User login
Study Finds Elevated Skin Cancer Risk Among US Veterans
of recent national data.
“US veterans are known to have increased risk of cancers and cancer morbidity compared to the general US population,” one of the study authors, Sepideh Ashrafzadeh, MD, a third-year dermatology resident at Massachusetts General Hospital, Boston, told this news organization following the annual meeting of the American Society for Dermatologic Surgery, where the results were presented. “There have been several studies that have shown that US veterans have an increased prevalence of melanoma compared to nonveterans,” she said, noting, however, that no study has investigated the prevalence of nonmelanoma skin cancers (NMSCs), which include basal cell carcinomas and squamous cell carcinomas, compared with the general population.
To address this knowledge gap, the researchers performed a national cross-sectional study of adults aged 18 years or older from the 2019-2023 National Health Interview Surveys to examine the prevalence of melanoma and NMSCs among veterans compared with the general US population. They aggregated and tabulated the data by veteran status, defined as having served at any point in the US armed forces, reserves, or national guard, and by demographic and socioeconomic status variables. Next, they performed multivariate logistic regression for skin cancer risk adjusted for age, sex, race, ethnicity, urbanicity, and disability status.
The study population consisted of 14,301 veterans and 209,936 nonveterans. Compared with nonveterans, veterans were more likely to have been diagnosed with skin cancer at some point in their lives (7% vs 2.4%; P < .001); had a higher mean age of skin cancer diagnosis (61.1 vs 55.8 years; P < .001); were more likely to have been diagnosed with melanoma (2.8% vs 0.9%; P < .001), and were more likely to have been diagnosed with NMSC (4.4% vs 1.6%; P < .001).
The researchers found that older age, White race, non-Hispanic ethnicity, and veteran status were all associated with higher odds of developing NMSCs, even after adjusting for relevant covariates. Specifically, veterans had 1.23 higher odds of developing NMSC than the general population, while two factors were protective for developing NMSCs: Living in a rural setting (adjusted odds ratio [aOR], 0.78) and receiving supplemental security income or disability income (aOR, 0.69).
In another part of the study, the researchers evaluated demographic and socioeconomic variables associated with developing melanoma among veterans. These included the following: Male (aOR, 1.16), older age (50-64 years: aOR, 6.82; 65-74 years: aOR, 12.55; and 75 years or older: aOR, 16.16), White race (aOR, 9.24), and non-Hispanic ethnicity (aOR, 7.15).
“Veterans may have occupational risks such as sun and chemical exposure, as well as behavioral habits for sun protection, that may contribute to their elevated risk of melanoma and NMSCs,” Ashrafzadeh said. “Therefore, US veterans would benefit from targeted and regular skin cancer screenings, sun protective preventative resources such as hats and sunscreen, and access to medical and surgical care for diagnosis and treatment of skin cancers.”
Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Connecticut, who was asked to comment on the findings, said that a key strength of the study is that it drew from a nationally representative sample. “A limitation is that skin cancer was self-reported rather than based on documented medical histories,” Ko said. “The study confirms that skin cancer risk is higher in older individuals (> 75 as compared to < 50) and in individuals of self-reported white race and non-Hispanic ethnicity,” she added.
Neither the researchers nor Ko reported having relevant disclosures.
A version of this article first appeared on Medscape.com.
of recent national data.
“US veterans are known to have increased risk of cancers and cancer morbidity compared to the general US population,” one of the study authors, Sepideh Ashrafzadeh, MD, a third-year dermatology resident at Massachusetts General Hospital, Boston, told this news organization following the annual meeting of the American Society for Dermatologic Surgery, where the results were presented. “There have been several studies that have shown that US veterans have an increased prevalence of melanoma compared to nonveterans,” she said, noting, however, that no study has investigated the prevalence of nonmelanoma skin cancers (NMSCs), which include basal cell carcinomas and squamous cell carcinomas, compared with the general population.
To address this knowledge gap, the researchers performed a national cross-sectional study of adults aged 18 years or older from the 2019-2023 National Health Interview Surveys to examine the prevalence of melanoma and NMSCs among veterans compared with the general US population. They aggregated and tabulated the data by veteran status, defined as having served at any point in the US armed forces, reserves, or national guard, and by demographic and socioeconomic status variables. Next, they performed multivariate logistic regression for skin cancer risk adjusted for age, sex, race, ethnicity, urbanicity, and disability status.
The study population consisted of 14,301 veterans and 209,936 nonveterans. Compared with nonveterans, veterans were more likely to have been diagnosed with skin cancer at some point in their lives (7% vs 2.4%; P < .001); had a higher mean age of skin cancer diagnosis (61.1 vs 55.8 years; P < .001); were more likely to have been diagnosed with melanoma (2.8% vs 0.9%; P < .001), and were more likely to have been diagnosed with NMSC (4.4% vs 1.6%; P < .001).
The researchers found that older age, White race, non-Hispanic ethnicity, and veteran status were all associated with higher odds of developing NMSCs, even after adjusting for relevant covariates. Specifically, veterans had 1.23 higher odds of developing NMSC than the general population, while two factors were protective for developing NMSCs: Living in a rural setting (adjusted odds ratio [aOR], 0.78) and receiving supplemental security income or disability income (aOR, 0.69).
In another part of the study, the researchers evaluated demographic and socioeconomic variables associated with developing melanoma among veterans. These included the following: Male (aOR, 1.16), older age (50-64 years: aOR, 6.82; 65-74 years: aOR, 12.55; and 75 years or older: aOR, 16.16), White race (aOR, 9.24), and non-Hispanic ethnicity (aOR, 7.15).
“Veterans may have occupational risks such as sun and chemical exposure, as well as behavioral habits for sun protection, that may contribute to their elevated risk of melanoma and NMSCs,” Ashrafzadeh said. “Therefore, US veterans would benefit from targeted and regular skin cancer screenings, sun protective preventative resources such as hats and sunscreen, and access to medical and surgical care for diagnosis and treatment of skin cancers.”
Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Connecticut, who was asked to comment on the findings, said that a key strength of the study is that it drew from a nationally representative sample. “A limitation is that skin cancer was self-reported rather than based on documented medical histories,” Ko said. “The study confirms that skin cancer risk is higher in older individuals (> 75 as compared to < 50) and in individuals of self-reported white race and non-Hispanic ethnicity,” she added.
Neither the researchers nor Ko reported having relevant disclosures.
A version of this article first appeared on Medscape.com.
of recent national data.
“US veterans are known to have increased risk of cancers and cancer morbidity compared to the general US population,” one of the study authors, Sepideh Ashrafzadeh, MD, a third-year dermatology resident at Massachusetts General Hospital, Boston, told this news organization following the annual meeting of the American Society for Dermatologic Surgery, where the results were presented. “There have been several studies that have shown that US veterans have an increased prevalence of melanoma compared to nonveterans,” she said, noting, however, that no study has investigated the prevalence of nonmelanoma skin cancers (NMSCs), which include basal cell carcinomas and squamous cell carcinomas, compared with the general population.
To address this knowledge gap, the researchers performed a national cross-sectional study of adults aged 18 years or older from the 2019-2023 National Health Interview Surveys to examine the prevalence of melanoma and NMSCs among veterans compared with the general US population. They aggregated and tabulated the data by veteran status, defined as having served at any point in the US armed forces, reserves, or national guard, and by demographic and socioeconomic status variables. Next, they performed multivariate logistic regression for skin cancer risk adjusted for age, sex, race, ethnicity, urbanicity, and disability status.
The study population consisted of 14,301 veterans and 209,936 nonveterans. Compared with nonveterans, veterans were more likely to have been diagnosed with skin cancer at some point in their lives (7% vs 2.4%; P < .001); had a higher mean age of skin cancer diagnosis (61.1 vs 55.8 years; P < .001); were more likely to have been diagnosed with melanoma (2.8% vs 0.9%; P < .001), and were more likely to have been diagnosed with NMSC (4.4% vs 1.6%; P < .001).
The researchers found that older age, White race, non-Hispanic ethnicity, and veteran status were all associated with higher odds of developing NMSCs, even after adjusting for relevant covariates. Specifically, veterans had 1.23 higher odds of developing NMSC than the general population, while two factors were protective for developing NMSCs: Living in a rural setting (adjusted odds ratio [aOR], 0.78) and receiving supplemental security income or disability income (aOR, 0.69).
In another part of the study, the researchers evaluated demographic and socioeconomic variables associated with developing melanoma among veterans. These included the following: Male (aOR, 1.16), older age (50-64 years: aOR, 6.82; 65-74 years: aOR, 12.55; and 75 years or older: aOR, 16.16), White race (aOR, 9.24), and non-Hispanic ethnicity (aOR, 7.15).
“Veterans may have occupational risks such as sun and chemical exposure, as well as behavioral habits for sun protection, that may contribute to their elevated risk of melanoma and NMSCs,” Ashrafzadeh said. “Therefore, US veterans would benefit from targeted and regular skin cancer screenings, sun protective preventative resources such as hats and sunscreen, and access to medical and surgical care for diagnosis and treatment of skin cancers.”
Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Connecticut, who was asked to comment on the findings, said that a key strength of the study is that it drew from a nationally representative sample. “A limitation is that skin cancer was self-reported rather than based on documented medical histories,” Ko said. “The study confirms that skin cancer risk is higher in older individuals (> 75 as compared to < 50) and in individuals of self-reported white race and non-Hispanic ethnicity,” she added.
Neither the researchers nor Ko reported having relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM ASDS 2024
‘Small Increase’ in Breast Cancer With Levonorgestrel IUD?
TOPLINE:
The use of a levonorgestrel-releasing intrauterine system (LNG-IUS) is associated with an increased risk for breast cancer. An analysis by Danish researchers found 14 extra cases of breast cancer per 10,000 women using this type of an intrauterine device (IUD) vs women not using hormonal contraceptives.
METHODOLOGY:
- The investigators used nationwide registries in Denmark to identify all women aged 15-49 years who were first-time initiators of any LNG-IUS between 2000 and 2019.
- They matched 78,595 new users of LNG-IUS 1:1 with women with the same birth year who were not taking hormonal contraceptives.
- Participants were followed through 2022 or until a diagnosis of breast cancer or another malignancy, pregnancy, the initiation of postmenopausal hormone therapy, emigration, or death.
- The investigators used a Cox proportional hazards model to examine the association between the continuous use of LNG-IUS and breast cancer. Their analysis adjusted for variables such as the duration of previous hormonal contraception, fertility drugs, parity, age at first delivery, polycystic ovarian syndrome, endometriosis, and education.
TAKEAWAY:
- Compared with the nonuse of hormonal contraceptives, the continuous use of LNG-IUS was associated with a hazard ratio for breast cancer of 1.4 (95% CI, 1.2-1.5).
- The use of a levonorgestrel IUD for 5 years or less was associated with a hazard ratio of 1.3 (95% CI, 1.1-1.5). With 5-10 years of use, the hazard ratio was 1.4 (95% CI, 1.1-1.7). And with 10-15 years of use, the hazard ratio was 1.8 (95% CI, 1.2-2.6). A test for trend was not significant, however, and “risk did not increase with duration of use,” the study authors wrote.
IN PRACTICE:
“Women should be aware that most types of hormonal contraceptive are associated with a small increased risk of breast cancer. This study adds another type of hormonal contraceptive to that list,” Amy Berrington de Gonzalez, DPhil, professor of clinical cancer epidemiology at The Institute of Cancer Research in London, England, said in comments on the research. “That has to be considered with the many benefits from hormonal contraceptives.”
Behaviors such as smoking could have differed between the groups in the study, and it has not been established that LNG-IUS use directly causes an increased risk for breast cancer, said Channa Jayasena, PhD, an endocrinologist at Imperial College London.
“Smoking, alcohol and obesity are much more important risk factors for breast cancer than contraceptive medications,” he said. “My advice for women is that breast cancer risk caused by LNG-IUS is not established but warrants a closer look.”
SOURCE:
Lina Steinrud Mørch, MSc, PhD, with the Danish Cancer Institute in Copenhagen, Denmark, was the corresponding author of the study. The researchers published their findings in JAMA.
LIMITATIONS:
Unmeasured confounding was possible, and the lack of a significant dose-response relationship “could indicate low statistical precision or no causal association,” the researchers noted.
DISCLOSURES:
The study was funded by Sundhedsdonationer.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
The use of a levonorgestrel-releasing intrauterine system (LNG-IUS) is associated with an increased risk for breast cancer. An analysis by Danish researchers found 14 extra cases of breast cancer per 10,000 women using this type of an intrauterine device (IUD) vs women not using hormonal contraceptives.
METHODOLOGY:
- The investigators used nationwide registries in Denmark to identify all women aged 15-49 years who were first-time initiators of any LNG-IUS between 2000 and 2019.
- They matched 78,595 new users of LNG-IUS 1:1 with women with the same birth year who were not taking hormonal contraceptives.
- Participants were followed through 2022 or until a diagnosis of breast cancer or another malignancy, pregnancy, the initiation of postmenopausal hormone therapy, emigration, or death.
- The investigators used a Cox proportional hazards model to examine the association between the continuous use of LNG-IUS and breast cancer. Their analysis adjusted for variables such as the duration of previous hormonal contraception, fertility drugs, parity, age at first delivery, polycystic ovarian syndrome, endometriosis, and education.
TAKEAWAY:
- Compared with the nonuse of hormonal contraceptives, the continuous use of LNG-IUS was associated with a hazard ratio for breast cancer of 1.4 (95% CI, 1.2-1.5).
- The use of a levonorgestrel IUD for 5 years or less was associated with a hazard ratio of 1.3 (95% CI, 1.1-1.5). With 5-10 years of use, the hazard ratio was 1.4 (95% CI, 1.1-1.7). And with 10-15 years of use, the hazard ratio was 1.8 (95% CI, 1.2-2.6). A test for trend was not significant, however, and “risk did not increase with duration of use,” the study authors wrote.
IN PRACTICE:
“Women should be aware that most types of hormonal contraceptive are associated with a small increased risk of breast cancer. This study adds another type of hormonal contraceptive to that list,” Amy Berrington de Gonzalez, DPhil, professor of clinical cancer epidemiology at The Institute of Cancer Research in London, England, said in comments on the research. “That has to be considered with the many benefits from hormonal contraceptives.”
Behaviors such as smoking could have differed between the groups in the study, and it has not been established that LNG-IUS use directly causes an increased risk for breast cancer, said Channa Jayasena, PhD, an endocrinologist at Imperial College London.
“Smoking, alcohol and obesity are much more important risk factors for breast cancer than contraceptive medications,” he said. “My advice for women is that breast cancer risk caused by LNG-IUS is not established but warrants a closer look.”
