User login
Smart health devices – promises and pitfalls
What needs to be done before the data deluge hits the office
Hurricane Sally recently crossed the Gulf of Mexico and landed with torrential rainfalls along the Alabama coast. A little rainfall is important for crops; too much leads to devastation. As physicians, we need data in order to help manage patients’ illnesses and to help to keep them healthy. Our fear though is that too much data provided too quickly may have the opposite effect.
Personal monitoring devices
When I bought my first Fitbit 7 years ago, I was enamored with the technology. The Fitbit was little more than a step tracker, yet I proudly wore its black rubber strap on my wrist. It was my first foray into wearable technology, and it felt quite empowering to have an objective way to track my fitness beyond just using my bathroom scale. Now less than a decade later, that Fitbit looks archaic in comparison with the wrist-top technology currently available.
As I write this, the world’s largest technology company is in the process of releasing its sixth-generation Apple Watch. In addition to acting as a smartphone, this new device, which is barely larger than a postage stamp, offers GPS-based movement tracking, the ability to detect falls, continuous heart rate monitoring, a built-in EKG capable of diagnosing atrial fibrillation, and an oxygen saturation sensor. These features weren’t added thoughtlessly. Apple is marketing this as a health-focused device, with their primary advertising campaign claiming that “the future of health is on your wrist,” and they aren’t the only company making this play.
Along with Apple, Samsung, Withings, Fitbit, and other companies continue to bring products to market that monitor our activity and provide new insights into our health. Typically linked to smartphone-based apps, these devices record all of their measurements for later review, while software helps interpret the findings to make them actionable. From heart rate tracking to sleep analysis, these options now provide access to volumes of data that promise to improve our wellness and change our lives. Of course, those promises will only be fulfilled if our behavior is altered as a consequence of having more detailed information. Whether that will happen remains to be seen.
Health system–linked devices
Major advancements in medical monitoring technology are now enabling physicians to get much deeper insight into their patients’ health status. Internet-connected scales, blood pressure cuffs, and exercise equipment offer the ability to upload information into patient portals and integrate that information into EHRs. New devices provide access to information that previously was impossible to obtain. For example, wearable continuous blood glucose monitors, such as the FreeStyle Libre or DexCom’s G6, allow patients and physicians to follow blood sugar readings 24 hours a day. This provides unprecedented awareness of diabetes control and relieves the pain and inconvenience of finger sticks and blood draws. It also aids with compliance because patients don’t need to remember to check their sugar levels on a schedule.
Other compliance-boosting breakthroughs, such as Bluetooth-enabled asthma inhalers and cellular-connected continuous positive airway pressure machines, assist patients with managing chronic respiratory conditions. Many companies are developing technologies to manage acute conditions as well. One such company, an on-demand telemedicine provider called TytoCare, has developed a $299 suite of instruments that includes a digital stethoscope, thermometer, and camera-based otoscope. In concert with a virtual visit, their providers can remotely use these tools to examine and assess sick individuals. This virtual “laying on of hands” may have sounded like science fiction and likely would have been rejected by patients just a few years ago. Now it is becoming commonplace and will soon be an expectation of many seeking care.
But if we are to be successful, everyone must acknowledge that this revolution in health care brings many challenges along with it. One of those is the deluge of data that connected devices provide.
Information overload
There is such a thing as “too much of a good thing.” Described by journalist David Shenk as “data smog” in his 1997 book of the same name, the idea is clear: There is only so much information we can assimilate.
Even after years of using EHRs and with government-implemented incentives that promote “meaningful use,” physicians are still struggling with EHRs. Additionally, many have expressed frustration with the connectedness that EHRs provide and lament their inability to ever really “leave the office.” As more and more data become available to physicians, the challenge of how to assimilate and act on those data will continue to grow. The addition of patient-provided health statistics will only make information overload worse, with clinicians will feeling an ever-growing burden to know, understand, and act on this information.
Unless we develop systems to sort, filter, and prioritize the flow of information, there is potential for liability from not acting on the amount of virtual information doctors receive. This new risk for already fatigued and overburdened physicians combined with an increase in the amount of virtual information at doctors’ fingertips may lead to the value of patient data being lost.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Follow him on Twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
What needs to be done before the data deluge hits the office
What needs to be done before the data deluge hits the office
Hurricane Sally recently crossed the Gulf of Mexico and landed with torrential rainfalls along the Alabama coast. A little rainfall is important for crops; too much leads to devastation. As physicians, we need data in order to help manage patients’ illnesses and to help to keep them healthy. Our fear though is that too much data provided too quickly may have the opposite effect.
Personal monitoring devices
When I bought my first Fitbit 7 years ago, I was enamored with the technology. The Fitbit was little more than a step tracker, yet I proudly wore its black rubber strap on my wrist. It was my first foray into wearable technology, and it felt quite empowering to have an objective way to track my fitness beyond just using my bathroom scale. Now less than a decade later, that Fitbit looks archaic in comparison with the wrist-top technology currently available.
As I write this, the world’s largest technology company is in the process of releasing its sixth-generation Apple Watch. In addition to acting as a smartphone, this new device, which is barely larger than a postage stamp, offers GPS-based movement tracking, the ability to detect falls, continuous heart rate monitoring, a built-in EKG capable of diagnosing atrial fibrillation, and an oxygen saturation sensor. These features weren’t added thoughtlessly. Apple is marketing this as a health-focused device, with their primary advertising campaign claiming that “the future of health is on your wrist,” and they aren’t the only company making this play.
Along with Apple, Samsung, Withings, Fitbit, and other companies continue to bring products to market that monitor our activity and provide new insights into our health. Typically linked to smartphone-based apps, these devices record all of their measurements for later review, while software helps interpret the findings to make them actionable. From heart rate tracking to sleep analysis, these options now provide access to volumes of data that promise to improve our wellness and change our lives. Of course, those promises will only be fulfilled if our behavior is altered as a consequence of having more detailed information. Whether that will happen remains to be seen.
Health system–linked devices
Major advancements in medical monitoring technology are now enabling physicians to get much deeper insight into their patients’ health status. Internet-connected scales, blood pressure cuffs, and exercise equipment offer the ability to upload information into patient portals and integrate that information into EHRs. New devices provide access to information that previously was impossible to obtain. For example, wearable continuous blood glucose monitors, such as the FreeStyle Libre or DexCom’s G6, allow patients and physicians to follow blood sugar readings 24 hours a day. This provides unprecedented awareness of diabetes control and relieves the pain and inconvenience of finger sticks and blood draws. It also aids with compliance because patients don’t need to remember to check their sugar levels on a schedule.
Other compliance-boosting breakthroughs, such as Bluetooth-enabled asthma inhalers and cellular-connected continuous positive airway pressure machines, assist patients with managing chronic respiratory conditions. Many companies are developing technologies to manage acute conditions as well. One such company, an on-demand telemedicine provider called TytoCare, has developed a $299 suite of instruments that includes a digital stethoscope, thermometer, and camera-based otoscope. In concert with a virtual visit, their providers can remotely use these tools to examine and assess sick individuals. This virtual “laying on of hands” may have sounded like science fiction and likely would have been rejected by patients just a few years ago. Now it is becoming commonplace and will soon be an expectation of many seeking care.
But if we are to be successful, everyone must acknowledge that this revolution in health care brings many challenges along with it. One of those is the deluge of data that connected devices provide.
Information overload
There is such a thing as “too much of a good thing.” Described by journalist David Shenk as “data smog” in his 1997 book of the same name, the idea is clear: There is only so much information we can assimilate.
Even after years of using EHRs and with government-implemented incentives that promote “meaningful use,” physicians are still struggling with EHRs. Additionally, many have expressed frustration with the connectedness that EHRs provide and lament their inability to ever really “leave the office.” As more and more data become available to physicians, the challenge of how to assimilate and act on those data will continue to grow. The addition of patient-provided health statistics will only make information overload worse, with clinicians will feeling an ever-growing burden to know, understand, and act on this information.
Unless we develop systems to sort, filter, and prioritize the flow of information, there is potential for liability from not acting on the amount of virtual information doctors receive. This new risk for already fatigued and overburdened physicians combined with an increase in the amount of virtual information at doctors’ fingertips may lead to the value of patient data being lost.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Follow him on Twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
Hurricane Sally recently crossed the Gulf of Mexico and landed with torrential rainfalls along the Alabama coast. A little rainfall is important for crops; too much leads to devastation. As physicians, we need data in order to help manage patients’ illnesses and to help to keep them healthy. Our fear though is that too much data provided too quickly may have the opposite effect.
Personal monitoring devices
When I bought my first Fitbit 7 years ago, I was enamored with the technology. The Fitbit was little more than a step tracker, yet I proudly wore its black rubber strap on my wrist. It was my first foray into wearable technology, and it felt quite empowering to have an objective way to track my fitness beyond just using my bathroom scale. Now less than a decade later, that Fitbit looks archaic in comparison with the wrist-top technology currently available.
As I write this, the world’s largest technology company is in the process of releasing its sixth-generation Apple Watch. In addition to acting as a smartphone, this new device, which is barely larger than a postage stamp, offers GPS-based movement tracking, the ability to detect falls, continuous heart rate monitoring, a built-in EKG capable of diagnosing atrial fibrillation, and an oxygen saturation sensor. These features weren’t added thoughtlessly. Apple is marketing this as a health-focused device, with their primary advertising campaign claiming that “the future of health is on your wrist,” and they aren’t the only company making this play.
Along with Apple, Samsung, Withings, Fitbit, and other companies continue to bring products to market that monitor our activity and provide new insights into our health. Typically linked to smartphone-based apps, these devices record all of their measurements for later review, while software helps interpret the findings to make them actionable. From heart rate tracking to sleep analysis, these options now provide access to volumes of data that promise to improve our wellness and change our lives. Of course, those promises will only be fulfilled if our behavior is altered as a consequence of having more detailed information. Whether that will happen remains to be seen.
Health system–linked devices
Major advancements in medical monitoring technology are now enabling physicians to get much deeper insight into their patients’ health status. Internet-connected scales, blood pressure cuffs, and exercise equipment offer the ability to upload information into patient portals and integrate that information into EHRs. New devices provide access to information that previously was impossible to obtain. For example, wearable continuous blood glucose monitors, such as the FreeStyle Libre or DexCom’s G6, allow patients and physicians to follow blood sugar readings 24 hours a day. This provides unprecedented awareness of diabetes control and relieves the pain and inconvenience of finger sticks and blood draws. It also aids with compliance because patients don’t need to remember to check their sugar levels on a schedule.
Other compliance-boosting breakthroughs, such as Bluetooth-enabled asthma inhalers and cellular-connected continuous positive airway pressure machines, assist patients with managing chronic respiratory conditions. Many companies are developing technologies to manage acute conditions as well. One such company, an on-demand telemedicine provider called TytoCare, has developed a $299 suite of instruments that includes a digital stethoscope, thermometer, and camera-based otoscope. In concert with a virtual visit, their providers can remotely use these tools to examine and assess sick individuals. This virtual “laying on of hands” may have sounded like science fiction and likely would have been rejected by patients just a few years ago. Now it is becoming commonplace and will soon be an expectation of many seeking care.
But if we are to be successful, everyone must acknowledge that this revolution in health care brings many challenges along with it. One of those is the deluge of data that connected devices provide.
Information overload
There is such a thing as “too much of a good thing.” Described by journalist David Shenk as “data smog” in his 1997 book of the same name, the idea is clear: There is only so much information we can assimilate.
Even after years of using EHRs and with government-implemented incentives that promote “meaningful use,” physicians are still struggling with EHRs. Additionally, many have expressed frustration with the connectedness that EHRs provide and lament their inability to ever really “leave the office.” As more and more data become available to physicians, the challenge of how to assimilate and act on those data will continue to grow. The addition of patient-provided health statistics will only make information overload worse, with clinicians will feeling an ever-growing burden to know, understand, and act on this information.
Unless we develop systems to sort, filter, and prioritize the flow of information, there is potential for liability from not acting on the amount of virtual information doctors receive. This new risk for already fatigued and overburdened physicians combined with an increase in the amount of virtual information at doctors’ fingertips may lead to the value of patient data being lost.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Follow him on Twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
EMA panel backs baricitinib for moderate to severe atopic dermatitis
The
Baricitinib (Olumiant) is already approved in the European Union and the United States to treat moderate to severe active rheumatoid arthritis.
If approved in Europe, it will be the first Janus kinase (JAK) inhibitor and first oral medication indicated to treat patients with AD.
The CHMP’s positive opinion on baricitinib for AD was based on three phase 3, randomized, double-blind, placebo-controlled studies where the JAK inhibitor was used alone or in combination with topical treatments in adults with moderate to severe AD for whom topical treatments were insufficient or not tolerated. In all three studies, baricitinib was shown to be more effective than placebo in achieving skin that is “clear” or “almost clear” at 16 weeks.
“Patients living with AD face difficulties on a daily basis, and this CHMP opinion marks an important milestone in providing adult AD patients with a new potential treatment option,” Thomas Bieber, MD, PhD, professor of dermatology and allergy, University of Bonn (Germany), said in a company news release.
The most common side effects with baricitinib in clinical trials include increased LDL cholesterol, upper respiratory tract infections, and headache.
Patients receiving baricitinib, particularly in combination with immunosuppressants, are at risk of developing serious infections that may lead to hospitalization or death. If a serious infection develops, baricitinib should be stopped until the infection is controlled.
The CHMP’s positive opinion will be sent to the European Commission, which will adopt a final decision regarding an European Union–wide marketing authorization. Once granted, each member state will make decisions about price and reimbursement, taking into account the potential role/use of baricitinib in the context of that country’s national health system.
A version of this story originally appeared on Medscape.com.
The
Baricitinib (Olumiant) is already approved in the European Union and the United States to treat moderate to severe active rheumatoid arthritis.
If approved in Europe, it will be the first Janus kinase (JAK) inhibitor and first oral medication indicated to treat patients with AD.
The CHMP’s positive opinion on baricitinib for AD was based on three phase 3, randomized, double-blind, placebo-controlled studies where the JAK inhibitor was used alone or in combination with topical treatments in adults with moderate to severe AD for whom topical treatments were insufficient or not tolerated. In all three studies, baricitinib was shown to be more effective than placebo in achieving skin that is “clear” or “almost clear” at 16 weeks.
“Patients living with AD face difficulties on a daily basis, and this CHMP opinion marks an important milestone in providing adult AD patients with a new potential treatment option,” Thomas Bieber, MD, PhD, professor of dermatology and allergy, University of Bonn (Germany), said in a company news release.
The most common side effects with baricitinib in clinical trials include increased LDL cholesterol, upper respiratory tract infections, and headache.
Patients receiving baricitinib, particularly in combination with immunosuppressants, are at risk of developing serious infections that may lead to hospitalization or death. If a serious infection develops, baricitinib should be stopped until the infection is controlled.
The CHMP’s positive opinion will be sent to the European Commission, which will adopt a final decision regarding an European Union–wide marketing authorization. Once granted, each member state will make decisions about price and reimbursement, taking into account the potential role/use of baricitinib in the context of that country’s national health system.
A version of this story originally appeared on Medscape.com.
The
Baricitinib (Olumiant) is already approved in the European Union and the United States to treat moderate to severe active rheumatoid arthritis.
If approved in Europe, it will be the first Janus kinase (JAK) inhibitor and first oral medication indicated to treat patients with AD.
The CHMP’s positive opinion on baricitinib for AD was based on three phase 3, randomized, double-blind, placebo-controlled studies where the JAK inhibitor was used alone or in combination with topical treatments in adults with moderate to severe AD for whom topical treatments were insufficient or not tolerated. In all three studies, baricitinib was shown to be more effective than placebo in achieving skin that is “clear” or “almost clear” at 16 weeks.
“Patients living with AD face difficulties on a daily basis, and this CHMP opinion marks an important milestone in providing adult AD patients with a new potential treatment option,” Thomas Bieber, MD, PhD, professor of dermatology and allergy, University of Bonn (Germany), said in a company news release.
The most common side effects with baricitinib in clinical trials include increased LDL cholesterol, upper respiratory tract infections, and headache.
Patients receiving baricitinib, particularly in combination with immunosuppressants, are at risk of developing serious infections that may lead to hospitalization or death. If a serious infection develops, baricitinib should be stopped until the infection is controlled.
The CHMP’s positive opinion will be sent to the European Commission, which will adopt a final decision regarding an European Union–wide marketing authorization. Once granted, each member state will make decisions about price and reimbursement, taking into account the potential role/use of baricitinib in the context of that country’s national health system.
A version of this story originally appeared on Medscape.com.
Palbociclib plus letrozole improves PFS in advanced endometrial cancer
Adding palbociclib to letrozole significantly prolonged progression-free survival (PFS) in a phase 2 trial of patients with advanced or recurrent estrogen receptor (ER)–positive endometrial cancer.
This was the first randomized trial to evaluate the efficacy of a CDK4/6 inhibitor in combination with an aromatase inhibitor in patients with advanced or recurrent ER-positive endometrial cancer, noted study investigator Mansoor Raza Mirza, MD, PhD, of Rigshospitalet Copenhagen University Hospital.
Dr. Mirza presented results from this study, ENGOT-EN3-NSGO/PALEO, at the European Society for Medical Oncology Virtual Congress 2020.
Palbociclib is a selective inhibitor of CDK4/, both of which are involved in cell-cycle transitions, Dr. Mirza explained. He observed that endometrial endometrioid adenocarcinomas are hormone dependent, and endocrine treatment with an aromatase inhibitor is well established. Palbociclib has been shown to be superior, when combined with letrozole, to letrozole alone in ER-positive breast cancer.
For the ENGOT-EN3-NSGO/PALEO study, investigators enrolled 77 patients with ER-positive advanced/recurrent endometrial cancer. Patients had received at least one prior systemic therapy, no prior endocrine therapy (except medroxyprogesterone acetate and megestrol acetate), and no prior CDK inhibitor.
Patients were randomized 1:1 to receive oral letrozole (2.5 mg on days 1-28) and either palbociclib (125 mg on days 1-21) or placebo (125 mg on days 1-21) in a 28-day cycle until progression. Baseline characteristics were similar between the treatment arms.
Efficacy
Of the 77 patients enrolled, 73 were evaluable. The primary endpoint was PFS.
The median PFS in the intention-to-treat population was 3.0 months in the placebo arm and 8.3 months in the palbociclib arm (hazard ratio, 0.56; 95% confidence interval, 0.32-0.98; P = .0376).
Looking at stratification factors, PFS was higher in the palbociclib arm among the large majority (about 85%) of patients who had received no prior medroxyprogesterone acetate or megestrol acetate (HR, 0.55; 95% CI, 0.29-01.04; P = .0615) and among patients with relapsed disease (HR, 0.61; 95% CI, 0.34-1.09; P = .0916).
The disease control rate at 24 weeks, a secondary endpoint, was 63.6% in the palbociclib arm and 37.8% in the placebo arm.
Safety
Adverse events were more frequent in the palbociclib arm, with neutropenia being the most common.
Rates of adverse event–related dose reduction (to 100 mg or 75 mg) were 36% in the palbociclib arm and 3% in the placebo arm.
Adverse event–related discontinuation rates were 25% and 19% for palbociclib and letrozole, respectively, in the palbociclib arm and 14% and 11% for placebo and letrozole, respectively, in the placebo arm.
“The toxicity of palbociclib plus letrozole combination therapy was manageable, with most patients remaining on treatment until disease progression,” Dr. Mirza said.
He noted that an analysis of patient-reported outcomes revealed no detrimental effect on quality of life with the combination therapy.
Next steps
“Compared with placebo plus letrozole, the combination of palbociclib plus letrozole demonstrated clinically meaningful improvement in PFS,” Dr. Mirza said. “These results merit a phase 3 validation trial.”
“There is a huge rationale for using this drug in endometrial cancer,” commented study discussant Domenica Lorusso, MD, PhD, of Fondazione Policlinico Universitario Agostino Gemelli IRCCS and Catholic University of Sacred Hearth in Rome.
Dr. Lorusso said hormone receptor expression, which has been identified as a strong predictor of CDK4/6 inhibitor activity, is present in up to 90% of patients with type 1 and in about 65% of patients with type 2 endometrial cancer.
Response rates, in experience with aromatase inhibitors, have been “quite disappointing, in the 10%-20% range,” Dr. Lorusso said, with “dismal prognosis” and guidelines stating that “no standard second-line treatment has been identified.”
This research was sponsored by investigators, but Pfizer provided a study grant. Dr. Mirza disclosed relationships with Pfizer, AstraZeneca, Biocad, Clovis Oncology, Genmab, Karyopharm Therapeutics, Merck, Oncology Adventure, Roche, Seattle Genetics, Sera Prognostics, Sotio, GlaxoSmithKline, Zai Lab, and Boehringer Ingelheim. Dr. Lorusso disclosed relationships with AstraZeneca, Biocad, Clovis Oncology, Genmab, Merck, Roche, Tesaro, Amgen, Immunogen, and Pharma Mar.
