User login
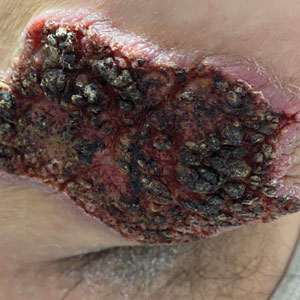
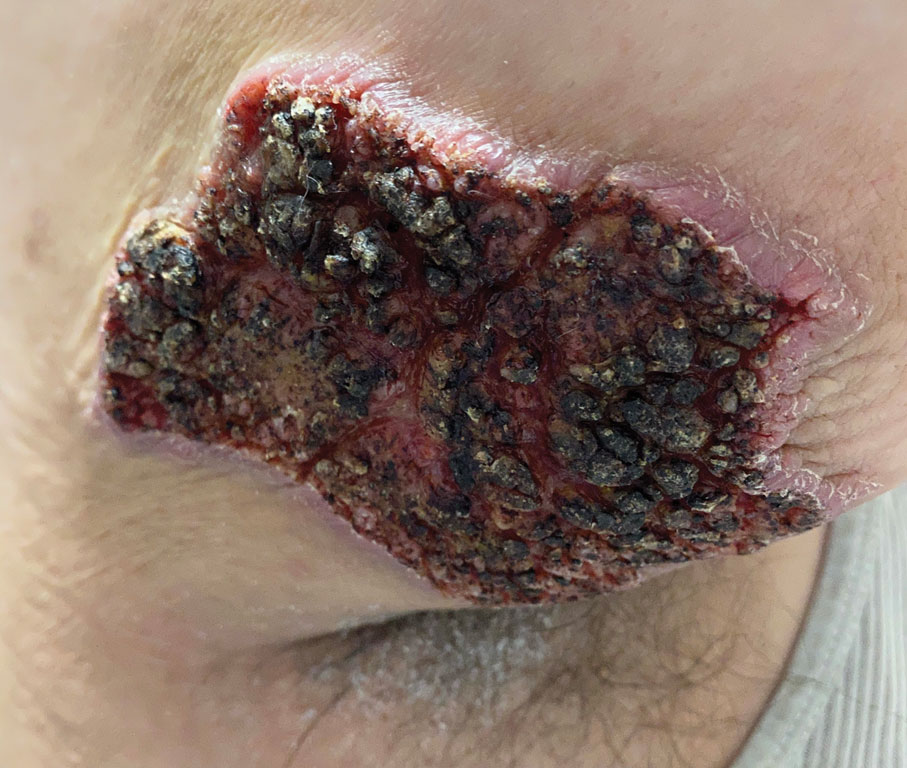
Fungated Eroded Plaque on the Arm
The Diagnosis: Cutaneous Blastomycosis
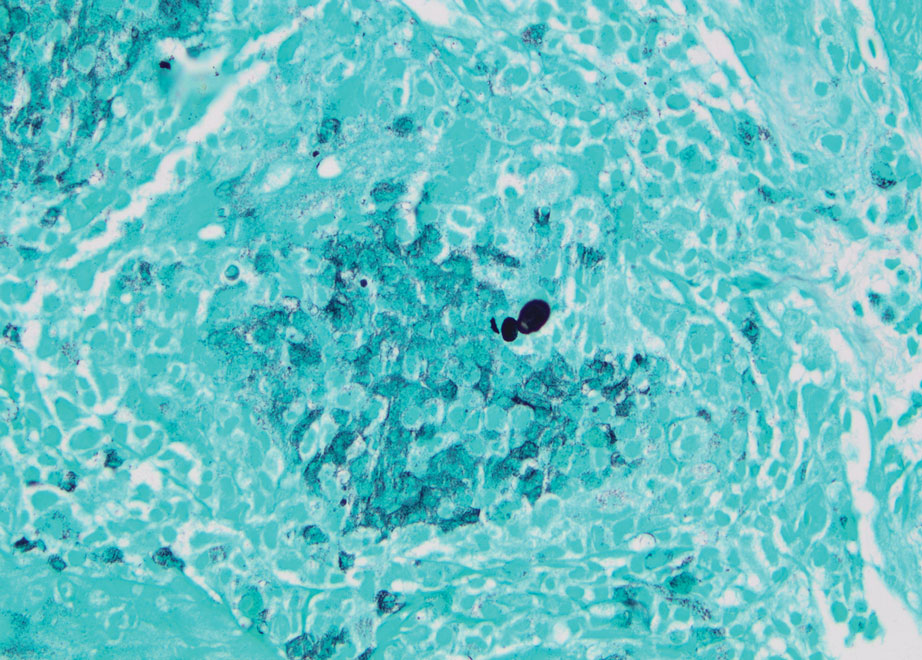
A skin biopsy and fungal cultures confirmed the diagnosis of cutaneous blastomycosis. Grocott- Gomori methenamine-silver staining highlighted fungal organisms with refractile walls and broad-based budding consistent with cutaneous blastomycosis (Figure 1). The histopathologic specimen also demonstrated marked pseudoepitheliomatous hyperplasia (Figure 2A) with neutrophilic microabscesses (Figure 2B). Acid-fast bacillus and Fite staining were negative for bacterial organisms. A fungal culture was positive for Blastomyces dermatitidis. Urine and serum blastomycosis antigen were positive. Although Histoplasma serum antigen also was positive, this likely was from cross-reactivity. Chest radiography was negative for lung involvement, and the patient displayed no neurologic symptoms. He was started on oral itraconazole therapy for the treatment of cutaneous blastomycosis.
Blastomyces dermatitidis, the causative organism of blastomycosis, is endemic to the Ohio and Mississippi River valleys, Great Lakes region, and southeastern United States. It is a thermally dimorphic fungus found in soils that grows as a mold at 25 °C and yeast at 37 °C. Primary infection of the lungs—blastomycosis pneumonia—often is the only clinical manifestation1; however, subsequent hematogenous dissemination to extrapulmonary sites such as the skin, bones, and genitourinary system can occur. Cutaneous blastomycosis, the most common extrapulmonary manifestation, typically follows pulmonary infection. In rare cases, it can occur from direct inoculation.2,3 Skin lesions can occur anywhere but frequently are found on exposed surfaces of the head, neck, and extremities. Lesions classically present as verrucous crusting plaques with draining microabscesses. Violaceous nodules, ulcers, and pustules also may occur.1
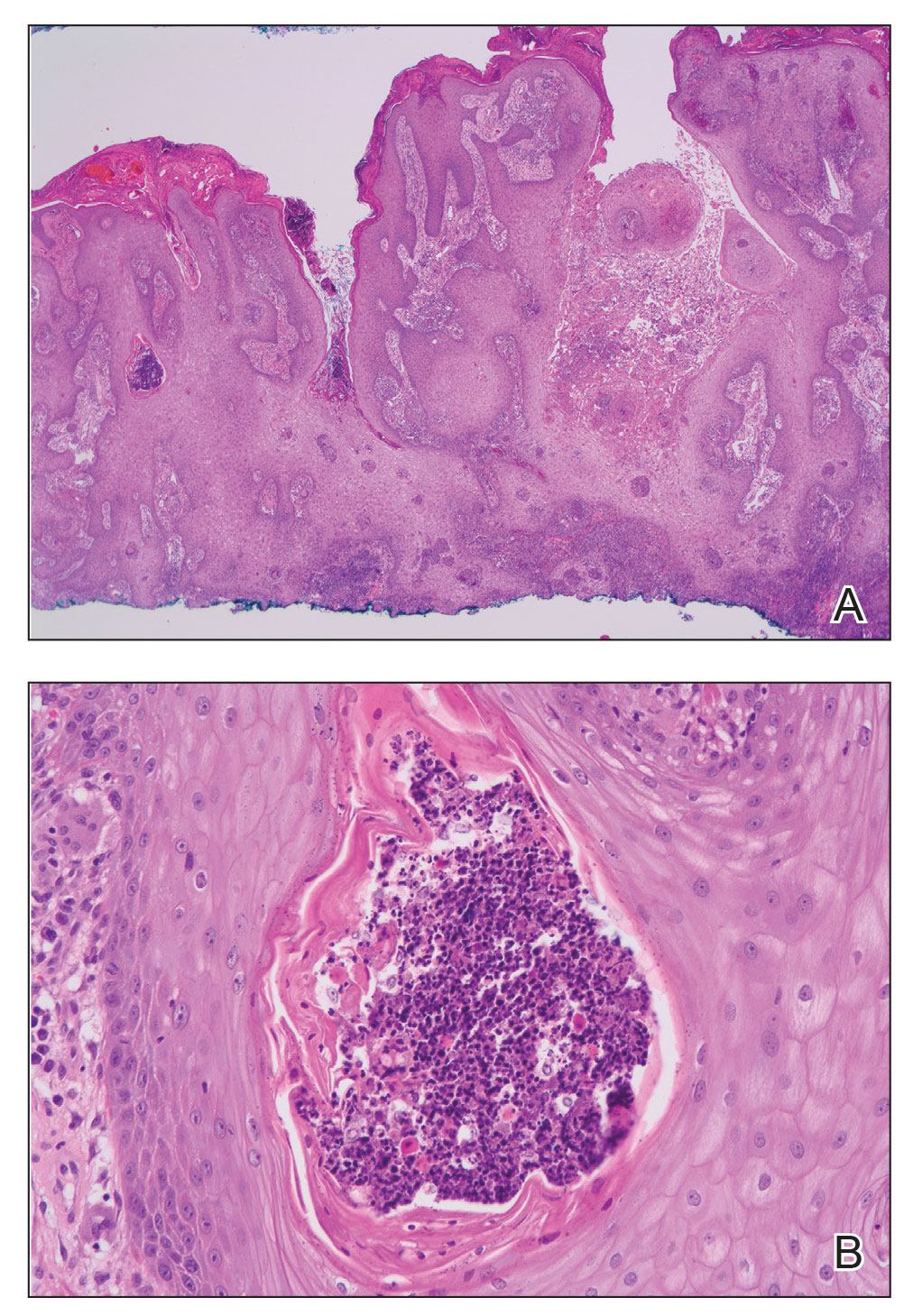
Diagnosis involves obtaining a thorough history of possible environmental exposures such as the patient’s geographic area of residence, occupation, and outdoor activities involving soil or decaying wood. Because blastomycosis can remain latent, remote exposures are relevant. Definitive diagnosis of cutaneous blastomycosis involves skin biopsy of the lesion with fungal culture, but the yeast’s distinctive thick wall and broad-based budding seen with periodic acid–Schiff or Grocott-Gomori methenamine-silver staining provides a rapid presumptive diagnosis.3 Pseudoepitheliomatous hyperplasia and microabscesses also are characteristic features.2 Urine antigen testing for a component of the polysaccharide cell wall has a sensitivity of 93% but a lower specificity of 79% due to cross-reactivity with histoplasmosis.4 Treatment consists of itraconazole for mild to moderate blastomycosis or amphotericin B for those with severe disease or central nervous system involvement or those who are immunosuppressed.1
The differential diagnosis for our patient’s lesion included infectious vs neoplastic etiologies. Histoplasma capsulatum, the dimorphic fungus that causes histoplasmosis, also is endemic to the Ohio and Mississippi River valleys. It is found in soil and droppings of some bats and birds such as chickens and pigeons. Similar to blastomycosis, the primary infection site most commonly is the lungs. It subsequently may disseminate to the skin or less commonly via direct inoculation of injured skin. It can present as papules, plaques, ulcers, purpura, or abscesses. Unlike blastomycosis, tissue biopsy of a cutaneous lesion reveals granuloma formation and distinctive oval, narrow-based budding yeast.5 Atypical mycobacteria are another source of infection to consider. For example, cutaneous Mycobacterium kansasii may present as papules and pustules forming verrucous or granulomatous plaques and ulceration. Histopathologic findings distinguishing mycobacterial infection from blastomycosis include granulomas and acid-fast bacilli in histiocytes.6
Noninfectious etiologies in the differential may include cutaneous squamous cell carcinoma or pemphigus vegetans. Squamous cell carcinoma may present with a broad range of clinical features—papules, plaques, or nodules with smooth, scaly, verrucous, or ulcerative secondary features all are possible presentations.7 Fairskinned individuals, such as our patient, would be at a higher risk in sun-damaged skin. Histologically, cutaneous squamous cell carcinoma is defined as an invasion of the dermis by neoplastic squamous epithelial cells in the form of cords, sheets, individual cells, nodules, or cystic structures.7 Pemphigus vegetans is the rarest variant of a group of autoimmune vesiculobullous diseases known as pemphigus. It can be differentiated from the most common variant—pemphigus vulgaris—by the presence of vegetative plaques in intertriginous areas. However, these verrucous vegetations can be misleading and make clinical diagnosis difficult. Histopathologic findings of hyperkeratosis, pseudoepitheliomatous hyperplasia, papillomatosis, and acantholysis with a suprabasal cleft would confirm the diagnosis.8
In summary, cutaneous blastomycosis classically presents as verrucous crusting plaques, as seen in our patient. It is important to conduct a thorough history for environmental exposures, but definitive diagnosis of cutaneous blastomycosis involves skin biopsy with fungal culture. Treatment depends on the severity of disease and organ involvement. Itraconazole would be appropriate for mild to moderate blastomycosis.
- Miceli A, Krishnamurthy K. Blastomycosis. StatPearls. StatPearls Publishing; 2022. Accessed June 21, 2022. https://www.ncbi.nlm.nih.gov/books/NBK441987/
- Gray NA, Baddour LM. Cutaneous inoculation blastomycosis. Clin Infect Dis. 2002;34:E44-E49.
- Schwartz IS, Kauffman CA. Blastomycosis. Semin Respir Crit Care Med. 2020;41:31-41. doi:10.1055/s-0039-3400281
- Castillo CG, Kauffman CA, Miceli MH. Blastomycosis. Infect Dis Clin North Am. 2016;30:247-264. doi:10.1016/j.idc.2015.10.002
- Raggio B. Primary cutaneous histoplasmosis. Ear Nose Throat J. 2018;97:346-348.
- Bhambri S, Bhambri A, Del Rosso JQ. Atypical mycobacterial cutaneous infections. Dermatol Clin. 2009;27:63-73. doi:10.1016/j.det.2008.07.009
- Parekh V, Seykora JT. Cutaneous squamous cell carcinoma. Clin Lab Med. 2017;37:503-525. doi:10.1016/j.cll.2017.06.003
- Messersmith L, Krauland K. Pemphigus vegetans. StatPearls. StatPearls Publishing; 2022. Accessed June 21, 2022. https://www.ncbi.nlm.nih.gov/books/NBK545229/
The Diagnosis: Cutaneous Blastomycosis
A skin biopsy and fungal cultures confirmed the diagnosis of cutaneous blastomycosis. Grocott- Gomori methenamine-silver staining highlighted fungal organisms with refractile walls and broad-based budding consistent with cutaneous blastomycosis (Figure 1). The histopathologic specimen also demonstrated marked pseudoepitheliomatous hyperplasia (Figure 2A) with neutrophilic microabscesses (Figure 2B). Acid-fast bacillus and Fite staining were negative for bacterial organisms. A fungal culture was positive for Blastomyces dermatitidis. Urine and serum blastomycosis antigen were positive. Although Histoplasma serum antigen also was positive, this likely was from cross-reactivity. Chest radiography was negative for lung involvement, and the patient displayed no neurologic symptoms. He was started on oral itraconazole therapy for the treatment of cutaneous blastomycosis.

Blastomyces dermatitidis, the causative organism of blastomycosis, is endemic to the Ohio and Mississippi River valleys, Great Lakes region, and southeastern United States. It is a thermally dimorphic fungus found in soils that grows as a mold at 25 °C and yeast at 37 °C. Primary infection of the lungs—blastomycosis pneumonia—often is the only clinical manifestation1; however, subsequent hematogenous dissemination to extrapulmonary sites such as the skin, bones, and genitourinary system can occur. Cutaneous blastomycosis, the most common extrapulmonary manifestation, typically follows pulmonary infection. In rare cases, it can occur from direct inoculation.2,3 Skin lesions can occur anywhere but frequently are found on exposed surfaces of the head, neck, and extremities. Lesions classically present as verrucous crusting plaques with draining microabscesses. Violaceous nodules, ulcers, and pustules also may occur.1

Diagnosis involves obtaining a thorough history of possible environmental exposures such as the patient’s geographic area of residence, occupation, and outdoor activities involving soil or decaying wood. Because blastomycosis can remain latent, remote exposures are relevant. Definitive diagnosis of cutaneous blastomycosis involves skin biopsy of the lesion with fungal culture, but the yeast’s distinctive thick wall and broad-based budding seen with periodic acid–Schiff or Grocott-Gomori methenamine-silver staining provides a rapid presumptive diagnosis.3 Pseudoepitheliomatous hyperplasia and microabscesses also are characteristic features.2 Urine antigen testing for a component of the polysaccharide cell wall has a sensitivity of 93% but a lower specificity of 79% due to cross-reactivity with histoplasmosis.4 Treatment consists of itraconazole for mild to moderate blastomycosis or amphotericin B for those with severe disease or central nervous system involvement or those who are immunosuppressed.1
The differential diagnosis for our patient’s lesion included infectious vs neoplastic etiologies. Histoplasma capsulatum, the dimorphic fungus that causes histoplasmosis, also is endemic to the Ohio and Mississippi River valleys. It is found in soil and droppings of some bats and birds such as chickens and pigeons. Similar to blastomycosis, the primary infection site most commonly is the lungs. It subsequently may disseminate to the skin or less commonly via direct inoculation of injured skin. It can present as papules, plaques, ulcers, purpura, or abscesses. Unlike blastomycosis, tissue biopsy of a cutaneous lesion reveals granuloma formation and distinctive oval, narrow-based budding yeast.5 Atypical mycobacteria are another source of infection to consider. For example, cutaneous Mycobacterium kansasii may present as papules and pustules forming verrucous or granulomatous plaques and ulceration. Histopathologic findings distinguishing mycobacterial infection from blastomycosis include granulomas and acid-fast bacilli in histiocytes.6
Noninfectious etiologies in the differential may include cutaneous squamous cell carcinoma or pemphigus vegetans. Squamous cell carcinoma may present with a broad range of clinical features—papules, plaques, or nodules with smooth, scaly, verrucous, or ulcerative secondary features all are possible presentations.7 Fairskinned individuals, such as our patient, would be at a higher risk in sun-damaged skin. Histologically, cutaneous squamous cell carcinoma is defined as an invasion of the dermis by neoplastic squamous epithelial cells in the form of cords, sheets, individual cells, nodules, or cystic structures.7 Pemphigus vegetans is the rarest variant of a group of autoimmune vesiculobullous diseases known as pemphigus. It can be differentiated from the most common variant—pemphigus vulgaris—by the presence of vegetative plaques in intertriginous areas. However, these verrucous vegetations can be misleading and make clinical diagnosis difficult. Histopathologic findings of hyperkeratosis, pseudoepitheliomatous hyperplasia, papillomatosis, and acantholysis with a suprabasal cleft would confirm the diagnosis.8
In summary, cutaneous blastomycosis classically presents as verrucous crusting plaques, as seen in our patient. It is important to conduct a thorough history for environmental exposures, but definitive diagnosis of cutaneous blastomycosis involves skin biopsy with fungal culture. Treatment depends on the severity of disease and organ involvement. Itraconazole would be appropriate for mild to moderate blastomycosis.
The Diagnosis: Cutaneous Blastomycosis
A skin biopsy and fungal cultures confirmed the diagnosis of cutaneous blastomycosis. Grocott- Gomori methenamine-silver staining highlighted fungal organisms with refractile walls and broad-based budding consistent with cutaneous blastomycosis (Figure 1). The histopathologic specimen also demonstrated marked pseudoepitheliomatous hyperplasia (Figure 2A) with neutrophilic microabscesses (Figure 2B). Acid-fast bacillus and Fite staining were negative for bacterial organisms. A fungal culture was positive for Blastomyces dermatitidis. Urine and serum blastomycosis antigen were positive. Although Histoplasma serum antigen also was positive, this likely was from cross-reactivity. Chest radiography was negative for lung involvement, and the patient displayed no neurologic symptoms. He was started on oral itraconazole therapy for the treatment of cutaneous blastomycosis.

Blastomyces dermatitidis, the causative organism of blastomycosis, is endemic to the Ohio and Mississippi River valleys, Great Lakes region, and southeastern United States. It is a thermally dimorphic fungus found in soils that grows as a mold at 25 °C and yeast at 37 °C. Primary infection of the lungs—blastomycosis pneumonia—often is the only clinical manifestation1; however, subsequent hematogenous dissemination to extrapulmonary sites such as the skin, bones, and genitourinary system can occur. Cutaneous blastomycosis, the most common extrapulmonary manifestation, typically follows pulmonary infection. In rare cases, it can occur from direct inoculation.2,3 Skin lesions can occur anywhere but frequently are found on exposed surfaces of the head, neck, and extremities. Lesions classically present as verrucous crusting plaques with draining microabscesses. Violaceous nodules, ulcers, and pustules also may occur.1

Diagnosis involves obtaining a thorough history of possible environmental exposures such as the patient’s geographic area of residence, occupation, and outdoor activities involving soil or decaying wood. Because blastomycosis can remain latent, remote exposures are relevant. Definitive diagnosis of cutaneous blastomycosis involves skin biopsy of the lesion with fungal culture, but the yeast’s distinctive thick wall and broad-based budding seen with periodic acid–Schiff or Grocott-Gomori methenamine-silver staining provides a rapid presumptive diagnosis.3 Pseudoepitheliomatous hyperplasia and microabscesses also are characteristic features.2 Urine antigen testing for a component of the polysaccharide cell wall has a sensitivity of 93% but a lower specificity of 79% due to cross-reactivity with histoplasmosis.4 Treatment consists of itraconazole for mild to moderate blastomycosis or amphotericin B for those with severe disease or central nervous system involvement or those who are immunosuppressed.1
The differential diagnosis for our patient’s lesion included infectious vs neoplastic etiologies. Histoplasma capsulatum, the dimorphic fungus that causes histoplasmosis, also is endemic to the Ohio and Mississippi River valleys. It is found in soil and droppings of some bats and birds such as chickens and pigeons. Similar to blastomycosis, the primary infection site most commonly is the lungs. It subsequently may disseminate to the skin or less commonly via direct inoculation of injured skin. It can present as papules, plaques, ulcers, purpura, or abscesses. Unlike blastomycosis, tissue biopsy of a cutaneous lesion reveals granuloma formation and distinctive oval, narrow-based budding yeast.5 Atypical mycobacteria are another source of infection to consider. For example, cutaneous Mycobacterium kansasii may present as papules and pustules forming verrucous or granulomatous plaques and ulceration. Histopathologic findings distinguishing mycobacterial infection from blastomycosis include granulomas and acid-fast bacilli in histiocytes.6
Noninfectious etiologies in the differential may include cutaneous squamous cell carcinoma or pemphigus vegetans. Squamous cell carcinoma may present with a broad range of clinical features—papules, plaques, or nodules with smooth, scaly, verrucous, or ulcerative secondary features all are possible presentations.7 Fairskinned individuals, such as our patient, would be at a higher risk in sun-damaged skin. Histologically, cutaneous squamous cell carcinoma is defined as an invasion of the dermis by neoplastic squamous epithelial cells in the form of cords, sheets, individual cells, nodules, or cystic structures.7 Pemphigus vegetans is the rarest variant of a group of autoimmune vesiculobullous diseases known as pemphigus. It can be differentiated from the most common variant—pemphigus vulgaris—by the presence of vegetative plaques in intertriginous areas. However, these verrucous vegetations can be misleading and make clinical diagnosis difficult. Histopathologic findings of hyperkeratosis, pseudoepitheliomatous hyperplasia, papillomatosis, and acantholysis with a suprabasal cleft would confirm the diagnosis.8
In summary, cutaneous blastomycosis classically presents as verrucous crusting plaques, as seen in our patient. It is important to conduct a thorough history for environmental exposures, but definitive diagnosis of cutaneous blastomycosis involves skin biopsy with fungal culture. Treatment depends on the severity of disease and organ involvement. Itraconazole would be appropriate for mild to moderate blastomycosis.
- Miceli A, Krishnamurthy K. Blastomycosis. StatPearls. StatPearls Publishing; 2022. Accessed June 21, 2022. https://www.ncbi.nlm.nih.gov/books/NBK441987/
- Gray NA, Baddour LM. Cutaneous inoculation blastomycosis. Clin Infect Dis. 2002;34:E44-E49.
- Schwartz IS, Kauffman CA. Blastomycosis. Semin Respir Crit Care Med. 2020;41:31-41. doi:10.1055/s-0039-3400281
- Castillo CG, Kauffman CA, Miceli MH. Blastomycosis. Infect Dis Clin North Am. 2016;30:247-264. doi:10.1016/j.idc.2015.10.002
- Raggio B. Primary cutaneous histoplasmosis. Ear Nose Throat J. 2018;97:346-348.
- Bhambri S, Bhambri A, Del Rosso JQ. Atypical mycobacterial cutaneous infections. Dermatol Clin. 2009;27:63-73. doi:10.1016/j.det.2008.07.009
- Parekh V, Seykora JT. Cutaneous squamous cell carcinoma. Clin Lab Med. 2017;37:503-525. doi:10.1016/j.cll.2017.06.003
- Messersmith L, Krauland K. Pemphigus vegetans. StatPearls. StatPearls Publishing; 2022. Accessed June 21, 2022. https://www.ncbi.nlm.nih.gov/books/NBK545229/
- Miceli A, Krishnamurthy K. Blastomycosis. StatPearls. StatPearls Publishing; 2022. Accessed June 21, 2022. https://www.ncbi.nlm.nih.gov/books/NBK441987/
- Gray NA, Baddour LM. Cutaneous inoculation blastomycosis. Clin Infect Dis. 2002;34:E44-E49.
- Schwartz IS, Kauffman CA. Blastomycosis. Semin Respir Crit Care Med. 2020;41:31-41. doi:10.1055/s-0039-3400281
- Castillo CG, Kauffman CA, Miceli MH. Blastomycosis. Infect Dis Clin North Am. 2016;30:247-264. doi:10.1016/j.idc.2015.10.002
- Raggio B. Primary cutaneous histoplasmosis. Ear Nose Throat J. 2018;97:346-348.
- Bhambri S, Bhambri A, Del Rosso JQ. Atypical mycobacterial cutaneous infections. Dermatol Clin. 2009;27:63-73. doi:10.1016/j.det.2008.07.009
- Parekh V, Seykora JT. Cutaneous squamous cell carcinoma. Clin Lab Med. 2017;37:503-525. doi:10.1016/j.cll.2017.06.003
- Messersmith L, Krauland K. Pemphigus vegetans. StatPearls. StatPearls Publishing; 2022. Accessed June 21, 2022. https://www.ncbi.nlm.nih.gov/books/NBK545229/
A 39-year-old man from Ohio presented with a tender, 10×6-cm, fungated, eroded plaque on the right medial upper arm that developed over the last 4 years. He initially noticed a firm lump under the right arm 4 years prior that was diagnosed as possible cellulitis at an outside clinic and treated with trimethoprim-sulfamethoxazole. The lesion then began to erode and became a chronic nonhealing wound. Approximately 1 year prior to the current presentation, the patient recalled unloading a truckload of soil around the same time the wound began to enlarge in diameter and depth. He denied any prior or current respiratory or systemic symptoms including fevers, chills, or weight loss.
How to overcome hesitancy for COVID-19 and other vaccines
The World Health Organization (WHO) named vaccine hesitancy as one of the top 10 threats to public health as of 2019.1 Although the COVID-19 vaccines manufactured by Pfizer-BioNTech and Moderna, first authorized for use in November 2020 and fully approved in August 2021,2 are widely available in most countries, vaccination uptake is insufficient.3
As of June 2022, 78% of the US population had received at least 1 vaccine dose and 66.8% were fully vaccinated against COVID-19.4 High confidence in vaccines is associated with greater uptake; thus, engendering confidence in patients is a critical area of intervention for increasing uptake of COVID-19 and other vaccines.5 Despite the steady increase in vaccine acceptance observed following the release of the COVID-19 vaccine, acceptance remains suboptimal.2,6
Demographic characteristics associated with lower vaccine acceptance include younger age, female sex, lower education and/or income, and Black race or Hispanic/Latinx ethnicity (compared to white or Asian non-Hispanic).6,7 Moreover, patients who are skeptical of vaccine safety and efficacy are associated with lower intentions to vaccinate. In contrast, patients with a history of receiving influenza vaccinations and those with a greater concern about COVID-19 and their risk of infection have increased vaccine intentions.6
Numerous strategies exist to increase vaccine acceptance; however, there does not appear to be a single “best” method to overcome individual or parental vaccine hesitancy for COVID-19 or other vaccines.8,9 There are no large-scale randomized controlled trials (RCTs) demonstrating one strategy as more effective than another. In this review, we outline a variety of evidenced-based strategies to help patients overcome vaccine hesitancy for COVID-19 and other vaccines, with a focus on practical tips for primary care physicians (PCPs).