SOURCE:
Lina Steinrud Mørch, MSc, PhD, with the Danish Cancer Institute in Copenhagen, Denmark, was the corresponding author of the study. The researchers published their findings in JAMA.
LIMITATIONS:
Unmeasured confounding was possible, and the lack of a significant dose-response relationship “could indicate low statistical precision or no causal association,” the researchers noted.
DISCLOSURES:
The study was funded by Sundhedsdonationer.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
The use of a levonorgestrel-releasing intrauterine system (LNG-IUS) is associated with an increased risk for breast cancer. An analysis by Danish researchers found 14 extra cases of breast cancer per 10,000 women using this type of an intrauterine device (IUD) vs women not using hormonal contraceptives.
METHODOLOGY:
- The investigators used nationwide registries in Denmark to identify all women aged 15-49 years who were first-time initiators of any LNG-IUS between 2000 and 2019.
- They matched 78,595 new users of LNG-IUS 1:1 with women with the same birth year who were not taking hormonal contraceptives.
- Participants were followed through 2022 or until a diagnosis of breast cancer or another malignancy, pregnancy, the initiation of postmenopausal hormone therapy, emigration, or death.
- The investigators used a Cox proportional hazards model to examine the association between the continuous use of LNG-IUS and breast cancer. Their analysis adjusted for variables such as the duration of previous hormonal contraception, fertility drugs, parity, age at first delivery, polycystic ovarian syndrome, endometriosis, and education.
TAKEAWAY:
- Compared with the nonuse of hormonal contraceptives, the continuous use of LNG-IUS was associated with a hazard ratio for breast cancer of 1.4 (95% CI, 1.2-1.5).
- The use of a levonorgestrel IUD for 5 years or less was associated with a hazard ratio of 1.3 (95% CI, 1.1-1.5). With 5-10 years of use, the hazard ratio was 1.4 (95% CI, 1.1-1.7). And with 10-15 years of use, the hazard ratio was 1.8 (95% CI, 1.2-2.6). A test for trend was not significant, however, and “risk did not increase with duration of use,” the study authors wrote.
IN PRACTICE:
“Women should be aware that most types of hormonal contraceptive are associated with a small increased risk of breast cancer. This study adds another type of hormonal contraceptive to that list,” Amy Berrington de Gonzalez, DPhil, professor of clinical cancer epidemiology at The Institute of Cancer Research in London, England, said in comments on the research. “That has to be considered with the many benefits from hormonal contraceptives.”
Behaviors such as smoking could have differed between the groups in the study, and it has not been established that LNG-IUS use directly causes an increased risk for breast cancer, said Channa Jayasena, PhD, an endocrinologist at Imperial College London.
“Smoking, alcohol and obesity are much more important risk factors for breast cancer than contraceptive medications,” he said. “My advice for women is that breast cancer risk caused by LNG-IUS is not established but warrants a closer look.”
SOURCE:
Lina Steinrud Mørch, MSc, PhD, with the Danish Cancer Institute in Copenhagen, Denmark, was the corresponding author of the study. The researchers published their findings in JAMA.
LIMITATIONS:
Unmeasured confounding was possible, and the lack of a significant dose-response relationship “could indicate low statistical precision or no causal association,” the researchers noted.
DISCLOSURES:
The study was funded by Sundhedsdonationer.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Cancer’s Other Toll: Long-Term Financial Fallout for Survivors
Overall, patients with cancer tend to face higher rates of debt collection, medical collections, and bankruptcies, as well as lower credit scores, according to two new studies presented at the American College of Surgeons Clinical Congress 2024.
“These are the first studies to provide numerical evidence of financial toxicity among cancer survivors,” Benjamin C. James, MD, with Beth Israel Deaconess Medical Center and Harvard Medical School, both in Boston, Massachusetts, who worked on both studies, said in a statement. “Previous data on this topic largely relies on subjective survey reviews.”
In one study, researchers used the Massachusetts Cancer Registry to identify 99,175 patients diagnosed with cancer between 2010 and 2019 and matched them with 188,875 control individuals without cancer. Researchers then assessed financial toxicity using Experian credit bureau data for participants.
Overall, patients with cancer faced a range of financial challenges that often lasted years following their diagnosis.
Patients were nearly five times more likely to experience bankruptcy and had average credit scores nearly 80 points lower than control individuals without cancer. The drop in credit scores was more pronounced for survivors of bladder, liver, lung, and colorectal cancer (CRC) and persisted for up to 9.5 years.
For certain cancer types, in particular, “we are looking years after a diagnosis, and we see that the credit score goes down and it never comes back up,” James said.
The other study, which used a sample of 7227 patients with CRC from Massachusetts, identified several factors that correlated with lower credit scores.
Compared with patients who only had surgery, peers who underwent radiation only experienced a 62-point drop in their credit score after their diagnosis, while those who had chemotherapy alone had just over a 14-point drop in their credit score. Among patients who had combination treatments, those who underwent both surgery and radiation experienced a nearly 16-point drop in their credit score and those who had surgery and chemoradiation actually experienced a 2.59 bump, compared with those who had surgery alone.
Financial toxicity was worse for patients younger than 62 years, those identifying as Black or Hispanic individuals, unmarried individuals, those with an annual income below $52,000, and those living in deprived areas.
The studies add to findings from the 2015 North American Thyroid Cancer Survivorship Study, which reported that 50% of thyroid cancer survivors encountered financial toxicity because of their diagnosis.
James said the persistent financial strain of cancer care, even in a state like Massachusetts, which mandates universal healthcare, underscores the need for “broader policy changes and reforms, including reconsidering debt collection practices.”
“Financial security should be a priority in cancer care,” he added.
The studies had no specific funding. The authors have disclosed no relevant conflict of interest.
A version of this article first appeared on Medscape.com.
Overall, patients with cancer tend to face higher rates of debt collection, medical collections, and bankruptcies, as well as lower credit scores, according to two new studies presented at the American College of Surgeons Clinical Congress 2024.
“These are the first studies to provide numerical evidence of financial toxicity among cancer survivors,” Benjamin C. James, MD, with Beth Israel Deaconess Medical Center and Harvard Medical School, both in Boston, Massachusetts, who worked on both studies, said in a statement. “Previous data on this topic largely relies on subjective survey reviews.”
In one study, researchers used the Massachusetts Cancer Registry to identify 99,175 patients diagnosed with cancer between 2010 and 2019 and matched them with 188,875 control individuals without cancer. Researchers then assessed financial toxicity using Experian credit bureau data for participants.
Overall, patients with cancer faced a range of financial challenges that often lasted years following their diagnosis.
Patients were nearly five times more likely to experience bankruptcy and had average credit scores nearly 80 points lower than control individuals without cancer. The drop in credit scores was more pronounced for survivors of bladder, liver, lung, and colorectal cancer (CRC) and persisted for up to 9.5 years.
For certain cancer types, in particular, “we are looking years after a diagnosis, and we see that the credit score goes down and it never comes back up,” James said.
The other study, which used a sample of 7227 patients with CRC from Massachusetts, identified several factors that correlated with lower credit scores.
Compared with patients who only had surgery, peers who underwent radiation only experienced a 62-point drop in their credit score after their diagnosis, while those who had chemotherapy alone had just over a 14-point drop in their credit score. Among patients who had combination treatments, those who underwent both surgery and radiation experienced a nearly 16-point drop in their credit score and those who had surgery and chemoradiation actually experienced a 2.59 bump, compared with those who had surgery alone.
Financial toxicity was worse for patients younger than 62 years, those identifying as Black or Hispanic individuals, unmarried individuals, those with an annual income below $52,000, and those living in deprived areas.
The studies add to findings from the 2015 North American Thyroid Cancer Survivorship Study, which reported that 50% of thyroid cancer survivors encountered financial toxicity because of their diagnosis.
James said the persistent financial strain of cancer care, even in a state like Massachusetts, which mandates universal healthcare, underscores the need for “broader policy changes and reforms, including reconsidering debt collection practices.”
“Financial security should be a priority in cancer care,” he added.
The studies had no specific funding. The authors have disclosed no relevant conflict of interest.
A version of this article first appeared on Medscape.com.
Overall, patients with cancer tend to face higher rates of debt collection, medical collections, and bankruptcies, as well as lower credit scores, according to two new studies presented at the American College of Surgeons Clinical Congress 2024.
“These are the first studies to provide numerical evidence of financial toxicity among cancer survivors,” Benjamin C. James, MD, with Beth Israel Deaconess Medical Center and Harvard Medical School, both in Boston, Massachusetts, who worked on both studies, said in a statement. “Previous data on this topic largely relies on subjective survey reviews.”
In one study, researchers used the Massachusetts Cancer Registry to identify 99,175 patients diagnosed with cancer between 2010 and 2019 and matched them with 188,875 control individuals without cancer. Researchers then assessed financial toxicity using Experian credit bureau data for participants.
Overall, patients with cancer faced a range of financial challenges that often lasted years following their diagnosis.
Patients were nearly five times more likely to experience bankruptcy and had average credit scores nearly 80 points lower than control individuals without cancer. The drop in credit scores was more pronounced for survivors of bladder, liver, lung, and colorectal cancer (CRC) and persisted for up to 9.5 years.
For certain cancer types, in particular, “we are looking years after a diagnosis, and we see that the credit score goes down and it never comes back up,” James said.
The other study, which used a sample of 7227 patients with CRC from Massachusetts, identified several factors that correlated with lower credit scores.
Compared with patients who only had surgery, peers who underwent radiation only experienced a 62-point drop in their credit score after their diagnosis, while those who had chemotherapy alone had just over a 14-point drop in their credit score. Among patients who had combination treatments, those who underwent both surgery and radiation experienced a nearly 16-point drop in their credit score and those who had surgery and chemoradiation actually experienced a 2.59 bump, compared with those who had surgery alone.
Financial toxicity was worse for patients younger than 62 years, those identifying as Black or Hispanic individuals, unmarried individuals, those with an annual income below $52,000, and those living in deprived areas.
The studies add to findings from the 2015 North American Thyroid Cancer Survivorship Study, which reported that 50% of thyroid cancer survivors encountered financial toxicity because of their diagnosis.
James said the persistent financial strain of cancer care, even in a state like Massachusetts, which mandates universal healthcare, underscores the need for “broader policy changes and reforms, including reconsidering debt collection practices.”
“Financial security should be a priority in cancer care,” he added.
The studies had no specific funding. The authors have disclosed no relevant conflict of interest.
A version of this article first appeared on Medscape.com.
FROM ACSCS 2024
Mortality Rates From Early-Onset CRC Have Risen Considerably Over Last 2 Decades
PHILADELPHIA — according to a new analysis of the two largest US mortality databases.
Data from the Centers for Disease Control and Prevention’s National Center of Health Statistics (NCHS) and the Surveillance, Epidemiology, and End Results (SEER) databases provide yet more evidence of the increasing prevalence of EO-CRC, which is defined as a diagnosis of CRC in patients younger than age 50 years.
Furthermore, the researchers reported that increased mortality occurred across all patients included in the study (aged 20-54) regardless of tumor stage at diagnosis.
These findings “prompt tailoring further efforts toward raising awareness of colorectal cancer symptoms and keeping a low clinical suspicion in younger patients presenting with anemia, gastrointestinal bleeding, or change in bowel habits,” Yazan Abboud, MD, internal medicine PGY-3, assistant chief resident, and chair of resident research at Rutgers New Jersey Medical School, Newark, said in an interview.
Abboud presented the findings at the American College of Gastroenterology (ACG) 2024 Annual Scientific Meeting.
Analyzing NCHS and SEER
Rising rates of EO-CRC had prompted US medical societies to recommend reducing the screening age to 45 years. The US Preventive Services Task Force officially lowered it to this age in 2021. This shift is supported by real-world evidence, which shows that earlier screening leads to a significantly reduced risk for colorectal cancer. However, because colorectal cancer cases are decreasing overall in older adults, there is considerable interest in discovering why young adults are experiencing a paradoxical uptick in EO-CRC, and what impact this is having on associated mortality.
Abboud and colleagues collected age-adjusted mortality rates for EO-CRC between 2000 and 2022 from the NCHS database. In addition, stage-specific incidence-based mortality rates between 2004-2020 were obtained from the SEER 22 database. The NCHS database covers approximately 100% of the US population, whereas the SEER 22 database, which is included within the NCHS, covers 42%.
The researchers divided patients into two cohorts based on age (20-44 years and 45-54 years) and tumor stage at diagnosis (early stage and late stage), and compared the annual percentage change (APC) and the average APC between the two groups. They also assessed trends for the entire cohort of patients aged 20-54 years.
In the NCHS database, there were 147,026 deaths in total across all ages studied resulting from EO-CRC, of which 27% (39,746) occurred in those 20-44 years of age. Although associated mortality rates decreased between 2000-2005 in all ages studied (APC, –1.56), they increased from 2005-2022 (APC, 0.87).
In the cohort aged 45-54 years, mortality decreased between 2000-2005 and increased thereafter, whereas in the cohort aged 20-44 years mortality increased steadily for the entire follow-up duration of 2000 to 2022 (APC, 0.93). A comparison of the age cohorts confirmed that those aged 20-44 years had a greater increase in mortality (average APC, 0.85; P < .001).
In the SEER 22 database, there were 4652 deaths in those with early-stage tumors across all age groups studied (average APC, 12.17). Mortality increased in patients aged 45-54 years (average APC, 11.52) with early-stage tumors, but there were insufficient numbers in those aged 20-44 years to determine this outcome.
There were 42,120 deaths in those with late-stage tumors across all age groups (average APC, 10.05) in the SEER 22 database. And increased mortality was observed in those with late-stage tumors in both age cohorts: 45-54 years (average APC, 9.58) and 20-44 years (average APC, 11.06).
“When evaluating the SEER database and stratifying the tumors by stage at diagnosis, we demonstrated increasing mortality of early-onset colorectal cancer in both early- and late-stage tumors on average over the study period,” Abboud said.
Identifying At-Risk Patients
In a comment, David A. Johnson, MD, professor of medicine and chief of gastroenterology at Eastern Virginia School of Medicine in Norfolk, said the findings speak to the need for evidence-based means of identifying younger individuals at a higher risk of EO-CRC.
“I suspect many of younger patients with CRC had their cancer detected when it was more advanced due to delayed presentation and diagnostic testing,” said Johnson, who was not involved in the study.
But it would be interesting to evaluate if the cancers in the cohort aged 20-44 years were more aggressive biologically or if these patients were dismissive of early signs or symptoms, he said.
