User login
Maternal BMI and Eating Disorders Tied to Mental Health in Kids
TOPLINE:
Children of mothers who had obesity or eating disorders before or during pregnancy may face higher risks for neurodevelopmental and psychiatric disorders.
METHODOLOGY:
- Researchers conducted a population-based cohort study to investigate the association of maternal eating disorders and high prepregnancy body mass index (BMI) with psychiatric disorder and neurodevelopmental diagnoses in offspring.
- They used Finnish national registers to assess all live births from 2004 through 2014, with follow-up until 2021.
- Data of 392,098 mothers (mean age, 30.15 years) and 649,956 offspring (48.86% girls) were included.
- Maternal eating disorders and prepregnancy BMI were the main exposures, with 1.60% of mothers having a history of eating disorders; 5.89% were underweight and 53.13% had obesity.
- Diagnoses of children were identified and grouped by ICD-10 codes of mental, behavioral, and neurodevelopmental disorders, mood disorders, anxiety disorders, sleep disorders, attention-deficit/hyperactivity disorder, and conduct disorders, among several others.
TAKEAWAY:
- From birth until 7-17 years of age, 16.43% of offspring were diagnosed with a neurodevelopmental or psychiatric disorder.
- Maternal eating disorders were associated with psychiatric disorders in the offspring, with the largest effect sizes observed for sleep disorders (hazard ratio [HR], 2.36) and social functioning and tic disorders (HR, 2.18; P < .001 for both).
- The offspring of mothers with severe prepregnancy obesity had a more than twofold increased risk for intellectual disabilities (HR, 2.04; 95% CI, 1.83-2.28); being underweight before pregnancy was also linked to many psychiatric disorders in offspring.
- The occurrence of adverse birth outcomes along with maternal eating disorders or high BMI further increased the risk for neurodevelopmental and psychiatric disorders in the offspring.
IN PRACTICE:
“The findings underline the risk of offspring mental illness associated with maternal eating disorders and prepregnancy BMI and suggest the need to consider these exposures clinically to help prevent offspring mental illness,” the authors wrote.
SOURCE:
This study was led by Ida A.K. Nilsson, PhD, of the Department of Molecular Medicine and Surgery at the Karolinska Institutet in Stockholm, Sweden, and was published online in JAMA Network Open.
LIMITATIONS:
A limitation of the study was the relatively short follow-up time, which restricted the inclusion of late-onset psychiatric disorder diagnoses, such as schizophrenia spectrum disorders. Paternal data and genetic information, which may have influenced the interpretation of the data, were not available. Another potential bias was that mothers with eating disorders may have been more perceptive to their child’s eating behavior, leading to greater access to care and diagnosis for these children.
DISCLOSURES:
This work was supported by the Swedish Research Council, the regional agreement on medical training and clinical research between Region Stockholm and the Karolinska Institutet, the Swedish Brain Foundation, and other sources. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Children of mothers who had obesity or eating disorders before or during pregnancy may face higher risks for neurodevelopmental and psychiatric disorders.
METHODOLOGY:
- Researchers conducted a population-based cohort study to investigate the association of maternal eating disorders and high prepregnancy body mass index (BMI) with psychiatric disorder and neurodevelopmental diagnoses in offspring.
- They used Finnish national registers to assess all live births from 2004 through 2014, with follow-up until 2021.
- Data of 392,098 mothers (mean age, 30.15 years) and 649,956 offspring (48.86% girls) were included.
- Maternal eating disorders and prepregnancy BMI were the main exposures, with 1.60% of mothers having a history of eating disorders; 5.89% were underweight and 53.13% had obesity.
- Diagnoses of children were identified and grouped by ICD-10 codes of mental, behavioral, and neurodevelopmental disorders, mood disorders, anxiety disorders, sleep disorders, attention-deficit/hyperactivity disorder, and conduct disorders, among several others.
TAKEAWAY:
- From birth until 7-17 years of age, 16.43% of offspring were diagnosed with a neurodevelopmental or psychiatric disorder.
- Maternal eating disorders were associated with psychiatric disorders in the offspring, with the largest effect sizes observed for sleep disorders (hazard ratio [HR], 2.36) and social functioning and tic disorders (HR, 2.18; P < .001 for both).
- The offspring of mothers with severe prepregnancy obesity had a more than twofold increased risk for intellectual disabilities (HR, 2.04; 95% CI, 1.83-2.28); being underweight before pregnancy was also linked to many psychiatric disorders in offspring.
- The occurrence of adverse birth outcomes along with maternal eating disorders or high BMI further increased the risk for neurodevelopmental and psychiatric disorders in the offspring.
IN PRACTICE:
“The findings underline the risk of offspring mental illness associated with maternal eating disorders and prepregnancy BMI and suggest the need to consider these exposures clinically to help prevent offspring mental illness,” the authors wrote.
SOURCE:
This study was led by Ida A.K. Nilsson, PhD, of the Department of Molecular Medicine and Surgery at the Karolinska Institutet in Stockholm, Sweden, and was published online in JAMA Network Open.
LIMITATIONS:
A limitation of the study was the relatively short follow-up time, which restricted the inclusion of late-onset psychiatric disorder diagnoses, such as schizophrenia spectrum disorders. Paternal data and genetic information, which may have influenced the interpretation of the data, were not available. Another potential bias was that mothers with eating disorders may have been more perceptive to their child’s eating behavior, leading to greater access to care and diagnosis for these children.
DISCLOSURES:
This work was supported by the Swedish Research Council, the regional agreement on medical training and clinical research between Region Stockholm and the Karolinska Institutet, the Swedish Brain Foundation, and other sources. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Children of mothers who had obesity or eating disorders before or during pregnancy may face higher risks for neurodevelopmental and psychiatric disorders.
METHODOLOGY:
- Researchers conducted a population-based cohort study to investigate the association of maternal eating disorders and high prepregnancy body mass index (BMI) with psychiatric disorder and neurodevelopmental diagnoses in offspring.
- They used Finnish national registers to assess all live births from 2004 through 2014, with follow-up until 2021.
- Data of 392,098 mothers (mean age, 30.15 years) and 649,956 offspring (48.86% girls) were included.
- Maternal eating disorders and prepregnancy BMI were the main exposures, with 1.60% of mothers having a history of eating disorders; 5.89% were underweight and 53.13% had obesity.
- Diagnoses of children were identified and grouped by ICD-10 codes of mental, behavioral, and neurodevelopmental disorders, mood disorders, anxiety disorders, sleep disorders, attention-deficit/hyperactivity disorder, and conduct disorders, among several others.
TAKEAWAY:
- From birth until 7-17 years of age, 16.43% of offspring were diagnosed with a neurodevelopmental or psychiatric disorder.
- Maternal eating disorders were associated with psychiatric disorders in the offspring, with the largest effect sizes observed for sleep disorders (hazard ratio [HR], 2.36) and social functioning and tic disorders (HR, 2.18; P < .001 for both).
- The offspring of mothers with severe prepregnancy obesity had a more than twofold increased risk for intellectual disabilities (HR, 2.04; 95% CI, 1.83-2.28); being underweight before pregnancy was also linked to many psychiatric disorders in offspring.
- The occurrence of adverse birth outcomes along with maternal eating disorders or high BMI further increased the risk for neurodevelopmental and psychiatric disorders in the offspring.
IN PRACTICE:
“The findings underline the risk of offspring mental illness associated with maternal eating disorders and prepregnancy BMI and suggest the need to consider these exposures clinically to help prevent offspring mental illness,” the authors wrote.
SOURCE:
This study was led by Ida A.K. Nilsson, PhD, of the Department of Molecular Medicine and Surgery at the Karolinska Institutet in Stockholm, Sweden, and was published online in JAMA Network Open.
LIMITATIONS:
A limitation of the study was the relatively short follow-up time, which restricted the inclusion of late-onset psychiatric disorder diagnoses, such as schizophrenia spectrum disorders. Paternal data and genetic information, which may have influenced the interpretation of the data, were not available. Another potential bias was that mothers with eating disorders may have been more perceptive to their child’s eating behavior, leading to greater access to care and diagnosis for these children.
DISCLOSURES:
This work was supported by the Swedish Research Council, the regional agreement on medical training and clinical research between Region Stockholm and the Karolinska Institutet, the Swedish Brain Foundation, and other sources. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Semiannual Time Changes Linked to Accidents, Heart Attacks
As people turn their clocks back an hour on November 3 to mark the end of daylight saving time and return to standard time, they should remain aware of their sleep health and of potential risks associated with shifts in sleep patterns, according to a University of Calgary psychology professor who researches circadian cycles.
said Michael Antle, PhD, head of the psychology department and member of the Hotchkiss Brain Institute at the Cumming School of Medicine, University of Calgary, Alberta, Canada.
In an interview, Antle explained the science behind the health risks associated with time changes, offered tips to prepare for the shift, and discussed scientists’ suggestion to move to year-round standard time. This interview has been condensed and edited for clarity.
Why is it important to pay attention to circadian rhythms?
Circadian rhythms are patterns of physiologic and behavioral changes that affect everything inside the body and everything we do, including when hormones are secreted, digestive juices are ready to digest, and growth hormones are released at night. The body is a carefully coordinated orchestra, and everything has to happen at the right time.
When we start messing with those rhythms, that’s when states of disease start coming on and we don’t feel well. You’ve probably experienced it — when you try to stay up late, eat at the wrong times, or have jet lag. Flying across one or two time zones is usually tolerable, but if you fly across the world, it can be profound and make you feel bad, even up to a week. Similar shifts happen with the time changes.
How do the time changes affect health risks?
The wintertime change is generally more tolerable, and you’ll hear people talk about “gaining an hour” of sleep. It’s better than that, because we’re realigning our social clocks — such as our work schedules and school schedules — with daylight. We tend to go to bed relative to the sun but wake up based on when our boss says to be at our desk, so an earlier sunset helps us to fall asleep earlier and is healthier for our body.
In the spring, the opposite happens, and the time change affects us much more than just one bad night of sleep. For some people, it can feel like losing an hour of sleep every day for weeks, and that abrupt change can lead to car accidents, workplace injuries, heart attacks, and strokes. Our body experiences extra strain when we’re not awake and ready for the day.
What does your research show?
Most of my work focuses on preclinical models to understand what’s going on in the brain and body. Because we can’t study this ethically in humans, we learn a lot from animal models, especially mice. In a recent study looking at mild circadian disruption — where we raised mice on days that were about 75 minutes shorter — we saw they started developing diabetes, heart disease, and insulin resistance within in a few months, or about the time they were a young adult.
Oftentimes, people think about their sleep rhythm as an arbitrary choice, but in fact, it does affect your health. We know that if your human circadian clock runs slow, morning light can help fix that and reset it, whereas evening light moves us in the other direction and makes it harder to get up in the morning.
Some people want to switch to one year-round time. What do you advocate?
In most cases, the standard time (or winter time) is the more natural time that fits better with our body cycle. If we follow a time where we get up before sunrise or have a later sunset, then it’s linked to more social jet lag, where people are less attentive at work, don’t learn as well at school, and have more accidents.
Instead of picking what sounds good or chasing the name — such as “daylight saving time” — we need to think about the right time for us and our circadian clock. Some places, such as Maine in the United States, would actually fit better with the Atlantic time zone or the Maritime provinces in Canada, whereas some parts of Alberta are geographically west of Los Angeles based on longitude and would fit better with the Pacific time zone. Sticking with a year-round daylight saving time in some cities in Alberta would mean people wouldn’t see the sun until 10:30 AM in the winter, which is really late and could affect activities such as skiing and hockey.
The Canadian Society for Chronobiology advocates for year-round standard time to align our social clocks with our biological clocks. Sleep and circadian rhythm experts in the US and globally have issued similar position statements.
What tips do you suggest to help people adjust their circadian clocks in November?
For people who know their bodies and that it will affect them more, give yourself extra time. If your schedule permits, plan ahead and change your clocks sooner, especially if you’re able to do so over the weekend. Don’t rush around while tired — rushing when you’re not ready leads to those increased accidents on the road or on the job. Know that the sun will still be mismatched for a bit and your body clock will take time to adjust, so you might feel out of sorts for a few days.
Antle reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
As people turn their clocks back an hour on November 3 to mark the end of daylight saving time and return to standard time, they should remain aware of their sleep health and of potential risks associated with shifts in sleep patterns, according to a University of Calgary psychology professor who researches circadian cycles.
said Michael Antle, PhD, head of the psychology department and member of the Hotchkiss Brain Institute at the Cumming School of Medicine, University of Calgary, Alberta, Canada.
In an interview, Antle explained the science behind the health risks associated with time changes, offered tips to prepare for the shift, and discussed scientists’ suggestion to move to year-round standard time. This interview has been condensed and edited for clarity.
Why is it important to pay attention to circadian rhythms?
Circadian rhythms are patterns of physiologic and behavioral changes that affect everything inside the body and everything we do, including when hormones are secreted, digestive juices are ready to digest, and growth hormones are released at night. The body is a carefully coordinated orchestra, and everything has to happen at the right time.
When we start messing with those rhythms, that’s when states of disease start coming on and we don’t feel well. You’ve probably experienced it — when you try to stay up late, eat at the wrong times, or have jet lag. Flying across one or two time zones is usually tolerable, but if you fly across the world, it can be profound and make you feel bad, even up to a week. Similar shifts happen with the time changes.
How do the time changes affect health risks?
The wintertime change is generally more tolerable, and you’ll hear people talk about “gaining an hour” of sleep. It’s better than that, because we’re realigning our social clocks — such as our work schedules and school schedules — with daylight. We tend to go to bed relative to the sun but wake up based on when our boss says to be at our desk, so an earlier sunset helps us to fall asleep earlier and is healthier for our body.
In the spring, the opposite happens, and the time change affects us much more than just one bad night of sleep. For some people, it can feel like losing an hour of sleep every day for weeks, and that abrupt change can lead to car accidents, workplace injuries, heart attacks, and strokes. Our body experiences extra strain when we’re not awake and ready for the day.
What does your research show?
Most of my work focuses on preclinical models to understand what’s going on in the brain and body. Because we can’t study this ethically in humans, we learn a lot from animal models, especially mice. In a recent study looking at mild circadian disruption — where we raised mice on days that were about 75 minutes shorter — we saw they started developing diabetes, heart disease, and insulin resistance within in a few months, or about the time they were a young adult.
Oftentimes, people think about their sleep rhythm as an arbitrary choice, but in fact, it does affect your health. We know that if your human circadian clock runs slow, morning light can help fix that and reset it, whereas evening light moves us in the other direction and makes it harder to get up in the morning.
Some people want to switch to one year-round time. What do you advocate?
In most cases, the standard time (or winter time) is the more natural time that fits better with our body cycle. If we follow a time where we get up before sunrise or have a later sunset, then it’s linked to more social jet lag, where people are less attentive at work, don’t learn as well at school, and have more accidents.
Instead of picking what sounds good or chasing the name — such as “daylight saving time” — we need to think about the right time for us and our circadian clock. Some places, such as Maine in the United States, would actually fit better with the Atlantic time zone or the Maritime provinces in Canada, whereas some parts of Alberta are geographically west of Los Angeles based on longitude and would fit better with the Pacific time zone. Sticking with a year-round daylight saving time in some cities in Alberta would mean people wouldn’t see the sun until 10:30 AM in the winter, which is really late and could affect activities such as skiing and hockey.
The Canadian Society for Chronobiology advocates for year-round standard time to align our social clocks with our biological clocks. Sleep and circadian rhythm experts in the US and globally have issued similar position statements.
What tips do you suggest to help people adjust their circadian clocks in November?
For people who know their bodies and that it will affect them more, give yourself extra time. If your schedule permits, plan ahead and change your clocks sooner, especially if you’re able to do so over the weekend. Don’t rush around while tired — rushing when you’re not ready leads to those increased accidents on the road or on the job. Know that the sun will still be mismatched for a bit and your body clock will take time to adjust, so you might feel out of sorts for a few days.
Antle reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
As people turn their clocks back an hour on November 3 to mark the end of daylight saving time and return to standard time, they should remain aware of their sleep health and of potential risks associated with shifts in sleep patterns, according to a University of Calgary psychology professor who researches circadian cycles.
said Michael Antle, PhD, head of the psychology department and member of the Hotchkiss Brain Institute at the Cumming School of Medicine, University of Calgary, Alberta, Canada.
In an interview, Antle explained the science behind the health risks associated with time changes, offered tips to prepare for the shift, and discussed scientists’ suggestion to move to year-round standard time. This interview has been condensed and edited for clarity.
Why is it important to pay attention to circadian rhythms?
Circadian rhythms are patterns of physiologic and behavioral changes that affect everything inside the body and everything we do, including when hormones are secreted, digestive juices are ready to digest, and growth hormones are released at night. The body is a carefully coordinated orchestra, and everything has to happen at the right time.
When we start messing with those rhythms, that’s when states of disease start coming on and we don’t feel well. You’ve probably experienced it — when you try to stay up late, eat at the wrong times, or have jet lag. Flying across one or two time zones is usually tolerable, but if you fly across the world, it can be profound and make you feel bad, even up to a week. Similar shifts happen with the time changes.
How do the time changes affect health risks?
The wintertime change is generally more tolerable, and you’ll hear people talk about “gaining an hour” of sleep. It’s better than that, because we’re realigning our social clocks — such as our work schedules and school schedules — with daylight. We tend to go to bed relative to the sun but wake up based on when our boss says to be at our desk, so an earlier sunset helps us to fall asleep earlier and is healthier for our body.
In the spring, the opposite happens, and the time change affects us much more than just one bad night of sleep. For some people, it can feel like losing an hour of sleep every day for weeks, and that abrupt change can lead to car accidents, workplace injuries, heart attacks, and strokes. Our body experiences extra strain when we’re not awake and ready for the day.
What does your research show?
Most of my work focuses on preclinical models to understand what’s going on in the brain and body. Because we can’t study this ethically in humans, we learn a lot from animal models, especially mice. In a recent study looking at mild circadian disruption — where we raised mice on days that were about 75 minutes shorter — we saw they started developing diabetes, heart disease, and insulin resistance within in a few months, or about the time they were a young adult.
Oftentimes, people think about their sleep rhythm as an arbitrary choice, but in fact, it does affect your health. We know that if your human circadian clock runs slow, morning light can help fix that and reset it, whereas evening light moves us in the other direction and makes it harder to get up in the morning.
Some people want to switch to one year-round time. What do you advocate?
In most cases, the standard time (or winter time) is the more natural time that fits better with our body cycle. If we follow a time where we get up before sunrise or have a later sunset, then it’s linked to more social jet lag, where people are less attentive at work, don’t learn as well at school, and have more accidents.
Instead of picking what sounds good or chasing the name — such as “daylight saving time” — we need to think about the right time for us and our circadian clock. Some places, such as Maine in the United States, would actually fit better with the Atlantic time zone or the Maritime provinces in Canada, whereas some parts of Alberta are geographically west of Los Angeles based on longitude and would fit better with the Pacific time zone. Sticking with a year-round daylight saving time in some cities in Alberta would mean people wouldn’t see the sun until 10:30 AM in the winter, which is really late and could affect activities such as skiing and hockey.
The Canadian Society for Chronobiology advocates for year-round standard time to align our social clocks with our biological clocks. Sleep and circadian rhythm experts in the US and globally have issued similar position statements.
What tips do you suggest to help people adjust their circadian clocks in November?
For people who know their bodies and that it will affect them more, give yourself extra time. If your schedule permits, plan ahead and change your clocks sooner, especially if you’re able to do so over the weekend. Don’t rush around while tired — rushing when you’re not ready leads to those increased accidents on the road or on the job. Know that the sun will still be mismatched for a bit and your body clock will take time to adjust, so you might feel out of sorts for a few days.
Antle reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID on the Floor Linked to Outbreaks on Two Hospital Wards
The viral burden of SARS-CoV-2 on floors, even in healthcare worker–only areas, was strongly associated with COVID-19 outbreaks in two acute-care hospitals, according to a new study from Ontario, Canada.
With every 10-fold increase in viral copies, the chance of an impending outbreak of COVID-19 rose 22-fold.
“These data add to the mounting evidence that built environment detection for SARS-CoV-2 may provide an additional layer of monitoring and could help inform local infection prevention and control measures,” they wrote.
The study was published online in Infection Control & Hospital Epidemiology.
Preventing Future Suffering
The current study builds on the researchers’ previous work, which found the same correlation between viral load on floors and COVID outbreaks in long-term care homes.
Currently, the best-known method of environmental surveillance for COVID is wastewater detection. “Swabbing the floors would be another approach to surveillance,” senior author Caroline Nott, MD, infectious disease physician at the Ottawa Hospital, said in an interview.
“We do have environmental surveillance with wastewater, but while this may tell you what’s going on in the city, it doesn’t tell you what is going on in a particular ward of a hospital, for instance,” she added.
Nott and her colleagues believe that swabbing, which is easy and relatively inexpensive, will become another tool to examine the built environment. “Instead of having to close a whole hospital, for example, we could just close one room instead of an entire ward if swabbing showed a high concentration of COVID,” Nott said.
The current study was conducted at two hospitals in Ontario between July 2022 and March 2023. The floors of healthcare worker–only areas on four inpatient adult wards were swabbed. These areas included changing rooms, meeting rooms, staff washrooms, nursing stations, and interdisciplinary team rooms.
SARS-CoV-2 RNA was detected on 537 of 760 floor swabs (71%). The overall positivity rate in the first hospital was 90% (n = 280). In the second hospital, the rate was 60% (n = 480).
Four COVID-19 outbreaks occurred in the first acute care hospital, and seven outbreaks occurred at the second hospital. Outbreaks occurred mostly among hospitalized patients (140 cases), but also in four hospital workers.
COVID-19 still requires vigilance, said Nott. “We weren’t prepared for COVID, and so as a result, many people died or have suffered long-term effects, especially vulnerable people like those being treated in hospital or in long-term care facilities. We want to develop methods to prevent similar suffering in the future, whether it’s a new COVID variant or a different pathogen altogether.”
Changing Surveillance Practice?
“This is a good study,” Steven Rogak, PhD, professor of mechanical engineering at the University of British Columbia (UBC) in Vancouver, Canada, said in an interivew. “The fundamental idea is that respiratory droplets and aerosols will deposit on the floor, and polymerase chain reaction [PCR] tests of swabs will provide a surrogate measurement of what might have been inhaled. There are solid statistics that it worked for the hospitals studied,” said Rogak, who studies aerosols at UBC’s Energy and Aerosols Laboratory. Rogak did not participate in the study.
“The authors note several limitations, including that increased healthcare worker testing may have been triggered by the higher values of PCR counts from the floor swabs. But this doesn’t seem to be a problem to me, because if the floor swabs motivate the hospital to test workers more, and that results in identifying outbreaks sooner, then great,” he said.
“Another limitation is that if the hospital has better HVAC or uses air purifiers, it could remove the most infectious aerosols, but the large droplets that fall quickly to the ground would remain, and this would still result in high PCR counts from floor swabs. In this case, perhaps the floor swabs would be a poorer indication of impending outbreaks,” said Rogak.
Determining the best timing and location for floor swabbing might be challenging and specific to the particular hospital, he added. ”For example, you would not want to take swabs from floors right after they are cleaned. Overall, I think this method deserves further development, and it could become a standard technique, but the details might require refinement for widespread application.”
Adrian Popp, MD, chair of the Infectious Disease Service at Huntington Hospital–Northwell Health in New York, said that, although interesting, the study would not change his current practice.
“I’m going to start testing the environment in different rooms in the hospital, and yes, I might find different amounts of COVID, but what does that mean? If pieces of RNA from COVID are on the floor, the likelihood is that they’re not infectious,” Popp said in an interview.
“Hospital workers do get sick with COVID, and sometimes they are asymptomatic and come to work. Patients may come into the hospital for another reason and be sick with COVID. There are many ways people who work in the hospital, as well as the patients, can get COVID. To me, it means that in that hospital and community there is a lot of COVID, but I can’t tell if there is causation here. Who is giving COVID to whom? What am I supposed to do with the information?”
The study was supported by the Northern Ontario Academic Medicine Association Clinical Innovation Opportunities Fund, the Ottawa Hospital Academic Medical Organization Innovation Fund, and a Canadian Institutes of Health Research Operating Grant. One author was a consultant for ProofDx, a startup company creating a point-of-care diagnostic test for COVID-19, and is an advisor for SIGNAL1, a startup company deploying machine-learning models to improve inpatient care. Nott, Rogak, and Popp reported having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The viral burden of SARS-CoV-2 on floors, even in healthcare worker–only areas, was strongly associated with COVID-19 outbreaks in two acute-care hospitals, according to a new study from Ontario, Canada.
With every 10-fold increase in viral copies, the chance of an impending outbreak of COVID-19 rose 22-fold.
“These data add to the mounting evidence that built environment detection for SARS-CoV-2 may provide an additional layer of monitoring and could help inform local infection prevention and control measures,” they wrote.
The study was published online in Infection Control & Hospital Epidemiology.
Preventing Future Suffering
The current study builds on the researchers’ previous work, which found the same correlation between viral load on floors and COVID outbreaks in long-term care homes.
Currently, the best-known method of environmental surveillance for COVID is wastewater detection. “Swabbing the floors would be another approach to surveillance,” senior author Caroline Nott, MD, infectious disease physician at the Ottawa Hospital, said in an interview.
“We do have environmental surveillance with wastewater, but while this may tell you what’s going on in the city, it doesn’t tell you what is going on in a particular ward of a hospital, for instance,” she added.
Nott and her colleagues believe that swabbing, which is easy and relatively inexpensive, will become another tool to examine the built environment. “Instead of having to close a whole hospital, for example, we could just close one room instead of an entire ward if swabbing showed a high concentration of COVID,” Nott said.
The current study was conducted at two hospitals in Ontario between July 2022 and March 2023. The floors of healthcare worker–only areas on four inpatient adult wards were swabbed. These areas included changing rooms, meeting rooms, staff washrooms, nursing stations, and interdisciplinary team rooms.
SARS-CoV-2 RNA was detected on 537 of 760 floor swabs (71%). The overall positivity rate in the first hospital was 90% (n = 280). In the second hospital, the rate was 60% (n = 480).
Four COVID-19 outbreaks occurred in the first acute care hospital, and seven outbreaks occurred at the second hospital. Outbreaks occurred mostly among hospitalized patients (140 cases), but also in four hospital workers.
COVID-19 still requires vigilance, said Nott. “We weren’t prepared for COVID, and so as a result, many people died or have suffered long-term effects, especially vulnerable people like those being treated in hospital or in long-term care facilities. We want to develop methods to prevent similar suffering in the future, whether it’s a new COVID variant or a different pathogen altogether.”
Changing Surveillance Practice?
“This is a good study,” Steven Rogak, PhD, professor of mechanical engineering at the University of British Columbia (UBC) in Vancouver, Canada, said in an interivew. “The fundamental idea is that respiratory droplets and aerosols will deposit on the floor, and polymerase chain reaction [PCR] tests of swabs will provide a surrogate measurement of what might have been inhaled. There are solid statistics that it worked for the hospitals studied,” said Rogak, who studies aerosols at UBC’s Energy and Aerosols Laboratory. Rogak did not participate in the study.
“The authors note several limitations, including that increased healthcare worker testing may have been triggered by the higher values of PCR counts from the floor swabs. But this doesn’t seem to be a problem to me, because if the floor swabs motivate the hospital to test workers more, and that results in identifying outbreaks sooner, then great,” he said.
“Another limitation is that if the hospital has better HVAC or uses air purifiers, it could remove the most infectious aerosols, but the large droplets that fall quickly to the ground would remain, and this would still result in high PCR counts from floor swabs. In this case, perhaps the floor swabs would be a poorer indication of impending outbreaks,” said Rogak.
Determining the best timing and location for floor swabbing might be challenging and specific to the particular hospital, he added. ”For example, you would not want to take swabs from floors right after they are cleaned. Overall, I think this method deserves further development, and it could become a standard technique, but the details might require refinement for widespread application.”
Adrian Popp, MD, chair of the Infectious Disease Service at Huntington Hospital–Northwell Health in New York, said that, although interesting, the study would not change his current practice.
“I’m going to start testing the environment in different rooms in the hospital, and yes, I might find different amounts of COVID, but what does that mean? If pieces of RNA from COVID are on the floor, the likelihood is that they’re not infectious,” Popp said in an interview.
“Hospital workers do get sick with COVID, and sometimes they are asymptomatic and come to work. Patients may come into the hospital for another reason and be sick with COVID. There are many ways people who work in the hospital, as well as the patients, can get COVID. To me, it means that in that hospital and community there is a lot of COVID, but I can’t tell if there is causation here. Who is giving COVID to whom? What am I supposed to do with the information?”
The study was supported by the Northern Ontario Academic Medicine Association Clinical Innovation Opportunities Fund, the Ottawa Hospital Academic Medical Organization Innovation Fund, and a Canadian Institutes of Health Research Operating Grant. One author was a consultant for ProofDx, a startup company creating a point-of-care diagnostic test for COVID-19, and is an advisor for SIGNAL1, a startup company deploying machine-learning models to improve inpatient care. Nott, Rogak, and Popp reported having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The viral burden of SARS-CoV-2 on floors, even in healthcare worker–only areas, was strongly associated with COVID-19 outbreaks in two acute-care hospitals, according to a new study from Ontario, Canada.
With every 10-fold increase in viral copies, the chance of an impending outbreak of COVID-19 rose 22-fold.
“These data add to the mounting evidence that built environment detection for SARS-CoV-2 may provide an additional layer of monitoring and could help inform local infection prevention and control measures,” they wrote.
The study was published online in Infection Control & Hospital Epidemiology.
Preventing Future Suffering
The current study builds on the researchers’ previous work, which found the same correlation between viral load on floors and COVID outbreaks in long-term care homes.
Currently, the best-known method of environmental surveillance for COVID is wastewater detection. “Swabbing the floors would be another approach to surveillance,” senior author Caroline Nott, MD, infectious disease physician at the Ottawa Hospital, said in an interview.
“We do have environmental surveillance with wastewater, but while this may tell you what’s going on in the city, it doesn’t tell you what is going on in a particular ward of a hospital, for instance,” she added.
Nott and her colleagues believe that swabbing, which is easy and relatively inexpensive, will become another tool to examine the built environment. “Instead of having to close a whole hospital, for example, we could just close one room instead of an entire ward if swabbing showed a high concentration of COVID,” Nott said.
The current study was conducted at two hospitals in Ontario between July 2022 and March 2023. The floors of healthcare worker–only areas on four inpatient adult wards were swabbed. These areas included changing rooms, meeting rooms, staff washrooms, nursing stations, and interdisciplinary team rooms.
SARS-CoV-2 RNA was detected on 537 of 760 floor swabs (71%). The overall positivity rate in the first hospital was 90% (n = 280). In the second hospital, the rate was 60% (n = 480).
Four COVID-19 outbreaks occurred in the first acute care hospital, and seven outbreaks occurred at the second hospital. Outbreaks occurred mostly among hospitalized patients (140 cases), but also in four hospital workers.
COVID-19 still requires vigilance, said Nott. “We weren’t prepared for COVID, and so as a result, many people died or have suffered long-term effects, especially vulnerable people like those being treated in hospital or in long-term care facilities. We want to develop methods to prevent similar suffering in the future, whether it’s a new COVID variant or a different pathogen altogether.”
Changing Surveillance Practice?
“This is a good study,” Steven Rogak, PhD, professor of mechanical engineering at the University of British Columbia (UBC) in Vancouver, Canada, said in an interivew. “The fundamental idea is that respiratory droplets and aerosols will deposit on the floor, and polymerase chain reaction [PCR] tests of swabs will provide a surrogate measurement of what might have been inhaled. There are solid statistics that it worked for the hospitals studied,” said Rogak, who studies aerosols at UBC’s Energy and Aerosols Laboratory. Rogak did not participate in the study.
“The authors note several limitations, including that increased healthcare worker testing may have been triggered by the higher values of PCR counts from the floor swabs. But this doesn’t seem to be a problem to me, because if the floor swabs motivate the hospital to test workers more, and that results in identifying outbreaks sooner, then great,” he said.
“Another limitation is that if the hospital has better HVAC or uses air purifiers, it could remove the most infectious aerosols, but the large droplets that fall quickly to the ground would remain, and this would still result in high PCR counts from floor swabs. In this case, perhaps the floor swabs would be a poorer indication of impending outbreaks,” said Rogak.
Determining the best timing and location for floor swabbing might be challenging and specific to the particular hospital, he added. ”For example, you would not want to take swabs from floors right after they are cleaned. Overall, I think this method deserves further development, and it could become a standard technique, but the details might require refinement for widespread application.”
Adrian Popp, MD, chair of the Infectious Disease Service at Huntington Hospital–Northwell Health in New York, said that, although interesting, the study would not change his current practice.
“I’m going to start testing the environment in different rooms in the hospital, and yes, I might find different amounts of COVID, but what does that mean? If pieces of RNA from COVID are on the floor, the likelihood is that they’re not infectious,” Popp said in an interview.
“Hospital workers do get sick with COVID, and sometimes they are asymptomatic and come to work. Patients may come into the hospital for another reason and be sick with COVID. There are many ways people who work in the hospital, as well as the patients, can get COVID. To me, it means that in that hospital and community there is a lot of COVID, but I can’t tell if there is causation here. Who is giving COVID to whom? What am I supposed to do with the information?”
The study was supported by the Northern Ontario Academic Medicine Association Clinical Innovation Opportunities Fund, the Ottawa Hospital Academic Medical Organization Innovation Fund, and a Canadian Institutes of Health Research Operating Grant. One author was a consultant for ProofDx, a startup company creating a point-of-care diagnostic test for COVID-19, and is an advisor for SIGNAL1, a startup company deploying machine-learning models to improve inpatient care. Nott, Rogak, and Popp reported having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM INFECTION CONTROL & HOSPITAL EPIDEMIOLOGY
Which Specialists Should Lead BP Control Efforts?
Current efforts to control high blood pressure (BP) are failing in the United States and globally.
The first World Health Organization (WHO) global report on hypertension found that only 54% of adults with hypertension are diagnosed, 42% get treatment, and just 21% have their hypertension controlled.
In the United States, almost half (48%) of adults have high BP, defined as a systolic BP > 130 mm Hg, or a diastolic BP > 80 mm Hg, or are taking medication for high BP, according to the Centers for Disease Control and Prevention. Only about one in four adults (22.5%) with high BP have their BP under control.
High BP is a major risk factor for coronary heart disease, heart failure, and stroke, and the problem of controlling it is only getting worse. In 2024, the American Heart Association estimates that, “among adults, prevalence of hypertension will increase from 51.2% in 2020 to 61.0% in 2050.”
Pharmacists Most Effective
Though many factors contribute to hypertension, researchers have found that the kind of specialist leading the hypertension team may play a role in success. Currently, most BP control teams are led by physicians in primary care.
In a recent meta-analysis involving 100 randomized controlled trials and more than 90,000 patients in Circulation, Katherine T. Mills, PhD, School of Public Health, Tulane University, New Orleans, Louisiana, and colleagues found that, while all the groups studied who led BP control efforts were successful in reducing BP, pharmacist- and community health worker–led teams saw the biggest reductions.
Those groups’ efforts resulted in the greatest systolic BP drops: −7.3 mm Hg (pharmacists) and −7.1 mm Hg (community health workers). Groups led by nurses and physicians saw systolic changes of −3 and −2.4 mm Hg, respectively.
Similarly, pharmacist- and community health worker–led efforts saw the greatest diastolic BP reductions (−3.8 and −3.1 mm Hg), compared with nurse-led (−1.6) and physician-led (−1.2) efforts.
Reductions Enough to Cut Cardiovascular Disease Risk
The reduction numbers for pharmacists are clinically meaningful, Mills said in an interview. “It’s greater than a lot of what we see from individual lifestyle changes,” such as reducing sodium intake or increasing physical activity.
“It’s a big enough blood pressure change to have meaningful reduction in risk of cardiovascular disease,” she said.
This evidence that the leader of the team matters is particularly important because the treatment of hypertension is not in doubt. Something else is not working the way it should.
“We have basically all the scientific evidence we need in terms of what interventions work. But there’s a big gap between that and what’s actually being done in the real world,” she said.
Mills said she was not surprised that pharmacists got the best results “because so much of it has to do with titrating medications and finding the right kind of medications for each patient.”
Additionally, BP management and control falls right into pharmacists’ wheelhouse, Mills noted, including evaluating medication side effects and talking to patients about medication adherence.
Why Pharmacists May Be More Successful
In an accompanying editorial, Ross T. Tsuyuki, PharmD, with the EPICORE Centre, Division of Cardiology, University of Alberta in Edmonton, Canada, and coauthors said the Mills study provides further data to support pharmacists leading BP control efforts, but it’s not the data that have been keeping the model from changing. The barriers include turf wars and lack of legislative change.
The editorialists also said having pharmacist-led BP teams is only the first step. “We need pharmacists to independently prescribe,” they wrote.
“Since individual states govern the scope of practice of pharmacists,” the editorialists wrote, “we have the enormous task of changing regulations to allow pharmacists to independently prescribe for hypertension. But it can be done. The Canadian province of Alberta allows pharmacists to prescribe. And more recently, Idaho. While most states allow some sort of collaborative (dependent) prescribing, that is only a first step.”
Allowing pharmacists to independently prescribe will help populations who do not have a physician or can’t get access to a physician, the editorialists wrote. But changing state legislation would be a lengthy and complex effort.
Physician-Led BP Control Model ‘Seems to Fail Miserably’
Coauthor of the editorial, Florian Rader, MD, MSc, medical director of the Hypertension Center of Excellence at Cedars-Sinai Medical Center in Los Angeles, California, said in an interview that, currently, physician-led teams are the norm, “and that model seems to fail miserably.”
He offered several key reasons for that. In primary care, patients with hypertension often have other problems — they may have high cholesterol or diabetes. “They may have acute illnesses that bother them as well as hypertension that doesn’t bother them,” he said.
Physicians tend to find excuses not to increase or add BP medications, Rader said. “We tend then to blame ‘white coat effect’ or say ‘you’re just nervous today.’ ”
Pharmacists, comparatively, are more protocol driven, he said. “They essentially look at blood pressure and they have an algorithm in their mind. If the blood pressure hits the guideline-stated bar, start this medication. If it hits another bar, increase or add another medication.”
Rader said turf wars are also keeping physician-led teams from changing, fueled by fears that patients will seek care from pharmacists instead of physicians.
“I don’t think the pharmacists will steal a single patient,” Rader said. “If a physician had a healthcare partner like a pharmacist to optimize blood pressure, then [patients] come back to the physician with normalized BP on the right medications. I think it’s a total win-win. I think we just have to get over that.”
Pharmacist-led warfarin clinics are very well established, Rader said, “but for whatever reason, when it comes to blood pressure, physicians are a little bit more hesitant.”
Collaboration Yes, Independent No
Hypertension expert Donald J. DiPette, MD, Health Sciences Distinguished Professor in the Department of Internal Medicine at University of South Carolina, Columbia, said he completely agrees with Mills and colleagues’ conclusion. “Pharmacists and community health workers are most effective at leading BP intervention implementation and should be prioritized in future hypertension control efforts.”
The conclusion “is in line with the thinking of major organizations,” said DiPette, who helped develop the WHO’s most recent pharmacological treatment of hypertension guidelines. “WHO suggests that pharmacological treatment of hypertension can be provided by nonphysician professionals such as pharmacists and nurses as long as the following conditions are met: Proper training, prescribing authority, specific management protocols, and physician oversight.”
DiPette strongly believes BP control efforts should be supervised by a physician, but that could come in different ways. He suggested a collaborative but physician-supervised development of a protocol. Everyone contributes, but the physician signs off on it.
As for the Idaho example of independent practice for pharmacists, DiPette said he doesn’t think that will make a big difference in control rates. “That’s still not team-based care.”
Community Health Workers Key
He said he was also glad to see community healthcare workers emerge as the next-most-effective group after pharmacists to lead BP control teams. This is particularly important as BP control efforts globally need to consider the cultural experience of individual communities. “The community worker is on the ground, and can help overcome some of the cultural barriers,” he said.
“The key is to focus on team-based care and moving away from silo practice,” DiPette said.
Physicians, he said, often fall into “clinical or therapeutic inertia,” where BP is concerned. “We fail to titrate or add additional hypertensive medications even when they’re clearly indicated by the blood pressure. This is a problem not with the individual patient or the healthcare system, this is on us as physicians.”
Nonphysicians are more aligned with following protocols and guidelines, irrespective of the dynamics of what’s going on, he said.
And following protocols rigidly is a good thing for hypertension. “We’re not overtreating hypertension,” he emphasized. “We’re undertreating it.”
Reversing the trend on hypertension will take a sea change in medicine — changing institutions, systems, and individuals who have been doing things the same way for decades, he said.
“Our hypertension control rates are dismal,” DiPette said. “What’s more alarming is they’re going down. That’s the urgency. That’s the burning platform. We must strongly consider doing something different.”
Tsuyuki has received investigator-initiated arm’s length research grants from Merck, Pfizer, AstraZeneca, and Sanofi. He has been a speaker/consultant for Merck, Emergent BioSolutions, and Shoppers Drug Mart/Loblaw Companies Limited. Rader has been a consultant for Bristol Meyers Squibb, Cytokinetics, Idorsia, Medtronic, and ReCor Medical. Mills and coauthors reported no relevant financial relationships. DiPette declared no relevant financial relationships. He was part of a leadership team that developed WHO guidelines on hypertension.
A version of this article first appeared on Medscape.com.
Current efforts to control high blood pressure (BP) are failing in the United States and globally.
The first World Health Organization (WHO) global report on hypertension found that only 54% of adults with hypertension are diagnosed, 42% get treatment, and just 21% have their hypertension controlled.
In the United States, almost half (48%) of adults have high BP, defined as a systolic BP > 130 mm Hg, or a diastolic BP > 80 mm Hg, or are taking medication for high BP, according to the Centers for Disease Control and Prevention. Only about one in four adults (22.5%) with high BP have their BP under control.
High BP is a major risk factor for coronary heart disease, heart failure, and stroke, and the problem of controlling it is only getting worse. In 2024, the American Heart Association estimates that, “among adults, prevalence of hypertension will increase from 51.2% in 2020 to 61.0% in 2050.”
Pharmacists Most Effective
Though many factors contribute to hypertension, researchers have found that the kind of specialist leading the hypertension team may play a role in success. Currently, most BP control teams are led by physicians in primary care.
In a recent meta-analysis involving 100 randomized controlled trials and more than 90,000 patients in Circulation, Katherine T. Mills, PhD, School of Public Health, Tulane University, New Orleans, Louisiana, and colleagues found that, while all the groups studied who led BP control efforts were successful in reducing BP, pharmacist- and community health worker–led teams saw the biggest reductions.
Those groups’ efforts resulted in the greatest systolic BP drops: −7.3 mm Hg (pharmacists) and −7.1 mm Hg (community health workers). Groups led by nurses and physicians saw systolic changes of −3 and −2.4 mm Hg, respectively.
Similarly, pharmacist- and community health worker–led efforts saw the greatest diastolic BP reductions (−3.8 and −3.1 mm Hg), compared with nurse-led (−1.6) and physician-led (−1.2) efforts.
Reductions Enough to Cut Cardiovascular Disease Risk
The reduction numbers for pharmacists are clinically meaningful, Mills said in an interview. “It’s greater than a lot of what we see from individual lifestyle changes,” such as reducing sodium intake or increasing physical activity.
“It’s a big enough blood pressure change to have meaningful reduction in risk of cardiovascular disease,” she said.
This evidence that the leader of the team matters is particularly important because the treatment of hypertension is not in doubt. Something else is not working the way it should.
“We have basically all the scientific evidence we need in terms of what interventions work. But there’s a big gap between that and what’s actually being done in the real world,” she said.
Mills said she was not surprised that pharmacists got the best results “because so much of it has to do with titrating medications and finding the right kind of medications for each patient.”
Additionally, BP management and control falls right into pharmacists’ wheelhouse, Mills noted, including evaluating medication side effects and talking to patients about medication adherence.
Why Pharmacists May Be More Successful
In an accompanying editorial, Ross T. Tsuyuki, PharmD, with the EPICORE Centre, Division of Cardiology, University of Alberta in Edmonton, Canada, and coauthors said the Mills study provides further data to support pharmacists leading BP control efforts, but it’s not the data that have been keeping the model from changing. The barriers include turf wars and lack of legislative change.
The editorialists also said having pharmacist-led BP teams is only the first step. “We need pharmacists to independently prescribe,” they wrote.
“Since individual states govern the scope of practice of pharmacists,” the editorialists wrote, “we have the enormous task of changing regulations to allow pharmacists to independently prescribe for hypertension. But it can be done. The Canadian province of Alberta allows pharmacists to prescribe. And more recently, Idaho. While most states allow some sort of collaborative (dependent) prescribing, that is only a first step.”
Allowing pharmacists to independently prescribe will help populations who do not have a physician or can’t get access to a physician, the editorialists wrote. But changing state legislation would be a lengthy and complex effort.
Physician-Led BP Control Model ‘Seems to Fail Miserably’
Coauthor of the editorial, Florian Rader, MD, MSc, medical director of the Hypertension Center of Excellence at Cedars-Sinai Medical Center in Los Angeles, California, said in an interview that, currently, physician-led teams are the norm, “and that model seems to fail miserably.”
He offered several key reasons for that. In primary care, patients with hypertension often have other problems — they may have high cholesterol or diabetes. “They may have acute illnesses that bother them as well as hypertension that doesn’t bother them,” he said.
Physicians tend to find excuses not to increase or add BP medications, Rader said. “We tend then to blame ‘white coat effect’ or say ‘you’re just nervous today.’ ”
Pharmacists, comparatively, are more protocol driven, he said. “They essentially look at blood pressure and they have an algorithm in their mind. If the blood pressure hits the guideline-stated bar, start this medication. If it hits another bar, increase or add another medication.”
Rader said turf wars are also keeping physician-led teams from changing, fueled by fears that patients will seek care from pharmacists instead of physicians.
“I don’t think the pharmacists will steal a single patient,” Rader said. “If a physician had a healthcare partner like a pharmacist to optimize blood pressure, then [patients] come back to the physician with normalized BP on the right medications. I think it’s a total win-win. I think we just have to get over that.”
Pharmacist-led warfarin clinics are very well established, Rader said, “but for whatever reason, when it comes to blood pressure, physicians are a little bit more hesitant.”
Collaboration Yes, Independent No
Hypertension expert Donald J. DiPette, MD, Health Sciences Distinguished Professor in the Department of Internal Medicine at University of South Carolina, Columbia, said he completely agrees with Mills and colleagues’ conclusion. “Pharmacists and community health workers are most effective at leading BP intervention implementation and should be prioritized in future hypertension control efforts.”
The conclusion “is in line with the thinking of major organizations,” said DiPette, who helped develop the WHO’s most recent pharmacological treatment of hypertension guidelines. “WHO suggests that pharmacological treatment of hypertension can be provided by nonphysician professionals such as pharmacists and nurses as long as the following conditions are met: Proper training, prescribing authority, specific management protocols, and physician oversight.”
DiPette strongly believes BP control efforts should be supervised by a physician, but that could come in different ways. He suggested a collaborative but physician-supervised development of a protocol. Everyone contributes, but the physician signs off on it.
As for the Idaho example of independent practice for pharmacists, DiPette said he doesn’t think that will make a big difference in control rates. “That’s still not team-based care.”
Community Health Workers Key
He said he was also glad to see community healthcare workers emerge as the next-most-effective group after pharmacists to lead BP control teams. This is particularly important as BP control efforts globally need to consider the cultural experience of individual communities. “The community worker is on the ground, and can help overcome some of the cultural barriers,” he said.
“The key is to focus on team-based care and moving away from silo practice,” DiPette said.
Physicians, he said, often fall into “clinical or therapeutic inertia,” where BP is concerned. “We fail to titrate or add additional hypertensive medications even when they’re clearly indicated by the blood pressure. This is a problem not with the individual patient or the healthcare system, this is on us as physicians.”
Nonphysicians are more aligned with following protocols and guidelines, irrespective of the dynamics of what’s going on, he said.
And following protocols rigidly is a good thing for hypertension. “We’re not overtreating hypertension,” he emphasized. “We’re undertreating it.”
Reversing the trend on hypertension will take a sea change in medicine — changing institutions, systems, and individuals who have been doing things the same way for decades, he said.
“Our hypertension control rates are dismal,” DiPette said. “What’s more alarming is they’re going down. That’s the urgency. That’s the burning platform. We must strongly consider doing something different.”
Tsuyuki has received investigator-initiated arm’s length research grants from Merck, Pfizer, AstraZeneca, and Sanofi. He has been a speaker/consultant for Merck, Emergent BioSolutions, and Shoppers Drug Mart/Loblaw Companies Limited. Rader has been a consultant for Bristol Meyers Squibb, Cytokinetics, Idorsia, Medtronic, and ReCor Medical. Mills and coauthors reported no relevant financial relationships. DiPette declared no relevant financial relationships. He was part of a leadership team that developed WHO guidelines on hypertension.
A version of this article first appeared on Medscape.com.
Current efforts to control high blood pressure (BP) are failing in the United States and globally.
The first World Health Organization (WHO) global report on hypertension found that only 54% of adults with hypertension are diagnosed, 42% get treatment, and just 21% have their hypertension controlled.
In the United States, almost half (48%) of adults have high BP, defined as a systolic BP > 130 mm Hg, or a diastolic BP > 80 mm Hg, or are taking medication for high BP, according to the Centers for Disease Control and Prevention. Only about one in four adults (22.5%) with high BP have their BP under control.
High BP is a major risk factor for coronary heart disease, heart failure, and stroke, and the problem of controlling it is only getting worse. In 2024, the American Heart Association estimates that, “among adults, prevalence of hypertension will increase from 51.2% in 2020 to 61.0% in 2050.”
Pharmacists Most Effective
Though many factors contribute to hypertension, researchers have found that the kind of specialist leading the hypertension team may play a role in success. Currently, most BP control teams are led by physicians in primary care.
In a recent meta-analysis involving 100 randomized controlled trials and more than 90,000 patients in Circulation, Katherine T. Mills, PhD, School of Public Health, Tulane University, New Orleans, Louisiana, and colleagues found that, while all the groups studied who led BP control efforts were successful in reducing BP, pharmacist- and community health worker–led teams saw the biggest reductions.
Those groups’ efforts resulted in the greatest systolic BP drops: −7.3 mm Hg (pharmacists) and −7.1 mm Hg (community health workers). Groups led by nurses and physicians saw systolic changes of −3 and −2.4 mm Hg, respectively.
Similarly, pharmacist- and community health worker–led efforts saw the greatest diastolic BP reductions (−3.8 and −3.1 mm Hg), compared with nurse-led (−1.6) and physician-led (−1.2) efforts.
Reductions Enough to Cut Cardiovascular Disease Risk
The reduction numbers for pharmacists are clinically meaningful, Mills said in an interview. “It’s greater than a lot of what we see from individual lifestyle changes,” such as reducing sodium intake or increasing physical activity.
“It’s a big enough blood pressure change to have meaningful reduction in risk of cardiovascular disease,” she said.
This evidence that the leader of the team matters is particularly important because the treatment of hypertension is not in doubt. Something else is not working the way it should.
“We have basically all the scientific evidence we need in terms of what interventions work. But there’s a big gap between that and what’s actually being done in the real world,” she said.
Mills said she was not surprised that pharmacists got the best results “because so much of it has to do with titrating medications and finding the right kind of medications for each patient.”
Additionally, BP management and control falls right into pharmacists’ wheelhouse, Mills noted, including evaluating medication side effects and talking to patients about medication adherence.
Why Pharmacists May Be More Successful
In an accompanying editorial, Ross T. Tsuyuki, PharmD, with the EPICORE Centre, Division of Cardiology, University of Alberta in Edmonton, Canada, and coauthors said the Mills study provides further data to support pharmacists leading BP control efforts, but it’s not the data that have been keeping the model from changing. The barriers include turf wars and lack of legislative change.
The editorialists also said having pharmacist-led BP teams is only the first step. “We need pharmacists to independently prescribe,” they wrote.
“Since individual states govern the scope of practice of pharmacists,” the editorialists wrote, “we have the enormous task of changing regulations to allow pharmacists to independently prescribe for hypertension. But it can be done. The Canadian province of Alberta allows pharmacists to prescribe. And more recently, Idaho. While most states allow some sort of collaborative (dependent) prescribing, that is only a first step.”
Allowing pharmacists to independently prescribe will help populations who do not have a physician or can’t get access to a physician, the editorialists wrote. But changing state legislation would be a lengthy and complex effort.
Physician-Led BP Control Model ‘Seems to Fail Miserably’
Coauthor of the editorial, Florian Rader, MD, MSc, medical director of the Hypertension Center of Excellence at Cedars-Sinai Medical Center in Los Angeles, California, said in an interview that, currently, physician-led teams are the norm, “and that model seems to fail miserably.”
He offered several key reasons for that. In primary care, patients with hypertension often have other problems — they may have high cholesterol or diabetes. “They may have acute illnesses that bother them as well as hypertension that doesn’t bother them,” he said.
Physicians tend to find excuses not to increase or add BP medications, Rader said. “We tend then to blame ‘white coat effect’ or say ‘you’re just nervous today.’ ”
Pharmacists, comparatively, are more protocol driven, he said. “They essentially look at blood pressure and they have an algorithm in their mind. If the blood pressure hits the guideline-stated bar, start this medication. If it hits another bar, increase or add another medication.”
Rader said turf wars are also keeping physician-led teams from changing, fueled by fears that patients will seek care from pharmacists instead of physicians.
“I don’t think the pharmacists will steal a single patient,” Rader said. “If a physician had a healthcare partner like a pharmacist to optimize blood pressure, then [patients] come back to the physician with normalized BP on the right medications. I think it’s a total win-win. I think we just have to get over that.”
Pharmacist-led warfarin clinics are very well established, Rader said, “but for whatever reason, when it comes to blood pressure, physicians are a little bit more hesitant.”
Collaboration Yes, Independent No
Hypertension expert Donald J. DiPette, MD, Health Sciences Distinguished Professor in the Department of Internal Medicine at University of South Carolina, Columbia, said he completely agrees with Mills and colleagues’ conclusion. “Pharmacists and community health workers are most effective at leading BP intervention implementation and should be prioritized in future hypertension control efforts.”
The conclusion “is in line with the thinking of major organizations,” said DiPette, who helped develop the WHO’s most recent pharmacological treatment of hypertension guidelines. “WHO suggests that pharmacological treatment of hypertension can be provided by nonphysician professionals such as pharmacists and nurses as long as the following conditions are met: Proper training, prescribing authority, specific management protocols, and physician oversight.”
DiPette strongly believes BP control efforts should be supervised by a physician, but that could come in different ways. He suggested a collaborative but physician-supervised development of a protocol. Everyone contributes, but the physician signs off on it.
As for the Idaho example of independent practice for pharmacists, DiPette said he doesn’t think that will make a big difference in control rates. “That’s still not team-based care.”
Community Health Workers Key
He said he was also glad to see community healthcare workers emerge as the next-most-effective group after pharmacists to lead BP control teams. This is particularly important as BP control efforts globally need to consider the cultural experience of individual communities. “The community worker is on the ground, and can help overcome some of the cultural barriers,” he said.
“The key is to focus on team-based care and moving away from silo practice,” DiPette said.
Physicians, he said, often fall into “clinical or therapeutic inertia,” where BP is concerned. “We fail to titrate or add additional hypertensive medications even when they’re clearly indicated by the blood pressure. This is a problem not with the individual patient or the healthcare system, this is on us as physicians.”
Nonphysicians are more aligned with following protocols and guidelines, irrespective of the dynamics of what’s going on, he said.
And following protocols rigidly is a good thing for hypertension. “We’re not overtreating hypertension,” he emphasized. “We’re undertreating it.”
Reversing the trend on hypertension will take a sea change in medicine — changing institutions, systems, and individuals who have been doing things the same way for decades, he said.
“Our hypertension control rates are dismal,” DiPette said. “What’s more alarming is they’re going down. That’s the urgency. That’s the burning platform. We must strongly consider doing something different.”
Tsuyuki has received investigator-initiated arm’s length research grants from Merck, Pfizer, AstraZeneca, and Sanofi. He has been a speaker/consultant for Merck, Emergent BioSolutions, and Shoppers Drug Mart/Loblaw Companies Limited. Rader has been a consultant for Bristol Meyers Squibb, Cytokinetics, Idorsia, Medtronic, and ReCor Medical. Mills and coauthors reported no relevant financial relationships. DiPette declared no relevant financial relationships. He was part of a leadership team that developed WHO guidelines on hypertension.
A version of this article first appeared on Medscape.com.
VHA Support for Home Health Agency Staff and Patients During Natural Disasters
As large-scale natural disasters become more common, health care coalitions and the engagement of health systems with local, state, and federal public health departments have effectively bolstered communities’ resilience via collective sharing and distribution of resources.1 These resources may include supplies and the dissemination of emergency information, education, and training.2 The COVID-19 pandemic demonstrated that larger health care systems including hospital networks and nursing homes are better connected to health care coalition resources than smaller, independent systems, such as community home health agencies.3 This leaves some organizations on their own to meet requirements that maintain continuity of care and support their patients and staff throughout a natural disaster.
Home health care workers play important roles in the care of older adults.4 Older adults experience high levels of disability and comorbidities that put them at risk during emergencies; they often require support from paid, family, and neighborhood caregivers to live independently.5 More than 9.3 million US adults receive paid care from 2.6 million home health care workers (eg, home health aides and personal care assistants).6 Many of these individuals are hired through small independent home health agencies (HHAs), while others may work directly for an individual. When neighborhood resources and family caregiving are disrupted during emergencies, the critical services these workers administer become even more essential to ensuring continued access to medical care and social services.
The importance of these services was underscored by the Centers for Medicare and Medicaid Services 2017 inclusion of HHAs in federal emergency preparedness guidelines.7,8 The fractured and decentralized nature of the home health care industry means many HHAs struggle to maintain continuous care during emergencies and protect their staff. HHAs, and health care workers in the home, are often isolated, under-resourced, and disconnected from broader emergency planning efforts. Additionally, home care jobs are largely part-time, unstable, and low paying, making the workers themselves vulnerable during emergencies.3,9-13
This is a significant issue for the Veterans Health Administration (VHA), which annually purchases 10.5 million home health care worker visits for 150,000 veterans from community-based HHAs to enable those individuals to live independently. Figure 1 illustrates the existing structure of directly provided and contracted VHA services for community-dwelling veterans, highlighting the circle of care around the veteran.8,9 Home health care workers anchored health care teams during the COVID-19 pandemic, observing and reporting on patients’ well-being to family caregivers, primary care practitioners, and HHAs. They also provided critical emotional support and companionship to patients isolated from family and friends.9 These workers also exposed themselves and their families to considerable risk and often lacked the protection afforded by personal protective equipment (PPE) in accordance with infection prevention guidance.3,12

