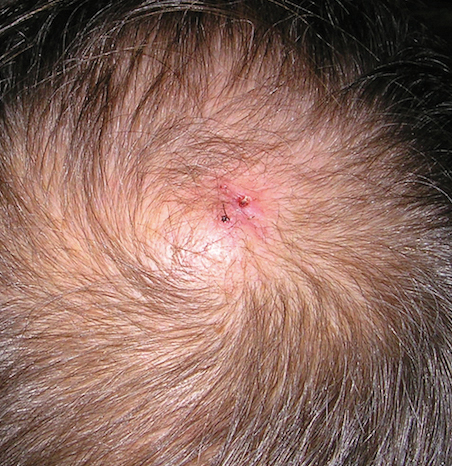
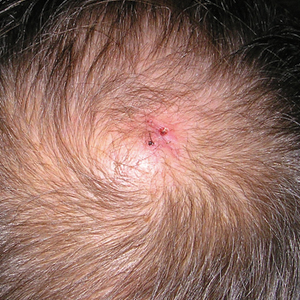
User login
For MD-IQ use only
New AHA scientific statement on menopause and CVD risk
Changes in hormones, body composition, lipids, and vascular health during the menopause transition can increase a woman’s chance of developing cardiovascular disease (CVD) after menopause, the American Heart Association said in a scientific statement.
“This statement aims to raise awareness of both healthcare providers and women about the menopause transition as a time of increasing heart disease risk,” Samar R. El Khoudary, PhD, MPH, who chaired the writing group, said in an interview.
“As such, it emphasizes the importance of monitoring women’s health during midlife and targeting this stage as a critical window for applying early intervention strategies that aim to maintain a healthy heart and reduce the risk of heart disease,” said Dr. El Khoudary, of the University of Pittsburgh.
The statement was published online Nov. 30 in Circulation.
Evolution in knowledge
During the past 20 years, knowledge of how menopause might contribute to CVD has evolved “dramatically,” Dr. El Khoudary noted. The accumulated data consistently point to the menopause transition as a time of change in heart health.
“Importantly,” she said, the latest AHA guidelines for CVD prevention in women, published in 2011, do not include data now available on the menopause transition as a time of increased CVD risk.
“As such, there is a compelling need to discuss the implications of the accumulating body of literature on this topic,” said Dr. El Khoudary.
The statement provides a contemporary synthesis of the existing data on menopause and how it relates to CVD, the leading cause of death of U.S. women.
Earlier age at natural menopause has generally been found to be a marker of greater CVD risk. Iatrogenically induced menopause (bilateral oophorectomy) during the premenopausal period is also associated with higher CVD risk, the data suggest.
Vasomotor symptoms are associated with worse levels of CVD risk factors and measures of subclinical atherosclerosis. Sleep disturbance has also been linked to greater risk for subclinical CVD and worse CV health indexes in women during midlife.
Increases in central/visceral fat and decreases in lean muscle mass are more pronounced during the menopause transition. This increased central adiposity is associated with increased risk for mortality, even among those with normal body mass index, the writing group found.
Increases in lipid levels (LDL cholesterol and apolipoprotein B), metabolic syndrome risk, and vascular remodeling at midlife are driven by the menopause transition more than aging, whereas increases in blood pressure, insulin level, and glucose level are likely more influenced by chronological aging, they reported.
Lifestyle interventions
The writing group noted that, because of the increase in overall life expectancy in the United States, a significant proportion of women will spend up to 40% of their lives after menopause.
Yet data suggest that only 7.2% of women transitioning to menopause are meeting physical activity guidelines and that fewer than 20% of those women are consistently maintaining a healthy diet.
Limited data from randomized, controlled trials suggest that a multidimensional lifestyle intervention during the menopause transition can prevent weight gain and reduce blood pressure and levels of triglycerides, blood glucose, and insulin and reduce the incidence of subclinical carotid atherosclerosis, they pointed out.
“Novel data” indicate a reversal in the associations of HDL cholesterol with CVD risk over the menopause transition, suggesting that higher HDL cholesterol levels may not consistently reflect good cardiovascular health in middle-aged women, the group noted.
There are also data suggesting that starting menopause hormone therapy when younger than 60 years or within 10 years of menopause is associated with reduced CVD risk.
The group said further research is needed into the cardiometabolic effects of menopause hormone therapy, including effects associated with form, route, and duration of administration, in women traversing menopause.
They also noted that data for the primary and secondary prevention of atherosclerotic CVD and improved survival with lipid-lowering interventions “remain elusive” for women and that further study is needed to develop evidence-based recommendations tailored specifically to women.
The research had no commercial funding. Dr. El Khoudary has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Changes in hormones, body composition, lipids, and vascular health during the menopause transition can increase a woman’s chance of developing cardiovascular disease (CVD) after menopause, the American Heart Association said in a scientific statement.
“This statement aims to raise awareness of both healthcare providers and women about the menopause transition as a time of increasing heart disease risk,” Samar R. El Khoudary, PhD, MPH, who chaired the writing group, said in an interview.
“As such, it emphasizes the importance of monitoring women’s health during midlife and targeting this stage as a critical window for applying early intervention strategies that aim to maintain a healthy heart and reduce the risk of heart disease,” said Dr. El Khoudary, of the University of Pittsburgh.
The statement was published online Nov. 30 in Circulation.
Evolution in knowledge
During the past 20 years, knowledge of how menopause might contribute to CVD has evolved “dramatically,” Dr. El Khoudary noted. The accumulated data consistently point to the menopause transition as a time of change in heart health.
“Importantly,” she said, the latest AHA guidelines for CVD prevention in women, published in 2011, do not include data now available on the menopause transition as a time of increased CVD risk.
“As such, there is a compelling need to discuss the implications of the accumulating body of literature on this topic,” said Dr. El Khoudary.
The statement provides a contemporary synthesis of the existing data on menopause and how it relates to CVD, the leading cause of death of U.S. women.
Earlier age at natural menopause has generally been found to be a marker of greater CVD risk. Iatrogenically induced menopause (bilateral oophorectomy) during the premenopausal period is also associated with higher CVD risk, the data suggest.
Vasomotor symptoms are associated with worse levels of CVD risk factors and measures of subclinical atherosclerosis. Sleep disturbance has also been linked to greater risk for subclinical CVD and worse CV health indexes in women during midlife.
Increases in central/visceral fat and decreases in lean muscle mass are more pronounced during the menopause transition. This increased central adiposity is associated with increased risk for mortality, even among those with normal body mass index, the writing group found.
Increases in lipid levels (LDL cholesterol and apolipoprotein B), metabolic syndrome risk, and vascular remodeling at midlife are driven by the menopause transition more than aging, whereas increases in blood pressure, insulin level, and glucose level are likely more influenced by chronological aging, they reported.
Lifestyle interventions
The writing group noted that, because of the increase in overall life expectancy in the United States, a significant proportion of women will spend up to 40% of their lives after menopause.
Yet data suggest that only 7.2% of women transitioning to menopause are meeting physical activity guidelines and that fewer than 20% of those women are consistently maintaining a healthy diet.
Limited data from randomized, controlled trials suggest that a multidimensional lifestyle intervention during the menopause transition can prevent weight gain and reduce blood pressure and levels of triglycerides, blood glucose, and insulin and reduce the incidence of subclinical carotid atherosclerosis, they pointed out.
“Novel data” indicate a reversal in the associations of HDL cholesterol with CVD risk over the menopause transition, suggesting that higher HDL cholesterol levels may not consistently reflect good cardiovascular health in middle-aged women, the group noted.
There are also data suggesting that starting menopause hormone therapy when younger than 60 years or within 10 years of menopause is associated with reduced CVD risk.
The group said further research is needed into the cardiometabolic effects of menopause hormone therapy, including effects associated with form, route, and duration of administration, in women traversing menopause.
They also noted that data for the primary and secondary prevention of atherosclerotic CVD and improved survival with lipid-lowering interventions “remain elusive” for women and that further study is needed to develop evidence-based recommendations tailored specifically to women.
The research had no commercial funding. Dr. El Khoudary has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Changes in hormones, body composition, lipids, and vascular health during the menopause transition can increase a woman’s chance of developing cardiovascular disease (CVD) after menopause, the American Heart Association said in a scientific statement.
“This statement aims to raise awareness of both healthcare providers and women about the menopause transition as a time of increasing heart disease risk,” Samar R. El Khoudary, PhD, MPH, who chaired the writing group, said in an interview.
“As such, it emphasizes the importance of monitoring women’s health during midlife and targeting this stage as a critical window for applying early intervention strategies that aim to maintain a healthy heart and reduce the risk of heart disease,” said Dr. El Khoudary, of the University of Pittsburgh.
The statement was published online Nov. 30 in Circulation.
Evolution in knowledge
During the past 20 years, knowledge of how menopause might contribute to CVD has evolved “dramatically,” Dr. El Khoudary noted. The accumulated data consistently point to the menopause transition as a time of change in heart health.
“Importantly,” she said, the latest AHA guidelines for CVD prevention in women, published in 2011, do not include data now available on the menopause transition as a time of increased CVD risk.
“As such, there is a compelling need to discuss the implications of the accumulating body of literature on this topic,” said Dr. El Khoudary.
The statement provides a contemporary synthesis of the existing data on menopause and how it relates to CVD, the leading cause of death of U.S. women.
Earlier age at natural menopause has generally been found to be a marker of greater CVD risk. Iatrogenically induced menopause (bilateral oophorectomy) during the premenopausal period is also associated with higher CVD risk, the data suggest.
Vasomotor symptoms are associated with worse levels of CVD risk factors and measures of subclinical atherosclerosis. Sleep disturbance has also been linked to greater risk for subclinical CVD and worse CV health indexes in women during midlife.
Increases in central/visceral fat and decreases in lean muscle mass are more pronounced during the menopause transition. This increased central adiposity is associated with increased risk for mortality, even among those with normal body mass index, the writing group found.
Increases in lipid levels (LDL cholesterol and apolipoprotein B), metabolic syndrome risk, and vascular remodeling at midlife are driven by the menopause transition more than aging, whereas increases in blood pressure, insulin level, and glucose level are likely more influenced by chronological aging, they reported.
Lifestyle interventions
The writing group noted that, because of the increase in overall life expectancy in the United States, a significant proportion of women will spend up to 40% of their lives after menopause.
Yet data suggest that only 7.2% of women transitioning to menopause are meeting physical activity guidelines and that fewer than 20% of those women are consistently maintaining a healthy diet.
Limited data from randomized, controlled trials suggest that a multidimensional lifestyle intervention during the menopause transition can prevent weight gain and reduce blood pressure and levels of triglycerides, blood glucose, and insulin and reduce the incidence of subclinical carotid atherosclerosis, they pointed out.
“Novel data” indicate a reversal in the associations of HDL cholesterol with CVD risk over the menopause transition, suggesting that higher HDL cholesterol levels may not consistently reflect good cardiovascular health in middle-aged women, the group noted.
There are also data suggesting that starting menopause hormone therapy when younger than 60 years or within 10 years of menopause is associated with reduced CVD risk.
The group said further research is needed into the cardiometabolic effects of menopause hormone therapy, including effects associated with form, route, and duration of administration, in women traversing menopause.
They also noted that data for the primary and secondary prevention of atherosclerotic CVD and improved survival with lipid-lowering interventions “remain elusive” for women and that further study is needed to develop evidence-based recommendations tailored specifically to women.
The research had no commercial funding. Dr. El Khoudary has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Practice-changing data at this year’s ASH meeting
Instead of flying out to San Diego in California and soaking up a bit of sunshine in between listening to new research presentations, hematologists from around the world will be glued to their computer screens next weekend, tuning into the 62nd American Society of Hematology annual meeting.
Like many other conferences this year, the ASH meeting will be virtual because of the continuing COVID-19 pandemic, although the dates remain the same: Dec. 5-8.
This is the premier hematology event of the year, and the largest hematology conference in the world, with around 3,500 abstracts presented this year, commented Aaron T. Gerds, MD, chair of ASH’s Committee on Communications.
Ruxolitinib in chronic GvHD
“One of the things that people come to ASH for is to hear about practice-changing clinical trials, and this year is no exception,” said ASH secretary Robert Brodsky, MD.
In a preview webinar, he highlighted four abstracts that offer opportunities to change practice and revamp the current standards of care.
One clinical trial that is “almost certainly a practice changer,” he said, is the REACH 3 study (abstract 77) of the JAK inhibitor ruxolitinib (Jakafi, Incyte) in patients with chronic graft-versus-host disease (GvHD) after a stem cell transplant.
“This has been really hard to treat in patients undergoing allogeneic bone marrow transplants,” said Brodsky. “Steroids are the first-line treatment, but after that, nothing else has shown any improvement, and even steroids don’t work that well.”
There is currently no approved second-line therapy for chronic forms of GvHD, he emphasized. The main endpoint of the trial was overall response rate, which was doubled with ruxolitinib compared to the best available therapy (50% vs 25%).
“This is the first successful phase 3 trial for chronic GvHD,” Brodsky commented.
Transplants for older patients with MDS
Transplant offers the only curative option for myelodysplastic syndromes (MDS), but typically this option is offered to younger patients because benefits for older adults have not been well-defined, Brodsky noted.
New data from a clinical trial conducted in patients with advanced MDS aged 50-75 years (abstract 75) offers the most definitive evidence to date that allogeneic hematopoietic cell transplantation (AHCT) can significantly improve outcomes for older adults.
It’s clear that transplant is the standard of care in younger patients, Brodsky commented, and although there is a trend of offering it to older patients, some are not getting referred and instead are being offered palliative care. “The thinking is that bone marrow transplant would be too toxic in this age group,” he said. “But what is very clear here is that, in an intent-to-treat analysis, there was a significant survival advantage – 48% versus 27% at 3 years for transplantation – and it was seen across all subgroups.”