Younger patients may dismiss “alarm” features that indicate CRC testing, said Johnson. “In particular, overt bleeding and iron deficiency need a focused evaluation in these younger cohorts.”
“Future research is needed to investigate the role of neoadjuvant chemotherapy in younger patients with early-stage colorectal cancer and evaluate patients’ outcomes,” Abboud added.
The study had no specific funding. Abboud reported no relevant financial relationships. Johnson reported serving as an adviser to ISOTHRIVE. He is also on the Medscape Gastroenterology editorial board.
A version of this article first appeared on Medscape.com.
PHILADELPHIA — according to a new analysis of the two largest US mortality databases.
Data from the Centers for Disease Control and Prevention’s National Center of Health Statistics (NCHS) and the Surveillance, Epidemiology, and End Results (SEER) databases provide yet more evidence of the increasing prevalence of EO-CRC, which is defined as a diagnosis of CRC in patients younger than age 50 years.
Furthermore, the researchers reported that increased mortality occurred across all patients included in the study (aged 20-54) regardless of tumor stage at diagnosis.
These findings “prompt tailoring further efforts toward raising awareness of colorectal cancer symptoms and keeping a low clinical suspicion in younger patients presenting with anemia, gastrointestinal bleeding, or change in bowel habits,” Yazan Abboud, MD, internal medicine PGY-3, assistant chief resident, and chair of resident research at Rutgers New Jersey Medical School, Newark, said in an interview.
Abboud presented the findings at the American College of Gastroenterology (ACG) 2024 Annual Scientific Meeting.
Analyzing NCHS and SEER
Rising rates of EO-CRC had prompted US medical societies to recommend reducing the screening age to 45 years. The US Preventive Services Task Force officially lowered it to this age in 2021. This shift is supported by real-world evidence, which shows that earlier screening leads to a significantly reduced risk for colorectal cancer. However, because colorectal cancer cases are decreasing overall in older adults, there is considerable interest in discovering why young adults are experiencing a paradoxical uptick in EO-CRC, and what impact this is having on associated mortality.
Abboud and colleagues collected age-adjusted mortality rates for EO-CRC between 2000 and 2022 from the NCHS database. In addition, stage-specific incidence-based mortality rates between 2004-2020 were obtained from the SEER 22 database. The NCHS database covers approximately 100% of the US population, whereas the SEER 22 database, which is included within the NCHS, covers 42%.
The researchers divided patients into two cohorts based on age (20-44 years and 45-54 years) and tumor stage at diagnosis (early stage and late stage), and compared the annual percentage change (APC) and the average APC between the two groups. They also assessed trends for the entire cohort of patients aged 20-54 years.
In the NCHS database, there were 147,026 deaths in total across all ages studied resulting from EO-CRC, of which 27% (39,746) occurred in those 20-44 years of age. Although associated mortality rates decreased between 2000-2005 in all ages studied (APC, –1.56), they increased from 2005-2022 (APC, 0.87).
In the cohort aged 45-54 years, mortality decreased between 2000-2005 and increased thereafter, whereas in the cohort aged 20-44 years mortality increased steadily for the entire follow-up duration of 2000 to 2022 (APC, 0.93). A comparison of the age cohorts confirmed that those aged 20-44 years had a greater increase in mortality (average APC, 0.85; P < .001).
In the SEER 22 database, there were 4652 deaths in those with early-stage tumors across all age groups studied (average APC, 12.17). Mortality increased in patients aged 45-54 years (average APC, 11.52) with early-stage tumors, but there were insufficient numbers in those aged 20-44 years to determine this outcome.
There were 42,120 deaths in those with late-stage tumors across all age groups (average APC, 10.05) in the SEER 22 database. And increased mortality was observed in those with late-stage tumors in both age cohorts: 45-54 years (average APC, 9.58) and 20-44 years (average APC, 11.06).
“When evaluating the SEER database and stratifying the tumors by stage at diagnosis, we demonstrated increasing mortality of early-onset colorectal cancer in both early- and late-stage tumors on average over the study period,” Abboud said.
Identifying At-Risk Patients
In a comment, David A. Johnson, MD, professor of medicine and chief of gastroenterology at Eastern Virginia School of Medicine in Norfolk, said the findings speak to the need for evidence-based means of identifying younger individuals at a higher risk of EO-CRC.
“I suspect many of younger patients with CRC had their cancer detected when it was more advanced due to delayed presentation and diagnostic testing,” said Johnson, who was not involved in the study.
But it would be interesting to evaluate if the cancers in the cohort aged 20-44 years were more aggressive biologically or if these patients were dismissive of early signs or symptoms, he said.
Younger patients may dismiss “alarm” features that indicate CRC testing, said Johnson. “In particular, overt bleeding and iron deficiency need a focused evaluation in these younger cohorts.”
“Future research is needed to investigate the role of neoadjuvant chemotherapy in younger patients with early-stage colorectal cancer and evaluate patients’ outcomes,” Abboud added.
The study had no specific funding. Abboud reported no relevant financial relationships. Johnson reported serving as an adviser to ISOTHRIVE. He is also on the Medscape Gastroenterology editorial board.
A version of this article first appeared on Medscape.com.
PHILADELPHIA — according to a new analysis of the two largest US mortality databases.
Data from the Centers for Disease Control and Prevention’s National Center of Health Statistics (NCHS) and the Surveillance, Epidemiology, and End Results (SEER) databases provide yet more evidence of the increasing prevalence of EO-CRC, which is defined as a diagnosis of CRC in patients younger than age 50 years.
Furthermore, the researchers reported that increased mortality occurred across all patients included in the study (aged 20-54) regardless of tumor stage at diagnosis.
These findings “prompt tailoring further efforts toward raising awareness of colorectal cancer symptoms and keeping a low clinical suspicion in younger patients presenting with anemia, gastrointestinal bleeding, or change in bowel habits,” Yazan Abboud, MD, internal medicine PGY-3, assistant chief resident, and chair of resident research at Rutgers New Jersey Medical School, Newark, said in an interview.
Abboud presented the findings at the American College of Gastroenterology (ACG) 2024 Annual Scientific Meeting.
Analyzing NCHS and SEER
Rising rates of EO-CRC had prompted US medical societies to recommend reducing the screening age to 45 years. The US Preventive Services Task Force officially lowered it to this age in 2021. This shift is supported by real-world evidence, which shows that earlier screening leads to a significantly reduced risk for colorectal cancer. However, because colorectal cancer cases are decreasing overall in older adults, there is considerable interest in discovering why young adults are experiencing a paradoxical uptick in EO-CRC, and what impact this is having on associated mortality.
Abboud and colleagues collected age-adjusted mortality rates for EO-CRC between 2000 and 2022 from the NCHS database. In addition, stage-specific incidence-based mortality rates between 2004-2020 were obtained from the SEER 22 database. The NCHS database covers approximately 100% of the US population, whereas the SEER 22 database, which is included within the NCHS, covers 42%.
The researchers divided patients into two cohorts based on age (20-44 years and 45-54 years) and tumor stage at diagnosis (early stage and late stage), and compared the annual percentage change (APC) and the average APC between the two groups. They also assessed trends for the entire cohort of patients aged 20-54 years.
In the NCHS database, there were 147,026 deaths in total across all ages studied resulting from EO-CRC, of which 27% (39,746) occurred in those 20-44 years of age. Although associated mortality rates decreased between 2000-2005 in all ages studied (APC, –1.56), they increased from 2005-2022 (APC, 0.87).
In the cohort aged 45-54 years, mortality decreased between 2000-2005 and increased thereafter, whereas in the cohort aged 20-44 years mortality increased steadily for the entire follow-up duration of 2000 to 2022 (APC, 0.93). A comparison of the age cohorts confirmed that those aged 20-44 years had a greater increase in mortality (average APC, 0.85; P < .001).
In the SEER 22 database, there were 4652 deaths in those with early-stage tumors across all age groups studied (average APC, 12.17). Mortality increased in patients aged 45-54 years (average APC, 11.52) with early-stage tumors, but there were insufficient numbers in those aged 20-44 years to determine this outcome.
There were 42,120 deaths in those with late-stage tumors across all age groups (average APC, 10.05) in the SEER 22 database. And increased mortality was observed in those with late-stage tumors in both age cohorts: 45-54 years (average APC, 9.58) and 20-44 years (average APC, 11.06).
“When evaluating the SEER database and stratifying the tumors by stage at diagnosis, we demonstrated increasing mortality of early-onset colorectal cancer in both early- and late-stage tumors on average over the study period,” Abboud said.
Identifying At-Risk Patients
In a comment, David A. Johnson, MD, professor of medicine and chief of gastroenterology at Eastern Virginia School of Medicine in Norfolk, said the findings speak to the need for evidence-based means of identifying younger individuals at a higher risk of EO-CRC.
“I suspect many of younger patients with CRC had their cancer detected when it was more advanced due to delayed presentation and diagnostic testing,” said Johnson, who was not involved in the study.
But it would be interesting to evaluate if the cancers in the cohort aged 20-44 years were more aggressive biologically or if these patients were dismissive of early signs or symptoms, he said.
Younger patients may dismiss “alarm” features that indicate CRC testing, said Johnson. “In particular, overt bleeding and iron deficiency need a focused evaluation in these younger cohorts.”
“Future research is needed to investigate the role of neoadjuvant chemotherapy in younger patients with early-stage colorectal cancer and evaluate patients’ outcomes,” Abboud added.
The study had no specific funding. Abboud reported no relevant financial relationships. Johnson reported serving as an adviser to ISOTHRIVE. He is also on the Medscape Gastroenterology editorial board.
A version of this article first appeared on Medscape.com.
FROM ACG 2024
Asteraceae Dermatitis: Everyday Plants With Allergenic Potential
The Asteraceae (formerly Compositae) family of plants is derived from the ancient Greek word aster, meaning “star,” referring to the starlike arrangement of flower petals around a central disc known as a capitulum. What initially appears as a single flower is actually a composite of several smaller flowers, hence the former name Compositae.1 Well-known members of the Asteraceae family include ornamental annuals (eg, sunflowers, marigolds, cosmos), herbaceous perennials (eg, chrysanthemums, dandelions), vegetables (eg, lettuce, chicory, artichokes), herbs (eg, chamomile, tarragon), and weeds (eg, ragweed, horseweed, capeweed)(Figure 1).2

There are more than 25,000 species of Asteraceae plants that thrive in a wide range of climates worldwide. Cases of Asteraceae-induced skin reactions have been reported in North America, Europe, Asia, and Australia.3 Members of the Asteraceae family are ubiquitous in gardens, along roadsides, and in the wilderness. Occupational exposure commonly affects gardeners, florists, farmers, and forestry workers through either direct contact with plants or via airborne pollen. Furthermore, plants of the Asteraceae family are used in various products, including pediculicides (eg, insect repellents), cosmetics (eg, eye creams, body washes), and food products (eg, cooking oils, sweetening agents, coffee substitutes, herbal teas).4-6 These plants have substantial allergic potential, resulting in numerous cutaneous reactions.
Allergic Potential
Asteraceae plants can elicit both immediate and delayed hypersensitivity reactions (HSRs); for instance, exposure to ragweed pollen may cause an IgE-mediated type 1 HSR manifesting as allergic rhinitis or a type IV HSR manifesting as airborne allergic contact dermatitis.7,8 The main contact allergens present in Asteraceae plants are sesquiterpene lactones, which are found in the leaves, stems, flowers, and pollen.9-11 Sesquiterpene lactones consist of an α-methyl group attached to a lactone ring combined with a sesquiterpene.12 Patch testing can be used to diagnose Asteraceae allergy; however, the results are not consistently reliable because there is no perfect screening allergen. Patch test preparations commonly used to detect Asteraceae allergy include Compositae mix (consisting of Anthemis nobilis extract, Chamomilla recutita extract, Achillea millefolium extract, Tanacetum vulgare extract, Arnica montana extract, and parthenolide) and sesquiterpene lactone mix (consisting of alantolactone, dehydrocostus lactone, and costunolide). In North America, the prevalence of positive patch tests to Compositae mix and sesquiterpene lactone mix is approximately 2% and 0.5%, respectively.13 When patch testing is performed, both Compositae mix and sesquiterpene lactone mix should be utilized to minimize the risk of missing Asteraceae allergy, as sesquiterpene lactone mix alone does not detect all Compositae-sensitized patients. Additionally, it may be necessary to test supplemental Asteraceae allergens, including preparations from specific plants to which the patient has been exposed. Exposure to Asteraceae-containing cosmetic products may lead to dermatitis, though this is highly dependent on the particular plant species involved. For instance, the prevalence of sensitization is high in arnica (tincture) and elecampane but low with more commonly used species such as German chamomile.14
Cutaneous Manifestations
Asteraceae dermatitis, which also is known as Australian bush dermatitis, weed dermatitis, and chrysanthemum dermatitis,2 can manifest on any area of the body that directly contacts the plant or is exposed to the pollen. Asteraceae dermatitis historically was reported in older adults with a recent history of plant exposure.6,15 However, recent data have shown a female preponderance and a younger mean age of onset (46–49 years).16
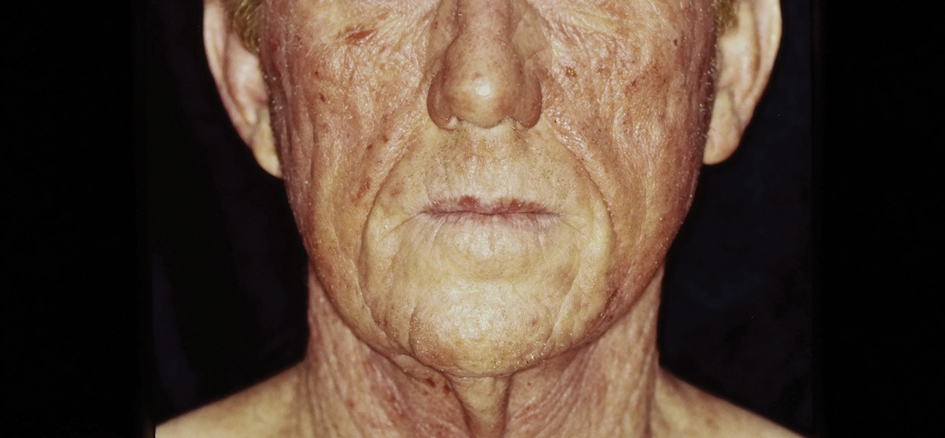
There are multiple distinct clinical manifestations of Asteraceae dermatitis. The most common cutaneous finding is localized vesicular or eczematous patches on the hands or wrists. Other variations include eczematous rashes on the exposed skin of the hands, arms, face, and neck; generalized eczema; and isolated facial eczema.16,17 These variations can be attributed to contact dermatitis caused by airborne pollen, which may mimic photodermatitis. However, airborne Asteraceae dermatitis can be distinguished clinically from photodermatitis by the involvement of sun-protected areas such as the skinfolds of the eyelids, retroauricular sulci, and nasolabial folds (Figure 2).2,9 In rare cases, systemic allergic contact dermatitis can occur if the Asteraceae allergen is ingested.2,18
Other diagnostic clues include dermatitis that flares during the summer, at the peak of the growing season, with remission in the cooler months. Potential risk factors include a childhood history of atopic dermatitis and allergic rhinitis.16 With prolonged exposure, patients may develop chronic actinic dermatitis, an immunologically mediated photodermatosis characterized by lichenified and pruritic eczematous plaques located predominantly on sun-exposed areas with notable sparing of the skin folds.19 The association between Asteraceae dermatitis and chronic actinic dermatitis is highly variable, with some studies reporting a 25% correlation and others finding a stronger association of up to 80%.2,15,20 Asteraceae allergy appears to be a relatively uncommon cause of photoallergy in North America. In one recent study, 16% (3/19) of patients with chronic actinic dermatitis had positive patch or photopatch tests to sesquiterpene lactone mix, but in another large study of photopatch testing it was reported to be a rare photoallergen.21,22
Parthenium dermatitis is an allergic contact dermatitis caused by exposure to Parthenium hysterophorus, a weed of the Asteraceae family that is responsible for 30% of cases of contact dermatitis in India.23,24 Unlike the more classic manifestation of Asteraceae dermatitis, which primarily affects the upper extremities in cases from North America and Europe, Parthenium dermatitis typically occurs in an airborne pattern distribution.24
Management
While complete avoidance of Asteraceae plants is ideal, it often is unrealistic due to their abundance in nature. Therefore, minimizing exposure to the causative plants is recommended. Primary preventive measures such as wearing protective gloves and clothing and applying bentonite clay prior to exposure should be taken when working outdoors. Promptly showering after contact with plants also can reduce the risk for Asteraceae dermatitis.