Abbreviations: HBPC, home based primary care; HHA, home health agency; VHA, Veterans Health Administration.
aAdapted with permission from Wyte-Lake and Franzosa.8,9
Through a combination of its national and local health care networks, the VHA has a robust and well-positioned emergency infrastructure to supportcommunity-dwelling older adults during disasters.14 This network is supported by the VHA Office of Emergency Management, which shares resources and guidance with local emergency managers at each facility as well as individual programs such as the VHA Home Based Primary Care (HBPC) program, which provides 38,000 seriously ill veterans with home medical visits.15 Working closely with their local and national hospital networks and emergency managers, individual VHA HBPC programs were able to maintain the safety of staff and continuity of care for patients enrolled in HBPC by rapidly administering COVID-19 vaccines to patients, caregivers, and staff, and providing emergency assistance during the 2017 hurricane season.16,17 These efforts were successful because HBPC practitioners and their patients, had access to a level of emergency-related information, resources, and technology that are often out of reach for individual community-based health care practitioners (HCPs). The US Department of Veterans Affairs (VA) also supports local communities through its Fourth Mission, which provides emergency resources to non-VHA health care facilities (ie, hospitals and nursing homes) during national emergencies and natural disasters.17 Although there has been an expansion in the definition of shared resources, such as extending behavioral health support to local communities, the VHA has not historically provided these resources to HHAs.14
This study examines opportunities to leverage VHA emergency management resources to support contracted HHAs and inform other large health system emergency planning efforts. The findings from the exploratory phase are described in this article. We interviewed VHA emergency managers, HBPC and VA staff who coordinate home health care worker services, as well as administrators at contracted HHAs within a Veterans Integrated Services Network (VISN). These findings will inform the second (single-site pilot study) and third (feasibility study) phases. Our intent was to (1) better understand the relationships between VA medical centers (VAMCs) and their contracted HHAs; (2) identify existing VHA emergency protocols to support community-dwelling older adults; and (3) determine opportunities to build on existing infrastructure and relationships to better support contracted HHAs and their staff in emergencies.
Methods
The 18 VISNs act as regional systems of care that are loosely connected to better meet local health needs and maximize access to care. This study was conducted at 6 of 9 VAMCs within VISN 2, the New York/New Jersey VHA Health Care Network.18 VAMCs that serve urban, rural, and mixed urban/rural catchment areas were included.
Each VAMC has an emergency management program led by an emergency manager, an HBPC program led by a program director and medical director, and a community care or purchased care office that has a liaison who manages contracted home health care worker services. The studyfocused on HBPC programs because they are most likely to interact with veterans’ home health care workers in the home and care for community-dwelling veterans during emergencies. Each VHA also contracts with a series of local HHAs that generally have a dedicated staff member who interfaces with the VHA liaison. Our goal was to interview ≥ 1 emergency manager, ≥ 1 HBPC team member, ≥ 1 community care staff person, and ≥ 1 contracted home health agency administrator at each site to gain multiple perspectives from the range of HCPs serving veterans in the community.
Recruitment and Data Collection
The 6 sites were selected in consultation with VISN 2 leadership for their strong HBPC and emergency management programs. To recruit respondents, we contacted VISN and VAMC leads and used our professional networks to identify a sample of multidisciplinary individuals who represent both community care and HBPC programs who were contacted via email.
Since each VAMC is organized differently, we utilized a snowball sampling approach to identify the appropriate contacts.19 At the completion of each interview, we asked the participant to suggest additional contacts and introduce us to any remaining stakeholders (eg, the emergency manager) at that site or colleagues at other VISN facilities. Because roles vary among VAMCs, we contacted the person who most closely resembled the identified role and asked them to direct us to a more appropriate contact, if necessary. We asked community care managers to identify 1 to 2 agencies serving the highest volume of patients who are veterans at their site and requested interviews with those liaisons. This resulted in the recruitment of key stakeholders from 4 teams across the 6 sites (Table).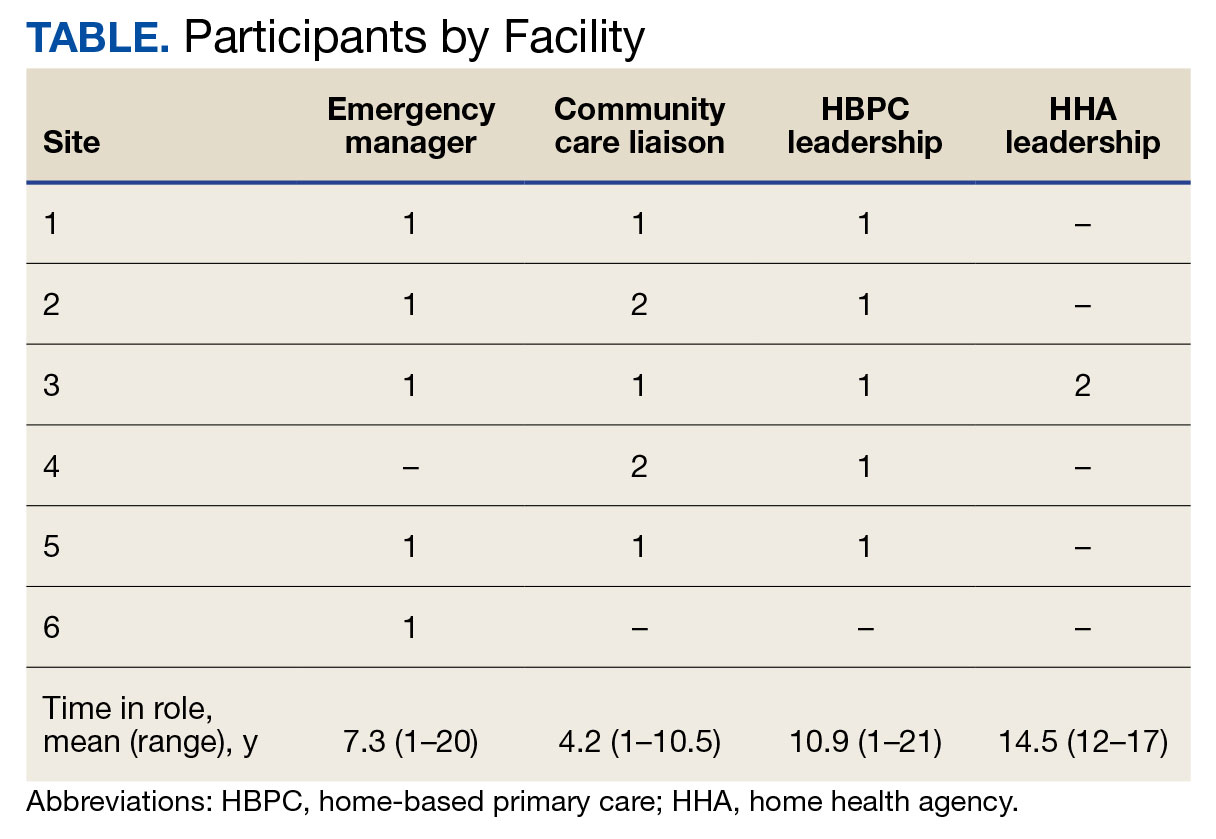
A semistructured interview guide was jointly developed based on constructs of interest, including relationships within VAMCs and between VAMCs and HHAs; existing emergency protocols and experience during disasters; and suggestions and opportunities for supporting agencies during emergencies and potential barriers. Two researchers (TWL and EF) who were trained in qualitative methods jointly conducted interviews using the interview guide, with 1 researcher leading and another taking notes and asking clarifying questions.
Interviews were conducted virtually via Microsoft Teams with respondents at their work locations between September 2022 and January 2023. Interviews were audio recorded and transcribed and 2 authors (TWL and ESO) reviewed transcripts for accuracy. Interviews averaged 47 minutes in length (range, 20-59).
The study was reviewed and determined to be exempt by institutional review boards at the James J. Peters VAMC and Greater Los Angeles VAMC. We asked participants for verbal consent to participate and preserved their confidentiality.
Analysis
Data were analyzed via an inductive approach, which involves drawing salient themes rather than imposing preconceived theories.20 Three researchers (TWL, EF, and ES) listened to and discussed 2 staff interviews and tagged text with specific codes (eg, communication between the VHA and HHA, internal communication, and barriers to case fulfillment) so the team could selectively return to the interview text for deeper analysis, allowing for the development of a final codebook. The project team synthesized the findings to identify higher-level themes, drawing comparisons across and within the respondent groups, including within and between health care systems. Throughout the analysis, we maintained analytic memos, documented discussions, and engaged in analyst triangulation to ensure trustworthiness.21,22 To ensure the analysis accurately reflected the participants’ understanding, we held 2 virtual member-checking sessions with participants to share preliminary findings and conclusions and solicit feedback. Analysis was conducted using ATLAS.ti version 20.
Results
VHA-based participants described internal emergency management systems that are deployed during a disaster to support patients and staff. Agency participants described their own internal emergency management protocols. Respondents discussed how and when the 2 intersected, as well as opportunities for future mutual support. The analysis identified several themes: (1) relationships between VAMC teams; (2) relationships between VHA and HHAs; (3) VHA and agencies responses during emergencies; (4) receptivity and opportunities for extending VHA resources into the community; and (5) barriers and facilitators to deeper engagement.
Relationships Within VHA (n = 17)
Staff at all VHA sites described close relationships between the internal emergency management and HBPC teams. HBPC teams identified patients who were most at risk during emergencies to triage those with the highest medical needs (eg, patients dependent on home infusion, oxygen, or electronic medical devices) and worked alongside emergency managers to develop plans to continue care during an emergency. HBPC representatives were part of their facilities’ local emergency response committees. Due to this close collaboration, VHA emergency managers were familiar with the needs of homebound veterans and caregivers. “I invite our [HBPC] program manager to attend [committee] meetings and … they’re part of the EOC [emergency operations center]," an emergency manager said. “We work together and I’m constantly in contact with that individual, especially during natural disasters and so forth, to ensure that everybody’s prepared in the community.”
On the other hand, community caremanagers—who described frequent interactions with HBPC teams, largely around coordinating and managing non-VHA home care services—were less likely to have direct relationships with their facility emergency managers. For example, when asked if they had a relationship with their emergency manager, a community care manager admitted, “I [only] know who he is.” They also did not report having structured protocols for veteran outreach during emergencies, “because all those veterans who are receiving [home health care worker] services also belong to a primary care team,” and considered the outreach to be the responsibility of the primary care team and HHA.
Relationships Between the VHA and HHAs (n = 17)
Communication between VAMCs and contracted agencies primarily went through community care managers, who described established long-term relationships with agency administrators. Communication was commonly restricted to operational activities, such as processing referrals and occasional troubleshooting. According to a community care manager most communication is “why haven’t you signed my orders?” There was a general sense from participants that communication was promptly answered, problems were addressed, and professional collegiality existed between the agencies as patients were referred and placed for services. One community care manager reported meeting with agencies regularly, noting, “I talk to them pretty much daily.”
If problems arose, community care managers described themselves as “the liaison” between agencies and VHA HCPs who ordered the referrals. This is particularly the case if the agency needed help finding a VHA clinician or addressing differences in care delivery protocols.
Responding During Emergencies (n = 19)
During emergencies, VHA and agency staff described following their own organization’s protocols and communicating with each other only on a case-by-case basis rather than through formal or systematic channels and had little knowledge of their counterpart’s emergency protocols. Beyond patient care, there was no evidence of information sharing between VHA and agency staff. Regarding sharing information with their local community, an HBPC Program Director said, “it’s almost like the VHA had become siloed” and operated on its own without engaging with community health systems or emergency managers.
Beyond the guidance provided by state departments of public health, HHAs described collaborating with other agencies in their network and relying on their informal professional network to manage the volume of information and updates they followed during emergencies like the COVID-19 pandemic. One agency administrator did not frequently communicate with VHA partners during the pandemic but explained that the local public health department helped work through challenges. However, “we realized pretty quickly they were overloaded and there was only so much they could do.” The agency administrator turned to a “sister agency” and local hospitals, noting, “Wherever you have connections in the field or in the industry, you know you’re going to reach out to people for guidance on policies and… protocol.”
Opportunities for Extending VHA Resources to the Community (n = 16)
All VHA emergency managers were receptive to extending support to community-based HCPS and, in some cases, felt strongly that they were an essential part of veterans’ care networks. Emergency managers offered examples for how they supportedcommunity-based HCPs, such as helping those in the VAMC medical foster home program develop and evaluate emergency plans. Many said they had not explicitly considered HHAs before (Appendix).
Emergency managers also described how supporting community-based HCPs could be considered within the scope of the VHA role and mission, specifically the Fourth Mission. “I think that we should be making our best effort to make sure that we’re also providing that same level [of protection] to the people taking care of the veteran [as our VHA staff],” an emergency manager said. “It’s our responsibility to provide the best for the staff that are going into those homes to take care of that patient.”
In many cases, emergency managers had already developed practical tools that could be easily shared outside the VHA, including weather alerts, trainings, emergency plan templates, and lists of community resources and shelters (Figure 2). A number of these examples built on existing communication channels. One emergency manager said that the extension of resources could be an opportunity to decrease the perceived isolation of home health care workers through regular