Which talking points are likely to resonate with your patients?
Intervention strategies promote vaccine acceptance by communicating personal benefit, collective benefit, or both to vaccine-hesitant patients. In a study sample of US undergraduate students, Kim and colleagues10 found that providing information about the benefits and risks of influenza vaccines resulted in significantly less vaccine intent compared to communicating information only on the benefits. Similarly, Shim and colleagues11 investigated how game theory (acting to maximize personal payoff regardless of payoff to others) and altruism affect influenza vaccination decisions. Through a survey-based study of 427 US university employees, researchers found altruistic motivation had a significant impact on the decision to vaccinate against influenza, resulting in a shift from self-interest to that of the good of the community.11
A German trial on COVID-19 vaccine acceptance by Sprengholz and colleagues12 found that communications about the benefits of vaccination, availability of financial compensation for vaccination, or a combination of both, did not increase a person’s willingness to get vaccinated. This trial, however, did not separate out individual vs collective benefit, and it was conducted prior to widespread COVID-19 vaccine availability.
In an online RCT conducted in early 2021, Freeman and colleagues13 randomized UK adults to 1 of 10 different “information conditions.” Participants read from 1 of 10 vaccine scripts that varied by the talking points they addressed. The topics that researchers drew from for these scripts included the personal or collective benefit from the COVID-19 vaccine, safety and effectiveness of the vaccine, and the seriousness of the pandemic. They found communications emphasizing personal benefit from vaccination and safety concerns were more effective in participants identified as being strongly hesitant (defined as those who said they would avoid getting the COVID-19 vaccine for as long as possible or who said they’d never get it). However, none of the information arms in this study decreased vaccine hesitancy among those who were doubtful of vaccination (defined as those who said they would delay vaccination or who didn’t know if they would get vaccinated).13
Continue to: When encountering patients who are strongly...
When encountering patients who are strongly hesitant to vaccination, an approach emphasizing concrete personal benefit may prove more effective than one stressing protection of others from illness. It is important to note, though, that findings from other countries may not be relevant to US patients due to differences in demographic factors, individual beliefs, and political climate.
It helps to explain herd immunity by providing concrete examples
Among the collective benefits of vaccination is the decreased risk of transmitting the disease to others (eg, family, friends, neighbors, colleagues), a quicker “return to normalcy,” and herd immunity.13 While individual health benefits may more strongly motivate people to get vaccinated than collective benefits, this may be due to a lack of understanding about herd immunity among the general public. The optimal method of communicating information on herd immunity is not known.14
Betsch and colleagues15 found that explaining herd immunity using interactive simulations increased vaccine intent, especially in countries that prioritize the self (rather than prioritizing the group over the individual). In addition to educating study participants about herd immunity, telling them how local vaccine coverage compared to the desired level of coverage helped to increase (influenza) vaccine intent among those who were least informed about herd immunity.16
Providing concrete examples of the collective benefits of vaccination (eg, protecting grandparents, children too young to be vaccinated, and those at increased risk for severe illness) or sharing stories about how other patients suffered from the disease in question may increase the likelihood of vaccination. One recent trial by Pfattheicher and colleagues17 found that empathy for those most vulnerable to COVID-19 and increased knowledge about herd immunity were 2 factors associated with greater vaccine intentions.
In this study, the authors induced empathy and increased COVID-19 vaccination intention by having participants read a short story about 2 close siblings who worked together in a nursing facility. In the story, participants learned that both siblings were given a diagnosis of COVID-19 at the same time but only 1 survived.17
Continue to: Try this 3-pronged approach
Try this 3-pronged approach. Consider explaining herd immunity to vaccine-hesitant patients, pairing this concept with information about local vaccine uptake, and appealing to the patient’s sense of empathy. You might share de-identified information on other patients in your practice or personal network who experienced severe illness, had long-term effects, or died from COVID-19 infection. Such concrete examples may help to increase motivation to vaccinate more than a general appeal to altruism.
Initiate the discussion by emphasizing that community immunity protects those who are vulnerable and lack immunity while providing specific empathetic examples (eg, newborns, cancer survivors) and asking patients to consider friends and family who might be at risk. Additionally, it is essential to explain that although community immunity can decrease the spread of infection, it can only be achieved when enough people are vaccinated.
Proceed with caution: Addressing conspiracy theories can backfire
Accurate information is critical to improving vaccine intentions; belief in conspiracy theories or misinformation related to COVID-19 is associated with reduced vaccine intentions and uptake.6 For example, a study by Loomba and colleagues18 showed that after exposure to misinformation, US and UK adults reported reduced intentions to vaccinate against COVID-19 once a vaccine became available.
Unfortunately, addressing myths about vaccines can sometimes backfire and unintentionally reinforce vaccine misperceptions.19,20 This is especially true for patients with the highest levels of concern or mistrust in vaccines. Nyhan and colleagues21,22 observed the backfire effect in 2 US studies looking at influenza and measles, mumps, and rubella vaccine misperceptions. Although corrective information significantly reduced belief in vaccine myths, they found individuals with the most concerns more strongly endorsed misperceptions when their beliefs were challenged.21,22
An Australian randomized study by Steffens and colleagues23 found repeating myths about childhood vaccines, followed by corrective text, to parents of children ages 0 to 5 years had no difference on parental intent to vaccinate their children compared to providing vaccine information as a statement or in a question/answer format. Furthermore, an RCT in Brazil by Carey and colleagues24 found that myth-correction messages about Zika virus failed to reduce misperceptions about the virus and actually reduced the belief in factual information about Zika—regardless of baseline beliefs in conspiracies. However, a similar experiment in the same study showed that myth-correction messages reduced false beliefs about yellow fever.
Continue to: The authors speculated...
The authors speculated that this may be because Zika is a relatively new virus when compared to yellow fever, and participants may have more pre-existing knowledge about yellow fever.24 These findings are important to keep in mind when addressing misinformation regarding COVID-19. When addressing myth perceptions with patients, consider pivoting the conversation from vaccine myths to the disease itself, focusing on the disease risk and severity of symptoms.19,20
Other studies have had positive results when addressing misinformation, including a digital RCT of older adults in the Netherlands by Yousuf and colleagues.25 In this study, participants were randomized to view 1 of 2 versions of an information video on vaccination featuring an informative discussion by celebrity scientists, government officials, and a cardiologist. Video 1 did not include debunking strategies, only information about vaccination; Video 2 provided the same information about vaccines but also described the myths surrounding vaccines and reiterated the truth to debunk the myths.
Findings demonstrated that a significantly higher number of participants in the Video 2 group overcame vaccination myths related to influenza and COVID-19.25 Notably, this study took place prior to the widespread availability of COVID-19 vaccines and did not measure intent to vaccinate against COVID-19.
Taken together, strategies for correcting vaccine misinformation may vary by population as well as type of vaccine; however, placing emphasis on facts delivered by trusted sources appears to be beneficial. When addressing misinformation, PCPs should first focus on key details (not all supporting information) and clearly explain why the misinformation is false before pointing out the actual myth and providing an alternative explanation.20 When caring for patients who express strong concerns over the vaccine in question or have avid beliefs in certain myths or conspiracy theories, it’s best to pivot the conversation back to the disease rather than address the misinformation to avoid a potential backfire effect.
Utilize these effective communication techniques
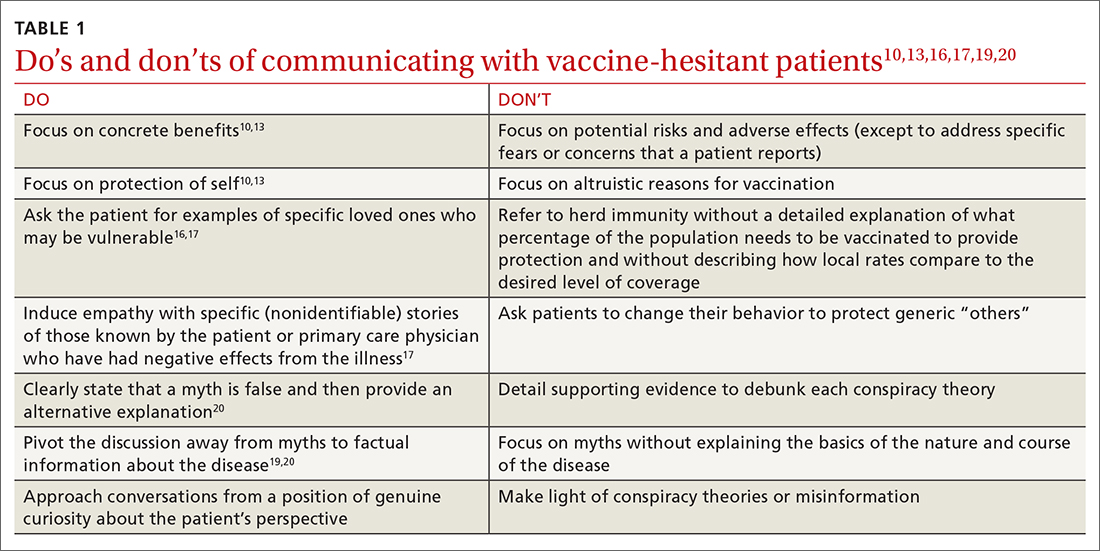
TABLE 110,13,16,17,19,20 summarizes the “do’s and don’ts” of communicating with vaccine-hesitant patients. PCPs should provide strong recommendations for vaccination, approaching it presumptively—ie, framing it as normative behavior.19,26 This approach is critical to building patient trust so that vaccine-hesitant patients feel the PCP is truly listening to them and addressing their concerns.27 Additionally, implementing motivational interviewing (MI) and self-determination theory (SDT)28 techniques when discussing vaccinations with patients can improve intentions and uptake.19,29TABLE 219,29 outlines specific techniques based on SDT and MI that PCPs may utilize to communicate with vaccine-hesitant individuals or parents.
Continue to: The takeaway
The takeaway
Strategies for increasing vaccine intentions include educating hesitant patients about the benefits and risks of vaccines, addressing misinformation, and explaining the personal and collective benefits of vaccination. These strategies appear to be more effective when delivered by a trusted source, such as a health care provider (HCP). Care should be taken when implementing vaccine-acceptance strategies to ensure that they are tailored to specific populations and vaccines.
At this stage in the COVID-19 pandemic, when several vaccines have been widely available for more than a year, we expect that the majority of patients desiring vaccination (ie, those with the greatest vaccine intent) have already received them. With the recent approval of COVID-19 vaccines for children younger than 5 years, we must now advocate for our patients to vaccinate not only themselves, but their children. Patients who remain unvaccinated may be hesitant or outright reject vaccination for a number of reasons, including fear or skepticism over the safety and efficacy of the vaccine, belief in conspiracy theories, belief that COVID-19 is not real or not severe, or mistrust of the government.6 Vaccine hesitation or rejection is also often political in nature.
Based on the studies included in this review, we have identified several strategies for reducing vaccine hesitancy, which can be used with vaccine-hesitant patients and parents. We suggest emphasizing the personal benefit of vaccination and focusing on specific disease risks. If time allows, you can also explain the collective benefit of vaccination through herd immunity, including the current levels of local vaccine uptake compared to the desired level for community immunity. Communicating the collective benefits of vaccination may be more effective when paired with a strategy intended to increase empathy and altruism, such as sharing actual stories about those who have suffered from a vaccine-preventable disease.
Addressing myths and misinformation related to COVID-19 and other vaccines, with emphasis placed on the correct information delivered by trusted sources may be beneficial for those who are uncertain but not strongly against vaccination. For those who remain staunchly hesitant against vaccination, we recommend focusing on the personal benefits of vaccination with a focus on delivering facts about the risk of the disease in question, rather than trying to refute misinformation.
COVID-19 vaccine acceptance in the United States is disturbingly low among health care workers, particularly nurses, technicians, and those in nonclinical roles, compared to physicians.6,30 Many of the strategies for addressing vaccine hesitancy among the general population can also apply to health care personnel (eg, vaccine education, addressing misinformation, delivering information from a trusted source). Health care personnel may also be subject to vaccine mandates by their employers, which have demonstrated increases in vaccination rates for influenza.31 Given that COVID-19 vaccination recommendations made by HCPs are associated with greater vaccine intentions and uptake,6 reducing hesitancy among health care workers is a critical first step to achieving optimal implementation.
CORRESPONDENCE
Nicole Mayo, PhD, 236 Pearl Street, Rochester, NY 14607; Nicole_Mayo@URMC.Rochester.edu
1. Ten threats to global health in 2019. World Health Organization. Accessed June 17, 2022. www.who.int/news-room/spotlight/ten-threats-to-global-health-in-2019
2. FDA approves first COVID-19 vaccine. US Food and Drug Administration. August 23, 2021. Accessed June 17, 2022. www.fda.gov/news-events/press-announcements/fda-approves-first-covid-19-vaccine
3. Mathieu E, Ritchie H, Ortiz-Ospina E, et al. A global database of COVID-19 vaccinations. Nat Hum Behav. 2021;5:947-953. doi: 10.1038/s41562-021-01122-8.
4. Ritchie H, Mathieu E, Rodés-Guirao L, et al. Coronavirus pandemic (COVID-19). Our world in data. Accessed June 17, 2022. https://ourworldindata.org/covid-vaccinations?country=USA
5. de Figueiredo A, Simas C, Karafillakis E, et al. Mapping global trends in vaccine confidence and investigating barriers to vaccine uptake: a large-scale retrospective temporal modelling study. Lancet. 2020;396:898-908. doi: 10.1016/S0140-6736(20)31558-0
6. Wang Y, Liu Y. Multilevel determinants of COVID-19 vaccination hesitancy in the United States: a rapid systematic review. Prev Med Rep. 2021;25:101673. doi: 10.1016/j.pmedr.2021.101673
7. Robinson E, Jones A, Lesser I, et al. International estimates of intended uptake and refusal of COVID-19 vaccines: a rapid systematic review and meta-analysis of large nationally representative samples. Vaccine. 2021;39:2024-2034. doi: 10.1016/j.vaccine.2021.02.005
8. Dubé E, Gagnon D, MacDonald NE; SAGE Working Group on Vaccine Hesitancy. Strategies intended to address vaccine hesitancy: review of published reviews. Vaccine. 2015;33:4191-4203. doi: 10.1016/j.vaccine.2015.04.041
9. Sadaf A, Richards JL, Glanz J, et al. A systematic review of interventions for reducing parental vaccine refusal and vaccine hesitancy. Vaccine. 2013;31:4293-4304. doi: 10.1016/j.vaccine.2013.07.013
10. Kim S, Pjesivac I, Jin Y. Effects of message framing on influenza vaccination: understanding the role of risk disclosure, perceived vaccine efficacy, and felt ambivalence. Health Commun. 2019;34:21-30. doi: 10.1080/10410236.2017.1384353
11. Shim E, Chapman GB, Townsend JP, et al. The influence of altruism on influenza vaccination decisions. J R Soc Interface. 2012;9:2234-2243. doi: 10.1098/rsif.2012.0115
12. Sprengholz P, Eitze S, Felgendreff L, et al. Money is not everything: experimental evidence that payments do not increase willingness to be vaccinated against COVID-19. J Med Ethics. 2021;47:547-548. doi: 10.1136/medethics-2020-107122
13. Freeman D, Loe BS, Yu LM, et al. Effects of different types of written vaccination information on COVID-19 vaccine hesitancy in the UK (OCEANS-III): a single-blind, parallel-group, randomised controlled trial. Lancet Public Health. 2021;6:e416-e427. doi: 10.1016/S2468-2667(21)00096-7
14. Hakim H, Provencher T, Chambers CT, et al. Interventions to help people understand community immunity: a systematic review. Vaccine. 2019;37:235-247. doi: 10.1016/j.vaccine.2018.11.016
15. Betsch C, Böhm R, Korn L, et al. On the benefits of explaining herd immunity in vaccine advocacy. Nat Hum Behav. 2017;1:1-6. doi: 10.1038/s41562-017-0056
16. Logan J, Nederhoff D, Koch B, et al. ‘What have you HEARD about the HERD?’ Does education about local influenza vaccination coverage and herd immunity affect willingness to vaccinate? Vaccine. 2018;36:4118-4125. doi: 10.1016/j.vaccine.2018.05.037
17. Pfattheicher S, Petersen MB, Böhm R. Information about herd immunity through vaccination and empathy promote COVID-19 vaccination intentions. Health Psychol. 2022;41:85-93. doi: 10.1037/hea0001096
18. Loomba S, de Figueiredo A, Piatek SJ, et al. Measuring the impact of COVID-19 vaccine misinformation on vaccination intent in the UK and USA. Nat Hum Behav. 2021;5:337-348. doi: 10.1038/s41562-021-01056-1
19. Limaye RJ, Opel DJ, Dempsey A, et al. Communicating with vaccine-hesitant parents: a narrative review. Acad Pediatr. 2021;21:S24-S29. doi: 10.1016/j.acap.2021.01.018
20. Omer SB, Amin AB, Limaye RJ. Communicating about vaccines in a fact-resistant world. JAMA Pediatr. 2017;171:929-930. doi: 10.1001/jamapediatrics.2017.2219
21. Nyhan B, Reifler J. Does correcting myths about the flu vaccine work? An experimental evaluation of the effects of corrective information. Vaccine. 2015;33:459-464. doi: 10.1016/j.vaccine.2014.11.017
22. Nyhan B, Reifler J, Richey S, et al. Effective messages in vaccine promotion: a randomized trial. Pediatrics. 2014;133:e835-e842. doi: 10.1542/peds.2013-2365
23. Steffens MS, Dunn AG, Marques MD, et al. Addressing myths and vaccine hesitancy: a randomized trial. Pediatrics. 2021;148:e2020049304. doi: 10.1542/peds.2020-049304
24. Carey JM, Chi V, Flynn DJ, et al. The effects of corrective information about disease epidemics and outbreaks: evidence from Zika and yellow fever in Brazil. Sci Adv. 2020;6:eaaw7449. doi: 10.1126/sciadv.aaw7449
25. Yousuf H, van der Linden S, Bredius L, et al. A media intervention applying debunking versus non-debunking content to combat vaccine misinformation in elderly in the Netherlands: a digital randomised trial. EClinicalMedicine. 2021;35:100881. doi: 10.1016/j.eclinm.2021.100881
26. Cambon L, Schwarzinger M, Alla F. Increasing acceptance of a vaccination program for coronavirus disease 2019 in France: a challenge for one of the world’s most vaccine-hesitant countries. Vaccine. 2022;40:178-182. doi: 10.1016/j.vaccine.2021.11.023
27. Leask J, Kinnersley P, Jackson C, et al. Communicating with parents about vaccination: a framework for health professionals. BMC Pediatr. 2012;12:154. doi: 10.1186/1471-2431-12-154
28. Martela F, Hankonen N, Ryan RM, et al. Motivating voluntary compliance to behavioural restrictions: self-determination theory–based checklist of principles for COVID-19 and other emergency communications. Eur Rev Soc Psychol. 2021:305-347. doi: 10.1080/10463283.2020.1857082
29. Boness CL, Nelson M, Douaihy AB. Motivational interviewing strategies for addressing COVID-19 vaccine hesitancy. J Am Board Fam Med. 2022;35:420-426. doi: 10.3122/jabfm.2022.02.210327
30. Salomoni MG, Di Valerio Z, Gabrielli E, et al. Hesitant or not hesitant? A systematic review on global COVID-19 vaccine acceptance in different populations. Vaccines (Basel). 2021;9:873. doi: 10.3390/vaccines9080873
31. Pitts SI, Maruthur NM, Millar KR, et al. A systematic review of mandatory influenza vaccination in healthcare personnel. Am J Prev Med. 2014;47:330-340. doi:
The World Health Organization (WHO) named vaccine hesitancy as one of the top 10 threats to public health as of 2019.1 Although the COVID-19 vaccines manufactured by Pfizer-BioNTech and Moderna, first authorized for use in November 2020 and fully approved in August 2021,2 are widely available in most countries, vaccination uptake is insufficient.3
As of June 2022, 78% of the US population had received at least 1 vaccine dose and 66.8% were fully vaccinated against COVID-19.4 High confidence in vaccines is associated with greater uptake; thus, engendering confidence in patients is a critical area of intervention for increasing uptake of COVID-19 and other vaccines.5 Despite the steady increase in vaccine acceptance observed following the release of the COVID-19 vaccine, acceptance remains suboptimal.2,6
Demographic characteristics associated with lower vaccine acceptance include younger age, female sex, lower education and/or income, and Black race or Hispanic/Latinx ethnicity (compared to white or Asian non-Hispanic).6,7 Moreover, patients who are skeptical of vaccine safety and efficacy are associated with lower intentions to vaccinate. In contrast, patients with a history of receiving influenza vaccinations and those with a greater concern about COVID-19 and their risk of infection have increased vaccine intentions.6
Numerous strategies exist to increase vaccine acceptance; however, there does not appear to be a single “best” method to overcome individual or parental vaccine hesitancy for COVID-19 or other vaccines.8,9 There are no large-scale randomized controlled trials (RCTs) demonstrating one strategy as more effective than another. In this review, we outline a variety of evidenced-based strategies to help patients overcome vaccine hesitancy for COVID-19 and other vaccines, with a focus on practical tips for primary care physicians (PCPs).
Which talking points are likely to resonate with your patients?
Intervention strategies promote vaccine acceptance by communicating personal benefit, collective benefit, or both to vaccine-hesitant patients. In a study sample of US undergraduate students, Kim and colleagues10 found that providing information about the benefits and risks of influenza vaccines resulted in significantly less vaccine intent compared to communicating information only on the benefits. Similarly, Shim and colleagues11 investigated how game theory (acting to maximize personal payoff regardless of payoff to others) and altruism affect influenza vaccination decisions. Through a survey-based study of 427 US university employees, researchers found altruistic motivation had a significant impact on the decision to vaccinate against influenza, resulting in a shift from self-interest to that of the good of the community.11
A German trial on COVID-19 vaccine acceptance by Sprengholz and colleagues12 found that communications about the benefits of vaccination, availability of financial compensation for vaccination, or a combination of both, did not increase a person’s willingness to get vaccinated. This trial, however, did not separate out individual vs collective benefit, and it was conducted prior to widespread COVID-19 vaccine availability.
In an online RCT conducted in early 2021, Freeman and colleagues13 randomized UK adults to 1 of 10 different “information conditions.” Participants read from 1 of 10 vaccine scripts that varied by the talking points they addressed. The topics that researchers drew from for these scripts included the personal or collective benefit from the COVID-19 vaccine, safety and effectiveness of the vaccine, and the seriousness of the pandemic. They found communications emphasizing personal benefit from vaccination and safety concerns were more effective in participants identified as being strongly hesitant (defined as those who said they would avoid getting the COVID-19 vaccine for as long as possible or who said they’d never get it). However, none of the information arms in this study decreased vaccine hesitancy among those who were doubtful of vaccination (defined as those who said they would delay vaccination or who didn’t know if they would get vaccinated).13
Continue to: When encountering patients who are strongly...
When encountering patients who are strongly hesitant to vaccination, an approach emphasizing concrete personal benefit may prove more effective than one stressing protection of others from illness. It is important to note, though, that findings from other countries may not be relevant to US patients due to differences in demographic factors, individual beliefs, and political climate.
It helps to explain herd immunity by providing concrete examples
Among the collective benefits of vaccination is the decreased risk of transmitting the disease to others (eg, family, friends, neighbors, colleagues), a quicker “return to normalcy,” and herd immunity.13 While individual health benefits may more strongly motivate people to get vaccinated than collective benefits, this may be due to a lack of understanding about herd immunity among the general public. The optimal method of communicating information on herd immunity is not known.14
Betsch and colleagues15 found that explaining herd immunity using interactive simulations increased vaccine intent, especially in countries that prioritize the self (rather than prioritizing the group over the individual). In addition to educating study participants about herd immunity, telling them how local vaccine coverage compared to the desired level of coverage helped to increase (influenza) vaccine intent among those who were least informed about herd immunity.16
Providing concrete examples of the collective benefits of vaccination (eg, protecting grandparents, children too young to be vaccinated, and those at increased risk for severe illness) or sharing stories about how other patients suffered from the disease in question may increase the likelihood of vaccination. One recent trial by Pfattheicher and colleagues17 found that empathy for those most vulnerable to COVID-19 and increased knowledge about herd immunity were 2 factors associated with greater vaccine intentions.
In this study, the authors induced empathy and increased COVID-19 vaccination intention by having participants read a short story about 2 close siblings who worked together in a nursing facility. In the story, participants learned that both siblings were given a diagnosis of COVID-19 at the same time but only 1 survived.17
Continue to: Try this 3-pronged approach
Try this 3-pronged approach. Consider explaining herd immunity to vaccine-hesitant patients, pairing this concept with information about local vaccine uptake, and appealing to the patient’s sense of empathy. You might share de-identified information on other patients in your practice or personal network who experienced severe illness, had long-term effects, or died from COVID-19 infection. Such concrete examples may help to increase motivation to vaccinate more than a general appeal to altruism.
Initiate the discussion by emphasizing that community immunity protects those who are vulnerable and lack immunity while providing specific empathetic examples (eg, newborns, cancer survivors) and asking patients to consider friends and family who might be at risk. Additionally, it is essential to explain that although community immunity can decrease the spread of infection, it can only be achieved when enough people are vaccinated.
Proceed with caution: Addressing conspiracy theories can backfire
Accurate information is critical to improving vaccine intentions; belief in conspiracy theories or misinformation related to COVID-19 is associated with reduced vaccine intentions and uptake.6 For example, a study by Loomba and colleagues18 showed that after exposure to misinformation, US and UK adults reported reduced intentions to vaccinate against COVID-19 once a vaccine became available.
Unfortunately, addressing myths about vaccines can sometimes backfire and unintentionally reinforce vaccine misperceptions.19,20 This is especially true for patients with the highest levels of concern or mistrust in vaccines. Nyhan and colleagues21,22 observed the backfire effect in 2 US studies looking at influenza and measles, mumps, and rubella vaccine misperceptions. Although corrective information significantly reduced belief in vaccine myths, they found individuals with the most concerns more strongly endorsed misperceptions when their beliefs were challenged.21,22
An Australian randomized study by Steffens and colleagues23 found repeating myths about childhood vaccines, followed by corrective text, to parents of children ages 0 to 5 years had no difference on parental intent to vaccinate their children compared to providing vaccine information as a statement or in a question/answer format. Furthermore, an RCT in Brazil by Carey and colleagues24 found that myth-correction messages about Zika virus failed to reduce misperceptions about the virus and actually reduced the belief in factual information about Zika—regardless of baseline beliefs in conspiracies. However, a similar experiment in the same study showed that myth-correction messages reduced false beliefs about yellow fever.
Continue to: The authors speculated...
The authors speculated that this may be because Zika is a relatively new virus when compared to yellow fever, and participants may have more pre-existing knowledge about yellow fever.24 These findings are important to keep in mind when addressing misinformation regarding COVID-19. When addressing myth perceptions with patients, consider pivoting the conversation from vaccine myths to the disease itself, focusing on the disease risk and severity of symptoms.19,20
Other studies have had positive results when addressing misinformation, including a digital RCT of older adults in the Netherlands by Yousuf and colleagues.25 In this study, participants were randomized to view 1 of 2 versions of an information video on vaccination featuring an informative discussion by celebrity scientists, government officials, and a cardiologist. Video 1 did not include debunking strategies, only information about vaccination; Video 2 provided the same information about vaccines but also described the myths surrounding vaccines and reiterated the truth to debunk the myths.
Findings demonstrated that a significantly higher number of participants in the Video 2 group overcame vaccination myths related to influenza and COVID-19.25 Notably, this study took place prior to the widespread availability of COVID-19 vaccines and did not measure intent to vaccinate against COVID-19.
Taken together, strategies for correcting vaccine misinformation may vary by population as well as type of vaccine; however, placing emphasis on facts delivered by trusted sources appears to be beneficial. When addressing misinformation, PCPs should first focus on key details (not all supporting information) and clearly explain why the misinformation is false before pointing out the actual myth and providing an alternative explanation.20 When caring for patients who express strong concerns over the vaccine in question or have avid beliefs in certain myths or conspiracy theories, it’s best to pivot the conversation back to the disease rather than address the misinformation to avoid a potential backfire effect.