Symptomatic treatment is appropriate for mild cases and includes topical corticosteroids and calcineurin inhibitors. For severe cases, systemic corticosteroids may be needed for acute flares, with azathioprine, mycophenolate, cyclosporine, or methotrexate available for recalcitrant disease. Verma et al25 found that treatment with azathioprine for 6 months resulted in greater than 60% clearance in all 12 patients, with a majority achieving 80% to 100% clearance. Methotrexate has been used at doses of 15 mg once weekly.26 Narrowband UVB and psoralen plus UVA have been effective in extensive cases; however, care should be exercised in patients with photosensitive dermatitis, who instead should practice strict photoprotection.27-29 Lakshmi et al30 reported the use of cyclosporine during the acute phase of Asteraceae dermatitis at a dose of 2.5 mg/kg daily for 4 to 8 weeks. There have been several case reports of dupilumab treating allergic contact dermatitis; however, there have been 3 cases of patients with atopic dermatitis developing Asteraceae dermatitis while taking dupilumab.31,32 Recently, oral Janus kinase inhibitors have shown success in treating refractory cases of airborne Asteraceae dermatitis.33,34 Further research is needed to determine the safety and efficacy of dupilumab and Janus kinase inhibitors for treatment of Asteraceae dermatitis.
Final Thoughts
The Asteraceae plant family is vast and diverse, with more than 200 species reported to cause allergic contact dermatitis.12 Common modes of contact include gardening, occupational exposure, airborne pollen, and use of pediculicides and cosmetics that contain components of Asteraceae plants. Educating patients on how to minimize contact with Asteraceae plants is the most effective management strategy; topical agents and oral immunosuppressives can be used for symptomatic treatment.
- Morhardt S, Morhardt E. California Desert Flowers: An Introduction to Families, Genera, and Species. University of California Press; 2004.
- Gordon LA. Compositae dermatitis. Australas J Dermatol. 1999;40:123-130. doi:10.1046/j.1440-0960.1999.00341.x
- Denisow-Pietrzyk M, Pietrzyk Ł, Denisow B. Asteraceae species as potential environmental factors of allergy. Environ Sci Pollut Res Int. 2019;26:6290-6300. doi:10.1007/s11356-019-04146-w
- Paulsen E, Chistensen LP, Andersen KE. Cosmetics and herbal remedies with Compositae plant extracts—are they tolerated by Compositae-allergic patients? Contact Dermatitis. 2008;58:15-23. doi:10.1111/j.1600-0536.2007.01250.x
- Burry JN, Reid JG, Kirk J. Australian bush dermatitis. Contact Dermatitis. 1975;1:263-264. doi:10.1111/j.1600-0536.1975.tb05422.x
- Punchihewa N, Palmer A, Nixon R. Allergic contact dermatitis to Compositae: an Australian case series. Contact Dermatitis. 2022;87:356-362. doi:10.1111/cod.14162
- Chen KW, Marusciac L, Tamas PT, et al. Ragweed pollen allergy: burden, characteristics, and management of an imported allergen source in Europe. Int Arch Allergy Immunol. 2018;176:163-180. doi:10.1159/000487997
- Schloemer JA, Zirwas MJ, Burkhart CG. Airborne contact dermatitis: common causes in the USA. Int J Dermatol. 2015;54:271-274. doi:10.1111/ijd.12692
- Arlette J, Mitchell JC. Compositae dermatitis. current aspects. Contact Dermatitis. 1981;7:129-136. doi:10.1111/j.1600-0536.1981.tb04584.x
- Mitchell JC, Dupuis G. Allergic contact dermatitis from sesquiterpenoids of the Compositae family of plants. Br J Dermatol. 1971;84:139-150. doi:10.1111/j.1365-2133.1971.tb06857.x
- Salapovic H, Geier J, Reznicek G. Quantification of Sesquiterpene lactones in Asteraceae plant extracts: evaluation of their allergenic potential. Sci Pharm. 2013;81:807-818. doi:10.3797/scipharm.1306-17
- Paulsen E. Compositae dermatitis: a survey. Contact Dermatitis. 1992;26:76-86. doi:10.1111/j.1600-0536.1992.tb00888.x. Published correction appears in Contact Dermatitis. 1992;27:208.
- DeKoven JG, Silverberg JI, Warshaw EM, et al. North American Contact Dermatitis Group patch test results: 2017-2018. Dermatitis. 2021;32:111-123. doi:10.1097/DER.0000000000000729
- Paulsen E. Contact sensitization from Compositae-containing herbal remedies and cosmetics. Contact Dermatitis. 2002;47:189-198. doi:10.1034/j.1600-0536.2002.470401.x
- Frain-Bell W, Johnson BE. Contact allergic sensitivity to plants and the photosensitivity dermatitis and actinic reticuloid syndrome. Br J Dermatol. 1979;101:503-512.
- Paulsen E, Andersen KE. Clinical patterns of Compositae dermatitis in Danish monosensitized patients. Contact Dermatitis. 2018;78:185-193. doi:10.1111/cod.12916
- Jovanovic´ M, Poljacki M. Compositae dermatitis. Med Pregl. 2003;56:43-49. doi:10.2298/mpns0302043j
- Krook G. Occupational dermatitis from Lactuca sativa (lettuce) and Cichorium (endive). simultaneous occurrence of immediate and delayed allergy as a cause of contact dermatitis. Contact Dermatitis. 1977;3:27-36. doi:10.1111/j.1600-0536.1977.tb03583.x
- Paek SY, Lim HW. Chronic actinic dermatitis. Dermatol Clin. 2014;32:355-361, viii-ix. doi:10.1016/j.det.2014.03.007
- du P Menagé H, Hawk JL, White IR. Sesquiterpene lactone mix contact sensitivity and its relationship to chronic actinic dermatitis: a follow-up study. Contact Dermatitis. 1998;39:119-122. doi:10.1111/j.1600-0536.1998.tb05859.x
- Wang CX, Belsito DV. Chronic actinic dermatitis revisited. Dermatitis. 2020;31:68-74. doi:10.1097/DER.0000000000000531
- DeLeo VA, Adler BL, Warshaw EM, et al. Photopatch test results of the North American contact dermatitis group, 1999-2009. Photodermatol Photoimmunol Photomed. 2022;38:288-291. doi:10.1111/phpp.12742
- McGovern TW, LaWarre S. Botanical briefs: the scourge of India—Parthenium hysterophorus L. Cutis. 2001;67:27-34. Published correction appears in Cutis. 2001;67:154.
- Sharma VK, Verma P, Maharaja K. Parthenium dermatitis. Photochem Photobiol Sci. 2013;12:85-94. doi:10.1039/c2pp25186h
- Verma KK, Bansal A, Sethuraman G. Parthenium dermatitis treated with azathioprine weekly pulse doses. Indian J Dermatol Venereol Leprol. 2006;72:24-27. doi:10.4103/0378-6323.19713
- Sharma VK, Bhat R, Sethuraman G, et al. Treatment of Parthenium dermatitis with methotrexate. Contact Dermatitis. 2007;57:118-119. doi:10.1111/j.1600-0536.2006.00950.x
- Burke DA, Corey G, Storrs FJ. Psoralen plus UVA protocol for Compositae photosensitivity. Am J Contact Dermat. 1996;7:171-176.
- Lovell CR. Allergic contact dermatitis due to plants. In: Plants and the Skin. Blackwell Scientific Publications; 1993:96-254.
- Dogra S, Parsad D, Handa S. Narrowband ultraviolet B in airborne contact dermatitis: a ray of hope! Br J Dermatol. 2004;150:373-374. doi:10.1111/j.1365-2133.2004.05724.x
- Lakshmi C, Srinivas CR, Jayaraman A. Ciclosporin in Parthenium dermatitis—a report of 2 cases. Contact Dermatitis. 2008;59:245-248. doi:10.1111/j.1600-0536.2007.01208.x
- Hendricks AJ, Yosipovitch G, Shi VY. Dupilumab use in dermatologic conditions beyond atopic dermatitis—a systematic review. J Dermatolog Treat. 2021;32:19-28. doi:10.1080/09546634.2019.1689227
- Napolitano M, Fabbrocini G, Patruno C. Allergic contact dermatitis to Compositae: a possible cause of dupilumab-associated facial and neck dermatitis in atopic dermatitis patients? Contact Dermatitis. 2021;85:473-474. doi:10.1111/cod.13898
- Muddebihal A, Sardana K, Sinha S, et al. Tofacitinib in refractory Parthenium-induced airborne allergic contact dermatitis. Contact Dermatitis. 2023;88:150-152. doi:10.1111/cod.14234
- Baltazar D, Shinamoto SR, Hamann CP, et al. Occupational airborne allergic contact dermatitis to invasive Compositae species treated with abrocitinib: a case report. Contact Dermatitis. 2022;87:542-544. doi:10.1111/cod.14204
The Asteraceae (formerly Compositae) family of plants is derived from the ancient Greek word aster, meaning “star,” referring to the starlike arrangement of flower petals around a central disc known as a capitulum. What initially appears as a single flower is actually a composite of several smaller flowers, hence the former name Compositae.1 Well-known members of the Asteraceae family include ornamental annuals (eg, sunflowers, marigolds, cosmos), herbaceous perennials (eg, chrysanthemums, dandelions), vegetables (eg, lettuce, chicory, artichokes), herbs (eg, chamomile, tarragon), and weeds (eg, ragweed, horseweed, capeweed)(Figure 1).2

There are more than 25,000 species of Asteraceae plants that thrive in a wide range of climates worldwide. Cases of Asteraceae-induced skin reactions have been reported in North America, Europe, Asia, and Australia.3 Members of the Asteraceae family are ubiquitous in gardens, along roadsides, and in the wilderness. Occupational exposure commonly affects gardeners, florists, farmers, and forestry workers through either direct contact with plants or via airborne pollen. Furthermore, plants of the Asteraceae family are used in various products, including pediculicides (eg, insect repellents), cosmetics (eg, eye creams, body washes), and food products (eg, cooking oils, sweetening agents, coffee substitutes, herbal teas).4-6 These plants have substantial allergic potential, resulting in numerous cutaneous reactions.
Allergic Potential
Asteraceae plants can elicit both immediate and delayed hypersensitivity reactions (HSRs); for instance, exposure to ragweed pollen may cause an IgE-mediated type 1 HSR manifesting as allergic rhinitis or a type IV HSR manifesting as airborne allergic contact dermatitis.7,8 The main contact allergens present in Asteraceae plants are sesquiterpene lactones, which are found in the leaves, stems, flowers, and pollen.9-11 Sesquiterpene lactones consist of an α-methyl group attached to a lactone ring combined with a sesquiterpene.12 Patch testing can be used to diagnose Asteraceae allergy; however, the results are not consistently reliable because there is no perfect screening allergen. Patch test preparations commonly used to detect Asteraceae allergy include Compositae mix (consisting of Anthemis nobilis extract, Chamomilla recutita extract, Achillea millefolium extract, Tanacetum vulgare extract, Arnica montana extract, and parthenolide) and sesquiterpene lactone mix (consisting of alantolactone, dehydrocostus lactone, and costunolide). In North America, the prevalence of positive patch tests to Compositae mix and sesquiterpene lactone mix is approximately 2% and 0.5%, respectively.13 When patch testing is performed, both Compositae mix and sesquiterpene lactone mix should be utilized to minimize the risk of missing Asteraceae allergy, as sesquiterpene lactone mix alone does not detect all Compositae-sensitized patients. Additionally, it may be necessary to test supplemental Asteraceae allergens, including preparations from specific plants to which the patient has been exposed. Exposure to Asteraceae-containing cosmetic products may lead to dermatitis, though this is highly dependent on the particular plant species involved. For instance, the prevalence of sensitization is high in arnica (tincture) and elecampane but low with more commonly used species such as German chamomile.14
Cutaneous Manifestations
Asteraceae dermatitis, which also is known as Australian bush dermatitis, weed dermatitis, and chrysanthemum dermatitis,2 can manifest on any area of the body that directly contacts the plant or is exposed to the pollen. Asteraceae dermatitis historically was reported in older adults with a recent history of plant exposure.6,15 However, recent data have shown a female preponderance and a younger mean age of onset (46–49 years).16
There are multiple distinct clinical manifestations of Asteraceae dermatitis. The most common cutaneous finding is localized vesicular or eczematous patches on the hands or wrists. Other variations include eczematous rashes on the exposed skin of the hands, arms, face, and neck; generalized eczema; and isolated facial eczema.16,17 These variations can be attributed to contact dermatitis caused by airborne pollen, which may mimic photodermatitis. However, airborne Asteraceae dermatitis can be distinguished clinically from photodermatitis by the involvement of sun-protected areas such as the skinfolds of the eyelids, retroauricular sulci, and nasolabial folds (Figure 2).2,9 In rare cases, systemic allergic contact dermatitis can occur if the Asteraceae allergen is ingested.2,18

Other diagnostic clues include dermatitis that flares during the summer, at the peak of the growing season, with remission in the cooler months. Potential risk factors include a childhood history of atopic dermatitis and allergic rhinitis.16 With prolonged exposure, patients may develop chronic actinic dermatitis, an immunologically mediated photodermatosis characterized by lichenified and pruritic eczematous plaques located predominantly on sun-exposed areas with notable sparing of the skin folds.19 The association between Asteraceae dermatitis and chronic actinic dermatitis is highly variable, with some studies reporting a 25% correlation and others finding a stronger association of up to 80%.2,15,20 Asteraceae allergy appears to be a relatively uncommon cause of photoallergy in North America. In one recent study, 16% (3/19) of patients with chronic actinic dermatitis had positive patch or photopatch tests to sesquiterpene lactone mix, but in another large study of photopatch testing it was reported to be a rare photoallergen.21,22
Parthenium dermatitis is an allergic contact dermatitis caused by exposure to Parthenium hysterophorus, a weed of the Asteraceae family that is responsible for 30% of cases of contact dermatitis in India.23,24 Unlike the more classic manifestation of Asteraceae dermatitis, which primarily affects the upper extremities in cases from North America and Europe, Parthenium dermatitis typically occurs in an airborne pattern distribution.24
Management
While complete avoidance of Asteraceae plants is ideal, it often is unrealistic due to their abundance in nature. Therefore, minimizing exposure to the causative plants is recommended. Primary preventive measures such as wearing protective gloves and clothing and applying bentonite clay prior to exposure should be taken when working outdoors. Promptly showering after contact with plants also can reduce the risk for Asteraceae dermatitis.