Abbreviations: PPE, personal protective equipment; VA, US Department of Veterans Affairs.
On the agency side, participants noted that some HHAs could benefit more from support than others. While some agencies are well staffed and have good protocols and keep up to date, “There are smaller agencies, agencies that are starting up that may not have the resources to just disseminate all the information. Those are the agencies [that] could well benefit from the VHA,” an HBPC medical director explained. Agency administrators suggested several areas where they would welcome support, including a deeper understanding of available community resources and access to PPE for staff. Regarding informational resources, an administrator said, “Anytime we can get information, it’s good to have it come to you and not always have to go out searching for it.”
Barriers and Facilitators to Partnering With Community Agencies (n = 16)
A primary barrier regarding resource sharing was potential misalignment between each organization’s policies. HHAs followed state and federal public health guidelines, which sometimes differed from VHA policies. Given that agencies care for both VHA and non-VHA clients, questions also arose around how agencies would prioritize information from the VHA, if they were already receiving information from other sources. When asked about information sharing, both VHA staff and agencies agreed staff time to support any additional activities should be weighed against the value of the information gained.
Six participants also shared that education around emergency preparedness could be an opportunity to bridge gaps between VAMCs and their surrounding communities.
Two emergency managers noted the need to be sensitive in the way they engaged with partners, respecting and building on the work that agencies were already doing in this area to ensure VHA was seen as a trusted partner and resource rather than trying to impose new policies or rules on community-based HCPs. “I know that like all leadership in various organizations, there’s a little bit of bristling going on when other people try and tell them what to do,” an HBPC medical director said. “However, if it is established that as a sort of greater level like a state level or a federal level, that VHA can be a resource. I think that as long as that’s recognized by their own professional organizations within each state, then I think that that would be a tremendous advantage to many agencies.”
In terms of sharing physical resources, emergency managers raised concerns around potential liability, although they also acknowledged this issue was important enough to think about potential workarounds. As one emergency manager said, “I want to know that my PPE is not compromised in any way shape or form and that I am in charge of that PPE, so to rely upon going to a home and hoping that [the PPE] wasn’t compromised … would kind of make me a little uneasy.” This emergency manager suggested possible solutions, such as creating a sealed PPE package to give directly to an aide.
Discussion
As the prevalence of climate-related disasters increases, the need to ensure the safety and independence of older adults during emergencies grows more urgent. Health systems must think beyond the direct services they provide and consider the community resources upon which their patients rely. While relationships did not formally exist between VHA emergency managers and community home health HCPs in the sample analyzed in this article, there is precedent and interest in supporting contracted home health agencies caring for veterans in the community. Although not historically part of the VA Fourth Mission, creating a pipeline of support for contracted HHAs by leveraging existing relationships and resources can potentially strengthen its mission to protect older veterans in emergencies, help them age safely in place, and provide a model for health systems to collaborate with community-based HCPs around emergency planning and response (Figure 3).23

Existing research on the value of health care coalitions highlights the need for established and growing partnerships with a focus on ensuring they are value-added, which echoes concerns we heard in interviews.24 Investment in community partnerships not only includes sharing supplies but also relying on bidirectional support that can be a trusted form of timely information.1,25 The findings in this study exhibit strong communication practices within the VHA during periods of nonemergency and underscore the untapped value of the pre-existing relationship between VAMCs and their contracted HHAs as an area of potential growth for health care coalitions.
Sharing resources in a way that does not put new demands on partners contributes to the sustainability and value-added nature of coalitions. Examples include establishing new low-investment practices (ie, information sharing) that support capacity and compliance with existing requirements rather than create new responsibilities for either member of the coalition. The relationship between the VHA emergency managers and the VHA HBPC program can act as a guide. The emergency managers interviewed for this study are currently engaged with HBPC programs and therefore understand the needs of homebound older adults and their caregivers. Extending the information already available to the HBPC teams via existing channels strengthens workforce practices and increased security for the shared patient, even without direct relationships between emergency managers and agencies. It is important to understand the limitations of these practices, including concerns around conflicting federal and state mandates, legal concerns around the liability of sharing physical resources (such as PPE), and awareness that the objective is not for the VHA to increase burdens (eg, increasing compliance requirements) but rather to serve as a resource for a mutual population in a shared community.
Offering training and practical resources to HHA home health care workers can help them meet disaster preparedness requirements. This is particularly important considering the growing home care workforce shortages, a topic mentioned by all HBPC and community care participants interviewed for this study.26,27 Home health care workers report feeling underprepared and isolated while on the job in normal conditions, a sentiment exacerbated by the COVID-19 pandemic.3,10 Supporting these individuals may help them feel more prepared and connected to their work, improving stability and quality of care.
While these issues are priorities within the VHA, there is growing recognition at the state and federal level of the importance of including older adults and their HCPs in disaster preparedness and response.5,28 The US Department of Health and Human Services, for example, includes older adults and organizations that serve them on its National Advisory Committee on Seniors and Disasters. The Senate version of the 2023 reauthorization of the Pandemic and All-Hazards Preparedness and Response Act included specific provisions to support community-dwelling older adults and people with disabilities, incorporating funding for community organizations to support continuity of services and avoid institutionalization in an emergency.29 Other proposed legislation includes the Real Emergency Access for Aging and Disability Inclusion for Disasters Act, which would ensure the needs of older adults and people with disabilities are explicitly included in all phases of emergency planning and response.30
The VHA expansion of the its VEText program to include disaster response is an effort to more efficiently extend outreach to older and vulnerable patients who are veterans.31 Given these growing efforts, the VHA and other health systems have an opportunity to expand internal emergency preparedness efforts to ensure the health and safety of individuals living in the community.
Limitations
VISN 2 has been a target of terrorism and other disasters. In addition to the sites being initially recruited for their strong emergency management protocols, this context may have biased respondents who are favorable to extending their resources into the community. At the time of recruitment, contracted HHAs were still experiencing staff shortages due to the COVID-19 pandemic, which limited the ability of agency staff to participate in interviews. Additionally, while the comprehensive exploration of VISN 2 facilities allows for confidence of the organizational structures described, the qualitative research design and small study sample, the study findings cannot be immediately generalized to all VISNs.
Conclusions
Many older veterans increasingly rely on home health care workers to age safely. The VHA, as a large national health care system and leader in emergency preparedness, could play an important role in supporting home health care workers and ameliorating their sense of isolation during emergencies and natural disasters. Leveraging existing resources and relationships may be a low-cost, low-effort opportunity to build higher-level interventions that support the needs of patients. Future research and work in this field, including the authors’ ongoing work, will expand agency participation and engage agency staff in conceptualizing pilot projects to ensure they are viable and feasible for the field.

- Barnett DJ, Knieser L, Errett NA, Rosenblum AJ, Seshamani M, Kirsch TD. Reexamining health-care coalitions in light of COVID-19. Disaster Med public Health Prep. 2022;16(3):859-863. doi:10.1017/dmp.2020.431
- Wulff K, Donato D, Lurie N. What is health resilience and how can we build it? Annu Rev Public Health. 2015;36:361-374. doi:10.1146/annurev-publhealth-031914-122829
- Franzosa E, Wyte-Lake T, Tsui EK, Reckrey JM, Sterling MR. Essential but excluded: building disaster preparedness capacity for home health care workers and home care agencies. J Am Med Dir Assoc. 2022;23(12):1990-1996. doi:10.1016/j.jamda.2022.09.012
- Miner S, Masci L, Chimenti C, Rin N, Mann A, Noonan B. An outreach phone call project: using home health to reach isolated community dwelling adults during the COVID 19 lockdown. J Community Health. 2022;47(2):266-272. doi:10.1007/s10900-021-01044-6
- National Institute on Aging. Protecting older adults from the effects of natural disasters and extreme weather. October 18, 2022. Accessed August 19, 2024. https://www.nia.nih.gov/news/protecting-older-adults-effects-natural-disasters-and-extreme-weather
- PHI. Direct Care Workers in the United States: Key Facts. September 7, 2021. Accessed August 19, 2024. https://www.phinational.org/resource/direct-care-workers-in-the-united-states-key-facts-2/
- Centers for Medicare & Medicaid Services. Emergency Preparedness Rule. September 8, 2016. Updated September 6, 2023. Accessed August 19, 2024. https://www.cms.gov/medicare/health-safety-standards/quality-safety-oversight-emergency-preparedness/emergency-preparedness-rule
- Wyte-Lake T, Claver M, Tubbesing S, Davis D, Dobalian A. Development of a home health patient assessment tool for disaster planning. Gerontology. 2019;65(4):353-361. doi:10.1159/000494971
- Franzosa E, Judon KM, Gottesman EM, et al. Home health aides’ increased role in supporting older veterans and primary healthcare teams during COVID-19: a qualitative analysis. J Gen Intern Med. 2022;37(8):1830-1837. doi:10.1007/s11606-021-07271-w
- Franzosa E, Tsui EK, Baron S. “Who’s caring for us?”: understanding and addressing the effects of emotional labor on home health aides’ well-being. Gerontologist. 2019;59(6):1055-1064. doi:10.1093/geront/gny099
- Osakwe ZT, Osborne JC, Samuel T, et al. All alone: a qualitative study of home health aides’ experiences during the COVID-19 pandemic in New York. Am J Infect Control. 2021;49(11):1362-1368. doi:10.1016/j.ajic.2021.08.004
- Feldman PH, Russell D, Onorato N, et al. Ensuring the safety of the home health aide workforce and the continuation of essential patient care through sustainable pandemic preparedness. July 2022. Accessed August 19, 2024. https://www.vnshealth.org/wp-content/uploads/2022/08/Pandemic_Preparedness_IB_07_21_22.pdf
- Sterling MR, Tseng E, Poon A, et al. Experiences of home health care workers in New York City during the coronavirus disease 2019 pandemic: a qualitative analysis. JAMA Internal Med. 2020;180(11):1453-1459. doi:10.1001/jamainternmed.2020.3930
- Wyte-Lake T, Schmitz S, Kornegay RJ, Acevedo F, Dobalian A. Three case studies of community behavioral health support from the US Department of Veterans Affairs after disasters. BMC Public Health. 2021;21(1):639. doi:10.1186/s12889-021-10650-x
- Beales JL, Edes T. Veteran’s affairs home based primary care. Clin Geriatr Med. 2009;25(1):149-ix. doi:10.1016/j.cger.2008.11.002
- Wyte-Lake T, Manheim C, Gillespie SM, Dobalian A, Haverhals LM. COVID-19 vaccination in VA home based primary care: experience of interdisciplinary team members. J Am Med Dir Assoc. 2022;23(6):917-922. doi:10.1016/j.jamda.2022.03.014
- Wyte-Lake T, Schmitz S, Cosme Torres-Sabater R, Dobalian A. Case study of VA Caribbean Healthcare System’s community response to Hurricane Maria. J Emerg Manag. 2022;19(8):189-199. doi:10.5055/jem.0536
- US Department of Veterans Affairs. New York/New Jersey VA Health Care Network, VISN 2 Locations. Updated January 3, 2024. Accessed August 19, 2024. https://www.visn2.va.gov/visn2/facilities.asp
- Noy C. Sampling knowledge: the hermeneutics of snowball sampling in qualitative research. Int J Soc Res Methodol. 2008;11(4):327-344. doi:10.1080/13645570701401305
- Ritchie J, Lewis J, Nicholls CM, Ormston R, eds. Qualitative Research Practice: A Guide for Social Science Students and Researchers. 2nd ed. Sage; 2013.
- Morrow SL. Quality and trustworthiness in qualitative research in counseling psychology. J Couns Psychol. 2005;52(2):250-260. doi:10.1037/0022-0167.52.2.250
- Rolfe G. Validity, trustworthiness and rigour: quality and the idea of qualitative research. J Adv Nurs. 2006;53(3):304-310. doi:10.1111/j.1365-2648.2006.03727.x
- Schmitz S, Wyte-Lake T, Dobalian A. Facilitators and barriers to preparedness partnerships: a veterans affairs medical center perspective. Disaster Med Public Health Prep. 2018;12(4):431-436. doi:10.1017/dmp.2017.92
- Koch AE, Bohn J, Corvin JA, Seaberg J. Maturing into high-functioning health-care coalitions: a qualitative Nationwide study of emergency preparedness and response leadership. Disaster Med Public Health Prep. 2022;17:e111. doi:10.1017/dmp.2022.13
- Lin JS, Webber EM, Bean SI, Martin AM, Davies MC. Rapid evidence review: policy actions for the integration of public health and health care in the United States. Front Public Health. 2023;11:1098431. doi:10.3389/fpubh.2023.1098431
- Watts MOM, Burns A, Ammula M. Ongoing impacts of the pandemic on medicaid home & community-based services (HCBS) programs: findings from a 50-state survey. November 28, 2022. Accessed August 19, 2024. https://www.kff.org/medicaid/issue-brief/ongoing-impacts-of-the-pandemic-on-medicaid-home-community-based-services-hcbs-programs-findings-from-a-50-state-survey/
- Kreider AR, Werner RM. The home care workforce has not kept pace with growth in home and community-based services. Health Aff (Millwood). 2023;42(5):650-657. doi:10.1377/hlthaff.2022.01351
- FEMA introduces disaster preparedness guide for older adults. News release. FEMA. September 20, 2023. Accessed August 19, 2024. https://www.fema.gov/press-release/20230920/fema-introduces-disaster-preparedness-guide-older-adults
- Pandemic and All-Hazards Preparedness and Response Act, S 2333, 118th Cong, 1st Sess (2023). https://www.congress.gov/bill/118th-congress/senate-bill/2333/text
- REAADI for Disasters Act, HR 2371, 118th Cong, 1st Sess (2023). https://www.congress.gov/bill/118th-congress/house-bill/2371
- Wyte-Lake T, Brewster P, Hubert T, Gin J, Davis D, Dobalian A. VA’s experience building capability to conduct outreach to vulnerable patients during emergencies. Innov Aging. 2023;7(suppl 1):209. doi:10.1093/geroni/igad104.0690
As large-scale natural disasters become more common, health care coalitions and the engagement of health systems with local, state, and federal public health departments have effectively bolstered communities’ resilience via collective sharing and distribution of resources.1 These resources may include supplies and the dissemination of emergency information, education, and training.2 The COVID-19 pandemic demonstrated that larger health care systems including hospital networks and nursing homes are better connected to health care coalition resources than smaller, independent systems, such as community home health agencies.3 This leaves some organizations on their own to meet requirements that maintain continuity of care and support their patients and staff throughout a natural disaster.
Home health care workers play important roles in the care of older adults.4 Older adults experience high levels of disability and comorbidities that put them at risk during emergencies; they often require support from paid, family, and neighborhood caregivers to live independently.5 More than 9.3 million US adults receive paid care from 2.6 million home health care workers (eg, home health aides and personal care assistants).6 Many of these individuals are hired through small independent home health agencies (HHAs), while others may work directly for an individual. When neighborhood resources and family caregiving are disrupted during emergencies, the critical services these workers administer become even more essential to ensuring continued access to medical care and social services.
The importance of these services was underscored by the Centers for Medicare and Medicaid Services 2017 inclusion of HHAs in federal emergency preparedness guidelines.7,8 The fractured and decentralized nature of the home health care industry means many HHAs struggle to maintain continuous care during emergencies and protect their staff. HHAs, and health care workers in the home, are often isolated, under-resourced, and disconnected from broader emergency planning efforts. Additionally, home care jobs are largely part-time, unstable, and low paying, making the workers themselves vulnerable during emergencies.3,9-13
This is a significant issue for the Veterans Health Administration (VHA), which annually purchases 10.5 million home health care worker visits for 150,000 veterans from community-based HHAs to enable those individuals to live independently. Figure 1 illustrates the existing structure of directly provided and contracted VHA services for community-dwelling veterans, highlighting the circle of care around the veteran.8,9 Home health care workers anchored health care teams during the COVID-19 pandemic, observing and reporting on patients’ well-being to family caregivers, primary care practitioners, and HHAs. They also provided critical emotional support and companionship to patients isolated from family and friends.9 These workers also exposed themselves and their families to considerable risk and often lacked the protection afforded by personal protective equipment (PPE) in accordance with infection prevention guidance.3,12

