User login
Vulvar and gluteal manifestations of Crohn disease
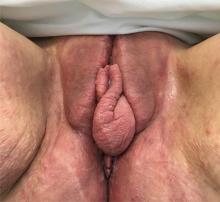
A 37-year-old woman presented with recurring painful swelling and erythema of the vulva over the last year. Despite a series of negative vaginal cultures, she was prescribed multiple courses of antifungal and antibacterial treatments, while her symptoms continued to worsen. She had no other relevant medical history except for occasional diarrhea and abdominal cramping, which were attributed to irritable bowel syndrome.
CROHN DISEASE OUTSIDE THE GASTROINTESTINAL TRACT
Crohn disease primarily affects the gastrointestinal tract but is associated with extraintestinal manifestations (in the oral cavity, eyes, skin, and joints) in up to 45% of patients.1
The most common mucocutaneous manifestations are granulomatous lesions that extend directly from the gastrointestinal tract, including perianal and peristomal skin tags, fistulas, and perineal ulcerations. In most cases, the onset of cutaneous manifestations follows intestinal disease, but vulvar Crohn disease may precede gastrointestinal symptoms in approximately 25% of patients, with the average age at onset in the mid-30s.1
The pathogenesis of vulvar Crohn disease remains unclear. One theory involves production of immune complexes from the gastrointestinal tract and a possible T-lymphocyte-mediated type IV hypersensitivity reaction.2
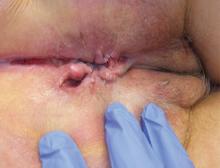
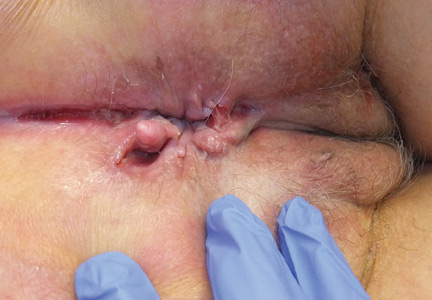
The diagnosis of vulvar Crohn disease should be considered in a patient who has vulvar pain, edema, and ulcerations not otherwise explained, whether or not gastrointestinal Crohn disease is present. The diagnosis is established with clinical history and characteristic histopathology on biopsy. Multiple biopsies may be needed, and early endoscopy is recommended to establish the diagnosis. The histologic features include noncaseating and nonnecrotizing granulomatous dermatitis or vulvitis with occasional reports of eosinophilic infiltrates and necrobiosis.5,6 An imaging study such as ultrasonography is sometimes used to differentiate between a specific cutaneous manifestation of Crohn disease and its complications such as perianal fistula or abscess.
Clinical vulvar lesions are nonspecific, and those of Crohn disease are frequently mistaken for infectious, inflammatory, or traumatic vulvitis. Diagnostic biopsy for histologic analysis is warranted.
- Andreani SM, Ratnasingham K, Dang HH, Gravante G, Giordano P. Crohn’s disease of the vulva. Int J Surg 2010; 8(1):2–5. doi:10.1016/j.ijsu.2009.09.012
- Siroy A, Wasman J. Metastatic Crohn disease: a rare cutaneous entity. Arch Pathol Lab Med 2012; 136(3):329–332. doi:10.5858/arpa.2010-0666-RS
- Foo WC, Papalas JA, Robboy SJ, Selim MA. Vulvar manifestations of Crohn’s disease. Am J Dermatopathol 2011; 33(6):588–593. doi:10.1097/DAD.0b013e31820a2635
- Amankwah Y, Haefner H. Vulvar edema. Dermatol Clin 2010; 28(4):765–777. doi:10.1016/j.det.2010.08.001
- Emanuel PO, Phelps RG. Metastatic Crohn’s disease: a histopathologic study of 12 cases. J Cutan Pathol 2008; 35(5):457–461. doi:10.1111/j.1600-0560.2007.00849.x
- Hackzell-Bradley M, Hedblad MA, Stephansson EA. Metastatic Crohn’s disease: report of 3 cases with special reference to histopathologic findings. Arch Dermatol 1996; 132(8):928–932.
A 37-year-old woman presented with recurring painful swelling and erythema of the vulva over the last year. Despite a series of negative vaginal cultures, she was prescribed multiple courses of antifungal and antibacterial treatments, while her symptoms continued to worsen. She had no other relevant medical history except for occasional diarrhea and abdominal cramping, which were attributed to irritable bowel syndrome.
CROHN DISEASE OUTSIDE THE GASTROINTESTINAL TRACT
Crohn disease primarily affects the gastrointestinal tract but is associated with extraintestinal manifestations (in the oral cavity, eyes, skin, and joints) in up to 45% of patients.1
The most common mucocutaneous manifestations are granulomatous lesions that extend directly from the gastrointestinal tract, including perianal and peristomal skin tags, fistulas, and perineal ulcerations. In most cases, the onset of cutaneous manifestations follows intestinal disease, but vulvar Crohn disease may precede gastrointestinal symptoms in approximately 25% of patients, with the average age at onset in the mid-30s.1
The pathogenesis of vulvar Crohn disease remains unclear. One theory involves production of immune complexes from the gastrointestinal tract and a possible T-lymphocyte-mediated type IV hypersensitivity reaction.2
The diagnosis of vulvar Crohn disease should be considered in a patient who has vulvar pain, edema, and ulcerations not otherwise explained, whether or not gastrointestinal Crohn disease is present. The diagnosis is established with clinical history and characteristic histopathology on biopsy. Multiple biopsies may be needed, and early endoscopy is recommended to establish the diagnosis. The histologic features include noncaseating and nonnecrotizing granulomatous dermatitis or vulvitis with occasional reports of eosinophilic infiltrates and necrobiosis.5,6 An imaging study such as ultrasonography is sometimes used to differentiate between a specific cutaneous manifestation of Crohn disease and its complications such as perianal fistula or abscess.
Clinical vulvar lesions are nonspecific, and those of Crohn disease are frequently mistaken for infectious, inflammatory, or traumatic vulvitis. Diagnostic biopsy for histologic analysis is warranted.
A 37-year-old woman presented with recurring painful swelling and erythema of the vulva over the last year. Despite a series of negative vaginal cultures, she was prescribed multiple courses of antifungal and antibacterial treatments, while her symptoms continued to worsen. She had no other relevant medical history except for occasional diarrhea and abdominal cramping, which were attributed to irritable bowel syndrome.
CROHN DISEASE OUTSIDE THE GASTROINTESTINAL TRACT
Crohn disease primarily affects the gastrointestinal tract but is associated with extraintestinal manifestations (in the oral cavity, eyes, skin, and joints) in up to 45% of patients.1
The most common mucocutaneous manifestations are granulomatous lesions that extend directly from the gastrointestinal tract, including perianal and peristomal skin tags, fistulas, and perineal ulcerations. In most cases, the onset of cutaneous manifestations follows intestinal disease, but vulvar Crohn disease may precede gastrointestinal symptoms in approximately 25% of patients, with the average age at onset in the mid-30s.1
The pathogenesis of vulvar Crohn disease remains unclear. One theory involves production of immune complexes from the gastrointestinal tract and a possible T-lymphocyte-mediated type IV hypersensitivity reaction.2
The diagnosis of vulvar Crohn disease should be considered in a patient who has vulvar pain, edema, and ulcerations not otherwise explained, whether or not gastrointestinal Crohn disease is present. The diagnosis is established with clinical history and characteristic histopathology on biopsy. Multiple biopsies may be needed, and early endoscopy is recommended to establish the diagnosis. The histologic features include noncaseating and nonnecrotizing granulomatous dermatitis or vulvitis with occasional reports of eosinophilic infiltrates and necrobiosis.5,6 An imaging study such as ultrasonography is sometimes used to differentiate between a specific cutaneous manifestation of Crohn disease and its complications such as perianal fistula or abscess.
Clinical vulvar lesions are nonspecific, and those of Crohn disease are frequently mistaken for infectious, inflammatory, or traumatic vulvitis. Diagnostic biopsy for histologic analysis is warranted.
- Andreani SM, Ratnasingham K, Dang HH, Gravante G, Giordano P. Crohn’s disease of the vulva. Int J Surg 2010; 8(1):2–5. doi:10.1016/j.ijsu.2009.09.012
- Siroy A, Wasman J. Metastatic Crohn disease: a rare cutaneous entity. Arch Pathol Lab Med 2012; 136(3):329–332. doi:10.5858/arpa.2010-0666-RS
- Foo WC, Papalas JA, Robboy SJ, Selim MA. Vulvar manifestations of Crohn’s disease. Am J Dermatopathol 2011; 33(6):588–593. doi:10.1097/DAD.0b013e31820a2635
- Amankwah Y, Haefner H. Vulvar edema. Dermatol Clin 2010; 28(4):765–777. doi:10.1016/j.det.2010.08.001
- Emanuel PO, Phelps RG. Metastatic Crohn’s disease: a histopathologic study of 12 cases. J Cutan Pathol 2008; 35(5):457–461. doi:10.1111/j.1600-0560.2007.00849.x
- Hackzell-Bradley M, Hedblad MA, Stephansson EA. Metastatic Crohn’s disease: report of 3 cases with special reference to histopathologic findings. Arch Dermatol 1996; 132(8):928–932.
- Andreani SM, Ratnasingham K, Dang HH, Gravante G, Giordano P. Crohn’s disease of the vulva. Int J Surg 2010; 8(1):2–5. doi:10.1016/j.ijsu.2009.09.012
- Siroy A, Wasman J. Metastatic Crohn disease: a rare cutaneous entity. Arch Pathol Lab Med 2012; 136(3):329–332. doi:10.5858/arpa.2010-0666-RS
- Foo WC, Papalas JA, Robboy SJ, Selim MA. Vulvar manifestations of Crohn’s disease. Am J Dermatopathol 2011; 33(6):588–593. doi:10.1097/DAD.0b013e31820a2635
- Amankwah Y, Haefner H. Vulvar edema. Dermatol Clin 2010; 28(4):765–777. doi:10.1016/j.det.2010.08.001
- Emanuel PO, Phelps RG. Metastatic Crohn’s disease: a histopathologic study of 12 cases. J Cutan Pathol 2008; 35(5):457–461. doi:10.1111/j.1600-0560.2007.00849.x
- Hackzell-Bradley M, Hedblad MA, Stephansson EA. Metastatic Crohn’s disease: report of 3 cases with special reference to histopathologic findings. Arch Dermatol 1996; 132(8):928–932.
USPSTF BRCA testing recs: 2 more groups require attention
Reference
1. US Preventive Services Task Force. Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer. JAMA. 2019;322:652-665.
Reference
1. US Preventive Services Task Force. Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer. JAMA. 2019;322:652-665.
Reference
1. US Preventive Services Task Force. Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer. JAMA. 2019;322:652-665.
AF risk is elevated after early-stage breast cancer diagnosis
Women with early-stage breast cancer are at elevated risk for atrial fibrillation (AF) short term and, to a lesser extent, long term, finds a large Canadian cohort study. Risk was higher for those who had received chemotherapy but not tied to specific cardiotoxic drugs or drug classes.
“Cardiovascular disease is a particularly pertinent clinical concern for women diagnosed with early-stage breast cancer,” note the investigators, led by Husam Abdel-Qadir, MD, PhD, FRCPC, Cardiology Clinic, Women’s College Hospital, Toronto. “Many early-stage breast cancer survivors are older than 65 years and have hypertension, diabetes, or left ventricular dysfunction. Accordingly, a diagnosis of AF would translate to a clinically relevant stroke risk for many early-stage breast cancer survivors.”
The investigators undertook a population-based retrospective cohort study of women in the province of Ontario with stage I-III breast cancer diagnosed between April 2007 and December 2016, matching them 1:3 to cancer-free control women on birth year and receipt of breast imaging.
An initial analysis, based on 95,539 breast cancer patients and 217,456 cancer-free controls, showed that the former and latter groups did not differ significantly on the prevalence of preexisting AF (5.3% vs. 5.2%; P = .21), according to results reported in JAMA Network Open.
Main analyses excluded women with preexisting AF, leaving 68,113 breast cancer patients and 204,330 cancer-free controls having a mean follow-up of 5.7 years. Both groups had a mean age of 60 years at baseline, and prevalences of cardiovascular comorbidities were similar. Within the breast cancer group, 50.4% had left-sided disease; overall, 53.2% received chemotherapy and 71.7% received radiation therapy.