Symptomatic treatment is appropriate for mild cases and includes topical corticosteroids and calcineurin inhibitors. For severe cases, systemic corticosteroids may be needed for acute flares, with azathioprine, mycophenolate, cyclosporine, or methotrexate available for recalcitrant disease. Verma et al25 found that treatment with azathioprine for 6 months resulted in greater than 60% clearance in all 12 patients, with a majority achieving 80% to 100% clearance. Methotrexate has been used at doses of 15 mg once weekly.26 Narrowband UVB and psoralen plus UVA have been effective in extensive cases; however, care should be exercised in patients with photosensitive dermatitis, who instead should practice strict photoprotection.27-29 Lakshmi et al30 reported the use of cyclosporine during the acute phase of Asteraceae dermatitis at a dose of 2.5 mg/kg daily for 4 to 8 weeks. There have been several case reports of dupilumab treating allergic contact dermatitis; however, there have been 3 cases of patients with atopic dermatitis developing Asteraceae dermatitis while taking dupilumab.31,32 Recently, oral Janus kinase inhibitors have shown success in treating refractory cases of airborne Asteraceae dermatitis.33,34 Further research is needed to determine the safety and efficacy of dupilumab and Janus kinase inhibitors for treatment of Asteraceae dermatitis.
Final Thoughts
The Asteraceae plant family is vast and diverse, with more than 200 species reported to cause allergic contact dermatitis.12 Common modes of contact include gardening, occupational exposure, airborne pollen, and use of pediculicides and cosmetics that contain components of Asteraceae plants. Educating patients on how to minimize contact with Asteraceae plants is the most effective management strategy; topical agents and oral immunosuppressives can be used for symptomatic treatment.
The Asteraceae (formerly Compositae) family of plants is derived from the ancient Greek word aster, meaning “star,” referring to the starlike arrangement of flower petals around a central disc known as a capitulum. What initially appears as a single flower is actually a composite of several smaller flowers, hence the former name Compositae.1 Well-known members of the Asteraceae family include ornamental annuals (eg, sunflowers, marigolds, cosmos), herbaceous perennials (eg, chrysanthemums, dandelions), vegetables (eg, lettuce, chicory, artichokes), herbs (eg, chamomile, tarragon), and weeds (eg, ragweed, horseweed, capeweed)(Figure 1).2

There are more than 25,000 species of Asteraceae plants that thrive in a wide range of climates worldwide. Cases of Asteraceae-induced skin reactions have been reported in North America, Europe, Asia, and Australia.3 Members of the Asteraceae family are ubiquitous in gardens, along roadsides, and in the wilderness. Occupational exposure commonly affects gardeners, florists, farmers, and forestry workers through either direct contact with plants or via airborne pollen. Furthermore, plants of the Asteraceae family are used in various products, including pediculicides (eg, insect repellents), cosmetics (eg, eye creams, body washes), and food products (eg, cooking oils, sweetening agents, coffee substitutes, herbal teas).4-6 These plants have substantial allergic potential, resulting in numerous cutaneous reactions.
Allergic Potential
Asteraceae plants can elicit both immediate and delayed hypersensitivity reactions (HSRs); for instance, exposure to ragweed pollen may cause an IgE-mediated type 1 HSR manifesting as allergic rhinitis or a type IV HSR manifesting as airborne allergic contact dermatitis.7,8 The main contact allergens present in Asteraceae plants are sesquiterpene lactones, which are found in the leaves, stems, flowers, and pollen.9-11 Sesquiterpene lactones consist of an α-methyl group attached to a lactone ring combined with a sesquiterpene.12 Patch testing can be used to diagnose Asteraceae allergy; however, the results are not consistently reliable because there is no perfect screening allergen. Patch test preparations commonly used to detect Asteraceae allergy include Compositae mix (consisting of Anthemis nobilis extract, Chamomilla recutita extract, Achillea millefolium extract, Tanacetum vulgare extract, Arnica montana extract, and parthenolide) and sesquiterpene lactone mix (consisting of alantolactone, dehydrocostus lactone, and costunolide). In North America, the prevalence of positive patch tests to Compositae mix and sesquiterpene lactone mix is approximately 2% and 0.5%, respectively.13 When patch testing is performed, both Compositae mix and sesquiterpene lactone mix should be utilized to minimize the risk of missing Asteraceae allergy, as sesquiterpene lactone mix alone does not detect all Compositae-sensitized patients. Additionally, it may be necessary to test supplemental Asteraceae allergens, including preparations from specific plants to which the patient has been exposed. Exposure to Asteraceae-containing cosmetic products may lead to dermatitis, though this is highly dependent on the particular plant species involved. For instance, the prevalence of sensitization is high in arnica (tincture) and elecampane but low with more commonly used species such as German chamomile.14
Cutaneous Manifestations
Asteraceae dermatitis, which also is known as Australian bush dermatitis, weed dermatitis, and chrysanthemum dermatitis,2 can manifest on any area of the body that directly contacts the plant or is exposed to the pollen. Asteraceae dermatitis historically was reported in older adults with a recent history of plant exposure.6,15 However, recent data have shown a female preponderance and a younger mean age of onset (46–49 years).16
There are multiple distinct clinical manifestations of Asteraceae dermatitis. The most common cutaneous finding is localized vesicular or eczematous patches on the hands or wrists. Other variations include eczematous rashes on the exposed skin of the hands, arms, face, and neck; generalized eczema; and isolated facial eczema.16,17 These variations can be attributed to contact dermatitis caused by airborne pollen, which may mimic photodermatitis. However, airborne Asteraceae dermatitis can be distinguished clinically from photodermatitis by the involvement of sun-protected areas such as the skinfolds of the eyelids, retroauricular sulci, and nasolabial folds (Figure 2).2,9 In rare cases, systemic allergic contact dermatitis can occur if the Asteraceae allergen is ingested.2,18

Other diagnostic clues include dermatitis that flares during the summer, at the peak of the growing season, with remission in the cooler months. Potential risk factors include a childhood history of atopic dermatitis and allergic rhinitis.16 With prolonged exposure, patients may develop chronic actinic dermatitis, an immunologically mediated photodermatosis characterized by lichenified and pruritic eczematous plaques located predominantly on sun-exposed areas with notable sparing of the skin folds.19 The association between Asteraceae dermatitis and chronic actinic dermatitis is highly variable, with some studies reporting a 25% correlation and others finding a stronger association of up to 80%.2,15,20 Asteraceae allergy appears to be a relatively uncommon cause of photoallergy in North America. In one recent study, 16% (3/19) of patients with chronic actinic dermatitis had positive patch or photopatch tests to sesquiterpene lactone mix, but in another large study of photopatch testing it was reported to be a rare photoallergen.21,22
Parthenium dermatitis is an allergic contact dermatitis caused by exposure to Parthenium hysterophorus, a weed of the Asteraceae family that is responsible for 30% of cases of contact dermatitis in India.23,24 Unlike the more classic manifestation of Asteraceae dermatitis, which primarily affects the upper extremities in cases from North America and Europe, Parthenium dermatitis typically occurs in an airborne pattern distribution.24
Management
While complete avoidance of Asteraceae plants is ideal, it often is unrealistic due to their abundance in nature. Therefore, minimizing exposure to the causative plants is recommended. Primary preventive measures such as wearing protective gloves and clothing and applying bentonite clay prior to exposure should be taken when working outdoors. Promptly showering after contact with plants also can reduce the risk for Asteraceae dermatitis.
Symptomatic treatment is appropriate for mild cases and includes topical corticosteroids and calcineurin inhibitors. For severe cases, systemic corticosteroids may be needed for acute flares, with azathioprine, mycophenolate, cyclosporine, or methotrexate available for recalcitrant disease. Verma et al25 found that treatment with azathioprine for 6 months resulted in greater than 60% clearance in all 12 patients, with a majority achieving 80% to 100% clearance. Methotrexate has been used at doses of 15 mg once weekly.26 Narrowband UVB and psoralen plus UVA have been effective in extensive cases; however, care should be exercised in patients with photosensitive dermatitis, who instead should practice strict photoprotection.27-29 Lakshmi et al30 reported the use of cyclosporine during the acute phase of Asteraceae dermatitis at a dose of 2.5 mg/kg daily for 4 to 8 weeks. There have been several case reports of dupilumab treating allergic contact dermatitis; however, there have been 3 cases of patients with atopic dermatitis developing Asteraceae dermatitis while taking dupilumab.31,32 Recently, oral Janus kinase inhibitors have shown success in treating refractory cases of airborne Asteraceae dermatitis.33,34 Further research is needed to determine the safety and efficacy of dupilumab and Janus kinase inhibitors for treatment of Asteraceae dermatitis.
Final Thoughts
The Asteraceae plant family is vast and diverse, with more than 200 species reported to cause allergic contact dermatitis.12 Common modes of contact include gardening, occupational exposure, airborne pollen, and use of pediculicides and cosmetics that contain components of Asteraceae plants. Educating patients on how to minimize contact with Asteraceae plants is the most effective management strategy; topical agents and oral immunosuppressives can be used for symptomatic treatment.
- Morhardt S, Morhardt E. California Desert Flowers: An Introduction to Families, Genera, and Species. University of California Press; 2004.
- Gordon LA. Compositae dermatitis. Australas J Dermatol. 1999;40:123-130. doi:10.1046/j.1440-0960.1999.00341.x
- Denisow-Pietrzyk M, Pietrzyk Ł, Denisow B. Asteraceae species as potential environmental factors of allergy. Environ Sci Pollut Res Int. 2019;26:6290-6300. doi:10.1007/s11356-019-04146-w
- Paulsen E, Chistensen LP, Andersen KE. Cosmetics and herbal remedies with Compositae plant extracts—are they tolerated by Compositae-allergic patients? Contact Dermatitis. 2008;58:15-23. doi:10.1111/j.1600-0536.2007.01250.x
- Burry JN, Reid JG, Kirk J. Australian bush dermatitis. Contact Dermatitis. 1975;1:263-264. doi:10.1111/j.1600-0536.1975.tb05422.x
- Punchihewa N, Palmer A, Nixon R. Allergic contact dermatitis to Compositae: an Australian case series. Contact Dermatitis. 2022;87:356-362. doi:10.1111/cod.14162
- Chen KW, Marusciac L, Tamas PT, et al. Ragweed pollen allergy: burden, characteristics, and management of an imported allergen source in Europe. Int Arch Allergy Immunol. 2018;176:163-180. doi:10.1159/000487997
- Schloemer JA, Zirwas MJ, Burkhart CG. Airborne contact dermatitis: common causes in the USA. Int J Dermatol. 2015;54:271-274. doi:10.1111/ijd.12692
- Arlette J, Mitchell JC. Compositae dermatitis. current aspects. Contact Dermatitis. 1981;7:129-136. doi:10.1111/j.1600-0536.1981.tb04584.x
- Mitchell JC, Dupuis G. Allergic contact dermatitis from sesquiterpenoids of the Compositae family of plants. Br J Dermatol. 1971;84:139-150. doi:10.1111/j.1365-2133.1971.tb06857.x
- Salapovic H, Geier J, Reznicek G. Quantification of Sesquiterpene lactones in Asteraceae plant extracts: evaluation of their allergenic potential. Sci Pharm. 2013;81:807-818. doi:10.3797/scipharm.1306-17
- Paulsen E. Compositae dermatitis: a survey. Contact Dermatitis. 1992;26:76-86. doi:10.1111/j.1600-0536.1992.tb00888.x. Published correction appears in Contact Dermatitis. 1992;27:208.
- DeKoven JG, Silverberg JI, Warshaw EM, et al. North American Contact Dermatitis Group patch test results: 2017-2018. Dermatitis. 2021;32:111-123. doi:10.1097/DER.0000000000000729
- Paulsen E. Contact sensitization from Compositae-containing herbal remedies and cosmetics. Contact Dermatitis. 2002;47:189-198. doi:10.1034/j.1600-0536.2002.470401.x
- Frain-Bell W, Johnson BE. Contact allergic sensitivity to plants and the photosensitivity dermatitis and actinic reticuloid syndrome. Br J Dermatol. 1979;101:503-512.
