moderation, reserving the phone for useful tasks without succumbing to the siren song of apps that waste time.
The most conspicuous culprit is social media, which Dr. Gupta considers to be among the most Pavlovian triggers of cell phone addiction. However, she acknowledged that participation in social media has its justifications.
“I, myself, use social media for my own branding and marketing,” Dr. Gupta said.
The problem that users have is distinguishing between screen time that does and does not have value, according to Dr. Gupta. She indicated that many of those overusing their smart devices are being driven by the dopaminergic reward system, which is generally divorced from the real goals of life, such as personal satisfaction and activity that is rewarding monetarily or in other ways.
“I am not asking for these devices to be thrown out the window. I am advocating for moderation, balance, and real-life engagement,” Dr. Gupta said at the meeting, held in Atlanta and virtually.
She outlined a long list of practical suggestions, including turning off the alarms, chimes, and messages that engage the user into the vicious dopaminergic-reward system loop. She suggested mindfulness so that the user can distinguish between valuable device use and activity that is simply procrastination.
“The devices are designed to be addictive. They are designed to manipulate our brain,” she said. “Eliminate the reward. Let’s try to make our devices boring, unappealing, or enticing so that they only work as tools.”
The medical literature is filled with data that support the potential harms of excessive screen use, leading many others to make some of the same points. In 2017, Thomas N. Robinson, MD, professor of child health at Stanford (Calif.) University, reviewed data showing an association between screen media exposure and obesity in children and adolescents.
“This is an area crying out for more research,” Dr. Robinson said in an interview. The problem of screen time, sedentary behavior, and weight gain has been an issue since the television was invented, which was the point he made in his 2017 paper, but he agreed that the problem is only getting worse.
“Digital technology has become ubiquitous, touching nearly every aspect of people’s lives,” he said. Yet, as evidence grows that overuse of this technology can be harmful, it is creating a problem without a clear solution.
“There are few data about the efficacy of specific strategies to reduce harmful impacts of digital screen use,” he said.
While some of the solutions that Dr. Gupta described make sense, they are more easily described than executed. The dopaminergic reward system is strong and largely experienced subconsciously. Recruiting patients to recognize that dopaminergic rewards are not rewards in any true sense is already a challenge. Enlisting patients to take the difficult steps to avoid the behavioral cues might be even more difficult.
Dr. Gupta and Dr. Robinson report no potential conflicts of interest.
FROM ENDO 2022
Low-carb, high-fat diet improves A1c, reduces liver fat
LONDON – A low-carbohydrate, high-fat (LCHF) diet reduced the progression of nonalcoholic fatty liver disease (NAFLD), and despite no calorie restriction, participants with both NAFLD and type 2 diabetes lost 5.8% of their body weight, according to a randomized controlled study.
“Based on these results, the LCHF diet may be recommended to people with NAFLD and type 2 diabetes,” said Camilla Dalby Hansen, MD, department of gastroenterology and hepatology, Odense University Hospital, Denmark, who presented the data at the International Liver Congress (ILC) 2022.
“Basically, if you have fat in your liver, you will benefit from eating fat,” she said.
The LCHF diet was compared with a low-fat, high-carbohydrate diet more typically followed for these conditions. The low-fat diet was also found to reduce the progression of NAFLD, but to a lesser extent than the LCHF diet.
Dr. Dalby Hansen called their study one of the most extensive investigations of the LCHF diet in patients with type 2 diabetes and fatty liver disease.
“Combining this [reduction in NAFLD score] with the huge weight loss, the lower HbA1c [blood sugar], the lowering of blood pressure in women, the rise in HDL levels, and reduction in triglycerides – all in all, this diet is very promising,” she said.
Stephen Harrison, MD, visiting professor, University of Oxford, United Kingdom, medical director of Pinnacle Clinical Research and president of Summit Clinical Research, San Antonio, commended Dr. Dalby Hansen on her methodology, which included before-and-after liver biopsies. “It’s a heinous effort to do paired liver biopsies in a lifestyle modification trial. That’s huge.”
“This study tells me that the way we manage patients doesn’t change – it is still lifestyle modification,” said Dr. Harrison, who was not involved with the study. “It’s eat less [rather] than more. It’s exercise and try to lose weight. In the long term, we give patients benefit, and we show that the disease has improved, and we offer something that means they can maintain a healthy life.”
He added that the relatively small and short trial was informative.
“They improved the NAFLD activity score [NAS],” he said. “I don’t know by how much. There was no change in fibrosis, but we wouldn’t expect this at 6 months.”
“It’s provocative work, and it gives us healthy information about how we can help manage our patients from a lifestyle perspective,” he concluded.
‘Do not lose weight. Eat until you are full’
In the study, 110 participants with type 2 diabetes and NAFLD, aged 18-78 years, were allocated to the LCHF diet, and 55 were allocated to the low-fat diet for 6 months.
The researchers performed liver biopsies at baseline and 6 months, which were blinded for scoring.
Participants had ongoing dietitian consultations, with follow-up visits at 3 and 6 months. Compliance was reported continuously through an online food diary platform.
The primary endpoint was change in glycemic control as measured by A1c level over 6 months. The secondary endpoints comprised the proportion of participants with changes in the NAS of at least 2 points over 6 months. Both these measures were compared between the two dietary groups.
The two groups were matched at baseline, with a mean age of 55-57 years, 58% were women, 89% with metabolic syndrome, and a mean BMI 34 kg/m2.