Subcutaneous daratumumab
New data on a subcutaneous formulation of daratumumab (Darzalex, Janssen), which is usually given by intravenous infusion, will be presented from the APOLLO trial (abstract 412) in patients with relapsed/refractory multiple myeloma.
Patients who received subcutaneous daratumumab combined with pomalidomide and dexamethasone had a 37% reduction in disease progression or death compared to those who received pomalidomide and dexamethasone alone.
“From previous years we’ve learned that daratumumab has had a major impact on outcomes in multiple myeloma,” said Brodsky. “The nice thing about the subcutaneous formulation is that it can be administered quickly and in an outpatient setting, which is especially important in the COVID era.”
Negative data with tranexamic acid
The fourth abstract highlighted by Brodsky is a negative study, but its findings can help guide clinical practice, he said. The a-TREAT study (abstract 2) showed that, despite being routinely used in the clinical setting, tranexamic acid does not prevent bleeding when administered prophylactically to severely thrombocytopenic patients undergoing treatment for hematologic malignancies.
“They found absolutely no difference in bleeding or need for transfusion,” said Brodsky. “What they did find was more catheter-associated blood clots in the tranexamic acid group. This is a practice changer in that it probably should not be given prophylactically to patients with thrombocytopenia.”
‘Very exciting’ news about gene therapy
Brodsky also highlighted several late-breaking abstract that will be presented at the meeting.
In particular, the first data on a gene therapy for hemophilia B (abstract LBA-6) are “very, very exciting,” he said. The HOPE-B trial showed a 96% response rate among patients with hemophilia B who were treated with etranacogene dezaparvovec, an investigational gene therapy composed of an adeno-associated virus serotype 5 (AAV5) vector containing a codon-optimized Padua variant human factor IX.
Brodsky pointed out that this was a large trial with 54 patients, but importantly, it included patients with pre-existing anti-AAV5 neutralizing antibodies. “About 40% of patients have naturally occurring antibodies to AAV5, and they have been excluded from previous trials because it was thought they wouldn’t take the vector,” said Brodsky. “But only one patient didn’t get a response.”
Following a single dose of etranacogene dezaparvovec, Factor IX activity increased into the mild to normal range without the need for prophylactic immunosuppression. Treated patients were able to discontinue prophylaxis and bleeding was controlled in most of the cohort.
“This is a big advance and we are getting very close to the point where gene therapy is going to be standard of care for some forms of hemophilia,” said Brodsky. However, he added that “we will still need to see more patients and have longer follow-up.”
He added that, with time, the technology behind gene therapy will probably become less expensive and more accessible to more patients, which will help become a standard of care.
This is also the hope for the technology behind chimeric antigen receptor T-cell (CAR-T) therapy, he added. At present, this cellular therapy is manufactured individually for each patient and is very expensive, but work on “off-the-shelf” products is underway. This topic will be explored during the presidential symposium, entitled, “Universal Donor Solutions in Hematology.”
New data on one of the currently available CAR-T cell products will be presented at the meeting. The phase 2 ZUMA-5 trial showed that axicabtagene ciloleucel (Axi-Cel) may be a viable option for some patients with high-risk non-Hodgkin lymphoma who have not responded to standard treatments (abstract 700).
At a median follow-up of almost 18 months, 92% of participants achieved an objective response, and 78% achieved a complete response to the treatment. By 12 months, 72% were still in response, and at 17.5 months, 64% were still in response.
“We were very impressed with the magnitude of the responses, and also the durability,” said senior study author Caron Jacobson, MD, of the Dana-Farber Cancer Institute, Boston, in a press release. “I was also struck early on by how favorable the safety profile was compared to what we’ve been seeing in the fast-growing lymphomas, such as large B cell lymphoma.”
Race and bloods cancers
ASH president Stephanie Lee, MD, MPH, highlighted several abstracts on disparities that will be presented at the meeting. One of these, which is to be presented during the plenary session, is an analysis of patient survival in acute myeloid leukemia (AML) (abstract 6).
It found that “self-reported race was the best indicator of survival,” noted Lee.
Overall survival at 3 years was 41% in White patients versus 32% in Black patients, a difference that was highly significant, she noted.
Part of the study also evaluated patients who were all on the same chemotherapy protocol, “so there was no effect of different treatment since they were on therapy determined by the trial,” said Lee.
Black patients were less likely to have normal cytogenetics compared with White patients (38% vs 51%; P = .01) and had a lower frequency of prognostically favorable NPM1 mutations (25% vs 38%; P = .04), but higher frequencies of spliceosome gene mutations (24% vs 12%; P = .009). Therefore, the results showed race was an independent prognosticator of poor survival in AML, aside from established molecular markers.
A special scientific session on race will be held on Dec. 5, Lee noted. While other abstracts consider race from the patient side, this session will focus on the scientist’s side, she explained, and address questions such as: “What are the implications of diversity and racism? And how does that impact scientists who are from underrepresented minorities?”
COVID-19 and blood disorders
Lee also highlighted a study (abstract 215) that analyzed emerging data from the ASH Research Collaborative COVID-19 Registry for Hematology, which was developed to look at outcomes of COVID-19 infection in patients with underlying blood disorders.
An analysis of data from 250 patients at 74 sites around the world found that overall mortality was 28%. “This supports the emerging consensus that patients with hematologic malignancies experience significant morbidity and mortality from COVID-19 infection,” say the authors.
“We do need real-world data to see how SARS-CoV-2 is affecting our patients with hematologic diseases or those who don’t have a hematologic disease but who are then infected with the coronavirus and develop a hematologic problem like blood clots,” said Lee.
“More data will be coming in, but this is a good example of trying to harness real-world information to learn things until we have more controlled trials.”
‘Fireside chat’ with Fauci
COVID-19 will be on the agenda for a special session billed as a “fireside chat” with Anthony Fauci, MD, of the National Institute of Allergy and Infectious Diseases, National Institutes of Health.
“This will be kicking off our meeting on Saturday morning,” said Lee.
This article first appeared on Medscape.com.
Instead of flying out to San Diego in California and soaking up a bit of sunshine in between listening to new research presentations, hematologists from around the world will be glued to their computer screens next weekend, tuning into the 62nd American Society of Hematology annual meeting.
Like many other conferences this year, the ASH meeting will be virtual because of the continuing COVID-19 pandemic, although the dates remain the same: Dec. 5-8.
This is the premier hematology event of the year, and the largest hematology conference in the world, with around 3,500 abstracts presented this year, commented Aaron T. Gerds, MD, chair of ASH’s Committee on Communications.
Ruxolitinib in chronic GvHD
“One of the things that people come to ASH for is to hear about practice-changing clinical trials, and this year is no exception,” said ASH secretary Robert Brodsky, MD.
In a preview webinar, he highlighted four abstracts that offer opportunities to change practice and revamp the current standards of care.
One clinical trial that is “almost certainly a practice changer,” he said, is the REACH 3 study (abstract 77) of the JAK inhibitor ruxolitinib (Jakafi, Incyte) in patients with chronic graft-versus-host disease (GvHD) after a stem cell transplant.
“This has been really hard to treat in patients undergoing allogeneic bone marrow transplants,” said Brodsky. “Steroids are the first-line treatment, but after that, nothing else has shown any improvement, and even steroids don’t work that well.”
There is currently no approved second-line therapy for chronic forms of GvHD, he emphasized. The main endpoint of the trial was overall response rate, which was doubled with ruxolitinib compared to the best available therapy (50% vs 25%).
“This is the first successful phase 3 trial for chronic GvHD,” Brodsky commented.
Transplants for older patients with MDS
Transplant offers the only curative option for myelodysplastic syndromes (MDS), but typically this option is offered to younger patients because benefits for older adults have not been well-defined, Brodsky noted.
New data from a clinical trial conducted in patients with advanced MDS aged 50-75 years (abstract 75) offers the most definitive evidence to date that allogeneic hematopoietic cell transplantation (AHCT) can significantly improve outcomes for older adults.
It’s clear that transplant is the standard of care in younger patients, Brodsky commented, and although there is a trend of offering it to older patients, some are not getting referred and instead are being offered palliative care. “The thinking is that bone marrow transplant would be too toxic in this age group,” he said. “But what is very clear here is that, in an intent-to-treat analysis, there was a significant survival advantage – 48% versus 27% at 3 years for transplantation – and it was seen across all subgroups.”
Subcutaneous daratumumab
New data on a subcutaneous formulation of daratumumab (Darzalex, Janssen), which is usually given by intravenous infusion, will be presented from the APOLLO trial (abstract 412) in patients with relapsed/refractory multiple myeloma.
Patients who received subcutaneous daratumumab combined with pomalidomide and dexamethasone had a 37% reduction in disease progression or death compared to those who received pomalidomide and dexamethasone alone.
“From previous years we’ve learned that daratumumab has had a major impact on outcomes in multiple myeloma,” said Brodsky. “The nice thing about the subcutaneous formulation is that it can be administered quickly and in an outpatient setting, which is especially important in the COVID era.”
Negative data with tranexamic acid
The fourth abstract highlighted by Brodsky is a negative study, but its findings can help guide clinical practice, he said. The a-TREAT study (abstract 2) showed that, despite being routinely used in the clinical setting, tranexamic acid does not prevent bleeding when administered prophylactically to severely thrombocytopenic patients undergoing treatment for hematologic malignancies.
“They found absolutely no difference in bleeding or need for transfusion,” said Brodsky. “What they did find was more catheter-associated blood clots in the tranexamic acid group. This is a practice changer in that it probably should not be given prophylactically to patients with thrombocytopenia.”
‘Very exciting’ news about gene therapy
Brodsky also highlighted several late-breaking abstract that will be presented at the meeting.
In particular, the first data on a gene therapy for hemophilia B (abstract LBA-6) are “very, very exciting,” he said. The HOPE-B trial showed a 96% response rate among patients with hemophilia B who were treated with etranacogene dezaparvovec, an investigational gene therapy composed of an adeno-associated virus serotype 5 (AAV5) vector containing a codon-optimized Padua variant human factor IX.
Brodsky pointed out that this was a large trial with 54 patients, but importantly, it included patients with pre-existing anti-AAV5 neutralizing antibodies. “About 40% of patients have naturally occurring antibodies to AAV5, and they have been excluded from previous trials because it was thought they wouldn’t take the vector,” said Brodsky. “But only one patient didn’t get a response.”
Following a single dose of etranacogene dezaparvovec, Factor IX activity increased into the mild to normal range without the need for prophylactic immunosuppression. Treated patients were able to discontinue prophylaxis and bleeding was controlled in most of the cohort.
“This is a big advance and we are getting very close to the point where gene therapy is going to be standard of care for some forms of hemophilia,” said Brodsky. However, he added that “we will still need to see more patients and have longer follow-up.”
He added that, with time, the technology behind gene therapy will probably become less expensive and more accessible to more patients, which will help become a standard of care.
This is also the hope for the technology behind chimeric antigen receptor T-cell (CAR-T) therapy, he added. At present, this cellular therapy is manufactured individually for each patient and is very expensive, but work on “off-the-shelf” products is underway. This topic will be explored during the presidential symposium, entitled, “Universal Donor Solutions in Hematology.”
New data on one of the currently available CAR-T cell products will be presented at the meeting. The phase 2 ZUMA-5 trial showed that axicabtagene ciloleucel (Axi-Cel) may be a viable option for some patients with high-risk non-Hodgkin lymphoma who have not responded to standard treatments (abstract 700).
At a median follow-up of almost 18 months, 92% of participants achieved an objective response, and 78% achieved a complete response to the treatment. By 12 months, 72% were still in response, and at 17.5 months, 64% were still in response.
“We were very impressed with the magnitude of the responses, and also the durability,” said senior study author Caron Jacobson, MD, of the Dana-Farber Cancer Institute, Boston, in a press release. “I was also struck early on by how favorable the safety profile was compared to what we’ve been seeing in the fast-growing lymphomas, such as large B cell lymphoma.”
Race and bloods cancers
ASH president Stephanie Lee, MD, MPH, highlighted several abstracts on disparities that will be presented at the meeting. One of these, which is to be presented during the plenary session, is an analysis of patient survival in acute myeloid leukemia (AML) (abstract 6).
It found that “self-reported race was the best indicator of survival,” noted Lee.
Overall survival at 3 years was 41% in White patients versus 32% in Black patients, a difference that was highly significant, she noted.
Part of the study also evaluated patients who were all on the same chemotherapy protocol, “so there was no effect of different treatment since they were on therapy determined by the trial,” said Lee.
Black patients were less likely to have normal cytogenetics compared with White patients (38% vs 51%; P = .01) and had a lower frequency of prognostically favorable NPM1 mutations (25% vs 38%; P = .04), but higher frequencies of spliceosome gene mutations (24% vs 12%; P = .009). Therefore, the results showed race was an independent prognosticator of poor survival in AML, aside from established molecular markers.
A special scientific session on race will be held on Dec. 5, Lee noted. While other abstracts consider race from the patient side, this session will focus on the scientist’s side, she explained, and address questions such as: “What are the implications of diversity and racism? And how does that impact scientists who are from underrepresented minorities?”
COVID-19 and blood disorders
Lee also highlighted a study (abstract 215) that analyzed emerging data from the ASH Research Collaborative COVID-19 Registry for Hematology, which was developed to look at outcomes of COVID-19 infection in patients with underlying blood disorders.
An analysis of data from 250 patients at 74 sites around the world found that overall mortality was 28%. “This supports the emerging consensus that patients with hematologic malignancies experience significant morbidity and mortality from COVID-19 infection,” say the authors.
“We do need real-world data to see how SARS-CoV-2 is affecting our patients with hematologic diseases or those who don’t have a hematologic disease but who are then infected with the coronavirus and develop a hematologic problem like blood clots,” said Lee.
“More data will be coming in, but this is a good example of trying to harness real-world information to learn things until we have more controlled trials.”