SOURCE: Mirza MR et al. ESMO 2020, Abstract LBA28.
Adding palbociclib to letrozole significantly prolonged progression-free survival (PFS) in a phase 2 trial of patients with advanced or recurrent estrogen receptor (ER)–positive endometrial cancer.
This was the first randomized trial to evaluate the efficacy of a CDK4/6 inhibitor in combination with an aromatase inhibitor in patients with advanced or recurrent ER-positive endometrial cancer, noted study investigator Mansoor Raza Mirza, MD, PhD, of Rigshospitalet Copenhagen University Hospital.
Dr. Mirza presented results from this study, ENGOT-EN3-NSGO/PALEO, at the European Society for Medical Oncology Virtual Congress 2020.
Palbociclib is a selective inhibitor of CDK4/, both of which are involved in cell-cycle transitions, Dr. Mirza explained. He observed that endometrial endometrioid adenocarcinomas are hormone dependent, and endocrine treatment with an aromatase inhibitor is well established. Palbociclib has been shown to be superior, when combined with letrozole, to letrozole alone in ER-positive breast cancer.
For the ENGOT-EN3-NSGO/PALEO study, investigators enrolled 77 patients with ER-positive advanced/recurrent endometrial cancer. Patients had received at least one prior systemic therapy, no prior endocrine therapy (except medroxyprogesterone acetate and megestrol acetate), and no prior CDK inhibitor.
Patients were randomized 1:1 to receive oral letrozole (2.5 mg on days 1-28) and either palbociclib (125 mg on days 1-21) or placebo (125 mg on days 1-21) in a 28-day cycle until progression. Baseline characteristics were similar between the treatment arms.
Efficacy
Of the 77 patients enrolled, 73 were evaluable. The primary endpoint was PFS.
The median PFS in the intention-to-treat population was 3.0 months in the placebo arm and 8.3 months in the palbociclib arm (hazard ratio, 0.56; 95% confidence interval, 0.32-0.98; P = .0376).
Looking at stratification factors, PFS was higher in the palbociclib arm among the large majority (about 85%) of patients who had received no prior medroxyprogesterone acetate or megestrol acetate (HR, 0.55; 95% CI, 0.29-01.04; P = .0615) and among patients with relapsed disease (HR, 0.61; 95% CI, 0.34-1.09; P = .0916).
The disease control rate at 24 weeks, a secondary endpoint, was 63.6% in the palbociclib arm and 37.8% in the placebo arm.
Safety
Adverse events were more frequent in the palbociclib arm, with neutropenia being the most common.
Rates of adverse event–related dose reduction (to 100 mg or 75 mg) were 36% in the palbociclib arm and 3% in the placebo arm.
Adverse event–related discontinuation rates were 25% and 19% for palbociclib and letrozole, respectively, in the palbociclib arm and 14% and 11% for placebo and letrozole, respectively, in the placebo arm.
“The toxicity of palbociclib plus letrozole combination therapy was manageable, with most patients remaining on treatment until disease progression,” Dr. Mirza said.
He noted that an analysis of patient-reported outcomes revealed no detrimental effect on quality of life with the combination therapy.
Next steps
“Compared with placebo plus letrozole, the combination of palbociclib plus letrozole demonstrated clinically meaningful improvement in PFS,” Dr. Mirza said. “These results merit a phase 3 validation trial.”
“There is a huge rationale for using this drug in endometrial cancer,” commented study discussant Domenica Lorusso, MD, PhD, of Fondazione Policlinico Universitario Agostino Gemelli IRCCS and Catholic University of Sacred Hearth in Rome.
Dr. Lorusso said hormone receptor expression, which has been identified as a strong predictor of CDK4/6 inhibitor activity, is present in up to 90% of patients with type 1 and in about 65% of patients with type 2 endometrial cancer.
Response rates, in experience with aromatase inhibitors, have been “quite disappointing, in the 10%-20% range,” Dr. Lorusso said, with “dismal prognosis” and guidelines stating that “no standard second-line treatment has been identified.”
This research was sponsored by investigators, but Pfizer provided a study grant. Dr. Mirza disclosed relationships with Pfizer, AstraZeneca, Biocad, Clovis Oncology, Genmab, Karyopharm Therapeutics, Merck, Oncology Adventure, Roche, Seattle Genetics, Sera Prognostics, Sotio, GlaxoSmithKline, Zai Lab, and Boehringer Ingelheim. Dr. Lorusso disclosed relationships with AstraZeneca, Biocad, Clovis Oncology, Genmab, Merck, Roche, Tesaro, Amgen, Immunogen, and Pharma Mar.
SOURCE: Mirza MR et al. ESMO 2020, Abstract LBA28.
Adding palbociclib to letrozole significantly prolonged progression-free survival (PFS) in a phase 2 trial of patients with advanced or recurrent estrogen receptor (ER)–positive endometrial cancer.
This was the first randomized trial to evaluate the efficacy of a CDK4/6 inhibitor in combination with an aromatase inhibitor in patients with advanced or recurrent ER-positive endometrial cancer, noted study investigator Mansoor Raza Mirza, MD, PhD, of Rigshospitalet Copenhagen University Hospital.
Dr. Mirza presented results from this study, ENGOT-EN3-NSGO/PALEO, at the European Society for Medical Oncology Virtual Congress 2020.
Palbociclib is a selective inhibitor of CDK4/, both of which are involved in cell-cycle transitions, Dr. Mirza explained. He observed that endometrial endometrioid adenocarcinomas are hormone dependent, and endocrine treatment with an aromatase inhibitor is well established. Palbociclib has been shown to be superior, when combined with letrozole, to letrozole alone in ER-positive breast cancer.
For the ENGOT-EN3-NSGO/PALEO study, investigators enrolled 77 patients with ER-positive advanced/recurrent endometrial cancer. Patients had received at least one prior systemic therapy, no prior endocrine therapy (except medroxyprogesterone acetate and megestrol acetate), and no prior CDK inhibitor.
Patients were randomized 1:1 to receive oral letrozole (2.5 mg on days 1-28) and either palbociclib (125 mg on days 1-21) or placebo (125 mg on days 1-21) in a 28-day cycle until progression. Baseline characteristics were similar between the treatment arms.
Efficacy
Of the 77 patients enrolled, 73 were evaluable. The primary endpoint was PFS.
The median PFS in the intention-to-treat population was 3.0 months in the placebo arm and 8.3 months in the palbociclib arm (hazard ratio, 0.56; 95% confidence interval, 0.32-0.98; P = .0376).
Looking at stratification factors, PFS was higher in the palbociclib arm among the large majority (about 85%) of patients who had received no prior medroxyprogesterone acetate or megestrol acetate (HR, 0.55; 95% CI, 0.29-01.04; P = .0615) and among patients with relapsed disease (HR, 0.61; 95% CI, 0.34-1.09; P = .0916).
The disease control rate at 24 weeks, a secondary endpoint, was 63.6% in the palbociclib arm and 37.8% in the placebo arm.
Safety
Adverse events were more frequent in the palbociclib arm, with neutropenia being the most common.
Rates of adverse event–related dose reduction (to 100 mg or 75 mg) were 36% in the palbociclib arm and 3% in the placebo arm.
Adverse event–related discontinuation rates were 25% and 19% for palbociclib and letrozole, respectively, in the palbociclib arm and 14% and 11% for placebo and letrozole, respectively, in the placebo arm.
“The toxicity of palbociclib plus letrozole combination therapy was manageable, with most patients remaining on treatment until disease progression,” Dr. Mirza said.
He noted that an analysis of patient-reported outcomes revealed no detrimental effect on quality of life with the combination therapy.
Next steps
“Compared with placebo plus letrozole, the combination of palbociclib plus letrozole demonstrated clinically meaningful improvement in PFS,” Dr. Mirza said. “These results merit a phase 3 validation trial.”
“There is a huge rationale for using this drug in endometrial cancer,” commented study discussant Domenica Lorusso, MD, PhD, of Fondazione Policlinico Universitario Agostino Gemelli IRCCS and Catholic University of Sacred Hearth in Rome.
Dr. Lorusso said hormone receptor expression, which has been identified as a strong predictor of CDK4/6 inhibitor activity, is present in up to 90% of patients with type 1 and in about 65% of patients with type 2 endometrial cancer.
Response rates, in experience with aromatase inhibitors, have been “quite disappointing, in the 10%-20% range,” Dr. Lorusso said, with “dismal prognosis” and guidelines stating that “no standard second-line treatment has been identified.”
This research was sponsored by investigators, but Pfizer provided a study grant. Dr. Mirza disclosed relationships with Pfizer, AstraZeneca, Biocad, Clovis Oncology, Genmab, Karyopharm Therapeutics, Merck, Oncology Adventure, Roche, Seattle Genetics, Sera Prognostics, Sotio, GlaxoSmithKline, Zai Lab, and Boehringer Ingelheim. Dr. Lorusso disclosed relationships with AstraZeneca, Biocad, Clovis Oncology, Genmab, Merck, Roche, Tesaro, Amgen, Immunogen, and Pharma Mar.
SOURCE: Mirza MR et al. ESMO 2020, Abstract LBA28.
FROM ESMO 2020
Vascular dementia risk particularly high in type 2 diabetes
Persons with type 2 diabetes may be at heightened risk for developing vascular dementia than other types of dementia, a team of international researchers has found.
Compared with a nondiabetic control population, those with type 2 diabetes had a statistically significant 35% increased chance of having vascular dementia in a large observational study.
By comparison, the risk for nonvascular dementia was increased by a “more modest” 8%, said the researchers from the University of Glasgow and the University of Gothenburg (Sweden), while the risk for Alzheimer’s dementia appeared to be reduced by 8%.
The link between type 2 diabetes and dementia is not new, observed Carlos Celis-Morales, PhD, who presented the study’s findings at the virtual annual meeting of the European Association for the Study of Diabetes. With people living longer thanks to improved preventative strategies and treatments, there is a risk for developing other chronic conditions, such as dementia.
“A third of all dementia cases may be attributable to modifiable risk factors, among them type 2 diabetes, which accounts for 3.2% of all dementia cases,” said Dr. Celis-Morales, a research fellow at the University of Glasgow’s Institute of Cardiovascular and Medical Sciences.
“Although we know that diabetes is linked to dementia, what we don’t know really well is how much of this association between diabetes and dementia outcomes are explained by modifiable and nonmodifiable risk factors,” Dr. Celis-Morales added.
“Diabetes and dementia share certain risk factors,” commented coinvestigator Naveed Sattar, MD, in a press release issued by the EASD. These include obesity, smoking, and lack of physical activity and might explain part of the association between the two conditions.
Dr. Sattar said that the heightened vascular dementia risk found in the study was “in itself an argument for preventive measures such as healthier lifestyle,” adding that “the importance of prevention is underscored by the fact that, for the majority of dementia diseases, there is no good treatment.”
Using data from the Swedish National Diabetes Register, the research team set out to determine the extent to which type 2 diabetes was associated with dementia and the incidence of different subtypes of dementia. They also looked to see if there were any associations with blood glucose control and what risk factors may be involved.
In total, data on 378,299 individuals with type 2 diabetes were compared with data on 1,886,022 similarly aged (average, 64 years) and gender-matched controls from the general population.
After a mean 7 years of follow-up, 10,143 people with and 46,479 people without type 2 diabetes developed dementia. Nonvascular dementia was the most common type of dementia recorded, followed by Alzheimer’s disease and then vascular dementia.
“Within type 2 diabetes individuals, poor glycemic [control] increased the risk of dementia especially for vascular dementia and nonvascular dementia. However, these associations were not as evident for Alzheimer’s disease,” Dr. Celis-Morales reported.
Comparing those with hemoglobin bA1c of less than 52 mmol/mol (7%) with those whose A1c was above 87 mmol/mol (10.1%), there was 93% increase in the risk for vascular dementia, a 67% increase in the risk for nonvascular dementia, and a 34% higher risk for Alzheimer’s disease–associated dementia.
“We have focused on high levels of HbA1c, but what happens if you have really low limits? It’s something we’re working on right now,” Dr. Celis-Morales said.
Importantly, cardiovascular-related risk factors – some of which, like systolic blood pressure and body weight, were potentially modifiable – accounted for more than 40% of the risk for dementia in type 2 diabetes. This suggests that a large percentage of the dementia risk could perhaps be addressed by identifying high-risk individuals and tailoring interventions accordingly.
“These are observational findings, so we need to be careful before we translate to any sort of recommendation,” Dr. Celis-Morales said.
The study was financed by the Swedish state under the agreement between the government and the county councils, the ALF agreement, as well as grant from the Novo Nordisk Foundation and the Swedish Association of Local Authorities and Regions. Dr. Celis-Morales and Dr. Sattar had no conflicts of interest.
SOURCE: Celis-Morales C et al. EASD 2020, Oral presentation 06.
Persons with type 2 diabetes may be at heightened risk for developing vascular dementia than other types of dementia, a team of international researchers has found.
Compared with a nondiabetic control population, those with type 2 diabetes had a statistically significant 35% increased chance of having vascular dementia in a large observational study.
By comparison, the risk for nonvascular dementia was increased by a “more modest” 8%, said the researchers from the University of Glasgow and the University of Gothenburg (Sweden), while the risk for Alzheimer’s dementia appeared to be reduced by 8%.
The link between type 2 diabetes and dementia is not new, observed Carlos Celis-Morales, PhD, who presented the study’s findings at the virtual annual meeting of the European Association for the Study of Diabetes. With people living longer thanks to improved preventative strategies and treatments, there is a risk for developing other chronic conditions, such as dementia.
“A third of all dementia cases may be attributable to modifiable risk factors, among them type 2 diabetes, which accounts for 3.2% of all dementia cases,” said Dr. Celis-Morales, a research fellow at the University of Glasgow’s Institute of Cardiovascular and Medical Sciences.
“Although we know that diabetes is linked to dementia, what we don’t know really well is how much of this association between diabetes and dementia outcomes are explained by modifiable and nonmodifiable risk factors,” Dr. Celis-Morales added.
“Diabetes and dementia share certain risk factors,” commented coinvestigator Naveed Sattar, MD, in a press release issued by the EASD. These include obesity, smoking, and lack of physical activity and might explain part of the association between the two conditions.
Dr. Sattar said that the heightened vascular dementia risk found in the study was “in itself an argument for preventive measures such as healthier lifestyle,” adding that “the importance of prevention is underscored by the fact that, for the majority of dementia diseases, there is no good treatment.”
Using data from the Swedish National Diabetes Register, the research team set out to determine the extent to which type 2 diabetes was associated with dementia and the incidence of different subtypes of dementia. They also looked to see if there were any associations with blood glucose control and what risk factors may be involved.
In total, data on 378,299 individuals with type 2 diabetes were compared with data on 1,886,022 similarly aged (average, 64 years) and gender-matched controls from the general population.
After a mean 7 years of follow-up, 10,143 people with and 46,479 people without type 2 diabetes developed dementia. Nonvascular dementia was the most common type of dementia recorded, followed by Alzheimer’s disease and then vascular dementia.
“Within type 2 diabetes individuals, poor glycemic [control] increased the risk of dementia especially for vascular dementia and nonvascular dementia. However, these associations were not as evident for Alzheimer’s disease,” Dr. Celis-Morales reported.
Comparing those with hemoglobin bA1c of less than 52 mmol/mol (7%) with those whose A1c was above 87 mmol/mol (10.1%), there was 93% increase in the risk for vascular dementia, a 67% increase in the risk for nonvascular dementia, and a 34% higher risk for Alzheimer’s disease–associated dementia.
“We have focused on high levels of HbA1c, but what happens if you have really low limits? It’s something we’re working on right now,” Dr. Celis-Morales said.
Importantly, cardiovascular-related risk factors – some of which, like systolic blood pressure and body weight, were potentially modifiable – accounted for more than 40% of the risk for dementia in type 2 diabetes. This suggests that a large percentage of the dementia risk could perhaps be addressed by identifying high-risk individuals and tailoring interventions accordingly.
“These are observational findings, so we need to be careful before we translate to any sort of recommendation,” Dr. Celis-Morales said.
The study was financed by the Swedish state under the agreement between the government and the county councils, the ALF agreement, as well as grant from the Novo Nordisk Foundation and the Swedish Association of Local Authorities and Regions. Dr. Celis-Morales and Dr. Sattar had no conflicts of interest.
SOURCE: Celis-Morales C et al. EASD 2020, Oral presentation 06.
Persons with type 2 diabetes may be at heightened risk for developing vascular dementia than other types of dementia, a team of international researchers has found.
Compared with a nondiabetic control population, those with type 2 diabetes had a statistically significant 35% increased chance of having vascular dementia in a large observational study.
By comparison, the risk for nonvascular dementia was increased by a “more modest” 8%, said the researchers from the University of Glasgow and the University of Gothenburg (Sweden), while the risk for Alzheimer’s dementia appeared to be reduced by 8%.
The link between type 2 diabetes and dementia is not new, observed Carlos Celis-Morales, PhD, who presented the study’s findings at the virtual annual meeting of the European Association for the Study of Diabetes. With people living longer thanks to improved preventative strategies and treatments, there is a risk for developing other chronic conditions, such as dementia.
“A third of all dementia cases may be attributable to modifiable risk factors, among them type 2 diabetes, which accounts for 3.2% of all dementia cases,” said Dr. Celis-Morales, a research fellow at the University of Glasgow’s Institute of Cardiovascular and Medical Sciences.
“Although we know that diabetes is linked to dementia, what we don’t know really well is how much of this association between diabetes and dementia outcomes are explained by modifiable and nonmodifiable risk factors,” Dr. Celis-Morales added.
“Diabetes and dementia share certain risk factors,” commented coinvestigator Naveed Sattar, MD, in a press release issued by the EASD. These include obesity, smoking, and lack of physical activity and might explain part of the association between the two conditions.
Dr. Sattar said that the heightened vascular dementia risk found in the study was “in itself an argument for preventive measures such as healthier lifestyle,” adding that “the importance of prevention is underscored by the fact that, for the majority of dementia diseases, there is no good treatment.”
Using data from the Swedish National Diabetes Register, the research team set out to determine the extent to which type 2 diabetes was associated with dementia and the incidence of different subtypes of dementia. They also looked to see if there were any associations with blood glucose control and what risk factors may be involved.
In total, data on 378,299 individuals with type 2 diabetes were compared with data on 1,886,022 similarly aged (average, 64 years) and gender-matched controls from the general population.
After a mean 7 years of follow-up, 10,143 people with and 46,479 people without type 2 diabetes developed dementia. Nonvascular dementia was the most common type of dementia recorded, followed by Alzheimer’s disease and then vascular dementia.
“Within type 2 diabetes individuals, poor glycemic [control] increased the risk of dementia especially for vascular dementia and nonvascular dementia. However, these associations were not as evident for Alzheimer’s disease,” Dr. Celis-Morales reported.
Comparing those with hemoglobin bA1c of less than 52 mmol/mol (7%) with those whose A1c was above 87 mmol/mol (10.1%), there was 93% increase in the risk for vascular dementia, a 67% increase in the risk for nonvascular dementia, and a 34% higher risk for Alzheimer’s disease–associated dementia.
“We have focused on high levels of HbA1c, but what happens if you have really low limits? It’s something we’re working on right now,” Dr. Celis-Morales said.
Importantly, cardiovascular-related risk factors – some of which, like systolic blood pressure and body weight, were potentially modifiable – accounted for more than 40% of the risk for dementia in type 2 diabetes. This suggests that a large percentage of the dementia risk could perhaps be addressed by identifying high-risk individuals and tailoring interventions accordingly.
“These are observational findings, so we need to be careful before we translate to any sort of recommendation,” Dr. Celis-Morales said.
The study was financed by the Swedish state under the agreement between the government and the county councils, the ALF agreement, as well as grant from the Novo Nordisk Foundation and the Swedish Association of Local Authorities and Regions. Dr. Celis-Morales and Dr. Sattar had no conflicts of interest.
SOURCE: Celis-Morales C et al. EASD 2020, Oral presentation 06.
FROM EASD 2020
Two new protein biomarkers may serve as prognostic indicators for outcomes in CLL
Two new protein biomarkers may serve as prognostic indicators for outcomes in chronic lymphocytic leukemia (CLL) patients, according to the results of a proteomic assessment of patients’ serum compared to their event-free survival (EFS).
The results were published in Experimental Hematology.
The study attempted to validate the prognostic ability of known proteomic markers measured pretreatment and to search for new proteomic markers that might be related to treatment response in CLL, according to Fatemeh Saberi Hosnijeh, MD, of Erasmus MC, University Medical Center, Rotterdam, The Netherlands, and colleagues.
Baseline serum samples were taken from 51 CLL patients who were then treated with chemoimmunotherapy. The samples were analyzed for 360 proteomic markers, and those results were compared with patient EFS.
Study subjects were selected from patients enrolled in the HOVON 109 clinical trial, a phase 1/2 trial designed to assess the efficacy and safety of first-line therapy involving chlorambucil, rituximab,and lenalidomide in elderly patients and young frail patients with advanced CLL.