Abbreviations: HBPC, home based primary care; HHA, home health agency; VHA, Veterans Health Administration.
aAdapted with permission from Wyte-Lake and Franzosa.8,9
Through a combination of its national and local health care networks, the VHA has a robust and well-positioned emergency infrastructure to supportcommunity-dwelling older adults during disasters.14 This network is supported by the VHA Office of Emergency Management, which shares resources and guidance with local emergency managers at each facility as well as individual programs such as the VHA Home Based Primary Care (HBPC) program, which provides 38,000 seriously ill veterans with home medical visits.15 Working closely with their local and national hospital networks and emergency managers, individual VHA HBPC programs were able to maintain the safety of staff and continuity of care for patients enrolled in HBPC by rapidly administering COVID-19 vaccines to patients, caregivers, and staff, and providing emergency assistance during the 2017 hurricane season.16,17 These efforts were successful because HBPC practitioners and their patients, had access to a level of emergency-related information, resources, and technology that are often out of reach for individual community-based health care practitioners (HCPs). The US Department of Veterans Affairs (VA) also supports local communities through its Fourth Mission, which provides emergency resources to non-VHA health care facilities (ie, hospitals and nursing homes) during national emergencies and natural disasters.17 Although there has been an expansion in the definition of shared resources, such as extending behavioral health support to local communities, the VHA has not historically provided these resources to HHAs.14
This study examines opportunities to leverage VHA emergency management resources to support contracted HHAs and inform other large health system emergency planning efforts. The findings from the exploratory phase are described in this article. We interviewed VHA emergency managers, HBPC and VA staff who coordinate home health care worker services, as well as administrators at contracted HHAs within a Veterans Integrated Services Network (VISN). These findings will inform the second (single-site pilot study) and third (feasibility study) phases. Our intent was to (1) better understand the relationships between VA medical centers (VAMCs) and their contracted HHAs; (2) identify existing VHA emergency protocols to support community-dwelling older adults; and (3) determine opportunities to build on existing infrastructure and relationships to better support contracted HHAs and their staff in emergencies.
Methods
The 18 VISNs act as regional systems of care that are loosely connected to better meet local health needs and maximize access to care. This study was conducted at 6 of 9 VAMCs within VISN 2, the New York/New Jersey VHA Health Care Network.18 VAMCs that serve urban, rural, and mixed urban/rural catchment areas were included.
Each VAMC has an emergency management program led by an emergency manager, an HBPC program led by a program director and medical director, and a community care or purchased care office that has a liaison who manages contracted home health care worker services. The studyfocused on HBPC programs because they are most likely to interact with veterans’ home health care workers in the home and care for community-dwelling veterans during emergencies. Each VHA also contracts with a series of local HHAs that generally have a dedicated staff member who interfaces with the VHA liaison. Our goal was to interview ≥ 1 emergency manager, ≥ 1 HBPC team member, ≥ 1 community care staff person, and ≥ 1 contracted home health agency administrator at each site to gain multiple perspectives from the range of HCPs serving veterans in the community.
Recruitment and Data Collection
The 6 sites were selected in consultation with VISN 2 leadership for their strong HBPC and emergency management programs. To recruit respondents, we contacted VISN and VAMC leads and used our professional networks to identify a sample of multidisciplinary individuals who represent both community care and HBPC programs who were contacted via email.
Since each VAMC is organized differently, we utilized a snowball sampling approach to identify the appropriate contacts.19 At the completion of each interview, we asked the participant to suggest additional contacts and introduce us to any remaining stakeholders (eg, the emergency manager) at that site or colleagues at other VISN facilities. Because roles vary among VAMCs, we contacted the person who most closely resembled the identified role and asked them to direct us to a more appropriate contact, if necessary. We asked community care managers to identify 1 to 2 agencies serving the highest volume of patients who are veterans at their site and requested interviews with those liaisons. This resulted in the recruitment of key stakeholders from 4 teams across the 6 sites (Table).
A semistructured interview guide was jointly developed based on constructs of interest, including relationships within VAMCs and between VAMCs and HHAs; existing emergency protocols and experience during disasters; and suggestions and opportunities for supporting agencies during emergencies and potential barriers. Two researchers (TWL and EF) who were trained in qualitative methods jointly conducted interviews using the interview guide, with 1 researcher leading and another taking notes and asking clarifying questions.
Interviews were conducted virtually via Microsoft Teams with respondents at their work locations between September 2022 and January 2023. Interviews were audio recorded and transcribed and 2 authors (TWL and ESO) reviewed transcripts for accuracy. Interviews averaged 47 minutes in length (range, 20-59).
The study was reviewed and determined to be exempt by institutional review boards at the James J. Peters VAMC and Greater Los Angeles VAMC. We asked participants for verbal consent to participate and preserved their confidentiality.
Analysis
Data were analyzed via an inductive approach, which involves drawing salient themes rather than imposing preconceived theories.20 Three researchers (TWL, EF, and ES) listened to and discussed 2 staff interviews and tagged text with specific codes (eg, communication between the VHA and HHA, internal communication, and barriers to case fulfillment) so the team could selectively return to the interview text for deeper analysis, allowing for the development of a final codebook. The project team synthesized the findings to identify higher-level themes, drawing comparisons across and within the respondent groups, including within and between health care systems. Throughout the analysis, we maintained analytic memos, documented discussions, and engaged in analyst triangulation to ensure trustworthiness.21,22 To ensure the analysis accurately reflected the participants’ understanding, we held 2 virtual member-checking sessions with participants to share preliminary findings and conclusions and solicit feedback. Analysis was conducted using ATLAS.ti version 20.
Results
VHA-based participants described internal emergency management systems that are deployed during a disaster to support patients and staff. Agency participants described their own internal emergency management protocols. Respondents discussed how and when the 2 intersected, as well as opportunities for future mutual support. The analysis identified several themes: (1) relationships between VAMC teams; (2) relationships between VHA and HHAs; (3) VHA and agencies responses during emergencies; (4) receptivity and opportunities for extending VHA resources into the community; and (5) barriers and facilitators to deeper engagement.
Relationships Within VHA (n = 17)
Staff at all VHA sites described close relationships between the internal emergency management and HBPC teams. HBPC teams identified patients who were most at risk during emergencies to triage those with the highest medical needs (eg, patients dependent on home infusion, oxygen, or electronic medical devices) and worked alongside emergency managers to develop plans to continue care during an emergency. HBPC representatives were part of their facilities’ local emergency response committees. Due to this close collaboration, VHA emergency managers were familiar with the needs of homebound veterans and caregivers. “I invite our [HBPC] program manager to attend [committee] meetings and … they’re part of the EOC [emergency operations center]," an emergency manager said. “We work together and I’m constantly in contact with that individual, especially during natural disasters and so forth, to ensure that everybody’s prepared in the community.”
On the other hand, community caremanagers—who described frequent interactions with HBPC teams, largely around coordinating and managing non-VHA home care services—were less likely to have direct relationships with their facility emergency managers. For example, when asked if they had a relationship with their emergency manager, a community care manager admitted, “I [only] know who he is.” They also did not report having structured protocols for veteran outreach during emergencies, “because all those veterans who are receiving [home health care worker] services also belong to a primary care team,” and considered the outreach to be the responsibility of the primary care team and HHA.
Relationships Between the VHA and HHAs (n = 17)
Communication between VAMCs and contracted agencies primarily went through community care managers, who described established long-term relationships with agency administrators. Communication was commonly restricted to operational activities, such as processing referrals and occasional troubleshooting. According to a community care manager most communication is “why haven’t you signed my orders?” There was a general sense from participants that communication was promptly answered, problems were addressed, and professional collegiality existed between the agencies as patients were referred and placed for services. One community care manager reported meeting with agencies regularly, noting, “I talk to them pretty much daily.”
If problems arose, community care managers described themselves as “the liaison” between agencies and VHA HCPs who ordered the referrals. This is particularly the case if the agency needed help finding a VHA clinician or addressing differences in care delivery protocols.
Responding During Emergencies (n = 19)
During emergencies, VHA and agency staff described following their own organization’s protocols and communicating with each other only on a case-by-case basis rather than through formal or systematic channels and had little knowledge of their counterpart’s emergency protocols. Beyond patient care, there was no evidence of information sharing between VHA and agency staff. Regarding sharing information with their local community, an HBPC Program Director said, “it’s almost like the VHA had become siloed” and operated on its own without engaging with community health systems or emergency managers.
Beyond the guidance provided by state departments of public health, HHAs described collaborating with other agencies in their network and relying on their informal professional network to manage the volume of information and updates they followed during emergencies like the COVID-19 pandemic. One agency administrator did not frequently communicate with VHA partners during the pandemic but explained that the local public health department helped work through challenges. However, “we realized pretty quickly they were overloaded and there was only so much they could do.” The agency administrator turned to a “sister agency” and local hospitals, noting, “Wherever you have connections in the field or in the industry, you know you’re going to reach out to people for guidance on policies and… protocol.”
Opportunities for Extending VHA Resources to the Community (n = 16)
All VHA emergency managers were receptive to extending support to community-based HCPS and, in some cases, felt strongly that they were an essential part of veterans’ care networks. Emergency managers offered examples for how they supportedcommunity-based HCPs, such as helping those in the VAMC medical foster home program develop and evaluate emergency plans. Many said they had not explicitly considered HHAs before (Appendix).
Emergency managers also described how supporting community-based HCPs could be considered within the scope of the VHA role and mission, specifically the Fourth Mission. “I think that we should be making our best effort to make sure that we’re also providing that same level [of protection] to the people taking care of the veteran [as our VHA staff],” an emergency manager said. “It’s our responsibility to provide the best for the staff that are going into those homes to take care of that patient.”
In many cases, emergency managers had already developed practical tools that could be easily shared outside the VHA, including weather alerts, trainings, emergency plan templates, and lists of community resources and shelters (Figure 2). A number of these examples built on existing communication channels. One emergency manager said that the extension of resources could be an opportunity to decrease the perceived isolation of home health care workers through regular

Abbreviations: PPE, personal protective equipment; VA, US Department of Veterans Affairs.
On the agency side, participants noted that some HHAs could benefit more from support than others. While some agencies are well staffed and have good protocols and keep up to date, “There are smaller agencies, agencies that are starting up that may not have the resources to just disseminate all the information. Those are the agencies [that] could well benefit from the VHA,” an HBPC medical director explained. Agency administrators suggested several areas where they would welcome support, including a deeper understanding of available community resources and access to PPE for staff. Regarding informational resources, an administrator said, “Anytime we can get information, it’s good to have it come to you and not always have to go out searching for it.”
Barriers and Facilitators to Partnering With Community Agencies (n = 16)
A primary barrier regarding resource sharing was potential misalignment between each organization’s policies. HHAs followed state and federal public health guidelines, which sometimes differed from VHA policies. Given that agencies care for both VHA and non-VHA clients, questions also arose around how agencies would prioritize information from the VHA, if they were already receiving information from other sources. When asked about information sharing, both VHA staff and agencies agreed staff time to support any additional activities should be weighed against the value of the information gained.
Six participants also shared that education around emergency preparedness could be an opportunity to bridge gaps between VAMCs and their surrounding communities.
Two emergency managers noted the need to be sensitive in the way they engaged with partners, respecting and building on the work that agencies were already doing in this area to ensure VHA was seen as a trusted partner and resource rather than trying to impose new policies or rules on community-based HCPs. “I know that like all leadership in various organizations, there’s a little bit of bristling going on when other people try and tell them what to do,” an HBPC medical director said. “However, if it is established that as a sort of greater level like a state level or a federal level, that VHA can be a resource. I think that as long as that’s recognized by their own professional organizations within each state, then I think that that would be a tremendous advantage to many agencies.”
In terms of sharing physical resources, emergency managers raised concerns around potential liability, although they also acknowledged this issue was important enough to think about potential workarounds. As one emergency manager said, “I want to know that my PPE is not compromised in any way shape or form and that I am in charge of that PPE, so to rely upon going to a home and hoping that [the PPE] wasn’t compromised … would kind of make me a little uneasy.” This emergency manager suggested possible solutions, such as creating a sealed PPE package to give directly to an aide.
Discussion
As the prevalence of climate-related disasters increases, the need to ensure the safety and independence of older adults during emergencies grows more urgent. Health systems must think beyond the direct services they provide and consider the community resources upon which their patients rely. While relationships did not formally exist between VHA emergency managers and community home health HCPs in the sample analyzed in this article, there is precedent and interest in supporting contracted home health agencies caring for veterans in the community. Although not historically part of the VA Fourth Mission, creating a pipeline of support for contracted HHAs by leveraging existing relationships and resources can potentially strengthen its mission to protect older veterans in emergencies, help them age safely in place, and provide a model for health systems to collaborate with community-based HCPs around emergency planning and response (Figure 3).23

Existing research on the value of health care coalitions highlights the need for established and growing partnerships with a focus on ensuring they are value-added, which echoes concerns we heard in interviews.24 Investment in community partnerships not only includes sharing supplies but also relying on bidirectional support that can be a trusted form of timely information.1,25 The findings in this study exhibit strong communication practices within the VHA during periods of nonemergency and underscore the untapped value of the pre-existing relationship between VAMCs and their contracted HHAs as an area of potential growth for health care coalitions.
Sharing resources in a way that does not put new demands on partners contributes to the sustainability and value-added nature of coalitions. Examples include establishing new low-investment practices (ie, information sharing) that support capacity and compliance with existing requirements rather than create new responsibilities for either member of the coalition. The relationship between the VHA emergency managers and the VHA HBPC program can act as a guide. The emergency managers interviewed for this study are currently engaged with HBPC programs and therefore understand the needs of homebound older adults and their caregivers. Extending the information already available to the HBPC teams via existing channels strengthens workforce practices and increased security for the shared patient, even without direct relationships between emergency managers and agencies. It is important to understand the limitations of these practices, including concerns around conflicting federal and state mandates, legal concerns around the liability of sharing physical resources (such as PPE), and awareness that the objective is not for the VHA to increase burdens (eg, increasing compliance requirements) but rather to serve as a resource for a mutual population in a shared community.
Offering training and practical resources to HHA home health care workers can help them meet disaster preparedness requirements. This is particularly important considering the growing home care workforce shortages, a topic mentioned by all HBPC and community care participants interviewed for this study.26,27 Home health care workers report feeling underprepared and isolated while on the job in normal conditions, a sentiment exacerbated by the COVID-19 pandemic.3,10 Supporting these individuals may help them feel more prepared and connected to their work, improving stability and quality of care.
While these issues are priorities within the VHA, there is growing recognition at the state and federal level of the importance of including older adults and their HCPs in disaster preparedness and response.5,28 The US Department of Health and Human Services, for example, includes older adults and organizations that serve them on its National Advisory Committee on Seniors and Disasters. The Senate version of the 2023 reauthorization of the Pandemic and All-Hazards Preparedness and Response Act included specific provisions to support community-dwelling older adults and people with disabilities, incorporating funding for community organizations to support continuity of services and avoid institutionalization in an emergency.29 Other proposed legislation includes the Real Emergency Access for Aging and Disability Inclusion for Disasters Act, which would ensure the needs of older adults and people with disabilities are explicitly included in all phases of emergency planning and response.30
The VHA expansion of the its VEText program to include disaster response is an effort to more efficiently extend outreach to older and vulnerable patients who are veterans.31 Given these growing efforts, the VHA and other health systems have an opportunity to expand internal emergency preparedness efforts to ensure the health and safety of individuals living in the community.
Limitations
VISN 2 has been a target of terrorism and other disasters. In addition to the sites being initially recruited for their strong emergency management protocols, this context may have biased respondents who are favorable to extending their resources into the community. At the time of recruitment, contracted HHAs were still experiencing staff shortages due to the COVID-19 pandemic, which limited the ability of agency staff to participate in interviews. Additionally, while the comprehensive exploration of VISN 2 facilities allows for confidence of the organizational structures described, the qualitative research design and small study sample, the study findings cannot be immediately generalized to all VISNs.
Conclusions
Many older veterans increasingly rely on home health care workers to age safely. The VHA, as a large national health care system and leader in emergency preparedness, could play an important role in supporting home health care workers and ameliorating their sense of isolation during emergencies and natural disasters. Leveraging existing resources and relationships may be a low-cost, low-effort opportunity to build higher-level interventions that support the needs of patients. Future research and work in this field, including the authors’ ongoing work, will expand agency participation and engage agency staff in conceptualizing pilot projects to ensure they are viable and feasible for the field.

As large-scale natural disasters become more common, health care coalitions and the engagement of health systems with local, state, and federal public health departments have effectively bolstered communities’ resilience via collective sharing and distribution of resources.1 These resources may include supplies and the dissemination of emergency information, education, and training.2 The COVID-19 pandemic demonstrated that larger health care systems including hospital networks and nursing homes are better connected to health care coalition resources than smaller, independent systems, such as community home health agencies.3 This leaves some organizations on their own to meet requirements that maintain continuity of care and support their patients and staff throughout a natural disaster.
Home health care workers play important roles in the care of older adults.4 Older adults experience high levels of disability and comorbidities that put them at risk during emergencies; they often require support from paid, family, and neighborhood caregivers to live independently.5 More than 9.3 million US adults receive paid care from 2.6 million home health care workers (eg, home health aides and personal care assistants).6 Many of these individuals are hired through small independent home health agencies (HHAs), while others may work directly for an individual. When neighborhood resources and family caregiving are disrupted during emergencies, the critical services these workers administer become even more essential to ensuring continued access to medical care and social services.
The importance of these services was underscored by the Centers for Medicare and Medicaid Services 2017 inclusion of HHAs in federal emergency preparedness guidelines.7,8 The fractured and decentralized nature of the home health care industry means many HHAs struggle to maintain continuous care during emergencies and protect their staff. HHAs, and health care workers in the home, are often isolated, under-resourced, and disconnected from broader emergency planning efforts. Additionally, home care jobs are largely part-time, unstable, and low paying, making the workers themselves vulnerable during emergencies.3,9-13
This is a significant issue for the Veterans Health Administration (VHA), which annually purchases 10.5 million home health care worker visits for 150,000 veterans from community-based HHAs to enable those individuals to live independently. Figure 1 illustrates the existing structure of directly provided and contracted VHA services for community-dwelling veterans, highlighting the circle of care around the veteran.8,9 Home health care workers anchored health care teams during the COVID-19 pandemic, observing and reporting on patients’ well-being to family caregivers, primary care practitioners, and HHAs. They also provided critical emotional support and companionship to patients isolated from family and friends.9 These workers also exposed themselves and their families to considerable risk and often lacked the protection afforded by personal protective equipment (PPE) in accordance with infection prevention guidance.3,12

Abbreviations: HBPC, home based primary care; HHA, home health agency; VHA, Veterans Health Administration.
aAdapted with permission from Wyte-Lake and Franzosa.8,9
Through a combination of its national and local health care networks, the VHA has a robust and well-positioned emergency infrastructure to supportcommunity-dwelling older adults during disasters.14 This network is supported by the VHA Office of Emergency Management, which shares resources and guidance with local emergency managers at each facility as well as individual programs such as the VHA Home Based Primary Care (HBPC) program, which provides 38,000 seriously ill veterans with home medical visits.15 Working closely with their local and national hospital networks and emergency managers, individual VHA HBPC programs were able to maintain the safety of staff and continuity of care for patients enrolled in HBPC by rapidly administering COVID-19 vaccines to patients, caregivers, and staff, and providing emergency assistance during the 2017 hurricane season.16,17 These efforts were successful because HBPC practitioners and their patients, had access to a level of emergency-related information, resources, and technology that are often out of reach for individual community-based health care practitioners (HCPs). The US Department of Veterans Affairs (VA) also supports local communities through its Fourth Mission, which provides emergency resources to non-VHA health care facilities (ie, hospitals and nursing homes) during national emergencies and natural disasters.17 Although there has been an expansion in the definition of shared resources, such as extending behavioral health support to local communities, the VHA has not historically provided these resources to HHAs.14
This study examines opportunities to leverage VHA emergency management resources to support contracted HHAs and inform other large health system emergency planning efforts. The findings from the exploratory phase are described in this article. We interviewed VHA emergency managers, HBPC and VA staff who coordinate home health care worker services, as well as administrators at contracted HHAs within a Veterans Integrated Services Network (VISN). These findings will inform the second (single-site pilot study) and third (feasibility study) phases. Our intent was to (1) better understand the relationships between VA medical centers (VAMCs) and their contracted HHAs; (2) identify existing VHA emergency protocols to support community-dwelling older adults; and (3) determine opportunities to build on existing infrastructure and relationships to better support contracted HHAs and their staff in emergencies.
Methods
The 18 VISNs act as regional systems of care that are loosely connected to better meet local health needs and maximize access to care. This study was conducted at 6 of 9 VAMCs within VISN 2, the New York/New Jersey VHA Health Care Network.18 VAMCs that serve urban, rural, and mixed urban/rural catchment areas were included.
Each VAMC has an emergency management program led by an emergency manager, an HBPC program led by a program director and medical director, and a community care or purchased care office that has a liaison who manages contracted home health care worker services. The studyfocused on HBPC programs because they are most likely to interact with veterans’ home health care workers in the home and care for community-dwelling veterans during emergencies. Each VHA also contracts with a series of local HHAs that generally have a dedicated staff member who interfaces with the VHA liaison. Our goal was to interview ≥ 1 emergency manager, ≥ 1 HBPC team member, ≥ 1 community care staff person, and ≥ 1 contracted home health agency administrator at each site to gain multiple perspectives from the range of HCPs serving veterans in the community.
Recruitment and Data Collection
The 6 sites were selected in consultation with VISN 2 leadership for their strong HBPC and emergency management programs. To recruit respondents, we contacted VISN and VAMC leads and used our professional networks to identify a sample of multidisciplinary individuals who represent both community care and HBPC programs who were contacted via email.
Since each VAMC is organized differently, we utilized a snowball sampling approach to identify the appropriate contacts.19 At the completion of each interview, we asked the participant to suggest additional contacts and introduce us to any remaining stakeholders (eg, the emergency manager) at that site or colleagues at other VISN facilities. Because roles vary among VAMCs, we contacted the person who most closely resembled the identified role and asked them to direct us to a more appropriate contact, if necessary. We asked community care managers to identify 1 to 2 agencies serving the highest volume of patients who are veterans at their site and requested interviews with those liaisons. This resulted in the recruitment of key stakeholders from 4 teams across the 6 sites (Table).
A semistructured interview guide was jointly developed based on constructs of interest, including relationships within VAMCs and between VAMCs and HHAs; existing emergency protocols and experience during disasters; and suggestions and opportunities for supporting agencies during emergencies and potential barriers. Two researchers (TWL and EF) who were trained in qualitative methods jointly conducted interviews using the interview guide, with 1 researcher leading and another taking notes and asking clarifying questions.
Interviews were conducted virtually via Microsoft Teams with respondents at their work locations between September 2022 and January 2023. Interviews were audio recorded and transcribed and 2 authors (TWL and ESO) reviewed transcripts for accuracy. Interviews averaged 47 minutes in length (range, 20-59).
The study was reviewed and determined to be exempt by institutional review boards at the James J. Peters VAMC and Greater Los Angeles VAMC. We asked participants for verbal consent to participate and preserved their confidentiality.
Analysis
Data were analyzed via an inductive approach, which involves drawing salient themes rather than imposing preconceived theories.20 Three researchers (TWL, EF, and ES) listened to and discussed 2 staff interviews and tagged text with specific codes (eg, communication between the VHA and HHA, internal communication, and barriers to case fulfillment) so the team could selectively return to the interview text for deeper analysis, allowing for the development of a final codebook. The project team synthesized the findings to identify higher-level themes, drawing comparisons across and within the respondent groups, including within and between health care systems. Throughout the analysis, we maintained analytic memos, documented discussions, and engaged in analyst triangulation to ensure trustworthiness.21,22 To ensure the analysis accurately reflected the participants’ understanding, we held 2 virtual member-checking sessions with participants to share preliminary findings and conclusions and solicit feedback. Analysis was conducted using ATLAS.ti version 20.
Results
VHA-based participants described internal emergency management systems that are deployed during a disaster to support patients and staff. Agency participants described their own internal emergency management protocols. Respondents discussed how and when the 2 intersected, as well as opportunities for future mutual support. The analysis identified several themes: (1) relationships between VAMC teams; (2) relationships between VHA and HHAs; (3) VHA and agencies responses during emergencies; (4) receptivity and opportunities for extending VHA resources into the community; and (5) barriers and facilitators to deeper engagement.
Relationships Within VHA (n = 17)
Staff at all VHA sites described close relationships between the internal emergency management and HBPC teams. HBPC teams identified patients who were most at risk during emergencies to triage those with the highest medical needs (eg, patients dependent on home infusion, oxygen, or electronic medical devices) and worked alongside emergency managers to develop plans to continue care during an emergency. HBPC representatives were part of their facilities’ local emergency response committees. Due to this close collaboration, VHA emergency managers were familiar with the needs of homebound veterans and caregivers. “I invite our [HBPC] program manager to attend [committee] meetings and … they’re part of the EOC [emergency operations center]," an emergency manager said. “We work together and I’m constantly in contact with that individual, especially during natural disasters and so forth, to ensure that everybody’s prepared in the community.”
On the other hand, community caremanagers—who described frequent interactions with HBPC teams, largely around coordinating and managing non-VHA home care services—were less likely to have direct relationships with their facility emergency managers. For example, when asked if they had a relationship with their emergency manager, a community care manager admitted, “I [only] know who he is.” They also did not report having structured protocols for veteran outreach during emergencies, “because all those veterans who are receiving [home health care worker] services also belong to a primary care team,” and considered the outreach to be the responsibility of the primary care team and HHA.
Relationships Between the VHA and HHAs (n = 17)
Communication between VAMCs and contracted agencies primarily went through community care managers, who described established long-term relationships with agency administrators. Communication was commonly restricted to operational activities, such as processing referrals and occasional troubleshooting. According to a community care manager most communication is “why haven’t you signed my orders?” There was a general sense from participants that communication was promptly answered, problems were addressed, and professional collegiality existed between the agencies as patients were referred and placed for services. One community care manager reported meeting with agencies regularly, noting, “I talk to them pretty much daily.”
If problems arose, community care managers described themselves as “the liaison” between agencies and VHA HCPs who ordered the referrals. This is particularly the case if the agency needed help finding a VHA clinician or addressing differences in care delivery protocols.
Responding During Emergencies (n = 19)
During emergencies, VHA and agency staff described following their own organization’s protocols and communicating with each other only on a case-by-case basis rather than through formal or systematic channels and had little knowledge of their counterpart’s emergency protocols. Beyond patient care, there was no evidence of information sharing between VHA and agency staff. Regarding sharing information with their local community, an HBPC Program Director said, “it’s almost like the VHA had become siloed” and operated on its own without engaging with community health systems or emergency managers.
Beyond the guidance provided by state departments of public health, HHAs described collaborating with other agencies in their network and relying on their informal professional network to manage the volume of information and updates they followed during emergencies like the COVID-19 pandemic. One agency administrator did not frequently communicate with VHA partners during the pandemic but explained that the local public health department helped work through challenges. However, “we realized pretty quickly they were overloaded and there was only so much they could do.” The agency administrator turned to a “sister agency” and local hospitals, noting, “Wherever you have connections in the field or in the industry, you know you’re going to reach out to people for guidance on policies and… protocol.”
Opportunities for Extending VHA Resources to the Community (n = 16)
All VHA emergency managers were receptive to extending support to community-based HCPS and, in some cases, felt strongly that they were an essential part of veterans’ care networks. Emergency managers offered examples for how they supportedcommunity-based HCPs, such as helping those in the VAMC medical foster home program develop and evaluate emergency plans. Many said they had not explicitly considered HHAs before (Appendix).
Emergency managers also described how supporting community-based HCPs could be considered within the scope of the VHA role and mission, specifically the Fourth Mission. “I think that we should be making our best effort to make sure that we’re also providing that same level [of protection] to the people taking care of the veteran [as our VHA staff],” an emergency manager said. “It’s our responsibility to provide the best for the staff that are going into those homes to take care of that patient.”
In many cases, emergency managers had already developed practical tools that could be easily shared outside the VHA, including weather alerts, trainings, emergency plan templates, and lists of community resources and shelters (Figure 2). A number of these examples built on existing communication channels. One emergency manager said that the extension of resources could be an opportunity to decrease the perceived isolation of home health care workers through regular

Abbreviations: PPE, personal protective equipment; VA, US Department of Veterans Affairs.
On the agency side, participants noted that some HHAs could benefit more from support than others. While some agencies are well staffed and have good protocols and keep up to date, “There are smaller agencies, agencies that are starting up that may not have the resources to just disseminate all the information. Those are the agencies [that] could well benefit from the VHA,” an HBPC medical director explained. Agency administrators suggested several areas where they would welcome support, including a deeper understanding of available community resources and access to PPE for staff. Regarding informational resources, an administrator said, “Anytime we can get information, it’s good to have it come to you and not always have to go out searching for it.”
Barriers and Facilitators to Partnering With Community Agencies (n = 16)
A primary barrier regarding resource sharing was potential misalignment between each organization’s policies. HHAs followed state and federal public health guidelines, which sometimes differed from VHA policies. Given that agencies care for both VHA and non-VHA clients, questions also arose around how agencies would prioritize information from the VHA, if they were already receiving information from other sources. When asked about information sharing, both VHA staff and agencies agreed staff time to support any additional activities should be weighed against the value of the information gained.
Six participants also shared that education around emergency preparedness could be an opportunity to bridge gaps between VAMCs and their surrounding communities.
Two emergency managers noted the need to be sensitive in the way they engaged with partners, respecting and building on the work that agencies were already doing in this area to ensure VHA was seen as a trusted partner and resource rather than trying to impose new policies or rules on community-based HCPs. “I know that like all leadership in various organizations, there’s a little bit of bristling going on when other people try and tell them what to do,” an HBPC medical director said. “However, if it is established that as a sort of greater level like a state level or a federal level, that VHA can be a resource. I think that as long as that’s recognized by their own professional organizations within each state, then I think that that would be a tremendous advantage to many agencies.”
In terms of sharing physical resources, emergency managers raised concerns around potential liability, although they also acknowledged this issue was important enough to think about potential workarounds. As one emergency manager said, “I want to know that my PPE is not compromised in any way shape or form and that I am in charge of that PPE, so to rely upon going to a home and hoping that [the PPE] wasn’t compromised … would kind of make me a little uneasy.” This emergency manager suggested possible solutions, such as creating a sealed PPE package to give directly to an aide.
Discussion
As the prevalence of climate-related disasters increases, the need to ensure the safety and independence of older adults during emergencies grows more urgent. Health systems must think beyond the direct services they provide and consider the community resources upon which their patients rely. While relationships did not formally exist between VHA emergency managers and community home health HCPs in the sample analyzed in this article, there is precedent and interest in supporting contracted home health agencies caring for veterans in the community. Although not historically part of the VA Fourth Mission, creating a pipeline of support for contracted HHAs by leveraging existing relationships and resources can potentially strengthen its mission to protect older veterans in emergencies, help them age safely in place, and provide a model for health systems to collaborate with community-based HCPs around emergency planning and response (Figure 3).23

Existing research on the value of health care coalitions highlights the need for established and growing partnerships with a focus on ensuring they are value-added, which echoes concerns we heard in interviews.24 Investment in community partnerships not only includes sharing supplies but also relying on bidirectional support that can be a trusted form of timely information.1,25 The findings in this study exhibit strong communication practices within the VHA during periods of nonemergency and underscore the untapped value of the pre-existing relationship between VAMCs and their contracted HHAs as an area of potential growth for health care coalitions.
Sharing resources in a way that does not put new demands on partners contributes to the sustainability and value-added nature of coalitions. Examples include establishing new low-investment practices (ie, information sharing) that support capacity and compliance with existing requirements rather than create new responsibilities for either member of the coalition. The relationship between the VHA emergency managers and the VHA HBPC program can act as a guide. The emergency managers interviewed for this study are currently engaged with HBPC programs and therefore understand the needs of homebound older adults and their caregivers. Extending the information already available to the HBPC teams via existing channels strengthens workforce practices and increased security for the shared patient, even without direct relationships between emergency managers and agencies. It is important to understand the limitations of these practices, including concerns around conflicting federal and state mandates, legal concerns around the liability of sharing physical resources (such as PPE), and awareness that the objective is not for the VHA to increase burdens (eg, increasing compliance requirements) but rather to serve as a resource for a mutual population in a shared community.
Offering training and practical resources to HHA home health care workers can help them meet disaster preparedness requirements. This is particularly important considering the growing home care workforce shortages, a topic mentioned by all HBPC and community care participants interviewed for this study.26,27 Home health care workers report feeling underprepared and isolated while on the job in normal conditions, a sentiment exacerbated by the COVID-19 pandemic.3,10 Supporting these individuals may help them feel more prepared and connected to their work, improving stability and quality of care.
While these issues are priorities within the VHA, there is growing recognition at the state and federal level of the importance of including older adults and their HCPs in disaster preparedness and response.5,28 The US Department of Health and Human Services, for example, includes older adults and organizations that serve them on its National Advisory Committee on Seniors and Disasters. The Senate version of the 2023 reauthorization of the Pandemic and All-Hazards Preparedness and Response Act included specific provisions to support community-dwelling older adults and people with disabilities, incorporating funding for community organizations to support continuity of services and avoid institutionalization in an emergency.29 Other proposed legislation includes the Real Emergency Access for Aging and Disability Inclusion for Disasters Act, which would ensure the needs of older adults and people with disabilities are explicitly included in all phases of emergency planning and response.30
The VHA expansion of the its VEText program to include disaster response is an effort to more efficiently extend outreach to older and vulnerable patients who are veterans.31 Given these growing efforts, the VHA and other health systems have an opportunity to expand internal emergency preparedness efforts to ensure the health and safety of individuals living in the community.
Limitations
VISN 2 has been a target of terrorism and other disasters. In addition to the sites being initially recruited for their strong emergency management protocols, this context may have biased respondents who are favorable to extending their resources into the community. At the time of recruitment, contracted HHAs were still experiencing staff shortages due to the COVID-19 pandemic, which limited the ability of agency staff to participate in interviews. Additionally, while the comprehensive exploration of VISN 2 facilities allows for confidence of the organizational structures described, the qualitative research design and small study sample, the study findings cannot be immediately generalized to all VISNs.
Conclusions
Many older veterans increasingly rely on home health care workers to age safely. The VHA, as a large national health care system and leader in emergency preparedness, could play an important role in supporting home health care workers and ameliorating their sense of isolation during emergencies and natural disasters. Leveraging existing resources and relationships may be a low-cost, low-effort opportunity to build higher-level interventions that support the needs of patients. Future research and work in this field, including the authors’ ongoing work, will expand agency participation and engage agency staff in conceptualizing pilot projects to ensure they are viable and feasible for the field.