‘Fireside chat’ with Fauci
COVID-19 will be on the agenda for a special session billed as a “fireside chat” with Anthony Fauci, MD, of the National Institute of Allergy and Infectious Diseases, National Institutes of Health.
“This will be kicking off our meeting on Saturday morning,” said Lee.
This article first appeared on Medscape.com.
Instead of flying out to San Diego in California and soaking up a bit of sunshine in between listening to new research presentations, hematologists from around the world will be glued to their computer screens next weekend, tuning into the 62nd American Society of Hematology annual meeting.
Like many other conferences this year, the ASH meeting will be virtual because of the continuing COVID-19 pandemic, although the dates remain the same: Dec. 5-8.
This is the premier hematology event of the year, and the largest hematology conference in the world, with around 3,500 abstracts presented this year, commented Aaron T. Gerds, MD, chair of ASH’s Committee on Communications.
Ruxolitinib in chronic GvHD
“One of the things that people come to ASH for is to hear about practice-changing clinical trials, and this year is no exception,” said ASH secretary Robert Brodsky, MD.
In a preview webinar, he highlighted four abstracts that offer opportunities to change practice and revamp the current standards of care.
One clinical trial that is “almost certainly a practice changer,” he said, is the REACH 3 study (abstract 77) of the JAK inhibitor ruxolitinib (Jakafi, Incyte) in patients with chronic graft-versus-host disease (GvHD) after a stem cell transplant.
“This has been really hard to treat in patients undergoing allogeneic bone marrow transplants,” said Brodsky. “Steroids are the first-line treatment, but after that, nothing else has shown any improvement, and even steroids don’t work that well.”
There is currently no approved second-line therapy for chronic forms of GvHD, he emphasized. The main endpoint of the trial was overall response rate, which was doubled with ruxolitinib compared to the best available therapy (50% vs 25%).
“This is the first successful phase 3 trial for chronic GvHD,” Brodsky commented.
Transplants for older patients with MDS
Transplant offers the only curative option for myelodysplastic syndromes (MDS), but typically this option is offered to younger patients because benefits for older adults have not been well-defined, Brodsky noted.
New data from a clinical trial conducted in patients with advanced MDS aged 50-75 years (abstract 75) offers the most definitive evidence to date that allogeneic hematopoietic cell transplantation (AHCT) can significantly improve outcomes for older adults.
It’s clear that transplant is the standard of care in younger patients, Brodsky commented, and although there is a trend of offering it to older patients, some are not getting referred and instead are being offered palliative care. “The thinking is that bone marrow transplant would be too toxic in this age group,” he said. “But what is very clear here is that, in an intent-to-treat analysis, there was a significant survival advantage – 48% versus 27% at 3 years for transplantation – and it was seen across all subgroups.”
Subcutaneous daratumumab
New data on a subcutaneous formulation of daratumumab (Darzalex, Janssen), which is usually given by intravenous infusion, will be presented from the APOLLO trial (abstract 412) in patients with relapsed/refractory multiple myeloma.
Patients who received subcutaneous daratumumab combined with pomalidomide and dexamethasone had a 37% reduction in disease progression or death compared to those who received pomalidomide and dexamethasone alone.
“From previous years we’ve learned that daratumumab has had a major impact on outcomes in multiple myeloma,” said Brodsky. “The nice thing about the subcutaneous formulation is that it can be administered quickly and in an outpatient setting, which is especially important in the COVID era.”
Negative data with tranexamic acid
The fourth abstract highlighted by Brodsky is a negative study, but its findings can help guide clinical practice, he said. The a-TREAT study (abstract 2) showed that, despite being routinely used in the clinical setting, tranexamic acid does not prevent bleeding when administered prophylactically to severely thrombocytopenic patients undergoing treatment for hematologic malignancies.
“They found absolutely no difference in bleeding or need for transfusion,” said Brodsky. “What they did find was more catheter-associated blood clots in the tranexamic acid group. This is a practice changer in that it probably should not be given prophylactically to patients with thrombocytopenia.”
‘Very exciting’ news about gene therapy
Brodsky also highlighted several late-breaking abstract that will be presented at the meeting.
In particular, the first data on a gene therapy for hemophilia B (abstract LBA-6) are “very, very exciting,” he said. The HOPE-B trial showed a 96% response rate among patients with hemophilia B who were treated with etranacogene dezaparvovec, an investigational gene therapy composed of an adeno-associated virus serotype 5 (AAV5) vector containing a codon-optimized Padua variant human factor IX.
Brodsky pointed out that this was a large trial with 54 patients, but importantly, it included patients with pre-existing anti-AAV5 neutralizing antibodies. “About 40% of patients have naturally occurring antibodies to AAV5, and they have been excluded from previous trials because it was thought they wouldn’t take the vector,” said Brodsky. “But only one patient didn’t get a response.”
Following a single dose of etranacogene dezaparvovec, Factor IX activity increased into the mild to normal range without the need for prophylactic immunosuppression. Treated patients were able to discontinue prophylaxis and bleeding was controlled in most of the cohort.
“This is a big advance and we are getting very close to the point where gene therapy is going to be standard of care for some forms of hemophilia,” said Brodsky. However, he added that “we will still need to see more patients and have longer follow-up.”
He added that, with time, the technology behind gene therapy will probably become less expensive and more accessible to more patients, which will help become a standard of care.
This is also the hope for the technology behind chimeric antigen receptor T-cell (CAR-T) therapy, he added. At present, this cellular therapy is manufactured individually for each patient and is very expensive, but work on “off-the-shelf” products is underway. This topic will be explored during the presidential symposium, entitled, “Universal Donor Solutions in Hematology.”
New data on one of the currently available CAR-T cell products will be presented at the meeting. The phase 2 ZUMA-5 trial showed that axicabtagene ciloleucel (Axi-Cel) may be a viable option for some patients with high-risk non-Hodgkin lymphoma who have not responded to standard treatments (abstract 700).
At a median follow-up of almost 18 months, 92% of participants achieved an objective response, and 78% achieved a complete response to the treatment. By 12 months, 72% were still in response, and at 17.5 months, 64% were still in response.
“We were very impressed with the magnitude of the responses, and also the durability,” said senior study author Caron Jacobson, MD, of the Dana-Farber Cancer Institute, Boston, in a press release. “I was also struck early on by how favorable the safety profile was compared to what we’ve been seeing in the fast-growing lymphomas, such as large B cell lymphoma.”
Race and bloods cancers
ASH president Stephanie Lee, MD, MPH, highlighted several abstracts on disparities that will be presented at the meeting. One of these, which is to be presented during the plenary session, is an analysis of patient survival in acute myeloid leukemia (AML) (abstract 6).
It found that “self-reported race was the best indicator of survival,” noted Lee.
Overall survival at 3 years was 41% in White patients versus 32% in Black patients, a difference that was highly significant, she noted.
Part of the study also evaluated patients who were all on the same chemotherapy protocol, “so there was no effect of different treatment since they were on therapy determined by the trial,” said Lee.
Black patients were less likely to have normal cytogenetics compared with White patients (38% vs 51%; P = .01) and had a lower frequency of prognostically favorable NPM1 mutations (25% vs 38%; P = .04), but higher frequencies of spliceosome gene mutations (24% vs 12%; P = .009). Therefore, the results showed race was an independent prognosticator of poor survival in AML, aside from established molecular markers.
A special scientific session on race will be held on Dec. 5, Lee noted. While other abstracts consider race from the patient side, this session will focus on the scientist’s side, she explained, and address questions such as: “What are the implications of diversity and racism? And how does that impact scientists who are from underrepresented minorities?”
COVID-19 and blood disorders
Lee also highlighted a study (abstract 215) that analyzed emerging data from the ASH Research Collaborative COVID-19 Registry for Hematology, which was developed to look at outcomes of COVID-19 infection in patients with underlying blood disorders.
An analysis of data from 250 patients at 74 sites around the world found that overall mortality was 28%. “This supports the emerging consensus that patients with hematologic malignancies experience significant morbidity and mortality from COVID-19 infection,” say the authors.
“We do need real-world data to see how SARS-CoV-2 is affecting our patients with hematologic diseases or those who don’t have a hematologic disease but who are then infected with the coronavirus and develop a hematologic problem like blood clots,” said Lee.
“More data will be coming in, but this is a good example of trying to harness real-world information to learn things until we have more controlled trials.”
‘Fireside chat’ with Fauci
COVID-19 will be on the agenda for a special session billed as a “fireside chat” with Anthony Fauci, MD, of the National Institute of Allergy and Infectious Diseases, National Institutes of Health.
“This will be kicking off our meeting on Saturday morning,” said Lee.
This article first appeared on Medscape.com.
CMS launches hospital-at-home program to free up hospital capacity
As an increasing number of health systems implement “hospital-at-home” (HaH) programs to increase their traditional hospital capacity, the Centers for Medicare & Medicaid Services has given the movement a boost by changing its regulations to allow acute care to be provided in a patient’s home under certain conditions.
The CMS announced Nov. 25 that it was launching its Acute Hospital Care at Home program “to increase the capacity of the American health care system” during the COVID-19 pandemic.
At the same time, the agency announced it was giving more flexibility to ambulatory surgery centers (ASCs) to provide hospital-level care.
The CMS said its new HaH program is an expansion of the Hospitals Without Walls initiative that was unveiled last March. Hospitals Without Walls is a set of “temporary new rules” that provide flexibility for hospitals to provide acute care outside of inpatient settings. Under those rules, hospitals are able to transfer patients to outside facilities, such as ASCs, inpatient rehabilitation hospitals, hotels, and dormitories, while still receiving Medicare hospital payments.
Under CMS’ new Acute Hospital Care at Home, which is not described as temporary, patients can be transferred from emergency departments or inpatient wards to hospital-level care at home. The CMS said the HaH program is designed for people with conditions such as the acute phases of asthma, heart failure, pneumonia, and chronic obstructive pulmonary disease. Altogether, the agency said, more than 60 acute conditions can be treated safely at home.
However, the agency didn’t say that facilities can’t admit COVID-19 patients to the hospital at home. Rami Karjian, MBA, cofounder and CEO of Medically Home, a firm that supplies health systems with technical services and software for HaH programs, said in an interview that several Medically Home clients plan to treat both COVID-19 and non-COVID-19 patients at home when they begin to participate in the CMS program in the near future.
The CMS said it consulted extensively with academic and private industry leaders in building its HaH program. Before rolling out the initiative, the agency noted, it conducted successful pilot programs in leading hospitals and health systems. The results of some of these pilots have been reported in academic journals.
Participating hospitals will be required to have specified screening protocols in place before beginning acute care at home, the CMS announced. An in-person physician evaluation will be required before starting care at home. A nurse will evaluate each patient once daily in person or remotely, and either nurses or paramedics will visit the patient in person twice a day.
In contrast, Medicare regulations require nursing staff to be available around the clock in traditional hospitals. So the CMS has to grant waivers to hospitals for HaH programs.
While not going into detail on the telemonitoring capabilities that will be required in the acute hospital care at home, the release said, “Today’s announcement builds upon the critical work by CMS to expand telehealth coverage to keep beneficiaries safe and prevent the spread of COVID-19.”
More flexibility for ASCs
The agency is also giving ASCs the flexibility to provide 24-hour nursing services only when one or more patients are receiving care on site. This flexibility will be available to any of the 5,700 ASCs that wish to participate, and will be immediately effective for the 85 ASCs currently participating in the Hospital Without Walls initiative, the CMS said.
The new ASC regulations, the CMS said, are aimed at allowing communities “to maintain surgical capacity and other life-saving non-COVID-19 [care], like cancer surgeries.” Patients who need such procedures will be able to receive them in ASCs without being exposed to known COVID-19 cases.
Similarly, the CMS said patients and families not diagnosed with COVID-19 may prefer to receive acute care at home if local hospitals are full of COVID-19 patients. In addition, the CMS said it anticipates patients may value the ability to be treated at home without the visitation restrictions of hospitals.
Early HaH participants
Six health systems with extensive experience in providing acute hospital care at home have been approved for the new HaH waivers from Medicare rules. They include Brigham and Women’s Hospital (Massachusetts); Huntsman Cancer Institute (Utah); Massachusetts General Hospital (Massachusetts); Mount Sinai Health System (New York City); Presbyterian Healthcare Services (New Mexico); and UnityPoint Health (Iowa).
The CMS said that it’s in discussions with other health care systems and expects new applications to be submitted soon.
To support these efforts, the CMS has launched an online portal to streamline the waiver request process. The agency said it will closely monitor the program to safeguard beneficiaries and will require participating hospitals to report quality and safety data on a regular basis.
Support from hospitals
The first health systems participating in the CMS HaH appear to be supportive of the program, with some hospital leaders submitting comments to the CMS about their view of the initiative.
“The CMS has taken an extraordinary step today, facilitating the rapid expansion of Hospitalization at Home, an innovative care model with proven results,” said Kenneth L. Davis, MD, president and CEO of the Mount Sinai Health System in New York City. “This important and timely move will enable hospitals across the country to use effective tools to safely care for patients during this pandemic.”
David Levine, MD, assistant professor of medicine and medical director of strategy and innovation for Brigham Health Home Hospital in Boston, was similarly laudatory: “Our research at Brigham Health Home has shown that we can deliver hospital-level care in our patients’ homes with lower readmission rates, more physical mobility, and a positive patient experience,” he said. “During these challenging times, a focus on the home is critical. We are so encouraged that CMS is taking this important step, which will allow hospitals across the country to increase their capacity while delivering the care all patients deserve.”
Scaling up quickly
If other hospitals and health systems recognize the value of HaH, how long might it take them to develop and implement these programs in the midst of a pandemic?
Atrium Health, a large health system in the Southeast, ramped up a hospital-at-home initiative last spring for its 10 hospitals in the Charlotte, N.C., area, in just 2 weeks. However, it had been working on the project for some time before the pandemic struck. Focusing mostly on COVID-19 patients, the initiative reduced the COVID-19 patient load by 20%-25% in Atrium’s hospitals.