In baseline liver disease, F1 level fibrosis was the most common (58%), followed by hepatic steatosis (S1, 47%; S2, 32%), with a median NAS of 3, and 19% had nonalcoholic steatohepatitis.
The special thing about these diets was that participants were told to “not lose weight, but eat until you are full,” remarked Dr. Dalby Hansen.
Those on the LCHF diet consumed an average of 61% energy from fat, 13% from carbohydrates, and 23% from protein, compared with the low-fat diet, which comprised an average of 29% energy from fat, 46% from carbohydrates, and 21% from protein.
“It’s a lot of fat and corresponds to a quarter of a liter of olive oil per day,” said Dr. Dalby Hansen. “They really had to change their mindset a lot, because it was difficult for them to start eating all these fats, especially since we’ve all been told for decades that it isn’t good. But we supported them, and they got into it.”
The LCHF diet was primarily comprised of unsaturated fats – for example, avocado, oil, nuts, and seeds – but also included saturated fats, such as cheese, cream, and high-fat dairy products. Participants were free to eat unsaturated and saturated fats, but Dr. Dalby Hansen and her team advised participants that “good” unsaturated fats were preferable.
“Also, this diet contained vegetables but no bread, no potatoes, no rice, and no pasta. It was low in carbohydrates, below 20%,” she added.
Improved glycemic control, reduced liver fat
“We found that the LCHF diet improved diabetes control, it reduced the fat in the liver, and, even though they’re eating as many calories as they were used to until they were full, they lost 5.8% of body weight,” said Dr. Dalby Hansen in reporting the results. Participants in the low-fat group lost only 1.8% of body weight.
However, mean calorie intake dropped in both groups, by –2.2% in the LCHF group and –8.7% in the low-fat group.
“The LCHF diet improved the primary outcome of A1c by 9.5 mmol/mol, which is similar to some anti-diabetic medications, such as DPP-4 inhibitors and SGLT2 inhibitors,” she said.
The low-fat group reduced A1c by 3.4 mmol/mol, resulting in a between-group difference of 6.1 mmol/mol.
“Upon follow-up of 3 months, after stopping the diets, on average the participants in both groups returned their HbA1c levels to nearly baseline values,” she said. Results were adjusted for weight loss and baseline values.
Both diets also improved the NAS. The proportion of participants who improved their NAS score by 2 or more points was 22% in the LCHF group versus 17% in the low-fat group (P = 0.58). Additionally, in the LCHF group, 70% of participants improved their score by 1 or more points, compared with 49% in the low-fat group and fewer in the LCHF group experienced a worsening of their score (1% vs. 23%, respectively).
One participant on LCHF had high triglycerides of 12 mmol/L after 3 months. Overall, the low-density lipoprotein increased marginally by 0.2 mmol per liter in the high-fat group, said Dr. Dalby Hansen.
Dr. Dalby Hansen noted some limitations. The findings might not be applicable in more severe NAFLD, dietary assessment relied on self-reporting, no food was provided, and participants had to cook themselves. It was also an open-label study because of the nature of the intervention.
Some hope for more sustainable dieting
Many diets are difficult to adhere to, remarked Dr. Dalby Hansen. “We thought this [diet] might be easier to comply with in the longer term, and we hope that these results might provide patients with more options.”
She added that most people who started the diet adapted and complied with it. “However, it might not be for everyone, but I think we can say that if people try, and it fits into their lives, then they go for it.”
However, “it is not about going out and eating whatever fat and how much of it you want. It’s important that you cut the carbohydrates too,” she said. “With this approach, we really saw amazing results.”
Dr. Dalby Hansen added that having various diets available, including the LCHF one, meant that as clinicians they could empower patients to take control of their metabolic health.
“We can ask them directly, ‘What would fit into their life?’” she said. “We know that one size does not fit at all, and I believe that if we could engage patients more, then they can take control of their own situation.”
Asked whether these findings were enough to change guidelines, Zobair Younossi, MD, professor and chairman, department of medicine, Inova Fairfax Medical Campus, Falls Church, Va., remarked that it was the sugar at work here.
“Dietary fat – it’s not the same as fat in the liver, and this diet has more to do with the sugar levels,” he said.
“I’m always reluctant to take results from a short-term study without long-term follow-up,” Dr. Younossi said. “I want to know will patients live longer, and long-term data are needed for this. Until I have that strong evidence that outcomes are going to change, or at least some sign that the outcome is going to change, it is too early to change any guidelines.”
Dr. Dalby Hansen reports no relevant financial relationships. Dr. Harrison reported financial relationships with numerous pharmaceutical companies. Dr. Younossi reports the following financial relationships: research funds and/or consultant to Abbott, Allergan, Bristol Myers Squibb, Echosens, Genfit, Gilead Sciences, Intercept, Madrigal, Merck, and Novo Nordisk.
A version of this article first appeared on Medscape.com.
LONDON – A low-carbohydrate, high-fat (LCHF) diet reduced the progression of nonalcoholic fatty liver disease (NAFLD), and despite no calorie restriction, participants with both NAFLD and type 2 diabetes lost 5.8% of their body weight, according to a randomized controlled study.
“Based on these results, the LCHF diet may be recommended to people with NAFLD and type 2 diabetes,” said Camilla Dalby Hansen, MD, department of gastroenterology and hepatology, Odense University Hospital, Denmark, who presented the data at the International Liver Congress (ILC) 2022.
“Basically, if you have fat in your liver, you will benefit from eating fat,” she said.
