User login
Specialists think it’s up to the PCP to recommend flu vaccines. But many patients don’t see a PCP every year
A new survey from the National Foundation for Infectious Diseases shows that, despite the recommendation that patients who have chronic illnesses receive annual flu vaccines, only 45% of these patients do get them. People with chronic diseases are at increased risk for serious flu-related complications, including hospitalization and death.
The survey looked at physicians’ practices toward flu vaccination and communication between health care providers (HCP) and their adult patients with chronic health conditions.
Overall, less than a third of HCPs (31%) said they recommend annual flu vaccination to all of their patients with chronic health conditions. There were some surprising differences between subspecialists. For example, 72% of patients with a heart problem who saw a cardiologist said that physician recommended the flu vaccine. The recommendation rate dropped to 32% of lung patients seeing a pulmonary physician and only 10% of people with diabetes who saw an endocrinologist.
There is quite a large gap between what physicians and patients say about their interactions. Fully 77% of HCPs who recommend annual flu vaccination say they tell patients when they are at higher risk of complications from influenza. Yet only 48% of patients say they have been given such information.
Although it is critically important information for patients to learn, their risk of influenza is often missing from the discussion. For example, patients with heart disease are six times more likely to have a heart attack within 7 days of flu infection. People with diabetes are six times more likely to be hospitalized from flu and three times more likely to die. Similarly, those with asthma or chronic obstructive pulmonary disorder are at a much higher risk of complications.
One problem is that Yet only 65% of patients with one of these chronic illnesses report seeing their primary care physician at least annually.
Much of the disparity between the patient’s perception of what they were told and the physician’s is “how the ‘recommendation’ is actually made,” William Schaffner, MD, NFID’s medical director and professor of medicine at Vanderbilt University, Nashville, Tenn., told this news organization. Dr. Schaffner offered the following example: At the end of the visit, the doctor might say: “It’s that time of the year again – you want to think about getting your flu shot.”
“The doctor thinks they’ve recommended that, but the doctor really has opened the door for you to think about it and leave [yourself] unvaccinated.”
Dr. Schaffner’s alternative? Tell the patient: “‘You’ll get your flu vaccine on the way out. Tom or Sally will give it to you.’ That’s a very different kind of recommendation. And it’s a much greater assurance of providing the vaccine.”
Another major problem, Dr. Schaffner said, is that many specialists “don’t think of vaccination as something that’s included with their routine care” even though they do direct much of the patient’s care. He said that physicians should be more “directive” in their care and that immunizations should be better integrated into routine practice.
Jody Lanard, MD, a retired risk communication consultant who spent many years working with the World Health Organization on disease outbreak communications, said in an interview that this disconnect between physician and patient reports “was really jarring. And it’s actionable!”
She offered several practical suggestions. For one, she said, “the messaging to the specialists has to be very, very empathic. We know you’re already overburdened. And here we’re asking you to do something that you think of as somebody else’s job.” But if your patient gets flu, then your job as the cardiologist or endocrinologist will become more complicated and time-consuming. So getting the patients vaccinated will be a good investment and will make your job easier.
Because of the disparity in patient and physician reports, Dr. Lanard suggested implementing a “feedback mechanism where they [the health care providers] give out the prescription, and then the office calls [the patient] to see if they’ve gotten the shot or not. Because that way it will help correct the mismatch between them thinking that they told the patient and the patient not hearing it.”
Asked about why there might be a big gap between what physicians report they said and what patients heard, Dr. Lanard explained that “physicians often communicate in [a manner] sort of like a checklist. And the patients are focused on one or two things that are high in their minds. And the physician was mentioning some things that are on a separate topic that are not on a patient’s list and it goes right past them.”
Dr. Lanard recommended brief storytelling instead of checklists. For example: “I’ve been treating your diabetes for 10 years. During this last flu season, several of my diabetic patients had a really hard time when they caught the flu. So now I’m trying harder to remember to remind you to get your flu shots.”
She urged HCPs to “make it more personal ... but it can still be scripted in advance as part of something that [you’re] remembering to do during the check.” She added that their professional associations may be able to send them suggested language they can adapt.
Finally, Dr. Lanard cautioned about vaccine myths. “The word myth is so insulting. It’s basically a word that sends the signal that you’re an idiot.”
She advised specialists to avoid the word “myth,” which will make the person defensive. Instead, say something like, “A lot of people, even some of my own family members, think the flu vaccine gives you the flu. ... But it doesn’t. And then you go into the reality.”
Dr. Lanard suggested that specialists implement the follow-up calls and close the feedback loop, saying: “If they did the survey a few years later, I bet that gap would narrow.”
Dr. Schaffner and Dr. Lanard disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new survey from the National Foundation for Infectious Diseases shows that, despite the recommendation that patients who have chronic illnesses receive annual flu vaccines, only 45% of these patients do get them. People with chronic diseases are at increased risk for serious flu-related complications, including hospitalization and death.
The survey looked at physicians’ practices toward flu vaccination and communication between health care providers (HCP) and their adult patients with chronic health conditions.
Overall, less than a third of HCPs (31%) said they recommend annual flu vaccination to all of their patients with chronic health conditions. There were some surprising differences between subspecialists. For example, 72% of patients with a heart problem who saw a cardiologist said that physician recommended the flu vaccine. The recommendation rate dropped to 32% of lung patients seeing a pulmonary physician and only 10% of people with diabetes who saw an endocrinologist.
There is quite a large gap between what physicians and patients say about their interactions. Fully 77% of HCPs who recommend annual flu vaccination say they tell patients when they are at higher risk of complications from influenza. Yet only 48% of patients say they have been given such information.
Although it is critically important information for patients to learn, their risk of influenza is often missing from the discussion. For example, patients with heart disease are six times more likely to have a heart attack within 7 days of flu infection. People with diabetes are six times more likely to be hospitalized from flu and three times more likely to die. Similarly, those with asthma or chronic obstructive pulmonary disorder are at a much higher risk of complications.
One problem is that Yet only 65% of patients with one of these chronic illnesses report seeing their primary care physician at least annually.
Much of the disparity between the patient’s perception of what they were told and the physician’s is “how the ‘recommendation’ is actually made,” William Schaffner, MD, NFID’s medical director and professor of medicine at Vanderbilt University, Nashville, Tenn., told this news organization. Dr. Schaffner offered the following example: At the end of the visit, the doctor might say: “It’s that time of the year again – you want to think about getting your flu shot.”
“The doctor thinks they’ve recommended that, but the doctor really has opened the door for you to think about it and leave [yourself] unvaccinated.”
Dr. Schaffner’s alternative? Tell the patient: “‘You’ll get your flu vaccine on the way out. Tom or Sally will give it to you.’ That’s a very different kind of recommendation. And it’s a much greater assurance of providing the vaccine.”
Another major problem, Dr. Schaffner said, is that many specialists “don’t think of vaccination as something that’s included with their routine care” even though they do direct much of the patient’s care. He said that physicians should be more “directive” in their care and that immunizations should be better integrated into routine practice.
Jody Lanard, MD, a retired risk communication consultant who spent many years working with the World Health Organization on disease outbreak communications, said in an interview that this disconnect between physician and patient reports “was really jarring. And it’s actionable!”
She offered several practical suggestions. For one, she said, “the messaging to the specialists has to be very, very empathic. We know you’re already overburdened. And here we’re asking you to do something that you think of as somebody else’s job.” But if your patient gets flu, then your job as the cardiologist or endocrinologist will become more complicated and time-consuming. So getting the patients vaccinated will be a good investment and will make your job easier.
Because of the disparity in patient and physician reports, Dr. Lanard suggested implementing a “feedback mechanism where they [the health care providers] give out the prescription, and then the office calls [the patient] to see if they’ve gotten the shot or not. Because that way it will help correct the mismatch between them thinking that they told the patient and the patient not hearing it.”
Asked about why there might be a big gap between what physicians report they said and what patients heard, Dr. Lanard explained that “physicians often communicate in [a manner] sort of like a checklist. And the patients are focused on one or two things that are high in their minds. And the physician was mentioning some things that are on a separate topic that are not on a patient’s list and it goes right past them.”
Dr. Lanard recommended brief storytelling instead of checklists. For example: “I’ve been treating your diabetes for 10 years. During this last flu season, several of my diabetic patients had a really hard time when they caught the flu. So now I’m trying harder to remember to remind you to get your flu shots.”
She urged HCPs to “make it more personal ... but it can still be scripted in advance as part of something that [you’re] remembering to do during the check.” She added that their professional associations may be able to send them suggested language they can adapt.
Finally, Dr. Lanard cautioned about vaccine myths. “The word myth is so insulting. It’s basically a word that sends the signal that you’re an idiot.”
She advised specialists to avoid the word “myth,” which will make the person defensive. Instead, say something like, “A lot of people, even some of my own family members, think the flu vaccine gives you the flu. ... But it doesn’t. And then you go into the reality.”
Dr. Lanard suggested that specialists implement the follow-up calls and close the feedback loop, saying: “If they did the survey a few years later, I bet that gap would narrow.”
Dr. Schaffner and Dr. Lanard disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new survey from the National Foundation for Infectious Diseases shows that, despite the recommendation that patients who have chronic illnesses receive annual flu vaccines, only 45% of these patients do get them. People with chronic diseases are at increased risk for serious flu-related complications, including hospitalization and death.
The survey looked at physicians’ practices toward flu vaccination and communication between health care providers (HCP) and their adult patients with chronic health conditions.
Overall, less than a third of HCPs (31%) said they recommend annual flu vaccination to all of their patients with chronic health conditions. There were some surprising differences between subspecialists. For example, 72% of patients with a heart problem who saw a cardiologist said that physician recommended the flu vaccine. The recommendation rate dropped to 32% of lung patients seeing a pulmonary physician and only 10% of people with diabetes who saw an endocrinologist.
There is quite a large gap between what physicians and patients say about their interactions. Fully 77% of HCPs who recommend annual flu vaccination say they tell patients when they are at higher risk of complications from influenza. Yet only 48% of patients say they have been given such information.
Although it is critically important information for patients to learn, their risk of influenza is often missing from the discussion. For example, patients with heart disease are six times more likely to have a heart attack within 7 days of flu infection. People with diabetes are six times more likely to be hospitalized from flu and three times more likely to die. Similarly, those with asthma or chronic obstructive pulmonary disorder are at a much higher risk of complications.
One problem is that Yet only 65% of patients with one of these chronic illnesses report seeing their primary care physician at least annually.
Much of the disparity between the patient’s perception of what they were told and the physician’s is “how the ‘recommendation’ is actually made,” William Schaffner, MD, NFID’s medical director and professor of medicine at Vanderbilt University, Nashville, Tenn., told this news organization. Dr. Schaffner offered the following example: At the end of the visit, the doctor might say: “It’s that time of the year again – you want to think about getting your flu shot.”
“The doctor thinks they’ve recommended that, but the doctor really has opened the door for you to think about it and leave [yourself] unvaccinated.”
Dr. Schaffner’s alternative? Tell the patient: “‘You’ll get your flu vaccine on the way out. Tom or Sally will give it to you.’ That’s a very different kind of recommendation. And it’s a much greater assurance of providing the vaccine.”
Another major problem, Dr. Schaffner said, is that many specialists “don’t think of vaccination as something that’s included with their routine care” even though they do direct much of the patient’s care. He said that physicians should be more “directive” in their care and that immunizations should be better integrated into routine practice.
Jody Lanard, MD, a retired risk communication consultant who spent many years working with the World Health Organization on disease outbreak communications, said in an interview that this disconnect between physician and patient reports “was really jarring. And it’s actionable!”
She offered several practical suggestions. For one, she said, “the messaging to the specialists has to be very, very empathic. We know you’re already overburdened. And here we’re asking you to do something that you think of as somebody else’s job.” But if your patient gets flu, then your job as the cardiologist or endocrinologist will become more complicated and time-consuming. So getting the patients vaccinated will be a good investment and will make your job easier.
Because of the disparity in patient and physician reports, Dr. Lanard suggested implementing a “feedback mechanism where they [the health care providers] give out the prescription, and then the office calls [the patient] to see if they’ve gotten the shot or not. Because that way it will help correct the mismatch between them thinking that they told the patient and the patient not hearing it.”
Asked about why there might be a big gap between what physicians report they said and what patients heard, Dr. Lanard explained that “physicians often communicate in [a manner] sort of like a checklist. And the patients are focused on one or two things that are high in their minds. And the physician was mentioning some things that are on a separate topic that are not on a patient’s list and it goes right past them.”
Dr. Lanard recommended brief storytelling instead of checklists. For example: “I’ve been treating your diabetes for 10 years. During this last flu season, several of my diabetic patients had a really hard time when they caught the flu. So now I’m trying harder to remember to remind you to get your flu shots.”
She urged HCPs to “make it more personal ... but it can still be scripted in advance as part of something that [you’re] remembering to do during the check.” She added that their professional associations may be able to send them suggested language they can adapt.
Finally, Dr. Lanard cautioned about vaccine myths. “The word myth is so insulting. It’s basically a word that sends the signal that you’re an idiot.”
She advised specialists to avoid the word “myth,” which will make the person defensive. Instead, say something like, “A lot of people, even some of my own family members, think the flu vaccine gives you the flu. ... But it doesn’t. And then you go into the reality.”
Dr. Lanard suggested that specialists implement the follow-up calls and close the feedback loop, saying: “If they did the survey a few years later, I bet that gap would narrow.”
Dr. Schaffner and Dr. Lanard disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Metabolites implicated in CHD development in African Americans
Selected metabolic biomarkers may influence disease risk and progression in African American and White persons in different ways, a cohort study of the landmark Jackson Heart Study has found.
The investigators identified 22 specific metabolites that seem to influence incident CHD risk in African American patients – 13 metabolites that were also replicated in a multiethnic population and 9 novel metabolites that include N-acylamides and leucine, a branched-chain amino acid.
“To our knowledge, this is the first time that an N-acylamide as a class of molecule has been shown to be associated with incident coronary heart disease,” lead study author Daniel E. Cruz, MD, an instructor at Harvard Medical School in the division of cardiovascular medicine at Beth Israel Deaconess Medical Center in Boston, said in an interview.
The researchers analyzed targeted plasma metabolomic profiles of 2,346 participants in the Jackson Heart Study, a prospective population-based cohort study in the Mississippi city that included 5,306 African American patients evaluated over 15 years. They then performed a replication analysis of CHD-associated metabolites among 1,588 multiethnic participants in the Women’s Health Initiative, another population-based cohort study that included 161,808 postmenopausal women, also over 15 years. In all, the study, published in JAMA Cardiology, identified 46 metabolites that were associated with incident CHD up to 16 years before the incident event
Dr. Cruz said the “most interesting” findings were the roles of the N-acylamide linoleoyl ethanolamide and leucine. The former is of interest “because it is a lipid-signaling molecule that has been shown to have anti-inflammatory effects on macrophages; the influence and effects on macrophages are of particular interest because of macrophages’ central role in atherosclerosis and coronary heart disease,” he said.
Leucine draws interest because, in this study population, it was linked to a reduced risk of incident CHD. The researchers cited four previous studies in predominantly non-Hispanic White populations that found no association between branched-chain amino acids and incident CHD in Circulation, Stroke Circulation: Genomic and Precision Medicine, and Atherosclerosis. Other branched-amino acids included in the analysis trended toward a decreased risk of CHD, but those didn’t achieve the same statistical significance as that of leucine, Dr. Cruz said.
“In some of the analyses we did, there was a subset of metabolites that the associations with CHD appeared to be different between self-identified African Americans in the Jackson cohort vs. self-identified non-Hispanic Whites, and leucine was one of them,” Dr. Cruz said.
He emphasized that this study “is not a genetic analysis” because the participants self-identified their race. “So our next step is to figure out why this difference appears between these self-identified groups,” Dr. Cruz said. “We suspect environmental factors play a role – psychological stress, diet, income level, to name a few – but we are also interested to see if there are genetic causes.”
The results “are not clinically applicable,” Dr. Cruz said, but they do point to a need for more ethnically and racially diverse study populations. “The big picture is that, before we go implementing novel biomarkers into clinical practice, we need to make sure that they are accurate across different populations of people,” he said. “The only way to do this is to study different groups with the same rigor and vigor and thoughtfulness as any other group.”
These findings fall in line with other studies that found other nonmetabolomic biomarkers have countervailing effects on CHD risk in African Americans and non-Hispanic Whites, said Christie M. Ballantyne, MD, chief of cardiology at Baylor College of Medicine in Houston. For example, African Americans have been found to have lower triglyceride and HDL cholesterol levels than those of Whites.
The study “points out that there may be important biological differences in the metabolic pathways and abnormalities in the development of CHD between races,” Dr. Ballantyne said. “This further emphasizes both the importance and challenge of testing therapies in multiple racial/ethnic groups and with more even representation between men and women.”
Combining metabolomic profiling along with other biomarkers and possibly genetics may be helpful to “personalize” therapies in the future, he added.
Dr. Cruz and Dr. Ballantyne have no relevant relationships to disclose.
Selected metabolic biomarkers may influence disease risk and progression in African American and White persons in different ways, a cohort study of the landmark Jackson Heart Study has found.
The investigators identified 22 specific metabolites that seem to influence incident CHD risk in African American patients – 13 metabolites that were also replicated in a multiethnic population and 9 novel metabolites that include N-acylamides and leucine, a branched-chain amino acid.
“To our knowledge, this is the first time that an N-acylamide as a class of molecule has been shown to be associated with incident coronary heart disease,” lead study author Daniel E. Cruz, MD, an instructor at Harvard Medical School in the division of cardiovascular medicine at Beth Israel Deaconess Medical Center in Boston, said in an interview.
The researchers analyzed targeted plasma metabolomic profiles of 2,346 participants in the Jackson Heart Study, a prospective population-based cohort study in the Mississippi city that included 5,306 African American patients evaluated over 15 years. They then performed a replication analysis of CHD-associated metabolites among 1,588 multiethnic participants in the Women’s Health Initiative, another population-based cohort study that included 161,808 postmenopausal women, also over 15 years. In all, the study, published in JAMA Cardiology, identified 46 metabolites that were associated with incident CHD up to 16 years before the incident event
Dr. Cruz said the “most interesting” findings were the roles of the N-acylamide linoleoyl ethanolamide and leucine. The former is of interest “because it is a lipid-signaling molecule that has been shown to have anti-inflammatory effects on macrophages; the influence and effects on macrophages are of particular interest because of macrophages’ central role in atherosclerosis and coronary heart disease,” he said.
Leucine draws interest because, in this study population, it was linked to a reduced risk of incident CHD. The researchers cited four previous studies in predominantly non-Hispanic White populations that found no association between branched-chain amino acids and incident CHD in Circulation, Stroke Circulation: Genomic and Precision Medicine, and Atherosclerosis. Other branched-amino acids included in the analysis trended toward a decreased risk of CHD, but those didn’t achieve the same statistical significance as that of leucine, Dr. Cruz said.
“In some of the analyses we did, there was a subset of metabolites that the associations with CHD appeared to be different between self-identified African Americans in the Jackson cohort vs. self-identified non-Hispanic Whites, and leucine was one of them,” Dr. Cruz said.
He emphasized that this study “is not a genetic analysis” because the participants self-identified their race. “So our next step is to figure out why this difference appears between these self-identified groups,” Dr. Cruz said. “We suspect environmental factors play a role – psychological stress, diet, income level, to name a few – but we are also interested to see if there are genetic causes.”
The results “are not clinically applicable,” Dr. Cruz said, but they do point to a need for more ethnically and racially diverse study populations. “The big picture is that, before we go implementing novel biomarkers into clinical practice, we need to make sure that they are accurate across different populations of people,” he said. “The only way to do this is to study different groups with the same rigor and vigor and thoughtfulness as any other group.”
These findings fall in line with other studies that found other nonmetabolomic biomarkers have countervailing effects on CHD risk in African Americans and non-Hispanic Whites, said Christie M. Ballantyne, MD, chief of cardiology at Baylor College of Medicine in Houston. For example, African Americans have been found to have lower triglyceride and HDL cholesterol levels than those of Whites.
The study “points out that there may be important biological differences in the metabolic pathways and abnormalities in the development of CHD between races,” Dr. Ballantyne said. “This further emphasizes both the importance and challenge of testing therapies in multiple racial/ethnic groups and with more even representation between men and women.”
Combining metabolomic profiling along with other biomarkers and possibly genetics may be helpful to “personalize” therapies in the future, he added.
Dr. Cruz and Dr. Ballantyne have no relevant relationships to disclose.
Selected metabolic biomarkers may influence disease risk and progression in African American and White persons in different ways, a cohort study of the landmark Jackson Heart Study has found.
The investigators identified 22 specific metabolites that seem to influence incident CHD risk in African American patients – 13 metabolites that were also replicated in a multiethnic population and 9 novel metabolites that include N-acylamides and leucine, a branched-chain amino acid.
“To our knowledge, this is the first time that an N-acylamide as a class of molecule has been shown to be associated with incident coronary heart disease,” lead study author Daniel E. Cruz, MD, an instructor at Harvard Medical School in the division of cardiovascular medicine at Beth Israel Deaconess Medical Center in Boston, said in an interview.
The researchers analyzed targeted plasma metabolomic profiles of 2,346 participants in the Jackson Heart Study, a prospective population-based cohort study in the Mississippi city that included 5,306 African American patients evaluated over 15 years. They then performed a replication analysis of CHD-associated metabolites among 1,588 multiethnic participants in the Women’s Health Initiative, another population-based cohort study that included 161,808 postmenopausal women, also over 15 years. In all, the study, published in JAMA Cardiology, identified 46 metabolites that were associated with incident CHD up to 16 years before the incident event
Dr. Cruz said the “most interesting” findings were the roles of the N-acylamide linoleoyl ethanolamide and leucine. The former is of interest “because it is a lipid-signaling molecule that has been shown to have anti-inflammatory effects on macrophages; the influence and effects on macrophages are of particular interest because of macrophages’ central role in atherosclerosis and coronary heart disease,” he said.
Leucine draws interest because, in this study population, it was linked to a reduced risk of incident CHD. The researchers cited four previous studies in predominantly non-Hispanic White populations that found no association between branched-chain amino acids and incident CHD in Circulation, Stroke Circulation: Genomic and Precision Medicine, and Atherosclerosis. Other branched-amino acids included in the analysis trended toward a decreased risk of CHD, but those didn’t achieve the same statistical significance as that of leucine, Dr. Cruz said.
“In some of the analyses we did, there was a subset of metabolites that the associations with CHD appeared to be different between self-identified African Americans in the Jackson cohort vs. self-identified non-Hispanic Whites, and leucine was one of them,” Dr. Cruz said.
He emphasized that this study “is not a genetic analysis” because the participants self-identified their race. “So our next step is to figure out why this difference appears between these self-identified groups,” Dr. Cruz said. “We suspect environmental factors play a role – psychological stress, diet, income level, to name a few – but we are also interested to see if there are genetic causes.”