Medically Home, the HaH infrastructure company, said in a news release that it “enables health systems to establish new hospital-at-home services in as little as 30 days.” Medically Home has partnered in this venture with Huron Consulting Group, which has about 200 HaH-trained consultants, and Cardinal Health, a large global medical supplies distributor.
Mr. Karjian said in an interview that he expects private insurers to follow CMS’ example, as they often do. “We think this decision will cause not only CMS but private insurers to cover hospital at home after the pandemic, if it becomes the standard of care, because patients have better outcomes when treated at home,” he said.
Asked for his view on why the CMS specified that patients could be admitted to an HaH only from emergency departments or inpatient settings, Mr. Karjian said that the CMS wants to make sure that patients have access to brick-and-mortar hospital care if that’s what they need. Also, he noted, this model is new to most hospitals, so the CMS wants to make sure it starts “with all the safety guardrails” in place.
Overall, Mr. Karjian said, “This is an exciting development for patients across the country. What CMS has done is terrific in terms of letting patients get the care they want, where they want it, and get the benefit of better outcomes while the nation is going through this capacity crunch for hospital beds.”
A version of this article originally appeared on Medscape.com.
As an increasing number of health systems implement “hospital-at-home” (HaH) programs to increase their traditional hospital capacity, the Centers for Medicare & Medicaid Services has given the movement a boost by changing its regulations to allow acute care to be provided in a patient’s home under certain conditions.
The CMS announced Nov. 25 that it was launching its Acute Hospital Care at Home program “to increase the capacity of the American health care system” during the COVID-19 pandemic.
At the same time, the agency announced it was giving more flexibility to ambulatory surgery centers (ASCs) to provide hospital-level care.
The CMS said its new HaH program is an expansion of the Hospitals Without Walls initiative that was unveiled last March. Hospitals Without Walls is a set of “temporary new rules” that provide flexibility for hospitals to provide acute care outside of inpatient settings. Under those rules, hospitals are able to transfer patients to outside facilities, such as ASCs, inpatient rehabilitation hospitals, hotels, and dormitories, while still receiving Medicare hospital payments.
Under CMS’ new Acute Hospital Care at Home, which is not described as temporary, patients can be transferred from emergency departments or inpatient wards to hospital-level care at home. The CMS said the HaH program is designed for people with conditions such as the acute phases of asthma, heart failure, pneumonia, and chronic obstructive pulmonary disease. Altogether, the agency said, more than 60 acute conditions can be treated safely at home.
However, the agency didn’t say that facilities can’t admit COVID-19 patients to the hospital at home. Rami Karjian, MBA, cofounder and CEO of Medically Home, a firm that supplies health systems with technical services and software for HaH programs, said in an interview that several Medically Home clients plan to treat both COVID-19 and non-COVID-19 patients at home when they begin to participate in the CMS program in the near future.
The CMS said it consulted extensively with academic and private industry leaders in building its HaH program. Before rolling out the initiative, the agency noted, it conducted successful pilot programs in leading hospitals and health systems. The results of some of these pilots have been reported in academic journals.
Participating hospitals will be required to have specified screening protocols in place before beginning acute care at home, the CMS announced. An in-person physician evaluation will be required before starting care at home. A nurse will evaluate each patient once daily in person or remotely, and either nurses or paramedics will visit the patient in person twice a day.
In contrast, Medicare regulations require nursing staff to be available around the clock in traditional hospitals. So the CMS has to grant waivers to hospitals for HaH programs.
While not going into detail on the telemonitoring capabilities that will be required in the acute hospital care at home, the release said, “Today’s announcement builds upon the critical work by CMS to expand telehealth coverage to keep beneficiaries safe and prevent the spread of COVID-19.”
More flexibility for ASCs
The agency is also giving ASCs the flexibility to provide 24-hour nursing services only when one or more patients are receiving care on site. This flexibility will be available to any of the 5,700 ASCs that wish to participate, and will be immediately effective for the 85 ASCs currently participating in the Hospital Without Walls initiative, the CMS said.
The new ASC regulations, the CMS said, are aimed at allowing communities “to maintain surgical capacity and other life-saving non-COVID-19 [care], like cancer surgeries.” Patients who need such procedures will be able to receive them in ASCs without being exposed to known COVID-19 cases.
Similarly, the CMS said patients and families not diagnosed with COVID-19 may prefer to receive acute care at home if local hospitals are full of COVID-19 patients. In addition, the CMS said it anticipates patients may value the ability to be treated at home without the visitation restrictions of hospitals.
Early HaH participants
Six health systems with extensive experience in providing acute hospital care at home have been approved for the new HaH waivers from Medicare rules. They include Brigham and Women’s Hospital (Massachusetts); Huntsman Cancer Institute (Utah); Massachusetts General Hospital (Massachusetts); Mount Sinai Health System (New York City); Presbyterian Healthcare Services (New Mexico); and UnityPoint Health (Iowa).
The CMS said that it’s in discussions with other health care systems and expects new applications to be submitted soon.
To support these efforts, the CMS has launched an online portal to streamline the waiver request process. The agency said it will closely monitor the program to safeguard beneficiaries and will require participating hospitals to report quality and safety data on a regular basis.
Support from hospitals
The first health systems participating in the CMS HaH appear to be supportive of the program, with some hospital leaders submitting comments to the CMS about their view of the initiative.
“The CMS has taken an extraordinary step today, facilitating the rapid expansion of Hospitalization at Home, an innovative care model with proven results,” said Kenneth L. Davis, MD, president and CEO of the Mount Sinai Health System in New York City. “This important and timely move will enable hospitals across the country to use effective tools to safely care for patients during this pandemic.”
David Levine, MD, assistant professor of medicine and medical director of strategy and innovation for Brigham Health Home Hospital in Boston, was similarly laudatory: “Our research at Brigham Health Home has shown that we can deliver hospital-level care in our patients’ homes with lower readmission rates, more physical mobility, and a positive patient experience,” he said. “During these challenging times, a focus on the home is critical. We are so encouraged that CMS is taking this important step, which will allow hospitals across the country to increase their capacity while delivering the care all patients deserve.”
Scaling up quickly
If other hospitals and health systems recognize the value of HaH, how long might it take them to develop and implement these programs in the midst of a pandemic?
Atrium Health, a large health system in the Southeast, ramped up a hospital-at-home initiative last spring for its 10 hospitals in the Charlotte, N.C., area, in just 2 weeks. However, it had been working on the project for some time before the pandemic struck. Focusing mostly on COVID-19 patients, the initiative reduced the COVID-19 patient load by 20%-25% in Atrium’s hospitals.
Medically Home, the HaH infrastructure company, said in a news release that it “enables health systems to establish new hospital-at-home services in as little as 30 days.” Medically Home has partnered in this venture with Huron Consulting Group, which has about 200 HaH-trained consultants, and Cardinal Health, a large global medical supplies distributor.
Mr. Karjian said in an interview that he expects private insurers to follow CMS’ example, as they often do. “We think this decision will cause not only CMS but private insurers to cover hospital at home after the pandemic, if it becomes the standard of care, because patients have better outcomes when treated at home,” he said.
Asked for his view on why the CMS specified that patients could be admitted to an HaH only from emergency departments or inpatient settings, Mr. Karjian said that the CMS wants to make sure that patients have access to brick-and-mortar hospital care if that’s what they need. Also, he noted, this model is new to most hospitals, so the CMS wants to make sure it starts “with all the safety guardrails” in place.
Overall, Mr. Karjian said, “This is an exciting development for patients across the country. What CMS has done is terrific in terms of letting patients get the care they want, where they want it, and get the benefit of better outcomes while the nation is going through this capacity crunch for hospital beds.”
A version of this article originally appeared on Medscape.com.
As an increasing number of health systems implement “hospital-at-home” (HaH) programs to increase their traditional hospital capacity, the Centers for Medicare & Medicaid Services has given the movement a boost by changing its regulations to allow acute care to be provided in a patient’s home under certain conditions.
The CMS announced Nov. 25 that it was launching its Acute Hospital Care at Home program “to increase the capacity of the American health care system” during the COVID-19 pandemic.
At the same time, the agency announced it was giving more flexibility to ambulatory surgery centers (ASCs) to provide hospital-level care.
The CMS said its new HaH program is an expansion of the Hospitals Without Walls initiative that was unveiled last March. Hospitals Without Walls is a set of “temporary new rules” that provide flexibility for hospitals to provide acute care outside of inpatient settings. Under those rules, hospitals are able to transfer patients to outside facilities, such as ASCs, inpatient rehabilitation hospitals, hotels, and dormitories, while still receiving Medicare hospital payments.
Under CMS’ new Acute Hospital Care at Home, which is not described as temporary, patients can be transferred from emergency departments or inpatient wards to hospital-level care at home. The CMS said the HaH program is designed for people with conditions such as the acute phases of asthma, heart failure, pneumonia, and chronic obstructive pulmonary disease. Altogether, the agency said, more than 60 acute conditions can be treated safely at home.
However, the agency didn’t say that facilities can’t admit COVID-19 patients to the hospital at home. Rami Karjian, MBA, cofounder and CEO of Medically Home, a firm that supplies health systems with technical services and software for HaH programs, said in an interview that several Medically Home clients plan to treat both COVID-19 and non-COVID-19 patients at home when they begin to participate in the CMS program in the near future.
The CMS said it consulted extensively with academic and private industry leaders in building its HaH program. Before rolling out the initiative, the agency noted, it conducted successful pilot programs in leading hospitals and health systems. The results of some of these pilots have been reported in academic journals.
Participating hospitals will be required to have specified screening protocols in place before beginning acute care at home, the CMS announced. An in-person physician evaluation will be required before starting care at home. A nurse will evaluate each patient once daily in person or remotely, and either nurses or paramedics will visit the patient in person twice a day.
In contrast, Medicare regulations require nursing staff to be available around the clock in traditional hospitals. So the CMS has to grant waivers to hospitals for HaH programs.
While not going into detail on the telemonitoring capabilities that will be required in the acute hospital care at home, the release said, “Today’s announcement builds upon the critical work by CMS to expand telehealth coverage to keep beneficiaries safe and prevent the spread of COVID-19.”
More flexibility for ASCs
The agency is also giving ASCs the flexibility to provide 24-hour nursing services only when one or more patients are receiving care on site. This flexibility will be available to any of the 5,700 ASCs that wish to participate, and will be immediately effective for the 85 ASCs currently participating in the Hospital Without Walls initiative, the CMS said.
The new ASC regulations, the CMS said, are aimed at allowing communities “to maintain surgical capacity and other life-saving non-COVID-19 [care], like cancer surgeries.” Patients who need such procedures will be able to receive them in ASCs without being exposed to known COVID-19 cases.
Similarly, the CMS said patients and families not diagnosed with COVID-19 may prefer to receive acute care at home if local hospitals are full of COVID-19 patients. In addition, the CMS said it anticipates patients may value the ability to be treated at home without the visitation restrictions of hospitals.
Early HaH participants
Six health systems with extensive experience in providing acute hospital care at home have been approved for the new HaH waivers from Medicare rules. They include Brigham and Women’s Hospital (Massachusetts); Huntsman Cancer Institute (Utah); Massachusetts General Hospital (Massachusetts); Mount Sinai Health System (New York City); Presbyterian Healthcare Services (New Mexico); and UnityPoint Health (Iowa).
The CMS said that it’s in discussions with other health care systems and expects new applications to be submitted soon.
To support these efforts, the CMS has launched an online portal to streamline the waiver request process. The agency said it will closely monitor the program to safeguard beneficiaries and will require participating hospitals to report quality and safety data on a regular basis.
Support from hospitals
The first health systems participating in the CMS HaH appear to be supportive of the program, with some hospital leaders submitting comments to the CMS about their view of the initiative.
“The CMS has taken an extraordinary step today, facilitating the rapid expansion of Hospitalization at Home, an innovative care model with proven results,” said Kenneth L. Davis, MD, president and CEO of the Mount Sinai Health System in New York City. “This important and timely move will enable hospitals across the country to use effective tools to safely care for patients during this pandemic.”
David Levine, MD, assistant professor of medicine and medical director of strategy and innovation for Brigham Health Home Hospital in Boston, was similarly laudatory: “Our research at Brigham Health Home has shown that we can deliver hospital-level care in our patients’ homes with lower readmission rates, more physical mobility, and a positive patient experience,” he said. “During these challenging times, a focus on the home is critical. We are so encouraged that CMS is taking this important step, which will allow hospitals across the country to increase their capacity while delivering the care all patients deserve.”
Scaling up quickly
If other hospitals and health systems recognize the value of HaH, how long might it take them to develop and implement these programs in the midst of a pandemic?
Atrium Health, a large health system in the Southeast, ramped up a hospital-at-home initiative last spring for its 10 hospitals in the Charlotte, N.C., area, in just 2 weeks. However, it had been working on the project for some time before the pandemic struck. Focusing mostly on COVID-19 patients, the initiative reduced the COVID-19 patient load by 20%-25% in Atrium’s hospitals.
Medically Home, the HaH infrastructure company, said in a news release that it “enables health systems to establish new hospital-at-home services in as little as 30 days.” Medically Home has partnered in this venture with Huron Consulting Group, which has about 200 HaH-trained consultants, and Cardinal Health, a large global medical supplies distributor.
Mr. Karjian said in an interview that he expects private insurers to follow CMS’ example, as they often do. “We think this decision will cause not only CMS but private insurers to cover hospital at home after the pandemic, if it becomes the standard of care, because patients have better outcomes when treated at home,” he said.
Asked for his view on why the CMS specified that patients could be admitted to an HaH only from emergency departments or inpatient settings, Mr. Karjian said that the CMS wants to make sure that patients have access to brick-and-mortar hospital care if that’s what they need. Also, he noted, this model is new to most hospitals, so the CMS wants to make sure it starts “with all the safety guardrails” in place.