The LCHF diet was compared with a low-fat, high-carbohydrate diet more typically followed for these conditions. The low-fat diet was also found to reduce the progression of NAFLD, but to a lesser extent than the LCHF diet.
Dr. Dalby Hansen called their study one of the most extensive investigations of the LCHF diet in patients with type 2 diabetes and fatty liver disease.
“Combining this [reduction in NAFLD score] with the huge weight loss, the lower HbA1c [blood sugar], the lowering of blood pressure in women, the rise in HDL levels, and reduction in triglycerides – all in all, this diet is very promising,” she said.
Stephen Harrison, MD, visiting professor, University of Oxford, United Kingdom, medical director of Pinnacle Clinical Research and president of Summit Clinical Research, San Antonio, commended Dr. Dalby Hansen on her methodology, which included before-and-after liver biopsies. “It’s a heinous effort to do paired liver biopsies in a lifestyle modification trial. That’s huge.”
“This study tells me that the way we manage patients doesn’t change – it is still lifestyle modification,” said Dr. Harrison, who was not involved with the study. “It’s eat less [rather] than more. It’s exercise and try to lose weight. In the long term, we give patients benefit, and we show that the disease has improved, and we offer something that means they can maintain a healthy life.”
He added that the relatively small and short trial was informative.
“They improved the NAFLD activity score [NAS],” he said. “I don’t know by how much. There was no change in fibrosis, but we wouldn’t expect this at 6 months.”
“It’s provocative work, and it gives us healthy information about how we can help manage our patients from a lifestyle perspective,” he concluded.
‘Do not lose weight. Eat until you are full’
In the study, 110 participants with type 2 diabetes and NAFLD, aged 18-78 years, were allocated to the LCHF diet, and 55 were allocated to the low-fat diet for 6 months.
The researchers performed liver biopsies at baseline and 6 months, which were blinded for scoring.
Participants had ongoing dietitian consultations, with follow-up visits at 3 and 6 months. Compliance was reported continuously through an online food diary platform.
The primary endpoint was change in glycemic control as measured by A1c level over 6 months. The secondary endpoints comprised the proportion of participants with changes in the NAS of at least 2 points over 6 months. Both these measures were compared between the two dietary groups.
The two groups were matched at baseline, with a mean age of 55-57 years, 58% were women, 89% with metabolic syndrome, and a mean BMI 34 kg/m2.
In baseline liver disease, F1 level fibrosis was the most common (58%), followed by hepatic steatosis (S1, 47%; S2, 32%), with a median NAS of 3, and 19% had nonalcoholic steatohepatitis.
The special thing about these diets was that participants were told to “not lose weight, but eat until you are full,” remarked Dr. Dalby Hansen.
Those on the LCHF diet consumed an average of 61% energy from fat, 13% from carbohydrates, and 23% from protein, compared with the low-fat diet, which comprised an average of 29% energy from fat, 46% from carbohydrates, and 21% from protein.
“It’s a lot of fat and corresponds to a quarter of a liter of olive oil per day,” said Dr. Dalby Hansen. “They really had to change their mindset a lot, because it was difficult for them to start eating all these fats, especially since we’ve all been told for decades that it isn’t good. But we supported them, and they got into it.”
The LCHF diet was primarily comprised of unsaturated fats – for example, avocado, oil, nuts, and seeds – but also included saturated fats, such as cheese, cream, and high-fat dairy products. Participants were free to eat unsaturated and saturated fats, but Dr. Dalby Hansen and her team advised participants that “good” unsaturated fats were preferable.
“Also, this diet contained vegetables but no bread, no potatoes, no rice, and no pasta. It was low in carbohydrates, below 20%,” she added.
Improved glycemic control, reduced liver fat
“We found that the LCHF diet improved diabetes control, it reduced the fat in the liver, and, even though they’re eating as many calories as they were used to until they were full, they lost 5.8% of body weight,” said Dr. Dalby Hansen in reporting the results. Participants in the low-fat group lost only 1.8% of body weight.
However, mean calorie intake dropped in both groups, by –2.2% in the LCHF group and –8.7% in the low-fat group.
“The LCHF diet improved the primary outcome of A1c by 9.5 mmol/mol, which is similar to some anti-diabetic medications, such as DPP-4 inhibitors and SGLT2 inhibitors,” she said.
The low-fat group reduced A1c by 3.4 mmol/mol, resulting in a between-group difference of 6.1 mmol/mol.
“Upon follow-up of 3 months, after stopping the diets, on average the participants in both groups returned their HbA1c levels to nearly baseline values,” she said. Results were adjusted for weight loss and baseline values.
Both diets also improved the NAS. The proportion of participants who improved their NAS score by 2 or more points was 22% in the LCHF group versus 17% in the low-fat group (P = 0.58). Additionally, in the LCHF group, 70% of participants improved their score by 1 or more points, compared with 49% in the low-fat group and fewer in the LCHF group experienced a worsening of their score (1% vs. 23%, respectively).
One participant on LCHF had high triglycerides of 12 mmol/L after 3 months. Overall, the low-density lipoprotein increased marginally by 0.2 mmol per liter in the high-fat group, said Dr. Dalby Hansen.
Dr. Dalby Hansen noted some limitations. The findings might not be applicable in more severe NAFLD, dietary assessment relied on self-reporting, no food was provided, and participants had to cook themselves. It was also an open-label study because of the nature of the intervention.
Some hope for more sustainable dieting
Many diets are difficult to adhere to, remarked Dr. Dalby Hansen. “We thought this [diet] might be easier to comply with in the longer term, and we hope that these results might provide patients with more options.”