At 10 years after diagnosis, breast cancer patients had a small but significant increase in AF incidence relative to control peers (7.4% vs. 6.8%; P less than .001). When the investigators looked at specific time periods, survivors had a significantly elevated AF risk in year 1 postdiagnosis (hazard ratio, 2.16) and after year 5 postdiagnosis (hazard ratio, 1.20), but not during years 2 through 5.
Analyses beginning 1 year after diagnosis showed a slightly smaller but still significant elevation of AF incidence for the breast cancer group at 9 years of follow-up (10 years after diagnosis) (7.0% vs. 6.5%; P less than .001).
Among breast cancer patients, those who received chemotherapy had a higher risk of AF than those who did not (adjusted hazard ratio, 1.23); however, this elevation of risk was not specifically tied to receipt of anthracyclines or trastuzumab (Herceptin) versus other chemotherapy. Risk was not elevated for those who received radiation therapy.
“Our study findings suggest that a diagnosis of early-stage breast cancer may be associated with a small increase in the risk of AF compared with that for cancer-free women,” Dr. Abdel-Qadir and coinvestigators noted. “Since the absolute risk is small, this finding does not warrant routine surveillance but rather should prompt consideration of AF in the differential diagnosis for women with compatible symptoms.
“The early and late periods of increased AF risk in early-stage breast cancer survivors warrant focused research to better understand the underlying causes and subsequent implications,” they concluded.
Dr. Abdel-Qadir reported receiving grants from the Canadian Cardiovascular Society during the conduct of the study, speaker fees from Amgen, and an honorarium for clinical events adjudication committee membership from the Canadian Vigour Centre for a study funded by AstraZeneca. The study was funded by the Canadian Cardiovascular Society Atrial Fibrillation Research Award.
SOURCE: Abdel-Qadir H et al. JAMA Netw Open. 2019 Sep 4;2(9):e1911838.
Women with early-stage breast cancer are at elevated risk for atrial fibrillation (AF) short term and, to a lesser extent, long term, finds a large Canadian cohort study. Risk was higher for those who had received chemotherapy but not tied to specific cardiotoxic drugs or drug classes.
“Cardiovascular disease is a particularly pertinent clinical concern for women diagnosed with early-stage breast cancer,” note the investigators, led by Husam Abdel-Qadir, MD, PhD, FRCPC, Cardiology Clinic, Women’s College Hospital, Toronto. “Many early-stage breast cancer survivors are older than 65 years and have hypertension, diabetes, or left ventricular dysfunction. Accordingly, a diagnosis of AF would translate to a clinically relevant stroke risk for many early-stage breast cancer survivors.”
The investigators undertook a population-based retrospective cohort study of women in the province of Ontario with stage I-III breast cancer diagnosed between April 2007 and December 2016, matching them 1:3 to cancer-free control women on birth year and receipt of breast imaging.
An initial analysis, based on 95,539 breast cancer patients and 217,456 cancer-free controls, showed that the former and latter groups did not differ significantly on the prevalence of preexisting AF (5.3% vs. 5.2%; P = .21), according to results reported in JAMA Network Open.
Main analyses excluded women with preexisting AF, leaving 68,113 breast cancer patients and 204,330 cancer-free controls having a mean follow-up of 5.7 years. Both groups had a mean age of 60 years at baseline, and prevalences of cardiovascular comorbidities were similar. Within the breast cancer group, 50.4% had left-sided disease; overall, 53.2% received chemotherapy and 71.7% received radiation therapy.
At 10 years after diagnosis, breast cancer patients had a small but significant increase in AF incidence relative to control peers (7.4% vs. 6.8%; P less than .001). When the investigators looked at specific time periods, survivors had a significantly elevated AF risk in year 1 postdiagnosis (hazard ratio, 2.16) and after year 5 postdiagnosis (hazard ratio, 1.20), but not during years 2 through 5.
Analyses beginning 1 year after diagnosis showed a slightly smaller but still significant elevation of AF incidence for the breast cancer group at 9 years of follow-up (10 years after diagnosis) (7.0% vs. 6.5%; P less than .001).
Among breast cancer patients, those who received chemotherapy had a higher risk of AF than those who did not (adjusted hazard ratio, 1.23); however, this elevation of risk was not specifically tied to receipt of anthracyclines or trastuzumab (Herceptin) versus other chemotherapy. Risk was not elevated for those who received radiation therapy.
“Our study findings suggest that a diagnosis of early-stage breast cancer may be associated with a small increase in the risk of AF compared with that for cancer-free women,” Dr. Abdel-Qadir and coinvestigators noted. “Since the absolute risk is small, this finding does not warrant routine surveillance but rather should prompt consideration of AF in the differential diagnosis for women with compatible symptoms.
“The early and late periods of increased AF risk in early-stage breast cancer survivors warrant focused research to better understand the underlying causes and subsequent implications,” they concluded.
Dr. Abdel-Qadir reported receiving grants from the Canadian Cardiovascular Society during the conduct of the study, speaker fees from Amgen, and an honorarium for clinical events adjudication committee membership from the Canadian Vigour Centre for a study funded by AstraZeneca. The study was funded by the Canadian Cardiovascular Society Atrial Fibrillation Research Award.
SOURCE: Abdel-Qadir H et al. JAMA Netw Open. 2019 Sep 4;2(9):e1911838.
Women with early-stage breast cancer are at elevated risk for atrial fibrillation (AF) short term and, to a lesser extent, long term, finds a large Canadian cohort study. Risk was higher for those who had received chemotherapy but not tied to specific cardiotoxic drugs or drug classes.
“Cardiovascular disease is a particularly pertinent clinical concern for women diagnosed with early-stage breast cancer,” note the investigators, led by Husam Abdel-Qadir, MD, PhD, FRCPC, Cardiology Clinic, Women’s College Hospital, Toronto. “Many early-stage breast cancer survivors are older than 65 years and have hypertension, diabetes, or left ventricular dysfunction. Accordingly, a diagnosis of AF would translate to a clinically relevant stroke risk for many early-stage breast cancer survivors.”
The investigators undertook a population-based retrospective cohort study of women in the province of Ontario with stage I-III breast cancer diagnosed between April 2007 and December 2016, matching them 1:3 to cancer-free control women on birth year and receipt of breast imaging.
An initial analysis, based on 95,539 breast cancer patients and 217,456 cancer-free controls, showed that the former and latter groups did not differ significantly on the prevalence of preexisting AF (5.3% vs. 5.2%; P = .21), according to results reported in JAMA Network Open.
Main analyses excluded women with preexisting AF, leaving 68,113 breast cancer patients and 204,330 cancer-free controls having a mean follow-up of 5.7 years. Both groups had a mean age of 60 years at baseline, and prevalences of cardiovascular comorbidities were similar. Within the breast cancer group, 50.4% had left-sided disease; overall, 53.2% received chemotherapy and 71.7% received radiation therapy.
At 10 years after diagnosis, breast cancer patients had a small but significant increase in AF incidence relative to control peers (7.4% vs. 6.8%; P less than .001). When the investigators looked at specific time periods, survivors had a significantly elevated AF risk in year 1 postdiagnosis (hazard ratio, 2.16) and after year 5 postdiagnosis (hazard ratio, 1.20), but not during years 2 through 5.
Analyses beginning 1 year after diagnosis showed a slightly smaller but still significant elevation of AF incidence for the breast cancer group at 9 years of follow-up (10 years after diagnosis) (7.0% vs. 6.5%; P less than .001).
Among breast cancer patients, those who received chemotherapy had a higher risk of AF than those who did not (adjusted hazard ratio, 1.23); however, this elevation of risk was not specifically tied to receipt of anthracyclines or trastuzumab (Herceptin) versus other chemotherapy. Risk was not elevated for those who received radiation therapy.
“Our study findings suggest that a diagnosis of early-stage breast cancer may be associated with a small increase in the risk of AF compared with that for cancer-free women,” Dr. Abdel-Qadir and coinvestigators noted. “Since the absolute risk is small, this finding does not warrant routine surveillance but rather should prompt consideration of AF in the differential diagnosis for women with compatible symptoms.
“The early and late periods of increased AF risk in early-stage breast cancer survivors warrant focused research to better understand the underlying causes and subsequent implications,” they concluded.
Dr. Abdel-Qadir reported receiving grants from the Canadian Cardiovascular Society during the conduct of the study, speaker fees from Amgen, and an honorarium for clinical events adjudication committee membership from the Canadian Vigour Centre for a study funded by AstraZeneca. The study was funded by the Canadian Cardiovascular Society Atrial Fibrillation Research Award.
SOURCE: Abdel-Qadir H et al. JAMA Netw Open. 2019 Sep 4;2(9):e1911838.
FROM JAMA NETWORK OPEN
Early maternal anxiety tied to adolescent hyperactivity
COPENHAGEN – Exposure to maternal somatic anxiety during pregnancy and toddlerhood increases a child’s risk of hyperactivity symptoms in adolescence, Blanca Bolea, MD, said at the annual congress of the European College of Neuropsychopharmacology.
In contrast, the children of mothers who were anxious were not at increased risk for subsequent inattention symptoms in an analysis of 8,725 mothers and their children participating in the Avon Longitudinal Study of Parents and Children, a prospective epidemiologic cohort study ongoing in southwest England since 1991, said Dr. Bolea, a psychiatrist at the University of Toronto.
These findings have practical implications for clinical care: “If we know that women who are anxious in the perinatal period put their children at risk for hyperactivity later on, then we can tackle their anxiety in pregnancy or toddlerhood. And that’s easy to do: You can do group [cognitive-behavioral therapy]; you can give medications, so there are things you can do to reduce that risk. That’s relevant, because we don’t know much about how to reduce levels of ADHD. We know it has a genetic component, but we can’t touch that. You cannot change your genes, so far. But environmental things, we can change. So if we can identify the mothers who are more anxious during pregnancy and toddlerhood and give them resources to reduce their anxiety, then we can potentially reduce hyperactivity later on,” she explained in an interview.
In the Avon study, maternal anxiety was serially assessed from early pregnancy up until a child’s 5th birthday.
“We looked for maternal symptoms similar to panic disorder: shortness of breath, dizziness, sweating, things like that. These are symptoms that any clinician can identify by asking the mothers, so it’s not hard to identify the mothers who could be at risk,” according to the psychiatrist.
Children in the Avon study were assessed for symptoms of inattention at age 8.5 years using the Sky Search, Sky Search Dual Test, and Opposite Worlds subtests of the Tests of Everyday Attention for Children. Hyperactivity symptoms were assessed at age 16 years via the Strengths and Difficulties Questionnaire.
In an analysis adjusted for potentially confounding sociodemographic factors, adolescents whose mothers were rated by investigators as having moderate or high somatic anxiety during pregnancy and the toddlerhood years were at 2.1-fold increased risk of hyperactivity symptoms compared to those whose mothers had low or no anxiety, but increased maternal anxiety wasn’t associated with scores on any of the three tests of inattention.
Dr. Bolea cautioned that, while these Avon study findings document an association between early maternal anxiety and subsequent adolescent hyperactivity, that doesn’t prove causality. The findings are consistent, however, with the fetal origins hypothesis put forth by the late British epidemiologist David J. Barker, MD, PhD, which postulates that stressful fetal circumstances have profound effects later in life.
she said.
The hypothesis has been borne out in animal studies: Stress a pregnant rat, and her offspring will display hyperactivity.
Dr. Bolea reported having no financial conflicts regarding her study. The Avon Longitudinal Study of Parents and Children is funded by the Medical Research Council and the Wellcome Trust.
COPENHAGEN – Exposure to maternal somatic anxiety during pregnancy and toddlerhood increases a child’s risk of hyperactivity symptoms in adolescence, Blanca Bolea, MD, said at the annual congress of the European College of Neuropsychopharmacology.
In contrast, the children of mothers who were anxious were not at increased risk for subsequent inattention symptoms in an analysis of 8,725 mothers and their children participating in the Avon Longitudinal Study of Parents and Children, a prospective epidemiologic cohort study ongoing in southwest England since 1991, said Dr. Bolea, a psychiatrist at the University of Toronto.
These findings have practical implications for clinical care: “If we know that women who are anxious in the perinatal period put their children at risk for hyperactivity later on, then we can tackle their anxiety in pregnancy or toddlerhood. And that’s easy to do: You can do group [cognitive-behavioral therapy]; you can give medications, so there are things you can do to reduce that risk. That’s relevant, because we don’t know much about how to reduce levels of ADHD. We know it has a genetic component, but we can’t touch that. You cannot change your genes, so far. But environmental things, we can change. So if we can identify the mothers who are more anxious during pregnancy and toddlerhood and give them resources to reduce their anxiety, then we can potentially reduce hyperactivity later on,” she explained in an interview.