The results “are not clinically applicable,” Dr. Cruz said, but they do point to a need for more ethnically and racially diverse study populations. “The big picture is that, before we go implementing novel biomarkers into clinical practice, we need to make sure that they are accurate across different populations of people,” he said. “The only way to do this is to study different groups with the same rigor and vigor and thoughtfulness as any other group.”
These findings fall in line with other studies that found other nonmetabolomic biomarkers have countervailing effects on CHD risk in African Americans and non-Hispanic Whites, said Christie M. Ballantyne, MD, chief of cardiology at Baylor College of Medicine in Houston. For example, African Americans have been found to have lower triglyceride and HDL cholesterol levels than those of Whites.
The study “points out that there may be important biological differences in the metabolic pathways and abnormalities in the development of CHD between races,” Dr. Ballantyne said. “This further emphasizes both the importance and challenge of testing therapies in multiple racial/ethnic groups and with more even representation between men and women.”
Combining metabolomic profiling along with other biomarkers and possibly genetics may be helpful to “personalize” therapies in the future, he added.
Dr. Cruz and Dr. Ballantyne have no relevant relationships to disclose.
FROM JAMA CARDIOLOGY
U.S. obesity rates soar in early adulthood
Obesity rates among “emerging adults” aged 18-25 have soared in the United States in recent decades with the mean body mass index (BMI) for these young adults now in the overweight category, according to research highlighting troubling trends in an often-overlooked age group.
While similar patterns have been observed in other age groups, including adolescents (ages 12-19) and young adults (ages 20-39) across recent decades, emerging adulthood tends to get less attention in the evaluation of obesity trends.
“Emerging adulthood may be a key period for preventing and treating obesity given that habits formed during this period often persist through the remainder of the life course,” write the authors of the study, which was published online Nov. 23 in JAMA.
“There is an urgent need for research on risk factors contributing to obesity during this developmental stage to inform the design of interventions as well as policies aimed at prevention,” they add.
They found that by 2018 a third of all young adults had obesity, compared with just 6% at the beginning of the study periods in 1976.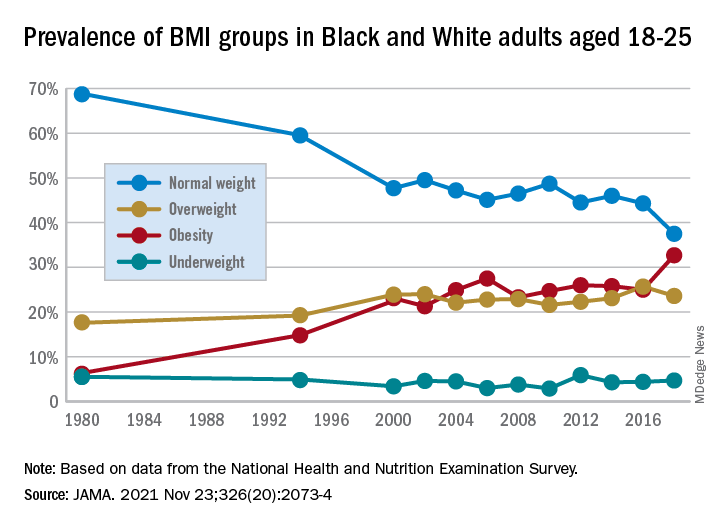
Studying the ages of transition
The findings are from an analysis of 8,015 emerging adults aged 18-25 in the cross-sectional National Health and Nutrition Examination Survey (NHANES), including NHANES II (1976-1980), NHANES III (1988-1994), and the continuous NHANES cycles from 1999 through 2018.
About half (3,965) of participants were female, 3,037 were non-Hispanic Black, and 2,386 met the criteria for household poverty.
The results showed substantial increases in mean BMI among emerging adults from a level in the normal range, at 23.1 kg/m2, in 1976-1980, increasing to 27.7 kg/m2 (overweight) in 2017-2018 (P = .006).
The prevalence of obesity (BMI 30.0 kg/m2 or higher) in the emerging adult age group soared from 6.2% between 1976-1980 to 32.7% in 2017-2018 (P = .007).
Meanwhile, the rate of those with normal/healthy weight (BMI 18.5-24.9 kg/m2) dropped from 68.7% to 37.5% (P = .005) over the same period.
Sensitivity analyses that were limited to continuous NHANES cycles showed similar results.
First author Alejandra Ellison-Barnes, MD, MPH, said the trends are consistent with rising obesity rates in the population as a whole – other studies have shown increases in obesity among children, adolescents, and adults over the same period – but are nevertheless striking, she stressed.
Young adults now fall into overweight category
“While we were not surprised by the general trend, given what is known about the increasing prevalence of obesity in both children and adults, we were surprised by the magnitude of the increase in prevalence and that the mean BMI in this age group now falls in the overweight range,” Dr. Ellison-Barnes, of the Division of General Internal Medicine, Johns Hopkins University School of Medicine, Baltimore, told this news organization.
She said she is not aware of other studies that have looked at obesity trends specifically among emerging adults.
However, considering the substantial life changes and growing independence, the life stage is important to understand in terms of dietary/lifestyle patterns.
“We theorize that emerging adulthood is a critical period for obesity development given that it is a time when individuals are often undergoing major life transitions such as leaving home, attending higher education, entering the workforce, and developing new relationships,” she emphasized.
As far as causes are concerned, “societal and cultural trends in these areas over the past several decades may have played a role in the observed changes,” she speculated.
The study population was limited to non-Hispanic Black and non-Hispanic White individuals due to changes in how NHANES assessed race and ethnicity over time. Therefore, a study limitation is that the patterns observed may not be generalizable to other races and ethnicities, the authors note.
However, considering the influence lifestyle changes can have, early adulthood “may be an ideal time to intervene in the clinical setting to prevent, manage, or reverse obesity to prevent adverse health outcomes in the future,” Dr. Ellison-Barnes said.
Dr. Ellison-Barnes has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Obesity rates among “emerging adults” aged 18-25 have soared in the United States in recent decades with the mean body mass index (BMI) for these young adults now in the overweight category, according to research highlighting troubling trends in an often-overlooked age group.
While similar patterns have been observed in other age groups, including adolescents (ages 12-19) and young adults (ages 20-39) across recent decades, emerging adulthood tends to get less attention in the evaluation of obesity trends.
“Emerging adulthood may be a key period for preventing and treating obesity given that habits formed during this period often persist through the remainder of the life course,” write the authors of the study, which was published online Nov. 23 in JAMA.
“There is an urgent need for research on risk factors contributing to obesity during this developmental stage to inform the design of interventions as well as policies aimed at prevention,” they add.
They found that by 2018 a third of all young adults had obesity, compared with just 6% at the beginning of the study periods in 1976.
Studying the ages of transition
The findings are from an analysis of 8,015 emerging adults aged 18-25 in the cross-sectional National Health and Nutrition Examination Survey (NHANES), including NHANES II (1976-1980), NHANES III (1988-1994), and the continuous NHANES cycles from 1999 through 2018.
About half (3,965) of participants were female, 3,037 were non-Hispanic Black, and 2,386 met the criteria for household poverty.
The results showed substantial increases in mean BMI among emerging adults from a level in the normal range, at 23.1 kg/m2, in 1976-1980, increasing to 27.7 kg/m2 (overweight) in 2017-2018 (P = .006).
The prevalence of obesity (BMI 30.0 kg/m2 or higher) in the emerging adult age group soared from 6.2% between 1976-1980 to 32.7% in 2017-2018 (P = .007).
Meanwhile, the rate of those with normal/healthy weight (BMI 18.5-24.9 kg/m2) dropped from 68.7% to 37.5% (P = .005) over the same period.
Sensitivity analyses that were limited to continuous NHANES cycles showed similar results.
First author Alejandra Ellison-Barnes, MD, MPH, said the trends are consistent with rising obesity rates in the population as a whole – other studies have shown increases in obesity among children, adolescents, and adults over the same period – but are nevertheless striking, she stressed.
Young adults now fall into overweight category
“While we were not surprised by the general trend, given what is known about the increasing prevalence of obesity in both children and adults, we were surprised by the magnitude of the increase in prevalence and that the mean BMI in this age group now falls in the overweight range,” Dr. Ellison-Barnes, of the Division of General Internal Medicine, Johns Hopkins University School of Medicine, Baltimore, told this news organization.
She said she is not aware of other studies that have looked at obesity trends specifically among emerging adults.
However, considering the substantial life changes and growing independence, the life stage is important to understand in terms of dietary/lifestyle patterns.
“We theorize that emerging adulthood is a critical period for obesity development given that it is a time when individuals are often undergoing major life transitions such as leaving home, attending higher education, entering the workforce, and developing new relationships,” she emphasized.
As far as causes are concerned, “societal and cultural trends in these areas over the past several decades may have played a role in the observed changes,” she speculated.
The study population was limited to non-Hispanic Black and non-Hispanic White individuals due to changes in how NHANES assessed race and ethnicity over time. Therefore, a study limitation is that the patterns observed may not be generalizable to other races and ethnicities, the authors note.
However, considering the influence lifestyle changes can have, early adulthood “may be an ideal time to intervene in the clinical setting to prevent, manage, or reverse obesity to prevent adverse health outcomes in the future,” Dr. Ellison-Barnes said.
Dr. Ellison-Barnes has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Obesity rates among “emerging adults” aged 18-25 have soared in the United States in recent decades with the mean body mass index (BMI) for these young adults now in the overweight category, according to research highlighting troubling trends in an often-overlooked age group.
While similar patterns have been observed in other age groups, including adolescents (ages 12-19) and young adults (ages 20-39) across recent decades, emerging adulthood tends to get less attention in the evaluation of obesity trends.
“Emerging adulthood may be a key period for preventing and treating obesity given that habits formed during this period often persist through the remainder of the life course,” write the authors of the study, which was published online Nov. 23 in JAMA.
“There is an urgent need for research on risk factors contributing to obesity during this developmental stage to inform the design of interventions as well as policies aimed at prevention,” they add.
They found that by 2018 a third of all young adults had obesity, compared with just 6% at the beginning of the study periods in 1976.
Studying the ages of transition
The findings are from an analysis of 8,015 emerging adults aged 18-25 in the cross-sectional National Health and Nutrition Examination Survey (NHANES), including NHANES II (1976-1980), NHANES III (1988-1994), and the continuous NHANES cycles from 1999 through 2018.
About half (3,965) of participants were female, 3,037 were non-Hispanic Black, and 2,386 met the criteria for household poverty.
The results showed substantial increases in mean BMI among emerging adults from a level in the normal range, at 23.1 kg/m2, in 1976-1980, increasing to 27.7 kg/m2 (overweight) in 2017-2018 (P = .006).
The prevalence of obesity (BMI 30.0 kg/m2 or higher) in the emerging adult age group soared from 6.2% between 1976-1980 to 32.7% in 2017-2018 (P = .007).
Meanwhile, the rate of those with normal/healthy weight (BMI 18.5-24.9 kg/m2) dropped from 68.7% to 37.5% (P = .005) over the same period.
Sensitivity analyses that were limited to continuous NHANES cycles showed similar results.
First author Alejandra Ellison-Barnes, MD, MPH, said the trends are consistent with rising obesity rates in the population as a whole – other studies have shown increases in obesity among children, adolescents, and adults over the same period – but are nevertheless striking, she stressed.
Young adults now fall into overweight category
“While we were not surprised by the general trend, given what is known about the increasing prevalence of obesity in both children and adults, we were surprised by the magnitude of the increase in prevalence and that the mean BMI in this age group now falls in the overweight range,” Dr. Ellison-Barnes, of the Division of General Internal Medicine, Johns Hopkins University School of Medicine, Baltimore, told this news organization.
She said she is not aware of other studies that have looked at obesity trends specifically among emerging adults.
However, considering the substantial life changes and growing independence, the life stage is important to understand in terms of dietary/lifestyle patterns.
“We theorize that emerging adulthood is a critical period for obesity development given that it is a time when individuals are often undergoing major life transitions such as leaving home, attending higher education, entering the workforce, and developing new relationships,” she emphasized.
As far as causes are concerned, “societal and cultural trends in these areas over the past several decades may have played a role in the observed changes,” she speculated.
The study population was limited to non-Hispanic Black and non-Hispanic White individuals due to changes in how NHANES assessed race and ethnicity over time. Therefore, a study limitation is that the patterns observed may not be generalizable to other races and ethnicities, the authors note.
However, considering the influence lifestyle changes can have, early adulthood “may be an ideal time to intervene in the clinical setting to prevent, manage, or reverse obesity to prevent adverse health outcomes in the future,” Dr. Ellison-Barnes said.
Dr. Ellison-Barnes has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Evaluation of Intermittent Energy Restriction and Continuous Energy Restriction on Weight Loss and Blood Pressure Control in Overweight and Obese Patients With Hypertension
Study Overview
Objective. To compare the effects of intermittent energy restriction (IER) with those of continuous energy restriction (CER) on blood pressure control and weight loss in overweight and obese patients with hypertension during a 6-month period.
Design. Randomized controlled trial.
Settings and participants. The trial was conducted at the Affiliated Hospital of Jiaxing University from June 1, 2020, to April 30, 2021. Chinese adults were recruited using advertisements and flyers posted in the hospital and local communities. Prior to participation in study activities, all participants gave informed consent prior to recruitment and were provided compensation in the form of a $38 voucher at 3 and 6 months for their time for participating in the study.
The main inclusion criteria were patients between the ages of 18 and 70 years, hypertension, and body mass index (BMI) ranging from 24 to 40 kg/m2. The exclusion criteria were systolic blood pressure (SBP) ≥ 180 mmHg or diastolic blood pressure (DBP) ≥ 120 mmHg, type 1 or 2 diabetes with a history of severe hypoglycemic episodes, pregnancy or breastfeeding, usage of glucagon-like peptide 1 receptor agonists, weight loss > 5 kg within the past 3 months or previous weight loss surgery, and inability to adhere to the dietary protocol.
Of the 294 participants screened for eligibility, 205 were randomized in a 1:1 ratio to the IER group (n = 102) or the CER group (n = 103), stratified by sex and BMI (as overweight or obese). All participants were required to have a stable medication regimen and weight in the 3 months prior to enrollment and not to use weight-loss drugs or vitamin supplements for the duration of the study. Researchers and participants were not blinded to the study group assignment.
Interventions. Participants randomly assigned to the IER group followed a 5:2 eating pattern: a very-low-energy diet of 500-600 kcal for 2 days of the week along with their usual diet for the other 5 days. The 2 days of calorie restriction could be consecutive or nonconsecutive, with a minimum of 0.8 g supplemental protein per kg of body weight per day, in accordance with the 2016 Dietary Guidelines for Chinese Residents. The CER group was advised to consume 1000 kcal/day for women and 1200 kcal/day for men on a 7-day energy restriction. That is, they were prescribed a daily 25% restriction based on the general principles of a Mediterranean-type diet (30% fat, 45-50% carbohydrate, and 20-25% protein).
Both groups received dietary education from a qualified dietitian and were recommended to maintain their current daily activity levels throughout the trial. Written dietary information brochures with portion advice and sample meal plans were provided to improve compliance in each group. All participants received a digital cooking scale to weigh foods to ensure accuracy of intake and were required to keep a food diary while following the recommended recipe on 2 days/week during calorie restriction to help with adherence. No food was provided. All participants were followed up by regular outpatient visits to both cardiologists and dietitians once a month. Diet checklists, activity schedules, and weight were reviewed to assess compliance with dietary advice at each visit.
Of note, participants were encouraged to measure and record their BP twice daily, and if 2 consecutive BP readings were < 110/70 mmHg and/or accompanied by hypotensive episodes with symptoms (dizziness, nausea, headache, and fatigue), they were asked to contact the investigators directly. Antihypertensive medication changes were then made in consultation with cardiologists. In addition, a medication management protocol (ie, doses of antidiabetic medications, including insulin and sulfonylurea) was designed to avoid hypoglycemia. Medication could be reduced in the CER group based on the basal dose at the endocrinologist’s discretion. In the IER group, insulin and sulfonylureas were discontinued on calorie restriction days only, and long-acting insulin was discontinued the night before the IER day. Insulin was not to be resumed until a full day’s caloric intake was achieved.
Measures and analysis. The primary outcomes of this study were changes in BP and weight (measured using an automatic digital sphygmomanometer and an electronic scale), and the secondary outcomes were changes in body composition (assessed by dual-energy x-ray absorptiometry scanning), as well as glycosylated hemoglobin A1c (HbA1c) levels and blood lipids after 6 months. All outcome measures were recorded at baseline and at each monthly visit. Incidence rates of hypoglycemia were based on blood glucose (defined as blood glucose < 70 mg/dL) and/or symptomatic hypoglycemia (symptoms of sweating, paleness, dizziness, and confusion). Two cardiologists who were blind to the patients’ diet condition measured and recorded all pertinent clinical parameters and adjudicated serious adverse events.
Data were compared using independent-samples t-tests or the Mann–Whitney U test for continuous variables, and Pearson’s χ2 test or Fisher’s exact test for categorial variables as appropriate. Repeated-measures ANOVA via a linear mixed model was employed to test the effects of diet, time, and their interaction. In subgroup analyses, differential effects of the intervention on the primary outcomes were evaluated with respect to patients’ level of education, domicile, and sex based on the statistical significance of the interaction term for the subgroup of interest in the multivariate model. Analyses were performed based on completers and on an intention-to-treat principle.
Main results. Among the 205 randomized participants, 118 were women and 87 were men; mean (SD) age was 50.5 (8.8) years; mean (SD) BMI was 28.7 (2.6); mean (SD) SBP was 143 (10) mmHg; and mean (SD) DBP was 91 (9) mmHg. At the end of the 6-month intervention, 173 (84.4%) completed the study (IER group: n = 88; CER group: n = 85). Both groups had similar dropout rates at 6 months (IER group: 14 participants [13.7%]; CER group: 18 participants [17.5%]; P = .83) and were well matched for baseline characteristics except for triglyceride levels.
In the completers analysis, both groups experienced significant reductions in weight (mean [SEM]), but there was no difference between treatment groups (−7.2 [0.6] kg in the IER group vs −7.1 [0.6] kg in the CER group; diet by time P = .72). Similarly, the change in SBP and DBP achieved was statistically significant over time, but there was also no difference between the dietary interventions (−8 [0.7] mmHg in the IER group vs −8 [0.6] mmHg in the CER group, diet by time P = .68; −6 [0.6] mmHg in the IER group vs −6 [0.5] mmHg in the CER group, diet by time P = .53]. Subgroup analyses of the association of the intervention with weight, SBP and DBP by sex, education, and domicile showed no significant between-group differences.
All measures of body composition decreased significantly at 6 months with both groups experiencing comparable reductions in total fat mass (−5.5 [0.6] kg in the IER group vs −4.8 [0.5] kg in the CER group, diet by time P = .08) and android fat mass (−1.1 [0.2] kg in the IER group vs −0.8 [0.2] kg in the CER group, diet by time P = .16). Of note, participants in the CER group lost significantly more total fat-free mass than did participants in the IER group (mean [SEM], −2.3 [0.2] kg vs −1.7 [0.2] kg; P = .03], and there was a trend toward a greater change in total fat mass in the IER group (P = .08). The secondary outcome of mean (SEM) HbA1c (−0.2% [0.1%]) and blood lipid levels (triglyceride level, −1.0 [0.3] mmol/L; total cholesterol level, −0.9 [0.2] mmol/L; low-density lipoprotein cholesterol level, −0.9 [0.2 mmol/L; high-density lipoprotein cholesterol level, 0.7 [0.3] mmol/L] improved with weight loss (P < .05), with no differences between groups (diet by time P > .05).
The intention-to-treat analysis demonstrated that IER and CER are equally effective for weight loss and blood pressure control: both groups experienced significant reductions in weight, SBP, and DBP, but with no difference between treatment groups – mean (SEM) weight change with IER was −7.0 (0.6) kg vs −6.8 (0.6) kg with CER; the mean (SEM) SBP with IER was −7 (0.7) mmHg vs −7 (0.6) mmHg with CER; and the mean (SEM) DBP with IER was −6 (0.5) mmHg vs −5 (0.5) mmHg with CER, (diet by time P = .62, .39, and .41, respectively). There were favorable improvements in
Conclusion. A 2-day severe energy restriction with 5 days of habitual eating compared to 7 days of CER provides an acceptable alternative for BP control and weight loss in overweight and obese individuals with hypertension after 6 months. IER may offer a useful alternative strategy for this population, who find continuous weight-loss diets too difficult to maintain.
Commentary
Globally, obesity represents a major health challenge as it substantially increases the risk of diseases such as hypertension, type 2 diabetes, and coronary heart disease.1 Lifestyle modifications, including weight loss and increased physical activity, are recommended in major guidelines as a first-step intervention in the treatment of hypertensive patients.2 However, lifestyle and behavioral interventions aimed at reducing calorie intake through low-calorie dieting is challenging as it is dependent on individual motivation and adherence to a strict, continuous protocol. Further, CER strategies have limited effectiveness because complex and persistent hormonal, metabolic, and neurochemical adaptations defend against weight loss and promote weight regain.3-4 IER has drawn attention in the popular media as an alternative to CER due to its feasibility and even potential for higher rates of compliance.5
This study adds to the literature as it is the first randomized controlled trial (to the knowledge of the authors at the time of publication) to explore 2 forms of energy restriction – CER and IER – and their impact on weight loss, BP, body composition, HbA1c, and blood lipid levels in overweight and obese patients with high blood pressure. Results from this study showed that IER is as effective as, but not superior to, CER (in terms of the outcomes measures assessed). Specifically, findings highlighted that the 5:2 diet is an effective strategy and noninferior to that of daily calorie restriction for BP and weight control. In addition, both weight loss and BP reduction were greater in a subgroup of obese compared with overweight participants, which indicates that obese populations may benefit more from energy restriction. As the authors highlight, this study both aligns with and expands on current related literature.
This study has both strengths and limitations, especially with regard to the design and data analysis strategy. A key strength is the randomized controlled trial design which enables increased internal validity and decreases several sources of bias, including selection bias and confounding. In addition, it was also designed as a pragmatic trial, with the protocol reflecting efforts to replicate the real-world environment by not supplying meal replacements or food. Notably, only 9 patients could not comply with the protocol, indicating that acceptability of the diet protocol was high. However, as this was only a 6-month long study, further studies are needed to determine whether a 5:2 diet is sustainable (and effective) in the long-term compared with CER, which the authors highlight. The study was also adequately powered to detect clinically meaningful differences in weight loss and SBP, and appropriate analyses were performed on both the basis of completers and on an intention-to-treat principle. However, further studies are needed that are adequately powered to also detect clinically meaningful differences in the other measures, ie, body composition, HbA1c, and blood lipid levels. Importantly, generalizability of findings from this study is limited as the study population comprises only Chinese adults, predominately middle-aged, overweight, and had mildly to moderately elevated SBP and DBP, and excluded diabetic patients. Thus, findings are not necessarily applicable to individuals with highly elevated blood pressure or poorly controlled diabetes.
