User login
JIA disease activity, disability linked to social factors
For children with polyarticular juvenile idiopathic arthritis (pJIA), functional disability lasts longer and disease activity is increased among those who belong to a racial/ethnic minority or come from homes with low household income or low family education, according to a study published online in Pediatric Rheumatology. The findings also initially revealed a higher likelihood of functional disability among those living in a poorer community, but that association lost statistical significance after adjustment for confounders.
“We chose community poverty level as the primary predictor for outcomes in pJIA because the socioeconomic context of communities and neighborhoods affects the characteristics of the social, service, and physical environments to which all residents are exposed regardless of their own socioeconomic position and may have a greater negative impact on those with fewer individual resources,” the authors write. “While community poverty level was not associated with an increase in odds of moderate-to-severe disease activity, those with high community poverty level did have higher disease activity scores (0.33 points greater on average than those with low community poverty level, in adjusted analysis).”
Nayimisha Balmuri, MD, an assistant professor of pediatrics at Johns Hopkins Medicine and study coauthor, told this news organization that anecdotal experience from everyday practice has shown that “patients with myriad social determinants of health stacked against them present sicker, take longer to present, and require far more aggressive therapies and follow-up,” which wreaks havoc in terms of disease activity. “It’s really difficult, then, to play catch-up to other cohorts of patients,” Dr. Balmuri added.
Disparities in outcomes persist
A key clinical take-home message from these findings is that the differences in clinical outcomes are relevant throughout the entire year of therapy, Dr. Balmuri said. “Patients get better; however, they don’t get better the same,” she said, and this is because of a variety of reasons. “Getting in the door is one of [those reasons] but then continuing to follow-up care is another.” For general practitioners, it’s especially important to refer patients who complain of joint pains to a specialist and to then follow up to be sure they’re improving and they’re getting the care they need.
For pediatric rheumatologists and subspecialists, “it’s important for us to realize that the disparity doesn’t end when patients come into your door to begin with,” Dr. Balmuri said. “It continues over the short term and far past that into adulthood.”
Candace Feldman, MD, MPH, ScD, an assistant professor of medicine in the Division of Rheumatology, Inflammation, and Immunity at Brigham and Women’s Hospital, Boston, told this news organization that the research “provides an important foundation to the study of the impact of social determinants of health on disease activity and disability among children with JIA. Individuals with rheumatic conditions should be screened for social determinants of health–related needs, and infrastructure should exist within the rheumatology clinic to help address the needs uncovered.” Dr. Feldman was not involved in the study.
In addition to the results’ clinical significance, Dr. Feldman also noted the policy implications of these findings. “Physicians should advocate for efforts to dismantle structural racism, to address income inequality, and to mitigate the effects of climate change, which also disproportionately affect historically marginalized populations,” Dr. Feldman said. Although this study focused predominantly on poverty, she noted that financial insecurity, food insecurity, homelessness, or housing instability were other social determinants of health to consider in future research.
Dr. Balmuri and William Daniel Soulsby, MD, a clinical fellow in pediatric rheumatology at the University of California, San Francisco, who is the study’s lead author, said they focused on poverty in this study not only because it’s so understudied in patients with pJIA but also because research in adults with lupus has found that leaving poverty was associated with a reversal of accrued disease damage.
Interactions of social determinants
The authors analyzed retrospective data from 1,684 pediatric patients in the Childhood Arthritis and Rheumatology Research Alliance (CARRA) registry covering the period of April 2015 to February 2020. All study participants had been diagnosed with pJIA. Symptom onset occurred before age 16, and at least five joints were involved. The authors excluded patients who had been diagnosed with other systemic inflammatory or autoimmune diseases.
The authors defined exposure to a high level of community poverty as living in a ZIP code where at least 20% of residents lived at or below the federal poverty level. The authors also collected data on household income, although these data were missing for more than a quarter of participants (27%) and were therefore included only in sensitivity analyses. They used the clinical Juvenile Arthritis Disease Activity Score–10 (cJADAS-10) and the Child Health Assessment Questionnaire (CHAQ) to assess disease activity and disability at baseline and 6 and 12 months later. A cutoff of 2.5 on the cJADAS-10 distinguished mild disease activity from moderate to high disease activity, and a CHAQ score of 0.25 was the cutoff for having functional disability.
Among those who reported household income, just over half the cohort had an income of at least $50,000. The study population was 74% White, and more non-White patients lived in high-poverty communities (36.4%) than did White patients (21.3%). Patients whose families had no more than a high school education (23.1% vs. 13.7%) and those with public insurance (43.0% vs. 21.5%) were also over-represented in poorer communities.
The median cJADAS-10 scores declined overall during patients’ first year of therapy. However, those with public insurance, a lower family education level, or residency in poorer communities made up the greatest proportion of patients who continued to have moderate to severe disease activity a year after diagnosis.
The unadjusted calculations showed that children living in high community poverty had 1.8 times greater odds of functional disability (odds ratio, 1.82; P < .001). However, after adjustment for age, sex, race/ethnicity, insurance status, family education, rheumatoid factor, and cyclic citrullinated peptide antibody, the association lost statistical significance (P = .3). Community poverty level was not associated with disease activity before or after adjustment.
“Race was adjusted for as a confounder; however, the association between race/ethnicity and social determinants of health is likely more complex,” Dr. Feldman said. “Interactions, for example, between individual race and area-level poverty could be investigated.”
Odds of persistent function disability were 1.5 times greater for children with public insurance (adjusted OR, 1.56; P = .023) and 1.9 times greater for those whose families had a lower education level (aOR, 1.89; P = .013). Children whose race/ethnicity was indicated as being other than White had more than double the odds of higher disease activity (aOR, 2.48; P = .002) and were nearly twice as likely to have persistent functional disability (aOR, 1.91; P = .031).
Future directions
Dr. Soulsby was struck by the difference in statistical significance between individual-level poverty, as measured by household income, and community-level poverty. “It’s interesting because it may suggest that both of these forms of poverty are different and have different impacts on disease,” he said. Dr. Balmuri elaborated on the nuances and interactions that exist with social determinants of health and how objective outcomes, such as disease activity as measured by clinical tools, can differ from subjective outcomes, such as patients’ reports of pain, daily disability, and social experiences.
“The human condition is far more complicated, unfortunately, than any dataset could have on their own collected,” Dr. Balmuri said. She said she plans to expand her pJIA research into other social determinants of health. “It’s first about getting people’s eyes and minds open to something we see every day that, for some reason, sometimes people are blinded to, [using] the data that we do have, and then our hope is to build upon that.”
Dr. Feldman noted that ZIP codes, which were used as a proxy for community poverty, may not provide the best perspective regarding a patient’s neighborhood, because significant variation may exist within a single ZIP code, which is something the authors noted as well. The investigators were limited in the data available from the registry, and Dr. Balmuri and Dr. Soulsby suggested that 9-digit ZIP codes or census tracts might better capture neighborhood deprivation.
The research was funded by the Arthritis Foundation and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Feldman has received research support from Pfizer and the Bristol-Myers Squibb Foundation. Dr. Soulsby and Dr. Balmuri have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For children with polyarticular juvenile idiopathic arthritis (pJIA), functional disability lasts longer and disease activity is increased among those who belong to a racial/ethnic minority or come from homes with low household income or low family education, according to a study published online in Pediatric Rheumatology. The findings also initially revealed a higher likelihood of functional disability among those living in a poorer community, but that association lost statistical significance after adjustment for confounders.
“We chose community poverty level as the primary predictor for outcomes in pJIA because the socioeconomic context of communities and neighborhoods affects the characteristics of the social, service, and physical environments to which all residents are exposed regardless of their own socioeconomic position and may have a greater negative impact on those with fewer individual resources,” the authors write. “While community poverty level was not associated with an increase in odds of moderate-to-severe disease activity, those with high community poverty level did have higher disease activity scores (0.33 points greater on average than those with low community poverty level, in adjusted analysis).”
Nayimisha Balmuri, MD, an assistant professor of pediatrics at Johns Hopkins Medicine and study coauthor, told this news organization that anecdotal experience from everyday practice has shown that “patients with myriad social determinants of health stacked against them present sicker, take longer to present, and require far more aggressive therapies and follow-up,” which wreaks havoc in terms of disease activity. “It’s really difficult, then, to play catch-up to other cohorts of patients,” Dr. Balmuri added.
Disparities in outcomes persist
A key clinical take-home message from these findings is that the differences in clinical outcomes are relevant throughout the entire year of therapy, Dr. Balmuri said. “Patients get better; however, they don’t get better the same,” she said, and this is because of a variety of reasons. “Getting in the door is one of [those reasons] but then continuing to follow-up care is another.” For general practitioners, it’s especially important to refer patients who complain of joint pains to a specialist and to then follow up to be sure they’re improving and they’re getting the care they need.
For pediatric rheumatologists and subspecialists, “it’s important for us to realize that the disparity doesn’t end when patients come into your door to begin with,” Dr. Balmuri said. “It continues over the short term and far past that into adulthood.”
Candace Feldman, MD, MPH, ScD, an assistant professor of medicine in the Division of Rheumatology, Inflammation, and Immunity at Brigham and Women’s Hospital, Boston, told this news organization that the research “provides an important foundation to the study of the impact of social determinants of health on disease activity and disability among children with JIA. Individuals with rheumatic conditions should be screened for social determinants of health–related needs, and infrastructure should exist within the rheumatology clinic to help address the needs uncovered.” Dr. Feldman was not involved in the study.
In addition to the results’ clinical significance, Dr. Feldman also noted the policy implications of these findings. “Physicians should advocate for efforts to dismantle structural racism, to address income inequality, and to mitigate the effects of climate change, which also disproportionately affect historically marginalized populations,” Dr. Feldman said. Although this study focused predominantly on poverty, she noted that financial insecurity, food insecurity, homelessness, or housing instability were other social determinants of health to consider in future research.
Dr. Balmuri and William Daniel Soulsby, MD, a clinical fellow in pediatric rheumatology at the University of California, San Francisco, who is the study’s lead author, said they focused on poverty in this study not only because it’s so understudied in patients with pJIA but also because research in adults with lupus has found that leaving poverty was associated with a reversal of accrued disease damage.
Interactions of social determinants
The authors analyzed retrospective data from 1,684 pediatric patients in the Childhood Arthritis and Rheumatology Research Alliance (CARRA) registry covering the period of April 2015 to February 2020. All study participants had been diagnosed with pJIA. Symptom onset occurred before age 16, and at least five joints were involved. The authors excluded patients who had been diagnosed with other systemic inflammatory or autoimmune diseases.
The authors defined exposure to a high level of community poverty as living in a ZIP code where at least 20% of residents lived at or below the federal poverty level. The authors also collected data on household income, although these data were missing for more than a quarter of participants (27%) and were therefore included only in sensitivity analyses. They used the clinical Juvenile Arthritis Disease Activity Score–10 (cJADAS-10) and the Child Health Assessment Questionnaire (CHAQ) to assess disease activity and disability at baseline and 6 and 12 months later. A cutoff of 2.5 on the cJADAS-10 distinguished mild disease activity from moderate to high disease activity, and a CHAQ score of 0.25 was the cutoff for having functional disability.
Among those who reported household income, just over half the cohort had an income of at least $50,000. The study population was 74% White, and more non-White patients lived in high-poverty communities (36.4%) than did White patients (21.3%). Patients whose families had no more than a high school education (23.1% vs. 13.7%) and those with public insurance (43.0% vs. 21.5%) were also over-represented in poorer communities.
The median cJADAS-10 scores declined overall during patients’ first year of therapy. However, those with public insurance, a lower family education level, or residency in poorer communities made up the greatest proportion of patients who continued to have moderate to severe disease activity a year after diagnosis.
The unadjusted calculations showed that children living in high community poverty had 1.8 times greater odds of functional disability (odds ratio, 1.82; P < .001). However, after adjustment for age, sex, race/ethnicity, insurance status, family education, rheumatoid factor, and cyclic citrullinated peptide antibody, the association lost statistical significance (P = .3). Community poverty level was not associated with disease activity before or after adjustment.
“Race was adjusted for as a confounder; however, the association between race/ethnicity and social determinants of health is likely more complex,” Dr. Feldman said. “Interactions, for example, between individual race and area-level poverty could be investigated.”
Odds of persistent function disability were 1.5 times greater for children with public insurance (adjusted OR, 1.56; P = .023) and 1.9 times greater for those whose families had a lower education level (aOR, 1.89; P = .013). Children whose race/ethnicity was indicated as being other than White had more than double the odds of higher disease activity (aOR, 2.48; P = .002) and were nearly twice as likely to have persistent functional disability (aOR, 1.91; P = .031).
Future directions
Dr. Soulsby was struck by the difference in statistical significance between individual-level poverty, as measured by household income, and community-level poverty. “It’s interesting because it may suggest that both of these forms of poverty are different and have different impacts on disease,” he said. Dr. Balmuri elaborated on the nuances and interactions that exist with social determinants of health and how objective outcomes, such as disease activity as measured by clinical tools, can differ from subjective outcomes, such as patients’ reports of pain, daily disability, and social experiences.
“The human condition is far more complicated, unfortunately, than any dataset could have on their own collected,” Dr. Balmuri said. She said she plans to expand her pJIA research into other social determinants of health. “It’s first about getting people’s eyes and minds open to something we see every day that, for some reason, sometimes people are blinded to, [using] the data that we do have, and then our hope is to build upon that.”
Dr. Feldman noted that ZIP codes, which were used as a proxy for community poverty, may not provide the best perspective regarding a patient’s neighborhood, because significant variation may exist within a single ZIP code, which is something the authors noted as well. The investigators were limited in the data available from the registry, and Dr. Balmuri and Dr. Soulsby suggested that 9-digit ZIP codes or census tracts might better capture neighborhood deprivation.
The research was funded by the Arthritis Foundation and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Feldman has received research support from Pfizer and the Bristol-Myers Squibb Foundation. Dr. Soulsby and Dr. Balmuri have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For children with polyarticular juvenile idiopathic arthritis (pJIA), functional disability lasts longer and disease activity is increased among those who belong to a racial/ethnic minority or come from homes with low household income or low family education, according to a study published online in Pediatric Rheumatology. The findings also initially revealed a higher likelihood of functional disability among those living in a poorer community, but that association lost statistical significance after adjustment for confounders.
“We chose community poverty level as the primary predictor for outcomes in pJIA because the socioeconomic context of communities and neighborhoods affects the characteristics of the social, service, and physical environments to which all residents are exposed regardless of their own socioeconomic position and may have a greater negative impact on those with fewer individual resources,” the authors write. “While community poverty level was not associated with an increase in odds of moderate-to-severe disease activity, those with high community poverty level did have higher disease activity scores (0.33 points greater on average than those with low community poverty level, in adjusted analysis).”
Nayimisha Balmuri, MD, an assistant professor of pediatrics at Johns Hopkins Medicine and study coauthor, told this news organization that anecdotal experience from everyday practice has shown that “patients with myriad social determinants of health stacked against them present sicker, take longer to present, and require far more aggressive therapies and follow-up,” which wreaks havoc in terms of disease activity. “It’s really difficult, then, to play catch-up to other cohorts of patients,” Dr. Balmuri added.
Disparities in outcomes persist
A key clinical take-home message from these findings is that the differences in clinical outcomes are relevant throughout the entire year of therapy, Dr. Balmuri said. “Patients get better; however, they don’t get better the same,” she said, and this is because of a variety of reasons. “Getting in the door is one of [those reasons] but then continuing to follow-up care is another.” For general practitioners, it’s especially important to refer patients who complain of joint pains to a specialist and to then follow up to be sure they’re improving and they’re getting the care they need.
For pediatric rheumatologists and subspecialists, “it’s important for us to realize that the disparity doesn’t end when patients come into your door to begin with,” Dr. Balmuri said. “It continues over the short term and far past that into adulthood.”
Candace Feldman, MD, MPH, ScD, an assistant professor of medicine in the Division of Rheumatology, Inflammation, and Immunity at Brigham and Women’s Hospital, Boston, told this news organization that the research “provides an important foundation to the study of the impact of social determinants of health on disease activity and disability among children with JIA. Individuals with rheumatic conditions should be screened for social determinants of health–related needs, and infrastructure should exist within the rheumatology clinic to help address the needs uncovered.” Dr. Feldman was not involved in the study.
In addition to the results’ clinical significance, Dr. Feldman also noted the policy implications of these findings. “Physicians should advocate for efforts to dismantle structural racism, to address income inequality, and to mitigate the effects of climate change, which also disproportionately affect historically marginalized populations,” Dr. Feldman said. Although this study focused predominantly on poverty, she noted that financial insecurity, food insecurity, homelessness, or housing instability were other social determinants of health to consider in future research.
Dr. Balmuri and William Daniel Soulsby, MD, a clinical fellow in pediatric rheumatology at the University of California, San Francisco, who is the study’s lead author, said they focused on poverty in this study not only because it’s so understudied in patients with pJIA but also because research in adults with lupus has found that leaving poverty was associated with a reversal of accrued disease damage.
Interactions of social determinants
The authors analyzed retrospective data from 1,684 pediatric patients in the Childhood Arthritis and Rheumatology Research Alliance (CARRA) registry covering the period of April 2015 to February 2020. All study participants had been diagnosed with pJIA. Symptom onset occurred before age 16, and at least five joints were involved. The authors excluded patients who had been diagnosed with other systemic inflammatory or autoimmune diseases.
The authors defined exposure to a high level of community poverty as living in a ZIP code where at least 20% of residents lived at or below the federal poverty level. The authors also collected data on household income, although these data were missing for more than a quarter of participants (27%) and were therefore included only in sensitivity analyses. They used the clinical Juvenile Arthritis Disease Activity Score–10 (cJADAS-10) and the Child Health Assessment Questionnaire (CHAQ) to assess disease activity and disability at baseline and 6 and 12 months later. A cutoff of 2.5 on the cJADAS-10 distinguished mild disease activity from moderate to high disease activity, and a CHAQ score of 0.25 was the cutoff for having functional disability.
Among those who reported household income, just over half the cohort had an income of at least $50,000. The study population was 74% White, and more non-White patients lived in high-poverty communities (36.4%) than did White patients (21.3%). Patients whose families had no more than a high school education (23.1% vs. 13.7%) and those with public insurance (43.0% vs. 21.5%) were also over-represented in poorer communities.
The median cJADAS-10 scores declined overall during patients’ first year of therapy. However, those with public insurance, a lower family education level, or residency in poorer communities made up the greatest proportion of patients who continued to have moderate to severe disease activity a year after diagnosis.
The unadjusted calculations showed that children living in high community poverty had 1.8 times greater odds of functional disability (odds ratio, 1.82; P < .001). However, after adjustment for age, sex, race/ethnicity, insurance status, family education, rheumatoid factor, and cyclic citrullinated peptide antibody, the association lost statistical significance (P = .3). Community poverty level was not associated with disease activity before or after adjustment.
“Race was adjusted for as a confounder; however, the association between race/ethnicity and social determinants of health is likely more complex,” Dr. Feldman said. “Interactions, for example, between individual race and area-level poverty could be investigated.”
Odds of persistent function disability were 1.5 times greater for children with public insurance (adjusted OR, 1.56; P = .023) and 1.9 times greater for those whose families had a lower education level (aOR, 1.89; P = .013). Children whose race/ethnicity was indicated as being other than White had more than double the odds of higher disease activity (aOR, 2.48; P = .002) and were nearly twice as likely to have persistent functional disability (aOR, 1.91; P = .031).
Future directions
Dr. Soulsby was struck by the difference in statistical significance between individual-level poverty, as measured by household income, and community-level poverty. “It’s interesting because it may suggest that both of these forms of poverty are different and have different impacts on disease,” he said. Dr. Balmuri elaborated on the nuances and interactions that exist with social determinants of health and how objective outcomes, such as disease activity as measured by clinical tools, can differ from subjective outcomes, such as patients’ reports of pain, daily disability, and social experiences.
“The human condition is far more complicated, unfortunately, than any dataset could have on their own collected,” Dr. Balmuri said. She said she plans to expand her pJIA research into other social determinants of health. “It’s first about getting people’s eyes and minds open to something we see every day that, for some reason, sometimes people are blinded to, [using] the data that we do have, and then our hope is to build upon that.”
Dr. Feldman noted that ZIP codes, which were used as a proxy for community poverty, may not provide the best perspective regarding a patient’s neighborhood, because significant variation may exist within a single ZIP code, which is something the authors noted as well. The investigators were limited in the data available from the registry, and Dr. Balmuri and Dr. Soulsby suggested that 9-digit ZIP codes or census tracts might better capture neighborhood deprivation.
The research was funded by the Arthritis Foundation and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Feldman has received research support from Pfizer and the Bristol-Myers Squibb Foundation. Dr. Soulsby and Dr. Balmuri have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM PEDIATRIC RHEUMATOLOGY
May 2022 - ICYMI
Gastroenterology
February 2022
How to Succeed in Digestive Research
Sonnenberg A, Inadomi JM. Gastroenterology. 2022 Feb;162(2):385-389. doi: 10.1053/j.gastro.2021.12.229.
Incidence and Mortality in Upper Gastrointestinal Cancer After Negative Endoscopy for Gastroesophageal Reflux Disease
Holmberg H et al. Gastroenterology. 2022 Feb;162(2):431-438.e4. doi: 10.1053/j.gastro.2021.10.003.
March 2022
Global Prevalence and Impact of Rumination Syndrome
Josefsson A et al. Gastroenterology. 2022 Mar;162(3):731-742.e9. doi: 10.1053/j.gastro.2021.11.008.
A Clinical Approach to Chronic Diarrhea
Dutra B et al. Gastroenterology. 2022 Mar;162(3):707-709. doi: 10.1053/j.gastro.2021.07.038.
Timeline of Development of Pancreatic Cancer and Implications for Successful Early Detection in High-Risk Individuals
Overbeek KA et al. Gastroenterology. 2022 Mar;162(3):772-785.e4. doi: 10.1053/j.gastro.2021.10.014.
April 2022
Artificial Intelligence and Deep Learning for Upper Gastrointestinal Neoplasia
Sharma P, Hassan C. Gastroenterology. 2022 Apr;162(4):1056-1066. doi: 10.1053/j.gastro.2021.11.040.
Associations of Body Mass Index at Different Ages With Early-Onset Colorectal Cancer
Li H et al. Gastroenterology. 2022 Apr;162(4):1088-1097.e3. doi: 10.1053/j.gastro.2021.12.239.
Inadequate Rectal Pressure and Insufficient Relaxation and Abdominopelvic Coordination in Defecatory Disorders
Deb B et al. Gastroenterology. 2022 Apr;162(4):1111-1122.e2. doi: 10.1053/j.gastro.2021.12.257.
AGA Clinical Practice Update on De-Prescribing of Proton Pump Inhibitors: Expert Review
Targownik LE et al. Gastroenterology. 2022 Apr;162(4):1334-1342. doi: 10.1053/j.gastro.2021.12.247.
Clinical Gastroenterology and Hepatology
February 2022
Restarting Warfarin vs Direct Oral Anticoagulants After Major Gastrointestinal Bleeding and Associated Outcomes in Atrial Fibrillation: A Cohort Study
Tapaskar N et al. Clin Gastroenterol Hepatol. 2022 Feb;20(2):381-389.e9. doi: 10.1016/j.cgh.2020.11.029.
Cancer Risk in 47,241 Individuals With Celiac Disease: A Nationwide Cohort Study
Lebwohl B et al. Clin Gastroenterol Hepatol. 2022 Feb;20(2):e111-e131. doi: 10.1016/j.cgh.2021.05.034.
Main Duct Thresholds for Malignancy Are Different in Intraductal Papillary Mucinous Neoplasms of the Pancreatic Head and Body-Tail
Crippa S et al. Clin Gastroenterol Hepatol. 2022 Feb;20(2):390-399.e7. doi: 10.1016/j.cgh.2020.12.028.
Frequency of Bowel Movements and Risk of Diverticulitis
Jovani M et al. Clin Gastroenterol Hepatol. 2022 Feb;20(2):325-333.e5. doi: 10.1016/j.cgh.2021.01.003.
March 2022
AGA Clinical Practice Update on Management of Medically Refractory Gastroparesis: Expert Review
Lacy BE et al. Clin Gastroenterol Hepatol. 2022 Mar;20(3):491-500. doi: 10.1016/j.cgh.2021.10.038.
Efficacy and Safety of Tofacitinib in Ulcerative Colitis Based on Prior Tumor Necrosis Factor Inhibitor Failure Status
Sandborn WJ et al. Clin Gastroenterol Hepatol. 2022 Mar;20(3):591-601.e8. doi: 10.1016/j.cgh.2021.02.043.
April 2022
What Faculty and Fellows Should Know About Milestones 2.0
Donnangelo JL, Brijen SJ. Clin Gastroenterol Hepatol. 2022 Apr;20(4):720-722. doi: 10.1016/j.cgh.2021.12.017.
Patient Experience in the Gastrointestinal Endoscopy Unit
Day LW, Savides TJ. Clin Gastroenterol Hepatol. 2022 Apr;20(4):723-726. doi: 10.1016/j.cgh.2021.12.001.
Tailoring Surveillance Colonoscopy in Patients With Advanced Adenomas
Kahi CJ et al. Clin Gastroenterol Hepatol. 2022 Apr;20(4):847-854.e1. doi: 10.1016/j.cgh.2021.03.027.
Techniques and Innovations in Gastrointestinal Endoscopy
Primary CT Angiography Vs Colonoscopy in Acute Lower Gastrointestinal Hemorrhage
Lipcsey MS et al. Tech Innov Gastrointest Endosc. 2022 Jan 01;24(1):2-9. doi: 10.1016/j.tige.2021.11.004.
Cellular and Molecular Gastroenterology and Hepatology
The Role of Microbiota in Gastrointestinal Cancer and Cancer Treatment: Chance or Curse?
Smet A et al. Cell Mol Gastroenterol Hepatol. 2022;13(3):857-874. doi: 10.1016/j.jcmgh.2021.08.013.
Gastroenterology
February 2022
How to Succeed in Digestive Research
Sonnenberg A, Inadomi JM. Gastroenterology. 2022 Feb;162(2):385-389. doi: 10.1053/j.gastro.2021.12.229.
Incidence and Mortality in Upper Gastrointestinal Cancer After Negative Endoscopy for Gastroesophageal Reflux Disease
Holmberg H et al. Gastroenterology. 2022 Feb;162(2):431-438.e4. doi: 10.1053/j.gastro.2021.10.003.
March 2022
Global Prevalence and Impact of Rumination Syndrome
Josefsson A et al. Gastroenterology. 2022 Mar;162(3):731-742.e9. doi: 10.1053/j.gastro.2021.11.008.
A Clinical Approach to Chronic Diarrhea
Dutra B et al. Gastroenterology. 2022 Mar;162(3):707-709. doi: 10.1053/j.gastro.2021.07.038.
Timeline of Development of Pancreatic Cancer and Implications for Successful Early Detection in High-Risk Individuals
Overbeek KA et al. Gastroenterology. 2022 Mar;162(3):772-785.e4. doi: 10.1053/j.gastro.2021.10.014.
April 2022
Artificial Intelligence and Deep Learning for Upper Gastrointestinal Neoplasia
Sharma P, Hassan C. Gastroenterology. 2022 Apr;162(4):1056-1066. doi: 10.1053/j.gastro.2021.11.040.
Associations of Body Mass Index at Different Ages With Early-Onset Colorectal Cancer
Li H et al. Gastroenterology. 2022 Apr;162(4):1088-1097.e3. doi: 10.1053/j.gastro.2021.12.239.
Inadequate Rectal Pressure and Insufficient Relaxation and Abdominopelvic Coordination in Defecatory Disorders
Deb B et al. Gastroenterology. 2022 Apr;162(4):1111-1122.e2. doi: 10.1053/j.gastro.2021.12.257.
AGA Clinical Practice Update on De-Prescribing of Proton Pump Inhibitors: Expert Review
Targownik LE et al. Gastroenterology. 2022 Apr;162(4):1334-1342. doi: 10.1053/j.gastro.2021.12.247.
Clinical Gastroenterology and Hepatology
February 2022
Restarting Warfarin vs Direct Oral Anticoagulants After Major Gastrointestinal Bleeding and Associated Outcomes in Atrial Fibrillation: A Cohort Study
Tapaskar N et al. Clin Gastroenterol Hepatol. 2022 Feb;20(2):381-389.e9. doi: 10.1016/j.cgh.2020.11.029.
Cancer Risk in 47,241 Individuals With Celiac Disease: A Nationwide Cohort Study
Lebwohl B et al. Clin Gastroenterol Hepatol. 2022 Feb;20(2):e111-e131. doi: 10.1016/j.cgh.2021.05.034.
Main Duct Thresholds for Malignancy Are Different in Intraductal Papillary Mucinous Neoplasms of the Pancreatic Head and Body-Tail
Crippa S et al. Clin Gastroenterol Hepatol. 2022 Feb;20(2):390-399.e7. doi: 10.1016/j.cgh.2020.12.028.
Frequency of Bowel Movements and Risk of Diverticulitis
Jovani M et al. Clin Gastroenterol Hepatol. 2022 Feb;20(2):325-333.e5. doi: 10.1016/j.cgh.2021.01.003.
March 2022
AGA Clinical Practice Update on Management of Medically Refractory Gastroparesis: Expert Review
Lacy BE et al. Clin Gastroenterol Hepatol. 2022 Mar;20(3):491-500. doi: 10.1016/j.cgh.2021.10.038.
Efficacy and Safety of Tofacitinib in Ulcerative Colitis Based on Prior Tumor Necrosis Factor Inhibitor Failure Status