In the Avon study, maternal anxiety was serially assessed from early pregnancy up until a child’s 5th birthday.
“We looked for maternal symptoms similar to panic disorder: shortness of breath, dizziness, sweating, things like that. These are symptoms that any clinician can identify by asking the mothers, so it’s not hard to identify the mothers who could be at risk,” according to the psychiatrist.
Children in the Avon study were assessed for symptoms of inattention at age 8.5 years using the Sky Search, Sky Search Dual Test, and Opposite Worlds subtests of the Tests of Everyday Attention for Children. Hyperactivity symptoms were assessed at age 16 years via the Strengths and Difficulties Questionnaire.
In an analysis adjusted for potentially confounding sociodemographic factors, adolescents whose mothers were rated by investigators as having moderate or high somatic anxiety during pregnancy and the toddlerhood years were at 2.1-fold increased risk of hyperactivity symptoms compared to those whose mothers had low or no anxiety, but increased maternal anxiety wasn’t associated with scores on any of the three tests of inattention.
Dr. Bolea cautioned that, while these Avon study findings document an association between early maternal anxiety and subsequent adolescent hyperactivity, that doesn’t prove causality. The findings are consistent, however, with the fetal origins hypothesis put forth by the late British epidemiologist David J. Barker, MD, PhD, which postulates that stressful fetal circumstances have profound effects later in life.
she said.
The hypothesis has been borne out in animal studies: Stress a pregnant rat, and her offspring will display hyperactivity.
Dr. Bolea reported having no financial conflicts regarding her study. The Avon Longitudinal Study of Parents and Children is funded by the Medical Research Council and the Wellcome Trust.
COPENHAGEN – Exposure to maternal somatic anxiety during pregnancy and toddlerhood increases a child’s risk of hyperactivity symptoms in adolescence, Blanca Bolea, MD, said at the annual congress of the European College of Neuropsychopharmacology.
In contrast, the children of mothers who were anxious were not at increased risk for subsequent inattention symptoms in an analysis of 8,725 mothers and their children participating in the Avon Longitudinal Study of Parents and Children, a prospective epidemiologic cohort study ongoing in southwest England since 1991, said Dr. Bolea, a psychiatrist at the University of Toronto.
These findings have practical implications for clinical care: “If we know that women who are anxious in the perinatal period put their children at risk for hyperactivity later on, then we can tackle their anxiety in pregnancy or toddlerhood. And that’s easy to do: You can do group [cognitive-behavioral therapy]; you can give medications, so there are things you can do to reduce that risk. That’s relevant, because we don’t know much about how to reduce levels of ADHD. We know it has a genetic component, but we can’t touch that. You cannot change your genes, so far. But environmental things, we can change. So if we can identify the mothers who are more anxious during pregnancy and toddlerhood and give them resources to reduce their anxiety, then we can potentially reduce hyperactivity later on,” she explained in an interview.
In the Avon study, maternal anxiety was serially assessed from early pregnancy up until a child’s 5th birthday.
“We looked for maternal symptoms similar to panic disorder: shortness of breath, dizziness, sweating, things like that. These are symptoms that any clinician can identify by asking the mothers, so it’s not hard to identify the mothers who could be at risk,” according to the psychiatrist.
Children in the Avon study were assessed for symptoms of inattention at age 8.5 years using the Sky Search, Sky Search Dual Test, and Opposite Worlds subtests of the Tests of Everyday Attention for Children. Hyperactivity symptoms were assessed at age 16 years via the Strengths and Difficulties Questionnaire.
In an analysis adjusted for potentially confounding sociodemographic factors, adolescents whose mothers were rated by investigators as having moderate or high somatic anxiety during pregnancy and the toddlerhood years were at 2.1-fold increased risk of hyperactivity symptoms compared to those whose mothers had low or no anxiety, but increased maternal anxiety wasn’t associated with scores on any of the three tests of inattention.
Dr. Bolea cautioned that, while these Avon study findings document an association between early maternal anxiety and subsequent adolescent hyperactivity, that doesn’t prove causality. The findings are consistent, however, with the fetal origins hypothesis put forth by the late British epidemiologist David J. Barker, MD, PhD, which postulates that stressful fetal circumstances have profound effects later in life.
she said.
The hypothesis has been borne out in animal studies: Stress a pregnant rat, and her offspring will display hyperactivity.
Dr. Bolea reported having no financial conflicts regarding her study. The Avon Longitudinal Study of Parents and Children is funded by the Medical Research Council and the Wellcome Trust.
REPORTING FROM ECNP 2019
Meeting the obstetrical needs of trans and gender nonconforming patients
Like their cisgender counterparts, transgender and gender nonconforming patients (trans patients) may reach a point in their lives where they want to build their own families. This may be achieved through adoption, alternative insemination with donor sperm, or assisted reproductive treatment with donor sperm or egg, cryopreserved sperm or egg, or surrogacy.1 Obstetricians can provide more equitable care to trans individuals by acknowledging these needs and providing gender-inclusive counseling and guidance.
The American Society for Reproductive Medicine recommends that medical providers counsel patients about the potential effects of medical transitioning on their fertility prior to the initiation of hormonal or surgical therapies.2 Patients should be educated about options for fertility preservation and reproduction since exogenous hormones and gonadectomy impact fertility.3 A referral to a fertility specialist should be placed for patients interested in oocyte or sperm cryopreservation, embryo cryopreservation, or ovarian tissue cryopreservation.2
If a trans patient presents to the obstetrician/gynecologist for preconception counseling after undergoing medical gender transition, they should be offered evidence-based guidance based on an organ inventory (surgical history with documentation of natal sex organs still in situ). A biologic pregnancy may be a fertility option for a patient who has a vagina, uterus, fallopian tubes, and ovaries and is not currently using testosterone. Gender-affirming testosterone therapy suppresses ovulation and causes amenorrhea in most patients, although this is often reversible once the exogenous hormone is discontinued.2 When the patient is ovulating on their own or undergoes ovulation induction, conception may be achieved via the same methods used with cisgender couples: Sperm is obtained from a partner or donor, followed by intercourse if the patient is comfortable with this, intrauterine insemination (IUI), or in vitro fertilization (IVF).
Conversely, a trans patient with a penis and testicles who has already undergone medical gender affirmation with estrogen should be counseled that prior exposure to estrogen may have caused irreversible testicular damage, making assisted reproductive treatment more challenging if sperm had not been cryopreserved prior to starting gender-affirming hormone therapy.2 If spermatogenesis is successful or sperm was previously cryopreserved, the next step in reproductive counseling for these patients centers on finding gestational carriers and egg donors if the patient does not already have a partner who is willing or able to carry the child. At this point in time, uterine transplantation has not been attempted in a trans patient and therefore is not considered a viable fertility option.
The trans patient who becomes pregnant will encounter physical changes that may trigger underlying gender dysphoria. One study found that transgender men who experience pregnancy exhibited varying degrees of gender dysphoria.4 Obstetrician/gynecologists should have an awareness about the possibility of heightened gender dysphoria and sensitively approach prenatal visits by avoiding triggering language or using inappropriate pronouns. Simply asking a trans patient about preferred pronouns and terminology for body parts can be the difference between a negative and positive pregnancy experience. For example, a transman may prefer a different term for vagina/vulva/cervix. This is especially important at the time of delivery, when exams may become more frequent for the patient. However, inclusive prenatal care starts from the first prenatal visit when the patient checks in and continues all the way through the doctor/patient experience. All office staff should be trained to use preferred names and pronouns and gender-neutral restrooms should be easily accessible. Likewise, waiting rooms should include visible support for the LGBTQ (lesbian, gay, bisexual, transgender and queer or questioning) patient population.
The anatomy ultrasound and “gender reveal” during the pregnancy and at the time of delivery can understandably also be a sensitive subject for a pregnant trans patient. Previous cultural practice has been to describe the gender of the fetus at the anatomy ultrasound, when in fact, gender can only be self-determined by an individual many years after birth. What the anatomy ultrasound does convey is the appearance of external genitalia to help predict the assigned sex. As obstetrician/gynecologists who practice evidence-based medicine, we are encouraged to challenge the cultural norm of announcing the gender of the baby at time of ultrasound and at time of birth. We should focus instead on conveying what objective information we do know. After the infant is born, we know the sex they are assigned based on the what external reproductive organs are seen.
In the postpartum period, trans patients who successfully carried a pregnancy may choose to feed their infant with their own human milk. For some trans patients, breastfeeding may be referred to as chestfeeding, since this terminology is more gender neutral. Having prior chest masculinization surgery does not exclude a transmasculine patient from lactating, although milk production may vary. Patients should be counseled that there is limited data on the safety of testosterone use while lactating.1 We found only one case report of induced lactation in a nonpuerperal transfeminine patient.5 In addition to addressing infant feeding concerns, obstetrician/gynecologists should counsel postpartum trans patients about contraceptive options and screen for perinatal mood disorders, especially those patients with a history of mood disorders before pregnancy.
Ultimately, trans patients seeking fertility options and obstetrical care have a right to obtain reliable information and access gender-inclusive treatment from their obstetrician/gynecologists. Each family makeup is unique and should be respected by all health care professionals taking care of the patient. As obstetrician/gynecologists, it is our duty to coordinate and advocate for the equitable care of our trans patients who want to grow their families.
Dr. Joyner is an assistant professor at Emory University, Atlanta, and is the director of gynecologic services in the Gender Center at Grady Memorial Hospital in Atlanta. Dr. Joyner identifies as a cisgender female and uses she/hers/her as her personal pronouns. Dr. Katie Riddle is an ob.gyn. in Connecticut who is passionate about LGBTQ health care. She recently completed her residency in Ann Arbor, Mich. Dr. Riddle identifies as a cisgender female and uses she/hers/her as her personal pronouns. Dr. Joyner and Dr. Riddle said they had no financial disclosures. Email them at obnews@mdedge.com.
References
1. Viloria RP. “Reproductive Health and Obstetric Care in Transgender Patients.” Fenway Health.
2. Amato P. “Fertility options for transgender persons.” University of California, San Francisco Transgender Care and Treatment Guidelines. 2016 Jun 17.
3. Fertil Steril. 2015 Nov. doi: 10.1016/j.fertnstert.2015.08.021.
4. Obstet Gynecol. 2014. doi: 10.1097/AOG.0000000000000540.
5. Transgend Health. 2018 Jan 1. doi: 10.1089/trgh.2017.0044.
Like their cisgender counterparts, transgender and gender nonconforming patients (trans patients) may reach a point in their lives where they want to build their own families. This may be achieved through adoption, alternative insemination with donor sperm, or assisted reproductive treatment with donor sperm or egg, cryopreserved sperm or egg, or surrogacy.1 Obstetricians can provide more equitable care to trans individuals by acknowledging these needs and providing gender-inclusive counseling and guidance.
The American Society for Reproductive Medicine recommends that medical providers counsel patients about the potential effects of medical transitioning on their fertility prior to the initiation of hormonal or surgical therapies.2 Patients should be educated about options for fertility preservation and reproduction since exogenous hormones and gonadectomy impact fertility.3 A referral to a fertility specialist should be placed for patients interested in oocyte or sperm cryopreservation, embryo cryopreservation, or ovarian tissue cryopreservation.2
If a trans patient presents to the obstetrician/gynecologist for preconception counseling after undergoing medical gender transition, they should be offered evidence-based guidance based on an organ inventory (surgical history with documentation of natal sex organs still in situ). A biologic pregnancy may be a fertility option for a patient who has a vagina, uterus, fallopian tubes, and ovaries and is not currently using testosterone. Gender-affirming testosterone therapy suppresses ovulation and causes amenorrhea in most patients, although this is often reversible once the exogenous hormone is discontinued.2 When the patient is ovulating on their own or undergoes ovulation induction, conception may be achieved via the same methods used with cisgender couples: Sperm is obtained from a partner or donor, followed by intercourse if the patient is comfortable with this, intrauterine insemination (IUI), or in vitro fertilization (IVF).
Conversely, a trans patient with a penis and testicles who has already undergone medical gender affirmation with estrogen should be counseled that prior exposure to estrogen may have caused irreversible testicular damage, making assisted reproductive treatment more challenging if sperm had not been cryopreserved prior to starting gender-affirming hormone therapy.2 If spermatogenesis is successful or sperm was previously cryopreserved, the next step in reproductive counseling for these patients centers on finding gestational carriers and egg donors if the patient does not already have a partner who is willing or able to carry the child. At this point in time, uterine transplantation has not been attempted in a trans patient and therefore is not considered a viable fertility option.