Applications for Clinical Practice
Results of this study demonstrated that IER is an effective alternative diet strategy for weight loss and blood pressure control in overweight and obese patients with hypertension and is comparable to CER. This is relevant for clinical practice as IER may be easier to maintain in this population compared to continuous weight-loss diets. Importantly, both types of calorie restriction require clinical oversight as medication changes and periodic monitoring of hypotensive and hypoglycemic episodes are needed. Clinicians should consider what is feasible and sustainable for their patients when recommending intermittent energy restriction.
Financial disclosures: None.
1. Blüher M. Obesity: global epidemiology and pathogenesis. Nat Rev Endocrinol. 2019;15(5):288-298. doi:10.1038/s41574-019-0176-8
2. Unger T, Borghi C, Charchar F, et al. 2020 International Society of Hypertension Global hypertension practice guidelines. J Hypertens. 2020;38(6):982-1004. doi:10.1097/HJH.0000000000002453
3. Müller MJ, Enderle J, Bosy-Westphal A. Changes in Energy Expenditure with Weight Gain and Weight Loss in Humans. Curr Obes Rep. 2016;5(4):413-423. doi:10.1007/s13679-016-0237-4
4. Sainsbury A, Wood RE, Seimon RV, et al. Rationale for novel intermittent dieting strategies to attenuate adaptive responses to energy restriction. Obes Rev. 2018;19 Suppl 1:47–60. doi:10.1111/obr.12787
5. Davis CS, Clarke RE, Coulter SN, et al. Intermittent energy restriction and weight loss: a systematic review. Eur J Clin Nutr. 2016;70(3):292-299. doi:10.1038/ejcn.2015.195
Study Overview
Objective. To compare the effects of intermittent energy restriction (IER) with those of continuous energy restriction (CER) on blood pressure control and weight loss in overweight and obese patients with hypertension during a 6-month period.
Design. Randomized controlled trial.
Settings and participants. The trial was conducted at the Affiliated Hospital of Jiaxing University from June 1, 2020, to April 30, 2021. Chinese adults were recruited using advertisements and flyers posted in the hospital and local communities. Prior to participation in study activities, all participants gave informed consent prior to recruitment and were provided compensation in the form of a $38 voucher at 3 and 6 months for their time for participating in the study.
The main inclusion criteria were patients between the ages of 18 and 70 years, hypertension, and body mass index (BMI) ranging from 24 to 40 kg/m2. The exclusion criteria were systolic blood pressure (SBP) ≥ 180 mmHg or diastolic blood pressure (DBP) ≥ 120 mmHg, type 1 or 2 diabetes with a history of severe hypoglycemic episodes, pregnancy or breastfeeding, usage of glucagon-like peptide 1 receptor agonists, weight loss > 5 kg within the past 3 months or previous weight loss surgery, and inability to adhere to the dietary protocol.
Of the 294 participants screened for eligibility, 205 were randomized in a 1:1 ratio to the IER group (n = 102) or the CER group (n = 103), stratified by sex and BMI (as overweight or obese). All participants were required to have a stable medication regimen and weight in the 3 months prior to enrollment and not to use weight-loss drugs or vitamin supplements for the duration of the study. Researchers and participants were not blinded to the study group assignment.
Interventions. Participants randomly assigned to the IER group followed a 5:2 eating pattern: a very-low-energy diet of 500-600 kcal for 2 days of the week along with their usual diet for the other 5 days. The 2 days of calorie restriction could be consecutive or nonconsecutive, with a minimum of 0.8 g supplemental protein per kg of body weight per day, in accordance with the 2016 Dietary Guidelines for Chinese Residents. The CER group was advised to consume 1000 kcal/day for women and 1200 kcal/day for men on a 7-day energy restriction. That is, they were prescribed a daily 25% restriction based on the general principles of a Mediterranean-type diet (30% fat, 45-50% carbohydrate, and 20-25% protein).
Both groups received dietary education from a qualified dietitian and were recommended to maintain their current daily activity levels throughout the trial. Written dietary information brochures with portion advice and sample meal plans were provided to improve compliance in each group. All participants received a digital cooking scale to weigh foods to ensure accuracy of intake and were required to keep a food diary while following the recommended recipe on 2 days/week during calorie restriction to help with adherence. No food was provided. All participants were followed up by regular outpatient visits to both cardiologists and dietitians once a month. Diet checklists, activity schedules, and weight were reviewed to assess compliance with dietary advice at each visit.
Of note, participants were encouraged to measure and record their BP twice daily, and if 2 consecutive BP readings were < 110/70 mmHg and/or accompanied by hypotensive episodes with symptoms (dizziness, nausea, headache, and fatigue), they were asked to contact the investigators directly. Antihypertensive medication changes were then made in consultation with cardiologists. In addition, a medication management protocol (ie, doses of antidiabetic medications, including insulin and sulfonylurea) was designed to avoid hypoglycemia. Medication could be reduced in the CER group based on the basal dose at the endocrinologist’s discretion. In the IER group, insulin and sulfonylureas were discontinued on calorie restriction days only, and long-acting insulin was discontinued the night before the IER day. Insulin was not to be resumed until a full day’s caloric intake was achieved.
Measures and analysis. The primary outcomes of this study were changes in BP and weight (measured using an automatic digital sphygmomanometer and an electronic scale), and the secondary outcomes were changes in body composition (assessed by dual-energy x-ray absorptiometry scanning), as well as glycosylated hemoglobin A1c (HbA1c) levels and blood lipids after 6 months. All outcome measures were recorded at baseline and at each monthly visit. Incidence rates of hypoglycemia were based on blood glucose (defined as blood glucose < 70 mg/dL) and/or symptomatic hypoglycemia (symptoms of sweating, paleness, dizziness, and confusion). Two cardiologists who were blind to the patients’ diet condition measured and recorded all pertinent clinical parameters and adjudicated serious adverse events.
Data were compared using independent-samples t-tests or the Mann–Whitney U test for continuous variables, and Pearson’s χ2 test or Fisher’s exact test for categorial variables as appropriate. Repeated-measures ANOVA via a linear mixed model was employed to test the effects of diet, time, and their interaction. In subgroup analyses, differential effects of the intervention on the primary outcomes were evaluated with respect to patients’ level of education, domicile, and sex based on the statistical significance of the interaction term for the subgroup of interest in the multivariate model. Analyses were performed based on completers and on an intention-to-treat principle.
Main results. Among the 205 randomized participants, 118 were women and 87 were men; mean (SD) age was 50.5 (8.8) years; mean (SD) BMI was 28.7 (2.6); mean (SD) SBP was 143 (10) mmHg; and mean (SD) DBP was 91 (9) mmHg. At the end of the 6-month intervention, 173 (84.4%) completed the study (IER group: n = 88; CER group: n = 85). Both groups had similar dropout rates at 6 months (IER group: 14 participants [13.7%]; CER group: 18 participants [17.5%]; P = .83) and were well matched for baseline characteristics except for triglyceride levels.
In the completers analysis, both groups experienced significant reductions in weight (mean [SEM]), but there was no difference between treatment groups (−7.2 [0.6] kg in the IER group vs −7.1 [0.6] kg in the CER group; diet by time P = .72). Similarly, the change in SBP and DBP achieved was statistically significant over time, but there was also no difference between the dietary interventions (−8 [0.7] mmHg in the IER group vs −8 [0.6] mmHg in the CER group, diet by time P = .68; −6 [0.6] mmHg in the IER group vs −6 [0.5] mmHg in the CER group, diet by time P = .53]. Subgroup analyses of the association of the intervention with weight, SBP and DBP by sex, education, and domicile showed no significant between-group differences.
All measures of body composition decreased significantly at 6 months with both groups experiencing comparable reductions in total fat mass (−5.5 [0.6] kg in the IER group vs −4.8 [0.5] kg in the CER group, diet by time P = .08) and android fat mass (−1.1 [0.2] kg in the IER group vs −0.8 [0.2] kg in the CER group, diet by time P = .16). Of note, participants in the CER group lost significantly more total fat-free mass than did participants in the IER group (mean [SEM], −2.3 [0.2] kg vs −1.7 [0.2] kg; P = .03], and there was a trend toward a greater change in total fat mass in the IER group (P = .08). The secondary outcome of mean (SEM) HbA1c (−0.2% [0.1%]) and blood lipid levels (triglyceride level, −1.0 [0.3] mmol/L; total cholesterol level, −0.9 [0.2] mmol/L; low-density lipoprotein cholesterol level, −0.9 [0.2 mmol/L; high-density lipoprotein cholesterol level, 0.7 [0.3] mmol/L] improved with weight loss (P < .05), with no differences between groups (diet by time P > .05).
The intention-to-treat analysis demonstrated that IER and CER are equally effective for weight loss and blood pressure control: both groups experienced significant reductions in weight, SBP, and DBP, but with no difference between treatment groups – mean (SEM) weight change with IER was −7.0 (0.6) kg vs −6.8 (0.6) kg with CER; the mean (SEM) SBP with IER was −7 (0.7) mmHg vs −7 (0.6) mmHg with CER; and the mean (SEM) DBP with IER was −6 (0.5) mmHg vs −5 (0.5) mmHg with CER, (diet by time P = .62, .39, and .41, respectively). There were favorable improvements in
Conclusion. A 2-day severe energy restriction with 5 days of habitual eating compared to 7 days of CER provides an acceptable alternative for BP control and weight loss in overweight and obese individuals with hypertension after 6 months. IER may offer a useful alternative strategy for this population, who find continuous weight-loss diets too difficult to maintain.
Commentary
Globally, obesity represents a major health challenge as it substantially increases the risk of diseases such as hypertension, type 2 diabetes, and coronary heart disease.1 Lifestyle modifications, including weight loss and increased physical activity, are recommended in major guidelines as a first-step intervention in the treatment of hypertensive patients.2 However, lifestyle and behavioral interventions aimed at reducing calorie intake through low-calorie dieting is challenging as it is dependent on individual motivation and adherence to a strict, continuous protocol. Further, CER strategies have limited effectiveness because complex and persistent hormonal, metabolic, and neurochemical adaptations defend against weight loss and promote weight regain.3-4 IER has drawn attention in the popular media as an alternative to CER due to its feasibility and even potential for higher rates of compliance.5
This study adds to the literature as it is the first randomized controlled trial (to the knowledge of the authors at the time of publication) to explore 2 forms of energy restriction – CER and IER – and their impact on weight loss, BP, body composition, HbA1c, and blood lipid levels in overweight and obese patients with high blood pressure. Results from this study showed that IER is as effective as, but not superior to, CER (in terms of the outcomes measures assessed). Specifically, findings highlighted that the 5:2 diet is an effective strategy and noninferior to that of daily calorie restriction for BP and weight control. In addition, both weight loss and BP reduction were greater in a subgroup of obese compared with overweight participants, which indicates that obese populations may benefit more from energy restriction. As the authors highlight, this study both aligns with and expands on current related literature.
This study has both strengths and limitations, especially with regard to the design and data analysis strategy. A key strength is the randomized controlled trial design which enables increased internal validity and decreases several sources of bias, including selection bias and confounding. In addition, it was also designed as a pragmatic trial, with the protocol reflecting efforts to replicate the real-world environment by not supplying meal replacements or food. Notably, only 9 patients could not comply with the protocol, indicating that acceptability of the diet protocol was high. However, as this was only a 6-month long study, further studies are needed to determine whether a 5:2 diet is sustainable (and effective) in the long-term compared with CER, which the authors highlight. The study was also adequately powered to detect clinically meaningful differences in weight loss and SBP, and appropriate analyses were performed on both the basis of completers and on an intention-to-treat principle. However, further studies are needed that are adequately powered to also detect clinically meaningful differences in the other measures, ie, body composition, HbA1c, and blood lipid levels. Importantly, generalizability of findings from this study is limited as the study population comprises only Chinese adults, predominately middle-aged, overweight, and had mildly to moderately elevated SBP and DBP, and excluded diabetic patients. Thus, findings are not necessarily applicable to individuals with highly elevated blood pressure or poorly controlled diabetes.
Applications for Clinical Practice
Results of this study demonstrated that IER is an effective alternative diet strategy for weight loss and blood pressure control in overweight and obese patients with hypertension and is comparable to CER. This is relevant for clinical practice as IER may be easier to maintain in this population compared to continuous weight-loss diets. Importantly, both types of calorie restriction require clinical oversight as medication changes and periodic monitoring of hypotensive and hypoglycemic episodes are needed. Clinicians should consider what is feasible and sustainable for their patients when recommending intermittent energy restriction.
Financial disclosures: None.
Study Overview
Objective. To compare the effects of intermittent energy restriction (IER) with those of continuous energy restriction (CER) on blood pressure control and weight loss in overweight and obese patients with hypertension during a 6-month period.
Design. Randomized controlled trial.
Settings and participants. The trial was conducted at the Affiliated Hospital of Jiaxing University from June 1, 2020, to April 30, 2021. Chinese adults were recruited using advertisements and flyers posted in the hospital and local communities. Prior to participation in study activities, all participants gave informed consent prior to recruitment and were provided compensation in the form of a $38 voucher at 3 and 6 months for their time for participating in the study.
The main inclusion criteria were patients between the ages of 18 and 70 years, hypertension, and body mass index (BMI) ranging from 24 to 40 kg/m2. The exclusion criteria were systolic blood pressure (SBP) ≥ 180 mmHg or diastolic blood pressure (DBP) ≥ 120 mmHg, type 1 or 2 diabetes with a history of severe hypoglycemic episodes, pregnancy or breastfeeding, usage of glucagon-like peptide 1 receptor agonists, weight loss > 5 kg within the past 3 months or previous weight loss surgery, and inability to adhere to the dietary protocol.
Of the 294 participants screened for eligibility, 205 were randomized in a 1:1 ratio to the IER group (n = 102) or the CER group (n = 103), stratified by sex and BMI (as overweight or obese). All participants were required to have a stable medication regimen and weight in the 3 months prior to enrollment and not to use weight-loss drugs or vitamin supplements for the duration of the study. Researchers and participants were not blinded to the study group assignment.
Interventions. Participants randomly assigned to the IER group followed a 5:2 eating pattern: a very-low-energy diet of 500-600 kcal for 2 days of the week along with their usual diet for the other 5 days. The 2 days of calorie restriction could be consecutive or nonconsecutive, with a minimum of 0.8 g supplemental protein per kg of body weight per day, in accordance with the 2016 Dietary Guidelines for Chinese Residents. The CER group was advised to consume 1000 kcal/day for women and 1200 kcal/day for men on a 7-day energy restriction. That is, they were prescribed a daily 25% restriction based on the general principles of a Mediterranean-type diet (30% fat, 45-50% carbohydrate, and 20-25% protein).
Both groups received dietary education from a qualified dietitian and were recommended to maintain their current daily activity levels throughout the trial. Written dietary information brochures with portion advice and sample meal plans were provided to improve compliance in each group. All participants received a digital cooking scale to weigh foods to ensure accuracy of intake and were required to keep a food diary while following the recommended recipe on 2 days/week during calorie restriction to help with adherence. No food was provided. All participants were followed up by regular outpatient visits to both cardiologists and dietitians once a month. Diet checklists, activity schedules, and weight were reviewed to assess compliance with dietary advice at each visit.
Of note, participants were encouraged to measure and record their BP twice daily, and if 2 consecutive BP readings were < 110/70 mmHg and/or accompanied by hypotensive episodes with symptoms (dizziness, nausea, headache, and fatigue), they were asked to contact the investigators directly. Antihypertensive medication changes were then made in consultation with cardiologists. In addition, a medication management protocol (ie, doses of antidiabetic medications, including insulin and sulfonylurea) was designed to avoid hypoglycemia. Medication could be reduced in the CER group based on the basal dose at the endocrinologist’s discretion. In the IER group, insulin and sulfonylureas were discontinued on calorie restriction days only, and long-acting insulin was discontinued the night before the IER day. Insulin was not to be resumed until a full day’s caloric intake was achieved.
Measures and analysis. The primary outcomes of this study were changes in BP and weight (measured using an automatic digital sphygmomanometer and an electronic scale), and the secondary outcomes were changes in body composition (assessed by dual-energy x-ray absorptiometry scanning), as well as glycosylated hemoglobin A1c (HbA1c) levels and blood lipids after 6 months. All outcome measures were recorded at baseline and at each monthly visit. Incidence rates of hypoglycemia were based on blood glucose (defined as blood glucose < 70 mg/dL) and/or symptomatic hypoglycemia (symptoms of sweating, paleness, dizziness, and confusion). Two cardiologists who were blind to the patients’ diet condition measured and recorded all pertinent clinical parameters and adjudicated serious adverse events.
Data were compared using independent-samples t-tests or the Mann–Whitney U test for continuous variables, and Pearson’s χ2 test or Fisher’s exact test for categorial variables as appropriate. Repeated-measures ANOVA via a linear mixed model was employed to test the effects of diet, time, and their interaction. In subgroup analyses, differential effects of the intervention on the primary outcomes were evaluated with respect to patients’ level of education, domicile, and sex based on the statistical significance of the interaction term for the subgroup of interest in the multivariate model. Analyses were performed based on completers and on an intention-to-treat principle.
Main results. Among the 205 randomized participants, 118 were women and 87 were men; mean (SD) age was 50.5 (8.8) years; mean (SD) BMI was 28.7 (2.6); mean (SD) SBP was 143 (10) mmHg; and mean (SD) DBP was 91 (9) mmHg. At the end of the 6-month intervention, 173 (84.4%) completed the study (IER group: n = 88; CER group: n = 85). Both groups had similar dropout rates at 6 months (IER group: 14 participants [13.7%]; CER group: 18 participants [17.5%]; P = .83) and were well matched for baseline characteristics except for triglyceride levels.
In the completers analysis, both groups experienced significant reductions in weight (mean [SEM]), but there was no difference between treatment groups (−7.2 [0.6] kg in the IER group vs −7.1 [0.6] kg in the CER group; diet by time P = .72). Similarly, the change in SBP and DBP achieved was statistically significant over time, but there was also no difference between the dietary interventions (−8 [0.7] mmHg in the IER group vs −8 [0.6] mmHg in the CER group, diet by time P = .68; −6 [0.6] mmHg in the IER group vs −6 [0.5] mmHg in the CER group, diet by time P = .53]. Subgroup analyses of the association of the intervention with weight, SBP and DBP by sex, education, and domicile showed no significant between-group differences.
All measures of body composition decreased significantly at 6 months with both groups experiencing comparable reductions in total fat mass (−5.5 [0.6] kg in the IER group vs −4.8 [0.5] kg in the CER group, diet by time P = .08) and android fat mass (−1.1 [0.2] kg in the IER group vs −0.8 [0.2] kg in the CER group, diet by time P = .16). Of note, participants in the CER group lost significantly more total fat-free mass than did participants in the IER group (mean [SEM], −2.3 [0.2] kg vs −1.7 [0.2] kg; P = .03], and there was a trend toward a greater change in total fat mass in the IER group (P = .08). The secondary outcome of mean (SEM) HbA1c (−0.2% [0.1%]) and blood lipid levels (triglyceride level, −1.0 [0.3] mmol/L; total cholesterol level, −0.9 [0.2] mmol/L; low-density lipoprotein cholesterol level, −0.9 [0.2 mmol/L; high-density lipoprotein cholesterol level, 0.7 [0.3] mmol/L] improved with weight loss (P < .05), with no differences between groups (diet by time P > .05).
The intention-to-treat analysis demonstrated that IER and CER are equally effective for weight loss and blood pressure control: both groups experienced significant reductions in weight, SBP, and DBP, but with no difference between treatment groups – mean (SEM) weight change with IER was −7.0 (0.6) kg vs −6.8 (0.6) kg with CER; the mean (SEM) SBP with IER was −7 (0.7) mmHg vs −7 (0.6) mmHg with CER; and the mean (SEM) DBP with IER was −6 (0.5) mmHg vs −5 (0.5) mmHg with CER, (diet by time P = .62, .39, and .41, respectively). There were favorable improvements in
Conclusion. A 2-day severe energy restriction with 5 days of habitual eating compared to 7 days of CER provides an acceptable alternative for BP control and weight loss in overweight and obese individuals with hypertension after 6 months. IER may offer a useful alternative strategy for this population, who find continuous weight-loss diets too difficult to maintain.
Commentary
Globally, obesity represents a major health challenge as it substantially increases the risk of diseases such as hypertension, type 2 diabetes, and coronary heart disease.1 Lifestyle modifications, including weight loss and increased physical activity, are recommended in major guidelines as a first-step intervention in the treatment of hypertensive patients.2 However, lifestyle and behavioral interventions aimed at reducing calorie intake through low-calorie dieting is challenging as it is dependent on individual motivation and adherence to a strict, continuous protocol. Further, CER strategies have limited effectiveness because complex and persistent hormonal, metabolic, and neurochemical adaptations defend against weight loss and promote weight regain.3-4 IER has drawn attention in the popular media as an alternative to CER due to its feasibility and even potential for higher rates of compliance.5
This study adds to the literature as it is the first randomized controlled trial (to the knowledge of the authors at the time of publication) to explore 2 forms of energy restriction – CER and IER – and their impact on weight loss, BP, body composition, HbA1c, and blood lipid levels in overweight and obese patients with high blood pressure. Results from this study showed that IER is as effective as, but not superior to, CER (in terms of the outcomes measures assessed). Specifically, findings highlighted that the 5:2 diet is an effective strategy and noninferior to that of daily calorie restriction for BP and weight control. In addition, both weight loss and BP reduction were greater in a subgroup of obese compared with overweight participants, which indicates that obese populations may benefit more from energy restriction. As the authors highlight, this study both aligns with and expands on current related literature.
This study has both strengths and limitations, especially with regard to the design and data analysis strategy. A key strength is the randomized controlled trial design which enables increased internal validity and decreases several sources of bias, including selection bias and confounding. In addition, it was also designed as a pragmatic trial, with the protocol reflecting efforts to replicate the real-world environment by not supplying meal replacements or food. Notably, only 9 patients could not comply with the protocol, indicating that acceptability of the diet protocol was high. However, as this was only a 6-month long study, further studies are needed to determine whether a 5:2 diet is sustainable (and effective) in the long-term compared with CER, which the authors highlight. The study was also adequately powered to detect clinically meaningful differences in weight loss and SBP, and appropriate analyses were performed on both the basis of completers and on an intention-to-treat principle. However, further studies are needed that are adequately powered to also detect clinically meaningful differences in the other measures, ie, body composition, HbA1c, and blood lipid levels. Importantly, generalizability of findings from this study is limited as the study population comprises only Chinese adults, predominately middle-aged, overweight, and had mildly to moderately elevated SBP and DBP, and excluded diabetic patients. Thus, findings are not necessarily applicable to individuals with highly elevated blood pressure or poorly controlled diabetes.