She added that most people who started the diet adapted and complied with it. “However, it might not be for everyone, but I think we can say that if people try, and it fits into their lives, then they go for it.”
However, “it is not about going out and eating whatever fat and how much of it you want. It’s important that you cut the carbohydrates too,” she said. “With this approach, we really saw amazing results.”
Dr. Dalby Hansen added that having various diets available, including the LCHF one, meant that as clinicians they could empower patients to take control of their metabolic health.
“We can ask them directly, ‘What would fit into their life?’” she said. “We know that one size does not fit at all, and I believe that if we could engage patients more, then they can take control of their own situation.”
Asked whether these findings were enough to change guidelines, Zobair Younossi, MD, professor and chairman, department of medicine, Inova Fairfax Medical Campus, Falls Church, Va., remarked that it was the sugar at work here.
“Dietary fat – it’s not the same as fat in the liver, and this diet has more to do with the sugar levels,” he said.
“I’m always reluctant to take results from a short-term study without long-term follow-up,” Dr. Younossi said. “I want to know will patients live longer, and long-term data are needed for this. Until I have that strong evidence that outcomes are going to change, or at least some sign that the outcome is going to change, it is too early to change any guidelines.”
Dr. Dalby Hansen reports no relevant financial relationships. Dr. Harrison reported financial relationships with numerous pharmaceutical companies. Dr. Younossi reports the following financial relationships: research funds and/or consultant to Abbott, Allergan, Bristol Myers Squibb, Echosens, Genfit, Gilead Sciences, Intercept, Madrigal, Merck, and Novo Nordisk.
A version of this article first appeared on Medscape.com.
LONDON – A low-carbohydrate, high-fat (LCHF) diet reduced the progression of nonalcoholic fatty liver disease (NAFLD), and despite no calorie restriction, participants with both NAFLD and type 2 diabetes lost 5.8% of their body weight, according to a randomized controlled study.
“Based on these results, the LCHF diet may be recommended to people with NAFLD and type 2 diabetes,” said Camilla Dalby Hansen, MD, department of gastroenterology and hepatology, Odense University Hospital, Denmark, who presented the data at the International Liver Congress (ILC) 2022.
“Basically, if you have fat in your liver, you will benefit from eating fat,” she said.
The LCHF diet was compared with a low-fat, high-carbohydrate diet more typically followed for these conditions. The low-fat diet was also found to reduce the progression of NAFLD, but to a lesser extent than the LCHF diet.
Dr. Dalby Hansen called their study one of the most extensive investigations of the LCHF diet in patients with type 2 diabetes and fatty liver disease.
“Combining this [reduction in NAFLD score] with the huge weight loss, the lower HbA1c [blood sugar], the lowering of blood pressure in women, the rise in HDL levels, and reduction in triglycerides – all in all, this diet is very promising,” she said.
Stephen Harrison, MD, visiting professor, University of Oxford, United Kingdom, medical director of Pinnacle Clinical Research and president of Summit Clinical Research, San Antonio, commended Dr. Dalby Hansen on her methodology, which included before-and-after liver biopsies. “It’s a heinous effort to do paired liver biopsies in a lifestyle modification trial. That’s huge.”
“This study tells me that the way we manage patients doesn’t change – it is still lifestyle modification,” said Dr. Harrison, who was not involved with the study. “It’s eat less [rather] than more. It’s exercise and try to lose weight. In the long term, we give patients benefit, and we show that the disease has improved, and we offer something that means they can maintain a healthy life.”
He added that the relatively small and short trial was informative.
“They improved the NAFLD activity score [NAS],” he said. “I don’t know by how much. There was no change in fibrosis, but we wouldn’t expect this at 6 months.”
“It’s provocative work, and it gives us healthy information about how we can help manage our patients from a lifestyle perspective,” he concluded.
‘Do not lose weight. Eat until you are full’
In the study, 110 participants with type 2 diabetes and NAFLD, aged 18-78 years, were allocated to the LCHF diet, and 55 were allocated to the low-fat diet for 6 months.
The researchers performed liver biopsies at baseline and 6 months, which were blinded for scoring.
Participants had ongoing dietitian consultations, with follow-up visits at 3 and 6 months. Compliance was reported continuously through an online food diary platform.
The primary endpoint was change in glycemic control as measured by A1c level over 6 months. The secondary endpoints comprised the proportion of participants with changes in the NAS of at least 2 points over 6 months. Both these measures were compared between the two dietary groups.
The two groups were matched at baseline, with a mean age of 55-57 years, 58% were women, 89% with metabolic syndrome, and a mean BMI 34 kg/m2.
In baseline liver disease, F1 level fibrosis was the most common (58%), followed by hepatic steatosis (S1, 47%; S2, 32%), with a median NAS of 3, and 19% had nonalcoholic steatohepatitis.
The special thing about these diets was that participants were told to “not lose weight, but eat until you are full,” remarked Dr. Dalby Hansen.
Those on the LCHF diet consumed an average of 61% energy from fat, 13% from carbohydrates, and 23% from protein, compared with the low-fat diet, which comprised an average of 29% energy from fat, 46% from carbohydrates, and 21% from protein.
“It’s a lot of fat and corresponds to a quarter of a liter of olive oil per day,” said Dr. Dalby Hansen. “They really had to change their mindset a lot, because it was difficult for them to start eating all these fats, especially since we’ve all been told for decades that it isn’t good. But we supported them, and they got into it.”