The trans patient who becomes pregnant will encounter physical changes that may trigger underlying gender dysphoria. One study found that transgender men who experience pregnancy exhibited varying degrees of gender dysphoria.4 Obstetrician/gynecologists should have an awareness about the possibility of heightened gender dysphoria and sensitively approach prenatal visits by avoiding triggering language or using inappropriate pronouns. Simply asking a trans patient about preferred pronouns and terminology for body parts can be the difference between a negative and positive pregnancy experience. For example, a transman may prefer a different term for vagina/vulva/cervix. This is especially important at the time of delivery, when exams may become more frequent for the patient. However, inclusive prenatal care starts from the first prenatal visit when the patient checks in and continues all the way through the doctor/patient experience. All office staff should be trained to use preferred names and pronouns and gender-neutral restrooms should be easily accessible. Likewise, waiting rooms should include visible support for the LGBTQ (lesbian, gay, bisexual, transgender and queer or questioning) patient population.
The anatomy ultrasound and “gender reveal” during the pregnancy and at the time of delivery can understandably also be a sensitive subject for a pregnant trans patient. Previous cultural practice has been to describe the gender of the fetus at the anatomy ultrasound, when in fact, gender can only be self-determined by an individual many years after birth. What the anatomy ultrasound does convey is the appearance of external genitalia to help predict the assigned sex. As obstetrician/gynecologists who practice evidence-based medicine, we are encouraged to challenge the cultural norm of announcing the gender of the baby at time of ultrasound and at time of birth. We should focus instead on conveying what objective information we do know. After the infant is born, we know the sex they are assigned based on the what external reproductive organs are seen.
In the postpartum period, trans patients who successfully carried a pregnancy may choose to feed their infant with their own human milk. For some trans patients, breastfeeding may be referred to as chestfeeding, since this terminology is more gender neutral. Having prior chest masculinization surgery does not exclude a transmasculine patient from lactating, although milk production may vary. Patients should be counseled that there is limited data on the safety of testosterone use while lactating.1 We found only one case report of induced lactation in a nonpuerperal transfeminine patient.5 In addition to addressing infant feeding concerns, obstetrician/gynecologists should counsel postpartum trans patients about contraceptive options and screen for perinatal mood disorders, especially those patients with a history of mood disorders before pregnancy.
Ultimately, trans patients seeking fertility options and obstetrical care have a right to obtain reliable information and access gender-inclusive treatment from their obstetrician/gynecologists. Each family makeup is unique and should be respected by all health care professionals taking care of the patient. As obstetrician/gynecologists, it is our duty to coordinate and advocate for the equitable care of our trans patients who want to grow their families.
Dr. Joyner is an assistant professor at Emory University, Atlanta, and is the director of gynecologic services in the Gender Center at Grady Memorial Hospital in Atlanta. Dr. Joyner identifies as a cisgender female and uses she/hers/her as her personal pronouns. Dr. Katie Riddle is an ob.gyn. in Connecticut who is passionate about LGBTQ health care. She recently completed her residency in Ann Arbor, Mich. Dr. Riddle identifies as a cisgender female and uses she/hers/her as her personal pronouns. Dr. Joyner and Dr. Riddle said they had no financial disclosures. Email them at obnews@mdedge.com.
References
1. Viloria RP. “Reproductive Health and Obstetric Care in Transgender Patients.” Fenway Health.
2. Amato P. “Fertility options for transgender persons.” University of California, San Francisco Transgender Care and Treatment Guidelines. 2016 Jun 17.
3. Fertil Steril. 2015 Nov. doi: 10.1016/j.fertnstert.2015.08.021.
4. Obstet Gynecol. 2014. doi: 10.1097/AOG.0000000000000540.
5. Transgend Health. 2018 Jan 1. doi: 10.1089/trgh.2017.0044.
Like their cisgender counterparts, transgender and gender nonconforming patients (trans patients) may reach a point in their lives where they want to build their own families. This may be achieved through adoption, alternative insemination with donor sperm, or assisted reproductive treatment with donor sperm or egg, cryopreserved sperm or egg, or surrogacy.1 Obstetricians can provide more equitable care to trans individuals by acknowledging these needs and providing gender-inclusive counseling and guidance.
The American Society for Reproductive Medicine recommends that medical providers counsel patients about the potential effects of medical transitioning on their fertility prior to the initiation of hormonal or surgical therapies.2 Patients should be educated about options for fertility preservation and reproduction since exogenous hormones and gonadectomy impact fertility.3 A referral to a fertility specialist should be placed for patients interested in oocyte or sperm cryopreservation, embryo cryopreservation, or ovarian tissue cryopreservation.2
If a trans patient presents to the obstetrician/gynecologist for preconception counseling after undergoing medical gender transition, they should be offered evidence-based guidance based on an organ inventory (surgical history with documentation of natal sex organs still in situ). A biologic pregnancy may be a fertility option for a patient who has a vagina, uterus, fallopian tubes, and ovaries and is not currently using testosterone. Gender-affirming testosterone therapy suppresses ovulation and causes amenorrhea in most patients, although this is often reversible once the exogenous hormone is discontinued.2 When the patient is ovulating on their own or undergoes ovulation induction, conception may be achieved via the same methods used with cisgender couples: Sperm is obtained from a partner or donor, followed by intercourse if the patient is comfortable with this, intrauterine insemination (IUI), or in vitro fertilization (IVF).
Conversely, a trans patient with a penis and testicles who has already undergone medical gender affirmation with estrogen should be counseled that prior exposure to estrogen may have caused irreversible testicular damage, making assisted reproductive treatment more challenging if sperm had not been cryopreserved prior to starting gender-affirming hormone therapy.2 If spermatogenesis is successful or sperm was previously cryopreserved, the next step in reproductive counseling for these patients centers on finding gestational carriers and egg donors if the patient does not already have a partner who is willing or able to carry the child. At this point in time, uterine transplantation has not been attempted in a trans patient and therefore is not considered a viable fertility option.
The trans patient who becomes pregnant will encounter physical changes that may trigger underlying gender dysphoria. One study found that transgender men who experience pregnancy exhibited varying degrees of gender dysphoria.4 Obstetrician/gynecologists should have an awareness about the possibility of heightened gender dysphoria and sensitively approach prenatal visits by avoiding triggering language or using inappropriate pronouns. Simply asking a trans patient about preferred pronouns and terminology for body parts can be the difference between a negative and positive pregnancy experience. For example, a transman may prefer a different term for vagina/vulva/cervix. This is especially important at the time of delivery, when exams may become more frequent for the patient. However, inclusive prenatal care starts from the first prenatal visit when the patient checks in and continues all the way through the doctor/patient experience. All office staff should be trained to use preferred names and pronouns and gender-neutral restrooms should be easily accessible. Likewise, waiting rooms should include visible support for the LGBTQ (lesbian, gay, bisexual, transgender and queer or questioning) patient population.
The anatomy ultrasound and “gender reveal” during the pregnancy and at the time of delivery can understandably also be a sensitive subject for a pregnant trans patient. Previous cultural practice has been to describe the gender of the fetus at the anatomy ultrasound, when in fact, gender can only be self-determined by an individual many years after birth. What the anatomy ultrasound does convey is the appearance of external genitalia to help predict the assigned sex. As obstetrician/gynecologists who practice evidence-based medicine, we are encouraged to challenge the cultural norm of announcing the gender of the baby at time of ultrasound and at time of birth. We should focus instead on conveying what objective information we do know. After the infant is born, we know the sex they are assigned based on the what external reproductive organs are seen.
In the postpartum period, trans patients who successfully carried a pregnancy may choose to feed their infant with their own human milk. For some trans patients, breastfeeding may be referred to as chestfeeding, since this terminology is more gender neutral. Having prior chest masculinization surgery does not exclude a transmasculine patient from lactating, although milk production may vary. Patients should be counseled that there is limited data on the safety of testosterone use while lactating.1 We found only one case report of induced lactation in a nonpuerperal transfeminine patient.5 In addition to addressing infant feeding concerns, obstetrician/gynecologists should counsel postpartum trans patients about contraceptive options and screen for perinatal mood disorders, especially those patients with a history of mood disorders before pregnancy.
Ultimately, trans patients seeking fertility options and obstetrical care have a right to obtain reliable information and access gender-inclusive treatment from their obstetrician/gynecologists. Each family makeup is unique and should be respected by all health care professionals taking care of the patient. As obstetrician/gynecologists, it is our duty to coordinate and advocate for the equitable care of our trans patients who want to grow their families.
Dr. Joyner is an assistant professor at Emory University, Atlanta, and is the director of gynecologic services in the Gender Center at Grady Memorial Hospital in Atlanta. Dr. Joyner identifies as a cisgender female and uses she/hers/her as her personal pronouns. Dr. Katie Riddle is an ob.gyn. in Connecticut who is passionate about LGBTQ health care. She recently completed her residency in Ann Arbor, Mich. Dr. Riddle identifies as a cisgender female and uses she/hers/her as her personal pronouns. Dr. Joyner and Dr. Riddle said they had no financial disclosures. Email them at obnews@mdedge.com.
References
1. Viloria RP. “Reproductive Health and Obstetric Care in Transgender Patients.” Fenway Health.
2. Amato P. “Fertility options for transgender persons.” University of California, San Francisco Transgender Care and Treatment Guidelines. 2016 Jun 17.
3. Fertil Steril. 2015 Nov. doi: 10.1016/j.fertnstert.2015.08.021.
4. Obstet Gynecol. 2014. doi: 10.1097/AOG.0000000000000540.
5. Transgend Health. 2018 Jan 1. doi: 10.1089/trgh.2017.0044.
Click for Credit: Psoriasis relief; Stress & CV problems; more
Here are 5 articles from the October issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Bronchiolitis is a feared complication of connective tissue disease
To take the posttest, go to: https://bit.ly/2klWpRb
Expires April 8, 2020
2. Stress incontinence surgery improves sexual dysfunction
To take the posttest, go to: https://bit.ly/2m0wb71
Expires April 10, 2020
3. Survey finds psoriasis patients seek relief with alternative therapies
To take the posttest, go to: https://bit.ly/2lZZDtO
Expires April 10, 2020
4. New data further suggest that stress does a number on the CV system
To take the posttest, go to: https://bit.ly/2lR31ax
Expires April 11, 2020
5. Rate of objects ingested by young children increased over last two decades
To take the posttest, go to: https://bit.ly/2mmYptb
Expires April 12, 2020
Here are 5 articles from the October issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Bronchiolitis is a feared complication of connective tissue disease
To take the posttest, go to: https://bit.ly/2klWpRb
Expires April 8, 2020
2. Stress incontinence surgery improves sexual dysfunction
To take the posttest, go to: https://bit.ly/2m0wb71
Expires April 10, 2020
3. Survey finds psoriasis patients seek relief with alternative therapies
To take the posttest, go to: https://bit.ly/2lZZDtO
Expires April 10, 2020
4. New data further suggest that stress does a number on the CV system
To take the posttest, go to: https://bit.ly/2lR31ax
Expires April 11, 2020
5. Rate of objects ingested by young children increased over last two decades
To take the posttest, go to: https://bit.ly/2mmYptb
Expires April 12, 2020
Here are 5 articles from the October issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Bronchiolitis is a feared complication of connective tissue disease
To take the posttest, go to: https://bit.ly/2klWpRb
Expires April 8, 2020
2. Stress incontinence surgery improves sexual dysfunction
To take the posttest, go to: https://bit.ly/2m0wb71
Expires April 10, 2020
3. Survey finds psoriasis patients seek relief with alternative therapies
To take the posttest, go to: https://bit.ly/2lZZDtO
Expires April 10, 2020
4. New data further suggest that stress does a number on the CV system
To take the posttest, go to: https://bit.ly/2lR31ax
Expires April 11, 2020
5. Rate of objects ingested by young children increased over last two decades
To take the posttest, go to: https://bit.ly/2mmYptb
Expires April 12, 2020
USPSTF: Screening pregnant women for asymptomatic bacteriuria cuts pyelonephritis risk
according to new recommendations set forth by the United States Preventive Services Task Force (USPSTF).
However, the investigating committee reported, there is evidence against screening nonpregnant women and adult men. In fact, the committee found “adequate” evidence of potential harm associated with treating asymptomatic bacteriuria in adults of both sexes, including adverse effects of antibiotics and on the microbiome.