Overall, Mr. Karjian said, “This is an exciting development for patients across the country. What CMS has done is terrific in terms of letting patients get the care they want, where they want it, and get the benefit of better outcomes while the nation is going through this capacity crunch for hospital beds.”
A version of this article originally appeared on Medscape.com.
Scientific power will save us
COVID-19 numbers again are increasing dramatically. Community infection rates have nearly doubled, and hospitals and health care workers are stretched beyond their limits. It is difficult not to feel anger about how poorly this pandemic was managed (mismanaged) by so many officials in charge and by a large segment of our population who still refuse protective actions to limit spread. While politics and ideology continue to cost American lives, scientific firepower will emerge as our saving grace.
My editorial board and I are entering our final year at the helm of GI & Hepatology News. AGA issued a search for the next Editor in Chief (EIC), who will take over October 2021. I urge anyone interested to apply (https://gastro.org/news/prestigious-aga-publications-seek-new-editors-in-chief/). As EIC, you will choose the next editorial board and forge professional friendships that are gratifying. You will assume responsibility for the content, where you must balance your own views with those of both the AGA and our readership.
As EIC, each month I am given space for 300 words to communicate interesting ideas and opinions. The AGA gives the newspaper great editorial freedom, and I hope we have supported AGA’s mission and values when we publish its official newspaper. I always have next month’s editorial in mind, and I look for useful phrases, quotes, ideas, and opinions. If you are interested in becoming EIC, please email ginews@gastro.org for more information.
I would be remiss not to acknowledge the contribution that Lora McGlade has made to GI & Hepatology News. She has been my partner, as the Frontline Medical Communications Editor in charge of GI & Hepatology News. Next month, she will move on to assume a new role. I cannot thank her enough for helping make this newspaper work. As the months go on, I will highlight the contributions of others from the AGA, our Board, and Frontline.
Please stay safe and do not let your guard down. COVID-19 is merciless and relentless. “If you think research is expensive, try disease.” – Mary Lasker.
John I. Allen, MD, MBA, AGA
Editor in Chief
COVID-19 numbers again are increasing dramatically. Community infection rates have nearly doubled, and hospitals and health care workers are stretched beyond their limits. It is difficult not to feel anger about how poorly this pandemic was managed (mismanaged) by so many officials in charge and by a large segment of our population who still refuse protective actions to limit spread. While politics and ideology continue to cost American lives, scientific firepower will emerge as our saving grace.
My editorial board and I are entering our final year at the helm of GI & Hepatology News. AGA issued a search for the next Editor in Chief (EIC), who will take over October 2021. I urge anyone interested to apply (https://gastro.org/news/prestigious-aga-publications-seek-new-editors-in-chief/). As EIC, you will choose the next editorial board and forge professional friendships that are gratifying. You will assume responsibility for the content, where you must balance your own views with those of both the AGA and our readership.
As EIC, each month I am given space for 300 words to communicate interesting ideas and opinions. The AGA gives the newspaper great editorial freedom, and I hope we have supported AGA’s mission and values when we publish its official newspaper. I always have next month’s editorial in mind, and I look for useful phrases, quotes, ideas, and opinions. If you are interested in becoming EIC, please email ginews@gastro.org for more information.
I would be remiss not to acknowledge the contribution that Lora McGlade has made to GI & Hepatology News. She has been my partner, as the Frontline Medical Communications Editor in charge of GI & Hepatology News. Next month, she will move on to assume a new role. I cannot thank her enough for helping make this newspaper work. As the months go on, I will highlight the contributions of others from the AGA, our Board, and Frontline.
Please stay safe and do not let your guard down. COVID-19 is merciless and relentless. “If you think research is expensive, try disease.” – Mary Lasker.
John I. Allen, MD, MBA, AGA
Editor in Chief
COVID-19 numbers again are increasing dramatically. Community infection rates have nearly doubled, and hospitals and health care workers are stretched beyond their limits. It is difficult not to feel anger about how poorly this pandemic was managed (mismanaged) by so many officials in charge and by a large segment of our population who still refuse protective actions to limit spread. While politics and ideology continue to cost American lives, scientific firepower will emerge as our saving grace.
My editorial board and I are entering our final year at the helm of GI & Hepatology News. AGA issued a search for the next Editor in Chief (EIC), who will take over October 2021. I urge anyone interested to apply (https://gastro.org/news/prestigious-aga-publications-seek-new-editors-in-chief/). As EIC, you will choose the next editorial board and forge professional friendships that are gratifying. You will assume responsibility for the content, where you must balance your own views with those of both the AGA and our readership.
As EIC, each month I am given space for 300 words to communicate interesting ideas and opinions. The AGA gives the newspaper great editorial freedom, and I hope we have supported AGA’s mission and values when we publish its official newspaper. I always have next month’s editorial in mind, and I look for useful phrases, quotes, ideas, and opinions. If you are interested in becoming EIC, please email ginews@gastro.org for more information.
I would be remiss not to acknowledge the contribution that Lora McGlade has made to GI & Hepatology News. She has been my partner, as the Frontline Medical Communications Editor in charge of GI & Hepatology News. Next month, she will move on to assume a new role. I cannot thank her enough for helping make this newspaper work. As the months go on, I will highlight the contributions of others from the AGA, our Board, and Frontline.
Please stay safe and do not let your guard down. COVID-19 is merciless and relentless. “If you think research is expensive, try disease.” – Mary Lasker.
John I. Allen, MD, MBA, AGA
Editor in Chief
How much longer?
SHM has changed direction as needed during the pandemic
“How much longer?” As a kid, I can remember the long holiday car ride with my parents from my home in Annapolis, Md., to Upstate New York where my grandparents lived. At the time, the ride felt like an eternity: endless miles of frozen landscape, limited food, and a brother who constantly crossed over the invisible line that was my side of the car.
We made our parents crazy asking, “how much longer?” every few minutes. This was the late 1970s, with no GPS or Google Maps to give you arrival times to the minute, traffic warnings, or reroutes when the inevitable delays occurred. We just plowed ahead, and my parents’ answer was always something vague like, “in a few hours” or “we’re about halfway through.” They did not know when we’d arrive with certainty either.
We at SHM have that same feeling about the pandemic. How much longer? No one can tell us when the COVID-19 threat will abate. The experts’ answers are understandably vague, and the tools for forecasting are non-existent. Months? That is the best we know for now.
At SHM, we believe we will make it through this journey by adapting to roadblocks, providing tools for success to our professional community, and identifying opportunities for us to connect with each other, even if that means virtually.
Like the rest of the planet, the spring of 2020 hit SHM with a shock. Hospital Medicine 2020 (HM20) in San Diego was shaping up to be the largest Annual Conference SHM ever had, the Pediatric Hospital Medicine 2020 (PHM20) conference was well planned and expected to be a huge success, regional SHM chapters were meeting (and growing), and membership was thriving. I was transitioning out of my roles at Johns Hopkins and looking forward to my new role as CEO of SHM. All in all, March 2020 began with a fantastic outlook.
Wow, what a difference a few weeks made. We watched as the pandemic spread across regions of the country, concerned for the wellbeing of our patients and our hospitalists. We saw how our members were at the forefront of patient care during this crisis and understood that SHM had to adapt rapidly to meet their needs in real time.
By May, SHM had canceled HM20, Chapter activity was halted, PHM20 was on its way to being canceled, SHM committee work was put on hold, and I was spending my last few months at Hopkins as the chief medical officer at the Baltimore Convention Center Field Hospital (which we got up and running in less than a month)! Whew.
But just like my dad could pivot our 1970s Chevy station wagon around a traffic jam in a flash, so too did SHM leadership start navigating around the COVID-19 landscape. As soon as HM20 was canceled, SHM immediately began planning for a virtual offering in August. We had hoped to attract at least 100 attendees and we were thrilled to have more than 1,000! PHM20 was switched from an in-person to a virtual meeting with 634 attendees. We launched numerous COVID-19 webinars and made our clinical and educational offerings open access. Our Public Policy Committee was active around both COVID-19 and hospitalist-related topics – immigration, telehealth, wellbeing, and financial impacts, to name a few. (And I even met with the POTUS & advocated for PPE.) The Journal of Hospital Medicine worked with authors to get important publications out at record speed. And of course, The Hospitalist connected all of us to our professional leaders and experts.
By the fall of 2020, SHM had actively adjusted to the “new normal” of this pandemic: SHM staff have settled into their new “work from home” environments, SHM Chapters are connecting members in the virtual world, SHM’s 2021 Annual Conference will be all virtual – rebranded as “SHM Converge” – and the State of Hospital Medicine Report (our every-other-year source for trends in hospital medicine) now has a COVID-19 supplement, which was developed at lightning speed. Even our SHM Board of Directors is meeting virtually! All this while advancing the routine work at SHM, which never faltered. Our work on resources for quality improvement, the opioid epidemic, wellbeing, diversity, equity and inclusion (DEI), leadership, professional development, advocacy, and so much more is as active as ever.
I don’t know how much longer we have on this very long pandemic journey, so I’ll use my father’s answer of “we’re about halfway through.” We have been immersed in it for months already, with months still ahead. But regardless of the upcoming twists and turns COVID-19 forces you, our patients, and our larger society to take, SHM is ready to change direction faster than a 1970s Chevy. The SHM staff, leadership, and members will be sure that hospitalists receive the tools to navigate these unprecedented times. Our patients need our skills to get through this as safely as possible. While we may not be able to tell them “how much longer,” we can certainly be prepared for the long road ahead as we begin 2021.
Dr. Howell is CEO of the Society of Hospital Medicine.
SHM has changed direction as needed during the pandemic
SHM has changed direction as needed during the pandemic
“How much longer?” As a kid, I can remember the long holiday car ride with my parents from my home in Annapolis, Md., to Upstate New York where my grandparents lived. At the time, the ride felt like an eternity: endless miles of frozen landscape, limited food, and a brother who constantly crossed over the invisible line that was my side of the car.
We made our parents crazy asking, “how much longer?” every few minutes. This was the late 1970s, with no GPS or Google Maps to give you arrival times to the minute, traffic warnings, or reroutes when the inevitable delays occurred. We just plowed ahead, and my parents’ answer was always something vague like, “in a few hours” or “we’re about halfway through.” They did not know when we’d arrive with certainty either.
We at SHM have that same feeling about the pandemic. How much longer? No one can tell us when the COVID-19 threat will abate. The experts’ answers are understandably vague, and the tools for forecasting are non-existent. Months? That is the best we know for now.
At SHM, we believe we will make it through this journey by adapting to roadblocks, providing tools for success to our professional community, and identifying opportunities for us to connect with each other, even if that means virtually.
Like the rest of the planet, the spring of 2020 hit SHM with a shock. Hospital Medicine 2020 (HM20) in San Diego was shaping up to be the largest Annual Conference SHM ever had, the Pediatric Hospital Medicine 2020 (PHM20) conference was well planned and expected to be a huge success, regional SHM chapters were meeting (and growing), and membership was thriving. I was transitioning out of my roles at Johns Hopkins and looking forward to my new role as CEO of SHM. All in all, March 2020 began with a fantastic outlook.
Wow, what a difference a few weeks made. We watched as the pandemic spread across regions of the country, concerned for the wellbeing of our patients and our hospitalists. We saw how our members were at the forefront of patient care during this crisis and understood that SHM had to adapt rapidly to meet their needs in real time.
By May, SHM had canceled HM20, Chapter activity was halted, PHM20 was on its way to being canceled, SHM committee work was put on hold, and I was spending my last few months at Hopkins as the chief medical officer at the Baltimore Convention Center Field Hospital (which we got up and running in less than a month)! Whew.
But just like my dad could pivot our 1970s Chevy station wagon around a traffic jam in a flash, so too did SHM leadership start navigating around the COVID-19 landscape. As soon as HM20 was canceled, SHM immediately began planning for a virtual offering in August. We had hoped to attract at least 100 attendees and we were thrilled to have more than 1,000! PHM20 was switched from an in-person to a virtual meeting with 634 attendees. We launched numerous COVID-19 webinars and made our clinical and educational offerings open access. Our Public Policy Committee was active around both COVID-19 and hospitalist-related topics – immigration, telehealth, wellbeing, and financial impacts, to name a few. (And I even met with the POTUS & advocated for PPE.) The Journal of Hospital Medicine worked with authors to get important publications out at record speed. And of course, The Hospitalist connected all of us to our professional leaders and experts.
By the fall of 2020, SHM had actively adjusted to the “new normal” of this pandemic: SHM staff have settled into their new “work from home” environments, SHM Chapters are connecting members in the virtual world, SHM’s 2021 Annual Conference will be all virtual – rebranded as “SHM Converge” – and the State of Hospital Medicine Report (our every-other-year source for trends in hospital medicine) now has a COVID-19 supplement, which was developed at lightning speed. Even our SHM Board of Directors is meeting virtually! All this while advancing the routine work at SHM, which never faltered. Our work on resources for quality improvement, the opioid epidemic, wellbeing, diversity, equity and inclusion (DEI), leadership, professional development, advocacy, and so much more is as active as ever.
I don’t know how much longer we have on this very long pandemic journey, so I’ll use my father’s answer of “we’re about halfway through.” We have been immersed in it for months already, with months still ahead. But regardless of the upcoming twists and turns COVID-19 forces you, our patients, and our larger society to take, SHM is ready to change direction faster than a 1970s Chevy. The SHM staff, leadership, and members will be sure that hospitalists receive the tools to navigate these unprecedented times. Our patients need our skills to get through this as safely as possible. While we may not be able to tell them “how much longer,” we can certainly be prepared for the long road ahead as we begin 2021.
Dr. Howell is CEO of the Society of Hospital Medicine.