Utilize these effective communication techniques
TABLE 110,13,16,17,19,20 summarizes the “do’s and don’ts” of communicating with vaccine-hesitant patients. PCPs should provide strong recommendations for vaccination, approaching it presumptively—ie, framing it as normative behavior.19,26 This approach is critical to building patient trust so that vaccine-hesitant patients feel the PCP is truly listening to them and addressing their concerns.27 Additionally, implementing motivational interviewing (MI) and self-determination theory (SDT)28 techniques when discussing vaccinations with patients can improve intentions and uptake.19,29TABLE 219,29 outlines specific techniques based on SDT and MI that PCPs may utilize to communicate with vaccine-hesitant individuals or parents.
Continue to: The takeaway
The takeaway
Strategies for increasing vaccine intentions include educating hesitant patients about the benefits and risks of vaccines, addressing misinformation, and explaining the personal and collective benefits of vaccination. These strategies appear to be more effective when delivered by a trusted source, such as a health care provider (HCP). Care should be taken when implementing vaccine-acceptance strategies to ensure that they are tailored to specific populations and vaccines.
At this stage in the COVID-19 pandemic, when several vaccines have been widely available for more than a year, we expect that the majority of patients desiring vaccination (ie, those with the greatest vaccine intent) have already received them. With the recent approval of COVID-19 vaccines for children younger than 5 years, we must now advocate for our patients to vaccinate not only themselves, but their children. Patients who remain unvaccinated may be hesitant or outright reject vaccination for a number of reasons, including fear or skepticism over the safety and efficacy of the vaccine, belief in conspiracy theories, belief that COVID-19 is not real or not severe, or mistrust of the government.6 Vaccine hesitation or rejection is also often political in nature.
Based on the studies included in this review, we have identified several strategies for reducing vaccine hesitancy, which can be used with vaccine-hesitant patients and parents. We suggest emphasizing the personal benefit of vaccination and focusing on specific disease risks. If time allows, you can also explain the collective benefit of vaccination through herd immunity, including the current levels of local vaccine uptake compared to the desired level for community immunity. Communicating the collective benefits of vaccination may be more effective when paired with a strategy intended to increase empathy and altruism, such as sharing actual stories about those who have suffered from a vaccine-preventable disease.
Addressing myths and misinformation related to COVID-19 and other vaccines, with emphasis placed on the correct information delivered by trusted sources may be beneficial for those who are uncertain but not strongly against vaccination. For those who remain staunchly hesitant against vaccination, we recommend focusing on the personal benefits of vaccination with a focus on delivering facts about the risk of the disease in question, rather than trying to refute misinformation.
COVID-19 vaccine acceptance in the United States is disturbingly low among health care workers, particularly nurses, technicians, and those in nonclinical roles, compared to physicians.6,30 Many of the strategies for addressing vaccine hesitancy among the general population can also apply to health care personnel (eg, vaccine education, addressing misinformation, delivering information from a trusted source). Health care personnel may also be subject to vaccine mandates by their employers, which have demonstrated increases in vaccination rates for influenza.31 Given that COVID-19 vaccination recommendations made by HCPs are associated with greater vaccine intentions and uptake,6 reducing hesitancy among health care workers is a critical first step to achieving optimal implementation.
CORRESPONDENCE
Nicole Mayo, PhD, 236 Pearl Street, Rochester, NY 14607; Nicole_Mayo@URMC.Rochester.edu
The World Health Organization (WHO) named vaccine hesitancy as one of the top 10 threats to public health as of 2019.1 Although the COVID-19 vaccines manufactured by Pfizer-BioNTech and Moderna, first authorized for use in November 2020 and fully approved in August 2021,2 are widely available in most countries, vaccination uptake is insufficient.3
As of June 2022, 78% of the US population had received at least 1 vaccine dose and 66.8% were fully vaccinated against COVID-19.4 High confidence in vaccines is associated with greater uptake; thus, engendering confidence in patients is a critical area of intervention for increasing uptake of COVID-19 and other vaccines.5 Despite the steady increase in vaccine acceptance observed following the release of the COVID-19 vaccine, acceptance remains suboptimal.2,6
Demographic characteristics associated with lower vaccine acceptance include younger age, female sex, lower education and/or income, and Black race or Hispanic/Latinx ethnicity (compared to white or Asian non-Hispanic).6,7 Moreover, patients who are skeptical of vaccine safety and efficacy are associated with lower intentions to vaccinate. In contrast, patients with a history of receiving influenza vaccinations and those with a greater concern about COVID-19 and their risk of infection have increased vaccine intentions.6
Numerous strategies exist to increase vaccine acceptance; however, there does not appear to be a single “best” method to overcome individual or parental vaccine hesitancy for COVID-19 or other vaccines.8,9 There are no large-scale randomized controlled trials (RCTs) demonstrating one strategy as more effective than another. In this review, we outline a variety of evidenced-based strategies to help patients overcome vaccine hesitancy for COVID-19 and other vaccines, with a focus on practical tips for primary care physicians (PCPs).
Which talking points are likely to resonate with your patients?
Intervention strategies promote vaccine acceptance by communicating personal benefit, collective benefit, or both to vaccine-hesitant patients. In a study sample of US undergraduate students, Kim and colleagues10 found that providing information about the benefits and risks of influenza vaccines resulted in significantly less vaccine intent compared to communicating information only on the benefits. Similarly, Shim and colleagues11 investigated how game theory (acting to maximize personal payoff regardless of payoff to others) and altruism affect influenza vaccination decisions. Through a survey-based study of 427 US university employees, researchers found altruistic motivation had a significant impact on the decision to vaccinate against influenza, resulting in a shift from self-interest to that of the good of the community.11
A German trial on COVID-19 vaccine acceptance by Sprengholz and colleagues12 found that communications about the benefits of vaccination, availability of financial compensation for vaccination, or a combination of both, did not increase a person’s willingness to get vaccinated. This trial, however, did not separate out individual vs collective benefit, and it was conducted prior to widespread COVID-19 vaccine availability.
In an online RCT conducted in early 2021, Freeman and colleagues13 randomized UK adults to 1 of 10 different “information conditions.” Participants read from 1 of 10 vaccine scripts that varied by the talking points they addressed. The topics that researchers drew from for these scripts included the personal or collective benefit from the COVID-19 vaccine, safety and effectiveness of the vaccine, and the seriousness of the pandemic. They found communications emphasizing personal benefit from vaccination and safety concerns were more effective in participants identified as being strongly hesitant (defined as those who said they would avoid getting the COVID-19 vaccine for as long as possible or who said they’d never get it). However, none of the information arms in this study decreased vaccine hesitancy among those who were doubtful of vaccination (defined as those who said they would delay vaccination or who didn’t know if they would get vaccinated).13
Continue to: When encountering patients who are strongly...
When encountering patients who are strongly hesitant to vaccination, an approach emphasizing concrete personal benefit may prove more effective than one stressing protection of others from illness. It is important to note, though, that findings from other countries may not be relevant to US patients due to differences in demographic factors, individual beliefs, and political climate.
It helps to explain herd immunity by providing concrete examples
Among the collective benefits of vaccination is the decreased risk of transmitting the disease to others (eg, family, friends, neighbors, colleagues), a quicker “return to normalcy,” and herd immunity.13 While individual health benefits may more strongly motivate people to get vaccinated than collective benefits, this may be due to a lack of understanding about herd immunity among the general public. The optimal method of communicating information on herd immunity is not known.14
Betsch and colleagues15 found that explaining herd immunity using interactive simulations increased vaccine intent, especially in countries that prioritize the self (rather than prioritizing the group over the individual). In addition to educating study participants about herd immunity, telling them how local vaccine coverage compared to the desired level of coverage helped to increase (influenza) vaccine intent among those who were least informed about herd immunity.16
Providing concrete examples of the collective benefits of vaccination (eg, protecting grandparents, children too young to be vaccinated, and those at increased risk for severe illness) or sharing stories about how other patients suffered from the disease in question may increase the likelihood of vaccination. One recent trial by Pfattheicher and colleagues17 found that empathy for those most vulnerable to COVID-19 and increased knowledge about herd immunity were 2 factors associated with greater vaccine intentions.
In this study, the authors induced empathy and increased COVID-19 vaccination intention by having participants read a short story about 2 close siblings who worked together in a nursing facility. In the story, participants learned that both siblings were given a diagnosis of COVID-19 at the same time but only 1 survived.17
Continue to: Try this 3-pronged approach
Try this 3-pronged approach. Consider explaining herd immunity to vaccine-hesitant patients, pairing this concept with information about local vaccine uptake, and appealing to the patient’s sense of empathy. You might share de-identified information on other patients in your practice or personal network who experienced severe illness, had long-term effects, or died from COVID-19 infection. Such concrete examples may help to increase motivation to vaccinate more than a general appeal to altruism.
Initiate the discussion by emphasizing that community immunity protects those who are vulnerable and lack immunity while providing specific empathetic examples (eg, newborns, cancer survivors) and asking patients to consider friends and family who might be at risk. Additionally, it is essential to explain that although community immunity can decrease the spread of infection, it can only be achieved when enough people are vaccinated.
Proceed with caution: Addressing conspiracy theories can backfire
Accurate information is critical to improving vaccine intentions; belief in conspiracy theories or misinformation related to COVID-19 is associated with reduced vaccine intentions and uptake.6 For example, a study by Loomba and colleagues18 showed that after exposure to misinformation, US and UK adults reported reduced intentions to vaccinate against COVID-19 once a vaccine became available.
Unfortunately, addressing myths about vaccines can sometimes backfire and unintentionally reinforce vaccine misperceptions.19,20 This is especially true for patients with the highest levels of concern or mistrust in vaccines. Nyhan and colleagues21,22 observed the backfire effect in 2 US studies looking at influenza and measles, mumps, and rubella vaccine misperceptions. Although corrective information significantly reduced belief in vaccine myths, they found individuals with the most concerns more strongly endorsed misperceptions when their beliefs were challenged.21,22
An Australian randomized study by Steffens and colleagues23 found repeating myths about childhood vaccines, followed by corrective text, to parents of children ages 0 to 5 years had no difference on parental intent to vaccinate their children compared to providing vaccine information as a statement or in a question/answer format. Furthermore, an RCT in Brazil by Carey and colleagues24 found that myth-correction messages about Zika virus failed to reduce misperceptions about the virus and actually reduced the belief in factual information about Zika—regardless of baseline beliefs in conspiracies. However, a similar experiment in the same study showed that myth-correction messages reduced false beliefs about yellow fever.
Continue to: The authors speculated...
The authors speculated that this may be because Zika is a relatively new virus when compared to yellow fever, and participants may have more pre-existing knowledge about yellow fever.24 These findings are important to keep in mind when addressing misinformation regarding COVID-19. When addressing myth perceptions with patients, consider pivoting the conversation from vaccine myths to the disease itself, focusing on the disease risk and severity of symptoms.19,20
Other studies have had positive results when addressing misinformation, including a digital RCT of older adults in the Netherlands by Yousuf and colleagues.25 In this study, participants were randomized to view 1 of 2 versions of an information video on vaccination featuring an informative discussion by celebrity scientists, government officials, and a cardiologist. Video 1 did not include debunking strategies, only information about vaccination; Video 2 provided the same information about vaccines but also described the myths surrounding vaccines and reiterated the truth to debunk the myths.
Findings demonstrated that a significantly higher number of participants in the Video 2 group overcame vaccination myths related to influenza and COVID-19.25 Notably, this study took place prior to the widespread availability of COVID-19 vaccines and did not measure intent to vaccinate against COVID-19.
Taken together, strategies for correcting vaccine misinformation may vary by population as well as type of vaccine; however, placing emphasis on facts delivered by trusted sources appears to be beneficial. When addressing misinformation, PCPs should first focus on key details (not all supporting information) and clearly explain why the misinformation is false before pointing out the actual myth and providing an alternative explanation.20 When caring for patients who express strong concerns over the vaccine in question or have avid beliefs in certain myths or conspiracy theories, it’s best to pivot the conversation back to the disease rather than address the misinformation to avoid a potential backfire effect.
Utilize these effective communication techniques
TABLE 110,13,16,17,19,20 summarizes the “do’s and don’ts” of communicating with vaccine-hesitant patients. PCPs should provide strong recommendations for vaccination, approaching it presumptively—ie, framing it as normative behavior.19,26 This approach is critical to building patient trust so that vaccine-hesitant patients feel the PCP is truly listening to them and addressing their concerns.27 Additionally, implementing motivational interviewing (MI) and self-determination theory (SDT)28 techniques when discussing vaccinations with patients can improve intentions and uptake.19,29TABLE 219,29 outlines specific techniques based on SDT and MI that PCPs may utilize to communicate with vaccine-hesitant individuals or parents.
Continue to: The takeaway
The takeaway
Strategies for increasing vaccine intentions include educating hesitant patients about the benefits and risks of vaccines, addressing misinformation, and explaining the personal and collective benefits of vaccination. These strategies appear to be more effective when delivered by a trusted source, such as a health care provider (HCP). Care should be taken when implementing vaccine-acceptance strategies to ensure that they are tailored to specific populations and vaccines.
At this stage in the COVID-19 pandemic, when several vaccines have been widely available for more than a year, we expect that the majority of patients desiring vaccination (ie, those with the greatest vaccine intent) have already received them. With the recent approval of COVID-19 vaccines for children younger than 5 years, we must now advocate for our patients to vaccinate not only themselves, but their children. Patients who remain unvaccinated may be hesitant or outright reject vaccination for a number of reasons, including fear or skepticism over the safety and efficacy of the vaccine, belief in conspiracy theories, belief that COVID-19 is not real or not severe, or mistrust of the government.6 Vaccine hesitation or rejection is also often political in nature.
Based on the studies included in this review, we have identified several strategies for reducing vaccine hesitancy, which can be used with vaccine-hesitant patients and parents. We suggest emphasizing the personal benefit of vaccination and focusing on specific disease risks. If time allows, you can also explain the collective benefit of vaccination through herd immunity, including the current levels of local vaccine uptake compared to the desired level for community immunity. Communicating the collective benefits of vaccination may be more effective when paired with a strategy intended to increase empathy and altruism, such as sharing actual stories about those who have suffered from a vaccine-preventable disease.
Addressing myths and misinformation related to COVID-19 and other vaccines, with emphasis placed on the correct information delivered by trusted sources may be beneficial for those who are uncertain but not strongly against vaccination. For those who remain staunchly hesitant against vaccination, we recommend focusing on the personal benefits of vaccination with a focus on delivering facts about the risk of the disease in question, rather than trying to refute misinformation.
COVID-19 vaccine acceptance in the United States is disturbingly low among health care workers, particularly nurses, technicians, and those in nonclinical roles, compared to physicians.6,30 Many of the strategies for addressing vaccine hesitancy among the general population can also apply to health care personnel (eg, vaccine education, addressing misinformation, delivering information from a trusted source). Health care personnel may also be subject to vaccine mandates by their employers, which have demonstrated increases in vaccination rates for influenza.31 Given that COVID-19 vaccination recommendations made by HCPs are associated with greater vaccine intentions and uptake,6 reducing hesitancy among health care workers is a critical first step to achieving optimal implementation.
CORRESPONDENCE
Nicole Mayo, PhD, 236 Pearl Street, Rochester, NY 14607; Nicole_Mayo@URMC.Rochester.edu
1. Ten threats to global health in 2019. World Health Organization. Accessed June 17, 2022. www.who.int/news-room/spotlight/ten-threats-to-global-health-in-2019
2. FDA approves first COVID-19 vaccine. US Food and Drug Administration. August 23, 2021. Accessed June 17, 2022. www.fda.gov/news-events/press-announcements/fda-approves-first-covid-19-vaccine
3. Mathieu E, Ritchie H, Ortiz-Ospina E, et al. A global database of COVID-19 vaccinations. Nat Hum Behav. 2021;5:947-953. doi: 10.1038/s41562-021-01122-8.
4. Ritchie H, Mathieu E, Rodés-Guirao L, et al. Coronavirus pandemic (COVID-19). Our world in data. Accessed June 17, 2022. https://ourworldindata.org/covid-vaccinations?country=USA
5. de Figueiredo A, Simas C, Karafillakis E, et al. Mapping global trends in vaccine confidence and investigating barriers to vaccine uptake: a large-scale retrospective temporal modelling study. Lancet. 2020;396:898-908. doi: 10.1016/S0140-6736(20)31558-0
6. Wang Y, Liu Y. Multilevel determinants of COVID-19 vaccination hesitancy in the United States: a rapid systematic review. Prev Med Rep. 2021;25:101673. doi: 10.1016/j.pmedr.2021.101673
7. Robinson E, Jones A, Lesser I, et al. International estimates of intended uptake and refusal of COVID-19 vaccines: a rapid systematic review and meta-analysis of large nationally representative samples. Vaccine. 2021;39:2024-2034. doi: 10.1016/j.vaccine.2021.02.005
8. Dubé E, Gagnon D, MacDonald NE; SAGE Working Group on Vaccine Hesitancy. Strategies intended to address vaccine hesitancy: review of published reviews. Vaccine. 2015;33:4191-4203. doi: 10.1016/j.vaccine.2015.04.041
9. Sadaf A, Richards JL, Glanz J, et al. A systematic review of interventions for reducing parental vaccine refusal and vaccine hesitancy. Vaccine. 2013;31:4293-4304. doi: 10.1016/j.vaccine.2013.07.013
10. Kim S, Pjesivac I, Jin Y. Effects of message framing on influenza vaccination: understanding the role of risk disclosure, perceived vaccine efficacy, and felt ambivalence. Health Commun. 2019;34:21-30. doi: 10.1080/10410236.2017.1384353
11. Shim E, Chapman GB, Townsend JP, et al. The influence of altruism on influenza vaccination decisions. J R Soc Interface. 2012;9:2234-2243. doi: 10.1098/rsif.2012.0115
12. Sprengholz P, Eitze S, Felgendreff L, et al. Money is not everything: experimental evidence that payments do not increase willingness to be vaccinated against COVID-19. J Med Ethics. 2021;47:547-548. doi: 10.1136/medethics-2020-107122
13. Freeman D, Loe BS, Yu LM, et al. Effects of different types of written vaccination information on COVID-19 vaccine hesitancy in the UK (OCEANS-III): a single-blind, parallel-group, randomised controlled trial. Lancet Public Health. 2021;6:e416-e427. doi: 10.1016/S2468-2667(21)00096-7
14. Hakim H, Provencher T, Chambers CT, et al. Interventions to help people understand community immunity: a systematic review. Vaccine. 2019;37:235-247. doi: 10.1016/j.vaccine.2018.11.016
15. Betsch C, Böhm R, Korn L, et al. On the benefits of explaining herd immunity in vaccine advocacy. Nat Hum Behav. 2017;1:1-6. doi: 10.1038/s41562-017-0056
16. Logan J, Nederhoff D, Koch B, et al. ‘What have you HEARD about the HERD?’ Does education about local influenza vaccination coverage and herd immunity affect willingness to vaccinate? Vaccine. 2018;36:4118-4125. doi: 10.1016/j.vaccine.2018.05.037
17. Pfattheicher S, Petersen MB, Böhm R. Information about herd immunity through vaccination and empathy promote COVID-19 vaccination intentions. Health Psychol. 2022;41:85-93. doi: 10.1037/hea0001096
18. Loomba S, de Figueiredo A, Piatek SJ, et al. Measuring the impact of COVID-19 vaccine misinformation on vaccination intent in the UK and USA. Nat Hum Behav. 2021;5:337-348. doi: 10.1038/s41562-021-01056-1
19. Limaye RJ, Opel DJ, Dempsey A, et al. Communicating with vaccine-hesitant parents: a narrative review. Acad Pediatr. 2021;21:S24-S29. doi: 10.1016/j.acap.2021.01.018
20. Omer SB, Amin AB, Limaye RJ. Communicating about vaccines in a fact-resistant world. JAMA Pediatr. 2017;171:929-930. doi: 10.1001/jamapediatrics.2017.2219
21. Nyhan B, Reifler J. Does correcting myths about the flu vaccine work? An experimental evaluation of the effects of corrective information. Vaccine. 2015;33:459-464. doi: 10.1016/j.vaccine.2014.11.017
22. Nyhan B, Reifler J, Richey S, et al. Effective messages in vaccine promotion: a randomized trial. Pediatrics. 2014;133:e835-e842. doi: 10.1542/peds.2013-2365
23. Steffens MS, Dunn AG, Marques MD, et al. Addressing myths and vaccine hesitancy: a randomized trial. Pediatrics. 2021;148:e2020049304. doi: 10.1542/peds.2020-049304
24. Carey JM, Chi V, Flynn DJ, et al. The effects of corrective information about disease epidemics and outbreaks: evidence from Zika and yellow fever in Brazil. Sci Adv. 2020;6:eaaw7449. doi: 10.1126/sciadv.aaw7449
25. Yousuf H, van der Linden S, Bredius L, et al. A media intervention applying debunking versus non-debunking content to combat vaccine misinformation in elderly in the Netherlands: a digital randomised trial. EClinicalMedicine. 2021;35:100881. doi: 10.1016/j.eclinm.2021.100881
26. Cambon L, Schwarzinger M, Alla F. Increasing acceptance of a vaccination program for coronavirus disease 2019 in France: a challenge for one of the world’s most vaccine-hesitant countries. Vaccine. 2022;40:178-182. doi: 10.1016/j.vaccine.2021.11.023
27. Leask J, Kinnersley P, Jackson C, et al. Communicating with parents about vaccination: a framework for health professionals. BMC Pediatr. 2012;12:154. doi: 10.1186/1471-2431-12-154
28. Martela F, Hankonen N, Ryan RM, et al. Motivating voluntary compliance to behavioural restrictions: self-determination theory–based checklist of principles for COVID-19 and other emergency communications. Eur Rev Soc Psychol. 2021:305-347. doi: 10.1080/10463283.2020.1857082
29. Boness CL, Nelson M, Douaihy AB. Motivational interviewing strategies for addressing COVID-19 vaccine hesitancy. J Am Board Fam Med. 2022;35:420-426. doi: 10.3122/jabfm.2022.02.210327
30. Salomoni MG, Di Valerio Z, Gabrielli E, et al. Hesitant or not hesitant? A systematic review on global COVID-19 vaccine acceptance in different populations. Vaccines (Basel). 2021;9:873. doi: 10.3390/vaccines9080873
31. Pitts SI, Maruthur NM, Millar KR, et al. A systematic review of mandatory influenza vaccination in healthcare personnel. Am J Prev Med. 2014;47:330-340. doi:
1. Ten threats to global health in 2019. World Health Organization. Accessed June 17, 2022. www.who.int/news-room/spotlight/ten-threats-to-global-health-in-2019
2. FDA approves first COVID-19 vaccine. US Food and Drug Administration. August 23, 2021. Accessed June 17, 2022. www.fda.gov/news-events/press-announcements/fda-approves-first-covid-19-vaccine
3. Mathieu E, Ritchie H, Ortiz-Ospina E, et al. A global database of COVID-19 vaccinations. Nat Hum Behav. 2021;5:947-953. doi: 10.1038/s41562-021-01122-8.
4. Ritchie H, Mathieu E, Rodés-Guirao L, et al. Coronavirus pandemic (COVID-19). Our world in data. Accessed June 17, 2022. https://ourworldindata.org/covid-vaccinations?country=USA
5. de Figueiredo A, Simas C, Karafillakis E, et al. Mapping global trends in vaccine confidence and investigating barriers to vaccine uptake: a large-scale retrospective temporal modelling study. Lancet. 2020;396:898-908. doi: 10.1016/S0140-6736(20)31558-0
6. Wang Y, Liu Y. Multilevel determinants of COVID-19 vaccination hesitancy in the United States: a rapid systematic review. Prev Med Rep. 2021;25:101673. doi: 10.1016/j.pmedr.2021.101673
7. Robinson E, Jones A, Lesser I, et al. International estimates of intended uptake and refusal of COVID-19 vaccines: a rapid systematic review and meta-analysis of large nationally representative samples. Vaccine. 2021;39:2024-2034. doi: 10.1016/j.vaccine.2021.02.005
8. Dubé E, Gagnon D, MacDonald NE; SAGE Working Group on Vaccine Hesitancy. Strategies intended to address vaccine hesitancy: review of published reviews. Vaccine. 2015;33:4191-4203. doi: 10.1016/j.vaccine.2015.04.041
9. Sadaf A, Richards JL, Glanz J, et al. A systematic review of interventions for reducing parental vaccine refusal and vaccine hesitancy. Vaccine. 2013;31:4293-4304. doi: 10.1016/j.vaccine.2013.07.013
10. Kim S, Pjesivac I, Jin Y. Effects of message framing on influenza vaccination: understanding the role of risk disclosure, perceived vaccine efficacy, and felt ambivalence. Health Commun. 2019;34:21-30. doi: 10.1080/10410236.2017.1384353
11. Shim E, Chapman GB, Townsend JP, et al. The influence of altruism on influenza vaccination decisions. J R Soc Interface. 2012;9:2234-2243. doi: 10.1098/rsif.2012.0115
12. Sprengholz P, Eitze S, Felgendreff L, et al. Money is not everything: experimental evidence that payments do not increase willingness to be vaccinated against COVID-19. J Med Ethics. 2021;47:547-548. doi: 10.1136/medethics-2020-107122
13. Freeman D, Loe BS, Yu LM, et al. Effects of different types of written vaccination information on COVID-19 vaccine hesitancy in the UK (OCEANS-III): a single-blind, parallel-group, randomised controlled trial. Lancet Public Health. 2021;6:e416-e427. doi: 10.1016/S2468-2667(21)00096-7
14. Hakim H, Provencher T, Chambers CT, et al. Interventions to help people understand community immunity: a systematic review. Vaccine. 2019;37:235-247. doi: 10.1016/j.vaccine.2018.11.016
15. Betsch C, Böhm R, Korn L, et al. On the benefits of explaining herd immunity in vaccine advocacy. Nat Hum Behav. 2017;1:1-6. doi: 10.1038/s41562-017-0056
16. Logan J, Nederhoff D, Koch B, et al. ‘What have you HEARD about the HERD?’ Does education about local influenza vaccination coverage and herd immunity affect willingness to vaccinate? Vaccine. 2018;36:4118-4125. doi: 10.1016/j.vaccine.2018.05.037
17. Pfattheicher S, Petersen MB, Böhm R. Information about herd immunity through vaccination and empathy promote COVID-19 vaccination intentions. Health Psychol. 2022;41:85-93. doi: 10.1037/hea0001096
18. Loomba S, de Figueiredo A, Piatek SJ, et al. Measuring the impact of COVID-19 vaccine misinformation on vaccination intent in the UK and USA. Nat Hum Behav. 2021;5:337-348. doi: 10.1038/s41562-021-01056-1
19. Limaye RJ, Opel DJ, Dempsey A, et al. Communicating with vaccine-hesitant parents: a narrative review. Acad Pediatr. 2021;21:S24-S29. doi: 10.1016/j.acap.2021.01.018
20. Omer SB, Amin AB, Limaye RJ. Communicating about vaccines in a fact-resistant world. JAMA Pediatr. 2017;171:929-930. doi: 10.1001/jamapediatrics.2017.2219
21. Nyhan B, Reifler J. Does correcting myths about the flu vaccine work? An experimental evaluation of the effects of corrective information. Vaccine. 2015;33:459-464. doi: 10.1016/j.vaccine.2014.11.017
22. Nyhan B, Reifler J, Richey S, et al. Effective messages in vaccine promotion: a randomized trial. Pediatrics. 2014;133:e835-e842. doi: 10.1542/peds.2013-2365
23. Steffens MS, Dunn AG, Marques MD, et al. Addressing myths and vaccine hesitancy: a randomized trial. Pediatrics. 2021;148:e2020049304. doi: 10.1542/peds.2020-049304
24. Carey JM, Chi V, Flynn DJ, et al. The effects of corrective information about disease epidemics and outbreaks: evidence from Zika and yellow fever in Brazil. Sci Adv. 2020;6:eaaw7449. doi: 10.1126/sciadv.aaw7449
25. Yousuf H, van der Linden S, Bredius L, et al. A media intervention applying debunking versus non-debunking content to combat vaccine misinformation in elderly in the Netherlands: a digital randomised trial. EClinicalMedicine. 2021;35:100881. doi: 10.1016/j.eclinm.2021.100881
26. Cambon L, Schwarzinger M, Alla F. Increasing acceptance of a vaccination program for coronavirus disease 2019 in France: a challenge for one of the world’s most vaccine-hesitant countries. Vaccine. 2022;40:178-182. doi: 10.1016/j.vaccine.2021.11.023
27. Leask J, Kinnersley P, Jackson C, et al. Communicating with parents about vaccination: a framework for health professionals. BMC Pediatr. 2012;12:154. doi: 10.1186/1471-2431-12-154
28. Martela F, Hankonen N, Ryan RM, et al. Motivating voluntary compliance to behavioural restrictions: self-determination theory–based checklist of principles for COVID-19 and other emergency communications. Eur Rev Soc Psychol. 2021:305-347. doi: 10.1080/10463283.2020.1857082
29. Boness CL, Nelson M, Douaihy AB. Motivational interviewing strategies for addressing COVID-19 vaccine hesitancy. J Am Board Fam Med. 2022;35:420-426. doi: 10.3122/jabfm.2022.02.210327
30. Salomoni MG, Di Valerio Z, Gabrielli E, et al. Hesitant or not hesitant? A systematic review on global COVID-19 vaccine acceptance in different populations. Vaccines (Basel). 2021;9:873. doi: 10.3390/vaccines9080873
31. Pitts SI, Maruthur NM, Millar KR, et al. A systematic review of mandatory influenza vaccination in healthcare personnel. Am J Prev Med. 2014;47:330-340. doi:
PRACTICE RECOMMENDATIONS
› Focus on personal benefits of vaccination with patients who express strong hesitancy and endorse vaccine myths; refocus the conversation away from myths and back to disease facts. C
› Emphasize personal and collective benefit to patients who are uncertain about vaccination; provide education about herd immunity and local vaccine coverage. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Diffuse annular lesions
A 24-YEAR-OLD WOMAN with a history of guttate psoriasis, for which she was taking adalimumab, presented with a 2-week history of diffuse papules and plaques on her neck, back, torso, and upper and lower extremities (FIGURE 1). She said that the lesions were pruritic and seemed similar to those that erupted during past outbreaks of psoriasis—although they were more numerous and progressive. So, the patient (a nurse) decided to take her biweekly dose (40 mg) of adalimumab 1 week early. After administration, the rash significantly worsened, spreading to the rest of her trunk and extremities.