Applications for Clinical Practice
Results of this study demonstrated that IER is an effective alternative diet strategy for weight loss and blood pressure control in overweight and obese patients with hypertension and is comparable to CER. This is relevant for clinical practice as IER may be easier to maintain in this population compared to continuous weight-loss diets. Importantly, both types of calorie restriction require clinical oversight as medication changes and periodic monitoring of hypotensive and hypoglycemic episodes are needed. Clinicians should consider what is feasible and sustainable for their patients when recommending intermittent energy restriction.
Financial disclosures: None.
1. Blüher M. Obesity: global epidemiology and pathogenesis. Nat Rev Endocrinol. 2019;15(5):288-298. doi:10.1038/s41574-019-0176-8
2. Unger T, Borghi C, Charchar F, et al. 2020 International Society of Hypertension Global hypertension practice guidelines. J Hypertens. 2020;38(6):982-1004. doi:10.1097/HJH.0000000000002453
3. Müller MJ, Enderle J, Bosy-Westphal A. Changes in Energy Expenditure with Weight Gain and Weight Loss in Humans. Curr Obes Rep. 2016;5(4):413-423. doi:10.1007/s13679-016-0237-4
4. Sainsbury A, Wood RE, Seimon RV, et al. Rationale for novel intermittent dieting strategies to attenuate adaptive responses to energy restriction. Obes Rev. 2018;19 Suppl 1:47–60. doi:10.1111/obr.12787
5. Davis CS, Clarke RE, Coulter SN, et al. Intermittent energy restriction and weight loss: a systematic review. Eur J Clin Nutr. 2016;70(3):292-299. doi:10.1038/ejcn.2015.195
1. Blüher M. Obesity: global epidemiology and pathogenesis. Nat Rev Endocrinol. 2019;15(5):288-298. doi:10.1038/s41574-019-0176-8
2. Unger T, Borghi C, Charchar F, et al. 2020 International Society of Hypertension Global hypertension practice guidelines. J Hypertens. 2020;38(6):982-1004. doi:10.1097/HJH.0000000000002453
3. Müller MJ, Enderle J, Bosy-Westphal A. Changes in Energy Expenditure with Weight Gain and Weight Loss in Humans. Curr Obes Rep. 2016;5(4):413-423. doi:10.1007/s13679-016-0237-4
4. Sainsbury A, Wood RE, Seimon RV, et al. Rationale for novel intermittent dieting strategies to attenuate adaptive responses to energy restriction. Obes Rev. 2018;19 Suppl 1:47–60. doi:10.1111/obr.12787
5. Davis CS, Clarke RE, Coulter SN, et al. Intermittent energy restriction and weight loss: a systematic review. Eur J Clin Nutr. 2016;70(3):292-299. doi:10.1038/ejcn.2015.195
Vegetable fats tied to lower stroke risk, animal fats to higher risk
Higher intake of vegetable fats from foods such as olive oil and nuts is associated with a lower risk for stroke, whereas people who eat more animal fats, especially processed red meats, may have a higher stroke risk, observational findings suggest.
In a study of more than 117,000 health professionals who were followed for 27 years, those whose diet was in the highest quintile for intake of vegetable fat had a 12% lower risk for stroke, compared with those who consumed the least amount of vegetable fats.
Conversely, having the highest intake of animal fat from nondairy sources was associated with a 16% increased risk of stroke.
Fenglei Wang, PhD, presented these results at the American Heart Association scientific sessions.
“Our findings support the Dietary Guidelines for Americans and dietary recommendations by AHA,” Dr. Wang, a postdoctoral fellow in the department of nutrition at Harvard University’s T.H. Chan School of Public Health in Boston, told this news organization.
“The main sources of vegetable fat have a large overlap with polyunsaturated fat, such as vegetable oils, nuts, walnuts, and peanut butter,” Dr. Wang noted, adding that fish, especially fatty fish, is a main source of polyunsaturated fat and is recommended for cardiovascular health.
“We would recommend that people reduce consumption of red and processed meat, minimize fatty parts of unprocessed meat if consumed, and replace lard or tallow (beef fat) with nontropical vegetable oils, such as olive oil, corn, or soybean oils in cooking, to lower their stroke risk,” she said.
Moreover, although the results from this study of dietary fat are informative, Dr. Wang continued, “there are other dietary factors (fruits, vegetables, salt, alcohol, et cetera), and lifestyle factors (physical activity, smoking, et cetera), that are associated with stroke risk and worthy of attention as well.”
“Many processed meats are high in salt and saturated fat, and low in vegetable fat,” Alice H. Lichtenstein, DSc, an AHA spokesperson who was not involved with this research, noted in a press release.
“Research shows that replacing processed meat with other protein sources, particularly plant sources, is associated with lower death rates,” added Dr. Lichtenstein, the Stanley N. Gershoff professor of nutrition science and policy at Tufts University in Boston, and lead author of the AHA’s 2021 scientific statement, Dietary Guidance to Improve Cardiovascular Health.
“Key features of a heart-healthy diet pattern,” she summarized, “are to balance calorie intake with calorie needs to achieve and maintain a healthy weight; choose whole grains, lean and plant-based protein, and a variety of fruits and vegetables; limit salt, sugar, animal fat, processed foods, and alcohol; and apply this guidance regardless of where the food is prepared or consumed.”
Replace processed meat with plant proteins
The focus on stroke in this study “is important” because, traditionally, studies of diet and cardiovascular health have focused on coronary heart disease, Andrew Mente, PhD, who also was not involved in this research, said in an email to this news organization.
“Overall, the take-home message from the study is that replacing processed meat with plant sources of protein in the diet is probably beneficial,” Dr. Mente, associate professor, health research methods, evidence, and impact, Faculty of Health Sciences, McMaster University, Hamilton, Ont., said.
The finding that people who ate the most vegetable fat had a modest 12% lower risk of stroke than those who ate the least vegetable fat “points to protective effects of foods like seeds, nuts, vegetables, and olive oil, which has been shown previously,” he continued.
The highest quintile of total red meat intake was associated with an 8% higher risk for stroke, but this was driven mainly by processed red meat (which was associated with a 12% higher risk for stroke). These findings are “generally consistent with cohort studies showing that processed meat, as with most highly processed foods for that matter, are associated with an increased risk of cardiovascular events,” Dr. Mente noted.
“Surprisingly, dairy products (such as cheese, butter, or milk) in the study were not connected with the risk of stroke,” he added. This finding differs from results of meta-analyses of multiple cohort studies of dairy intake and stroke and the recent large international PURE study, which showed that dairy intake was associated with a lower risk for stroke.
“What is needed to move the field forward,” according to Dr. Mente, “is to employ new methods that use cutting-edge technology to study nutritional biomarkers and health outcomes.”
“When dealing with modest associations as usually encountered in nutrition, it is a challenge to make causal connections based on dietary questionnaires, which are fraught with measurement error,” he added. “The use of novel methods is where the field is headed.”
Total dietary fat, different types, and different food sources
Dr. Wang and colleagues investigated how total dietary fat, different types of fat, and fats from different foods were associated with incident stroke in 73,867 women in the 1984-2016 Nurses’ Health Study and 43,269 men who participated in the 1986-2016 Health Professionals Follow-up Study.
The participants had an average age of 50 years, 63% were women, and 97% were White. They replied to food-frequency questionnaires every 4 years.
Total red meat included beef, pork, or lamb (as a main dish or in sandwiches or mixed dishes) as well as processed red meats (such as bacon, sausage, bologna, hot dogs, and salami).
Animal fat sources included meat, beef tallow, lard, and full-fat dairy products, such as full-fat milk and cheese.
The median percentage of total daily calories from different sources of fat ranged from 10% to 20% for vegetable fat, 3% to 10% for dairy fat, and 7% to 17% for nondairy animal fat (for lowest to highest quintiles).
The median percentage of total daily calories from different types of fat ranged from 5% to 8% for polyunsaturated fat, 4% to 7% for n-6 polyunsaturated fat, 9% to 15% for monounsaturated fat, 8% to 14% for saturated fat, and 1% to 2% for trans fat.
During follow-up, there were 6,189 incident strokes, including 2,967 ischemic strokes and 814 hemorrhagic strokes.
The researchers found that intake in the highest quintile of vegetable fat was associated with a lower risk for total stroke, compared with the lowest quintile (hazard ratio, 0.88; 95% confidence interval, 0.81-0.96; P for trend < .001).
Similarly, the highest intake of polyunsaturated fat was also associated with lower total stroke (HR, 0.88; 95% CI, 0.80-0.96; P for trend = .002).
Highest intake of nondairy animal fat, however, was associated with an increased risk for total stroke (HR, 1.16; 95% CI, 1.05-1.29; P for trend < .001). They observed “similar associations” for ischemic stroke, but the only positive association for nondairy animal fat was with hemorrhagic stroke, the abstract notes.
The risk for stroke was lower by 9% per serving per day for vegetable oil but increased by 8% and 12%, respectively, per serving of total red meat or processed red meat.
The association for vegetable oil was attenuated after adjustment for vegetable fat or polyunsaturated fat, whereas adjustment for nondairy animal fat rendered the association for total red meat and processed red meat nonsignificant.
The study was funded by the National Heart, Lung, and Blood Institute of the National Institutes of Health. Dr. Wang has no relevant financial disclosures. Dr. Mente has received research funding from the Dairy Farmers of Canada and the National Dairy Council to analyze data on dairy consumption and health outcomes in the PURE study, which is funded by the Population Health Research Institute, Hamilton Health Sciences Research Institute, and more than 70 other sources (government and pharmaceutical).
A version of this article first appeared on Medscape.com.
Higher intake of vegetable fats from foods such as olive oil and nuts is associated with a lower risk for stroke, whereas people who eat more animal fats, especially processed red meats, may have a higher stroke risk, observational findings suggest.
In a study of more than 117,000 health professionals who were followed for 27 years, those whose diet was in the highest quintile for intake of vegetable fat had a 12% lower risk for stroke, compared with those who consumed the least amount of vegetable fats.
Conversely, having the highest intake of animal fat from nondairy sources was associated with a 16% increased risk of stroke.
Fenglei Wang, PhD, presented these results at the American Heart Association scientific sessions.
“Our findings support the Dietary Guidelines for Americans and dietary recommendations by AHA,” Dr. Wang, a postdoctoral fellow in the department of nutrition at Harvard University’s T.H. Chan School of Public Health in Boston, told this news organization.
“The main sources of vegetable fat have a large overlap with polyunsaturated fat, such as vegetable oils, nuts, walnuts, and peanut butter,” Dr. Wang noted, adding that fish, especially fatty fish, is a main source of polyunsaturated fat and is recommended for cardiovascular health.
“We would recommend that people reduce consumption of red and processed meat, minimize fatty parts of unprocessed meat if consumed, and replace lard or tallow (beef fat) with nontropical vegetable oils, such as olive oil, corn, or soybean oils in cooking, to lower their stroke risk,” she said.
Moreover, although the results from this study of dietary fat are informative, Dr. Wang continued, “there are other dietary factors (fruits, vegetables, salt, alcohol, et cetera), and lifestyle factors (physical activity, smoking, et cetera), that are associated with stroke risk and worthy of attention as well.”
“Many processed meats are high in salt and saturated fat, and low in vegetable fat,” Alice H. Lichtenstein, DSc, an AHA spokesperson who was not involved with this research, noted in a press release.
“Research shows that replacing processed meat with other protein sources, particularly plant sources, is associated with lower death rates,” added Dr. Lichtenstein, the Stanley N. Gershoff professor of nutrition science and policy at Tufts University in Boston, and lead author of the AHA’s 2021 scientific statement, Dietary Guidance to Improve Cardiovascular Health.
“Key features of a heart-healthy diet pattern,” she summarized, “are to balance calorie intake with calorie needs to achieve and maintain a healthy weight; choose whole grains, lean and plant-based protein, and a variety of fruits and vegetables; limit salt, sugar, animal fat, processed foods, and alcohol; and apply this guidance regardless of where the food is prepared or consumed.”
Replace processed meat with plant proteins
The focus on stroke in this study “is important” because, traditionally, studies of diet and cardiovascular health have focused on coronary heart disease, Andrew Mente, PhD, who also was not involved in this research, said in an email to this news organization.
“Overall, the take-home message from the study is that replacing processed meat with plant sources of protein in the diet is probably beneficial,” Dr. Mente, associate professor, health research methods, evidence, and impact, Faculty of Health Sciences, McMaster University, Hamilton, Ont., said.
The finding that people who ate the most vegetable fat had a modest 12% lower risk of stroke than those who ate the least vegetable fat “points to protective effects of foods like seeds, nuts, vegetables, and olive oil, which has been shown previously,” he continued.
The highest quintile of total red meat intake was associated with an 8% higher risk for stroke, but this was driven mainly by processed red meat (which was associated with a 12% higher risk for stroke). These findings are “generally consistent with cohort studies showing that processed meat, as with most highly processed foods for that matter, are associated with an increased risk of cardiovascular events,” Dr. Mente noted.
“Surprisingly, dairy products (such as cheese, butter, or milk) in the study were not connected with the risk of stroke,” he added. This finding differs from results of meta-analyses of multiple cohort studies of dairy intake and stroke and the recent large international PURE study, which showed that dairy intake was associated with a lower risk for stroke.
“What is needed to move the field forward,” according to Dr. Mente, “is to employ new methods that use cutting-edge technology to study nutritional biomarkers and health outcomes.”
“When dealing with modest associations as usually encountered in nutrition, it is a challenge to make causal connections based on dietary questionnaires, which are fraught with measurement error,” he added. “The use of novel methods is where the field is headed.”
Total dietary fat, different types, and different food sources
Dr. Wang and colleagues investigated how total dietary fat, different types of fat, and fats from different foods were associated with incident stroke in 73,867 women in the 1984-2016 Nurses’ Health Study and 43,269 men who participated in the 1986-2016 Health Professionals Follow-up Study.
The participants had an average age of 50 years, 63% were women, and 97% were White. They replied to food-frequency questionnaires every 4 years.
Total red meat included beef, pork, or lamb (as a main dish or in sandwiches or mixed dishes) as well as processed red meats (such as bacon, sausage, bologna, hot dogs, and salami).
Animal fat sources included meat, beef tallow, lard, and full-fat dairy products, such as full-fat milk and cheese.
The median percentage of total daily calories from different sources of fat ranged from 10% to 20% for vegetable fat, 3% to 10% for dairy fat, and 7% to 17% for nondairy animal fat (for lowest to highest quintiles).
The median percentage of total daily calories from different types of fat ranged from 5% to 8% for polyunsaturated fat, 4% to 7% for n-6 polyunsaturated fat, 9% to 15% for monounsaturated fat, 8% to 14% for saturated fat, and 1% to 2% for trans fat.
During follow-up, there were 6,189 incident strokes, including 2,967 ischemic strokes and 814 hemorrhagic strokes.
The researchers found that intake in the highest quintile of vegetable fat was associated with a lower risk for total stroke, compared with the lowest quintile (hazard ratio, 0.88; 95% confidence interval, 0.81-0.96; P for trend < .001).
Similarly, the highest intake of polyunsaturated fat was also associated with lower total stroke (HR, 0.88; 95% CI, 0.80-0.96; P for trend = .002).
Highest intake of nondairy animal fat, however, was associated with an increased risk for total stroke (HR, 1.16; 95% CI, 1.05-1.29; P for trend < .001). They observed “similar associations” for ischemic stroke, but the only positive association for nondairy animal fat was with hemorrhagic stroke, the abstract notes.
The risk for stroke was lower by 9% per serving per day for vegetable oil but increased by 8% and 12%, respectively, per serving of total red meat or processed red meat.
The association for vegetable oil was attenuated after adjustment for vegetable fat or polyunsaturated fat, whereas adjustment for nondairy animal fat rendered the association for total red meat and processed red meat nonsignificant.
The study was funded by the National Heart, Lung, and Blood Institute of the National Institutes of Health. Dr. Wang has no relevant financial disclosures. Dr. Mente has received research funding from the Dairy Farmers of Canada and the National Dairy Council to analyze data on dairy consumption and health outcomes in the PURE study, which is funded by the Population Health Research Institute, Hamilton Health Sciences Research Institute, and more than 70 other sources (government and pharmaceutical).
A version of this article first appeared on Medscape.com.
Higher intake of vegetable fats from foods such as olive oil and nuts is associated with a lower risk for stroke, whereas people who eat more animal fats, especially processed red meats, may have a higher stroke risk, observational findings suggest.
In a study of more than 117,000 health professionals who were followed for 27 years, those whose diet was in the highest quintile for intake of vegetable fat had a 12% lower risk for stroke, compared with those who consumed the least amount of vegetable fats.
Conversely, having the highest intake of animal fat from nondairy sources was associated with a 16% increased risk of stroke.
Fenglei Wang, PhD, presented these results at the American Heart Association scientific sessions.
“Our findings support the Dietary Guidelines for Americans and dietary recommendations by AHA,” Dr. Wang, a postdoctoral fellow in the department of nutrition at Harvard University’s T.H. Chan School of Public Health in Boston, told this news organization.
“The main sources of vegetable fat have a large overlap with polyunsaturated fat, such as vegetable oils, nuts, walnuts, and peanut butter,” Dr. Wang noted, adding that fish, especially fatty fish, is a main source of polyunsaturated fat and is recommended for cardiovascular health.
“We would recommend that people reduce consumption of red and processed meat, minimize fatty parts of unprocessed meat if consumed, and replace lard or tallow (beef fat) with nontropical vegetable oils, such as olive oil, corn, or soybean oils in cooking, to lower their stroke risk,” she said.
Moreover, although the results from this study of dietary fat are informative, Dr. Wang continued, “there are other dietary factors (fruits, vegetables, salt, alcohol, et cetera), and lifestyle factors (physical activity, smoking, et cetera), that are associated with stroke risk and worthy of attention as well.”
“Many processed meats are high in salt and saturated fat, and low in vegetable fat,” Alice H. Lichtenstein, DSc, an AHA spokesperson who was not involved with this research, noted in a press release.
“Research shows that replacing processed meat with other protein sources, particularly plant sources, is associated with lower death rates,” added Dr. Lichtenstein, the Stanley N. Gershoff professor of nutrition science and policy at Tufts University in Boston, and lead author of the AHA’s 2021 scientific statement, Dietary Guidance to Improve Cardiovascular Health.
“Key features of a heart-healthy diet pattern,” she summarized, “are to balance calorie intake with calorie needs to achieve and maintain a healthy weight; choose whole grains, lean and plant-based protein, and a variety of fruits and vegetables; limit salt, sugar, animal fat, processed foods, and alcohol; and apply this guidance regardless of where the food is prepared or consumed.”
Replace processed meat with plant proteins
The focus on stroke in this study “is important” because, traditionally, studies of diet and cardiovascular health have focused on coronary heart disease, Andrew Mente, PhD, who also was not involved in this research, said in an email to this news organization.
“Overall, the take-home message from the study is that replacing processed meat with plant sources of protein in the diet is probably beneficial,” Dr. Mente, associate professor, health research methods, evidence, and impact, Faculty of Health Sciences, McMaster University, Hamilton, Ont., said.
The finding that people who ate the most vegetable fat had a modest 12% lower risk of stroke than those who ate the least vegetable fat “points to protective effects of foods like seeds, nuts, vegetables, and olive oil, which has been shown previously,” he continued.
The highest quintile of total red meat intake was associated with an 8% higher risk for stroke, but this was driven mainly by processed red meat (which was associated with a 12% higher risk for stroke). These findings are “generally consistent with cohort studies showing that processed meat, as with most highly processed foods for that matter, are associated with an increased risk of cardiovascular events,” Dr. Mente noted.
“Surprisingly, dairy products (such as cheese, butter, or milk) in the study were not connected with the risk of stroke,” he added. This finding differs from results of meta-analyses of multiple cohort studies of dairy intake and stroke and the recent large international PURE study, which showed that dairy intake was associated with a lower risk for stroke.
“What is needed to move the field forward,” according to Dr. Mente, “is to employ new methods that use cutting-edge technology to study nutritional biomarkers and health outcomes.”
“When dealing with modest associations as usually encountered in nutrition, it is a challenge to make causal connections based on dietary questionnaires, which are fraught with measurement error,” he added. “The use of novel methods is where the field is headed.”
Total dietary fat, different types, and different food sources
Dr. Wang and colleagues investigated how total dietary fat, different types of fat, and fats from different foods were associated with incident stroke in 73,867 women in the 1984-2016 Nurses’ Health Study and 43,269 men who participated in the 1986-2016 Health Professionals Follow-up Study.
The participants had an average age of 50 years, 63% were women, and 97% were White. They replied to food-frequency questionnaires every 4 years.
Total red meat included beef, pork, or lamb (as a main dish or in sandwiches or mixed dishes) as well as processed red meats (such as bacon, sausage, bologna, hot dogs, and salami).
Animal fat sources included meat, beef tallow, lard, and full-fat dairy products, such as full-fat milk and cheese.
The median percentage of total daily calories from different sources of fat ranged from 10% to 20% for vegetable fat, 3% to 10% for dairy fat, and 7% to 17% for nondairy animal fat (for lowest to highest quintiles).
The median percentage of total daily calories from different types of fat ranged from 5% to 8% for polyunsaturated fat, 4% to 7% for n-6 polyunsaturated fat, 9% to 15% for monounsaturated fat, 8% to 14% for saturated fat, and 1% to 2% for trans fat.
During follow-up, there were 6,189 incident strokes, including 2,967 ischemic strokes and 814 hemorrhagic strokes.
The researchers found that intake in the highest quintile of vegetable fat was associated with a lower risk for total stroke, compared with the lowest quintile (hazard ratio, 0.88; 95% confidence interval, 0.81-0.96; P for trend < .001).
Similarly, the highest intake of polyunsaturated fat was also associated with lower total stroke (HR, 0.88; 95% CI, 0.80-0.96; P for trend = .002).
Highest intake of nondairy animal fat, however, was associated with an increased risk for total stroke (HR, 1.16; 95% CI, 1.05-1.29; P for trend < .001). They observed “similar associations” for ischemic stroke, but the only positive association for nondairy animal fat was with hemorrhagic stroke, the abstract notes.
The risk for stroke was lower by 9% per serving per day for vegetable oil but increased by 8% and 12%, respectively, per serving of total red meat or processed red meat.
The association for vegetable oil was attenuated after adjustment for vegetable fat or polyunsaturated fat, whereas adjustment for nondairy animal fat rendered the association for total red meat and processed red meat nonsignificant.
The study was funded by the National Heart, Lung, and Blood Institute of the National Institutes of Health. Dr. Wang has no relevant financial disclosures. Dr. Mente has received research funding from the Dairy Farmers of Canada and the National Dairy Council to analyze data on dairy consumption and health outcomes in the PURE study, which is funded by the Population Health Research Institute, Hamilton Health Sciences Research Institute, and more than 70 other sources (government and pharmaceutical).
A version of this article first appeared on Medscape.com.