Sandborn WJ et al. Clin Gastroenterol Hepatol. 2022 Mar;20(3):591-601.e8. doi: 10.1016/j.cgh.2021.02.043.
April 2022
What Faculty and Fellows Should Know About Milestones 2.0
Donnangelo JL, Brijen SJ. Clin Gastroenterol Hepatol. 2022 Apr;20(4):720-722. doi: 10.1016/j.cgh.2021.12.017.
Patient Experience in the Gastrointestinal Endoscopy Unit
Day LW, Savides TJ. Clin Gastroenterol Hepatol. 2022 Apr;20(4):723-726. doi: 10.1016/j.cgh.2021.12.001.
Tailoring Surveillance Colonoscopy in Patients With Advanced Adenomas
Kahi CJ et al. Clin Gastroenterol Hepatol. 2022 Apr;20(4):847-854.e1. doi: 10.1016/j.cgh.2021.03.027.
Techniques and Innovations in Gastrointestinal Endoscopy
Primary CT Angiography Vs Colonoscopy in Acute Lower Gastrointestinal Hemorrhage
Lipcsey MS et al. Tech Innov Gastrointest Endosc. 2022 Jan 01;24(1):2-9. doi: 10.1016/j.tige.2021.11.004.
Cellular and Molecular Gastroenterology and Hepatology
The Role of Microbiota in Gastrointestinal Cancer and Cancer Treatment: Chance or Curse?
Smet A et al. Cell Mol Gastroenterol Hepatol. 2022;13(3):857-874. doi: 10.1016/j.jcmgh.2021.08.013.
Gastroenterology
February 2022
How to Succeed in Digestive Research
Sonnenberg A, Inadomi JM. Gastroenterology. 2022 Feb;162(2):385-389. doi: 10.1053/j.gastro.2021.12.229.
Incidence and Mortality in Upper Gastrointestinal Cancer After Negative Endoscopy for Gastroesophageal Reflux Disease
Holmberg H et al. Gastroenterology. 2022 Feb;162(2):431-438.e4. doi: 10.1053/j.gastro.2021.10.003.
March 2022
Global Prevalence and Impact of Rumination Syndrome
Josefsson A et al. Gastroenterology. 2022 Mar;162(3):731-742.e9. doi: 10.1053/j.gastro.2021.11.008.
A Clinical Approach to Chronic Diarrhea
Dutra B et al. Gastroenterology. 2022 Mar;162(3):707-709. doi: 10.1053/j.gastro.2021.07.038.
Timeline of Development of Pancreatic Cancer and Implications for Successful Early Detection in High-Risk Individuals
Overbeek KA et al. Gastroenterology. 2022 Mar;162(3):772-785.e4. doi: 10.1053/j.gastro.2021.10.014.
April 2022
Artificial Intelligence and Deep Learning for Upper Gastrointestinal Neoplasia
Sharma P, Hassan C. Gastroenterology. 2022 Apr;162(4):1056-1066. doi: 10.1053/j.gastro.2021.11.040.
Associations of Body Mass Index at Different Ages With Early-Onset Colorectal Cancer
Li H et al. Gastroenterology. 2022 Apr;162(4):1088-1097.e3. doi: 10.1053/j.gastro.2021.12.239.
Inadequate Rectal Pressure and Insufficient Relaxation and Abdominopelvic Coordination in Defecatory Disorders
Deb B et al. Gastroenterology. 2022 Apr;162(4):1111-1122.e2. doi: 10.1053/j.gastro.2021.12.257.
AGA Clinical Practice Update on De-Prescribing of Proton Pump Inhibitors: Expert Review
Targownik LE et al. Gastroenterology. 2022 Apr;162(4):1334-1342. doi: 10.1053/j.gastro.2021.12.247.
Clinical Gastroenterology and Hepatology
February 2022
Restarting Warfarin vs Direct Oral Anticoagulants After Major Gastrointestinal Bleeding and Associated Outcomes in Atrial Fibrillation: A Cohort Study
Tapaskar N et al. Clin Gastroenterol Hepatol. 2022 Feb;20(2):381-389.e9. doi: 10.1016/j.cgh.2020.11.029.
Cancer Risk in 47,241 Individuals With Celiac Disease: A Nationwide Cohort Study
Lebwohl B et al. Clin Gastroenterol Hepatol. 2022 Feb;20(2):e111-e131. doi: 10.1016/j.cgh.2021.05.034.
Main Duct Thresholds for Malignancy Are Different in Intraductal Papillary Mucinous Neoplasms of the Pancreatic Head and Body-Tail
Crippa S et al. Clin Gastroenterol Hepatol. 2022 Feb;20(2):390-399.e7. doi: 10.1016/j.cgh.2020.12.028.
Frequency of Bowel Movements and Risk of Diverticulitis
Jovani M et al. Clin Gastroenterol Hepatol. 2022 Feb;20(2):325-333.e5. doi: 10.1016/j.cgh.2021.01.003.
March 2022
AGA Clinical Practice Update on Management of Medically Refractory Gastroparesis: Expert Review
Lacy BE et al. Clin Gastroenterol Hepatol. 2022 Mar;20(3):491-500. doi: 10.1016/j.cgh.2021.10.038.
Efficacy and Safety of Tofacitinib in Ulcerative Colitis Based on Prior Tumor Necrosis Factor Inhibitor Failure Status
Sandborn WJ et al. Clin Gastroenterol Hepatol. 2022 Mar;20(3):591-601.e8. doi: 10.1016/j.cgh.2021.02.043.
April 2022
What Faculty and Fellows Should Know About Milestones 2.0
Donnangelo JL, Brijen SJ. Clin Gastroenterol Hepatol. 2022 Apr;20(4):720-722. doi: 10.1016/j.cgh.2021.12.017.
Patient Experience in the Gastrointestinal Endoscopy Unit
Day LW, Savides TJ. Clin Gastroenterol Hepatol. 2022 Apr;20(4):723-726. doi: 10.1016/j.cgh.2021.12.001.
Tailoring Surveillance Colonoscopy in Patients With Advanced Adenomas
Kahi CJ et al. Clin Gastroenterol Hepatol. 2022 Apr;20(4):847-854.e1. doi: 10.1016/j.cgh.2021.03.027.
Techniques and Innovations in Gastrointestinal Endoscopy
Primary CT Angiography Vs Colonoscopy in Acute Lower Gastrointestinal Hemorrhage
Lipcsey MS et al. Tech Innov Gastrointest Endosc. 2022 Jan 01;24(1):2-9. doi: 10.1016/j.tige.2021.11.004.
Cellular and Molecular Gastroenterology and Hepatology
The Role of Microbiota in Gastrointestinal Cancer and Cancer Treatment: Chance or Curse?
Smet A et al. Cell Mol Gastroenterol Hepatol. 2022;13(3):857-874. doi: 10.1016/j.jcmgh.2021.08.013.
Direct-to-Consumer Teledermatology Growth: A Review and Outlook for the Future
In recent years, direct-to-consumer (DTC) teledermatology platforms have gained popularity as telehealth business models, allowing patients to directly initiate visits with physicians and purchase medications from single platforms. A shortage of dermatologists, improved technology, drug patent expirations, and rising health care costs accelerated the growth of DTC dermatology.1 During the COVID-19 pandemic, teledermatology adoption surged due to the need to provide care while social distancing and minimizing viral exposure. These needs prompted additional federal funding and loosened regulatory provisions.2 As the userbase of these companies has grown, so have their valuations.3 Although the DTC model has attracted the attention of patients and investors, its rise provokes many questions about patients acting as consumers in health care. Indeed, DTC telemedicine offers greater autonomy and convenience for patients, but it may impact the quality of care and the nature of physician-patient relationships, perhaps making them more transactional.
Evolution of DTC in Health Care
The DTC model emphasizes individual choice and accessible health care. Although the definition has evolved, the core idea is not new.4 Over decades, pharmaceutical companies have spent billions of dollars on DTC advertising, circumventing physicians by directly reaching patients with campaigns on prescription drugs and laboratory tests and shaping public definitions of diseases.5
The DTC model of care is fundamentally different from traditional care models in that it changes the roles of the patient and physician. Whereas early telehealth models required a health care provider to initiate teleconsultations with specialists, DTC telemedicine bypasses this step (eg, the patient can consult a dermatologist without needing a primary care provider’s input first). This care can then be provided by dermatologists with whom patients may or may not have pre-established relationships.4,6
Dermatology was an early adopter of DTC telemedicine. The shortage of dermatologists in the United States created demand for increasing accessibility to dermatologic care. Additionally, the visual nature of diagnosing dermatologic disease was ideal for platforms supporting image sharing.7 Early DTC providers were primarily individual companies offering teledermatology. However, many dermatologists can now offer DTC capabilities via companies such as Amwell and Teladoc Health.8
Over the last 2 decades, start-ups such as Warby Parker (eyeglasses) and Casper (mattresses) defined the DTC industry using borrowed supply chains, cohesive branding, heavy social media marketing, and web-only retail. Scalability, lack of competition, and abundant venture capital created competition across numerous markets.9 Health care capitalized on this DTC model, creating a $700 billion market for products ranging from hearing aids to over-the-counter medications.10
Borrowing from this DTC playbook, platforms were created to offer delivery of generic prescription drugs to patients’ doorsteps. However, unlike with other products bought online, a consumer cannot simply add prescription drugs to their shopping cart and check out. In all models of American medical practice, physicians still serve as gatekeepers, providing a safeguard for patients to ensure appropriate prescription and avoid negative consequences of unnecessary drug use. This new model effectively streamlines diagnosis, prescription, and drug delivery without the patient ever having to leave home. Combining the prescribing and selling of medications (2 tasks that traditionally have been separated) potentially creates financial conflicts of interest (COIs). Additionally, high utilization of health care, including more prescriptions and visits, does not necessarily equal high quality of care. The companies stand to benefit from extra care regardless of need, and thus these models must be scrutinized for any incentives driving unnecessary care and prescriptions.
Ultimately, DTC has evolved to encompass multiple definitions in health care (Table 1). Although all models provide health care, each offers a different modality of delivery. The primary service may be the sale of prescription drugs or simply telemedicine visits. This review primarily discusses DTC pharmaceutical telemedicine platforms that sell private-label drugs and also offer telemedicine services to streamline care. However, the history, risks, and benefits discussed may apply to all models.
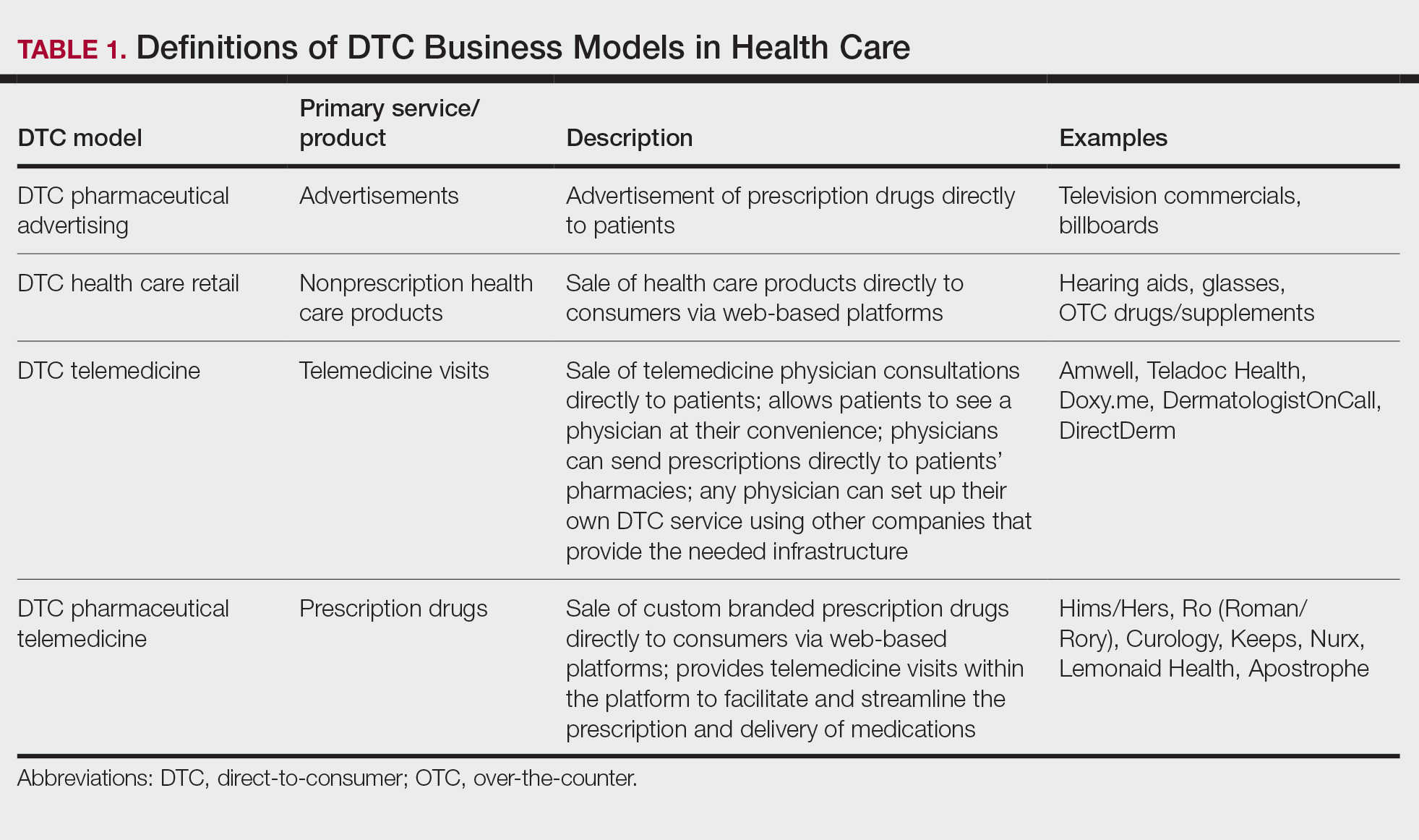
The DTC Landscape
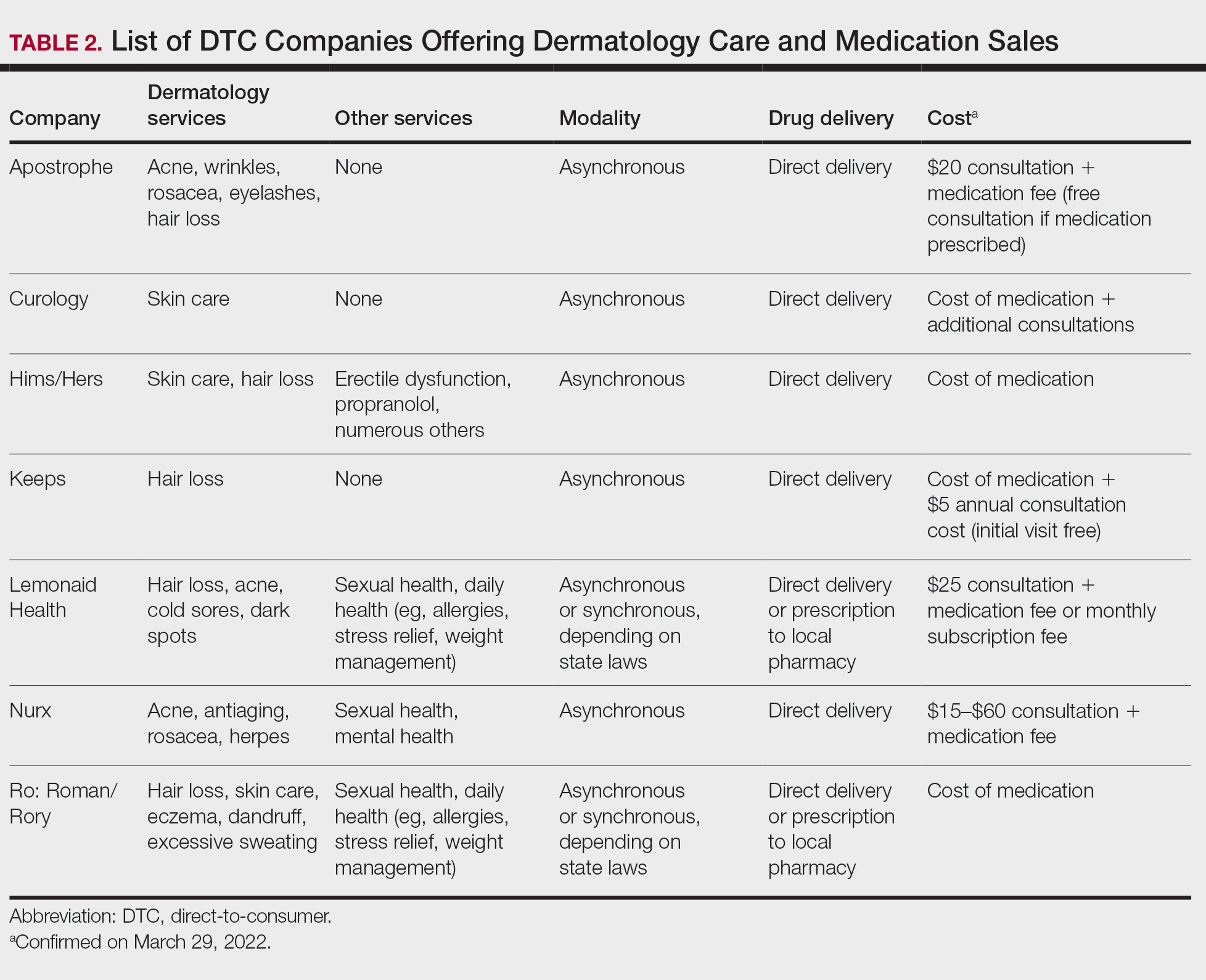
Most DTC companies employ variations on a model with the same 3 main components: a triage questionnaire, telehealth services, and prescription/drug delivery (Figure). The triage questionnaire elicits a history of the patient’s presentation and medical history. Some companies may use artificial intelligence (AI) algorithms to tailor questions to patient needs. There are 2 modalities for patient-provider communication: synchronous and asynchronous. Synchronous communication entails real-time patient-physician conversations via audio only or video call. Asynchronous (or store-and-forward) communication refers to consultations provided via messaging or text-based modality, where a provider may respond to a patient within 24 hours.6 Direct-to-consumer platforms primarily use asynchronous visits (Table 2). However, some also use synchronous modalities if the provider deems it necessary or if state laws require it.
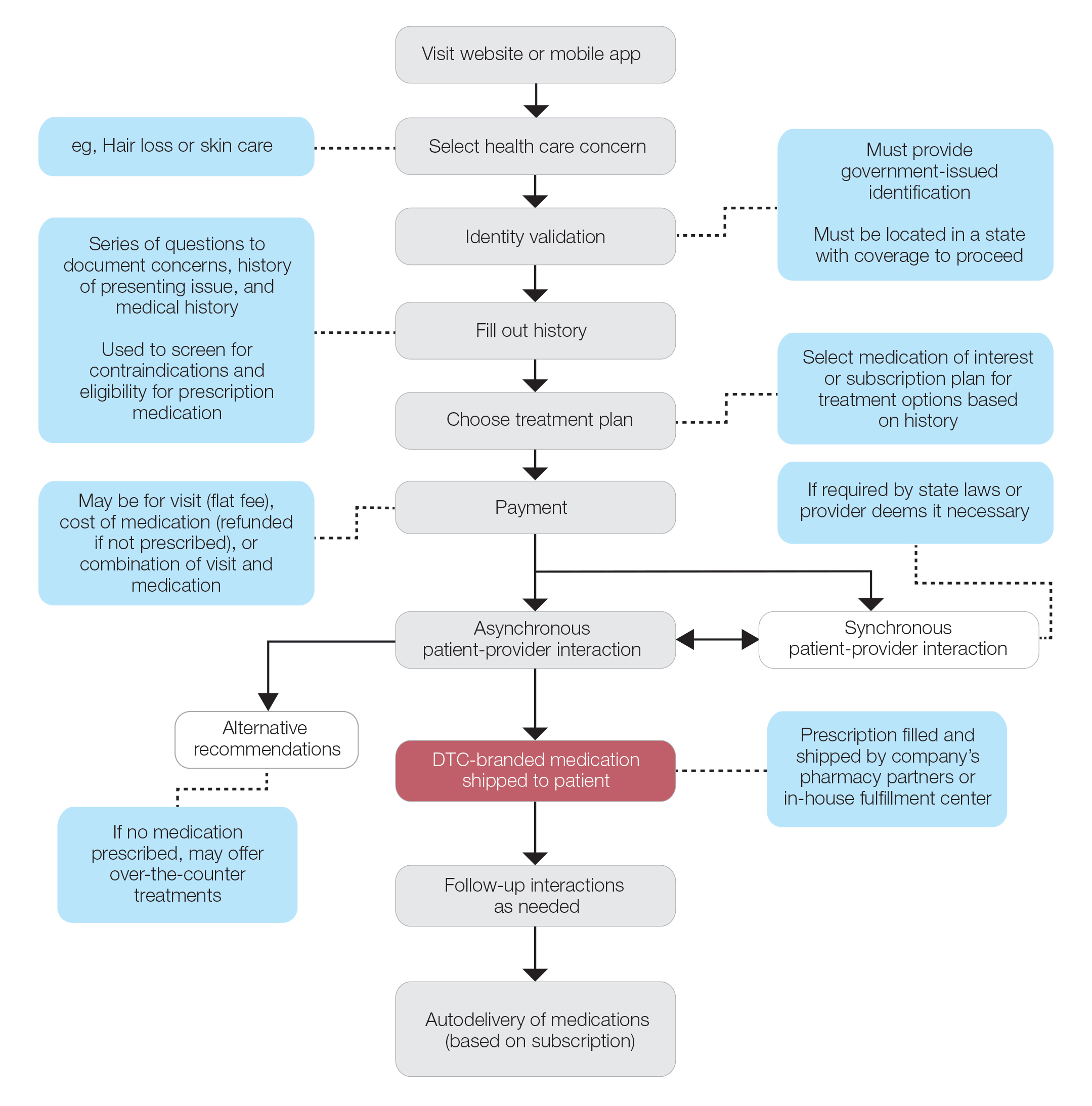
Once a provider has consulted with the patient, they can prescribe medication as needed. In certain cases, with adequate history, a prescription may be issued without a full physician visit. Furthermore, DTC companies require purchase of their custom-branded generic drugs. Prescriptions are fulfilled by the company’s pharmacy network and directly shipped to patients; few will allow patients to transfer a prescription to a pharmacy of their choice. Some platforms also sell supplements and over-the-counter medications.
Payment models vary among these companies, and most do not accept insurance (Table 2). Select models may provide free consultations and only require payment for pharmaceuticals. Others charge for consultations but reallocate payment to the cost of medication if prescribed. Another model involves flat rates for consultations and additional charges for drugs but unlimited messaging with providers for the duration of the prescription. Moreover, patients can subscribe to monthly deliveries of their medications.
Foundation of DTC
Technological advances have enabled patients to receive remote treatment from a single platform offering video calls, AI, electronic medical record interoperability, and integration of drug supply chains. Even in its simplest form, AI is increasingly used, as it allows for programs and chatbots to screen and triage patients.11 Technology also has improved at targeted mass marketing through social media platforms and search engines (eg, companies can use age, interests, location, and other parameters to target individuals likely needing acne treatment).
Drug patent expirations are a key catalyst for the rise of DTC companies, creating an attractive business model with generic drugs as the core product. Since 2008, patents for medications treating chronic conditions, such as erectile dysfunction, have expired. These patent expirations are responsible for $198 billion in projected prescription sales between 2019 and 2024.1 Thus, it follows that DTC companies have seized this opportunity to act as middlemen, taking advantage of these generic medications’ lower costs to create platforms focused on personalization and accessibility.
Rising deductibles have led patients to consider cheaper out-of-pocket alternatives that are not covered by insurance.1 For example, insurers typically do not cover finasteride treatment for conditions deemed cosmetic, such as androgenetic alopecia.12 The low cost of generic drugs creates an attractive business model for patients and investors. According to GoodRx, the average retail price for a 30-day supply of brand-name finasteride (Propecia [Merck]) is $135.92, whereas generic finasteride is $75.24.13 Direct-to-consumer pharmaceutical companies offer a 30-day supply of generic finasteride ranging from $8.33 to $30.14 The average wholesale cost for retailers is an estimated $2.31 for 30 days.15 Although profit margins on generic medications may be lower, more affordable drugs increase the size of the total market. These prescriptions are available as subscription plans, resulting in recurring revenue.
Lax US pharmaceutical marketing regulations allow direct advertising to the general public.16 In 1997, the US Food and Drug Administration allowed DTC advertisements to replace summaries of serious and common adverse effects with short statements covering important risks or referrals to other sources for complete information. In 2015, the US Food and Drug Administration guidelines preventing encouragement of self-diagnosis and self-treatment were withdrawn.5 These changes enable DTC companies to launch large advertising campaigns and to accelerate customer acquisition, as the industry often describes it, with ease.
Rapid Growth and Implications
Increasing generic drug availability and improving telemedicine capabilities have the potential to reduce costs and barriers but also have the potential for financial gain. Venture capital funds have recognized this opportunity, reflected by millions of dollars of investments, and accelerated the growth of DTC health care start-ups. For example, Ro has raised $376 million from venture capital, valuing the company at $1.5 billion.3
Direct-to-consumer companies require a heavy focus on marketing campaigns for customer acquisition. Their aesthetically pleasing websites and aggressive campaigns target specific audiences based on demographics, digital use habits, and purchasing behavior.4 Some campaigns celebrate the ease of obtaining prescriptions.17 Companies have been effective in recruiting so-called millennial and Generation Z patients, known to search the internet for remedies prior to seeking physician consultations.18 Recognizing these needs, some platforms offer guides on diseases they treat, creating effective customer-acquisition funnels. Recruitment of these technology-friendly patients has proven effective, especially given the largely positive media coverage of DTC platforms––potentially serving as a surrogate for medical credibility for patients.18
Some DTC companies also market physically; skin care ads may be strategically placed in social media feeds, or even found near mirrors in public bathrooms.19 Marketing campaigns also involve disease awareness; such efforts serve to increase diagnoses and prescribed treatments while destigmatizing diseases. Although DTC companies argue this strategy empowers patients, these marketing habits have the potential to take advantage of uninformed patients. Campaigns could potentially medicalize normal experiences and expand disease definitions resulting in overdiagnosis, overtreatment, and wasted resources.5 For example, off-label propranolol use has been advertised to attract patients who might have “nerves that come creeping before an important presentation.”17 Disease awareness campaigns also may lead people to falsely believe unproven drug benefits.5 According to studies, DTC pharmaceutical advertisements are low in informational quality and result in increased patient visits and prescriptions despite cost-effective alternatives.5,20-22
Fragmentation of the health care system is another possible complication of DTC teledermatology. These companies operate as for-profit organizations separated from the rest of the health care system, raising concerns about care coordination.8 Vital health data may not be conveyed as patients move among different providers and pharmacies. One study found DTC teledermatology rarely offered to provide medical records or facilitate a referral to a local physician.23 Such a lack of communication is concerning, as medication errors are the leading cause of avoidable harm in health care.24
Direct-to-consumer care models also seemingly redefine the physician-patient relationship by turning patients into consumers. Patient interactions may seem transactional and streamlined toward sales. For these platforms, a visit often is set up as an evaluation of a patient’s suitability for a prescription, not necessarily for the best treatment modality for the problem. These companies primarily make money through the sale of prescription drugs, creating a potential COI that may undermine the patient-physician relationship. Although some companies have made it clear that medical care and pharmaceutical sales are provided by legally separate business entities and that they do not pay physicians on commission, a conflict may still exist given the financial importance of physicians prescribing medication to the success of the business.16
Even as DTC models advertise upon expanded access and choice, the companies largely prohibit patients from choosing their own pharmacy. Instead, they encourage patients to fill prescriptions with the company’s pharmacy network by claiming lower costs compared with competitors. One DTC company, Hims, is launching a prescription-fulfillment center to further consolidate their business.17,19,25 The inherent COI of issuing and fulfilling prescriptions raises concerns of patient harm.26 For example, when Dermatology.com launched as a DTC prescription skin medication shop backed by Bausch Health Companies Inc, its model included telemedicine consultation. Although consultations were provided by RxDefine, a third party, only Dermatology.com drugs were prescribed. Given the poor quality of care and obvious financial COI, an uproar in the dermatology community and advocacy by the American Academy of Dermatology led to the shutdown of Dermatology.com’s online prescription services.26
The quality of care among DTC telemedicine platforms has been equivocal. Some studies have reported equivalent care in person and online, while others have reported poor adherence to guidelines, overuse of antibiotics, and misdiagnosis.8,23 A vital portion of the DTC experience is the history questionnaire, which is geared to diagnosis and risk assessment.25 Resneck et al23 found diagnostic quality to be adequate for simple dermatologic clinical scenarios but poor for scenarios requiring more than basic histories. Although Ro has reported leveraging data from millions of interactions to ask the right questions and streamline visits, it is still unclear whether history questionnaires are adequate.17,27 Additionally, consultations may lack sufficient counseling on adverse effects, risks, or pregnancy warnings, as well as discussions on alternative treatments and preventative care.17,23 Finally, patients often are limited in their choice of dermatologist; the lack of a fully developed relationship increases concerns of follow-up and monitoring practices. Although some DTC platforms offer unlimited interactions with physicians for the duration of a prescription, it is unknown how often these services are utilized or how adequate the quality of these interactions is. This potential for lax follow-up is especially concerning for prescriptions that autorenew on a monthly basis and could result in unnecessary overtreatment.
Postpandemic and Future Outlook
The COVID-19 pandemic dramatically impacted the use of telemedicine. To minimize COVID-19 transmission, the Centers for Medicare & Medicaid Services and private payers expanded telehealth coverage and eliminated reimbursement and licensing barriers.28 A decade’s worth of regulatory changes and consumer adoption was accelerated to weeks, resulting in telemedicine companies reaching record-high visit numbers.29 McKinsey & Company estimated that telehealth visit numbers surged 50- to 175-fold compared with pre–COVID-19 numbers. Additionally, 76% of patients were interested in future telehealth use, and 64% of providers were more comfortable using telehealth than before the pandemic.30 For their part, US dermatologists reported an increase in telemedicine use from 14.1% to 96.9% since COVID-19.31
Exactly how much DTC pharmaceutical telemedicine companies are growing is unclear, but private investments may be an indication. A record $14.7 billion was invested in the digital health sector in the first half of 2021; the majority went to telehealth companies.30 Ro, which reported $230 million in revenue in 2020 and has served 6 million visits, raised $200 milllion in July 2020 and $500 million in March 2021.32 Although post–COVID-19 health care will certainly involve increased telemedicine, the extent remains unclear, as telehealth vendors saw decreased usage upon reopening of state economies. Ultimately, the postpandemic regulatory landscape is hard to predict.30
Although COVID-19 appears to have caused rapid growth for DTC platforms, it also may have spurred competition. Telemedicine providers have given independent dermatologists and health care systems the infrastructure to implement custom DTC services.33 Although systems do not directly sell prescription drugs, the target market is essentially the same: patients looking for instant virtual dermatologic care. Therefore, sustained telemedicine services offered by traditional practices and systems may prove detrimental to DTC companies. However, unlike most telemedicine services, DTC models are less affected by certain changes in regulation since they do not rely on insurance. If regulations are tightened and reimbursements for telehealth are not attractive for dermatologists, teledermatology services may see an overall decrease. If so, patients who appreciate teledermatology may shift to using DTC platforms, even if their insurance does not cover them. Still, a nationwide survey found 56% of respondents felt an established relationship with a physician prior to a telemedicine visit is important, which may create a barrier for DTC adoption.34
Conclusion
Direct-to-consumer teledermatology represents a growing for-profit model of health care that provides patients with seemingly affordable and convenient care. However, there is potential for overtreatment, misdiagnosis, and fragmentation of health care. It will be important to monitor and evaluate the quality of care that DTC teledermatology offers and advocate for appropriate regulations and oversight. Eventually, more patients will have medications prescribed and dermatologic care administered through DTC companies. Dermatologists will benefit from this knowledge of DTC models to properly counsel patients on the risks and benefits of their use.
- Vennare J. The DTC healthcare report. Fitt Insider. September 15, 2019. Accessed February 23, 2022. https://insider.fitt.co/direct-to-consumer-healthcare-startups/
- Kannampallil T, Ma J. Digital translucence: adapting telemedicine delivery post-COVID-19. Telemed J E Health. 2020;26:1120-1122.
- Farr C. Ro, a 3-year-old online health provider, just raised a new round that values it at $1.5 billion. CNBC. July 27, 2020. Accessed February 23, 2022. https://www.cnbc.com/2020/07/27/ro-raises-200-million-at-1point5-billion-valuation-250-million-sales.html
- Elliott T, Shih J. Direct to consumer telemedicine. Curr Allergy Asthma Rep. 2019;19:1.
- Schwartz LM, Woloshin S. Medical marketing in the United States, 1997-2016. JAMA. 2019;321:80-96.
- Peart JM, Kovarik C. Direct-to-patient teledermatology practices. J Am Acad Dermatol. 2015;72:907-909.
- Coates SJ, Kvedar J, Granstein RD. Teledermatology: from historical perspective to emerging techniques of the modern era. J Am Acad Dermatol. 2015;72:563-574.
- Rheuban KS, Krupinski EA, eds. Understanding Telehealth. McGraw-Hill Education; 2017.
- Schlesinger LA, Higgins M, Roseman S. Reinventing the direct-to-consumer business model. Harvard Business Review. March 31, 2020. Accessed February 23, 2022. https://hbr.org/2020/03/reinventing-the-direct-to-consumer-business-model
- Cohen AB, Mathews SC, Dorsey ER, et al. Direct-to-consumer digital health. Lancet Digit Health. 2020;2:E163-E165.
- 6 telehealth trends for 2020. Wolters Kluwer. Published January 27, 2021. Accessed February 23, 2022. https://www.wolterskluwer.com/en/expert-insights/6-telehealth-trends-for-2020
- Jadoo SA, Lipoff JB. Prescribing to save patients money: ethical considerations. J Am Acad Dermatol. 2018;78:826-828.
- Propecia. GoodRx. Accessed February 23, 2022. https://www.goodrx.com/propecia
- Lauer A. The truth about online hair-loss treatments like Roman and Hims, according to a dermatologist. InsideHook. January 13, 2020. Accessed February 23, 2022. https://www.insidehook.com/article/grooming/men-hair-loss-treatments-dermatologist-review
- Friedman Y. Drug price trends for NDC 16729-0089. DrugPatentWatch. Accessed February 23, 2022. https://www.drugpatentwatch.com/p/drug-price/ndc/index.php?query=16729-0089
- Curtis H, Milner J. Ethical concerns with online direct-to-consumer pharmaceutical companies. J Med Ethics. 2020;46:168-171.
- Jain T, Lu RJ, Mehrotra A. Prescriptions on demand: the growth of direct-to-consumer telemedicine companies. JAMA. 2019;322:925-926.
- Shahinyan RH, Amighi A, Carey AN, et al. Direct-to-consumer internet prescription platforms overlook crucial pathology found during traditional office evaluation of young men with erectile dysfunction. Urology. 2020;143:165-172.
- Ali M. Andrew Dudum—bold strategies that propelled Hims & Hers into unicorn status. Exit Strategy with Moiz Ali. Published April 2020. Accessed February 23, 2022. https://open.spotify.com/episode/6DtaJxwZDjvZSJI88DTf24?si=b3FHQiUIQY62YjfRHmnJBQ
- Klara K, Kim J, Ross JS. Direct-to-consumer broadcast advertisements for pharmaceuticals: off-label promotion and adherence to FDA guidelines. J Gen Intern Med. 2018;33:651-658.
- Sullivan HW, Aikin KJ, Poehlman J. Communicating risk information in direct-to-consumer prescription drug television ads: a content analysis. Health Commun. 2019;34:212-219.
- Applequist J, Ball JG. An updated analysis of direct-to-consumer television advertisements for prescription drugs. Ann Fam Med. 2018;16:211-216.
- Resneck JS Jr, Abrouk M, Steuer M, et al. Choice, transparency, coordination, and quality among direct-to-consumer telemedicine websites and apps treating skin disease. JAMA Dermatol. 2016;152:768-775.
- Patient safety. World Health Organization. Published September 13, 2019. Accessed February 1, 2022. https://www.who.int/news-room/fact-sheets/detail/patient-safety
- Bollmeier SG, Stevenson E, Finnegan P, et al. Direct to consumer telemedicine: is healthcare from home best? Mo Med. 2020;117:303-309.
26. Court E. Bausch yanked online prescribing after dermatologist backlash. Bloomberg.com. Published March 11, 2020. Accessed September 25, 2020. https://www.bloomberg.com/news/articles/2020-03-11/bausch-yanked-online-prescribing-after-dermatologist-backlash
27. Reitano Z. The future of healthcare: how Ro helps providers treat patients 2 minutes, 2 days, 2 weeks, and 2 years at a time. Medium. Published March 4, 2019. Accessed February 1, 2022. https://medium.com/ro-co/the-future-of-healthcare-how-ro-helps-providers-treat-patients-2-mins-2-days-2-weeks-and-2-10efc0679d7
28. Lee I, Kovarik C, Tejasvi T, et al. Telehealth: helping your patients and practice survive and thrive during the COVID-19 crisis with rapid quality implementation. J Am Acad Dermatol. 2020;82:1213-1214.
29. Pifer R. “Weeks where decades happen”: telehealth 6 months into COVID-19. Healthcare Dive. Published July 27, 2020. Accessed February 23, 2022. https://www.healthcaredive.com/news/telehealth-6-months-coronavirus/581447/
30. Bestsennyy O, Gilbert G, Harris A, et al. Telehealth: a quarter-trillion-dollar post-COVID-19 reality? McKinsey & Company. Updated July 9, 2021. Accessed February 23, 2022. https://www.mckinsey.com/industries/healthcare-systems-and-services/our-insights/telehealth-a-quarter-trillion-dollar-post-covid-19-reality
31. Kennedy J, Arey S, Hopkins Z, et al. Dermatologist perceptions of teledermatology implementation and future use after COVID-19: demographics, barriers, and insights. JAMA Dermatol. 2021;157:595-597.
32. Jennings K. Digital health startup Ro raised $500 million at $5 billion valuation. Forbes. March 22, 2021. Accessed March 29, 2022. https://www.forbes.com/sites/katiejennings/2021/03/22/digital-health-startup-ro-raised-500-million-at-5-billion-valuation/?sh=695be0e462f5
33. Hollander JE, Carr BG. Virtually perfect? telemedicine for COVID-19. N Engl J Med. 2020;382:1679-1681.
34. Welch BM, Harvey J, O’Connell NS, et al. Patient preferences for direct-to-consumer telemedicine services: a nationwide survey. BMC Health Serv Res. 2017;17:784.
In recent years, direct-to-consumer (DTC) teledermatology platforms have gained popularity as telehealth business models, allowing patients to directly initiate visits with physicians and purchase medications from single platforms. A shortage of dermatologists, improved technology, drug patent expirations, and rising health care costs accelerated the growth of DTC dermatology.1 During the COVID-19 pandemic, teledermatology adoption surged due to the need to provide care while social distancing and minimizing viral exposure. These needs prompted additional federal funding and loosened regulatory provisions.2 As the userbase of these companies has grown, so have their valuations.3 Although the DTC model has attracted the attention of patients and investors, its rise provokes many questions about patients acting as consumers in health care. Indeed, DTC telemedicine offers greater autonomy and convenience for patients, but it may impact the quality of care and the nature of physician-patient relationships, perhaps making them more transactional.
Evolution of DTC in Health Care
The DTC model emphasizes individual choice and accessible health care. Although the definition has evolved, the core idea is not new.4 Over decades, pharmaceutical companies have spent billions of dollars on DTC advertising, circumventing physicians by directly reaching patients with campaigns on prescription drugs and laboratory tests and shaping public definitions of diseases.5
The DTC model of care is fundamentally different from traditional care models in that it changes the roles of the patient and physician. Whereas early telehealth models required a health care provider to initiate teleconsultations with specialists, DTC telemedicine bypasses this step (eg, the patient can consult a dermatologist without needing a primary care provider’s input first). This care can then be provided by dermatologists with whom patients may or may not have pre-established relationships.4,6
Dermatology was an early adopter of DTC telemedicine. The shortage of dermatologists in the United States created demand for increasing accessibility to dermatologic care. Additionally, the visual nature of diagnosing dermatologic disease was ideal for platforms supporting image sharing.7 Early DTC providers were primarily individual companies offering teledermatology. However, many dermatologists can now offer DTC capabilities via companies such as Amwell and Teladoc Health.8
Over the last 2 decades, start-ups such as Warby Parker (eyeglasses) and Casper (mattresses) defined the DTC industry using borrowed supply chains, cohesive branding, heavy social media marketing, and web-only retail. Scalability, lack of competition, and abundant venture capital created competition across numerous markets.9 Health care capitalized on this DTC model, creating a $700 billion market for products ranging from hearing aids to over-the-counter medications.10
Borrowing from this DTC playbook, platforms were created to offer delivery of generic prescription drugs to patients’ doorsteps. However, unlike with other products bought online, a consumer cannot simply add prescription drugs to their shopping cart and check out. In all models of American medical practice, physicians still serve as gatekeepers, providing a safeguard for patients to ensure appropriate prescription and avoid negative consequences of unnecessary drug use. This new model effectively streamlines diagnosis, prescription, and drug delivery without the patient ever having to leave home. Combining the prescribing and selling of medications (2 tasks that traditionally have been separated) potentially creates financial conflicts of interest (COIs). Additionally, high utilization of health care, including more prescriptions and visits, does not necessarily equal high quality of care. The companies stand to benefit from extra care regardless of need, and thus these models must be scrutinized for any incentives driving unnecessary care and prescriptions.
Ultimately, DTC has evolved to encompass multiple definitions in health care (Table 1). Although all models provide health care, each offers a different modality of delivery. The primary service may be the sale of prescription drugs or simply telemedicine visits. This review primarily discusses DTC pharmaceutical telemedicine platforms that sell private-label drugs and also offer telemedicine services to streamline care. However, the history, risks, and benefits discussed may apply to all models.