Physical exam was notable for multiple erythematous papules and plaques with central clearing and light peripheral scaling on both arms and legs, as well as her chest and back. The patient also indicated she’d adopted a stray cat 2 weeks prior. Given the patient’s pet exposure and the annular nature of the lesions, a potassium hydroxide (KOH) preparation was done.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Tinea corporis
The KOH preparation was positive for hyphae in 4 separate sites (trunk, left arm, left leg, and left neck), confirming the diagnosis of severe extensive tinea corporis (FIGURE 2).
Dermatophyte (tinea) infections are caused by fungi that invade and reproduce in the skin, hair, and nails. Dermatophytes, which include the genera Trichophyton, Microsporum, and Epidermophyton, are the most common cause of superficial mycotic infections. As of 2016, the worldwide prevalence of superficial mycotic infections was 20% to 25%.1 Tinea corporis can result from contact with people, animals, or soil. Infections resulting from animal-to-human contact are often transmitted by domestic animals. In this case, the patient’s exposure was from her new cat.
Tinea corporis classically manifests as pruritic, erythematous patches or plaques with central clearing, giving it an annular appearance. The response to a tinea infection depends on the immune system of the host and can range in severity from superficial to severe.2 There are 2 forms of severe dermatophytosis: invasive, which involves localized perifollicular sites or deep dermatophytosis, and extensive, which is confined to the stratum corneum but results in numerous lesions.3
The diagnosis of tinea corporis is commonly confirmed using direct microscopic examination with 10% to 20% KOH preparation, which will show branching and septate hyphal filaments.4
Several conditions with annular lesions comprise the differential
The findings of pruritic annular erythematous lesions on the patient’s neck, chest, trunk, and bilateral extremities led the patient to suspect this was a worsening case of her guttate psoriasis. Other possible diagnoses included pityriasis rosea, subacute cutaneous lupus erythematosus (SCLE), and secondary syphilis.
Continue to: Guttate psoriasis
Guttate psoriasis would not typically progress during treatment with adalimumab, although tumor necrosis factor (TNF) inhibitors have been associated with worsening psoriasis. Guttate psoriasis manifests with small, pink to red, scaly raindrop-shaped patches over the trunk and extremities.
Pityriasis rosea, a rash that resembles branches of a Christmas tree, was strongly considered given the appearance of the lesions on the patient’s back. It commonly manifests as round to oval lesions with a subtle advancing border and central fine scaling, similar in shape and color to the lesions seen in tinea corporis.
SCLE has been associated with use of TNF inhibitors, but our patient had no other lupus-like symptoms, such as fatigue, fever, headaches, or joint pain. SCLE lesions are often annular with raised pink to red borders similar in appearance to tinea corporis.
Secondary syphilis was ruled out in this patient because she had a negative rapid plasma reagin test. Secondary syphilis most commonly manifests with diffuse, nonpruritic pink to red-brown lesions on the palms and soles of patients. Patients often have prodromal symptoms that include fever, weight loss, myalgias, headache, and sore throat.
Terbinafine, Yes, but for how long?
Historically, terbinafine has been prescribed at 250 mg once daily for 2 weeks for extensive tinea corporis. However, recent studies in India suggest that terbinafine should be dosed at 250 mg twice daily, with longer durations of treatment, due to resistance.5 In the United States, it is reasonable to prescribe oral terbinafine 250 mg once daily for 4 weeks and then re-evaluate the patient in a case of extensive tinea corporis.
Other oral antifungals that can effectively treat extensive tinea corporis include itraconazole, fluconazole, and griseofulvin.1 Itraconazole and terbinafine are equally effective and safe in the treatment of tinea corporis, although itraconazole is significantly more expensive.6 Furthermore, a recent study found that combination therapy with oral terbinafine and itraconazole is as safe as monotherapy and is an option when terbinafine resistance is suspected.7
Our patient was initially started on oral terbinafine 250 mg/d. After the first dose, the patient requested a change in medication because there was no improvement in the rash. The patient was then prescribed oral fluconazole 300 mg daily and the tinea cleared after 2 months of daily therapy. (We surmise the treatment course may have been prolonged due to the possible immunosuppressant effects of adalimumab.) At the completion of treatment for the tinea corporis, the patient was restarted on adalimumab 40 mg biweekly for her psoriasis.
1. Sahoo AK, Mahajan R. Management of tinea corporis, tinea cruris, and tinea pedis: a comprehensive review. Indian Dermatol Online J. 2016;7:77-86. doi: 10.4103/2229-5178.178099
2. Weitzman I, Summerbell RC. The dermatophytes. Clin Microbial Rev. 1995:8:240-259. doi: 10.1128/CMR.8.2.240
3. Rouzaud C, Hay R, Chosidow O, et al. Severe dermatophytosis and acquired or innate immunodeficiency: a review. J Fungi (Basel). 2015;2:4. doi: 10.3390/jof2010004
4. Kurade SM, Amladi SA, Miskeen AK. Skin scraping and a potassium hydroxide mount. Indian J Dermatol Venereol Leprol. 2006;72:238-41. doi: 10.4103/0378-6323.25794
5. Khurana A, Sardana K, Chowdhary A. Antifungal resistance in dermatophytes: recent trends and therapeutic implications. Fungal Genet Biol. 2019;132:103255. doi: 10.1016/j.fgb.2019.103255
6. Bhatia A, Kanish B, Badyal DK, et al. Efficacy of oral terbinafine versus itraconazole in treatment of dermatophytic infection of skin - a prospective, randomized comparative study. Indian J Pharmacol. 2019;51:116-119.
7. Sharma P, Bhalla M, Thami GP, et al. Evaluation of efficacy and safety of oral terbinafine and itraconazole combination therapy in the management of dermatophytosis. J Dermatolog Treat. 2020;31:749-753. doi: 10.1080/09546634.2019.1612835
A 24-YEAR-OLD WOMAN with a history of guttate psoriasis, for which she was taking adalimumab, presented with a 2-week history of diffuse papules and plaques on her neck, back, torso, and upper and lower extremities (FIGURE 1). She said that the lesions were pruritic and seemed similar to those that erupted during past outbreaks of psoriasis—although they were more numerous and progressive. So, the patient (a nurse) decided to take her biweekly dose (40 mg) of adalimumab 1 week early. After administration, the rash significantly worsened, spreading to the rest of her trunk and extremities.
Physical exam was notable for multiple erythematous papules and plaques with central clearing and light peripheral scaling on both arms and legs, as well as her chest and back. The patient also indicated she’d adopted a stray cat 2 weeks prior. Given the patient’s pet exposure and the annular nature of the lesions, a potassium hydroxide (KOH) preparation was done.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Tinea corporis
The KOH preparation was positive for hyphae in 4 separate sites (trunk, left arm, left leg, and left neck), confirming the diagnosis of severe extensive tinea corporis (FIGURE 2).

Dermatophyte (tinea) infections are caused by fungi that invade and reproduce in the skin, hair, and nails. Dermatophytes, which include the genera Trichophyton, Microsporum, and Epidermophyton, are the most common cause of superficial mycotic infections. As of 2016, the worldwide prevalence of superficial mycotic infections was 20% to 25%.1 Tinea corporis can result from contact with people, animals, or soil. Infections resulting from animal-to-human contact are often transmitted by domestic animals. In this case, the patient’s exposure was from her new cat.
Tinea corporis classically manifests as pruritic, erythematous patches or plaques with central clearing, giving it an annular appearance. The response to a tinea infection depends on the immune system of the host and can range in severity from superficial to severe.2 There are 2 forms of severe dermatophytosis: invasive, which involves localized perifollicular sites or deep dermatophytosis, and extensive, which is confined to the stratum corneum but results in numerous lesions.3
The diagnosis of tinea corporis is commonly confirmed using direct microscopic examination with 10% to 20% KOH preparation, which will show branching and septate hyphal filaments.4
Several conditions with annular lesions comprise the differential
The findings of pruritic annular erythematous lesions on the patient’s neck, chest, trunk, and bilateral extremities led the patient to suspect this was a worsening case of her guttate psoriasis. Other possible diagnoses included pityriasis rosea, subacute cutaneous lupus erythematosus (SCLE), and secondary syphilis.
Continue to: Guttate psoriasis
Guttate psoriasis would not typically progress during treatment with adalimumab, although tumor necrosis factor (TNF) inhibitors have been associated with worsening psoriasis. Guttate psoriasis manifests with small, pink to red, scaly raindrop-shaped patches over the trunk and extremities.
Pityriasis rosea, a rash that resembles branches of a Christmas tree, was strongly considered given the appearance of the lesions on the patient’s back. It commonly manifests as round to oval lesions with a subtle advancing border and central fine scaling, similar in shape and color to the lesions seen in tinea corporis.
SCLE has been associated with use of TNF inhibitors, but our patient had no other lupus-like symptoms, such as fatigue, fever, headaches, or joint pain. SCLE lesions are often annular with raised pink to red borders similar in appearance to tinea corporis.
Secondary syphilis was ruled out in this patient because she had a negative rapid plasma reagin test. Secondary syphilis most commonly manifests with diffuse, nonpruritic pink to red-brown lesions on the palms and soles of patients. Patients often have prodromal symptoms that include fever, weight loss, myalgias, headache, and sore throat.
Terbinafine, Yes, but for how long?
Historically, terbinafine has been prescribed at 250 mg once daily for 2 weeks for extensive tinea corporis. However, recent studies in India suggest that terbinafine should be dosed at 250 mg twice daily, with longer durations of treatment, due to resistance.5 In the United States, it is reasonable to prescribe oral terbinafine 250 mg once daily for 4 weeks and then re-evaluate the patient in a case of extensive tinea corporis.
Other oral antifungals that can effectively treat extensive tinea corporis include itraconazole, fluconazole, and griseofulvin.1 Itraconazole and terbinafine are equally effective and safe in the treatment of tinea corporis, although itraconazole is significantly more expensive.6 Furthermore, a recent study found that combination therapy with oral terbinafine and itraconazole is as safe as monotherapy and is an option when terbinafine resistance is suspected.7
Our patient was initially started on oral terbinafine 250 mg/d. After the first dose, the patient requested a change in medication because there was no improvement in the rash. The patient was then prescribed oral fluconazole 300 mg daily and the tinea cleared after 2 months of daily therapy. (We surmise the treatment course may have been prolonged due to the possible immunosuppressant effects of adalimumab.) At the completion of treatment for the tinea corporis, the patient was restarted on adalimumab 40 mg biweekly for her psoriasis.
A 24-YEAR-OLD WOMAN with a history of guttate psoriasis, for which she was taking adalimumab, presented with a 2-week history of diffuse papules and plaques on her neck, back, torso, and upper and lower extremities (FIGURE 1). She said that the lesions were pruritic and seemed similar to those that erupted during past outbreaks of psoriasis—although they were more numerous and progressive. So, the patient (a nurse) decided to take her biweekly dose (40 mg) of adalimumab 1 week early. After administration, the rash significantly worsened, spreading to the rest of her trunk and extremities.
Physical exam was notable for multiple erythematous papules and plaques with central clearing and light peripheral scaling on both arms and legs, as well as her chest and back. The patient also indicated she’d adopted a stray cat 2 weeks prior. Given the patient’s pet exposure and the annular nature of the lesions, a potassium hydroxide (KOH) preparation was done.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Tinea corporis
The KOH preparation was positive for hyphae in 4 separate sites (trunk, left arm, left leg, and left neck), confirming the diagnosis of severe extensive tinea corporis (FIGURE 2).

Dermatophyte (tinea) infections are caused by fungi that invade and reproduce in the skin, hair, and nails. Dermatophytes, which include the genera Trichophyton, Microsporum, and Epidermophyton, are the most common cause of superficial mycotic infections. As of 2016, the worldwide prevalence of superficial mycotic infections was 20% to 25%.1 Tinea corporis can result from contact with people, animals, or soil. Infections resulting from animal-to-human contact are often transmitted by domestic animals. In this case, the patient’s exposure was from her new cat.
Tinea corporis classically manifests as pruritic, erythematous patches or plaques with central clearing, giving it an annular appearance. The response to a tinea infection depends on the immune system of the host and can range in severity from superficial to severe.2 There are 2 forms of severe dermatophytosis: invasive, which involves localized perifollicular sites or deep dermatophytosis, and extensive, which is confined to the stratum corneum but results in numerous lesions.3
The diagnosis of tinea corporis is commonly confirmed using direct microscopic examination with 10% to 20% KOH preparation, which will show branching and septate hyphal filaments.4
Several conditions with annular lesions comprise the differential
The findings of pruritic annular erythematous lesions on the patient’s neck, chest, trunk, and bilateral extremities led the patient to suspect this was a worsening case of her guttate psoriasis. Other possible diagnoses included pityriasis rosea, subacute cutaneous lupus erythematosus (SCLE), and secondary syphilis.
Continue to: Guttate psoriasis
Guttate psoriasis would not typically progress during treatment with adalimumab, although tumor necrosis factor (TNF) inhibitors have been associated with worsening psoriasis. Guttate psoriasis manifests with small, pink to red, scaly raindrop-shaped patches over the trunk and extremities.
Pityriasis rosea, a rash that resembles branches of a Christmas tree, was strongly considered given the appearance of the lesions on the patient’s back. It commonly manifests as round to oval lesions with a subtle advancing border and central fine scaling, similar in shape and color to the lesions seen in tinea corporis.
SCLE has been associated with use of TNF inhibitors, but our patient had no other lupus-like symptoms, such as fatigue, fever, headaches, or joint pain. SCLE lesions are often annular with raised pink to red borders similar in appearance to tinea corporis.
Secondary syphilis was ruled out in this patient because she had a negative rapid plasma reagin test. Secondary syphilis most commonly manifests with diffuse, nonpruritic pink to red-brown lesions on the palms and soles of patients. Patients often have prodromal symptoms that include fever, weight loss, myalgias, headache, and sore throat.
Terbinafine, Yes, but for how long?
Historically, terbinafine has been prescribed at 250 mg once daily for 2 weeks for extensive tinea corporis. However, recent studies in India suggest that terbinafine should be dosed at 250 mg twice daily, with longer durations of treatment, due to resistance.5 In the United States, it is reasonable to prescribe oral terbinafine 250 mg once daily for 4 weeks and then re-evaluate the patient in a case of extensive tinea corporis.
Other oral antifungals that can effectively treat extensive tinea corporis include itraconazole, fluconazole, and griseofulvin.1 Itraconazole and terbinafine are equally effective and safe in the treatment of tinea corporis, although itraconazole is significantly more expensive.6 Furthermore, a recent study found that combination therapy with oral terbinafine and itraconazole is as safe as monotherapy and is an option when terbinafine resistance is suspected.7
Our patient was initially started on oral terbinafine 250 mg/d. After the first dose, the patient requested a change in medication because there was no improvement in the rash. The patient was then prescribed oral fluconazole 300 mg daily and the tinea cleared after 2 months of daily therapy. (We surmise the treatment course may have been prolonged due to the possible immunosuppressant effects of adalimumab.) At the completion of treatment for the tinea corporis, the patient was restarted on adalimumab 40 mg biweekly for her psoriasis.
1. Sahoo AK, Mahajan R. Management of tinea corporis, tinea cruris, and tinea pedis: a comprehensive review. Indian Dermatol Online J. 2016;7:77-86. doi: 10.4103/2229-5178.178099
2. Weitzman I, Summerbell RC. The dermatophytes. Clin Microbial Rev. 1995:8:240-259. doi: 10.1128/CMR.8.2.240
3. Rouzaud C, Hay R, Chosidow O, et al. Severe dermatophytosis and acquired or innate immunodeficiency: a review. J Fungi (Basel). 2015;2:4. doi: 10.3390/jof2010004
4. Kurade SM, Amladi SA, Miskeen AK. Skin scraping and a potassium hydroxide mount. Indian J Dermatol Venereol Leprol. 2006;72:238-41. doi: 10.4103/0378-6323.25794
5. Khurana A, Sardana K, Chowdhary A. Antifungal resistance in dermatophytes: recent trends and therapeutic implications. Fungal Genet Biol. 2019;132:103255. doi: 10.1016/j.fgb.2019.103255
6. Bhatia A, Kanish B, Badyal DK, et al. Efficacy of oral terbinafine versus itraconazole in treatment of dermatophytic infection of skin - a prospective, randomized comparative study. Indian J Pharmacol. 2019;51:116-119.
7. Sharma P, Bhalla M, Thami GP, et al. Evaluation of efficacy and safety of oral terbinafine and itraconazole combination therapy in the management of dermatophytosis. J Dermatolog Treat. 2020;31:749-753. doi: 10.1080/09546634.2019.1612835
1. Sahoo AK, Mahajan R. Management of tinea corporis, tinea cruris, and tinea pedis: a comprehensive review. Indian Dermatol Online J. 2016;7:77-86. doi: 10.4103/2229-5178.178099
2. Weitzman I, Summerbell RC. The dermatophytes. Clin Microbial Rev. 1995:8:240-259. doi: 10.1128/CMR.8.2.240
3. Rouzaud C, Hay R, Chosidow O, et al. Severe dermatophytosis and acquired or innate immunodeficiency: a review. J Fungi (Basel). 2015;2:4. doi: 10.3390/jof2010004
4. Kurade SM, Amladi SA, Miskeen AK. Skin scraping and a potassium hydroxide mount. Indian J Dermatol Venereol Leprol. 2006;72:238-41. doi: 10.4103/0378-6323.25794
5. Khurana A, Sardana K, Chowdhary A. Antifungal resistance in dermatophytes: recent trends and therapeutic implications. Fungal Genet Biol. 2019;132:103255. doi: 10.1016/j.fgb.2019.103255
6. Bhatia A, Kanish B, Badyal DK, et al. Efficacy of oral terbinafine versus itraconazole in treatment of dermatophytic infection of skin - a prospective, randomized comparative study. Indian J Pharmacol. 2019;51:116-119.
7. Sharma P, Bhalla M, Thami GP, et al. Evaluation of efficacy and safety of oral terbinafine and itraconazole combination therapy in the management of dermatophytosis. J Dermatolog Treat. 2020;31:749-753. doi: 10.1080/09546634.2019.1612835
Berdazimer gel beats vehicle for molluscum contagiosum in phase 3 study
Treatment with .
Molluscum contagiosum (MC) remains a common infection that, despite being self-limiting, may persist for months or years, and is associated with quality of life concerns and the need for ongoing therapy, wrote John C. Browning, MD, of Texas Dermatology and Laser Specialists, San Antonio, and colleagues, who conducted the phase 3 randomized study.
The infection is most common in children aged 1-14 years, and treatment may be needed in part to avoid infecting peers and family members, they said. No treatments for molluscum are currently approved by the Food and Drug Administration.
In the study, which was published in JAMA Dermatology, the researchers randomized 444 patients to berdazimer gel 10.3% and 447 to a placebo gel, applied once daily in a thin layer on all MC lesions for 12 weeks. The study was conducted at 55 clinics across the United States between Sept. 1, 2020, and July 21, 2021. The mean age of the patients was about 6.5 years (range was 0.9-49 years), and about 85% were White. Participants had 3-70 raised MC lesions; those with sexually transmitted MC or MC in the periocular area were excluded. The primary endpoint was complete clearance of MC lesions after 12 weeks of treatment. At 12 weeks, significantly more patients treated with berdazimer gel achieved complete clearance than those on vehicle (32.4% vs. 19.7%; P < .001). A total of 64 (14.4%) patients in the berdazimer group discontinued treatment because of MC clearance, compared with 40 patients (8.9%) in the vehicle group.
Most adverse events were mild or moderate, and rates of adverse events resulting in treatment discontinuation were low overall for both groups; the most common adverse events were application-site pain and erythema, which were mostly mild. Overall, 4.1% of berdazimer-treated patients and 0.7% of placebo patients discontinued the study because of adverse events.
The study findings were limited by several factors, including the small number of patients in subgroups for race, ethnicity, and age; and the lack of data on patients with sexually transmitted MC and on concomitant use with other topical MC therapies, the researchers noted.
However, the results represent the largest randomized clinical trial of berdazimer 10.3% to date, and support its potential as a first-line therapy for MC patients aged 6 months and older, according to the authors. “Berdazimer is under consideration as a first in-class therapeutic agent for MC and may provide a topical prescription alternative to other therapies used for this highly contagious and psychosocially challenging skin condition,” they said.
Having a reliable, steroid-free, safe, and efficacious medication to treat molluscum in the pediatric population, as early as age 6 months, that can be used at home would “change the whole therapeutic paradigm,” one of the study authors, Adelaide Hebert, MD, said in an interview at the Society for Pediatric Dermatology annual meeting in July, where she presented phase 2 data on berdazimer gel. “This is a common problem and the rate of infections among siblings if it goes untreated is 41%. Affected kids have a sense of isolation; they don’t get invited to swimming parties.”
The lack of a safe and effective topical therapy “has been challenging,” added Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. She noted that treatments that have been used but have not been successful include imiquimod. “I’m not impressed with tretinoin,” although it is prescribed for MC, and the most common treatment prescribed by pediatricians for molluscum – mupirocin – is “usually not effective,” she said.
Another MC treatment in trials
Another investigative treatment for molluscum contagiosum, VP-102, a drug-device combination of cantharidin 0.7% administered through a single-use precision applicator, has been evaluated in phase 3 studies of patients with MC aged 2 years and older. The results of two phase 3 studies were published in 2020.
In May 2022, Verrica Pharmaceuticals, which is developing VP-102, announced that Food and Drug Administration approval had been delayed because of deficiencies identified at a contract manufacturing organization, and that the company was working with the agency to bring VP-102 to the market as soon as possible.
A step in the right direction
Although MC is self-resolving, cases last an average of 13.5 months, and “many families look to fast-forward their child’s experience with the infection,” Vikash S. Oza, MD, a pediatric dermatologist at New York University, New York, wrote in an editorial that accompanied the berdazimer study.
“To truly create a paradigm shift in the decision to treat MC, a therapeutic treatment would need to be developed that would lead to resolution of the infection over a short time frame (ideally, weeks) with minimal discomfort,” Dr. Oza noted. “Both VP-102 and berdazimer gel, 10.3%, have the potential to be the first-ever MC therapies approved by the U.S. Food and Drug Administration,” and families seeking to reduce MC in visible areas would welcome this option for a home therapy, he said.
However, Dr. Oza emphasized that potential barriers to widespread use of these therapies include whether the efficacy can be maintained in patients who fail to comply with daily application, and the ongoing need for office-based therapy to manage sexually transmitted MC in adults and periocular and perianal MC in children. The study was funded by Novan. Lead author Dr. Browning disclosed grants from Novan during the conduct of the study; Dr. Hebert reported grants from the University of Texas Health Science Center McGovern Medical School-Houston during the conduct of the study. Disclosures of other authors included having reported equity in Novan during the conduct of the study and receiving a grant from Novan. Dr. Oza had no financial conflicts to disclose.
Treatment with .
Molluscum contagiosum (MC) remains a common infection that, despite being self-limiting, may persist for months or years, and is associated with quality of life concerns and the need for ongoing therapy, wrote John C. Browning, MD, of Texas Dermatology and Laser Specialists, San Antonio, and colleagues, who conducted the phase 3 randomized study.
The infection is most common in children aged 1-14 years, and treatment may be needed in part to avoid infecting peers and family members, they said. No treatments for molluscum are currently approved by the Food and Drug Administration.
In the study, which was published in JAMA Dermatology, the researchers randomized 444 patients to berdazimer gel 10.3% and 447 to a placebo gel, applied once daily in a thin layer on all MC lesions for 12 weeks. The study was conducted at 55 clinics across the United States between Sept. 1, 2020, and July 21, 2021. The mean age of the patients was about 6.5 years (range was 0.9-49 years), and about 85% were White. Participants had 3-70 raised MC lesions; those with sexually transmitted MC or MC in the periocular area were excluded. The primary endpoint was complete clearance of MC lesions after 12 weeks of treatment. At 12 weeks, significantly more patients treated with berdazimer gel achieved complete clearance than those on vehicle (32.4% vs. 19.7%; P < .001). A total of 64 (14.4%) patients in the berdazimer group discontinued treatment because of MC clearance, compared with 40 patients (8.9%) in the vehicle group.
Most adverse events were mild or moderate, and rates of adverse events resulting in treatment discontinuation were low overall for both groups; the most common adverse events were application-site pain and erythema, which were mostly mild. Overall, 4.1% of berdazimer-treated patients and 0.7% of placebo patients discontinued the study because of adverse events.
The study findings were limited by several factors, including the small number of patients in subgroups for race, ethnicity, and age; and the lack of data on patients with sexually transmitted MC and on concomitant use with other topical MC therapies, the researchers noted.
However, the results represent the largest randomized clinical trial of berdazimer 10.3% to date, and support its potential as a first-line therapy for MC patients aged 6 months and older, according to the authors. “Berdazimer is under consideration as a first in-class therapeutic agent for MC and may provide a topical prescription alternative to other therapies used for this highly contagious and psychosocially challenging skin condition,” they said.
Having a reliable, steroid-free, safe, and efficacious medication to treat molluscum in the pediatric population, as early as age 6 months, that can be used at home would “change the whole therapeutic paradigm,” one of the study authors, Adelaide Hebert, MD, said in an interview at the Society for Pediatric Dermatology annual meeting in July, where she presented phase 2 data on berdazimer gel. “This is a common problem and the rate of infections among siblings if it goes untreated is 41%. Affected kids have a sense of isolation; they don’t get invited to swimming parties.”
The lack of a safe and effective topical therapy “has been challenging,” added Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. She noted that treatments that have been used but have not been successful include imiquimod. “I’m not impressed with tretinoin,” although it is prescribed for MC, and the most common treatment prescribed by pediatricians for molluscum – mupirocin – is “usually not effective,” she said.
Another MC treatment in trials
Another investigative treatment for molluscum contagiosum, VP-102, a drug-device combination of cantharidin 0.7% administered through a single-use precision applicator, has been evaluated in phase 3 studies of patients with MC aged 2 years and older. The results of two phase 3 studies were published in 2020.
In May 2022, Verrica Pharmaceuticals, which is developing VP-102, announced that Food and Drug Administration approval had been delayed because of deficiencies identified at a contract manufacturing organization, and that the company was working with the agency to bring VP-102 to the market as soon as possible.
A step in the right direction
Although MC is self-resolving, cases last an average of 13.5 months, and “many families look to fast-forward their child’s experience with the infection,” Vikash S. Oza, MD, a pediatric dermatologist at New York University, New York, wrote in an editorial that accompanied the berdazimer study.
“To truly create a paradigm shift in the decision to treat MC, a therapeutic treatment would need to be developed that would lead to resolution of the infection over a short time frame (ideally, weeks) with minimal discomfort,” Dr. Oza noted. “Both VP-102 and berdazimer gel, 10.3%, have the potential to be the first-ever MC therapies approved by the U.S. Food and Drug Administration,” and families seeking to reduce MC in visible areas would welcome this option for a home therapy, he said.