The patients assessed comprised 30 men and 21 women, and the median EFS for all patients was 23 months (ranging from 1.25 to 60.9 months).
Promising biomarkers
The researchers found that patients who had high serum levels of the proteins sCD23 (P = .026), sCD27 (P = .04), the serine peptidase inhibitor SPINT1 (P = .001), and the surface antigen protein LY9 (P = .0003) had a shorter EFS than those with marker levels below the median.
“Taken together, our results validate the prognostic impact of sCD23 and highlight SPINT1 and LY9 as possible promising markers for treatment response in CLL patients,” the researchers stated.
“Despite the relatively small number of available cases, which had an impact on statistical power, our pilot study identified SPINT1 and LY9 as promising independent prognostic proteomic markers next to sCD23 and sCD27 in patients treated for CLL. Further studies with larger sample sizes are required to validate these results,” the researchers concluded.
This research was supported by a grant from Gilead Sciences and an EU TRANSCAN/Dutch Cancer Society grant. The authors declared that they had no conflicts of interest.
SOURCE: Hosnijeh FS et al. Exp Hematol. 2020;89:55-60.
Two new protein biomarkers may serve as prognostic indicators for outcomes in chronic lymphocytic leukemia (CLL) patients, according to the results of a proteomic assessment of patients’ serum compared to their event-free survival (EFS).
The results were published in Experimental Hematology.
The study attempted to validate the prognostic ability of known proteomic markers measured pretreatment and to search for new proteomic markers that might be related to treatment response in CLL, according to Fatemeh Saberi Hosnijeh, MD, of Erasmus MC, University Medical Center, Rotterdam, The Netherlands, and colleagues.
Baseline serum samples were taken from 51 CLL patients who were then treated with chemoimmunotherapy. The samples were analyzed for 360 proteomic markers, and those results were compared with patient EFS.
Study subjects were selected from patients enrolled in the HOVON 109 clinical trial, a phase 1/2 trial designed to assess the efficacy and safety of first-line therapy involving chlorambucil, rituximab,and lenalidomide in elderly patients and young frail patients with advanced CLL.
The patients assessed comprised 30 men and 21 women, and the median EFS for all patients was 23 months (ranging from 1.25 to 60.9 months).
Promising biomarkers
The researchers found that patients who had high serum levels of the proteins sCD23 (P = .026), sCD27 (P = .04), the serine peptidase inhibitor SPINT1 (P = .001), and the surface antigen protein LY9 (P = .0003) had a shorter EFS than those with marker levels below the median.
“Taken together, our results validate the prognostic impact of sCD23 and highlight SPINT1 and LY9 as possible promising markers for treatment response in CLL patients,” the researchers stated.
“Despite the relatively small number of available cases, which had an impact on statistical power, our pilot study identified SPINT1 and LY9 as promising independent prognostic proteomic markers next to sCD23 and sCD27 in patients treated for CLL. Further studies with larger sample sizes are required to validate these results,” the researchers concluded.
This research was supported by a grant from Gilead Sciences and an EU TRANSCAN/Dutch Cancer Society grant. The authors declared that they had no conflicts of interest.
SOURCE: Hosnijeh FS et al. Exp Hematol. 2020;89:55-60.
Two new protein biomarkers may serve as prognostic indicators for outcomes in chronic lymphocytic leukemia (CLL) patients, according to the results of a proteomic assessment of patients’ serum compared to their event-free survival (EFS).
The results were published in Experimental Hematology.
The study attempted to validate the prognostic ability of known proteomic markers measured pretreatment and to search for new proteomic markers that might be related to treatment response in CLL, according to Fatemeh Saberi Hosnijeh, MD, of Erasmus MC, University Medical Center, Rotterdam, The Netherlands, and colleagues.
Baseline serum samples were taken from 51 CLL patients who were then treated with chemoimmunotherapy. The samples were analyzed for 360 proteomic markers, and those results were compared with patient EFS.
Study subjects were selected from patients enrolled in the HOVON 109 clinical trial, a phase 1/2 trial designed to assess the efficacy and safety of first-line therapy involving chlorambucil, rituximab,and lenalidomide in elderly patients and young frail patients with advanced CLL.
The patients assessed comprised 30 men and 21 women, and the median EFS for all patients was 23 months (ranging from 1.25 to 60.9 months).
Promising biomarkers
The researchers found that patients who had high serum levels of the proteins sCD23 (P = .026), sCD27 (P = .04), the serine peptidase inhibitor SPINT1 (P = .001), and the surface antigen protein LY9 (P = .0003) had a shorter EFS than those with marker levels below the median.
“Taken together, our results validate the prognostic impact of sCD23 and highlight SPINT1 and LY9 as possible promising markers for treatment response in CLL patients,” the researchers stated.
“Despite the relatively small number of available cases, which had an impact on statistical power, our pilot study identified SPINT1 and LY9 as promising independent prognostic proteomic markers next to sCD23 and sCD27 in patients treated for CLL. Further studies with larger sample sizes are required to validate these results,” the researchers concluded.
This research was supported by a grant from Gilead Sciences and an EU TRANSCAN/Dutch Cancer Society grant. The authors declared that they had no conflicts of interest.
SOURCE: Hosnijeh FS et al. Exp Hematol. 2020;89:55-60.
FROM EXPERIMENTAL HEMATOLOGY
Survival after kidney transplantation lags in diabetes patients
Survival of U.S. patients who received a kidney transplant improved during 2000-2018, but the extent of improvement among patients whose end-stage kidney disease linked with diabetes lagged behind patients with renal disease unrelated to diabetes, based on a review of more than 250,000 U.S. renal transplant recipients from that period.
After adjustment for several demographic and clinical baseline differences, as well as for several characteristics of the organ donor, the analysis showed that patients with type 2 diabetes (T2D) had a significant 64% higher mortality rate following kidney transplant compared with patients without diabetes, while patients with type 1 diabetes (T1D) had a significant 94% increased relative rate of death, Jessica Harding, PhD, said at the virtual annual meeting of the European Association for the Study of Diabetes.
The analyses that Dr. Harding reported also showed that, throughout the period examined, mortality rates following kidney transplant remained several times greater than the death rate of similar Americans who did not undergo renal replacement. By 2017, the standardized mortality ratio for patients with T2D following a kidney transplant was roughly fourfold greater than in similarly aged Americans in the general population who did undergo a transplant, while for patients with T1D the standardized mortality ratio compared with the general population was about sevenfold higher.
“Important disparities” for survival following kidney transplantation based on a specific diabetes etiology exist among U.S. patients, and further research should examine ways to better reduce posttransplant mortality in patients with diabetes, especially those with T1D, concluded Dr. Harding, an epidemiologist in the division of transplantation, department of surgery, at Emory University, Atlanta.
Issues surrounding kidney transplantation and postsurgical survival among patients with diabetes are important because these patients remain very susceptible to developing end-stage kidney disease and need for renal replacement. Adequate management of hyperglycemia, hypertension, and the adverse cardiovascular effects of immunosuppressive drugs might provide effective strategies for further mortality reductions among patients with diabetes following kidney transplant, she suggested.
The study used data collected in the United States Renal Data System during January 2000–August 2018, and included 258,188 adults who underwent a first-time, single kidney transplant at a U.S. center. About 20,000 patients had T1D (8%), about 59,000 (23%) had T2D, and the remaining 69% had no diabetes diagnosis. The data allowed for survival monitoring during a median follow-up of just over 6 years, during which more than 72,000 of the tracked patients (28%) died. The Renal Data System entries for 2017 also showed that 47% of U.S. patients with new end-stage renal disease had a diabetes etiology, Dr. Harding said.
The study received no commercial funding. Dr. Harding had no disclosures.
SOURCE: Harding J. EASD 2020. Oral presentation 66.
Survival of U.S. patients who received a kidney transplant improved during 2000-2018, but the extent of improvement among patients whose end-stage kidney disease linked with diabetes lagged behind patients with renal disease unrelated to diabetes, based on a review of more than 250,000 U.S. renal transplant recipients from that period.
After adjustment for several demographic and clinical baseline differences, as well as for several characteristics of the organ donor, the analysis showed that patients with type 2 diabetes (T2D) had a significant 64% higher mortality rate following kidney transplant compared with patients without diabetes, while patients with type 1 diabetes (T1D) had a significant 94% increased relative rate of death, Jessica Harding, PhD, said at the virtual annual meeting of the European Association for the Study of Diabetes.
The analyses that Dr. Harding reported also showed that, throughout the period examined, mortality rates following kidney transplant remained several times greater than the death rate of similar Americans who did not undergo renal replacement. By 2017, the standardized mortality ratio for patients with T2D following a kidney transplant was roughly fourfold greater than in similarly aged Americans in the general population who did undergo a transplant, while for patients with T1D the standardized mortality ratio compared with the general population was about sevenfold higher.
“Important disparities” for survival following kidney transplantation based on a specific diabetes etiology exist among U.S. patients, and further research should examine ways to better reduce posttransplant mortality in patients with diabetes, especially those with T1D, concluded Dr. Harding, an epidemiologist in the division of transplantation, department of surgery, at Emory University, Atlanta.
Issues surrounding kidney transplantation and postsurgical survival among patients with diabetes are important because these patients remain very susceptible to developing end-stage kidney disease and need for renal replacement. Adequate management of hyperglycemia, hypertension, and the adverse cardiovascular effects of immunosuppressive drugs might provide effective strategies for further mortality reductions among patients with diabetes following kidney transplant, she suggested.
The study used data collected in the United States Renal Data System during January 2000–August 2018, and included 258,188 adults who underwent a first-time, single kidney transplant at a U.S. center. About 20,000 patients had T1D (8%), about 59,000 (23%) had T2D, and the remaining 69% had no diabetes diagnosis. The data allowed for survival monitoring during a median follow-up of just over 6 years, during which more than 72,000 of the tracked patients (28%) died. The Renal Data System entries for 2017 also showed that 47% of U.S. patients with new end-stage renal disease had a diabetes etiology, Dr. Harding said.
The study received no commercial funding. Dr. Harding had no disclosures.
SOURCE: Harding J. EASD 2020. Oral presentation 66.
Survival of U.S. patients who received a kidney transplant improved during 2000-2018, but the extent of improvement among patients whose end-stage kidney disease linked with diabetes lagged behind patients with renal disease unrelated to diabetes, based on a review of more than 250,000 U.S. renal transplant recipients from that period.
After adjustment for several demographic and clinical baseline differences, as well as for several characteristics of the organ donor, the analysis showed that patients with type 2 diabetes (T2D) had a significant 64% higher mortality rate following kidney transplant compared with patients without diabetes, while patients with type 1 diabetes (T1D) had a significant 94% increased relative rate of death, Jessica Harding, PhD, said at the virtual annual meeting of the European Association for the Study of Diabetes.
The analyses that Dr. Harding reported also showed that, throughout the period examined, mortality rates following kidney transplant remained several times greater than the death rate of similar Americans who did not undergo renal replacement. By 2017, the standardized mortality ratio for patients with T2D following a kidney transplant was roughly fourfold greater than in similarly aged Americans in the general population who did undergo a transplant, while for patients with T1D the standardized mortality ratio compared with the general population was about sevenfold higher.
“Important disparities” for survival following kidney transplantation based on a specific diabetes etiology exist among U.S. patients, and further research should examine ways to better reduce posttransplant mortality in patients with diabetes, especially those with T1D, concluded Dr. Harding, an epidemiologist in the division of transplantation, department of surgery, at Emory University, Atlanta.
Issues surrounding kidney transplantation and postsurgical survival among patients with diabetes are important because these patients remain very susceptible to developing end-stage kidney disease and need for renal replacement. Adequate management of hyperglycemia, hypertension, and the adverse cardiovascular effects of immunosuppressive drugs might provide effective strategies for further mortality reductions among patients with diabetes following kidney transplant, she suggested.
The study used data collected in the United States Renal Data System during January 2000–August 2018, and included 258,188 adults who underwent a first-time, single kidney transplant at a U.S. center. About 20,000 patients had T1D (8%), about 59,000 (23%) had T2D, and the remaining 69% had no diabetes diagnosis. The data allowed for survival monitoring during a median follow-up of just over 6 years, during which more than 72,000 of the tracked patients (28%) died. The Renal Data System entries for 2017 also showed that 47% of U.S. patients with new end-stage renal disease had a diabetes etiology, Dr. Harding said.
The study received no commercial funding. Dr. Harding had no disclosures.
SOURCE: Harding J. EASD 2020. Oral presentation 66.
FROM EASD 2020
Children’s share of COVID-19 burden continues to increase
Children continue to represent an increasing proportion of reported COVID-19 cases in the United States, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
The previous week, children represented 10.0% of all cases, and that proportion has continued to rise throughout the pandemic, the AAP and CHA report shows.
Looking at just new cases for the latest week, the 38,000+ pediatric cases made up almost 17% of the 228,396 cases reported for all ages, compared with 16% and 15% the two previous weeks. For the weeks ending Aug. 13 and Aug. 6, the corresponding figures were 8% and 13%, based on the data in the AAP/CHA report, which cover 49 states (New York City but not New York state), the District of Columbia, Puerto Rico, and Guam.
The state with the highest proportion of child COVID-19 cases as of Sept. 17 was Wyoming, with 20.6%, followed by North Dakota at 18.3% and Tennessee at 17.9%. New York City has a cumulative rate of just 3.4%, but New Jersey is the state with the lowest rate at 3.6%. Florida comes in at 5.9% but is using an age range of 0-14 years for children, and Texas has a rate of 6.0% but has reported ages for only 8% of confirmed cases, the AAP and CHA noted.
Severe illness, however, continues to be rare in children. The overall hospitalization rate for children was down to 1.7% among the 26 jurisdictions providing ages as Sept. 17 – down from 1.8% the week before and 2.3% on Aug. 20. The death rate is just 0.02% among 43 jurisdictions, the report said.
Children continue to represent an increasing proportion of reported COVID-19 cases in the United States, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
The previous week, children represented 10.0% of all cases, and that proportion has continued to rise throughout the pandemic, the AAP and CHA report shows.
Looking at just new cases for the latest week, the 38,000+ pediatric cases made up almost 17% of the 228,396 cases reported for all ages, compared with 16% and 15% the two previous weeks. For the weeks ending Aug. 13 and Aug. 6, the corresponding figures were 8% and 13%, based on the data in the AAP/CHA report, which cover 49 states (New York City but not New York state), the District of Columbia, Puerto Rico, and Guam.
The state with the highest proportion of child COVID-19 cases as of Sept. 17 was Wyoming, with 20.6%, followed by North Dakota at 18.3% and Tennessee at 17.9%. New York City has a cumulative rate of just 3.4%, but New Jersey is the state with the lowest rate at 3.6%. Florida comes in at 5.9% but is using an age range of 0-14 years for children, and Texas has a rate of 6.0% but has reported ages for only 8% of confirmed cases, the AAP and CHA noted.
Severe illness, however, continues to be rare in children. The overall hospitalization rate for children was down to 1.7% among the 26 jurisdictions providing ages as Sept. 17 – down from 1.8% the week before and 2.3% on Aug. 20. The death rate is just 0.02% among 43 jurisdictions, the report said.
Children continue to represent an increasing proportion of reported COVID-19 cases in the United States, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
The previous week, children represented 10.0% of all cases, and that proportion has continued to rise throughout the pandemic, the AAP and CHA report shows.
Looking at just new cases for the latest week, the 38,000+ pediatric cases made up almost 17% of the 228,396 cases reported for all ages, compared with 16% and 15% the two previous weeks. For the weeks ending Aug. 13 and Aug. 6, the corresponding figures were 8% and 13%, based on the data in the AAP/CHA report, which cover 49 states (New York City but not New York state), the District of Columbia, Puerto Rico, and Guam.
The state with the highest proportion of child COVID-19 cases as of Sept. 17 was Wyoming, with 20.6%, followed by North Dakota at 18.3% and Tennessee at 17.9%. New York City has a cumulative rate of just 3.4%, but New Jersey is the state with the lowest rate at 3.6%. Florida comes in at 5.9% but is using an age range of 0-14 years for children, and Texas has a rate of 6.0% but has reported ages for only 8% of confirmed cases, the AAP and CHA noted.
Severe illness, however, continues to be rare in children. The overall hospitalization rate for children was down to 1.7% among the 26 jurisdictions providing ages as Sept. 17 – down from 1.8% the week before and 2.3% on Aug. 20. The death rate is just 0.02% among 43 jurisdictions, the report said.
Breathing enriched oxygen improves major depression
Maybe the hipsters patronizing trendy oxygen bars seeking elevation of mood back in the prepandemic era were actually onto something – because Israeli investigators have now shown in a pilot double-blind, placebo-controlled, randomized trial that breathing enriched oxygen on a nightly basis resulted in clinically meaningful symptomatic improvement in mild to moderate major depression.
“We saw a highly significant effect of normobaric hyperoxia therapy in lowering Hamilton Rating Scale for Depression scores,” R. Haim Belmaker, MD, reported at the virtual congress of the European College of Neuropsychopharmacology.
In addition, the patients on enriched oxygen also showed statistically significant and clinically meaningful improvements relative to sham-treated controls on the secondary endpoints of Clinical Global Impressions Scale, the World Health Organization–Five Well-Being Index, the Sheehan Disability Scale, and the Sense of Coherence Scale, added Dr. Belmaker, professor emeritus of psychiatry at the Ben Gurion University of the Negev in Beersheva, Israel.
Numerous PET imaging studies have documented diminished brain mitochondrial function in patients with depression or schizophrenia. And mitochondria need oxygen to do their work. Yet, the idea of administering enriched oxygen in an effort to boost mitochondrial energy metabolism has long been viewed with skepticism – even though it’s a simple and well-tolerated intervention – because of the fact that 90%-95% of the oxygen supply is carried bound to hemoglobin, and oxygen enrichment doesn’t further increase hemoglobin saturation in individuals with normal lung function. However, recently it has been shown that inspired enriched oxygen roughly doubles arterial oxygen tension, and while this doesn’t translate into anything close to a doubled oxygen supply to tissues, it may result in increased oxygen diffusion into brain tissue, the psychiatrist explained.
Normobaric hyperoxia therapy is not to be confused with hyperbaric oxygen therapy, which requires a special chamber to handle markedly increased atmospheric pressures and has some inherent dangers. Mobile bedside oxygen generator units for oxygen enrichment are commercially available over the counter. Those used in the Israeli study were about the size of a vacuum cleaner and weighed a little more than 40 lb. Much smaller, more convenient units are available as well, but are costlier.
Dr. Belmaker reported on 51 adults with mild or moderate symptoms of major depressive disorder and a mean 11-year disease history who were randomized double blind to breathe either 35% oxygen or normal air – that is, 21% oxygen – at 1 atm pressure delivered from an investigator-supplied oxygen generator through standard plastic nasal prongs at a flow rate of 5 L/min for 7 hours nightly for 1 month.
“Controls heard the same flow and felt the same feeling on the face but were receiving 21% oxygen,” he noted.
Oxygen generator units are capable of enriching air to more than 90% oxygen; however, the investigators wanted to be cautious in a pilot study of an untested therapy, and they found evidence from both animal and human studies that 40% oxygen is reassuringly safe. Study exclusion criteria included obesity, acute or chronic respiratory disease, psychosis, and suicidality.
The primary study endpoint was the change in Hamilton Rating Scale for Depression score at 1 month. From a mean baseline of 14.6, the score in the normobaric hyperoxia group dropped by more than 4 points while remaining unchanged in controls. In a subscale analysis, it was apparent that most of the improvement occurred in the anxiety and cognitive disturbance subscale domains, according to Dr. Belmaker.
Of note, all patients rated by blinded investigators as much improved or very much improved on the Clinical Global Impression scale came from the enriched oxygen group.
No treatment-related adverse events occurred in the study.
“We don’t know the mechanism of the benefit of oxygen on the brain. It’s complex. In stroke and acute MI we used to think oxygen was beneficial, but scientists now feel that it’s not,” the psychiatrist said. “This early data deserve replication with higher concentrations of oxygen, different time periods of application, and in different patient groups.”
He emphasized that, since individuals with physical illnesses – including sleep apnea and chronic obstructive pulmonary disease – were excluded from the study, it’s not possible to say whether normobaric hyperoxia therapy would have an antidepressant effect in such patients.
“I would be especially careful with the normobaric oxygen in any patients with any cardiovascular or hypertensive disease because the increased oxygen pressure can have the side effect of contracting cardiac capillaries as a reflex action. So I certainly cannot recommend applying this study in any patients with a physical disease at this point,” Dr. Belmaker emphasized.
He reported having no financial conflicts regarding the study, funded by a grant from the Brain and Behavior Research Foundation.
SOURCE: Belmaker RH. ECNP 2020, Session S.12.
Maybe the hipsters patronizing trendy oxygen bars seeking elevation of mood back in the prepandemic era were actually onto something – because Israeli investigators have now shown in a pilot double-blind, placebo-controlled, randomized trial that breathing enriched oxygen on a nightly basis resulted in clinically meaningful symptomatic improvement in mild to moderate major depression.