The DTC Landscape
Most DTC companies employ variations on a model with the same 3 main components: a triage questionnaire, telehealth services, and prescription/drug delivery (Figure). The triage questionnaire elicits a history of the patient’s presentation and medical history. Some companies may use artificial intelligence (AI) algorithms to tailor questions to patient needs. There are 2 modalities for patient-provider communication: synchronous and asynchronous. Synchronous communication entails real-time patient-physician conversations via audio only or video call. Asynchronous (or store-and-forward) communication refers to consultations provided via messaging or text-based modality, where a provider may respond to a patient within 24 hours.6 Direct-to-consumer platforms primarily use asynchronous visits (Table 2). However, some also use synchronous modalities if the provider deems it necessary or if state laws require it.

Once a provider has consulted with the patient, they can prescribe medication as needed. In certain cases, with adequate history, a prescription may be issued without a full physician visit. Furthermore, DTC companies require purchase of their custom-branded generic drugs. Prescriptions are fulfilled by the company’s pharmacy network and directly shipped to patients; few will allow patients to transfer a prescription to a pharmacy of their choice. Some platforms also sell supplements and over-the-counter medications.

Payment models vary among these companies, and most do not accept insurance (Table 2). Select models may provide free consultations and only require payment for pharmaceuticals. Others charge for consultations but reallocate payment to the cost of medication if prescribed. Another model involves flat rates for consultations and additional charges for drugs but unlimited messaging with providers for the duration of the prescription. Moreover, patients can subscribe to monthly deliveries of their medications.
Foundation of DTC
Technological advances have enabled patients to receive remote treatment from a single platform offering video calls, AI, electronic medical record interoperability, and integration of drug supply chains. Even in its simplest form, AI is increasingly used, as it allows for programs and chatbots to screen and triage patients.11 Technology also has improved at targeted mass marketing through social media platforms and search engines (eg, companies can use age, interests, location, and other parameters to target individuals likely needing acne treatment).
Drug patent expirations are a key catalyst for the rise of DTC companies, creating an attractive business model with generic drugs as the core product. Since 2008, patents for medications treating chronic conditions, such as erectile dysfunction, have expired. These patent expirations are responsible for $198 billion in projected prescription sales between 2019 and 2024.1 Thus, it follows that DTC companies have seized this opportunity to act as middlemen, taking advantage of these generic medications’ lower costs to create platforms focused on personalization and accessibility.
Rising deductibles have led patients to consider cheaper out-of-pocket alternatives that are not covered by insurance.1 For example, insurers typically do not cover finasteride treatment for conditions deemed cosmetic, such as androgenetic alopecia.12 The low cost of generic drugs creates an attractive business model for patients and investors. According to GoodRx, the average retail price for a 30-day supply of brand-name finasteride (Propecia [Merck]) is $135.92, whereas generic finasteride is $75.24.13 Direct-to-consumer pharmaceutical companies offer a 30-day supply of generic finasteride ranging from $8.33 to $30.14 The average wholesale cost for retailers is an estimated $2.31 for 30 days.15 Although profit margins on generic medications may be lower, more affordable drugs increase the size of the total market. These prescriptions are available as subscription plans, resulting in recurring revenue.
Lax US pharmaceutical marketing regulations allow direct advertising to the general public.16 In 1997, the US Food and Drug Administration allowed DTC advertisements to replace summaries of serious and common adverse effects with short statements covering important risks or referrals to other sources for complete information. In 2015, the US Food and Drug Administration guidelines preventing encouragement of self-diagnosis and self-treatment were withdrawn.5 These changes enable DTC companies to launch large advertising campaigns and to accelerate customer acquisition, as the industry often describes it, with ease.
Rapid Growth and Implications
Increasing generic drug availability and improving telemedicine capabilities have the potential to reduce costs and barriers but also have the potential for financial gain. Venture capital funds have recognized this opportunity, reflected by millions of dollars of investments, and accelerated the growth of DTC health care start-ups. For example, Ro has raised $376 million from venture capital, valuing the company at $1.5 billion.3
Direct-to-consumer companies require a heavy focus on marketing campaigns for customer acquisition. Their aesthetically pleasing websites and aggressive campaigns target specific audiences based on demographics, digital use habits, and purchasing behavior.4 Some campaigns celebrate the ease of obtaining prescriptions.17 Companies have been effective in recruiting so-called millennial and Generation Z patients, known to search the internet for remedies prior to seeking physician consultations.18 Recognizing these needs, some platforms offer guides on diseases they treat, creating effective customer-acquisition funnels. Recruitment of these technology-friendly patients has proven effective, especially given the largely positive media coverage of DTC platforms––potentially serving as a surrogate for medical credibility for patients.18
Some DTC companies also market physically; skin care ads may be strategically placed in social media feeds, or even found near mirrors in public bathrooms.19 Marketing campaigns also involve disease awareness; such efforts serve to increase diagnoses and prescribed treatments while destigmatizing diseases. Although DTC companies argue this strategy empowers patients, these marketing habits have the potential to take advantage of uninformed patients. Campaigns could potentially medicalize normal experiences and expand disease definitions resulting in overdiagnosis, overtreatment, and wasted resources.5 For example, off-label propranolol use has been advertised to attract patients who might have “nerves that come creeping before an important presentation.”17 Disease awareness campaigns also may lead people to falsely believe unproven drug benefits.5 According to studies, DTC pharmaceutical advertisements are low in informational quality and result in increased patient visits and prescriptions despite cost-effective alternatives.5,20-22
Fragmentation of the health care system is another possible complication of DTC teledermatology. These companies operate as for-profit organizations separated from the rest of the health care system, raising concerns about care coordination.8 Vital health data may not be conveyed as patients move among different providers and pharmacies. One study found DTC teledermatology rarely offered to provide medical records or facilitate a referral to a local physician.23 Such a lack of communication is concerning, as medication errors are the leading cause of avoidable harm in health care.24
Direct-to-consumer care models also seemingly redefine the physician-patient relationship by turning patients into consumers. Patient interactions may seem transactional and streamlined toward sales. For these platforms, a visit often is set up as an evaluation of a patient’s suitability for a prescription, not necessarily for the best treatment modality for the problem. These companies primarily make money through the sale of prescription drugs, creating a potential COI that may undermine the patient-physician relationship. Although some companies have made it clear that medical care and pharmaceutical sales are provided by legally separate business entities and that they do not pay physicians on commission, a conflict may still exist given the financial importance of physicians prescribing medication to the success of the business.16
Even as DTC models advertise upon expanded access and choice, the companies largely prohibit patients from choosing their own pharmacy. Instead, they encourage patients to fill prescriptions with the company’s pharmacy network by claiming lower costs compared with competitors. One DTC company, Hims, is launching a prescription-fulfillment center to further consolidate their business.17,19,25 The inherent COI of issuing and fulfilling prescriptions raises concerns of patient harm.26 For example, when Dermatology.com launched as a DTC prescription skin medication shop backed by Bausch Health Companies Inc, its model included telemedicine consultation. Although consultations were provided by RxDefine, a third party, only Dermatology.com drugs were prescribed. Given the poor quality of care and obvious financial COI, an uproar in the dermatology community and advocacy by the American Academy of Dermatology led to the shutdown of Dermatology.com’s online prescription services.26
The quality of care among DTC telemedicine platforms has been equivocal. Some studies have reported equivalent care in person and online, while others have reported poor adherence to guidelines, overuse of antibiotics, and misdiagnosis.8,23 A vital portion of the DTC experience is the history questionnaire, which is geared to diagnosis and risk assessment.25 Resneck et al23 found diagnostic quality to be adequate for simple dermatologic clinical scenarios but poor for scenarios requiring more than basic histories. Although Ro has reported leveraging data from millions of interactions to ask the right questions and streamline visits, it is still unclear whether history questionnaires are adequate.17,27 Additionally, consultations may lack sufficient counseling on adverse effects, risks, or pregnancy warnings, as well as discussions on alternative treatments and preventative care.17,23 Finally, patients often are limited in their choice of dermatologist; the lack of a fully developed relationship increases concerns of follow-up and monitoring practices. Although some DTC platforms offer unlimited interactions with physicians for the duration of a prescription, it is unknown how often these services are utilized or how adequate the quality of these interactions is. This potential for lax follow-up is especially concerning for prescriptions that autorenew on a monthly basis and could result in unnecessary overtreatment.
Postpandemic and Future Outlook
The COVID-19 pandemic dramatically impacted the use of telemedicine. To minimize COVID-19 transmission, the Centers for Medicare & Medicaid Services and private payers expanded telehealth coverage and eliminated reimbursement and licensing barriers.28 A decade’s worth of regulatory changes and consumer adoption was accelerated to weeks, resulting in telemedicine companies reaching record-high visit numbers.29 McKinsey & Company estimated that telehealth visit numbers surged 50- to 175-fold compared with pre–COVID-19 numbers. Additionally, 76% of patients were interested in future telehealth use, and 64% of providers were more comfortable using telehealth than before the pandemic.30 For their part, US dermatologists reported an increase in telemedicine use from 14.1% to 96.9% since COVID-19.31
Exactly how much DTC pharmaceutical telemedicine companies are growing is unclear, but private investments may be an indication. A record $14.7 billion was invested in the digital health sector in the first half of 2021; the majority went to telehealth companies.30 Ro, which reported $230 million in revenue in 2020 and has served 6 million visits, raised $200 milllion in July 2020 and $500 million in March 2021.32 Although post–COVID-19 health care will certainly involve increased telemedicine, the extent remains unclear, as telehealth vendors saw decreased usage upon reopening of state economies. Ultimately, the postpandemic regulatory landscape is hard to predict.30
Although COVID-19 appears to have caused rapid growth for DTC platforms, it also may have spurred competition. Telemedicine providers have given independent dermatologists and health care systems the infrastructure to implement custom DTC services.33 Although systems do not directly sell prescription drugs, the target market is essentially the same: patients looking for instant virtual dermatologic care. Therefore, sustained telemedicine services offered by traditional practices and systems may prove detrimental to DTC companies. However, unlike most telemedicine services, DTC models are less affected by certain changes in regulation since they do not rely on insurance. If regulations are tightened and reimbursements for telehealth are not attractive for dermatologists, teledermatology services may see an overall decrease. If so, patients who appreciate teledermatology may shift to using DTC platforms, even if their insurance does not cover them. Still, a nationwide survey found 56% of respondents felt an established relationship with a physician prior to a telemedicine visit is important, which may create a barrier for DTC adoption.34
Conclusion
Direct-to-consumer teledermatology represents a growing for-profit model of health care that provides patients with seemingly affordable and convenient care. However, there is potential for overtreatment, misdiagnosis, and fragmentation of health care. It will be important to monitor and evaluate the quality of care that DTC teledermatology offers and advocate for appropriate regulations and oversight. Eventually, more patients will have medications prescribed and dermatologic care administered through DTC companies. Dermatologists will benefit from this knowledge of DTC models to properly counsel patients on the risks and benefits of their use.
In recent years, direct-to-consumer (DTC) teledermatology platforms have gained popularity as telehealth business models, allowing patients to directly initiate visits with physicians and purchase medications from single platforms. A shortage of dermatologists, improved technology, drug patent expirations, and rising health care costs accelerated the growth of DTC dermatology.1 During the COVID-19 pandemic, teledermatology adoption surged due to the need to provide care while social distancing and minimizing viral exposure. These needs prompted additional federal funding and loosened regulatory provisions.2 As the userbase of these companies has grown, so have their valuations.3 Although the DTC model has attracted the attention of patients and investors, its rise provokes many questions about patients acting as consumers in health care. Indeed, DTC telemedicine offers greater autonomy and convenience for patients, but it may impact the quality of care and the nature of physician-patient relationships, perhaps making them more transactional.
Evolution of DTC in Health Care
The DTC model emphasizes individual choice and accessible health care. Although the definition has evolved, the core idea is not new.4 Over decades, pharmaceutical companies have spent billions of dollars on DTC advertising, circumventing physicians by directly reaching patients with campaigns on prescription drugs and laboratory tests and shaping public definitions of diseases.5
The DTC model of care is fundamentally different from traditional care models in that it changes the roles of the patient and physician. Whereas early telehealth models required a health care provider to initiate teleconsultations with specialists, DTC telemedicine bypasses this step (eg, the patient can consult a dermatologist without needing a primary care provider’s input first). This care can then be provided by dermatologists with whom patients may or may not have pre-established relationships.4,6
Dermatology was an early adopter of DTC telemedicine. The shortage of dermatologists in the United States created demand for increasing accessibility to dermatologic care. Additionally, the visual nature of diagnosing dermatologic disease was ideal for platforms supporting image sharing.7 Early DTC providers were primarily individual companies offering teledermatology. However, many dermatologists can now offer DTC capabilities via companies such as Amwell and Teladoc Health.8
Over the last 2 decades, start-ups such as Warby Parker (eyeglasses) and Casper (mattresses) defined the DTC industry using borrowed supply chains, cohesive branding, heavy social media marketing, and web-only retail. Scalability, lack of competition, and abundant venture capital created competition across numerous markets.9 Health care capitalized on this DTC model, creating a $700 billion market for products ranging from hearing aids to over-the-counter medications.10
Borrowing from this DTC playbook, platforms were created to offer delivery of generic prescription drugs to patients’ doorsteps. However, unlike with other products bought online, a consumer cannot simply add prescription drugs to their shopping cart and check out. In all models of American medical practice, physicians still serve as gatekeepers, providing a safeguard for patients to ensure appropriate prescription and avoid negative consequences of unnecessary drug use. This new model effectively streamlines diagnosis, prescription, and drug delivery without the patient ever having to leave home. Combining the prescribing and selling of medications (2 tasks that traditionally have been separated) potentially creates financial conflicts of interest (COIs). Additionally, high utilization of health care, including more prescriptions and visits, does not necessarily equal high quality of care. The companies stand to benefit from extra care regardless of need, and thus these models must be scrutinized for any incentives driving unnecessary care and prescriptions.
Ultimately, DTC has evolved to encompass multiple definitions in health care (Table 1). Although all models provide health care, each offers a different modality of delivery. The primary service may be the sale of prescription drugs or simply telemedicine visits. This review primarily discusses DTC pharmaceutical telemedicine platforms that sell private-label drugs and also offer telemedicine services to streamline care. However, the history, risks, and benefits discussed may apply to all models.

The DTC Landscape
Most DTC companies employ variations on a model with the same 3 main components: a triage questionnaire, telehealth services, and prescription/drug delivery (Figure). The triage questionnaire elicits a history of the patient’s presentation and medical history. Some companies may use artificial intelligence (AI) algorithms to tailor questions to patient needs. There are 2 modalities for patient-provider communication: synchronous and asynchronous. Synchronous communication entails real-time patient-physician conversations via audio only or video call. Asynchronous (or store-and-forward) communication refers to consultations provided via messaging or text-based modality, where a provider may respond to a patient within 24 hours.6 Direct-to-consumer platforms primarily use asynchronous visits (Table 2). However, some also use synchronous modalities if the provider deems it necessary or if state laws require it.

Once a provider has consulted with the patient, they can prescribe medication as needed. In certain cases, with adequate history, a prescription may be issued without a full physician visit. Furthermore, DTC companies require purchase of their custom-branded generic drugs. Prescriptions are fulfilled by the company’s pharmacy network and directly shipped to patients; few will allow patients to transfer a prescription to a pharmacy of their choice. Some platforms also sell supplements and over-the-counter medications.