However, Dr. Oza emphasized that potential barriers to widespread use of these therapies include whether the efficacy can be maintained in patients who fail to comply with daily application, and the ongoing need for office-based therapy to manage sexually transmitted MC in adults and periocular and perianal MC in children. The study was funded by Novan. Lead author Dr. Browning disclosed grants from Novan during the conduct of the study; Dr. Hebert reported grants from the University of Texas Health Science Center McGovern Medical School-Houston during the conduct of the study. Disclosures of other authors included having reported equity in Novan during the conduct of the study and receiving a grant from Novan. Dr. Oza had no financial conflicts to disclose.
Treatment with .
Molluscum contagiosum (MC) remains a common infection that, despite being self-limiting, may persist for months or years, and is associated with quality of life concerns and the need for ongoing therapy, wrote John C. Browning, MD, of Texas Dermatology and Laser Specialists, San Antonio, and colleagues, who conducted the phase 3 randomized study.
The infection is most common in children aged 1-14 years, and treatment may be needed in part to avoid infecting peers and family members, they said. No treatments for molluscum are currently approved by the Food and Drug Administration.
In the study, which was published in JAMA Dermatology, the researchers randomized 444 patients to berdazimer gel 10.3% and 447 to a placebo gel, applied once daily in a thin layer on all MC lesions for 12 weeks. The study was conducted at 55 clinics across the United States between Sept. 1, 2020, and July 21, 2021. The mean age of the patients was about 6.5 years (range was 0.9-49 years), and about 85% were White. Participants had 3-70 raised MC lesions; those with sexually transmitted MC or MC in the periocular area were excluded. The primary endpoint was complete clearance of MC lesions after 12 weeks of treatment. At 12 weeks, significantly more patients treated with berdazimer gel achieved complete clearance than those on vehicle (32.4% vs. 19.7%; P < .001). A total of 64 (14.4%) patients in the berdazimer group discontinued treatment because of MC clearance, compared with 40 patients (8.9%) in the vehicle group.
Most adverse events were mild or moderate, and rates of adverse events resulting in treatment discontinuation were low overall for both groups; the most common adverse events were application-site pain and erythema, which were mostly mild. Overall, 4.1% of berdazimer-treated patients and 0.7% of placebo patients discontinued the study because of adverse events.
The study findings were limited by several factors, including the small number of patients in subgroups for race, ethnicity, and age; and the lack of data on patients with sexually transmitted MC and on concomitant use with other topical MC therapies, the researchers noted.
However, the results represent the largest randomized clinical trial of berdazimer 10.3% to date, and support its potential as a first-line therapy for MC patients aged 6 months and older, according to the authors. “Berdazimer is under consideration as a first in-class therapeutic agent for MC and may provide a topical prescription alternative to other therapies used for this highly contagious and psychosocially challenging skin condition,” they said.
Having a reliable, steroid-free, safe, and efficacious medication to treat molluscum in the pediatric population, as early as age 6 months, that can be used at home would “change the whole therapeutic paradigm,” one of the study authors, Adelaide Hebert, MD, said in an interview at the Society for Pediatric Dermatology annual meeting in July, where she presented phase 2 data on berdazimer gel. “This is a common problem and the rate of infections among siblings if it goes untreated is 41%. Affected kids have a sense of isolation; they don’t get invited to swimming parties.”
The lack of a safe and effective topical therapy “has been challenging,” added Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. She noted that treatments that have been used but have not been successful include imiquimod. “I’m not impressed with tretinoin,” although it is prescribed for MC, and the most common treatment prescribed by pediatricians for molluscum – mupirocin – is “usually not effective,” she said.
Another MC treatment in trials
Another investigative treatment for molluscum contagiosum, VP-102, a drug-device combination of cantharidin 0.7% administered through a single-use precision applicator, has been evaluated in phase 3 studies of patients with MC aged 2 years and older. The results of two phase 3 studies were published in 2020.
In May 2022, Verrica Pharmaceuticals, which is developing VP-102, announced that Food and Drug Administration approval had been delayed because of deficiencies identified at a contract manufacturing organization, and that the company was working with the agency to bring VP-102 to the market as soon as possible.
A step in the right direction
Although MC is self-resolving, cases last an average of 13.5 months, and “many families look to fast-forward their child’s experience with the infection,” Vikash S. Oza, MD, a pediatric dermatologist at New York University, New York, wrote in an editorial that accompanied the berdazimer study.
“To truly create a paradigm shift in the decision to treat MC, a therapeutic treatment would need to be developed that would lead to resolution of the infection over a short time frame (ideally, weeks) with minimal discomfort,” Dr. Oza noted. “Both VP-102 and berdazimer gel, 10.3%, have the potential to be the first-ever MC therapies approved by the U.S. Food and Drug Administration,” and families seeking to reduce MC in visible areas would welcome this option for a home therapy, he said.
However, Dr. Oza emphasized that potential barriers to widespread use of these therapies include whether the efficacy can be maintained in patients who fail to comply with daily application, and the ongoing need for office-based therapy to manage sexually transmitted MC in adults and periocular and perianal MC in children. The study was funded by Novan. Lead author Dr. Browning disclosed grants from Novan during the conduct of the study; Dr. Hebert reported grants from the University of Texas Health Science Center McGovern Medical School-Houston during the conduct of the study. Disclosures of other authors included having reported equity in Novan during the conduct of the study and receiving a grant from Novan. Dr. Oza had no financial conflicts to disclose.
FROM JAMA DERMATOLOGY
Moderate drinking shows more benefit for older vs. younger adults
The health risks and benefits of moderate alcohol consumption are complex and remain a hot topic of debate. The data suggest that small amounts of alcohol may reduce the risk of certain health outcomes over time, but increase the risk of others, wrote Dana Bryazka, MS, a researcher at the Institute for Health Metrics and Evaluation (IHME) at the University of Washington, Seattle, and colleagues, in a paper published in the Lancet.
“The amount of alcohol that minimizes health loss is likely to depend on the distribution of underlying causes of disease burden in a given population. Since this distribution varies widely by geography, age, sex, and time, the level of alcohol consumption associated with the lowest risk to health would depend on the age structure and disease composition of that population,” the researchers wrote.
“We estimate that 1.78 million people worldwide died due to alcohol use in 2020,” Ms. Bryazka said in an interview. “It is important that alcohol consumption guidelines and policies are updated to minimize this harm, particularly in the populations at greatest risk,” she said.
“Existing alcohol consumption guidelines frequently vary by sex, with higher consumption thresholds set for males compared to females. Interestingly, with the currently available data we do not see evidence that risk of alcohol use varies by sex,” she noted.
Methods and results
In the study, the researchers conducted a systematic analysis of burden-weighted dose-response relative risk curves across 22 health outcomes. They used disease rates from the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2020 for the years 1990-2020 for 21 regions, including 204 countries and territories. The data were analyzed by 5-year age group, sex, and year for individuals aged 15-95 years and older. The researchers estimated the theoretical minimum risk exposure level (TMREL) and nondrinker equivalent (NDE), meaning the amount of alcohol at which the health risk equals that of a nondrinker.
One standard drink was defined as 10 g of pure alcohol, equivalent to a small glass of red wine (100 mL or 3.4 fluid ounces) at 13% alcohol by volume, a can or bottle of beer (375 mL or 12 fluid ounces) at 3.5% alcohol by volume, or a shot of whiskey or other spirits (30 mL or 1.0 fluid ounces) at 40% alcohol by volume.
Overall, the TMREL was low regardless of age, sex, time, or geography, and varied from 0 to 1.87 standard drinks per day. However, it was lowest for males aged 15-39 years (0.136 drinks per day) and only slightly higher for females aged 15-39 (0.273), representing 1-2 tenths of a standard drink.
For adults aged 40 and older without any underlying health conditions, drinking a small amount of alcohol may provide some benefits, such as reducing the risk of ischemic heart disease, stroke, and diabetes, the researchers noted. In general, for individuals aged 40-64 years, TMRELs ranged from about half a standard drink per day (0.527 drinks for males and 0.562 standard drinks per day for females) to almost two standard drinks (1.69 standard drinks per day for males and 1.82 for females). For those older than 65 years, the TMRELs represented just over 3 standard drinks per day (3.19 for males and 3.51 for females). For individuals aged 40 years and older, the distribution of disease burden varied by region, but was J-shaped across all regions, the researchers noted.
The researchers also found that those individuals consuming harmful amounts of alcohol were most likely to be aged 15-39 (59.1%) and male (76.9%).
The study findings were limited by several factors including the observational design and lack of data on drinking patterns, such as binge drinking, the researchers noted. Other limitations include the lack of data reflecting patterns of alcohol consumption during the COVID-19 pandemic, and exclusion of outcomes often associated with alcohol use, such as depression, anxiety, and dementia, that might reduce estimates of TMREL and NDE.
However, the results add to the ongoing discussion of the relationship between moderate alcohol consumption and health, the researchers said.
“The findings of this study support the development of tailored guidelines and recommendations on alcohol consumption by age and across regions and highlight that existing low consumption thresholds are too high for younger populations in all regions,” they concluded.
Consider individual factors when counseling patients
The takeaway message for primary care is that alcohol consumed in moderation can reduce the risk of ischemic heart disease, stroke, and diabetes, Ms. Bryazka noted. “However, it also increases the risk of many cancers, intentional and unintentional injuries, and infectious diseases like tuberculosis,” she said. “Of these health outcomes, young people are most likely to experience injuries, and as a result, we find that there are significant health risks associated with consuming alcohol for young people. Among older individuals, the relative proportions of these outcomes vary by geography, and so do the risks associated with consuming alcohol,” she explained.
“Importantly, our analysis was conducted at the population level; when evaluating risk at the individual level, it is also important to consider other factors such as the presence of comorbidities and interactions between alcohol and medications,” she emphasized.
Health and alcohol interaction is complicated
“These findings seemingly contradict a previous [Global Burden of Diseases, Injuries, and Risk Factors Study] estimate published in The Lancet, which emphasized that any alcohol use, regardless of amount, leads to health loss across populations,” wrote Robyn Burton, PhD, and Nick Sheron, MD, both of King’s College, London, in an accompanying comment.
However, the novel methods of weighting relative risk curves according to levels of underlying disease drive the difference in results, along with disaggregated estimates by age, sex, and region, they said.
“Across most geographical regions in this latest analysis, injuries accounted for most alcohol-related harm in younger age groups. This led to a minimum risk level of zero, or very close to zero, among individuals aged 15-39 years across all geographical regions,” which is lower than the level for older adults because of the shift in alcohol-related disease burden towards cardiovascular disease and cancers, they said. “This highlights the need to consider existing rates of disease in a population when trying to determine the total harm posed by alcohol,” the commentators wrote.
In an additional commentary, Tony Rao, MD, a visiting clinical research fellow in psychiatry at King’s College, London, noted that “the elephant in the room with this study is the interpretation of risk based on outcomes for cardiovascular disease – particularly in older people. We know that any purported health benefits from alcohol on the heart and circulation are balanced out by the increased risk from other conditions such as cancer, liver disease, and mental disorders such as depression and dementia,” Dr. Rao said. “If we are to simply draw the conclusion that older people should continue or start drinking small amounts because it protects against diseases affecting heart and circulation – which still remains controversial – other lifestyle changes or the use of drugs targeted at individual cardiovascular disorders seem like a less harmful way of improving health and wellbeing.”
Data can guide clinical practice
No previous study has examined the effect of the theoretical minimum risk of alcohol consumption by geography, age, sex, and time in the context of background disease rates, said Noel Deep, MD, in an interview.
“This study enabled the researchers to quantify the proportion of the population that consumed alcohol in amounts that exceeded the thresholds by location, age, sex, and year, and this can serve as a guide in our efforts to target the control of alcohol intake by individuals,” said Dr. Deep, a general internist in private practice in Antigo, Wisc. He also serves as chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo.
The first take-home message for clinicians is that even low levels of alcohol consumption can have deleterious effects on the health of patients, and patients should be advised accordingly based on the prevalence of diseases in that community and geographic area, Dr. Deep said. “Secondly, clinicians should also consider the risk of alcohol consumption on all forms of health impacts in a given population rather than just focusing on alcohol-related health conditions,” he added.
“This study provides us with the data to tailor our efforts in educating the clinicians and the public about the relationship between alcohol consumption and disease outcomes based on the observed disease rates in each population,” Dr. Deep explained. “The data should provide another reason for physicians to advise their younger patients, especially the younger males, to avoid or minimize alcohol use,” he said. The data also can help clinicians formulate public health messaging and community education to reduce harmful alcohol use, he added.
As for additional research, Dr. Deep said he would like to see data on the difference in the health-related effects of alcohol in binge-drinkers vs. those who regularly consume alcohol on a daily basis. “It would probably also be helpful to figure out what type of alcohol is being studied and the quality of the alcohol,” he said.
The study was supported by the Bill and Melinda Gates Foundation. Ms. Bryazka and colleagues had no financial conflicts to disclose. Dr. Burton disclosed serving as a consultant to the World Health Organization European Office for the Prevention and Control of Noncommunicable Diseases. Dr. Sheron had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Internal Medicine News.
The study was supported by the Bill and Melinda Gates Foundation.
The health risks and benefits of moderate alcohol consumption are complex and remain a hot topic of debate. The data suggest that small amounts of alcohol may reduce the risk of certain health outcomes over time, but increase the risk of others, wrote Dana Bryazka, MS, a researcher at the Institute for Health Metrics and Evaluation (IHME) at the University of Washington, Seattle, and colleagues, in a paper published in the Lancet.
“The amount of alcohol that minimizes health loss is likely to depend on the distribution of underlying causes of disease burden in a given population. Since this distribution varies widely by geography, age, sex, and time, the level of alcohol consumption associated with the lowest risk to health would depend on the age structure and disease composition of that population,” the researchers wrote.
“We estimate that 1.78 million people worldwide died due to alcohol use in 2020,” Ms. Bryazka said in an interview. “It is important that alcohol consumption guidelines and policies are updated to minimize this harm, particularly in the populations at greatest risk,” she said.
“Existing alcohol consumption guidelines frequently vary by sex, with higher consumption thresholds set for males compared to females. Interestingly, with the currently available data we do not see evidence that risk of alcohol use varies by sex,” she noted.
Methods and results
In the study, the researchers conducted a systematic analysis of burden-weighted dose-response relative risk curves across 22 health outcomes. They used disease rates from the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2020 for the years 1990-2020 for 21 regions, including 204 countries and territories. The data were analyzed by 5-year age group, sex, and year for individuals aged 15-95 years and older. The researchers estimated the theoretical minimum risk exposure level (TMREL) and nondrinker equivalent (NDE), meaning the amount of alcohol at which the health risk equals that of a nondrinker.
One standard drink was defined as 10 g of pure alcohol, equivalent to a small glass of red wine (100 mL or 3.4 fluid ounces) at 13% alcohol by volume, a can or bottle of beer (375 mL or 12 fluid ounces) at 3.5% alcohol by volume, or a shot of whiskey or other spirits (30 mL or 1.0 fluid ounces) at 40% alcohol by volume.
Overall, the TMREL was low regardless of age, sex, time, or geography, and varied from 0 to 1.87 standard drinks per day. However, it was lowest for males aged 15-39 years (0.136 drinks per day) and only slightly higher for females aged 15-39 (0.273), representing 1-2 tenths of a standard drink.
For adults aged 40 and older without any underlying health conditions, drinking a small amount of alcohol may provide some benefits, such as reducing the risk of ischemic heart disease, stroke, and diabetes, the researchers noted. In general, for individuals aged 40-64 years, TMRELs ranged from about half a standard drink per day (0.527 drinks for males and 0.562 standard drinks per day for females) to almost two standard drinks (1.69 standard drinks per day for males and 1.82 for females). For those older than 65 years, the TMRELs represented just over 3 standard drinks per day (3.19 for males and 3.51 for females). For individuals aged 40 years and older, the distribution of disease burden varied by region, but was J-shaped across all regions, the researchers noted.
The researchers also found that those individuals consuming harmful amounts of alcohol were most likely to be aged 15-39 (59.1%) and male (76.9%).
The study findings were limited by several factors including the observational design and lack of data on drinking patterns, such as binge drinking, the researchers noted. Other limitations include the lack of data reflecting patterns of alcohol consumption during the COVID-19 pandemic, and exclusion of outcomes often associated with alcohol use, such as depression, anxiety, and dementia, that might reduce estimates of TMREL and NDE.
However, the results add to the ongoing discussion of the relationship between moderate alcohol consumption and health, the researchers said.
“The findings of this study support the development of tailored guidelines and recommendations on alcohol consumption by age and across regions and highlight that existing low consumption thresholds are too high for younger populations in all regions,” they concluded.
Consider individual factors when counseling patients
The takeaway message for primary care is that alcohol consumed in moderation can reduce the risk of ischemic heart disease, stroke, and diabetes, Ms. Bryazka noted. “However, it also increases the risk of many cancers, intentional and unintentional injuries, and infectious diseases like tuberculosis,” she said. “Of these health outcomes, young people are most likely to experience injuries, and as a result, we find that there are significant health risks associated with consuming alcohol for young people. Among older individuals, the relative proportions of these outcomes vary by geography, and so do the risks associated with consuming alcohol,” she explained.
“Importantly, our analysis was conducted at the population level; when evaluating risk at the individual level, it is also important to consider other factors such as the presence of comorbidities and interactions between alcohol and medications,” she emphasized.
Health and alcohol interaction is complicated
“These findings seemingly contradict a previous [Global Burden of Diseases, Injuries, and Risk Factors Study] estimate published in The Lancet, which emphasized that any alcohol use, regardless of amount, leads to health loss across populations,” wrote Robyn Burton, PhD, and Nick Sheron, MD, both of King’s College, London, in an accompanying comment.
However, the novel methods of weighting relative risk curves according to levels of underlying disease drive the difference in results, along with disaggregated estimates by age, sex, and region, they said.
“Across most geographical regions in this latest analysis, injuries accounted for most alcohol-related harm in younger age groups. This led to a minimum risk level of zero, or very close to zero, among individuals aged 15-39 years across all geographical regions,” which is lower than the level for older adults because of the shift in alcohol-related disease burden towards cardiovascular disease and cancers, they said. “This highlights the need to consider existing rates of disease in a population when trying to determine the total harm posed by alcohol,” the commentators wrote.
In an additional commentary, Tony Rao, MD, a visiting clinical research fellow in psychiatry at King’s College, London, noted that “the elephant in the room with this study is the interpretation of risk based on outcomes for cardiovascular disease – particularly in older people. We know that any purported health benefits from alcohol on the heart and circulation are balanced out by the increased risk from other conditions such as cancer, liver disease, and mental disorders such as depression and dementia,” Dr. Rao said. “If we are to simply draw the conclusion that older people should continue or start drinking small amounts because it protects against diseases affecting heart and circulation – which still remains controversial – other lifestyle changes or the use of drugs targeted at individual cardiovascular disorders seem like a less harmful way of improving health and wellbeing.”
Data can guide clinical practice
No previous study has examined the effect of the theoretical minimum risk of alcohol consumption by geography, age, sex, and time in the context of background disease rates, said Noel Deep, MD, in an interview.
“This study enabled the researchers to quantify the proportion of the population that consumed alcohol in amounts that exceeded the thresholds by location, age, sex, and year, and this can serve as a guide in our efforts to target the control of alcohol intake by individuals,” said Dr. Deep, a general internist in private practice in Antigo, Wisc. He also serves as chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo.
The first take-home message for clinicians is that even low levels of alcohol consumption can have deleterious effects on the health of patients, and patients should be advised accordingly based on the prevalence of diseases in that community and geographic area, Dr. Deep said. “Secondly, clinicians should also consider the risk of alcohol consumption on all forms of health impacts in a given population rather than just focusing on alcohol-related health conditions,” he added.
“This study provides us with the data to tailor our efforts in educating the clinicians and the public about the relationship between alcohol consumption and disease outcomes based on the observed disease rates in each population,” Dr. Deep explained. “The data should provide another reason for physicians to advise their younger patients, especially the younger males, to avoid or minimize alcohol use,” he said. The data also can help clinicians formulate public health messaging and community education to reduce harmful alcohol use, he added.
As for additional research, Dr. Deep said he would like to see data on the difference in the health-related effects of alcohol in binge-drinkers vs. those who regularly consume alcohol on a daily basis. “It would probably also be helpful to figure out what type of alcohol is being studied and the quality of the alcohol,” he said.
The study was supported by the Bill and Melinda Gates Foundation. Ms. Bryazka and colleagues had no financial conflicts to disclose. Dr. Burton disclosed serving as a consultant to the World Health Organization European Office for the Prevention and Control of Noncommunicable Diseases. Dr. Sheron had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Internal Medicine News.
The study was supported by the Bill and Melinda Gates Foundation.
The health risks and benefits of moderate alcohol consumption are complex and remain a hot topic of debate. The data suggest that small amounts of alcohol may reduce the risk of certain health outcomes over time, but increase the risk of others, wrote Dana Bryazka, MS, a researcher at the Institute for Health Metrics and Evaluation (IHME) at the University of Washington, Seattle, and colleagues, in a paper published in the Lancet.
“The amount of alcohol that minimizes health loss is likely to depend on the distribution of underlying causes of disease burden in a given population. Since this distribution varies widely by geography, age, sex, and time, the level of alcohol consumption associated with the lowest risk to health would depend on the age structure and disease composition of that population,” the researchers wrote.
“We estimate that 1.78 million people worldwide died due to alcohol use in 2020,” Ms. Bryazka said in an interview. “It is important that alcohol consumption guidelines and policies are updated to minimize this harm, particularly in the populations at greatest risk,” she said.
“Existing alcohol consumption guidelines frequently vary by sex, with higher consumption thresholds set for males compared to females. Interestingly, with the currently available data we do not see evidence that risk of alcohol use varies by sex,” she noted.
Methods and results
In the study, the researchers conducted a systematic analysis of burden-weighted dose-response relative risk curves across 22 health outcomes. They used disease rates from the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2020 for the years 1990-2020 for 21 regions, including 204 countries and territories. The data were analyzed by 5-year age group, sex, and year for individuals aged 15-95 years and older. The researchers estimated the theoretical minimum risk exposure level (TMREL) and nondrinker equivalent (NDE), meaning the amount of alcohol at which the health risk equals that of a nondrinker.
One standard drink was defined as 10 g of pure alcohol, equivalent to a small glass of red wine (100 mL or 3.4 fluid ounces) at 13% alcohol by volume, a can or bottle of beer (375 mL or 12 fluid ounces) at 3.5% alcohol by volume, or a shot of whiskey or other spirits (30 mL or 1.0 fluid ounces) at 40% alcohol by volume.
Overall, the TMREL was low regardless of age, sex, time, or geography, and varied from 0 to 1.87 standard drinks per day. However, it was lowest for males aged 15-39 years (0.136 drinks per day) and only slightly higher for females aged 15-39 (0.273), representing 1-2 tenths of a standard drink.
For adults aged 40 and older without any underlying health conditions, drinking a small amount of alcohol may provide some benefits, such as reducing the risk of ischemic heart disease, stroke, and diabetes, the researchers noted. In general, for individuals aged 40-64 years, TMRELs ranged from about half a standard drink per day (0.527 drinks for males and 0.562 standard drinks per day for females) to almost two standard drinks (1.69 standard drinks per day for males and 1.82 for females). For those older than 65 years, the TMRELs represented just over 3 standard drinks per day (3.19 for males and 3.51 for females). For individuals aged 40 years and older, the distribution of disease burden varied by region, but was J-shaped across all regions, the researchers noted.
The researchers also found that those individuals consuming harmful amounts of alcohol were most likely to be aged 15-39 (59.1%) and male (76.9%).
The study findings were limited by several factors including the observational design and lack of data on drinking patterns, such as binge drinking, the researchers noted. Other limitations include the lack of data reflecting patterns of alcohol consumption during the COVID-19 pandemic, and exclusion of outcomes often associated with alcohol use, such as depression, anxiety, and dementia, that might reduce estimates of TMREL and NDE.
However, the results add to the ongoing discussion of the relationship between moderate alcohol consumption and health, the researchers said.
“The findings of this study support the development of tailored guidelines and recommendations on alcohol consumption by age and across regions and highlight that existing low consumption thresholds are too high for younger populations in all regions,” they concluded.
Consider individual factors when counseling patients
The takeaway message for primary care is that alcohol consumed in moderation can reduce the risk of ischemic heart disease, stroke, and diabetes, Ms. Bryazka noted. “However, it also increases the risk of many cancers, intentional and unintentional injuries, and infectious diseases like tuberculosis,” she said. “Of these health outcomes, young people are most likely to experience injuries, and as a result, we find that there are significant health risks associated with consuming alcohol for young people. Among older individuals, the relative proportions of these outcomes vary by geography, and so do the risks associated with consuming alcohol,” she explained.
“Importantly, our analysis was conducted at the population level; when evaluating risk at the individual level, it is also important to consider other factors such as the presence of comorbidities and interactions between alcohol and medications,” she emphasized.
Health and alcohol interaction is complicated
“These findings seemingly contradict a previous [Global Burden of Diseases, Injuries, and Risk Factors Study] estimate published in The Lancet, which emphasized that any alcohol use, regardless of amount, leads to health loss across populations,” wrote Robyn Burton, PhD, and Nick Sheron, MD, both of King’s College, London, in an accompanying comment.
However, the novel methods of weighting relative risk curves according to levels of underlying disease drive the difference in results, along with disaggregated estimates by age, sex, and region, they said.
“Across most geographical regions in this latest analysis, injuries accounted for most alcohol-related harm in younger age groups. This led to a minimum risk level of zero, or very close to zero, among individuals aged 15-39 years across all geographical regions,” which is lower than the level for older adults because of the shift in alcohol-related disease burden towards cardiovascular disease and cancers, they said. “This highlights the need to consider existing rates of disease in a population when trying to determine the total harm posed by alcohol,” the commentators wrote.
In an additional commentary, Tony Rao, MD, a visiting clinical research fellow in psychiatry at King’s College, London, noted that “the elephant in the room with this study is the interpretation of risk based on outcomes for cardiovascular disease – particularly in older people. We know that any purported health benefits from alcohol on the heart and circulation are balanced out by the increased risk from other conditions such as cancer, liver disease, and mental disorders such as depression and dementia,” Dr. Rao said. “If we are to simply draw the conclusion that older people should continue or start drinking small amounts because it protects against diseases affecting heart and circulation – which still remains controversial – other lifestyle changes or the use of drugs targeted at individual cardiovascular disorders seem like a less harmful way of improving health and wellbeing.”
Data can guide clinical practice
No previous study has examined the effect of the theoretical minimum risk of alcohol consumption by geography, age, sex, and time in the context of background disease rates, said Noel Deep, MD, in an interview.
“This study enabled the researchers to quantify the proportion of the population that consumed alcohol in amounts that exceeded the thresholds by location, age, sex, and year, and this can serve as a guide in our efforts to target the control of alcohol intake by individuals,” said Dr. Deep, a general internist in private practice in Antigo, Wisc. He also serves as chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo.
The first take-home message for clinicians is that even low levels of alcohol consumption can have deleterious effects on the health of patients, and patients should be advised accordingly based on the prevalence of diseases in that community and geographic area, Dr. Deep said. “Secondly, clinicians should also consider the risk of alcohol consumption on all forms of health impacts in a given population rather than just focusing on alcohol-related health conditions,” he added.
“This study provides us with the data to tailor our efforts in educating the clinicians and the public about the relationship between alcohol consumption and disease outcomes based on the observed disease rates in each population,” Dr. Deep explained. “The data should provide another reason for physicians to advise their younger patients, especially the younger males, to avoid or minimize alcohol use,” he said. The data also can help clinicians formulate public health messaging and community education to reduce harmful alcohol use, he added.
As for additional research, Dr. Deep said he would like to see data on the difference in the health-related effects of alcohol in binge-drinkers vs. those who regularly consume alcohol on a daily basis. “It would probably also be helpful to figure out what type of alcohol is being studied and the quality of the alcohol,” he said.
The study was supported by the Bill and Melinda Gates Foundation. Ms. Bryazka and colleagues had no financial conflicts to disclose. Dr. Burton disclosed serving as a consultant to the World Health Organization European Office for the Prevention and Control of Noncommunicable Diseases. Dr. Sheron had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Internal Medicine News.
The study was supported by the Bill and Melinda Gates Foundation.
FROM THE LANCET
Liver cancer risk persists after direct-acting antiviral treatment for HCV
, according to a new report.