- Barnett DJ, Knieser L, Errett NA, Rosenblum AJ, Seshamani M, Kirsch TD. Reexamining health-care coalitions in light of COVID-19. Disaster Med public Health Prep. 2022;16(3):859-863. doi:10.1017/dmp.2020.431
- Wulff K, Donato D, Lurie N. What is health resilience and how can we build it? Annu Rev Public Health. 2015;36:361-374. doi:10.1146/annurev-publhealth-031914-122829
- Franzosa E, Wyte-Lake T, Tsui EK, Reckrey JM, Sterling MR. Essential but excluded: building disaster preparedness capacity for home health care workers and home care agencies. J Am Med Dir Assoc. 2022;23(12):1990-1996. doi:10.1016/j.jamda.2022.09.012
- Miner S, Masci L, Chimenti C, Rin N, Mann A, Noonan B. An outreach phone call project: using home health to reach isolated community dwelling adults during the COVID 19 lockdown. J Community Health. 2022;47(2):266-272. doi:10.1007/s10900-021-01044-6
- National Institute on Aging. Protecting older adults from the effects of natural disasters and extreme weather. October 18, 2022. Accessed August 19, 2024. https://www.nia.nih.gov/news/protecting-older-adults-effects-natural-disasters-and-extreme-weather
- PHI. Direct Care Workers in the United States: Key Facts. September 7, 2021. Accessed August 19, 2024. https://www.phinational.org/resource/direct-care-workers-in-the-united-states-key-facts-2/
- Centers for Medicare & Medicaid Services. Emergency Preparedness Rule. September 8, 2016. Updated September 6, 2023. Accessed August 19, 2024. https://www.cms.gov/medicare/health-safety-standards/quality-safety-oversight-emergency-preparedness/emergency-preparedness-rule
- Wyte-Lake T, Claver M, Tubbesing S, Davis D, Dobalian A. Development of a home health patient assessment tool for disaster planning. Gerontology. 2019;65(4):353-361. doi:10.1159/000494971
- Franzosa E, Judon KM, Gottesman EM, et al. Home health aides’ increased role in supporting older veterans and primary healthcare teams during COVID-19: a qualitative analysis. J Gen Intern Med. 2022;37(8):1830-1837. doi:10.1007/s11606-021-07271-w
- Franzosa E, Tsui EK, Baron S. “Who’s caring for us?”: understanding and addressing the effects of emotional labor on home health aides’ well-being. Gerontologist. 2019;59(6):1055-1064. doi:10.1093/geront/gny099
- Osakwe ZT, Osborne JC, Samuel T, et al. All alone: a qualitative study of home health aides’ experiences during the COVID-19 pandemic in New York. Am J Infect Control. 2021;49(11):1362-1368. doi:10.1016/j.ajic.2021.08.004
- Feldman PH, Russell D, Onorato N, et al. Ensuring the safety of the home health aide workforce and the continuation of essential patient care through sustainable pandemic preparedness. July 2022. Accessed August 19, 2024. https://www.vnshealth.org/wp-content/uploads/2022/08/Pandemic_Preparedness_IB_07_21_22.pdf
- Sterling MR, Tseng E, Poon A, et al. Experiences of home health care workers in New York City during the coronavirus disease 2019 pandemic: a qualitative analysis. JAMA Internal Med. 2020;180(11):1453-1459. doi:10.1001/jamainternmed.2020.3930
- Wyte-Lake T, Schmitz S, Kornegay RJ, Acevedo F, Dobalian A. Three case studies of community behavioral health support from the US Department of Veterans Affairs after disasters. BMC Public Health. 2021;21(1):639. doi:10.1186/s12889-021-10650-x
- Beales JL, Edes T. Veteran’s affairs home based primary care. Clin Geriatr Med. 2009;25(1):149-ix. doi:10.1016/j.cger.2008.11.002
- Wyte-Lake T, Manheim C, Gillespie SM, Dobalian A, Haverhals LM. COVID-19 vaccination in VA home based primary care: experience of interdisciplinary team members. J Am Med Dir Assoc. 2022;23(6):917-922. doi:10.1016/j.jamda.2022.03.014
- Wyte-Lake T, Schmitz S, Cosme Torres-Sabater R, Dobalian A. Case study of VA Caribbean Healthcare System’s community response to Hurricane Maria. J Emerg Manag. 2022;19(8):189-199. doi:10.5055/jem.0536
- US Department of Veterans Affairs. New York/New Jersey VA Health Care Network, VISN 2 Locations. Updated January 3, 2024. Accessed August 19, 2024. https://www.visn2.va.gov/visn2/facilities.asp
- Noy C. Sampling knowledge: the hermeneutics of snowball sampling in qualitative research. Int J Soc Res Methodol. 2008;11(4):327-344. doi:10.1080/13645570701401305
- Ritchie J, Lewis J, Nicholls CM, Ormston R, eds. Qualitative Research Practice: A Guide for Social Science Students and Researchers. 2nd ed. Sage; 2013.
- Morrow SL. Quality and trustworthiness in qualitative research in counseling psychology. J Couns Psychol. 2005;52(2):250-260. doi:10.1037/0022-0167.52.2.250
- Rolfe G. Validity, trustworthiness and rigour: quality and the idea of qualitative research. J Adv Nurs. 2006;53(3):304-310. doi:10.1111/j.1365-2648.2006.03727.x
- Schmitz S, Wyte-Lake T, Dobalian A. Facilitators and barriers to preparedness partnerships: a veterans affairs medical center perspective. Disaster Med Public Health Prep. 2018;12(4):431-436. doi:10.1017/dmp.2017.92
- Koch AE, Bohn J, Corvin JA, Seaberg J. Maturing into high-functioning health-care coalitions: a qualitative Nationwide study of emergency preparedness and response leadership. Disaster Med Public Health Prep. 2022;17:e111. doi:10.1017/dmp.2022.13
- Lin JS, Webber EM, Bean SI, Martin AM, Davies MC. Rapid evidence review: policy actions for the integration of public health and health care in the United States. Front Public Health. 2023;11:1098431. doi:10.3389/fpubh.2023.1098431
- Watts MOM, Burns A, Ammula M. Ongoing impacts of the pandemic on medicaid home & community-based services (HCBS) programs: findings from a 50-state survey. November 28, 2022. Accessed August 19, 2024. https://www.kff.org/medicaid/issue-brief/ongoing-impacts-of-the-pandemic-on-medicaid-home-community-based-services-hcbs-programs-findings-from-a-50-state-survey/
- Kreider AR, Werner RM. The home care workforce has not kept pace with growth in home and community-based services. Health Aff (Millwood). 2023;42(5):650-657. doi:10.1377/hlthaff.2022.01351
- FEMA introduces disaster preparedness guide for older adults. News release. FEMA. September 20, 2023. Accessed August 19, 2024. https://www.fema.gov/press-release/20230920/fema-introduces-disaster-preparedness-guide-older-adults
- Pandemic and All-Hazards Preparedness and Response Act, S 2333, 118th Cong, 1st Sess (2023). https://www.congress.gov/bill/118th-congress/senate-bill/2333/text
- REAADI for Disasters Act, HR 2371, 118th Cong, 1st Sess (2023). https://www.congress.gov/bill/118th-congress/house-bill/2371
- Wyte-Lake T, Brewster P, Hubert T, Gin J, Davis D, Dobalian A. VA’s experience building capability to conduct outreach to vulnerable patients during emergencies. Innov Aging. 2023;7(suppl 1):209. doi:10.1093/geroni/igad104.0690
- Barnett DJ, Knieser L, Errett NA, Rosenblum AJ, Seshamani M, Kirsch TD. Reexamining health-care coalitions in light of COVID-19. Disaster Med public Health Prep. 2022;16(3):859-863. doi:10.1017/dmp.2020.431
- Wulff K, Donato D, Lurie N. What is health resilience and how can we build it? Annu Rev Public Health. 2015;36:361-374. doi:10.1146/annurev-publhealth-031914-122829
- Franzosa E, Wyte-Lake T, Tsui EK, Reckrey JM, Sterling MR. Essential but excluded: building disaster preparedness capacity for home health care workers and home care agencies. J Am Med Dir Assoc. 2022;23(12):1990-1996. doi:10.1016/j.jamda.2022.09.012
- Miner S, Masci L, Chimenti C, Rin N, Mann A, Noonan B. An outreach phone call project: using home health to reach isolated community dwelling adults during the COVID 19 lockdown. J Community Health. 2022;47(2):266-272. doi:10.1007/s10900-021-01044-6
- National Institute on Aging. Protecting older adults from the effects of natural disasters and extreme weather. October 18, 2022. Accessed August 19, 2024. https://www.nia.nih.gov/news/protecting-older-adults-effects-natural-disasters-and-extreme-weather
- PHI. Direct Care Workers in the United States: Key Facts. September 7, 2021. Accessed August 19, 2024. https://www.phinational.org/resource/direct-care-workers-in-the-united-states-key-facts-2/
- Centers for Medicare & Medicaid Services. Emergency Preparedness Rule. September 8, 2016. Updated September 6, 2023. Accessed August 19, 2024. https://www.cms.gov/medicare/health-safety-standards/quality-safety-oversight-emergency-preparedness/emergency-preparedness-rule
- Wyte-Lake T, Claver M, Tubbesing S, Davis D, Dobalian A. Development of a home health patient assessment tool for disaster planning. Gerontology. 2019;65(4):353-361. doi:10.1159/000494971
- Franzosa E, Judon KM, Gottesman EM, et al. Home health aides’ increased role in supporting older veterans and primary healthcare teams during COVID-19: a qualitative analysis. J Gen Intern Med. 2022;37(8):1830-1837. doi:10.1007/s11606-021-07271-w
- Franzosa E, Tsui EK, Baron S. “Who’s caring for us?”: understanding and addressing the effects of emotional labor on home health aides’ well-being. Gerontologist. 2019;59(6):1055-1064. doi:10.1093/geront/gny099
- Osakwe ZT, Osborne JC, Samuel T, et al. All alone: a qualitative study of home health aides’ experiences during the COVID-19 pandemic in New York. Am J Infect Control. 2021;49(11):1362-1368. doi:10.1016/j.ajic.2021.08.004
- Feldman PH, Russell D, Onorato N, et al. Ensuring the safety of the home health aide workforce and the continuation of essential patient care through sustainable pandemic preparedness. July 2022. Accessed August 19, 2024. https://www.vnshealth.org/wp-content/uploads/2022/08/Pandemic_Preparedness_IB_07_21_22.pdf
- Sterling MR, Tseng E, Poon A, et al. Experiences of home health care workers in New York City during the coronavirus disease 2019 pandemic: a qualitative analysis. JAMA Internal Med. 2020;180(11):1453-1459. doi:10.1001/jamainternmed.2020.3930
- Wyte-Lake T, Schmitz S, Kornegay RJ, Acevedo F, Dobalian A. Three case studies of community behavioral health support from the US Department of Veterans Affairs after disasters. BMC Public Health. 2021;21(1):639. doi:10.1186/s12889-021-10650-x
- Beales JL, Edes T. Veteran’s affairs home based primary care. Clin Geriatr Med. 2009;25(1):149-ix. doi:10.1016/j.cger.2008.11.002
- Wyte-Lake T, Manheim C, Gillespie SM, Dobalian A, Haverhals LM. COVID-19 vaccination in VA home based primary care: experience of interdisciplinary team members. J Am Med Dir Assoc. 2022;23(6):917-922. doi:10.1016/j.jamda.2022.03.014
- Wyte-Lake T, Schmitz S, Cosme Torres-Sabater R, Dobalian A. Case study of VA Caribbean Healthcare System’s community response to Hurricane Maria. J Emerg Manag. 2022;19(8):189-199. doi:10.5055/jem.0536
- US Department of Veterans Affairs. New York/New Jersey VA Health Care Network, VISN 2 Locations. Updated January 3, 2024. Accessed August 19, 2024. https://www.visn2.va.gov/visn2/facilities.asp
- Noy C. Sampling knowledge: the hermeneutics of snowball sampling in qualitative research. Int J Soc Res Methodol. 2008;11(4):327-344. doi:10.1080/13645570701401305
- Ritchie J, Lewis J, Nicholls CM, Ormston R, eds. Qualitative Research Practice: A Guide for Social Science Students and Researchers. 2nd ed. Sage; 2013.
- Morrow SL. Quality and trustworthiness in qualitative research in counseling psychology. J Couns Psychol. 2005;52(2):250-260. doi:10.1037/0022-0167.52.2.250
- Rolfe G. Validity, trustworthiness and rigour: quality and the idea of qualitative research. J Adv Nurs. 2006;53(3):304-310. doi:10.1111/j.1365-2648.2006.03727.x
- Schmitz S, Wyte-Lake T, Dobalian A. Facilitators and barriers to preparedness partnerships: a veterans affairs medical center perspective. Disaster Med Public Health Prep. 2018;12(4):431-436. doi:10.1017/dmp.2017.92
- Koch AE, Bohn J, Corvin JA, Seaberg J. Maturing into high-functioning health-care coalitions: a qualitative Nationwide study of emergency preparedness and response leadership. Disaster Med Public Health Prep. 2022;17:e111. doi:10.1017/dmp.2022.13
- Lin JS, Webber EM, Bean SI, Martin AM, Davies MC. Rapid evidence review: policy actions for the integration of public health and health care in the United States. Front Public Health. 2023;11:1098431. doi:10.3389/fpubh.2023.1098431
- Watts MOM, Burns A, Ammula M. Ongoing impacts of the pandemic on medicaid home & community-based services (HCBS) programs: findings from a 50-state survey. November 28, 2022. Accessed August 19, 2024. https://www.kff.org/medicaid/issue-brief/ongoing-impacts-of-the-pandemic-on-medicaid-home-community-based-services-hcbs-programs-findings-from-a-50-state-survey/
- Kreider AR, Werner RM. The home care workforce has not kept pace with growth in home and community-based services. Health Aff (Millwood). 2023;42(5):650-657. doi:10.1377/hlthaff.2022.01351
- FEMA introduces disaster preparedness guide for older adults. News release. FEMA. September 20, 2023. Accessed August 19, 2024. https://www.fema.gov/press-release/20230920/fema-introduces-disaster-preparedness-guide-older-adults
- Pandemic and All-Hazards Preparedness and Response Act, S 2333, 118th Cong, 1st Sess (2023). https://www.congress.gov/bill/118th-congress/senate-bill/2333/text
- REAADI for Disasters Act, HR 2371, 118th Cong, 1st Sess (2023). https://www.congress.gov/bill/118th-congress/house-bill/2371
- Wyte-Lake T, Brewster P, Hubert T, Gin J, Davis D, Dobalian A. VA’s experience building capability to conduct outreach to vulnerable patients during emergencies. Innov Aging. 2023;7(suppl 1):209. doi:10.1093/geroni/igad104.0690
Over-the-Counter UTI Products: What Primary Care Clinicians Need to Know
When it comes to managing urinary tract infections (UTIs), patients often turn to over-the-counter (OTC) products in search of quick relief. However, recent research suggests some products promise more than they can deliver and can vary widely in price and ingredients. For primary care clinicians, understanding these differences could make all the difference in offering effective, cost-conscious advice to patients.
Researchers from the University of Wisconsin analyzed OTC products marketed for urinary tract health in three major US drugstores and found significant price variations and a wide array of active ingredients.
Their study, presented at the American Urogynecologic Society’s PFD Week conference, found that the price of OTC products fluctuates dramatically. Phenazopyridine hydrochloride, commonly used for UTI symptom relief, ranged from $0.17 to $0.83 per tablet, the study found. Methenamine/sodium salicylate combinations, which are marketed for UTI prevention, varied from $0.13 to $0.33 per tablet. Cranberry supplements — often touted for their preventive benefits — showed the most extreme price range, from as low as $0.07 to as high as $1.00 per serving.
The study also looked into the ingredients, which were categorized into five main groups: Phenazopyridine hydrochloride, methenamine/sodium salicylate, cranberry supplements, D-mannose, and cranberry/D-mannose combinations.
These ingredients vary not only in price but also in the strength of scientific evidence supporting their use.
The researchers concluded:
- Phenazopyridine hydrochloride offers effective symptom relief but is not a UTI treatment.
- Methenamine/sodium salicylate shows potential for preventing recurrent UTIs in certain patients.
- Cranberry supplements have limited evidence for preventing UTIs, with no proof they treat infections.
- D-mannose has shown promise for short-term use in preventing recurrent UTIs, though more research is needed to weigh its effectiveness in the long run.
“No OTC product within its respective category is superior to another,” said Ushma J. Patel, MD, a fellow in Urogynecology and Reconstructive Pelvic Surgery at the University of Wisconsin School of Medicine and Public Health, Madison, and lead author of the study.
Patel and her coresearcher also found that many products are falsely marketed as treatments for UTI.
“The products in each type of category are for symptom relief or UTI prevention — not treatment,” said Patel. “These products within the categories described are interchangeable, and consumers should make cost-effective choices as no product is superior to another within its respective category.”
This presents the opportunity for clinicians to guide individuals to pick the right products while explaining that symptom relief doesn’t necessarily mean an infection is being treated, Patel said.
Indeed, Patel proposed that clinicians utilize a summary table created from their findings to offer patients vetted information about OTC UTI products.
And, while OTC products can provide benefits, they should not replace proper medical evaluation and treatment of UTIs.
“If patients are experiencing ongoing symptoms or develop new-onset symptoms despite trialing an over-the-counter product, they should contact a healthcare provider,” said Patel. “OTC products can provide symptom relief until patients are able to see a healthcare provider.”
The researchers reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
When it comes to managing urinary tract infections (UTIs), patients often turn to over-the-counter (OTC) products in search of quick relief. However, recent research suggests some products promise more than they can deliver and can vary widely in price and ingredients. For primary care clinicians, understanding these differences could make all the difference in offering effective, cost-conscious advice to patients.
Researchers from the University of Wisconsin analyzed OTC products marketed for urinary tract health in three major US drugstores and found significant price variations and a wide array of active ingredients.
Their study, presented at the American Urogynecologic Society’s PFD Week conference, found that the price of OTC products fluctuates dramatically. Phenazopyridine hydrochloride, commonly used for UTI symptom relief, ranged from $0.17 to $0.83 per tablet, the study found. Methenamine/sodium salicylate combinations, which are marketed for UTI prevention, varied from $0.13 to $0.33 per tablet. Cranberry supplements — often touted for their preventive benefits — showed the most extreme price range, from as low as $0.07 to as high as $1.00 per serving.
The study also looked into the ingredients, which were categorized into five main groups: Phenazopyridine hydrochloride, methenamine/sodium salicylate, cranberry supplements, D-mannose, and cranberry/D-mannose combinations.
These ingredients vary not only in price but also in the strength of scientific evidence supporting their use.
The researchers concluded:
- Phenazopyridine hydrochloride offers effective symptom relief but is not a UTI treatment.
- Methenamine/sodium salicylate shows potential for preventing recurrent UTIs in certain patients.
- Cranberry supplements have limited evidence for preventing UTIs, with no proof they treat infections.
- D-mannose has shown promise for short-term use in preventing recurrent UTIs, though more research is needed to weigh its effectiveness in the long run.
“No OTC product within its respective category is superior to another,” said Ushma J. Patel, MD, a fellow in Urogynecology and Reconstructive Pelvic Surgery at the University of Wisconsin School of Medicine and Public Health, Madison, and lead author of the study.
Patel and her coresearcher also found that many products are falsely marketed as treatments for UTI.
“The products in each type of category are for symptom relief or UTI prevention — not treatment,” said Patel. “These products within the categories described are interchangeable, and consumers should make cost-effective choices as no product is superior to another within its respective category.”
This presents the opportunity for clinicians to guide individuals to pick the right products while explaining that symptom relief doesn’t necessarily mean an infection is being treated, Patel said.
Indeed, Patel proposed that clinicians utilize a summary table created from their findings to offer patients vetted information about OTC UTI products.
And, while OTC products can provide benefits, they should not replace proper medical evaluation and treatment of UTIs.
“If patients are experiencing ongoing symptoms or develop new-onset symptoms despite trialing an over-the-counter product, they should contact a healthcare provider,” said Patel. “OTC products can provide symptom relief until patients are able to see a healthcare provider.”
The researchers reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
When it comes to managing urinary tract infections (UTIs), patients often turn to over-the-counter (OTC) products in search of quick relief. However, recent research suggests some products promise more than they can deliver and can vary widely in price and ingredients. For primary care clinicians, understanding these differences could make all the difference in offering effective, cost-conscious advice to patients.
Researchers from the University of Wisconsin analyzed OTC products marketed for urinary tract health in three major US drugstores and found significant price variations and a wide array of active ingredients.
Their study, presented at the American Urogynecologic Society’s PFD Week conference, found that the price of OTC products fluctuates dramatically. Phenazopyridine hydrochloride, commonly used for UTI symptom relief, ranged from $0.17 to $0.83 per tablet, the study found. Methenamine/sodium salicylate combinations, which are marketed for UTI prevention, varied from $0.13 to $0.33 per tablet. Cranberry supplements — often touted for their preventive benefits — showed the most extreme price range, from as low as $0.07 to as high as $1.00 per serving.
The study also looked into the ingredients, which were categorized into five main groups: Phenazopyridine hydrochloride, methenamine/sodium salicylate, cranberry supplements, D-mannose, and cranberry/D-mannose combinations.
These ingredients vary not only in price but also in the strength of scientific evidence supporting their use.
The researchers concluded:
- Phenazopyridine hydrochloride offers effective symptom relief but is not a UTI treatment.
- Methenamine/sodium salicylate shows potential for preventing recurrent UTIs in certain patients.
- Cranberry supplements have limited evidence for preventing UTIs, with no proof they treat infections.
- D-mannose has shown promise for short-term use in preventing recurrent UTIs, though more research is needed to weigh its effectiveness in the long run.
“No OTC product within its respective category is superior to another,” said Ushma J. Patel, MD, a fellow in Urogynecology and Reconstructive Pelvic Surgery at the University of Wisconsin School of Medicine and Public Health, Madison, and lead author of the study.
Patel and her coresearcher also found that many products are falsely marketed as treatments for UTI.
“The products in each type of category are for symptom relief or UTI prevention — not treatment,” said Patel. “These products within the categories described are interchangeable, and consumers should make cost-effective choices as no product is superior to another within its respective category.”
This presents the opportunity for clinicians to guide individuals to pick the right products while explaining that symptom relief doesn’t necessarily mean an infection is being treated, Patel said.
Indeed, Patel proposed that clinicians utilize a summary table created from their findings to offer patients vetted information about OTC UTI products.
And, while OTC products can provide benefits, they should not replace proper medical evaluation and treatment of UTIs.
“If patients are experiencing ongoing symptoms or develop new-onset symptoms despite trialing an over-the-counter product, they should contact a healthcare provider,” said Patel. “OTC products can provide symptom relief until patients are able to see a healthcare provider.”
The researchers reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM PFD WEEK 2024
FDA OKs New Drug for Urinary Tract Infections
The Food and Drug Administration (FDA) has approved Orlynvah, a new oral treatment for uncomplicated urinary tract infections (UTIs) in women who have limited options for effective antibiotic therapy.
Uncomplicated UTIs are bladder infections that typically affect women who don’t have other issues like kidney disease or urinary tract abnormalities. These infections are common, affecting around half of all women at least once in their lives.
Treating UTIs can be difficult when standard antibiotics don’t work well, often because of antibiotic resistance or certain health conditions. Orlynvah offers a promising new option by combining two drugs, sulopenem etzadroxil and probenecid, in one oral tablet. This combination helps keep the antibiotic in the body longer, making it work better, especially against bacteria that resist traditional treatments. Orlynvah is specifically approved to target infections from bacteria like Escherichia coli, Klebsiella pneumoniae, and Proteus mirabilis, which can be harder to treat.
Marjorie Golden, MD, an infectious disease specialist at Yale New Haven Hospital in Connecticut, described Orlynvah as a much-needed alternative for women struggling with difficult-to-treat UTIs.
“Orlynvah has the potential to be an important treatment option for those who need it,” she said in a news release from Iterum Therapeutics, the drug’s maker.
The FDA approved Orlynvah based on two large clinical trials involving over 3,800 women. In these studies, Orlynvah worked as well as or better than antibiotics like ciprofloxacin and Augmentin. It was generally well-tolerated, though common side effects included diarrhea, nausea, yeast infections, and headaches.
The FDA advises people to discuss their medical history with their doctor before taking Orlynvah, especially if they have conditions like gout, kidney stones, or allergies to other antibiotics.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration (FDA) has approved Orlynvah, a new oral treatment for uncomplicated urinary tract infections (UTIs) in women who have limited options for effective antibiotic therapy.
Uncomplicated UTIs are bladder infections that typically affect women who don’t have other issues like kidney disease or urinary tract abnormalities. These infections are common, affecting around half of all women at least once in their lives.
Treating UTIs can be difficult when standard antibiotics don’t work well, often because of antibiotic resistance or certain health conditions. Orlynvah offers a promising new option by combining two drugs, sulopenem etzadroxil and probenecid, in one oral tablet. This combination helps keep the antibiotic in the body longer, making it work better, especially against bacteria that resist traditional treatments. Orlynvah is specifically approved to target infections from bacteria like Escherichia coli, Klebsiella pneumoniae, and Proteus mirabilis, which can be harder to treat.
Marjorie Golden, MD, an infectious disease specialist at Yale New Haven Hospital in Connecticut, described Orlynvah as a much-needed alternative for women struggling with difficult-to-treat UTIs.
“Orlynvah has the potential to be an important treatment option for those who need it,” she said in a news release from Iterum Therapeutics, the drug’s maker.
The FDA approved Orlynvah based on two large clinical trials involving over 3,800 women. In these studies, Orlynvah worked as well as or better than antibiotics like ciprofloxacin and Augmentin. It was generally well-tolerated, though common side effects included diarrhea, nausea, yeast infections, and headaches.
The FDA advises people to discuss their medical history with their doctor before taking Orlynvah, especially if they have conditions like gout, kidney stones, or allergies to other antibiotics.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration (FDA) has approved Orlynvah, a new oral treatment for uncomplicated urinary tract infections (UTIs) in women who have limited options for effective antibiotic therapy.
Uncomplicated UTIs are bladder infections that typically affect women who don’t have other issues like kidney disease or urinary tract abnormalities. These infections are common, affecting around half of all women at least once in their lives.
Treating UTIs can be difficult when standard antibiotics don’t work well, often because of antibiotic resistance or certain health conditions. Orlynvah offers a promising new option by combining two drugs, sulopenem etzadroxil and probenecid, in one oral tablet. This combination helps keep the antibiotic in the body longer, making it work better, especially against bacteria that resist traditional treatments. Orlynvah is specifically approved to target infections from bacteria like Escherichia coli, Klebsiella pneumoniae, and Proteus mirabilis, which can be harder to treat.
Marjorie Golden, MD, an infectious disease specialist at Yale New Haven Hospital in Connecticut, described Orlynvah as a much-needed alternative for women struggling with difficult-to-treat UTIs.
“Orlynvah has the potential to be an important treatment option for those who need it,” she said in a news release from Iterum Therapeutics, the drug’s maker.
The FDA approved Orlynvah based on two large clinical trials involving over 3,800 women. In these studies, Orlynvah worked as well as or better than antibiotics like ciprofloxacin and Augmentin. It was generally well-tolerated, though common side effects included diarrhea, nausea, yeast infections, and headaches.
The FDA advises people to discuss their medical history with their doctor before taking Orlynvah, especially if they have conditions like gout, kidney stones, or allergies to other antibiotics.
A version of this article first appeared on Medscape.com.
Treating Digestive Disease Across the Lifespan
Pediatric gastroenterologists are a vital, yet often overlooked segment of the GI workforce and an important part of AGA’s diverse membership. Per the American Board of Pediatrics, 2,232 pediatricians have been board certified in pediatric gastroenterology since formal certification was first offered in 1990, and AGA Institute Council’s Pediatric Gastroenterology and Developmental Biology Section has nearly 1,900 members.
According to a recently published study in the journal Pediatrics, the pediatric GI workforce is expected to double by 2040, growing at a rate faster than that of most other pediatric subspecialties. This is largely due to the increased scope and complexity of the field driven by scientific advances and the increasing prevalence of digestive and liver diseases in children, including inflammatory bowel and other diseases.
In this month’s Member Spotlight, we highlight Dr. Yoyo Zhang, a pediatric gastroenterologist at Stanford Children’s Health specializing in intestinal and liver transplantation. Her passion for her profession and for improving the lives of her patients shines brightly, and her interview provides fascinating insights into the complexities and rewards of the rapidly expanding field of pediatric gastroenterology.
Also in our November issue, we update you on the FDA’s recent approval of the “next-gen” Cologuard test and query a panel of primary care and GI experts on their thoughts regarding the role that newly FDA-approved (but not yet guideline-recommended) Guardant blood-based CRC screening test should play in CRC screening moving forward.
In our Perspectives feature, we offer expert insights on how to appropriately screen patients for certain rare malignancies. Is it worthwhile screening for pancreatic cancer, and if so, how should it be done? Likewise, diagnosing cholangiocarcinoma is challenging; how best should one evaluate for this in higher-risk populations?
We hope you enjoy all the content in our November issue – as always, thanks for reading!
Megan A. Adams, MD, JD, MSc
Editor in Chief
Pediatric gastroenterologists are a vital, yet often overlooked segment of the GI workforce and an important part of AGA’s diverse membership. Per the American Board of Pediatrics, 2,232 pediatricians have been board certified in pediatric gastroenterology since formal certification was first offered in 1990, and AGA Institute Council’s Pediatric Gastroenterology and Developmental Biology Section has nearly 1,900 members.
According to a recently published study in the journal Pediatrics, the pediatric GI workforce is expected to double by 2040, growing at a rate faster than that of most other pediatric subspecialties. This is largely due to the increased scope and complexity of the field driven by scientific advances and the increasing prevalence of digestive and liver diseases in children, including inflammatory bowel and other diseases.
In this month’s Member Spotlight, we highlight Dr. Yoyo Zhang, a pediatric gastroenterologist at Stanford Children’s Health specializing in intestinal and liver transplantation. Her passion for her profession and for improving the lives of her patients shines brightly, and her interview provides fascinating insights into the complexities and rewards of the rapidly expanding field of pediatric gastroenterology.
Also in our November issue, we update you on the FDA’s recent approval of the “next-gen” Cologuard test and query a panel of primary care and GI experts on their thoughts regarding the role that newly FDA-approved (but not yet guideline-recommended) Guardant blood-based CRC screening test should play in CRC screening moving forward.
In our Perspectives feature, we offer expert insights on how to appropriately screen patients for certain rare malignancies. Is it worthwhile screening for pancreatic cancer, and if so, how should it be done? Likewise, diagnosing cholangiocarcinoma is challenging; how best should one evaluate for this in higher-risk populations?
We hope you enjoy all the content in our November issue – as always, thanks for reading!
Megan A. Adams, MD, JD, MSc
Editor in Chief
Pediatric gastroenterologists are a vital, yet often overlooked segment of the GI workforce and an important part of AGA’s diverse membership. Per the American Board of Pediatrics, 2,232 pediatricians have been board certified in pediatric gastroenterology since formal certification was first offered in 1990, and AGA Institute Council’s Pediatric Gastroenterology and Developmental Biology Section has nearly 1,900 members.
According to a recently published study in the journal Pediatrics, the pediatric GI workforce is expected to double by 2040, growing at a rate faster than that of most other pediatric subspecialties. This is largely due to the increased scope and complexity of the field driven by scientific advances and the increasing prevalence of digestive and liver diseases in children, including inflammatory bowel and other diseases.
In this month’s Member Spotlight, we highlight Dr. Yoyo Zhang, a pediatric gastroenterologist at Stanford Children’s Health specializing in intestinal and liver transplantation. Her passion for her profession and for improving the lives of her patients shines brightly, and her interview provides fascinating insights into the complexities and rewards of the rapidly expanding field of pediatric gastroenterology.
Also in our November issue, we update you on the FDA’s recent approval of the “next-gen” Cologuard test and query a panel of primary care and GI experts on their thoughts regarding the role that newly FDA-approved (but not yet guideline-recommended) Guardant blood-based CRC screening test should play in CRC screening moving forward.
In our Perspectives feature, we offer expert insights on how to appropriately screen patients for certain rare malignancies. Is it worthwhile screening for pancreatic cancer, and if so, how should it be done? Likewise, diagnosing cholangiocarcinoma is challenging; how best should one evaluate for this in higher-risk populations?
We hope you enjoy all the content in our November issue – as always, thanks for reading!
Megan A. Adams, MD, JD, MSc
Editor in Chief
Screening Options for Rare Malignancies
Dear colleagues,
As gastroenterologists and endoscopists, we spend significant time preventing and diagnosing GI malignancies.
For instance, is it worthwhile screening for pancreatic cancer, and, if so, how should this be done? Likewise, diagnosing cholangiocarcinoma is challenging; how best should one evaluate for this in higher risk populations, such as primary sclerosing cholangitis? And what about the costs, financial and otherwise, associated with screening?
In this issue of Perspectives, Dr. Darshan Kothari and Dr. Daniel Bernstein discuss their approach to pancreatic cancer screening, including who is eligible, the preferred screening modalities, and the barriers to screening. In the accompanying perspective, Dr. Aparna Goel and Dr. Judah Kupferman focus on cholangiocarcinoma screening, identifying high-risk populations and discussing some of the concerns with screening, necessitating shared decision-making.
We welcome your thoughts on this issue. Share with us on X at @AGA_GIHN.
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, and chief of endoscopy at West Haven VA Medical Center, both in Connecticut. He is an associate editor for GI & Hepatology News.
An Approach to Pancreatic Cancer Screening
BY DANIEL A. BERNSTEIN, MD, AND DARSHAN KOTHARI, MD
Pancreatic cancer carries a dismal prognosis, now accounting for the third-most cancer-related mortality in the United States. A small proportion of patients are diagnosed at a local stage of disease, with over half found to have metastatic disease at presentation. Given the low overall incidence and lifetime risk in the general population, population-based screening is not justified.
About 10% of cases of pancreas cancer are associated with germ-line mutations and/or with a strong family history of pancreatic cancer. Several academic societies and expert committees now recommend regular screening for pancreatic cancer in patients who are considered high-risk individuals, as they carry a fivefold relative risk for pancreatic cancer. Moreover, studies suggest that screening has the potential to identify early-stage resectable disease and decrease mortality in this patient population.
Patients who benefit from pancreatic cancer screening are those who carry an increased lifetime risk (in excess of 5%) of pancreatic cancer. High-risk individuals include those with germ-line mutations and/or those with a family history of pancreatic cancer in first-degree relatives. Consensus guidelines by the International Cancer of the Pancreas Screening Consortium and the American Society for Gastrointestinal Endoscopy provide medical centers with detailed recommendations on who and when to start screening.
High-risk individuals fall into three categories:
- Patients with high-risk germline mutations including: familial atypical multiple mole melanoma syndrome (CDKN2A), hereditary breast and ovarian cancer syndromes (BRCA1, BRCA2, and PALB2), Peutz-Jeghers syndrome (STK11), and hereditary pancreatitis (PRSS1 and SPINK1)
- Patients with low- to moderate-risk germ-line mutations with at least one first-degree relative with pancreatic cancer: Lynch Syndrome (particularly MLH1 mutation), ataxia-telangiectasia (ATM), or Li-Fraumeni syndrome (p53)
- Patients with one first-degree relative with pancreatic cancer who in turn has one first-degree relative with pancreatic cancer (eg, a patient’s mother and maternal aunt or a patient’s father and patient’s sister)
Consistent with established guidelines, we recommend screening for high-risk patients beginning at age 50, or 10 years before the youngest age at which pancreas cancer was diagnosed in an affected relative. Screening is recommended earlier in patients with particularly high risk: at age 40 for patients with CDKN2A and STKI11 mutations and age 40 for patients with PRSS1 mutation or 20 years after the first attack of acute pancreatitis. For patients with a strong family history of pancreas cancer, we recommend comprehensive evaluation by a certified genetic counselor at a high-volume cancer center.
In practice, patients at our institution who are identified as high risk based on the above criteria are referred for an initial consultation at our pancreas center. In most cases, this should occur no sooner than 5 years prior to the recommended starting age for screening. All patients who are identified as high risk should be screened annually for diabetes given the growing evidence base supporting an association between new-onset diabetes and pancreatic cancer.
After an initial visit and discussion of the risks and benefits of screening, most screening protocols start with a baseline endoscopic ultrasound (EUS) and contrast-enhanced magnetic resonance abdomen with magnetic resonance cholangiopancreatography (MRI/MRCP), which will be repeated annually or sooner as the clinical condition warrants. A sooner-interval EUS should be considered for patients already undergoing screening who are newly found to have diabetes.
At our institution, we start with an in-person clinic evaluation followed by EUS. Thereafter, patients undergo MRI/MRCP (synchronized with a same-day clinic visit) alternating with EUS every 6 months to ensure patients are seen twice a year, though there is no specific data to support this approach. Non-diabetics also undergo yearly diabetes screening which will trigger an EUS if patients become diabetic.
We engage in shared decision-making with our high-risk individuals undergoing pancreatic cancer screening and at each visit we review their concurrent medical conditions and suitability to continue screening. We consider discontinuing screening after age 75, at the onset of any life-limiting illness, or after a discussion of risks and benefits if comorbidities lead to a substantial deterioration in a patient’s overall health status.
While a growing body of evidence exists to support the application of pancreatic cancer screening in high-risk individuals, this preventive service remains underutilized. Recent analysis of the screening cohort at our institution showed a demographically homogeneous group of mostly highly educated, high-income White females. These findings are consistent with the patient cohorts described in other pancreatic cancer screening programs and represent only a fraction of people who would qualify for pancreatic cancer screening.
A survey of patients undergoing screening at our institution identified cost, travel, and time associated with pancreatic cancer screening to be frequent challenges to participation. Further studies are needed to fully explore the barriers and psychological burden of pancreas cancer screening in high-risk individuals, and to identify ways to enrich the cohort of patients undergoing screening. This may involve novel methods to identify family members of patients with a new diagnosis of pancreas cancer and increasing health literacy around pancreatic cancer screening among patients and providers.
Pancreatic cancer screening has the potential to identify early-stage disease in patients who are at high risk because of germ-line mutations and/or family history. We recommend that patients engage in pancreatic cancer screening at high-volume centers with well-supported oncology, genetics, and research infrastructure.
Dr. Bernstein is a gastroenterology fellow at Duke University School of Medicine, Durham, North Carolina. Dr. Kothari is an associate professor of medicine in gastroenterology and hepatology at Duke University School of Medicine.
Screening for Cholangiocarcinoma
BY JUDAH KUPFERMAN, MD, AND APARNA GOEL, MD
Cholangiocarcinoma is a rare but aggressive cancer of the bile ducts that poses many diagnostic challenges. Approximately 3% of gastrointestinal cancers are attributed to cholangiocarcinoma, and while the annual incidence of disease in the United States is about 1.26 per 100,000 people, the incidence of intrahepatic disease has been rising considerably.1,2 Screening for cholangiocarcinoma is reserved for high-risk individuals — such as those with primary sclerosing cholangitis (PSC), secondary sclerosing cholangitis (SSC), and biliary tract disorders such as choledochal cysts or Caroli’s disease. The goal is to balance the benefits of early diagnosis with the costs and risks associated with screening, particularly given the limitations of available tools like MRI with cholangiopancreatography (MRCP), which has a sensitivity of 70%-85%. In general, we recommend annual cholangiocarcinoma screening for high-risk individuals with MRI and MRCP as well as with cancer antigen (CA) 19-9. .
Screening in Patients with Primary Sclerosing Cholangitis
The lifetime risk of cholangiocarcinoma in patients with PSC is 10%-15% with an annual risk of 0.5%-1.5%. In our experience, this is often the most feared complication for PSC patients, even more so than the risk of liver transplantation. We recommend annual MRI with MRCP in addition to CA 19-9 for patients with PSC in the first decade of their diagnosis, as most cancers are diagnosed during this period. If a patient’s imaging has remained stable for over a decade and there is minimal hepatic fibrosis, we discuss the option of reducing screening frequency to every 2 years to minimize costs and exposure to MRI contrast risks.
If MRI reveals a concerning new large duct stricture, we will evaluate this with an endoscopic retrograde cholangiopancreatography (ERCP), as differentiating benign and malignant strictures is quite challenging with MRI. We generally recommend ERCP with brush cytology and fluorescence in situ hybridization to improve diagnostic yield. Depending on imaging findings and location of the new large duct stricture, we may consider cholangioscopy during ERCP for direct visualization of the bile duct and directed tissue biopsies. Unfortunately, even in young, asymptomatic patients who undergo regular screening, cholangiocarcinoma is frequently diagnosed at an advanced stage.
Screening in Patients with Secondary Sclerosing Cholangitis
Patients with SSC may develop cholangiocarcinoma because of chronic inflammatory and fibrotic processes, such as IgG4-associated cholangiopathy, sarcoidosis, ischemic cholangiopathy, cystic fibrosis, recurrent pyogenic cholangitis, severe sepsis (as recently seen from SARS-CoV-2), surgical complications, or other etiologies. When the condition is reversible, such as with IgG4-associated cholangiopathy, cancer screening may not be necessary. However, when irreversible damage occurs, the cancer risk increases, though it varies by disease type and severity. In most cases, we recommend routine screening for cholangiocarcinoma with MRI and CA 19-9 in this population.
Screening in Patients with Biliary Tract Disorders
Biliary tract disorders such as choledochal cysts and Caroli’s disease also harbor an increased risk of cholangiocarcinoma. Choledochal cysts are congenital cystic dilations of the bile duct that have a 10%-30% lifetime risk of malignant transformation to cholangiocarcinoma. Surgical intervention to remove the cyst is often recommended because of this high risk. However, some patients may be unable or unwilling to undergo this surgery or they may have residual cysts. We recommend ongoing screening with MRI and CA 19-9 for these patients. Similarly, Caroli’s disease is a congenital disease associated with intrahepatic and extrahepatic bile duct cysts and associated with a 5%-15% lifetime risk of cholangiocarcinoma. MRI with MRCP and CA 19-9 should be performed routinely for patients with Caroli’s disease and syndrome.
Risks and Challenges in Cholangiocarcinoma Screening
While MRI with MRCP is the gold standard for cholangiocarcinoma screening, its limitations must be carefully considered. One growing concern is the potential for gadolinium retention in the brain, bones, or skin following repeated MRI scans. Though the long-term effects of gadolinium retention are not fully understood, we factor this into screening decisions, particularly for younger patients who may undergo decades of regular imaging.
MRI is not always feasible for certain patients, including those with metal implants, on hemodialysis, or with severe allergic reactions. In such cases, CT or ultrasound may serve as alternatives, though with lower sensitivity for detecting cholangiocarcinoma. Additionally, claustrophobia during MRI can be addressed with sedation, but this underscores the importance of shared decision-making.
From our perspective, cholangiocarcinoma screening in high-risk patients is crucial but not without challenges. Our current screening methods, while essential, are far from perfect, often missing early cancers or leading to unnecessary interventions. Because of these limitations, the window for treatment of localized disease can easily be missed. In our practice, we tailor screening strategies to each patient’s specific needs, weighing the potential benefits against the risks, costs, and the inherent uncertainty of early detection tools. We believe it is essential to involve patients in this decision-making process to provide a balanced, individualized approach that considers both clinical evidence and the personal preferences of each person.
Dr. Kupferman is a gastroenterology fellow at Stanford University School of Medicine in California. Dr. Goel is a transplant hepatologist and a clinical associate professor in gastroenterology & hepatology at Stanford.
References
1. Vithayathil M and Khan SA. J Hepatol. 2022 Dec. doi: 10.1016/j.jhep.2022.07.022.
2. Patel N and Benipal B. Cureus. 2019 Jan. doi: 10.7759/cureus.3962.
Dear colleagues,
As gastroenterologists and endoscopists, we spend significant time preventing and diagnosing GI malignancies.
For instance, is it worthwhile screening for pancreatic cancer, and, if so, how should this be done? Likewise, diagnosing cholangiocarcinoma is challenging; how best should one evaluate for this in higher risk populations, such as primary sclerosing cholangitis? And what about the costs, financial and otherwise, associated with screening?
In this issue of Perspectives, Dr. Darshan Kothari and Dr. Daniel Bernstein discuss their approach to pancreatic cancer screening, including who is eligible, the preferred screening modalities, and the barriers to screening. In the accompanying perspective, Dr. Aparna Goel and Dr. Judah Kupferman focus on cholangiocarcinoma screening, identifying high-risk populations and discussing some of the concerns with screening, necessitating shared decision-making.
We welcome your thoughts on this issue. Share with us on X at @AGA_GIHN.
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, and chief of endoscopy at West Haven VA Medical Center, both in Connecticut. He is an associate editor for GI & Hepatology News.
An Approach to Pancreatic Cancer Screening
BY DANIEL A. BERNSTEIN, MD, AND DARSHAN KOTHARI, MD
Pancreatic cancer carries a dismal prognosis, now accounting for the third-most cancer-related mortality in the United States. A small proportion of patients are diagnosed at a local stage of disease, with over half found to have metastatic disease at presentation. Given the low overall incidence and lifetime risk in the general population, population-based screening is not justified.
About 10% of cases of pancreas cancer are associated with germ-line mutations and/or with a strong family history of pancreatic cancer. Several academic societies and expert committees now recommend regular screening for pancreatic cancer in patients who are considered high-risk individuals, as they carry a fivefold relative risk for pancreatic cancer. Moreover, studies suggest that screening has the potential to identify early-stage resectable disease and decrease mortality in this patient population.
Patients who benefit from pancreatic cancer screening are those who carry an increased lifetime risk (in excess of 5%) of pancreatic cancer. High-risk individuals include those with germ-line mutations and/or those with a family history of pancreatic cancer in first-degree relatives. Consensus guidelines by the International Cancer of the Pancreas Screening Consortium and the American Society for Gastrointestinal Endoscopy provide medical centers with detailed recommendations on who and when to start screening.
High-risk individuals fall into three categories:
- Patients with high-risk germline mutations including: familial atypical multiple mole melanoma syndrome (CDKN2A), hereditary breast and ovarian cancer syndromes (BRCA1, BRCA2, and PALB2), Peutz-Jeghers syndrome (STK11), and hereditary pancreatitis (PRSS1 and SPINK1)
- Patients with low- to moderate-risk germ-line mutations with at least one first-degree relative with pancreatic cancer: Lynch Syndrome (particularly MLH1 mutation), ataxia-telangiectasia (ATM), or Li-Fraumeni syndrome (p53)
- Patients with one first-degree relative with pancreatic cancer who in turn has one first-degree relative with pancreatic cancer (eg, a patient’s mother and maternal aunt or a patient’s father and patient’s sister)
Consistent with established guidelines, we recommend screening for high-risk patients beginning at age 50, or 10 years before the youngest age at which pancreas cancer was diagnosed in an affected relative. Screening is recommended earlier in patients with particularly high risk: at age 40 for patients with CDKN2A and STKI11 mutations and age 40 for patients with PRSS1 mutation or 20 years after the first attack of acute pancreatitis. For patients with a strong family history of pancreas cancer, we recommend comprehensive evaluation by a certified genetic counselor at a high-volume cancer center.
In practice, patients at our institution who are identified as high risk based on the above criteria are referred for an initial consultation at our pancreas center. In most cases, this should occur no sooner than 5 years prior to the recommended starting age for screening. All patients who are identified as high risk should be screened annually for diabetes given the growing evidence base supporting an association between new-onset diabetes and pancreatic cancer.
After an initial visit and discussion of the risks and benefits of screening, most screening protocols start with a baseline endoscopic ultrasound (EUS) and contrast-enhanced magnetic resonance abdomen with magnetic resonance cholangiopancreatography (MRI/MRCP), which will be repeated annually or sooner as the clinical condition warrants. A sooner-interval EUS should be considered for patients already undergoing screening who are newly found to have diabetes.
At our institution, we start with an in-person clinic evaluation followed by EUS. Thereafter, patients undergo MRI/MRCP (synchronized with a same-day clinic visit) alternating with EUS every 6 months to ensure patients are seen twice a year, though there is no specific data to support this approach. Non-diabetics also undergo yearly diabetes screening which will trigger an EUS if patients become diabetic.
We engage in shared decision-making with our high-risk individuals undergoing pancreatic cancer screening and at each visit we review their concurrent medical conditions and suitability to continue screening. We consider discontinuing screening after age 75, at the onset of any life-limiting illness, or after a discussion of risks and benefits if comorbidities lead to a substantial deterioration in a patient’s overall health status.
While a growing body of evidence exists to support the application of pancreatic cancer screening in high-risk individuals, this preventive service remains underutilized. Recent analysis of the screening cohort at our institution showed a demographically homogeneous group of mostly highly educated, high-income White females. These findings are consistent with the patient cohorts described in other pancreatic cancer screening programs and represent only a fraction of people who would qualify for pancreatic cancer screening.
A survey of patients undergoing screening at our institution identified cost, travel, and time associated with pancreatic cancer screening to be frequent challenges to participation. Further studies are needed to fully explore the barriers and psychological burden of pancreas cancer screening in high-risk individuals, and to identify ways to enrich the cohort of patients undergoing screening. This may involve novel methods to identify family members of patients with a new diagnosis of pancreas cancer and increasing health literacy around pancreatic cancer screening among patients and providers.
Pancreatic cancer screening has the potential to identify early-stage disease in patients who are at high risk because of germ-line mutations and/or family history. We recommend that patients engage in pancreatic cancer screening at high-volume centers with well-supported oncology, genetics, and research infrastructure.
Dr. Bernstein is a gastroenterology fellow at Duke University School of Medicine, Durham, North Carolina. Dr. Kothari is an associate professor of medicine in gastroenterology and hepatology at Duke University School of Medicine.
Screening for Cholangiocarcinoma
BY JUDAH KUPFERMAN, MD, AND APARNA GOEL, MD
Cholangiocarcinoma is a rare but aggressive cancer of the bile ducts that poses many diagnostic challenges. Approximately 3% of gastrointestinal cancers are attributed to cholangiocarcinoma, and while the annual incidence of disease in the United States is about 1.26 per 100,000 people, the incidence of intrahepatic disease has been rising considerably.1,2 Screening for cholangiocarcinoma is reserved for high-risk individuals — such as those with primary sclerosing cholangitis (PSC), secondary sclerosing cholangitis (SSC), and biliary tract disorders such as choledochal cysts or Caroli’s disease. The goal is to balance the benefits of early diagnosis with the costs and risks associated with screening, particularly given the limitations of available tools like MRI with cholangiopancreatography (MRCP), which has a sensitivity of 70%-85%. In general, we recommend annual cholangiocarcinoma screening for high-risk individuals with MRI and MRCP as well as with cancer antigen (CA) 19-9. .
Screening in Patients with Primary Sclerosing Cholangitis
The lifetime risk of cholangiocarcinoma in patients with PSC is 10%-15% with an annual risk of 0.5%-1.5%. In our experience, this is often the most feared complication for PSC patients, even more so than the risk of liver transplantation. We recommend annual MRI with MRCP in addition to CA 19-9 for patients with PSC in the first decade of their diagnosis, as most cancers are diagnosed during this period. If a patient’s imaging has remained stable for over a decade and there is minimal hepatic fibrosis, we discuss the option of reducing screening frequency to every 2 years to minimize costs and exposure to MRI contrast risks.
If MRI reveals a concerning new large duct stricture, we will evaluate this with an endoscopic retrograde cholangiopancreatography (ERCP), as differentiating benign and malignant strictures is quite challenging with MRI. We generally recommend ERCP with brush cytology and fluorescence in situ hybridization to improve diagnostic yield. Depending on imaging findings and location of the new large duct stricture, we may consider cholangioscopy during ERCP for direct visualization of the bile duct and directed tissue biopsies. Unfortunately, even in young, asymptomatic patients who undergo regular screening, cholangiocarcinoma is frequently diagnosed at an advanced stage.
Screening in Patients with Secondary Sclerosing Cholangitis
Patients with SSC may develop cholangiocarcinoma because of chronic inflammatory and fibrotic processes, such as IgG4-associated cholangiopathy, sarcoidosis, ischemic cholangiopathy, cystic fibrosis, recurrent pyogenic cholangitis, severe sepsis (as recently seen from SARS-CoV-2), surgical complications, or other etiologies. When the condition is reversible, such as with IgG4-associated cholangiopathy, cancer screening may not be necessary. However, when irreversible damage occurs, the cancer risk increases, though it varies by disease type and severity. In most cases, we recommend routine screening for cholangiocarcinoma with MRI and CA 19-9 in this population.
Screening in Patients with Biliary Tract Disorders
Biliary tract disorders such as choledochal cysts and Caroli’s disease also harbor an increased risk of cholangiocarcinoma. Choledochal cysts are congenital cystic dilations of the bile duct that have a 10%-30% lifetime risk of malignant transformation to cholangiocarcinoma. Surgical intervention to remove the cyst is often recommended because of this high risk. However, some patients may be unable or unwilling to undergo this surgery or they may have residual cysts. We recommend ongoing screening with MRI and CA 19-9 for these patients. Similarly, Caroli’s disease is a congenital disease associated with intrahepatic and extrahepatic bile duct cysts and associated with a 5%-15% lifetime risk of cholangiocarcinoma. MRI with MRCP and CA 19-9 should be performed routinely for patients with Caroli’s disease and syndrome.
Risks and Challenges in Cholangiocarcinoma Screening
While MRI with MRCP is the gold standard for cholangiocarcinoma screening, its limitations must be carefully considered. One growing concern is the potential for gadolinium retention in the brain, bones, or skin following repeated MRI scans. Though the long-term effects of gadolinium retention are not fully understood, we factor this into screening decisions, particularly for younger patients who may undergo decades of regular imaging.
MRI is not always feasible for certain patients, including those with metal implants, on hemodialysis, or with severe allergic reactions. In such cases, CT or ultrasound may serve as alternatives, though with lower sensitivity for detecting cholangiocarcinoma. Additionally, claustrophobia during MRI can be addressed with sedation, but this underscores the importance of shared decision-making.
From our perspective, cholangiocarcinoma screening in high-risk patients is crucial but not without challenges. Our current screening methods, while essential, are far from perfect, often missing early cancers or leading to unnecessary interventions. Because of these limitations, the window for treatment of localized disease can easily be missed. In our practice, we tailor screening strategies to each patient’s specific needs, weighing the potential benefits against the risks, costs, and the inherent uncertainty of early detection tools. We believe it is essential to involve patients in this decision-making process to provide a balanced, individualized approach that considers both clinical evidence and the personal preferences of each person.
Dr. Kupferman is a gastroenterology fellow at Stanford University School of Medicine in California. Dr. Goel is a transplant hepatologist and a clinical associate professor in gastroenterology & hepatology at Stanford.
References
1. Vithayathil M and Khan SA. J Hepatol. 2022 Dec. doi: 10.1016/j.jhep.2022.07.022.
2. Patel N and Benipal B. Cureus. 2019 Jan. doi: 10.7759/cureus.3962.
Dear colleagues,
As gastroenterologists and endoscopists, we spend significant time preventing and diagnosing GI malignancies.
For instance, is it worthwhile screening for pancreatic cancer, and, if so, how should this be done? Likewise, diagnosing cholangiocarcinoma is challenging; how best should one evaluate for this in higher risk populations, such as primary sclerosing cholangitis? And what about the costs, financial and otherwise, associated with screening?
In this issue of Perspectives, Dr. Darshan Kothari and Dr. Daniel Bernstein discuss their approach to pancreatic cancer screening, including who is eligible, the preferred screening modalities, and the barriers to screening. In the accompanying perspective, Dr. Aparna Goel and Dr. Judah Kupferman focus on cholangiocarcinoma screening, identifying high-risk populations and discussing some of the concerns with screening, necessitating shared decision-making.
We welcome your thoughts on this issue. Share with us on X at @AGA_GIHN.
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, and chief of endoscopy at West Haven VA Medical Center, both in Connecticut. He is an associate editor for GI & Hepatology News.
An Approach to Pancreatic Cancer Screening
BY DANIEL A. BERNSTEIN, MD, AND DARSHAN KOTHARI, MD
Pancreatic cancer carries a dismal prognosis, now accounting for the third-most cancer-related mortality in the United States. A small proportion of patients are diagnosed at a local stage of disease, with over half found to have metastatic disease at presentation. Given the low overall incidence and lifetime risk in the general population, population-based screening is not justified.
About 10% of cases of pancreas cancer are associated with germ-line mutations and/or with a strong family history of pancreatic cancer. Several academic societies and expert committees now recommend regular screening for pancreatic cancer in patients who are considered high-risk individuals, as they carry a fivefold relative risk for pancreatic cancer. Moreover, studies suggest that screening has the potential to identify early-stage resectable disease and decrease mortality in this patient population.
Patients who benefit from pancreatic cancer screening are those who carry an increased lifetime risk (in excess of 5%) of pancreatic cancer. High-risk individuals include those with germ-line mutations and/or those with a family history of pancreatic cancer in first-degree relatives. Consensus guidelines by the International Cancer of the Pancreas Screening Consortium and the American Society for Gastrointestinal Endoscopy provide medical centers with detailed recommendations on who and when to start screening.
High-risk individuals fall into three categories:
- Patients with high-risk germline mutations including: familial atypical multiple mole melanoma syndrome (CDKN2A), hereditary breast and ovarian cancer syndromes (BRCA1, BRCA2, and PALB2), Peutz-Jeghers syndrome (STK11), and hereditary pancreatitis (PRSS1 and SPINK1)
- Patients with low- to moderate-risk germ-line mutations with at least one first-degree relative with pancreatic cancer: Lynch Syndrome (particularly MLH1 mutation), ataxia-telangiectasia (ATM), or Li-Fraumeni syndrome (p53)
- Patients with one first-degree relative with pancreatic cancer who in turn has one first-degree relative with pancreatic cancer (eg, a patient’s mother and maternal aunt or a patient’s father and patient’s sister)
Consistent with established guidelines, we recommend screening for high-risk patients beginning at age 50, or 10 years before the youngest age at which pancreas cancer was diagnosed in an affected relative. Screening is recommended earlier in patients with particularly high risk: at age 40 for patients with CDKN2A and STKI11 mutations and age 40 for patients with PRSS1 mutation or 20 years after the first attack of acute pancreatitis. For patients with a strong family history of pancreas cancer, we recommend comprehensive evaluation by a certified genetic counselor at a high-volume cancer center.
In practice, patients at our institution who are identified as high risk based on the above criteria are referred for an initial consultation at our pancreas center. In most cases, this should occur no sooner than 5 years prior to the recommended starting age for screening. All patients who are identified as high risk should be screened annually for diabetes given the growing evidence base supporting an association between new-onset diabetes and pancreatic cancer.
After an initial visit and discussion of the risks and benefits of screening, most screening protocols start with a baseline endoscopic ultrasound (EUS) and contrast-enhanced magnetic resonance abdomen with magnetic resonance cholangiopancreatography (MRI/MRCP), which will be repeated annually or sooner as the clinical condition warrants. A sooner-interval EUS should be considered for patients already undergoing screening who are newly found to have diabetes.
At our institution, we start with an in-person clinic evaluation followed by EUS. Thereafter, patients undergo MRI/MRCP (synchronized with a same-day clinic visit) alternating with EUS every 6 months to ensure patients are seen twice a year, though there is no specific data to support this approach. Non-diabetics also undergo yearly diabetes screening which will trigger an EUS if patients become diabetic.
We engage in shared decision-making with our high-risk individuals undergoing pancreatic cancer screening and at each visit we review their concurrent medical conditions and suitability to continue screening. We consider discontinuing screening after age 75, at the onset of any life-limiting illness, or after a discussion of risks and benefits if comorbidities lead to a substantial deterioration in a patient’s overall health status.
While a growing body of evidence exists to support the application of pancreatic cancer screening in high-risk individuals, this preventive service remains underutilized. Recent analysis of the screening cohort at our institution showed a demographically homogeneous group of mostly highly educated, high-income White females. These findings are consistent with the patient cohorts described in other pancreatic cancer screening programs and represent only a fraction of people who would qualify for pancreatic cancer screening.
A survey of patients undergoing screening at our institution identified cost, travel, and time associated with pancreatic cancer screening to be frequent challenges to participation. Further studies are needed to fully explore the barriers and psychological burden of pancreas cancer screening in high-risk individuals, and to identify ways to enrich the cohort of patients undergoing screening. This may involve novel methods to identify family members of patients with a new diagnosis of pancreas cancer and increasing health literacy around pancreatic cancer screening among patients and providers.
Pancreatic cancer screening has the potential to identify early-stage disease in patients who are at high risk because of germ-line mutations and/or family history. We recommend that patients engage in pancreatic cancer screening at high-volume centers with well-supported oncology, genetics, and research infrastructure.
Dr. Bernstein is a gastroenterology fellow at Duke University School of Medicine, Durham, North Carolina. Dr. Kothari is an associate professor of medicine in gastroenterology and hepatology at Duke University School of Medicine.
Screening for Cholangiocarcinoma
BY JUDAH KUPFERMAN, MD, AND APARNA GOEL, MD
Cholangiocarcinoma is a rare but aggressive cancer of the bile ducts that poses many diagnostic challenges. Approximately 3% of gastrointestinal cancers are attributed to cholangiocarcinoma, and while the annual incidence of disease in the United States is about 1.26 per 100,000 people, the incidence of intrahepatic disease has been rising considerably.1,2 Screening for cholangiocarcinoma is reserved for high-risk individuals — such as those with primary sclerosing cholangitis (PSC), secondary sclerosing cholangitis (SSC), and biliary tract disorders such as choledochal cysts or Caroli’s disease. The goal is to balance the benefits of early diagnosis with the costs and risks associated with screening, particularly given the limitations of available tools like MRI with cholangiopancreatography (MRCP), which has a sensitivity of 70%-85%. In general, we recommend annual cholangiocarcinoma screening for high-risk individuals with MRI and MRCP as well as with cancer antigen (CA) 19-9. .
Screening in Patients with Primary Sclerosing Cholangitis
The lifetime risk of cholangiocarcinoma in patients with PSC is 10%-15% with an annual risk of 0.5%-1.5%. In our experience, this is often the most feared complication for PSC patients, even more so than the risk of liver transplantation. We recommend annual MRI with MRCP in addition to CA 19-9 for patients with PSC in the first decade of their diagnosis, as most cancers are diagnosed during this period. If a patient’s imaging has remained stable for over a decade and there is minimal hepatic fibrosis, we discuss the option of reducing screening frequency to every 2 years to minimize costs and exposure to MRI contrast risks.
If MRI reveals a concerning new large duct stricture, we will evaluate this with an endoscopic retrograde cholangiopancreatography (ERCP), as differentiating benign and malignant strictures is quite challenging with MRI. We generally recommend ERCP with brush cytology and fluorescence in situ hybridization to improve diagnostic yield. Depending on imaging findings and location of the new large duct stricture, we may consider cholangioscopy during ERCP for direct visualization of the bile duct and directed tissue biopsies. Unfortunately, even in young, asymptomatic patients who undergo regular screening, cholangiocarcinoma is frequently diagnosed at an advanced stage.
Screening in Patients with Secondary Sclerosing Cholangitis
Patients with SSC may develop cholangiocarcinoma because of chronic inflammatory and fibrotic processes, such as IgG4-associated cholangiopathy, sarcoidosis, ischemic cholangiopathy, cystic fibrosis, recurrent pyogenic cholangitis, severe sepsis (as recently seen from SARS-CoV-2), surgical complications, or other etiologies. When the condition is reversible, such as with IgG4-associated cholangiopathy, cancer screening may not be necessary. However, when irreversible damage occurs, the cancer risk increases, though it varies by disease type and severity. In most cases, we recommend routine screening for cholangiocarcinoma with MRI and CA 19-9 in this population.
Screening in Patients with Biliary Tract Disorders
Biliary tract disorders such as choledochal cysts and Caroli’s disease also harbor an increased risk of cholangiocarcinoma. Choledochal cysts are congenital cystic dilations of the bile duct that have a 10%-30% lifetime risk of malignant transformation to cholangiocarcinoma. Surgical intervention to remove the cyst is often recommended because of this high risk. However, some patients may be unable or unwilling to undergo this surgery or they may have residual cysts. We recommend ongoing screening with MRI and CA 19-9 for these patients. Similarly, Caroli’s disease is a congenital disease associated with intrahepatic and extrahepatic bile duct cysts and associated with a 5%-15% lifetime risk of cholangiocarcinoma. MRI with MRCP and CA 19-9 should be performed routinely for patients with Caroli’s disease and syndrome.
Risks and Challenges in Cholangiocarcinoma Screening
While MRI with MRCP is the gold standard for cholangiocarcinoma screening, its limitations must be carefully considered. One growing concern is the potential for gadolinium retention in the brain, bones, or skin following repeated MRI scans. Though the long-term effects of gadolinium retention are not fully understood, we factor this into screening decisions, particularly for younger patients who may undergo decades of regular imaging.
MRI is not always feasible for certain patients, including those with metal implants, on hemodialysis, or with severe allergic reactions. In such cases, CT or ultrasound may serve as alternatives, though with lower sensitivity for detecting cholangiocarcinoma. Additionally, claustrophobia during MRI can be addressed with sedation, but this underscores the importance of shared decision-making.
From our perspective, cholangiocarcinoma screening in high-risk patients is crucial but not without challenges. Our current screening methods, while essential, are far from perfect, often missing early cancers or leading to unnecessary interventions. Because of these limitations, the window for treatment of localized disease can easily be missed. In our practice, we tailor screening strategies to each patient’s specific needs, weighing the potential benefits against the risks, costs, and the inherent uncertainty of early detection tools. We believe it is essential to involve patients in this decision-making process to provide a balanced, individualized approach that considers both clinical evidence and the personal preferences of each person.
Dr. Kupferman is a gastroenterology fellow at Stanford University School of Medicine in California. Dr. Goel is a transplant hepatologist and a clinical associate professor in gastroenterology & hepatology at Stanford.
References
1. Vithayathil M and Khan SA. J Hepatol. 2022 Dec. doi: 10.1016/j.jhep.2022.07.022.
2. Patel N and Benipal B. Cureus. 2019 Jan. doi: 10.7759/cureus.3962.
Medical, Endoscopic, and Surgical Management of Gastroesophageal Reflux Disease
Introduction
Gastroesophageal reflux disease (GERD) is a frequently encountered condition, and rising annually.1 A recent meta-analysis suggests nearly 14% (1.03 billion) of the population are affected worldwide. Differences may range by region from 12% in Latin America to 20% in North America, and by country from 4% in China to 23% in Turkey.1 In the United States, 21% of the population are afflicted with weekly GERD symptoms.2 Novel medical therapies and endoscopic options provide clinicians with opportunities to help patients with GERD.3
Diagnosis
Definition
GERD was originally defined by the Montreal consensus as a condition that develops when the reflux of stomach contents causes troublesome symptoms and/or complications.4 Heartburn and regurgitation are common symptoms of GERD, with a sensitivity of 30%-76% and specificity of 62%-96% for erosive esophagitis (EE), which occurs when the reflux of stomach content causes esophageal mucosal breaks.5 The presence of characteristic mucosal injury observed during an upper endoscopy or abnormal esophageal acid exposure on ambulatory reflux monitoring are objective evidence of GERD. A trial of a proton pump inhibitor (PPI) may function as a diagnostic test for patients exhibiting the typical symptoms of GERD without any alarm symptoms.3,6
Endoscopic Evaluation and Confirmation
The 2022 American Gastroenterological Association (AGA) clinical practice update recommends diagnostic endoscopy, after PPIs are stopped for 2-4 weeks, in patients whose GERD symptoms do not respond adequately to an empiric trial of a PPI.3 Those with GERD and alarm symptoms such as dysphagia, weight loss, bleeding, and vomiting should undergo endoscopy as soon as possible. Endoscopic findings of EE (Los Angeles Grade B or more severe) and long-segment Barrett’s esophagus (> 3-cm segment with intestinal metaplasia on biopsy) are diagnostic of GERD.3
Reflux Monitoring
With ambulatory reflux monitoring (pH or impedance-pH), esophageal acid exposure (or neutral refluxate in impedance testing) can be measured to confirm GERD diagnosis and to correlate symptoms with reflux episodes. Patients with atypical GERD symptoms or patients with a confirmed diagnosis of GERD whose symptoms have not improved sufficiently with twice-daily PPI therapy should have esophageal impedance-pH monitoring while on PPIs.6,7
Esophageal Manometry
High-resolution esophageal manometry can be used to assess motility abnormalities associated with GERD.
Although no manometric abnormality is unique to GERD, weak lower esophageal sphincter (LES) resting pressure and ineffective esophageal motility frequently coexist with severe GERD.6
Manometry is particularly useful in patients considering surgical or endoscopic anti-reflux procedures to evaluate for achalasia,3 an important contraindication to surgery.
Medical Management
Management of GERD requires a multidisciplinary and personalized approach based on symptom presentation, body mass index, endoscopic findings (e.g., presence of EE, Barrett’s esophagus, hiatal hernia), and physiological abnormalities (e.g., gastroparesis or ineffective motility).3
Lifestyle Modifications
Recommended lifestyle modifications include weight loss for patients with obesity, stress reduction, tobacco and alcohol cessation, elevating the head of the bed, staying upright during and after meals, avoidance of food intake < 3 hours before bedtime, and cessation of foods that potentially aggravate reflux symptoms such as coffee, chocolate, carbonated beverages, spicy foods, acidic foods, and foods with high fat content.6,8
Medications
Pharmacologic therapy for GERD includes medications that primarily aim to neutralize or reduce gastric acid -- we summarize options in Table 1.3,8
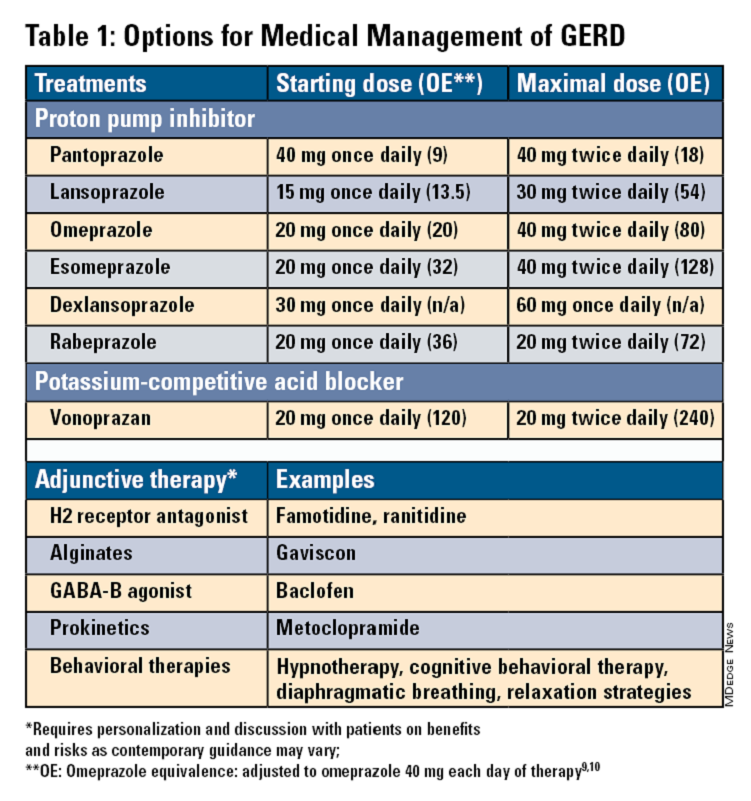
Proton Pump Inhibitors
Most guidelines suggest a trial of 4-8 weeks of once-daily enteric-coated PPI before meals in patients with typical GERD symptoms and no alarm symptoms. Escalation to double-dose PPI may be considered in the case of persistent symptoms. The relative potencies of standard-dose pantoprazole, lansoprazole, esomeprazole, and rabeprazole are presented in Table 1.9 When a PPI switch is needed, rabeprazole may be considered as it is a PPI that does not rely on CYP2C19 for primary metabolism.9
Acid suppression should be weaned down to the lowest effective dose or converted to H2RAs or other antacids once symptoms are sufficiently controlled unless patients have EE, Barrett’s esophagus, or peptic stricture.3 Patients with severe GERD may require long-term PPI therapy or an invasive anti-reflux procedure.
Recent studies have shown that potassium-competitive acid blockers (PCAB) like vonoprazan may offer more effective gastric acid inhibition. While not included in the latest clinical practice update, vonoprazan is thought to be superior to lansoprazole for those with LA Grade C/D esophagitis for both symptom relief and healing at 2 weeks.10
Adjunctive Therapies
Alginates can function as a physical barrier to even neutral reflux and may be helpful for patients with postprandial or nighttime symptoms as well as those with hiatal hernia.3 H2RAs can also help mitigate nighttime symptoms.3 Baclofen is a gamma-aminobutyric acid–B agonist which inhibits transient lower esophageal sphincter relaxation (TLESR) and may be effective for patients with belching.3 Prokinetics may be helpful for GERD with concomitant gastroparesis.3 Sucralfate is a mucosal protective agent, but there is a lack of data supporting its efficacy in GERD treatment. Consider referral to a behavioral therapist for supplemental therapies, hypnotherapy, cognitive-behavior therapy, diaphragmatic breathing, and relaxation strategies for functional heartburn or reflux-associated esophageal hypervigilance or reflux hypersensitivity.3
When to Refer to Higher Level of Care
For patients who do not wish to remain on longer-term pharmacologic therapy or would benefit from anatomic repair, clinicians should have a discussion of risks and benefits prior to consideration of referral for anti-reflux procedures.3,6,8 We advise this conversation should include review of patient health status, postsurgical side effects such as increased flatus, bloating and dysphagia as well as the potential need to still resume PPI post operation.8
Endoscopic Management
Patient Selection And Evaluation
For the groups indicated for a higher level of care, we agree with AGA recommendations, multi-society guidelines, and expert review,3,7,11,12 and highlight potential options in Table 2. Step-up options should be based on patient characteristics and reviewed carefully with patients. Endoscopic therapies are less invasive than surgery and may be considered for those who do not require anatomic repair of hiatal hernia, do not want surgery, or are not suitable for surgery.
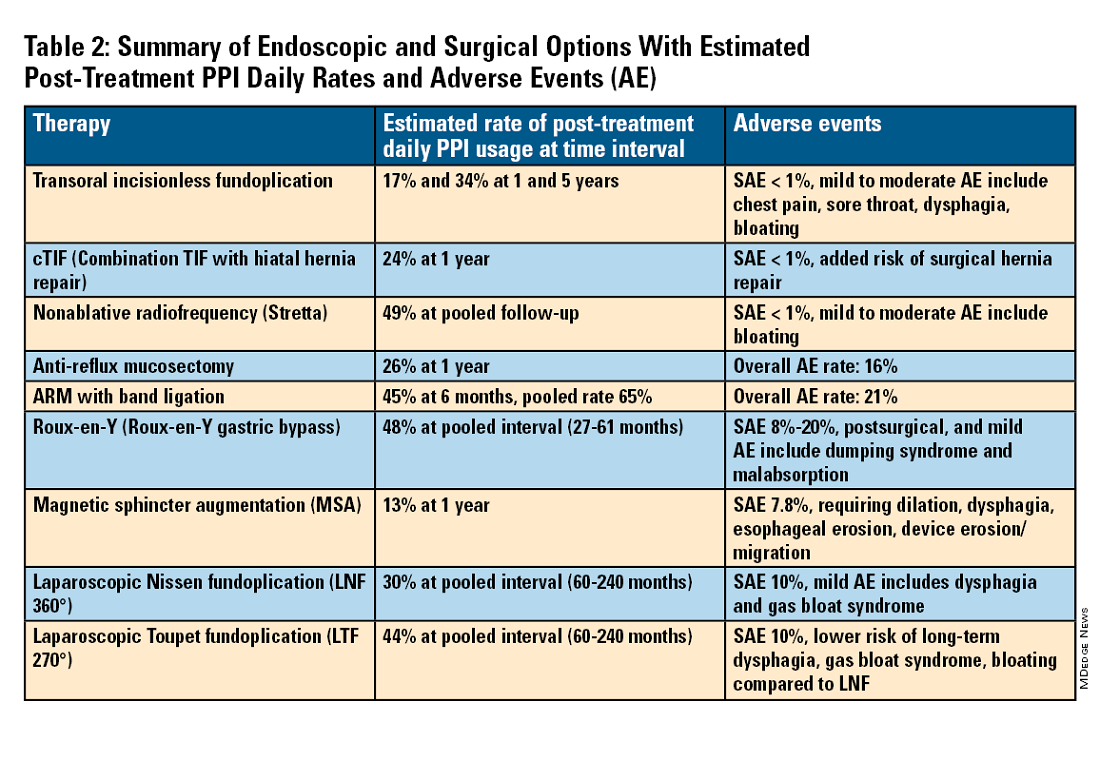
The pathophysiology of GERD is from a loss of the anti-reflux barrier of the esophageal gastric junction (EGJ) at the lower esophageal sphincter (LES) leading to unintended retrograde movement of gastric contents.6 Anatomically, the LES is composed of muscles of the distal esophagus and sling fibers of the proximal stomach, the “external valve” from the diaphragmatic crura, and the “internal valve” from the gastroesophageal flap valve (GEFV). GERD occurs from mechanical failure of the LES. First, there may be disproportional dilation of the diaphragmatic crura as categorized by Hill Grade of the GEFV as seen by a retroflexed view of EGJ after 30-45 seconds of insufflation.13 Second, there may be a migration of the LES away from the diaphragmatic crura as in the case of a hiatal hernia. Provocative maneuvers may reveal a sliding hernia by gentle retraction of the endoscope while under retroflexed view.13 Third, there may be more frequent TLESR associated with GERD.12
The aim of most interventions is to restore competency of the LES by reconstruction of the GEFV via suture or staple-based approximation of tissue.11,12 Intraluminal therapy may only target the GEFV at the internal valve. Therefore, most endoscopic interventions are limited to patients with intact diaphragmatic crura (ie, small to no hiatal hernia and GEFV Hill Grade 1 to 2). Contraindications for endoscopic therapy are moderate to severe reflux (ie, LA Grade C/ D), hiatus hernia 2 cm or larger, strictures, or long-segment Barrett’s esophagus.
Utility, Safety, and Outcomes of TIF
Historically, endoscopic therapy targeting endoscopic fundoplication started with EndoLuminal gastro-gastric fundoplication (ELF, 2005) which was a proof of concept of safe manipulation and suture for gastro-gastric plication to below the Z-line. Transoral incisionless fundoplication (TIF) 1.0 was suggested in 2007 for clinical application by proposing a longitudinal oriented esophago-gastric plication 1 cm above the Z-line.
In 2009, TIF2.0 was proposed as a rotational 270° wrap of the cardia and fundus to a full-thickness esophago-gastric fundoplication around 2-4 cm of the distal esophagus. Like a surgical fundoplication, this reinforces sling fibers, increases the Angle of His and improves the cardiac notch. TIF 2.0 is indicated for those with small (< 2 cm) or no hiatal hernia and a GEFV Hill Grade 1 or 2. The present iteration of TIF2.0 uses EsophyX-Z (EndoGastric Solutions; Redmond, Washington) which features dual fastener deployment and a simplified firing mechanism. Plication is secured via nonresorbable polypropylene T-fasteners with strength equivalence of 3-0 sutures.
Compared with the original, TIF2.0 represents a decrease of severe adverse events from 2%-2.5% to 0.4%-1%.11,14 Based on longitudinal TEMPO data, patient satisfaction ranges between 70% and 90% and rates of patients reverting to daily PPI use are 17% and 34% at 1 and 5 years. A 5% reintervention rate was noted to be comparable with surgical reoperation for fundoplication.15 One retrospective evaluation of patients with failed TIF followed by successful cTIF noted that in all failures there was a documented underestimation of a much larger crura defect at time of index procedure.16 Chest pain is common post procedure and patients and collaborating providers should be counseled on the expected course. In our practice, we admit patients for at least 1 postprocedure day and consider scheduling symptom control medications for those with significant pain.
TIF2.0 for Special Populations
Indications for TIF2.0 continue to evolve. In 2017, concomitant TIF2.0 with hiatal hernia repair (cTIF or HH-TIF) for hernia > 2 cm was accepted for expanded use. In one study, cTIF has been shown to have similar outcomes for postprocedural PPI use, dysphagia, wrap disruption, and hiatal hernia recurrence, compared with hiatal hernia repair paired with laparoscopic Nissen fundoplication with possibly shorter postadmission stay, serious adverse events, and bloating.17 A cTIF may be performed in a single general anesthetic session typically with a surgical hiatal hernia repair followed by TIF2.0.
Other Endoscopic Procedures
Several other endoscopic interventions have been proposed for GERD management. The following procedures are under continuous study and should be considered only by those with expertise.
Stretta
The Stretta device (Restech; Houston, Texas) was approved in 2000 for use of a radiofrequency (RF) generator and catheter applied to the squamocolumnar junction under irrigation. Ideal candidates for this nonablative procedure may include patients with confirmed GERD, low-grade EE, without Barrett’s esophagus, small hiatal hernia, and a competent LES with pressure > 5 mmHg. Meta-analysis has yielded conflicting results in terms of its efficacy, compared with TIF2.0, and recent multi-society guidance suggests fundoplication over Stretta.7
ARM, MASE, and RAP
Anti-reflux mucosectomy (ARM) has been proposed based on the observation that patients undergoing mucosectomy for neoplasms in the cardia had improvement of reflux symptoms.11,12 Systematic review has suggested a clinical response of 80% of either PPI discontinuation or reduction, but 17% of adverse events include development of strictures. Iterations of ARM continue to be studied including ARM with band ligation (L-ARM) and endoscopic submucosal dissection for GERD (ESD-G).12
Experts have proposed incorporating endoscopic suturing of the EGJ to modulate the LES. Mucosal ablation and suturing of the EG junction (MASE) has been proposed by first priming tissue via argon plasma coagulation (APC) prior to endoscopic overstitch of two to three interrupted sutures below the EGJ to narrow and elongate the EGJ. The resection and plication (RAP) procedure performs a mucosal resection prior to full-thickness plication of the LES and cardia.11,12 Expert opinion has suggested that RAP may be used in patients with altered anatomy whereas MASE may be used when resection is not possible (eg, prior scarring, resection or ablation).12
Surgical Management
We agree with a recent multi-society guideline recommending that an interdisciplinary consultation with surgery for indicated patients with refractory GERD and underlying hiatal hernia, or who do not want lifelong medical therapy.
Fundoplication creates a surgical wrap to reinforce the LES and may be performed laparoscopically. Contraindications include body mass index (BMI) >35 kg/m2 and significantly impaired dysmotility. Fundoplication of 180°, 270°, and 360° may achieve comparable outcomes, but a laparoscopic toupet fundoplication (LTF 270°) may have fewer postsurgical issues of dysphagia and bloating. Advantages for both anterior and posterior partial fundoplications have been demonstrated by network meta-analysis. Therefore, a multi-society guideline for GERD suggests partial over complete fundoplication.7 Compared with posterior techniques, anterior fundoplication (Watson fundoplication) led to more recurrent reflux symptoms but less dysphagia and other side effects.19
Magnetic sphincter augmentation (MSA) is a surgical option that strengthens the LES with magnets to improve sphincter competence. In addition to listed contraindications of fundoplication, patients with an allergy to nickel and/or titanium are also contraindicated to receive MSA.7 MSA has been suggested to be equivalent to LNF although there may be less gas bloat and greater ability to belch on follow up.20
Surgical Options for Special Populations
Patients with medically refractory GERD and a BMI ≥ 35 kg/m2 may benefit from either Roux-en-Y gastric bypass (RYGB) or fundoplication, however sleeve gastrectomy is not advised.7 In patients with BMI > 50 kg/m2, RYGB may provide an optimal choice. We agree with consultation with a bariatric surgeon when reviewing these situations.
Conclusion
Patients with GERD are commonly encountered worldwide. Empiric PPI are effective mainstays for medical treatment of GERD. Novel PCABs (e.g., vonoprazan) may present new options for GERD with LA Grade C/D esophagitis EE and merit more study. In refractory cases or for patients who do not want long term medical therapy, step-up therapy may be considered via endoscopic or surgical interventions. Patient anatomy and comorbidities should be considered by the clinician to inform treatment options. Surgery may have the most durable outcomes for those requiring step-up therapy. Improvements in technique, devices and patient selection have allowed TIF2.0 to grow as a viable offering with excellent 5-year outcomes for indicated patients.
Dr. Chang, Dr. Tintara, and Dr. Phan are based in the Division of Gastrointestinal and Liver Disease at the University of Southern California in Los Angeles. They have no conflicts of interest to declare.
References
1. Richter JE andRubenstein JH. Gastroenterology. 2018 Jan. doi: 10.1053/j.gastro.2017.07.045.
2. El-Serag HB et al. Gut. 2014 Jun. doi: 10.1136/gutjnl-2012-304269.
3. Yadlapati R et al. Clin Gastroenterol Hepatol. 2022 May. doi: 10.1016/j.cgh.2022.01.025.
4. Vakil N et al. Am J Gastroenterol. 2006 Aug. doi: 10.1111/j.1572-0241.2006.00630.x.
5. Numans ME et al. Ann Intern Med. 2004 Apr. doi: 10.7326/0003-4819-140-7-200404060-00011.
6. Kahrilas PJ et al. Gastroenterology. 2008 Oct. doi: 10.1053/j.gastro.2008.08.045.
7. Slater BJ et al. Surg Endosc. 2023 Feb. doi: 10.1007/s00464-022-09817-3.
8. Gyawali CP et al. Gut. 2018 Jul. doi:10.1136/gutjnl-2017-314722.
9. Graham DY and Tansel A. Clin Gastroenterol Hepatol. 2018 Jun. doi: 10.1016/j.cgh.2017.09.033.
10. Graham DY and Dore MP. Gastroenterology. 2018 Feb. doi:10.1053/j.gastro.2018.01.018.
11. Haseeb M and Thompson CC. Curr Opin Gastroenterol. 2023 Sep. doi: 10.1097/MOG.0000000000000968.
12. Kolb JM and Chang KJ. Curr Opin Gastroenterol. 2023 Jul. doi:10.1097/MOG.0000000000000944.
13. Nguyen NT et al. Foregut. 2022 Sep. doi: 10.1177/26345161221126961.
14. Mazzoleni G et al. Endosc Int Open. 2021 Feb. doi: 10.1055/a-1322-2209.
15. Trad KS et al. Surg Innov. 2018 Apr. doi: 10.1177/1553350618755214.
16. Kolb JM et al. Gastroenterology. 2021 May. doi: 10.1016/S0016-5085(21)02953-X.
17. Jaruvongvanich VK et al. Endosc Int Open. 2023 Jan. doi: 10.1055/a-1972-9190.
18. Lee Y et al. Surg Endosc. 2023 Jul. doi: 10.1007/s00464-023-10151-5.
19. Andreou A et al. Surg Endosc. 2020 Feb. doi: 10.1007/s00464-019-07208-9.
20. Guidozzi N et al. Dis Esophagus. 2019 Nov. doi: 10.1093/dote/doz031.
Introduction
Gastroesophageal reflux disease (GERD) is a frequently encountered condition, and rising annually.1 A recent meta-analysis suggests nearly 14% (1.03 billion) of the population are affected worldwide. Differences may range by region from 12% in Latin America to 20% in North America, and by country from 4% in China to 23% in Turkey.1 In the United States, 21% of the population are afflicted with weekly GERD symptoms.2 Novel medical therapies and endoscopic options provide clinicians with opportunities to help patients with GERD.3
Diagnosis
Definition
GERD was originally defined by the Montreal consensus as a condition that develops when the reflux of stomach contents causes troublesome symptoms and/or complications.4 Heartburn and regurgitation are common symptoms of GERD, with a sensitivity of 30%-76% and specificity of 62%-96% for erosive esophagitis (EE), which occurs when the reflux of stomach content causes esophageal mucosal breaks.5 The presence of characteristic mucosal injury observed during an upper endoscopy or abnormal esophageal acid exposure on ambulatory reflux monitoring are objective evidence of GERD. A trial of a proton pump inhibitor (PPI) may function as a diagnostic test for patients exhibiting the typical symptoms of GERD without any alarm symptoms.3,6
Endoscopic Evaluation and Confirmation
The 2022 American Gastroenterological Association (AGA) clinical practice update recommends diagnostic endoscopy, after PPIs are stopped for 2-4 weeks, in patients whose GERD symptoms do not respond adequately to an empiric trial of a PPI.3 Those with GERD and alarm symptoms such as dysphagia, weight loss, bleeding, and vomiting should undergo endoscopy as soon as possible. Endoscopic findings of EE (Los Angeles Grade B or more severe) and long-segment Barrett’s esophagus (> 3-cm segment with intestinal metaplasia on biopsy) are diagnostic of GERD.3
Reflux Monitoring
With ambulatory reflux monitoring (pH or impedance-pH), esophageal acid exposure (or neutral refluxate in impedance testing) can be measured to confirm GERD diagnosis and to correlate symptoms with reflux episodes. Patients with atypical GERD symptoms or patients with a confirmed diagnosis of GERD whose symptoms have not improved sufficiently with twice-daily PPI therapy should have esophageal impedance-pH monitoring while on PPIs.6,7
Esophageal Manometry
High-resolution esophageal manometry can be used to assess motility abnormalities associated with GERD.
Although no manometric abnormality is unique to GERD, weak lower esophageal sphincter (LES) resting pressure and ineffective esophageal motility frequently coexist with severe GERD.6
Manometry is particularly useful in patients considering surgical or endoscopic anti-reflux procedures to evaluate for achalasia,3 an important contraindication to surgery.
Medical Management
Management of GERD requires a multidisciplinary and personalized approach based on symptom presentation, body mass index, endoscopic findings (e.g., presence of EE, Barrett’s esophagus, hiatal hernia), and physiological abnormalities (e.g., gastroparesis or ineffective motility).3
Lifestyle Modifications
Recommended lifestyle modifications include weight loss for patients with obesity, stress reduction, tobacco and alcohol cessation, elevating the head of the bed, staying upright during and after meals, avoidance of food intake < 3 hours before bedtime, and cessation of foods that potentially aggravate reflux symptoms such as coffee, chocolate, carbonated beverages, spicy foods, acidic foods, and foods with high fat content.6,8
Medications
Pharmacologic therapy for GERD includes medications that primarily aim to neutralize or reduce gastric acid -- we summarize options in Table 1.3,8