The LCHF diet was primarily comprised of unsaturated fats – for example, avocado, oil, nuts, and seeds – but also included saturated fats, such as cheese, cream, and high-fat dairy products. Participants were free to eat unsaturated and saturated fats, but Dr. Dalby Hansen and her team advised participants that “good” unsaturated fats were preferable.
“Also, this diet contained vegetables but no bread, no potatoes, no rice, and no pasta. It was low in carbohydrates, below 20%,” she added.
Improved glycemic control, reduced liver fat
“We found that the LCHF diet improved diabetes control, it reduced the fat in the liver, and, even though they’re eating as many calories as they were used to until they were full, they lost 5.8% of body weight,” said Dr. Dalby Hansen in reporting the results. Participants in the low-fat group lost only 1.8% of body weight.
However, mean calorie intake dropped in both groups, by –2.2% in the LCHF group and –8.7% in the low-fat group.
“The LCHF diet improved the primary outcome of A1c by 9.5 mmol/mol, which is similar to some anti-diabetic medications, such as DPP-4 inhibitors and SGLT2 inhibitors,” she said.
The low-fat group reduced A1c by 3.4 mmol/mol, resulting in a between-group difference of 6.1 mmol/mol.
“Upon follow-up of 3 months, after stopping the diets, on average the participants in both groups returned their HbA1c levels to nearly baseline values,” she said. Results were adjusted for weight loss and baseline values.
Both diets also improved the NAS. The proportion of participants who improved their NAS score by 2 or more points was 22% in the LCHF group versus 17% in the low-fat group (P = 0.58). Additionally, in the LCHF group, 70% of participants improved their score by 1 or more points, compared with 49% in the low-fat group and fewer in the LCHF group experienced a worsening of their score (1% vs. 23%, respectively).
One participant on LCHF had high triglycerides of 12 mmol/L after 3 months. Overall, the low-density lipoprotein increased marginally by 0.2 mmol per liter in the high-fat group, said Dr. Dalby Hansen.
Dr. Dalby Hansen noted some limitations. The findings might not be applicable in more severe NAFLD, dietary assessment relied on self-reporting, no food was provided, and participants had to cook themselves. It was also an open-label study because of the nature of the intervention.
Some hope for more sustainable dieting
Many diets are difficult to adhere to, remarked Dr. Dalby Hansen. “We thought this [diet] might be easier to comply with in the longer term, and we hope that these results might provide patients with more options.”
She added that most people who started the diet adapted and complied with it. “However, it might not be for everyone, but I think we can say that if people try, and it fits into their lives, then they go for it.”
However, “it is not about going out and eating whatever fat and how much of it you want. It’s important that you cut the carbohydrates too,” she said. “With this approach, we really saw amazing results.”
Dr. Dalby Hansen added that having various diets available, including the LCHF one, meant that as clinicians they could empower patients to take control of their metabolic health.
“We can ask them directly, ‘What would fit into their life?’” she said. “We know that one size does not fit at all, and I believe that if we could engage patients more, then they can take control of their own situation.”
Asked whether these findings were enough to change guidelines, Zobair Younossi, MD, professor and chairman, department of medicine, Inova Fairfax Medical Campus, Falls Church, Va., remarked that it was the sugar at work here.
“Dietary fat – it’s not the same as fat in the liver, and this diet has more to do with the sugar levels,” he said.
“I’m always reluctant to take results from a short-term study without long-term follow-up,” Dr. Younossi said. “I want to know will patients live longer, and long-term data are needed for this. Until I have that strong evidence that outcomes are going to change, or at least some sign that the outcome is going to change, it is too early to change any guidelines.”
Dr. Dalby Hansen reports no relevant financial relationships. Dr. Harrison reported financial relationships with numerous pharmaceutical companies. Dr. Younossi reports the following financial relationships: research funds and/or consultant to Abbott, Allergan, Bristol Myers Squibb, Echosens, Genfit, Gilead Sciences, Intercept, Madrigal, Merck, and Novo Nordisk.
A version of this article first appeared on Medscape.com.
AT ILC 2022
Pemvidutide promising for fatty liver disease
LONDON – Weight loss, lipid reductions, and “robust improvements” in lipid species associated with nonalcoholic fatty liver disease were achieved in patients who were treated with pemvidutide in a first-in-human, phase 1 clinical trial reported at the annual International Liver Congress, sponsored by the European Association for the Study of the Liver.
The presenting study investigator, Stephen A. Harrison, MD, said that pemvidutide, which is also being developed for the treatment of obesity, appeared to be well tolerated. There were no serious or severe adverse events, and no patient had to discontinue treatment because of side effects.
Overall, “pemvidutide represents a promising new agent,” said Dr. Harrison, medical director of Pinnacle Research in San Antonio, Texas.
Dual incretin effect
Pemvidutide is a “balanced” dual agonist of glucagon-like peptide 1 (GLP-1) and glucagon, Dr. Harrison explained in his oral abstract.
“With glucagon, we are working to drive energy expenditure up, and with GLP-1, we’re decreasing food intake,” Dr. Harrison said.
What might set pemvidutide apart from other incretins lies within its structure, Dr. Harrison suggested. The structure has two main regions – one with greater GLP-1 specificity and the other with greater glucagon specificity, and these two areas are linked by a propriety technology called a EuPort™ domain. This is an area which allows the drug to bind to albumin, which increases its serum half-life and enables weekly dosing while slowing its entry into the bloodstream.
“Ultimately, we think that this has impacts, hypothetically, on tolerability and potentially mitigating the need for dose escalation,” said Dr. Harrison.