“How much longer?” As a kid, I can remember the long holiday car ride with my parents from my home in Annapolis, Md., to Upstate New York where my grandparents lived. At the time, the ride felt like an eternity: endless miles of frozen landscape, limited food, and a brother who constantly crossed over the invisible line that was my side of the car.
We made our parents crazy asking, “how much longer?” every few minutes. This was the late 1970s, with no GPS or Google Maps to give you arrival times to the minute, traffic warnings, or reroutes when the inevitable delays occurred. We just plowed ahead, and my parents’ answer was always something vague like, “in a few hours” or “we’re about halfway through.” They did not know when we’d arrive with certainty either.
We at SHM have that same feeling about the pandemic. How much longer? No one can tell us when the COVID-19 threat will abate. The experts’ answers are understandably vague, and the tools for forecasting are non-existent. Months? That is the best we know for now.
At SHM, we believe we will make it through this journey by adapting to roadblocks, providing tools for success to our professional community, and identifying opportunities for us to connect with each other, even if that means virtually.
Like the rest of the planet, the spring of 2020 hit SHM with a shock. Hospital Medicine 2020 (HM20) in San Diego was shaping up to be the largest Annual Conference SHM ever had, the Pediatric Hospital Medicine 2020 (PHM20) conference was well planned and expected to be a huge success, regional SHM chapters were meeting (and growing), and membership was thriving. I was transitioning out of my roles at Johns Hopkins and looking forward to my new role as CEO of SHM. All in all, March 2020 began with a fantastic outlook.
Wow, what a difference a few weeks made. We watched as the pandemic spread across regions of the country, concerned for the wellbeing of our patients and our hospitalists. We saw how our members were at the forefront of patient care during this crisis and understood that SHM had to adapt rapidly to meet their needs in real time.
By May, SHM had canceled HM20, Chapter activity was halted, PHM20 was on its way to being canceled, SHM committee work was put on hold, and I was spending my last few months at Hopkins as the chief medical officer at the Baltimore Convention Center Field Hospital (which we got up and running in less than a month)! Whew.
But just like my dad could pivot our 1970s Chevy station wagon around a traffic jam in a flash, so too did SHM leadership start navigating around the COVID-19 landscape. As soon as HM20 was canceled, SHM immediately began planning for a virtual offering in August. We had hoped to attract at least 100 attendees and we were thrilled to have more than 1,000! PHM20 was switched from an in-person to a virtual meeting with 634 attendees. We launched numerous COVID-19 webinars and made our clinical and educational offerings open access. Our Public Policy Committee was active around both COVID-19 and hospitalist-related topics – immigration, telehealth, wellbeing, and financial impacts, to name a few. (And I even met with the POTUS & advocated for PPE.) The Journal of Hospital Medicine worked with authors to get important publications out at record speed. And of course, The Hospitalist connected all of us to our professional leaders and experts.
By the fall of 2020, SHM had actively adjusted to the “new normal” of this pandemic: SHM staff have settled into their new “work from home” environments, SHM Chapters are connecting members in the virtual world, SHM’s 2021 Annual Conference will be all virtual – rebranded as “SHM Converge” – and the State of Hospital Medicine Report (our every-other-year source for trends in hospital medicine) now has a COVID-19 supplement, which was developed at lightning speed. Even our SHM Board of Directors is meeting virtually! All this while advancing the routine work at SHM, which never faltered. Our work on resources for quality improvement, the opioid epidemic, wellbeing, diversity, equity and inclusion (DEI), leadership, professional development, advocacy, and so much more is as active as ever.
I don’t know how much longer we have on this very long pandemic journey, so I’ll use my father’s answer of “we’re about halfway through.” We have been immersed in it for months already, with months still ahead. But regardless of the upcoming twists and turns COVID-19 forces you, our patients, and our larger society to take, SHM is ready to change direction faster than a 1970s Chevy. The SHM staff, leadership, and members will be sure that hospitalists receive the tools to navigate these unprecedented times. Our patients need our skills to get through this as safely as possible. While we may not be able to tell them “how much longer,” we can certainly be prepared for the long road ahead as we begin 2021.
Dr. Howell is CEO of the Society of Hospital Medicine.
December 2020 - Quick Quiz Question 2
Q2. Correct answer: D
Rationale
Deficient intake of fiber and folate may originate in the food choice of the individual, whereas some deficiencies of intake, such as thiamine, appear to be celiac specific. The provider should encourage intake of nutrient-dense foods including wholegrain foods, enriched if possible, legumes, fruits, vegetables, lean meat, fish, chicken, and eggs. It is not necessary to prioritize micronutrient supplements over achieving nutritional adequacy through dietary intake. Iron deficiency is an effect of untreated celiac disease.
Reference
1. Shepherd SJ, Gibson PR. J Human Nutr Dietet. 2012;26:349-58.
Q2. Correct answer: D
Rationale
Deficient intake of fiber and folate may originate in the food choice of the individual, whereas some deficiencies of intake, such as thiamine, appear to be celiac specific. The provider should encourage intake of nutrient-dense foods including wholegrain foods, enriched if possible, legumes, fruits, vegetables, lean meat, fish, chicken, and eggs. It is not necessary to prioritize micronutrient supplements over achieving nutritional adequacy through dietary intake. Iron deficiency is an effect of untreated celiac disease.
Reference
1. Shepherd SJ, Gibson PR. J Human Nutr Dietet. 2012;26:349-58.
Q2. Correct answer: D
Rationale
Deficient intake of fiber and folate may originate in the food choice of the individual, whereas some deficiencies of intake, such as thiamine, appear to be celiac specific. The provider should encourage intake of nutrient-dense foods including wholegrain foods, enriched if possible, legumes, fruits, vegetables, lean meat, fish, chicken, and eggs. It is not necessary to prioritize micronutrient supplements over achieving nutritional adequacy through dietary intake. Iron deficiency is an effect of untreated celiac disease.
Reference
1. Shepherd SJ, Gibson PR. J Human Nutr Dietet. 2012;26:349-58.
Question 2
December 2020 - Quick Quiz Question 1
Correct answer: C
Rationale
According to the Multi-Society Task Force on Colorectal Cancer, colonoscopy should be performed 1 year after resection, and again 3 years later, in order to decrease the risk of metachronous colorectal cancer.
Reference
1. Kahi CJ, Boland CR, Dominitz JA. Gastroenterology. 2016. 150(3):758-68.e11.
Correct answer: C
Rationale
According to the Multi-Society Task Force on Colorectal Cancer, colonoscopy should be performed 1 year after resection, and again 3 years later, in order to decrease the risk of metachronous colorectal cancer.
Reference
1. Kahi CJ, Boland CR, Dominitz JA. Gastroenterology. 2016. 150(3):758-68.e11.
Correct answer: C
Rationale
According to the Multi-Society Task Force on Colorectal Cancer, colonoscopy should be performed 1 year after resection, and again 3 years later, in order to decrease the risk of metachronous colorectal cancer.
Reference
1. Kahi CJ, Boland CR, Dominitz JA. Gastroenterology. 2016. 150(3):758-68.e11.
You perform a colonoscopy for a patient who underwent sigmoid resection for stage 2 colorectal cancer 1 year ago. The colonoscopy reveals one diminutive adenoma in the cecum, which you remove with a cold snare.
Pigment traits, sun sensitivity associated with risk of non-Hodgkin lymphomas and chronic lymphocytic leukemia
Risk factors for keratinocyte carcinomas, primarily pigment traits and sun sensitivity, were associated with the risk of developing non-Hodgkin lymphomas (NHL) and chronic lymphocytic leukemia (CLL) in an analysis of 92,097 women in France.
The presence of “many or very many nevi [moles]” was particularly associated with the risk of CLL among individuals in the E3N cohort, according to a report published online in Cancer Medicine. E3N is a prospective cohort of French women aged 40-65 years at inclusion in 1990. Researchers collected cancer data at baseline and every 2-3 years.
Hazard ratios and 95% confidence intervals for associations between patients pigmentary traits and sun exposure and their risk for CLL/NHL were estimated using Cox models, according to study author Louis-Marie Garcin, MD, of the Université Paris-Saclay, Villejuif, and colleagues.
Common etiology?
Among the 92,097 women included in the study, 622 incident cases of CLL/NHL were observed over a median of 24-years’ follow-up.
The presence of nevi was associated with CLL/NHL risk. The HR for “many or very many nevi” relative to “no nevi” was 1.56. The association with number of nevi was strongest for the risk of CLL, with an HR for “many or very many nevi” of 3.00 vs. 1.32 for NHL. In addition, the researchers found that women whose skin was highly sensitive to sunburn also had a higher risk of CLL (HR, 1.96), while no increased risk of NHL was observed. All HR values were within their respective 95% confidence intervals.
Relevant characteristics that were found to not be associated with added CLL/NHL risk were skin or hair color, number of freckles, and average daily UV dose during spring and summer in the location of residence at birth or at inclusion.
These observations suggest that CLL in particular may share some constitutional risk factors with keratinocyte cancers, according to the researchers.
“We report an association between nevi frequency and CLL/NHL risk, suggesting a partly common genetic etiology of these tumors. Future research should investigate common pathophysiological pathways that could promote the development of both skin carcinoma and CLL/NHL,” the researchers concluded.
The study was sponsored by the French government. The authors stated that they had no conflicts of interest.
SOURCE: Garcin L-M et al. Cancer Med. 2020. doi: 10.1002/cam4.3586.
Risk factors for keratinocyte carcinomas, primarily pigment traits and sun sensitivity, were associated with the risk of developing non-Hodgkin lymphomas (NHL) and chronic lymphocytic leukemia (CLL) in an analysis of 92,097 women in France.
The presence of “many or very many nevi [moles]” was particularly associated with the risk of CLL among individuals in the E3N cohort, according to a report published online in Cancer Medicine. E3N is a prospective cohort of French women aged 40-65 years at inclusion in 1990. Researchers collected cancer data at baseline and every 2-3 years.
Hazard ratios and 95% confidence intervals for associations between patients pigmentary traits and sun exposure and their risk for CLL/NHL were estimated using Cox models, according to study author Louis-Marie Garcin, MD, of the Université Paris-Saclay, Villejuif, and colleagues.
Common etiology?
Among the 92,097 women included in the study, 622 incident cases of CLL/NHL were observed over a median of 24-years’ follow-up.
The presence of nevi was associated with CLL/NHL risk. The HR for “many or very many nevi” relative to “no nevi” was 1.56. The association with number of nevi was strongest for the risk of CLL, with an HR for “many or very many nevi” of 3.00 vs. 1.32 for NHL. In addition, the researchers found that women whose skin was highly sensitive to sunburn also had a higher risk of CLL (HR, 1.96), while no increased risk of NHL was observed. All HR values were within their respective 95% confidence intervals.
Relevant characteristics that were found to not be associated with added CLL/NHL risk were skin or hair color, number of freckles, and average daily UV dose during spring and summer in the location of residence at birth or at inclusion.
These observations suggest that CLL in particular may share some constitutional risk factors with keratinocyte cancers, according to the researchers.
“We report an association between nevi frequency and CLL/NHL risk, suggesting a partly common genetic etiology of these tumors. Future research should investigate common pathophysiological pathways that could promote the development of both skin carcinoma and CLL/NHL,” the researchers concluded.
The study was sponsored by the French government. The authors stated that they had no conflicts of interest.
SOURCE: Garcin L-M et al. Cancer Med. 2020. doi: 10.1002/cam4.3586.
Risk factors for keratinocyte carcinomas, primarily pigment traits and sun sensitivity, were associated with the risk of developing non-Hodgkin lymphomas (NHL) and chronic lymphocytic leukemia (CLL) in an analysis of 92,097 women in France.
The presence of “many or very many nevi [moles]” was particularly associated with the risk of CLL among individuals in the E3N cohort, according to a report published online in Cancer Medicine. E3N is a prospective cohort of French women aged 40-65 years at inclusion in 1990. Researchers collected cancer data at baseline and every 2-3 years.
Hazard ratios and 95% confidence intervals for associations between patients pigmentary traits and sun exposure and their risk for CLL/NHL were estimated using Cox models, according to study author Louis-Marie Garcin, MD, of the Université Paris-Saclay, Villejuif, and colleagues.
Common etiology?
Among the 92,097 women included in the study, 622 incident cases of CLL/NHL were observed over a median of 24-years’ follow-up.
The presence of nevi was associated with CLL/NHL risk. The HR for “many or very many nevi” relative to “no nevi” was 1.56. The association with number of nevi was strongest for the risk of CLL, with an HR for “many or very many nevi” of 3.00 vs. 1.32 for NHL. In addition, the researchers found that women whose skin was highly sensitive to sunburn also had a higher risk of CLL (HR, 1.96), while no increased risk of NHL was observed. All HR values were within their respective 95% confidence intervals.
Relevant characteristics that were found to not be associated with added CLL/NHL risk were skin or hair color, number of freckles, and average daily UV dose during spring and summer in the location of residence at birth or at inclusion.
These observations suggest that CLL in particular may share some constitutional risk factors with keratinocyte cancers, according to the researchers.
“We report an association between nevi frequency and CLL/NHL risk, suggesting a partly common genetic etiology of these tumors. Future research should investigate common pathophysiological pathways that could promote the development of both skin carcinoma and CLL/NHL,” the researchers concluded.
The study was sponsored by the French government. The authors stated that they had no conflicts of interest.
SOURCE: Garcin L-M et al. Cancer Med. 2020. doi: 10.1002/cam4.3586.
FROM CANCER MEDICINE
Photosensitivity diagnosis made simple
of the University of Southern California, Los Angeles.
“When a patient comes in who makes you suspect a photosensitivity, there will be two different presentations,” he said in a virtual presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
In some cases, the patient presents with a reaction they believe is sun related, although they don’t have a rash currently, he said. In other cases, “you as a good clinician suspect photosensitivity because the eruption is in a photo distribution,” although the patient may or may not relate it to sun exposure, he added.