Proton Pump Inhibitors
Most guidelines suggest a trial of 4-8 weeks of once-daily enteric-coated PPI before meals in patients with typical GERD symptoms and no alarm symptoms. Escalation to double-dose PPI may be considered in the case of persistent symptoms. The relative potencies of standard-dose pantoprazole, lansoprazole, esomeprazole, and rabeprazole are presented in Table 1.9 When a PPI switch is needed, rabeprazole may be considered as it is a PPI that does not rely on CYP2C19 for primary metabolism.9
Acid suppression should be weaned down to the lowest effective dose or converted to H2RAs or other antacids once symptoms are sufficiently controlled unless patients have EE, Barrett’s esophagus, or peptic stricture.3 Patients with severe GERD may require long-term PPI therapy or an invasive anti-reflux procedure.
Recent studies have shown that potassium-competitive acid blockers (PCAB) like vonoprazan may offer more effective gastric acid inhibition. While not included in the latest clinical practice update, vonoprazan is thought to be superior to lansoprazole for those with LA Grade C/D esophagitis for both symptom relief and healing at 2 weeks.10
Adjunctive Therapies
Alginates can function as a physical barrier to even neutral reflux and may be helpful for patients with postprandial or nighttime symptoms as well as those with hiatal hernia.3 H2RAs can also help mitigate nighttime symptoms.3 Baclofen is a gamma-aminobutyric acid–B agonist which inhibits transient lower esophageal sphincter relaxation (TLESR) and may be effective for patients with belching.3 Prokinetics may be helpful for GERD with concomitant gastroparesis.3 Sucralfate is a mucosal protective agent, but there is a lack of data supporting its efficacy in GERD treatment. Consider referral to a behavioral therapist for supplemental therapies, hypnotherapy, cognitive-behavior therapy, diaphragmatic breathing, and relaxation strategies for functional heartburn or reflux-associated esophageal hypervigilance or reflux hypersensitivity.3
When to Refer to Higher Level of Care
For patients who do not wish to remain on longer-term pharmacologic therapy or would benefit from anatomic repair, clinicians should have a discussion of risks and benefits prior to consideration of referral for anti-reflux procedures.3,6,8 We advise this conversation should include review of patient health status, postsurgical side effects such as increased flatus, bloating and dysphagia as well as the potential need to still resume PPI post operation.8
Endoscopic Management
Patient Selection And Evaluation
For the groups indicated for a higher level of care, we agree with AGA recommendations, multi-society guidelines, and expert review,3,7,11,12 and highlight potential options in Table 2. Step-up options should be based on patient characteristics and reviewed carefully with patients. Endoscopic therapies are less invasive than surgery and may be considered for those who do not require anatomic repair of hiatal hernia, do not want surgery, or are not suitable for surgery.