Weight loss results
The phase 1 study Dr. Harrison presented had a randomized, double-blind, placebo-controlled design with single and multiple ascending doses (SAD/MAD) of pemvidutide being tested. He presented data on the MAD phase only, noting that the SAD phase had been used to determine what doses to use in the latter.
Seventy individuals with a body mass index of between 25 and 40 kg/m2 were recruited and 34 of these were enrolled in the MAD phase of the study. Three doses of pemvidutide were used, given subcutaneously once a week for 12 weeks: Seven participants received 1.2 mg, 9 were given 1.8 mg, 11 had 2.4 mg, and 7 subjects were treated with placebo. Dr. Harrison noted that there were no caloric restrictions in the trial and no lifestyle modifications or interventions.
The average age of study participants ranged from 27 to 35 years and the mean BMI was 30-31 kg/m2 across each group, with their lipid parameters in the upper range of normal.
Clear weight loss reductions were seen across all the pemvidutide groups versus placebo, with the greatest percentage changes in weight loss seen with the two higher doses used. At week 12, there was a 4.9%, 10.3% and 9.0% weight loss in the 1.2-mg, 1.8-mg and 2.4-mg pemvidutide groups compared to 1.6% in placebo-treated individuals.
All patients in the 1.8-mg group achieved a 5% or greater weight loss, Dr. Harrison observed, but there “was a plateauing” effect with the 2.4-mg dose with 89% of patients achieving this target. In comparison, a third of patients on the lowest dose and 20% of those on placebo achieved this target.
The trajectory of weight loss seen in the trial suggests that “the rate of weight loss would continue beyond 12 weeks if we were to continue the therapy” Dr. Harrison said.
Lipid changes and liver fat reductions
Levels of serum lipids from baseline to week 12 fell to a greater extent with pemvidutide treatment than with placebo, in the range of –27% for total cholesterol in the two highest dose groups, –25% for LDL-cholesterol for those groups, –37% for triglycerides for the 1.2- and 1.8-mg groups, and reductions in apolipoprotein B were seen.
“We saw an initial decline in HDL [high-density lipoprotein],” Dr. Harrison said, noting that “this is consistent with prior studies looking at rapid weight loss, and over time, this mitigates as you continue to treat at least based on other mechanisms of action or other drugs with similar mechanisms.”
Pemvidutide treatment was also associated with increased lipid oxidation and decreased lipid synthesis, and “there was a robust decrease in lipids implicated in NASH inflammation,” Dr. Harrison pointed out.
Importantly, in five of eight participants who had high levels of liver fat at baseline – defined as a 5% or greater magnetic resonance imaging–derived proton-density-fat-fraction (MRI-PDFF) – showed a decrease to undetectable limits (1.5% or less). This was a greater than 90% reduction in liver fat, Dr. Harrison said. All five patients were in the 1.8-mg and 2.4-mg groups.
As for side effects, these were “predominantly upper GI, with nausea and vomiting.” These were mild in most cases, but he pointed out that five patients treated with the 1.8-mg dose experienced moderate nausea and three experienced moderate vomiting. Mild diarrhea and constipation were also seen in two of patients given this dose but was not reported in any of the other groups.
During the discussion following the presentation, it was pointed out that there was no clear dose-dependent effect considering the 1.8-mg dose seemed to have a stronger effect in some areas than the 2.4-mg dose. That’s a fair point, Dr. Harrison responded, reiterating it was a small study with a short treatment duration, but that there did look like a plateauing effect, “at least in patients with a mean BMI of between 30 and 31.”
Dr. Harrison was asked about potential effects on insulin levels and if that was a worry because, if glucagon is stimulated, it could increase insulin. That in turn might encourage insulin resistance and promote worse outcomes.
“If you look outside of just this program, glucagon agonism has been dosed in a lot of patients over time, and we haven’t seen that,” Dr. Harrison replied. Pemvidutide is an agonist rather than antagonist, so perhaps the [nonalcoholic steatohepatitis]–inducing effects seen before with glucagon antagonism won’t occur, he suggested.
Dr. Harrison disclosed ties to Altimmune (the study sponsor), Akero, Axcella, Bristol Myers Squibb, Cirius, CiVi Biopharma, Conatus, Corcept, CymaBay, Enyo, Galectin, Genentech, Genfit, Gilead, Hepion, Hightide, HistoIndex, Intercept, Madrigal, Metacrine, NGM Bio, Novartis, Novo Nordisk, NorthSea, Pfizer, Sagimet, Viking, and 89Bio.
LONDON – Weight loss, lipid reductions, and “robust improvements” in lipid species associated with nonalcoholic fatty liver disease were achieved in patients who were treated with pemvidutide in a first-in-human, phase 1 clinical trial reported at the annual International Liver Congress, sponsored by the European Association for the Study of the Liver.
The presenting study investigator, Stephen A. Harrison, MD, said that pemvidutide, which is also being developed for the treatment of obesity, appeared to be well tolerated. There were no serious or severe adverse events, and no patient had to discontinue treatment because of side effects.
Overall, “pemvidutide represents a promising new agent,” said Dr. Harrison, medical director of Pinnacle Research in San Antonio, Texas.
Dual incretin effect
Pemvidutide is a “balanced” dual agonist of glucagon-like peptide 1 (GLP-1) and glucagon, Dr. Harrison explained in his oral abstract.
“With glucagon, we are working to drive energy expenditure up, and with GLP-1, we’re decreasing food intake,” Dr. Harrison said.