“We saw a highly significant effect of normobaric hyperoxia therapy in lowering Hamilton Rating Scale for Depression scores,” R. Haim Belmaker, MD, reported at the virtual congress of the European College of Neuropsychopharmacology.
In addition, the patients on enriched oxygen also showed statistically significant and clinically meaningful improvements relative to sham-treated controls on the secondary endpoints of Clinical Global Impressions Scale, the World Health Organization–Five Well-Being Index, the Sheehan Disability Scale, and the Sense of Coherence Scale, added Dr. Belmaker, professor emeritus of psychiatry at the Ben Gurion University of the Negev in Beersheva, Israel.
Numerous PET imaging studies have documented diminished brain mitochondrial function in patients with depression or schizophrenia. And mitochondria need oxygen to do their work. Yet, the idea of administering enriched oxygen in an effort to boost mitochondrial energy metabolism has long been viewed with skepticism – even though it’s a simple and well-tolerated intervention – because of the fact that 90%-95% of the oxygen supply is carried bound to hemoglobin, and oxygen enrichment doesn’t further increase hemoglobin saturation in individuals with normal lung function. However, recently it has been shown that inspired enriched oxygen roughly doubles arterial oxygen tension, and while this doesn’t translate into anything close to a doubled oxygen supply to tissues, it may result in increased oxygen diffusion into brain tissue, the psychiatrist explained.
Normobaric hyperoxia therapy is not to be confused with hyperbaric oxygen therapy, which requires a special chamber to handle markedly increased atmospheric pressures and has some inherent dangers. Mobile bedside oxygen generator units for oxygen enrichment are commercially available over the counter. Those used in the Israeli study were about the size of a vacuum cleaner and weighed a little more than 40 lb. Much smaller, more convenient units are available as well, but are costlier.
Dr. Belmaker reported on 51 adults with mild or moderate symptoms of major depressive disorder and a mean 11-year disease history who were randomized double blind to breathe either 35% oxygen or normal air – that is, 21% oxygen – at 1 atm pressure delivered from an investigator-supplied oxygen generator through standard plastic nasal prongs at a flow rate of 5 L/min for 7 hours nightly for 1 month.
“Controls heard the same flow and felt the same feeling on the face but were receiving 21% oxygen,” he noted.
Oxygen generator units are capable of enriching air to more than 90% oxygen; however, the investigators wanted to be cautious in a pilot study of an untested therapy, and they found evidence from both animal and human studies that 40% oxygen is reassuringly safe. Study exclusion criteria included obesity, acute or chronic respiratory disease, psychosis, and suicidality.
The primary study endpoint was the change in Hamilton Rating Scale for Depression score at 1 month. From a mean baseline of 14.6, the score in the normobaric hyperoxia group dropped by more than 4 points while remaining unchanged in controls. In a subscale analysis, it was apparent that most of the improvement occurred in the anxiety and cognitive disturbance subscale domains, according to Dr. Belmaker.
Of note, all patients rated by blinded investigators as much improved or very much improved on the Clinical Global Impression scale came from the enriched oxygen group.
No treatment-related adverse events occurred in the study.
“We don’t know the mechanism of the benefit of oxygen on the brain. It’s complex. In stroke and acute MI we used to think oxygen was beneficial, but scientists now feel that it’s not,” the psychiatrist said. “This early data deserve replication with higher concentrations of oxygen, different time periods of application, and in different patient groups.”
He emphasized that, since individuals with physical illnesses – including sleep apnea and chronic obstructive pulmonary disease – were excluded from the study, it’s not possible to say whether normobaric hyperoxia therapy would have an antidepressant effect in such patients.
“I would be especially careful with the normobaric oxygen in any patients with any cardiovascular or hypertensive disease because the increased oxygen pressure can have the side effect of contracting cardiac capillaries as a reflex action. So I certainly cannot recommend applying this study in any patients with a physical disease at this point,” Dr. Belmaker emphasized.
He reported having no financial conflicts regarding the study, funded by a grant from the Brain and Behavior Research Foundation.
SOURCE: Belmaker RH. ECNP 2020, Session S.12.
Maybe the hipsters patronizing trendy oxygen bars seeking elevation of mood back in the prepandemic era were actually onto something – because Israeli investigators have now shown in a pilot double-blind, placebo-controlled, randomized trial that breathing enriched oxygen on a nightly basis resulted in clinically meaningful symptomatic improvement in mild to moderate major depression.
“We saw a highly significant effect of normobaric hyperoxia therapy in lowering Hamilton Rating Scale for Depression scores,” R. Haim Belmaker, MD, reported at the virtual congress of the European College of Neuropsychopharmacology.
In addition, the patients on enriched oxygen also showed statistically significant and clinically meaningful improvements relative to sham-treated controls on the secondary endpoints of Clinical Global Impressions Scale, the World Health Organization–Five Well-Being Index, the Sheehan Disability Scale, and the Sense of Coherence Scale, added Dr. Belmaker, professor emeritus of psychiatry at the Ben Gurion University of the Negev in Beersheva, Israel.
Numerous PET imaging studies have documented diminished brain mitochondrial function in patients with depression or schizophrenia. And mitochondria need oxygen to do their work. Yet, the idea of administering enriched oxygen in an effort to boost mitochondrial energy metabolism has long been viewed with skepticism – even though it’s a simple and well-tolerated intervention – because of the fact that 90%-95% of the oxygen supply is carried bound to hemoglobin, and oxygen enrichment doesn’t further increase hemoglobin saturation in individuals with normal lung function. However, recently it has been shown that inspired enriched oxygen roughly doubles arterial oxygen tension, and while this doesn’t translate into anything close to a doubled oxygen supply to tissues, it may result in increased oxygen diffusion into brain tissue, the psychiatrist explained.
Normobaric hyperoxia therapy is not to be confused with hyperbaric oxygen therapy, which requires a special chamber to handle markedly increased atmospheric pressures and has some inherent dangers. Mobile bedside oxygen generator units for oxygen enrichment are commercially available over the counter. Those used in the Israeli study were about the size of a vacuum cleaner and weighed a little more than 40 lb. Much smaller, more convenient units are available as well, but are costlier.
Dr. Belmaker reported on 51 adults with mild or moderate symptoms of major depressive disorder and a mean 11-year disease history who were randomized double blind to breathe either 35% oxygen or normal air – that is, 21% oxygen – at 1 atm pressure delivered from an investigator-supplied oxygen generator through standard plastic nasal prongs at a flow rate of 5 L/min for 7 hours nightly for 1 month.
“Controls heard the same flow and felt the same feeling on the face but were receiving 21% oxygen,” he noted.
Oxygen generator units are capable of enriching air to more than 90% oxygen; however, the investigators wanted to be cautious in a pilot study of an untested therapy, and they found evidence from both animal and human studies that 40% oxygen is reassuringly safe. Study exclusion criteria included obesity, acute or chronic respiratory disease, psychosis, and suicidality.
The primary study endpoint was the change in Hamilton Rating Scale for Depression score at 1 month. From a mean baseline of 14.6, the score in the normobaric hyperoxia group dropped by more than 4 points while remaining unchanged in controls. In a subscale analysis, it was apparent that most of the improvement occurred in the anxiety and cognitive disturbance subscale domains, according to Dr. Belmaker.
Of note, all patients rated by blinded investigators as much improved or very much improved on the Clinical Global Impression scale came from the enriched oxygen group.
No treatment-related adverse events occurred in the study.
“We don’t know the mechanism of the benefit of oxygen on the brain. It’s complex. In stroke and acute MI we used to think oxygen was beneficial, but scientists now feel that it’s not,” the psychiatrist said. “This early data deserve replication with higher concentrations of oxygen, different time periods of application, and in different patient groups.”
He emphasized that, since individuals with physical illnesses – including sleep apnea and chronic obstructive pulmonary disease – were excluded from the study, it’s not possible to say whether normobaric hyperoxia therapy would have an antidepressant effect in such patients.
“I would be especially careful with the normobaric oxygen in any patients with any cardiovascular or hypertensive disease because the increased oxygen pressure can have the side effect of contracting cardiac capillaries as a reflex action. So I certainly cannot recommend applying this study in any patients with a physical disease at this point,” Dr. Belmaker emphasized.
He reported having no financial conflicts regarding the study, funded by a grant from the Brain and Behavior Research Foundation.
SOURCE: Belmaker RH. ECNP 2020, Session S.12.
FROM ECNP 2020
Dr. Len Calabrese gives advice on vaccinating adult patients with rheumatic disease
When it comes to preventing infection in rheumatology patients, “vaccination is the best mode of infection protection” and works synergistically with masks and hand washing, according to Leonard H. Calabrese, DO.
“Patients with rheumatic diseases have increased morbidity and mortality [from infection] and a lot of risk factors, including age, comorbidities, cytopenias, and extra-articular disease immunosuppression,” he said in a virtual presentation at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
Unfortunately, vaccination uptake remains “much lower than we would like in this country,” he said. Notably, influenza vaccination remains well below the World Health Organization target of 75%, he said.
Influenza vaccination
Flu vaccination will be even more important this year in the context of the COVID-19 pandemic, said Dr. Calabrese, professor of medicine and the RJ Fasenmyer Chair of Clinical Immunology at the Cleveland Clinic in Ohio. “For everyone who comes in with a respiratory illness, we will have to figure out whether it is flu or COVID,” he emphasized.
The Centers for Disease Control and Prevention recommendations include a detailed special considerations section for patients with immunocompromising conditions; “the notes have everything you need to know” about advising rheumatology patients, most of whom can safely receive a flu vaccine, he said.
One concern that always comes up is whether an antibody response will be suppressed based on therapy, Dr. Calabrese noted. Two major drugs with the greatest ability to reduce response are methotrexate and rituximab, he said. His tip: “Withhold methotrexate for two doses following seasonal flu vaccination.” This advice stems from a series of “practice-changing” studies by Park et al. published in 2017, 2018, and 2019 that showed benefit in withholding methotrexate for two doses following vaccination.
In the past, high-dose trivalent flu vaccines have been more expensive, and not necessarily practice changing, with studies showing varying clinical effectiveness and cost-effectiveness, Dr. Calabrese said. This year, a high-dose quadrivalent vaccine should be available that showed a 24% improvement in protection from all strains of influenza, compared with the standard vaccine in a head-to-head, randomized, controlled trial, he noted.
“All patients in rheumatology practices should get a flu vaccine,” with a 2-week hold on methotrexate following vaccination, he advised, and those aged 65 years and older should receive the high-dose quadrivalent. Younger patients on immunosuppressive therapy also might be considered for the high-dose vaccine, he said.
Pneumococcal vaccination
Dr. Calabrese also emphasized the value of pneumococcal vaccines for rheumatology patients. “The mortality for invasive disease ranges from 5% to 32%, but patients with immunocompromising conditions are at increased risk.”
Dr. Calabrese added a note on safety: Patients with cryopyrin-associated periodic syndrome (CAPS), a rare hereditary inflammatory disorder with cutaneous, neurologic, ophthalmologic, and rheumatologic manifestations, may have severe local and systemic reactions to the 23-valent polysaccharide vaccine (PPSV23), he said.
However, immunization against pneumococcal disease is safe and effective for most patients with autoimmune and inflammatory disorders regardless of their current therapy, he said. As with influenza, the CDC’s vaccination recommendations provide details for special situations, including immunocompromised individuals, he noted.
Dr. Calabrese recommended the 13-valent pneumococcal conjugate vaccine (PCV13) as soon as possible for rheumatology patients who have never been vaccinated, with follow-up doses of the 23-valent polysaccharide vaccine (PPSV23) at least 8 weeks later, and a PPSV23 booster 5 years after the first PPSV23 dose.
Protecting against shingles
When it comes to managing the varicella zoster virus (VZV) in immunocompromised patients, “prevention is preferable to treatment, as our patients are particularly vulnerable because of age and declining immunity,” Dr. Calabrese said.
Prevention is important because “once herpes zoster develops, the available treatments, including antiviral therapy, do not prevent postherpetic neuralgia in all patients,” he emphasized. “The treatments are complicated and not always effective,” he added.
The complications of zoster are well known, but recent data show an increased risk of cardiovascular disease as well, Dr. Calabrese said. “All the more reason to protect rheumatology patients from incident zoster,” he said.
Currently, the nonlive recombinant subunit zoster vaccine (Shingrix) is the preferred option for VZV vaccination according to the CDC’s Advisory Committee on Immunization Practices, Dr. Calabrese said. The CDC initially recommended its use to prevent herpes zoster and related complications in all immunocompetent adults aged 50 years and older; in an update, a C-level recommendation extends to “all patients aged 50 with or without immunosuppressive illnesses regardless of previous Zostavax exposure,” Dr. Calabrese said. “All patients on or starting [Janus] kinase inhibitors, regardless of age, should be considered” to receive the herpes zoster vaccine, he noted.
In general, promoting vaccination for rheumatology patients and for all patients is a multipronged effort that might include reminders, rewards, education, and standing orders, Dr. Calabrese said. Clinicians must continue to educate patients not only by strongly recommending the appropriate vaccines, but dispelling myths about vaccination, addressing fears, and providing current and accurate information, he said.
Dr. Calabrese disclosed relationships with AbbVie, Bristol-Myers Squibb, Crescendo, Genentech, Gilead, GlaxoSmithKline, Janssen, Novartis, Pfizer, Sanofi-Regeneron, and UCB.
Global Academy for Medical Education and this news organization are owned by the same parent company.
When it comes to preventing infection in rheumatology patients, “vaccination is the best mode of infection protection” and works synergistically with masks and hand washing, according to Leonard H. Calabrese, DO.
“Patients with rheumatic diseases have increased morbidity and mortality [from infection] and a lot of risk factors, including age, comorbidities, cytopenias, and extra-articular disease immunosuppression,” he said in a virtual presentation at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
Unfortunately, vaccination uptake remains “much lower than we would like in this country,” he said. Notably, influenza vaccination remains well below the World Health Organization target of 75%, he said.
Influenza vaccination
Flu vaccination will be even more important this year in the context of the COVID-19 pandemic, said Dr. Calabrese, professor of medicine and the RJ Fasenmyer Chair of Clinical Immunology at the Cleveland Clinic in Ohio. “For everyone who comes in with a respiratory illness, we will have to figure out whether it is flu or COVID,” he emphasized.
The Centers for Disease Control and Prevention recommendations include a detailed special considerations section for patients with immunocompromising conditions; “the notes have everything you need to know” about advising rheumatology patients, most of whom can safely receive a flu vaccine, he said.
One concern that always comes up is whether an antibody response will be suppressed based on therapy, Dr. Calabrese noted. Two major drugs with the greatest ability to reduce response are methotrexate and rituximab, he said. His tip: “Withhold methotrexate for two doses following seasonal flu vaccination.” This advice stems from a series of “practice-changing” studies by Park et al. published in 2017, 2018, and 2019 that showed benefit in withholding methotrexate for two doses following vaccination.
In the past, high-dose trivalent flu vaccines have been more expensive, and not necessarily practice changing, with studies showing varying clinical effectiveness and cost-effectiveness, Dr. Calabrese said. This year, a high-dose quadrivalent vaccine should be available that showed a 24% improvement in protection from all strains of influenza, compared with the standard vaccine in a head-to-head, randomized, controlled trial, he noted.
“All patients in rheumatology practices should get a flu vaccine,” with a 2-week hold on methotrexate following vaccination, he advised, and those aged 65 years and older should receive the high-dose quadrivalent. Younger patients on immunosuppressive therapy also might be considered for the high-dose vaccine, he said.
Pneumococcal vaccination
Dr. Calabrese also emphasized the value of pneumococcal vaccines for rheumatology patients. “The mortality for invasive disease ranges from 5% to 32%, but patients with immunocompromising conditions are at increased risk.”
Dr. Calabrese added a note on safety: Patients with cryopyrin-associated periodic syndrome (CAPS), a rare hereditary inflammatory disorder with cutaneous, neurologic, ophthalmologic, and rheumatologic manifestations, may have severe local and systemic reactions to the 23-valent polysaccharide vaccine (PPSV23), he said.
However, immunization against pneumococcal disease is safe and effective for most patients with autoimmune and inflammatory disorders regardless of their current therapy, he said. As with influenza, the CDC’s vaccination recommendations provide details for special situations, including immunocompromised individuals, he noted.
Dr. Calabrese recommended the 13-valent pneumococcal conjugate vaccine (PCV13) as soon as possible for rheumatology patients who have never been vaccinated, with follow-up doses of the 23-valent polysaccharide vaccine (PPSV23) at least 8 weeks later, and a PPSV23 booster 5 years after the first PPSV23 dose.
Protecting against shingles
When it comes to managing the varicella zoster virus (VZV) in immunocompromised patients, “prevention is preferable to treatment, as our patients are particularly vulnerable because of age and declining immunity,” Dr. Calabrese said.
Prevention is important because “once herpes zoster develops, the available treatments, including antiviral therapy, do not prevent postherpetic neuralgia in all patients,” he emphasized. “The treatments are complicated and not always effective,” he added.
The complications of zoster are well known, but recent data show an increased risk of cardiovascular disease as well, Dr. Calabrese said. “All the more reason to protect rheumatology patients from incident zoster,” he said.
Currently, the nonlive recombinant subunit zoster vaccine (Shingrix) is the preferred option for VZV vaccination according to the CDC’s Advisory Committee on Immunization Practices, Dr. Calabrese said. The CDC initially recommended its use to prevent herpes zoster and related complications in all immunocompetent adults aged 50 years and older; in an update, a C-level recommendation extends to “all patients aged 50 with or without immunosuppressive illnesses regardless of previous Zostavax exposure,” Dr. Calabrese said. “All patients on or starting [Janus] kinase inhibitors, regardless of age, should be considered” to receive the herpes zoster vaccine, he noted.
In general, promoting vaccination for rheumatology patients and for all patients is a multipronged effort that might include reminders, rewards, education, and standing orders, Dr. Calabrese said. Clinicians must continue to educate patients not only by strongly recommending the appropriate vaccines, but dispelling myths about vaccination, addressing fears, and providing current and accurate information, he said.
Dr. Calabrese disclosed relationships with AbbVie, Bristol-Myers Squibb, Crescendo, Genentech, Gilead, GlaxoSmithKline, Janssen, Novartis, Pfizer, Sanofi-Regeneron, and UCB.
Global Academy for Medical Education and this news organization are owned by the same parent company.
When it comes to preventing infection in rheumatology patients, “vaccination is the best mode of infection protection” and works synergistically with masks and hand washing, according to Leonard H. Calabrese, DO.
“Patients with rheumatic diseases have increased morbidity and mortality [from infection] and a lot of risk factors, including age, comorbidities, cytopenias, and extra-articular disease immunosuppression,” he said in a virtual presentation at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
Unfortunately, vaccination uptake remains “much lower than we would like in this country,” he said. Notably, influenza vaccination remains well below the World Health Organization target of 75%, he said.
Influenza vaccination
Flu vaccination will be even more important this year in the context of the COVID-19 pandemic, said Dr. Calabrese, professor of medicine and the RJ Fasenmyer Chair of Clinical Immunology at the Cleveland Clinic in Ohio. “For everyone who comes in with a respiratory illness, we will have to figure out whether it is flu or COVID,” he emphasized.
The Centers for Disease Control and Prevention recommendations include a detailed special considerations section for patients with immunocompromising conditions; “the notes have everything you need to know” about advising rheumatology patients, most of whom can safely receive a flu vaccine, he said.
One concern that always comes up is whether an antibody response will be suppressed based on therapy, Dr. Calabrese noted. Two major drugs with the greatest ability to reduce response are methotrexate and rituximab, he said. His tip: “Withhold methotrexate for two doses following seasonal flu vaccination.” This advice stems from a series of “practice-changing” studies by Park et al. published in 2017, 2018, and 2019 that showed benefit in withholding methotrexate for two doses following vaccination.
In the past, high-dose trivalent flu vaccines have been more expensive, and not necessarily practice changing, with studies showing varying clinical effectiveness and cost-effectiveness, Dr. Calabrese said. This year, a high-dose quadrivalent vaccine should be available that showed a 24% improvement in protection from all strains of influenza, compared with the standard vaccine in a head-to-head, randomized, controlled trial, he noted.
“All patients in rheumatology practices should get a flu vaccine,” with a 2-week hold on methotrexate following vaccination, he advised, and those aged 65 years and older should receive the high-dose quadrivalent. Younger patients on immunosuppressive therapy also might be considered for the high-dose vaccine, he said.
Pneumococcal vaccination
Dr. Calabrese also emphasized the value of pneumococcal vaccines for rheumatology patients. “The mortality for invasive disease ranges from 5% to 32%, but patients with immunocompromising conditions are at increased risk.”
Dr. Calabrese added a note on safety: Patients with cryopyrin-associated periodic syndrome (CAPS), a rare hereditary inflammatory disorder with cutaneous, neurologic, ophthalmologic, and rheumatologic manifestations, may have severe local and systemic reactions to the 23-valent polysaccharide vaccine (PPSV23), he said.