- Paulsen E, Andersen KE. Clinical patterns of Compositae dermatitis in Danish monosensitized patients. Contact Dermatitis. 2018;78:185-193. doi:10.1111/cod.12916
- Jovanovic´ M, Poljacki M. Compositae dermatitis. Med Pregl. 2003;56:43-49. doi:10.2298/mpns0302043j
- Krook G. Occupational dermatitis from Lactuca sativa (lettuce) and Cichorium (endive). simultaneous occurrence of immediate and delayed allergy as a cause of contact dermatitis. Contact Dermatitis. 1977;3:27-36. doi:10.1111/j.1600-0536.1977.tb03583.x
- Paek SY, Lim HW. Chronic actinic dermatitis. Dermatol Clin. 2014;32:355-361, viii-ix. doi:10.1016/j.det.2014.03.007
- du P Menagé H, Hawk JL, White IR. Sesquiterpene lactone mix contact sensitivity and its relationship to chronic actinic dermatitis: a follow-up study. Contact Dermatitis. 1998;39:119-122. doi:10.1111/j.1600-0536.1998.tb05859.x
- Wang CX, Belsito DV. Chronic actinic dermatitis revisited. Dermatitis. 2020;31:68-74. doi:10.1097/DER.0000000000000531
- DeLeo VA, Adler BL, Warshaw EM, et al. Photopatch test results of the North American contact dermatitis group, 1999-2009. Photodermatol Photoimmunol Photomed. 2022;38:288-291. doi:10.1111/phpp.12742
- McGovern TW, LaWarre S. Botanical briefs: the scourge of India—Parthenium hysterophorus L. Cutis. 2001;67:27-34. Published correction appears in Cutis. 2001;67:154.
- Sharma VK, Verma P, Maharaja K. Parthenium dermatitis. Photochem Photobiol Sci. 2013;12:85-94. doi:10.1039/c2pp25186h
- Verma KK, Bansal A, Sethuraman G. Parthenium dermatitis treated with azathioprine weekly pulse doses. Indian J Dermatol Venereol Leprol. 2006;72:24-27. doi:10.4103/0378-6323.19713
- Sharma VK, Bhat R, Sethuraman G, et al. Treatment of Parthenium dermatitis with methotrexate. Contact Dermatitis. 2007;57:118-119. doi:10.1111/j.1600-0536.2006.00950.x
- Burke DA, Corey G, Storrs FJ. Psoralen plus UVA protocol for Compositae photosensitivity. Am J Contact Dermat. 1996;7:171-176.
- Lovell CR. Allergic contact dermatitis due to plants. In: Plants and the Skin. Blackwell Scientific Publications; 1993:96-254.
- Dogra S, Parsad D, Handa S. Narrowband ultraviolet B in airborne contact dermatitis: a ray of hope! Br J Dermatol. 2004;150:373-374. doi:10.1111/j.1365-2133.2004.05724.x
- Lakshmi C, Srinivas CR, Jayaraman A. Ciclosporin in Parthenium dermatitis—a report of 2 cases. Contact Dermatitis. 2008;59:245-248. doi:10.1111/j.1600-0536.2007.01208.x
- Hendricks AJ, Yosipovitch G, Shi VY. Dupilumab use in dermatologic conditions beyond atopic dermatitis—a systematic review. J Dermatolog Treat. 2021;32:19-28. doi:10.1080/09546634.2019.1689227
- Napolitano M, Fabbrocini G, Patruno C. Allergic contact dermatitis to Compositae: a possible cause of dupilumab-associated facial and neck dermatitis in atopic dermatitis patients? Contact Dermatitis. 2021;85:473-474. doi:10.1111/cod.13898
- Muddebihal A, Sardana K, Sinha S, et al. Tofacitinib in refractory Parthenium-induced airborne allergic contact dermatitis. Contact Dermatitis. 2023;88:150-152. doi:10.1111/cod.14234
- Baltazar D, Shinamoto SR, Hamann CP, et al. Occupational airborne allergic contact dermatitis to invasive Compositae species treated with abrocitinib: a case report. Contact Dermatitis. 2022;87:542-544. doi:10.1111/cod.14204
- Morhardt S, Morhardt E. California Desert Flowers: An Introduction to Families, Genera, and Species. University of California Press; 2004.
- Gordon LA. Compositae dermatitis. Australas J Dermatol. 1999;40:123-130. doi:10.1046/j.1440-0960.1999.00341.x
- Denisow-Pietrzyk M, Pietrzyk Ł, Denisow B. Asteraceae species as potential environmental factors of allergy. Environ Sci Pollut Res Int. 2019;26:6290-6300. doi:10.1007/s11356-019-04146-w
- Paulsen E, Chistensen LP, Andersen KE. Cosmetics and herbal remedies with Compositae plant extracts—are they tolerated by Compositae-allergic patients? Contact Dermatitis. 2008;58:15-23. doi:10.1111/j.1600-0536.2007.01250.x
- Burry JN, Reid JG, Kirk J. Australian bush dermatitis. Contact Dermatitis. 1975;1:263-264. doi:10.1111/j.1600-0536.1975.tb05422.x
- Punchihewa N, Palmer A, Nixon R. Allergic contact dermatitis to Compositae: an Australian case series. Contact Dermatitis. 2022;87:356-362. doi:10.1111/cod.14162
- Chen KW, Marusciac L, Tamas PT, et al. Ragweed pollen allergy: burden, characteristics, and management of an imported allergen source in Europe. Int Arch Allergy Immunol. 2018;176:163-180. doi:10.1159/000487997
- Schloemer JA, Zirwas MJ, Burkhart CG. Airborne contact dermatitis: common causes in the USA. Int J Dermatol. 2015;54:271-274. doi:10.1111/ijd.12692
- Arlette J, Mitchell JC. Compositae dermatitis. current aspects. Contact Dermatitis. 1981;7:129-136. doi:10.1111/j.1600-0536.1981.tb04584.x
- Mitchell JC, Dupuis G. Allergic contact dermatitis from sesquiterpenoids of the Compositae family of plants. Br J Dermatol. 1971;84:139-150. doi:10.1111/j.1365-2133.1971.tb06857.x
- Salapovic H, Geier J, Reznicek G. Quantification of Sesquiterpene lactones in Asteraceae plant extracts: evaluation of their allergenic potential. Sci Pharm. 2013;81:807-818. doi:10.3797/scipharm.1306-17
- Paulsen E. Compositae dermatitis: a survey. Contact Dermatitis. 1992;26:76-86. doi:10.1111/j.1600-0536.1992.tb00888.x. Published correction appears in Contact Dermatitis. 1992;27:208.
- DeKoven JG, Silverberg JI, Warshaw EM, et al. North American Contact Dermatitis Group patch test results: 2017-2018. Dermatitis. 2021;32:111-123. doi:10.1097/DER.0000000000000729
- Paulsen E. Contact sensitization from Compositae-containing herbal remedies and cosmetics. Contact Dermatitis. 2002;47:189-198. doi:10.1034/j.1600-0536.2002.470401.x
- Frain-Bell W, Johnson BE. Contact allergic sensitivity to plants and the photosensitivity dermatitis and actinic reticuloid syndrome. Br J Dermatol. 1979;101:503-512.
- Paulsen E, Andersen KE. Clinical patterns of Compositae dermatitis in Danish monosensitized patients. Contact Dermatitis. 2018;78:185-193. doi:10.1111/cod.12916
- Jovanovic´ M, Poljacki M. Compositae dermatitis. Med Pregl. 2003;56:43-49. doi:10.2298/mpns0302043j
- Krook G. Occupational dermatitis from Lactuca sativa (lettuce) and Cichorium (endive). simultaneous occurrence of immediate and delayed allergy as a cause of contact dermatitis. Contact Dermatitis. 1977;3:27-36. doi:10.1111/j.1600-0536.1977.tb03583.x
- Paek SY, Lim HW. Chronic actinic dermatitis. Dermatol Clin. 2014;32:355-361, viii-ix. doi:10.1016/j.det.2014.03.007
- du P Menagé H, Hawk JL, White IR. Sesquiterpene lactone mix contact sensitivity and its relationship to chronic actinic dermatitis: a follow-up study. Contact Dermatitis. 1998;39:119-122. doi:10.1111/j.1600-0536.1998.tb05859.x
- Wang CX, Belsito DV. Chronic actinic dermatitis revisited. Dermatitis. 2020;31:68-74. doi:10.1097/DER.0000000000000531
- DeLeo VA, Adler BL, Warshaw EM, et al. Photopatch test results of the North American contact dermatitis group, 1999-2009. Photodermatol Photoimmunol Photomed. 2022;38:288-291. doi:10.1111/phpp.12742
- McGovern TW, LaWarre S. Botanical briefs: the scourge of India—Parthenium hysterophorus L. Cutis. 2001;67:27-34. Published correction appears in Cutis. 2001;67:154.
- Sharma VK, Verma P, Maharaja K. Parthenium dermatitis. Photochem Photobiol Sci. 2013;12:85-94. doi:10.1039/c2pp25186h
- Verma KK, Bansal A, Sethuraman G. Parthenium dermatitis treated with azathioprine weekly pulse doses. Indian J Dermatol Venereol Leprol. 2006;72:24-27. doi:10.4103/0378-6323.19713
- Sharma VK, Bhat R, Sethuraman G, et al. Treatment of Parthenium dermatitis with methotrexate. Contact Dermatitis. 2007;57:118-119. doi:10.1111/j.1600-0536.2006.00950.x
- Burke DA, Corey G, Storrs FJ. Psoralen plus UVA protocol for Compositae photosensitivity. Am J Contact Dermat. 1996;7:171-176.
- Lovell CR. Allergic contact dermatitis due to plants. In: Plants and the Skin. Blackwell Scientific Publications; 1993:96-254.
- Dogra S, Parsad D, Handa S. Narrowband ultraviolet B in airborne contact dermatitis: a ray of hope! Br J Dermatol. 2004;150:373-374. doi:10.1111/j.1365-2133.2004.05724.x
- Lakshmi C, Srinivas CR, Jayaraman A. Ciclosporin in Parthenium dermatitis—a report of 2 cases. Contact Dermatitis. 2008;59:245-248. doi:10.1111/j.1600-0536.2007.01208.x
- Hendricks AJ, Yosipovitch G, Shi VY. Dupilumab use in dermatologic conditions beyond atopic dermatitis—a systematic review. J Dermatolog Treat. 2021;32:19-28. doi:10.1080/09546634.2019.1689227
- Napolitano M, Fabbrocini G, Patruno C. Allergic contact dermatitis to Compositae: a possible cause of dupilumab-associated facial and neck dermatitis in atopic dermatitis patients? Contact Dermatitis. 2021;85:473-474. doi:10.1111/cod.13898
- Muddebihal A, Sardana K, Sinha S, et al. Tofacitinib in refractory Parthenium-induced airborne allergic contact dermatitis. Contact Dermatitis. 2023;88:150-152. doi:10.1111/cod.14234
- Baltazar D, Shinamoto SR, Hamann CP, et al. Occupational airborne allergic contact dermatitis to invasive Compositae species treated with abrocitinib: a case report. Contact Dermatitis. 2022;87:542-544. doi:10.1111/cod.14204
Practice Points
- Asteraceae dermatitis can occur from direct contact with plants of the Asteraceae family; through airborne pollen; or from exposure to topical medications, cooking products, and cosmetics.
- Patient education on primary prevention, especially protective clothing, is crucial, as these plants are ubiquitous outdoors and have diverse phenotypes.
- Management of mild Asteraceae dermatitis consists primarily of topical corticosteroids and calcineurin inhibitors, while systemic corticosteroids and other immunosuppressive agents are utilized for severe or recalcitrant cases.
Atopic Dermatitis: Upadacitinib Effectiveness Maintained Through 76 weeks Among Adolescents
TOPLINE:
across three phase 3 trials.
METHODOLOGY:
- Researchers conducted three double-blind, placebo-controlled phase 3 randomized clinical trials (Measure Up 1, Measure Up 2, and AD Up) involving 542 adolescents aged 12-17 years with moderate to severe AD.
- Participants were randomized to receive the oral Janus kinase inhibitor upadacitinib (15 mg or 30 mg once daily) or placebo, with or without topical corticosteroids, for 16 weeks, followed by rerandomization of patients in the placebo group to upadacitinib for up to 76 weeks.
- Study endpoints were at least a 75%, 90%, or 100% reduction in the Eczema Area and Severity Index (EASI-75, EASI-90, and EASI-100, respectively), Validated Investigator Global Assessment for AD (vIGA-AD) score of 0 or 1, and a ≥ 4-point improvement in the Worst Pruritus Numerical Rating Scale (WP-NRS).
- Adverse events were monitored, including serious infections, herpes zoster, and creatine kinase elevation.
TAKEAWAY:
- Among those who continued treatment on upadacitinib, 15 mg and 30 mg, EASI-75 response rates were maintained or improved through week 76 in all three studies. Patients who switched from placebo to upadacitinib also experienced improvements in EASI-75 through week 76.
- The proportion of patients who achieved EASI-90 and EASI-100 responses increased, and in general, were maintained from week 16 through week 76 in all three studies; the proportion was numerically higher among patients on 30 mg for all three studies.
- The proportion of adolescents achieving vIGA-AD score of 0 or 1 and WP-NRS improvement of ≥ 4 points was sustained or improved through 76 weeks.
- Serious infections were reported in five patients or fewer in each treatment group for all three studies. All opportunistic infections were eczema herpeticum; most cases were not serious, or were mild or moderate, and in general, did not require stopping treatment.
IN PRACTICE:
“These results through 76 weeks demonstrated that upadacitinib, with a favorable benefit-risk profile, was an effective long-term treatment option for adolescents with moderate to severe AD,” the authors wrote.
SOURCE:
The study was led by Amy S. Paller, MD, professor and chair of dermatology, Northwestern University, Chicago, and was published online on October 23 in JAMA Dermatology.
LIMITATIONS:
The study limitations included a small sample size, and the findings did not extend to patients under 12 years or those weighing < 40 kg.
DISCLOSURES:
This study was supported by AbbVie. Paller received grants and personal fees from pharmaceutical companies including AbbVie during the conduct of the study. Several authors reported financial ties with various sources, including AbbVie.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
across three phase 3 trials.
METHODOLOGY:
- Researchers conducted three double-blind, placebo-controlled phase 3 randomized clinical trials (Measure Up 1, Measure Up 2, and AD Up) involving 542 adolescents aged 12-17 years with moderate to severe AD.
- Participants were randomized to receive the oral Janus kinase inhibitor upadacitinib (15 mg or 30 mg once daily) or placebo, with or without topical corticosteroids, for 16 weeks, followed by rerandomization of patients in the placebo group to upadacitinib for up to 76 weeks.
- Study endpoints were at least a 75%, 90%, or 100% reduction in the Eczema Area and Severity Index (EASI-75, EASI-90, and EASI-100, respectively), Validated Investigator Global Assessment for AD (vIGA-AD) score of 0 or 1, and a ≥ 4-point improvement in the Worst Pruritus Numerical Rating Scale (WP-NRS).