FROM AHA 2021
More STEP data: Semaglutide cuts weight, cravings, beats liraglutide
The STEP 5 clinical trial extends favorable weight loss from 1 year out to 2 years for the glucagon-like peptide-1 (GLP-1) agonist semaglutide (Wegovy, Novo Nordisk), given as a once-weekly 2.4-mg subcutaneous injection, and some food cravings were improved in a subgroup analysis.
In another study, STEP 8, weight loss was greater at 68 weeks with semaglutide subcutaneous injection than with a 3-mg daily subcutaneous injection of another GLP-1 agonist, liraglutide (Saxenda, Novo Nordisk), approved earlier for weight loss.
Researchers presented these promising outcomes, with no new safety signals, at ObesityWeek® 2021.
However, there is more to learn about the drug class, researchers agree. Follow-up is still relatively short for a chronic disease and many patients have gastrointestinal side effects with semaglutide, one expert cautions.
The key findings were:
In STEP 5, combined with lifestyle intervention (a reduced-calorie meal plan and advice about physical activity), weekly injection of 2.4 mg semaglutide led to:
- 15.2% weight loss, compared with 2.6% weight loss with placebo at 2 years (P < .0001);
- 77% of patients losing at least 5% of their weight, compared with 34% of patients in the placebo group at 2 years (P < .0001);
- Significantly greater improvement in overall control of cravings, and craving for savory foods, in a subset of patients, versus placebo, but questionnaire scores for positive mood and craving for sweet foods were similar in both groups.
- In STEP 8, mean body weight at 68 weeks was 15.8% lower with 2.4 mg/week subcutaneous semaglutide plus lifestyle changes versus 6.4% lower with 3.0 mg/day subcutaneous liraglutide plus lifestyle changes (P < .001).
Can treat to a target weight-loss range
The undiminished weight loss efficacy in the 2-year data for STEP 5 “portends well,” said W. Timothy Garvey, MD, following his presentation of the results.
“I think this is a new era in obesity care,” said Dr. Garvey, director of the diabetes research center at the University of Alabama at Birmingham. Semaglutide “essentially doubles weight loss efficacy” compared to the other approved pharmacotherapies for obesity.
With this degree of potential weight loss, clinicians “can use weight as a biomarker and treat to a target [weight-loss] range,” he said.
Expounding on this in an interview, Dr. Garvey noted that, as stated in the 2016 American Association of Clinical Endocrinologists (AACE) and American College of Endocrinology (ACE) clinical practice guidelines for medical care of patients with obesity, of which he was lead author, “the objective of care in obesity is to increase health of patients and prevent or treat complications.”
Semaglutide “can treat to a range of weight loss of 10% to 20% in the majority of patients,” which is associated with improvements in cardiovascular and metabolic risk factors.
In STEP 5, of the 51% of patients in the semaglutide group who had prediabetes at enrollment, 80% had normal glycemia at 2 years; however, the trial was not powered nor designed to investigate this.
More data are needed to inform long-term care decisions. The ongoing SELECT cardiovascular outcomes trial of semaglutide, with expected primary study completion on Sept. 28, 2023, should provide more information.
Weight loss plus reduced cravings
In another presentation, Sean Wharton, MD, PharmD, said, “In adults with overweight or obesity, substantial weight loss with semaglutide 2.4 mg was accompanied by short- and long-term improvements in control of eating.”
“Most patients living with obesity who are attempting to decrease calories will have food cravings, based on the biological parameters of weight preservation,” Dr. Wharton, medical director at the Wharton Medical Clinic, in Hamilton, Ont., explained in an email.
The degree of craving varies from patient to patient, likely based on genetics, he added. Research in this field is still emerging.
“I believe that semaglutide 2.4 mg is a game-changer in the field of weight management, and it will change the dialogue for insurance plans and with policymakers regarding coverage for this medication,” said Dr. Wharton.
“The data from the STEP programs are very strong. I am certainly hoping for a change to bias against covering these medications that we have seen in the past,” he said.
Clinically meaningful weight loss
When presenting the STEP 8 findings, Domenica M. Rubino, MD, said: “Participants were significantly more likely to achieve clinically meaningful weight loss thresholds with semaglutide 2.4 mg versus liraglutide 3.0 mg, accompanied by greater improvements in cardiometabolic risk factors.”
For example, patients can have better mobility, which is important for quality of life, Dr. Rubino, director of the Washington Center for Weight Management and Research, Arlington, Virginia, noted.
A smaller percentage of patients respond to liraglutide, she added. Clinicians need to individualize treatment.
When asked, “How do you choose which medical therapy?” Dr. Rubino responded: “We sit and talk.” Finding the medical therapy that fits the patient depends on things such as the patient’s insurance coverage and ability to tolerate side effects such as dehydration, diarrhea, and nausea.
When asked, “How do you switch from liraglutide to semaglutide?” she noted that there are no current guidelines for this. “You have to be careful. Start on the lowest dose of Wegovy. Be cautious, conservative.”
Still early days, caveats remain
“The STEP trials as a group appear to be making the case that obesity may now be considered a medically manageable disease, based on the experience with semaglutide,” Julie R. Ingelfinger, MD, who was not involved with the research, commented in an email.
“STEP 5 and 8 may suggest that weight loss occurs and is sustainable in overweight persons without diabetes with one or more comorbidities or in obese persons without diabetes,” added Dr. Ingelfinger, professor of pediatrics, Harvard Medical School, consultant in pediatric nephrology, Massachusetts General Hospital, Boston, and deputy editor, The New England Journal of Medicine.
However, “even 2 years, in the case of STEP 5, and ~68 weeks in the case of STEP 8, may not be long enough to know whether semaglutide is as promising as these brief summaries (abstracts) suggest,” she cautioned.
“Obesity is a chronic condition, and very long-term therapy and management are required,” Dr. Ingelfinger continued.
“Further, it is hard to generalize when gastrointestinal adverse events are common in a study,” she said. For example, in STEP 8, they were just as common with semaglutide as with the comparator liraglutide, she noted.
“The racial and ethnic representativeness of these studies does not reflect population distributions in the U.S., limiting generalization,” she continued.
“So, there remain caveats in interpreting these data.”
STEP 5 weight loss efficacy and safety at 2 years
Garvey reported that STEP 5 was a phase 3b trial that randomized 304 adults in the United States, Canada, Hungary, Italy, and Spain, who were 18 years and older, with a body mass index (BMI) ≥27 kg/m2 with at least one weight-related comorbidity (hypertension, dyslipidemia, obstructive sleep apnea, or cardiovascular disease) or a BMI ≥30 kg/m2, without type 2 diabetes, to receive semaglutide or placebo plus lifestyle intervention.
Most participants were women (78%) and White (93%). On average, they were 47 years old, weighed 106 kg (223.7 pounds), had a BMI of 38.5 kg/m2, a waist circumference of 115.7 cm (45.6 inches), and an A1c of 5.7%.
A total of 87% of patients in the semaglutide group and 73% of patients in the placebo group completed the trial.
At 104 weeks, participants were more likely to lose ≥10%, ≥15%, and ≥20% of body weight with semaglutide versus placebo (61.8% vs. 13.3%, 52.1% vs. 7.0%, and 36.1% vs. 2.3%, respectively; P < .0001 for all).
Patients in the semaglutide group had greater health improvements in cardiovascular risk factors (waist circumference, systolic and diastolic blood pressure, and C-reactive protein) and metabolic risk factors (A1c, fasting plasma glucose, fasting serum insulin, and triglycerides) than those in the placebo group (P < .05 for all).
Safety and tolerability were consistent with adverse events seen with this drug class, with no new safety signals.
Control of eating questionnaire findings at 2 years in STEP 5
Dr. Wharton and colleagues assessed changes in responses to the Control of Eating questionnaire at baseline and at 20, 52, and 104 weeks in patients from the U.S. and Canada in the STEP 5 trial (88 patients in the semaglutide group and 86 patients in the placebo group).
The questionnaire consisted of 19 questions grouped into four categories: control of food cravings, craving for savory foods (salty and spicy, dairy, or starchy foods), craving for sweet foods (chocolate, sweet foods, or fruit/fruit juice), and positive mood.
At week 104, patients in the semaglutide group had significantly greater improvements in scores for craving for salty and spicy, dairy, and starchy foods, and resisting cravings.
Semaglutide versus liraglutide, 68-week efficacy and safety in STEP 8
STEP 8 randomized 338 U.S. adults without diabetes and a BMI of ≥27 kg/m2 plus one or more weight-related comorbidities or a BMI of ≥30 kg/m2 3:1 to semaglutide 2.4 mg once weekly (n = 126) or matching placebo, or 3:1 liraglutide 3.0 mg once daily (n = 127) or matching placebo, plus lifestyle intervention.
Most participants were women (78%) and were a mean age of 49, had a mean body weight of 104.5 kg, and had a mean BMI of 37.5 kg/m2.
In STEP 8, more participants achieved ≥10%, ≥15%, and ≥20% weight loss with semaglutide than with liraglutide (70.9% vs. 25.6%, 55.6% vs. 12.0%, and 38.5% vs. 6.0%, respectively; P < .001 for all odds ratios).
Semaglutide improved waist circumference, A1c, and C-reactive protein versus liraglutide (unadjusted P < .001 for all).
Gastrointestinal adverse events were reported by 84% and 83% of participants receiving semaglutide and liraglutide, respectively. Most events were mild/moderate and transient, with prevalence declining over time.
Fewer participants stopped treatment due to adverse events with semaglutide than liraglutide (3.2% vs. 12.6%).
Dr. Garvey has reported serving as a site principal investigator for multicentered clinical trials sponsored by his university and funded by Eli Lilly, Novo Nordisk, and Pfizer. Dr. Wharton has reported financial ties to Novo Nordisk, Bausch Health Canada, Eli Lily, and Boehringer Ingelheim Canada. Dr. Rubino has reported ties to Boehringer Ingelheim and AstraZeneca. Dr. Ingelfinger has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The STEP 5 clinical trial extends favorable weight loss from 1 year out to 2 years for the glucagon-like peptide-1 (GLP-1) agonist semaglutide (Wegovy, Novo Nordisk), given as a once-weekly 2.4-mg subcutaneous injection, and some food cravings were improved in a subgroup analysis.
In another study, STEP 8, weight loss was greater at 68 weeks with semaglutide subcutaneous injection than with a 3-mg daily subcutaneous injection of another GLP-1 agonist, liraglutide (Saxenda, Novo Nordisk), approved earlier for weight loss.
Researchers presented these promising outcomes, with no new safety signals, at ObesityWeek® 2021.
However, there is more to learn about the drug class, researchers agree. Follow-up is still relatively short for a chronic disease and many patients have gastrointestinal side effects with semaglutide, one expert cautions.
The key findings were:
In STEP 5, combined with lifestyle intervention (a reduced-calorie meal plan and advice about physical activity), weekly injection of 2.4 mg semaglutide led to:
- 15.2% weight loss, compared with 2.6% weight loss with placebo at 2 years (P < .0001);
- 77% of patients losing at least 5% of their weight, compared with 34% of patients in the placebo group at 2 years (P < .0001);
- Significantly greater improvement in overall control of cravings, and craving for savory foods, in a subset of patients, versus placebo, but questionnaire scores for positive mood and craving for sweet foods were similar in both groups.
- In STEP 8, mean body weight at 68 weeks was 15.8% lower with 2.4 mg/week subcutaneous semaglutide plus lifestyle changes versus 6.4% lower with 3.0 mg/day subcutaneous liraglutide plus lifestyle changes (P < .001).
Can treat to a target weight-loss range
The undiminished weight loss efficacy in the 2-year data for STEP 5 “portends well,” said W. Timothy Garvey, MD, following his presentation of the results.
“I think this is a new era in obesity care,” said Dr. Garvey, director of the diabetes research center at the University of Alabama at Birmingham. Semaglutide “essentially doubles weight loss efficacy” compared to the other approved pharmacotherapies for obesity.
With this degree of potential weight loss, clinicians “can use weight as a biomarker and treat to a target [weight-loss] range,” he said.
Expounding on this in an interview, Dr. Garvey noted that, as stated in the 2016 American Association of Clinical Endocrinologists (AACE) and American College of Endocrinology (ACE) clinical practice guidelines for medical care of patients with obesity, of which he was lead author, “the objective of care in obesity is to increase health of patients and prevent or treat complications.”
Semaglutide “can treat to a range of weight loss of 10% to 20% in the majority of patients,” which is associated with improvements in cardiovascular and metabolic risk factors.
In STEP 5, of the 51% of patients in the semaglutide group who had prediabetes at enrollment, 80% had normal glycemia at 2 years; however, the trial was not powered nor designed to investigate this.
More data are needed to inform long-term care decisions. The ongoing SELECT cardiovascular outcomes trial of semaglutide, with expected primary study completion on Sept. 28, 2023, should provide more information.
Weight loss plus reduced cravings
In another presentation, Sean Wharton, MD, PharmD, said, “In adults with overweight or obesity, substantial weight loss with semaglutide 2.4 mg was accompanied by short- and long-term improvements in control of eating.”
“Most patients living with obesity who are attempting to decrease calories will have food cravings, based on the biological parameters of weight preservation,” Dr. Wharton, medical director at the Wharton Medical Clinic, in Hamilton, Ont., explained in an email.
The degree of craving varies from patient to patient, likely based on genetics, he added. Research in this field is still emerging.
“I believe that semaglutide 2.4 mg is a game-changer in the field of weight management, and it will change the dialogue for insurance plans and with policymakers regarding coverage for this medication,” said Dr. Wharton.
“The data from the STEP programs are very strong. I am certainly hoping for a change to bias against covering these medications that we have seen in the past,” he said.
Clinically meaningful weight loss
When presenting the STEP 8 findings, Domenica M. Rubino, MD, said: “Participants were significantly more likely to achieve clinically meaningful weight loss thresholds with semaglutide 2.4 mg versus liraglutide 3.0 mg, accompanied by greater improvements in cardiometabolic risk factors.”
For example, patients can have better mobility, which is important for quality of life, Dr. Rubino, director of the Washington Center for Weight Management and Research, Arlington, Virginia, noted.
A smaller percentage of patients respond to liraglutide, she added. Clinicians need to individualize treatment.
When asked, “How do you choose which medical therapy?” Dr. Rubino responded: “We sit and talk.” Finding the medical therapy that fits the patient depends on things such as the patient’s insurance coverage and ability to tolerate side effects such as dehydration, diarrhea, and nausea.
When asked, “How do you switch from liraglutide to semaglutide?” she noted that there are no current guidelines for this. “You have to be careful. Start on the lowest dose of Wegovy. Be cautious, conservative.”
Still early days, caveats remain
“The STEP trials as a group appear to be making the case that obesity may now be considered a medically manageable disease, based on the experience with semaglutide,” Julie R. Ingelfinger, MD, who was not involved with the research, commented in an email.
“STEP 5 and 8 may suggest that weight loss occurs and is sustainable in overweight persons without diabetes with one or more comorbidities or in obese persons without diabetes,” added Dr. Ingelfinger, professor of pediatrics, Harvard Medical School, consultant in pediatric nephrology, Massachusetts General Hospital, Boston, and deputy editor, The New England Journal of Medicine.
However, “even 2 years, in the case of STEP 5, and ~68 weeks in the case of STEP 8, may not be long enough to know whether semaglutide is as promising as these brief summaries (abstracts) suggest,” she cautioned.
“Obesity is a chronic condition, and very long-term therapy and management are required,” Dr. Ingelfinger continued.
“Further, it is hard to generalize when gastrointestinal adverse events are common in a study,” she said. For example, in STEP 8, they were just as common with semaglutide as with the comparator liraglutide, she noted.
“The racial and ethnic representativeness of these studies does not reflect population distributions in the U.S., limiting generalization,” she continued.
“So, there remain caveats in interpreting these data.”
STEP 5 weight loss efficacy and safety at 2 years
Garvey reported that STEP 5 was a phase 3b trial that randomized 304 adults in the United States, Canada, Hungary, Italy, and Spain, who were 18 years and older, with a body mass index (BMI) ≥27 kg/m2 with at least one weight-related comorbidity (hypertension, dyslipidemia, obstructive sleep apnea, or cardiovascular disease) or a BMI ≥30 kg/m2, without type 2 diabetes, to receive semaglutide or placebo plus lifestyle intervention.
Most participants were women (78%) and White (93%). On average, they were 47 years old, weighed 106 kg (223.7 pounds), had a BMI of 38.5 kg/m2, a waist circumference of 115.7 cm (45.6 inches), and an A1c of 5.7%.
A total of 87% of patients in the semaglutide group and 73% of patients in the placebo group completed the trial.
At 104 weeks, participants were more likely to lose ≥10%, ≥15%, and ≥20% of body weight with semaglutide versus placebo (61.8% vs. 13.3%, 52.1% vs. 7.0%, and 36.1% vs. 2.3%, respectively; P < .0001 for all).
Patients in the semaglutide group had greater health improvements in cardiovascular risk factors (waist circumference, systolic and diastolic blood pressure, and C-reactive protein) and metabolic risk factors (A1c, fasting plasma glucose, fasting serum insulin, and triglycerides) than those in the placebo group (P < .05 for all).
Safety and tolerability were consistent with adverse events seen with this drug class, with no new safety signals.
Control of eating questionnaire findings at 2 years in STEP 5
Dr. Wharton and colleagues assessed changes in responses to the Control of Eating questionnaire at baseline and at 20, 52, and 104 weeks in patients from the U.S. and Canada in the STEP 5 trial (88 patients in the semaglutide group and 86 patients in the placebo group).
The questionnaire consisted of 19 questions grouped into four categories: control of food cravings, craving for savory foods (salty and spicy, dairy, or starchy foods), craving for sweet foods (chocolate, sweet foods, or fruit/fruit juice), and positive mood.
At week 104, patients in the semaglutide group had significantly greater improvements in scores for craving for salty and spicy, dairy, and starchy foods, and resisting cravings.
Semaglutide versus liraglutide, 68-week efficacy and safety in STEP 8
STEP 8 randomized 338 U.S. adults without diabetes and a BMI of ≥27 kg/m2 plus one or more weight-related comorbidities or a BMI of ≥30 kg/m2 3:1 to semaglutide 2.4 mg once weekly (n = 126) or matching placebo, or 3:1 liraglutide 3.0 mg once daily (n = 127) or matching placebo, plus lifestyle intervention.
Most participants were women (78%) and were a mean age of 49, had a mean body weight of 104.5 kg, and had a mean BMI of 37.5 kg/m2.
In STEP 8, more participants achieved ≥10%, ≥15%, and ≥20% weight loss with semaglutide than with liraglutide (70.9% vs. 25.6%, 55.6% vs. 12.0%, and 38.5% vs. 6.0%, respectively; P < .001 for all odds ratios).
Semaglutide improved waist circumference, A1c, and C-reactive protein versus liraglutide (unadjusted P < .001 for all).
Gastrointestinal adverse events were reported by 84% and 83% of participants receiving semaglutide and liraglutide, respectively. Most events were mild/moderate and transient, with prevalence declining over time.
Fewer participants stopped treatment due to adverse events with semaglutide than liraglutide (3.2% vs. 12.6%).
Dr. Garvey has reported serving as a site principal investigator for multicentered clinical trials sponsored by his university and funded by Eli Lilly, Novo Nordisk, and Pfizer. Dr. Wharton has reported financial ties to Novo Nordisk, Bausch Health Canada, Eli Lily, and Boehringer Ingelheim Canada. Dr. Rubino has reported ties to Boehringer Ingelheim and AstraZeneca. Dr. Ingelfinger has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The STEP 5 clinical trial extends favorable weight loss from 1 year out to 2 years for the glucagon-like peptide-1 (GLP-1) agonist semaglutide (Wegovy, Novo Nordisk), given as a once-weekly 2.4-mg subcutaneous injection, and some food cravings were improved in a subgroup analysis.
In another study, STEP 8, weight loss was greater at 68 weeks with semaglutide subcutaneous injection than with a 3-mg daily subcutaneous injection of another GLP-1 agonist, liraglutide (Saxenda, Novo Nordisk), approved earlier for weight loss.
Researchers presented these promising outcomes, with no new safety signals, at ObesityWeek® 2021.
However, there is more to learn about the drug class, researchers agree. Follow-up is still relatively short for a chronic disease and many patients have gastrointestinal side effects with semaglutide, one expert cautions.
The key findings were:
In STEP 5, combined with lifestyle intervention (a reduced-calorie meal plan and advice about physical activity), weekly injection of 2.4 mg semaglutide led to:
- 15.2% weight loss, compared with 2.6% weight loss with placebo at 2 years (P < .0001);
- 77% of patients losing at least 5% of their weight, compared with 34% of patients in the placebo group at 2 years (P < .0001);
- Significantly greater improvement in overall control of cravings, and craving for savory foods, in a subset of patients, versus placebo, but questionnaire scores for positive mood and craving for sweet foods were similar in both groups.
- In STEP 8, mean body weight at 68 weeks was 15.8% lower with 2.4 mg/week subcutaneous semaglutide plus lifestyle changes versus 6.4% lower with 3.0 mg/day subcutaneous liraglutide plus lifestyle changes (P < .001).
Can treat to a target weight-loss range
The undiminished weight loss efficacy in the 2-year data for STEP 5 “portends well,” said W. Timothy Garvey, MD, following his presentation of the results.
“I think this is a new era in obesity care,” said Dr. Garvey, director of the diabetes research center at the University of Alabama at Birmingham. Semaglutide “essentially doubles weight loss efficacy” compared to the other approved pharmacotherapies for obesity.
With this degree of potential weight loss, clinicians “can use weight as a biomarker and treat to a target [weight-loss] range,” he said.
Expounding on this in an interview, Dr. Garvey noted that, as stated in the 2016 American Association of Clinical Endocrinologists (AACE) and American College of Endocrinology (ACE) clinical practice guidelines for medical care of patients with obesity, of which he was lead author, “the objective of care in obesity is to increase health of patients and prevent or treat complications.”
Semaglutide “can treat to a range of weight loss of 10% to 20% in the majority of patients,” which is associated with improvements in cardiovascular and metabolic risk factors.
In STEP 5, of the 51% of patients in the semaglutide group who had prediabetes at enrollment, 80% had normal glycemia at 2 years; however, the trial was not powered nor designed to investigate this.
More data are needed to inform long-term care decisions. The ongoing SELECT cardiovascular outcomes trial of semaglutide, with expected primary study completion on Sept. 28, 2023, should provide more information.
Weight loss plus reduced cravings
In another presentation, Sean Wharton, MD, PharmD, said, “In adults with overweight or obesity, substantial weight loss with semaglutide 2.4 mg was accompanied by short- and long-term improvements in control of eating.”
“Most patients living with obesity who are attempting to decrease calories will have food cravings, based on the biological parameters of weight preservation,” Dr. Wharton, medical director at the Wharton Medical Clinic, in Hamilton, Ont., explained in an email.
The degree of craving varies from patient to patient, likely based on genetics, he added. Research in this field is still emerging.
“I believe that semaglutide 2.4 mg is a game-changer in the field of weight management, and it will change the dialogue for insurance plans and with policymakers regarding coverage for this medication,” said Dr. Wharton.