The new document downgrades from A to B the group’s prior recommendation that urine culture screening for asymptomatic bacteriuria should be performed among pregnant women at 12-16 weeks’ gestation or at their first prenatal visit. The USPSTF recommendation to not screen nonpregnant adults retained its D rating, Jerome A. Leis, MD and Christine Soong, MD said in an accompanying editorial.
“Not screening or treating asymptomatic bacteriuria in this population has long been an ironclad recommendation endorsed by the Infectious Diseases Society of America, as well as numerous professional societies as part of the Choosing Wisely campaign,” wrote Dr. Leis of Sunnybrook Health Sciences Centre, Toronto, and Dr. Soong of the University of Toronto. “Restating this steadfast and pervasive recommendation may seem unremarkable and almost pedantic, yet it remains stubbornly disregarded by clinicians across multiple settings.”
The new recommendations were based on a review of 19 studies involving almost 8,500 pregnant and nonpregnant women, as well as a small number of adult men. Most were carried out in the 1960s or 1970s. The most recent ones were published in 2002 and 2015. The dearth of more recent data may have limited some conclusions and certainly highlighted the need for more research, said Jillian T. Henderson, PhD, chair of the committee assigned to investigate the evidence.
“Few studies of asymptomatic bacteriuria screening or treatment in pregnant populations have been conducted in the past 40 years,” wrote Dr. Henderson of Kaiser Permanente Northwest, Portland, and associates. “Historical evidence established asymptomatic bacteriuria screening and treatment as standard obstetric practice in the United States.” But these trials typically were less rigorous than modern studies, and the results are out of touch with modern clinical settings and treatment protocols, the team noted.
Additionally, Dr. Henderson and coauthors said, rates of pyelonephritis were about 10 times higher then than they are now. In the more recent studies, pyelonephritis rates in control groups were 2.2% and 2.5%; in most of the older studies, control group rates ranged from 33% to 36%.
In commissioning the investigation, the task force looked at the following four questions:
Does screening improve health outcomes?
Neither of two studies involving 5,289 women, one from Spain and one from Turkey, addressed this question in nonpregnant women; however, studies that looked at pregnant women generally found that screening did reduce the risk of pyelonephritis by about 70%. The investigators cautioned that these studies were out of date and perhaps methodologically flawed.
The only study that looked at newborn outcomes found no difference in birth weights or premature births between the screened and unscreened cohorts.
No study examined this question in nonpregnant women or men.
What are the harms of such screening?
A single study of 372 pregnant women described potential prenatal and perinatal harms associated with screening and treatment. It found a slight increase in congenital abnormalities in the screened cohort (1.6%), compared with those who were not screened (1.1%). However, those who were not screened were presumably not prescribed antibiotics.
Does treatment of screening-detected asymptomatic bacteriuria improve health outcomes?
Twelve trials of pregnant women (2,377) addressed this issue. All but two were conducted in the 1960s and 1970s. Treatment varied widely; sulfonamides were the most common, including the now discarded sulfamethazine and sulfadimethoxine. Dosages and duration of treatment also were considerably higher and longer than current practice.
In all but one study, there were higher rates of pyelonephritis in the control group. A pooled risk analysis indicated that treatment reduced the risk of pyelonephritis by nearly 80% (relative risk, 0.24).
Seven studies found higher rates of low birth weight in infants born to mothers who were treated, but two studies reported a significant reduction in the risk of low birth weight.
Among the six trials that examined perinatal mortality, none found significant associations with treatment.
Five studies examined treatment in nonpregnant women with screening-detected asymptomatic bacteriuria, and one included men as well. Of the four that reported the rate of symptomatic infection or pyelonephritis, none found a significant difference between treatment and control groups. The single study that included men also found no significant difference between treatment and control groups.
Among the three studies that focused on older adults, there also were no significant between-group differences in outcomes.
What harms are associated with treatment of screening-detected asymptomatic bacteriuria?
Seven studies comprised pregnant women. Five reported congenital malformations in the intervention and control groups. Overall, there were very few cases of malformations, with more – although not significantly more – in the control groups.
Evidence related to other infant and maternal harms was “sparsely and inconsistently reported,” Dr. Henderson and coauthors noted, “and there was a lack of evidence on long-term neonatal outcomes after antibiotic treatment of asymptomatic bacteriuria in pregnancy.”
Two studies listed maternal adverse events associated with different treatments including vaginitis and diarrhea with ampicillin and rashes and nausea with nalidixic acid.
In terms of nonpregnant women and men, four studies reported adverse events. None occurred with nitrofurantoin or trimethoprim treatment; however, one study that included daily treatment with ofloxacin noted that 6% withdrew because of adverse events – vertigo and gastrointestinal symptoms.
Treatments didn’t affect hematocrit, bilirubin, serum urea, or nitrogen, although some studies found a slight reduction in serum creatinine.
Although there’s a need for additional research into this question, the new recommendations provide a good reason to further reduce unnecessary antibiotic exposure, Lindsey E. Nicolle, MD, wrote in a second commentary.
These updated recommendations “contribute to the evolution of management of asymptomatic bacteriuria in healthy women,” wrote Dr. Nicolle of the University of Manitoba, Winnipeg. “However, questions remain about the risks and benefits of universal screening for and treatment of asymptomatic bacteriuria in pregnant women in the context of current clinical practice. The effects of changes in fetal-maternal care, of low- compared with high-risk pregnancies, and of health care access need to be understood. In the short term, application of current diagnostic recommendations for identification of persistent symptomatic bacteriuria with a second urine culture may provide an immediate opportunity to limit unnecessary antimicrobial use for some pregnant women.”
No conflicts of interest were reported by the USPSTF authors, nor by Dr. Leis, Dr. Soong, or Dr. Nicolle. The USPSTF report was funded by the Agency for Healthcare Research and Quality.
SOURCES: U.S. Preventive Services Task Force. JAMA. 2019;322(12):1188-94; Henderson JT et al. JAMA. 2019;322(12):1195-205; Leis JA and Soong C. JAMA. 2019. doi: 10.1001/jamainternmed.2019.4515; Nicolle LE. JAMA. 2019;322(12):1152-4.
according to new recommendations set forth by the United States Preventive Services Task Force (USPSTF).
However, the investigating committee reported, there is evidence against screening nonpregnant women and adult men. In fact, the committee found “adequate” evidence of potential harm associated with treating asymptomatic bacteriuria in adults of both sexes, including adverse effects of antibiotics and on the microbiome.
The new document downgrades from A to B the group’s prior recommendation that urine culture screening for asymptomatic bacteriuria should be performed among pregnant women at 12-16 weeks’ gestation or at their first prenatal visit. The USPSTF recommendation to not screen nonpregnant adults retained its D rating, Jerome A. Leis, MD and Christine Soong, MD said in an accompanying editorial.
“Not screening or treating asymptomatic bacteriuria in this population has long been an ironclad recommendation endorsed by the Infectious Diseases Society of America, as well as numerous professional societies as part of the Choosing Wisely campaign,” wrote Dr. Leis of Sunnybrook Health Sciences Centre, Toronto, and Dr. Soong of the University of Toronto. “Restating this steadfast and pervasive recommendation may seem unremarkable and almost pedantic, yet it remains stubbornly disregarded by clinicians across multiple settings.”
The new recommendations were based on a review of 19 studies involving almost 8,500 pregnant and nonpregnant women, as well as a small number of adult men. Most were carried out in the 1960s or 1970s. The most recent ones were published in 2002 and 2015. The dearth of more recent data may have limited some conclusions and certainly highlighted the need for more research, said Jillian T. Henderson, PhD, chair of the committee assigned to investigate the evidence.
“Few studies of asymptomatic bacteriuria screening or treatment in pregnant populations have been conducted in the past 40 years,” wrote Dr. Henderson of Kaiser Permanente Northwest, Portland, and associates. “Historical evidence established asymptomatic bacteriuria screening and treatment as standard obstetric practice in the United States.” But these trials typically were less rigorous than modern studies, and the results are out of touch with modern clinical settings and treatment protocols, the team noted.
Additionally, Dr. Henderson and coauthors said, rates of pyelonephritis were about 10 times higher then than they are now. In the more recent studies, pyelonephritis rates in control groups were 2.2% and 2.5%; in most of the older studies, control group rates ranged from 33% to 36%.
In commissioning the investigation, the task force looked at the following four questions:
Does screening improve health outcomes?
Neither of two studies involving 5,289 women, one from Spain and one from Turkey, addressed this question in nonpregnant women; however, studies that looked at pregnant women generally found that screening did reduce the risk of pyelonephritis by about 70%. The investigators cautioned that these studies were out of date and perhaps methodologically flawed.
The only study that looked at newborn outcomes found no difference in birth weights or premature births between the screened and unscreened cohorts.
No study examined this question in nonpregnant women or men.
What are the harms of such screening?
A single study of 372 pregnant women described potential prenatal and perinatal harms associated with screening and treatment. It found a slight increase in congenital abnormalities in the screened cohort (1.6%), compared with those who were not screened (1.1%). However, those who were not screened were presumably not prescribed antibiotics.
Does treatment of screening-detected asymptomatic bacteriuria improve health outcomes?
Twelve trials of pregnant women (2,377) addressed this issue. All but two were conducted in the 1960s and 1970s. Treatment varied widely; sulfonamides were the most common, including the now discarded sulfamethazine and sulfadimethoxine. Dosages and duration of treatment also were considerably higher and longer than current practice.
In all but one study, there were higher rates of pyelonephritis in the control group. A pooled risk analysis indicated that treatment reduced the risk of pyelonephritis by nearly 80% (relative risk, 0.24).
Seven studies found higher rates of low birth weight in infants born to mothers who were treated, but two studies reported a significant reduction in the risk of low birth weight.
Among the six trials that examined perinatal mortality, none found significant associations with treatment.
Five studies examined treatment in nonpregnant women with screening-detected asymptomatic bacteriuria, and one included men as well. Of the four that reported the rate of symptomatic infection or pyelonephritis, none found a significant difference between treatment and control groups. The single study that included men also found no significant difference between treatment and control groups.
Among the three studies that focused on older adults, there also were no significant between-group differences in outcomes.
What harms are associated with treatment of screening-detected asymptomatic bacteriuria?
Seven studies comprised pregnant women. Five reported congenital malformations in the intervention and control groups. Overall, there were very few cases of malformations, with more – although not significantly more – in the control groups.
Evidence related to other infant and maternal harms was “sparsely and inconsistently reported,” Dr. Henderson and coauthors noted, “and there was a lack of evidence on long-term neonatal outcomes after antibiotic treatment of asymptomatic bacteriuria in pregnancy.”
Two studies listed maternal adverse events associated with different treatments including vaginitis and diarrhea with ampicillin and rashes and nausea with nalidixic acid.
In terms of nonpregnant women and men, four studies reported adverse events. None occurred with nitrofurantoin or trimethoprim treatment; however, one study that included daily treatment with ofloxacin noted that 6% withdrew because of adverse events – vertigo and gastrointestinal symptoms.
Treatments didn’t affect hematocrit, bilirubin, serum urea, or nitrogen, although some studies found a slight reduction in serum creatinine.
Although there’s a need for additional research into this question, the new recommendations provide a good reason to further reduce unnecessary antibiotic exposure, Lindsey E. Nicolle, MD, wrote in a second commentary.
These updated recommendations “contribute to the evolution of management of asymptomatic bacteriuria in healthy women,” wrote Dr. Nicolle of the University of Manitoba, Winnipeg. “However, questions remain about the risks and benefits of universal screening for and treatment of asymptomatic bacteriuria in pregnant women in the context of current clinical practice. The effects of changes in fetal-maternal care, of low- compared with high-risk pregnancies, and of health care access need to be understood. In the short term, application of current diagnostic recommendations for identification of persistent symptomatic bacteriuria with a second urine culture may provide an immediate opportunity to limit unnecessary antimicrobial use for some pregnant women.”
No conflicts of interest were reported by the USPSTF authors, nor by Dr. Leis, Dr. Soong, or Dr. Nicolle. The USPSTF report was funded by the Agency for Healthcare Research and Quality.
SOURCES: U.S. Preventive Services Task Force. JAMA. 2019;322(12):1188-94; Henderson JT et al. JAMA. 2019;322(12):1195-205; Leis JA and Soong C. JAMA. 2019. doi: 10.1001/jamainternmed.2019.4515; Nicolle LE. JAMA. 2019;322(12):1152-4.
according to new recommendations set forth by the United States Preventive Services Task Force (USPSTF).