- Adverse events were monitored, including serious infections, herpes zoster, and creatine kinase elevation.
TAKEAWAY:
- Among those who continued treatment on upadacitinib, 15 mg and 30 mg, EASI-75 response rates were maintained or improved through week 76 in all three studies. Patients who switched from placebo to upadacitinib also experienced improvements in EASI-75 through week 76.
- The proportion of patients who achieved EASI-90 and EASI-100 responses increased, and in general, were maintained from week 16 through week 76 in all three studies; the proportion was numerically higher among patients on 30 mg for all three studies.
- The proportion of adolescents achieving vIGA-AD score of 0 or 1 and WP-NRS improvement of ≥ 4 points was sustained or improved through 76 weeks.
- Serious infections were reported in five patients or fewer in each treatment group for all three studies. All opportunistic infections were eczema herpeticum; most cases were not serious, or were mild or moderate, and in general, did not require stopping treatment.
IN PRACTICE:
“These results through 76 weeks demonstrated that upadacitinib, with a favorable benefit-risk profile, was an effective long-term treatment option for adolescents with moderate to severe AD,” the authors wrote.
SOURCE:
The study was led by Amy S. Paller, MD, professor and chair of dermatology, Northwestern University, Chicago, and was published online on October 23 in JAMA Dermatology.
LIMITATIONS:
The study limitations included a small sample size, and the findings did not extend to patients under 12 years or those weighing < 40 kg.
DISCLOSURES:
This study was supported by AbbVie. Paller received grants and personal fees from pharmaceutical companies including AbbVie during the conduct of the study. Several authors reported financial ties with various sources, including AbbVie.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
across three phase 3 trials.
METHODOLOGY:
- Researchers conducted three double-blind, placebo-controlled phase 3 randomized clinical trials (Measure Up 1, Measure Up 2, and AD Up) involving 542 adolescents aged 12-17 years with moderate to severe AD.
- Participants were randomized to receive the oral Janus kinase inhibitor upadacitinib (15 mg or 30 mg once daily) or placebo, with or without topical corticosteroids, for 16 weeks, followed by rerandomization of patients in the placebo group to upadacitinib for up to 76 weeks.
- Study endpoints were at least a 75%, 90%, or 100% reduction in the Eczema Area and Severity Index (EASI-75, EASI-90, and EASI-100, respectively), Validated Investigator Global Assessment for AD (vIGA-AD) score of 0 or 1, and a ≥ 4-point improvement in the Worst Pruritus Numerical Rating Scale (WP-NRS).
- Adverse events were monitored, including serious infections, herpes zoster, and creatine kinase elevation.
TAKEAWAY:
- Among those who continued treatment on upadacitinib, 15 mg and 30 mg, EASI-75 response rates were maintained or improved through week 76 in all three studies. Patients who switched from placebo to upadacitinib also experienced improvements in EASI-75 through week 76.
- The proportion of patients who achieved EASI-90 and EASI-100 responses increased, and in general, were maintained from week 16 through week 76 in all three studies; the proportion was numerically higher among patients on 30 mg for all three studies.
- The proportion of adolescents achieving vIGA-AD score of 0 or 1 and WP-NRS improvement of ≥ 4 points was sustained or improved through 76 weeks.
- Serious infections were reported in five patients or fewer in each treatment group for all three studies. All opportunistic infections were eczema herpeticum; most cases were not serious, or were mild or moderate, and in general, did not require stopping treatment.
IN PRACTICE:
“These results through 76 weeks demonstrated that upadacitinib, with a favorable benefit-risk profile, was an effective long-term treatment option for adolescents with moderate to severe AD,” the authors wrote.
SOURCE:
The study was led by Amy S. Paller, MD, professor and chair of dermatology, Northwestern University, Chicago, and was published online on October 23 in JAMA Dermatology.
LIMITATIONS:
The study limitations included a small sample size, and the findings did not extend to patients under 12 years or those weighing < 40 kg.
DISCLOSURES:
This study was supported by AbbVie. Paller received grants and personal fees from pharmaceutical companies including AbbVie during the conduct of the study. Several authors reported financial ties with various sources, including AbbVie.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Study Compares Punch Excision vs. Core Excision for Recalcitrant Keloids
according to the results of a small retrospective study.
The method “offers similar efficacy, faster healing, and fewer complications,” one of the study authors, Jinwoong Jung, MD, said in an interview following the annual meeting of the American Society for Dermatologic Surgery, where he presented the study results during an oral abstract session.
For the study, Jung, a dermatologist at Yonsei University College of Medicine, Seoul, South Korea, and colleagues retrospectively analyzed 22 patients with recalcitrant keloids treated with cryotherapy immediately following either PE or CE between May 2019 and March 2024. They used the Vancouver Scar Scale (VSS) to assess treatment efficacy.
Of the 22 patients, 16 underwent treatment with CE and 6 underwent treatment with PE. Pretreatment VSS scores showed no significant differences between the groups (P = .535). The CE group had a reduction in the VSS score from 8.13 to 4.00, while the PE group had a reduction from 7.83 to 3.67, but these declines did not differ significantly (P = .737). The PE group exhibited a shorter healing time than the CE group (a mean of 43.5 vs 63.87 days, respectively), though this difference was not statistically significant (P = .129).
“The uniqueness of this work lies in its simplified use of PE for recalcitrant keloids, which demonstrated efficacy comparable to CE, with the potential advantage of faster healing times,” Jung said. “Future studies with larger sample sizes and extended follow-up periods could help establish this approach as a standard treatment method.”
He acknowledged certain limitations of the study, including its small sample size and the lack of long-term follow-up data. The researchers reported having no relevant disclosures.
A version of this article first appeared on Medscape.com.
according to the results of a small retrospective study.
The method “offers similar efficacy, faster healing, and fewer complications,” one of the study authors, Jinwoong Jung, MD, said in an interview following the annual meeting of the American Society for Dermatologic Surgery, where he presented the study results during an oral abstract session.
For the study, Jung, a dermatologist at Yonsei University College of Medicine, Seoul, South Korea, and colleagues retrospectively analyzed 22 patients with recalcitrant keloids treated with cryotherapy immediately following either PE or CE between May 2019 and March 2024. They used the Vancouver Scar Scale (VSS) to assess treatment efficacy.
Of the 22 patients, 16 underwent treatment with CE and 6 underwent treatment with PE. Pretreatment VSS scores showed no significant differences between the groups (P = .535). The CE group had a reduction in the VSS score from 8.13 to 4.00, while the PE group had a reduction from 7.83 to 3.67, but these declines did not differ significantly (P = .737). The PE group exhibited a shorter healing time than the CE group (a mean of 43.5 vs 63.87 days, respectively), though this difference was not statistically significant (P = .129).
“The uniqueness of this work lies in its simplified use of PE for recalcitrant keloids, which demonstrated efficacy comparable to CE, with the potential advantage of faster healing times,” Jung said. “Future studies with larger sample sizes and extended follow-up periods could help establish this approach as a standard treatment method.”
He acknowledged certain limitations of the study, including its small sample size and the lack of long-term follow-up data. The researchers reported having no relevant disclosures.
A version of this article first appeared on Medscape.com.
according to the results of a small retrospective study.
The method “offers similar efficacy, faster healing, and fewer complications,” one of the study authors, Jinwoong Jung, MD, said in an interview following the annual meeting of the American Society for Dermatologic Surgery, where he presented the study results during an oral abstract session.
For the study, Jung, a dermatologist at Yonsei University College of Medicine, Seoul, South Korea, and colleagues retrospectively analyzed 22 patients with recalcitrant keloids treated with cryotherapy immediately following either PE or CE between May 2019 and March 2024. They used the Vancouver Scar Scale (VSS) to assess treatment efficacy.
Of the 22 patients, 16 underwent treatment with CE and 6 underwent treatment with PE. Pretreatment VSS scores showed no significant differences between the groups (P = .535). The CE group had a reduction in the VSS score from 8.13 to 4.00, while the PE group had a reduction from 7.83 to 3.67, but these declines did not differ significantly (P = .737). The PE group exhibited a shorter healing time than the CE group (a mean of 43.5 vs 63.87 days, respectively), though this difference was not statistically significant (P = .129).
“The uniqueness of this work lies in its simplified use of PE for recalcitrant keloids, which demonstrated efficacy comparable to CE, with the potential advantage of faster healing times,” Jung said. “Future studies with larger sample sizes and extended follow-up periods could help establish this approach as a standard treatment method.”
He acknowledged certain limitations of the study, including its small sample size and the lack of long-term follow-up data. The researchers reported having no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM ASDS 2024
Bipartisan Bill to Provide Free Gun Lockboxes to Veterans
About 7 of every 10 veterans who die by suicide involve the use of a firearm. A reason for this high rate is access, as half of veterans report owning ≥ 1 personal firearms. Of those individuals, more than half report storing firearms loaded and/or unsecured and one-third of veterans who store their firearms loaded and unlocked do not own a lockbox or safe.
Suicide death prevention has improved as firearms have become more difficult to obtain. That’s why Navy veteran Rep. Chris Deluzio (D-PA), former FBI Special Agent and federal prosecutor Rep. Brian Fitzpatrick (R-PA), and Rep. Greg Landsman (D-OH) have teamed up to introduce the Saving Our Veterans Lives Act of 2024. Under the proposed act, any veteran would be able to get a free lockbox from the US Department of Veterans Affairs (VA).
Suicidal crises can be brief. According to the VA, if a person experiencing a suicidal crisis can’t access the method they planned to use, they generally do not seek out other lethal means. Lockboxes are a way of “putting space between thought and trigger,” the VA said.
The VA Suicide Prevention Program distributes free firearm cable locks to any veteran who requests one. However, many veterans favor lockboxes and safes to secure their guns. A VA pilot program offers free lockboxes to veterans enrolled in the Veterans Health Administration who are at an elevated risk for suicide. The program is set to launch in late 2024 and is a collaboration between the Rocky Mountain Mental Illness Research, Education, and Clinical Center for Suicide Prevention, VA National Prosthetics Service, and VA Office of Suicide Prevention.
The proposed bill would make the lockboxes (which typically cost between $25 and $350) free to any veteran, regardless of VA enrollment status or diagnosis. It ensures “sufficient funding for many tens of thousands of lockboxes to be distributed.” The bill would also direct the VA to create a public education campaign on the availability of lockboxes and the importance of secure firearm storage in suicide prevention.
“The alarming and tragic reality is that our veterans face a suicide rate 57% higher than that of civilians,” Rep. Fitzpatrick said. “This commonsense, bipartisan initiative is more than a solution—it's a lifeline.”
The representatives report that the bill has been endorsed by an “unprecedented” number of organizations, including the National Shooting Sports Foundation, Disabled American Veterans, The American Legion, GIFFORDS, Everytown for Gun Safety, Brady, American Psychological Association, American Foundation for Suicide Prevention, and Association of VA Psychologist Leaders.
“Did you know that in some cases only 10 minutes elapse between an individual having suicidal ideation and acting?” American Legion National Commander James LaCoursiere said in the representatives’ press release. “The Saving Our Veterans Lives Act is an important part of preventing suicide as it will provide veterans with the information and means to securely store their firearms to prevent suicide, while still protecting their Second Amendment rights. The Legion commends Rep. Deluzio and his team for bringing this bill forward and for their continued dedication to the welfare of our nation’s veterans.”
"I hear colleagues all the time talk about veteran suicide," Rep. Deluzio said in an interview with Military.com. "It is a problem in my community. It's a problem across the country. Let's take action. This is a chance where we can do it that I think can cut through the politics that normally divide us on these [gun] issues. And I think the coalition supporting the bill tells you, we've got a path to pass it."
About 7 of every 10 veterans who die by suicide involve the use of a firearm. A reason for this high rate is access, as half of veterans report owning ≥ 1 personal firearms. Of those individuals, more than half report storing firearms loaded and/or unsecured and one-third of veterans who store their firearms loaded and unlocked do not own a lockbox or safe.
Suicide death prevention has improved as firearms have become more difficult to obtain. That’s why Navy veteran Rep. Chris Deluzio (D-PA), former FBI Special Agent and federal prosecutor Rep. Brian Fitzpatrick (R-PA), and Rep. Greg Landsman (D-OH) have teamed up to introduce the Saving Our Veterans Lives Act of 2024. Under the proposed act, any veteran would be able to get a free lockbox from the US Department of Veterans Affairs (VA).
Suicidal crises can be brief. According to the VA, if a person experiencing a suicidal crisis can’t access the method they planned to use, they generally do not seek out other lethal means. Lockboxes are a way of “putting space between thought and trigger,” the VA said.
The VA Suicide Prevention Program distributes free firearm cable locks to any veteran who requests one. However, many veterans favor lockboxes and safes to secure their guns. A VA pilot program offers free lockboxes to veterans enrolled in the Veterans Health Administration who are at an elevated risk for suicide. The program is set to launch in late 2024 and is a collaboration between the Rocky Mountain Mental Illness Research, Education, and Clinical Center for Suicide Prevention, VA National Prosthetics Service, and VA Office of Suicide Prevention.
The proposed bill would make the lockboxes (which typically cost between $25 and $350) free to any veteran, regardless of VA enrollment status or diagnosis. It ensures “sufficient funding for many tens of thousands of lockboxes to be distributed.” The bill would also direct the VA to create a public education campaign on the availability of lockboxes and the importance of secure firearm storage in suicide prevention.
“The alarming and tragic reality is that our veterans face a suicide rate 57% higher than that of civilians,” Rep. Fitzpatrick said. “This commonsense, bipartisan initiative is more than a solution—it's a lifeline.”
The representatives report that the bill has been endorsed by an “unprecedented” number of organizations, including the National Shooting Sports Foundation, Disabled American Veterans, The American Legion, GIFFORDS, Everytown for Gun Safety, Brady, American Psychological Association, American Foundation for Suicide Prevention, and Association of VA Psychologist Leaders.
“Did you know that in some cases only 10 minutes elapse between an individual having suicidal ideation and acting?” American Legion National Commander James LaCoursiere said in the representatives’ press release. “The Saving Our Veterans Lives Act is an important part of preventing suicide as it will provide veterans with the information and means to securely store their firearms to prevent suicide, while still protecting their Second Amendment rights. The Legion commends Rep. Deluzio and his team for bringing this bill forward and for their continued dedication to the welfare of our nation’s veterans.”
"I hear colleagues all the time talk about veteran suicide," Rep. Deluzio said in an interview with Military.com. "It is a problem in my community. It's a problem across the country. Let's take action. This is a chance where we can do it that I think can cut through the politics that normally divide us on these [gun] issues. And I think the coalition supporting the bill tells you, we've got a path to pass it."