Among patients with cirrhosis and fibrosis-4 (FIB-4) scores of 3.25 or higher, the incidence of hepatocellular carcinoma appeared to decline progressively each year up to 7 years after a sustained virologic response, although the rate remained above the 1% per year threshold that warrants screening.
“The majority of patients with hepatitis C have been treated and cured in the United States,” George Ioannou, MD, the senior study author and professor of medicine at the University of Washington, Seattle, said in an interview. “After hepatitis C eradication, these patients generally do very well from the liver standpoint, but the one thing they have to continue worrying about is development of liver cancer.”
Dr. Ioannou, who is also director of hepatology at the Veterans Affairs Puget Sound Health Care System, Seattle, noted that patients may be screened “indefinitely,” which places a burden on the patients and the health care system.
“We are still not sure to what extent the risk of liver cancer declines after hepatitis C eradication as more and more time accrues,” he said. “In those who had cirrhosis of the liver prior to hepatitis C cure, we are still not certain if there is a time point after hepatitis C cure when we can tell a patient that their risk of liver cancer is now very low and we no longer need to keep screening for liver cancer.”
The study was published online in Gastroenterology.
Risk calculations
In a previous study, Dr. Ioannou and colleagues found that hepatocellular carcinoma risk declined during the first 4 years of follow-up after a sustained virologic response from direct-acting antiviral medications. But the follow-up time wasn’t long enough to determine whether the cancer risk continues to decline to levels low enough to forgo screening.
In this study, Dr. Ioannou and colleagues extended the follow-up to 7 years. They were curious to see whether the cancer risk declines enough to drop the screening requirement, particularly as related to pretreatment cirrhosis and fibrosis-4 scores.
The research team analyzed electronic health records from the Veterans Affairs Corporate Data Warehouse, a national repository of Veterans Health Administration records developed specifically for research purposes.
The researchers included 29,033 patients in the Veterans Affairs health care system who had been infected with hepatitis C virus and were treated with direct-acting antivirals between January 2013 and December 2015. The patients had a sustained virologic response, which is defined as a viral load below the lower limit of detection at least 12 weeks after therapy completion.
The patients were followed for incident hepatocellular carcinoma until December 2021. The researchers then calculated the annual incidence during each year of follow-up after treatment.
About 96.6% of patients were men, and 52.2% were non-Hispanic White persons. The average age was 61 years. The most common conditions were alcohol use disorder (43.7%), substance use disorder (37.7%), and diabetes (28.9%).
Among the 7,533 patients with pretreatment cirrhosis, 948 (12.6%) developed hepatocellular carcinoma during a mean follow-up period of 4.9 years. Among patients with FIB-4 scores of 3.25 or higher, the annual incidence decreased from 3.8% in the first year to 1.4% in the seventh year but remained substantial up to 7 years after sustained virologic response. Among patients with both cirrhosis and a high FIB-4 score, the annual rate ranged from 0.7% to 1.3% and didn’t change significantly over time.
Among the 21,500 patients without pretreatment cirrhosis, 541 (or 2.5%) developed hepatocellular carcinoma during a mean follow-up period of 5.4 years. The incidence rate was significantly higher for patients with high FIB-4 scores. Among patients without cirrhosis but who had a high FIB-4 score, the annual rate remained stable but substantial (from 0.8% to 1.3%) for up to 7 years.
In a subgroup analysis that examined incidence according to changes in FIB-4 scores before and after treatment, the rate remained high among those with cirrhosis regardless of a score change. Among those without cirrhosis but who had a persistently high FIB-4 score, the incidence was high. In those without cirrhosis whose FIB-4 score dropped, the incidence was lower.
“The study demonstrates a clear decline in the risk of liver cancer over time after hepatitis C cure in the highest-risk group. This is very positive news for patients,” Dr. Ioannou said. “However, even with that decline in risk up to 7 years after eradication of hepatitis C with direct-acting antivirals, the risk is still high enough to warrant liver cancer screening.”
Future concerns
For a follow-up study, Dr. Ioannou and colleagues plan to adjust their analyses for other factors that influence the risk of liver cancer, such as age and nonalcoholic fatty liver disease. Other studies could increase the follow-up time beyond 7 years and assess how changes in diabetes, weight management, and alcohol use might affect liver cancer risk.
“With the availability of safe and effective direct-acting antiviral treatments, a growing number of patients have been or will be treated and cured of their hepatitis C infection,” Nicole Kim, MD, one of the lead authors and a transplant hepatology fellow at the University of Washington, Seattle, told this news organization.
“It is therefore important for us to develop a better understanding of how liver cancer risk might change after treatment, so we can improve the care we provide to this patient population,” she said.
The results require validation in nonveteran cohorts, the study authors write, as well as follow-up after the COVID-19 pandemic, when screening and diagnostic practices were restricted.
“Several studies have demonstrated that HCC [hepatocellular carcinoma] surveillance is underused in clinical practice, including in patients after [sustained virologic response],” Amit Singal, MD, clinical chief of hepatology and medical director of the liver tumor program at the University of Texas Southwestern Medical Center, told this news organization.
Dr. Singal, who wasn’t involved with this study, is evaluating several intervention strategies to increase surveillance utilization. His research group is conducting a multicenter randomized trial using mailed outreach invitations and is also evaluating a biomarker, PLSec-AFP, to identify patients with the highest risks who may warrant more intensive surveillance strategies.
“We have recently validated the performance of this biomarker in a large cohort of patients with cirrhosis, including some with cured hepatitis C virus infection,” he said.
The study was funded by an NIH/NCI grant and a VA CSR under Dr. Ioannou. The manuscript writing was supported by the NIH under Dr. Kim and co-author Philip Vutien. Dr. Singal has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to a new report.
Among patients with cirrhosis and fibrosis-4 (FIB-4) scores of 3.25 or higher, the incidence of hepatocellular carcinoma appeared to decline progressively each year up to 7 years after a sustained virologic response, although the rate remained above the 1% per year threshold that warrants screening.
“The majority of patients with hepatitis C have been treated and cured in the United States,” George Ioannou, MD, the senior study author and professor of medicine at the University of Washington, Seattle, said in an interview. “After hepatitis C eradication, these patients generally do very well from the liver standpoint, but the one thing they have to continue worrying about is development of liver cancer.”
Dr. Ioannou, who is also director of hepatology at the Veterans Affairs Puget Sound Health Care System, Seattle, noted that patients may be screened “indefinitely,” which places a burden on the patients and the health care system.
“We are still not sure to what extent the risk of liver cancer declines after hepatitis C eradication as more and more time accrues,” he said. “In those who had cirrhosis of the liver prior to hepatitis C cure, we are still not certain if there is a time point after hepatitis C cure when we can tell a patient that their risk of liver cancer is now very low and we no longer need to keep screening for liver cancer.”
The study was published online in Gastroenterology.
Risk calculations
In a previous study, Dr. Ioannou and colleagues found that hepatocellular carcinoma risk declined during the first 4 years of follow-up after a sustained virologic response from direct-acting antiviral medications. But the follow-up time wasn’t long enough to determine whether the cancer risk continues to decline to levels low enough to forgo screening.
In this study, Dr. Ioannou and colleagues extended the follow-up to 7 years. They were curious to see whether the cancer risk declines enough to drop the screening requirement, particularly as related to pretreatment cirrhosis and fibrosis-4 scores.
The research team analyzed electronic health records from the Veterans Affairs Corporate Data Warehouse, a national repository of Veterans Health Administration records developed specifically for research purposes.
The researchers included 29,033 patients in the Veterans Affairs health care system who had been infected with hepatitis C virus and were treated with direct-acting antivirals between January 2013 and December 2015. The patients had a sustained virologic response, which is defined as a viral load below the lower limit of detection at least 12 weeks after therapy completion.
The patients were followed for incident hepatocellular carcinoma until December 2021. The researchers then calculated the annual incidence during each year of follow-up after treatment.
About 96.6% of patients were men, and 52.2% were non-Hispanic White persons. The average age was 61 years. The most common conditions were alcohol use disorder (43.7%), substance use disorder (37.7%), and diabetes (28.9%).
Among the 7,533 patients with pretreatment cirrhosis, 948 (12.6%) developed hepatocellular carcinoma during a mean follow-up period of 4.9 years. Among patients with FIB-4 scores of 3.25 or higher, the annual incidence decreased from 3.8% in the first year to 1.4% in the seventh year but remained substantial up to 7 years after sustained virologic response. Among patients with both cirrhosis and a high FIB-4 score, the annual rate ranged from 0.7% to 1.3% and didn’t change significantly over time.
Among the 21,500 patients without pretreatment cirrhosis, 541 (or 2.5%) developed hepatocellular carcinoma during a mean follow-up period of 5.4 years. The incidence rate was significantly higher for patients with high FIB-4 scores. Among patients without cirrhosis but who had a high FIB-4 score, the annual rate remained stable but substantial (from 0.8% to 1.3%) for up to 7 years.
In a subgroup analysis that examined incidence according to changes in FIB-4 scores before and after treatment, the rate remained high among those with cirrhosis regardless of a score change. Among those without cirrhosis but who had a persistently high FIB-4 score, the incidence was high. In those without cirrhosis whose FIB-4 score dropped, the incidence was lower.
“The study demonstrates a clear decline in the risk of liver cancer over time after hepatitis C cure in the highest-risk group. This is very positive news for patients,” Dr. Ioannou said. “However, even with that decline in risk up to 7 years after eradication of hepatitis C with direct-acting antivirals, the risk is still high enough to warrant liver cancer screening.”
Future concerns
For a follow-up study, Dr. Ioannou and colleagues plan to adjust their analyses for other factors that influence the risk of liver cancer, such as age and nonalcoholic fatty liver disease. Other studies could increase the follow-up time beyond 7 years and assess how changes in diabetes, weight management, and alcohol use might affect liver cancer risk.
“With the availability of safe and effective direct-acting antiviral treatments, a growing number of patients have been or will be treated and cured of their hepatitis C infection,” Nicole Kim, MD, one of the lead authors and a transplant hepatology fellow at the University of Washington, Seattle, told this news organization.
“It is therefore important for us to develop a better understanding of how liver cancer risk might change after treatment, so we can improve the care we provide to this patient population,” she said.
The results require validation in nonveteran cohorts, the study authors write, as well as follow-up after the COVID-19 pandemic, when screening and diagnostic practices were restricted.
“Several studies have demonstrated that HCC [hepatocellular carcinoma] surveillance is underused in clinical practice, including in patients after [sustained virologic response],” Amit Singal, MD, clinical chief of hepatology and medical director of the liver tumor program at the University of Texas Southwestern Medical Center, told this news organization.
Dr. Singal, who wasn’t involved with this study, is evaluating several intervention strategies to increase surveillance utilization. His research group is conducting a multicenter randomized trial using mailed outreach invitations and is also evaluating a biomarker, PLSec-AFP, to identify patients with the highest risks who may warrant more intensive surveillance strategies.
“We have recently validated the performance of this biomarker in a large cohort of patients with cirrhosis, including some with cured hepatitis C virus infection,” he said.
The study was funded by an NIH/NCI grant and a VA CSR under Dr. Ioannou. The manuscript writing was supported by the NIH under Dr. Kim and co-author Philip Vutien. Dr. Singal has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to a new report.
Among patients with cirrhosis and fibrosis-4 (FIB-4) scores of 3.25 or higher, the incidence of hepatocellular carcinoma appeared to decline progressively each year up to 7 years after a sustained virologic response, although the rate remained above the 1% per year threshold that warrants screening.
“The majority of patients with hepatitis C have been treated and cured in the United States,” George Ioannou, MD, the senior study author and professor of medicine at the University of Washington, Seattle, said in an interview. “After hepatitis C eradication, these patients generally do very well from the liver standpoint, but the one thing they have to continue worrying about is development of liver cancer.”
Dr. Ioannou, who is also director of hepatology at the Veterans Affairs Puget Sound Health Care System, Seattle, noted that patients may be screened “indefinitely,” which places a burden on the patients and the health care system.
“We are still not sure to what extent the risk of liver cancer declines after hepatitis C eradication as more and more time accrues,” he said. “In those who had cirrhosis of the liver prior to hepatitis C cure, we are still not certain if there is a time point after hepatitis C cure when we can tell a patient that their risk of liver cancer is now very low and we no longer need to keep screening for liver cancer.”
The study was published online in Gastroenterology.
Risk calculations
In a previous study, Dr. Ioannou and colleagues found that hepatocellular carcinoma risk declined during the first 4 years of follow-up after a sustained virologic response from direct-acting antiviral medications. But the follow-up time wasn’t long enough to determine whether the cancer risk continues to decline to levels low enough to forgo screening.
In this study, Dr. Ioannou and colleagues extended the follow-up to 7 years. They were curious to see whether the cancer risk declines enough to drop the screening requirement, particularly as related to pretreatment cirrhosis and fibrosis-4 scores.
The research team analyzed electronic health records from the Veterans Affairs Corporate Data Warehouse, a national repository of Veterans Health Administration records developed specifically for research purposes.
The researchers included 29,033 patients in the Veterans Affairs health care system who had been infected with hepatitis C virus and were treated with direct-acting antivirals between January 2013 and December 2015. The patients had a sustained virologic response, which is defined as a viral load below the lower limit of detection at least 12 weeks after therapy completion.
The patients were followed for incident hepatocellular carcinoma until December 2021. The researchers then calculated the annual incidence during each year of follow-up after treatment.
About 96.6% of patients were men, and 52.2% were non-Hispanic White persons. The average age was 61 years. The most common conditions were alcohol use disorder (43.7%), substance use disorder (37.7%), and diabetes (28.9%).
Among the 7,533 patients with pretreatment cirrhosis, 948 (12.6%) developed hepatocellular carcinoma during a mean follow-up period of 4.9 years. Among patients with FIB-4 scores of 3.25 or higher, the annual incidence decreased from 3.8% in the first year to 1.4% in the seventh year but remained substantial up to 7 years after sustained virologic response. Among patients with both cirrhosis and a high FIB-4 score, the annual rate ranged from 0.7% to 1.3% and didn’t change significantly over time.
Among the 21,500 patients without pretreatment cirrhosis, 541 (or 2.5%) developed hepatocellular carcinoma during a mean follow-up period of 5.4 years. The incidence rate was significantly higher for patients with high FIB-4 scores. Among patients without cirrhosis but who had a high FIB-4 score, the annual rate remained stable but substantial (from 0.8% to 1.3%) for up to 7 years.
In a subgroup analysis that examined incidence according to changes in FIB-4 scores before and after treatment, the rate remained high among those with cirrhosis regardless of a score change. Among those without cirrhosis but who had a persistently high FIB-4 score, the incidence was high. In those without cirrhosis whose FIB-4 score dropped, the incidence was lower.
“The study demonstrates a clear decline in the risk of liver cancer over time after hepatitis C cure in the highest-risk group. This is very positive news for patients,” Dr. Ioannou said. “However, even with that decline in risk up to 7 years after eradication of hepatitis C with direct-acting antivirals, the risk is still high enough to warrant liver cancer screening.”
Future concerns
For a follow-up study, Dr. Ioannou and colleagues plan to adjust their analyses for other factors that influence the risk of liver cancer, such as age and nonalcoholic fatty liver disease. Other studies could increase the follow-up time beyond 7 years and assess how changes in diabetes, weight management, and alcohol use might affect liver cancer risk.
“With the availability of safe and effective direct-acting antiviral treatments, a growing number of patients have been or will be treated and cured of their hepatitis C infection,” Nicole Kim, MD, one of the lead authors and a transplant hepatology fellow at the University of Washington, Seattle, told this news organization.
“It is therefore important for us to develop a better understanding of how liver cancer risk might change after treatment, so we can improve the care we provide to this patient population,” she said.
The results require validation in nonveteran cohorts, the study authors write, as well as follow-up after the COVID-19 pandemic, when screening and diagnostic practices were restricted.
“Several studies have demonstrated that HCC [hepatocellular carcinoma] surveillance is underused in clinical practice, including in patients after [sustained virologic response],” Amit Singal, MD, clinical chief of hepatology and medical director of the liver tumor program at the University of Texas Southwestern Medical Center, told this news organization.
Dr. Singal, who wasn’t involved with this study, is evaluating several intervention strategies to increase surveillance utilization. His research group is conducting a multicenter randomized trial using mailed outreach invitations and is also evaluating a biomarker, PLSec-AFP, to identify patients with the highest risks who may warrant more intensive surveillance strategies.
“We have recently validated the performance of this biomarker in a large cohort of patients with cirrhosis, including some with cured hepatitis C virus infection,” he said.
The study was funded by an NIH/NCI grant and a VA CSR under Dr. Ioannou. The manuscript writing was supported by the NIH under Dr. Kim and co-author Philip Vutien. Dr. Singal has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM GASTROENTEROLOGY
LGBTQ+ Youth Consult: Let’s talk about PrEP!
As pediatricians, almost all of our clinic visits include some anticipatory guidance and recommendations on ways to promote well-being and prevent illness and injury for our patients. Because of minority stress, discrimination, and increased exposure to adverse childhood experiences, LGBTQ+ patients are disproportionately affected by certain health conditions including depression, anxiety, substance use, homelessness, as well as HIV and other sexually transmitted infections (STIs).1 While LGBTQ+ youth could benefit from additional guidance, counseling, and interventions related to these health disparities and have expressed interest in talking about these topics with their providers, sexual and gender minority youth also stress that they want to be treated as any other youth.2 Extending counseling for preventive care measures such as preexposure prophylaxis (PrEP) for HIV to all sexually active youth could help to destigmatize LGBTQ+ youth as being “different” from other youth and also help to increase overall access to HIV prevention services.3
Described by some as the “birth control” for HIV infection, PrEP is taken on an ongoing basis by those who are HIV negative before potential exposures to HIV in order to prevent new HIV infections. PrEP was first approved as a daily pill for adults in 2015 by the Food and Drug Administration with extension in 2018 to all individuals at risk for HIV weighing at least 35 kg after safety and efficacy data showed it could be used routinely for adolescents.4 When taken daily, oral PrEP can decrease the risk of HIV from sexual contact by more than 90% and from injection drug use by around 70%. As PrEP is highly effective with low risk for side effects, the U.S. Preventive Services Task Force (USPSTF) gave PrEP a “Grade A” recommendation for use in those at high risk for HIV infection in 2019.5 Since efficacy is closely tied to adherence, the first injectable PrEP (given at 0, 1, and 2 months with dosing then every 2 months) was also recently FDA approved in late 2021.6
Since HIV infection disproportionately affects LBGTQ+ individuals, and particularly LBGTQ+ youth of color, counseling related to PrEP has been largely targeted to these groups.7 Insurance and financial barriers to PrEP have been greatly reduced over the past several years through changes in insurance coverage (strengthened by the USPSTF recommendation), supplemental insurance programs, and pharmaceutical copay programs. Many states (but not all) also include HIV in the definition of STIs and allow minors to consent to PrEP services without a parent or guardian. Unfortunately, despite the high efficacy of PrEP and efforts to decrease barriers, rates of PrEP use continue to be extremely low, especially in youth, with only 15.6% of those aged 16-24 who are at risk for HIV in the United States actually taking PrEP in 2019.8 Many barriers to PrEP continue to exist including lack of awareness of PrEP, stigma surrounding HIV and PrEP, and lack of PrEP providers.
In order to address these low rates of PrEP uptake, the Centers for Disease Control and Prevention now recommends that medical providers discuss PrEP with all sexually active patients.6 PrEP should not be seen or discussed as something only relevant to LBGTQ+ populations, but rather as another tool in everyone’s “sexual health toolbox” that can allow us to experience human connection and pleasure through sexual activity while also having more control over what happens to our bodies. Not only will this allow more patients to access PrEP directly, it will also decrease the stigma of talking about HIV and PrEP and strengthen youths’ sense of autonomy and control over their own sexual health.
Since PrEP is a relatively new medical service, many providers will need to learn more about PrEP to at least have initial discussions with patients and to feel comfortable prescribing this themselves (See Resources). Below are also some suggestions to incorporate into your practice in order to advocate for the health and well-being of all your patients, including LGBTQ+ youth.
- Once your patients are 13 years and older, spend time with them alone to confidentially discuss more sensitive topics such as sexual health, mental health, and substance use.
- For all patients who are sexually active or considering sexual activity in the near future, discuss topics to help them control what happens to their bodies including consent, condoms, birth control, PrEP, and routine STI screening.
- Recommend PrEP to anyone who is sexually active and may be at increased risk for HIV infection or who is interested in taking PrEP for HIV prevention.
- Learn more about PrEP and start prescribing it to your own patients or become familiar with providers in your area to whom you could refer patients who are interested. While no certification is needed to prescribe PrEP, programs exist to help providers become more familiar with how to prescribe PrEP.
Dr. Warus is an adolescent medicine physician who specializes in care for transgender and gender-nonconforming youth, HIV prevention for adolescents and young adults, and LGBTQ health for youth at Children’s Hospital of Los Angeles. He is an assistant professor of clinical pediatrics and a University of Southern California faculty member.
Resources
CDC PrEP resources for clinicians: www.cdc.gov/hiv/clinicians/prevention/prep.html.Health HIV’s HIV Prevention Certified Provider Certification Program: https://healthhiv.org/programs/hpcp/.PrEP providers in the United States: https://preplocator.org/.Adolescent Health Working Group’s Sexual and Reproductive Health Toolkit for Adolescent Providers: https://ahwg.org/download/sexual-and-reproductive-health-toolkit-for-adolescent-providers/.
References
1. Lund EM and Burgess CM. Prim Care Clin Office Pract. 2021:48:179-89.
2. Hoffman ND et al. J Adolesc Health. 2009;45:222-9.
3. Mayer KH et al. Adv Ther. 2020;37:1778-811.
4. Hosek SG et al. JAMA Pediatr. 2017;171(11):1063-71.
5. U.S. Preventive Services Task Force; Owens DK et al. JAMA. 2019;321(22):2203-13.
6. Centers for Disease Control and Prevention: U.S. Public Health Service: Preexposure Prophylaxis for the Prevention of HIV Infection in the United States – 2021 Update: A Clinical Practice Guideline. Published 2021. Accessed July 10, 2022.
7. Centers for Disease Control and Prevention. Estimated HIV Incidence and Prevalence in the United States, 2015-2019. HIV Surveillance Supplemental Report. 2021;26(1). Published May 2021. Accessed July 10, 2022.
8. Centers for Disease Control and Prevention. Monitoring Selected National HIV Prevention and Care Objectives by Using HIV Surveillance Data–United States and 6 Dependent Areas, 2020. HIV Surveillance Supplemental Report. 2022;27(3).
As pediatricians, almost all of our clinic visits include some anticipatory guidance and recommendations on ways to promote well-being and prevent illness and injury for our patients. Because of minority stress, discrimination, and increased exposure to adverse childhood experiences, LGBTQ+ patients are disproportionately affected by certain health conditions including depression, anxiety, substance use, homelessness, as well as HIV and other sexually transmitted infections (STIs).1 While LGBTQ+ youth could benefit from additional guidance, counseling, and interventions related to these health disparities and have expressed interest in talking about these topics with their providers, sexual and gender minority youth also stress that they want to be treated as any other youth.2 Extending counseling for preventive care measures such as preexposure prophylaxis (PrEP) for HIV to all sexually active youth could help to destigmatize LGBTQ+ youth as being “different” from other youth and also help to increase overall access to HIV prevention services.3
Described by some as the “birth control” for HIV infection, PrEP is taken on an ongoing basis by those who are HIV negative before potential exposures to HIV in order to prevent new HIV infections. PrEP was first approved as a daily pill for adults in 2015 by the Food and Drug Administration with extension in 2018 to all individuals at risk for HIV weighing at least 35 kg after safety and efficacy data showed it could be used routinely for adolescents.4 When taken daily, oral PrEP can decrease the risk of HIV from sexual contact by more than 90% and from injection drug use by around 70%. As PrEP is highly effective with low risk for side effects, the U.S. Preventive Services Task Force (USPSTF) gave PrEP a “Grade A” recommendation for use in those at high risk for HIV infection in 2019.5 Since efficacy is closely tied to adherence, the first injectable PrEP (given at 0, 1, and 2 months with dosing then every 2 months) was also recently FDA approved in late 2021.6
Since HIV infection disproportionately affects LBGTQ+ individuals, and particularly LBGTQ+ youth of color, counseling related to PrEP has been largely targeted to these groups.7 Insurance and financial barriers to PrEP have been greatly reduced over the past several years through changes in insurance coverage (strengthened by the USPSTF recommendation), supplemental insurance programs, and pharmaceutical copay programs. Many states (but not all) also include HIV in the definition of STIs and allow minors to consent to PrEP services without a parent or guardian. Unfortunately, despite the high efficacy of PrEP and efforts to decrease barriers, rates of PrEP use continue to be extremely low, especially in youth, with only 15.6% of those aged 16-24 who are at risk for HIV in the United States actually taking PrEP in 2019.8 Many barriers to PrEP continue to exist including lack of awareness of PrEP, stigma surrounding HIV and PrEP, and lack of PrEP providers.
In order to address these low rates of PrEP uptake, the Centers for Disease Control and Prevention now recommends that medical providers discuss PrEP with all sexually active patients.6 PrEP should not be seen or discussed as something only relevant to LBGTQ+ populations, but rather as another tool in everyone’s “sexual health toolbox” that can allow us to experience human connection and pleasure through sexual activity while also having more control over what happens to our bodies. Not only will this allow more patients to access PrEP directly, it will also decrease the stigma of talking about HIV and PrEP and strengthen youths’ sense of autonomy and control over their own sexual health.
Since PrEP is a relatively new medical service, many providers will need to learn more about PrEP to at least have initial discussions with patients and to feel comfortable prescribing this themselves (See Resources). Below are also some suggestions to incorporate into your practice in order to advocate for the health and well-being of all your patients, including LGBTQ+ youth.
- Once your patients are 13 years and older, spend time with them alone to confidentially discuss more sensitive topics such as sexual health, mental health, and substance use.
- For all patients who are sexually active or considering sexual activity in the near future, discuss topics to help them control what happens to their bodies including consent, condoms, birth control, PrEP, and routine STI screening.
- Recommend PrEP to anyone who is sexually active and may be at increased risk for HIV infection or who is interested in taking PrEP for HIV prevention.
- Learn more about PrEP and start prescribing it to your own patients or become familiar with providers in your area to whom you could refer patients who are interested. While no certification is needed to prescribe PrEP, programs exist to help providers become more familiar with how to prescribe PrEP.
Dr. Warus is an adolescent medicine physician who specializes in care for transgender and gender-nonconforming youth, HIV prevention for adolescents and young adults, and LGBTQ health for youth at Children’s Hospital of Los Angeles. He is an assistant professor of clinical pediatrics and a University of Southern California faculty member.
Resources
CDC PrEP resources for clinicians: www.cdc.gov/hiv/clinicians/prevention/prep.html.Health HIV’s HIV Prevention Certified Provider Certification Program: https://healthhiv.org/programs/hpcp/.PrEP providers in the United States: https://preplocator.org/.Adolescent Health Working Group’s Sexual and Reproductive Health Toolkit for Adolescent Providers: https://ahwg.org/download/sexual-and-reproductive-health-toolkit-for-adolescent-providers/.
References
1. Lund EM and Burgess CM. Prim Care Clin Office Pract. 2021:48:179-89.
2. Hoffman ND et al. J Adolesc Health. 2009;45:222-9.
3. Mayer KH et al. Adv Ther. 2020;37:1778-811.
4. Hosek SG et al. JAMA Pediatr. 2017;171(11):1063-71.
5. U.S. Preventive Services Task Force; Owens DK et al. JAMA. 2019;321(22):2203-13.
6. Centers for Disease Control and Prevention: U.S. Public Health Service: Preexposure Prophylaxis for the Prevention of HIV Infection in the United States – 2021 Update: A Clinical Practice Guideline. Published 2021. Accessed July 10, 2022.
7. Centers for Disease Control and Prevention. Estimated HIV Incidence and Prevalence in the United States, 2015-2019. HIV Surveillance Supplemental Report. 2021;26(1). Published May 2021. Accessed July 10, 2022.
8. Centers for Disease Control and Prevention. Monitoring Selected National HIV Prevention and Care Objectives by Using HIV Surveillance Data–United States and 6 Dependent Areas, 2020. HIV Surveillance Supplemental Report. 2022;27(3).