However, the investigating committee reported, there is evidence against screening nonpregnant women and adult men. In fact, the committee found “adequate” evidence of potential harm associated with treating asymptomatic bacteriuria in adults of both sexes, including adverse effects of antibiotics and on the microbiome.
The new document downgrades from A to B the group’s prior recommendation that urine culture screening for asymptomatic bacteriuria should be performed among pregnant women at 12-16 weeks’ gestation or at their first prenatal visit. The USPSTF recommendation to not screen nonpregnant adults retained its D rating, Jerome A. Leis, MD and Christine Soong, MD said in an accompanying editorial.
“Not screening or treating asymptomatic bacteriuria in this population has long been an ironclad recommendation endorsed by the Infectious Diseases Society of America, as well as numerous professional societies as part of the Choosing Wisely campaign,” wrote Dr. Leis of Sunnybrook Health Sciences Centre, Toronto, and Dr. Soong of the University of Toronto. “Restating this steadfast and pervasive recommendation may seem unremarkable and almost pedantic, yet it remains stubbornly disregarded by clinicians across multiple settings.”
The new recommendations were based on a review of 19 studies involving almost 8,500 pregnant and nonpregnant women, as well as a small number of adult men. Most were carried out in the 1960s or 1970s. The most recent ones were published in 2002 and 2015. The dearth of more recent data may have limited some conclusions and certainly highlighted the need for more research, said Jillian T. Henderson, PhD, chair of the committee assigned to investigate the evidence.
“Few studies of asymptomatic bacteriuria screening or treatment in pregnant populations have been conducted in the past 40 years,” wrote Dr. Henderson of Kaiser Permanente Northwest, Portland, and associates. “Historical evidence established asymptomatic bacteriuria screening and treatment as standard obstetric practice in the United States.” But these trials typically were less rigorous than modern studies, and the results are out of touch with modern clinical settings and treatment protocols, the team noted.
Additionally, Dr. Henderson and coauthors said, rates of pyelonephritis were about 10 times higher then than they are now. In the more recent studies, pyelonephritis rates in control groups were 2.2% and 2.5%; in most of the older studies, control group rates ranged from 33% to 36%.
In commissioning the investigation, the task force looked at the following four questions:
Does screening improve health outcomes?
Neither of two studies involving 5,289 women, one from Spain and one from Turkey, addressed this question in nonpregnant women; however, studies that looked at pregnant women generally found that screening did reduce the risk of pyelonephritis by about 70%. The investigators cautioned that these studies were out of date and perhaps methodologically flawed.
The only study that looked at newborn outcomes found no difference in birth weights or premature births between the screened and unscreened cohorts.
No study examined this question in nonpregnant women or men.
What are the harms of such screening?
A single study of 372 pregnant women described potential prenatal and perinatal harms associated with screening and treatment. It found a slight increase in congenital abnormalities in the screened cohort (1.6%), compared with those who were not screened (1.1%). However, those who were not screened were presumably not prescribed antibiotics.
Does treatment of screening-detected asymptomatic bacteriuria improve health outcomes?
Twelve trials of pregnant women (2,377) addressed this issue. All but two were conducted in the 1960s and 1970s. Treatment varied widely; sulfonamides were the most common, including the now discarded sulfamethazine and sulfadimethoxine. Dosages and duration of treatment also were considerably higher and longer than current practice.
In all but one study, there were higher rates of pyelonephritis in the control group. A pooled risk analysis indicated that treatment reduced the risk of pyelonephritis by nearly 80% (relative risk, 0.24).
Seven studies found higher rates of low birth weight in infants born to mothers who were treated, but two studies reported a significant reduction in the risk of low birth weight.
Among the six trials that examined perinatal mortality, none found significant associations with treatment.
Five studies examined treatment in nonpregnant women with screening-detected asymptomatic bacteriuria, and one included men as well. Of the four that reported the rate of symptomatic infection or pyelonephritis, none found a significant difference between treatment and control groups. The single study that included men also found no significant difference between treatment and control groups.
Among the three studies that focused on older adults, there also were no significant between-group differences in outcomes.
What harms are associated with treatment of screening-detected asymptomatic bacteriuria?
Seven studies comprised pregnant women. Five reported congenital malformations in the intervention and control groups. Overall, there were very few cases of malformations, with more – although not significantly more – in the control groups.
Evidence related to other infant and maternal harms was “sparsely and inconsistently reported,” Dr. Henderson and coauthors noted, “and there was a lack of evidence on long-term neonatal outcomes after antibiotic treatment of asymptomatic bacteriuria in pregnancy.”
Two studies listed maternal adverse events associated with different treatments including vaginitis and diarrhea with ampicillin and rashes and nausea with nalidixic acid.
In terms of nonpregnant women and men, four studies reported adverse events. None occurred with nitrofurantoin or trimethoprim treatment; however, one study that included daily treatment with ofloxacin noted that 6% withdrew because of adverse events – vertigo and gastrointestinal symptoms.
Treatments didn’t affect hematocrit, bilirubin, serum urea, or nitrogen, although some studies found a slight reduction in serum creatinine.
Although there’s a need for additional research into this question, the new recommendations provide a good reason to further reduce unnecessary antibiotic exposure, Lindsey E. Nicolle, MD, wrote in a second commentary.
These updated recommendations “contribute to the evolution of management of asymptomatic bacteriuria in healthy women,” wrote Dr. Nicolle of the University of Manitoba, Winnipeg. “However, questions remain about the risks and benefits of universal screening for and treatment of asymptomatic bacteriuria in pregnant women in the context of current clinical practice. The effects of changes in fetal-maternal care, of low- compared with high-risk pregnancies, and of health care access need to be understood. In the short term, application of current diagnostic recommendations for identification of persistent symptomatic bacteriuria with a second urine culture may provide an immediate opportunity to limit unnecessary antimicrobial use for some pregnant women.”
No conflicts of interest were reported by the USPSTF authors, nor by Dr. Leis, Dr. Soong, or Dr. Nicolle. The USPSTF report was funded by the Agency for Healthcare Research and Quality.
SOURCES: U.S. Preventive Services Task Force. JAMA. 2019;322(12):1188-94; Henderson JT et al. JAMA. 2019;322(12):1195-205; Leis JA and Soong C. JAMA. 2019. doi: 10.1001/jamainternmed.2019.4515; Nicolle LE. JAMA. 2019;322(12):1152-4.
FROM JAMA
Vitamin D does not improve bone density, structure in healthy patients
ORLANDO – after 2 years of daily use, according to data presented at the annual meeting of the American Society for Bone and Mineral Research.
“Participants may have already reached the vitamin D level needed for bone health,” Meryl S. LeBoff, MD, of Brigham and Women’s Hospital in Boston, said in her presentation.
Dr. LeBoff presented results from 771 patients (mean age, 63.8 years) in the Bone Health Subcohort of VITAL (Vitamin D and OmegA-3 TriaL) who were not on any bone active medications and were randomized to receive daily vitamin D3 at a dose of 2,000 IU or placebo. Patients received bone imaging at baseline and at 2 years; areal bone mineral density (aBMD) of the whole body, femoral neck, total hip, and spine was assessed via dual x-ray absorptiometry scan. Total 25-hydroxyvitamin D (25[OH]D) levels were measured via liquid chromatography tandem mass spectrometry, and free 25(OH)D levels were measured via the ELISA assay. The baseline characteristics of the vitamin D3 supplementation and placebo groups were similar. Overall, 52% of patients had osteopenia and 10.4% had osteoporosis.
Between baseline and 2 years, the vitamin D group’s total 25(OH)D levels increased from a mean 27.0 ng/mL to 39.5 ng/mL (46%) and the free 25(OH)D levels increased from 5.8 pg/mL to 9.0 pg/mL (55%), whereas levels in the placebo stayed the same. The researchers found no significant absolute percentage changes over 2 years in aBMD of the whole body (P = .60), femoral neck (P = .16), total hip (P = .23) and spine (P = .55), compared with patients in the placebo group.
In a secondary analysis, Dr. LeBoff and colleagues found no benefit to volumetric BMD (vBMD) of the radius and the tibia at 2 years, and the results persisted after they performed a sensitivity analysis. Adverse events, such as hypercalciuria, kidney stones, and gastrointestinal symptoms, were not significantly different in the vitamin D group, compared with the placebo group.
Dr. LeBoff noted among the limitations of the study that it evaluated one dose level of vitamin D and was not designed to determine whether vitamin D supplementation was effective in people with vitamin D insufficiency, and the results are not generalizable to patients with osteoporosis or osteomalacia. Future studies should also examine whether free 25(OH)D levels can be used to detect which patients can benefit from vitamin D supplementation, she added.
Risk of falls
In a separate abstract, which Dr. LeBoff presented in a different session, 12,927 patients who received vitamin D supplementation for 5 years, were studied for risk of falls, compared with 12,994 individuals in a placebo group. At baseline, 33.3% of patients had fallen at least once in the previous year, and overall 6,605 patients reported 13,235 falls. At 5.3 years of follow-up, there were no significant differences in number of falls between groups, falls leading to injury, and falls leading to a doctor or a hospital visit.
There are ongoing parallel studies examining the incidence of fractures between groups in the total population of the VITAL study (25,871 participants); bone turnover markers; bone microarchitecture measurements through high-resolution peripheral quantitative computed tomography; and examining the connection between free 25(OH)D, parathyroid hormone, and vitamin D binding protein, said Dr. LeBoff.
The study was funded in part by grants from the National Cancer Institute, the National Heart, Lung and Blood Institute, the Office of Dietary Supplements, the National Institute of Neurological Disorders and Stroke, and the National Center for Complementary and Integrative Health. Dr. LeBoff reported receiving grants from the National Institute of Arthritis Musculoskeletal and Skin Diseases. Two authors reported nonfinancial support Pharmavite LLC of Northridge, Calif., Pronova BioPharma of Norway and BASF, and Quest Diagnostics. The remaining authors reported no conflicts of interest.
SOURCE: LeBoff M et al. ASBMR 2019, Abstracts 1046 and 1057.
ORLANDO – after 2 years of daily use, according to data presented at the annual meeting of the American Society for Bone and Mineral Research.
“Participants may have already reached the vitamin D level needed for bone health,” Meryl S. LeBoff, MD, of Brigham and Women’s Hospital in Boston, said in her presentation.
Dr. LeBoff presented results from 771 patients (mean age, 63.8 years) in the Bone Health Subcohort of VITAL (Vitamin D and OmegA-3 TriaL) who were not on any bone active medications and were randomized to receive daily vitamin D3 at a dose of 2,000 IU or placebo. Patients received bone imaging at baseline and at 2 years; areal bone mineral density (aBMD) of the whole body, femoral neck, total hip, and spine was assessed via dual x-ray absorptiometry scan. Total 25-hydroxyvitamin D (25[OH]D) levels were measured via liquid chromatography tandem mass spectrometry, and free 25(OH)D levels were measured via the ELISA assay. The baseline characteristics of the vitamin D3 supplementation and placebo groups were similar. Overall, 52% of patients had osteopenia and 10.4% had osteoporosis.
Between baseline and 2 years, the vitamin D group’s total 25(OH)D levels increased from a mean 27.0 ng/mL to 39.5 ng/mL (46%) and the free 25(OH)D levels increased from 5.8 pg/mL to 9.0 pg/mL (55%), whereas levels in the placebo stayed the same. The researchers found no significant absolute percentage changes over 2 years in aBMD of the whole body (P = .60), femoral neck (P = .16), total hip (P = .23) and spine (P = .55), compared with patients in the placebo group.
In a secondary analysis, Dr. LeBoff and colleagues found no benefit to volumetric BMD (vBMD) of the radius and the tibia at 2 years, and the results persisted after they performed a sensitivity analysis. Adverse events, such as hypercalciuria, kidney stones, and gastrointestinal symptoms, were not significantly different in the vitamin D group, compared with the placebo group.
Dr. LeBoff noted among the limitations of the study that it evaluated one dose level of vitamin D and was not designed to determine whether vitamin D supplementation was effective in people with vitamin D insufficiency, and the results are not generalizable to patients with osteoporosis or osteomalacia. Future studies should also examine whether free 25(OH)D levels can be used to detect which patients can benefit from vitamin D supplementation, she added.
Risk of falls
In a separate abstract, which Dr. LeBoff presented in a different session, 12,927 patients who received vitamin D supplementation for 5 years, were studied for risk of falls, compared with 12,994 individuals in a placebo group. At baseline, 33.3% of patients had fallen at least once in the previous year, and overall 6,605 patients reported 13,235 falls. At 5.3 years of follow-up, there were no significant differences in number of falls between groups, falls leading to injury, and falls leading to a doctor or a hospital visit.