About 7 of every 10 veterans who die by suicide involve the use of a firearm. A reason for this high rate is access, as half of veterans report owning ≥ 1 personal firearms. Of those individuals, more than half report storing firearms loaded and/or unsecured and one-third of veterans who store their firearms loaded and unlocked do not own a lockbox or safe.
Suicide death prevention has improved as firearms have become more difficult to obtain. That’s why Navy veteran Rep. Chris Deluzio (D-PA), former FBI Special Agent and federal prosecutor Rep. Brian Fitzpatrick (R-PA), and Rep. Greg Landsman (D-OH) have teamed up to introduce the Saving Our Veterans Lives Act of 2024. Under the proposed act, any veteran would be able to get a free lockbox from the US Department of Veterans Affairs (VA).
Suicidal crises can be brief. According to the VA, if a person experiencing a suicidal crisis can’t access the method they planned to use, they generally do not seek out other lethal means. Lockboxes are a way of “putting space between thought and trigger,” the VA said.
The VA Suicide Prevention Program distributes free firearm cable locks to any veteran who requests one. However, many veterans favor lockboxes and safes to secure their guns. A VA pilot program offers free lockboxes to veterans enrolled in the Veterans Health Administration who are at an elevated risk for suicide. The program is set to launch in late 2024 and is a collaboration between the Rocky Mountain Mental Illness Research, Education, and Clinical Center for Suicide Prevention, VA National Prosthetics Service, and VA Office of Suicide Prevention.
The proposed bill would make the lockboxes (which typically cost between $25 and $350) free to any veteran, regardless of VA enrollment status or diagnosis. It ensures “sufficient funding for many tens of thousands of lockboxes to be distributed.” The bill would also direct the VA to create a public education campaign on the availability of lockboxes and the importance of secure firearm storage in suicide prevention.
“The alarming and tragic reality is that our veterans face a suicide rate 57% higher than that of civilians,” Rep. Fitzpatrick said. “This commonsense, bipartisan initiative is more than a solution—it's a lifeline.”
The representatives report that the bill has been endorsed by an “unprecedented” number of organizations, including the National Shooting Sports Foundation, Disabled American Veterans, The American Legion, GIFFORDS, Everytown for Gun Safety, Brady, American Psychological Association, American Foundation for Suicide Prevention, and Association of VA Psychologist Leaders.
“Did you know that in some cases only 10 minutes elapse between an individual having suicidal ideation and acting?” American Legion National Commander James LaCoursiere said in the representatives’ press release. “The Saving Our Veterans Lives Act is an important part of preventing suicide as it will provide veterans with the information and means to securely store their firearms to prevent suicide, while still protecting their Second Amendment rights. The Legion commends Rep. Deluzio and his team for bringing this bill forward and for their continued dedication to the welfare of our nation’s veterans.”
"I hear colleagues all the time talk about veteran suicide," Rep. Deluzio said in an interview with Military.com. "It is a problem in my community. It's a problem across the country. Let's take action. This is a chance where we can do it that I think can cut through the politics that normally divide us on these [gun] issues. And I think the coalition supporting the bill tells you, we've got a path to pass it."
Air Pollution Exposure Linked to Higher Breast Cancer Risk
TOPLINE:
A recent study found that long-term exposure to fine particulate matter ≤ 2.5 μm (PM2.5) is associated with an increased risk for breast cancer, with the highest risk observed among White women.
METHODOLOGY:
- Studies have suggested that exposure to air pollution — specifically PM2.5 — may increase the risk for breast cancer, but data are largely in populations of White women.
- The current analysis explored the potential risk among a more racially and ethnically diverse group.
- The study included 58,358 women (median age, 60.4 years at enrollment) from the California Cancer Registry, followed over an average of 19.3 years. Overall, 35% were African American, 39% were Latino, 15% were White, and 10% were Japanese American.
- Researchers measured PM2.5 exposure using satellite-based data and geocoded addresses. Other pollutants, such as PM10, NO2, NOX, and CO, were also tracked using Environmental Protection Agency data.
TAKEAWAY:
- A total of 3524 invasive breast cancer cases were diagnosed over an average follow-up period of 19.3 years. PM2.5 exposure was associated with a 28% increased risk for breast cancer overall (hazard ratio [HR], 1.28; 95% CI, 1.08-1.51).
- When looking at risk by racial/ethnic group, the association between PM2.5 exposure and breast cancer risk was strongest among White women (HR, 1.67). PM2.5 exposure was also associated with a higher risk for breast cancer among African American women (HR, 1.14; 95% CI, 0.89-1.46) and Latino women (HR, 1.34; 95% CI, 0.94-1.92), but the associations were not significant.
- Overall breast cancer incidence was also positively associated with exposure to NO2, NOX, and CO (HRs, 1.09-1.11), but the associations were not significant. A meta-analysis of this study and ten other cohorts estimated a 5% increased breast cancer incidence per 10-unit increase in PM2.5 (HR, 1.05).
IN PRACTICE:
“Collective findings suggest that PM2.5 exposure should be considered a risk factor for breast cancer, and curtailing air pollution exposures at the population level using regulatory strategies should be a priority,” the authors concluded.
SOURCE:
The study, led by Anna H. Wu, PhD, MPH, Keck School of Medicine, University of Southern California, Los Angeles, was published online in the Journal of Clinical Oncology.
LIMITATIONS:
The study did not include data on nonresidential exposures or residential history before cohort entry, which limited the assessment of earlier exposures. The study also lacked information on specific sources of PM emissions, as well as an explanation for why White women had the highest breast cancer risk compared with other racial/ethnic groups.
DISCLOSURES:
The study was supported by grants from the Health Effects Air Pollution Foundation, the National Cancer Institute, USC Environmental Exposures, Host Factors, and Human Disease, and the California Air Resource Board. One author disclosed being an associate editor for the Journal of Clinical Oncology. No other potential conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
A recent study found that long-term exposure to fine particulate matter ≤ 2.5 μm (PM2.5) is associated with an increased risk for breast cancer, with the highest risk observed among White women.
METHODOLOGY:
- Studies have suggested that exposure to air pollution — specifically PM2.5 — may increase the risk for breast cancer, but data are largely in populations of White women.
- The current analysis explored the potential risk among a more racially and ethnically diverse group.
- The study included 58,358 women (median age, 60.4 years at enrollment) from the California Cancer Registry, followed over an average of 19.3 years. Overall, 35% were African American, 39% were Latino, 15% were White, and 10% were Japanese American.
- Researchers measured PM2.5 exposure using satellite-based data and geocoded addresses. Other pollutants, such as PM10, NO2, NOX, and CO, were also tracked using Environmental Protection Agency data.
TAKEAWAY:
- A total of 3524 invasive breast cancer cases were diagnosed over an average follow-up period of 19.3 years. PM2.5 exposure was associated with a 28% increased risk for breast cancer overall (hazard ratio [HR], 1.28; 95% CI, 1.08-1.51).
- When looking at risk by racial/ethnic group, the association between PM2.5 exposure and breast cancer risk was strongest among White women (HR, 1.67). PM2.5 exposure was also associated with a higher risk for breast cancer among African American women (HR, 1.14; 95% CI, 0.89-1.46) and Latino women (HR, 1.34; 95% CI, 0.94-1.92), but the associations were not significant.
- Overall breast cancer incidence was also positively associated with exposure to NO2, NOX, and CO (HRs, 1.09-1.11), but the associations were not significant. A meta-analysis of this study and ten other cohorts estimated a 5% increased breast cancer incidence per 10-unit increase in PM2.5 (HR, 1.05).
IN PRACTICE:
“Collective findings suggest that PM2.5 exposure should be considered a risk factor for breast cancer, and curtailing air pollution exposures at the population level using regulatory strategies should be a priority,” the authors concluded.
SOURCE:
The study, led by Anna H. Wu, PhD, MPH, Keck School of Medicine, University of Southern California, Los Angeles, was published online in the Journal of Clinical Oncology.
LIMITATIONS:
The study did not include data on nonresidential exposures or residential history before cohort entry, which limited the assessment of earlier exposures. The study also lacked information on specific sources of PM emissions, as well as an explanation for why White women had the highest breast cancer risk compared with other racial/ethnic groups.
DISCLOSURES:
The study was supported by grants from the Health Effects Air Pollution Foundation, the National Cancer Institute, USC Environmental Exposures, Host Factors, and Human Disease, and the California Air Resource Board. One author disclosed being an associate editor for the Journal of Clinical Oncology. No other potential conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
A recent study found that long-term exposure to fine particulate matter ≤ 2.5 μm (PM2.5) is associated with an increased risk for breast cancer, with the highest risk observed among White women.
METHODOLOGY:
- Studies have suggested that exposure to air pollution — specifically PM2.5 — may increase the risk for breast cancer, but data are largely in populations of White women.
- The current analysis explored the potential risk among a more racially and ethnically diverse group.
- The study included 58,358 women (median age, 60.4 years at enrollment) from the California Cancer Registry, followed over an average of 19.3 years. Overall, 35% were African American, 39% were Latino, 15% were White, and 10% were Japanese American.
- Researchers measured PM2.5 exposure using satellite-based data and geocoded addresses. Other pollutants, such as PM10, NO2, NOX, and CO, were also tracked using Environmental Protection Agency data.
TAKEAWAY:
- A total of 3524 invasive breast cancer cases were diagnosed over an average follow-up period of 19.3 years. PM2.5 exposure was associated with a 28% increased risk for breast cancer overall (hazard ratio [HR], 1.28; 95% CI, 1.08-1.51).
- When looking at risk by racial/ethnic group, the association between PM2.5 exposure and breast cancer risk was strongest among White women (HR, 1.67). PM2.5 exposure was also associated with a higher risk for breast cancer among African American women (HR, 1.14; 95% CI, 0.89-1.46) and Latino women (HR, 1.34; 95% CI, 0.94-1.92), but the associations were not significant.
- Overall breast cancer incidence was also positively associated with exposure to NO2, NOX, and CO (HRs, 1.09-1.11), but the associations were not significant. A meta-analysis of this study and ten other cohorts estimated a 5% increased breast cancer incidence per 10-unit increase in PM2.5 (HR, 1.05).
IN PRACTICE:
“Collective findings suggest that PM2.5 exposure should be considered a risk factor for breast cancer, and curtailing air pollution exposures at the population level using regulatory strategies should be a priority,” the authors concluded.
SOURCE:
The study, led by Anna H. Wu, PhD, MPH, Keck School of Medicine, University of Southern California, Los Angeles, was published online in the Journal of Clinical Oncology.
LIMITATIONS:
The study did not include data on nonresidential exposures or residential history before cohort entry, which limited the assessment of earlier exposures. The study also lacked information on specific sources of PM emissions, as well as an explanation for why White women had the highest breast cancer risk compared with other racial/ethnic groups.
DISCLOSURES:
The study was supported by grants from the Health Effects Air Pollution Foundation, the National Cancer Institute, USC Environmental Exposures, Host Factors, and Human Disease, and the California Air Resource Board. One author disclosed being an associate editor for the Journal of Clinical Oncology. No other potential conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
FDA Approves OnabotulinumtoxinA for Improving Platysma Bands
The in adults.
According to a press release from Allergan Aesthetics, which developed onabotulinumtoxinA, by injecting along the jawline and the vertical bands connecting the jaw and neck with one of the FDA-approved doses of the product based on severity, onabotulinumtoxinA temporarily reduces underlying muscle activity.
The company cited results from phase 3 clinical studies, which demonstrated statistical significance for the improvement in appearance of platysma bands from baseline with onabotulinumtoxinA compared with placebo on both investigator and patient assessment (P < .0001).
All secondary endpoints were also met, as measured by multiple validated, proprietary patient-reported outcome instruments. In two of the clinical studies, for example, 65% and 62% of patients reported being “very satisfied” or “satisfied,” respectively, with their neck and jawline definition 14 days after treatment with a dose of 26, 31, or 36 units of onabotulinumtoxinA, compared with 12% with placebo in both studies.
The development marks the fourth indication for onabotulinumtoxinA. The others are for moderate to severe glabellar lines associated with corrugator and/or procerus muscle activity, moderate to severe lateral canthal lines associated with orbicularis oculi activity, and moderate to severe forehead lines associated with frontalis activity.
A version of this article appeared on Medscape.com.
The in adults.
According to a press release from Allergan Aesthetics, which developed onabotulinumtoxinA, by injecting along the jawline and the vertical bands connecting the jaw and neck with one of the FDA-approved doses of the product based on severity, onabotulinumtoxinA temporarily reduces underlying muscle activity.
The company cited results from phase 3 clinical studies, which demonstrated statistical significance for the improvement in appearance of platysma bands from baseline with onabotulinumtoxinA compared with placebo on both investigator and patient assessment (P < .0001).
All secondary endpoints were also met, as measured by multiple validated, proprietary patient-reported outcome instruments. In two of the clinical studies, for example, 65% and 62% of patients reported being “very satisfied” or “satisfied,” respectively, with their neck and jawline definition 14 days after treatment with a dose of 26, 31, or 36 units of onabotulinumtoxinA, compared with 12% with placebo in both studies.
The development marks the fourth indication for onabotulinumtoxinA. The others are for moderate to severe glabellar lines associated with corrugator and/or procerus muscle activity, moderate to severe lateral canthal lines associated with orbicularis oculi activity, and moderate to severe forehead lines associated with frontalis activity.
A version of this article appeared on Medscape.com.
The in adults.
According to a press release from Allergan Aesthetics, which developed onabotulinumtoxinA, by injecting along the jawline and the vertical bands connecting the jaw and neck with one of the FDA-approved doses of the product based on severity, onabotulinumtoxinA temporarily reduces underlying muscle activity.
The company cited results from phase 3 clinical studies, which demonstrated statistical significance for the improvement in appearance of platysma bands from baseline with onabotulinumtoxinA compared with placebo on both investigator and patient assessment (P < .0001).
All secondary endpoints were also met, as measured by multiple validated, proprietary patient-reported outcome instruments. In two of the clinical studies, for example, 65% and 62% of patients reported being “very satisfied” or “satisfied,” respectively, with their neck and jawline definition 14 days after treatment with a dose of 26, 31, or 36 units of onabotulinumtoxinA, compared with 12% with placebo in both studies.
The development marks the fourth indication for onabotulinumtoxinA. The others are for moderate to severe glabellar lines associated with corrugator and/or procerus muscle activity, moderate to severe lateral canthal lines associated with orbicularis oculi activity, and moderate to severe forehead lines associated with frontalis activity.
A version of this article appeared on Medscape.com.