Payment models vary among these companies, and most do not accept insurance (Table 2). Select models may provide free consultations and only require payment for pharmaceuticals. Others charge for consultations but reallocate payment to the cost of medication if prescribed. Another model involves flat rates for consultations and additional charges for drugs but unlimited messaging with providers for the duration of the prescription. Moreover, patients can subscribe to monthly deliveries of their medications.
Foundation of DTC
Technological advances have enabled patients to receive remote treatment from a single platform offering video calls, AI, electronic medical record interoperability, and integration of drug supply chains. Even in its simplest form, AI is increasingly used, as it allows for programs and chatbots to screen and triage patients.11 Technology also has improved at targeted mass marketing through social media platforms and search engines (eg, companies can use age, interests, location, and other parameters to target individuals likely needing acne treatment).
Drug patent expirations are a key catalyst for the rise of DTC companies, creating an attractive business model with generic drugs as the core product. Since 2008, patents for medications treating chronic conditions, such as erectile dysfunction, have expired. These patent expirations are responsible for $198 billion in projected prescription sales between 2019 and 2024.1 Thus, it follows that DTC companies have seized this opportunity to act as middlemen, taking advantage of these generic medications’ lower costs to create platforms focused on personalization and accessibility.
Rising deductibles have led patients to consider cheaper out-of-pocket alternatives that are not covered by insurance.1 For example, insurers typically do not cover finasteride treatment for conditions deemed cosmetic, such as androgenetic alopecia.12 The low cost of generic drugs creates an attractive business model for patients and investors. According to GoodRx, the average retail price for a 30-day supply of brand-name finasteride (Propecia [Merck]) is $135.92, whereas generic finasteride is $75.24.13 Direct-to-consumer pharmaceutical companies offer a 30-day supply of generic finasteride ranging from $8.33 to $30.14 The average wholesale cost for retailers is an estimated $2.31 for 30 days.15 Although profit margins on generic medications may be lower, more affordable drugs increase the size of the total market. These prescriptions are available as subscription plans, resulting in recurring revenue.
Lax US pharmaceutical marketing regulations allow direct advertising to the general public.16 In 1997, the US Food and Drug Administration allowed DTC advertisements to replace summaries of serious and common adverse effects with short statements covering important risks or referrals to other sources for complete information. In 2015, the US Food and Drug Administration guidelines preventing encouragement of self-diagnosis and self-treatment were withdrawn.5 These changes enable DTC companies to launch large advertising campaigns and to accelerate customer acquisition, as the industry often describes it, with ease.
Rapid Growth and Implications
Increasing generic drug availability and improving telemedicine capabilities have the potential to reduce costs and barriers but also have the potential for financial gain. Venture capital funds have recognized this opportunity, reflected by millions of dollars of investments, and accelerated the growth of DTC health care start-ups. For example, Ro has raised $376 million from venture capital, valuing the company at $1.5 billion.3
Direct-to-consumer companies require a heavy focus on marketing campaigns for customer acquisition. Their aesthetically pleasing websites and aggressive campaigns target specific audiences based on demographics, digital use habits, and purchasing behavior.4 Some campaigns celebrate the ease of obtaining prescriptions.17 Companies have been effective in recruiting so-called millennial and Generation Z patients, known to search the internet for remedies prior to seeking physician consultations.18 Recognizing these needs, some platforms offer guides on diseases they treat, creating effective customer-acquisition funnels. Recruitment of these technology-friendly patients has proven effective, especially given the largely positive media coverage of DTC platforms––potentially serving as a surrogate for medical credibility for patients.18
Some DTC companies also market physically; skin care ads may be strategically placed in social media feeds, or even found near mirrors in public bathrooms.19 Marketing campaigns also involve disease awareness; such efforts serve to increase diagnoses and prescribed treatments while destigmatizing diseases. Although DTC companies argue this strategy empowers patients, these marketing habits have the potential to take advantage of uninformed patients. Campaigns could potentially medicalize normal experiences and expand disease definitions resulting in overdiagnosis, overtreatment, and wasted resources.5 For example, off-label propranolol use has been advertised to attract patients who might have “nerves that come creeping before an important presentation.”17 Disease awareness campaigns also may lead people to falsely believe unproven drug benefits.5 According to studies, DTC pharmaceutical advertisements are low in informational quality and result in increased patient visits and prescriptions despite cost-effective alternatives.5,20-22
Fragmentation of the health care system is another possible complication of DTC teledermatology. These companies operate as for-profit organizations separated from the rest of the health care system, raising concerns about care coordination.8 Vital health data may not be conveyed as patients move among different providers and pharmacies. One study found DTC teledermatology rarely offered to provide medical records or facilitate a referral to a local physician.23 Such a lack of communication is concerning, as medication errors are the leading cause of avoidable harm in health care.24
Direct-to-consumer care models also seemingly redefine the physician-patient relationship by turning patients into consumers. Patient interactions may seem transactional and streamlined toward sales. For these platforms, a visit often is set up as an evaluation of a patient’s suitability for a prescription, not necessarily for the best treatment modality for the problem. These companies primarily make money through the sale of prescription drugs, creating a potential COI that may undermine the patient-physician relationship. Although some companies have made it clear that medical care and pharmaceutical sales are provided by legally separate business entities and that they do not pay physicians on commission, a conflict may still exist given the financial importance of physicians prescribing medication to the success of the business.16
Even as DTC models advertise upon expanded access and choice, the companies largely prohibit patients from choosing their own pharmacy. Instead, they encourage patients to fill prescriptions with the company’s pharmacy network by claiming lower costs compared with competitors. One DTC company, Hims, is launching a prescription-fulfillment center to further consolidate their business.17,19,25 The inherent COI of issuing and fulfilling prescriptions raises concerns of patient harm.26 For example, when Dermatology.com launched as a DTC prescription skin medication shop backed by Bausch Health Companies Inc, its model included telemedicine consultation. Although consultations were provided by RxDefine, a third party, only Dermatology.com drugs were prescribed. Given the poor quality of care and obvious financial COI, an uproar in the dermatology community and advocacy by the American Academy of Dermatology led to the shutdown of Dermatology.com’s online prescription services.26
The quality of care among DTC telemedicine platforms has been equivocal. Some studies have reported equivalent care in person and online, while others have reported poor adherence to guidelines, overuse of antibiotics, and misdiagnosis.8,23 A vital portion of the DTC experience is the history questionnaire, which is geared to diagnosis and risk assessment.25 Resneck et al23 found diagnostic quality to be adequate for simple dermatologic clinical scenarios but poor for scenarios requiring more than basic histories. Although Ro has reported leveraging data from millions of interactions to ask the right questions and streamline visits, it is still unclear whether history questionnaires are adequate.17,27 Additionally, consultations may lack sufficient counseling on adverse effects, risks, or pregnancy warnings, as well as discussions on alternative treatments and preventative care.17,23 Finally, patients often are limited in their choice of dermatologist; the lack of a fully developed relationship increases concerns of follow-up and monitoring practices. Although some DTC platforms offer unlimited interactions with physicians for the duration of a prescription, it is unknown how often these services are utilized or how adequate the quality of these interactions is. This potential for lax follow-up is especially concerning for prescriptions that autorenew on a monthly basis and could result in unnecessary overtreatment.
Postpandemic and Future Outlook
The COVID-19 pandemic dramatically impacted the use of telemedicine. To minimize COVID-19 transmission, the Centers for Medicare & Medicaid Services and private payers expanded telehealth coverage and eliminated reimbursement and licensing barriers.28 A decade’s worth of regulatory changes and consumer adoption was accelerated to weeks, resulting in telemedicine companies reaching record-high visit numbers.29 McKinsey & Company estimated that telehealth visit numbers surged 50- to 175-fold compared with pre–COVID-19 numbers. Additionally, 76% of patients were interested in future telehealth use, and 64% of providers were more comfortable using telehealth than before the pandemic.30 For their part, US dermatologists reported an increase in telemedicine use from 14.1% to 96.9% since COVID-19.31
Exactly how much DTC pharmaceutical telemedicine companies are growing is unclear, but private investments may be an indication. A record $14.7 billion was invested in the digital health sector in the first half of 2021; the majority went to telehealth companies.30 Ro, which reported $230 million in revenue in 2020 and has served 6 million visits, raised $200 milllion in July 2020 and $500 million in March 2021.32 Although post–COVID-19 health care will certainly involve increased telemedicine, the extent remains unclear, as telehealth vendors saw decreased usage upon reopening of state economies. Ultimately, the postpandemic regulatory landscape is hard to predict.30
Although COVID-19 appears to have caused rapid growth for DTC platforms, it also may have spurred competition. Telemedicine providers have given independent dermatologists and health care systems the infrastructure to implement custom DTC services.33 Although systems do not directly sell prescription drugs, the target market is essentially the same: patients looking for instant virtual dermatologic care. Therefore, sustained telemedicine services offered by traditional practices and systems may prove detrimental to DTC companies. However, unlike most telemedicine services, DTC models are less affected by certain changes in regulation since they do not rely on insurance. If regulations are tightened and reimbursements for telehealth are not attractive for dermatologists, teledermatology services may see an overall decrease. If so, patients who appreciate teledermatology may shift to using DTC platforms, even if their insurance does not cover them. Still, a nationwide survey found 56% of respondents felt an established relationship with a physician prior to a telemedicine visit is important, which may create a barrier for DTC adoption.34
Conclusion
Direct-to-consumer teledermatology represents a growing for-profit model of health care that provides patients with seemingly affordable and convenient care. However, there is potential for overtreatment, misdiagnosis, and fragmentation of health care. It will be important to monitor and evaluate the quality of care that DTC teledermatology offers and advocate for appropriate regulations and oversight. Eventually, more patients will have medications prescribed and dermatologic care administered through DTC companies. Dermatologists will benefit from this knowledge of DTC models to properly counsel patients on the risks and benefits of their use.
- Vennare J. The DTC healthcare report. Fitt Insider. September 15, 2019. Accessed February 23, 2022. https://insider.fitt.co/direct-to-consumer-healthcare-startups/
- Kannampallil T, Ma J. Digital translucence: adapting telemedicine delivery post-COVID-19. Telemed J E Health. 2020;26:1120-1122.
- Farr C. Ro, a 3-year-old online health provider, just raised a new round that values it at $1.5 billion. CNBC. July 27, 2020. Accessed February 23, 2022. https://www.cnbc.com/2020/07/27/ro-raises-200-million-at-1point5-billion-valuation-250-million-sales.html
- Elliott T, Shih J. Direct to consumer telemedicine. Curr Allergy Asthma Rep. 2019;19:1.
- Schwartz LM, Woloshin S. Medical marketing in the United States, 1997-2016. JAMA. 2019;321:80-96.
- Peart JM, Kovarik C. Direct-to-patient teledermatology practices. J Am Acad Dermatol. 2015;72:907-909.
- Coates SJ, Kvedar J, Granstein RD. Teledermatology: from historical perspective to emerging techniques of the modern era. J Am Acad Dermatol. 2015;72:563-574.
- Rheuban KS, Krupinski EA, eds. Understanding Telehealth. McGraw-Hill Education; 2017.
- Schlesinger LA, Higgins M, Roseman S. Reinventing the direct-to-consumer business model. Harvard Business Review. March 31, 2020. Accessed February 23, 2022. https://hbr.org/2020/03/reinventing-the-direct-to-consumer-business-model
- Cohen AB, Mathews SC, Dorsey ER, et al. Direct-to-consumer digital health. Lancet Digit Health. 2020;2:E163-E165.
- 6 telehealth trends for 2020. Wolters Kluwer. Published January 27, 2021. Accessed February 23, 2022. https://www.wolterskluwer.com/en/expert-insights/6-telehealth-trends-for-2020
- Jadoo SA, Lipoff JB. Prescribing to save patients money: ethical considerations. J Am Acad Dermatol. 2018;78:826-828.
- Propecia. GoodRx. Accessed February 23, 2022. https://www.goodrx.com/propecia
- Lauer A. The truth about online hair-loss treatments like Roman and Hims, according to a dermatologist. InsideHook. January 13, 2020. Accessed February 23, 2022. https://www.insidehook.com/article/grooming/men-hair-loss-treatments-dermatologist-review
- Friedman Y. Drug price trends for NDC 16729-0089. DrugPatentWatch. Accessed February 23, 2022. https://www.drugpatentwatch.com/p/drug-price/ndc/index.php?query=16729-0089
- Curtis H, Milner J. Ethical concerns with online direct-to-consumer pharmaceutical companies. J Med Ethics. 2020;46:168-171.
- Jain T, Lu RJ, Mehrotra A. Prescriptions on demand: the growth of direct-to-consumer telemedicine companies. JAMA. 2019;322:925-926.
- Shahinyan RH, Amighi A, Carey AN, et al. Direct-to-consumer internet prescription platforms overlook crucial pathology found during traditional office evaluation of young men with erectile dysfunction. Urology. 2020;143:165-172.
- Ali M. Andrew Dudum—bold strategies that propelled Hims & Hers into unicorn status. Exit Strategy with Moiz Ali. Published April 2020. Accessed February 23, 2022. https://open.spotify.com/episode/6DtaJxwZDjvZSJI88DTf24?si=b3FHQiUIQY62YjfRHmnJBQ
- Klara K, Kim J, Ross JS. Direct-to-consumer broadcast advertisements for pharmaceuticals: off-label promotion and adherence to FDA guidelines. J Gen Intern Med. 2018;33:651-658.
- Sullivan HW, Aikin KJ, Poehlman J. Communicating risk information in direct-to-consumer prescription drug television ads: a content analysis. Health Commun. 2019;34:212-219.
- Applequist J, Ball JG. An updated analysis of direct-to-consumer television advertisements for prescription drugs. Ann Fam Med. 2018;16:211-216.
- Resneck JS Jr, Abrouk M, Steuer M, et al. Choice, transparency, coordination, and quality among direct-to-consumer telemedicine websites and apps treating skin disease. JAMA Dermatol. 2016;152:768-775.
- Patient safety. World Health Organization. Published September 13, 2019. Accessed February 1, 2022. https://www.who.int/news-room/fact-sheets/detail/patient-safety
- Bollmeier SG, Stevenson E, Finnegan P, et al. Direct to consumer telemedicine: is healthcare from home best? Mo Med. 2020;117:303-309.
26. Court E. Bausch yanked online prescribing after dermatologist backlash. Bloomberg.com. Published March 11, 2020. Accessed September 25, 2020. https://www.bloomberg.com/news/articles/2020-03-11/bausch-yanked-online-prescribing-after-dermatologist-backlash
27. Reitano Z. The future of healthcare: how Ro helps providers treat patients 2 minutes, 2 days, 2 weeks, and 2 years at a time. Medium. Published March 4, 2019. Accessed February 1, 2022. https://medium.com/ro-co/the-future-of-healthcare-how-ro-helps-providers-treat-patients-2-mins-2-days-2-weeks-and-2-10efc0679d7
28. Lee I, Kovarik C, Tejasvi T, et al. Telehealth: helping your patients and practice survive and thrive during the COVID-19 crisis with rapid quality implementation. J Am Acad Dermatol. 2020;82:1213-1214.
29. Pifer R. “Weeks where decades happen”: telehealth 6 months into COVID-19. Healthcare Dive. Published July 27, 2020. Accessed February 23, 2022. https://www.healthcaredive.com/news/telehealth-6-months-coronavirus/581447/
30. Bestsennyy O, Gilbert G, Harris A, et al. Telehealth: a quarter-trillion-dollar post-COVID-19 reality? McKinsey & Company. Updated July 9, 2021. Accessed February 23, 2022. https://www.mckinsey.com/industries/healthcare-systems-and-services/our-insights/telehealth-a-quarter-trillion-dollar-post-covid-19-reality
31. Kennedy J, Arey S, Hopkins Z, et al. Dermatologist perceptions of teledermatology implementation and future use after COVID-19: demographics, barriers, and insights. JAMA Dermatol. 2021;157:595-597.
32. Jennings K. Digital health startup Ro raised $500 million at $5 billion valuation. Forbes. March 22, 2021. Accessed March 29, 2022. https://www.forbes.com/sites/katiejennings/2021/03/22/digital-health-startup-ro-raised-500-million-at-5-billion-valuation/?sh=695be0e462f5
33. Hollander JE, Carr BG. Virtually perfect? telemedicine for COVID-19. N Engl J Med. 2020;382:1679-1681.
34. Welch BM, Harvey J, O’Connell NS, et al. Patient preferences for direct-to-consumer telemedicine services: a nationwide survey. BMC Health Serv Res. 2017;17:784.
- Vennare J. The DTC healthcare report. Fitt Insider. September 15, 2019. Accessed February 23, 2022. https://insider.fitt.co/direct-to-consumer-healthcare-startups/
- Kannampallil T, Ma J. Digital translucence: adapting telemedicine delivery post-COVID-19. Telemed J E Health. 2020;26:1120-1122.
- Farr C. Ro, a 3-year-old online health provider, just raised a new round that values it at $1.5 billion. CNBC. July 27, 2020. Accessed February 23, 2022. https://www.cnbc.com/2020/07/27/ro-raises-200-million-at-1point5-billion-valuation-250-million-sales.html
- Elliott T, Shih J. Direct to consumer telemedicine. Curr Allergy Asthma Rep. 2019;19:1.
- Schwartz LM, Woloshin S. Medical marketing in the United States, 1997-2016. JAMA. 2019;321:80-96.
- Peart JM, Kovarik C. Direct-to-patient teledermatology practices. J Am Acad Dermatol. 2015;72:907-909.
- Coates SJ, Kvedar J, Granstein RD. Teledermatology: from historical perspective to emerging techniques of the modern era. J Am Acad Dermatol. 2015;72:563-574.
- Rheuban KS, Krupinski EA, eds. Understanding Telehealth. McGraw-Hill Education; 2017.
- Schlesinger LA, Higgins M, Roseman S. Reinventing the direct-to-consumer business model. Harvard Business Review. March 31, 2020. Accessed February 23, 2022. https://hbr.org/2020/03/reinventing-the-direct-to-consumer-business-model
- Cohen AB, Mathews SC, Dorsey ER, et al. Direct-to-consumer digital health. Lancet Digit Health. 2020;2:E163-E165.
- 6 telehealth trends for 2020. Wolters Kluwer. Published January 27, 2021. Accessed February 23, 2022. https://www.wolterskluwer.com/en/expert-insights/6-telehealth-trends-for-2020
- Jadoo SA, Lipoff JB. Prescribing to save patients money: ethical considerations. J Am Acad Dermatol. 2018;78:826-828.
- Propecia. GoodRx. Accessed February 23, 2022. https://www.goodrx.com/propecia
- Lauer A. The truth about online hair-loss treatments like Roman and Hims, according to a dermatologist. InsideHook. January 13, 2020. Accessed February 23, 2022. https://www.insidehook.com/article/grooming/men-hair-loss-treatments-dermatologist-review
- Friedman Y. Drug price trends for NDC 16729-0089. DrugPatentWatch. Accessed February 23, 2022. https://www.drugpatentwatch.com/p/drug-price/ndc/index.php?query=16729-0089
- Curtis H, Milner J. Ethical concerns with online direct-to-consumer pharmaceutical companies. J Med Ethics. 2020;46:168-171.
- Jain T, Lu RJ, Mehrotra A. Prescriptions on demand: the growth of direct-to-consumer telemedicine companies. JAMA. 2019;322:925-926.
- Shahinyan RH, Amighi A, Carey AN, et al. Direct-to-consumer internet prescription platforms overlook crucial pathology found during traditional office evaluation of young men with erectile dysfunction. Urology. 2020;143:165-172.
- Ali M. Andrew Dudum—bold strategies that propelled Hims & Hers into unicorn status. Exit Strategy with Moiz Ali. Published April 2020. Accessed February 23, 2022. https://open.spotify.com/episode/6DtaJxwZDjvZSJI88DTf24?si=b3FHQiUIQY62YjfRHmnJBQ
- Klara K, Kim J, Ross JS. Direct-to-consumer broadcast advertisements for pharmaceuticals: off-label promotion and adherence to FDA guidelines. J Gen Intern Med. 2018;33:651-658.
- Sullivan HW, Aikin KJ, Poehlman J. Communicating risk information in direct-to-consumer prescription drug television ads: a content analysis. Health Commun. 2019;34:212-219.
- Applequist J, Ball JG. An updated analysis of direct-to-consumer television advertisements for prescription drugs. Ann Fam Med. 2018;16:211-216.
- Resneck JS Jr, Abrouk M, Steuer M, et al. Choice, transparency, coordination, and quality among direct-to-consumer telemedicine websites and apps treating skin disease. JAMA Dermatol. 2016;152:768-775.
- Patient safety. World Health Organization. Published September 13, 2019. Accessed February 1, 2022. https://www.who.int/news-room/fact-sheets/detail/patient-safety
- Bollmeier SG, Stevenson E, Finnegan P, et al. Direct to consumer telemedicine: is healthcare from home best? Mo Med. 2020;117:303-309.
26. Court E. Bausch yanked online prescribing after dermatologist backlash. Bloomberg.com. Published March 11, 2020. Accessed September 25, 2020. https://www.bloomberg.com/news/articles/2020-03-11/bausch-yanked-online-prescribing-after-dermatologist-backlash
27. Reitano Z. The future of healthcare: how Ro helps providers treat patients 2 minutes, 2 days, 2 weeks, and 2 years at a time. Medium. Published March 4, 2019. Accessed February 1, 2022. https://medium.com/ro-co/the-future-of-healthcare-how-ro-helps-providers-treat-patients-2-mins-2-days-2-weeks-and-2-10efc0679d7
28. Lee I, Kovarik C, Tejasvi T, et al. Telehealth: helping your patients and practice survive and thrive during the COVID-19 crisis with rapid quality implementation. J Am Acad Dermatol. 2020;82:1213-1214.
29. Pifer R. “Weeks where decades happen”: telehealth 6 months into COVID-19. Healthcare Dive. Published July 27, 2020. Accessed February 23, 2022. https://www.healthcaredive.com/news/telehealth-6-months-coronavirus/581447/
30. Bestsennyy O, Gilbert G, Harris A, et al. Telehealth: a quarter-trillion-dollar post-COVID-19 reality? McKinsey & Company. Updated July 9, 2021. Accessed February 23, 2022. https://www.mckinsey.com/industries/healthcare-systems-and-services/our-insights/telehealth-a-quarter-trillion-dollar-post-covid-19-reality
31. Kennedy J, Arey S, Hopkins Z, et al. Dermatologist perceptions of teledermatology implementation and future use after COVID-19: demographics, barriers, and insights. JAMA Dermatol. 2021;157:595-597.
32. Jennings K. Digital health startup Ro raised $500 million at $5 billion valuation. Forbes. March 22, 2021. Accessed March 29, 2022. https://www.forbes.com/sites/katiejennings/2021/03/22/digital-health-startup-ro-raised-500-million-at-5-billion-valuation/?sh=695be0e462f5
33. Hollander JE, Carr BG. Virtually perfect? telemedicine for COVID-19. N Engl J Med. 2020;382:1679-1681.
34. Welch BM, Harvey J, O’Connell NS, et al. Patient preferences for direct-to-consumer telemedicine services: a nationwide survey. BMC Health Serv Res. 2017;17:784.
Practice Points
- Direct-to-consumer (DTC) teledermatology platforms are for-profit companies that provide telemedicine visits and sell prescription drugs directly to patients.
- Although they are growing in popularity, DTC teledermatology platforms may lead to overdiagnosis, overtreatment, and fragmentation of health care. Knowledge of teledermatology will be vital to counsel patients on the risks and benefits of these platforms.
Live-donor liver transplants for patients with CRC liver mets
These patients usually have a poor prognosis, and for many, palliative chemotherapy is the standard of care.
“For the first time, we have been able to demonstrate [outside of Norway] that liver transplantation for patients with unresectable liver metastases is feasible with good outcomes,” lead author Gonzalo Sapisochin, MD, PhD, an assistant professor of surgery at the University of Toronto, said in an interview.
“Furthermore, this is the first time we are able to prove that living donation may be a good strategy in this setting,” Dr. Sapisochin said of the series of 10 cases that they published in JAMA Surgery.
The series showed “excellent perioperative outcomes for both donors and recipients,” noted the authors of an accompanying commentary. They said the team “should be commended for adding liver-donor live transplantation to the armamentarium of surgical options for patients with CRC liver metastases.”
However, they express concern about the relatively short follow-up of 1.5 years and the “very high” recurrence rate of 30%.
Commenting in an interview, lead editorialist Shimul Shah, MD, an associate professor of surgery and the chief of solid organ transplantation at the University of Cincinnati, said: “I agree that overall survival is an important measure to look at, but it’s hard to look at overall survival with [1.5] years of follow-up.”
Other key areas of concern are the need for more standardized practices and for more data on how liver transplantation compares with patients who just continue to receive chemotherapy.
“I certainly think that there’s a role for liver transplantation in these patients, and I am a big fan of this,” Dr. Shah emphasized, noting that four patients at his own center have recently received liver transplants, including three from deceased donors.
“However, I just think that as a community, we need to be cautious and not get too excited too early,” he said. “We need to keep studying it and take it one step at a time.”
Moving from deceased to living donors
Nearly 70% of patients with CRC develop liver metastases, and when these are unresectable, the prognosis is poor, with 5-year survival rates of less than 10%.
The option of liver transplantation was first reported in 2015 by a group in Norway. Their study included 21 patients with CRC and unresectable liver tumors. They reported a striking improvement in overall survival at 5 years (56% vs. 9% among patients who started first-line chemotherapy).
But with shortages of donor livers, this approach has not caught on. Deceased-donor liver allografts are in short supply in most countries, and recent allocation changes have further shifted available organs away from patients with liver tumors.
An alternative is to use living donors. In a recent study, Dr. Sapisochin and colleagues showed viability and a survival advantage, compared with deceased-donor liver transplantation.
Building on that work, they established treatment protocols at three centers – the University of Rochester (N.Y.) Medical Center, the Cleveland Clinic, , and the University Health Network in Toronto.
Of 91 evaluated patients who were prospectively enrolled with liver-confined, unresectable CRC liver metastases, 10 met all inclusion criteria and received living-donor liver transplants between December 2017 and May 2021. The median age of the patients was 45 years; six were men, and four were women.
These patients all had primary tumors greater than stage T2 (six T3 and four T4b). Lymphovascular invasion was present in two patients, and perineural invasion was present in one patient.
The median time from diagnosis of the liver metastases to liver transplant was 1.7 years (range, 1.1-7.8 years).
At a median follow-up of 1.5 years (range, 0.4-2.9 years), recurrences occurred in three patients, with a rate of recurrence-free survival, using Kaplan-Meier estimates, of 62% and a rate of overall survival of 100%.
Rates of morbidity associated with transplantation were no higher than those observed in established standards for the donors or recipients, the authors noted.
Among transplant recipients, three patients had no Clavien-Dindo complications; three had grade II, and four had grade III complications. Among donors, five had no complications, four had grade I, and one had grade III complications.
All 10 donors were discharged from the hospital 4-7 days after surgery and recovered fully.
All three patients who experienced recurrences were treated with palliative chemotherapy. One died of disease after 3 months of treatment. As of the time of publication of the study, the other two had survived for 2 or more years following their live donor liver transplant.
Patient selection key
The authors are now investigating tumor subtypes, responses in CRC liver metastases, and other factors, with the aim of developing a novel screening method to identify appropriate candidates more quickly.
In the meantime, they emphasized that indicators of disease biology, such as the Oslo Score, the Clinical Risk Score, and sustained clinical response to systemic therapy, “remain the key filters through which to select patients who have sufficient opportunity for long-term cancer control, which is necessary to justify the risk to a living donor.”
Dr. Sapisochin reported receiving grants from Roche and Bayer and personal fees from Integra, Roche, AstraZeneca, and Novartis outside the submitted work. Dr. Shah disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
These patients usually have a poor prognosis, and for many, palliative chemotherapy is the standard of care.
“For the first time, we have been able to demonstrate [outside of Norway] that liver transplantation for patients with unresectable liver metastases is feasible with good outcomes,” lead author Gonzalo Sapisochin, MD, PhD, an assistant professor of surgery at the University of Toronto, said in an interview.
“Furthermore, this is the first time we are able to prove that living donation may be a good strategy in this setting,” Dr. Sapisochin said of the series of 10 cases that they published in JAMA Surgery.
The series showed “excellent perioperative outcomes for both donors and recipients,” noted the authors of an accompanying commentary. They said the team “should be commended for adding liver-donor live transplantation to the armamentarium of surgical options for patients with CRC liver metastases.”
However, they express concern about the relatively short follow-up of 1.5 years and the “very high” recurrence rate of 30%.
Commenting in an interview, lead editorialist Shimul Shah, MD, an associate professor of surgery and the chief of solid organ transplantation at the University of Cincinnati, said: “I agree that overall survival is an important measure to look at, but it’s hard to look at overall survival with [1.5] years of follow-up.”
Other key areas of concern are the need for more standardized practices and for more data on how liver transplantation compares with patients who just continue to receive chemotherapy.
“I certainly think that there’s a role for liver transplantation in these patients, and I am a big fan of this,” Dr. Shah emphasized, noting that four patients at his own center have recently received liver transplants, including three from deceased donors.
“However, I just think that as a community, we need to be cautious and not get too excited too early,” he said. “We need to keep studying it and take it one step at a time.”
Moving from deceased to living donors
Nearly 70% of patients with CRC develop liver metastases, and when these are unresectable, the prognosis is poor, with 5-year survival rates of less than 10%.
The option of liver transplantation was first reported in 2015 by a group in Norway. Their study included 21 patients with CRC and unresectable liver tumors. They reported a striking improvement in overall survival at 5 years (56% vs. 9% among patients who started first-line chemotherapy).
But with shortages of donor livers, this approach has not caught on. Deceased-donor liver allografts are in short supply in most countries, and recent allocation changes have further shifted available organs away from patients with liver tumors.
An alternative is to use living donors. In a recent study, Dr. Sapisochin and colleagues showed viability and a survival advantage, compared with deceased-donor liver transplantation.
Building on that work, they established treatment protocols at three centers – the University of Rochester (N.Y.) Medical Center, the Cleveland Clinic, , and the University Health Network in Toronto.
Of 91 evaluated patients who were prospectively enrolled with liver-confined, unresectable CRC liver metastases, 10 met all inclusion criteria and received living-donor liver transplants between December 2017 and May 2021. The median age of the patients was 45 years; six were men, and four were women.
These patients all had primary tumors greater than stage T2 (six T3 and four T4b). Lymphovascular invasion was present in two patients, and perineural invasion was present in one patient.
The median time from diagnosis of the liver metastases to liver transplant was 1.7 years (range, 1.1-7.8 years).
At a median follow-up of 1.5 years (range, 0.4-2.9 years), recurrences occurred in three patients, with a rate of recurrence-free survival, using Kaplan-Meier estimates, of 62% and a rate of overall survival of 100%.
Rates of morbidity associated with transplantation were no higher than those observed in established standards for the donors or recipients, the authors noted.
Among transplant recipients, three patients had no Clavien-Dindo complications; three had grade II, and four had grade III complications. Among donors, five had no complications, four had grade I, and one had grade III complications.
All 10 donors were discharged from the hospital 4-7 days after surgery and recovered fully.
All three patients who experienced recurrences were treated with palliative chemotherapy. One died of disease after 3 months of treatment. As of the time of publication of the study, the other two had survived for 2 or more years following their live donor liver transplant.
Patient selection key
The authors are now investigating tumor subtypes, responses in CRC liver metastases, and other factors, with the aim of developing a novel screening method to identify appropriate candidates more quickly.
In the meantime, they emphasized that indicators of disease biology, such as the Oslo Score, the Clinical Risk Score, and sustained clinical response to systemic therapy, “remain the key filters through which to select patients who have sufficient opportunity for long-term cancer control, which is necessary to justify the risk to a living donor.”
Dr. Sapisochin reported receiving grants from Roche and Bayer and personal fees from Integra, Roche, AstraZeneca, and Novartis outside the submitted work. Dr. Shah disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
These patients usually have a poor prognosis, and for many, palliative chemotherapy is the standard of care.
“For the first time, we have been able to demonstrate [outside of Norway] that liver transplantation for patients with unresectable liver metastases is feasible with good outcomes,” lead author Gonzalo Sapisochin, MD, PhD, an assistant professor of surgery at the University of Toronto, said in an interview.
“Furthermore, this is the first time we are able to prove that living donation may be a good strategy in this setting,” Dr. Sapisochin said of the series of 10 cases that they published in JAMA Surgery.
The series showed “excellent perioperative outcomes for both donors and recipients,” noted the authors of an accompanying commentary. They said the team “should be commended for adding liver-donor live transplantation to the armamentarium of surgical options for patients with CRC liver metastases.”
However, they express concern about the relatively short follow-up of 1.5 years and the “very high” recurrence rate of 30%.
Commenting in an interview, lead editorialist Shimul Shah, MD, an associate professor of surgery and the chief of solid organ transplantation at the University of Cincinnati, said: “I agree that overall survival is an important measure to look at, but it’s hard to look at overall survival with [1.5] years of follow-up.”
Other key areas of concern are the need for more standardized practices and for more data on how liver transplantation compares with patients who just continue to receive chemotherapy.
“I certainly think that there’s a role for liver transplantation in these patients, and I am a big fan of this,” Dr. Shah emphasized, noting that four patients at his own center have recently received liver transplants, including three from deceased donors.
“However, I just think that as a community, we need to be cautious and not get too excited too early,” he said. “We need to keep studying it and take it one step at a time.”
Moving from deceased to living donors
Nearly 70% of patients with CRC develop liver metastases, and when these are unresectable, the prognosis is poor, with 5-year survival rates of less than 10%.
The option of liver transplantation was first reported in 2015 by a group in Norway. Their study included 21 patients with CRC and unresectable liver tumors. They reported a striking improvement in overall survival at 5 years (56% vs. 9% among patients who started first-line chemotherapy).
But with shortages of donor livers, this approach has not caught on. Deceased-donor liver allografts are in short supply in most countries, and recent allocation changes have further shifted available organs away from patients with liver tumors.
An alternative is to use living donors. In a recent study, Dr. Sapisochin and colleagues showed viability and a survival advantage, compared with deceased-donor liver transplantation.
Building on that work, they established treatment protocols at three centers – the University of Rochester (N.Y.) Medical Center, the Cleveland Clinic, , and the University Health Network in Toronto.
Of 91 evaluated patients who were prospectively enrolled with liver-confined, unresectable CRC liver metastases, 10 met all inclusion criteria and received living-donor liver transplants between December 2017 and May 2021. The median age of the patients was 45 years; six were men, and four were women.
These patients all had primary tumors greater than stage T2 (six T3 and four T4b). Lymphovascular invasion was present in two patients, and perineural invasion was present in one patient.
The median time from diagnosis of the liver metastases to liver transplant was 1.7 years (range, 1.1-7.8 years).
At a median follow-up of 1.5 years (range, 0.4-2.9 years), recurrences occurred in three patients, with a rate of recurrence-free survival, using Kaplan-Meier estimates, of 62% and a rate of overall survival of 100%.
Rates of morbidity associated with transplantation were no higher than those observed in established standards for the donors or recipients, the authors noted.
Among transplant recipients, three patients had no Clavien-Dindo complications; three had grade II, and four had grade III complications. Among donors, five had no complications, four had grade I, and one had grade III complications.
All 10 donors were discharged from the hospital 4-7 days after surgery and recovered fully.
All three patients who experienced recurrences were treated with palliative chemotherapy. One died of disease after 3 months of treatment. As of the time of publication of the study, the other two had survived for 2 or more years following their live donor liver transplant.
Patient selection key
The authors are now investigating tumor subtypes, responses in CRC liver metastases, and other factors, with the aim of developing a novel screening method to identify appropriate candidates more quickly.
In the meantime, they emphasized that indicators of disease biology, such as the Oslo Score, the Clinical Risk Score, and sustained clinical response to systemic therapy, “remain the key filters through which to select patients who have sufficient opportunity for long-term cancer control, which is necessary to justify the risk to a living donor.”
Dr. Sapisochin reported receiving grants from Roche and Bayer and personal fees from Integra, Roche, AstraZeneca, and Novartis outside the submitted work. Dr. Shah disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA SURGERY
Acral Papulovesicular Eruption in a Soldier Following Smallpox Vaccination
Following the attacks of September 11, 2001, heightened concerns over bioterrorism and the potential use of smallpox as a biological weapon made smallpox vaccination a critical component of military readiness. Therefore, the US Military resumed its smallpox vaccination program in 2002 using the first-generation smallpox vaccine (Dryvax, Wyeth Pharmaceuticals), a live vaccinia virus vaccine created in the late 19th century. This vaccine was developed by pooling vaccinia strains from the skin of infected cows1 and had previously been used during the worldwide vaccination campaign in the 1970s. Dryvax was associated with various cardiac and cutaneous complications, from benign hypersensitivity reactions to life-threatening eczema vaccinatum and progressive vaccinia.
Due to concerns that the remaining supply of Dryvax was insufficient to vaccinate the US population in the case of a bioterrorism attack, investigators developed the second-generation smallpox vaccine (ACAM2000, Sanofi Pasteur Biologics Co) using advances in vaccine technology.2 ACAM2000 is a plaque-purified isolate of vaccinia virus propagated in cell culture, thereby reducing contaminants and lot-to-lot variation.1 Clinical trials demonstrated comparable immunogenicity and frequency of adverse events compared with Dryvax,2 and ACAM2000 replaced Dryvax in 2008. However, these trials focused on serious adverse events, such as cardiac complications and postvaccinal encephalitis, with less specific characterization and description of cutaneous eruptions.3
Since 2008, there have been few reports of cutaneous adverse reactions following vaccination with ACAM2000. Beachkofsky et al4 described 7 cases of papulovesicular eruptions and 1 case of generalized vaccinia. Freeman and Lenz5 described 4 cases of papulovesicular eruptions, and there has been 1 case of progressive vaccinia reported in a soldier with newly diagnosed acute myelogenous leukemia.6 Kramer7 described a patient with multiple vesiculopustular lesions secondary to autoinoculation. The distinct pruritic acral papulovesicular eruptions following ACAM2000 vaccination have occurred in healthy military service members at different locations since the introduction of ACAM2000. We describe an additional case of this unique cutaneous eruption, followed by a review of previously described cutaneous adverse events associated with smallpox vaccination.
Case Report
A 21-year-old female soldier who was otherwise healthy presented to the dermatology clinic with a pruritic papular eruption involving the upper and lower extremities of 1 week’s duration. The lesions first appeared 8 days after she received the ACAM2000 vaccine. She received no other concurrent vaccines, had no history of atopic dermatitis, and had no systemic symptoms. Physical examination revealed numerous erythematous indurated papules involving the dorsolateral hands and fingers, as well as the extensor surfaces of the elbows, knees, and thighs (Figures 1 and 2). Based on the clinical presentation, the differential diagnosis included lichen planus, verruca plana, dyshidrotic eczema, and smallpox vaccine reaction. Erythema multiforme was considered; however, the absence of palmoplantar involvement and typical targetoid lesions made this diagnosis less likely.
Biopsies of lesions on the arm and thigh were performed. Histologic findings revealed interface and spongiotic dermatitis with scattered necrotic keratinocytes and extravasated erythrocytes (Figure 3). There was no evidence of viral cytopathic effects. Similar clinical and histologic findings have been reported in the literature as acral papulovesicular eruptions following smallpox vaccination or papular spongiotic dermatitis of smallpox vaccination.8 The presence of eosinophils was not conspicuous in the current case and was only a notable finding in 1 of 2 cases previously described by Gaertner et al.8 This may simply be due to an idiosyncratic drug reaction. Furthermore, in the cases described by Beachkofsky et al,4 there were essentially 2 histologic groups. The first group demonstrated a dermal hypersensitivity-type reaction, and the second group demonstrated a lymphocytic capillaritis.
Based on these findings, the patient was diagnosed with an acral papulovesicular eruption following smallpox vaccination. Of note, the patient’s presentation was not consistent with other described smallpox vaccine reactions, which included eczema vaccinatum, autoinoculation, generalized vaccinia, and progressive vaccinia. The patient was treated supportively with triamcinolone acetonide cream 0.1%, cool compresses, and oral diphenhydramine as needed for pruritus. The lesions notably improved within the first week of treatment.
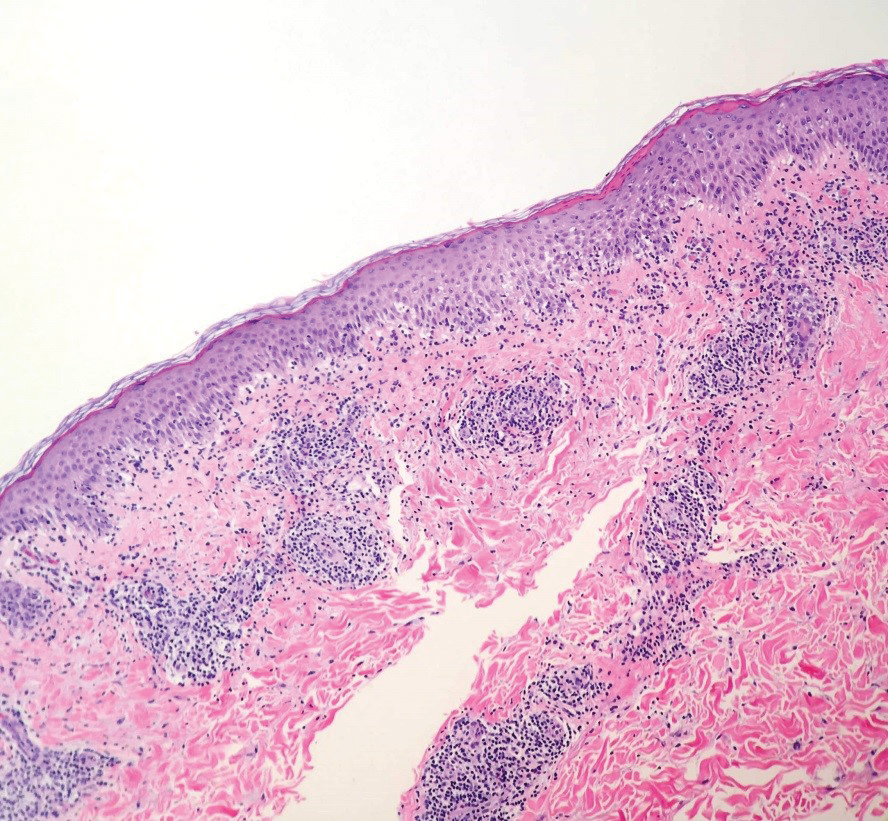
Comment
Reported cases of acral papulovesicular eruption4-6 demonstrated an onset of cutaneous symptoms an average of 14 days following vaccination (range, 8–18 days postvaccination). Lesions were benign and self-limited in all cases, with resolution within an average of 25 days (range, 7–71 days). All patients were active-duty military adults with a mean age of 24 years. Supportive treatment varied from topical steroids and oral antihistamines to tapering oral prednisone doses. Of note, all previously reported cases of this reaction occurred in patients who also had received other concurrent or near-concurrent vaccines, including anthrax, hepatitis B, influenza, and typhoid. Our patient represents a unique case of a papulovesicular eruption following smallpox vaccination with no history of concurrent vaccines.
Since the 1970s, smallpox vaccination has been associated with numerous cutaneous reactions, most of which have been reported with the first-generation Dryvax. Minor local reactions occurred in approximately 2% to 6% of vaccinees in clinical trials.9 These reactions included local edema involving the upper arm, satellite lesions within 2.5 cm of the vaccination site, local lymphadenopathy, intense inflammation or viral cellulitis surrounding the inoculation site, and viral lymphangitis tracking to axillary lymph nodes. In clinical trials, these reactions were self-limited and required only symptomatic treatment.9
Autoinoculation is another cutaneous reaction that can occur because Dryvax and ACAM2000 both contain live-attenuated replicating vaccinia virus. Accidental implantation may occur when the high titers of virus present at the vaccine site are subsequently transferred to other sites, especially abnormal mucosa or skin, resulting in an additional primary inoculation site.10
Eczema vaccinatum is a potentially life-threatening reaction that may occur in patients with disruptive skin disorders, such as atopic dermatitis. These patients are at risk for massive confluent vaccinia infection of the skin.10 In patients with atopic dermatitis, the virus rapidly disseminates due to both skin barrier dysfunction and impaired immunomodulation, resulting in large confluent skin lesions and the potential for viremia, septic shock, and death.10,11 Mortality from eczema vaccinatum may be reduced by administration of vaccinia immune globulin.10
The vaccinia virus also may spread hematogenously in healthy individuals,10 resulting in a benign reaction called generalized vaccinia. These patients develop pustules on areas of the skin other than the vaccination site. Although typically benign and self-limited, Beachkofsky et al4 described a case of generalized vaccinia in a healthy 34-year-old man resulting in a rapidly progressive vesiculopustular eruption with associated fever and pancytopenia. The patient made a complete recovery over the course of the following month.4
Alternatively, progressive vaccinia is a severe complication of smallpox vaccination seen in patients with impaired cell-mediated immunity. It also is known as vaccinia gangrenosum or vaccinia necrosum. These patients develop expanding ulcers due to exaggerated viral replication and cell-to-cell spread of the vaccinia virus.10,11 Hematogenous spread may result in viral implantation at distant sites of the body. This disease slowly progresses over weeks to months, and it often is resistant to treatment and fatal in patients with severe T-cell deficiency.10
Acral papulovesicular eruption is a distinct cutaneous adverse event following smallpox vaccination. Although further research is needed to discern the pathogenesis of this reaction, it is benign and self-limited, and patients have fully recovered with supportive care. In addition, a modified vaccinia Ankara vaccine (Bavarian Nordic) was approved by the US Food and Drug Administration in 2019.12,13 It is a nonreplicating attenuated viral vaccine that had fewer adverse events compared to ACAM2000 in clinical trials.13 To date, papulovesicular eruptions have not been reported following vaccination with the modified vaccinia Ankara vaccine; however, continued monitoring will help to further characterize any cutaneous reactions to this newer vaccine.
- Nalca A, Zumbrun EE. ACAM2000: the new smallpox vaccine for United States Strategic National Stockpile. Drug Des Devel Ther. 2010;4:71-79.
- Monath TP, Caldwell JR, Mundt W, et al. ACAM2000 clonal Vero cell culture vaccinia virus (New York City Board of Health strain)—a second-generation smallpox vaccine for biological defense. Int J Infect Dis. 2004;8:S31-S44.
- Thomas TN, Reef S, Neff L, et al. A review of the smallpox vaccine adverse events active surveillance system. Clin Infect Dis. 2008;46:S212-S220.
- Beachkofsky TM, Carrizales SC, Bidinger JJ, et al. Adverse events following smallpox vaccination with ACAM2000 in a military population. Arch Dermatol. 2010;146:656-661.
- Freeman R, Lenz B. Cutaneous reactions associated with ACAM2000 smallpox vaccination in a deploying U.S. Army unit. Mil Med. 2015;180:E152-E156.
- Centers for Disease Control and Prevention. Progressive vaccinia in a military smallpox vaccinee—United States, 2009. MMWR Morb Mortal Wkly Rep. 2009;58:532-536.
- Kramer TR. Post–smallpox vaccination skin eruption in a marine. Mil Med. 2018;183:E649-E653.
- Gaertner EM, Groo S, Kim J. Papular spongiotic dermatitis of smallpox vaccination: report of 2 cases with review of the literature. Arch Pathol Lab Med. 2004;128:1173-1175.
- Fulginiti VA, Papier A, Lane JM, et al. Smallpox vaccination: a review, part I. background, vaccination technique, normal vaccination and revaccination, and expected normal reactions. Clin Infect Dis. 2003;37:241-250.
- Fulginiti VA, Papier A, Lane JM, et al. Smallpox vaccination: a review, part II. adverse events. Clin Infect Dis. 2003;37:251-271.
- Bray M. Understanding smallpox vaccination. J Infect Dis. 2011;203:1037-1039.
- Greenberg RN, Hay CM, Stapleton JT, et al. A randomized, double-blind, placebo-controlled phase II trial investigating the safety and immunogenicity of modified vaccinia ankara smallpox vaccine (MVA-BN®) in 56-80-year-old subjects. PLoS One. 2016;11:E0157335.
- Pittman PR, Hahn M, Lee HS, et al. Phase 3 efficacy trial of modified vaccinia Ankara as a vaccine against smallpox. N Engl J Med. 2019;381:1897-1908.
Following the attacks of September 11, 2001, heightened concerns over bioterrorism and the potential use of smallpox as a biological weapon made smallpox vaccination a critical component of military readiness. Therefore, the US Military resumed its smallpox vaccination program in 2002 using the first-generation smallpox vaccine (Dryvax, Wyeth Pharmaceuticals), a live vaccinia virus vaccine created in the late 19th century. This vaccine was developed by pooling vaccinia strains from the skin of infected cows1 and had previously been used during the worldwide vaccination campaign in the 1970s. Dryvax was associated with various cardiac and cutaneous complications, from benign hypersensitivity reactions to life-threatening eczema vaccinatum and progressive vaccinia.
Due to concerns that the remaining supply of Dryvax was insufficient to vaccinate the US population in the case of a bioterrorism attack, investigators developed the second-generation smallpox vaccine (ACAM2000, Sanofi Pasteur Biologics Co) using advances in vaccine technology.2 ACAM2000 is a plaque-purified isolate of vaccinia virus propagated in cell culture, thereby reducing contaminants and lot-to-lot variation.1 Clinical trials demonstrated comparable immunogenicity and frequency of adverse events compared with Dryvax,2 and ACAM2000 replaced Dryvax in 2008. However, these trials focused on serious adverse events, such as cardiac complications and postvaccinal encephalitis, with less specific characterization and description of cutaneous eruptions.3
Since 2008, there have been few reports of cutaneous adverse reactions following vaccination with ACAM2000. Beachkofsky et al4 described 7 cases of papulovesicular eruptions and 1 case of generalized vaccinia. Freeman and Lenz5 described 4 cases of papulovesicular eruptions, and there has been 1 case of progressive vaccinia reported in a soldier with newly diagnosed acute myelogenous leukemia.6 Kramer7 described a patient with multiple vesiculopustular lesions secondary to autoinoculation. The distinct pruritic acral papulovesicular eruptions following ACAM2000 vaccination have occurred in healthy military service members at different locations since the introduction of ACAM2000. We describe an additional case of this unique cutaneous eruption, followed by a review of previously described cutaneous adverse events associated with smallpox vaccination.
Case Report
A 21-year-old female soldier who was otherwise healthy presented to the dermatology clinic with a pruritic papular eruption involving the upper and lower extremities of 1 week’s duration. The lesions first appeared 8 days after she received the ACAM2000 vaccine. She received no other concurrent vaccines, had no history of atopic dermatitis, and had no systemic symptoms. Physical examination revealed numerous erythematous indurated papules involving the dorsolateral hands and fingers, as well as the extensor surfaces of the elbows, knees, and thighs (Figures 1 and 2). Based on the clinical presentation, the differential diagnosis included lichen planus, verruca plana, dyshidrotic eczema, and smallpox vaccine reaction. Erythema multiforme was considered; however, the absence of palmoplantar involvement and typical targetoid lesions made this diagnosis less likely.