What might set pemvidutide apart from other incretins lies within its structure, Dr. Harrison suggested. The structure has two main regions – one with greater GLP-1 specificity and the other with greater glucagon specificity, and these two areas are linked by a propriety technology called a EuPort™ domain. This is an area which allows the drug to bind to albumin, which increases its serum half-life and enables weekly dosing while slowing its entry into the bloodstream.
“Ultimately, we think that this has impacts, hypothetically, on tolerability and potentially mitigating the need for dose escalation,” said Dr. Harrison.
Weight loss results
The phase 1 study Dr. Harrison presented had a randomized, double-blind, placebo-controlled design with single and multiple ascending doses (SAD/MAD) of pemvidutide being tested. He presented data on the MAD phase only, noting that the SAD phase had been used to determine what doses to use in the latter.
Seventy individuals with a body mass index of between 25 and 40 kg/m2 were recruited and 34 of these were enrolled in the MAD phase of the study. Three doses of pemvidutide were used, given subcutaneously once a week for 12 weeks: Seven participants received 1.2 mg, 9 were given 1.8 mg, 11 had 2.4 mg, and 7 subjects were treated with placebo. Dr. Harrison noted that there were no caloric restrictions in the trial and no lifestyle modifications or interventions.
The average age of study participants ranged from 27 to 35 years and the mean BMI was 30-31 kg/m2 across each group, with their lipid parameters in the upper range of normal.
Clear weight loss reductions were seen across all the pemvidutide groups versus placebo, with the greatest percentage changes in weight loss seen with the two higher doses used. At week 12, there was a 4.9%, 10.3% and 9.0% weight loss in the 1.2-mg, 1.8-mg and 2.4-mg pemvidutide groups compared to 1.6% in placebo-treated individuals.
All patients in the 1.8-mg group achieved a 5% or greater weight loss, Dr. Harrison observed, but there “was a plateauing” effect with the 2.4-mg dose with 89% of patients achieving this target. In comparison, a third of patients on the lowest dose and 20% of those on placebo achieved this target.
The trajectory of weight loss seen in the trial suggests that “the rate of weight loss would continue beyond 12 weeks if we were to continue the therapy” Dr. Harrison said.
Lipid changes and liver fat reductions
Levels of serum lipids from baseline to week 12 fell to a greater extent with pemvidutide treatment than with placebo, in the range of –27% for total cholesterol in the two highest dose groups, –25% for LDL-cholesterol for those groups, –37% for triglycerides for the 1.2- and 1.8-mg groups, and reductions in apolipoprotein B were seen.
“We saw an initial decline in HDL [high-density lipoprotein],” Dr. Harrison said, noting that “this is consistent with prior studies looking at rapid weight loss, and over time, this mitigates as you continue to treat at least based on other mechanisms of action or other drugs with similar mechanisms.”
Pemvidutide treatment was also associated with increased lipid oxidation and decreased lipid synthesis, and “there was a robust decrease in lipids implicated in NASH inflammation,” Dr. Harrison pointed out.
Importantly, in five of eight participants who had high levels of liver fat at baseline – defined as a 5% or greater magnetic resonance imaging–derived proton-density-fat-fraction (MRI-PDFF) – showed a decrease to undetectable limits (1.5% or less). This was a greater than 90% reduction in liver fat, Dr. Harrison said. All five patients were in the 1.8-mg and 2.4-mg groups.
As for side effects, these were “predominantly upper GI, with nausea and vomiting.” These were mild in most cases, but he pointed out that five patients treated with the 1.8-mg dose experienced moderate nausea and three experienced moderate vomiting. Mild diarrhea and constipation were also seen in two of patients given this dose but was not reported in any of the other groups.
During the discussion following the presentation, it was pointed out that there was no clear dose-dependent effect considering the 1.8-mg dose seemed to have a stronger effect in some areas than the 2.4-mg dose. That’s a fair point, Dr. Harrison responded, reiterating it was a small study with a short treatment duration, but that there did look like a plateauing effect, “at least in patients with a mean BMI of between 30 and 31.”
Dr. Harrison was asked about potential effects on insulin levels and if that was a worry because, if glucagon is stimulated, it could increase insulin. That in turn might encourage insulin resistance and promote worse outcomes.
“If you look outside of just this program, glucagon agonism has been dosed in a lot of patients over time, and we haven’t seen that,” Dr. Harrison replied. Pemvidutide is an agonist rather than antagonist, so perhaps the [nonalcoholic steatohepatitis]–inducing effects seen before with glucagon antagonism won’t occur, he suggested.
Dr. Harrison disclosed ties to Altimmune (the study sponsor), Akero, Axcella, Bristol Myers Squibb, Cirius, CiVi Biopharma, Conatus, Corcept, CymaBay, Enyo, Galectin, Genentech, Genfit, Gilead, Hepion, Hightide, HistoIndex, Intercept, Madrigal, Metacrine, NGM Bio, Novartis, Novo Nordisk, NorthSea, Pfizer, Sagimet, Viking, and 89Bio.
LONDON – Weight loss, lipid reductions, and “robust improvements” in lipid species associated with nonalcoholic fatty liver disease were achieved in patients who were treated with pemvidutide in a first-in-human, phase 1 clinical trial reported at the annual International Liver Congress, sponsored by the European Association for the Study of the Liver.
The presenting study investigator, Stephen A. Harrison, MD, said that pemvidutide, which is also being developed for the treatment of obesity, appeared to be well tolerated. There were no serious or severe adverse events, and no patient had to discontinue treatment because of side effects.