“The data from the STEP programs are very strong. I am certainly hoping for a change to bias against covering these medications that we have seen in the past,” he said.
Clinically meaningful weight loss
When presenting the STEP 8 findings, Domenica M. Rubino, MD, said: “Participants were significantly more likely to achieve clinically meaningful weight loss thresholds with semaglutide 2.4 mg versus liraglutide 3.0 mg, accompanied by greater improvements in cardiometabolic risk factors.”
For example, patients can have better mobility, which is important for quality of life, Dr. Rubino, director of the Washington Center for Weight Management and Research, Arlington, Virginia, noted.
A smaller percentage of patients respond to liraglutide, she added. Clinicians need to individualize treatment.
When asked, “How do you choose which medical therapy?” Dr. Rubino responded: “We sit and talk.” Finding the medical therapy that fits the patient depends on things such as the patient’s insurance coverage and ability to tolerate side effects such as dehydration, diarrhea, and nausea.
When asked, “How do you switch from liraglutide to semaglutide?” she noted that there are no current guidelines for this. “You have to be careful. Start on the lowest dose of Wegovy. Be cautious, conservative.”
Still early days, caveats remain
“The STEP trials as a group appear to be making the case that obesity may now be considered a medically manageable disease, based on the experience with semaglutide,” Julie R. Ingelfinger, MD, who was not involved with the research, commented in an email.
“STEP 5 and 8 may suggest that weight loss occurs and is sustainable in overweight persons without diabetes with one or more comorbidities or in obese persons without diabetes,” added Dr. Ingelfinger, professor of pediatrics, Harvard Medical School, consultant in pediatric nephrology, Massachusetts General Hospital, Boston, and deputy editor, The New England Journal of Medicine.
However, “even 2 years, in the case of STEP 5, and ~68 weeks in the case of STEP 8, may not be long enough to know whether semaglutide is as promising as these brief summaries (abstracts) suggest,” she cautioned.
“Obesity is a chronic condition, and very long-term therapy and management are required,” Dr. Ingelfinger continued.
“Further, it is hard to generalize when gastrointestinal adverse events are common in a study,” she said. For example, in STEP 8, they were just as common with semaglutide as with the comparator liraglutide, she noted.
“The racial and ethnic representativeness of these studies does not reflect population distributions in the U.S., limiting generalization,” she continued.
“So, there remain caveats in interpreting these data.”
STEP 5 weight loss efficacy and safety at 2 years
Garvey reported that STEP 5 was a phase 3b trial that randomized 304 adults in the United States, Canada, Hungary, Italy, and Spain, who were 18 years and older, with a body mass index (BMI) ≥27 kg/m2 with at least one weight-related comorbidity (hypertension, dyslipidemia, obstructive sleep apnea, or cardiovascular disease) or a BMI ≥30 kg/m2, without type 2 diabetes, to receive semaglutide or placebo plus lifestyle intervention.
Most participants were women (78%) and White (93%). On average, they were 47 years old, weighed 106 kg (223.7 pounds), had a BMI of 38.5 kg/m2, a waist circumference of 115.7 cm (45.6 inches), and an A1c of 5.7%.
A total of 87% of patients in the semaglutide group and 73% of patients in the placebo group completed the trial.
At 104 weeks, participants were more likely to lose ≥10%, ≥15%, and ≥20% of body weight with semaglutide versus placebo (61.8% vs. 13.3%, 52.1% vs. 7.0%, and 36.1% vs. 2.3%, respectively; P < .0001 for all).
Patients in the semaglutide group had greater health improvements in cardiovascular risk factors (waist circumference, systolic and diastolic blood pressure, and C-reactive protein) and metabolic risk factors (A1c, fasting plasma glucose, fasting serum insulin, and triglycerides) than those in the placebo group (P < .05 for all).
Safety and tolerability were consistent with adverse events seen with this drug class, with no new safety signals.
Control of eating questionnaire findings at 2 years in STEP 5
Dr. Wharton and colleagues assessed changes in responses to the Control of Eating questionnaire at baseline and at 20, 52, and 104 weeks in patients from the U.S. and Canada in the STEP 5 trial (88 patients in the semaglutide group and 86 patients in the placebo group).
The questionnaire consisted of 19 questions grouped into four categories: control of food cravings, craving for savory foods (salty and spicy, dairy, or starchy foods), craving for sweet foods (chocolate, sweet foods, or fruit/fruit juice), and positive mood.
At week 104, patients in the semaglutide group had significantly greater improvements in scores for craving for salty and spicy, dairy, and starchy foods, and resisting cravings.
Semaglutide versus liraglutide, 68-week efficacy and safety in STEP 8
STEP 8 randomized 338 U.S. adults without diabetes and a BMI of ≥27 kg/m2 plus one or more weight-related comorbidities or a BMI of ≥30 kg/m2 3:1 to semaglutide 2.4 mg once weekly (n = 126) or matching placebo, or 3:1 liraglutide 3.0 mg once daily (n = 127) or matching placebo, plus lifestyle intervention.
Most participants were women (78%) and were a mean age of 49, had a mean body weight of 104.5 kg, and had a mean BMI of 37.5 kg/m2.
In STEP 8, more participants achieved ≥10%, ≥15%, and ≥20% weight loss with semaglutide than with liraglutide (70.9% vs. 25.6%, 55.6% vs. 12.0%, and 38.5% vs. 6.0%, respectively; P < .001 for all odds ratios).
Semaglutide improved waist circumference, A1c, and C-reactive protein versus liraglutide (unadjusted P < .001 for all).
Gastrointestinal adverse events were reported by 84% and 83% of participants receiving semaglutide and liraglutide, respectively. Most events were mild/moderate and transient, with prevalence declining over time.
Fewer participants stopped treatment due to adverse events with semaglutide than liraglutide (3.2% vs. 12.6%).
Dr. Garvey has reported serving as a site principal investigator for multicentered clinical trials sponsored by his university and funded by Eli Lilly, Novo Nordisk, and Pfizer. Dr. Wharton has reported financial ties to Novo Nordisk, Bausch Health Canada, Eli Lily, and Boehringer Ingelheim Canada. Dr. Rubino has reported ties to Boehringer Ingelheim and AstraZeneca. Dr. Ingelfinger has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Liraglutide effective against weight regain after gastric bypass
The glucagon-like peptide-1 (GLP-1) receptor agonist liraglutide (Saxenda, Novo Nordisk) was safe and effective for treating weight regain after Roux-en-Y gastric bypass (RYGB), in a randomized controlled trial.
That is, 132 patients who had lost at least 25% of their initial weight after RYGB and then gained at least 10% back were randomized 2:1 to receive liraglutide plus frequent lifestyle advice from a registered dietitian or lifestyle advice alone.
After a year, 69%, 48%, and 24% of patients who had received liraglutide lost at least 5%, 10%, and 15% of their study entry weight, respectively. In contrast, only 5% of patients in the control group lost at least 5% of their weight and none lost at least 10% of their weight.
“Liraglutide 3.0 mg/day, with lifestyle modification, was significantly more effective than placebo in treating weight regain after RYGB without increased risk of serious adverse events,” Holly F. Lofton, MD, summarized this week in an oral session at ObesityWeek®, the annual meeting of The Obesity Society.
Dr. Lofton, a clinical associate professor of surgery and medicine, and director, weight management program, NYU, Langone Health, explained to this news organization that she initiated the study after attending a “packed” session about post bariatric surgery weight regain at a prior American Society of Metabolic and Bariatric Surgery conference.
“The lecturers recommended conservative measures (such as reiterating the diet recommendations, exercise, [and] counseling), and revisional surgeries,” she said in an email, but at the time “there was no literature that provided direction on which pharmacotherapies are best for this population.”
It was known that decreases in endogenous GLP-1 levels coincide with weight regain, and liraglutide (Saxenda) was the only GLP-1 agonist approved for chronic weight management at the time, so she devised the current study protocol.
The findings are especially helpful for patients who are not candidates for bariatric surgery revisions, she noted. Further research is needed to investigate the effect of newer GLP-1 agonists, such as semaglutide (Wegovy), on weight regain following different types of bariatric surgery.
Asked to comment, Wendy C. King, PhD, who was not involved with this research, said that more than two-thirds of patients treated with 3 mg/day subcutaneous liraglutide injections in the current study lost at least 5% of their initial weight a year later, and 20% of them attained a weight as low as, or lower than, their lowest weight after bariatric surgery (nadir weight).
“The fact that both groups received lifestyle counseling from registered dietitians for just over a year, but only patients in the liraglutide group lost weight, on average, speaks to the difficulty of losing weight following weight regain post–bariatric surgery,” added Dr. King, an associate professor of epidemiology at the University of Pittsburgh, Pennsylvania.
This study “provides data that may help clinicians and patients understand the potential effect of adding liraglutide 3.0 mg/day to their weight loss efforts,” she told this news organization in an email.
However, “given that 42% of those on liraglutide reported gastrointestinal-related side effects, patients should also be counseled on this potential outcome and given suggestions for how to minimize such side effects,” Dr. King suggested.
Weight regain common, repeat surgery entails risk
Weight regain is common even years after bariatric surgery. Repeat surgery entails some risk, and lifestyle approaches alone are rarely successful in reversing weight regain, Dr. Lofton told the audience.
The researchers enrolled 132 adults who had a mean weight of 134 kg (295 pounds) when they underwent RYGB, and who lost at least 25% of their initial weight (mean weight loss of 38%) after the surgery, but who also regained at least 10% of their initial weight.
At enrollment of the current study (baseline), the patients had had RYGB 18 months to 10 years earlier (mean 5.7 years earlier) and now had a mean weight of 99 kg (218 pounds) and a mean BMI of 35.6 kg/m2. None of the patients had diabetes.
The patients were randomized to receive liraglutide (n = 89, 84% women) or placebo (n = 43, 88% women) for 56 weeks.
They were a mean age of 48 years, and about 59% were White and 25% were Black.
All patients had clinic visits every 3 months where they received lifestyle counseling from a registered dietitian.
At 12 months, patients in the liraglutide group had lost a mean of 8.8% of their baseline weight, whereas those in the placebo group had gained a mean of 1.48% of their baseline weight.
There were no significant between-group differences in cardiometabolic variables.
None of the patients in the control group attained a weight that was as low as their nadir weight after RYGB.
The rates of nausea (25%), constipation (16%), and abdominal pain (10%) in the liraglutide group were higher than in the placebo group (7%, 14%, and 5%, respectively) but similar to rates of gastrointestinal side effects in other trials of this agent.
Dr. Lofton has disclosed receiving consulting fees and being on a speaker bureau for Novo Nordisk and receiving research funds from Boehringer Ingelheim, Eli Lilly, and Novo Nordisk. Dr. King has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The glucagon-like peptide-1 (GLP-1) receptor agonist liraglutide (Saxenda, Novo Nordisk) was safe and effective for treating weight regain after Roux-en-Y gastric bypass (RYGB), in a randomized controlled trial.
That is, 132 patients who had lost at least 25% of their initial weight after RYGB and then gained at least 10% back were randomized 2:1 to receive liraglutide plus frequent lifestyle advice from a registered dietitian or lifestyle advice alone.
After a year, 69%, 48%, and 24% of patients who had received liraglutide lost at least 5%, 10%, and 15% of their study entry weight, respectively. In contrast, only 5% of patients in the control group lost at least 5% of their weight and none lost at least 10% of their weight.
“Liraglutide 3.0 mg/day, with lifestyle modification, was significantly more effective than placebo in treating weight regain after RYGB without increased risk of serious adverse events,” Holly F. Lofton, MD, summarized this week in an oral session at ObesityWeek®, the annual meeting of The Obesity Society.
Dr. Lofton, a clinical associate professor of surgery and medicine, and director, weight management program, NYU, Langone Health, explained to this news organization that she initiated the study after attending a “packed” session about post bariatric surgery weight regain at a prior American Society of Metabolic and Bariatric Surgery conference.
“The lecturers recommended conservative measures (such as reiterating the diet recommendations, exercise, [and] counseling), and revisional surgeries,” she said in an email, but at the time “there was no literature that provided direction on which pharmacotherapies are best for this population.”
It was known that decreases in endogenous GLP-1 levels coincide with weight regain, and liraglutide (Saxenda) was the only GLP-1 agonist approved for chronic weight management at the time, so she devised the current study protocol.
The findings are especially helpful for patients who are not candidates for bariatric surgery revisions, she noted. Further research is needed to investigate the effect of newer GLP-1 agonists, such as semaglutide (Wegovy), on weight regain following different types of bariatric surgery.
Asked to comment, Wendy C. King, PhD, who was not involved with this research, said that more than two-thirds of patients treated with 3 mg/day subcutaneous liraglutide injections in the current study lost at least 5% of their initial weight a year later, and 20% of them attained a weight as low as, or lower than, their lowest weight after bariatric surgery (nadir weight).
“The fact that both groups received lifestyle counseling from registered dietitians for just over a year, but only patients in the liraglutide group lost weight, on average, speaks to the difficulty of losing weight following weight regain post–bariatric surgery,” added Dr. King, an associate professor of epidemiology at the University of Pittsburgh, Pennsylvania.
This study “provides data that may help clinicians and patients understand the potential effect of adding liraglutide 3.0 mg/day to their weight loss efforts,” she told this news organization in an email.
However, “given that 42% of those on liraglutide reported gastrointestinal-related side effects, patients should also be counseled on this potential outcome and given suggestions for how to minimize such side effects,” Dr. King suggested.
Weight regain common, repeat surgery entails risk
Weight regain is common even years after bariatric surgery. Repeat surgery entails some risk, and lifestyle approaches alone are rarely successful in reversing weight regain, Dr. Lofton told the audience.
The researchers enrolled 132 adults who had a mean weight of 134 kg (295 pounds) when they underwent RYGB, and who lost at least 25% of their initial weight (mean weight loss of 38%) after the surgery, but who also regained at least 10% of their initial weight.
At enrollment of the current study (baseline), the patients had had RYGB 18 months to 10 years earlier (mean 5.7 years earlier) and now had a mean weight of 99 kg (218 pounds) and a mean BMI of 35.6 kg/m2. None of the patients had diabetes.
The patients were randomized to receive liraglutide (n = 89, 84% women) or placebo (n = 43, 88% women) for 56 weeks.
They were a mean age of 48 years, and about 59% were White and 25% were Black.
All patients had clinic visits every 3 months where they received lifestyle counseling from a registered dietitian.
At 12 months, patients in the liraglutide group had lost a mean of 8.8% of their baseline weight, whereas those in the placebo group had gained a mean of 1.48% of their baseline weight.
There were no significant between-group differences in cardiometabolic variables.
None of the patients in the control group attained a weight that was as low as their nadir weight after RYGB.
The rates of nausea (25%), constipation (16%), and abdominal pain (10%) in the liraglutide group were higher than in the placebo group (7%, 14%, and 5%, respectively) but similar to rates of gastrointestinal side effects in other trials of this agent.
Dr. Lofton has disclosed receiving consulting fees and being on a speaker bureau for Novo Nordisk and receiving research funds from Boehringer Ingelheim, Eli Lilly, and Novo Nordisk. Dr. King has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The glucagon-like peptide-1 (GLP-1) receptor agonist liraglutide (Saxenda, Novo Nordisk) was safe and effective for treating weight regain after Roux-en-Y gastric bypass (RYGB), in a randomized controlled trial.
That is, 132 patients who had lost at least 25% of their initial weight after RYGB and then gained at least 10% back were randomized 2:1 to receive liraglutide plus frequent lifestyle advice from a registered dietitian or lifestyle advice alone.
After a year, 69%, 48%, and 24% of patients who had received liraglutide lost at least 5%, 10%, and 15% of their study entry weight, respectively. In contrast, only 5% of patients in the control group lost at least 5% of their weight and none lost at least 10% of their weight.
“Liraglutide 3.0 mg/day, with lifestyle modification, was significantly more effective than placebo in treating weight regain after RYGB without increased risk of serious adverse events,” Holly F. Lofton, MD, summarized this week in an oral session at ObesityWeek®, the annual meeting of The Obesity Society.
Dr. Lofton, a clinical associate professor of surgery and medicine, and director, weight management program, NYU, Langone Health, explained to this news organization that she initiated the study after attending a “packed” session about post bariatric surgery weight regain at a prior American Society of Metabolic and Bariatric Surgery conference.
“The lecturers recommended conservative measures (such as reiterating the diet recommendations, exercise, [and] counseling), and revisional surgeries,” she said in an email, but at the time “there was no literature that provided direction on which pharmacotherapies are best for this population.”
It was known that decreases in endogenous GLP-1 levels coincide with weight regain, and liraglutide (Saxenda) was the only GLP-1 agonist approved for chronic weight management at the time, so she devised the current study protocol.
The findings are especially helpful for patients who are not candidates for bariatric surgery revisions, she noted. Further research is needed to investigate the effect of newer GLP-1 agonists, such as semaglutide (Wegovy), on weight regain following different types of bariatric surgery.
Asked to comment, Wendy C. King, PhD, who was not involved with this research, said that more than two-thirds of patients treated with 3 mg/day subcutaneous liraglutide injections in the current study lost at least 5% of their initial weight a year later, and 20% of them attained a weight as low as, or lower than, their lowest weight after bariatric surgery (nadir weight).
“The fact that both groups received lifestyle counseling from registered dietitians for just over a year, but only patients in the liraglutide group lost weight, on average, speaks to the difficulty of losing weight following weight regain post–bariatric surgery,” added Dr. King, an associate professor of epidemiology at the University of Pittsburgh, Pennsylvania.
This study “provides data that may help clinicians and patients understand the potential effect of adding liraglutide 3.0 mg/day to their weight loss efforts,” she told this news organization in an email.
However, “given that 42% of those on liraglutide reported gastrointestinal-related side effects, patients should also be counseled on this potential outcome and given suggestions for how to minimize such side effects,” Dr. King suggested.
Weight regain common, repeat surgery entails risk
Weight regain is common even years after bariatric surgery. Repeat surgery entails some risk, and lifestyle approaches alone are rarely successful in reversing weight regain, Dr. Lofton told the audience.
The researchers enrolled 132 adults who had a mean weight of 134 kg (295 pounds) when they underwent RYGB, and who lost at least 25% of their initial weight (mean weight loss of 38%) after the surgery, but who also regained at least 10% of their initial weight.
At enrollment of the current study (baseline), the patients had had RYGB 18 months to 10 years earlier (mean 5.7 years earlier) and now had a mean weight of 99 kg (218 pounds) and a mean BMI of 35.6 kg/m2. None of the patients had diabetes.
The patients were randomized to receive liraglutide (n = 89, 84% women) or placebo (n = 43, 88% women) for 56 weeks.
They were a mean age of 48 years, and about 59% were White and 25% were Black.
All patients had clinic visits every 3 months where they received lifestyle counseling from a registered dietitian.
At 12 months, patients in the liraglutide group had lost a mean of 8.8% of their baseline weight, whereas those in the placebo group had gained a mean of 1.48% of their baseline weight.
There were no significant between-group differences in cardiometabolic variables.
None of the patients in the control group attained a weight that was as low as their nadir weight after RYGB.
The rates of nausea (25%), constipation (16%), and abdominal pain (10%) in the liraglutide group were higher than in the placebo group (7%, 14%, and 5%, respectively) but similar to rates of gastrointestinal side effects in other trials of this agent.
Dr. Lofton has disclosed receiving consulting fees and being on a speaker bureau for Novo Nordisk and receiving research funds from Boehringer Ingelheim, Eli Lilly, and Novo Nordisk. Dr. King has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM OBESITY WEEK 2021
‘If obesity were diabetes or cancer, how would you approach it?’
“When considering the challenges of obesity, ask yourself: ‘If it were diabetes, cancer, HIV, or Alzheimer’s, how would you discuss it, approach it, assess it, treat it?’” Lee M. Kaplan, MD, PhD, asked the audience of health care professionals during ObesityWeek®, the annual meeting of The Obesity Society.
“And then do it for obesity, using the full spectrum of tools at our disposal,” he advised.
This was the takeaway that Dr. Kaplan, director of the Obesity, Metabolism, and Nutrition Institute at Massachusetts General Hospital and associate professor, Harvard Medical School, Boston, left the audience with at the end of his lecture entitled, “What does the future of obesity care look like?”
Invited to summarize his main points, Dr. Kaplan told this news organization in an interview that practitioners caring for patients with obesity need to first “recognize that obesity is a disease” caused by dysfunction of the metabolic system that regulates body fat – in the same way immune dysregulation can lead to asthma.
Second, “we are finally developing noninvasive therapies that are more effective,” he noted, referring to the recently approved semaglutide, and even more potent weight-loss therapies that could be on the market within 3 years, so that weight-loss outcomes with pharmacotherapy are approaching those with bariatric surgery.
Third, it is important that patients with obesity get “broad and equitable access” to treatment, and health care practitioners need to be on the same page and have a “shared understanding” of which treatments are appropriate for individual patients, “just as we do for other diseases.”
Need for a shared understanding
“Dr. Kaplan really brought home the idea that we all need a shared understanding of what obesity is – and what it is not,” agreed symposium moderator Donna H. Ryan, MD, in an email.
“He underscored the biologic basis of obesity,” noted Dr. Ryan, professor emerita at Pennington Biomedical Research Center in Baton Rouge, Louisiana, and associate editor-in-chief of Obesity, the official journal of The Obesity Society.
“It is a dysregulation of the body’s weight (especially adipose tissue) regulatory system,” she continued. “The body responds to powerful environmental pressures that produce excess energy balance, and we store that as fat and defend our highest fat mass. This makes obesity a disease, a chronic disease that requires a medical approach to reverse. It’s not a cosmetic problem, it’s a medical problem,” she emphasized.
There is so much misinformation out there about obesity, according to Dr. Ryan.
“People think it’s a lack of willpower, and even patients blame themselves for not being able to lose weight and keep it off. It’s not their fault! It’s biology.”
Although the supplement industry and fad diets falsely promise fast results, there is no magic diet, she continued.
“But we have made progress based on understanding the biologic basis of obesity and have new medications that offer real hope for patients.”
“With 42% of U.S. adults having a BMI that qualifies as obesity, we need a concerted and broad effort to address this problem, and that starts with everybody on the same page as to what obesity is ... a shared understanding of the biologic basis of obesity. It’s time to take obesity seriously,” she summarized, echoing Dr. Kaplan.
A question of biology
“Obesity results from inappropriate pathophysiological regulation of body fat mass,” when the body defends adiposity, Dr. Kaplan explained at the start of his lecture.
The treatment strategy for obesity has always been a stepwise approach starting with lifestyle changes, then pharmacotherapy, then possibly bariatric surgery – each step with a potentially greater chance of weight loss. But now, he explained, medicine is on the verge of having an armamentarium of more potent weight-loss medications.
Compared with phentermine/topiramate, orlistat, naltrexone/bupropion, and liraglutide – which roughly might provide 5% to 10% weight loss, the glucagon-like peptide-1 (GLP-1) agonist semaglutide 2.4 mg/week (Wegovy, Novo Nordisk), approved by the U.S. Food and Drug Association in June, provides almost double this potential weight loss.