However, immunization against pneumococcal disease is safe and effective for most patients with autoimmune and inflammatory disorders regardless of their current therapy, he said. As with influenza, the CDC’s vaccination recommendations provide details for special situations, including immunocompromised individuals, he noted.
Dr. Calabrese recommended the 13-valent pneumococcal conjugate vaccine (PCV13) as soon as possible for rheumatology patients who have never been vaccinated, with follow-up doses of the 23-valent polysaccharide vaccine (PPSV23) at least 8 weeks later, and a PPSV23 booster 5 years after the first PPSV23 dose.
Protecting against shingles
When it comes to managing the varicella zoster virus (VZV) in immunocompromised patients, “prevention is preferable to treatment, as our patients are particularly vulnerable because of age and declining immunity,” Dr. Calabrese said.
Prevention is important because “once herpes zoster develops, the available treatments, including antiviral therapy, do not prevent postherpetic neuralgia in all patients,” he emphasized. “The treatments are complicated and not always effective,” he added.
The complications of zoster are well known, but recent data show an increased risk of cardiovascular disease as well, Dr. Calabrese said. “All the more reason to protect rheumatology patients from incident zoster,” he said.
Currently, the nonlive recombinant subunit zoster vaccine (Shingrix) is the preferred option for VZV vaccination according to the CDC’s Advisory Committee on Immunization Practices, Dr. Calabrese said. The CDC initially recommended its use to prevent herpes zoster and related complications in all immunocompetent adults aged 50 years and older; in an update, a C-level recommendation extends to “all patients aged 50 with or without immunosuppressive illnesses regardless of previous Zostavax exposure,” Dr. Calabrese said. “All patients on or starting [Janus] kinase inhibitors, regardless of age, should be considered” to receive the herpes zoster vaccine, he noted.
In general, promoting vaccination for rheumatology patients and for all patients is a multipronged effort that might include reminders, rewards, education, and standing orders, Dr. Calabrese said. Clinicians must continue to educate patients not only by strongly recommending the appropriate vaccines, but dispelling myths about vaccination, addressing fears, and providing current and accurate information, he said.
Dr. Calabrese disclosed relationships with AbbVie, Bristol-Myers Squibb, Crescendo, Genentech, Gilead, GlaxoSmithKline, Janssen, Novartis, Pfizer, Sanofi-Regeneron, and UCB.
Global Academy for Medical Education and this news organization are owned by the same parent company.
FROM PRD 2020
Improving Identification of Patients at Low Risk for Major Cardiac Events After Noncardiac Surgery Using Intraoperative Data
Annually, more than 40 million noncardiac surgeries take place in the US,1 with 1%-3% of patients experiencing a major adverse cardiovascular event (MACE) such as acute myocardial infarction (AMI) or cardiac arrest postoperatively.2 Such patients are at markedly increased risk of both perioperative and long-term death.2-5
Over the past 40 years, efforts to model the risk of cardiac complications after noncardiac surgery have examined relationships between preoperative risk factors and postoperative cardiovascular events. The resulting risk-stratification tools, such as the Lee Revised Cardiac Risk Index (RCRI), have been used to inform perioperative care, including strategies for risk factor management prior to surgery, testing for cardiac events after surgery, and decisions regarding postoperative disposition.6 However, tools used in practice have not incorporated intraoperative data on hemodynamics or medication administration in the transition to postoperative care, which is often provided by nonsurgical clinicians such as hospitalists. Presently, there is active debate about the optimal approach to postoperative evaluation and management of MACE, particularly with regard to indications for cardiac biomarker testing after surgery in patients without signs or symptoms of acute cardiac syndromes. The lack of consensus is reflected in differences among guidelines for postoperative cardiac biomarker testing across professional societies in Europe, Canada, and the United States.7-9
In this study, we examined whether the addition of intraoperative data to preoperative data (together, perioperative data) improved prediction of MACE after noncardiac surgery when compared with RCRI. Additionally, to investigate how such a model could be applied in practice, we compared risk stratification based on our model to a published risk factor–based guideline algorithm for postoperative cardiac biomarker testing.7 In particular, we evaluated to what extent patients recommended for postoperative cardiac biomarkers under the risk factor–based guideline algorithm would be reclassified as low risk by the model using perioperative data. Conducting biomarker tests on these patients would potentially represent low-value care. We hypothesized that adding intraoperative data would (a) lead to improved prediction of MACE complications when compared with RCRI and (b) more effectively identify, compared with a risk factor–based guideline algorithm, patients for whom cardiac biomarker testing would or would not be clinically meaningful.
METHODS
We followed the Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis (TRIPOD) reporting guideline.10
Study Data
Baseline, preoperative, and intraoperative data were collected for patients undergoing surgery between January 2014 and April 2018 within the University of Pennsylvania Health System (UPHS) electronic health record (EHR), and these data were then integrated into a comprehensive perioperative dataset (data containing administrative, preoperative, intraoperative, and postoperative information related to surgeries) created through a collaboration with the Multicenter Perioperative Outcomes Group.11 The University of Pennsylvania Institutional Review Board approved this study.
Study Population
Patients aged 18 years or older who underwent inpatient major noncardiac surgery across four tertiary academic medical centers within UPHS in Pennsylvania during the study period were included in the cohort (see Appendix for inclusion/exclusion criteria).12,13 Noncardiac surgery was identified using primary Current Procedural Terminology (CPT) code specification ranges for noncardiac surgeries 10021-32999 and 34001-69990. The study sample was divided randomly into a training set (60%), validation (20%), and test set (20%),14 with similar rates of MACE in the resulting sets. We used a holdout test set for all final analyses to avoid overfitting during model selection.
Outcomes
The composite outcome used to develop the risk-stratification models was in-hospital MACE after major noncardiac surgery. Following prior literature, MACE was defined using billing codes for ST-elevation/non–ST-elevation myocardial infarction (STEMI/NSTEMI, ICD-9-CM 410.xx, ICD-10-CM I21.xx), cardiac arrest (ICD-9-CM 427.5, ICD-10-CM I46.x, I97.121), or all-cause in-hospital death.2,15-17
Variables
Variables were selected from baseline administrative, preoperative clinical, and intraoperative clinical data sources (full list in Appendix). Baseline variables included demographics, insurance type, and Elixhauser comorbidities.18,19 Preoperative variables included surgery type, laboratory results, and American Society of Anesthesiologists (ASA) Physical Status classification.20 Intraoperative variables included vital signs, estimated blood loss, fluid administration, and vasopressor use. We winsorized outlier values and used multiple imputation to address missingness. Rates of missing data can be found in Appendix Table 1.
Risk-Stratification Models Used as Comparisons
Briefly, RCRI variables include the presence of high-risk surgery,21 comorbid cardiovascular diseases (ie, ischemic heart disease, congestive heart failure, and cerebrovascular disease), preoperative use of insulin, and elevated preoperative serum creatinine.6 RCRI uses the inputs to calculate a point score that equates to different risk strata and is based on a stepwise logistic regression model with postoperative cardiovascular complications as the dependent outcome variable. For this study, we implemented the weighted version of the RCRI algorithm and computed the point scores (Appendix).6,7,22
We also applied a risk factor–based algorithm for postoperative cardiac biomarker testing published in 2017 by the Canadian Cardiovascular Society (CCS) guidelines to each patient in the study sample.7 Specifically, this algorithm recommends daily troponin surveillance for 48 to 72 hours after surgery among patients who have (1) an elevated NT-proBNP/BNP measurement or no NT-proBNP/BNP measurement before surgery, (2) have a Revised Cardiac Risk Index score of 1 or greater, (3) are aged 65 years and older, (4) are aged 45 to 64 years with significant cardiovascular disease undergoing elective surgery, or (5) are aged 18 to 64 years with significant cardiovascular disease undergoing semiurgent, urgent, or emergent surgery.
Statistical Analysis
We compared patient characteristics and outcomes between those who did and those who did not experience MACE during hospitalization. Chi-square tests were used to compare categorical variables and Mann Whitney tests were used to compare continuous variables.
To create the perioperative risk-stratification model based on baseline, preoperative, and intraoperative data, we used a logistic regression with elastic net selection using a dichotomous dependent variable indicating MACE and independent variables described earlier. This perioperative model was fit on the training set and the model coefficients were then applied to the patients in the test set. The area under the receiver operating characteristic curve (AUC) was reported and the outcomes were reported by predicted risk decile, with higher deciles indicating higher risk (ie, higher numbers of patients with MACE outcomes in higher deciles implied better risk stratification). Because predicted risk of postoperative MACE may not have been distributed evenly across deciles, we also examined the distribution of the predicted probability of MACE and examined the number of patients below thresholds of risk corresponding to 0.1% or less, 0.25% or less, 0.5% or less, and 1% or less. These thresholds were chosen because they were close to the overall rate of MACE within our cohort.
We tested for differences in predictive performance between the RCRI logistic regression model AUC and the perioperative model AUC using DeLong’s test.23 Additionally, we illustrated differences between the perioperative and RCRI models’ performance in two ways by stratifying patients into deciles based on predicted risk. First, we compared rates of MACE and MACE component events by predicted decile of the perioperative and RCRI models. Second, we further classified patients as RCRI high or low risk (per RCRI score classification in which RCRI score of 1 or greater is high risk and RCRI score of 0 is low risk) and examined numbers of surgical cases and MACE complications within these categories stratified by perioperative model predicted decile.
To compare the perioperative model’s performance with that of a risk factor–based guideline algorithm, we classified patients according to CCS guidelines as high risk (those for whom the CCS guidelines algorithm would recommend postoperative troponin surveillance testing) and low risk (those for whom the CCS guidelines algorithm would not recommend surveillance testing). We also used a logistic regression to examine if the predicted risk from our model was independently associated with MACE above and beyond the testing recommendation of the CCS guidelines algorithm. This model used MACE as the dependent variable and model-predicted risk and a CCS guidelines–defined high-risk indicator as predictors. We computed the association between a 10 percentage–point increase in predicted risk on observed MACE outcome rates.24
In sensitivity analyses, we used a random forest machine learning classifier to test an alternate model specification, used complete case analysis, varied RCRI thresholds, and limited to patients aged 50 years or older. We also varied the penalty parameter in the elastic net model and plotted AUC versus the number of variables included to examine parsimonious models. SAS v9.4 (SAS Institute Inc) was used for main analyses. Data preparations and sensitivity analysis were done in Python v3.6 with Pandas v0.24.2 and Scikit-learn v0.19.1.
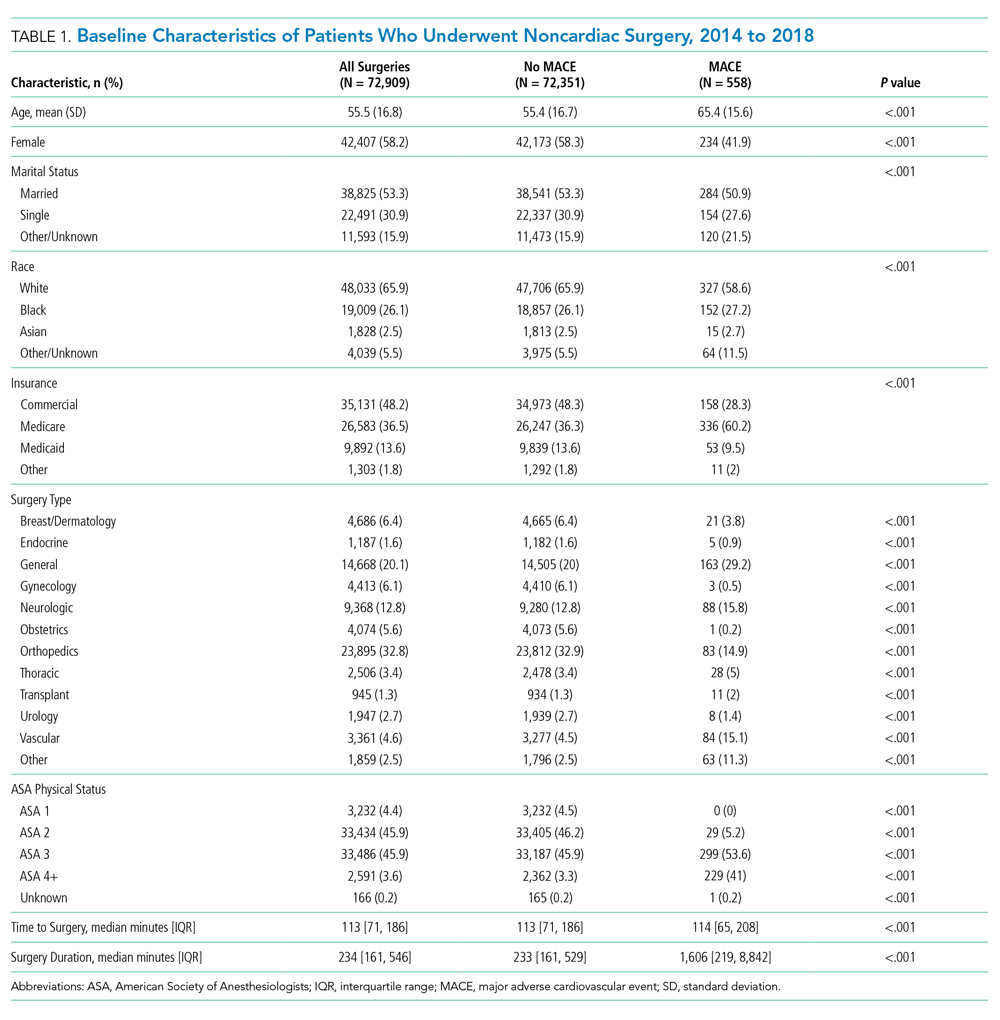
RESULTS
Study Sample
Patients who underwent major noncardiac surgery in our sample (n = 72,909) were approximately a mean age of 56 years, 58% female, 66% of White race and 26% of Black race, and most likely to have received orthopedic surgery (33%) or general surgery (20%). Those who experienced MACE (n = 558; 0.77%) differed along several characteristics (Table 1). For example, those with MACE were older (mean age, 65.4 vs 55.4 years; P < .001) and less likely to be female (41.9% vs 58.3%; P < .001).
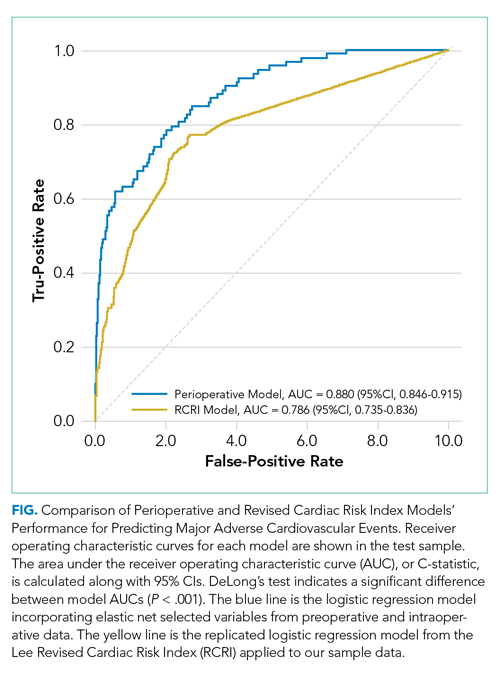
Model Performance After Intraoperative Data Inclusion
In the perioperative model combining preoperative and intraoperative data, 26 variables were included after elastic net selection (Appendix Table 2). Model discrimination in the test set of patients demonstrated an AUC of 0.88 (95% CI, 0.85-0.92; Figure). When examining outcome rates by predicted decile, the outcome rates of in-hospital MACE complications were higher in the highest decile than in the lowest decile, notably with 58 of 92 (63%) cases with MACE complications within the top decile of predicted risk (Table 2). The majority of patients had low predicted risk of MACE, with 5,309 (36.1%), 8,796 (59.7%), 11,335 (77.0%), and 12,972 (88.1%) below the risk thresholds of to 0.1%, 0.25%, 0.5%, and 1.0% respectively. The associated MACE rates were 0.04%, 0.10%, 0.17%, and 0.25% (average rate in sample was 0.63%) (Appendix Table 3).
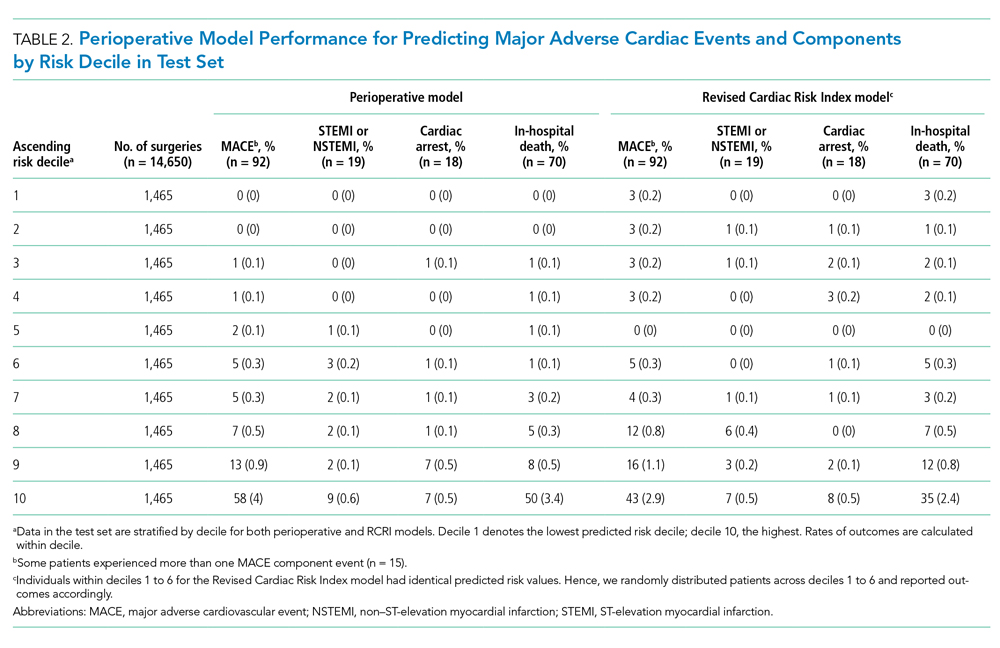
Model Performance Comparisons
The perioperative model AUC of 0.88 was higher when compared with RCRI’s AUC of 0.79 (95% CI, 0.74-0.84; P < .001). The number of MACE complications was more concentrated in the top decile of predicted risk of the perioperative model than it was in that of the RCRI model (58 vs 43 of 92 events, respectively; 63% vs 47%; Table 2). Furthermore, there were fewer cases with MACE complications in the low-risk deciles (ie, deciles 1 to 5) of the perioperative model than in the those of the RCRI model. These relative differences were consistent for MACE component outcomes of STEMI/NSTEMI, cardiac arrest, and in-hospital death, as well.
There was substantial heterogeneity in the perioperative model predicted risk of patients classified as either RCRI low risk or high risk (ie, each category included patients with low and high predicted risk) categories (Table 3). Patients in the bottom (low-risk) five deciles of the perioperative model’s predicted risk who were in the RCRI model’s high-risk group were very unlikely to experience MACE complications (3 out of 722 cases; 0.42%). Furthermore, among those classified as low risk by the RCRI model but were in the top decile of the perioperative model’s predicted risk, the MACE complication rate was 3.5% (8 out of 229), which was 6 times the sample mean MACE complication rate.
The perioperative model identified more patients as low risk than did the CCS guidelines’ risk factor–based algorithm (Table 3). For example, 2,341 of the patients the CCS guidelines algorithm identified as high risk were in the bottom 50% of the perioperative model’s predicted risk for experiencing MACE (below a 0.18% chance of a MACE complication); only four of these patients (0.17%) actually experienced MACE. This indicates that the 2,341 of 7,597 (31%) high-risk patients identified as low risk in the perioperative model would have been recommended for postoperative troponin testing by CCS guidelines based on preoperative risk factors alone—but did not go on to experience a MACE. Regression results indicated that both CCS guidelines risk-factor classification and the perioperative model’s predicted risk were predictive of MACE outcomes. A change in the perioperative model’s predicted risk of 10 percentage points was associated with an increase in the probability of a MACE outcomes of 0.45 percentage points (95% CI, 0.35-0.55 percentage points; P < .001) and moving from CCS guidelines’ low- to high-risk categories was associated with an increased probability of MACE by 0.96 percentage points (95% CI, 0.75-1.16 percentage points; P < .001).
Results were consistent with the main analysis across all sensitivity analyses (Appendix Tables 4-7). Parsimonious models with variables as few as eight variables retained strong predictive power (AUC, 0.870; Appendix Figure 1 and Table 8).
DISCUSSION
In this study, the addition of intraoperative data improved risk stratification for MACE complications when compared with standard risk tools such as RCRI. This approach also outperformed a guidelines-based approach and identified additional patients at low risk of cardiovascular complications. This study has three main implications.