Biopsies of lesions on the arm and thigh were performed. Histologic findings revealed interface and spongiotic dermatitis with scattered necrotic keratinocytes and extravasated erythrocytes (Figure 3). There was no evidence of viral cytopathic effects. Similar clinical and histologic findings have been reported in the literature as acral papulovesicular eruptions following smallpox vaccination or papular spongiotic dermatitis of smallpox vaccination.8 The presence of eosinophils was not conspicuous in the current case and was only a notable finding in 1 of 2 cases previously described by Gaertner et al.8 This may simply be due to an idiosyncratic drug reaction. Furthermore, in the cases described by Beachkofsky et al,4 there were essentially 2 histologic groups. The first group demonstrated a dermal hypersensitivity-type reaction, and the second group demonstrated a lymphocytic capillaritis.

Based on these findings, the patient was diagnosed with an acral papulovesicular eruption following smallpox vaccination. Of note, the patient’s presentation was not consistent with other described smallpox vaccine reactions, which included eczema vaccinatum, autoinoculation, generalized vaccinia, and progressive vaccinia. The patient was treated supportively with triamcinolone acetonide cream 0.1%, cool compresses, and oral diphenhydramine as needed for pruritus. The lesions notably improved within the first week of treatment.

Comment
Reported cases of acral papulovesicular eruption4-6 demonstrated an onset of cutaneous symptoms an average of 14 days following vaccination (range, 8–18 days postvaccination). Lesions were benign and self-limited in all cases, with resolution within an average of 25 days (range, 7–71 days). All patients were active-duty military adults with a mean age of 24 years. Supportive treatment varied from topical steroids and oral antihistamines to tapering oral prednisone doses. Of note, all previously reported cases of this reaction occurred in patients who also had received other concurrent or near-concurrent vaccines, including anthrax, hepatitis B, influenza, and typhoid. Our patient represents a unique case of a papulovesicular eruption following smallpox vaccination with no history of concurrent vaccines.
Since the 1970s, smallpox vaccination has been associated with numerous cutaneous reactions, most of which have been reported with the first-generation Dryvax. Minor local reactions occurred in approximately 2% to 6% of vaccinees in clinical trials.9 These reactions included local edema involving the upper arm, satellite lesions within 2.5 cm of the vaccination site, local lymphadenopathy, intense inflammation or viral cellulitis surrounding the inoculation site, and viral lymphangitis tracking to axillary lymph nodes. In clinical trials, these reactions were self-limited and required only symptomatic treatment.9
Autoinoculation is another cutaneous reaction that can occur because Dryvax and ACAM2000 both contain live-attenuated replicating vaccinia virus. Accidental implantation may occur when the high titers of virus present at the vaccine site are subsequently transferred to other sites, especially abnormal mucosa or skin, resulting in an additional primary inoculation site.10
Eczema vaccinatum is a potentially life-threatening reaction that may occur in patients with disruptive skin disorders, such as atopic dermatitis. These patients are at risk for massive confluent vaccinia infection of the skin.10 In patients with atopic dermatitis, the virus rapidly disseminates due to both skin barrier dysfunction and impaired immunomodulation, resulting in large confluent skin lesions and the potential for viremia, septic shock, and death.10,11 Mortality from eczema vaccinatum may be reduced by administration of vaccinia immune globulin.10
The vaccinia virus also may spread hematogenously in healthy individuals,10 resulting in a benign reaction called generalized vaccinia. These patients develop pustules on areas of the skin other than the vaccination site. Although typically benign and self-limited, Beachkofsky et al4 described a case of generalized vaccinia in a healthy 34-year-old man resulting in a rapidly progressive vesiculopustular eruption with associated fever and pancytopenia. The patient made a complete recovery over the course of the following month.4
Alternatively, progressive vaccinia is a severe complication of smallpox vaccination seen in patients with impaired cell-mediated immunity. It also is known as vaccinia gangrenosum or vaccinia necrosum. These patients develop expanding ulcers due to exaggerated viral replication and cell-to-cell spread of the vaccinia virus.10,11 Hematogenous spread may result in viral implantation at distant sites of the body. This disease slowly progresses over weeks to months, and it often is resistant to treatment and fatal in patients with severe T-cell deficiency.10
Acral papulovesicular eruption is a distinct cutaneous adverse event following smallpox vaccination. Although further research is needed to discern the pathogenesis of this reaction, it is benign and self-limited, and patients have fully recovered with supportive care. In addition, a modified vaccinia Ankara vaccine (Bavarian Nordic) was approved by the US Food and Drug Administration in 2019.12,13 It is a nonreplicating attenuated viral vaccine that had fewer adverse events compared to ACAM2000 in clinical trials.13 To date, papulovesicular eruptions have not been reported following vaccination with the modified vaccinia Ankara vaccine; however, continued monitoring will help to further characterize any cutaneous reactions to this newer vaccine.
Following the attacks of September 11, 2001, heightened concerns over bioterrorism and the potential use of smallpox as a biological weapon made smallpox vaccination a critical component of military readiness. Therefore, the US Military resumed its smallpox vaccination program in 2002 using the first-generation smallpox vaccine (Dryvax, Wyeth Pharmaceuticals), a live vaccinia virus vaccine created in the late 19th century. This vaccine was developed by pooling vaccinia strains from the skin of infected cows1 and had previously been used during the worldwide vaccination campaign in the 1970s. Dryvax was associated with various cardiac and cutaneous complications, from benign hypersensitivity reactions to life-threatening eczema vaccinatum and progressive vaccinia.
Due to concerns that the remaining supply of Dryvax was insufficient to vaccinate the US population in the case of a bioterrorism attack, investigators developed the second-generation smallpox vaccine (ACAM2000, Sanofi Pasteur Biologics Co) using advances in vaccine technology.2 ACAM2000 is a plaque-purified isolate of vaccinia virus propagated in cell culture, thereby reducing contaminants and lot-to-lot variation.1 Clinical trials demonstrated comparable immunogenicity and frequency of adverse events compared with Dryvax,2 and ACAM2000 replaced Dryvax in 2008. However, these trials focused on serious adverse events, such as cardiac complications and postvaccinal encephalitis, with less specific characterization and description of cutaneous eruptions.3
Since 2008, there have been few reports of cutaneous adverse reactions following vaccination with ACAM2000. Beachkofsky et al4 described 7 cases of papulovesicular eruptions and 1 case of generalized vaccinia. Freeman and Lenz5 described 4 cases of papulovesicular eruptions, and there has been 1 case of progressive vaccinia reported in a soldier with newly diagnosed acute myelogenous leukemia.6 Kramer7 described a patient with multiple vesiculopustular lesions secondary to autoinoculation. The distinct pruritic acral papulovesicular eruptions following ACAM2000 vaccination have occurred in healthy military service members at different locations since the introduction of ACAM2000. We describe an additional case of this unique cutaneous eruption, followed by a review of previously described cutaneous adverse events associated with smallpox vaccination.
Case Report
A 21-year-old female soldier who was otherwise healthy presented to the dermatology clinic with a pruritic papular eruption involving the upper and lower extremities of 1 week’s duration. The lesions first appeared 8 days after she received the ACAM2000 vaccine. She received no other concurrent vaccines, had no history of atopic dermatitis, and had no systemic symptoms. Physical examination revealed numerous erythematous indurated papules involving the dorsolateral hands and fingers, as well as the extensor surfaces of the elbows, knees, and thighs (Figures 1 and 2). Based on the clinical presentation, the differential diagnosis included lichen planus, verruca plana, dyshidrotic eczema, and smallpox vaccine reaction. Erythema multiforme was considered; however, the absence of palmoplantar involvement and typical targetoid lesions made this diagnosis less likely.

Biopsies of lesions on the arm and thigh were performed. Histologic findings revealed interface and spongiotic dermatitis with scattered necrotic keratinocytes and extravasated erythrocytes (Figure 3). There was no evidence of viral cytopathic effects. Similar clinical and histologic findings have been reported in the literature as acral papulovesicular eruptions following smallpox vaccination or papular spongiotic dermatitis of smallpox vaccination.8 The presence of eosinophils was not conspicuous in the current case and was only a notable finding in 1 of 2 cases previously described by Gaertner et al.8 This may simply be due to an idiosyncratic drug reaction. Furthermore, in the cases described by Beachkofsky et al,4 there were essentially 2 histologic groups. The first group demonstrated a dermal hypersensitivity-type reaction, and the second group demonstrated a lymphocytic capillaritis.

Based on these findings, the patient was diagnosed with an acral papulovesicular eruption following smallpox vaccination. Of note, the patient’s presentation was not consistent with other described smallpox vaccine reactions, which included eczema vaccinatum, autoinoculation, generalized vaccinia, and progressive vaccinia. The patient was treated supportively with triamcinolone acetonide cream 0.1%, cool compresses, and oral diphenhydramine as needed for pruritus. The lesions notably improved within the first week of treatment.

Comment
Reported cases of acral papulovesicular eruption4-6 demonstrated an onset of cutaneous symptoms an average of 14 days following vaccination (range, 8–18 days postvaccination). Lesions were benign and self-limited in all cases, with resolution within an average of 25 days (range, 7–71 days). All patients were active-duty military adults with a mean age of 24 years. Supportive treatment varied from topical steroids and oral antihistamines to tapering oral prednisone doses. Of note, all previously reported cases of this reaction occurred in patients who also had received other concurrent or near-concurrent vaccines, including anthrax, hepatitis B, influenza, and typhoid. Our patient represents a unique case of a papulovesicular eruption following smallpox vaccination with no history of concurrent vaccines.
Since the 1970s, smallpox vaccination has been associated with numerous cutaneous reactions, most of which have been reported with the first-generation Dryvax. Minor local reactions occurred in approximately 2% to 6% of vaccinees in clinical trials.9 These reactions included local edema involving the upper arm, satellite lesions within 2.5 cm of the vaccination site, local lymphadenopathy, intense inflammation or viral cellulitis surrounding the inoculation site, and viral lymphangitis tracking to axillary lymph nodes. In clinical trials, these reactions were self-limited and required only symptomatic treatment.9
Autoinoculation is another cutaneous reaction that can occur because Dryvax and ACAM2000 both contain live-attenuated replicating vaccinia virus. Accidental implantation may occur when the high titers of virus present at the vaccine site are subsequently transferred to other sites, especially abnormal mucosa or skin, resulting in an additional primary inoculation site.10
Eczema vaccinatum is a potentially life-threatening reaction that may occur in patients with disruptive skin disorders, such as atopic dermatitis. These patients are at risk for massive confluent vaccinia infection of the skin.10 In patients with atopic dermatitis, the virus rapidly disseminates due to both skin barrier dysfunction and impaired immunomodulation, resulting in large confluent skin lesions and the potential for viremia, septic shock, and death.10,11 Mortality from eczema vaccinatum may be reduced by administration of vaccinia immune globulin.10
The vaccinia virus also may spread hematogenously in healthy individuals,10 resulting in a benign reaction called generalized vaccinia. These patients develop pustules on areas of the skin other than the vaccination site. Although typically benign and self-limited, Beachkofsky et al4 described a case of generalized vaccinia in a healthy 34-year-old man resulting in a rapidly progressive vesiculopustular eruption with associated fever and pancytopenia. The patient made a complete recovery over the course of the following month.4
Alternatively, progressive vaccinia is a severe complication of smallpox vaccination seen in patients with impaired cell-mediated immunity. It also is known as vaccinia gangrenosum or vaccinia necrosum. These patients develop expanding ulcers due to exaggerated viral replication and cell-to-cell spread of the vaccinia virus.10,11 Hematogenous spread may result in viral implantation at distant sites of the body. This disease slowly progresses over weeks to months, and it often is resistant to treatment and fatal in patients with severe T-cell deficiency.10
Acral papulovesicular eruption is a distinct cutaneous adverse event following smallpox vaccination. Although further research is needed to discern the pathogenesis of this reaction, it is benign and self-limited, and patients have fully recovered with supportive care. In addition, a modified vaccinia Ankara vaccine (Bavarian Nordic) was approved by the US Food and Drug Administration in 2019.12,13 It is a nonreplicating attenuated viral vaccine that had fewer adverse events compared to ACAM2000 in clinical trials.13 To date, papulovesicular eruptions have not been reported following vaccination with the modified vaccinia Ankara vaccine; however, continued monitoring will help to further characterize any cutaneous reactions to this newer vaccine.
- Nalca A, Zumbrun EE. ACAM2000: the new smallpox vaccine for United States Strategic National Stockpile. Drug Des Devel Ther. 2010;4:71-79.
- Monath TP, Caldwell JR, Mundt W, et al. ACAM2000 clonal Vero cell culture vaccinia virus (New York City Board of Health strain)—a second-generation smallpox vaccine for biological defense. Int J Infect Dis. 2004;8:S31-S44.
- Thomas TN, Reef S, Neff L, et al. A review of the smallpox vaccine adverse events active surveillance system. Clin Infect Dis. 2008;46:S212-S220.
- Beachkofsky TM, Carrizales SC, Bidinger JJ, et al. Adverse events following smallpox vaccination with ACAM2000 in a military population. Arch Dermatol. 2010;146:656-661.
- Freeman R, Lenz B. Cutaneous reactions associated with ACAM2000 smallpox vaccination in a deploying U.S. Army unit. Mil Med. 2015;180:E152-E156.
- Centers for Disease Control and Prevention. Progressive vaccinia in a military smallpox vaccinee—United States, 2009. MMWR Morb Mortal Wkly Rep. 2009;58:532-536.
- Kramer TR. Post–smallpox vaccination skin eruption in a marine. Mil Med. 2018;183:E649-E653.
- Gaertner EM, Groo S, Kim J. Papular spongiotic dermatitis of smallpox vaccination: report of 2 cases with review of the literature. Arch Pathol Lab Med. 2004;128:1173-1175.
- Fulginiti VA, Papier A, Lane JM, et al. Smallpox vaccination: a review, part I. background, vaccination technique, normal vaccination and revaccination, and expected normal reactions. Clin Infect Dis. 2003;37:241-250.
- Fulginiti VA, Papier A, Lane JM, et al. Smallpox vaccination: a review, part II. adverse events. Clin Infect Dis. 2003;37:251-271.
- Bray M. Understanding smallpox vaccination. J Infect Dis. 2011;203:1037-1039.
- Greenberg RN, Hay CM, Stapleton JT, et al. A randomized, double-blind, placebo-controlled phase II trial investigating the safety and immunogenicity of modified vaccinia ankara smallpox vaccine (MVA-BN®) in 56-80-year-old subjects. PLoS One. 2016;11:E0157335.
- Pittman PR, Hahn M, Lee HS, et al. Phase 3 efficacy trial of modified vaccinia Ankara as a vaccine against smallpox. N Engl J Med. 2019;381:1897-1908.
- Nalca A, Zumbrun EE. ACAM2000: the new smallpox vaccine for United States Strategic National Stockpile. Drug Des Devel Ther. 2010;4:71-79.
- Monath TP, Caldwell JR, Mundt W, et al. ACAM2000 clonal Vero cell culture vaccinia virus (New York City Board of Health strain)—a second-generation smallpox vaccine for biological defense. Int J Infect Dis. 2004;8:S31-S44.
- Thomas TN, Reef S, Neff L, et al. A review of the smallpox vaccine adverse events active surveillance system. Clin Infect Dis. 2008;46:S212-S220.
- Beachkofsky TM, Carrizales SC, Bidinger JJ, et al. Adverse events following smallpox vaccination with ACAM2000 in a military population. Arch Dermatol. 2010;146:656-661.
- Freeman R, Lenz B. Cutaneous reactions associated with ACAM2000 smallpox vaccination in a deploying U.S. Army unit. Mil Med. 2015;180:E152-E156.
- Centers for Disease Control and Prevention. Progressive vaccinia in a military smallpox vaccinee—United States, 2009. MMWR Morb Mortal Wkly Rep. 2009;58:532-536.
- Kramer TR. Post–smallpox vaccination skin eruption in a marine. Mil Med. 2018;183:E649-E653.
- Gaertner EM, Groo S, Kim J. Papular spongiotic dermatitis of smallpox vaccination: report of 2 cases with review of the literature. Arch Pathol Lab Med. 2004;128:1173-1175.
- Fulginiti VA, Papier A, Lane JM, et al. Smallpox vaccination: a review, part I. background, vaccination technique, normal vaccination and revaccination, and expected normal reactions. Clin Infect Dis. 2003;37:241-250.
- Fulginiti VA, Papier A, Lane JM, et al. Smallpox vaccination: a review, part II. adverse events. Clin Infect Dis. 2003;37:251-271.
- Bray M. Understanding smallpox vaccination. J Infect Dis. 2011;203:1037-1039.
- Greenberg RN, Hay CM, Stapleton JT, et al. A randomized, double-blind, placebo-controlled phase II trial investigating the safety and immunogenicity of modified vaccinia ankara smallpox vaccine (MVA-BN®) in 56-80-year-old subjects. PLoS One. 2016;11:E0157335.
- Pittman PR, Hahn M, Lee HS, et al. Phase 3 efficacy trial of modified vaccinia Ankara as a vaccine against smallpox. N Engl J Med. 2019;381:1897-1908.
Practice Points
- There are several potential cutaneous adverse reactions associated with smallpox vaccination, ranging from benign self-limited hypersensitivity reactions to life-threatening eczema vaccinatum and progressive vaccinia.
- Acral papulovesicular eruption is a distinct presentation that has been described in the US Military following vaccination with the second-generation live smallpox vaccine (ACAM2000).
Vesicular Eruption Secondary to Bites by Larval Amblyomma americanum
Case Report
A 58-year-old woman presented to the dermatology office with a widespread pruritic eruption of 3 days’ duration that started in the groin and spread to the rest of the body. No treatments had been attempted. She had no notable medical history, and she denied any recent illness, change in personal care products, or new medications or supplements. She reported a camping trip 2 weeks prior to presentation on the east end of Long Island, New York. She later learned that others on the same trip developed a similar, albeit less widespread, eruption.
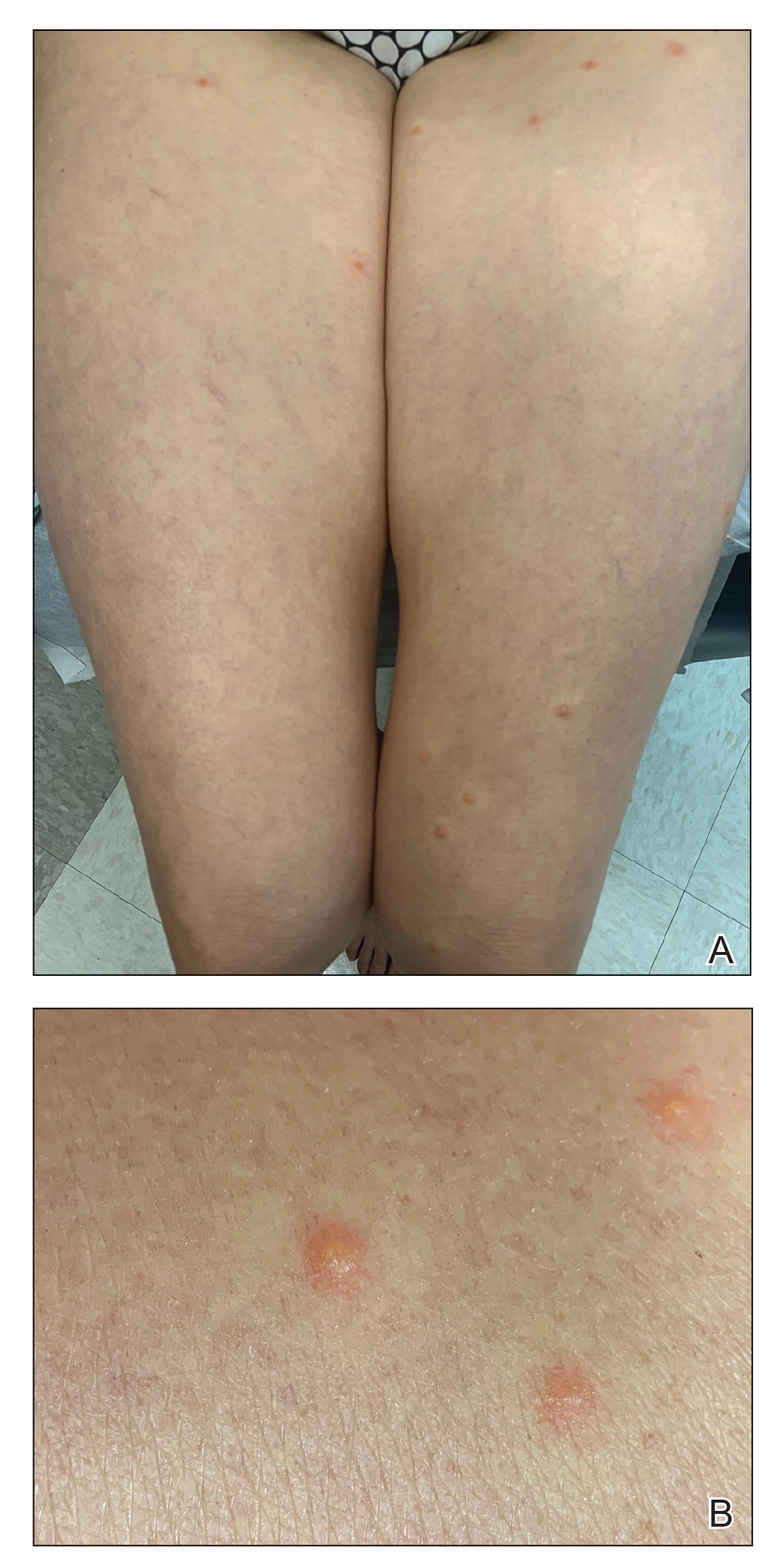
Physical examination revealed clear vesicles on the arms, legs, trunk, and pubic area (Figure 1). Dermoscopy revealed a small lone star tick larva in the center of one of the vesicles (Figure 2). The type of tick larva was identified using resources from the Centers for Disease Control and Prevention (Figure 3).1 Careful inspection revealed dark marks on various vesicles, mostly in the perineum, yielding nearly 20 larvae, which were removed with forceps. The patient was counseled to cover herself in petrolatum for 2 to 3 hours with the hope of smothering any remaining tick larvae. She was given triamcinolone cream and was encouraged to take a nonsedating antihistamine for itch. The patient was seen back in clinic 2 weeks later and the eruption had resolved.
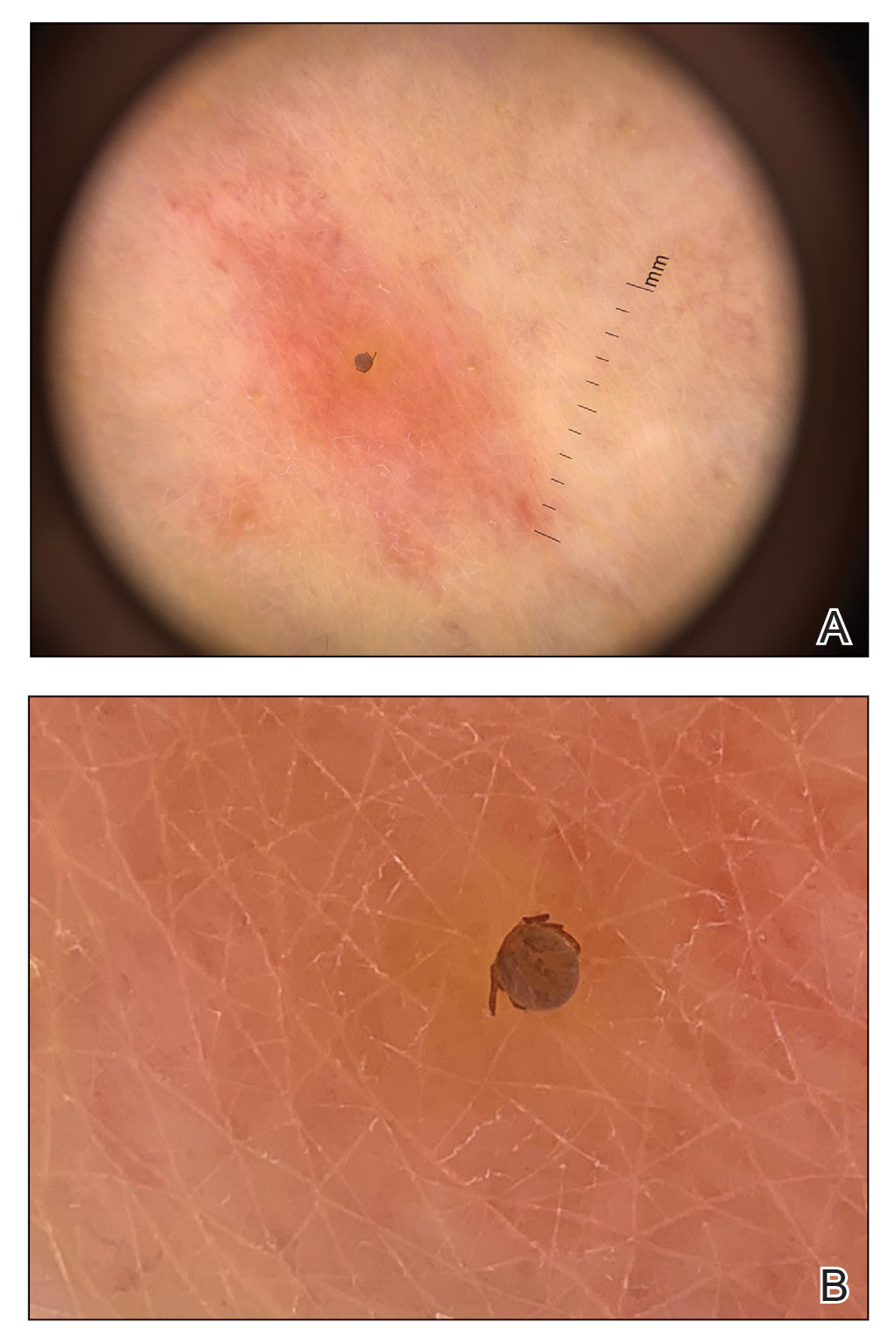
Comment
Spread of Tick-Borne Disease—Ticks and tick-borne disease are increasing major health concerns for humans, domesticated animals, and livestock. Reported cases of bacterial and protozoan tick-borne disease doubled in the United States between 2004 and 2016. Ninety percent of the nearly 60,000 cases of nationally notifiable vector-borne diseases reported in 2017 were linked to ticks.2 Geographic ranges of multiple tick species continue to expand, which is thought to be secondary to rising global temperatures, ecologic changes, reforestation, and increases in commerce and travel (Figure 4).3 Not only have warming temperatures contributed to geographic range expansion, they also may extend ticks’ active season. The lone star tick (Amblyomma americanum) is widely distributed throughout much of the eastern United States.4 The range of A americanum has expanded north in recent years from its prior core range in the southeastern United States.2 One study found that from 2006 to 2016, the vector tick species most commonly collected from humans and submitted to a tick surveillance system in New Jersey shifted from Ixodes scapularis to A americanum.5
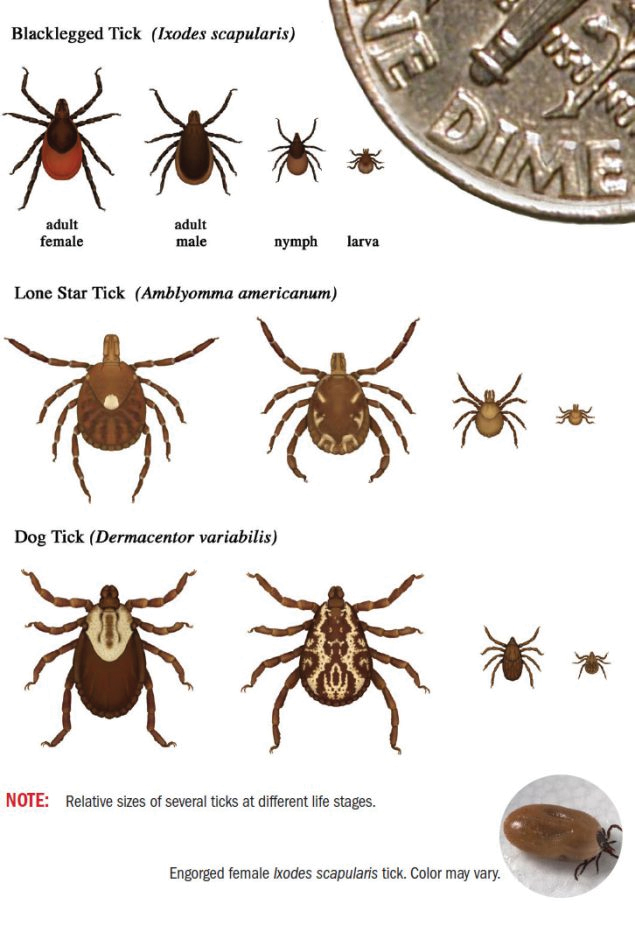
Bites by Amblyomma Ticks—As with most hard ticks, the life cycle of A americanum lasts 2 years and includes the egg, the 6-legged larva or “seed tick,” the 8-legged immature nymph, and the 8-legged reproductively mature adult (Figure 3). Amblyomma americanum can lay several thousand eggs.2 Because our patient had numerous bites, it is plausible that she came into contact with a nest of newly hatched tick larvae. Morphogenesis from larva to nymph, then nymph to adult, requires a blood meal.6,7 The larvae emerge from eggs deposited on the ground and then crawl up low vegetation where they can easily attach to passing hosts. The tick clings to hair or clothing and waits until the host is at rest before moving to a favorable location and then bites.8 When attaching, ticks inject an anesthetic akin to lidocaine, making the bite painless. A tick may spend up to 24 hours on the host prior to biting and then feed for 2 hours to 7 days before releasing.9 For the majority of tick-borne illnesses, the tick must remain attached for 24 to 48 hours before disease is transmitted.10
All stages of
Even when the ticks do not transmit disease, tick bites can cause impressive local reactions. Uncomplicated bites can be painful and leave a puncture wound that can take 1 to 2 weeks to heal.13 Rarely, bites can cause a delayed hypersensitivity reaction including fever, pruritus, and urticaria. Granulomas can develop if a tick is improperly removed.9 Other reports describe prurigo lesions, skin hemorrhage, papular urticaria, diffuse papules, vesicles and bullae, necrotic ulcers, and patchy alopecia.14,15 A 2015 systematic controlled study of human bite reactions from A americanum demonstrated the development of itchy erythematous papules and vesicles within 48 hours of larval tick attachment to research participants. The study found tissue damage from A americanum mouthparts, and degranulating mast cells may be evident in as little as 15 minutes.16 The severity of individual skin reaction is hypothesized to depend on several variables, such as the duration of feeding, size of mouthparts, type of tick secretions, changes in secretions during feeding, and prior exposures of the host.14
Tick Removal—If patients present to clinic with ticks attached, removal can be challenging. Removal recommendations call for use of blunt forceps or tweezers. Ticks should be grasped near the skin with consistent pressure, and the tick should be pulled straight out, perpendicular to the skin. Twisting motions can cause the head to separate from the body and remain in the bite wound. Immediately following removal, the area should be cleansed with a disinfectant.10,17 After the tick is removed, some studies recommend storing the tick at −20 °C; should the patient develop disease, the tick could be sent for evaluation.6,17 If there is no clinical or serologic evidence of infection, testing for the presence of antibodies against tick-borne bacteria at presentation and at 3 and 6 weeks is not recommended due to low sensitivity, low positive predictive value, and cost. Clinicians must only observe and treat if disease occurs.17
Prevention of Tick Bites—Tick bites are best prevented by avoiding tick-infested areas; when these areas are unavoidable, tick bites may be prevented by wearing long pants with the pant legs tucked into boots. In addition, applying topical DEET (N,N-diethyl-m-toluamide) repellent to exposed skin and treating clothing with permethrin can be helpful.17 When used alone, DEET provides greater than 90% protection for up to 2.7 hours against A americanum.18 Permethrin-treated clothing alone is 79% to 100% effective at killing A americanum ticks or disabling them for several hours.19
Conclusion
Tick-borne illness is an increasingly important cause of human infectious disease. In addition to their role as a disease vector, ticks can produce primary skin disorders. This case posed a diagnostic challenge because of the unusually large number and wide distribution of bites as well as the subsequent vesicular reaction that ensued. It is important to keep tick larvae or adult tick bites in the differential when evaluating a patient to expedite tick removal and begin clinical monitoring. Recognition of A americanum larvae as a potential cause of pruritic papules may be helpful in similar cases. In addition, it is important for dermatologists to be aware of the tick species in their area.
- Centers for Disease Control and Prevention. Tick ID. Accessed February 21, 2022. https://www.cdc.gov/ticks/tickbornediseases/tickID.html
- Molaei G, Little EAH, Williams SC, et al. Bracing for the worst—range expansion of the lone star tick in the northeastern United States. N Engl J Med. 2019;381:2189-2192.
- Centers for Disease Control and Prevention, Division of Vector-Borne Diseases. Lone star tick (Amblyomma americanum). Accessed March 23, 2022. https://www.cdc.gov/ticks/maps/lone_star_tick.pdf
- Reynolds HH, Elston DM. What’s eating you? lone star tick (Amblyomma americanum). Cutis. 2017;99:111-114.
- Jordan RA, Egizi A. The growing importance of lone star ticks in a Lyme disease endemic county: passive tick surveillance in Monmouth County, NJ, 2006–2016. PLoS One. 2019;14:E0211778.
- Singh-Behl D, La Rosa SP, Tomecki KJ. Tick-borne infections. Dermatol Clin. 2003;21:237-244, v.
- Spach DH, Liles WC, Campbell GL, et al. Tick-borne diseases in the United States. N Engl J Med. 1993;329:936-947.
- Duckworth PF Jr, Hayden GF, Reed CN. Human infestation by Amblyomma americanum larvae (“seed ticks”). South Med J. 1985;78:751-753.
- Middleton DB. Tick-borne infections. what starts as a tiny bite may have a serious outcome. Postgrad Med. 1994;95:131-139.
- Moody EK, Barker RW, White JL, et al. Ticks and tick-borne diseases in Oklahoma. J Okla State Med Assoc. 1998;91:438-445.
- Jones BE. Human ‘seed tick’ infestation. Amblyomma americanum larvae. Arch Dermatol. 1981;117:812-814.
- Centers for Disease Control and Prevention. Tick bite prophylaxis. Accessed February 21, 2022. https://www.cdc.gov/ticks/tickbornediseases/tick-bite-prophylaxis.html
- Fisher EJ, Mo J, Lucky AW. Multiple pruritic papules from lone star tick larvae bites. Arch Dermatol. 2006;142:491-494.
- Krinsky WL. Dermatoses associated with the bites of mites and ticks (Arthropoda: Acari). Int J Dermatol. 1983;22:75-91.
- Yesudian P, Thambiah AS. Persistent papules after tick-bites. Dermatologica. 1973;147:214-218.
- Goddard J, Portugal JS. Cutaneous lesions due to bites by larval Amblyomma americanum ticks. JAMA Dermatol. 2015;151:1373-1375.
- Parola P, Raoult D. Ticks and tickborne bacterial diseases in humans: an emerging infectious threat. Clin Infect Dis. 2001;32:897-928.
- Solberg VB, Klein TA, McPherson KR, et al. Field evaluation of DEET and a piperidine repellent (AI3-37220) against Amblyomma americanum (Acari: Ixodidae). J Med Entomol. 1995;32:870-875.
- Evans SR, Korch GW Jr, Lawson MA. Comparative field evaluation of permethrin and DEET-treated military uniforms for personal protection against ticks (Acari). J Med Entomol. 1990;27:829-834.
Case Report
A 58-year-old woman presented to the dermatology office with a widespread pruritic eruption of 3 days’ duration that started in the groin and spread to the rest of the body. No treatments had been attempted. She had no notable medical history, and she denied any recent illness, change in personal care products, or new medications or supplements. She reported a camping trip 2 weeks prior to presentation on the east end of Long Island, New York. She later learned that others on the same trip developed a similar, albeit less widespread, eruption.