Overall, “pemvidutide represents a promising new agent,” said Dr. Harrison, medical director of Pinnacle Research in San Antonio, Texas.
Dual incretin effect
Pemvidutide is a “balanced” dual agonist of glucagon-like peptide 1 (GLP-1) and glucagon, Dr. Harrison explained in his oral abstract.
“With glucagon, we are working to drive energy expenditure up, and with GLP-1, we’re decreasing food intake,” Dr. Harrison said.
What might set pemvidutide apart from other incretins lies within its structure, Dr. Harrison suggested. The structure has two main regions – one with greater GLP-1 specificity and the other with greater glucagon specificity, and these two areas are linked by a propriety technology called a EuPort™ domain. This is an area which allows the drug to bind to albumin, which increases its serum half-life and enables weekly dosing while slowing its entry into the bloodstream.
“Ultimately, we think that this has impacts, hypothetically, on tolerability and potentially mitigating the need for dose escalation,” said Dr. Harrison.
Weight loss results
The phase 1 study Dr. Harrison presented had a randomized, double-blind, placebo-controlled design with single and multiple ascending doses (SAD/MAD) of pemvidutide being tested. He presented data on the MAD phase only, noting that the SAD phase had been used to determine what doses to use in the latter.
Seventy individuals with a body mass index of between 25 and 40 kg/m2 were recruited and 34 of these were enrolled in the MAD phase of the study. Three doses of pemvidutide were used, given subcutaneously once a week for 12 weeks: Seven participants received 1.2 mg, 9 were given 1.8 mg, 11 had 2.4 mg, and 7 subjects were treated with placebo. Dr. Harrison noted that there were no caloric restrictions in the trial and no lifestyle modifications or interventions.
The average age of study participants ranged from 27 to 35 years and the mean BMI was 30-31 kg/m2 across each group, with their lipid parameters in the upper range of normal.
Clear weight loss reductions were seen across all the pemvidutide groups versus placebo, with the greatest percentage changes in weight loss seen with the two higher doses used. At week 12, there was a 4.9%, 10.3% and 9.0% weight loss in the 1.2-mg, 1.8-mg and 2.4-mg pemvidutide groups compared to 1.6% in placebo-treated individuals.
All patients in the 1.8-mg group achieved a 5% or greater weight loss, Dr. Harrison observed, but there “was a plateauing” effect with the 2.4-mg dose with 89% of patients achieving this target. In comparison, a third of patients on the lowest dose and 20% of those on placebo achieved this target.
The trajectory of weight loss seen in the trial suggests that “the rate of weight loss would continue beyond 12 weeks if we were to continue the therapy” Dr. Harrison said.
Lipid changes and liver fat reductions
Levels of serum lipids from baseline to week 12 fell to a greater extent with pemvidutide treatment than with placebo, in the range of –27% for total cholesterol in the two highest dose groups, –25% for LDL-cholesterol for those groups, –37% for triglycerides for the 1.2- and 1.8-mg groups, and reductions in apolipoprotein B were seen.
“We saw an initial decline in HDL [high-density lipoprotein],” Dr. Harrison said, noting that “this is consistent with prior studies looking at rapid weight loss, and over time, this mitigates as you continue to treat at least based on other mechanisms of action or other drugs with similar mechanisms.”
Pemvidutide treatment was also associated with increased lipid oxidation and decreased lipid synthesis, and “there was a robust decrease in lipids implicated in NASH inflammation,” Dr. Harrison pointed out.
Importantly, in five of eight participants who had high levels of liver fat at baseline – defined as a 5% or greater magnetic resonance imaging–derived proton-density-fat-fraction (MRI-PDFF) – showed a decrease to undetectable limits (1.5% or less). This was a greater than 90% reduction in liver fat, Dr. Harrison said. All five patients were in the 1.8-mg and 2.4-mg groups.
As for side effects, these were “predominantly upper GI, with nausea and vomiting.” These were mild in most cases, but he pointed out that five patients treated with the 1.8-mg dose experienced moderate nausea and three experienced moderate vomiting. Mild diarrhea and constipation were also seen in two of patients given this dose but was not reported in any of the other groups.
During the discussion following the presentation, it was pointed out that there was no clear dose-dependent effect considering the 1.8-mg dose seemed to have a stronger effect in some areas than the 2.4-mg dose. That’s a fair point, Dr. Harrison responded, reiterating it was a small study with a short treatment duration, but that there did look like a plateauing effect, “at least in patients with a mean BMI of between 30 and 31.”
Dr. Harrison was asked about potential effects on insulin levels and if that was a worry because, if glucagon is stimulated, it could increase insulin. That in turn might encourage insulin resistance and promote worse outcomes.
“If you look outside of just this program, glucagon agonism has been dosed in a lot of patients over time, and we haven’t seen that,” Dr. Harrison replied. Pemvidutide is an agonist rather than antagonist, so perhaps the [nonalcoholic steatohepatitis]–inducing effects seen before with glucagon antagonism won’t occur, he suggested.
Dr. Harrison disclosed ties to Altimmune (the study sponsor), Akero, Axcella, Bristol Myers Squibb, Cirius, CiVi Biopharma, Conatus, Corcept, CymaBay, Enyo, Galectin, Genentech, Genfit, Gilead, Hepion, Hightide, HistoIndex, Intercept, Madrigal, Metacrine, NGM Bio, Novartis, Novo Nordisk, NorthSea, Pfizer, Sagimet, Viking, and 89Bio.
AT ILC 2022