First, this study demonstrated the additional value of combining intraoperative data with preoperative data in risk prediction for postoperative cardiovascular events. The intraoperative data most strongly associated with MACE, which likely were responsible for the performance improvement, included administration of medications (eg, sodium bicarbonate or calcium chloride) and blood products (eg, platelets and packed red blood cells), vitals (ie, heart rate), and intraoperative procedures (ie, arterial line placement); all model variables and coefficients are reported in Appendix Table 9. The risk-stratification model using intraoperative clinical data outperformed validated standard models such as RCRI. While this model should not be used in causal inference and cannot be used to inform decisions about risk-benefit tradeoffs of undergoing surgery, its improved performance relative to prior models highlights the potential in using real-time data. Preliminary illustrative analysis demonstrated that parsimonious models with as few as eight variables perform well, whose implementation as risk scores in EHRs is likely straightforward (Appendix Table 8). This is particularly important for longitudinal care in the hospital, in which patients frequently are cared for by multiple clinical services and experience handoffs. For example, many orthopedic surgery patients with significant medical comorbidity are managed postoperatively by hospitalist physicians after initial surgical care.
Second, our study aligns well with the cardiac risk-stratification literature more broadly. For example, the patient characteristics and clinical variables most associated with cardiovascular complications were age, history of ischemic heart disease, American Society of Anesthesiologists physical status, use of intraoperative sodium bicarbonate or vasopressors, lowest intraoperative heart rate measured, and lowest intraoperative mean arterial pressure measured. While many of these variables overlap with those included in the RCRI model, others (such as American Society of Anesthesiologists physical status) are not included in RCRI but have been shown to be important in risk prediction in other studies using different data variables.6,25,26
Third, we illustrated a clinical application of this model in identifying patients at low risk of cardiovascular complications, although benefit may extend to other patients as well. This is particularly germane to clinicians who frequently manage patients in the postsurgical or postprocedural setting. Moreover, the clinical relevance to these clinicians is underscored by the lack of consensus among professional societies across Europe, Canada, and the United States about which subgroups of patients undergoing noncardiac surgery should receive postoperative cardiac biomarker surveillance testing in the 48 to 72 hours after surgery.6-9 This may be in part caused by differences in clinical objectives. For example, the CCS guidelines in part aim to detect myocardial injury after noncardiac surgery (MINS) up to 30 days after surgery, which may be more sensitive to myocardial injury but less strongly associated with outcomes like MACE. The results of this study suggest that adopting such risk factor–based testing would likely lead to additional testing of low risk patients, which may represent low value surveillance tests. For example, there were 2,257 patients without postoperative cardiac biomarker testing in our data who would have been categorized as high risk by risk factor guidelines and therefore recommended to receive at least one postoperative cardiac biomarker surveillance test but were classified as low-risk individuals using a predicted probability of MACE less than 0.18% per our perioperative risk stratification model (Appendix Table 4). If each of these patients received one troponin biomarker test, the associated cost increase would be $372,405 (using the $165 cost per test reported at our institution). These costs would multiply if daily surveillance troponin biomarker tests were ordered for 48 to 72 hours after surgery, as recommended by the risk factor–based testing guidelines. This would be a departure from testing among patients using clinician discretion that may avoid low-value testing.
Applying the perioperative model developed in this paper to clinical practice still requires several steps. The technical aspects of finding a parsimonious model that can be implemented in the EHR is likely quite straightforward. Our preliminary analysis illustrates that doing so will not require accessing large numbers of intraoperative variables. Perhaps more important steps include prospective validation of the safety, usability, and clinical benefit of such an algorithm-based risk score.27
The study has several limitations. First, it was an observational study using EHR data subject to missingness and data quality issues that may have persisted despite our methods. Furthermore, EHR data is not generated randomly, and unmeasured variables observed by clinicians but not by researchers could confound the results. However, our approach used the statistical model to examine risk, not causal inference. Second, this is a single institution study and the availability of EHR data, as well as practice patterns, may vary at other institutions. Furthermore, it is possible that performance of the RCRI score, the model fitting RCRI classification of high vs low risk on the sample data, and our model’s performance may not generalize to other clinical settings. However, we utilized data from multiple hospitals within a health system with different surgery and anesthesia groups and providers, and a similar AUC was reported for RCRI in original validation study.6 Third, our follow up period was limited to the hospital setting and we do not capture longitudinal outcomes, such as 30-day MACE. This may impact the ability to risk stratify for other important longer-term outcomes, limit clinical utility, and hinder comparability to other studies. Fourth, results may vary for other important cardiovascular outcomes that may be more sensitive to myocardial injury, such as MINS. Fifth, we used a limited number of modeling strategies.
CONCLUSION
Addition of intraoperative data to preoperative data improves prediction of cardiovascular complications after noncardiac surgery. Improving the identification of patients at low risk for such complications could potentially be applied to reduce unnecessary postoperative cardiac biomarker testing after noncardiac surgery, but it will require further validation in prospective clinical settings.
Disclosures
Dr Navathe reports grants from the following entities: Hawaii Medical Service Association, Anthem Public Policy Institute, Commonwealth Fund, Oscar Health, Cigna Corporation, Robert Wood Johnson Foundation, Donaghue Foundation, Pennsylvania Department of Health, Ochsner Health System, United Healthcare, Blue Cross Blue Shield of NC, Blue Shield of CA; personal fees from the following: Navvis Healthcare, Agathos, Inc, Navahealth, YNHHSC/CORE, Maine Health Accountable Care Organization, Maine Department of Health and Human Services, National University Health System - Singapore, Ministry of Health - Singapore, Social Security Administration - France, Elsevier Press, Medicare Payment Advisory Commission, Cleveland Clinic, Embedded Healthcare; and other support from Integrated Services, Inc, outside of the submitted work. Dr Volpp reports grants from Humana during the conduct of the study; grants from Hawaii Medical Services Agency, Discovery (South Africa), Merck, Weight Watchers, and CVS outside of the submitted work; he has received consulting income from CVS and VALHealth and is a principal in VALHealth, a behavioral economics consulting firm. Dr Holmes receives funding from the Pennsylvania Department of Health, US Public Health Service, and the Cardiovascular Medicine Research and Education Foundation. All other authors declare no conflicts of interest.
Prior Presentations
2019 Academy Health Annual Research Meeting, Poster Abstract Presentation, June 2 to June 4, 2019, Washington, DC.
Funding
This project was funded, in part, under a grant with the Pennsylvania Department of Health. This research was independent from the funder. The funder had no role in the study design; in the collection, analysis, and interpretation of data; in the writing of the report; or in the decision to submit the article for publication. The department specifically disclaims responsibility for any analyses, interpretations, or conclusions.
1. National Center for Health Statistics. National Hospital Discharge Survey: 2010 Table, Number of all-listed procedures for discharges from short-stay hospitals, by procedure category and age: United States, 2010. Centers for Disease Control and Prevention; 2010. Accessed November 11, 2018. https://www.cdc.gov/nchs/data/nhds/4procedures/2010pro4_numberprocedureage.pdf
2. Devereaux PJ, Goldman L, Cook DJ, Gilbert K, Leslie K, Guyatt GH. Perioperative cardiac events in patients undergoing noncardiac surgery: a review of the magnitude of the problem, the pathophysiology of the events and methods to estimate and communicate risk. CMAJ. 2005;173(6):627-634. https://doi.org/10.1503/cmaj.050011
3. Charlson M, Peterson J, Szatrowski TP, MacKenzie R, Gold J. Long-term prognosis after peri-operative cardiac complications. J Clin Epidemiol. 1994;47(12):1389-1400. https://doi.org/10.1016/0895-4356(94)90083-3
4. Devereaux PJ, Sessler DI. Cardiac complications in patients undergoing major noncardiac surgery. N Engl J Med. 2015;373(23):2258-2269. https://doi.org/10.1056/nejmra1502824
5. Sprung J, Warner ME, Contreras MG, et al. Predictors of survival following cardiac arrest in patients undergoing noncardiac surgery: a study of 518,294 patients at a tertiary referral center. Anesthesiology. 2003;99(2):259-269. https://doi.org/10.1097/00000542-200308000-00006
6. Lee TH, Marcantonio ER, Mangione CM, et al. Derivation and prospective validation of a simple index for prediction of cardiac risk of major noncardiac surgery. Circulation. 1999;100(10):1043-1049. https://doi.org/10.1161/01.cir.100.10.1043
7. Duceppe E, Parlow J, MacDonald P, et al. Canadian Cardiovascular Society guidelines on perioperative cardiac risk assessment and management for patients who undergo noncardiac surgery. Can J Cardiol. 2017;33(1):17-32. https://doi.org/10.1016/j.cjca.2016.09.008
8. Fleisher LA, Fleischmann KE, Auerbach AD, et al. 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines. J Am Coll Cardiol. 2014;64(22):e77-e137. https://doi.org/10.1016/j.jacc.2014.07.944
9. Kristensen SD, Knuuti J, Saraste A, et al. 2014 ESC/ESA guidelines on non-cardiac surgery: cardiovascular assessment and management: The Joint Task Force on non-cardiac surgery: cardiovascular assessment and management of the European Society of Cardiology (ESC) and the European Society of Anaesthesiology (ESA). Euro Heart J. 2014;35(35):2383-2431. https://doi.org/10.1093/eurheartj/ehu282
10. Collins GS, Reitsma JB, Altman DG, Moons KG. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement. Ann Intern Med. 2015;162(1):55-63. https://doi.org/10.7326/m14-0697
11. Freundlich RE, Kheterpal S. Perioperative effectiveness research using large databases. Best Pract Res Clin Anaesthesiol. 2011;25(4):489-498. https://doi.org/10.1016/j.bpa.2011.08.008
12. CPT® (Current Procedural Terminology). American Medical Association. 2018. Accessed November 11, 2018. https://www.ama-assn.org/practice-management/cpt-current-procedural-terminology
13. Surgery Flag Software for ICD-9-CM. AHRQ Healthcare Cost and Utilization Project; 2017. Accessed November 11, 2018. https://www.hcup-us.ahrq.gov/toolssoftware/surgflags/surgeryflags.jsp
14. Hastie T, Tibshirani R, Friedman J. The Elements of Statistical Learning: Data Mining, Inference, and Prediction. 2nd ed. Springer; 2009. https://www.springer.com/gp/book/9780387848570
15. Bucy R, Hanisko KA, Ewing LA, et al. Abstract 281: Validity of in-hospital cardiac arrest ICD-9-CM codes in veterans. Circ Cardiovasc Qual Outcomes. 2015;8(suppl_2):A281-A281.
16. Institute of Medicine; Board on Health Sciences Policy; Committee on the Treatment of Cardiac Arrest: Current Status and Future Directions. Graham R, McCoy MA, Schultz AM, eds. Strategies to Improve Cardiac Arrest Survival: A Time to Act. The National Academies Press; 2015. https://doi.org/10.17226/21723
17. Pladevall M, Goff DC, Nichaman MZ, et al. An assessment of the validity of ICD Code 410 to identify hospital admissions for myocardial infarction: The Corpus Christi Heart Project. Int J Epidemiol. 1996;25(5):948-952. https://doi.org/10.1093/ije/25.5.948
18. Elixhauser A, Steiner C, Harris DR, Coffey RM. Comorbidity measures for use with administrative data. Med Care. 1998;36(1):8-27. https://doi.org/10.1097/00005650-199801000-00004
19. Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130-1139. https://doi.org/10.1097/01.mlr.0000182534.19832.83
20. Keats AS. The ASA classification of physical status--a recapitulation. Anesthesiology. 1978;49(4):233-236. https://doi.org/10.1097/00000542-197810000-00001
21. Schwarze ML, Barnato AE, Rathouz PJ, et al. Development of a list of high-risk operations for patients 65 years and older. JAMA Surg. 2015;150(4):325-331. https://doi.org/10.1001/jamasurg.2014.1819
22. VISION Pilot Study Investigators, Devereaux PJ, Bradley D, et al. An international prospective cohort study evaluating major vascular complications among patients undergoing noncardiac surgery: the VISION Pilot Study. Open Med. 2011;5(4):e193-e200.
23. DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44(3):837-845.
24. Norton EC, Dowd BE, Maciejewski ML. Marginal effects-quantifying the effect of changes in risk factors in logistic regression models. JAMA. 2019;321(13):1304‐1305. https://doi.org/10.1001/jama.2019.1954
25. Bilimoria KY, Liu Y, Paruch JL, et al. Development and evaluation of the universal ACS NSQIP surgical risk calculator: a decision aid and informed consent tool for patients and surgeons. J Am Coll Surg. 2013;217(5):833-842. https://doi.org/10.1016/j.jamcollsurg.2013.07.385
26. Gawande AA, Kwaan MR, Regenbogen SE, Lipsitz SA, Zinner MJ. An Apgar score for surgery. J Am Coll Surg. 2007;204(2):201-208. https://doi.org/10.1016/j.jamcollsurg.2006.11.011
27. Parikh RB, Obermeyer Z, Navathe AS. Regulation of predictive analytics in medicine. Science. 2019;363(6429):810-812. https://doi.org/10.1126/science.aaw0029
Annually, more than 40 million noncardiac surgeries take place in the US,1 with 1%-3% of patients experiencing a major adverse cardiovascular event (MACE) such as acute myocardial infarction (AMI) or cardiac arrest postoperatively.2 Such patients are at markedly increased risk of both perioperative and long-term death.2-5
Over the past 40 years, efforts to model the risk of cardiac complications after noncardiac surgery have examined relationships between preoperative risk factors and postoperative cardiovascular events. The resulting risk-stratification tools, such as the Lee Revised Cardiac Risk Index (RCRI), have been used to inform perioperative care, including strategies for risk factor management prior to surgery, testing for cardiac events after surgery, and decisions regarding postoperative disposition.6 However, tools used in practice have not incorporated intraoperative data on hemodynamics or medication administration in the transition to postoperative care, which is often provided by nonsurgical clinicians such as hospitalists. Presently, there is active debate about the optimal approach to postoperative evaluation and management of MACE, particularly with regard to indications for cardiac biomarker testing after surgery in patients without signs or symptoms of acute cardiac syndromes. The lack of consensus is reflected in differences among guidelines for postoperative cardiac biomarker testing across professional societies in Europe, Canada, and the United States.7-9
In this study, we examined whether the addition of intraoperative data to preoperative data (together, perioperative data) improved prediction of MACE after noncardiac surgery when compared with RCRI. Additionally, to investigate how such a model could be applied in practice, we compared risk stratification based on our model to a published risk factor–based guideline algorithm for postoperative cardiac biomarker testing.7 In particular, we evaluated to what extent patients recommended for postoperative cardiac biomarkers under the risk factor–based guideline algorithm would be reclassified as low risk by the model using perioperative data. Conducting biomarker tests on these patients would potentially represent low-value care. We hypothesized that adding intraoperative data would (a) lead to improved prediction of MACE complications when compared with RCRI and (b) more effectively identify, compared with a risk factor–based guideline algorithm, patients for whom cardiac biomarker testing would or would not be clinically meaningful.
METHODS
We followed the Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis (TRIPOD) reporting guideline.10
Study Data
Baseline, preoperative, and intraoperative data were collected for patients undergoing surgery between January 2014 and April 2018 within the University of Pennsylvania Health System (UPHS) electronic health record (EHR), and these data were then integrated into a comprehensive perioperative dataset (data containing administrative, preoperative, intraoperative, and postoperative information related to surgeries) created through a collaboration with the Multicenter Perioperative Outcomes Group.11 The University of Pennsylvania Institutional Review Board approved this study.
Study Population
Patients aged 18 years or older who underwent inpatient major noncardiac surgery across four tertiary academic medical centers within UPHS in Pennsylvania during the study period were included in the cohort (see Appendix for inclusion/exclusion criteria).12,13 Noncardiac surgery was identified using primary Current Procedural Terminology (CPT) code specification ranges for noncardiac surgeries 10021-32999 and 34001-69990. The study sample was divided randomly into a training set (60%), validation (20%), and test set (20%),14 with similar rates of MACE in the resulting sets. We used a holdout test set for all final analyses to avoid overfitting during model selection.
Outcomes
The composite outcome used to develop the risk-stratification models was in-hospital MACE after major noncardiac surgery. Following prior literature, MACE was defined using billing codes for ST-elevation/non–ST-elevation myocardial infarction (STEMI/NSTEMI, ICD-9-CM 410.xx, ICD-10-CM I21.xx), cardiac arrest (ICD-9-CM 427.5, ICD-10-CM I46.x, I97.121), or all-cause in-hospital death.2,15-17
Variables
Variables were selected from baseline administrative, preoperative clinical, and intraoperative clinical data sources (full list in Appendix). Baseline variables included demographics, insurance type, and Elixhauser comorbidities.18,19 Preoperative variables included surgery type, laboratory results, and American Society of Anesthesiologists (ASA) Physical Status classification.20 Intraoperative variables included vital signs, estimated blood loss, fluid administration, and vasopressor use. We winsorized outlier values and used multiple imputation to address missingness. Rates of missing data can be found in Appendix Table 1.
Risk-Stratification Models Used as Comparisons
Briefly, RCRI variables include the presence of high-risk surgery,21 comorbid cardiovascular diseases (ie, ischemic heart disease, congestive heart failure, and cerebrovascular disease), preoperative use of insulin, and elevated preoperative serum creatinine.6 RCRI uses the inputs to calculate a point score that equates to different risk strata and is based on a stepwise logistic regression model with postoperative cardiovascular complications as the dependent outcome variable. For this study, we implemented the weighted version of the RCRI algorithm and computed the point scores (Appendix).6,7,22
We also applied a risk factor–based algorithm for postoperative cardiac biomarker testing published in 2017 by the Canadian Cardiovascular Society (CCS) guidelines to each patient in the study sample.7 Specifically, this algorithm recommends daily troponin surveillance for 48 to 72 hours after surgery among patients who have (1) an elevated NT-proBNP/BNP measurement or no NT-proBNP/BNP measurement before surgery, (2) have a Revised Cardiac Risk Index score of 1 or greater, (3) are aged 65 years and older, (4) are aged 45 to 64 years with significant cardiovascular disease undergoing elective surgery, or (5) are aged 18 to 64 years with significant cardiovascular disease undergoing semiurgent, urgent, or emergent surgery.
Statistical Analysis
We compared patient characteristics and outcomes between those who did and those who did not experience MACE during hospitalization. Chi-square tests were used to compare categorical variables and Mann Whitney tests were used to compare continuous variables.
To create the perioperative risk-stratification model based on baseline, preoperative, and intraoperative data, we used a logistic regression with elastic net selection using a dichotomous dependent variable indicating MACE and independent variables described earlier. This perioperative model was fit on the training set and the model coefficients were then applied to the patients in the test set. The area under the receiver operating characteristic curve (AUC) was reported and the outcomes were reported by predicted risk decile, with higher deciles indicating higher risk (ie, higher numbers of patients with MACE outcomes in higher deciles implied better risk stratification). Because predicted risk of postoperative MACE may not have been distributed evenly across deciles, we also examined the distribution of the predicted probability of MACE and examined the number of patients below thresholds of risk corresponding to 0.1% or less, 0.25% or less, 0.5% or less, and 1% or less. These thresholds were chosen because they were close to the overall rate of MACE within our cohort.
We tested for differences in predictive performance between the RCRI logistic regression model AUC and the perioperative model AUC using DeLong’s test.23 Additionally, we illustrated differences between the perioperative and RCRI models’ performance in two ways by stratifying patients into deciles based on predicted risk. First, we compared rates of MACE and MACE component events by predicted decile of the perioperative and RCRI models. Second, we further classified patients as RCRI high or low risk (per RCRI score classification in which RCRI score of 1 or greater is high risk and RCRI score of 0 is low risk) and examined numbers of surgical cases and MACE complications within these categories stratified by perioperative model predicted decile.
To compare the perioperative model’s performance with that of a risk factor–based guideline algorithm, we classified patients according to CCS guidelines as high risk (those for whom the CCS guidelines algorithm would recommend postoperative troponin surveillance testing) and low risk (those for whom the CCS guidelines algorithm would not recommend surveillance testing). We also used a logistic regression to examine if the predicted risk from our model was independently associated with MACE above and beyond the testing recommendation of the CCS guidelines algorithm. This model used MACE as the dependent variable and model-predicted risk and a CCS guidelines–defined high-risk indicator as predictors. We computed the association between a 10 percentage–point increase in predicted risk on observed MACE outcome rates.24
In sensitivity analyses, we used a random forest machine learning classifier to test an alternate model specification, used complete case analysis, varied RCRI thresholds, and limited to patients aged 50 years or older. We also varied the penalty parameter in the elastic net model and plotted AUC versus the number of variables included to examine parsimonious models. SAS v9.4 (SAS Institute Inc) was used for main analyses. Data preparations and sensitivity analysis were done in Python v3.6 with Pandas v0.24.2 and Scikit-learn v0.19.1.