The pathophysiology of GERD is from a loss of the anti-reflux barrier of the esophageal gastric junction (EGJ) at the lower esophageal sphincter (LES) leading to unintended retrograde movement of gastric contents.6 Anatomically, the LES is composed of muscles of the distal esophagus and sling fibers of the proximal stomach, the “external valve” from the diaphragmatic crura, and the “internal valve” from the gastroesophageal flap valve (GEFV). GERD occurs from mechanical failure of the LES. First, there may be disproportional dilation of the diaphragmatic crura as categorized by Hill Grade of the GEFV as seen by a retroflexed view of EGJ after 30-45 seconds of insufflation.13 Second, there may be a migration of the LES away from the diaphragmatic crura as in the case of a hiatal hernia. Provocative maneuvers may reveal a sliding hernia by gentle retraction of the endoscope while under retroflexed view.13 Third, there may be more frequent TLESR associated with GERD.12
The aim of most interventions is to restore competency of the LES by reconstruction of the GEFV via suture or staple-based approximation of tissue.11,12 Intraluminal therapy may only target the GEFV at the internal valve. Therefore, most endoscopic interventions are limited to patients with intact diaphragmatic crura (ie, small to no hiatal hernia and GEFV Hill Grade 1 to 2). Contraindications for endoscopic therapy are moderate to severe reflux (ie, LA Grade C/ D), hiatus hernia 2 cm or larger, strictures, or long-segment Barrett’s esophagus.
Utility, Safety, and Outcomes of TIF
Historically, endoscopic therapy targeting endoscopic fundoplication started with EndoLuminal gastro-gastric fundoplication (ELF, 2005) which was a proof of concept of safe manipulation and suture for gastro-gastric plication to below the Z-line. Transoral incisionless fundoplication (TIF) 1.0 was suggested in 2007 for clinical application by proposing a longitudinal oriented esophago-gastric plication 1 cm above the Z-line.
In 2009, TIF2.0 was proposed as a rotational 270° wrap of the cardia and fundus to a full-thickness esophago-gastric fundoplication around 2-4 cm of the distal esophagus. Like a surgical fundoplication, this reinforces sling fibers, increases the Angle of His and improves the cardiac notch. TIF 2.0 is indicated for those with small (< 2 cm) or no hiatal hernia and a GEFV Hill Grade 1 or 2. The present iteration of TIF2.0 uses EsophyX-Z (EndoGastric Solutions; Redmond, Washington) which features dual fastener deployment and a simplified firing mechanism. Plication is secured via nonresorbable polypropylene T-fasteners with strength equivalence of 3-0 sutures.
Compared with the original, TIF2.0 represents a decrease of severe adverse events from 2%-2.5% to 0.4%-1%.11,14 Based on longitudinal TEMPO data, patient satisfaction ranges between 70% and 90% and rates of patients reverting to daily PPI use are 17% and 34% at 1 and 5 years. A 5% reintervention rate was noted to be comparable with surgical reoperation for fundoplication.15 One retrospective evaluation of patients with failed TIF followed by successful cTIF noted that in all failures there was a documented underestimation of a much larger crura defect at time of index procedure.16 Chest pain is common post procedure and patients and collaborating providers should be counseled on the expected course. In our practice, we admit patients for at least 1 postprocedure day and consider scheduling symptom control medications for those with significant pain.
TIF2.0 for Special Populations
Indications for TIF2.0 continue to evolve. In 2017, concomitant TIF2.0 with hiatal hernia repair (cTIF or HH-TIF) for hernia > 2 cm was accepted for expanded use. In one study, cTIF has been shown to have similar outcomes for postprocedural PPI use, dysphagia, wrap disruption, and hiatal hernia recurrence, compared with hiatal hernia repair paired with laparoscopic Nissen fundoplication with possibly shorter postadmission stay, serious adverse events, and bloating.17 A cTIF may be performed in a single general anesthetic session typically with a surgical hiatal hernia repair followed by TIF2.0.
Other Endoscopic Procedures
Several other endoscopic interventions have been proposed for GERD management. The following procedures are under continuous study and should be considered only by those with expertise.
Stretta
The Stretta device (Restech; Houston, Texas) was approved in 2000 for use of a radiofrequency (RF) generator and catheter applied to the squamocolumnar junction under irrigation. Ideal candidates for this nonablative procedure may include patients with confirmed GERD, low-grade EE, without Barrett’s esophagus, small hiatal hernia, and a competent LES with pressure > 5 mmHg. Meta-analysis has yielded conflicting results in terms of its efficacy, compared with TIF2.0, and recent multi-society guidance suggests fundoplication over Stretta.7
ARM, MASE, and RAP
Anti-reflux mucosectomy (ARM) has been proposed based on the observation that patients undergoing mucosectomy for neoplasms in the cardia had improvement of reflux symptoms.11,12 Systematic review has suggested a clinical response of 80% of either PPI discontinuation or reduction, but 17% of adverse events include development of strictures. Iterations of ARM continue to be studied including ARM with band ligation (L-ARM) and endoscopic submucosal dissection for GERD (ESD-G).12
Experts have proposed incorporating endoscopic suturing of the EGJ to modulate the LES. Mucosal ablation and suturing of the EG junction (MASE) has been proposed by first priming tissue via argon plasma coagulation (APC) prior to endoscopic overstitch of two to three interrupted sutures below the EGJ to narrow and elongate the EGJ. The resection and plication (RAP) procedure performs a mucosal resection prior to full-thickness plication of the LES and cardia.11,12 Expert opinion has suggested that RAP may be used in patients with altered anatomy whereas MASE may be used when resection is not possible (eg, prior scarring, resection or ablation).12
Surgical Management
We agree with a recent multi-society guideline recommending that an interdisciplinary consultation with surgery for indicated patients with refractory GERD and underlying hiatal hernia, or who do not want lifelong medical therapy.
Fundoplication creates a surgical wrap to reinforce the LES and may be performed laparoscopically. Contraindications include body mass index (BMI) >35 kg/m2 and significantly impaired dysmotility. Fundoplication of 180°, 270°, and 360° may achieve comparable outcomes, but a laparoscopic toupet fundoplication (LTF 270°) may have fewer postsurgical issues of dysphagia and bloating. Advantages for both anterior and posterior partial fundoplications have been demonstrated by network meta-analysis. Therefore, a multi-society guideline for GERD suggests partial over complete fundoplication.7 Compared with posterior techniques, anterior fundoplication (Watson fundoplication) led to more recurrent reflux symptoms but less dysphagia and other side effects.19
Magnetic sphincter augmentation (MSA) is a surgical option that strengthens the LES with magnets to improve sphincter competence. In addition to listed contraindications of fundoplication, patients with an allergy to nickel and/or titanium are also contraindicated to receive MSA.7 MSA has been suggested to be equivalent to LNF although there may be less gas bloat and greater ability to belch on follow up.20
Surgical Options for Special Populations
Patients with medically refractory GERD and a BMI ≥ 35 kg/m2 may benefit from either Roux-en-Y gastric bypass (RYGB) or fundoplication, however sleeve gastrectomy is not advised.7 In patients with BMI > 50 kg/m2, RYGB may provide an optimal choice. We agree with consultation with a bariatric surgeon when reviewing these situations.
Conclusion
Patients with GERD are commonly encountered worldwide. Empiric PPI are effective mainstays for medical treatment of GERD. Novel PCABs (e.g., vonoprazan) may present new options for GERD with LA Grade C/D esophagitis EE and merit more study. In refractory cases or for patients who do not want long term medical therapy, step-up therapy may be considered via endoscopic or surgical interventions. Patient anatomy and comorbidities should be considered by the clinician to inform treatment options. Surgery may have the most durable outcomes for those requiring step-up therapy. Improvements in technique, devices and patient selection have allowed TIF2.0 to grow as a viable offering with excellent 5-year outcomes for indicated patients.
Dr. Chang, Dr. Tintara, and Dr. Phan are based in the Division of Gastrointestinal and Liver Disease at the University of Southern California in Los Angeles. They have no conflicts of interest to declare.
References
1. Richter JE andRubenstein JH. Gastroenterology. 2018 Jan. doi: 10.1053/j.gastro.2017.07.045.
2. El-Serag HB et al. Gut. 2014 Jun. doi: 10.1136/gutjnl-2012-304269.
3. Yadlapati R et al. Clin Gastroenterol Hepatol. 2022 May. doi: 10.1016/j.cgh.2022.01.025.
4. Vakil N et al. Am J Gastroenterol. 2006 Aug. doi: 10.1111/j.1572-0241.2006.00630.x.
5. Numans ME et al. Ann Intern Med. 2004 Apr. doi: 10.7326/0003-4819-140-7-200404060-00011.
6. Kahrilas PJ et al. Gastroenterology. 2008 Oct. doi: 10.1053/j.gastro.2008.08.045.
7. Slater BJ et al. Surg Endosc. 2023 Feb. doi: 10.1007/s00464-022-09817-3.
8. Gyawali CP et al. Gut. 2018 Jul. doi:10.1136/gutjnl-2017-314722.
9. Graham DY and Tansel A. Clin Gastroenterol Hepatol. 2018 Jun. doi: 10.1016/j.cgh.2017.09.033.
10. Graham DY and Dore MP. Gastroenterology. 2018 Feb. doi:10.1053/j.gastro.2018.01.018.
11. Haseeb M and Thompson CC. Curr Opin Gastroenterol. 2023 Sep. doi: 10.1097/MOG.0000000000000968.
12. Kolb JM and Chang KJ. Curr Opin Gastroenterol. 2023 Jul. doi:10.1097/MOG.0000000000000944.
13. Nguyen NT et al. Foregut. 2022 Sep. doi: 10.1177/26345161221126961.
14. Mazzoleni G et al. Endosc Int Open. 2021 Feb. doi: 10.1055/a-1322-2209.
15. Trad KS et al. Surg Innov. 2018 Apr. doi: 10.1177/1553350618755214.
16. Kolb JM et al. Gastroenterology. 2021 May. doi: 10.1016/S0016-5085(21)02953-X.
17. Jaruvongvanich VK et al. Endosc Int Open. 2023 Jan. doi: 10.1055/a-1972-9190.
18. Lee Y et al. Surg Endosc. 2023 Jul. doi: 10.1007/s00464-023-10151-5.
19. Andreou A et al. Surg Endosc. 2020 Feb. doi: 10.1007/s00464-019-07208-9.
20. Guidozzi N et al. Dis Esophagus. 2019 Nov. doi: 10.1093/dote/doz031.
Introduction
Gastroesophageal reflux disease (GERD) is a frequently encountered condition, and rising annually.1 A recent meta-analysis suggests nearly 14% (1.03 billion) of the population are affected worldwide. Differences may range by region from 12% in Latin America to 20% in North America, and by country from 4% in China to 23% in Turkey.1 In the United States, 21% of the population are afflicted with weekly GERD symptoms.2 Novel medical therapies and endoscopic options provide clinicians with opportunities to help patients with GERD.3
Diagnosis
Definition
GERD was originally defined by the Montreal consensus as a condition that develops when the reflux of stomach contents causes troublesome symptoms and/or complications.4 Heartburn and regurgitation are common symptoms of GERD, with a sensitivity of 30%-76% and specificity of 62%-96% for erosive esophagitis (EE), which occurs when the reflux of stomach content causes esophageal mucosal breaks.5 The presence of characteristic mucosal injury observed during an upper endoscopy or abnormal esophageal acid exposure on ambulatory reflux monitoring are objective evidence of GERD. A trial of a proton pump inhibitor (PPI) may function as a diagnostic test for patients exhibiting the typical symptoms of GERD without any alarm symptoms.3,6
Endoscopic Evaluation and Confirmation
The 2022 American Gastroenterological Association (AGA) clinical practice update recommends diagnostic endoscopy, after PPIs are stopped for 2-4 weeks, in patients whose GERD symptoms do not respond adequately to an empiric trial of a PPI.3 Those with GERD and alarm symptoms such as dysphagia, weight loss, bleeding, and vomiting should undergo endoscopy as soon as possible. Endoscopic findings of EE (Los Angeles Grade B or more severe) and long-segment Barrett’s esophagus (> 3-cm segment with intestinal metaplasia on biopsy) are diagnostic of GERD.3
Reflux Monitoring
With ambulatory reflux monitoring (pH or impedance-pH), esophageal acid exposure (or neutral refluxate in impedance testing) can be measured to confirm GERD diagnosis and to correlate symptoms with reflux episodes. Patients with atypical GERD symptoms or patients with a confirmed diagnosis of GERD whose symptoms have not improved sufficiently with twice-daily PPI therapy should have esophageal impedance-pH monitoring while on PPIs.6,7
Esophageal Manometry
High-resolution esophageal manometry can be used to assess motility abnormalities associated with GERD.
Although no manometric abnormality is unique to GERD, weak lower esophageal sphincter (LES) resting pressure and ineffective esophageal motility frequently coexist with severe GERD.6
Manometry is particularly useful in patients considering surgical or endoscopic anti-reflux procedures to evaluate for achalasia,3 an important contraindication to surgery.
Medical Management
Management of GERD requires a multidisciplinary and personalized approach based on symptom presentation, body mass index, endoscopic findings (e.g., presence of EE, Barrett’s esophagus, hiatal hernia), and physiological abnormalities (e.g., gastroparesis or ineffective motility).3
Lifestyle Modifications
Recommended lifestyle modifications include weight loss for patients with obesity, stress reduction, tobacco and alcohol cessation, elevating the head of the bed, staying upright during and after meals, avoidance of food intake < 3 hours before bedtime, and cessation of foods that potentially aggravate reflux symptoms such as coffee, chocolate, carbonated beverages, spicy foods, acidic foods, and foods with high fat content.6,8
Medications
Pharmacologic therapy for GERD includes medications that primarily aim to neutralize or reduce gastric acid -- we summarize options in Table 1.3,8