And two new agents that could provide “never seen before weight loss” of 25% could potentially enter the marketplace by 2025: the amylin agonist cagrilintide (Novo Nordisk) and the twincretin tirzepatide (Eli Lilly) (a combined glucose-dependent insulinotropic polypeptide [GIP] and GLP-1 receptor agonist).
In addition, when liraglutide comes off patent, a generic version could potentially be introduced, and combined generic liraglutide plus generic phentermine/topiramate could be a less expensive weight-loss treatment option in the future, he noted.
One size does not fit all
Importantly, weight loss varies widely among individual patients.
A graph of potential weight loss with different treatments (for example, bariatric surgery or liraglutide) versus the percentage of patients that attain the weight losses is roughly bell-shaped, Dr. Kaplan explained. For example, in the STEP1 trial of semaglutide, roughly 7.1% of patients lost less than 5% of their initial weight, 25% of patients lost 20% to 30%, and 10.8% of patients lost 30% or more; that is, patients at the higher end had weight loss comparable to that seen with bariatric surgery
Adding pharmacotherapy after bariatric surgery could be synergistic. For example, in the GRAVITAS study of patients with type 2 diabetes who had gastric bypass surgery, those who received liraglutide after surgery had augmented weight loss compared with those who received placebo.
People at a cocktail party might come up to him and say, “I’d like to lose 5 pounds, 10 pounds,” Dr. Kaplan related in the Q&A session.
“That’s not obesity,” he emphasized. Obesity is excess body fat that poses a risk to health. A person with obesity may have 50 or more excess pounds, and the body is trying to defend this weight.
“If we want to treat obesity more effectively, we have to fully understand why it is a disease and how that disease differs from the cultural desire for thinness,” he reiterated.
“We have to keep the needs and goals of all people living with obesity foremost in our minds, even if many of them have been previously misled by the bias, stigma, blame, and discrimination that surrounds them.”
“We need to re-evaluate what we think we know about obesity and open our minds to new ideas,” he added.
Dr. Kaplan has reported financial ties to Eli Lilly, Gelesis, GI Dynamics, IntelliHealth, Johnson & Johnson, Novo Nordisk, Pfizer, and Rhythm Pharmaceuticals. Dr. Ryan has ties to numerous Novo Nordisk, Pfizer, and several other pharmaceutical companies, including having an ownership interest in Gila Therapeutics, Xeno Biosciences, Epitomee, Calibrate, Roman, and Scientific Intake.
A version of this article first appeared on Medscape.com.
“When considering the challenges of obesity, ask yourself: ‘If it were diabetes, cancer, HIV, or Alzheimer’s, how would you discuss it, approach it, assess it, treat it?’” Lee M. Kaplan, MD, PhD, asked the audience of health care professionals during ObesityWeek®, the annual meeting of The Obesity Society.
“And then do it for obesity, using the full spectrum of tools at our disposal,” he advised.
This was the takeaway that Dr. Kaplan, director of the Obesity, Metabolism, and Nutrition Institute at Massachusetts General Hospital and associate professor, Harvard Medical School, Boston, left the audience with at the end of his lecture entitled, “What does the future of obesity care look like?”
Invited to summarize his main points, Dr. Kaplan told this news organization in an interview that practitioners caring for patients with obesity need to first “recognize that obesity is a disease” caused by dysfunction of the metabolic system that regulates body fat – in the same way immune dysregulation can lead to asthma.
Second, “we are finally developing noninvasive therapies that are more effective,” he noted, referring to the recently approved semaglutide, and even more potent weight-loss therapies that could be on the market within 3 years, so that weight-loss outcomes with pharmacotherapy are approaching those with bariatric surgery.
Third, it is important that patients with obesity get “broad and equitable access” to treatment, and health care practitioners need to be on the same page and have a “shared understanding” of which treatments are appropriate for individual patients, “just as we do for other diseases.”
Need for a shared understanding
“Dr. Kaplan really brought home the idea that we all need a shared understanding of what obesity is – and what it is not,” agreed symposium moderator Donna H. Ryan, MD, in an email.
“He underscored the biologic basis of obesity,” noted Dr. Ryan, professor emerita at Pennington Biomedical Research Center in Baton Rouge, Louisiana, and associate editor-in-chief of Obesity, the official journal of The Obesity Society.
“It is a dysregulation of the body’s weight (especially adipose tissue) regulatory system,” she continued. “The body responds to powerful environmental pressures that produce excess energy balance, and we store that as fat and defend our highest fat mass. This makes obesity a disease, a chronic disease that requires a medical approach to reverse. It’s not a cosmetic problem, it’s a medical problem,” she emphasized.
There is so much misinformation out there about obesity, according to Dr. Ryan.
“People think it’s a lack of willpower, and even patients blame themselves for not being able to lose weight and keep it off. It’s not their fault! It’s biology.”
Although the supplement industry and fad diets falsely promise fast results, there is no magic diet, she continued.
“But we have made progress based on understanding the biologic basis of obesity and have new medications that offer real hope for patients.”
“With 42% of U.S. adults having a BMI that qualifies as obesity, we need a concerted and broad effort to address this problem, and that starts with everybody on the same page as to what obesity is ... a shared understanding of the biologic basis of obesity. It’s time to take obesity seriously,” she summarized, echoing Dr. Kaplan.
A question of biology
“Obesity results from inappropriate pathophysiological regulation of body fat mass,” when the body defends adiposity, Dr. Kaplan explained at the start of his lecture.
The treatment strategy for obesity has always been a stepwise approach starting with lifestyle changes, then pharmacotherapy, then possibly bariatric surgery – each step with a potentially greater chance of weight loss. But now, he explained, medicine is on the verge of having an armamentarium of more potent weight-loss medications.
Compared with phentermine/topiramate, orlistat, naltrexone/bupropion, and liraglutide – which roughly might provide 5% to 10% weight loss, the glucagon-like peptide-1 (GLP-1) agonist semaglutide 2.4 mg/week (Wegovy, Novo Nordisk), approved by the U.S. Food and Drug Association in June, provides almost double this potential weight loss.
And two new agents that could provide “never seen before weight loss” of 25% could potentially enter the marketplace by 2025: the amylin agonist cagrilintide (Novo Nordisk) and the twincretin tirzepatide (Eli Lilly) (a combined glucose-dependent insulinotropic polypeptide [GIP] and GLP-1 receptor agonist).
In addition, when liraglutide comes off patent, a generic version could potentially be introduced, and combined generic liraglutide plus generic phentermine/topiramate could be a less expensive weight-loss treatment option in the future, he noted.
One size does not fit all
Importantly, weight loss varies widely among individual patients.
A graph of potential weight loss with different treatments (for example, bariatric surgery or liraglutide) versus the percentage of patients that attain the weight losses is roughly bell-shaped, Dr. Kaplan explained. For example, in the STEP1 trial of semaglutide, roughly 7.1% of patients lost less than 5% of their initial weight, 25% of patients lost 20% to 30%, and 10.8% of patients lost 30% or more; that is, patients at the higher end had weight loss comparable to that seen with bariatric surgery
Adding pharmacotherapy after bariatric surgery could be synergistic. For example, in the GRAVITAS study of patients with type 2 diabetes who had gastric bypass surgery, those who received liraglutide after surgery had augmented weight loss compared with those who received placebo.
People at a cocktail party might come up to him and say, “I’d like to lose 5 pounds, 10 pounds,” Dr. Kaplan related in the Q&A session.
“That’s not obesity,” he emphasized. Obesity is excess body fat that poses a risk to health. A person with obesity may have 50 or more excess pounds, and the body is trying to defend this weight.
“If we want to treat obesity more effectively, we have to fully understand why it is a disease and how that disease differs from the cultural desire for thinness,” he reiterated.
“We have to keep the needs and goals of all people living with obesity foremost in our minds, even if many of them have been previously misled by the bias, stigma, blame, and discrimination that surrounds them.”
“We need to re-evaluate what we think we know about obesity and open our minds to new ideas,” he added.
Dr. Kaplan has reported financial ties to Eli Lilly, Gelesis, GI Dynamics, IntelliHealth, Johnson & Johnson, Novo Nordisk, Pfizer, and Rhythm Pharmaceuticals. Dr. Ryan has ties to numerous Novo Nordisk, Pfizer, and several other pharmaceutical companies, including having an ownership interest in Gila Therapeutics, Xeno Biosciences, Epitomee, Calibrate, Roman, and Scientific Intake.
A version of this article first appeared on Medscape.com.
“When considering the challenges of obesity, ask yourself: ‘If it were diabetes, cancer, HIV, or Alzheimer’s, how would you discuss it, approach it, assess it, treat it?’” Lee M. Kaplan, MD, PhD, asked the audience of health care professionals during ObesityWeek®, the annual meeting of The Obesity Society.
“And then do it for obesity, using the full spectrum of tools at our disposal,” he advised.
This was the takeaway that Dr. Kaplan, director of the Obesity, Metabolism, and Nutrition Institute at Massachusetts General Hospital and associate professor, Harvard Medical School, Boston, left the audience with at the end of his lecture entitled, “What does the future of obesity care look like?”
Invited to summarize his main points, Dr. Kaplan told this news organization in an interview that practitioners caring for patients with obesity need to first “recognize that obesity is a disease” caused by dysfunction of the metabolic system that regulates body fat – in the same way immune dysregulation can lead to asthma.
Second, “we are finally developing noninvasive therapies that are more effective,” he noted, referring to the recently approved semaglutide, and even more potent weight-loss therapies that could be on the market within 3 years, so that weight-loss outcomes with pharmacotherapy are approaching those with bariatric surgery.
Third, it is important that patients with obesity get “broad and equitable access” to treatment, and health care practitioners need to be on the same page and have a “shared understanding” of which treatments are appropriate for individual patients, “just as we do for other diseases.”
Need for a shared understanding
“Dr. Kaplan really brought home the idea that we all need a shared understanding of what obesity is – and what it is not,” agreed symposium moderator Donna H. Ryan, MD, in an email.
“He underscored the biologic basis of obesity,” noted Dr. Ryan, professor emerita at Pennington Biomedical Research Center in Baton Rouge, Louisiana, and associate editor-in-chief of Obesity, the official journal of The Obesity Society.
“It is a dysregulation of the body’s weight (especially adipose tissue) regulatory system,” she continued. “The body responds to powerful environmental pressures that produce excess energy balance, and we store that as fat and defend our highest fat mass. This makes obesity a disease, a chronic disease that requires a medical approach to reverse. It’s not a cosmetic problem, it’s a medical problem,” she emphasized.
There is so much misinformation out there about obesity, according to Dr. Ryan.
“People think it’s a lack of willpower, and even patients blame themselves for not being able to lose weight and keep it off. It’s not their fault! It’s biology.”
Although the supplement industry and fad diets falsely promise fast results, there is no magic diet, she continued.
“But we have made progress based on understanding the biologic basis of obesity and have new medications that offer real hope for patients.”
“With 42% of U.S. adults having a BMI that qualifies as obesity, we need a concerted and broad effort to address this problem, and that starts with everybody on the same page as to what obesity is ... a shared understanding of the biologic basis of obesity. It’s time to take obesity seriously,” she summarized, echoing Dr. Kaplan.
A question of biology
“Obesity results from inappropriate pathophysiological regulation of body fat mass,” when the body defends adiposity, Dr. Kaplan explained at the start of his lecture.
The treatment strategy for obesity has always been a stepwise approach starting with lifestyle changes, then pharmacotherapy, then possibly bariatric surgery – each step with a potentially greater chance of weight loss. But now, he explained, medicine is on the verge of having an armamentarium of more potent weight-loss medications.
Compared with phentermine/topiramate, orlistat, naltrexone/bupropion, and liraglutide – which roughly might provide 5% to 10% weight loss, the glucagon-like peptide-1 (GLP-1) agonist semaglutide 2.4 mg/week (Wegovy, Novo Nordisk), approved by the U.S. Food and Drug Association in June, provides almost double this potential weight loss.
And two new agents that could provide “never seen before weight loss” of 25% could potentially enter the marketplace by 2025: the amylin agonist cagrilintide (Novo Nordisk) and the twincretin tirzepatide (Eli Lilly) (a combined glucose-dependent insulinotropic polypeptide [GIP] and GLP-1 receptor agonist).
In addition, when liraglutide comes off patent, a generic version could potentially be introduced, and combined generic liraglutide plus generic phentermine/topiramate could be a less expensive weight-loss treatment option in the future, he noted.
One size does not fit all
Importantly, weight loss varies widely among individual patients.
A graph of potential weight loss with different treatments (for example, bariatric surgery or liraglutide) versus the percentage of patients that attain the weight losses is roughly bell-shaped, Dr. Kaplan explained. For example, in the STEP1 trial of semaglutide, roughly 7.1% of patients lost less than 5% of their initial weight, 25% of patients lost 20% to 30%, and 10.8% of patients lost 30% or more; that is, patients at the higher end had weight loss comparable to that seen with bariatric surgery
Adding pharmacotherapy after bariatric surgery could be synergistic. For example, in the GRAVITAS study of patients with type 2 diabetes who had gastric bypass surgery, those who received liraglutide after surgery had augmented weight loss compared with those who received placebo.
People at a cocktail party might come up to him and say, “I’d like to lose 5 pounds, 10 pounds,” Dr. Kaplan related in the Q&A session.
“That’s not obesity,” he emphasized. Obesity is excess body fat that poses a risk to health. A person with obesity may have 50 or more excess pounds, and the body is trying to defend this weight.
“If we want to treat obesity more effectively, we have to fully understand why it is a disease and how that disease differs from the cultural desire for thinness,” he reiterated.
“We have to keep the needs and goals of all people living with obesity foremost in our minds, even if many of them have been previously misled by the bias, stigma, blame, and discrimination that surrounds them.”
“We need to re-evaluate what we think we know about obesity and open our minds to new ideas,” he added.
Dr. Kaplan has reported financial ties to Eli Lilly, Gelesis, GI Dynamics, IntelliHealth, Johnson & Johnson, Novo Nordisk, Pfizer, and Rhythm Pharmaceuticals. Dr. Ryan has ties to numerous Novo Nordisk, Pfizer, and several other pharmaceutical companies, including having an ownership interest in Gila Therapeutics, Xeno Biosciences, Epitomee, Calibrate, Roman, and Scientific Intake.
A version of this article first appeared on Medscape.com.
FROM OBESITY WEEK 2020
Obesity interventions tied to colon cancer risk reduction
LAS VEGAS – People with obesity may be able to reduce their risk of colorectal cancer with weight loss surgery or medication, researchers say.
“We need to have conversations with our patients in the clinic and educate them that they have these resources available,” said Aakash Desai, MD, a hospitalist at MetroHealth Medical Center, Cleveland, in an interview with this news organization.
Dr. Desai and colleagues found that sleeve gastrectomy and four medications were associated with a reduced risk of colorectal cancer but Roux-en-Y gastrojejunostomy and orlistat were not.
Coauthor Zryan Shwani, MD, a gastroenterology fellow at Sibley Memorial Hospital, Washington, D.C., presented the findings here at the American College of Gastroenterology (ACG) 2021 Annual Scientific Meeting.
Working with an underserved population with high rates of obesity in northeastern Ohio, the researchers wondered how surgery and medication could affect these patients.
They analyzed data from the IBM Explorys clinical database, which compiles and standardizes data from electronic medical records on about 74 million patients from more than 300 U.S. hospitals. Consistent with previous studies, they determined that patients with obesity in the database were 2.5 times more likely than people with a healthy weight to be diagnosed with colorectal cancer (odds ratio, 2.48; 95% CI, 2.45-2.51).
Zeroing in on people who had weight loss interventions, they included adults aged 18-75 years who had undergone either Roux-en-Y gastrojejunostomy or sleeve gastrectomy, or had taken the medications liraglutide, orlistat, phentermine/topiramate, bupropion/naltrexone, or lorcaserin.
They excluded patients with Lynch syndrome, intestinal polyposis syndrome, a family history of gastrointestinal malignancy, inflammatory bowel disease, or tobacco or alcohol abuse. Patients who had taken one of the weight loss medications and also had type 2 diabetes were excluded. They did not include patients who had undergone gastric banding because it has become less popular.
For the weight loss medication group, they found 117,730 patients who met their criteria. For the surgery group, 43,050 patients met the criteria.
In analyzing the colorectal cancer rates, they included only diagnoses of malignant neoplasms made 2 years after the interventions.
They compared these patients to a control group of 52,540 people matched in age, with a body mass index (BMI) greater than 30 kg/m2 who did not undergo weight loss surgery or take weight loss medication.
Among the 9,370 patients who underwent Roux-en-Y gastrojejunostomy, 50 were diagnosed with colorectal cancer and 400 had benign polyps. Their rate of colorectal cancer was not statistically different from people who didn’t have surgery (OR, 1.09; 95% CI, 0.82-1.43). The rate of benign polyps after Roux-en-Y gastrojejunostomy was greater (OR, 1.72; 95% CI, 1.55-1.90).
On the other hand, among the 33,680 patients who underwent sleeve gastrectomy, 50 were diagnosed with colorectal cancer, a lower rate than in the population who didn’t have surgery (OR, 0.30; 95% CI, 0.22-0.39). Their risk of benign polyps was also reduced (OR, 0.45; 95% CI, 0.40-0.50).
All of the medications were significantly associated with a lower risk of colorectal cancer, except orlistat (OR, 0.94; 95% CI, 0.72-1.25).
The finding on Roux-en-Y gastrojejunostomy agreed with studies from England and Nordic countries showing double the risk of colorectal cancer in those patients but conflicted with a French study showing decreased risk, Dr. Shwani said.
While the study doesn’t establish a reason why Roux-en-Y gastrojejunostomy was less beneficial, other researchers have associated the procedure with biomarkers of inflammation, Dr. Shwani said. “It’s inconsistent, and I don’t think we have a clear answer why.”
As a retrospective analysis, the study could not establish a cause-and-effect relationship between surgery or medication and cancer, or adjust for such factors as diet, exercise, or genes, he acknowledged.
Colorectal cancer is just one outcome to consider when deciding whether to undergo weight loss surgery or take weight loss drugs, said session moderator Mohammad Yaghoobi, MD, an associate professor of medicine at McMaster University, Hamilton, Ont.
“The most important outcome that should be investigated is the survival of the patients after obesity surgery,” he told this news organization. “The second would be the quality of life of those patients. Colon cancer is preventable if you are having regular colonoscopies.”
Other studies have not shown much difference between patients who have weight loss surgery and those who don’t, he added.
The study was funded by Merck. Dr. Desai and Dr. Shwani have reported receiving grant funding from Merck. Dr. Yaghoobi has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
LAS VEGAS – People with obesity may be able to reduce their risk of colorectal cancer with weight loss surgery or medication, researchers say.
“We need to have conversations with our patients in the clinic and educate them that they have these resources available,” said Aakash Desai, MD, a hospitalist at MetroHealth Medical Center, Cleveland, in an interview with this news organization.
Dr. Desai and colleagues found that sleeve gastrectomy and four medications were associated with a reduced risk of colorectal cancer but Roux-en-Y gastrojejunostomy and orlistat were not.
Coauthor Zryan Shwani, MD, a gastroenterology fellow at Sibley Memorial Hospital, Washington, D.C., presented the findings here at the American College of Gastroenterology (ACG) 2021 Annual Scientific Meeting.
Working with an underserved population with high rates of obesity in northeastern Ohio, the researchers wondered how surgery and medication could affect these patients.
They analyzed data from the IBM Explorys clinical database, which compiles and standardizes data from electronic medical records on about 74 million patients from more than 300 U.S. hospitals. Consistent with previous studies, they determined that patients with obesity in the database were 2.5 times more likely than people with a healthy weight to be diagnosed with colorectal cancer (odds ratio, 2.48; 95% CI, 2.45-2.51).
Zeroing in on people who had weight loss interventions, they included adults aged 18-75 years who had undergone either Roux-en-Y gastrojejunostomy or sleeve gastrectomy, or had taken the medications liraglutide, orlistat, phentermine/topiramate, bupropion/naltrexone, or lorcaserin.
They excluded patients with Lynch syndrome, intestinal polyposis syndrome, a family history of gastrointestinal malignancy, inflammatory bowel disease, or tobacco or alcohol abuse. Patients who had taken one of the weight loss medications and also had type 2 diabetes were excluded. They did not include patients who had undergone gastric banding because it has become less popular.
For the weight loss medication group, they found 117,730 patients who met their criteria. For the surgery group, 43,050 patients met the criteria.
In analyzing the colorectal cancer rates, they included only diagnoses of malignant neoplasms made 2 years after the interventions.
They compared these patients to a control group of 52,540 people matched in age, with a body mass index (BMI) greater than 30 kg/m2 who did not undergo weight loss surgery or take weight loss medication.
Among the 9,370 patients who underwent Roux-en-Y gastrojejunostomy, 50 were diagnosed with colorectal cancer and 400 had benign polyps. Their rate of colorectal cancer was not statistically different from people who didn’t have surgery (OR, 1.09; 95% CI, 0.82-1.43). The rate of benign polyps after Roux-en-Y gastrojejunostomy was greater (OR, 1.72; 95% CI, 1.55-1.90).
On the other hand, among the 33,680 patients who underwent sleeve gastrectomy, 50 were diagnosed with colorectal cancer, a lower rate than in the population who didn’t have surgery (OR, 0.30; 95% CI, 0.22-0.39). Their risk of benign polyps was also reduced (OR, 0.45; 95% CI, 0.40-0.50).
All of the medications were significantly associated with a lower risk of colorectal cancer, except orlistat (OR, 0.94; 95% CI, 0.72-1.25).
The finding on Roux-en-Y gastrojejunostomy agreed with studies from England and Nordic countries showing double the risk of colorectal cancer in those patients but conflicted with a French study showing decreased risk, Dr. Shwani said.
While the study doesn’t establish a reason why Roux-en-Y gastrojejunostomy was less beneficial, other researchers have associated the procedure with biomarkers of inflammation, Dr. Shwani said. “It’s inconsistent, and I don’t think we have a clear answer why.”
As a retrospective analysis, the study could not establish a cause-and-effect relationship between surgery or medication and cancer, or adjust for such factors as diet, exercise, or genes, he acknowledged.
Colorectal cancer is just one outcome to consider when deciding whether to undergo weight loss surgery or take weight loss drugs, said session moderator Mohammad Yaghoobi, MD, an associate professor of medicine at McMaster University, Hamilton, Ont.
“The most important outcome that should be investigated is the survival of the patients after obesity surgery,” he told this news organization. “The second would be the quality of life of those patients. Colon cancer is preventable if you are having regular colonoscopies.”
Other studies have not shown much difference between patients who have weight loss surgery and those who don’t, he added.