There are ongoing parallel studies examining the incidence of fractures between groups in the total population of the VITAL study (25,871 participants); bone turnover markers; bone microarchitecture measurements through high-resolution peripheral quantitative computed tomography; and examining the connection between free 25(OH)D, parathyroid hormone, and vitamin D binding protein, said Dr. LeBoff.
The study was funded in part by grants from the National Cancer Institute, the National Heart, Lung and Blood Institute, the Office of Dietary Supplements, the National Institute of Neurological Disorders and Stroke, and the National Center for Complementary and Integrative Health. Dr. LeBoff reported receiving grants from the National Institute of Arthritis Musculoskeletal and Skin Diseases. Two authors reported nonfinancial support Pharmavite LLC of Northridge, Calif., Pronova BioPharma of Norway and BASF, and Quest Diagnostics. The remaining authors reported no conflicts of interest.
SOURCE: LeBoff M et al. ASBMR 2019, Abstracts 1046 and 1057.
ORLANDO – after 2 years of daily use, according to data presented at the annual meeting of the American Society for Bone and Mineral Research.
“Participants may have already reached the vitamin D level needed for bone health,” Meryl S. LeBoff, MD, of Brigham and Women’s Hospital in Boston, said in her presentation.
Dr. LeBoff presented results from 771 patients (mean age, 63.8 years) in the Bone Health Subcohort of VITAL (Vitamin D and OmegA-3 TriaL) who were not on any bone active medications and were randomized to receive daily vitamin D3 at a dose of 2,000 IU or placebo. Patients received bone imaging at baseline and at 2 years; areal bone mineral density (aBMD) of the whole body, femoral neck, total hip, and spine was assessed via dual x-ray absorptiometry scan. Total 25-hydroxyvitamin D (25[OH]D) levels were measured via liquid chromatography tandem mass spectrometry, and free 25(OH)D levels were measured via the ELISA assay. The baseline characteristics of the vitamin D3 supplementation and placebo groups were similar. Overall, 52% of patients had osteopenia and 10.4% had osteoporosis.
Between baseline and 2 years, the vitamin D group’s total 25(OH)D levels increased from a mean 27.0 ng/mL to 39.5 ng/mL (46%) and the free 25(OH)D levels increased from 5.8 pg/mL to 9.0 pg/mL (55%), whereas levels in the placebo stayed the same. The researchers found no significant absolute percentage changes over 2 years in aBMD of the whole body (P = .60), femoral neck (P = .16), total hip (P = .23) and spine (P = .55), compared with patients in the placebo group.
In a secondary analysis, Dr. LeBoff and colleagues found no benefit to volumetric BMD (vBMD) of the radius and the tibia at 2 years, and the results persisted after they performed a sensitivity analysis. Adverse events, such as hypercalciuria, kidney stones, and gastrointestinal symptoms, were not significantly different in the vitamin D group, compared with the placebo group.
Dr. LeBoff noted among the limitations of the study that it evaluated one dose level of vitamin D and was not designed to determine whether vitamin D supplementation was effective in people with vitamin D insufficiency, and the results are not generalizable to patients with osteoporosis or osteomalacia. Future studies should also examine whether free 25(OH)D levels can be used to detect which patients can benefit from vitamin D supplementation, she added.
Risk of falls
In a separate abstract, which Dr. LeBoff presented in a different session, 12,927 patients who received vitamin D supplementation for 5 years, were studied for risk of falls, compared with 12,994 individuals in a placebo group. At baseline, 33.3% of patients had fallen at least once in the previous year, and overall 6,605 patients reported 13,235 falls. At 5.3 years of follow-up, there were no significant differences in number of falls between groups, falls leading to injury, and falls leading to a doctor or a hospital visit.
There are ongoing parallel studies examining the incidence of fractures between groups in the total population of the VITAL study (25,871 participants); bone turnover markers; bone microarchitecture measurements through high-resolution peripheral quantitative computed tomography; and examining the connection between free 25(OH)D, parathyroid hormone, and vitamin D binding protein, said Dr. LeBoff.
The study was funded in part by grants from the National Cancer Institute, the National Heart, Lung and Blood Institute, the Office of Dietary Supplements, the National Institute of Neurological Disorders and Stroke, and the National Center for Complementary and Integrative Health. Dr. LeBoff reported receiving grants from the National Institute of Arthritis Musculoskeletal and Skin Diseases. Two authors reported nonfinancial support Pharmavite LLC of Northridge, Calif., Pronova BioPharma of Norway and BASF, and Quest Diagnostics. The remaining authors reported no conflicts of interest.
SOURCE: LeBoff M et al. ASBMR 2019, Abstracts 1046 and 1057.
REPORTING FROM ASBMR 2019
Closure of women’s health clinics may negatively impact cervical cancer outcomes
.
States with a decreased number of women’s clinics per capita between 2010 and 2013 were found to have less screening for cervical cancer, more advanced stage of cervical cancer at presentation, and higher mortality from cervical cancer than states with no decrease in clinics, reported lead author Amar J. Srivastava, MD, of Washington University in St. Louis, who also noted that these changes occurred within a relatively short time frame.
“We know that women are generally diagnosed through the utilization of Pap smears,” Dr. Srivastava said during a presentation at the annual meeting of the American Society for Radiation Oncology. “These are low-cost tests that are available at multiple low-cost women’s health clinics. Unfortunately ... over the course of the past decade, we’ve seen a significant reduction of these clinics throughout the United States.”
“Between 2010 and 2013, which is the period of interest in this study, we know that about 100 of these women’s health clinics closed,” Dr. Srivastava said. “This was due to a combination of several factors; some of it was due to funding, some of it was due to restructuring of the clinics, and there were also laws passed throughout many states that ultimately led to the closure of many clinics.”
To determine the impact of these closures, the investigators first divided states into those that had women’s clinic closures between 2010 and 2013 and those that did not. Comparisons between these two cohorts involved the use of two databases. The first was the Behavioral Risk Factors Surveillance Study (BRFSS), which provided data from 197,143 cases, enabling assessment of differences between screening availability. The second database was the Surveillance, Epidemiology, and End Results (SEER) registry, which provided data from 10,652 patients, facilitating comparisons of stage at time of diagnosis and mortality rate.
Results were described in terms of relative differences between the two cohorts. For instance, screening rate among women with cervical cancer in states that had a decreased number of clinics was 1.63% lower than in states that did not lose clinics. This disparity was more pronounced in specific demographic subgroups, including Hispanic women (–5.82%), women aged between 21 and 34 years (–5.19%), unmarried women (–4.10%), and uninsured women (–6.88%).
“Historically, these are marginalized, underserved groups, and unfortunately, it comes as no surprise that these were the groups of women who were most dramatically hit by these changes,” Dr. Srivastava said.
Early-stage diagnosis was also significantly less common in states that had a decreased number of clinics, by a margin of 13.2%. Finally, the overall mortality rate among women with cervical cancer was 36% higher in states with clinic closures, a difference that climbed to 40% when comparing only metro residents.
Connecting the dots, Dr. Srivastava suggested that the decreased availability of screening may have led to fewer diagnoses at an early stage, which is more curable than late-stage disease, ultimately translating to a higher mortality rate. After noting that this chain of causality cannot be confirmed, owing to the retrospective nature of the study, Dr. Srivastava finished his presentation with a call to action.
“These findings should really give us some pause,” he said, “as physicians, as people who care about other people, to spend some time, try to figure out what’s going on, and try to address this disparity.”
After the presentation, Geraldine M. Jacobsen, MD, chair of radiation oncology at West Virginia University Cancer Institute, in Morgantown, W.V., echoed Dr. Srivastava’s concern.
“This study really raises broader questions,” Dr. Jacobsen said. “In the United States we’re always engaged in an ongoing dialogue about health care, health care policy, [and] health care costs. But a study like this brings to us the human face of what these dialogues mean. Policy affects people, and if we make changes in health care policy or health care legislation, we’re impacting people’s health and people’s lives.”
The investigators disclosed relationships with Phelps County Regional Medical Center, the Elsa U. Pardee Foundation, the American Society of Clinical Oncology, and ASTRO.
SOURCE: Srivastava AJ et al. ASTRO 2019, Abstract 202.
.
States with a decreased number of women’s clinics per capita between 2010 and 2013 were found to have less screening for cervical cancer, more advanced stage of cervical cancer at presentation, and higher mortality from cervical cancer than states with no decrease in clinics, reported lead author Amar J. Srivastava, MD, of Washington University in St. Louis, who also noted that these changes occurred within a relatively short time frame.
“We know that women are generally diagnosed through the utilization of Pap smears,” Dr. Srivastava said during a presentation at the annual meeting of the American Society for Radiation Oncology. “These are low-cost tests that are available at multiple low-cost women’s health clinics. Unfortunately ... over the course of the past decade, we’ve seen a significant reduction of these clinics throughout the United States.”
“Between 2010 and 2013, which is the period of interest in this study, we know that about 100 of these women’s health clinics closed,” Dr. Srivastava said. “This was due to a combination of several factors; some of it was due to funding, some of it was due to restructuring of the clinics, and there were also laws passed throughout many states that ultimately led to the closure of many clinics.”
To determine the impact of these closures, the investigators first divided states into those that had women’s clinic closures between 2010 and 2013 and those that did not. Comparisons between these two cohorts involved the use of two databases. The first was the Behavioral Risk Factors Surveillance Study (BRFSS), which provided data from 197,143 cases, enabling assessment of differences between screening availability. The second database was the Surveillance, Epidemiology, and End Results (SEER) registry, which provided data from 10,652 patients, facilitating comparisons of stage at time of diagnosis and mortality rate.
Results were described in terms of relative differences between the two cohorts. For instance, screening rate among women with cervical cancer in states that had a decreased number of clinics was 1.63% lower than in states that did not lose clinics. This disparity was more pronounced in specific demographic subgroups, including Hispanic women (–5.82%), women aged between 21 and 34 years (–5.19%), unmarried women (–4.10%), and uninsured women (–6.88%).
“Historically, these are marginalized, underserved groups, and unfortunately, it comes as no surprise that these were the groups of women who were most dramatically hit by these changes,” Dr. Srivastava said.
Early-stage diagnosis was also significantly less common in states that had a decreased number of clinics, by a margin of 13.2%. Finally, the overall mortality rate among women with cervical cancer was 36% higher in states with clinic closures, a difference that climbed to 40% when comparing only metro residents.
Connecting the dots, Dr. Srivastava suggested that the decreased availability of screening may have led to fewer diagnoses at an early stage, which is more curable than late-stage disease, ultimately translating to a higher mortality rate. After noting that this chain of causality cannot be confirmed, owing to the retrospective nature of the study, Dr. Srivastava finished his presentation with a call to action.
“These findings should really give us some pause,” he said, “as physicians, as people who care about other people, to spend some time, try to figure out what’s going on, and try to address this disparity.”
After the presentation, Geraldine M. Jacobsen, MD, chair of radiation oncology at West Virginia University Cancer Institute, in Morgantown, W.V., echoed Dr. Srivastava’s concern.
“This study really raises broader questions,” Dr. Jacobsen said. “In the United States we’re always engaged in an ongoing dialogue about health care, health care policy, [and] health care costs. But a study like this brings to us the human face of what these dialogues mean. Policy affects people, and if we make changes in health care policy or health care legislation, we’re impacting people’s health and people’s lives.”
The investigators disclosed relationships with Phelps County Regional Medical Center, the Elsa U. Pardee Foundation, the American Society of Clinical Oncology, and ASTRO.
SOURCE: Srivastava AJ et al. ASTRO 2019, Abstract 202.
.
States with a decreased number of women’s clinics per capita between 2010 and 2013 were found to have less screening for cervical cancer, more advanced stage of cervical cancer at presentation, and higher mortality from cervical cancer than states with no decrease in clinics, reported lead author Amar J. Srivastava, MD, of Washington University in St. Louis, who also noted that these changes occurred within a relatively short time frame.
“We know that women are generally diagnosed through the utilization of Pap smears,” Dr. Srivastava said during a presentation at the annual meeting of the American Society for Radiation Oncology. “These are low-cost tests that are available at multiple low-cost women’s health clinics. Unfortunately ... over the course of the past decade, we’ve seen a significant reduction of these clinics throughout the United States.”
“Between 2010 and 2013, which is the period of interest in this study, we know that about 100 of these women’s health clinics closed,” Dr. Srivastava said. “This was due to a combination of several factors; some of it was due to funding, some of it was due to restructuring of the clinics, and there were also laws passed throughout many states that ultimately led to the closure of many clinics.”