Dr. DeLeo noted a few key points to include when taking the history in patients with likely photosensitivity, whether or not they present with a rash.
“I always ask patients when did the episode occur? Is it chronic?” Also ask about timing: Does the reaction occur in the sun, or later? Does it occur quickly and go away within hours, or occur within days or weeks of exposure?
“Always take a good drug history, as photosensitivity can often be related to drugs,” Dr. DeLeo noted. For example, approximately 50% of individuals on amiodarone will have some type of photosensitivity, he said.
Other drug-induced photosensitive conditions include drug-induced subacute cutaneous lupus and pseudoporphyria from NSAIDs, as well as hyperpigmentation from diltiazem, which most often occurs in Black women, he said.
“Photodrug reactions are usually related to UVA radiation, and that is important because you can develop it through the window while driving in your car”: The car windows do not protect against UVA, Dr. DeLeo said. If you have a patient who tells you about a photosensitivity or has a rash and they are on a photosensitizing drug, first rule out connective tissue disease, then discontinue the drug in collaboration with the patient’s internist and wait for the reaction to disappear, and it should, he said.
Some photosensitivity rashes have characteristic patterns, notably connective tissue disease patterns in lupus and dermatomyositis patients, bullous eruptions in cases of porphyria or phototoxic contact dermatitis, and eczematous eruptions, Dr. DeLeo noted.
Patients who present without a rash, but report a history of a reaction that they believe is related to sun exposure, fall into two categories: some had a rash that occurred while in the sun and disappeared quickly, and some had one that occurred hours or days after exposure and lasted a few days to weeks, said Dr. DeLeo.
The differential diagnosis in the patient with immediate photosensitivity is fairly clear: These patients usually have solar urticaria, he said. However, some lupus patients may report this reaction so it is important to rule out connective tissue disease. The diagnosis can be made with phototesting or do a simple test by having the patient sit out in the sunshine, he said.
For the patient who has a delayed reactivity after sun exposure, and doesn’t have the reaction when they come to the office, the differential diagnosis in a simply applied way is that, if the reaction spared the face, it is likely polymorphous light eruption (PMLE); but if the face is involved, the patient likely has photoallergic contact dermatitis, Dr. DeLeo explained. However, always consider the alternatives of connective tissue disease, drug reactions, and contact dermatitis that is not photoallergic, he noted.
PMLE “is the most common photosensitivity reaction that we see in the United States,” and it almost always occurs when people are away from home, usually on vacation, said Dr. DeLeo. The differential diagnosis for patients with recurrent or delayed rash involving the face could be photoallergic contact dermatitis, but rule out airborne contact dermatitis, personal care product contact dermatitis, and chronic actinic dermatitis, he said. A work-up for these patients could include a photo test, photopatch test, or patch test.
Dr. DeLeo disclosed serving as a consultant for Estee Lauder.
MedscapeLive and this news organization are owned by the same parent company.
of the University of Southern California, Los Angeles.
“When a patient comes in who makes you suspect a photosensitivity, there will be two different presentations,” he said in a virtual presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
In some cases, the patient presents with a reaction they believe is sun related, although they don’t have a rash currently, he said. In other cases, “you as a good clinician suspect photosensitivity because the eruption is in a photo distribution,” although the patient may or may not relate it to sun exposure, he added.
Dr. DeLeo noted a few key points to include when taking the history in patients with likely photosensitivity, whether or not they present with a rash.
“I always ask patients when did the episode occur? Is it chronic?” Also ask about timing: Does the reaction occur in the sun, or later? Does it occur quickly and go away within hours, or occur within days or weeks of exposure?
“Always take a good drug history, as photosensitivity can often be related to drugs,” Dr. DeLeo noted. For example, approximately 50% of individuals on amiodarone will have some type of photosensitivity, he said.
Other drug-induced photosensitive conditions include drug-induced subacute cutaneous lupus and pseudoporphyria from NSAIDs, as well as hyperpigmentation from diltiazem, which most often occurs in Black women, he said.
“Photodrug reactions are usually related to UVA radiation, and that is important because you can develop it through the window while driving in your car”: The car windows do not protect against UVA, Dr. DeLeo said. If you have a patient who tells you about a photosensitivity or has a rash and they are on a photosensitizing drug, first rule out connective tissue disease, then discontinue the drug in collaboration with the patient’s internist and wait for the reaction to disappear, and it should, he said.
Some photosensitivity rashes have characteristic patterns, notably connective tissue disease patterns in lupus and dermatomyositis patients, bullous eruptions in cases of porphyria or phototoxic contact dermatitis, and eczematous eruptions, Dr. DeLeo noted.
Patients who present without a rash, but report a history of a reaction that they believe is related to sun exposure, fall into two categories: some had a rash that occurred while in the sun and disappeared quickly, and some had one that occurred hours or days after exposure and lasted a few days to weeks, said Dr. DeLeo.
The differential diagnosis in the patient with immediate photosensitivity is fairly clear: These patients usually have solar urticaria, he said. However, some lupus patients may report this reaction so it is important to rule out connective tissue disease. The diagnosis can be made with phototesting or do a simple test by having the patient sit out in the sunshine, he said.
For the patient who has a delayed reactivity after sun exposure, and doesn’t have the reaction when they come to the office, the differential diagnosis in a simply applied way is that, if the reaction spared the face, it is likely polymorphous light eruption (PMLE); but if the face is involved, the patient likely has photoallergic contact dermatitis, Dr. DeLeo explained. However, always consider the alternatives of connective tissue disease, drug reactions, and contact dermatitis that is not photoallergic, he noted.
PMLE “is the most common photosensitivity reaction that we see in the United States,” and it almost always occurs when people are away from home, usually on vacation, said Dr. DeLeo. The differential diagnosis for patients with recurrent or delayed rash involving the face could be photoallergic contact dermatitis, but rule out airborne contact dermatitis, personal care product contact dermatitis, and chronic actinic dermatitis, he said. A work-up for these patients could include a photo test, photopatch test, or patch test.
Dr. DeLeo disclosed serving as a consultant for Estee Lauder.
MedscapeLive and this news organization are owned by the same parent company.
of the University of Southern California, Los Angeles.
“When a patient comes in who makes you suspect a photosensitivity, there will be two different presentations,” he said in a virtual presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
In some cases, the patient presents with a reaction they believe is sun related, although they don’t have a rash currently, he said. In other cases, “you as a good clinician suspect photosensitivity because the eruption is in a photo distribution,” although the patient may or may not relate it to sun exposure, he added.
Dr. DeLeo noted a few key points to include when taking the history in patients with likely photosensitivity, whether or not they present with a rash.
“I always ask patients when did the episode occur? Is it chronic?” Also ask about timing: Does the reaction occur in the sun, or later? Does it occur quickly and go away within hours, or occur within days or weeks of exposure?
“Always take a good drug history, as photosensitivity can often be related to drugs,” Dr. DeLeo noted. For example, approximately 50% of individuals on amiodarone will have some type of photosensitivity, he said.
Other drug-induced photosensitive conditions include drug-induced subacute cutaneous lupus and pseudoporphyria from NSAIDs, as well as hyperpigmentation from diltiazem, which most often occurs in Black women, he said.
“Photodrug reactions are usually related to UVA radiation, and that is important because you can develop it through the window while driving in your car”: The car windows do not protect against UVA, Dr. DeLeo said. If you have a patient who tells you about a photosensitivity or has a rash and they are on a photosensitizing drug, first rule out connective tissue disease, then discontinue the drug in collaboration with the patient’s internist and wait for the reaction to disappear, and it should, he said.
Some photosensitivity rashes have characteristic patterns, notably connective tissue disease patterns in lupus and dermatomyositis patients, bullous eruptions in cases of porphyria or phototoxic contact dermatitis, and eczematous eruptions, Dr. DeLeo noted.
Patients who present without a rash, but report a history of a reaction that they believe is related to sun exposure, fall into two categories: some had a rash that occurred while in the sun and disappeared quickly, and some had one that occurred hours or days after exposure and lasted a few days to weeks, said Dr. DeLeo.
The differential diagnosis in the patient with immediate photosensitivity is fairly clear: These patients usually have solar urticaria, he said. However, some lupus patients may report this reaction so it is important to rule out connective tissue disease. The diagnosis can be made with phototesting or do a simple test by having the patient sit out in the sunshine, he said.
For the patient who has a delayed reactivity after sun exposure, and doesn’t have the reaction when they come to the office, the differential diagnosis in a simply applied way is that, if the reaction spared the face, it is likely polymorphous light eruption (PMLE); but if the face is involved, the patient likely has photoallergic contact dermatitis, Dr. DeLeo explained. However, always consider the alternatives of connective tissue disease, drug reactions, and contact dermatitis that is not photoallergic, he noted.
PMLE “is the most common photosensitivity reaction that we see in the United States,” and it almost always occurs when people are away from home, usually on vacation, said Dr. DeLeo. The differential diagnosis for patients with recurrent or delayed rash involving the face could be photoallergic contact dermatitis, but rule out airborne contact dermatitis, personal care product contact dermatitis, and chronic actinic dermatitis, he said. A work-up for these patients could include a photo test, photopatch test, or patch test.
Dr. DeLeo disclosed serving as a consultant for Estee Lauder.
MedscapeLive and this news organization are owned by the same parent company.
FROM MEDSCAPELIVE LAS VEGAS DERMATOLOGY SEMINAR
Scalp Arteriovenous Fistula With Intracranial Communication
To the Editor:
A 71-year-old man presented with a nodule on the vertex of the scalp of 1 year’s duration. The lesion had become soft and tender during the week prior to presentation. He noted that he was experiencing headaches and a buzzing sound in his head. He denied all other neurologic symptoms. The patient was given amoxicillin from a primary care physician and was referred to our institution for evaluation of a presumed inflamed cyst.
The patient’s medical history included an intracranial arteriovenous fistula (AVF) treated with endovascular embolization 1 year prior to presentation, 2 substantial falls in childhood with head trauma and loss of consciousness, essential hypertension, and an aortic aneurysm. His medications included amlodipine, lisinopril, amoxicillin, a multivitamin, and grape seed extract.
Physical examination revealed a 2-cm, pink, somewhat rubbery, subcutaneous, nonmobile nodule on the vertex of the scalp (Figure 1). The lesion was not consistent with a common pilar cyst, and an excisional biopsy was performed to exclude malignancy. Upon superficial incision, the lesion bled moderately, and the procedure was immediately discontinued. Hemostasis was obtained, and the patient was sent for ultrasonography of the lesion.
Ultrasonography demonstrated a small hypoechoic nodule measuring up to 0.5 cm containing a tangle of vessels in the subcutaneous soft tissue corresponding to the palpable abnormality. A cerebral angiogram demonstrated a dural AVF of the superior sagittal sinus with multifocal supply that connected with this scalp nodule (Figure 2). The patient was treated by interventional neuroradiology with endovascular embolization, which resulted in complete resolution of the scalp nodule.
Scalp arteriovenous fistulas (S-AVFs) are characterized by abnormal connections between supplying arteries and draining veins in the subcutaneous plane of the scalp.1,2 The veins of an S-AVF undergo progressive aneurysmal dilatation from abnormal hemodynamics.1-3 Scalp arteriovenous fistulas are rare and may present as either an innocuous-looking scalp nodule or a progressively enlarging pulsatile mass on the scalp.2-4 Associated symptoms often include headache, local pain, bruits, tinnitus, and thrill.1,3,4 Recurrent hemorrhage, scalp necrosis, congestive heart failure, epilepsy, mental retardation, and intracranial ischemia also may occur.4
Scalp AVFs may occur with or without intracranial communication.4 Spontaneous S-AVFs with intracranial communication are uncommon, and their etiology is unclear. They may form as congenital malformations or may be idiopathic. Factors increasing circulation through the S-AVF such as trauma, pregnancy, hormonal changes, and inflammation prompt the development of symptoms.4 Scalp AVFs also may be caused by trauma.3 Scalp AVFs without intracranial communication have been reported following hair transplantation.1 Scalp AVFs with intracranial communication have been reported months to years after skull fracture or craniotomy.2 True spontaneous S-AVFs are difficult to differentiate from traumatic S-AVFs other than by history alone.2
Increased venous pressure has been shown to generate AVFs in rats.5 It has been suggested that S-AVFs can become enlarged by capturing subcutaneous or intracranial feeder vessels and that the consequent hemodynamic stress may induce de novo aneurysms in S-AVFs. Additionally, intracranial AVFs may alter the intracranial hemodynamics, leading to increased venous pressure in the superior sagittal sinus and the formation of communicating S-AVFs.5 Interestingly, our patient had an intracranial AVF treated with endovascular embolization 1 year prior to the formation of the S-AVF. An angiogram at the time of this embolization procedure did not demonstrate any S-AVFs. Furthermore, our patient has a history of 2 substantial falls in childhood with head trauma and loss of consciousness. Perhaps these traumas initiated a channel through the cranium where an S-AVF with intracranial communication was able to form and may have only become clinically or radiographically detectable once it enlarged due to the altered hemodynamics caused by the intracranial AVF 1 year prior.
The diagnosis of an S-AVF is confirmed with imaging studies. Doppler ultrasonography initially will help to detect that a lesion is vascular in nature. Intra-arterial digital subtraction angiography is the gold-standard imaging technique and is necessary to delineate the feeding arteries and the draining channels as well as possible communication with intracranial vasculature.1,2 There is controversy regarding the appropriate treatment of S-AVFs.2 Each S-AVF possesses unique anatomic features that dictate appropriate management. The prognosis for an S-AVF is extremely variable, and the decision to treat is based on the patient’s symptoms and risk for exsanguinating hemorrhage.2,4 Neurosurgical approaches include ligation of the feeding arteries, surgical resection, electrothrombosis, direct intralesional injection of sclerosing agents, and endovascular embolization. Endovascular intervention increasingly is utilized as a primary treatment or as a preoperative adjunct to surgery.2,4 Large S-AVFs have a high risk for recurrence after treatment with endovascular embolization alone. In cases with intracranial communication, the intracranial component is treated first.2
This case emphasizes the importance of including S-AVFs on the dermatologic differential diagnosis of a scalp nodule, especially in patients with any history of intracranial AVF. A thorough history, detailed intake of potential signs and symptoms of AVF, and palpation for bruits is recommended as part of the surgical evaluation of a scalp nodule. Imaging of scalp nodules also should be considered for patients with any history of intracranial AVF; S-AVFs should be referred to neurosurgery or interventional neuroradiology for evaluation and possible treatment.