RESULTS
Study Sample
Patients who underwent major noncardiac surgery in our sample (n = 72,909) were approximately a mean age of 56 years, 58% female, 66% of White race and 26% of Black race, and most likely to have received orthopedic surgery (33%) or general surgery (20%). Those who experienced MACE (n = 558; 0.77%) differed along several characteristics (Table 1). For example, those with MACE were older (mean age, 65.4 vs 55.4 years; P < .001) and less likely to be female (41.9% vs 58.3%; P < .001).

Model Performance After Intraoperative Data Inclusion
In the perioperative model combining preoperative and intraoperative data, 26 variables were included after elastic net selection (Appendix Table 2). Model discrimination in the test set of patients demonstrated an AUC of 0.88 (95% CI, 0.85-0.92; Figure). When examining outcome rates by predicted decile, the outcome rates of in-hospital MACE complications were higher in the highest decile than in the lowest decile, notably with 58 of 92 (63%) cases with MACE complications within the top decile of predicted risk (Table 2). The majority of patients had low predicted risk of MACE, with 5,309 (36.1%), 8,796 (59.7%), 11,335 (77.0%), and 12,972 (88.1%) below the risk thresholds of to 0.1%, 0.25%, 0.5%, and 1.0% respectively. The associated MACE rates were 0.04%, 0.10%, 0.17%, and 0.25% (average rate in sample was 0.63%) (Appendix Table 3).

Model Performance Comparisons
The perioperative model AUC of 0.88 was higher when compared with RCRI’s AUC of 0.79 (95% CI, 0.74-0.84; P < .001). The number of MACE complications was more concentrated in the top decile of predicted risk of the perioperative model than it was in that of the RCRI model (58 vs 43 of 92 events, respectively; 63% vs 47%; Table 2). Furthermore, there were fewer cases with MACE complications in the low-risk deciles (ie, deciles 1 to 5) of the perioperative model than in the those of the RCRI model. These relative differences were consistent for MACE component outcomes of STEMI/NSTEMI, cardiac arrest, and in-hospital death, as well.
There was substantial heterogeneity in the perioperative model predicted risk of patients classified as either RCRI low risk or high risk (ie, each category included patients with low and high predicted risk) categories (Table 3). Patients in the bottom (low-risk) five deciles of the perioperative model’s predicted risk who were in the RCRI model’s high-risk group were very unlikely to experience MACE complications (3 out of 722 cases; 0.42%). Furthermore, among those classified as low risk by the RCRI model but were in the top decile of the perioperative model’s predicted risk, the MACE complication rate was 3.5% (8 out of 229), which was 6 times the sample mean MACE complication rate.

The perioperative model identified more patients as low risk than did the CCS guidelines’ risk factor–based algorithm (Table 3). For example, 2,341 of the patients the CCS guidelines algorithm identified as high risk were in the bottom 50% of the perioperative model’s predicted risk for experiencing MACE (below a 0.18% chance of a MACE complication); only four of these patients (0.17%) actually experienced MACE. This indicates that the 2,341 of 7,597 (31%) high-risk patients identified as low risk in the perioperative model would have been recommended for postoperative troponin testing by CCS guidelines based on preoperative risk factors alone—but did not go on to experience a MACE. Regression results indicated that both CCS guidelines risk-factor classification and the perioperative model’s predicted risk were predictive of MACE outcomes. A change in the perioperative model’s predicted risk of 10 percentage points was associated with an increase in the probability of a MACE outcomes of 0.45 percentage points (95% CI, 0.35-0.55 percentage points; P < .001) and moving from CCS guidelines’ low- to high-risk categories was associated with an increased probability of MACE by 0.96 percentage points (95% CI, 0.75-1.16 percentage points; P < .001).
Results were consistent with the main analysis across all sensitivity analyses (Appendix Tables 4-7). Parsimonious models with variables as few as eight variables retained strong predictive power (AUC, 0.870; Appendix Figure 1 and Table 8).
DISCUSSION
In this study, the addition of intraoperative data improved risk stratification for MACE complications when compared with standard risk tools such as RCRI. This approach also outperformed a guidelines-based approach and identified additional patients at low risk of cardiovascular complications. This study has three main implications.
First, this study demonstrated the additional value of combining intraoperative data with preoperative data in risk prediction for postoperative cardiovascular events. The intraoperative data most strongly associated with MACE, which likely were responsible for the performance improvement, included administration of medications (eg, sodium bicarbonate or calcium chloride) and blood products (eg, platelets and packed red blood cells), vitals (ie, heart rate), and intraoperative procedures (ie, arterial line placement); all model variables and coefficients are reported in Appendix Table 9. The risk-stratification model using intraoperative clinical data outperformed validated standard models such as RCRI. While this model should not be used in causal inference and cannot be used to inform decisions about risk-benefit tradeoffs of undergoing surgery, its improved performance relative to prior models highlights the potential in using real-time data. Preliminary illustrative analysis demonstrated that parsimonious models with as few as eight variables perform well, whose implementation as risk scores in EHRs is likely straightforward (Appendix Table 8). This is particularly important for longitudinal care in the hospital, in which patients frequently are cared for by multiple clinical services and experience handoffs. For example, many orthopedic surgery patients with significant medical comorbidity are managed postoperatively by hospitalist physicians after initial surgical care.
Second, our study aligns well with the cardiac risk-stratification literature more broadly. For example, the patient characteristics and clinical variables most associated with cardiovascular complications were age, history of ischemic heart disease, American Society of Anesthesiologists physical status, use of intraoperative sodium bicarbonate or vasopressors, lowest intraoperative heart rate measured, and lowest intraoperative mean arterial pressure measured. While many of these variables overlap with those included in the RCRI model, others (such as American Society of Anesthesiologists physical status) are not included in RCRI but have been shown to be important in risk prediction in other studies using different data variables.6,25,26
Third, we illustrated a clinical application of this model in identifying patients at low risk of cardiovascular complications, although benefit may extend to other patients as well. This is particularly germane to clinicians who frequently manage patients in the postsurgical or postprocedural setting. Moreover, the clinical relevance to these clinicians is underscored by the lack of consensus among professional societies across Europe, Canada, and the United States about which subgroups of patients undergoing noncardiac surgery should receive postoperative cardiac biomarker surveillance testing in the 48 to 72 hours after surgery.6-9 This may be in part caused by differences in clinical objectives. For example, the CCS guidelines in part aim to detect myocardial injury after noncardiac surgery (MINS) up to 30 days after surgery, which may be more sensitive to myocardial injury but less strongly associated with outcomes like MACE. The results of this study suggest that adopting such risk factor–based testing would likely lead to additional testing of low risk patients, which may represent low value surveillance tests. For example, there were 2,257 patients without postoperative cardiac biomarker testing in our data who would have been categorized as high risk by risk factor guidelines and therefore recommended to receive at least one postoperative cardiac biomarker surveillance test but were classified as low-risk individuals using a predicted probability of MACE less than 0.18% per our perioperative risk stratification model (Appendix Table 4). If each of these patients received one troponin biomarker test, the associated cost increase would be $372,405 (using the $165 cost per test reported at our institution). These costs would multiply if daily surveillance troponin biomarker tests were ordered for 48 to 72 hours after surgery, as recommended by the risk factor–based testing guidelines. This would be a departure from testing among patients using clinician discretion that may avoid low-value testing.
Applying the perioperative model developed in this paper to clinical practice still requires several steps. The technical aspects of finding a parsimonious model that can be implemented in the EHR is likely quite straightforward. Our preliminary analysis illustrates that doing so will not require accessing large numbers of intraoperative variables. Perhaps more important steps include prospective validation of the safety, usability, and clinical benefit of such an algorithm-based risk score.27
The study has several limitations. First, it was an observational study using EHR data subject to missingness and data quality issues that may have persisted despite our methods. Furthermore, EHR data is not generated randomly, and unmeasured variables observed by clinicians but not by researchers could confound the results. However, our approach used the statistical model to examine risk, not causal inference. Second, this is a single institution study and the availability of EHR data, as well as practice patterns, may vary at other institutions. Furthermore, it is possible that performance of the RCRI score, the model fitting RCRI classification of high vs low risk on the sample data, and our model’s performance may not generalize to other clinical settings. However, we utilized data from multiple hospitals within a health system with different surgery and anesthesia groups and providers, and a similar AUC was reported for RCRI in original validation study.6 Third, our follow up period was limited to the hospital setting and we do not capture longitudinal outcomes, such as 30-day MACE. This may impact the ability to risk stratify for other important longer-term outcomes, limit clinical utility, and hinder comparability to other studies. Fourth, results may vary for other important cardiovascular outcomes that may be more sensitive to myocardial injury, such as MINS. Fifth, we used a limited number of modeling strategies.
CONCLUSION
Addition of intraoperative data to preoperative data improves prediction of cardiovascular complications after noncardiac surgery. Improving the identification of patients at low risk for such complications could potentially be applied to reduce unnecessary postoperative cardiac biomarker testing after noncardiac surgery, but it will require further validation in prospective clinical settings.
Disclosures
Dr Navathe reports grants from the following entities: Hawaii Medical Service Association, Anthem Public Policy Institute, Commonwealth Fund, Oscar Health, Cigna Corporation, Robert Wood Johnson Foundation, Donaghue Foundation, Pennsylvania Department of Health, Ochsner Health System, United Healthcare, Blue Cross Blue Shield of NC, Blue Shield of CA; personal fees from the following: Navvis Healthcare, Agathos, Inc, Navahealth, YNHHSC/CORE, Maine Health Accountable Care Organization, Maine Department of Health and Human Services, National University Health System - Singapore, Ministry of Health - Singapore, Social Security Administration - France, Elsevier Press, Medicare Payment Advisory Commission, Cleveland Clinic, Embedded Healthcare; and other support from Integrated Services, Inc, outside of the submitted work. Dr Volpp reports grants from Humana during the conduct of the study; grants from Hawaii Medical Services Agency, Discovery (South Africa), Merck, Weight Watchers, and CVS outside of the submitted work; he has received consulting income from CVS and VALHealth and is a principal in VALHealth, a behavioral economics consulting firm. Dr Holmes receives funding from the Pennsylvania Department of Health, US Public Health Service, and the Cardiovascular Medicine Research and Education Foundation. All other authors declare no conflicts of interest.
Prior Presentations
2019 Academy Health Annual Research Meeting, Poster Abstract Presentation, June 2 to June 4, 2019, Washington, DC.
Funding
This project was funded, in part, under a grant with the Pennsylvania Department of Health. This research was independent from the funder. The funder had no role in the study design; in the collection, analysis, and interpretation of data; in the writing of the report; or in the decision to submit the article for publication. The department specifically disclaims responsibility for any analyses, interpretations, or conclusions.
Annually, more than 40 million noncardiac surgeries take place in the US,1 with 1%-3% of patients experiencing a major adverse cardiovascular event (MACE) such as acute myocardial infarction (AMI) or cardiac arrest postoperatively.2 Such patients are at markedly increased risk of both perioperative and long-term death.2-5
Over the past 40 years, efforts to model the risk of cardiac complications after noncardiac surgery have examined relationships between preoperative risk factors and postoperative cardiovascular events. The resulting risk-stratification tools, such as the Lee Revised Cardiac Risk Index (RCRI), have been used to inform perioperative care, including strategies for risk factor management prior to surgery, testing for cardiac events after surgery, and decisions regarding postoperative disposition.6 However, tools used in practice have not incorporated intraoperative data on hemodynamics or medication administration in the transition to postoperative care, which is often provided by nonsurgical clinicians such as hospitalists. Presently, there is active debate about the optimal approach to postoperative evaluation and management of MACE, particularly with regard to indications for cardiac biomarker testing after surgery in patients without signs or symptoms of acute cardiac syndromes. The lack of consensus is reflected in differences among guidelines for postoperative cardiac biomarker testing across professional societies in Europe, Canada, and the United States.7-9
In this study, we examined whether the addition of intraoperative data to preoperative data (together, perioperative data) improved prediction of MACE after noncardiac surgery when compared with RCRI. Additionally, to investigate how such a model could be applied in practice, we compared risk stratification based on our model to a published risk factor–based guideline algorithm for postoperative cardiac biomarker testing.7 In particular, we evaluated to what extent patients recommended for postoperative cardiac biomarkers under the risk factor–based guideline algorithm would be reclassified as low risk by the model using perioperative data. Conducting biomarker tests on these patients would potentially represent low-value care. We hypothesized that adding intraoperative data would (a) lead to improved prediction of MACE complications when compared with RCRI and (b) more effectively identify, compared with a risk factor–based guideline algorithm, patients for whom cardiac biomarker testing would or would not be clinically meaningful.
METHODS
We followed the Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis (TRIPOD) reporting guideline.10
Study Data
Baseline, preoperative, and intraoperative data were collected for patients undergoing surgery between January 2014 and April 2018 within the University of Pennsylvania Health System (UPHS) electronic health record (EHR), and these data were then integrated into a comprehensive perioperative dataset (data containing administrative, preoperative, intraoperative, and postoperative information related to surgeries) created through a collaboration with the Multicenter Perioperative Outcomes Group.11 The University of Pennsylvania Institutional Review Board approved this study.
Study Population
Patients aged 18 years or older who underwent inpatient major noncardiac surgery across four tertiary academic medical centers within UPHS in Pennsylvania during the study period were included in the cohort (see Appendix for inclusion/exclusion criteria).12,13 Noncardiac surgery was identified using primary Current Procedural Terminology (CPT) code specification ranges for noncardiac surgeries 10021-32999 and 34001-69990. The study sample was divided randomly into a training set (60%), validation (20%), and test set (20%),14 with similar rates of MACE in the resulting sets. We used a holdout test set for all final analyses to avoid overfitting during model selection.
Outcomes
The composite outcome used to develop the risk-stratification models was in-hospital MACE after major noncardiac surgery. Following prior literature, MACE was defined using billing codes for ST-elevation/non–ST-elevation myocardial infarction (STEMI/NSTEMI, ICD-9-CM 410.xx, ICD-10-CM I21.xx), cardiac arrest (ICD-9-CM 427.5, ICD-10-CM I46.x, I97.121), or all-cause in-hospital death.2,15-17
Variables
Variables were selected from baseline administrative, preoperative clinical, and intraoperative clinical data sources (full list in Appendix). Baseline variables included demographics, insurance type, and Elixhauser comorbidities.18,19 Preoperative variables included surgery type, laboratory results, and American Society of Anesthesiologists (ASA) Physical Status classification.20 Intraoperative variables included vital signs, estimated blood loss, fluid administration, and vasopressor use. We winsorized outlier values and used multiple imputation to address missingness. Rates of missing data can be found in Appendix Table 1.
Risk-Stratification Models Used as Comparisons
Briefly, RCRI variables include the presence of high-risk surgery,21 comorbid cardiovascular diseases (ie, ischemic heart disease, congestive heart failure, and cerebrovascular disease), preoperative use of insulin, and elevated preoperative serum creatinine.6 RCRI uses the inputs to calculate a point score that equates to different risk strata and is based on a stepwise logistic regression model with postoperative cardiovascular complications as the dependent outcome variable. For this study, we implemented the weighted version of the RCRI algorithm and computed the point scores (Appendix).6,7,22
We also applied a risk factor–based algorithm for postoperative cardiac biomarker testing published in 2017 by the Canadian Cardiovascular Society (CCS) guidelines to each patient in the study sample.7 Specifically, this algorithm recommends daily troponin surveillance for 48 to 72 hours after surgery among patients who have (1) an elevated NT-proBNP/BNP measurement or no NT-proBNP/BNP measurement before surgery, (2) have a Revised Cardiac Risk Index score of 1 or greater, (3) are aged 65 years and older, (4) are aged 45 to 64 years with significant cardiovascular disease undergoing elective surgery, or (5) are aged 18 to 64 years with significant cardiovascular disease undergoing semiurgent, urgent, or emergent surgery.
Statistical Analysis
We compared patient characteristics and outcomes between those who did and those who did not experience MACE during hospitalization. Chi-square tests were used to compare categorical variables and Mann Whitney tests were used to compare continuous variables.
To create the perioperative risk-stratification model based on baseline, preoperative, and intraoperative data, we used a logistic regression with elastic net selection using a dichotomous dependent variable indicating MACE and independent variables described earlier. This perioperative model was fit on the training set and the model coefficients were then applied to the patients in the test set. The area under the receiver operating characteristic curve (AUC) was reported and the outcomes were reported by predicted risk decile, with higher deciles indicating higher risk (ie, higher numbers of patients with MACE outcomes in higher deciles implied better risk stratification). Because predicted risk of postoperative MACE may not have been distributed evenly across deciles, we also examined the distribution of the predicted probability of MACE and examined the number of patients below thresholds of risk corresponding to 0.1% or less, 0.25% or less, 0.5% or less, and 1% or less. These thresholds were chosen because they were close to the overall rate of MACE within our cohort.
We tested for differences in predictive performance between the RCRI logistic regression model AUC and the perioperative model AUC using DeLong’s test.23 Additionally, we illustrated differences between the perioperative and RCRI models’ performance in two ways by stratifying patients into deciles based on predicted risk. First, we compared rates of MACE and MACE component events by predicted decile of the perioperative and RCRI models. Second, we further classified patients as RCRI high or low risk (per RCRI score classification in which RCRI score of 1 or greater is high risk and RCRI score of 0 is low risk) and examined numbers of surgical cases and MACE complications within these categories stratified by perioperative model predicted decile.
To compare the perioperative model’s performance with that of a risk factor–based guideline algorithm, we classified patients according to CCS guidelines as high risk (those for whom the CCS guidelines algorithm would recommend postoperative troponin surveillance testing) and low risk (those for whom the CCS guidelines algorithm would not recommend surveillance testing). We also used a logistic regression to examine if the predicted risk from our model was independently associated with MACE above and beyond the testing recommendation of the CCS guidelines algorithm. This model used MACE as the dependent variable and model-predicted risk and a CCS guidelines–defined high-risk indicator as predictors. We computed the association between a 10 percentage–point increase in predicted risk on observed MACE outcome rates.24
In sensitivity analyses, we used a random forest machine learning classifier to test an alternate model specification, used complete case analysis, varied RCRI thresholds, and limited to patients aged 50 years or older. We also varied the penalty parameter in the elastic net model and plotted AUC versus the number of variables included to examine parsimonious models. SAS v9.4 (SAS Institute Inc) was used for main analyses. Data preparations and sensitivity analysis were done in Python v3.6 with Pandas v0.24.2 and Scikit-learn v0.19.1.

RESULTS
Study Sample
Patients who underwent major noncardiac surgery in our sample (n = 72,909) were approximately a mean age of 56 years, 58% female, 66% of White race and 26% of Black race, and most likely to have received orthopedic surgery (33%) or general surgery (20%). Those who experienced MACE (n = 558; 0.77%) differed along several characteristics (Table 1). For example, those with MACE were older (mean age, 65.4 vs 55.4 years; P < .001) and less likely to be female (41.9% vs 58.3%; P < .001).

Model Performance After Intraoperative Data Inclusion
In the perioperative model combining preoperative and intraoperative data, 26 variables were included after elastic net selection (Appendix Table 2). Model discrimination in the test set of patients demonstrated an AUC of 0.88 (95% CI, 0.85-0.92; Figure). When examining outcome rates by predicted decile, the outcome rates of in-hospital MACE complications were higher in the highest decile than in the lowest decile, notably with 58 of 92 (63%) cases with MACE complications within the top decile of predicted risk (Table 2). The majority of patients had low predicted risk of MACE, with 5,309 (36.1%), 8,796 (59.7%), 11,335 (77.0%), and 12,972 (88.1%) below the risk thresholds of to 0.1%, 0.25%, 0.5%, and 1.0% respectively. The associated MACE rates were 0.04%, 0.10%, 0.17%, and 0.25% (average rate in sample was 0.63%) (Appendix Table 3).

Model Performance Comparisons
The perioperative model AUC of 0.88 was higher when compared with RCRI’s AUC of 0.79 (95% CI, 0.74-0.84; P < .001). The number of MACE complications was more concentrated in the top decile of predicted risk of the perioperative model than it was in that of the RCRI model (58 vs 43 of 92 events, respectively; 63% vs 47%; Table 2). Furthermore, there were fewer cases with MACE complications in the low-risk deciles (ie, deciles 1 to 5) of the perioperative model than in the those of the RCRI model. These relative differences were consistent for MACE component outcomes of STEMI/NSTEMI, cardiac arrest, and in-hospital death, as well.
There was substantial heterogeneity in the perioperative model predicted risk of patients classified as either RCRI low risk or high risk (ie, each category included patients with low and high predicted risk) categories (Table 3). Patients in the bottom (low-risk) five deciles of the perioperative model’s predicted risk who were in the RCRI model’s high-risk group were very unlikely to experience MACE complications (3 out of 722 cases; 0.42%). Furthermore, among those classified as low risk by the RCRI model but were in the top decile of the perioperative model’s predicted risk, the MACE complication rate was 3.5% (8 out of 229), which was 6 times the sample mean MACE complication rate.