To determine the impact of these closures, the investigators first divided states into those that had women’s clinic closures between 2010 and 2013 and those that did not. Comparisons between these two cohorts involved the use of two databases. The first was the Behavioral Risk Factors Surveillance Study (BRFSS), which provided data from 197,143 cases, enabling assessment of differences between screening availability. The second database was the Surveillance, Epidemiology, and End Results (SEER) registry, which provided data from 10,652 patients, facilitating comparisons of stage at time of diagnosis and mortality rate.
Results were described in terms of relative differences between the two cohorts. For instance, screening rate among women with cervical cancer in states that had a decreased number of clinics was 1.63% lower than in states that did not lose clinics. This disparity was more pronounced in specific demographic subgroups, including Hispanic women (–5.82%), women aged between 21 and 34 years (–5.19%), unmarried women (–4.10%), and uninsured women (–6.88%).
“Historically, these are marginalized, underserved groups, and unfortunately, it comes as no surprise that these were the groups of women who were most dramatically hit by these changes,” Dr. Srivastava said.
Early-stage diagnosis was also significantly less common in states that had a decreased number of clinics, by a margin of 13.2%. Finally, the overall mortality rate among women with cervical cancer was 36% higher in states with clinic closures, a difference that climbed to 40% when comparing only metro residents.
Connecting the dots, Dr. Srivastava suggested that the decreased availability of screening may have led to fewer diagnoses at an early stage, which is more curable than late-stage disease, ultimately translating to a higher mortality rate. After noting that this chain of causality cannot be confirmed, owing to the retrospective nature of the study, Dr. Srivastava finished his presentation with a call to action.
“These findings should really give us some pause,” he said, “as physicians, as people who care about other people, to spend some time, try to figure out what’s going on, and try to address this disparity.”
After the presentation, Geraldine M. Jacobsen, MD, chair of radiation oncology at West Virginia University Cancer Institute, in Morgantown, W.V., echoed Dr. Srivastava’s concern.
“This study really raises broader questions,” Dr. Jacobsen said. “In the United States we’re always engaged in an ongoing dialogue about health care, health care policy, [and] health care costs. But a study like this brings to us the human face of what these dialogues mean. Policy affects people, and if we make changes in health care policy or health care legislation, we’re impacting people’s health and people’s lives.”
The investigators disclosed relationships with Phelps County Regional Medical Center, the Elsa U. Pardee Foundation, the American Society of Clinical Oncology, and ASTRO.
SOURCE: Srivastava AJ et al. ASTRO 2019, Abstract 202.
REPORTING FROM ASTRO 2019
No link found between sleep position, pregnancy outcomes
such as stillbirth, small-for-gestational-age (SGA) newborns, and gestational hypertensive disorders. The finding, published in the October issue of Obstetrics & Gynecology, conflicts with previous retrospective case-control studies that suggested left-side sleeping may lead to reduced risk. The disagreement may be due to the prospective nature of the new study, which followed 8,706 women over 4 years.
Right-side or back sleeping had attracted suspicion because of its potential to compress uterine blood vessels and decrease uterine blood flow, and various public health campaigns urge pregnant women to sleep on their left side. Although case-control studies backed up those worries, those retrospective approaches can suffer from limitations including recall bias, in which grieving mothers overreport suspect sleeping behaviors, perhaps in search of an explanation for their loss.
Prospective analyses can counter some of those limitations. The researchers, led by Robert Silver, MD, of the University of Utah, Salt Lake City, conducted a secondary analysis of the nuMoM2b study, examining adverse pregnancy outcomes and risk factors. It was a multicenter observational cohort study of 8,706 nulliparous women with singleton gestations who completed two sleep questionnaires: one between 6 and 13 weeks of gestation, and one between 22 and 29 weeks.
Adverse outcomes occurred in 1,903 women, including 178 cases of both SGA and hypertensive disorders, 8 with SGA plus stillbirth, 3 with hypertensive disorders plus stillbirth, and 2 cases with all three complications.
The researchers found no association between any adverse outcomes and sleep position either at the first visit in early pregnancy (adjusted odds ratio, 1.00; 95% confidence interval, 0.89-1.14) or the third visit in midpregnancy (aOR, 0.99; 95% CI, 0.89-1.11). Propensity score matching to adjust non-left lateral positioning to the composite outcome also showed no association.
In midpregnancy, there was an association between non-left lateral sleeping and reduced risk of stillbirth (aOR, 0.27; 95% CI, 0.09-0.75). “This observation is likely spurious owing to small numbers of stillbirths, Dr. Silver and associates said.
A post hoc analysis indicated that the study was sufficiently powered to detect clinically meaningful risks; ORs of 1.2 for hypertensive disorders, 1.23 for SGA, 2.4 for stillbirth, and 1.2 for the composite outcome.
Let sleeping mothers lie
Pregnant women have enough on their minds. They shouldn’t have to worry about sleep position as well, according to Nathan Fox, MD, professor of obstetrics, gynecology, and reproductive science at Icahn School of Medicine at Mount Sinai, New York, and Emily Oster, PhD, economist at Brown University, Providence, R.I., who wrote an accompanying editorial.
It may seem harmless to direct women to sleep on their left side even if it has no benefit, but restricting a woman’s sleep options may leave her less well rested at a time when she is about to enter a period of sleep deprivation, as well as contribute to general discomfort, in their opinion.
Also, in the rare cases of a bad outcome, advice based on limited or poor quality evidence contributes to “devastating and unwarranted feelings of responsibility and guilt, and this harm to women already suffering from sadness and despair must not be minimized,” they said.
The study received funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and grants from multiple universities. One author received research funding from Kyndermed through her institution. Another receives royalties from UpToDate.com. Dr. Fox and Dr. Oster have no relevant financial disclosures.
SOURCES: Silver RM et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003458; Fox N and Oster E. Obstet. Gynecol. 2019 Oct. doi: 10.1097/AOG.0000000000003466
such as stillbirth, small-for-gestational-age (SGA) newborns, and gestational hypertensive disorders. The finding, published in the October issue of Obstetrics & Gynecology, conflicts with previous retrospective case-control studies that suggested left-side sleeping may lead to reduced risk. The disagreement may be due to the prospective nature of the new study, which followed 8,706 women over 4 years.
Right-side or back sleeping had attracted suspicion because of its potential to compress uterine blood vessels and decrease uterine blood flow, and various public health campaigns urge pregnant women to sleep on their left side. Although case-control studies backed up those worries, those retrospective approaches can suffer from limitations including recall bias, in which grieving mothers overreport suspect sleeping behaviors, perhaps in search of an explanation for their loss.
Prospective analyses can counter some of those limitations. The researchers, led by Robert Silver, MD, of the University of Utah, Salt Lake City, conducted a secondary analysis of the nuMoM2b study, examining adverse pregnancy outcomes and risk factors. It was a multicenter observational cohort study of 8,706 nulliparous women with singleton gestations who completed two sleep questionnaires: one between 6 and 13 weeks of gestation, and one between 22 and 29 weeks.
Adverse outcomes occurred in 1,903 women, including 178 cases of both SGA and hypertensive disorders, 8 with SGA plus stillbirth, 3 with hypertensive disorders plus stillbirth, and 2 cases with all three complications.
The researchers found no association between any adverse outcomes and sleep position either at the first visit in early pregnancy (adjusted odds ratio, 1.00; 95% confidence interval, 0.89-1.14) or the third visit in midpregnancy (aOR, 0.99; 95% CI, 0.89-1.11). Propensity score matching to adjust non-left lateral positioning to the composite outcome also showed no association.
In midpregnancy, there was an association between non-left lateral sleeping and reduced risk of stillbirth (aOR, 0.27; 95% CI, 0.09-0.75). “This observation is likely spurious owing to small numbers of stillbirths, Dr. Silver and associates said.
A post hoc analysis indicated that the study was sufficiently powered to detect clinically meaningful risks; ORs of 1.2 for hypertensive disorders, 1.23 for SGA, 2.4 for stillbirth, and 1.2 for the composite outcome.
Let sleeping mothers lie
Pregnant women have enough on their minds. They shouldn’t have to worry about sleep position as well, according to Nathan Fox, MD, professor of obstetrics, gynecology, and reproductive science at Icahn School of Medicine at Mount Sinai, New York, and Emily Oster, PhD, economist at Brown University, Providence, R.I., who wrote an accompanying editorial.
It may seem harmless to direct women to sleep on their left side even if it has no benefit, but restricting a woman’s sleep options may leave her less well rested at a time when she is about to enter a period of sleep deprivation, as well as contribute to general discomfort, in their opinion.
Also, in the rare cases of a bad outcome, advice based on limited or poor quality evidence contributes to “devastating and unwarranted feelings of responsibility and guilt, and this harm to women already suffering from sadness and despair must not be minimized,” they said.
The study received funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and grants from multiple universities. One author received research funding from Kyndermed through her institution. Another receives royalties from UpToDate.com. Dr. Fox and Dr. Oster have no relevant financial disclosures.
SOURCES: Silver RM et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003458; Fox N and Oster E. Obstet. Gynecol. 2019 Oct. doi: 10.1097/AOG.0000000000003466
such as stillbirth, small-for-gestational-age (SGA) newborns, and gestational hypertensive disorders. The finding, published in the October issue of Obstetrics & Gynecology, conflicts with previous retrospective case-control studies that suggested left-side sleeping may lead to reduced risk. The disagreement may be due to the prospective nature of the new study, which followed 8,706 women over 4 years.
Right-side or back sleeping had attracted suspicion because of its potential to compress uterine blood vessels and decrease uterine blood flow, and various public health campaigns urge pregnant women to sleep on their left side. Although case-control studies backed up those worries, those retrospective approaches can suffer from limitations including recall bias, in which grieving mothers overreport suspect sleeping behaviors, perhaps in search of an explanation for their loss.
Prospective analyses can counter some of those limitations. The researchers, led by Robert Silver, MD, of the University of Utah, Salt Lake City, conducted a secondary analysis of the nuMoM2b study, examining adverse pregnancy outcomes and risk factors. It was a multicenter observational cohort study of 8,706 nulliparous women with singleton gestations who completed two sleep questionnaires: one between 6 and 13 weeks of gestation, and one between 22 and 29 weeks.
Adverse outcomes occurred in 1,903 women, including 178 cases of both SGA and hypertensive disorders, 8 with SGA plus stillbirth, 3 with hypertensive disorders plus stillbirth, and 2 cases with all three complications.
The researchers found no association between any adverse outcomes and sleep position either at the first visit in early pregnancy (adjusted odds ratio, 1.00; 95% confidence interval, 0.89-1.14) or the third visit in midpregnancy (aOR, 0.99; 95% CI, 0.89-1.11). Propensity score matching to adjust non-left lateral positioning to the composite outcome also showed no association.
In midpregnancy, there was an association between non-left lateral sleeping and reduced risk of stillbirth (aOR, 0.27; 95% CI, 0.09-0.75). “This observation is likely spurious owing to small numbers of stillbirths, Dr. Silver and associates said.
A post hoc analysis indicated that the study was sufficiently powered to detect clinically meaningful risks; ORs of 1.2 for hypertensive disorders, 1.23 for SGA, 2.4 for stillbirth, and 1.2 for the composite outcome.
Let sleeping mothers lie
Pregnant women have enough on their minds. They shouldn’t have to worry about sleep position as well, according to Nathan Fox, MD, professor of obstetrics, gynecology, and reproductive science at Icahn School of Medicine at Mount Sinai, New York, and Emily Oster, PhD, economist at Brown University, Providence, R.I., who wrote an accompanying editorial.
It may seem harmless to direct women to sleep on their left side even if it has no benefit, but restricting a woman’s sleep options may leave her less well rested at a time when she is about to enter a period of sleep deprivation, as well as contribute to general discomfort, in their opinion.
Also, in the rare cases of a bad outcome, advice based on limited or poor quality evidence contributes to “devastating and unwarranted feelings of responsibility and guilt, and this harm to women already suffering from sadness and despair must not be minimized,” they said.
The study received funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and grants from multiple universities. One author received research funding from Kyndermed through her institution. Another receives royalties from UpToDate.com. Dr. Fox and Dr. Oster have no relevant financial disclosures.
SOURCES: Silver RM et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003458; Fox N and Oster E. Obstet. Gynecol. 2019 Oct. doi: 10.1097/AOG.0000000000003466
FROM OBSTETRICS & GYNECOLOGY