The study was funded by Merck. Dr. Desai and Dr. Shwani have reported receiving grant funding from Merck. Dr. Yaghoobi has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
LAS VEGAS – People with obesity may be able to reduce their risk of colorectal cancer with weight loss surgery or medication, researchers say.
“We need to have conversations with our patients in the clinic and educate them that they have these resources available,” said Aakash Desai, MD, a hospitalist at MetroHealth Medical Center, Cleveland, in an interview with this news organization.
Dr. Desai and colleagues found that sleeve gastrectomy and four medications were associated with a reduced risk of colorectal cancer but Roux-en-Y gastrojejunostomy and orlistat were not.
Coauthor Zryan Shwani, MD, a gastroenterology fellow at Sibley Memorial Hospital, Washington, D.C., presented the findings here at the American College of Gastroenterology (ACG) 2021 Annual Scientific Meeting.
Working with an underserved population with high rates of obesity in northeastern Ohio, the researchers wondered how surgery and medication could affect these patients.
They analyzed data from the IBM Explorys clinical database, which compiles and standardizes data from electronic medical records on about 74 million patients from more than 300 U.S. hospitals. Consistent with previous studies, they determined that patients with obesity in the database were 2.5 times more likely than people with a healthy weight to be diagnosed with colorectal cancer (odds ratio, 2.48; 95% CI, 2.45-2.51).
Zeroing in on people who had weight loss interventions, they included adults aged 18-75 years who had undergone either Roux-en-Y gastrojejunostomy or sleeve gastrectomy, or had taken the medications liraglutide, orlistat, phentermine/topiramate, bupropion/naltrexone, or lorcaserin.
They excluded patients with Lynch syndrome, intestinal polyposis syndrome, a family history of gastrointestinal malignancy, inflammatory bowel disease, or tobacco or alcohol abuse. Patients who had taken one of the weight loss medications and also had type 2 diabetes were excluded. They did not include patients who had undergone gastric banding because it has become less popular.
For the weight loss medication group, they found 117,730 patients who met their criteria. For the surgery group, 43,050 patients met the criteria.
In analyzing the colorectal cancer rates, they included only diagnoses of malignant neoplasms made 2 years after the interventions.
They compared these patients to a control group of 52,540 people matched in age, with a body mass index (BMI) greater than 30 kg/m2 who did not undergo weight loss surgery or take weight loss medication.
Among the 9,370 patients who underwent Roux-en-Y gastrojejunostomy, 50 were diagnosed with colorectal cancer and 400 had benign polyps. Their rate of colorectal cancer was not statistically different from people who didn’t have surgery (OR, 1.09; 95% CI, 0.82-1.43). The rate of benign polyps after Roux-en-Y gastrojejunostomy was greater (OR, 1.72; 95% CI, 1.55-1.90).
On the other hand, among the 33,680 patients who underwent sleeve gastrectomy, 50 were diagnosed with colorectal cancer, a lower rate than in the population who didn’t have surgery (OR, 0.30; 95% CI, 0.22-0.39). Their risk of benign polyps was also reduced (OR, 0.45; 95% CI, 0.40-0.50).
All of the medications were significantly associated with a lower risk of colorectal cancer, except orlistat (OR, 0.94; 95% CI, 0.72-1.25).
The finding on Roux-en-Y gastrojejunostomy agreed with studies from England and Nordic countries showing double the risk of colorectal cancer in those patients but conflicted with a French study showing decreased risk, Dr. Shwani said.
While the study doesn’t establish a reason why Roux-en-Y gastrojejunostomy was less beneficial, other researchers have associated the procedure with biomarkers of inflammation, Dr. Shwani said. “It’s inconsistent, and I don’t think we have a clear answer why.”
As a retrospective analysis, the study could not establish a cause-and-effect relationship between surgery or medication and cancer, or adjust for such factors as diet, exercise, or genes, he acknowledged.
Colorectal cancer is just one outcome to consider when deciding whether to undergo weight loss surgery or take weight loss drugs, said session moderator Mohammad Yaghoobi, MD, an associate professor of medicine at McMaster University, Hamilton, Ont.
“The most important outcome that should be investigated is the survival of the patients after obesity surgery,” he told this news organization. “The second would be the quality of life of those patients. Colon cancer is preventable if you are having regular colonoscopies.”
Other studies have not shown much difference between patients who have weight loss surgery and those who don’t, he added.
The study was funded by Merck. Dr. Desai and Dr. Shwani have reported receiving grant funding from Merck. Dr. Yaghoobi has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ACG 2021
AHA dietary guidance cites structural challenges to heart-healthy patterns
In a new scientific statement on diet and lifestyle recommendations, the American Heart Association is highlighting, for the first time, structural challenges that impede the adoption of heart-healthy dietary patterns.
This is in addition to stressing aspects of diet that improve cardiovascular health and reduce cardiovascular risk, with an emphasis on dietary patterns and food-based guidance beyond naming individual foods or nutrients.
The 2021 Dietary Guidance to Improve Cardiovascular Health scientific statement, developed under Alice H. Lichtenstein, DSc, chair of the AHA writing group, provides 10 evidence-based guidance recommendations to promote cardiometabolic health.
“The way to make heart-healthy choices every day,” said Dr. Lichtenstein, of the Jean Mayer USDA Human Nutrition Research Center on Aging at Tufts University in Boston, in a statement, “is to step back, look at the environment in which you eat, whether it be at home, at work, during social interaction, and then identify what the best choices are. And if there are no good choices, then think about how you can modify your environment so that there are good choices.”
The statement, published in Circulation, underscores growing evidence that nutrition-related chronic diseases have maternal-nutritional origins, and that prevention of pediatric obesity is a key to preserving and prolonging ideal cardiovascular health.
The features are as follows:
- Adjust energy intake and expenditure to achieve and maintain a healthy body weight. To counter the shift toward higher energy intake and more sedentary lifestyles over the past 3 decades, the statement recommends at least 150 minutes of moderate physical activity per week, adjusted for individual’s age, activity level, sex, and size.
- Eat plenty of fruits and vegetables; choose a wide variety. Observational and intervention studies document that dietary patterns rich in varied fruits and vegetables, with the exception of white potatoes, are linked to a lower risk of cardiovascular disease (CVD). Also, whole fruits and vegetables, which more readily provide fiber and satiety, are preferred over juices.
- Choose whole grain foods and products made mostly with whole grains rather than refined grains. Evidence from observational, interventional, and clinical studies confirm the benefits of frequent consumption of whole grains over infrequent consumption or over refined grains in terms of CVD risk, coronary heart disease (CHD), stroke, metabolic syndrome, cardiometabolic risk factors, laxation, and gut microbiota.
- Choose healthy sources of protein, mostly from plants (legumes and nuts).
- Higher intake of legumes, which are rich in protein and fiber, is associated with lower CVD risk, while higher nut intake is associated with lower risks of CVD, CHD, and stroke mortality/incidence. Replacing animal-source foods with plant-source whole foods, beyond health benefits, lowers the diet’s carbon footprint. Meat alternatives are often ultraprocessed and evidence on their short- and long-term health effects is limited. Unsaturated fats are preferred, as are lean, nonprocessed meats.
- Use liquid plant oils rather than tropical oils (coconut, palm, and palm kernel), animal fats (butter and lard), and partially hydrogenated fats. Saturated and trans fats (animal and dairy fats, and partially hydrogenated fat) should be replaced with nontropical liquid plant oils. Evidence supports cardiovascular benefits of dietary unsaturated fats, especially polyunsaturated fats primarily from plant oils (e.g. soybean, corn, safflower and sunflower oils, walnuts, and flax seeds).
- Choose minimally processed foods instead of ultraprocessed foods. Because of their proven association with adverse health outcomes, including overweight and obesity, cardiometabolic disorders (type 2 diabetes, CVD), and all-cause mortality, the consumption of many ultraprocessed foods is of concern. Ultraprocessed foods include artificial colors and flavors and preservatives that promote shelf stability, preserve texture, and increase palatability. A general principle is to emphasize unprocessed or minimally processed foods.
- Minimize intake of beverages and foods with added sugars. Added sugars (commonly glucose, dextrose, sucrose, corn syrup, honey, maple syrup, and concentrated fruit juice) are tied to elevated risk for type 2 diabetes, high cholesterol, and excess body weight. Findings from meta-analyses on body weight and metabolic outcomes for replacing added sugars with low-energy sweeteners are mixed, and the possibility of reverse causality has been raised.
- Choose and prepare foods with little or no salt. In general, the effects of sodium reduction on blood pressure tend to be higher in Black people, middle-aged and older people, and those with hypertension. In the United States, the main combined sources of sodium intake are processed foods, those prepared outside the home, packaged foods, and restaurant foods. Potassium-enriched salts are a promising alternative.
- If you don’t drink alcohol, don’t start; if you choose to drink, limit intake.
- While relationships between alcohol intake and cardiovascular outcomes are complex, the 2020 Dietary Guidelines Advisory Committee recently concluded that those who do drink should consume no more than one drink per day and should not drink alcohol in binges; the 2020 Dietary Guidelines for Americans continues to recommend no more than one drink per day for women and two drinks per day for men.
- Adhere to the guidance regardless in all settings. Food-based dietary guidance applies to all foods and beverages, regardless of where prepared, procured, and consumed. Policies should be enacted that encourage healthier default options (for example, whole grains, minimized sodium and sugar content).
Recognizing impediments
The AHA/ASA scientific statement closes with the declaration: “Creating an environment that facilitates, rather than impedes, adherence to heart-healthy dietary patterns among all individuals is a public health imperative.” It points to the National Institutes of Health’s 2020-2030 Strategic Plan for National Institutes of Health Nutrition Research, which focuses on precision nutrition as a means “to determine the impact on health of not only what individuals eat, but also of why, when, and how they eat throughout the life course.”
Ultimately, precision nutrition may provide personalized diets for CVD prevention. But the “food environment,” often conditioned by “rampant nutrition misinformation” through local, state, and federal practices and policies, may impede the adoption of heart-healthy dietary patterns. Factors such as targeted food marketing (for example, of processed food and beverages in minority neighborhoods), structural racism, neighborhood segregation, unhealthy built environments, and food insecurity create environments in which unhealthy foods are the default option.”
These factors compound adverse dietary and health effects, and underscore a need to “directly combat nutrition misinformation among the public and health care professionals.” They also explain why, despite widespread knowledge of heart-healthy dietary pattern components, little progress has been made in achieving dietary goals in the United States.
Dr. Lichtenstein’s office, in response to a request regarding AHA advocacy and consumer programs, provided the following links: Voices for Healthy Kids initiative site and choosing healthier processed foods and one on fresh, frozen, and canned fruits and vegetables.
Dr. Lichtenstein had no disclosures. Disclosures for the writing group members are included in the statement.
In a new scientific statement on diet and lifestyle recommendations, the American Heart Association is highlighting, for the first time, structural challenges that impede the adoption of heart-healthy dietary patterns.
This is in addition to stressing aspects of diet that improve cardiovascular health and reduce cardiovascular risk, with an emphasis on dietary patterns and food-based guidance beyond naming individual foods or nutrients.
The 2021 Dietary Guidance to Improve Cardiovascular Health scientific statement, developed under Alice H. Lichtenstein, DSc, chair of the AHA writing group, provides 10 evidence-based guidance recommendations to promote cardiometabolic health.
“The way to make heart-healthy choices every day,” said Dr. Lichtenstein, of the Jean Mayer USDA Human Nutrition Research Center on Aging at Tufts University in Boston, in a statement, “is to step back, look at the environment in which you eat, whether it be at home, at work, during social interaction, and then identify what the best choices are. And if there are no good choices, then think about how you can modify your environment so that there are good choices.”
The statement, published in Circulation, underscores growing evidence that nutrition-related chronic diseases have maternal-nutritional origins, and that prevention of pediatric obesity is a key to preserving and prolonging ideal cardiovascular health.
The features are as follows:
- Adjust energy intake and expenditure to achieve and maintain a healthy body weight. To counter the shift toward higher energy intake and more sedentary lifestyles over the past 3 decades, the statement recommends at least 150 minutes of moderate physical activity per week, adjusted for individual’s age, activity level, sex, and size.
- Eat plenty of fruits and vegetables; choose a wide variety. Observational and intervention studies document that dietary patterns rich in varied fruits and vegetables, with the exception of white potatoes, are linked to a lower risk of cardiovascular disease (CVD). Also, whole fruits and vegetables, which more readily provide fiber and satiety, are preferred over juices.
- Choose whole grain foods and products made mostly with whole grains rather than refined grains. Evidence from observational, interventional, and clinical studies confirm the benefits of frequent consumption of whole grains over infrequent consumption or over refined grains in terms of CVD risk, coronary heart disease (CHD), stroke, metabolic syndrome, cardiometabolic risk factors, laxation, and gut microbiota.
- Choose healthy sources of protein, mostly from plants (legumes and nuts).
- Higher intake of legumes, which are rich in protein and fiber, is associated with lower CVD risk, while higher nut intake is associated with lower risks of CVD, CHD, and stroke mortality/incidence. Replacing animal-source foods with plant-source whole foods, beyond health benefits, lowers the diet’s carbon footprint. Meat alternatives are often ultraprocessed and evidence on their short- and long-term health effects is limited. Unsaturated fats are preferred, as are lean, nonprocessed meats.
- Use liquid plant oils rather than tropical oils (coconut, palm, and palm kernel), animal fats (butter and lard), and partially hydrogenated fats. Saturated and trans fats (animal and dairy fats, and partially hydrogenated fat) should be replaced with nontropical liquid plant oils. Evidence supports cardiovascular benefits of dietary unsaturated fats, especially polyunsaturated fats primarily from plant oils (e.g. soybean, corn, safflower and sunflower oils, walnuts, and flax seeds).
- Choose minimally processed foods instead of ultraprocessed foods. Because of their proven association with adverse health outcomes, including overweight and obesity, cardiometabolic disorders (type 2 diabetes, CVD), and all-cause mortality, the consumption of many ultraprocessed foods is of concern. Ultraprocessed foods include artificial colors and flavors and preservatives that promote shelf stability, preserve texture, and increase palatability. A general principle is to emphasize unprocessed or minimally processed foods.
- Minimize intake of beverages and foods with added sugars. Added sugars (commonly glucose, dextrose, sucrose, corn syrup, honey, maple syrup, and concentrated fruit juice) are tied to elevated risk for type 2 diabetes, high cholesterol, and excess body weight. Findings from meta-analyses on body weight and metabolic outcomes for replacing added sugars with low-energy sweeteners are mixed, and the possibility of reverse causality has been raised.
- Choose and prepare foods with little or no salt. In general, the effects of sodium reduction on blood pressure tend to be higher in Black people, middle-aged and older people, and those with hypertension. In the United States, the main combined sources of sodium intake are processed foods, those prepared outside the home, packaged foods, and restaurant foods. Potassium-enriched salts are a promising alternative.
- If you don’t drink alcohol, don’t start; if you choose to drink, limit intake.
- While relationships between alcohol intake and cardiovascular outcomes are complex, the 2020 Dietary Guidelines Advisory Committee recently concluded that those who do drink should consume no more than one drink per day and should not drink alcohol in binges; the 2020 Dietary Guidelines for Americans continues to recommend no more than one drink per day for women and two drinks per day for men.
- Adhere to the guidance regardless in all settings. Food-based dietary guidance applies to all foods and beverages, regardless of where prepared, procured, and consumed. Policies should be enacted that encourage healthier default options (for example, whole grains, minimized sodium and sugar content).
Recognizing impediments
The AHA/ASA scientific statement closes with the declaration: “Creating an environment that facilitates, rather than impedes, adherence to heart-healthy dietary patterns among all individuals is a public health imperative.” It points to the National Institutes of Health’s 2020-2030 Strategic Plan for National Institutes of Health Nutrition Research, which focuses on precision nutrition as a means “to determine the impact on health of not only what individuals eat, but also of why, when, and how they eat throughout the life course.”
Ultimately, precision nutrition may provide personalized diets for CVD prevention. But the “food environment,” often conditioned by “rampant nutrition misinformation” through local, state, and federal practices and policies, may impede the adoption of heart-healthy dietary patterns. Factors such as targeted food marketing (for example, of processed food and beverages in minority neighborhoods), structural racism, neighborhood segregation, unhealthy built environments, and food insecurity create environments in which unhealthy foods are the default option.”
These factors compound adverse dietary and health effects, and underscore a need to “directly combat nutrition misinformation among the public and health care professionals.” They also explain why, despite widespread knowledge of heart-healthy dietary pattern components, little progress has been made in achieving dietary goals in the United States.
Dr. Lichtenstein’s office, in response to a request regarding AHA advocacy and consumer programs, provided the following links: Voices for Healthy Kids initiative site and choosing healthier processed foods and one on fresh, frozen, and canned fruits and vegetables.
Dr. Lichtenstein had no disclosures. Disclosures for the writing group members are included in the statement.
In a new scientific statement on diet and lifestyle recommendations, the American Heart Association is highlighting, for the first time, structural challenges that impede the adoption of heart-healthy dietary patterns.
This is in addition to stressing aspects of diet that improve cardiovascular health and reduce cardiovascular risk, with an emphasis on dietary patterns and food-based guidance beyond naming individual foods or nutrients.
The 2021 Dietary Guidance to Improve Cardiovascular Health scientific statement, developed under Alice H. Lichtenstein, DSc, chair of the AHA writing group, provides 10 evidence-based guidance recommendations to promote cardiometabolic health.
“The way to make heart-healthy choices every day,” said Dr. Lichtenstein, of the Jean Mayer USDA Human Nutrition Research Center on Aging at Tufts University in Boston, in a statement, “is to step back, look at the environment in which you eat, whether it be at home, at work, during social interaction, and then identify what the best choices are. And if there are no good choices, then think about how you can modify your environment so that there are good choices.”
The statement, published in Circulation, underscores growing evidence that nutrition-related chronic diseases have maternal-nutritional origins, and that prevention of pediatric obesity is a key to preserving and prolonging ideal cardiovascular health.
The features are as follows:
- Adjust energy intake and expenditure to achieve and maintain a healthy body weight. To counter the shift toward higher energy intake and more sedentary lifestyles over the past 3 decades, the statement recommends at least 150 minutes of moderate physical activity per week, adjusted for individual’s age, activity level, sex, and size.
- Eat plenty of fruits and vegetables; choose a wide variety. Observational and intervention studies document that dietary patterns rich in varied fruits and vegetables, with the exception of white potatoes, are linked to a lower risk of cardiovascular disease (CVD). Also, whole fruits and vegetables, which more readily provide fiber and satiety, are preferred over juices.
- Choose whole grain foods and products made mostly with whole grains rather than refined grains. Evidence from observational, interventional, and clinical studies confirm the benefits of frequent consumption of whole grains over infrequent consumption or over refined grains in terms of CVD risk, coronary heart disease (CHD), stroke, metabolic syndrome, cardiometabolic risk factors, laxation, and gut microbiota.
- Choose healthy sources of protein, mostly from plants (legumes and nuts).
- Higher intake of legumes, which are rich in protein and fiber, is associated with lower CVD risk, while higher nut intake is associated with lower risks of CVD, CHD, and stroke mortality/incidence. Replacing animal-source foods with plant-source whole foods, beyond health benefits, lowers the diet’s carbon footprint. Meat alternatives are often ultraprocessed and evidence on their short- and long-term health effects is limited. Unsaturated fats are preferred, as are lean, nonprocessed meats.
- Use liquid plant oils rather than tropical oils (coconut, palm, and palm kernel), animal fats (butter and lard), and partially hydrogenated fats. Saturated and trans fats (animal and dairy fats, and partially hydrogenated fat) should be replaced with nontropical liquid plant oils. Evidence supports cardiovascular benefits of dietary unsaturated fats, especially polyunsaturated fats primarily from plant oils (e.g. soybean, corn, safflower and sunflower oils, walnuts, and flax seeds).
- Choose minimally processed foods instead of ultraprocessed foods. Because of their proven association with adverse health outcomes, including overweight and obesity, cardiometabolic disorders (type 2 diabetes, CVD), and all-cause mortality, the consumption of many ultraprocessed foods is of concern. Ultraprocessed foods include artificial colors and flavors and preservatives that promote shelf stability, preserve texture, and increase palatability. A general principle is to emphasize unprocessed or minimally processed foods.
- Minimize intake of beverages and foods with added sugars. Added sugars (commonly glucose, dextrose, sucrose, corn syrup, honey, maple syrup, and concentrated fruit juice) are tied to elevated risk for type 2 diabetes, high cholesterol, and excess body weight. Findings from meta-analyses on body weight and metabolic outcomes for replacing added sugars with low-energy sweeteners are mixed, and the possibility of reverse causality has been raised.
- Choose and prepare foods with little or no salt. In general, the effects of sodium reduction on blood pressure tend to be higher in Black people, middle-aged and older people, and those with hypertension. In the United States, the main combined sources of sodium intake are processed foods, those prepared outside the home, packaged foods, and restaurant foods. Potassium-enriched salts are a promising alternative.
- If you don’t drink alcohol, don’t start; if you choose to drink, limit intake.
- While relationships between alcohol intake and cardiovascular outcomes are complex, the 2020 Dietary Guidelines Advisory Committee recently concluded that those who do drink should consume no more than one drink per day and should not drink alcohol in binges; the 2020 Dietary Guidelines for Americans continues to recommend no more than one drink per day for women and two drinks per day for men.
- Adhere to the guidance regardless in all settings. Food-based dietary guidance applies to all foods and beverages, regardless of where prepared, procured, and consumed. Policies should be enacted that encourage healthier default options (for example, whole grains, minimized sodium and sugar content).
Recognizing impediments
The AHA/ASA scientific statement closes with the declaration: “Creating an environment that facilitates, rather than impedes, adherence to heart-healthy dietary patterns among all individuals is a public health imperative.” It points to the National Institutes of Health’s 2020-2030 Strategic Plan for National Institutes of Health Nutrition Research, which focuses on precision nutrition as a means “to determine the impact on health of not only what individuals eat, but also of why, when, and how they eat throughout the life course.”
Ultimately, precision nutrition may provide personalized diets for CVD prevention. But the “food environment,” often conditioned by “rampant nutrition misinformation” through local, state, and federal practices and policies, may impede the adoption of heart-healthy dietary patterns. Factors such as targeted food marketing (for example, of processed food and beverages in minority neighborhoods), structural racism, neighborhood segregation, unhealthy built environments, and food insecurity create environments in which unhealthy foods are the default option.”
These factors compound adverse dietary and health effects, and underscore a need to “directly combat nutrition misinformation among the public and health care professionals.” They also explain why, despite widespread knowledge of heart-healthy dietary pattern components, little progress has been made in achieving dietary goals in the United States.
Dr. Lichtenstein’s office, in response to a request regarding AHA advocacy and consumer programs, provided the following links: Voices for Healthy Kids initiative site and choosing healthier processed foods and one on fresh, frozen, and canned fruits and vegetables.
Dr. Lichtenstein had no disclosures. Disclosures for the writing group members are included in the statement.
FROM CIRCULATION