Proton Pump Inhibitors
Most guidelines suggest a trial of 4-8 weeks of once-daily enteric-coated PPI before meals in patients with typical GERD symptoms and no alarm symptoms. Escalation to double-dose PPI may be considered in the case of persistent symptoms. The relative potencies of standard-dose pantoprazole, lansoprazole, esomeprazole, and rabeprazole are presented in Table 1.9 When a PPI switch is needed, rabeprazole may be considered as it is a PPI that does not rely on CYP2C19 for primary metabolism.9
Acid suppression should be weaned down to the lowest effective dose or converted to H2RAs or other antacids once symptoms are sufficiently controlled unless patients have EE, Barrett’s esophagus, or peptic stricture.3 Patients with severe GERD may require long-term PPI therapy or an invasive anti-reflux procedure.
Recent studies have shown that potassium-competitive acid blockers (PCAB) like vonoprazan may offer more effective gastric acid inhibition. While not included in the latest clinical practice update, vonoprazan is thought to be superior to lansoprazole for those with LA Grade C/D esophagitis for both symptom relief and healing at 2 weeks.10
Adjunctive Therapies
Alginates can function as a physical barrier to even neutral reflux and may be helpful for patients with postprandial or nighttime symptoms as well as those with hiatal hernia.3 H2RAs can also help mitigate nighttime symptoms.3 Baclofen is a gamma-aminobutyric acid–B agonist which inhibits transient lower esophageal sphincter relaxation (TLESR) and may be effective for patients with belching.3 Prokinetics may be helpful for GERD with concomitant gastroparesis.3 Sucralfate is a mucosal protective agent, but there is a lack of data supporting its efficacy in GERD treatment. Consider referral to a behavioral therapist for supplemental therapies, hypnotherapy, cognitive-behavior therapy, diaphragmatic breathing, and relaxation strategies for functional heartburn or reflux-associated esophageal hypervigilance or reflux hypersensitivity.3
When to Refer to Higher Level of Care
For patients who do not wish to remain on longer-term pharmacologic therapy or would benefit from anatomic repair, clinicians should have a discussion of risks and benefits prior to consideration of referral for anti-reflux procedures.3,6,8 We advise this conversation should include review of patient health status, postsurgical side effects such as increased flatus, bloating and dysphagia as well as the potential need to still resume PPI post operation.8
Endoscopic Management
Patient Selection And Evaluation
For the groups indicated for a higher level of care, we agree with AGA recommendations, multi-society guidelines, and expert review,3,7,11,12 and highlight potential options in Table 2. Step-up options should be based on patient characteristics and reviewed carefully with patients. Endoscopic therapies are less invasive than surgery and may be considered for those who do not require anatomic repair of hiatal hernia, do not want surgery, or are not suitable for surgery.

The pathophysiology of GERD is from a loss of the anti-reflux barrier of the esophageal gastric junction (EGJ) at the lower esophageal sphincter (LES) leading to unintended retrograde movement of gastric contents.6 Anatomically, the LES is composed of muscles of the distal esophagus and sling fibers of the proximal stomach, the “external valve” from the diaphragmatic crura, and the “internal valve” from the gastroesophageal flap valve (GEFV). GERD occurs from mechanical failure of the LES. First, there may be disproportional dilation of the diaphragmatic crura as categorized by Hill Grade of the GEFV as seen by a retroflexed view of EGJ after 30-45 seconds of insufflation.13 Second, there may be a migration of the LES away from the diaphragmatic crura as in the case of a hiatal hernia. Provocative maneuvers may reveal a sliding hernia by gentle retraction of the endoscope while under retroflexed view.13 Third, there may be more frequent TLESR associated with GERD.12
The aim of most interventions is to restore competency of the LES by reconstruction of the GEFV via suture or staple-based approximation of tissue.11,12 Intraluminal therapy may only target the GEFV at the internal valve. Therefore, most endoscopic interventions are limited to patients with intact diaphragmatic crura (ie, small to no hiatal hernia and GEFV Hill Grade 1 to 2). Contraindications for endoscopic therapy are moderate to severe reflux (ie, LA Grade C/ D), hiatus hernia 2 cm or larger, strictures, or long-segment Barrett’s esophagus.
Utility, Safety, and Outcomes of TIF
Historically, endoscopic therapy targeting endoscopic fundoplication started with EndoLuminal gastro-gastric fundoplication (ELF, 2005) which was a proof of concept of safe manipulation and suture for gastro-gastric plication to below the Z-line. Transoral incisionless fundoplication (TIF) 1.0 was suggested in 2007 for clinical application by proposing a longitudinal oriented esophago-gastric plication 1 cm above the Z-line.
In 2009, TIF2.0 was proposed as a rotational 270° wrap of the cardia and fundus to a full-thickness esophago-gastric fundoplication around 2-4 cm of the distal esophagus. Like a surgical fundoplication, this reinforces sling fibers, increases the Angle of His and improves the cardiac notch. TIF 2.0 is indicated for those with small (< 2 cm) or no hiatal hernia and a GEFV Hill Grade 1 or 2. The present iteration of TIF2.0 uses EsophyX-Z (EndoGastric Solutions; Redmond, Washington) which features dual fastener deployment and a simplified firing mechanism. Plication is secured via nonresorbable polypropylene T-fasteners with strength equivalence of 3-0 sutures.
Compared with the original, TIF2.0 represents a decrease of severe adverse events from 2%-2.5% to 0.4%-1%.11,14 Based on longitudinal TEMPO data, patient satisfaction ranges between 70% and 90% and rates of patients reverting to daily PPI use are 17% and 34% at 1 and 5 years. A 5% reintervention rate was noted to be comparable with surgical reoperation for fundoplication.15 One retrospective evaluation of patients with failed TIF followed by successful cTIF noted that in all failures there was a documented underestimation of a much larger crura defect at time of index procedure.16 Chest pain is common post procedure and patients and collaborating providers should be counseled on the expected course. In our practice, we admit patients for at least 1 postprocedure day and consider scheduling symptom control medications for those with significant pain.
TIF2.0 for Special Populations
Indications for TIF2.0 continue to evolve. In 2017, concomitant TIF2.0 with hiatal hernia repair (cTIF or HH-TIF) for hernia > 2 cm was accepted for expanded use. In one study, cTIF has been shown to have similar outcomes for postprocedural PPI use, dysphagia, wrap disruption, and hiatal hernia recurrence, compared with hiatal hernia repair paired with laparoscopic Nissen fundoplication with possibly shorter postadmission stay, serious adverse events, and bloating.17 A cTIF may be performed in a single general anesthetic session typically with a surgical hiatal hernia repair followed by TIF2.0.
Other Endoscopic Procedures
Several other endoscopic interventions have been proposed for GERD management. The following procedures are under continuous study and should be considered only by those with expertise.
Stretta
The Stretta device (Restech; Houston, Texas) was approved in 2000 for use of a radiofrequency (RF) generator and catheter applied to the squamocolumnar junction under irrigation. Ideal candidates for this nonablative procedure may include patients with confirmed GERD, low-grade EE, without Barrett’s esophagus, small hiatal hernia, and a competent LES with pressure > 5 mmHg. Meta-analysis has yielded conflicting results in terms of its efficacy, compared with TIF2.0, and recent multi-society guidance suggests fundoplication over Stretta.7
ARM, MASE, and RAP
Anti-reflux mucosectomy (ARM) has been proposed based on the observation that patients undergoing mucosectomy for neoplasms in the cardia had improvement of reflux symptoms.11,12 Systematic review has suggested a clinical response of 80% of either PPI discontinuation or reduction, but 17% of adverse events include development of strictures. Iterations of ARM continue to be studied including ARM with band ligation (L-ARM) and endoscopic submucosal dissection for GERD (ESD-G).12
Experts have proposed incorporating endoscopic suturing of the EGJ to modulate the LES. Mucosal ablation and suturing of the EG junction (MASE) has been proposed by first priming tissue via argon plasma coagulation (APC) prior to endoscopic overstitch of two to three interrupted sutures below the EGJ to narrow and elongate the EGJ. The resection and plication (RAP) procedure performs a mucosal resection prior to full-thickness plication of the LES and cardia.11,12 Expert opinion has suggested that RAP may be used in patients with altered anatomy whereas MASE may be used when resection is not possible (eg, prior scarring, resection or ablation).12
Surgical Management
We agree with a recent multi-society guideline recommending that an interdisciplinary consultation with surgery for indicated patients with refractory GERD and underlying hiatal hernia, or who do not want lifelong medical therapy.
Fundoplication creates a surgical wrap to reinforce the LES and may be performed laparoscopically. Contraindications include body mass index (BMI) >35 kg/m2 and significantly impaired dysmotility. Fundoplication of 180°, 270°, and 360° may achieve comparable outcomes, but a laparoscopic toupet fundoplication (LTF 270°) may have fewer postsurgical issues of dysphagia and bloating. Advantages for both anterior and posterior partial fundoplications have been demonstrated by network meta-analysis. Therefore, a multi-society guideline for GERD suggests partial over complete fundoplication.7 Compared with posterior techniques, anterior fundoplication (Watson fundoplication) led to more recurrent reflux symptoms but less dysphagia and other side effects.19
Magnetic sphincter augmentation (MSA) is a surgical option that strengthens the LES with magnets to improve sphincter competence. In addition to listed contraindications of fundoplication, patients with an allergy to nickel and/or titanium are also contraindicated to receive MSA.7 MSA has been suggested to be equivalent to LNF although there may be less gas bloat and greater ability to belch on follow up.20
Surgical Options for Special Populations
Patients with medically refractory GERD and a BMI ≥ 35 kg/m2 may benefit from either Roux-en-Y gastric bypass (RYGB) or fundoplication, however sleeve gastrectomy is not advised.7 In patients with BMI > 50 kg/m2, RYGB may provide an optimal choice. We agree with consultation with a bariatric surgeon when reviewing these situations.
Conclusion
Patients with GERD are commonly encountered worldwide. Empiric PPI are effective mainstays for medical treatment of GERD. Novel PCABs (e.g., vonoprazan) may present new options for GERD with LA Grade C/D esophagitis EE and merit more study. In refractory cases or for patients who do not want long term medical therapy, step-up therapy may be considered via endoscopic or surgical interventions. Patient anatomy and comorbidities should be considered by the clinician to inform treatment options. Surgery may have the most durable outcomes for those requiring step-up therapy. Improvements in technique, devices and patient selection have allowed TIF2.0 to grow as a viable offering with excellent 5-year outcomes for indicated patients.
Dr. Chang, Dr. Tintara, and Dr. Phan are based in the Division of Gastrointestinal and Liver Disease at the University of Southern California in Los Angeles. They have no conflicts of interest to declare.
References
1. Richter JE andRubenstein JH. Gastroenterology. 2018 Jan. doi: 10.1053/j.gastro.2017.07.045.
2. El-Serag HB et al. Gut. 2014 Jun. doi: 10.1136/gutjnl-2012-304269.
3. Yadlapati R et al. Clin Gastroenterol Hepatol. 2022 May. doi: 10.1016/j.cgh.2022.01.025.
4. Vakil N et al. Am J Gastroenterol. 2006 Aug. doi: 10.1111/j.1572-0241.2006.00630.x.
5. Numans ME et al. Ann Intern Med. 2004 Apr. doi: 10.7326/0003-4819-140-7-200404060-00011.
6. Kahrilas PJ et al. Gastroenterology. 2008 Oct. doi: 10.1053/j.gastro.2008.08.045.
7. Slater BJ et al. Surg Endosc. 2023 Feb. doi: 10.1007/s00464-022-09817-3.
8. Gyawali CP et al. Gut. 2018 Jul. doi:10.1136/gutjnl-2017-314722.
9. Graham DY and Tansel A. Clin Gastroenterol Hepatol. 2018 Jun. doi: 10.1016/j.cgh.2017.09.033.
10. Graham DY and Dore MP. Gastroenterology. 2018 Feb. doi:10.1053/j.gastro.2018.01.018.
11. Haseeb M and Thompson CC. Curr Opin Gastroenterol. 2023 Sep. doi: 10.1097/MOG.0000000000000968.
12. Kolb JM and Chang KJ. Curr Opin Gastroenterol. 2023 Jul. doi:10.1097/MOG.0000000000000944.
13. Nguyen NT et al. Foregut. 2022 Sep. doi: 10.1177/26345161221126961.
14. Mazzoleni G et al. Endosc Int Open. 2021 Feb. doi: 10.1055/a-1322-2209.
15. Trad KS et al. Surg Innov. 2018 Apr. doi: 10.1177/1553350618755214.
16. Kolb JM et al. Gastroenterology. 2021 May. doi: 10.1016/S0016-5085(21)02953-X.
17. Jaruvongvanich VK et al. Endosc Int Open. 2023 Jan. doi: 10.1055/a-1972-9190.
18. Lee Y et al. Surg Endosc. 2023 Jul. doi: 10.1007/s00464-023-10151-5.
19. Andreou A et al. Surg Endosc. 2020 Feb. doi: 10.1007/s00464-019-07208-9.
20. Guidozzi N et al. Dis Esophagus. 2019 Nov. doi: 10.1093/dote/doz031.