The perioperative model identified more patients as low risk than did the CCS guidelines’ risk factor–based algorithm (Table 3). For example, 2,341 of the patients the CCS guidelines algorithm identified as high risk were in the bottom 50% of the perioperative model’s predicted risk for experiencing MACE (below a 0.18% chance of a MACE complication); only four of these patients (0.17%) actually experienced MACE. This indicates that the 2,341 of 7,597 (31%) high-risk patients identified as low risk in the perioperative model would have been recommended for postoperative troponin testing by CCS guidelines based on preoperative risk factors alone—but did not go on to experience a MACE. Regression results indicated that both CCS guidelines risk-factor classification and the perioperative model’s predicted risk were predictive of MACE outcomes. A change in the perioperative model’s predicted risk of 10 percentage points was associated with an increase in the probability of a MACE outcomes of 0.45 percentage points (95% CI, 0.35-0.55 percentage points; P < .001) and moving from CCS guidelines’ low- to high-risk categories was associated with an increased probability of MACE by 0.96 percentage points (95% CI, 0.75-1.16 percentage points; P < .001).
Results were consistent with the main analysis across all sensitivity analyses (Appendix Tables 4-7). Parsimonious models with variables as few as eight variables retained strong predictive power (AUC, 0.870; Appendix Figure 1 and Table 8).
DISCUSSION
In this study, the addition of intraoperative data improved risk stratification for MACE complications when compared with standard risk tools such as RCRI. This approach also outperformed a guidelines-based approach and identified additional patients at low risk of cardiovascular complications. This study has three main implications.
First, this study demonstrated the additional value of combining intraoperative data with preoperative data in risk prediction for postoperative cardiovascular events. The intraoperative data most strongly associated with MACE, which likely were responsible for the performance improvement, included administration of medications (eg, sodium bicarbonate or calcium chloride) and blood products (eg, platelets and packed red blood cells), vitals (ie, heart rate), and intraoperative procedures (ie, arterial line placement); all model variables and coefficients are reported in Appendix Table 9. The risk-stratification model using intraoperative clinical data outperformed validated standard models such as RCRI. While this model should not be used in causal inference and cannot be used to inform decisions about risk-benefit tradeoffs of undergoing surgery, its improved performance relative to prior models highlights the potential in using real-time data. Preliminary illustrative analysis demonstrated that parsimonious models with as few as eight variables perform well, whose implementation as risk scores in EHRs is likely straightforward (Appendix Table 8). This is particularly important for longitudinal care in the hospital, in which patients frequently are cared for by multiple clinical services and experience handoffs. For example, many orthopedic surgery patients with significant medical comorbidity are managed postoperatively by hospitalist physicians after initial surgical care.
Second, our study aligns well with the cardiac risk-stratification literature more broadly. For example, the patient characteristics and clinical variables most associated with cardiovascular complications were age, history of ischemic heart disease, American Society of Anesthesiologists physical status, use of intraoperative sodium bicarbonate or vasopressors, lowest intraoperative heart rate measured, and lowest intraoperative mean arterial pressure measured. While many of these variables overlap with those included in the RCRI model, others (such as American Society of Anesthesiologists physical status) are not included in RCRI but have been shown to be important in risk prediction in other studies using different data variables.6,25,26
Third, we illustrated a clinical application of this model in identifying patients at low risk of cardiovascular complications, although benefit may extend to other patients as well. This is particularly germane to clinicians who frequently manage patients in the postsurgical or postprocedural setting. Moreover, the clinical relevance to these clinicians is underscored by the lack of consensus among professional societies across Europe, Canada, and the United States about which subgroups of patients undergoing noncardiac surgery should receive postoperative cardiac biomarker surveillance testing in the 48 to 72 hours after surgery.6-9 This may be in part caused by differences in clinical objectives. For example, the CCS guidelines in part aim to detect myocardial injury after noncardiac surgery (MINS) up to 30 days after surgery, which may be more sensitive to myocardial injury but less strongly associated with outcomes like MACE. The results of this study suggest that adopting such risk factor–based testing would likely lead to additional testing of low risk patients, which may represent low value surveillance tests. For example, there were 2,257 patients without postoperative cardiac biomarker testing in our data who would have been categorized as high risk by risk factor guidelines and therefore recommended to receive at least one postoperative cardiac biomarker surveillance test but were classified as low-risk individuals using a predicted probability of MACE less than 0.18% per our perioperative risk stratification model (Appendix Table 4). If each of these patients received one troponin biomarker test, the associated cost increase would be $372,405 (using the $165 cost per test reported at our institution). These costs would multiply if daily surveillance troponin biomarker tests were ordered for 48 to 72 hours after surgery, as recommended by the risk factor–based testing guidelines. This would be a departure from testing among patients using clinician discretion that may avoid low-value testing.
Applying the perioperative model developed in this paper to clinical practice still requires several steps. The technical aspects of finding a parsimonious model that can be implemented in the EHR is likely quite straightforward. Our preliminary analysis illustrates that doing so will not require accessing large numbers of intraoperative variables. Perhaps more important steps include prospective validation of the safety, usability, and clinical benefit of such an algorithm-based risk score.27
The study has several limitations. First, it was an observational study using EHR data subject to missingness and data quality issues that may have persisted despite our methods. Furthermore, EHR data is not generated randomly, and unmeasured variables observed by clinicians but not by researchers could confound the results. However, our approach used the statistical model to examine risk, not causal inference. Second, this is a single institution study and the availability of EHR data, as well as practice patterns, may vary at other institutions. Furthermore, it is possible that performance of the RCRI score, the model fitting RCRI classification of high vs low risk on the sample data, and our model’s performance may not generalize to other clinical settings. However, we utilized data from multiple hospitals within a health system with different surgery and anesthesia groups and providers, and a similar AUC was reported for RCRI in original validation study.6 Third, our follow up period was limited to the hospital setting and we do not capture longitudinal outcomes, such as 30-day MACE. This may impact the ability to risk stratify for other important longer-term outcomes, limit clinical utility, and hinder comparability to other studies. Fourth, results may vary for other important cardiovascular outcomes that may be more sensitive to myocardial injury, such as MINS. Fifth, we used a limited number of modeling strategies.
CONCLUSION
Addition of intraoperative data to preoperative data improves prediction of cardiovascular complications after noncardiac surgery. Improving the identification of patients at low risk for such complications could potentially be applied to reduce unnecessary postoperative cardiac biomarker testing after noncardiac surgery, but it will require further validation in prospective clinical settings.
Disclosures
Dr Navathe reports grants from the following entities: Hawaii Medical Service Association, Anthem Public Policy Institute, Commonwealth Fund, Oscar Health, Cigna Corporation, Robert Wood Johnson Foundation, Donaghue Foundation, Pennsylvania Department of Health, Ochsner Health System, United Healthcare, Blue Cross Blue Shield of NC, Blue Shield of CA; personal fees from the following: Navvis Healthcare, Agathos, Inc, Navahealth, YNHHSC/CORE, Maine Health Accountable Care Organization, Maine Department of Health and Human Services, National University Health System - Singapore, Ministry of Health - Singapore, Social Security Administration - France, Elsevier Press, Medicare Payment Advisory Commission, Cleveland Clinic, Embedded Healthcare; and other support from Integrated Services, Inc, outside of the submitted work. Dr Volpp reports grants from Humana during the conduct of the study; grants from Hawaii Medical Services Agency, Discovery (South Africa), Merck, Weight Watchers, and CVS outside of the submitted work; he has received consulting income from CVS and VALHealth and is a principal in VALHealth, a behavioral economics consulting firm. Dr Holmes receives funding from the Pennsylvania Department of Health, US Public Health Service, and the Cardiovascular Medicine Research and Education Foundation. All other authors declare no conflicts of interest.
Prior Presentations
2019 Academy Health Annual Research Meeting, Poster Abstract Presentation, June 2 to June 4, 2019, Washington, DC.
Funding
This project was funded, in part, under a grant with the Pennsylvania Department of Health. This research was independent from the funder. The funder had no role in the study design; in the collection, analysis, and interpretation of data; in the writing of the report; or in the decision to submit the article for publication. The department specifically disclaims responsibility for any analyses, interpretations, or conclusions.
1. National Center for Health Statistics. National Hospital Discharge Survey: 2010 Table, Number of all-listed procedures for discharges from short-stay hospitals, by procedure category and age: United States, 2010. Centers for Disease Control and Prevention; 2010. Accessed November 11, 2018. https://www.cdc.gov/nchs/data/nhds/4procedures/2010pro4_numberprocedureage.pdf
2. Devereaux PJ, Goldman L, Cook DJ, Gilbert K, Leslie K, Guyatt GH. Perioperative cardiac events in patients undergoing noncardiac surgery: a review of the magnitude of the problem, the pathophysiology of the events and methods to estimate and communicate risk. CMAJ. 2005;173(6):627-634. https://doi.org/10.1503/cmaj.050011
3. Charlson M, Peterson J, Szatrowski TP, MacKenzie R, Gold J. Long-term prognosis after peri-operative cardiac complications. J Clin Epidemiol. 1994;47(12):1389-1400. https://doi.org/10.1016/0895-4356(94)90083-3
4. Devereaux PJ, Sessler DI. Cardiac complications in patients undergoing major noncardiac surgery. N Engl J Med. 2015;373(23):2258-2269. https://doi.org/10.1056/nejmra1502824
5. Sprung J, Warner ME, Contreras MG, et al. Predictors of survival following cardiac arrest in patients undergoing noncardiac surgery: a study of 518,294 patients at a tertiary referral center. Anesthesiology. 2003;99(2):259-269. https://doi.org/10.1097/00000542-200308000-00006
6. Lee TH, Marcantonio ER, Mangione CM, et al. Derivation and prospective validation of a simple index for prediction of cardiac risk of major noncardiac surgery. Circulation. 1999;100(10):1043-1049. https://doi.org/10.1161/01.cir.100.10.1043
7. Duceppe E, Parlow J, MacDonald P, et al. Canadian Cardiovascular Society guidelines on perioperative cardiac risk assessment and management for patients who undergo noncardiac surgery. Can J Cardiol. 2017;33(1):17-32. https://doi.org/10.1016/j.cjca.2016.09.008
8. Fleisher LA, Fleischmann KE, Auerbach AD, et al. 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines. J Am Coll Cardiol. 2014;64(22):e77-e137. https://doi.org/10.1016/j.jacc.2014.07.944
9. Kristensen SD, Knuuti J, Saraste A, et al. 2014 ESC/ESA guidelines on non-cardiac surgery: cardiovascular assessment and management: The Joint Task Force on non-cardiac surgery: cardiovascular assessment and management of the European Society of Cardiology (ESC) and the European Society of Anaesthesiology (ESA). Euro Heart J. 2014;35(35):2383-2431. https://doi.org/10.1093/eurheartj/ehu282
10. Collins GS, Reitsma JB, Altman DG, Moons KG. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement. Ann Intern Med. 2015;162(1):55-63. https://doi.org/10.7326/m14-0697
11. Freundlich RE, Kheterpal S. Perioperative effectiveness research using large databases. Best Pract Res Clin Anaesthesiol. 2011;25(4):489-498. https://doi.org/10.1016/j.bpa.2011.08.008
12. CPT® (Current Procedural Terminology). American Medical Association. 2018. Accessed November 11, 2018. https://www.ama-assn.org/practice-management/cpt-current-procedural-terminology
13. Surgery Flag Software for ICD-9-CM. AHRQ Healthcare Cost and Utilization Project; 2017. Accessed November 11, 2018. https://www.hcup-us.ahrq.gov/toolssoftware/surgflags/surgeryflags.jsp
14. Hastie T, Tibshirani R, Friedman J. The Elements of Statistical Learning: Data Mining, Inference, and Prediction. 2nd ed. Springer; 2009. https://www.springer.com/gp/book/9780387848570
15. Bucy R, Hanisko KA, Ewing LA, et al. Abstract 281: Validity of in-hospital cardiac arrest ICD-9-CM codes in veterans. Circ Cardiovasc Qual Outcomes. 2015;8(suppl_2):A281-A281.
16. Institute of Medicine; Board on Health Sciences Policy; Committee on the Treatment of Cardiac Arrest: Current Status and Future Directions. Graham R, McCoy MA, Schultz AM, eds. Strategies to Improve Cardiac Arrest Survival: A Time to Act. The National Academies Press; 2015. https://doi.org/10.17226/21723
17. Pladevall M, Goff DC, Nichaman MZ, et al. An assessment of the validity of ICD Code 410 to identify hospital admissions for myocardial infarction: The Corpus Christi Heart Project. Int J Epidemiol. 1996;25(5):948-952. https://doi.org/10.1093/ije/25.5.948
18. Elixhauser A, Steiner C, Harris DR, Coffey RM. Comorbidity measures for use with administrative data. Med Care. 1998;36(1):8-27. https://doi.org/10.1097/00005650-199801000-00004
19. Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130-1139. https://doi.org/10.1097/01.mlr.0000182534.19832.83
20. Keats AS. The ASA classification of physical status--a recapitulation. Anesthesiology. 1978;49(4):233-236. https://doi.org/10.1097/00000542-197810000-00001
21. Schwarze ML, Barnato AE, Rathouz PJ, et al. Development of a list of high-risk operations for patients 65 years and older. JAMA Surg. 2015;150(4):325-331. https://doi.org/10.1001/jamasurg.2014.1819
22. VISION Pilot Study Investigators, Devereaux PJ, Bradley D, et al. An international prospective cohort study evaluating major vascular complications among patients undergoing noncardiac surgery: the VISION Pilot Study. Open Med. 2011;5(4):e193-e200.
23. DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44(3):837-845.
24. Norton EC, Dowd BE, Maciejewski ML. Marginal effects-quantifying the effect of changes in risk factors in logistic regression models. JAMA. 2019;321(13):1304‐1305. https://doi.org/10.1001/jama.2019.1954
25. Bilimoria KY, Liu Y, Paruch JL, et al. Development and evaluation of the universal ACS NSQIP surgical risk calculator: a decision aid and informed consent tool for patients and surgeons. J Am Coll Surg. 2013;217(5):833-842. https://doi.org/10.1016/j.jamcollsurg.2013.07.385
26. Gawande AA, Kwaan MR, Regenbogen SE, Lipsitz SA, Zinner MJ. An Apgar score for surgery. J Am Coll Surg. 2007;204(2):201-208. https://doi.org/10.1016/j.jamcollsurg.2006.11.011
27. Parikh RB, Obermeyer Z, Navathe AS. Regulation of predictive analytics in medicine. Science. 2019;363(6429):810-812. https://doi.org/10.1126/science.aaw0029
1. National Center for Health Statistics. National Hospital Discharge Survey: 2010 Table, Number of all-listed procedures for discharges from short-stay hospitals, by procedure category and age: United States, 2010. Centers for Disease Control and Prevention; 2010. Accessed November 11, 2018. https://www.cdc.gov/nchs/data/nhds/4procedures/2010pro4_numberprocedureage.pdf
2. Devereaux PJ, Goldman L, Cook DJ, Gilbert K, Leslie K, Guyatt GH. Perioperative cardiac events in patients undergoing noncardiac surgery: a review of the magnitude of the problem, the pathophysiology of the events and methods to estimate and communicate risk. CMAJ. 2005;173(6):627-634. https://doi.org/10.1503/cmaj.050011
3. Charlson M, Peterson J, Szatrowski TP, MacKenzie R, Gold J. Long-term prognosis after peri-operative cardiac complications. J Clin Epidemiol. 1994;47(12):1389-1400. https://doi.org/10.1016/0895-4356(94)90083-3
4. Devereaux PJ, Sessler DI. Cardiac complications in patients undergoing major noncardiac surgery. N Engl J Med. 2015;373(23):2258-2269. https://doi.org/10.1056/nejmra1502824
5. Sprung J, Warner ME, Contreras MG, et al. Predictors of survival following cardiac arrest in patients undergoing noncardiac surgery: a study of 518,294 patients at a tertiary referral center. Anesthesiology. 2003;99(2):259-269. https://doi.org/10.1097/00000542-200308000-00006
6. Lee TH, Marcantonio ER, Mangione CM, et al. Derivation and prospective validation of a simple index for prediction of cardiac risk of major noncardiac surgery. Circulation. 1999;100(10):1043-1049. https://doi.org/10.1161/01.cir.100.10.1043
7. Duceppe E, Parlow J, MacDonald P, et al. Canadian Cardiovascular Society guidelines on perioperative cardiac risk assessment and management for patients who undergo noncardiac surgery. Can J Cardiol. 2017;33(1):17-32. https://doi.org/10.1016/j.cjca.2016.09.008
8. Fleisher LA, Fleischmann KE, Auerbach AD, et al. 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines. J Am Coll Cardiol. 2014;64(22):e77-e137. https://doi.org/10.1016/j.jacc.2014.07.944
9. Kristensen SD, Knuuti J, Saraste A, et al. 2014 ESC/ESA guidelines on non-cardiac surgery: cardiovascular assessment and management: The Joint Task Force on non-cardiac surgery: cardiovascular assessment and management of the European Society of Cardiology (ESC) and the European Society of Anaesthesiology (ESA). Euro Heart J. 2014;35(35):2383-2431. https://doi.org/10.1093/eurheartj/ehu282
10. Collins GS, Reitsma JB, Altman DG, Moons KG. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement. Ann Intern Med. 2015;162(1):55-63. https://doi.org/10.7326/m14-0697
11. Freundlich RE, Kheterpal S. Perioperative effectiveness research using large databases. Best Pract Res Clin Anaesthesiol. 2011;25(4):489-498. https://doi.org/10.1016/j.bpa.2011.08.008
12. CPT® (Current Procedural Terminology). American Medical Association. 2018. Accessed November 11, 2018. https://www.ama-assn.org/practice-management/cpt-current-procedural-terminology
13. Surgery Flag Software for ICD-9-CM. AHRQ Healthcare Cost and Utilization Project; 2017. Accessed November 11, 2018. https://www.hcup-us.ahrq.gov/toolssoftware/surgflags/surgeryflags.jsp
14. Hastie T, Tibshirani R, Friedman J. The Elements of Statistical Learning: Data Mining, Inference, and Prediction. 2nd ed. Springer; 2009. https://www.springer.com/gp/book/9780387848570
15. Bucy R, Hanisko KA, Ewing LA, et al. Abstract 281: Validity of in-hospital cardiac arrest ICD-9-CM codes in veterans. Circ Cardiovasc Qual Outcomes. 2015;8(suppl_2):A281-A281.
16. Institute of Medicine; Board on Health Sciences Policy; Committee on the Treatment of Cardiac Arrest: Current Status and Future Directions. Graham R, McCoy MA, Schultz AM, eds. Strategies to Improve Cardiac Arrest Survival: A Time to Act. The National Academies Press; 2015. https://doi.org/10.17226/21723
17. Pladevall M, Goff DC, Nichaman MZ, et al. An assessment of the validity of ICD Code 410 to identify hospital admissions for myocardial infarction: The Corpus Christi Heart Project. Int J Epidemiol. 1996;25(5):948-952. https://doi.org/10.1093/ije/25.5.948
18. Elixhauser A, Steiner C, Harris DR, Coffey RM. Comorbidity measures for use with administrative data. Med Care. 1998;36(1):8-27. https://doi.org/10.1097/00005650-199801000-00004
19. Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130-1139. https://doi.org/10.1097/01.mlr.0000182534.19832.83
20. Keats AS. The ASA classification of physical status--a recapitulation. Anesthesiology. 1978;49(4):233-236. https://doi.org/10.1097/00000542-197810000-00001
21. Schwarze ML, Barnato AE, Rathouz PJ, et al. Development of a list of high-risk operations for patients 65 years and older. JAMA Surg. 2015;150(4):325-331. https://doi.org/10.1001/jamasurg.2014.1819
22. VISION Pilot Study Investigators, Devereaux PJ, Bradley D, et al. An international prospective cohort study evaluating major vascular complications among patients undergoing noncardiac surgery: the VISION Pilot Study. Open Med. 2011;5(4):e193-e200.
23. DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44(3):837-845.
24. Norton EC, Dowd BE, Maciejewski ML. Marginal effects-quantifying the effect of changes in risk factors in logistic regression models. JAMA. 2019;321(13):1304‐1305. https://doi.org/10.1001/jama.2019.1954
25. Bilimoria KY, Liu Y, Paruch JL, et al. Development and evaluation of the universal ACS NSQIP surgical risk calculator: a decision aid and informed consent tool for patients and surgeons. J Am Coll Surg. 2013;217(5):833-842. https://doi.org/10.1016/j.jamcollsurg.2013.07.385
26. Gawande AA, Kwaan MR, Regenbogen SE, Lipsitz SA, Zinner MJ. An Apgar score for surgery. J Am Coll Surg. 2007;204(2):201-208. https://doi.org/10.1016/j.jamcollsurg.2006.11.011
27. Parikh RB, Obermeyer Z, Navathe AS. Regulation of predictive analytics in medicine. Science. 2019;363(6429):810-812. https://doi.org/10.1126/science.aaw0029
© 2020 Society of Hospital Medicine