As pediatricians, almost all of our clinic visits include some anticipatory guidance and recommendations on ways to promote well-being and prevent illness and injury for our patients. Because of minority stress, discrimination, and increased exposure to adverse childhood experiences, LGBTQ+ patients are disproportionately affected by certain health conditions including depression, anxiety, substance use, homelessness, as well as HIV and other sexually transmitted infections (STIs).1 While LGBTQ+ youth could benefit from additional guidance, counseling, and interventions related to these health disparities and have expressed interest in talking about these topics with their providers, sexual and gender minority youth also stress that they want to be treated as any other youth.2 Extending counseling for preventive care measures such as preexposure prophylaxis (PrEP) for HIV to all sexually active youth could help to destigmatize LGBTQ+ youth as being “different” from other youth and also help to increase overall access to HIV prevention services.3
Described by some as the “birth control” for HIV infection, PrEP is taken on an ongoing basis by those who are HIV negative before potential exposures to HIV in order to prevent new HIV infections. PrEP was first approved as a daily pill for adults in 2015 by the Food and Drug Administration with extension in 2018 to all individuals at risk for HIV weighing at least 35 kg after safety and efficacy data showed it could be used routinely for adolescents.4 When taken daily, oral PrEP can decrease the risk of HIV from sexual contact by more than 90% and from injection drug use by around 70%. As PrEP is highly effective with low risk for side effects, the U.S. Preventive Services Task Force (USPSTF) gave PrEP a “Grade A” recommendation for use in those at high risk for HIV infection in 2019.5 Since efficacy is closely tied to adherence, the first injectable PrEP (given at 0, 1, and 2 months with dosing then every 2 months) was also recently FDA approved in late 2021.6
Since HIV infection disproportionately affects LBGTQ+ individuals, and particularly LBGTQ+ youth of color, counseling related to PrEP has been largely targeted to these groups.7 Insurance and financial barriers to PrEP have been greatly reduced over the past several years through changes in insurance coverage (strengthened by the USPSTF recommendation), supplemental insurance programs, and pharmaceutical copay programs. Many states (but not all) also include HIV in the definition of STIs and allow minors to consent to PrEP services without a parent or guardian. Unfortunately, despite the high efficacy of PrEP and efforts to decrease barriers, rates of PrEP use continue to be extremely low, especially in youth, with only 15.6% of those aged 16-24 who are at risk for HIV in the United States actually taking PrEP in 2019.8 Many barriers to PrEP continue to exist including lack of awareness of PrEP, stigma surrounding HIV and PrEP, and lack of PrEP providers.
In order to address these low rates of PrEP uptake, the Centers for Disease Control and Prevention now recommends that medical providers discuss PrEP with all sexually active patients.6 PrEP should not be seen or discussed as something only relevant to LBGTQ+ populations, but rather as another tool in everyone’s “sexual health toolbox” that can allow us to experience human connection and pleasure through sexual activity while also having more control over what happens to our bodies. Not only will this allow more patients to access PrEP directly, it will also decrease the stigma of talking about HIV and PrEP and strengthen youths’ sense of autonomy and control over their own sexual health.
Since PrEP is a relatively new medical service, many providers will need to learn more about PrEP to at least have initial discussions with patients and to feel comfortable prescribing this themselves (See Resources). Below are also some suggestions to incorporate into your practice in order to advocate for the health and well-being of all your patients, including LGBTQ+ youth.
- Once your patients are 13 years and older, spend time with them alone to confidentially discuss more sensitive topics such as sexual health, mental health, and substance use.
- For all patients who are sexually active or considering sexual activity in the near future, discuss topics to help them control what happens to their bodies including consent, condoms, birth control, PrEP, and routine STI screening.
- Recommend PrEP to anyone who is sexually active and may be at increased risk for HIV infection or who is interested in taking PrEP for HIV prevention.
- Learn more about PrEP and start prescribing it to your own patients or become familiar with providers in your area to whom you could refer patients who are interested. While no certification is needed to prescribe PrEP, programs exist to help providers become more familiar with how to prescribe PrEP.
Dr. Warus is an adolescent medicine physician who specializes in care for transgender and gender-nonconforming youth, HIV prevention for adolescents and young adults, and LGBTQ health for youth at Children’s Hospital of Los Angeles. He is an assistant professor of clinical pediatrics and a University of Southern California faculty member.
Resources
CDC PrEP resources for clinicians: www.cdc.gov/hiv/clinicians/prevention/prep.html.Health HIV’s HIV Prevention Certified Provider Certification Program: https://healthhiv.org/programs/hpcp/.PrEP providers in the United States: https://preplocator.org/.Adolescent Health Working Group’s Sexual and Reproductive Health Toolkit for Adolescent Providers: https://ahwg.org/download/sexual-and-reproductive-health-toolkit-for-adolescent-providers/.
References
1. Lund EM and Burgess CM. Prim Care Clin Office Pract. 2021:48:179-89.
2. Hoffman ND et al. J Adolesc Health. 2009;45:222-9.
3. Mayer KH et al. Adv Ther. 2020;37:1778-811.
4. Hosek SG et al. JAMA Pediatr. 2017;171(11):1063-71.
5. U.S. Preventive Services Task Force; Owens DK et al. JAMA. 2019;321(22):2203-13.
6. Centers for Disease Control and Prevention: U.S. Public Health Service: Preexposure Prophylaxis for the Prevention of HIV Infection in the United States – 2021 Update: A Clinical Practice Guideline. Published 2021. Accessed July 10, 2022.
7. Centers for Disease Control and Prevention. Estimated HIV Incidence and Prevalence in the United States, 2015-2019. HIV Surveillance Supplemental Report. 2021;26(1). Published May 2021. Accessed July 10, 2022.
8. Centers for Disease Control and Prevention. Monitoring Selected National HIV Prevention and Care Objectives by Using HIV Surveillance Data–United States and 6 Dependent Areas, 2020. HIV Surveillance Supplemental Report. 2022;27(3).
Link between pediatric hepatitis and adenovirus 41 still unclear
While two new studies reiterate a possible relationship between adenovirus 41 and acute hepatitis of unknown cause in children, whether these infections are significant or merely bystanders remains unclear.
In both studies – one conducted in Alabama and the other conducted in the United Kingdom – researchers found that 90% of children with acute hepatitis of unknown cause tested positive for adenovirus 41. The virus subtype is not an uncommon infection, but it usually causes gastroenteritis in children.
“Across the world, adenovirus continues to be a common signal” in these pediatric hepatitis cases, said Helena Gutierrez, MD, the medical director of the Pediatric Liver Transplant Program at the University of Alabama at Birmingham, in an interview. She led one of the studies. More data are necessary to understand what role this virus may play in these cases, she said.
In November, the Alabama Department of Public Health and the U.S. Centers for Disease Control and Prevention began investigating a cluster of severe pediatric hepatitis cases at the Children’s of Alabama hospital in Birmingham. These children also tested positive for adenovirus. In April, the United Kingdom announced they were investigating similar cases, and the CDC expanded their search nationally. As of July 8, 1,010 cases in 35 countries have been reported to the World Health Organization. There are 263 confirmed cases in the United Kingdom and 332 cases under investigation by the CDC in the United States, according to the most recent counts.
The two studies, both published in the New England Journal of Medicine, provide additional clinical data on a number of these mysterious hepatitis cases. Dr. Gutierrez’s study looked at nine children admitted for hepatitis of unknown origin between October 1 and February 28. Patients had a median age of 2 years 11 months and two required liver transplants, and there were no deaths.
Eight out of nine patients (89%) tested positive for adenovirus, and all five of the samples that were of sufficient quality for gene sequencing tested positive for adenovirus 41. None of the six liver biopsies performed found signs of adenovirus infection, but the liver tissue samples of three patients tested positive for adenovirus via PCR.
The second study involved 44 children referred to a liver transplantation center in the United Kingdom between January 1 and April 11, 2022. The median age for patients was 4 years. Six children required liver transplants, and there were no deaths. Of the 30 patients who underwent molecular adenovirus testing, 27 (90%) were positive for adenovirus 41. Liver samples of nine children (3 from biopsies and 6 from explanted livers) all tested negative for adenovirus antibodies.
In both studies, however, the median adenovirus viral load of patients needing a transplant was much higher than the viral loads in children who did not require liver transplants.
Although most of the clinical features and test results of these cases suggest that adenovirus may be involved, the negative results in histology are “intriguing,” Chayarani Kelgeri, MD, a consultant pediatric hepatologist at the Birmingham Women’s and Children’s Hospital, U.K., said in an email. She is the lead author of the U.K. study. “Whether this is because the liver injury we see is an aftermath of the viral infection, the mechanism of injury is immune mediated, and if other cofactors are involved is being explored,” she added. “Further investigations being undertaken by UK Health Security Agency will add to our understanding of this illness.”
Although there is a high adenovirus positivity rate amongst these cases, there is not enough evidence yet to say adenovirus 41 is a new cause of pediatric hepatitis in previously healthy children, said Saul Karpen, MD, PhD, the division chief of pediatric gastroenterology, hepatology, and nutrition at Emory University School of Medicine, Atlanta. He wrote an editorial accompanying the two NEJM studies.
The CDC has not yet found an increase in pediatric hepatitis cases, according to a recent analysis, though the United Kingdom has found an uptick in cases this year, he told this news organization. Also, the cases highlighted in both articles showed no histological evidence of adenovirus in liver biopsies. “That’s completely opposite of what we generally see in adenoviral hepatitis that can be quite severe,” he said, adding that in general, there are detectable viral particles and antigens in affected livers.
“These two important reports indicate to those inside and outside the field of pediatric hepatology that registries and clinical studies of acute hepatitis in children are sorely needed,” Dr. Karpen writes in the editorial; “It is likely that with greater attention to collecting data on cases and biospecimens from children with acute hepatitis, we will be able to determine whether this one virus, human adenovirus 41, is of relevance to this important and serious condition in children.”
Dr. Gutierrez, Dr. Kelgeri, and Dr. Karpen report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
While two new studies reiterate a possible relationship between adenovirus 41 and acute hepatitis of unknown cause in children, whether these infections are significant or merely bystanders remains unclear.
In both studies – one conducted in Alabama and the other conducted in the United Kingdom – researchers found that 90% of children with acute hepatitis of unknown cause tested positive for adenovirus 41. The virus subtype is not an uncommon infection, but it usually causes gastroenteritis in children.
“Across the world, adenovirus continues to be a common signal” in these pediatric hepatitis cases, said Helena Gutierrez, MD, the medical director of the Pediatric Liver Transplant Program at the University of Alabama at Birmingham, in an interview. She led one of the studies. More data are necessary to understand what role this virus may play in these cases, she said.
In November, the Alabama Department of Public Health and the U.S. Centers for Disease Control and Prevention began investigating a cluster of severe pediatric hepatitis cases at the Children’s of Alabama hospital in Birmingham. These children also tested positive for adenovirus. In April, the United Kingdom announced they were investigating similar cases, and the CDC expanded their search nationally. As of July 8, 1,010 cases in 35 countries have been reported to the World Health Organization. There are 263 confirmed cases in the United Kingdom and 332 cases under investigation by the CDC in the United States, according to the most recent counts.
The two studies, both published in the New England Journal of Medicine, provide additional clinical data on a number of these mysterious hepatitis cases. Dr. Gutierrez’s study looked at nine children admitted for hepatitis of unknown origin between October 1 and February 28. Patients had a median age of 2 years 11 months and two required liver transplants, and there were no deaths.
Eight out of nine patients (89%) tested positive for adenovirus, and all five of the samples that were of sufficient quality for gene sequencing tested positive for adenovirus 41. None of the six liver biopsies performed found signs of adenovirus infection, but the liver tissue samples of three patients tested positive for adenovirus via PCR.
The second study involved 44 children referred to a liver transplantation center in the United Kingdom between January 1 and April 11, 2022. The median age for patients was 4 years. Six children required liver transplants, and there were no deaths. Of the 30 patients who underwent molecular adenovirus testing, 27 (90%) were positive for adenovirus 41. Liver samples of nine children (3 from biopsies and 6 from explanted livers) all tested negative for adenovirus antibodies.
In both studies, however, the median adenovirus viral load of patients needing a transplant was much higher than the viral loads in children who did not require liver transplants.
Although most of the clinical features and test results of these cases suggest that adenovirus may be involved, the negative results in histology are “intriguing,” Chayarani Kelgeri, MD, a consultant pediatric hepatologist at the Birmingham Women’s and Children’s Hospital, U.K., said in an email. She is the lead author of the U.K. study. “Whether this is because the liver injury we see is an aftermath of the viral infection, the mechanism of injury is immune mediated, and if other cofactors are involved is being explored,” she added. “Further investigations being undertaken by UK Health Security Agency will add to our understanding of this illness.”
Although there is a high adenovirus positivity rate amongst these cases, there is not enough evidence yet to say adenovirus 41 is a new cause of pediatric hepatitis in previously healthy children, said Saul Karpen, MD, PhD, the division chief of pediatric gastroenterology, hepatology, and nutrition at Emory University School of Medicine, Atlanta. He wrote an editorial accompanying the two NEJM studies.
The CDC has not yet found an increase in pediatric hepatitis cases, according to a recent analysis, though the United Kingdom has found an uptick in cases this year, he told this news organization. Also, the cases highlighted in both articles showed no histological evidence of adenovirus in liver biopsies. “That’s completely opposite of what we generally see in adenoviral hepatitis that can be quite severe,” he said, adding that in general, there are detectable viral particles and antigens in affected livers.
“These two important reports indicate to those inside and outside the field of pediatric hepatology that registries and clinical studies of acute hepatitis in children are sorely needed,” Dr. Karpen writes in the editorial; “It is likely that with greater attention to collecting data on cases and biospecimens from children with acute hepatitis, we will be able to determine whether this one virus, human adenovirus 41, is of relevance to this important and serious condition in children.”
Dr. Gutierrez, Dr. Kelgeri, and Dr. Karpen report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
While two new studies reiterate a possible relationship between adenovirus 41 and acute hepatitis of unknown cause in children, whether these infections are significant or merely bystanders remains unclear.
In both studies – one conducted in Alabama and the other conducted in the United Kingdom – researchers found that 90% of children with acute hepatitis of unknown cause tested positive for adenovirus 41. The virus subtype is not an uncommon infection, but it usually causes gastroenteritis in children.
“Across the world, adenovirus continues to be a common signal” in these pediatric hepatitis cases, said Helena Gutierrez, MD, the medical director of the Pediatric Liver Transplant Program at the University of Alabama at Birmingham, in an interview. She led one of the studies. More data are necessary to understand what role this virus may play in these cases, she said.
In November, the Alabama Department of Public Health and the U.S. Centers for Disease Control and Prevention began investigating a cluster of severe pediatric hepatitis cases at the Children’s of Alabama hospital in Birmingham. These children also tested positive for adenovirus. In April, the United Kingdom announced they were investigating similar cases, and the CDC expanded their search nationally. As of July 8, 1,010 cases in 35 countries have been reported to the World Health Organization. There are 263 confirmed cases in the United Kingdom and 332 cases under investigation by the CDC in the United States, according to the most recent counts.
The two studies, both published in the New England Journal of Medicine, provide additional clinical data on a number of these mysterious hepatitis cases. Dr. Gutierrez’s study looked at nine children admitted for hepatitis of unknown origin between October 1 and February 28. Patients had a median age of 2 years 11 months and two required liver transplants, and there were no deaths.
Eight out of nine patients (89%) tested positive for adenovirus, and all five of the samples that were of sufficient quality for gene sequencing tested positive for adenovirus 41. None of the six liver biopsies performed found signs of adenovirus infection, but the liver tissue samples of three patients tested positive for adenovirus via PCR.
The second study involved 44 children referred to a liver transplantation center in the United Kingdom between January 1 and April 11, 2022. The median age for patients was 4 years. Six children required liver transplants, and there were no deaths. Of the 30 patients who underwent molecular adenovirus testing, 27 (90%) were positive for adenovirus 41. Liver samples of nine children (3 from biopsies and 6 from explanted livers) all tested negative for adenovirus antibodies.
In both studies, however, the median adenovirus viral load of patients needing a transplant was much higher than the viral loads in children who did not require liver transplants.
Although most of the clinical features and test results of these cases suggest that adenovirus may be involved, the negative results in histology are “intriguing,” Chayarani Kelgeri, MD, a consultant pediatric hepatologist at the Birmingham Women’s and Children’s Hospital, U.K., said in an email. She is the lead author of the U.K. study. “Whether this is because the liver injury we see is an aftermath of the viral infection, the mechanism of injury is immune mediated, and if other cofactors are involved is being explored,” she added. “Further investigations being undertaken by UK Health Security Agency will add to our understanding of this illness.”
Although there is a high adenovirus positivity rate amongst these cases, there is not enough evidence yet to say adenovirus 41 is a new cause of pediatric hepatitis in previously healthy children, said Saul Karpen, MD, PhD, the division chief of pediatric gastroenterology, hepatology, and nutrition at Emory University School of Medicine, Atlanta. He wrote an editorial accompanying the two NEJM studies.
The CDC has not yet found an increase in pediatric hepatitis cases, according to a recent analysis, though the United Kingdom has found an uptick in cases this year, he told this news organization. Also, the cases highlighted in both articles showed no histological evidence of adenovirus in liver biopsies. “That’s completely opposite of what we generally see in adenoviral hepatitis that can be quite severe,” he said, adding that in general, there are detectable viral particles and antigens in affected livers.
“These two important reports indicate to those inside and outside the field of pediatric hepatology that registries and clinical studies of acute hepatitis in children are sorely needed,” Dr. Karpen writes in the editorial; “It is likely that with greater attention to collecting data on cases and biospecimens from children with acute hepatitis, we will be able to determine whether this one virus, human adenovirus 41, is of relevance to this important and serious condition in children.”
Dr. Gutierrez, Dr. Kelgeri, and Dr. Karpen report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Coming soon: More breathable, more comfortable face masks
Sitting at his desk in Sea Girt, N.J., John Schwind is eager to demonstrate his ReadiMask 365. He holds up what looks like a white sheet of memo paper, peels off a protective liner, and sticks the mask first to his nose. He glides his fingers down his face, over his cheeks, and to his chin, sealing the mask and then demonstrating how easy it is to talk with it in place.
The mask’s medical adhesive sticks directly to the face, without causing breakouts, he said. It doesn’t let air leak and won’t fog his glasses. It’s strapless, so it won’t hurt his ears or make them stick out.
This fall, Mr. Schwind, the CEO of Global Safety First, is hoping to take home $150,000 as one of the two top winners of the federal Mask Innovation Challenge. He has made it to the top 10 but realizes he still has a ton of competition.
After the challenge launched in late 2021, nearly 1,500 submissions were received, says Kumiko Lippold, PhD, a health scientist and manager of the Mask Innovation Challenge. The challenge is run by Dr. Lippold and others at the Division of Research, Innovation, and Ventures (DRIVe), which is part of the Biomedical Advanced Research and Development Authority (BARDA) at the U.S. Department of Health & Human Services.
Like the rest of us, Dr. Lippold knows that masks desperately need a makeover. The aim is not only to get us through this pandemic but also future pandemics and other public health emergencies. “We are focused on building masks for the next pandemic, the next wildfires,” she says.
The project is a partnership among BARDA’s DRIVe, the National Institute for Occupational Safety and Health (NIOSH), and the National Institute of Standards and Technology (NIST).
While NIOSH is a partner in the challenge, giving feedback to mask developers, “the mask challenge is entirely separate from the NIOSH approval process,” Dr. Lippold says. Companies can then pursue NIOSH approval on their own, later, if they wish. The agency certifies only masks and respirators.
Preview of masks to come
“We’ve seen some really amazing things,” Dr. Lippold said of the new designs. She didn’t want to play favorites, so she gave an overview of innovations. Some designs have transparent materials, or partially see-through materials, so facial expressions can be read. “We’ve also seen really unique bio-based materials that are derived from natural products. We’ve seen sensors in some.”
One mask model has origami folds, which increase overall surface and breathing area. Some 3D-printed masks promise a custom fit and take into account whether a person’s nose bridge is low or high.
And the finalists are ...
ReadiMask 365: “I can wear this all day long,” Mr. Schwind said of his new design. It has a nano fiber filter and is flexible. Besides the one in the BARDA challenge, the company has other ReadiMasks on the market. “The most important thing is comfort,” he says. “Second is protection. If they don’t feel they have a good seal, users don’t have confidence in the mask.”
He offers various sizes of ReadiMasks, from small sizes designed for women with smaller faces to extra-large, “for NFL linemen.”
ClearMask: “We are the original clear mask,” says Aaron Hsu, CEO and co-founder of ClearMask in Baltimore. The company began in 2017, and the clear design was inspired by a company co-founder who is deaf. She was scheduled to have surgery, and her sign language interpreter did not show up, leaving her to try to communicate in the operating room with masked health care providers. There were no transparent masks available then, Mr. Hsu says.
“Being able to work with BARDA and getting their wisdom is invaluable,” he says.
The makers of ClearMask think masks are here to stay, at least for some. “I think a certain percentage of the population will continue to wear them, regardless,” said Mr. Hsu. He predicts health care settings will become stricter about wearing masks.
“Even now, when you even walk in to a hospital, you might be required to wear a mask,” he says, even as a visitor. His company’s masks are easy to adjust and are secured around the head, so your ears don’t get sore, he says.
4C Air: The BreSafe transparent mask is semi-transparent and is made of a nanomaterial that provides high levels of filtration and breathability with some transparency.
Air99: Based on origami principles, the Airgami mask is meant to improve fit, breathability, and aesthetics over existing masks. “Airgami fits better, works better and looks better,” says Min Xiao, a company spokesperson. “It won’t fall off the nose or collapse onto the mouth, and eyeglasses fog less, she says. Voices are less muffled.” It’s also reusable, rinseable and can be heat disinfected, she says. It went on the market in November 2020.
Air Flo Labs: Flo Mask Pro, like the company’s other designs, conducted over 100 3D facial scans across many ethnicities to produce a better fit, says Kevin Ngo, its creator. For the adult masks, two nose bridge sizes are offered. And users can choose a Pro Filter, with 99% filtration, or an Everyday, which is meant to be much more breathable than other masks. “Our silicone gasket is incredibly soft and gentle on the skin,” Mr. Ngo says. “In addition,we offer indents for glasses, which prevent any fogging.” The company began shipping in May; several thousand masks are in use now, Mr. Ngo said.
Georgetown University: This team’s smart mask is made of metallic foams that can be cleaned and reused.
Levi Strauss: The form of the mask can be made by any basic garment factory. It aims to activate the apparel supply chain as another source of low-cost, high-performance masks.
Matregenix: This mask, made of a transparent nanofiber, allows for easier communication while having high filtration.
SEAL Lab: The SINEW mask stands for Smart, Individualized, Near-Face, Extended Wear. The mask used technology to overcome flaws of traditional respirators, with the same degree of protection. It doesn’t make contact with the skin of the wearer’s face.
StaySafeNow: A team from Harvard University developed Crystal Guard, a reusable, cost-effective clear mask. Its developers say it’s meant to be especially useful for essential workers, teachers, and others who have to communicate to do their work.
Bye-bye N95?
“From our perspective, our goal with the mask challenge was not to replace the N95 respirator,” Dr. Lippold says. N95 masks, which NIOSH certifies, are valuable and protect people in high-risk settings. “With the mask challenge, our goal was really to provide the public with a comparable alternative that really meets their specific level of risk.” Working in a health care setting carries a different risk, she says, than going to the grocery store.
“A common complaint with the N95 is that they are very uncomfortable.” It’s a major barrier to compliance, “and we wanted to address that gap. We didn’t directly compare [the entries] to an N95,” she says, although their testing was similar to NIOSH’s. A number of finalists say they will pursue NIOSH approval, she says.
Meanwhile, some of the finalists’ masks are for sale. Air Flo Labs, for instance, has its Flo Mask Pro for sale online, noting that BARDA allowed it to release the test results from NIOSH and NIST.
Getting from 1,500 to 10
In the first phase of the challenge, Dr. Lippold says, “the goal was to engage as wide an audience as possible.” With the second phase, the bar was set a bit higher. Instead of just submitting ideas on paper, companies had to submit prototypes for lab testing. “We got about 80 submissions,” she says.
Those 80 were whittled down to 10 finalists. Teams had sent prototypes, and experts, including those from NIOSH and NIST, rated them, sometimes looking at multiple copies of the masks. Experts looked at how well the masks filtered the air, how breathable they were, and other data. Once the feedback was given to the mask companies, they entered a redesign period. “Scientists can take this data and basically make these prototypes better,” Dr. Lippold says.
The final round of testing will be in September, and the winners will be announced in the fall. The opportunity allowed companies to have their products go through testing they might not otherwise have been able to get, she says.
A version of this article first appeared on WebMD.com.
Sitting at his desk in Sea Girt, N.J., John Schwind is eager to demonstrate his ReadiMask 365. He holds up what looks like a white sheet of memo paper, peels off a protective liner, and sticks the mask first to his nose. He glides his fingers down his face, over his cheeks, and to his chin, sealing the mask and then demonstrating how easy it is to talk with it in place.
The mask’s medical adhesive sticks directly to the face, without causing breakouts, he said. It doesn’t let air leak and won’t fog his glasses. It’s strapless, so it won’t hurt his ears or make them stick out.
This fall, Mr. Schwind, the CEO of Global Safety First, is hoping to take home $150,000 as one of the two top winners of the federal Mask Innovation Challenge. He has made it to the top 10 but realizes he still has a ton of competition.
After the challenge launched in late 2021, nearly 1,500 submissions were received, says Kumiko Lippold, PhD, a health scientist and manager of the Mask Innovation Challenge. The challenge is run by Dr. Lippold and others at the Division of Research, Innovation, and Ventures (DRIVe), which is part of the Biomedical Advanced Research and Development Authority (BARDA) at the U.S. Department of Health & Human Services.
Like the rest of us, Dr. Lippold knows that masks desperately need a makeover. The aim is not only to get us through this pandemic but also future pandemics and other public health emergencies. “We are focused on building masks for the next pandemic, the next wildfires,” she says.
The project is a partnership among BARDA’s DRIVe, the National Institute for Occupational Safety and Health (NIOSH), and the National Institute of Standards and Technology (NIST).
While NIOSH is a partner in the challenge, giving feedback to mask developers, “the mask challenge is entirely separate from the NIOSH approval process,” Dr. Lippold says. Companies can then pursue NIOSH approval on their own, later, if they wish. The agency certifies only masks and respirators.
Preview of masks to come
“We’ve seen some really amazing things,” Dr. Lippold said of the new designs. She didn’t want to play favorites, so she gave an overview of innovations. Some designs have transparent materials, or partially see-through materials, so facial expressions can be read. “We’ve also seen really unique bio-based materials that are derived from natural products. We’ve seen sensors in some.”
One mask model has origami folds, which increase overall surface and breathing area. Some 3D-printed masks promise a custom fit and take into account whether a person’s nose bridge is low or high.
And the finalists are ...
ReadiMask 365: “I can wear this all day long,” Mr. Schwind said of his new design. It has a nano fiber filter and is flexible. Besides the one in the BARDA challenge, the company has other ReadiMasks on the market. “The most important thing is comfort,” he says. “Second is protection. If they don’t feel they have a good seal, users don’t have confidence in the mask.”
He offers various sizes of ReadiMasks, from small sizes designed for women with smaller faces to extra-large, “for NFL linemen.”
ClearMask: “We are the original clear mask,” says Aaron Hsu, CEO and co-founder of ClearMask in Baltimore. The company began in 2017, and the clear design was inspired by a company co-founder who is deaf. She was scheduled to have surgery, and her sign language interpreter did not show up, leaving her to try to communicate in the operating room with masked health care providers. There were no transparent masks available then, Mr. Hsu says.
“Being able to work with BARDA and getting their wisdom is invaluable,” he says.
The makers of ClearMask think masks are here to stay, at least for some. “I think a certain percentage of the population will continue to wear them, regardless,” said Mr. Hsu. He predicts health care settings will become stricter about wearing masks.
“Even now, when you even walk in to a hospital, you might be required to wear a mask,” he says, even as a visitor. His company’s masks are easy to adjust and are secured around the head, so your ears don’t get sore, he says.
4C Air: The BreSafe transparent mask is semi-transparent and is made of a nanomaterial that provides high levels of filtration and breathability with some transparency.
Air99: Based on origami principles, the Airgami mask is meant to improve fit, breathability, and aesthetics over existing masks. “Airgami fits better, works better and looks better,” says Min Xiao, a company spokesperson. “It won’t fall off the nose or collapse onto the mouth, and eyeglasses fog less, she says. Voices are less muffled.” It’s also reusable, rinseable and can be heat disinfected, she says. It went on the market in November 2020.
Air Flo Labs: Flo Mask Pro, like the company’s other designs, conducted over 100 3D facial scans across many ethnicities to produce a better fit, says Kevin Ngo, its creator. For the adult masks, two nose bridge sizes are offered. And users can choose a Pro Filter, with 99% filtration, or an Everyday, which is meant to be much more breathable than other masks. “Our silicone gasket is incredibly soft and gentle on the skin,” Mr. Ngo says. “In addition,we offer indents for glasses, which prevent any fogging.” The company began shipping in May; several thousand masks are in use now, Mr. Ngo said.