- Bernstein J, Podnos S, Leavitt M. Arteriovenous fistula following hair transplantation. Dermatol Surg. 2011;37:873-875.
- Kumar R, Sharma G, Sharma BS. Management of scalp arterio-venous malformation: case series and review of literature. Br J Neurosurg. 2012;26:371-377.
- Gurkanlar D, Gonul M, Solmaz I, et al. Cirsoid aneurysms of the scalp. Neurosurg Rev. 2006;29:208-212.
- Senoglu M, Yasim A, Gokce M, et al. Nontraumatic scalp arteriovenous fistula in an adult: technical report on an illustrative case. Surg Neurol. 2008;70:194-197.
- Lanzino G, Passacantilli E, Lemole G, et al. Scalp arteriovenous malformation draining into the superior sagittal sinus associated with an intracranial arteriovenous malformation: just a coincidence? case report. Neurosurgery. 2003;52:440-443.
To the Editor:
A 71-year-old man presented with a nodule on the vertex of the scalp of 1 year’s duration. The lesion had become soft and tender during the week prior to presentation. He noted that he was experiencing headaches and a buzzing sound in his head. He denied all other neurologic symptoms. The patient was given amoxicillin from a primary care physician and was referred to our institution for evaluation of a presumed inflamed cyst.
The patient’s medical history included an intracranial arteriovenous fistula (AVF) treated with endovascular embolization 1 year prior to presentation, 2 substantial falls in childhood with head trauma and loss of consciousness, essential hypertension, and an aortic aneurysm. His medications included amlodipine, lisinopril, amoxicillin, a multivitamin, and grape seed extract.
Physical examination revealed a 2-cm, pink, somewhat rubbery, subcutaneous, nonmobile nodule on the vertex of the scalp (Figure 1). The lesion was not consistent with a common pilar cyst, and an excisional biopsy was performed to exclude malignancy. Upon superficial incision, the lesion bled moderately, and the procedure was immediately discontinued. Hemostasis was obtained, and the patient was sent for ultrasonography of the lesion.
Ultrasonography demonstrated a small hypoechoic nodule measuring up to 0.5 cm containing a tangle of vessels in the subcutaneous soft tissue corresponding to the palpable abnormality. A cerebral angiogram demonstrated a dural AVF of the superior sagittal sinus with multifocal supply that connected with this scalp nodule (Figure 2). The patient was treated by interventional neuroradiology with endovascular embolization, which resulted in complete resolution of the scalp nodule.
Scalp arteriovenous fistulas (S-AVFs) are characterized by abnormal connections between supplying arteries and draining veins in the subcutaneous plane of the scalp.1,2 The veins of an S-AVF undergo progressive aneurysmal dilatation from abnormal hemodynamics.1-3 Scalp arteriovenous fistulas are rare and may present as either an innocuous-looking scalp nodule or a progressively enlarging pulsatile mass on the scalp.2-4 Associated symptoms often include headache, local pain, bruits, tinnitus, and thrill.1,3,4 Recurrent hemorrhage, scalp necrosis, congestive heart failure, epilepsy, mental retardation, and intracranial ischemia also may occur.4
Scalp AVFs may occur with or without intracranial communication.4 Spontaneous S-AVFs with intracranial communication are uncommon, and their etiology is unclear. They may form as congenital malformations or may be idiopathic. Factors increasing circulation through the S-AVF such as trauma, pregnancy, hormonal changes, and inflammation prompt the development of symptoms.4 Scalp AVFs also may be caused by trauma.3 Scalp AVFs without intracranial communication have been reported following hair transplantation.1 Scalp AVFs with intracranial communication have been reported months to years after skull fracture or craniotomy.2 True spontaneous S-AVFs are difficult to differentiate from traumatic S-AVFs other than by history alone.2
Increased venous pressure has been shown to generate AVFs in rats.5 It has been suggested that S-AVFs can become enlarged by capturing subcutaneous or intracranial feeder vessels and that the consequent hemodynamic stress may induce de novo aneurysms in S-AVFs. Additionally, intracranial AVFs may alter the intracranial hemodynamics, leading to increased venous pressure in the superior sagittal sinus and the formation of communicating S-AVFs.5 Interestingly, our patient had an intracranial AVF treated with endovascular embolization 1 year prior to the formation of the S-AVF. An angiogram at the time of this embolization procedure did not demonstrate any S-AVFs. Furthermore, our patient has a history of 2 substantial falls in childhood with head trauma and loss of consciousness. Perhaps these traumas initiated a channel through the cranium where an S-AVF with intracranial communication was able to form and may have only become clinically or radiographically detectable once it enlarged due to the altered hemodynamics caused by the intracranial AVF 1 year prior.
The diagnosis of an S-AVF is confirmed with imaging studies. Doppler ultrasonography initially will help to detect that a lesion is vascular in nature. Intra-arterial digital subtraction angiography is the gold-standard imaging technique and is necessary to delineate the feeding arteries and the draining channels as well as possible communication with intracranial vasculature.1,2 There is controversy regarding the appropriate treatment of S-AVFs.2 Each S-AVF possesses unique anatomic features that dictate appropriate management. The prognosis for an S-AVF is extremely variable, and the decision to treat is based on the patient’s symptoms and risk for exsanguinating hemorrhage.2,4 Neurosurgical approaches include ligation of the feeding arteries, surgical resection, electrothrombosis, direct intralesional injection of sclerosing agents, and endovascular embolization. Endovascular intervention increasingly is utilized as a primary treatment or as a preoperative adjunct to surgery.2,4 Large S-AVFs have a high risk for recurrence after treatment with endovascular embolization alone. In cases with intracranial communication, the intracranial component is treated first.2
This case emphasizes the importance of including S-AVFs on the dermatologic differential diagnosis of a scalp nodule, especially in patients with any history of intracranial AVF. A thorough history, detailed intake of potential signs and symptoms of AVF, and palpation for bruits is recommended as part of the surgical evaluation of a scalp nodule. Imaging of scalp nodules also should be considered for patients with any history of intracranial AVF; S-AVFs should be referred to neurosurgery or interventional neuroradiology for evaluation and possible treatment.
To the Editor:
A 71-year-old man presented with a nodule on the vertex of the scalp of 1 year’s duration. The lesion had become soft and tender during the week prior to presentation. He noted that he was experiencing headaches and a buzzing sound in his head. He denied all other neurologic symptoms. The patient was given amoxicillin from a primary care physician and was referred to our institution for evaluation of a presumed inflamed cyst.
The patient’s medical history included an intracranial arteriovenous fistula (AVF) treated with endovascular embolization 1 year prior to presentation, 2 substantial falls in childhood with head trauma and loss of consciousness, essential hypertension, and an aortic aneurysm. His medications included amlodipine, lisinopril, amoxicillin, a multivitamin, and grape seed extract.
Physical examination revealed a 2-cm, pink, somewhat rubbery, subcutaneous, nonmobile nodule on the vertex of the scalp (Figure 1). The lesion was not consistent with a common pilar cyst, and an excisional biopsy was performed to exclude malignancy. Upon superficial incision, the lesion bled moderately, and the procedure was immediately discontinued. Hemostasis was obtained, and the patient was sent for ultrasonography of the lesion.
Ultrasonography demonstrated a small hypoechoic nodule measuring up to 0.5 cm containing a tangle of vessels in the subcutaneous soft tissue corresponding to the palpable abnormality. A cerebral angiogram demonstrated a dural AVF of the superior sagittal sinus with multifocal supply that connected with this scalp nodule (Figure 2). The patient was treated by interventional neuroradiology with endovascular embolization, which resulted in complete resolution of the scalp nodule.
Scalp arteriovenous fistulas (S-AVFs) are characterized by abnormal connections between supplying arteries and draining veins in the subcutaneous plane of the scalp.1,2 The veins of an S-AVF undergo progressive aneurysmal dilatation from abnormal hemodynamics.1-3 Scalp arteriovenous fistulas are rare and may present as either an innocuous-looking scalp nodule or a progressively enlarging pulsatile mass on the scalp.2-4 Associated symptoms often include headache, local pain, bruits, tinnitus, and thrill.1,3,4 Recurrent hemorrhage, scalp necrosis, congestive heart failure, epilepsy, mental retardation, and intracranial ischemia also may occur.4
Scalp AVFs may occur with or without intracranial communication.4 Spontaneous S-AVFs with intracranial communication are uncommon, and their etiology is unclear. They may form as congenital malformations or may be idiopathic. Factors increasing circulation through the S-AVF such as trauma, pregnancy, hormonal changes, and inflammation prompt the development of symptoms.4 Scalp AVFs also may be caused by trauma.3 Scalp AVFs without intracranial communication have been reported following hair transplantation.1 Scalp AVFs with intracranial communication have been reported months to years after skull fracture or craniotomy.2 True spontaneous S-AVFs are difficult to differentiate from traumatic S-AVFs other than by history alone.2
Increased venous pressure has been shown to generate AVFs in rats.5 It has been suggested that S-AVFs can become enlarged by capturing subcutaneous or intracranial feeder vessels and that the consequent hemodynamic stress may induce de novo aneurysms in S-AVFs. Additionally, intracranial AVFs may alter the intracranial hemodynamics, leading to increased venous pressure in the superior sagittal sinus and the formation of communicating S-AVFs.5 Interestingly, our patient had an intracranial AVF treated with endovascular embolization 1 year prior to the formation of the S-AVF. An angiogram at the time of this embolization procedure did not demonstrate any S-AVFs. Furthermore, our patient has a history of 2 substantial falls in childhood with head trauma and loss of consciousness. Perhaps these traumas initiated a channel through the cranium where an S-AVF with intracranial communication was able to form and may have only become clinically or radiographically detectable once it enlarged due to the altered hemodynamics caused by the intracranial AVF 1 year prior.
The diagnosis of an S-AVF is confirmed with imaging studies. Doppler ultrasonography initially will help to detect that a lesion is vascular in nature. Intra-arterial digital subtraction angiography is the gold-standard imaging technique and is necessary to delineate the feeding arteries and the draining channels as well as possible communication with intracranial vasculature.1,2 There is controversy regarding the appropriate treatment of S-AVFs.2 Each S-AVF possesses unique anatomic features that dictate appropriate management. The prognosis for an S-AVF is extremely variable, and the decision to treat is based on the patient’s symptoms and risk for exsanguinating hemorrhage.2,4 Neurosurgical approaches include ligation of the feeding arteries, surgical resection, electrothrombosis, direct intralesional injection of sclerosing agents, and endovascular embolization. Endovascular intervention increasingly is utilized as a primary treatment or as a preoperative adjunct to surgery.2,4 Large S-AVFs have a high risk for recurrence after treatment with endovascular embolization alone. In cases with intracranial communication, the intracranial component is treated first.2
This case emphasizes the importance of including S-AVFs on the dermatologic differential diagnosis of a scalp nodule, especially in patients with any history of intracranial AVF. A thorough history, detailed intake of potential signs and symptoms of AVF, and palpation for bruits is recommended as part of the surgical evaluation of a scalp nodule. Imaging of scalp nodules also should be considered for patients with any history of intracranial AVF; S-AVFs should be referred to neurosurgery or interventional neuroradiology for evaluation and possible treatment.
- Bernstein J, Podnos S, Leavitt M. Arteriovenous fistula following hair transplantation. Dermatol Surg. 2011;37:873-875.
- Kumar R, Sharma G, Sharma BS. Management of scalp arterio-venous malformation: case series and review of literature. Br J Neurosurg. 2012;26:371-377.
- Gurkanlar D, Gonul M, Solmaz I, et al. Cirsoid aneurysms of the scalp. Neurosurg Rev. 2006;29:208-212.
- Senoglu M, Yasim A, Gokce M, et al. Nontraumatic scalp arteriovenous fistula in an adult: technical report on an illustrative case. Surg Neurol. 2008;70:194-197.
- Lanzino G, Passacantilli E, Lemole G, et al. Scalp arteriovenous malformation draining into the superior sagittal sinus associated with an intracranial arteriovenous malformation: just a coincidence? case report. Neurosurgery. 2003;52:440-443.
- Bernstein J, Podnos S, Leavitt M. Arteriovenous fistula following hair transplantation. Dermatol Surg. 2011;37:873-875.
- Kumar R, Sharma G, Sharma BS. Management of scalp arterio-venous malformation: case series and review of literature. Br J Neurosurg. 2012;26:371-377.
- Gurkanlar D, Gonul M, Solmaz I, et al. Cirsoid aneurysms of the scalp. Neurosurg Rev. 2006;29:208-212.
- Senoglu M, Yasim A, Gokce M, et al. Nontraumatic scalp arteriovenous fistula in an adult: technical report on an illustrative case. Surg Neurol. 2008;70:194-197.
- Lanzino G, Passacantilli E, Lemole G, et al. Scalp arteriovenous malformation draining into the superior sagittal sinus associated with an intracranial arteriovenous malformation: just a coincidence? case report. Neurosurgery. 2003;52:440-443.
Practice Points
- Scalp arteriovenous fistulas may be traumatic or spontaneous and present as either an innocuous-looking scalp nodule or as a progressively enlarging pulsatile mass on the scalp.
- Clinical detection followed by appropriate imaging and referral to neurosurgery or interventional neuroradiology is vital to patient safety.