Physical examination revealed clear vesicles on the arms, legs, trunk, and pubic area (Figure 1). Dermoscopy revealed a small lone star tick larva in the center of one of the vesicles (Figure 2). The type of tick larva was identified using resources from the Centers for Disease Control and Prevention (Figure 3).1 Careful inspection revealed dark marks on various vesicles, mostly in the perineum, yielding nearly 20 larvae, which were removed with forceps. The patient was counseled to cover herself in petrolatum for 2 to 3 hours with the hope of smothering any remaining tick larvae. She was given triamcinolone cream and was encouraged to take a nonsedating antihistamine for itch. The patient was seen back in clinic 2 weeks later and the eruption had resolved.

Comment
Spread of Tick-Borne Disease—Ticks and tick-borne disease are increasing major health concerns for humans, domesticated animals, and livestock. Reported cases of bacterial and protozoan tick-borne disease doubled in the United States between 2004 and 2016. Ninety percent of the nearly 60,000 cases of nationally notifiable vector-borne diseases reported in 2017 were linked to ticks.2 Geographic ranges of multiple tick species continue to expand, which is thought to be secondary to rising global temperatures, ecologic changes, reforestation, and increases in commerce and travel (Figure 4).3 Not only have warming temperatures contributed to geographic range expansion, they also may extend ticks’ active season. The lone star tick (Amblyomma americanum) is widely distributed throughout much of the eastern United States.4 The range of A americanum has expanded north in recent years from its prior core range in the southeastern United States.2 One study found that from 2006 to 2016, the vector tick species most commonly collected from humans and submitted to a tick surveillance system in New Jersey shifted from Ixodes scapularis to A americanum.5

Bites by Amblyomma Ticks—As with most hard ticks, the life cycle of A americanum lasts 2 years and includes the egg, the 6-legged larva or “seed tick,” the 8-legged immature nymph, and the 8-legged reproductively mature adult (Figure 3). Amblyomma americanum can lay several thousand eggs.2 Because our patient had numerous bites, it is plausible that she came into contact with a nest of newly hatched tick larvae. Morphogenesis from larva to nymph, then nymph to adult, requires a blood meal.6,7 The larvae emerge from eggs deposited on the ground and then crawl up low vegetation where they can easily attach to passing hosts. The tick clings to hair or clothing and waits until the host is at rest before moving to a favorable location and then bites.8 When attaching, ticks inject an anesthetic akin to lidocaine, making the bite painless. A tick may spend up to 24 hours on the host prior to biting and then feed for 2 hours to 7 days before releasing.9 For the majority of tick-borne illnesses, the tick must remain attached for 24 to 48 hours before disease is transmitted.10

All stages of
Even when the ticks do not transmit disease, tick bites can cause impressive local reactions. Uncomplicated bites can be painful and leave a puncture wound that can take 1 to 2 weeks to heal.13 Rarely, bites can cause a delayed hypersensitivity reaction including fever, pruritus, and urticaria. Granulomas can develop if a tick is improperly removed.9 Other reports describe prurigo lesions, skin hemorrhage, papular urticaria, diffuse papules, vesicles and bullae, necrotic ulcers, and patchy alopecia.14,15 A 2015 systematic controlled study of human bite reactions from A americanum demonstrated the development of itchy erythematous papules and vesicles within 48 hours of larval tick attachment to research participants. The study found tissue damage from A americanum mouthparts, and degranulating mast cells may be evident in as little as 15 minutes.16 The severity of individual skin reaction is hypothesized to depend on several variables, such as the duration of feeding, size of mouthparts, type of tick secretions, changes in secretions during feeding, and prior exposures of the host.14
Tick Removal—If patients present to clinic with ticks attached, removal can be challenging. Removal recommendations call for use of blunt forceps or tweezers. Ticks should be grasped near the skin with consistent pressure, and the tick should be pulled straight out, perpendicular to the skin. Twisting motions can cause the head to separate from the body and remain in the bite wound. Immediately following removal, the area should be cleansed with a disinfectant.10,17 After the tick is removed, some studies recommend storing the tick at −20 °C; should the patient develop disease, the tick could be sent for evaluation.6,17 If there is no clinical or serologic evidence of infection, testing for the presence of antibodies against tick-borne bacteria at presentation and at 3 and 6 weeks is not recommended due to low sensitivity, low positive predictive value, and cost. Clinicians must only observe and treat if disease occurs.17
Prevention of Tick Bites—Tick bites are best prevented by avoiding tick-infested areas; when these areas are unavoidable, tick bites may be prevented by wearing long pants with the pant legs tucked into boots. In addition, applying topical DEET (N,N-diethyl-m-toluamide) repellent to exposed skin and treating clothing with permethrin can be helpful.17 When used alone, DEET provides greater than 90% protection for up to 2.7 hours against A americanum.18 Permethrin-treated clothing alone is 79% to 100% effective at killing A americanum ticks or disabling them for several hours.19
Conclusion
Tick-borne illness is an increasingly important cause of human infectious disease. In addition to their role as a disease vector, ticks can produce primary skin disorders. This case posed a diagnostic challenge because of the unusually large number and wide distribution of bites as well as the subsequent vesicular reaction that ensued. It is important to keep tick larvae or adult tick bites in the differential when evaluating a patient to expedite tick removal and begin clinical monitoring. Recognition of A americanum larvae as a potential cause of pruritic papules may be helpful in similar cases. In addition, it is important for dermatologists to be aware of the tick species in their area.
Case Report
A 58-year-old woman presented to the dermatology office with a widespread pruritic eruption of 3 days’ duration that started in the groin and spread to the rest of the body. No treatments had been attempted. She had no notable medical history, and she denied any recent illness, change in personal care products, or new medications or supplements. She reported a camping trip 2 weeks prior to presentation on the east end of Long Island, New York. She later learned that others on the same trip developed a similar, albeit less widespread, eruption.

Physical examination revealed clear vesicles on the arms, legs, trunk, and pubic area (Figure 1). Dermoscopy revealed a small lone star tick larva in the center of one of the vesicles (Figure 2). The type of tick larva was identified using resources from the Centers for Disease Control and Prevention (Figure 3).1 Careful inspection revealed dark marks on various vesicles, mostly in the perineum, yielding nearly 20 larvae, which were removed with forceps. The patient was counseled to cover herself in petrolatum for 2 to 3 hours with the hope of smothering any remaining tick larvae. She was given triamcinolone cream and was encouraged to take a nonsedating antihistamine for itch. The patient was seen back in clinic 2 weeks later and the eruption had resolved.

Comment
Spread of Tick-Borne Disease—Ticks and tick-borne disease are increasing major health concerns for humans, domesticated animals, and livestock. Reported cases of bacterial and protozoan tick-borne disease doubled in the United States between 2004 and 2016. Ninety percent of the nearly 60,000 cases of nationally notifiable vector-borne diseases reported in 2017 were linked to ticks.2 Geographic ranges of multiple tick species continue to expand, which is thought to be secondary to rising global temperatures, ecologic changes, reforestation, and increases in commerce and travel (Figure 4).3 Not only have warming temperatures contributed to geographic range expansion, they also may extend ticks’ active season. The lone star tick (Amblyomma americanum) is widely distributed throughout much of the eastern United States.4 The range of A americanum has expanded north in recent years from its prior core range in the southeastern United States.2 One study found that from 2006 to 2016, the vector tick species most commonly collected from humans and submitted to a tick surveillance system in New Jersey shifted from Ixodes scapularis to A americanum.5

Bites by Amblyomma Ticks—As with most hard ticks, the life cycle of A americanum lasts 2 years and includes the egg, the 6-legged larva or “seed tick,” the 8-legged immature nymph, and the 8-legged reproductively mature adult (Figure 3). Amblyomma americanum can lay several thousand eggs.2 Because our patient had numerous bites, it is plausible that she came into contact with a nest of newly hatched tick larvae. Morphogenesis from larva to nymph, then nymph to adult, requires a blood meal.6,7 The larvae emerge from eggs deposited on the ground and then crawl up low vegetation where they can easily attach to passing hosts. The tick clings to hair or clothing and waits until the host is at rest before moving to a favorable location and then bites.8 When attaching, ticks inject an anesthetic akin to lidocaine, making the bite painless. A tick may spend up to 24 hours on the host prior to biting and then feed for 2 hours to 7 days before releasing.9 For the majority of tick-borne illnesses, the tick must remain attached for 24 to 48 hours before disease is transmitted.10