Georgetown University: This team’s smart mask is made of metallic foams that can be cleaned and reused.
Levi Strauss: The form of the mask can be made by any basic garment factory. It aims to activate the apparel supply chain as another source of low-cost, high-performance masks.
Matregenix: This mask, made of a transparent nanofiber, allows for easier communication while having high filtration.
SEAL Lab: The SINEW mask stands for Smart, Individualized, Near-Face, Extended Wear. The mask used technology to overcome flaws of traditional respirators, with the same degree of protection. It doesn’t make contact with the skin of the wearer’s face.
StaySafeNow: A team from Harvard University developed Crystal Guard, a reusable, cost-effective clear mask. Its developers say it’s meant to be especially useful for essential workers, teachers, and others who have to communicate to do their work.
Bye-bye N95?
“From our perspective, our goal with the mask challenge was not to replace the N95 respirator,” Dr. Lippold says. N95 masks, which NIOSH certifies, are valuable and protect people in high-risk settings. “With the mask challenge, our goal was really to provide the public with a comparable alternative that really meets their specific level of risk.” Working in a health care setting carries a different risk, she says, than going to the grocery store.
“A common complaint with the N95 is that they are very uncomfortable.” It’s a major barrier to compliance, “and we wanted to address that gap. We didn’t directly compare [the entries] to an N95,” she says, although their testing was similar to NIOSH’s. A number of finalists say they will pursue NIOSH approval, she says.
Meanwhile, some of the finalists’ masks are for sale. Air Flo Labs, for instance, has its Flo Mask Pro for sale online, noting that BARDA allowed it to release the test results from NIOSH and NIST.
Getting from 1,500 to 10
In the first phase of the challenge, Dr. Lippold says, “the goal was to engage as wide an audience as possible.” With the second phase, the bar was set a bit higher. Instead of just submitting ideas on paper, companies had to submit prototypes for lab testing. “We got about 80 submissions,” she says.
Those 80 were whittled down to 10 finalists. Teams had sent prototypes, and experts, including those from NIOSH and NIST, rated them, sometimes looking at multiple copies of the masks. Experts looked at how well the masks filtered the air, how breathable they were, and other data. Once the feedback was given to the mask companies, they entered a redesign period. “Scientists can take this data and basically make these prototypes better,” Dr. Lippold says.
The final round of testing will be in September, and the winners will be announced in the fall. The opportunity allowed companies to have their products go through testing they might not otherwise have been able to get, she says.
A version of this article first appeared on WebMD.com.
Sitting at his desk in Sea Girt, N.J., John Schwind is eager to demonstrate his ReadiMask 365. He holds up what looks like a white sheet of memo paper, peels off a protective liner, and sticks the mask first to his nose. He glides his fingers down his face, over his cheeks, and to his chin, sealing the mask and then demonstrating how easy it is to talk with it in place.
The mask’s medical adhesive sticks directly to the face, without causing breakouts, he said. It doesn’t let air leak and won’t fog his glasses. It’s strapless, so it won’t hurt his ears or make them stick out.
This fall, Mr. Schwind, the CEO of Global Safety First, is hoping to take home $150,000 as one of the two top winners of the federal Mask Innovation Challenge. He has made it to the top 10 but realizes he still has a ton of competition.
After the challenge launched in late 2021, nearly 1,500 submissions were received, says Kumiko Lippold, PhD, a health scientist and manager of the Mask Innovation Challenge. The challenge is run by Dr. Lippold and others at the Division of Research, Innovation, and Ventures (DRIVe), which is part of the Biomedical Advanced Research and Development Authority (BARDA) at the U.S. Department of Health & Human Services.
Like the rest of us, Dr. Lippold knows that masks desperately need a makeover. The aim is not only to get us through this pandemic but also future pandemics and other public health emergencies. “We are focused on building masks for the next pandemic, the next wildfires,” she says.
The project is a partnership among BARDA’s DRIVe, the National Institute for Occupational Safety and Health (NIOSH), and the National Institute of Standards and Technology (NIST).
While NIOSH is a partner in the challenge, giving feedback to mask developers, “the mask challenge is entirely separate from the NIOSH approval process,” Dr. Lippold says. Companies can then pursue NIOSH approval on their own, later, if they wish. The agency certifies only masks and respirators.
Preview of masks to come
“We’ve seen some really amazing things,” Dr. Lippold said of the new designs. She didn’t want to play favorites, so she gave an overview of innovations. Some designs have transparent materials, or partially see-through materials, so facial expressions can be read. “We’ve also seen really unique bio-based materials that are derived from natural products. We’ve seen sensors in some.”
One mask model has origami folds, which increase overall surface and breathing area. Some 3D-printed masks promise a custom fit and take into account whether a person’s nose bridge is low or high.
And the finalists are ...
ReadiMask 365: “I can wear this all day long,” Mr. Schwind said of his new design. It has a nano fiber filter and is flexible. Besides the one in the BARDA challenge, the company has other ReadiMasks on the market. “The most important thing is comfort,” he says. “Second is protection. If they don’t feel they have a good seal, users don’t have confidence in the mask.”
He offers various sizes of ReadiMasks, from small sizes designed for women with smaller faces to extra-large, “for NFL linemen.”
ClearMask: “We are the original clear mask,” says Aaron Hsu, CEO and co-founder of ClearMask in Baltimore. The company began in 2017, and the clear design was inspired by a company co-founder who is deaf. She was scheduled to have surgery, and her sign language interpreter did not show up, leaving her to try to communicate in the operating room with masked health care providers. There were no transparent masks available then, Mr. Hsu says.
“Being able to work with BARDA and getting their wisdom is invaluable,” he says.
The makers of ClearMask think masks are here to stay, at least for some. “I think a certain percentage of the population will continue to wear them, regardless,” said Mr. Hsu. He predicts health care settings will become stricter about wearing masks.
“Even now, when you even walk in to a hospital, you might be required to wear a mask,” he says, even as a visitor. His company’s masks are easy to adjust and are secured around the head, so your ears don’t get sore, he says.
4C Air: The BreSafe transparent mask is semi-transparent and is made of a nanomaterial that provides high levels of filtration and breathability with some transparency.
Air99: Based on origami principles, the Airgami mask is meant to improve fit, breathability, and aesthetics over existing masks. “Airgami fits better, works better and looks better,” says Min Xiao, a company spokesperson. “It won’t fall off the nose or collapse onto the mouth, and eyeglasses fog less, she says. Voices are less muffled.” It’s also reusable, rinseable and can be heat disinfected, she says. It went on the market in November 2020.
Air Flo Labs: Flo Mask Pro, like the company’s other designs, conducted over 100 3D facial scans across many ethnicities to produce a better fit, says Kevin Ngo, its creator. For the adult masks, two nose bridge sizes are offered. And users can choose a Pro Filter, with 99% filtration, or an Everyday, which is meant to be much more breathable than other masks. “Our silicone gasket is incredibly soft and gentle on the skin,” Mr. Ngo says. “In addition,we offer indents for glasses, which prevent any fogging.” The company began shipping in May; several thousand masks are in use now, Mr. Ngo said.
Georgetown University: This team’s smart mask is made of metallic foams that can be cleaned and reused.
Levi Strauss: The form of the mask can be made by any basic garment factory. It aims to activate the apparel supply chain as another source of low-cost, high-performance masks.
Matregenix: This mask, made of a transparent nanofiber, allows for easier communication while having high filtration.
SEAL Lab: The SINEW mask stands for Smart, Individualized, Near-Face, Extended Wear. The mask used technology to overcome flaws of traditional respirators, with the same degree of protection. It doesn’t make contact with the skin of the wearer’s face.
StaySafeNow: A team from Harvard University developed Crystal Guard, a reusable, cost-effective clear mask. Its developers say it’s meant to be especially useful for essential workers, teachers, and others who have to communicate to do their work.
Bye-bye N95?
“From our perspective, our goal with the mask challenge was not to replace the N95 respirator,” Dr. Lippold says. N95 masks, which NIOSH certifies, are valuable and protect people in high-risk settings. “With the mask challenge, our goal was really to provide the public with a comparable alternative that really meets their specific level of risk.” Working in a health care setting carries a different risk, she says, than going to the grocery store.
“A common complaint with the N95 is that they are very uncomfortable.” It’s a major barrier to compliance, “and we wanted to address that gap. We didn’t directly compare [the entries] to an N95,” she says, although their testing was similar to NIOSH’s. A number of finalists say they will pursue NIOSH approval, she says.
Meanwhile, some of the finalists’ masks are for sale. Air Flo Labs, for instance, has its Flo Mask Pro for sale online, noting that BARDA allowed it to release the test results from NIOSH and NIST.
Getting from 1,500 to 10
In the first phase of the challenge, Dr. Lippold says, “the goal was to engage as wide an audience as possible.” With the second phase, the bar was set a bit higher. Instead of just submitting ideas on paper, companies had to submit prototypes for lab testing. “We got about 80 submissions,” she says.
Those 80 were whittled down to 10 finalists. Teams had sent prototypes, and experts, including those from NIOSH and NIST, rated them, sometimes looking at multiple copies of the masks. Experts looked at how well the masks filtered the air, how breathable they were, and other data. Once the feedback was given to the mask companies, they entered a redesign period. “Scientists can take this data and basically make these prototypes better,” Dr. Lippold says.
The final round of testing will be in September, and the winners will be announced in the fall. The opportunity allowed companies to have their products go through testing they might not otherwise have been able to get, she says.
A version of this article first appeared on WebMD.com.
Hospital-acquired pneumonia is killing patients, yet there is a simple way to stop it
Four years ago, when Dr. Karen Giuliano went to a Boston hospital for hip replacement surgery, she was given a pale-pink bucket of toiletries issued to patients in many hospitals. Inside were tissues, bar soap, deodorant, toothpaste, and, without a doubt, the worst toothbrush she’d ever seen.
“I couldn’t believe it. I got a toothbrush with no bristles,” she said. “It must have not gone through the bristle machine. It was just a stick.”
To most patients, a useless hospital toothbrush would be a mild inconvenience. But to Dr. Giuliano, a nursing professor at the University of Massachusetts, Amherst, it was a reminder of a pervasive “blind spot” in U.S. hospitals: the stunning consequences of unbrushed teeth.
Hospital patients not getting their teeth brushed, or not brushing their teeth themselves, is believed to be a leading cause of hundreds of thousands of cases of pneumonia a year in patients who have not been put on a ventilator. Pneumonia is among the most common infections that occur in health care facilities, and a majority of cases are nonventilator hospital-acquired pneumonia, or NVHAP, which kills up to 30% of those infected, Dr. Giuliano and other experts said.
But unlike many infections that strike within hospitals, the federal government doesn’t require hospitals to report cases of NVHAP. As a result, few hospitals understand the origin of the illness, track its occurrence, or actively work to prevent it, the experts said.
, according to a growing body of peer-reviewed research papers. Instead, many hospitals often skip teeth brushing to prioritize other tasks and provide only cheap, ineffective toothbrushes, often unaware of the consequences, said Dr. Dian Baker, a Sacramento (Calif.) State nursing professor who has spent more than a decade studying NVHAP.
“I’ll tell you that today the vast majority of the tens of thousands of nurses in hospitals have no idea that pneumonia comes from germs in the mouth,” Dr. Baker said.
Pneumonia occurs when germs trigger an infection in the lungs. Although NVHAP accounts for most of the cases that occur in hospitals, it historically has not received the same attention as pneumonia tied to ventilators, which is easier to identify and study because it occurs among a narrow subset of patients.
NVHAP, a risk for virtually all hospital patients, is often caused by bacteria from the mouth that gathers in the scummy biofilm on unbrushed teeth and is aspirated into the lungs. Patients face a higher risk if they lie flat or remain immobile for long periods, so NVHAP can also be prevented by elevating their heads and getting them out of bed more often.
According to the National Organization for NV-HAP Prevention, which was founded in 2020, this pneumonia infects about 1 in every 100 hospital patients and kills 15%-30% of them. For those who survive, the illness often extends their hospital stay by up to 15 days and makes it much more likely they will be readmitted within a month or transferred to an intensive care unit.
John McCleary, 83, of Millinocket, Maine, contracted a likely case of NVHAP in 2008 after he fractured his ankle in a fall and spent 12 days in rehabilitation at a hospital, said his daughter, Kathy Day, a retired nurse and advocate with the Patient Safety Action Network.
Mr. McCleary recovered from the fracture but not from pneumonia. Two days after he returned home, the infection in his lungs caused him to be rushed back to the hospital, where he went into sepsis and spent weeks in treatment before moving to an isolation unit in a nursing home.
He died weeks later, emaciated, largely deaf, unable to eat, and often “too weak to get water through a straw,” his daughter said. After contracting pneumonia, he never walked again.
“It was an astounding assault on his body, from him being here visiting me the week before his fall, to his death just a few months later,” Ms. Day said. “And the whole thing was avoidable.”
While experts describe NVHAP as a largely ignored threat, that appears to be changing.
Last year, a group of researchers – including Dr. Giuliano and Dr. Baker, plus officials from the Centers for Disease Control and Prevention, the Veterans Health Administration, and the Joint Commission – published a “call-to-action” research paper hoping to launch “a national health care conversation about NVHAP prevention.”
The Joint Commission, a nonprofit organization whose accreditation can make or break hospitals, is considering broadening the infection control standards to include more ailments, including NVHAP, said Sylvia Garcia-Houchins, its director of infection prevention and control.
Separately, ECRI, a nonprofit focused on health care safety, this year pinpointed NVHAP as one of its top patient safety concerns.
James Davis, an ECRI infection expert, said the prevalence of NVHAP, while already alarming, is likely “underestimated” and probably worsened as hospitals swelled with patients during the coronavirus pandemic.
“We only know what’s reported,” Mr. Davis said. “Could this be the tip of the iceberg? I would say, in my opinion, probably.”
To better measure the condition, some researchers call for a standardized surveillance definition of NVHAP, which could in time open the door for the federal government to mandate reporting of cases or incentivize prevention. With increasing urgency, researchers are pushing for hospitals not to wait for the federal government to act against NVHAP.
Dr. Baker said she has spoken with hundreds of hospitals about how to prevent NVHAP, but thousands more have yet to take up the cause.
“We are not asking for some big, $300,000 piece of equipment,” Dr. Baker said. “The two things that show the best evidence of preventing this harm are things that should be happening in standard care anyway – brushing teeth and getting patients mobilized.”
That evidence comes from a smattering of studies that show those two strategies can lead to sharp reductions in infection rates.
In California, a study at 21 Kaiser Permanente hospitals used a reprioritization of oral care and getting patients out of bed to reduce rates of hospital-acquired pneumonia by around 70%. At Sutter Medical Center in Sacramento, better oral care reduced NVHAP cases by a yearly average of 35%.
At Orlando Regional Medical Center in Florida, a medical unit and a surgical unit where patients received enhanced oral care reduced NVHAP rates by 85% and 56%, respectively, when compared with similar units that received normal care. A similar study is underway at two hospitals in Illinois.
And the most compelling results come from a veterans’ hospital in Salem, Va., where a 2016 oral care pilot program reduced rates of NVHAP by 92% – saving an estimated 13 lives in just 19 months. The program, the HAPPEN Initiative, has been expanded across the Veterans Health Administration, and experts say it could serve as a model for all U.S. hospitals.
Dr. Michelle Lucatorto, a nursing official who leads HAPPEN, said the program trains nurses to most effectively brush patients’ teeth and educates patients and families on the link between oral care and preventing NVHAP. While teeth brushing may not seem to require training, Dr. Lucatorto made comparisons to how the coronavirus revealed many Americans were doing a lackluster job of another routine hygienic practice: washing their hands.
“Sometimes we are searching for the most complicated intervention,” she said. “We are always looking for that new bypass surgery, or some new technical equipment. And sometimes I think we fail to look at the simple things we can do in our practice to save people’s lives.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Four years ago, when Dr. Karen Giuliano went to a Boston hospital for hip replacement surgery, she was given a pale-pink bucket of toiletries issued to patients in many hospitals. Inside were tissues, bar soap, deodorant, toothpaste, and, without a doubt, the worst toothbrush she’d ever seen.
“I couldn’t believe it. I got a toothbrush with no bristles,” she said. “It must have not gone through the bristle machine. It was just a stick.”
To most patients, a useless hospital toothbrush would be a mild inconvenience. But to Dr. Giuliano, a nursing professor at the University of Massachusetts, Amherst, it was a reminder of a pervasive “blind spot” in U.S. hospitals: the stunning consequences of unbrushed teeth.
Hospital patients not getting their teeth brushed, or not brushing their teeth themselves, is believed to be a leading cause of hundreds of thousands of cases of pneumonia a year in patients who have not been put on a ventilator. Pneumonia is among the most common infections that occur in health care facilities, and a majority of cases are nonventilator hospital-acquired pneumonia, or NVHAP, which kills up to 30% of those infected, Dr. Giuliano and other experts said.
But unlike many infections that strike within hospitals, the federal government doesn’t require hospitals to report cases of NVHAP. As a result, few hospitals understand the origin of the illness, track its occurrence, or actively work to prevent it, the experts said.
, according to a growing body of peer-reviewed research papers. Instead, many hospitals often skip teeth brushing to prioritize other tasks and provide only cheap, ineffective toothbrushes, often unaware of the consequences, said Dr. Dian Baker, a Sacramento (Calif.) State nursing professor who has spent more than a decade studying NVHAP.
“I’ll tell you that today the vast majority of the tens of thousands of nurses in hospitals have no idea that pneumonia comes from germs in the mouth,” Dr. Baker said.
Pneumonia occurs when germs trigger an infection in the lungs. Although NVHAP accounts for most of the cases that occur in hospitals, it historically has not received the same attention as pneumonia tied to ventilators, which is easier to identify and study because it occurs among a narrow subset of patients.
NVHAP, a risk for virtually all hospital patients, is often caused by bacteria from the mouth that gathers in the scummy biofilm on unbrushed teeth and is aspirated into the lungs. Patients face a higher risk if they lie flat or remain immobile for long periods, so NVHAP can also be prevented by elevating their heads and getting them out of bed more often.
According to the National Organization for NV-HAP Prevention, which was founded in 2020, this pneumonia infects about 1 in every 100 hospital patients and kills 15%-30% of them. For those who survive, the illness often extends their hospital stay by up to 15 days and makes it much more likely they will be readmitted within a month or transferred to an intensive care unit.
John McCleary, 83, of Millinocket, Maine, contracted a likely case of NVHAP in 2008 after he fractured his ankle in a fall and spent 12 days in rehabilitation at a hospital, said his daughter, Kathy Day, a retired nurse and advocate with the Patient Safety Action Network.
Mr. McCleary recovered from the fracture but not from pneumonia. Two days after he returned home, the infection in his lungs caused him to be rushed back to the hospital, where he went into sepsis and spent weeks in treatment before moving to an isolation unit in a nursing home.
He died weeks later, emaciated, largely deaf, unable to eat, and often “too weak to get water through a straw,” his daughter said. After contracting pneumonia, he never walked again.
“It was an astounding assault on his body, from him being here visiting me the week before his fall, to his death just a few months later,” Ms. Day said. “And the whole thing was avoidable.”
While experts describe NVHAP as a largely ignored threat, that appears to be changing.
Last year, a group of researchers – including Dr. Giuliano and Dr. Baker, plus officials from the Centers for Disease Control and Prevention, the Veterans Health Administration, and the Joint Commission – published a “call-to-action” research paper hoping to launch “a national health care conversation about NVHAP prevention.”
The Joint Commission, a nonprofit organization whose accreditation can make or break hospitals, is considering broadening the infection control standards to include more ailments, including NVHAP, said Sylvia Garcia-Houchins, its director of infection prevention and control.
Separately, ECRI, a nonprofit focused on health care safety, this year pinpointed NVHAP as one of its top patient safety concerns.
James Davis, an ECRI infection expert, said the prevalence of NVHAP, while already alarming, is likely “underestimated” and probably worsened as hospitals swelled with patients during the coronavirus pandemic.
“We only know what’s reported,” Mr. Davis said. “Could this be the tip of the iceberg? I would say, in my opinion, probably.”
To better measure the condition, some researchers call for a standardized surveillance definition of NVHAP, which could in time open the door for the federal government to mandate reporting of cases or incentivize prevention. With increasing urgency, researchers are pushing for hospitals not to wait for the federal government to act against NVHAP.
Dr. Baker said she has spoken with hundreds of hospitals about how to prevent NVHAP, but thousands more have yet to take up the cause.
“We are not asking for some big, $300,000 piece of equipment,” Dr. Baker said. “The two things that show the best evidence of preventing this harm are things that should be happening in standard care anyway – brushing teeth and getting patients mobilized.”
That evidence comes from a smattering of studies that show those two strategies can lead to sharp reductions in infection rates.
In California, a study at 21 Kaiser Permanente hospitals used a reprioritization of oral care and getting patients out of bed to reduce rates of hospital-acquired pneumonia by around 70%. At Sutter Medical Center in Sacramento, better oral care reduced NVHAP cases by a yearly average of 35%.
At Orlando Regional Medical Center in Florida, a medical unit and a surgical unit where patients received enhanced oral care reduced NVHAP rates by 85% and 56%, respectively, when compared with similar units that received normal care. A similar study is underway at two hospitals in Illinois.
And the most compelling results come from a veterans’ hospital in Salem, Va., where a 2016 oral care pilot program reduced rates of NVHAP by 92% – saving an estimated 13 lives in just 19 months. The program, the HAPPEN Initiative, has been expanded across the Veterans Health Administration, and experts say it could serve as a model for all U.S. hospitals.
Dr. Michelle Lucatorto, a nursing official who leads HAPPEN, said the program trains nurses to most effectively brush patients’ teeth and educates patients and families on the link between oral care and preventing NVHAP. While teeth brushing may not seem to require training, Dr. Lucatorto made comparisons to how the coronavirus revealed many Americans were doing a lackluster job of another routine hygienic practice: washing their hands.
“Sometimes we are searching for the most complicated intervention,” she said. “We are always looking for that new bypass surgery, or some new technical equipment. And sometimes I think we fail to look at the simple things we can do in our practice to save people’s lives.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Four years ago, when Dr. Karen Giuliano went to a Boston hospital for hip replacement surgery, she was given a pale-pink bucket of toiletries issued to patients in many hospitals. Inside were tissues, bar soap, deodorant, toothpaste, and, without a doubt, the worst toothbrush she’d ever seen.
“I couldn’t believe it. I got a toothbrush with no bristles,” she said. “It must have not gone through the bristle machine. It was just a stick.”
To most patients, a useless hospital toothbrush would be a mild inconvenience. But to Dr. Giuliano, a nursing professor at the University of Massachusetts, Amherst, it was a reminder of a pervasive “blind spot” in U.S. hospitals: the stunning consequences of unbrushed teeth.
Hospital patients not getting their teeth brushed, or not brushing their teeth themselves, is believed to be a leading cause of hundreds of thousands of cases of pneumonia a year in patients who have not been put on a ventilator. Pneumonia is among the most common infections that occur in health care facilities, and a majority of cases are nonventilator hospital-acquired pneumonia, or NVHAP, which kills up to 30% of those infected, Dr. Giuliano and other experts said.
But unlike many infections that strike within hospitals, the federal government doesn’t require hospitals to report cases of NVHAP. As a result, few hospitals understand the origin of the illness, track its occurrence, or actively work to prevent it, the experts said.
, according to a growing body of peer-reviewed research papers. Instead, many hospitals often skip teeth brushing to prioritize other tasks and provide only cheap, ineffective toothbrushes, often unaware of the consequences, said Dr. Dian Baker, a Sacramento (Calif.) State nursing professor who has spent more than a decade studying NVHAP.
“I’ll tell you that today the vast majority of the tens of thousands of nurses in hospitals have no idea that pneumonia comes from germs in the mouth,” Dr. Baker said.
Pneumonia occurs when germs trigger an infection in the lungs. Although NVHAP accounts for most of the cases that occur in hospitals, it historically has not received the same attention as pneumonia tied to ventilators, which is easier to identify and study because it occurs among a narrow subset of patients.
NVHAP, a risk for virtually all hospital patients, is often caused by bacteria from the mouth that gathers in the scummy biofilm on unbrushed teeth and is aspirated into the lungs. Patients face a higher risk if they lie flat or remain immobile for long periods, so NVHAP can also be prevented by elevating their heads and getting them out of bed more often.
According to the National Organization for NV-HAP Prevention, which was founded in 2020, this pneumonia infects about 1 in every 100 hospital patients and kills 15%-30% of them. For those who survive, the illness often extends their hospital stay by up to 15 days and makes it much more likely they will be readmitted within a month or transferred to an intensive care unit.
John McCleary, 83, of Millinocket, Maine, contracted a likely case of NVHAP in 2008 after he fractured his ankle in a fall and spent 12 days in rehabilitation at a hospital, said his daughter, Kathy Day, a retired nurse and advocate with the Patient Safety Action Network.
Mr. McCleary recovered from the fracture but not from pneumonia. Two days after he returned home, the infection in his lungs caused him to be rushed back to the hospital, where he went into sepsis and spent weeks in treatment before moving to an isolation unit in a nursing home.
He died weeks later, emaciated, largely deaf, unable to eat, and often “too weak to get water through a straw,” his daughter said. After contracting pneumonia, he never walked again.
“It was an astounding assault on his body, from him being here visiting me the week before his fall, to his death just a few months later,” Ms. Day said. “And the whole thing was avoidable.”
While experts describe NVHAP as a largely ignored threat, that appears to be changing.
Last year, a group of researchers – including Dr. Giuliano and Dr. Baker, plus officials from the Centers for Disease Control and Prevention, the Veterans Health Administration, and the Joint Commission – published a “call-to-action” research paper hoping to launch “a national health care conversation about NVHAP prevention.”
The Joint Commission, a nonprofit organization whose accreditation can make or break hospitals, is considering broadening the infection control standards to include more ailments, including NVHAP, said Sylvia Garcia-Houchins, its director of infection prevention and control.
Separately, ECRI, a nonprofit focused on health care safety, this year pinpointed NVHAP as one of its top patient safety concerns.
James Davis, an ECRI infection expert, said the prevalence of NVHAP, while already alarming, is likely “underestimated” and probably worsened as hospitals swelled with patients during the coronavirus pandemic.
“We only know what’s reported,” Mr. Davis said. “Could this be the tip of the iceberg? I would say, in my opinion, probably.”
To better measure the condition, some researchers call for a standardized surveillance definition of NVHAP, which could in time open the door for the federal government to mandate reporting of cases or incentivize prevention. With increasing urgency, researchers are pushing for hospitals not to wait for the federal government to act against NVHAP.
Dr. Baker said she has spoken with hundreds of hospitals about how to prevent NVHAP, but thousands more have yet to take up the cause.
“We are not asking for some big, $300,000 piece of equipment,” Dr. Baker said. “The two things that show the best evidence of preventing this harm are things that should be happening in standard care anyway – brushing teeth and getting patients mobilized.”
That evidence comes from a smattering of studies that show those two strategies can lead to sharp reductions in infection rates.
In California, a study at 21 Kaiser Permanente hospitals used a reprioritization of oral care and getting patients out of bed to reduce rates of hospital-acquired pneumonia by around 70%. At Sutter Medical Center in Sacramento, better oral care reduced NVHAP cases by a yearly average of 35%.
At Orlando Regional Medical Center in Florida, a medical unit and a surgical unit where patients received enhanced oral care reduced NVHAP rates by 85% and 56%, respectively, when compared with similar units that received normal care. A similar study is underway at two hospitals in Illinois.
And the most compelling results come from a veterans’ hospital in Salem, Va., where a 2016 oral care pilot program reduced rates of NVHAP by 92% – saving an estimated 13 lives in just 19 months. The program, the HAPPEN Initiative, has been expanded across the Veterans Health Administration, and experts say it could serve as a model for all U.S. hospitals.
Dr. Michelle Lucatorto, a nursing official who leads HAPPEN, said the program trains nurses to most effectively brush patients’ teeth and educates patients and families on the link between oral care and preventing NVHAP. While teeth brushing may not seem to require training, Dr. Lucatorto made comparisons to how the coronavirus revealed many Americans were doing a lackluster job of another routine hygienic practice: washing their hands.
“Sometimes we are searching for the most complicated intervention,” she said. “We are always looking for that new bypass surgery, or some new technical equipment. And sometimes I think we fail to look at the simple things we can do in our practice to save people’s lives.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.