All stages of
Even when the ticks do not transmit disease, tick bites can cause impressive local reactions. Uncomplicated bites can be painful and leave a puncture wound that can take 1 to 2 weeks to heal.13 Rarely, bites can cause a delayed hypersensitivity reaction including fever, pruritus, and urticaria. Granulomas can develop if a tick is improperly removed.9 Other reports describe prurigo lesions, skin hemorrhage, papular urticaria, diffuse papules, vesicles and bullae, necrotic ulcers, and patchy alopecia.14,15 A 2015 systematic controlled study of human bite reactions from A americanum demonstrated the development of itchy erythematous papules and vesicles within 48 hours of larval tick attachment to research participants. The study found tissue damage from A americanum mouthparts, and degranulating mast cells may be evident in as little as 15 minutes.16 The severity of individual skin reaction is hypothesized to depend on several variables, such as the duration of feeding, size of mouthparts, type of tick secretions, changes in secretions during feeding, and prior exposures of the host.14
Tick Removal—If patients present to clinic with ticks attached, removal can be challenging. Removal recommendations call for use of blunt forceps or tweezers. Ticks should be grasped near the skin with consistent pressure, and the tick should be pulled straight out, perpendicular to the skin. Twisting motions can cause the head to separate from the body and remain in the bite wound. Immediately following removal, the area should be cleansed with a disinfectant.10,17 After the tick is removed, some studies recommend storing the tick at −20 °C; should the patient develop disease, the tick could be sent for evaluation.6,17 If there is no clinical or serologic evidence of infection, testing for the presence of antibodies against tick-borne bacteria at presentation and at 3 and 6 weeks is not recommended due to low sensitivity, low positive predictive value, and cost. Clinicians must only observe and treat if disease occurs.17
Prevention of Tick Bites—Tick bites are best prevented by avoiding tick-infested areas; when these areas are unavoidable, tick bites may be prevented by wearing long pants with the pant legs tucked into boots. In addition, applying topical DEET (N,N-diethyl-m-toluamide) repellent to exposed skin and treating clothing with permethrin can be helpful.17 When used alone, DEET provides greater than 90% protection for up to 2.7 hours against A americanum.18 Permethrin-treated clothing alone is 79% to 100% effective at killing A americanum ticks or disabling them for several hours.19
Conclusion
Tick-borne illness is an increasingly important cause of human infectious disease. In addition to their role as a disease vector, ticks can produce primary skin disorders. This case posed a diagnostic challenge because of the unusually large number and wide distribution of bites as well as the subsequent vesicular reaction that ensued. It is important to keep tick larvae or adult tick bites in the differential when evaluating a patient to expedite tick removal and begin clinical monitoring. Recognition of A americanum larvae as a potential cause of pruritic papules may be helpful in similar cases. In addition, it is important for dermatologists to be aware of the tick species in their area.
- Centers for Disease Control and Prevention. Tick ID. Accessed February 21, 2022. https://www.cdc.gov/ticks/tickbornediseases/tickID.html
- Molaei G, Little EAH, Williams SC, et al. Bracing for the worst—range expansion of the lone star tick in the northeastern United States. N Engl J Med. 2019;381:2189-2192.
- Centers for Disease Control and Prevention, Division of Vector-Borne Diseases. Lone star tick (Amblyomma americanum). Accessed March 23, 2022. https://www.cdc.gov/ticks/maps/lone_star_tick.pdf
- Reynolds HH, Elston DM. What’s eating you? lone star tick (Amblyomma americanum). Cutis. 2017;99:111-114.
- Jordan RA, Egizi A. The growing importance of lone star ticks in a Lyme disease endemic county: passive tick surveillance in Monmouth County, NJ, 2006–2016. PLoS One. 2019;14:E0211778.
- Singh-Behl D, La Rosa SP, Tomecki KJ. Tick-borne infections. Dermatol Clin. 2003;21:237-244, v.
- Spach DH, Liles WC, Campbell GL, et al. Tick-borne diseases in the United States. N Engl J Med. 1993;329:936-947.
- Duckworth PF Jr, Hayden GF, Reed CN. Human infestation by Amblyomma americanum larvae (“seed ticks”). South Med J. 1985;78:751-753.
- Middleton DB. Tick-borne infections. what starts as a tiny bite may have a serious outcome. Postgrad Med. 1994;95:131-139.
- Moody EK, Barker RW, White JL, et al. Ticks and tick-borne diseases in Oklahoma. J Okla State Med Assoc. 1998;91:438-445.
- Jones BE. Human ‘seed tick’ infestation. Amblyomma americanum larvae. Arch Dermatol. 1981;117:812-814.
- Centers for Disease Control and Prevention. Tick bite prophylaxis. Accessed February 21, 2022. https://www.cdc.gov/ticks/tickbornediseases/tick-bite-prophylaxis.html
- Fisher EJ, Mo J, Lucky AW. Multiple pruritic papules from lone star tick larvae bites. Arch Dermatol. 2006;142:491-494.
- Krinsky WL. Dermatoses associated with the bites of mites and ticks (Arthropoda: Acari). Int J Dermatol. 1983;22:75-91.
- Yesudian P, Thambiah AS. Persistent papules after tick-bites. Dermatologica. 1973;147:214-218.
- Goddard J, Portugal JS. Cutaneous lesions due to bites by larval Amblyomma americanum ticks. JAMA Dermatol. 2015;151:1373-1375.
- Parola P, Raoult D. Ticks and tickborne bacterial diseases in humans: an emerging infectious threat. Clin Infect Dis. 2001;32:897-928.
- Solberg VB, Klein TA, McPherson KR, et al. Field evaluation of DEET and a piperidine repellent (AI3-37220) against Amblyomma americanum (Acari: Ixodidae). J Med Entomol. 1995;32:870-875.
- Evans SR, Korch GW Jr, Lawson MA. Comparative field evaluation of permethrin and DEET-treated military uniforms for personal protection against ticks (Acari). J Med Entomol. 1990;27:829-834.
- Centers for Disease Control and Prevention. Tick ID. Accessed February 21, 2022. https://www.cdc.gov/ticks/tickbornediseases/tickID.html
- Molaei G, Little EAH, Williams SC, et al. Bracing for the worst—range expansion of the lone star tick in the northeastern United States. N Engl J Med. 2019;381:2189-2192.
- Centers for Disease Control and Prevention, Division of Vector-Borne Diseases. Lone star tick (Amblyomma americanum). Accessed March 23, 2022. https://www.cdc.gov/ticks/maps/lone_star_tick.pdf
- Reynolds HH, Elston DM. What’s eating you? lone star tick (Amblyomma americanum). Cutis. 2017;99:111-114.
- Jordan RA, Egizi A. The growing importance of lone star ticks in a Lyme disease endemic county: passive tick surveillance in Monmouth County, NJ, 2006–2016. PLoS One. 2019;14:E0211778.
- Singh-Behl D, La Rosa SP, Tomecki KJ. Tick-borne infections. Dermatol Clin. 2003;21:237-244, v.
- Spach DH, Liles WC, Campbell GL, et al. Tick-borne diseases in the United States. N Engl J Med. 1993;329:936-947.
- Duckworth PF Jr, Hayden GF, Reed CN. Human infestation by Amblyomma americanum larvae (“seed ticks”). South Med J. 1985;78:751-753.
- Middleton DB. Tick-borne infections. what starts as a tiny bite may have a serious outcome. Postgrad Med. 1994;95:131-139.
- Moody EK, Barker RW, White JL, et al. Ticks and tick-borne diseases in Oklahoma. J Okla State Med Assoc. 1998;91:438-445.
- Jones BE. Human ‘seed tick’ infestation. Amblyomma americanum larvae. Arch Dermatol. 1981;117:812-814.
- Centers for Disease Control and Prevention. Tick bite prophylaxis. Accessed February 21, 2022. https://www.cdc.gov/ticks/tickbornediseases/tick-bite-prophylaxis.html
- Fisher EJ, Mo J, Lucky AW. Multiple pruritic papules from lone star tick larvae bites. Arch Dermatol. 2006;142:491-494.
- Krinsky WL. Dermatoses associated with the bites of mites and ticks (Arthropoda: Acari). Int J Dermatol. 1983;22:75-91.
- Yesudian P, Thambiah AS. Persistent papules after tick-bites. Dermatologica. 1973;147:214-218.
- Goddard J, Portugal JS. Cutaneous lesions due to bites by larval Amblyomma americanum ticks. JAMA Dermatol. 2015;151:1373-1375.
- Parola P, Raoult D. Ticks and tickborne bacterial diseases in humans: an emerging infectious threat. Clin Infect Dis. 2001;32:897-928.
- Solberg VB, Klein TA, McPherson KR, et al. Field evaluation of DEET and a piperidine repellent (AI3-37220) against Amblyomma americanum (Acari: Ixodidae). J Med Entomol. 1995;32:870-875.
- Evans SR, Korch GW Jr, Lawson MA. Comparative field evaluation of permethrin and DEET-treated military uniforms for personal protection against ticks (Acari). J Med Entomol. 1990;27:829-834.
Practice Points
- The range of Amblyomma americanum has expanded north in recent years from its core range in the southeastern United States. Warming temperatures also have increased the duration of the ticks’ active season.
- Amblyomma americanum can lay several thousand eggs. A person happening upon a newly hatched nest of larval ticks could sustain a widespread vesicular eruption secondary to tick bites.
- It is important to keep larval tick infestation in the differential when evaluating a patient with a new widespread vesicular eruption to expedite prompt removal of the offending ticks and to begin clinical monitoring.
The Residency Application Process: Current and Future Landscape
Amid increasing numbers of applications, decreasing match rates, and ongoing lack of diversity in the dermatology trainee workforce, the COVID-19 pandemic introduced additional challenges to the dermatology residency application process and laid bare systemic inequities and inherent problems that must be addressed. Historically, dermatology applicants have excelled in academic metrics, such as US Medical Licensing Examination (USMLE) scores and nomination to the Alpha Omega Alpha honor society. As biases associated with these academic metrics are being elucidated, they have in turn become less available. With the upcoming change in USMLE Step 1 reporting to pass/fail only, as well as the elimination of Alpha Omega Alpha nomination for students, clinical grades, and/or class ranks at many medical schools, other elements of the application, such as volunteer experiences and research publications, may be weighed more heavily in the selection process. This may serve to exacerbate the application arms race, characterized by a steady rise in volunteer experiences, research publications, and research gap years that has already begun and likely will continue, particularly among dermatology applicants.
These issues are not unique to dermatology and are occurring across all medical specialties to varying degrees. The monetary and opportunity costs of the application process have become astronomical for both applicants and faculty. Faculty are overburdened with administrative duties related to resident recruitment and advising, and students are experiencing heightened match-related anxiety earlier and more acutely. These factors may contribute to burnout among trainees and faculty and may have deleterious effects on medical education. It is clear that transformative work must be pursued to ensure an equitable and sustainable residency application process moving forward. In this column, we review the notable work being done within dermatology and across specialties to reform the residency application process.
Coalition Recommendations
In August 2021, the Coalition for Physician Accountability (CoPA) released recommendations for comprehensive improvement of the undergraduate medical education (UME) to graduate medical education transition, which includes residency application. Of the 9 principal themes addressed, 2 focus on the residency application process: (1) equitable mission-driven application review, and (2) optimization of the application, interview, and selection processes, which relates to application volume as well as interview offers and formats.1
In the area of application review, CoPA recommends replacing all letters of recommendation with structured evaluative letters as a universal tool in the application process.1 These letters would include specialty-specific questions based on core competencies and would be completed by an evaluator who directly observed the student. Additionally, the group recommends revising the content and structure of the medical student performance evaluation to improve access to longitudinal assessment data about students. Ideally, developing UME competency outcomes to apply across learners would decrease reliance on traditional but potentially problematic application elements, such as licensing examination scores, clinical grades, and narrative evaluations.1
To optimize residency application processes, CoPA recommends exploring innovative approaches to reduce application volume and maximize applicants interviewing and matching at programs where mutual interest is high.1 Suggestions to address these issues include preference signaling, application caps, and/or additional rounds of application or matching. Standardization of the interview process also is recommended to improve equity, minimize educational disruption, and improve applicant well-being. Suggestions include the use of common interview offer and scheduling platforms, policies to govern interview offers and scheduling timelines, interview caps, and ongoing study of the impact of virtual interviews.1
Residency Application Innovations Implemented by Other Specialties
A number of specialties have developed innovations in the residency application process to improve equity and fairness as well as optimize applicant-program fit. Emergency medicine created a now widely adopted, specialty-specific standardized letter of evaluation (SLOE).2 It compares applicants across a number of measures that include personal qualities, clinical skills, and a global assessment. The SLOE is designed to assess and compare applicants across institutions rather than provide recommendations. The emergency medicine SLOE also provides useful information about the letter writer, including duration and depth of interaction with the applicant and distribution of rankings of prior applicants.2
In 2019, obstetrics and gynecology launched a standardized application and interview process, which set a specialty-wide application deadline, limited interview invitations to the number of interview positions available, encouraged coordinated release of interview offers, and allowed applicants 72 hours to respond to invitations.3 These measures were implemented to improve fairness, transparency, and applicant well-being, as well as to promote equitable distribution of interviews. Data following this launch suggested that universal offer dates reduced excessive interviewing among competitive applicants.3
Last year, otolaryngology implemented a process known as preference signaling in which applicants were able to signal up to 5 preferred programs at the time of application. A signal allowed applicants to demonstrate interest in specific programs and could be used by programs during their application review process. Most applicants opted to submit signals, and programs received 0 to 71 signals (mean, 22).4 Almost all programs received at least 1 signal. The rate of receiving an interview was significantly higher for signaled programs (58%) compared to nonsignaled programs (14%)(P<.001), indicating that preference signaling may be beneficial for both programs and applicants for interview selection.4
Residency Application Innovations Implemented by Dermatology
Over the last 2 application cycles, dermatology has implemented several innovations to the residency application process. Initial work included release of guidelines for residency programs to conduct holistic application review,5 recommendations for website updates to share program-specific information with prospective trainees,6 and informational webinars and statements to update dermatology applicants about changes to the process and to answer application-related questions.7-9
In 2020, dermatology initiated a coordinated interview invitation release in which interview offers were released on prespecified dates and applicants were given 48 hours prior to scheduling. Approximately 50% of residency programs participated in the first year, yet nearly all programs released on 1 of 2 universal dates in the current cycle. In a recent survey of dermatology applicants, nearly 90% supported coordinated release.10 Several other specialties also have incorporated universal release dates into their processes.
For the 2021-2022 application cycle, dermatology—along with internal medicine and general surgery—participated in the Association of American Medical Colleges’ pilot supplemental Electronic Residency Application Service (ERAS) application.11 The pilot was designed as a first step to updating the ERAS content by allowing students to share more information about their extracurricular, research, and clinical activities, as well as geographic and program preferences to optimize applicant-program fit. Preference signaling, similar to the otolaryngology process, was included in the supplemental application, with dermatology applicants choosing up to 3 preferred programs to signal, excluding their home programs and any programs where they completed in-person away rotations. Preliminary data suggest that the vast majority of dermatology programs and applicants participated in the supplemental application.12 Ongoing analysis of survey data from applicants, advisors, and program directors will help inform future directions. Dermatology has been an integral partner in the development, implementation, and evaluation of this pilot.
Proposed Innovations to the Application Process
Given the challenges of the current application process, there has been a long list of proposed innovations to ameliorate applicant, advisor, and program concerns.13 Many of these approaches are intended to respond to increasing costs to programs and applicants as well as the lack of equity in the process. Application caps and an early result acceptance program have both been proposed to address the ever-increasing volume of applications.14,15 Neither of these proposals has been adopted by a specialty yet, but obstetrics and gynecology stakeholders have shown broad support for an early result acceptance program, signaling a possible future pilot.16
Interview caps also have been proposed to promote more equitable distribution of interview positions.17 Ophthalmology implemented this approach in the 2021-2022 application cycle, with applicants limited to a maximum of 18 interviews.18 Data from this pilot will help determine the effect of interview caps as well as the optimal limit, which will vary by specialty.
Changes to the application content itself could better facilitate holistic review and optimize applicant-program fit. This is the principle driving the pilot supplemental ERAS application, but it also has been addressed in other specialties. Ophthalmology replaced the traditional personal statement with a shorter autobiographical statement as well as 2 short personal essay questions. Plastic surgery designed a common supplemental application, currently in its second iteration, that highlights specialty-specific information from applicants to promote holistic review and eventually reduce application costs.19
Final Thoughts
The reforms introduced and proposed by dermatology and other specialties represent initial steps to address the issues inherent to the current residency application process. Providing faculty with better tools to holistically assess applicants during the review process and increasing transparency between programs and applicants should help optimize applicant-program fit and increase diversity in the dermatology workforce. Streamlining the application process to allow students to highlight their unique qualities in a user-friendly format as well as addressing potential inequities in interview distribution and access to the application process hopefully will contribute to better outcomes for both programs and applicants. However, many of these steps are likely to create additional administrative burdens on program faculty and are unlikely to allay student fears about matching.
The underlying issue for many specialties, and particularly for dermatology, is that demand far outstrips supply. With stable numbers of residency positions and an ever-increasing number of applicants, the match rate will continue to decrease, leading to increased anxiety among those interested in pursuing dermatology. Although USMLE Step 1 scores have been shown to have racial bias20 and there are no data correlating scores with clinical performance, the elimination of a scoring system may affect the number of applicants entering dermatology with downstream effects on match rates. Heightened anxiety places increased pressure on students to choose a specialty earlier in their training and impacts the activities they pursue during medical school. Overemphasis on specialty choice and the match process can lead to higher rates of burnout among students and trainees, as students may focus on activities designed to increase their chances of matching at the expense of pursuing activities that could lead to greater engagement and passion in their careers—a key protective factor against burnout.
The goal of the residency application process is to optimize fit between candidates and programs by aligning goals, values, and learning environment. Students and programs working together as honest brokers can lead to transformative change in the process, freeing both parties to highlight their unique qualities and contributions. Programs benefit from optimal fit by being able to hone their particular mission and recruit and retain residents and faculty engaged in that mission. Residents will thrive in programs that support their learning and career goals and will ultimately be better positioned to meaningfully contribute to their chosen field in whatever capacity they choose.
Acknowledgments—The views presented in this column reflect those of the 9 elected members of the Association of Professors of Dermatology Residency Program Directors Section steering committee, all of whom are program directors at their institutions (listed in parentheses): Ammar Ahmed, MD (The University of Texas at Austin, Austin, Texas); Yolanda Helfrich, MD (University of Michigan, Ann Arbor, Michigan); Jo-Ann M. Latkowksi, MD (New York University, New York); Kiran Motaparthi, MD (University of Florida, Gainesville, Florida); Adena E. Rosenblatt, MD, PhD (The University of Chicago, Chicago, Illinois); Ilana S. Rosman, MD (Washington University, St. Louis, Missouri); Travis Vandergriff, MD (University of Texas Southwestern, Dallas, Texas); Diane Whitaker-Worth, MD (University of Connecticut, Farmington, Connecticut); Scott Worswick, MD (University of Southern California, Los Angeles, California).
- Coalition for Physician Accountability. The Coalition for Physician Accountability’s Undergraduate Medical Education–Graduate Medical Education Review Committee (UGRC): recommendations for comprehensive improvement of the UME-GME transition. Accessed March 7, 2022. https://physicianaccountability.org/wp-content/uploads/2021/08/UGRC-Coalition-Report-FINAL.pdf
- Jackson JS, Bond M, Love JN, et al. Emergency medicine standardized letter of evaluation (SLOE): findings from the new electronic SLOE format. J Grad Med Educ. 2019;11:182-186.
- Santos-Parker KS, Morgan HK, Katz NT, et al. Can standardized dates for interview offers mitigate excessive interviewing? J Surg Educ. 2021;78:1091-1096.
- Pletcher SD, Chang CWD, Thorne MC, et al. The otolaryngology residency program preference signaling experience [published online October 5, 2021]. Acad Med. doi:10.1097/ACM.0000000000004441
- Association of Professors of Dermatology. Holistic review. Accessed March 7, 2022. https://www.dermatologyprofessors.org/files/3_Holistic%20review_Oct2020.pdf
- Rosmarin D, Friedman AJ, Burkemper NM, et al. The Association of Professors of Dermatology Program Directors Task Force and Residency Program Transparency Work Group guidelines on residency program transparency. J Drugs Dermatol. 2020;19:1117-1118.
- Rosman IS, Schadt CR, Samimi SS, et al. Approaching the dermatology residency application process during a pandemic. J Am Acad Dermatol. 2020;83:E351-E352.
- Association of Professors of Dermatology. Program director resources. Accessed March 7, 2022. https://www.dermatologyprofessors.org/programdirectors_resources.php
- Brumfiel CM, Jefferson IS, Wu AG, et al. A national webinar for dermatology applicants during the COVID-19 pandemic. J Am Acad Dermatol. 2021;84:574-575.
- Brumfiel CM, Jefferson IS, Rinderknecht FA, et al. Current perspectives of and potential reforms to the dermatology residency application process: a nationwide survey of program directors and applicants. Clin Dermatol. In press.
- Association of American Medical Colleges. Supplemental ERAS application (for the ERAS 2022 cycle). Accessed March 7, 2022. https://students-residents.aamc.org/applying-residencies-eras/supplementalerasapplication
- Association of American Medical Colleges. AAMC supplemental ERAS application: key findings from the 2022 application cycle. Accessed March 11, 2022. https://www.aamc.org/media/58891/download
- Warm EJ, Kinnear B, Pereira A, et al. The residency match: escaping the prisoner’s dilemma. J Grad Med Educ. 2021;13:616-625.
- Carmody JB, Rosman IS, Carlson JC. Application fever: reviewing the causes, costs, and cures for residency application inflation. Cureus. 2021;13:E13804.
- Hammoud MM, Andrews J, Skochelak SE. Improving the residency application and selection process: an optional early result acceptance program. JAMA. 2020;323:503-504.
- Winkel AF, Morgan HK, Akingbola O, et al. Perspectives of stakeholders about an early release acceptance program to complement the residency match in obstetrics and gynecology. JAMA Netw Open. 2021;4:E2124158.
- Morgan HK, Winkel AF, Standiford T, et al. The case for capping residency interviews. J Surg Educ. 2021;78:755-762.
- Association of University Professors of Ophthalmology. 2021-22 ophthalmology residency match FAQs. Accessed March 7, 2022. https://aupo.org/sites/default/files/2021-06/Residency%20Match%20FAQs_2021.pdf
- American Council of Academic Plastic Surgeons. Applying to plastic surgery (PSCA). Accessed March 7, 2022. https://acaplasticsurgeons.org/PSCA/
- Rubright JD, Jodoin M, Barone MA. Examining demographics, prior academic performance, and United States Medical Licensing Examination Scores. Acad Med. 2019;94:364-370.
Amid increasing numbers of applications, decreasing match rates, and ongoing lack of diversity in the dermatology trainee workforce, the COVID-19 pandemic introduced additional challenges to the dermatology residency application process and laid bare systemic inequities and inherent problems that must be addressed. Historically, dermatology applicants have excelled in academic metrics, such as US Medical Licensing Examination (USMLE) scores and nomination to the Alpha Omega Alpha honor society. As biases associated with these academic metrics are being elucidated, they have in turn become less available. With the upcoming change in USMLE Step 1 reporting to pass/fail only, as well as the elimination of Alpha Omega Alpha nomination for students, clinical grades, and/or class ranks at many medical schools, other elements of the application, such as volunteer experiences and research publications, may be weighed more heavily in the selection process. This may serve to exacerbate the application arms race, characterized by a steady rise in volunteer experiences, research publications, and research gap years that has already begun and likely will continue, particularly among dermatology applicants.
These issues are not unique to dermatology and are occurring across all medical specialties to varying degrees. The monetary and opportunity costs of the application process have become astronomical for both applicants and faculty. Faculty are overburdened with administrative duties related to resident recruitment and advising, and students are experiencing heightened match-related anxiety earlier and more acutely. These factors may contribute to burnout among trainees and faculty and may have deleterious effects on medical education. It is clear that transformative work must be pursued to ensure an equitable and sustainable residency application process moving forward. In this column, we review the notable work being done within dermatology and across specialties to reform the residency application process.
Coalition Recommendations
In August 2021, the Coalition for Physician Accountability (CoPA) released recommendations for comprehensive improvement of the undergraduate medical education (UME) to graduate medical education transition, which includes residency application. Of the 9 principal themes addressed, 2 focus on the residency application process: (1) equitable mission-driven application review, and (2) optimization of the application, interview, and selection processes, which relates to application volume as well as interview offers and formats.1
In the area of application review, CoPA recommends replacing all letters of recommendation with structured evaluative letters as a universal tool in the application process.1 These letters would include specialty-specific questions based on core competencies and would be completed by an evaluator who directly observed the student. Additionally, the group recommends revising the content and structure of the medical student performance evaluation to improve access to longitudinal assessment data about students. Ideally, developing UME competency outcomes to apply across learners would decrease reliance on traditional but potentially problematic application elements, such as licensing examination scores, clinical grades, and narrative evaluations.1
To optimize residency application processes, CoPA recommends exploring innovative approaches to reduce application volume and maximize applicants interviewing and matching at programs where mutual interest is high.1 Suggestions to address these issues include preference signaling, application caps, and/or additional rounds of application or matching. Standardization of the interview process also is recommended to improve equity, minimize educational disruption, and improve applicant well-being. Suggestions include the use of common interview offer and scheduling platforms, policies to govern interview offers and scheduling timelines, interview caps, and ongoing study of the impact of virtual interviews.1
Residency Application Innovations Implemented by Other Specialties
A number of specialties have developed innovations in the residency application process to improve equity and fairness as well as optimize applicant-program fit. Emergency medicine created a now widely adopted, specialty-specific standardized letter of evaluation (SLOE).2 It compares applicants across a number of measures that include personal qualities, clinical skills, and a global assessment. The SLOE is designed to assess and compare applicants across institutions rather than provide recommendations. The emergency medicine SLOE also provides useful information about the letter writer, including duration and depth of interaction with the applicant and distribution of rankings of prior applicants.2
In 2019, obstetrics and gynecology launched a standardized application and interview process, which set a specialty-wide application deadline, limited interview invitations to the number of interview positions available, encouraged coordinated release of interview offers, and allowed applicants 72 hours to respond to invitations.3 These measures were implemented to improve fairness, transparency, and applicant well-being, as well as to promote equitable distribution of interviews. Data following this launch suggested that universal offer dates reduced excessive interviewing among competitive applicants.3
Last year, otolaryngology implemented a process known as preference signaling in which applicants were able to signal up to 5 preferred programs at the time of application. A signal allowed applicants to demonstrate interest in specific programs and could be used by programs during their application review process. Most applicants opted to submit signals, and programs received 0 to 71 signals (mean, 22).4 Almost all programs received at least 1 signal. The rate of receiving an interview was significantly higher for signaled programs (58%) compared to nonsignaled programs (14%)(P<.001), indicating that preference signaling may be beneficial for both programs and applicants for interview selection.4
Residency Application Innovations Implemented by Dermatology
Over the last 2 application cycles, dermatology has implemented several innovations to the residency application process. Initial work included release of guidelines for residency programs to conduct holistic application review,5 recommendations for website updates to share program-specific information with prospective trainees,6 and informational webinars and statements to update dermatology applicants about changes to the process and to answer application-related questions.7-9
In 2020, dermatology initiated a coordinated interview invitation release in which interview offers were released on prespecified dates and applicants were given 48 hours prior to scheduling. Approximately 50% of residency programs participated in the first year, yet nearly all programs released on 1 of 2 universal dates in the current cycle. In a recent survey of dermatology applicants, nearly 90% supported coordinated release.10 Several other specialties also have incorporated universal release dates into their processes.
For the 2021-2022 application cycle, dermatology—along with internal medicine and general surgery—participated in the Association of American Medical Colleges’ pilot supplemental Electronic Residency Application Service (ERAS) application.11 The pilot was designed as a first step to updating the ERAS content by allowing students to share more information about their extracurricular, research, and clinical activities, as well as geographic and program preferences to optimize applicant-program fit. Preference signaling, similar to the otolaryngology process, was included in the supplemental application, with dermatology applicants choosing up to 3 preferred programs to signal, excluding their home programs and any programs where they completed in-person away rotations. Preliminary data suggest that the vast majority of dermatology programs and applicants participated in the supplemental application.12 Ongoing analysis of survey data from applicants, advisors, and program directors will help inform future directions. Dermatology has been an integral partner in the development, implementation, and evaluation of this pilot.
Proposed Innovations to the Application Process
Given the challenges of the current application process, there has been a long list of proposed innovations to ameliorate applicant, advisor, and program concerns.13 Many of these approaches are intended to respond to increasing costs to programs and applicants as well as the lack of equity in the process. Application caps and an early result acceptance program have both been proposed to address the ever-increasing volume of applications.14,15 Neither of these proposals has been adopted by a specialty yet, but obstetrics and gynecology stakeholders have shown broad support for an early result acceptance program, signaling a possible future pilot.16
Interview caps also have been proposed to promote more equitable distribution of interview positions.17 Ophthalmology implemented this approach in the 2021-2022 application cycle, with applicants limited to a maximum of 18 interviews.18 Data from this pilot will help determine the effect of interview caps as well as the optimal limit, which will vary by specialty.
Changes to the application content itself could better facilitate holistic review and optimize applicant-program fit. This is the principle driving the pilot supplemental ERAS application, but it also has been addressed in other specialties. Ophthalmology replaced the traditional personal statement with a shorter autobiographical statement as well as 2 short personal essay questions. Plastic surgery designed a common supplemental application, currently in its second iteration, that highlights specialty-specific information from applicants to promote holistic review and eventually reduce application costs.19
Final Thoughts
The reforms introduced and proposed by dermatology and other specialties represent initial steps to address the issues inherent to the current residency application process. Providing faculty with better tools to holistically assess applicants during the review process and increasing transparency between programs and applicants should help optimize applicant-program fit and increase diversity in the dermatology workforce. Streamlining the application process to allow students to highlight their unique qualities in a user-friendly format as well as addressing potential inequities in interview distribution and access to the application process hopefully will contribute to better outcomes for both programs and applicants. However, many of these steps are likely to create additional administrative burdens on program faculty and are unlikely to allay student fears about matching.
The underlying issue for many specialties, and particularly for dermatology, is that demand far outstrips supply. With stable numbers of residency positions and an ever-increasing number of applicants, the match rate will continue to decrease, leading to increased anxiety among those interested in pursuing dermatology. Although USMLE Step 1 scores have been shown to have racial bias20 and there are no data correlating scores with clinical performance, the elimination of a scoring system may affect the number of applicants entering dermatology with downstream effects on match rates. Heightened anxiety places increased pressure on students to choose a specialty earlier in their training and impacts the activities they pursue during medical school. Overemphasis on specialty choice and the match process can lead to higher rates of burnout among students and trainees, as students may focus on activities designed to increase their chances of matching at the expense of pursuing activities that could lead to greater engagement and passion in their careers—a key protective factor against burnout.
The goal of the residency application process is to optimize fit between candidates and programs by aligning goals, values, and learning environment. Students and programs working together as honest brokers can lead to transformative change in the process, freeing both parties to highlight their unique qualities and contributions. Programs benefit from optimal fit by being able to hone their particular mission and recruit and retain residents and faculty engaged in that mission. Residents will thrive in programs that support their learning and career goals and will ultimately be better positioned to meaningfully contribute to their chosen field in whatever capacity they choose.
Acknowledgments—The views presented in this column reflect those of the 9 elected members of the Association of Professors of Dermatology Residency Program Directors Section steering committee, all of whom are program directors at their institutions (listed in parentheses): Ammar Ahmed, MD (The University of Texas at Austin, Austin, Texas); Yolanda Helfrich, MD (University of Michigan, Ann Arbor, Michigan); Jo-Ann M. Latkowksi, MD (New York University, New York); Kiran Motaparthi, MD (University of Florida, Gainesville, Florida); Adena E. Rosenblatt, MD, PhD (The University of Chicago, Chicago, Illinois); Ilana S. Rosman, MD (Washington University, St. Louis, Missouri); Travis Vandergriff, MD (University of Texas Southwestern, Dallas, Texas); Diane Whitaker-Worth, MD (University of Connecticut, Farmington, Connecticut); Scott Worswick, MD (University of Southern California, Los Angeles, California).
Amid increasing numbers of applications, decreasing match rates, and ongoing lack of diversity in the dermatology trainee workforce, the COVID-19 pandemic introduced additional challenges to the dermatology residency application process and laid bare systemic inequities and inherent problems that must be addressed. Historically, dermatology applicants have excelled in academic metrics, such as US Medical Licensing Examination (USMLE) scores and nomination to the Alpha Omega Alpha honor society. As biases associated with these academic metrics are being elucidated, they have in turn become less available. With the upcoming change in USMLE Step 1 reporting to pass/fail only, as well as the elimination of Alpha Omega Alpha nomination for students, clinical grades, and/or class ranks at many medical schools, other elements of the application, such as volunteer experiences and research publications, may be weighed more heavily in the selection process. This may serve to exacerbate the application arms race, characterized by a steady rise in volunteer experiences, research publications, and research gap years that has already begun and likely will continue, particularly among dermatology applicants.
These issues are not unique to dermatology and are occurring across all medical specialties to varying degrees. The monetary and opportunity costs of the application process have become astronomical for both applicants and faculty. Faculty are overburdened with administrative duties related to resident recruitment and advising, and students are experiencing heightened match-related anxiety earlier and more acutely. These factors may contribute to burnout among trainees and faculty and may have deleterious effects on medical education. It is clear that transformative work must be pursued to ensure an equitable and sustainable residency application process moving forward. In this column, we review the notable work being done within dermatology and across specialties to reform the residency application process.
Coalition Recommendations
In August 2021, the Coalition for Physician Accountability (CoPA) released recommendations for comprehensive improvement of the undergraduate medical education (UME) to graduate medical education transition, which includes residency application. Of the 9 principal themes addressed, 2 focus on the residency application process: (1) equitable mission-driven application review, and (2) optimization of the application, interview, and selection processes, which relates to application volume as well as interview offers and formats.1
In the area of application review, CoPA recommends replacing all letters of recommendation with structured evaluative letters as a universal tool in the application process.1 These letters would include specialty-specific questions based on core competencies and would be completed by an evaluator who directly observed the student. Additionally, the group recommends revising the content and structure of the medical student performance evaluation to improve access to longitudinal assessment data about students. Ideally, developing UME competency outcomes to apply across learners would decrease reliance on traditional but potentially problematic application elements, such as licensing examination scores, clinical grades, and narrative evaluations.1
To optimize residency application processes, CoPA recommends exploring innovative approaches to reduce application volume and maximize applicants interviewing and matching at programs where mutual interest is high.1 Suggestions to address these issues include preference signaling, application caps, and/or additional rounds of application or matching. Standardization of the interview process also is recommended to improve equity, minimize educational disruption, and improve applicant well-being. Suggestions include the use of common interview offer and scheduling platforms, policies to govern interview offers and scheduling timelines, interview caps, and ongoing study of the impact of virtual interviews.1
Residency Application Innovations Implemented by Other Specialties
A number of specialties have developed innovations in the residency application process to improve equity and fairness as well as optimize applicant-program fit. Emergency medicine created a now widely adopted, specialty-specific standardized letter of evaluation (SLOE).2 It compares applicants across a number of measures that include personal qualities, clinical skills, and a global assessment. The SLOE is designed to assess and compare applicants across institutions rather than provide recommendations. The emergency medicine SLOE also provides useful information about the letter writer, including duration and depth of interaction with the applicant and distribution of rankings of prior applicants.2
In 2019, obstetrics and gynecology launched a standardized application and interview process, which set a specialty-wide application deadline, limited interview invitations to the number of interview positions available, encouraged coordinated release of interview offers, and allowed applicants 72 hours to respond to invitations.3 These measures were implemented to improve fairness, transparency, and applicant well-being, as well as to promote equitable distribution of interviews. Data following this launch suggested that universal offer dates reduced excessive interviewing among competitive applicants.3
Last year, otolaryngology implemented a process known as preference signaling in which applicants were able to signal up to 5 preferred programs at the time of application. A signal allowed applicants to demonstrate interest in specific programs and could be used by programs during their application review process. Most applicants opted to submit signals, and programs received 0 to 71 signals (mean, 22).4 Almost all programs received at least 1 signal. The rate of receiving an interview was significantly higher for signaled programs (58%) compared to nonsignaled programs (14%)(P<.001), indicating that preference signaling may be beneficial for both programs and applicants for interview selection.4
Residency Application Innovations Implemented by Dermatology
Over the last 2 application cycles, dermatology has implemented several innovations to the residency application process. Initial work included release of guidelines for residency programs to conduct holistic application review,5 recommendations for website updates to share program-specific information with prospective trainees,6 and informational webinars and statements to update dermatology applicants about changes to the process and to answer application-related questions.7-9
In 2020, dermatology initiated a coordinated interview invitation release in which interview offers were released on prespecified dates and applicants were given 48 hours prior to scheduling. Approximately 50% of residency programs participated in the first year, yet nearly all programs released on 1 of 2 universal dates in the current cycle. In a recent survey of dermatology applicants, nearly 90% supported coordinated release.10 Several other specialties also have incorporated universal release dates into their processes.
For the 2021-2022 application cycle, dermatology—along with internal medicine and general surgery—participated in the Association of American Medical Colleges’ pilot supplemental Electronic Residency Application Service (ERAS) application.11 The pilot was designed as a first step to updating the ERAS content by allowing students to share more information about their extracurricular, research, and clinical activities, as well as geographic and program preferences to optimize applicant-program fit. Preference signaling, similar to the otolaryngology process, was included in the supplemental application, with dermatology applicants choosing up to 3 preferred programs to signal, excluding their home programs and any programs where they completed in-person away rotations. Preliminary data suggest that the vast majority of dermatology programs and applicants participated in the supplemental application.12 Ongoing analysis of survey data from applicants, advisors, and program directors will help inform future directions. Dermatology has been an integral partner in the development, implementation, and evaluation of this pilot.
Proposed Innovations to the Application Process
Given the challenges of the current application process, there has been a long list of proposed innovations to ameliorate applicant, advisor, and program concerns.13 Many of these approaches are intended to respond to increasing costs to programs and applicants as well as the lack of equity in the process. Application caps and an early result acceptance program have both been proposed to address the ever-increasing volume of applications.14,15 Neither of these proposals has been adopted by a specialty yet, but obstetrics and gynecology stakeholders have shown broad support for an early result acceptance program, signaling a possible future pilot.16
Interview caps also have been proposed to promote more equitable distribution of interview positions.17 Ophthalmology implemented this approach in the 2021-2022 application cycle, with applicants limited to a maximum of 18 interviews.18 Data from this pilot will help determine the effect of interview caps as well as the optimal limit, which will vary by specialty.
Changes to the application content itself could better facilitate holistic review and optimize applicant-program fit. This is the principle driving the pilot supplemental ERAS application, but it also has been addressed in other specialties. Ophthalmology replaced the traditional personal statement with a shorter autobiographical statement as well as 2 short personal essay questions. Plastic surgery designed a common supplemental application, currently in its second iteration, that highlights specialty-specific information from applicants to promote holistic review and eventually reduce application costs.19
Final Thoughts
The reforms introduced and proposed by dermatology and other specialties represent initial steps to address the issues inherent to the current residency application process. Providing faculty with better tools to holistically assess applicants during the review process and increasing transparency between programs and applicants should help optimize applicant-program fit and increase diversity in the dermatology workforce. Streamlining the application process to allow students to highlight their unique qualities in a user-friendly format as well as addressing potential inequities in interview distribution and access to the application process hopefully will contribute to better outcomes for both programs and applicants. However, many of these steps are likely to create additional administrative burdens on program faculty and are unlikely to allay student fears about matching.
The underlying issue for many specialties, and particularly for dermatology, is that demand far outstrips supply. With stable numbers of residency positions and an ever-increasing number of applicants, the match rate will continue to decrease, leading to increased anxiety among those interested in pursuing dermatology. Although USMLE Step 1 scores have been shown to have racial bias20 and there are no data correlating scores with clinical performance, the elimination of a scoring system may affect the number of applicants entering dermatology with downstream effects on match rates. Heightened anxiety places increased pressure on students to choose a specialty earlier in their training and impacts the activities they pursue during medical school. Overemphasis on specialty choice and the match process can lead to higher rates of burnout among students and trainees, as students may focus on activities designed to increase their chances of matching at the expense of pursuing activities that could lead to greater engagement and passion in their careers—a key protective factor against burnout.
The goal of the residency application process is to optimize fit between candidates and programs by aligning goals, values, and learning environment. Students and programs working together as honest brokers can lead to transformative change in the process, freeing both parties to highlight their unique qualities and contributions. Programs benefit from optimal fit by being able to hone their particular mission and recruit and retain residents and faculty engaged in that mission. Residents will thrive in programs that support their learning and career goals and will ultimately be better positioned to meaningfully contribute to their chosen field in whatever capacity they choose.
Acknowledgments—The views presented in this column reflect those of the 9 elected members of the Association of Professors of Dermatology Residency Program Directors Section steering committee, all of whom are program directors at their institutions (listed in parentheses): Ammar Ahmed, MD (The University of Texas at Austin, Austin, Texas); Yolanda Helfrich, MD (University of Michigan, Ann Arbor, Michigan); Jo-Ann M. Latkowksi, MD (New York University, New York); Kiran Motaparthi, MD (University of Florida, Gainesville, Florida); Adena E. Rosenblatt, MD, PhD (The University of Chicago, Chicago, Illinois); Ilana S. Rosman, MD (Washington University, St. Louis, Missouri); Travis Vandergriff, MD (University of Texas Southwestern, Dallas, Texas); Diane Whitaker-Worth, MD (University of Connecticut, Farmington, Connecticut); Scott Worswick, MD (University of Southern California, Los Angeles, California).
- Coalition for Physician Accountability. The Coalition for Physician Accountability’s Undergraduate Medical Education–Graduate Medical Education Review Committee (UGRC): recommendations for comprehensive improvement of the UME-GME transition. Accessed March 7, 2022. https://physicianaccountability.org/wp-content/uploads/2021/08/UGRC-Coalition-Report-FINAL.pdf
- Jackson JS, Bond M, Love JN, et al. Emergency medicine standardized letter of evaluation (SLOE): findings from the new electronic SLOE format. J Grad Med Educ. 2019;11:182-186.
- Santos-Parker KS, Morgan HK, Katz NT, et al. Can standardized dates for interview offers mitigate excessive interviewing? J Surg Educ. 2021;78:1091-1096.
- Pletcher SD, Chang CWD, Thorne MC, et al. The otolaryngology residency program preference signaling experience [published online October 5, 2021]. Acad Med. doi:10.1097/ACM.0000000000004441
- Association of Professors of Dermatology. Holistic review. Accessed March 7, 2022. https://www.dermatologyprofessors.org/files/3_Holistic%20review_Oct2020.pdf
- Rosmarin D, Friedman AJ, Burkemper NM, et al. The Association of Professors of Dermatology Program Directors Task Force and Residency Program Transparency Work Group guidelines on residency program transparency. J Drugs Dermatol. 2020;19:1117-1118.
- Rosman IS, Schadt CR, Samimi SS, et al. Approaching the dermatology residency application process during a pandemic. J Am Acad Dermatol. 2020;83:E351-E352.
- Association of Professors of Dermatology. Program director resources. Accessed March 7, 2022. https://www.dermatologyprofessors.org/programdirectors_resources.php
- Brumfiel CM, Jefferson IS, Wu AG, et al. A national webinar for dermatology applicants during the COVID-19 pandemic. J Am Acad Dermatol. 2021;84:574-575.
- Brumfiel CM, Jefferson IS, Rinderknecht FA, et al. Current perspectives of and potential reforms to the dermatology residency application process: a nationwide survey of program directors and applicants. Clin Dermatol. In press.
- Association of American Medical Colleges. Supplemental ERAS application (for the ERAS 2022 cycle). Accessed March 7, 2022. https://students-residents.aamc.org/applying-residencies-eras/supplementalerasapplication
- Association of American Medical Colleges. AAMC supplemental ERAS application: key findings from the 2022 application cycle. Accessed March 11, 2022. https://www.aamc.org/media/58891/download
- Warm EJ, Kinnear B, Pereira A, et al. The residency match: escaping the prisoner’s dilemma. J Grad Med Educ. 2021;13:616-625.
- Carmody JB, Rosman IS, Carlson JC. Application fever: reviewing the causes, costs, and cures for residency application inflation. Cureus. 2021;13:E13804.
- Hammoud MM, Andrews J, Skochelak SE. Improving the residency application and selection process: an optional early result acceptance program. JAMA. 2020;323:503-504.
- Winkel AF, Morgan HK, Akingbola O, et al. Perspectives of stakeholders about an early release acceptance program to complement the residency match in obstetrics and gynecology. JAMA Netw Open. 2021;4:E2124158.
- Morgan HK, Winkel AF, Standiford T, et al. The case for capping residency interviews. J Surg Educ. 2021;78:755-762.
- Association of University Professors of Ophthalmology. 2021-22 ophthalmology residency match FAQs. Accessed March 7, 2022. https://aupo.org/sites/default/files/2021-06/Residency%20Match%20FAQs_2021.pdf
- American Council of Academic Plastic Surgeons. Applying to plastic surgery (PSCA). Accessed March 7, 2022. https://acaplasticsurgeons.org/PSCA/
- Rubright JD, Jodoin M, Barone MA. Examining demographics, prior academic performance, and United States Medical Licensing Examination Scores. Acad Med. 2019;94:364-370.
- Coalition for Physician Accountability. The Coalition for Physician Accountability’s Undergraduate Medical Education–Graduate Medical Education Review Committee (UGRC): recommendations for comprehensive improvement of the UME-GME transition. Accessed March 7, 2022. https://physicianaccountability.org/wp-content/uploads/2021/08/UGRC-Coalition-Report-FINAL.pdf
- Jackson JS, Bond M, Love JN, et al. Emergency medicine standardized letter of evaluation (SLOE): findings from the new electronic SLOE format. J Grad Med Educ. 2019;11:182-186.
- Santos-Parker KS, Morgan HK, Katz NT, et al. Can standardized dates for interview offers mitigate excessive interviewing? J Surg Educ. 2021;78:1091-1096.
- Pletcher SD, Chang CWD, Thorne MC, et al. The otolaryngology residency program preference signaling experience [published online October 5, 2021]. Acad Med. doi:10.1097/ACM.0000000000004441
- Association of Professors of Dermatology. Holistic review. Accessed March 7, 2022. https://www.dermatologyprofessors.org/files/3_Holistic%20review_Oct2020.pdf
- Rosmarin D, Friedman AJ, Burkemper NM, et al. The Association of Professors of Dermatology Program Directors Task Force and Residency Program Transparency Work Group guidelines on residency program transparency. J Drugs Dermatol. 2020;19:1117-1118.
- Rosman IS, Schadt CR, Samimi SS, et al. Approaching the dermatology residency application process during a pandemic. J Am Acad Dermatol. 2020;83:E351-E352.
- Association of Professors of Dermatology. Program director resources. Accessed March 7, 2022. https://www.dermatologyprofessors.org/programdirectors_resources.php
- Brumfiel CM, Jefferson IS, Wu AG, et al. A national webinar for dermatology applicants during the COVID-19 pandemic. J Am Acad Dermatol. 2021;84:574-575.
- Brumfiel CM, Jefferson IS, Rinderknecht FA, et al. Current perspectives of and potential reforms to the dermatology residency application process: a nationwide survey of program directors and applicants. Clin Dermatol. In press.
- Association of American Medical Colleges. Supplemental ERAS application (for the ERAS 2022 cycle). Accessed March 7, 2022. https://students-residents.aamc.org/applying-residencies-eras/supplementalerasapplication
- Association of American Medical Colleges. AAMC supplemental ERAS application: key findings from the 2022 application cycle. Accessed March 11, 2022. https://www.aamc.org/media/58891/download
- Warm EJ, Kinnear B, Pereira A, et al. The residency match: escaping the prisoner’s dilemma. J Grad Med Educ. 2021;13:616-625.
- Carmody JB, Rosman IS, Carlson JC. Application fever: reviewing the causes, costs, and cures for residency application inflation. Cureus. 2021;13:E13804.
- Hammoud MM, Andrews J, Skochelak SE. Improving the residency application and selection process: an optional early result acceptance program. JAMA. 2020;323:503-504.
- Winkel AF, Morgan HK, Akingbola O, et al. Perspectives of stakeholders about an early release acceptance program to complement the residency match in obstetrics and gynecology. JAMA Netw Open. 2021;4:E2124158.
- Morgan HK, Winkel AF, Standiford T, et al. The case for capping residency interviews. J Surg Educ. 2021;78:755-762.
- Association of University Professors of Ophthalmology. 2021-22 ophthalmology residency match FAQs. Accessed March 7, 2022. https://aupo.org/sites/default/files/2021-06/Residency%20Match%20FAQs_2021.pdf
- American Council of Academic Plastic Surgeons. Applying to plastic surgery (PSCA). Accessed March 7, 2022. https://acaplasticsurgeons.org/PSCA/
- Rubright JD, Jodoin M, Barone MA. Examining demographics, prior academic performance, and United States Medical Licensing Examination Scores. Acad Med. 2019;94:364-370.
Practice Points
- Dermatology has implemented several reforms to the residency application process, including coordinated interview invitation release, mechanisms for enhanced transparency between programs and applicants, and a new common supplemental application.
- Across specialties, additional innovations to the residency application process have been implemented and proposed, including preference signaling, an early result acceptance process, and interview and application limits.
- Current efforts to improve the residency application process are ongoing with cross-specialty collaboration.
Residency Roundup: Introducing a New Partnership Between Cutis and the APD-RPDS
We are excited to announce a new partnership between Cutis and the Association of Professors of Dermatology Residency Program Directors Section (APD-RPDS). The new APD-RPDS column Residency Roundup will contain quarterly communications and submissions that we hope will facilitate greater dissemination of information that is useful to the dermatology teaching community.
The APD is a group of academic dermatologists whose membership comprises chairs, chiefs, residency and fellowship program directors, and teaching faculty. Each fall, the group convenes in Chicago, Illinois, for a 2-day meeting centered around departmental and program leadership with a focus on education. The APD-RPDS was formed in 2020 and is led by a steering committee of 9 members, including our current Chair, Ilana S. Rosman, MD (Washington University School of Medicine, St. Louis, Missouri), and Vice Chair, Jo-Ann M. Latkowski, MD (New York University, New York). Committee members are elected from and by the APD membership and must serve in program leadership at their home programs. The APD-RPDS helps plan and coordinate breakout sessions and lectures at the annual APD meeting, which typically relate to program director duties, changing policies within the American Board of Dermatology or Accreditation Council for Graduate Medical Education, ideas for future growth, and changes in our specialty and in resident education. Members of the APD-RPDS have access to the APD listserv, a valuable resource for discussing issues affecting residency training. We also have work groups led by our members, which include diversity, equity, and inclusion; resource development; communications; and the annual survey. To join the APD, the RPDS, and/or any of our workgroups, please reach out to us or visit the APD website (https://www.dermatologyprofessors.org).
We look forward to welcoming and expediently reviewing members’ submissions to the new Residency Roundup column falling into 2 principal categories within the scope of dermatologic recruitment, didactic education, and clinical training. The first category will feature novel tools, programs, and platforms to improve dermatology training through collaboration. This could entail a description of a new platform designed for sharing resources among programs and specialties to enhance learning for trainees and faculty alike. For example, if a database is created that contains prerecorded lectures pertaining to alopecia, a potential article submission might introduce the database and provide information on what topics are covered and how to access these lectures for readers worldwide. Likewise, if a new technology emerges that allows for easier collaboration among programs, a possible submission would introduce the technology and discuss its potential benefits to trainees, faculty, and practicing dermatologists.
Secondly and more commonly, we anticipate the Residency Roundup column will feature articles that delve into the critical issues and challenges currently impacting recruitment, training, and administration in dermatology residency programs. Specific topics may include but are not limited to recruitment of underrepresented in medicine applicants to dermatology, technological advances to improve teaching methods within training programs, surveys delving into the dermatology match process, and educational gaps or future directions in the specialty. The column occasionally may be used to disseminate information from our section of the APD, including consensus statements or editorials related to changes implemented in the dermatology residency application process. A prospective editorial on this subject could explore varying viewpoints of implemented and proposed changes as well as the reasons behind the changes.
Our group is collaborative, and our aim is to improve education, equity, management of program director responsibilities, and the dermatology application process for programs and applicants alike. With your input, experience, and varied perspectives, we look forward to moving the field of dermatology to a better future by working together.
We are excited to announce a new partnership between Cutis and the Association of Professors of Dermatology Residency Program Directors Section (APD-RPDS). The new APD-RPDS column Residency Roundup will contain quarterly communications and submissions that we hope will facilitate greater dissemination of information that is useful to the dermatology teaching community.
The APD is a group of academic dermatologists whose membership comprises chairs, chiefs, residency and fellowship program directors, and teaching faculty. Each fall, the group convenes in Chicago, Illinois, for a 2-day meeting centered around departmental and program leadership with a focus on education. The APD-RPDS was formed in 2020 and is led by a steering committee of 9 members, including our current Chair, Ilana S. Rosman, MD (Washington University School of Medicine, St. Louis, Missouri), and Vice Chair, Jo-Ann M. Latkowski, MD (New York University, New York). Committee members are elected from and by the APD membership and must serve in program leadership at their home programs. The APD-RPDS helps plan and coordinate breakout sessions and lectures at the annual APD meeting, which typically relate to program director duties, changing policies within the American Board of Dermatology or Accreditation Council for Graduate Medical Education, ideas for future growth, and changes in our specialty and in resident education. Members of the APD-RPDS have access to the APD listserv, a valuable resource for discussing issues affecting residency training. We also have work groups led by our members, which include diversity, equity, and inclusion; resource development; communications; and the annual survey. To join the APD, the RPDS, and/or any of our workgroups, please reach out to us or visit the APD website (https://www.dermatologyprofessors.org).
We look forward to welcoming and expediently reviewing members’ submissions to the new Residency Roundup column falling into 2 principal categories within the scope of dermatologic recruitment, didactic education, and clinical training. The first category will feature novel tools, programs, and platforms to improve dermatology training through collaboration. This could entail a description of a new platform designed for sharing resources among programs and specialties to enhance learning for trainees and faculty alike. For example, if a database is created that contains prerecorded lectures pertaining to alopecia, a potential article submission might introduce the database and provide information on what topics are covered and how to access these lectures for readers worldwide. Likewise, if a new technology emerges that allows for easier collaboration among programs, a possible submission would introduce the technology and discuss its potential benefits to trainees, faculty, and practicing dermatologists.
Secondly and more commonly, we anticipate the Residency Roundup column will feature articles that delve into the critical issues and challenges currently impacting recruitment, training, and administration in dermatology residency programs. Specific topics may include but are not limited to recruitment of underrepresented in medicine applicants to dermatology, technological advances to improve teaching methods within training programs, surveys delving into the dermatology match process, and educational gaps or future directions in the specialty. The column occasionally may be used to disseminate information from our section of the APD, including consensus statements or editorials related to changes implemented in the dermatology residency application process. A prospective editorial on this subject could explore varying viewpoints of implemented and proposed changes as well as the reasons behind the changes.
Our group is collaborative, and our aim is to improve education, equity, management of program director responsibilities, and the dermatology application process for programs and applicants alike. With your input, experience, and varied perspectives, we look forward to moving the field of dermatology to a better future by working together.
We are excited to announce a new partnership between Cutis and the Association of Professors of Dermatology Residency Program Directors Section (APD-RPDS). The new APD-RPDS column Residency Roundup will contain quarterly communications and submissions that we hope will facilitate greater dissemination of information that is useful to the dermatology teaching community.
The APD is a group of academic dermatologists whose membership comprises chairs, chiefs, residency and fellowship program directors, and teaching faculty. Each fall, the group convenes in Chicago, Illinois, for a 2-day meeting centered around departmental and program leadership with a focus on education. The APD-RPDS was formed in 2020 and is led by a steering committee of 9 members, including our current Chair, Ilana S. Rosman, MD (Washington University School of Medicine, St. Louis, Missouri), and Vice Chair, Jo-Ann M. Latkowski, MD (New York University, New York). Committee members are elected from and by the APD membership and must serve in program leadership at their home programs. The APD-RPDS helps plan and coordinate breakout sessions and lectures at the annual APD meeting, which typically relate to program director duties, changing policies within the American Board of Dermatology or Accreditation Council for Graduate Medical Education, ideas for future growth, and changes in our specialty and in resident education. Members of the APD-RPDS have access to the APD listserv, a valuable resource for discussing issues affecting residency training. We also have work groups led by our members, which include diversity, equity, and inclusion; resource development; communications; and the annual survey. To join the APD, the RPDS, and/or any of our workgroups, please reach out to us or visit the APD website (https://www.dermatologyprofessors.org).
We look forward to welcoming and expediently reviewing members’ submissions to the new Residency Roundup column falling into 2 principal categories within the scope of dermatologic recruitment, didactic education, and clinical training. The first category will feature novel tools, programs, and platforms to improve dermatology training through collaboration. This could entail a description of a new platform designed for sharing resources among programs and specialties to enhance learning for trainees and faculty alike. For example, if a database is created that contains prerecorded lectures pertaining to alopecia, a potential article submission might introduce the database and provide information on what topics are covered and how to access these lectures for readers worldwide. Likewise, if a new technology emerges that allows for easier collaboration among programs, a possible submission would introduce the technology and discuss its potential benefits to trainees, faculty, and practicing dermatologists.
Secondly and more commonly, we anticipate the Residency Roundup column will feature articles that delve into the critical issues and challenges currently impacting recruitment, training, and administration in dermatology residency programs. Specific topics may include but are not limited to recruitment of underrepresented in medicine applicants to dermatology, technological advances to improve teaching methods within training programs, surveys delving into the dermatology match process, and educational gaps or future directions in the specialty. The column occasionally may be used to disseminate information from our section of the APD, including consensus statements or editorials related to changes implemented in the dermatology residency application process. A prospective editorial on this subject could explore varying viewpoints of implemented and proposed changes as well as the reasons behind the changes.
Our group is collaborative, and our aim is to improve education, equity, management of program director responsibilities, and the dermatology application process for programs and applicants alike. With your input, experience, and varied perspectives, we look forward to moving the field of dermatology to a better future by working together.
Some leukemias detectable up to 16 years before diagnosis?
Previous analyses showed that monoclonal B-cell lymphocytosis (MBL), a CLL precursor state, has been detected up to 6 years before CLL diagnosis, the investigators explained, noting that “[a]nother prognostically relevant immunogenetic feature of CLL concerns the stereotype of the B-cell receptor immunoglobulins (BcR IG).”
“Indeed, distinct stereotyped subsets can be defined by the expression of shared sequence motifs and are associated with particular presentation and outcomes,” P. Martijn Kolijn, PhD, a researcher in the department of immunology at Erasmus Medical Center, Rotterdam, the Netherlands, and colleagues wrote in a brief report published online in Blood. In an effort to “gain insight into the composition of the BcR IG repertoire during the early stages of CLL,” the investigators utilized next-generation sequencing to analyze 124 blood samples taken from healthy individuals up to 22 years before they received a diagnosis of CLL or small lymphocytic leukemia (SLL). An additional 118 matched control samples were also analyzed.
Study subjects were participants in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort.
“First, unsurprisingly, we observed a significant difference in the frequency of the dominant clonotype in CLL patients versus controls with a median frequency of 54.9%, compared to only 0.38% in controls,” they wrote.
Among 28 patients whose lymphocyte counts were measured at baseline, 10 showed evidence of lymphocytosis up to 8 years before CLL diagnosis.
This suggests undiagnosed instances of high-count MBL (cases with a cell count above 0.5x 109 cells/L, which can progress to CLL) or asymptomatic CLL, they explained.
“In contrast, next-generation sequencing results showed detectable skewing of the IGH gene repertoire in 21/28 patients up to 15 years before CLL diagnosis, often in the absence of elevated lymphocyte counts,” they wrote. “Remarkably, some patients with CLL requiring treatment and clinical transformation to an aggressive B-cell lymphoma displayed considerable skewing in the IGH gene repertoire even 16 years before CLL diagnosis.”
Patients with a prediagnostic IGHV-unmutated dominant clonotype had significantly shorter overall survival after CLL diagnosis than did those with an IGHV-mutated clonotype, they noted.
“Furthermore, at early timepoints (>10 years before diagnosis), patients with a high dominant clonotype frequency were more likely to be IGHV mutated, whereas closer to diagnosis this tendency was lost, indicating that the prediagnostic phase may be even longer than 16 years for [mutated] CLL patients,” they added.
The investigators also found that:
- Twenty-five patients carried stereotyped BcR IG up to 17 years prior to CLL diagnosis, and of these, 10 clonotypes were assigned to minor subsets and 15 to major CLL subsets. Among the latter, 14 of the 15 belonged to high-risk subsets, and most of those showed a trend for faster disease evolution.
- High frequency of the dominant clonotype was evident in samples obtained less than 6 years before diagnosis, whereas high-risk stereotyped clonotypes found longer before diagnosis (as early as 16 years) tended to have a lower dominant clonotype frequency (<20% of IGH gene repertoire)
- The stereotyped BcR IG matched the clonotype at diagnosis for both patients with diagnostic material.
- No stereotyped subsets were identified among the dominant clonotypes of the healthy controls.
“To our knowledge, the dynamics of the emergence of biclonality in an MBL patient and subsequent progression to CLL have never been captured in such a convincing manner,” they noted.
The findings “extend current knowledge on the evolution of the IGH repertoire prior to CLL diagnosis, highlighting that even high-risk CLL subtypes may display a prolonged indolent preclinical stage,” they added, speculating that “somatic genetic aberrations, (auto)stimulation, epigenetic and/or microenvironmental influences are required for the transformation into overt CLL.”
The investigators also noted that since the observed skewing in the IGH gene repertoire often occurs prior to B-cell lymphocytosis, they consider the findings “a novel extension to the characterization of MBL.”
“Further studies may prove invaluable in the clinical distinction between ‘progressing’ MBL versus ‘stable’ MBL. Notwithstanding the above, we emphasize that early detection is only warranted if it provides clear benefits to patient care,” they concluded.
In a related commentary, Gerald Marti, MD, PhD, of the National Heart, Lung, and Blood Institute, emphasized that the findings “represent the earliest detection of a clonotypic precursor cell for CLL.” .
They also raise new questions and point to new directions for research, Dr. Marti noted.
“Where do we go from here? CLL has a long evolutionary history in which early branching may start as an oligoclonal process (antigen stimulation) and include driver mutations,” he wrote. “A long-term analysis of the B-cell repertoire in familial CLL might shed light on this process. Further clarification of the mechanisms of age-related immune senescence is also of interest.”
The study authors and Dr. Marti reported having no competing financial interests.
Previous analyses showed that monoclonal B-cell lymphocytosis (MBL), a CLL precursor state, has been detected up to 6 years before CLL diagnosis, the investigators explained, noting that “[a]nother prognostically relevant immunogenetic feature of CLL concerns the stereotype of the B-cell receptor immunoglobulins (BcR IG).”
“Indeed, distinct stereotyped subsets can be defined by the expression of shared sequence motifs and are associated with particular presentation and outcomes,” P. Martijn Kolijn, PhD, a researcher in the department of immunology at Erasmus Medical Center, Rotterdam, the Netherlands, and colleagues wrote in a brief report published online in Blood. In an effort to “gain insight into the composition of the BcR IG repertoire during the early stages of CLL,” the investigators utilized next-generation sequencing to analyze 124 blood samples taken from healthy individuals up to 22 years before they received a diagnosis of CLL or small lymphocytic leukemia (SLL). An additional 118 matched control samples were also analyzed.
Study subjects were participants in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort.
“First, unsurprisingly, we observed a significant difference in the frequency of the dominant clonotype in CLL patients versus controls with a median frequency of 54.9%, compared to only 0.38% in controls,” they wrote.
Among 28 patients whose lymphocyte counts were measured at baseline, 10 showed evidence of lymphocytosis up to 8 years before CLL diagnosis.
This suggests undiagnosed instances of high-count MBL (cases with a cell count above 0.5x 109 cells/L, which can progress to CLL) or asymptomatic CLL, they explained.
“In contrast, next-generation sequencing results showed detectable skewing of the IGH gene repertoire in 21/28 patients up to 15 years before CLL diagnosis, often in the absence of elevated lymphocyte counts,” they wrote. “Remarkably, some patients with CLL requiring treatment and clinical transformation to an aggressive B-cell lymphoma displayed considerable skewing in the IGH gene repertoire even 16 years before CLL diagnosis.”
Patients with a prediagnostic IGHV-unmutated dominant clonotype had significantly shorter overall survival after CLL diagnosis than did those with an IGHV-mutated clonotype, they noted.
“Furthermore, at early timepoints (>10 years before diagnosis), patients with a high dominant clonotype frequency were more likely to be IGHV mutated, whereas closer to diagnosis this tendency was lost, indicating that the prediagnostic phase may be even longer than 16 years for [mutated] CLL patients,” they added.
The investigators also found that:
- Twenty-five patients carried stereotyped BcR IG up to 17 years prior to CLL diagnosis, and of these, 10 clonotypes were assigned to minor subsets and 15 to major CLL subsets. Among the latter, 14 of the 15 belonged to high-risk subsets, and most of those showed a trend for faster disease evolution.
- High frequency of the dominant clonotype was evident in samples obtained less than 6 years before diagnosis, whereas high-risk stereotyped clonotypes found longer before diagnosis (as early as 16 years) tended to have a lower dominant clonotype frequency (<20% of IGH gene repertoire)
- The stereotyped BcR IG matched the clonotype at diagnosis for both patients with diagnostic material.
- No stereotyped subsets were identified among the dominant clonotypes of the healthy controls.
“To our knowledge, the dynamics of the emergence of biclonality in an MBL patient and subsequent progression to CLL have never been captured in such a convincing manner,” they noted.
The findings “extend current knowledge on the evolution of the IGH repertoire prior to CLL diagnosis, highlighting that even high-risk CLL subtypes may display a prolonged indolent preclinical stage,” they added, speculating that “somatic genetic aberrations, (auto)stimulation, epigenetic and/or microenvironmental influences are required for the transformation into overt CLL.”
The investigators also noted that since the observed skewing in the IGH gene repertoire often occurs prior to B-cell lymphocytosis, they consider the findings “a novel extension to the characterization of MBL.”
“Further studies may prove invaluable in the clinical distinction between ‘progressing’ MBL versus ‘stable’ MBL. Notwithstanding the above, we emphasize that early detection is only warranted if it provides clear benefits to patient care,” they concluded.
In a related commentary, Gerald Marti, MD, PhD, of the National Heart, Lung, and Blood Institute, emphasized that the findings “represent the earliest detection of a clonotypic precursor cell for CLL.” .
They also raise new questions and point to new directions for research, Dr. Marti noted.
“Where do we go from here? CLL has a long evolutionary history in which early branching may start as an oligoclonal process (antigen stimulation) and include driver mutations,” he wrote. “A long-term analysis of the B-cell repertoire in familial CLL might shed light on this process. Further clarification of the mechanisms of age-related immune senescence is also of interest.”
The study authors and Dr. Marti reported having no competing financial interests.
Previous analyses showed that monoclonal B-cell lymphocytosis (MBL), a CLL precursor state, has been detected up to 6 years before CLL diagnosis, the investigators explained, noting that “[a]nother prognostically relevant immunogenetic feature of CLL concerns the stereotype of the B-cell receptor immunoglobulins (BcR IG).”
“Indeed, distinct stereotyped subsets can be defined by the expression of shared sequence motifs and are associated with particular presentation and outcomes,” P. Martijn Kolijn, PhD, a researcher in the department of immunology at Erasmus Medical Center, Rotterdam, the Netherlands, and colleagues wrote in a brief report published online in Blood. In an effort to “gain insight into the composition of the BcR IG repertoire during the early stages of CLL,” the investigators utilized next-generation sequencing to analyze 124 blood samples taken from healthy individuals up to 22 years before they received a diagnosis of CLL or small lymphocytic leukemia (SLL). An additional 118 matched control samples were also analyzed.
Study subjects were participants in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort.
“First, unsurprisingly, we observed a significant difference in the frequency of the dominant clonotype in CLL patients versus controls with a median frequency of 54.9%, compared to only 0.38% in controls,” they wrote.
Among 28 patients whose lymphocyte counts were measured at baseline, 10 showed evidence of lymphocytosis up to 8 years before CLL diagnosis.
This suggests undiagnosed instances of high-count MBL (cases with a cell count above 0.5x 109 cells/L, which can progress to CLL) or asymptomatic CLL, they explained.
“In contrast, next-generation sequencing results showed detectable skewing of the IGH gene repertoire in 21/28 patients up to 15 years before CLL diagnosis, often in the absence of elevated lymphocyte counts,” they wrote. “Remarkably, some patients with CLL requiring treatment and clinical transformation to an aggressive B-cell lymphoma displayed considerable skewing in the IGH gene repertoire even 16 years before CLL diagnosis.”
Patients with a prediagnostic IGHV-unmutated dominant clonotype had significantly shorter overall survival after CLL diagnosis than did those with an IGHV-mutated clonotype, they noted.
“Furthermore, at early timepoints (>10 years before diagnosis), patients with a high dominant clonotype frequency were more likely to be IGHV mutated, whereas closer to diagnosis this tendency was lost, indicating that the prediagnostic phase may be even longer than 16 years for [mutated] CLL patients,” they added.
The investigators also found that:
- Twenty-five patients carried stereotyped BcR IG up to 17 years prior to CLL diagnosis, and of these, 10 clonotypes were assigned to minor subsets and 15 to major CLL subsets. Among the latter, 14 of the 15 belonged to high-risk subsets, and most of those showed a trend for faster disease evolution.
- High frequency of the dominant clonotype was evident in samples obtained less than 6 years before diagnosis, whereas high-risk stereotyped clonotypes found longer before diagnosis (as early as 16 years) tended to have a lower dominant clonotype frequency (<20% of IGH gene repertoire)
- The stereotyped BcR IG matched the clonotype at diagnosis for both patients with diagnostic material.
- No stereotyped subsets were identified among the dominant clonotypes of the healthy controls.
“To our knowledge, the dynamics of the emergence of biclonality in an MBL patient and subsequent progression to CLL have never been captured in such a convincing manner,” they noted.
The findings “extend current knowledge on the evolution of the IGH repertoire prior to CLL diagnosis, highlighting that even high-risk CLL subtypes may display a prolonged indolent preclinical stage,” they added, speculating that “somatic genetic aberrations, (auto)stimulation, epigenetic and/or microenvironmental influences are required for the transformation into overt CLL.”
The investigators also noted that since the observed skewing in the IGH gene repertoire often occurs prior to B-cell lymphocytosis, they consider the findings “a novel extension to the characterization of MBL.”
“Further studies may prove invaluable in the clinical distinction between ‘progressing’ MBL versus ‘stable’ MBL. Notwithstanding the above, we emphasize that early detection is only warranted if it provides clear benefits to patient care,” they concluded.
In a related commentary, Gerald Marti, MD, PhD, of the National Heart, Lung, and Blood Institute, emphasized that the findings “represent the earliest detection of a clonotypic precursor cell for CLL.” .
They also raise new questions and point to new directions for research, Dr. Marti noted.
“Where do we go from here? CLL has a long evolutionary history in which early branching may start as an oligoclonal process (antigen stimulation) and include driver mutations,” he wrote. “A long-term analysis of the B-cell repertoire in familial CLL might shed light on this process. Further clarification of the mechanisms of age-related immune senescence is also of interest.”
The study authors and Dr. Marti reported having no competing financial interests.
FROM BLOOD
AGA Clinical Practice Update: Expert review on personalizing GERD management
A recent American Gastroenterological Association Clinical Practice Update for evaluation and management of gastroesophageal reflux disease (GERD) focuses on delivering personalized diagnostic and therapeutic strategies.
The document includes new advice on use of upfront objective testing for isolated extraesophageal symptoms, confirmation of GERD diagnosis prior to long-term GERD therapy even in PPI responders, as well as important elements focused on personalization of therapy.
Although GERD is common, with an estimated 30% of people in the United States experiencing symptoms, up to half of all individuals on proton pump inhibitor (PPI) therapy report incomplete symptom improvement. That could be due to the heterogeneous nature of symptoms, which may include heartburn and regurgitation, chest pain, and cough or sore throat, among others. Other conditions may produce similar symptoms or could be exacerbated by the presence of GERD.
The authors of the expert review, published in Clinical Gastroenterology and Hepatology, note that these considerations have driven increased interest in personalized approaches to the management of GERD. The practice update includes sections on how to approach GERD symptoms in the clinic, personalized diagnosis related to GERD symptoms, and precision management.
In the initial management, the authors offer advice on involving the patient in creating a care plan, patient education, and conducting a 4- to 8-week PPI trial in patients with heartburn, regurgitation, or noncardiac chest pains without accompanying alarm signals. If symptoms don’t improve to the patient’s satisfaction, dosing can be boosted to twice per day, or a more effective acid suppressor can be substituted and continued at a once-daily dose. When the response to PPIs is adequate, the dose should be reduced until the lowest effective dose is reached, or the patient could potentially be moved to H2 receptor antagonists or other antacids. However, patients with erosive esophagitis, biopsy-confirmed Barrett’s esophagus, or peptic stricture must stay on long-term PPI therapy.
The authors also gave advice on when to conduct objective testing. When a PPI trial doesn’t adequately address troublesome heartburn, regurgitation, and/or noncardiac chest pain, or if alarm systems are present, endoscopy should be employed to look for erosive reflux disease or long-segment Barrett’s esophagus as conclusive evidence for GERD. If these are absent, prolonged wireless pH monitoring while a patient is off medication is suggested. In addition, patients with extraesophageal symptoms suspected to be caused by reflux should undergo upfront objective reflux testing while off PPI therapy rather than doing an empiric PPI trial.
The authors advise that, if patients don’t have proven GERD and are continued on PPI therapy, they should be evaluated within 12 months to ensure that the therapy and dose are appropriate. Physicians should offer endoscopy with prolonged wireless reflux monitoring in the absence of PPI therapy (ideally after 2-4 weeks of withdrawal) to confirm that long-term PPI therapy is needed.
In the section on personalization of disease management, the authors note that ambulatory reflux monitoring and upper gastrointestinal endoscopy can be used to guide management of GERD. When upper GI endoscopy reveals no erosive findings and esophageal acid exposure time (AET) is less than 4% throughout all days of prolonged wireless pH monitoring, the physician can conclude that the patient has no pathologic gastroesophageal reflux and is likely to have a functional esophageal disorder. In contrast, erosive findings during upper GI endoscopy and/or AET more than 4% across at least 1 day of wireless pH monitoring suggests a GERD diagnosis.
Optimization of PPI is important among patients with GERD, and the authors stress that patients should be educated about the safety of PPI use.
Adjunctive pharmacotherapy is useful and can include alginate antacids for breakthrough symptoms, H2RAs for nocturnal symptoms, baclofen to counter regurgitation or belching, and prokinetics for accompanying gastroparesis. The choice of medications depends on the phenotype, and they should not be used empirically.
For patients with functional heartburn or reflux disease linked to esophageal hypervigilance, reflux sensitivity, or behavioral disorders, options include pharmacologic neuromodulation, hypnotherapy provided by a behavioral therapist, cognitive behavioral therapy, and diaphragmatic breathing and relaxation.
If symptoms persist despite efforts at optimization of treatments and lifestyle factors, ambulatory 24-hour pH-impedance monitoring on PPI can be used to investigate mechanistic causes, especially when there is no known antireflux barrier abnormality, but the technique requires expertise to correctly interpret. This can ensure that the symptoms are not due to reflux hypersensitivity, rumination syndrome, or a belching disorder. When symptoms are confirmed to be treatment resistant, therapy should be escalated, using a strategy that incorporates a pattern of reflux, integrity of the antireflux barrier, obesity if present, and psychological factors.
Surgical options for confirmed GERD include laparoscopic fundoplication and magnetic sphincter augmentation. Transoral incisionless fundoplication can be performed endoscopically in selected patients. For obese patients with confirmed GERD, Roux-en-Y gastric bypass is effective at reducing reflux and can be used as a salvage treatment for nonobese patients. Sleeve gastrectomy may exacerbate GERD.
The authors reported relationships with Medtronic, Diversatek, Ironwood, and Takeda. The authors also reported funding from National Institutes of Health grants.
A recent American Gastroenterological Association Clinical Practice Update for evaluation and management of gastroesophageal reflux disease (GERD) focuses on delivering personalized diagnostic and therapeutic strategies.
The document includes new advice on use of upfront objective testing for isolated extraesophageal symptoms, confirmation of GERD diagnosis prior to long-term GERD therapy even in PPI responders, as well as important elements focused on personalization of therapy.
Although GERD is common, with an estimated 30% of people in the United States experiencing symptoms, up to half of all individuals on proton pump inhibitor (PPI) therapy report incomplete symptom improvement. That could be due to the heterogeneous nature of symptoms, which may include heartburn and regurgitation, chest pain, and cough or sore throat, among others. Other conditions may produce similar symptoms or could be exacerbated by the presence of GERD.
The authors of the expert review, published in Clinical Gastroenterology and Hepatology, note that these considerations have driven increased interest in personalized approaches to the management of GERD. The practice update includes sections on how to approach GERD symptoms in the clinic, personalized diagnosis related to GERD symptoms, and precision management.
In the initial management, the authors offer advice on involving the patient in creating a care plan, patient education, and conducting a 4- to 8-week PPI trial in patients with heartburn, regurgitation, or noncardiac chest pains without accompanying alarm signals. If symptoms don’t improve to the patient’s satisfaction, dosing can be boosted to twice per day, or a more effective acid suppressor can be substituted and continued at a once-daily dose. When the response to PPIs is adequate, the dose should be reduced until the lowest effective dose is reached, or the patient could potentially be moved to H2 receptor antagonists or other antacids. However, patients with erosive esophagitis, biopsy-confirmed Barrett’s esophagus, or peptic stricture must stay on long-term PPI therapy.
The authors also gave advice on when to conduct objective testing. When a PPI trial doesn’t adequately address troublesome heartburn, regurgitation, and/or noncardiac chest pain, or if alarm systems are present, endoscopy should be employed to look for erosive reflux disease or long-segment Barrett’s esophagus as conclusive evidence for GERD. If these are absent, prolonged wireless pH monitoring while a patient is off medication is suggested. In addition, patients with extraesophageal symptoms suspected to be caused by reflux should undergo upfront objective reflux testing while off PPI therapy rather than doing an empiric PPI trial.
The authors advise that, if patients don’t have proven GERD and are continued on PPI therapy, they should be evaluated within 12 months to ensure that the therapy and dose are appropriate. Physicians should offer endoscopy with prolonged wireless reflux monitoring in the absence of PPI therapy (ideally after 2-4 weeks of withdrawal) to confirm that long-term PPI therapy is needed.
In the section on personalization of disease management, the authors note that ambulatory reflux monitoring and upper gastrointestinal endoscopy can be used to guide management of GERD. When upper GI endoscopy reveals no erosive findings and esophageal acid exposure time (AET) is less than 4% throughout all days of prolonged wireless pH monitoring, the physician can conclude that the patient has no pathologic gastroesophageal reflux and is likely to have a functional esophageal disorder. In contrast, erosive findings during upper GI endoscopy and/or AET more than 4% across at least 1 day of wireless pH monitoring suggests a GERD diagnosis.
Optimization of PPI is important among patients with GERD, and the authors stress that patients should be educated about the safety of PPI use.
Adjunctive pharmacotherapy is useful and can include alginate antacids for breakthrough symptoms, H2RAs for nocturnal symptoms, baclofen to counter regurgitation or belching, and prokinetics for accompanying gastroparesis. The choice of medications depends on the phenotype, and they should not be used empirically.
For patients with functional heartburn or reflux disease linked to esophageal hypervigilance, reflux sensitivity, or behavioral disorders, options include pharmacologic neuromodulation, hypnotherapy provided by a behavioral therapist, cognitive behavioral therapy, and diaphragmatic breathing and relaxation.
If symptoms persist despite efforts at optimization of treatments and lifestyle factors, ambulatory 24-hour pH-impedance monitoring on PPI can be used to investigate mechanistic causes, especially when there is no known antireflux barrier abnormality, but the technique requires expertise to correctly interpret. This can ensure that the symptoms are not due to reflux hypersensitivity, rumination syndrome, or a belching disorder. When symptoms are confirmed to be treatment resistant, therapy should be escalated, using a strategy that incorporates a pattern of reflux, integrity of the antireflux barrier, obesity if present, and psychological factors.
Surgical options for confirmed GERD include laparoscopic fundoplication and magnetic sphincter augmentation. Transoral incisionless fundoplication can be performed endoscopically in selected patients. For obese patients with confirmed GERD, Roux-en-Y gastric bypass is effective at reducing reflux and can be used as a salvage treatment for nonobese patients. Sleeve gastrectomy may exacerbate GERD.
The authors reported relationships with Medtronic, Diversatek, Ironwood, and Takeda. The authors also reported funding from National Institutes of Health grants.
A recent American Gastroenterological Association Clinical Practice Update for evaluation and management of gastroesophageal reflux disease (GERD) focuses on delivering personalized diagnostic and therapeutic strategies.
The document includes new advice on use of upfront objective testing for isolated extraesophageal symptoms, confirmation of GERD diagnosis prior to long-term GERD therapy even in PPI responders, as well as important elements focused on personalization of therapy.
Although GERD is common, with an estimated 30% of people in the United States experiencing symptoms, up to half of all individuals on proton pump inhibitor (PPI) therapy report incomplete symptom improvement. That could be due to the heterogeneous nature of symptoms, which may include heartburn and regurgitation, chest pain, and cough or sore throat, among others. Other conditions may produce similar symptoms or could be exacerbated by the presence of GERD.
The authors of the expert review, published in Clinical Gastroenterology and Hepatology, note that these considerations have driven increased interest in personalized approaches to the management of GERD. The practice update includes sections on how to approach GERD symptoms in the clinic, personalized diagnosis related to GERD symptoms, and precision management.
In the initial management, the authors offer advice on involving the patient in creating a care plan, patient education, and conducting a 4- to 8-week PPI trial in patients with heartburn, regurgitation, or noncardiac chest pains without accompanying alarm signals. If symptoms don’t improve to the patient’s satisfaction, dosing can be boosted to twice per day, or a more effective acid suppressor can be substituted and continued at a once-daily dose. When the response to PPIs is adequate, the dose should be reduced until the lowest effective dose is reached, or the patient could potentially be moved to H2 receptor antagonists or other antacids. However, patients with erosive esophagitis, biopsy-confirmed Barrett’s esophagus, or peptic stricture must stay on long-term PPI therapy.
The authors also gave advice on when to conduct objective testing. When a PPI trial doesn’t adequately address troublesome heartburn, regurgitation, and/or noncardiac chest pain, or if alarm systems are present, endoscopy should be employed to look for erosive reflux disease or long-segment Barrett’s esophagus as conclusive evidence for GERD. If these are absent, prolonged wireless pH monitoring while a patient is off medication is suggested. In addition, patients with extraesophageal symptoms suspected to be caused by reflux should undergo upfront objective reflux testing while off PPI therapy rather than doing an empiric PPI trial.
The authors advise that, if patients don’t have proven GERD and are continued on PPI therapy, they should be evaluated within 12 months to ensure that the therapy and dose are appropriate. Physicians should offer endoscopy with prolonged wireless reflux monitoring in the absence of PPI therapy (ideally after 2-4 weeks of withdrawal) to confirm that long-term PPI therapy is needed.
In the section on personalization of disease management, the authors note that ambulatory reflux monitoring and upper gastrointestinal endoscopy can be used to guide management of GERD. When upper GI endoscopy reveals no erosive findings and esophageal acid exposure time (AET) is less than 4% throughout all days of prolonged wireless pH monitoring, the physician can conclude that the patient has no pathologic gastroesophageal reflux and is likely to have a functional esophageal disorder. In contrast, erosive findings during upper GI endoscopy and/or AET more than 4% across at least 1 day of wireless pH monitoring suggests a GERD diagnosis.
Optimization of PPI is important among patients with GERD, and the authors stress that patients should be educated about the safety of PPI use.
Adjunctive pharmacotherapy is useful and can include alginate antacids for breakthrough symptoms, H2RAs for nocturnal symptoms, baclofen to counter regurgitation or belching, and prokinetics for accompanying gastroparesis. The choice of medications depends on the phenotype, and they should not be used empirically.
For patients with functional heartburn or reflux disease linked to esophageal hypervigilance, reflux sensitivity, or behavioral disorders, options include pharmacologic neuromodulation, hypnotherapy provided by a behavioral therapist, cognitive behavioral therapy, and diaphragmatic breathing and relaxation.
If symptoms persist despite efforts at optimization of treatments and lifestyle factors, ambulatory 24-hour pH-impedance monitoring on PPI can be used to investigate mechanistic causes, especially when there is no known antireflux barrier abnormality, but the technique requires expertise to correctly interpret. This can ensure that the symptoms are not due to reflux hypersensitivity, rumination syndrome, or a belching disorder. When symptoms are confirmed to be treatment resistant, therapy should be escalated, using a strategy that incorporates a pattern of reflux, integrity of the antireflux barrier, obesity if present, and psychological factors.
Surgical options for confirmed GERD include laparoscopic fundoplication and magnetic sphincter augmentation. Transoral incisionless fundoplication can be performed endoscopically in selected patients. For obese patients with confirmed GERD, Roux-en-Y gastric bypass is effective at reducing reflux and can be used as a salvage treatment for nonobese patients. Sleeve gastrectomy may exacerbate GERD.
The authors reported relationships with Medtronic, Diversatek, Ironwood, and Takeda. The authors also reported funding from National Institutes of Health grants.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY